Chapter 14 Neisseriaceae, Veillonella, parvobacteria and Capnocytophaga
Neisseriaceae
The Neisseriaceae include the genera Neisseria and Moraxella. Two species of Neisseria are human pathogens:
There are a number of non-pathogenic species, such as Neisseria sicca, Neisseria mucosa and Neisseria lactamica, which are members of the indigenous flora, including the oral mucosa. Hence, it is important to differentiate these from the pathogenic species from oral samples.
N. gonorrhoeae is the agent of gonorrhoea, the most frequently diagnosed venereal disease in western Europe and the USA. Gonococci frequently cause pelvic inflammatory disease (PID) and sterility in women, in addition to arthritis and sometimes septicaemia. N. meningitidis is the aetiological agent of meningococcal meningitis, a highly contagious disease associated with a mortality rate approximating 80% when untreated.
General characteristics
Non-motile, Gram-negative cocci ranging from 0.6 to 1.0 µm in diameter. On microscopy, the cocci are seen as pairs with concave adjacent sides (bean-shaped); tetrads, short chains and clusters are occasionally seen but all show the characteristic pairing.
Pathogenic Neisseria species are nutritionally fastidious, especially on initial isolation from clinical specimens; the non-pathogenic species are less so. Though aerobic, most strains of N. gonorrhoeae are capnophilic (they require increased carbon dioxide for growth); haemolysed blood and solubilized starch enhance growth.
Members of this genus grow optimally at 36–39°C, although the non-pathogenic species can grow at temperatures below 24°C.
Neisseria gonorrhoeae
Habitat and transmission
The human urogenital tract is the usual habitat; oral, nasopharyngeal and rectal carriage in healthy individuals is not uncommon. Spread is by both homosexual and heterosexual intercourse or intimate contact.
Culture and identification
Specimens are usually inoculated onto an enriched medium (lysed blood or chocolate agar normally) and incubated under 5–10% carbon dioxide (as the species is capnophilic). Small, grey, oxidase-positive colonies initially become large and opaque on prolonged incubation. Subsequent staining by fluorescent antibody techniques, and the production of acid from glucose but not from maltose or sucrose, confirms the identification. Gram-stained smears (of urethral exudate from men and the cervix in women) usually reveal Gram-negative, kidney-shaped intracellular cocci in pairs.
Neisseria meningitidis
Habitat and transmission
The main reservoir is the nasopharynx in healthy individuals (10–25%). Droplet spread is the most common transmission mode.
Culture and identification
As for N. gonorrhoeae. Presumptive identification is made by observing Gram-negative cocci in pairs in nasopharyngeal discharge, cerebrospinal fluid or blood smears. Selective media are not required as the organism is found pure in cerebrospinal fluid. Identified by the carbohydrate utilization test: produces acid from the oxidation of glucose and maltose. Serology is useful.
Pathogenicity
In susceptible individuals, meningococci spread from the nasopharynx into the blood stream (septicaemia), and then to the meninges. Septicaemia is accompanied by a rash. Eventual death may be due to meningitis or adrenal haemorrhage (Waterhouse–Friderichsen syndrome). The antiphagocytic properties of the capsule help dissemination, while the toxic effects are mainly due to the meningococcal endotoxin.
Commensal Neisseria species
Commensal Neisseria species are common in the oral cavity, nose and pharynx, and sometimes in the female genital tract. The taxonomy of the group is confused. The three main species are Neisseria subflava, N. mucosa and N. sicca. The main difference between these and the pathogenic Neisseria species is the ability of the commensal species to grow on ordinary agar at room temperature in the absence of carbon dioxide supplements.
These organisms are essentially non-pathogenic and are almost always found in oral specimens contaminated with saliva or mucosa. Neisseria species are among the earliest colonizers of a clean tooth surface. They consume oxygen during the early plaque formation and facilitate subsequent growth of facultative and obligate anaerobic late colonizers.
Moraxella
Moraxella (formerly Branhamella) are Gram-negative cocci closely related to the non-pathogenic Neisseria species, but asaccharolytic and non-pigmented. They are commensals of the human respiratory tract and are recognized opportunistic pathogens causing meningitis, endocarditis, otitis media, maxillary sinusitis and chronic obstructive pulmonary disease. As the majority of strains produce β-lactamase, they may indirectly ‘protect’ other pathogens and thus complicate antibiotic therapy.
Veillonella
Veillonella species are obligate anaerobic, Gram-negative cocci frequently isolated from oral samples. Three oral species are recognized: Veillonella parvula (the type species), Veillonella dispar and Veillonella atypica.
Veillonella parvula
Gram-negative, small anaerobic cocci. Found in the human oral cavity, mostly in dental plaque, they are considered as ‘benevolent organisms’ in relation to dental caries as they metabolize the lactic acid produced by cariogenic bacteria into weaker acids (acetic and propionic) with a reduced ability to solubilize enamel. No known pathogenic potential.
Parvobacteria
Parvobacteria are so called because of their size (Latin parvus: small). They are a miscellaneous, heterogeneous group of small, Gram-negative bacilli that cause a number of different diseases. They include the following genera:
Of these, Haemophilus and Bordetella spp. are of particular interest, as the former causes significant morbidity in the general population and the latter is the agent of whooping cough. Additionally, Haemophilus spp. and Aggregatibacter spp. are common inhabitants of the oral cavity; the latter being an important periodontopathogen.
Haemophilus spp.
The genus Haemophilus is composed of tiny, non-motile, aerobic, Gram-negative coccobacilli; some are capsulated. One of its major distinguishing features is the requirement of two growth factors:
Haemophilus species cause a variety of diseases, as shown in Table 14.1.
Table 14.1 Some characteristics of Haemophilus spp.a
| Species | Factor requirement | Diseases caused |
|---|---|---|
| H. influenzae | X and V | Acute exacerbation of chronic bronchitis, epiglottitis, meningitis, sinusitis, otitis media, osteomyelitis, arthritis |
| H. parainfluenzae | V | Commensals of the oral cavity and upper respiratory tract; rarely cause disease |
| H. parahaemolyticus | V | |
| H. haemolyticus | X and V | |
| H. aegyptius | X and V | Conjunctivitis |
| H. ducreyi | X | Chancroid (a sexually transmitted disease – a soft sore) |
a H. aphrophilus has been recently renamed as Aggregatibacter aphrophilus hence not included here, although it is a frequent oral commensal.
Haemophilus influenzae
Habitat and transmission
An upper respiratory tract commensal of humans and associated animals, Haemophilus influenzae is a major aetiological agent of upper respiratory tract infections and acute exacerbations of chronic bronchitis. Although not the cause, H. influenzae is a common secondary colonizer of the respiratory tract after a bout of influenza (the agent of which is the influenza virus).
Characteristics
Small, Gram-negative, non-sporing, non-motile rods; predominantly coccobacillary in nature with a few long bacilli and filamentous forms. Virulent strains (for instance, isolated from the cerebrospinal fluid in meningitis) are capsulated.
Culture and identification
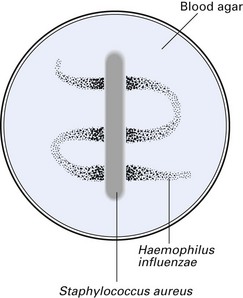
Requires both V factor (NADP) and X factor (haematin) for growth on nutrient agar, but grows on blood-enriched media containing these nutrients. Typically forms large colonies around colonies of other organisms that secrete the V factor – a phenomenon called satellitism. For example, if a blood agar plate (containing the X factor) seeded with H. influenzae is streaked with S. aureus (which secretes the V factor) and incubated overnight at 37°C, the former will grow as large colonies adjacent to the streak of S. aureus (Fig. 14.1).
Pathogenicity
H. influenzae causes four major infections, often accompanied by septicaemia, especially in children and the elderly:
The most important virulence factor of H. influenzae is the polysaccharide capsule. An IgA protease and a factor that causes slowing and incoordination of (respiratory tract) ciliary beating are produced; the outer membrane and endotoxin may contribute to the pathogenesis; there are no known exotoxins. Non-capsulated strains cause exacerbation of chronic bronchitis.
Treatment and prevention
Ampicillin is given for β-lactamase-negative strains. There are many other alternative antibiotics. Prevention by vaccination (Hib) against invasive H. influenzae type b infection has been introduced in some countries. Close contacts of meningitis cases should be given rifampicin as a prophylactic measure.
Bordetella
There are three species in the genus Bordetella, of which Bordetella pertussis, the agent of whooping cough, is the most important.
Bordetella pertussis
Habitat and transmission
Found in the human respiratory tract in diseased individuals; healthy carriage is not known. Spread is by the airborne route.
Characteristics
Short, sometimes oval, Gram-negative rods; fresh isolates may be capsulated. Fastidious growth requirements.
Culture and identification
Requires a special enriched medium for growth, i.e. Bordet–Gengou medium or blood-charcoal agar supplemented with antibiotics. On incubation for 3–5 days at 35°C, under high humidity, iridescent colonies resembling mercury drops appear on Bordet–Gengou medium. Identification is confirmed serologically.
Pathogenicity
Causes whooping cough, especially in preschool children (severe in those under 12 months). The characteristic symptom is the bout of paroxysmal coughs followed by the ‘whoop’ of rapid inhalation after coughing. Virulence factors identified are tracheal cytotoxin, fimbrial antigen and endotoxin.
Treatment and prevention
Erythromycin is the drug of choice for patients and close contacts but antibiotics have little effect on the course of infection, although they may reduce spread and minimize superinfection.
Prevention is by immunization with whole-cell inactivated vaccine, a component of the diphtheria–tetanus–pertussis (DTP) vaccine of childhood. New acellular, subunit vaccines appear effective.
Aggregatibacter actinomycetemcomitans
This relatively new genus Aggregatibacter (formerly called Actinobacillus) includes species isolated from humans and mammals. (Latin aggregare: to come together, aggregate; bacter: bacterial rod; Aggregatibacter: rod-shaped bacterium that aggregates with others).
The only species of this genus routinely isolated from the oral cavity is A. actinomycetemcomitans, so named because it is frequently isolated with Actinomyces spp. from actinomycotic lesions. The reason for this association is unknown. Multiple biotypes and up to six serotypes (a–e) have been described. This species is a major infective agent in particularly aggressive forms of periodontal disease in adolescents (localized aggressive periodontitis) and rapidly destructive periodontal disease in adults.
Habitat and transmission
Primary habitat is unknown but is likely to be subgingival sites of humans and mammals. Infection is endogenous.
Characteristics
Small, short (0.4–1 µm), straight or curved rods with rounded ends. Electron microscopic studies have revealed bleb-like structures on the cell surface, which appear to be released from the cells. Fresh isolates possess fimbriae (lost on subculture).
Culture and identification
Grows as white, translucent, smooth, non-haemolytic colonies on blood agar; grows best aerobically with 5–10% carbon dioxide. Selective media are available for identification; the tryptone-soy-serum-bacitracin-vancomycin agar yields white, translucent colonies with a star-shaped or crossed cigar-shaped internal structure on first isolation, but this is not a consistent finding (Fig. 14.2). There are two phenotypes: smooth and rough. The latter phenotype is related to fimbriation and to the production of hexoseamine-containing exopolysaccharide. Cells from rough colonies grow in broth as granular, autoaggregated cells that adhere to the glass and leave a clear broth. Identified by sugar fermentation and assimilation reactions and acid end products of carbohydrate metabolism.
Pathogenicity
A number of virulence factors, including lipopolysaccharide (endotoxin), a leukotoxin, collagenase, cytolethal distending toxin (cdt), epitheliotoxin-bone resorption inducing factor and a protease-cleaving IgG have all been isolated from A. actinomycetemcomitans. The leukotoxin, in particular, is thought to play a significant role in subverting the host immune response in the gingival crevice. It also has the potency to invade epithelial and vascular endothelial cells in vitro and buccal epithelial cells in vivo. Together with other coagents, A. actinomycetemcomitans is involved in localized aggressive periodontitis and destructive periodontal disease in adults. Also isolated from cases of infective endocarditis, and from brain and subcutaneous abscesses.
Eikenella
Members of the genus Eikenella resemble Haemophilus spp. and are commensals of the human oral cavity and the intestine. Although in the past their presence was linked to periodontal diseases, this has now been disproved. The type species is Eikenella corrodens. These organisms are capnophilic, Gram-negative, short coccobacillary forms that are asaccharolytic. When grown on non-selective media, they corrode the agar surface (hence the name corrodens). Human infection usually results from predisposing factors, such as trauma to a mucosal surface, which allow the organism access to the surrounding tissues; thus, they may cause extraoral infections, including brain and abdominal abscesses, peritonitis, endocarditis, osteomyelitis and meningitis. Also associated with human bites or fist-fight injuries.
Capnocytophaga
The genus Capnocytophaga was created for fusiform species isolated from periodontal pockets, which, unlike Fusobacterium and Bacteroides spp., grow under capnophilic conditions. They have a characteristic ability to glide over routine blood agar (compare ‘swarming’ of Proteus spp.). Species recognized include Capnocytophaga ochracea (type species), Capnocytophaga sputigena, Capnocytophaga gingivalis, Capnocytophaga granulosa and Capnocytophaga haemolytica.
Characteristics
Long, thin fusiform organisms that demonstrate gliding motility seen on bright-field microscopy.
Culture and identification
Facultative anaerobes, but most strains require carbon dioxide for growth. Colonies spread over the agar surface with uneven edges and may be pink, yellow or white. Identification is by gliding characteristic, cell morphology, biochemical reactions and acid end products.
Pathogenicity
Opportunistic pathogens, sometimes associated with gingivitis and other systemic infections in immunocompromised patients; some strains produce an IgAl protease.
Key facts
Duerden B.I., Drasar B.S., editors. Anaerobes in human disease. London: Edward Arnold, 1991.
Greenwood D., Slack R., Peutherer J., editors. Medical microbiology, 16th ed., Edinburgh: Churchill Livingstone, 2003. Chs 24 and 37
Haffajee A.D., Sockransky S.S. Microbial aetiological agents of destructive periodontal diseases. In: Sockransky S.S., Haffajee A.D., editors. Periodontology 2000, Vol. 5. London: Blackwell; 1994:78-111.
Nørskov-Lauritsen N, Kilian M. Reclassification of Actinobacillus actinomycetemcomitans, Haemophilus aphrophilus, Haemophilus paraphrophilus and Haemophilus segnis as Aggregatibacter actinomycetemcomitans gen. nov., comb. nov., Aggregatibacter aphrophilus comb. nov. and Aggregatibacter segnis comb. nov., and emended description of Aggregatibacter aphrophilus to include V factor-dependent and V factor-independent isolates. International Journal of Systematic and Evolutionary Microbiology. 2006;56:2135-2146.
Review questions (answers on p. 352)
Please indicate which answers are true, and which are false.