Fascial fitness
Suggestions for a fascia-oriented training approach in sports and movement therapies
Introduction
Whenever a football player is not able to take the field because of a recurrent calf spasm, a tennis star gives up early on a match due to knee problems, or a sprinter limps across the finish line with a torn Achilles tendon, the problem is most often neither in the musculature nor the skeleton. Instead, it is the structure of the connective tissue – ligaments, tendons, joint capsules, etc. – which has been loaded beyond its capacity (Renström & Johnson 1985; Hyman & Rodeo 2000; Mackey et al. 2008; Counsel & Breidahl 2010). A focused training of the fascial network could be of great importance for athletes, dancers and other movement advocates. If one’s fascial body is well trained, that is to say optimally elastic and resilient, then it can be relied on to perform effectively and at the same time to offer a high degree of injury prevention (Kjaer et al. 2009). Until now, most of the emphasis in sports has been focused on the classic triad of muscular strength, cardiovascular conditioning, and neuromuscular coordination (Jenkins 2005). Some alternative physical training activities – such as Pilates, yoga, Continuum Movement, and martial art – are already taking the connective tissue network into account. Here the importance of the fasciae is often specifically discussed, though modern insights in the field of fascia research have often not been specifically included. We suggest that in order to build up an injury-resistant and elastic fascial body network it is essential to translate current insights in the field of fascia research into a practical training program. Our intention is to encourage physical therapists, sports trainers and movement enthusiasts to incorporate the principles presented in this chapter, and to apply them to their specific context.
Fascial remodeling
A recognized characteristic of connective tissue is its impressive adaptability: when regularly put under increasing yet physiological strain, it changes its architectural properties to meet the demand. For example, through our everyday biped locomotion the fascia on the lateral side of the thigh develops a palpable firmness. If we were instead to spend that same amount of time with our legs straddling a horse, then the opposite would happen, i.e., after a few months the fascia on the inner side of the legs would become more developed and strong (El-Labban et al. 1993).
The varied capacities of fibrous collagenous connective tissues make it possible for these materials to continuously adapt to the regularly occurring strain, particularly in relation to changes in length, strength and ability to shear. Not only the density of bone changes, for example, as happens with astronauts who spend time in zero gravity wherein the bones become more porous (Ingber 2008); fascial tissues also react to their dominant loading patterns. With the help of the fibroblasts, they react to everyday strain as well as to specific training, steadily remodeling the arrangement of their collagenous fiber network (Kjaer et al. 2009). For example, with each passing year half the collagen fibrils are replaced in a healthy body (Neuberger & Slack 1953). The intention of fascial fitness is to influence this replacement via specific training activities which will, after 6 to 24 months, result in a ‘silk-like bodysuit’ which is not only strong but also allows for a smoothly gliding joint mobility over wide angular ranges.
Interestingly, the fascial tissues of young people show stronger undulations within their collagen fibers, reminiscent of elastic springs, whereas in older people the collagen fibers appear as rather flattened (Staubesand et al. 1997). Research has confirmed the previously optimistic assumption that proper exercise loading – if applied regularly – can induce a more youthful collagen architecture, which shows a more wavy fiber arrangement (Wood et al. 1988; Jarniven et al. 2002) and which also expresses a significant increased elastic storage capacity (Fig. 7.24.1) (Reeves et al. 2006). However, it seems to matter which kind of exercise movements are applied: a controlled exercise study using slow-velocity and low-load contractions only demonstrated an increase in muscular strength and volume; however, it failed to yield any change in the elastic storage capacity of the collagenous structures (Kubo et al. 2003).
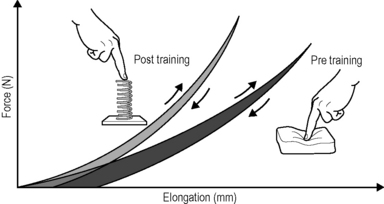
Fig. 7.24.1 • Increased elastic storage capacity.
Regular oscillatory exercise, such as daily rapid running, induces a higher storage capacity in the tendinous tissues of rats, compared with their nonrunning peers. This is expressed in a more spring-like recoil movement, as shown on the left. The area between the respective loading versus unloading curves represents the amount of ’hysteresis’: the smaller hysteresis of the trained animals (grey) reveals their more ’elastic’ tissue storage capacity, whereas the larger hysteresis of their peers signifies their more ’viscoelastic’ tissue properties, also called inertia. Illustration modified after Reeves 2006. Reeves ND, Narici MV, Maganaris CN (2006) Myotendinous plasticity to ageing and resistance exercise in humans. Exp Physiol 91(3): 483–498.
The catapult mechanism: elastic recoil of fascial tissues
Kangaroos can jump much farther than can be explained by the force of the contraction of their leg muscles. Under closer scrutiny, scientists discovered that a spring-like action is behind the unique ability: the so-called ‘catapult mechanism’ (Kram & Dawson 1998). Here, the tendons and the fascia of the legs are tensioned like elastic bands. The release of this stored energy is what makes the amazing jumps possible. The discovery soon thereafter that the same mechanism is also utilized by gazelles was hardly surprising. These animals are also capable of impressive leaping as well as running, though their musculature is not especially powerful. On the contrary, gazelles are generally considered to be rather delicate, making the springy ease of their incredible jumps all the more interesting.
The possibility of high-resolution ultrasound examination made it possible to discover similar orchestration of loading between muscle and fascia in human movement. Surprisingly, it has been found that the fasciae of humans have a similar kinetic storage capacity to that of kangaroos and gazelles (Sawicki et al. 2009). This is not only made use of when we jump or run but also with simple walking, as a significant part of the energy of the movement comes from the same springiness described above. This new discovery has led to an active revision of long-accepted principles in the field of movement science.
In the past, it was assumed that in a muscular joint movement, the skeletal muscles involved shorten and this energy passes through passive tendons, which results in the movement of the joint. This classic form of energy transfer is still true – according to these recent measurements – for steady movements such as bicycling. Here, the muscle fibers actively change in length, while the tendons and aponeuroses scarcely grow longer. The fascial elements remain quite passive. This is in contrast to oscillatory movements with an elastic spring quality, in which the length of the muscle fibers changes little. Here, the muscle fibers contract in an almost isometric fashion (they stiffen temporarily without any significant change of their length) while the fascial elements function in an elastic way with a movement similar to that of a yo-yo (Fig. 7.24.2). It is this lengthening and shortening of the fascial elements that ‘produces’ the actual movement (Fukunaga et al. 2002; Kawakami et al. 2002).
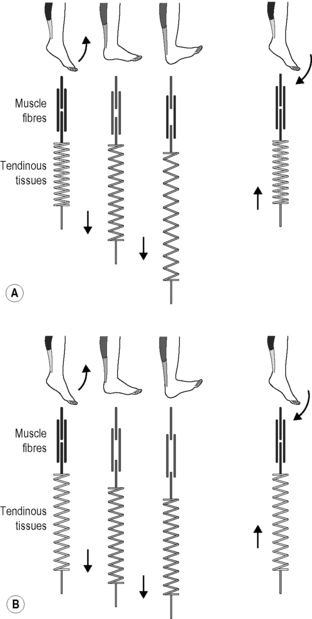
Fig. 7.24.2 • Length changes of fascial elements and muscle fibres in an oscillatory movement with elastic recoil properties (A) and in conventional muscle training (B). The elastic tendinous (or fascial) elements are shown as springs, the myofibers as straight lines above. Note that during a conventional movement (B) the fascial elements do not change their length significantly while the muscle fibers clearly change their length. During movements like hopping or jumping, however, the muscle fibers contract almost isometrically while the fascial elements lengthen and shorten like an elastic yoyo spring. In Kawakami Y, Muraoka T, Ito S, Kanehisa H, Fukunaga T 2002 In vivo muscle fibre behaviour during countermovement exercise in humans reveals a significant role for tendon elasticity. From Kawakami et al. 2002 J Physiol 540 (2): 635–646.
It is of interest that the elastic movement quality in young people is associated with a typical two-directional lattice arrangement of their fasciae, similar to a woman’s stocking (Staubesand et al. 1997). In contrast, as we age and usually lose the springiness in our gait, the fascial architecture takes on a more haphazard and multidirectional arrangement. Animal experiments have also shown that lack of movement quickly fosters the development of additional cross-links in fascial tissues. The fibers lose their elasticity and do not glide against one another as they once did; instead, they become stuck together and form tissue adhesions, and in the worst cases they actually become matted together (Fig. 7.24.3) (Jarvinen et al. 2002). The goal of the fascial fitness training is to stimulate fascial fibroblasts to lay down a more youthful and ‘gazelle-like’ fiber architecture. This is done through movements that load the fascial tissues over multiple extension ranges while utilizing their elastic springiness.
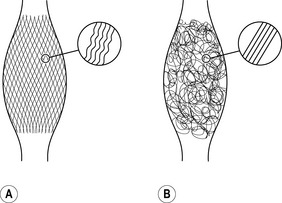
Fig. 7.24.3 • Collagen architecture responds to loading.
Fasciae of young people – shown on the left – express more often a clear two-directional (lattice) orientation of their collagen fiber network. In addition, the individual collagen fibers show a stronger crimp formation. In contrast, fasciae from older persons show a more irregular alignment with less crimp formation. As evidenced by animal studies, application of proper exercise can induce an altered architecture with increased crimp-formation. Lack of exercise, on the other hand, has been shown to induce formation of a multidirectional fiber network and a decreased crimp formation.
Figure 7.24.4 illustrates different fascial elements affected by various loading regimens. Classic weight training loads the muscle in its normal range of motion, thereby strengthening the fascial tissues, which are arranged in series with the active muscle fibers. In addition, the transverse fibers across the muscular envelope are stimulated as well. However, little effect can be expected on extramuscular fasciae as well as on those intramuscular fascial fibers that are arranged in parallel to the active muscle fibers (Huijing 1999).
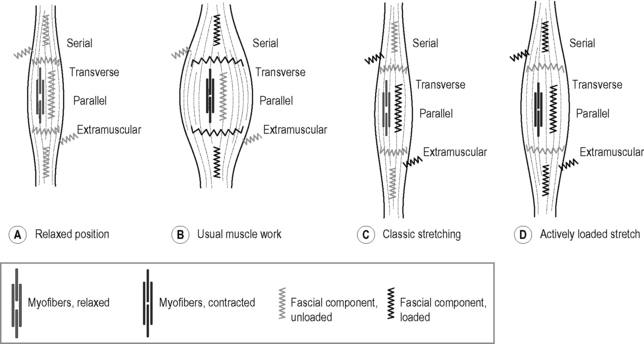
Fig. 7.24.4 • Loading of different fascial components.
A Relaxed position: the myofibers are relaxed and the muscle is at normal length. None of the fascial elements is being stretched. B Usual muscle work: myofibers contracted and muscle at normal length range. Fascial tissues, which are either arranged in series with the myofibers or transverse to them, are loaded. C Classic stretching: myofibers relaxed and muscle elongated. Fascial tissues oriented parallel to the myofibers are loaded as well, as are extramuscular connections. However, fascial tissues oriented in series with the myofibers are not sufficiently loaded, since most of the elongation in that serially arranged force chain is taken up by the relaxed myofibers. D Actively loaded stretch: muscle active and loaded at long end range. Most of the fascial components are being stretched and stimulated in that loading pattern. Note that various mixtures and combinations between the four different fascial components exist. This simplified abstraction therefore serves as a basic orientation only.
Classic Hatha yoga stretches, on the other hand, will show little effect on those fascial tissues which are arranged in series with the muscle fibers, since the relaxed myofibers are much softer than their serially arranged tendinous extensions and will therefore ‘swallow’ most of the elongation (Jami 1992). However, such stretching provides good stimulation for fascial tissues which are hardly reached by classic muscle training, such as the extramuscular fasciae and the intramuscular fasciae oriented in parallel to the myofibers. Finally, a dynamic muscular loading pattern in which the muscle is both activated and extended promises a more comprehensive stimulation of fascial tissues. This can be achieved by muscular activation (e.g. against resistance) in a lengthened position while requiring small or medium amounts of muscle force only. Soft elastic bounces in the end ranges of available motion can also be utilized for that purpose. The following guidelines are developed to make such training more efficient.
Training principles
Here, we make use of the catapult effect described above. Before we perform the actual movement, we start with a slight pretensioning in the opposite direction. This is comparable with using a bow to shoot an arrow; just as the bow has to have sufficient tension in order for the arrow to reach its goal, the fascia becomes actively pretensioned in the opposite direction. In a sample exercise called ‘the flying sword’, the pretensioning is achieved as the body’s axis is slightly tilted backward for a brief moment, while at the same time there is an upward lengthening (Fig. 7.24.5). This increases the elastic tension in the fascial bodysuit and as a result allows the upper body and the arms to spring forward and down like a catapult as the weight is shifted in this direction.


Fig. 7.24.5 • Training example: the flying sword.
A Tension the bow: the preparatory countermovement (pre-stretch) initiates the elastic-dynamic spring in an anterior and inferior direction. Free weights can also be used. B To return to an upright position, the ‘catapulting back fascia’ is loaded as the upper body is briefly bounced dynamically downwards followed by an elastic swing back up. The attention of the person doing the exercise should be on the optimal timing and calibration of the movement in order to create the smoothest movement possible.
The opposite is true for straightening up – we activate the catapult capacity of the fascia through an active pretensioning of the fascia of the back. When standing up from a forward bending position, the muscles on the front of the body are first briefly activated. This momentarily pulls the body even further forward and down and at the same time the fascia on the posterior fascia is loaded with greater tension. The energy which is stored in the fascia is dynamically released via a passive recoil effect as the upper body ‘swings’ back to the original position. To be sure that the individual is not relying on muscle work, but rather on dynamic recoil action of the fascia, requires a focus on timing – much the same as when playing with a yo-yo. It is necessary to determine the ideal swing, which is apparent when the action is fluid and pleasurable.
The Ninja principle
This principle is inspired by the legendary Japanese warriors who reputedly moved as silently as cats and left no trace. When performing bouncy movements such as hopping, running and dancing, special attention needs to be paid to executing the movement as smoothly and softly as possible. A change in direction is preceded by a gradual deceleration of the movement before the turn and a gradual acceleration afterwards, each movement flowing from the last; any extraneous or jerky movements should therefore be avoided (Fig. 7.24.6).


Fig. 7.24.6 • Training example: elastic wall bounces.
Imitating the elastic bounces of a gazelle, soft-bouncing movements off a wall are explored in standing. Proper pretension in the whole body will avoid any collapsing into a ‘banana posture.’ Making the least sound and avoiding any abrupt movement qualities are imperative. Only with the mastery of these qualities can a progression into further load increases – e.g., bouncing off a table or window sill instead of a wall – eventually be explored by stronger individuals. For example, this person should not yet be permitted to progress to higher loads, as his neck and shoulder region already show slight compression on the left picture.
Normal stairs become training equipment when they are used appropriately, employing gentle stepping. The production of ‘as little noise as possible’ provides the most useful feedback – the more the fascial spring effect is utilized, the quieter and gentler the process will be. It may be useful to reflect on the way a cat moves as it prepares to jump. The feline first sends a condensed impulse down through its paws in order to accelerate softly and quietly, landing with precision.
Dynamic stretching
Rather than a motionless waiting in a static stretch position, a more flowing stretch is suggested. In fascial fitness there is a differentiation between two kinds of dynamic stretching: fast and slow. The fast variation may be familiar to many people as it was part of physical training in the past. For several decades, this bouncing stretch was considered to be generally harmful to the tissue, but the method’s merits have been confirmed in recent research. Although stretching immediately before competition can be counterproductive, it seems that long-term and regular use of such dynamic stretching can positively influence the architecture of the connective tissue in that it becomes more elastic when correctly performed (Decoster et al. 2005). Muscles and tissue should first be warmed up, and jerking or abrupt movements should be avoided. The motion should have a sinusoidal deceleration and acceleration shape in each direction turn; this goes along with the perception of a smooth and ‘elegant’ quality of movement. Dynamic, fast stretching has even more effect on the fascia when combined with a preparatory countermovement, as was previously described (Fukashiro et al. 2006). For example, when stretching the hip flexors, a brief backward movement should be introduced before dynamically lengthening and stretching forwards.
The long myofascial chains are the preferred focus when doing slow dynamic stretches. Instead of stretching isolated muscle groups, the aim is finding body movements that engage the longest possible myofascial chains (Myers 1997). This is not done by passively waiting, as in a lengthening classic Hatha yoga pose, or in a conventional isolated muscle stretch. Multidirectional movements, with slight changes in angle are utilized; this might include sideways or diagonal movement variations as well as spiraling rotations. With this method, large areas of the fascial network are simultaneously involved (Fig. 7.24.7).


Fig. 7.24.7 • Training example: the big cat stretch.
A This is a slow stretching movement of the long posterior chain, from the fingertips to the sitbones, from the coccyx to the top of the head and to the heels. The movement goes in opposing directions at the same time – think of a cat stretching its long body. By changing the angle slightly, different aspects of the fascial web are addressed with slow and steady movements. B In the next step one rotates and lengthens the pelvis or chest towards one side (here shown with the pelvis starting to rotate to the right). The intensity of the feeling of stretch on that entire side of the body is then gently reversed. Note the feeling of increased length afterwards.
Proprioceptive refinement
The importance of proprioception for movement control is made clear by the case of Ian Waterman, a man repeatedly mentioned in scientific literature. This impressive man contracted a viral infection at the age of 19 which resulted in a so-called ‘sensory neuropathy.’ In this rare pathology, the sensory peripheral nerves which provide the somatomotor cortex with information about the movements of the body are destroyed, while the motor nerves remain completely intact. This meant than Mr. Waterman could move, but he could not ‘feel’ his movements. After some time, this giant of a man became virtually lifeless. Only with an iron will and years of practice did he finally succeed in making up for these normal physical sensations, a capacity that is commonly taken for granted. He did so with conscious control that primarily relies on visual feedback. He is currently the only person known with this affliction who is able to stand unaided, as well as being able to walk (Cole 1995).
The way Waterman moves is similar to the way patients with chronic back pain move. When in a public place, if the lights unexpectedly go out, he clumsily falls to the ground (see BBC documentary: The man who lost his body, http://bbc-horizon-1998-the-man-who-lost-his-7812922.cooga.net). Springy, swinging movements are possible for him only with obvious and jerky changes in direction. If doing a ‘classic’ stretching program with static or active stretches, he would appear normal. As for the dynamic stretching that is part of our fascial training, he is clearly not capable, as he lacks the proprioception needed for fine coordination. It is interesting to note that the classic ‘joint receptors’ – located in joint capsules and associated ligaments – have been shown to be of lesser importance for normal proprioception, since they are usually stimulated at extreme joint ranges only, and not during physiological motions (Lu et al. 2005; Ianuzzi et al. 2011). On the contrary, proprioceptive nerve endings located in the more superficial layers are more optimally situated, as here even small angular joint movements lead to relatively distinct shearing motions. Recent findings indicate that the superficial fascial layers of the body are, in fact, more densely populated with mechanoreceptive nerve endings than tissue situated more internally (Stecco et al. 2008; Tesarz et al., in Press).
For this reason, we encourage a perceptual refinement of shear, gliding, and tensioning motions in superficial fascial membranes. In doing this, it is important to limit the filtering function of the reticular formation, as it can markedly restrict the transfer of sensations from movements which are repetitive and predictable. To prevent such a sensory dampening, the idea of varied and creative experiencing becomes important. In addition to the slow and fast dynamic stretches noted above, as well as utilizing elastic recoil properties, we recommend (based on experience) inclusion of ‘fascial refinement’ training in which various qualities of movement are experimented with, e.g., extreme slow-motion and very quick micro-movements which may not even be visible to an observer, and large macro-movements involving the whole body. To this end, it is common to place the body into unfamiliar positions while working with the awareness of gravity, or possibly through exploring the weight of a training partner.
The micro-movements are inspired by Continuum Movement (Conrad 2007). Such movement is active and specific and can have effects which are not possible with larger movements. In doing these coordinated fascial movements, it appears possible to specifically address adhesions, for example, between muscle septa deep in the body. In addition, such tiny and specific movements can be used to illuminate and bring awareness to perceptually neglected areas of the body (Fig. 7.24.8). Thomas Hanna uses the label ‘sensory-motor amnesia’ when referring to such places in the body (Hanna 1998).


Fig. 7.24.8 • Training example: octopus tentacle.
With the image of an octopus tentacle in mind, a multitude of extensional movements through the whole leg are explored in slow motion. Through creative changes in muscular activation patterns, the tensional fascial proprioception is activated. This goes along with a deep myofascial stimulation that aims to reach not only the fascial envelopes but also into the septa between muscles. While avoiding any jerky movement, the action of these tentacle-like micro-movements leads to a feeling of flowing strength in the leg.
Hydration and renewal
The video recordings of fascia by Dr. Guimbertau (see Chapter 3.6) have helped our understanding of the plasticity and changing elasticity of the water-filled fascia. This awareness has proven to be especially effective when incorporated into the slow dynamic stretching and fascial refinement work. An essential basic principle of these exercises is the understanding that the fascial tissue is predominantly made up of free-moving and bound-water molecules. During the strain of stretching, the water is pushed out of the more stressed zones, similar to squeezing a sponge (Schleip & Klingler 2007). With the release that follows, this area is again filled with new fluid which comes from surrounding tissue as well as the lymphatic and vascular network. The sponge-like connective tissue can lack adequate hydration at neglected places. The goal of exercise is to refresh such places in the body with improved hydration through specific stretching to encourage fluid movement.
Proper timing of the duration of individual loading and release phases is very important. As part of modern running training, it is often recommended to frequently interrupt the running with short walking intervals (Galloway 2002). There is good reason for this: under strain, the fluid is pressed out of the fascial tissues and these begin to function less optimally as their elastic and springy resilience slowly decreases. The short walking pauses then serve to rehydrate the tissue, as it is given a chance to take up nourishing fluid. For an average beginning runner, for example, the authors recommend walking pauses of 1 to 3 minutes every 10 minutes. More advanced runners with more developed body awareness can adjust the optimal timing and duration of those breaks based on the presence (or lack) of that youthful and dynamic rebound: if the running movement begins to feel and look more dampened and less springy, it is likely time for a short pause. Similarly, if after a brief walking break there is a noticeable return of that gazelle-like rebound, then the rest period was adequate.
This cyclic training, with periods of more intense effort interspersed with purposeful breaks, is recommended in all facets of fascia training. The person training then learns to pay attention to the dynamic properties of their fascial ‘bodysuit’ while exercising, and to adjust the exercises based on this new body awareness. This also carries over to an increased ‘fascial embodiment’ in everyday life. Preliminary anecdotal reports also indicate a preventative effect of a fascia-oriented training in relation to connective tissue overuse injuries.
The use of special foam rollers can be a useful tool for inducing a localized sponge-like temporary tissue dehydration with resultant renewed hydration. However, the firmness of the roller and application of the body weight needs to be individually monitored. If properly applied and including very slow and finely tuned directional changes only, the tissue forces and potential benefits could be similar to those of manual myofascial release treatments (Chaudhry et al. 2008). In addition, the localized tissue stimulation might serve to stimulate and fine-tune possibly inhibited or desensitized fascial proprioceptors in more hidden tissue locations (Fig. 7.24.9).


Fig. 7.24.9 • Training example: fascial release.
The use of particular foam rollers may allow the application of localized tissue stimulations with similar forces and possibly similar benefits as in a manual myofascial release session. However, the stiffness of the roller and application of the body weight needs to be adjusted and monitored for each person. To foster a sponge-like tissue dehydration with subsequent renewed local hydration, only slow-motion, subtle changes in the applied forces and vectors are recommended
Sustainability: the power of a thousand tiny steps
An additional and important aspect is the concept of the slow and long-term renewal of the fascial network. In contrast to muscular strength training in which big gains occur early on and then a plateau is quickly reached wherein only very small gains are possible, fascia changes more slowly and the results are more lasting. It is possible to work without a great deal of strain – so that consistent and regular training pays off. When training the fascia, improvements in the first few weeks may be small and less obvious on the outside. However, improvements have a lasting cumulative effect which, after years, can be expected to result in marked improvements in the strength and elasticity of the global fascial net (Fig. 7.24.10) (Kjaer et al. 2009). As the fascial proprioception becomes refined, improved coordination is probable.
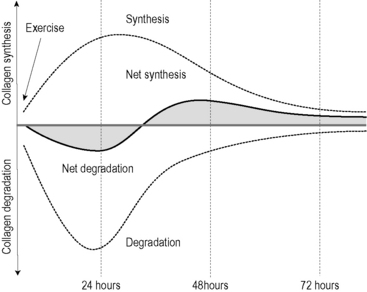
Fig.7.24.10 • Collagen turnover after exercise.
The upper curve shows collagen synthesis in tendons is increasing after exercise. However, the stimulated fibroblasts also increase their rate of collagen degradation. Interestingly, during the first 1–2 days following exercise, collagen degradation outweighs the collagen synthesis, whereas afterwards this situation is reversed. To increase tendon strength, the proposed fascial fitness training therefore suggests an appropriate tissue stimulation 1 to 2 times per week only. While the increased tendon strength is not achieved by an increase in tendon diameter, recent examinations by Kjaer et al. (2009) indicated that it is probably the result of altered cross-link formations between collagen fibers. (From Magnusson et al. 2010, with permission.) Magnusson SP, Langberg H, Kjaer M (2010) The pathogenesis of tendinopathy: balancing the response to loading. Nat Rev Rheumatol 6(5): 262–268
It is suggested that training should be consistent, and that only a few minutes of appropriate exercises, performed once or twice per week, is sufficient for collagen remodeling. The related renewal process will take between 6 months and 2 years and will yield a lithe, flexible and resilient collagenous matrix. For those who do yoga or martial arts, such a focus on a long-term goal is nothing new. For the person who is new to physical training, such knowledge of fascial properties can go a long way in convincing them to train their connective tissues. Of course, fascial fitness training should not replace muscular strength work, cardiovascular training and coordination exercises; instead, it should be thought of as an important addition to a comprehensive training program.
References
Chaudhry H., Schleip R., Ji Z., et al. Three-dimensional mathematical model for deformation of human fasciae in manual therapy. J. Am. Osteopath. Assoc.. 2008;108(8):379–390.
Cole J. Pride and a daily marathon. London: MIT Press; 1995.
Conrad E. Life on land. Berkely: North Atlantic Books; 2007.
Counsel P., Breidahl W. Muscle injuries of the lower leg. Semin. Musculoskelet. Radiol.. 2010;14(2):162–175.
Decoster L.C., Cleland J., Altieri C., et al. The effects of hamstring stretching on range of motion: a systematic literature review. J. Orthop. Sports Phys. Ther.. 2005;35(6):377–387.
EI-Labban N.G., Hopper C., Barber P. Ultrastructural finding of vascular degeneration in myositis ossificans circumscripta (fibrodysplasia ossificans). J. Oral Pathol. Med.. 1993;22(9):428–431.
Fukashiro S., Hay D.C, Nagano A. Biomechanical behaviorof muscle-tendon complex during dynamic human movements. J. Appl.Biomech.. 1993;22(2):131–147.
Fukunaga T., Kawakami Y., Kubo K., et al. Muscle and tendon interaction during human movements. Exerc. Sport. Sci. Rev.. 2002;30(3):106–110.
Galloway J. Galloway’s book on running. Bolinas, California: Shelter Publications; 2002.
Hanna T. Somatics: reawakening the mind’s control of movement, flexibility, and health. Cambridge MA: Da Capo Press; 1998.
Huijing P.A. Muscle as a collagen fiber reinforced composite: a review of force transmission in muscle and whole limb. J. Biomech.. 1999;32(4):329–345.
Hyman J., Rodeo S.A. Injury and repair of tendons and ligaments. Phys. Med. Rehabil. Clin. N. Am.. 2000;11(2):267–288.
Ianuzzi A., Pickar J.G., Khalsa P.S. Relationships between joint motion and facet joint capsule strain during cat and human lumbar spinal motions. J. Manipulative Physiol. Ther.. 2011;34(7):420–431.
Ingber D.E. Tensegrity and mechanotransduction. J. Bodyw. Mov. Ther.. 2008;12(3):198–200.
Jami A. Golgi tendon organs in mammalian skeletal muscles: functional properties and central actions. Physiol. Rev.. 1992;72(3):623–666.
Jarvinen T.A., Jozsa L., Kannus P., et al. Organization and distribution of intramuscular connective tissue in normal and immobilized skeletal muscles. An immunohistochemical, polarization and scanning electron microscopic study. J. Muscle Res. Cell Motil.. 2002;23(3):245–254.
Jenkins S. Sports science handbook: Volume 1: The essential guide to kinesiology, sport & exercise science. Essex, UK: Multi-Science Publishing Co. Ltd.; 2005.
Kawakami Y., Muraoka T., Ito S., et al. In vivo muscle fibre behaviour during countermovement exercise in humans reveals a significant role for tendon elasticity. J. Physiol.. 2002;540(2):635–646.
Kjaer M., Langberg H., Heinemeier K., et al. From mechanical loading to collagen synthesis, structural changes and function in human tendon. Scand. J. Med. Sci. Sports. 2009;19(4):500–510.
Kram R., Dawson T.J. Energetics and biomechanics of locomotion by red kangaroos (Macropus rufus). Comp. Biochem. Physiol. B. Biochem. Mol. Biol.. 1998;120(1):41–49.
Kubo K., Kanehisa H., Miyatani M., et al. Effect of low-load resistance training on the tendon properties in middle-aged and elderly women. Acta Physiol. Scand.. 2003;178(1):25–32.
Lu Y., Chen C., Kallakuri S., et al. Neural response of cervical facet joint capsule to stretch: a study of whiplash pain mechanism. Stapp Car Crash J.. 2005;49:49–65.
Mackey A.L., Heinemeier K.M., Koskinen S.O., et al. Dynamic adaptation of tendon and muscle connective tissue to mechanical loading. Connect. Tissue Res.. 2008;49(3):165–168.
Magnusson S.P., Langberg H., Kjaer M. The pathogenesis of tendinopathy: balancing the response to loading. Nat. Rev. Rheumatol.. 2010;6(5):262–268.
Myers T.W. The ‘anatomy trains’. J. Bodyw. Mov. Ther.. 1997;1(2):91–101.
Neuberger A., Slack H. The metabolism of collagen from liver, bones, skin and tendon in normal rats. Biochem. J.. 1953;53:47–52.
Reeves N.D., Narici M.V., Maganaris C.N. Myotendinous plasticity to ageing and resistance exercise in humans. Exp. Physiol.. 2006;91(3):483–498.
Renström P., Johnson R.J. Overuse injuries in sports. A review. Sports Med.. 1985;2(5):316–333.
Sawicki G.S., Lewis C.L., Ferris D.P. It pays to have a spring in your step. Exerc. Sport Sci. Rev.. 2009;37(3):130–138.
Schleip R., Klingler W. Fascial strain hardening correlates with matrix hydration changes. In: Findley T.W., Schleip R., eds. Fascia research – basic science and implications to conventional and complementary health care. Munich: Elsevier GmbH; 2007:51.
Staubesand J., Baumbach K.U.K., Li Y. La structure fine de l’aponévrose jambie˘re. Phlebol. 1997;50:105–113.
Stecco C., Porzionato A., Lancerotto L., et al. Histological study of the deep fasciae of the limbs. J. Bodyw. Mov. Ther.. 2008;12(3):225–230.
Tesarz, J., Hoheisel, U., Wiedenhöfer, et al., Sensory innervation of the thoracolumbar fascia in rats and humans. Neuroscience, in Press. doi:10.1016/j.neuroscience.2011.07.066
Wood T.O., Cooke P.H., Goodship A.E. The effect of exercise and anabolic steroids on the mechanical properties and crimp morphology of the rat tendon. Am. J. Sports Med.. 1988;16(2):153–158.