Chapter 18 Breast
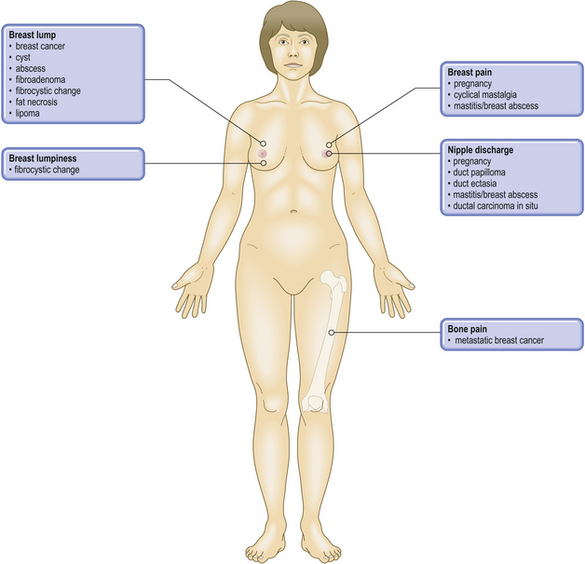
COMMON CLINICAL PROBLEMS FROM BREAST DISEASE
Pathological basis of breast signs and symptoms
| Sign or symptom | Pathological basis |
|---|---|
| Lump | |
| Fibrosis, epithelial hyperplasia and cysts in fibrocystic change | |
| Neoplasm or solitary cyst | |
| Benign neoplasm (usually fibroadenoma) | |
| Invasive neoplasm (carcinoma) | |
| Skin features | |
| Impaired lymphatic drainage due to carcinoma | |
| Invasion of skin by carcinoma | |
| Increased blood flow due to inflammation or tumour | |
| Nipple | |
| Tethering by invasive carcinoma | |
| Paget’s disease of nipple or eczema | |
| Breast pain | |
| Benign breast changes | |
| Inflammatory lesion (e.g. mastitis) | |
| Microcalcification (on mammography) | Dystrophic calcification associated with benign changes, e.g. cysts, sclerosing adenosis, or in situ or invasive carcinoma |
| Axillary node enlargement | Often due to metastatic breast carcinoma |
| Bone pain or fracture | Possibly due to metastatic breast carcinoma or associated with hypercalcaemia |
NORMAL STRUCTURE AND FUNCTION
The physiological and pathological changes in a woman’s breasts vary during different phases of her life. This is due to the variations in hormone levels that occur before, during and after the period of reproductive life; hormones are important in the regulation of growth, development and function of the breast.
Development
Before puberty, the breast consists of a few ducts which are connected to the nipple and open to the surface, but there are no glandular structures. Shortly before menarche, lengthening and branching of the ducts occurs and the terminal buds appear. There is increased volume of fat and connective tissue. With the onset of menses, further growth takes place and continues until at least the age of 25, unless accelerated by the intervention of pregnancy.
Developmental abnormalities
Failure of breast development in the female is very rare; in some cases it is due to ovarian agenesis (Turner’s syndrome). Accessory nipples are the commonest abnormality; these can occur anywhere along the ‘milk line’, from axilla to groin. Juvenile hypertrophy is characterised by rapid and disproportionate breast growth during puberty; it can cause psychological distress and may warrant surgical reduction.
Hormonal regulation
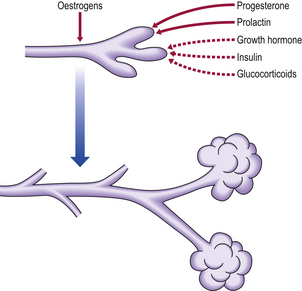
Development of the breast requires the co-ordinated action of many hormones. The precise role of each hormone is difficult to determine because they may have both growth and secretory effects, and may regulate the activity of each other (Fig. 18.1).
Structure
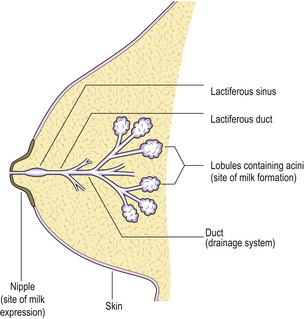
The main function of the breast is the production and expression of milk (Fig. 18.2).
Lobules
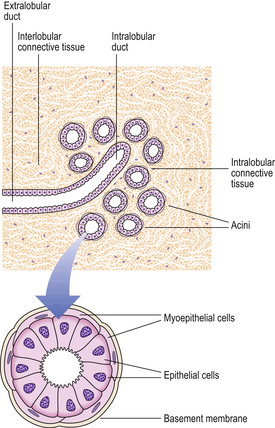
The lobules are the secretory units of the breast. Each lobule consists of a variable number of acini, or glands, embedded within loose connective tissue and connecting to the intralobular duct (Fig. 18.3). Each acinus is composed of two types of cell, epithelial and myoepithelial. The epithelial cells are secretory. Although synthesising milk only during the later stages of pregnancy and post-partum, they continuously secrete a variety of glycoproteins into the glandular lumens. They are surrounded by myoepithelial cells which contact with the basement membrane and may directly or indirectly control luminal cell function. The intralobular duct connects with the extralobular duct and this, together with the lobule, is called the terminal ductal lobular unit.
Ducts
The extralobular ducts within the same area link together to form subsegmental ducts, which link in turn to form segmental ducts. These drain into the lactiferous ducts and sinuses (Fig. 18.2) which empty on to the surface of the nipple through separate orifices. There are 15 to 20 lactiferous ducts, each draining a segment of breast. The ducts are lined by epithelial cells surrounded by myoepithelial cells. The connective tissue in which they lie is denser than that of the lobules, and they are surrounded by elastic tissue which helps in the drainage function of the ducts.
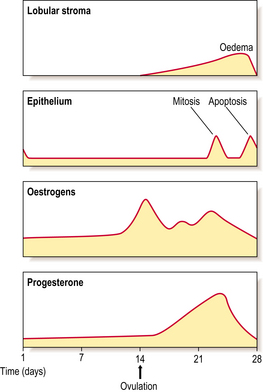
Cyclical variations
The breast undergoes minor changes during each menstrual cycle but these will vary if there is a failure of ovulation or if pregnancy intervenes. The breast is sensitive to changes in the levels of sex steroids during the different phases (Fig. 18.4). The lobular stroma becomes oedematous during the secretory phase, due to the effects of oestrogens, and this accounts for the breast fullness often felt in the premenstrual phase. An increase in the number of cells in mitosis occurs at days 22–24 of the cycle, coincident with the high peaks of oestrogen and progesterone; however, the numbers are never very high. A loss of cells occurs by apoptosis (Ch. 5) at the end of the cycle, due to a fall in hormone levels, so that an overall balance is maintained. In view of the changes that can occur in the breast in the second half of the menstrual cycle, it is better to examine clinically the breasts of a pre-menopausal woman in the first half of the cycle.
Pregnancy and lactation
During pregnancy, the lobules undergo controlled proliferation and enlargement in preparation for the synthetic and secretory activity of lactation. By the third trimester the number of acini in each lobule and the overall size of the lobules have markedly increased. The epithelial cells have become differentiated and they synthesise and secrete milk (Fig. 18.5). The various components of milk (casein, alpha-lactalbumin and milk fat globule membranes derived from the luminal surface of breast cells) are useful markers of the state of differentiation of breast cells, and because of this they have been extensively studied in breast disease.
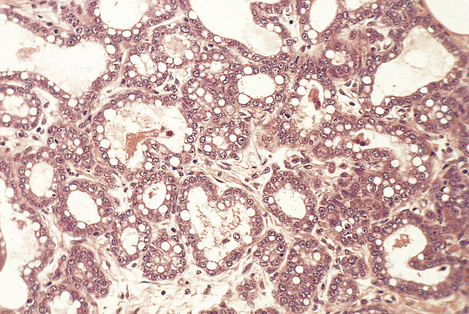
Fig. 18.5 Lactation. Breast histology from a woman 30 weeks pregnant, showing the acini lined by cells containing secretory vacuoles, and with secretions in their lumens.
Oestrogens, progesterone and prolactin, together with other hormones shown in Figure 18.1, are important in the development of the breast during pregnancy; however, once delivery occurs, the levels of sex steroids fall and it is prolactin that is necessary for the initiation of lactation. When breast feeding ceases there is a rapid involution of the differentiated lobular structure, and the breast returns to the pre-pregnancy structure.
Involution
Changes occur in the breast with increasing age; these involutional changes relate to the altered sex steroid levels that accompany decreasing ovarian function. The connective tissue of the lobules changes from a loose to a dense structure, the basement membranes around acini become thicker, and the lining cells of the acini are lost. These changes start in the pre-menopausal period and continue past the menopause; they often occur at an uneven rate, producing clinically palpable lumps. In elderly women, the major component of the breast is adipose tissue.
CLINICAL FEATURES OF BREAST LESIONS
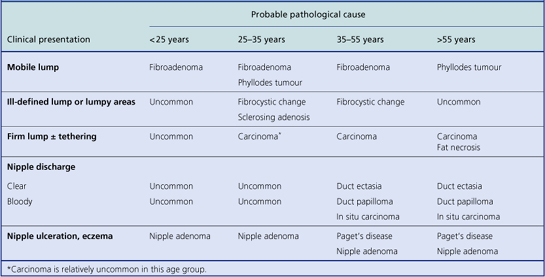
Most pathological lesions of the breast present as a lump or lumps. These can vary in their nature depending on their cause: well-circumscribed or ill-defined; single or multiple small nodules; soft or firm; mobile or attached to skin or underlying muscle. These features assist in the clinical distinction between benign breast lesions and breast carcinomas, but they are relatively weak discriminators on their own. Below the age of about 35, benign breast lumps are much more common than carcinomas. Most women with breast cancer are peri- or post-menopausal. The most likely type of lesion will vary with the age of the patient, although overlaps occur (Table 18.1). However, there can be exceptions and histological examination is mandatory for a definite diagnosis.
Table 18.1The probable pathological causes of presenting clinical lesions at different ages in women
Physiological conditions can be confused with, or mimic, pathological conditions. A degree of tenderness and swelling of the breast in the premenstrual phase is common. Some women have naturally ‘lumpy’ breasts and this may become exaggerated in this phase of the menstrual cycle. Uneven proliferation of the glandular substance during pregnancy, and irregular involution after pregnancy and during and after the menopause, can result in lumps that are the outcome of physiological and not pathological events. Other manifestations of a pathological lesion within the breast are discharge from the nipple, eczema and ulceration of the skin of the nipple.
DIAGNOSTIC METHODS
Several methods are used to investigate breast lesions. The aim is to get a diagnosis and either negate surgery or be able to discuss management with the patient (i.e. pre-operative diagnosis). They include:
Mammography and ultrasonography
Radiography of the breasts (mammography) is used to help in the diagnosis of both palpable and impalpable lesions, looking for deformities and/or calcifications. This technique forms the basis of screening programmes. It is of less value in younger women due to the dense nature of their breasts. Ultrasound imaging is of value for younger patients for defining the edge of lesions, e.g. cysts. Both investigations should be undertaken before a needle is inserted.
Fine-needle aspiration cytology
This technique is used in the clinic. A needle is inserted into the lump or area in the breast with the abnormality (guided if necessary by ultrasonography or mammography). Cells are aspirated, and after staining are examined by a pathologist; if the sample is adequate a diagnosis can be made. Women with benign conditions can be reassured and surgery may not be necessary. It is possible to prepare slides and a report while the patient is in the clinic.
Core biopsy
A core of tissue is removed from the lump/lesion using a biopsy needle, under local anaesthetic. This can be done under mammographic control and is of particular value for diagnosis of calcifications seen on mammograms. It can also be ultrasonographically guided. Core biopsies are of value when sufficient cells cannot be obtained or a diagnosis cannot be made on cytology; for calcifications; if a patient has an obvious cancer and is going to be treated with chemotherapy prior to surgery, as marker studies can be done. Some centres prefer core biopsies to cytology.
Other
If a diagnosis cannot be made then the patient will have a larger piece of tissue removed, under general anaesthetic. It is uncommon nowadays for frozen sections to be performed. This involves a small sample of tissue being frozen, sectioned, stained and interpreted by a pathologist at the time of surgery, while the patient is under general anaesthetic. It is only considered if a diagnosis cannot be made pre-operatively and there are medical reasons for the patient not to have two anaesthetics.
Screening for breast cancer
In several developed countries with a high incidence of breast cancer, such as the UK, screening programmes for the detection of early breast cancer have been introduced. Trials in Sweden and the USA strongly suggest that women whose cancers have been detected by regular mammographic screening have an increased survival rate. This is because the tumours are detected when they are either pre-invasive (in situ carcinoma) or invasive but small, with less risk of metastasis. Unscreened women present when the tumour has grown to a size sufficient to be felt, at which stage there is a higher probability of metastases.
In the UK, women between the ages of 50 and 69 are invited to attend for breast screening by mammography every 3 years. Suspicious features on the mammographic image, such as microcalcification and localised densities, are further investigated by ultrasonography and clinical examination, with histology of biopsy samples, and very occasionally cytology, providing the definitive diagnosis. Impalpable lesions detected in this way require an X-ray-directed guidewire to be inserted into them before surgery to help the surgeon find the right area.
Besides being smaller, the invasive tumours have a higher frequency of being of a more favourable histological type and lower grade. This, along with the lower incidence of lymph node metastasis, will contribute to the improved prognosis. The surgery for these early lesions is more likely to be conservative.
The greater density of the pre-menopausal breast means that mammography may be less reliable for screening women under 50 years.
INFLAMMATORY CONDITIONS
Acute pyogenic mastitis
Acute pyogenic mastitis is a painful acute inflammatory condition which usually occurs in the first few weeks after delivery, and Staphylococcus aureus is the commonest organism. The usual portal of entry is a crack in the nipple, although persistence of the keratotic plug at the orifice of a duct may be a factor. The organisms spread via the lymphatics, and the infection tends to be confined to one segment of the breast, resulting in localised swelling and erythema. The infection can spread to other segments and, if Streptococcus pyogenes is the causative organism, a more widespread inflammation occurs with systemic symptoms. If antibiotics are given but there is inadequate drainage, a localised breast abscess will result.
Other infections
Tuberculosis
This is rare and usually results from haematogenous spread. The infection results in a fibrocaseous mass with the formation of sinuses, although a marked fibrous reaction can occur, giving a firm mass that will mimic a carcinoma.
Actinomycosis
Also rare, actinomycosis can be due to extension of infection from the lung through the thoracic cage, or occur as a primary infection. The usual presentation is as a hard lump beneath the nipple, which may be painful, but with no temperature change, so mimicking a tumour. It results in abscess formation, within which are the bacterial colonies.
Mammary duct ectasia
Mammary duct ectasia involves the larger ducts within the breast but, in severe cases, can also extend to the smaller interlobular ducts. It occurs predominantly in women in the second half of reproductive life and after the menopause, and mild degrees of the condition are often an incidental finding in breast tissue excised for other conditions. Severe forms, in which it is the primary presenting condition, are less frequent. Severe cases can be mistaken clinically for a carcinoma as there may be a discharge from the nipple which may be bloodstained. Fibrosis around the ducts may result in nipple retraction, and there may be a firm palpable mass. However, mammary duct ectasia is a purely inflammatory condition with no relationship to malignancy.
The aetiology is unknown but the affected women are usually parous. The ducts are dilated and filled with white–green viscid matter; this material may be discharged from the nipple. The matter can usually be seen with the naked eye in excised tissue. The tissue around the ducts contains lymphocytes, plasma cells and macrophages, with a significant degree of fibrosis. Due to the inflammatory reaction, the condition is sometimes known as periductal mastitis.
Fat necrosis
Trauma, e.g. seat belt injury, is thought to be the cause of fat necrosis, although a history is not always obtained. It is more frequent in obese women and after the menopause, when the breast has a proportionally greater amount of adipose tissue. It usually presents as a discrete lump and can therefore mimic a carcinoma clinically.
Macroscopically, the tissue is yellow and haemorrhagic, with flecks of calcification. Fibrous tissue is also present, the amount depending on the duration of the condition.
Histologically, the appearances are the same as those of any adipose tissue that undergoes necrosis (Ch. 6): collections of macrophages and giant cells containing lipid material may be seen, and there is an associated reaction with lymphocytes, fibroblasts and small vascular channels. The necrotic fat acts as a persistent irritant, resulting in a chronic inflammatory process and hence fibrous tissue formation.
Similar foreign body reactions can occur in the breast around ruptured prosthetic implants, in which silicone fluid is frequently used; a very dense fibrous tissue reaction can result, causing considerable distortion.
PROLIFERATIVE CONDITIONS OF THE BREAST
Proliferative conditions of the breast include a wide variety of morphological changes with consequently varied clinical features; because of this there has been much confusion about the terminology and significance of these conditions.
Fibrocystic change
The commonest proliferative condition of the breast is fibrocystic change. Although benign and non-neoplastic, it is important because:
Terminology
The old term for proliferative conditions of the breast was ‘chronic mastitis’; this is incorrect, as these are not inflammatory conditions, but the name may have arisen because of the tenderness that can occur in some cases. Other names include fibroadenosis, epithelial hyperplasia, fibrocystic disease, cystic hyperplasia and mammary dysplasia. Since some of the features are similar to physiological changes the term ‘fibrocystic change’, rather than ‘disease’, is now used.
The terms fibroadenosis and epithelial hyperplasia describe the proliferative changes that occur in the condition (see below), and are appropriate terms for the changes that occur in the 30–45-year age group. Fibrocystic change and cystic hyperplasia are descriptive of the changes that occur from 40–45 years to the menopause, when cysts are more prominent. The term ‘mammary dysplasia’ is not really correct, as true dysplasia occurs only in a few cases.
Incidence
Estimates indicate that at least 10% of women develop clinically apparent benign proliferative breast disease, although breast tissue from women at postmortem shows such changes to be present in 50% or more, suggesting that lesser degrees of change are much more common.
Aetiology and pathogenesis
Although benign proliferative breast disease is not uncommon, the aetiology is poorly understood. There is no doubt that ovarian hormones participate in its causation but the means by which the changes are produced are still obscure.
The fact that the incidence of benign proliferative changes increases as the menopause gets nearer, and that failure of ovulation also increases in this time period, suggests that the relative imbalance between oestrogen and progesterone in each menstrual cycle could be an important aetiological factor. The disturbance may involve interaction of the pituitary and the ovaries. Alternatively, the fault may lie in the responsiveness of the breast tissue to the hormonal influences. Not all parts of the breast are equally affected by the hormonal changes occurring in each menstrual cycle, and this may account for the focal nature of the changes.
Cystic change is considered to be due to an imbalance between epithelial hyperplasia, together with ductal and lobular dilatation, that occurs with each menstrual cycle, and subsequent regressive changes. The cystic dilatation thus occurs because of a distortion of cyclical changes rather than as a consequence of obstruction, which is the usual cause in other organs.
Clinical and gross features
Proliferative lesions and their associated tissue responses generally occur between the ages of 30 and 55, with a marked decrease in incidence after the menopause. The incidence reaches a maximum in the years just before the menopause (Fig. 18.6).
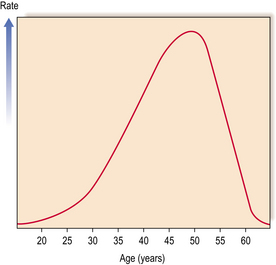
Fig. 18.6 Incidence rates of benign proliferative breast changes occurring in women at different ages.
The clinical features tend to vary with the age of the patient and the underlying pathological changes. In younger women, there is usually a diffuse granularity in one or more segments of the breast, with nodules up to 5 mm in diameter. The area may be tender, particularly in the premenstrual period. In women nearer the menopause, there is usually an ill-defined rubbery mass. The finding of discrete swelling indicates the presence of cysts. If fibrosis is a component of the proliferative lesion, the lump will be firm and therefore more difficult to differentiate clinically from carcinoma.
Surgery for benign conditions is now uncommon. If undertaken it is more common to find nodules of soft pink or grey tissue, up to 3 mm in diameter in younger women, whereas in women nearer the menopause cysts are frequently seen. These cysts can vary in size from 2 to 20 mm (Fig. 18.7) and, rarely, a solitary large cyst can be seen. The small cysts are often multiple. They frequently have a dark blue surface and, on opening, contain clear, yellowish or blood-stained fluid. The intervening tissue is usually firm due to the increase in fibrous tissue but the softer foci of epithelial proliferation can be seen and felt.
Histological features
A variety of histological changes can occur (Fig. 18.8). These are:
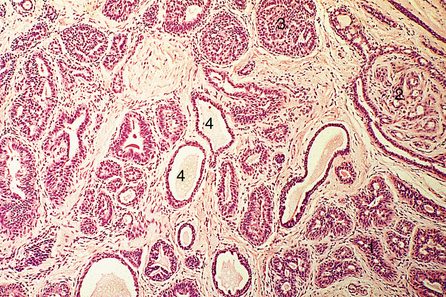
Fig. 18.8 Fibrocystic change. There is adenosis (1), papilloma formation (2), epithelial hyperplasia (3) and small cysts (4).
An individual woman may show one, some or all of these changes. However, the types of change do tend to vary with the age of the patient.
Adenosis
Adenosis is enlargement of the lobules which contain many, up to hundreds, of acini. In other respects they are structurally normal. The term is often used to refer to blunt duct adenosis in which the acini of the lobules are larger than normal and are lined by cells that are increased in size and may also be more numerous, although the acinar lumen is always clearly seen. The lobular stroma may also increase. The changes are not confined to the epithelium and can involve the surrounding myoepithelium. Such areas correspond to the grey–pink nodules seen macroscopically and the fine nodules felt clinically.
Sclerosing adenosis
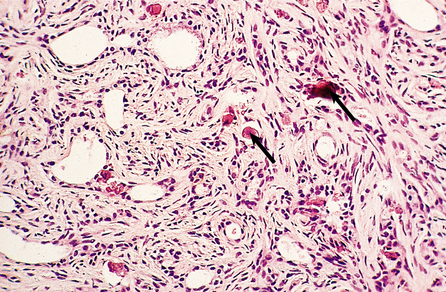
In sclerosing adenosis there is lobular proliferation but the acini become distorted. The proliferation involves both epithelium and myoepithelium, but the latter tends to predominate. Large amounts of collagen can intervene between the glandular components, although the extent of this varies both within the same breast and between patients (Fig. 18.9). Due to the collagen component these lesions can mimic carcinomas clinically, and on mammograms where the associated calcification can be seen.
Epithelial hyperplasia
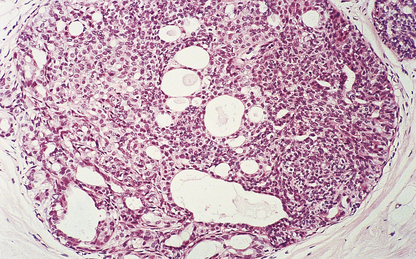
Epithelial hyperplasia, previously called epitheliosis, is the proliferation of epithelial cells that occurs in the small interlobular ducts, the intralobular ducts and the acini, resulting in a solid or almost solid mass obliterating the lumens (Fig. 18.10).
Papillomatosis
Papillomatosis comprises simple papillary processes projecting into the lumens of dilated ducts or small cysts. The papillae have a fine connective tissue core and are covered by one or two layers of epithelium; they may undergo branching.
Cysts
Cysts develop through dilatation of the acini of the lobules andthe terminal ducts. These cysts may remain small, or enlarge to sizes up to 20–30 mm. They may be fairly evenly distributed, with little variation in size, or show quite marked variation in size, shape or number. They may be lined by simple cuboidal or flattened epithelium, or focal proliferative change may occur. Occasionally cysts can rupture, causing an inflammatory reaction.
Apocrine metaplasia
Frequently the cysts, both large and small, are lined entirely or partly by cells that resemble the epithelium of the apocrine sweat glands. This condition is called apocrine metaplasia. The lining cells are large columnar cells with pink-staining (eosinophilic) cytoplasm, hence the alternative name ‘pink cell metaplasia’. It has no special clinical or prognostic significance.
Fibrosis
Fibrosis can occur in association with the various proliferative conditions, or as an isolated lesion. When associated with proliferative conditions it is probably due to the hormonal imbalances causing changes in the typical loose connective tissue of the lobules, making it denser with fewer glycosaminoglycans. The solitary form of fibrosis produces a poorly defined area of rubbery consistency consisting of dense connective tissue with few atrophic epithelial areas. This condition is found mainly in women with a clear history of hormone imbalance.
Atypical hyperplasia
The epithelial hyperplasia that can result in total or partial occlusion of the acini and small ducts may sometimes show abnormalities of cellular growth, with disordered orientation of cells, nuclear pleomorphism and occasional mitotic figures. This is termed either atypical ductal or atypical lobular hyperplasia, depending on its situation. It is important for the pathologist to identify these cellular changes (see below).
Lesions in women aged 30–45 years
In the 30–45-year age group, lesions are more likely to consist of areas of adenosis, possibly with epithelial hyperplasia, and a mild degree of fibrosis. Sclerosing adenosis may also be present. Microcysts with apocrine metaplasia start to develop in the late thirties, but are generally not a major feature. Between 40 and 45 years the changes may be predominantly proliferative, with adenosis and epithelial hyperplasia, or may be more cystic.
Lesions in women aged 45–55 years
Cysts are the more prominent feature in this age group and can be quite large. The terms ‘blue domed cyst’ and ‘Bloodgood’s cyst’ used to be applied. Apocrine metaplasia is often present. Proliferative features, such as adenosis, epithelial hyperplasia and papillomatosis, can be seen, and fibrosis is quite common.
Radial scars
Radial scars are benign focal lesions commonly detected by mammography. They are stellate fibrous structures with foci of ductal epithelial proliferation. Their structure mimics radiologically the appearance of invasive carcinoma. When larger than 10 mm, they are named complex sclerosing lesions.
Significance of proliferative lesions
Clinically, the presence of a lump can cause anxiety in the patient, who may believe it is a cancer when it is benign; diagnostic investigations can reassure her.
Previous studies have shown that up to 70% of women who undergo breast biopsy for benign fibrocystic change are not at an increased risk of developing cancer. However, if the biopsy contains areas of atypical hyperplasia, the woman has a risk of developing cancer five times higher than that of a woman with non-proliferative lesions, and the risk increases if there is a family history of breast cancer. Cysts alone do not appear to increase the risk.
Lesions in men: gynaecomastia
The breast tissue in men contains only ductular structures with no evidence of acini; it is similar in appearance to the pre-pubertal female breast.
Gynaecomastia is benign enlargement of the male breast tissue. The breast may resemble that of a young adolescent female in appearance and consistency, or there may be a firm, mobile disc beneath the nipple. The condition is unilateral in 75% of cases. The ducts are dilated and there is a variable degree of epithelial proliferation. The stroma around the ducts is often oedematous and myxoid, but in longstanding cases the stroma becomes dense and hyalinised (Fig. 18.11).
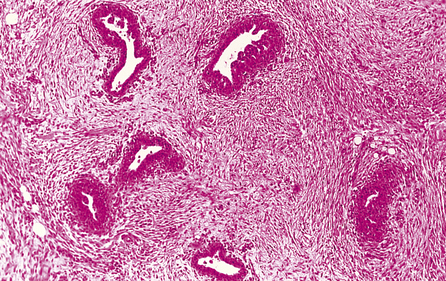
Fig. 18.11 Gynaecomastia. Male breast in which the ducts are lined by an increased number of cells, and are surrounded by loose connective tissue.
Gynaecomastia occurs most commonly in adolescence and in older age groups. In both of these groups it is probably due to some hormonal effects relating to oestrogens, possibly a result of endocrine disturbances such as hyperthyroidism, pituitary disorders and tumours of the adrenals and testis. Both of the latter can secrete oestrogens. In the older age group, diethylstilbestrol therapy of prostatic carcinoma can cause gynaecomastia. Other causes include Klinefelter’s syndrome, malnutrition and cirrhosis, as well as the drugs chlorpromazine and spironolactone, and digitalis therapy.
BENIGN TUMOURS
Unlike the situation in other glandular tissues, the commonest type of benign tumour of the breast is a combined product of both connective tissue and epithelial cells; purely epithelial tumours are less frequent.
The benign breast tumours comprise:
Fibroadenoma
Fibroadenomas are the commonest type of benign tumour of the breast, and are the commonest primary tumour in younger age groups. In a study in New York, fibroadenomas were seen with a quarter of the frequency of carcinomas, but six times more frequently than duct papillomas. However, not all fibroadenomas are excised, so their actual frequency may be higher.
The greatest incidence of fibroadenomas is in the third decade, although they can occur at any time from puberty onwards. The tumours are usually solitary, although some women do develop multiple fibroadenomas.
Fibroadenomas arise from the breast lobule, from both the loose connective tissue stroma and the glands. As they are mixed tumours, fibroadenomas will undergo some of the same hormonally induced changes as the surrounding breast. Thus, during pregnancy the glands will show lactational changes, and in older women the stroma will become more dense and fibrous. During pregnancy, fibroadenomas may grow rapidly in size, but this is due to hormonal effects and is not a sign of malignancy.
Gross appearance
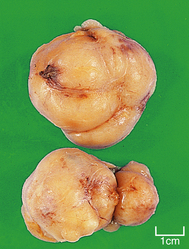
Fibroadenomas are well circumscribed with a lobulated appearance (Fig. 18.12), and range in size from 10 to 40 mm in diameter, although larger tumours can occur in juvenile fibroadenoma (see below). The surrounding breast tissue can become compressed, but the tumour is not tethered; this lack of fixation accounts for its mobility on clinical examination, and the nickname of ‘breast mouse’. In young women, the tumours are soft and have a slightly gelatinous cut surface due to the loose connective tissue component; however, in older women they tend to be firmer as the connective tissue becomes more fibrous and sometimes calcified.
Histology
Fibroadenomas show duct-like structures or elongated and thinned ductular structures associated with overgrown connective tissue masses (Fig. 18.13). Fibroadenoma does not progress to malignancy, although very occasionally a tumour, such as lobular carcinoma, will involve a fibroadenoma.
Duct papilloma
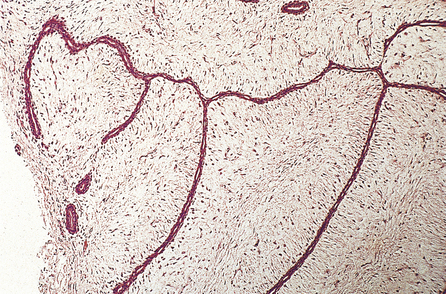
Duct papillomas are considerably less frequent than fibroadenomas. They also differ in several other respects. Although they can occur in the young and the elderly, they more frequently arise in middle-aged women. Duct papillomas are the commonest cause of nipple discharge. About 80% of patients present with a discharge, which is often blood-stained, and a mass can often be felt. The tumours arise from ductal epithelium.
Duct papillomas arise as a solitary lesion within a large duct, up to 40 mm from the nipple. They appear either as an elongated structure extending along a duct, or as a spheroid which causes distension of the duct, making it cyst-like. The tumours have soft, pink or white outgrowths except when haemorrhage has occurred, in which case the surface will be brown from altered blood. Duct papillomas consist of branching fibrovascular cores covered by epithelium, which is cytologically benign (Fig. 18.14). Solitary duct papillomas are not premalignant; there is no increased risk of carcinoma.
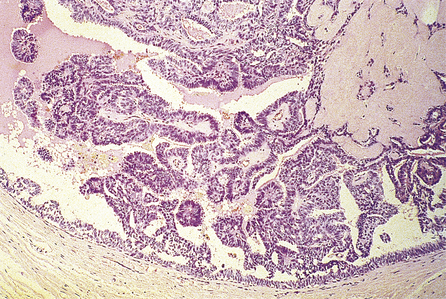
Fig. 18.14 Duct papilloma. A duct containing finger-like projections covered by a layer of epithelial and underlying myoepithelial cells, with a fibrous core.
There is a rare condition in which multiple ductal papillomas occur, but these arise in the smaller ducts, away from the nipple, and so present as a mass rather than as nipple discharge. These tend to occur in a younger age group than do solitary papillomas and there is an increased risk of carcinoma developing.
Adenomas
Adenomas are much rarer than fibroadenomas and duct papillomas. Tubular adenomas are well-circumscribed tumours between 10 and 40 mm in diameter, occurring mainly in women in their early twenties. They are composed of closely packed, uniform tubular structures with little connective tissue in between; hence the only tumorous component is the glands.
Lactating adenomas are tubular adenomas that undergo secretory changes during pregnancy.
Nipple adenomas occur as a nodule under the nipple, usually less than 15 mm in diameter, in women of any age. The overlying skin is often ulcerated, and there may be a blood-stained discharge, so that clinically nipple adenomas may be mistaken for Paget’s disease. They are well circumscribed, and contain small and larger ducts filled with masses of cells and surrounded by a dense stroma.
BREAST CARCINOMA
In North America, north-west Europe and Australia, breast cancer is the commonest type of malignancy in women. In the UK it accounts for 31% of all cancers, with 80% occurring in women aged above 50years. It is the commonest cause of death among women in the 35–55-year age group. Recent reports (2004) show there are 44659 new cases each year in the UK. It is estimated, in the high-risk areas, that any individual woman has a 1 in 9 chance of developing the disease in her lifetime.
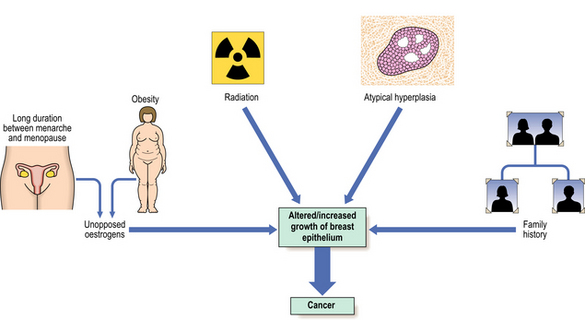
Many risk factors have been identified, and these, together with advances in the analysis of genetic and hormonal factors, have resulted in several aetiological hypotheses (Fig. 18.15). An understanding of these can help in the development of programmes directed towards the prevention of breast cancer. Schemes aimed at the early detection of breast cancer have been introduced in several countries.
Risk factors
The risk factors identified to date are:
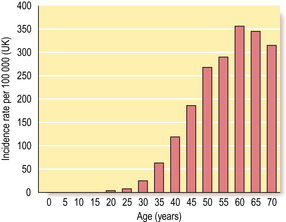
Female sex and age
Around 1% of all breast cancers occur in men, so being female is an important risk factor. As with all carcinomas, increasing age is another significant factor. Up to the age of 50 years, the rate of increase is steep; it then slows down, although the incidence of breast cancer continues to increase into old age (Fig. 18.16).
Age at menarche and menopause
There is a significantly higher risk of developing breast cancer among women with an early age at menarche. At the other end of the reproductive life, women whose natural menopause occurs before 45 years have only half the breast cancer risk of those whose menopause occurs after 55 years. Therefore women with 40 or more years of active menstruation have twice the breast cancer risk of those with fewer than 30 years of menstrual activity.
Age at first full-term pregnancy
Nulliparous women have an increased risk of developing breast cancer. However, among parous women protection is related to early age for the first full-term pregnancy. If the first birth is delayed to the mid or late thirties, the woman is at a greater risk of developing breast cancer than is a nulliparous woman.
Weight and diet
For women of above average weight but below 50 years of age there is little or no increased risk of developing breast cancer. However, women aged 60 or over whose weight is increased have a higher cancer risk. Diet, obviously, can be a determinant for weight. In rodents, a high-fat diet increases the incidence of breast tumours, and international breast cancer incidence rates correlate with the consumption of fat. Although these observations suggest that a high-fat diet may be a risk factor, the evidence is not as clear as it is for weight.
Radiation
Women treated for Hodgkin’s disease by mantle radiation are at an increased risk of developing breast cancer and now undergo surviellance.
Family history and genetic factors
Breast cancer is common, thus a history of a relative having breast cancer can be found in at least 10% of new cases. However, a proportion of these will be sporadic cancers and not due to familial (inherited genetic) factors. The risk of developing breast cancer is increased in first-degree relatives (e.g. sister, daughter) of breast cancer cases, particularly if that person is pre-menopausal. For example, the risk increases to nine-fold for first-degree relatives of pre-menopausal women with bilateral breast cancer. Up to five-fold increases in risk have been found for women with multiple first-degree relatives with breast cancer.
There are rare familial syndromes such as Li–Fraumeni, in which there is an association between sarcomas, brain tumours and breast cancer at a young age. This is linked in some families to abnormalities of the p53 gene. Approximately 4–6% of breast cancers are associated with a very strong family history and in certain families there is breast and ovarian cancer. Inherited mutations of a gene on the long arm of chromosome 17 (BRCA1) are responsible for families with susceptibility to female breast and ovarian cancer. Another susceptibility gene, BRCA2, located on chromosome 13q12–13, is linked to families with early-onset breast cancer, including male breast cancer. Products of both genes are involved in DNA repair mechanisms. An inherited deletion in CHEK2 increases breast cancer risk.
There are likely to be yet more moderate- or low-risk genes that confer susceptibility. It must be remembered that this explains only a small proportion of breast cancers.
Geographic variation
There is a marked variation in breast cancer rates between different countries. The highest rates are in North America, north-west Europe, Australia and New Zealand, with the lowest in South-East Asia and Africa. Several factors probably contribute to this difference: age at menarche, age at first full-term pregnancy, age at menopause and post-menopausal weight. The length of time between age at menarche and first pregnancy may be quite short in some of these low-incidence countries.
Atypical hyperplasia
Women with benign breast disease whose breast biopsies show atypical epithelial hyperplasia have a definite increased risk of developing breast cancer. Ordinary epithelial hyperplasia is associated with a slightly increased risk. The risk is augmented by a family history of breast cancer.
Aetiological mechanisms
Hormones
The association of breast cancer risk with menarche, menopause and first full-term pregnancy indicates that hormones must have some role in the development of carcinomas, but they are more likely to be promoters than initiators.
Oestrogen activity appears to be important, with overexposure to oestrogens and underexposure to progesterone being significant. Early menarche and late menopause will result in a higher number of menstrual cycles, with repeated surges of oestrogen having a stimulatory effect on breast epithelium. The beneficial effect of early full-term pregnancy could be due to the high concentrations of progesterone and/or prolactin protecting the breast cells against oestrogens in the long term. The risks associated with obesity may be partly due to the ability of fat cells to synthesise oestrogens, or to altered levels of sex hormone-binding protein levels.
Oral contraceptives/hormone replacement therapy
There is a slightly increased risk for current and recent users of oral contraceptives but no long-term increase. Combined oestrogen and progesterone hormone replacement therapy increases the relative risk of developing breast cancer for current users by two-fold, and is greater the longer the duration of treatment. The risk decreases with cessation. Oestrogen-only preparations have a lower risk.
Hormone receptors
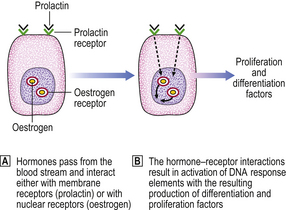
Hormones have an effect on cells only after interacting with specific receptors present on or in their target cells. The sex steroid, oestrogen, interacts with a nuclear receptor. Subsequent interaction with DNA results in the formation of differentiation- and proliferation-associated factors. Prolactin and other polypeptides interact with receptors on the cell surface (Fig. 18.17).
Oestrogen receptors can be detected in varying amounts in about 75% of breast cancers. The progesterone receptor, which can normally be formed only when the oestrogen receptor is present and active, is present in about 50% of tumours, and women whose tumours contain both types of receptor are more likely to respond to some form of hormone manipulation therapy. This suggests that hormones are important in the growth and maintenance of these carcinomas.
Non-invasive carcinomas
Virtually all breast carcinomas are adenocarcinomas derived from the epithelial cells of the ducts or glands.
The term ‘non-invasive’ means that the malignant cells are confined to either the ducts or the acini of the lobules, with no evidence of penetration of the tumour cells through the basement membranes around these two types of structure into the surrounding fibrous tissue. There are two forms of non-invasive carcinoma:
Ductal carcinoma in situ
Ductal carcinoma in situ can occur in both pre- and post-menopausal women, usually in the 40–60-year age group. It can present as a palpable mass, especially if extensive and associated with fibrosis. If the larger ducts are involved, presentation can be as a nipple discharge, or as Paget’s disease of the nipple. The disease can be found incidentally in surgical biopsies or be detected by mammography screening due to the presence of calcification. Pure ductal carcinoma in situ accounts for about 5% of breast carcinomas that present clinically.
The size of the area involved in the breast can range from 10 to 100 mm in length. It is usually unifocal, being confined within one quadrant of the breast, although multicentricity can occur with larger lesions. Bilateral disease is uncommon. The macroscopic appearances depend on the architecture of the ductal carcinoma in situ. Creamy necrotic material can exude from the cut surface of the breast, rather similar in appearance to comedones.
Histologically, the changes are to be found in the small and medium-sized ducts, although, in older women, the larger ducts can be involved. The ducts contain cells that show cytoplasmic and nuclear pleomorphism to varying degrees. Mitotic figures may be frequent and can be abnormal. These features are used to classify ductal carcinoma in situ into high grade (more aggressive features) and non-high grade lesions. The ducts may be completely filled with cells (solid pattern), or have central necrosis (comedo pattern; Fig. 18.18) which may calcify, rendering the lesion mammographically detectable. The cribriform pattern of ductal carcinoma in situ has numerous gland-like structures within the sheets of cells. Ductal carcinoma in situ can spread along the duct system or into the lobules.
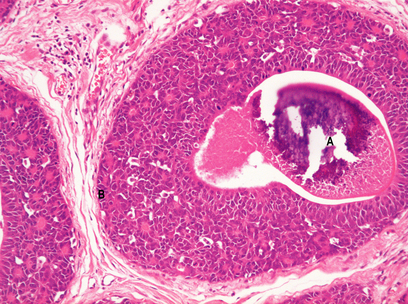
Fig. 18.18 Ductal carcinoma in situ. Both ducts are expanded. One has (A) a central necrotic area which has calcified and would show on a mammogram. The basement membrane (B) is intact.
The previous management of ductal carcinoma in situ was generally mastectomy, so it is difficult to know the fate of these lesions if left. Estimates of residual carcinoma changing from non-invasive to invasive range from one-third to one-half, based on studies where there was local incomplete excision. If the tumour is completely removed the woman’s prognosis is excellent.
Lobular carcinoma in situ
Lobular carcinoma in situ occurs more frequently in pre-menopausal women, but can be found in biopsies taken to investigate mammographic (screening) abnormalities. A major problem is that it does not present as a palpable lump and is usually found in biopsies removed for other reasons. A further important clinical feature is that it is often multifocal within the one breast and is frequently bilateral. Not surprisingly, there are no specific radiological or macroscopic features.
Histologically, the changes are found in the acini—hence the term ‘lobular’—although they may extend into extralobular ducts and replace ductal epithelium (Fig. 18.19). Within the acini, the normal cells are replaced by relatively uniform cells with clear cytoplasm that appear loose and non-cohesive. The overall size of the acini increases, but the lobular shape is retained. Unlike the situation in ductal carcinoma in situ, necrosis is unusual.
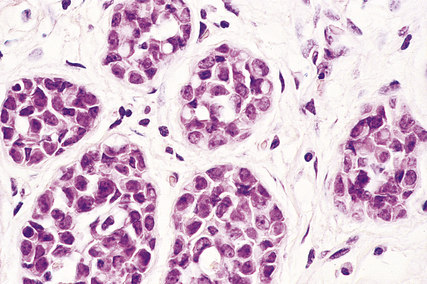
Fig. 18.19 Lobular carcinoma in situ. A breast lobule in which the acini are expanded. There is complete loss of the lumen and of the two-cell layer.
About one-quarter to one-third of all patients with lobular carcinoma in situ who are treated by biopsy alone will go on to develop an invasive carcinoma. This may occur in either or both breasts and there may be a long time interval.
Invasive carcinomas
An ‘invasive’ tumour is one whose cells have broken through the basement membrane around the breast structure in which they have arisen, and spread into the surrounding tissue. Invasive carcinomas are categorised into different histological types, but the name given to them does not always mean that the tumour arises only from that site; for example, invasive (infiltrating) duct or ductal carcinomas and invasive (infiltrating) lobular carcinomas may both arise from the cells at the junction of the extralobular and intralobular ducts. If an invasive tumour develops in a patient with previous lobular carcinoma in situ it can be ductal in morphology.
The histological types of invasive carcinoma and their relative incidence for palpable tumours are:
There is a higher frequency of tubular carcinoma in mammographically detected tumours.
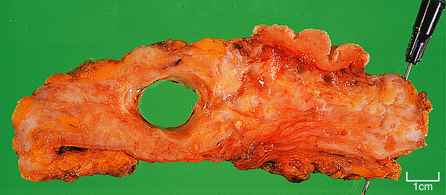
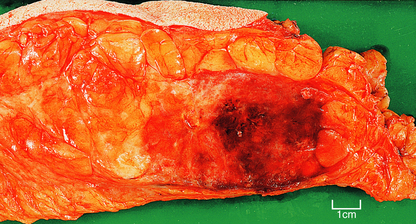
Carcinomas vary in size from less than 10 mm in diameter to over 80 mm, depending on whether detected by mammography or presenting clinically, but with the latter are often 20–30 mm in diameter. Clinically, they are firm on palpation and may show evidence of tethering to the overlying skin (Fig. 18.20) or underlying muscle. The skin may also show ‘peau d’orange’—dimpling due to lymphatic permeation. The nipple may be retracted due to tethering and contraction of the intramammary ligaments.
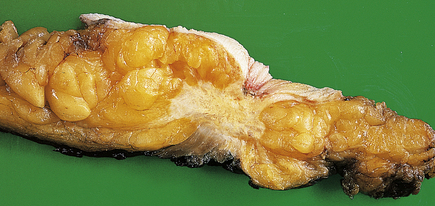
Fig. 18.20 Invasive carcinoma of breast. Mastectomy covered by skin and including the nipple. Beneath the nipple there is an irregular white area which has caused contraction. This is a carcinoma and the white tissue represents the fibrous (scirrhous) reaction. The rest of the breast is fat.
Gross features
The macroscopic appearance of the tumours tends to depend on the amount or type of stroma within the carcinoma. It is this that gave rise to the terms previously applied to tumours: scirrhous, medullary (or encephaloid) and mucinous (or colloid).
The term scirrhous implies that there is a prominent fibrous tissue reaction, usually in the central part of the tumour. This results in the carcinoma having a dense white appearance, which grates when cut. Yellow streaks may be seen; these are due to the presence of elastic tissue within the tumour. Carcinomas with a prominent stromal reaction usually have irregular edges, extending into the adjacent fat or other structures (Fig. 18.20).
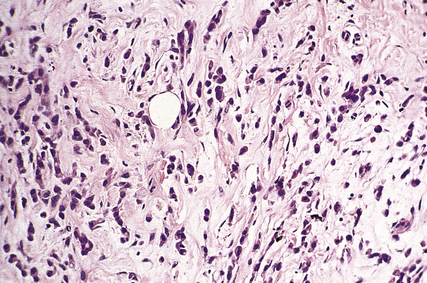
Medullary (brain-like) tumours are very cellular with little stroma. The edges of the carcinoma are often more rounded and discrete than those of the scirrhous tumours (Fig. 18.21). Necrosis is common. When palpated the tumours feel much softer.
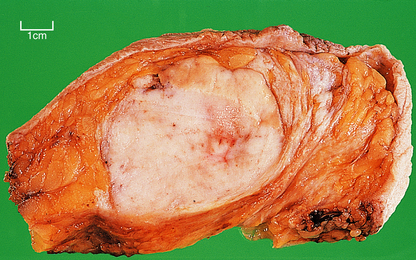
Fig. 18.21 Medullary carcinoma. Breast tissue containing a 60 mm diameter carcinoma with a rounded edge, and no evidence of a fibrous reaction.
Mucinous carcinomas have a predominance of mucin, or jelly-like, material within them. They usually have a well-defined edge.
Some of the changes that occur within carcinomas explain their clinical features, for example skin and nipple retraction due to the fibrous reaction.
Infiltrating ductal carcinomas
Infiltrating duct or ductal carcinomas of no special type comprise the majority (up to 75%) of infiltrating breast carcinomas. Macroscopically, they usually have a scirrhous consistency. The size of the tumours varies between patients. They can occur in both pre- and post-menopausal women.
Histologically, the tumour cells are arranged in groups, cords and gland-like structures. Quite marked variations can be seen between different carcinomas even though they are of the same type (Fig. 18.22). For example, the size of the solid groups of cells can be variable, and ductal carcinoma in situ is often present. The amount of stroma between the tumour cells can also vary, but in those carcinomas in which it is prominent it is most marked at the centre, with the periphery being more cellular. Collections of elastic tissue (elastosis) around ducts or within the stroma are common in tumours with a scirrhous reaction.
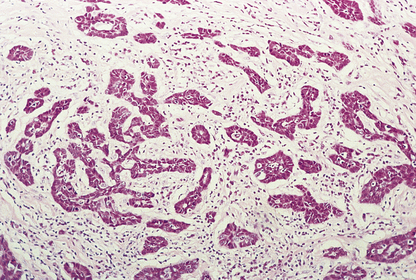
Fig. 18.22 Infiltrating ductal carcinoma. The lesion is composed of irregular solid groups of cells in a dense fibrous stroma, with an associated lymphocytic infiltrate.
The degree of differentiation or grade of the tumour is based on the extent to which it resembles non-tumorous breast: whether the cells are in a gland-like pattern or as solid sheets; the degree of nuclear pleomorphism; and the number of mitotic figures present. A well-differentiated (grade I) infiltrating duct carcinoma tends to behave less aggressively than a poorly differentiated (grade III) tumour, which is composed of sheets of pleomorphic cells with large numbers of mitotic figures.
Infiltrating lobular carcinomas
While lobular carcinoma in situ usually occurs in pre-menopausal women, the infiltrating lesion can also occur in post-menopausal women. In the UK, infiltrating lobular carcinomas constitute about 10% of invasive breast carcinomas, but the incidence may vary in other parts of the world.
Infiltrating lobular carcinomas have abundant fibrous stroma, so that macroscopically they are always scirrhous. While infiltrating ductal carcinomas usually form at one focus in the breast, infiltrating lobular carcinomas can be multifocal throughout the breast.
Histologically the cells are small and uniform and are dispersed singly, or in columns one cell wide (‘Indian files’; Fig. 18.23), in a dense stroma. Elastosis can be present. The cells infiltrate around pre-existing breast ducts and acini, rather than destroying them as occurs with invasive duct carcinomas. This method of infiltration may account for the occasional multifocal nature of the tumours. The cells in some carcinomas may appear signet-ring in shape due to the accumulation of mucin within an intracytoplasmic acinus, displacing the nucleus to one side. A characteristic feature of these tumours is that the cells lack the cell adhesion molecule E-cadherin, which may account for their pattern of spread. Residual lobular carcinoma in situ can sometimes be found in the invasive tumours.
Mucinous carcinomas
Mucinous carcinomas (also known as colloid, mucoid and gelatinous carcinomas) usually arise in post-menopausal women and comprise 2–3% of invasive carcinomas.
Macroscopically, the tumours are well circumscribed and have a soft, grey, gelatinous cut surface. They vary in size from 10 to 50 mm in diameter. Since there is no dense stroma and the edges are rounded, these tumours do not cause retraction of the nipple or tethering of the skin.
These carcinomas comprise small nests and cords of tumour cells, which show little pleomorphism, embedded in large amounts of mucin (Fig. 18.24). The latter is composed of neutral or weakly acidic glycoproteins, which are secreted by the tumour cells and are different from the proteoglycans of the stroma.
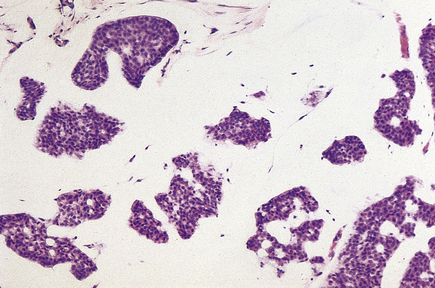
Fig. 18.24 Mucinous carcinoma. Small solid and tubular groups of cells lie in pools of mucin, or jelly-like material.
The survival of women with mucinous carcinomas is better than that of those having invasive duct or lobular carcinomas.
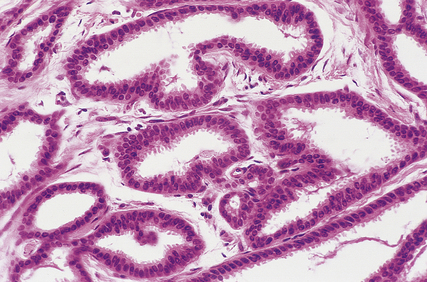
Tubular carcinomas
As the name implies, tubular carcinomas are well-differentiated carcinomas composed of cells arranged as tubules. They are often small lesions, less than 10 mm in diameter, and are firm, gritty tumours with irregular outlines. Tubular carcinomas form 1–2% of invasive carcinomas but constitute a higher proportion of screen-detected tumours.
Histologically, they are composed of well-formed tubular structures, the cells of which show little pleomorphism or mitotic activity. The stroma is dense, often with elastosis (Fig. 18.25).
Patients with tubular carcinomas do extremely well—better than those with well-differentiated invasive duct carcinomas.
Medullary carcinomas
The incidence of medullary carcinomas is difficult to assess because not all of the criteria for diagnosis have been strictly adhered to in some studies; hence figures have ranged from very rare to 5%. These tumours usually occur in post-menopausal women.
Medullary carcinomas are circumscribed and often large. Histologically, they are composed of large tracts of confluent cells with little stroma in between them. The cells show quite marked nuclear pleomorphism, and mitotic figures are frequent. There is never evidence of gland formation. These cytological appearances put them into the ‘poorly differentiated’ category. Around the islands of tumour cells there is a prominent lymphocytic infiltrate, predominantly T-lymphocytes, with macrophages (Fig. 18.26).
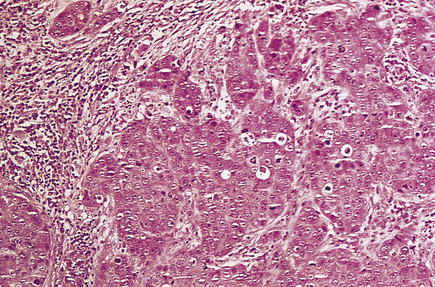
Fig. 18.26 Medullary carcinoma. Large groups of irregularly shaped tumour cells are surrounded by an infiltrate of lymphocytes.
Despite the aggressive cytological features of these tumours, the patients have a significantly better 10-year survival than women with invasive duct carcinomas. It may be that the lymphocytic and macrophage infiltrate has a beneficial effect, and this has stimulated much research into the immunological responses to tumours generally.
Papillary carcinomas
Papillary carcinomas are rare tumours that occur in post-menopausal women. They are usually circumscribed and can be focally necrotic, with little stromal reaction. The tumours are in the form of papillary structures, and areas of intraductal papillary growth are usually found.
The prognosis of patients with these carcinomas is probably better than that of the much more common invasive duct carcinoma.
Other types
Much rarer types of breast carcinoma include: adenoid cystic carcinomas; secretory carcinomas, which occur predominantly in juveniles; apocrine carcinomas, which are composed of cells with abundant eosinophilic cytoplasm; and carcinomas showing metaplasia, e.g. squamous, spindle cell, cartilaginous and osseous features.
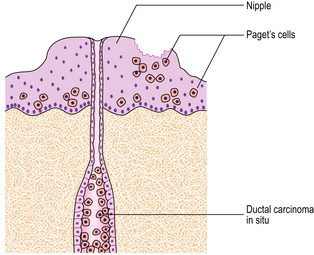
Paget’s disease of the nipple
Paget’s disease of the nipple was first described by Sir James Paget in 1874. Clinically, there is roughening, reddening and slight ulceration of the nipple, similar to the skin changes of eczema. Recognition is important, as it is associated with an underlying carcinoma, mainly in the subareolar region. Paget’s disease of the nipple occurs with about 2% of all breast carcinomas, and is associated with a higher frequency of multicentric breast carcinomas.
Within the epidermis of the nipple, large, pale-staining malignant cells can be seen histologically and these cause the changes seen clinically. The malignant cells are derived from the adjacent breast carcinomas. A direct connection may not be seen. The relationship between Paget’s disease of the nipple and an underlying carcinoma is shown in Figure 18.27.
Spread of breast carcinomas
Breast carcinomas can infiltrate locally (direct spread) or metastasise to more distant sites via lymphatics and the blood stream and to pleura (Fig. 18.28).
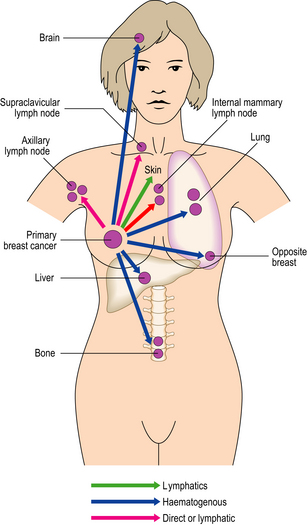
Fig. 18.28 The common sites of metastasis from breast carcinoma via the lymphatic system or blood stream.
Direct spread. Local infiltration (direct spread) into the underlying muscles and the overlying skin can be detected clinically, the latter because of ulceration or tethering.
Via lymphatics. Permeation of the lymphatic channels of the skin results in the clinical sign of ‘peau d’orange’. The axillary lymph nodes are the commonest initial site of metastasis via lymphatics, and between 40% and 50% of women with symptomatic breast carcinoma will have axillary lymph node metastases at the time of presentation. Figures will differ for those detected by mammography. It is important that the lymph nodes are examined histologically, as clinical palpation is not always reliable. Sentinel node removal is an increasingly used method for determining node status. Metastasis to intramammary, supraclavicular and tracheobronchial lymph nodes also occurs.
Via blood stream. Blood-borne metastasis most frequently involves the lungs and bones, but the liver, adrenals and brain are also common sites. The pleura on the same side as the breast carcinoma can be a site of metastasis, causing an effusion.
Infiltrating lobular carcinomas can metastasise to more unusual sites, and this may be due to their single-cell method of spread as seen within the breast.
Extensive infiltration of bone marrow can cause leukoerythroblastic anaemia. Destruction of bone can result in hypercalcaemia, with renal complications.
Breast carcinomas exhibit quite marked variation in the length of time between presentation of the primary carcinoma and the appearance of recurrent/metastatic disease. Some breast carcinomas never recur; in some patients reappearance of the disease may not be until as much as 20 years after the original excision, while for others it can be within 2–5 years. Tumour can recur at the site of the original excision and/or as distant metastases. The mechanisms by which a metastasis becomes clinically apparent after a long time interval are not known. They may relate to changes in tumour cells that have been lying dormant at that site, causing them to alter their behaviour, and/or to changes in the host response to the tumour.
Prognostic factors
Some women have carcinomas for several years before seeking medical help; in this time the tumour may ulcerate into the skin and become large. However, despite the horrifying features the tumour may present, such patients may survive for many years after treatment. Other women seek medical help promptly after palpating a lump but die from the disease within a short time. There are thus obviously quite marked differences between individual breast carcinomas and in the host response of patients to them.
Several factors have been identified that may help to predict how an individual carcinoma will behave, and may help in planning therapy. However, despite the great effort expended in this area, the only major changes made clinically have been in lengthening the disease-free interval (time before development of recurrence/metastasis) rather than in improving patient survival.
Type of carcinoma
Medullary, mucinous, tubular and, possibly, invasive lobular carcinomas generally behave less aggressively than other types, but these constitute the minority of types so that this knowledge is of value to only a few patients.
Histological grade
As described above, tumours can be graded for their degree of differentiation. Patients whose tumours are well differentiated (grade I), showing greater resemblance to non-malignant breast, do better, while those whose tumours are poorly differentiated (grade III) do worse; however, prediction of how the group with moderately differentiated carcinomas will do is more difficult.
Stage
When a woman presents with a breast carcinoma, staging is undertaken so as to assess the absence or presence and extent of spread both locally and distantly. The management of the patient will depend on the stage of the disease. The two main systems used are the International Classification of Staging and the TNM (Tumour, Node, Metastasis) system (Table 18.2).
Table 18.2 The main staging systems used to assess the extent of spread of breast carcinomas
| Stage | Extent of spread |
|---|---|
| International classification | |
| I | Lump with slight tethering to skin, but node negative |
| II | Lump with lymph node metastasis or skin tethering |
| III | Tumour that is extensively adherent to skin and/or underlying muscles, or ulcerating or lymph nodes are fixed |
| IV | Distant metastases |
| TNM | |
| T1 | Tumour 20 mm or less; no fixation or nipple retraction. Includes Paget’s disease |
| T2 | Tumour 20–50 mm, or less than 20 mm but with tethering |
| T3 | Tumour greater than 50 mm but less than 100 mm; or less than 50 mm but with infiltration, ulceration or fixation |
| T4 | Any tumour with ulceration or infiltration wide of it, or chest wall fixation, or greater than 100 mm in diameter |
| N0 | Node-negative |
| N1 | Axillary nodes mobile |
| N2 | Axillary nodes fixed |
| N3 | Supraclavicular nodes or oedema of arm |
| M0 | No distant metastases |
| M1 | Distant metastases |
If there is evidence of metastatic spread to axillary lymph nodes when the patient presents with the primary carcinoma, both the 5- and 10-year survival figures are worse than in those with no evidence of metastasis. The outlook for the patient is also worse if there is evidence of more distant spread.
Oestrogen receptors
The presence of oestrogen receptors within a carcinoma indicates that the tumour cells have a higher degree of functional differentiation. It is thus not surprising that women whose tumours are oestrogen receptor-positive have better survival figures than those whose carcinomas are oestrogen receptor-negative. More importantly, they are more likely to benefit from tamoxifen, an oestrogen receptor antagonist.
Growth kinetics
The growth activity of carcinomas can be measured by several methods; that of breast carcinomas may be low, medium or high. Tumours with lower cell growth rates tend to behave better clinically. It must be remembered that tumours with a high rate of division may also have a high cell death rate by apoptosis, and that not all cell divisions result in doubling of the population as the division may be abnormal.
HER-2
The oncogene c-erbB-2/HER-2 is altered in approximately 20% of invasive breast carcinomas. There is amplification of the gene with resultant overexpression of the membrane-related protein. Patients whose carcinomas have this alteration have a poorer prognosis. A humanised monoclonal antibody, trastuzumab (Herceptin) has been developed, which can be used as adjuvant treatment and to treat women with metastatic disease, if the cancers have the molecular alteration.
Examples of the effects some of these prognostic factors may have on survival are shown in Figure 18.29.
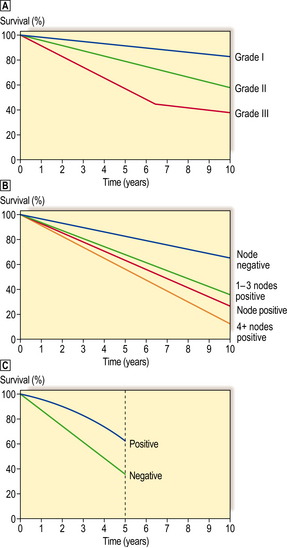
Fig. 18.29 Relationship of various prognostic factors with survival for patients with operable breast cancer.  Grade or degree of tumour differentiation (grade I = well differentiated; grade II = moderately differentiated; grade III = poorly differentiated). 10-year survival.
Grade or degree of tumour differentiation (grade I = well differentiated; grade II = moderately differentiated; grade III = poorly differentiated). 10-year survival.  Presence or absence of lymph node metastasis, and relationship to number of lymph nodes involved. 10-year survival.
Presence or absence of lymph node metastasis, and relationship to number of lymph nodes involved. 10-year survival.  Presence or absence of oestrogen receptor within the tumours. 5-year survival (less significant after this period).
Presence or absence of oestrogen receptor within the tumours. 5-year survival (less significant after this period).
Breast carcinomas in men
About 1% of breast carcinomas occur in males, but the incidence varies throughout the world. The tumour is rare in young men. There is an increased risk in patients with Klinefelter’s syndrome, and for carriers of BRCA2 mutations.
The tumour usually presents as a lump, but there can be nipple discharge or retraction. Paget’s disease is relatively commoner in men, probably because of the small size of the male breast.
Ductal carcinoma in situ and all types of invasive carcinoma can occur, although lobular carcinoma in situ has not been reported. The prognosis in males is similar to that in females, and is affected by such factors as lymph node status and size. Oestrogen receptors can be detected in male breast carcinomas.
OTHER TUMOURS
Phyllodes tumours
Phyllodes tumours can occur at any age, but the median age is 45 years. This is older than for fibroadenoma and the incidence of phyllodes tumours is considerably lower. Phyllodes tumours present clinically as a discrete lump. Macroscopically, they are circumscribed and vary in size up to as much as 60 mm in diameter. They may have both soft and firm areas.
Phyllodes tumours have two characteristic parts, epithelium and stroma. The epithelium covers large, club-like projections which push into cystic spaces. The stroma is much more cellular than that of fibroadenomas (Fig. 18.30) and can vary in type within the same tumour. The cells may resemble fibroblasts, or they may show marked pleomorphism with mitotic figures. In some tumours, the stromal changes are so marked that they have the appearances of sarcomas.
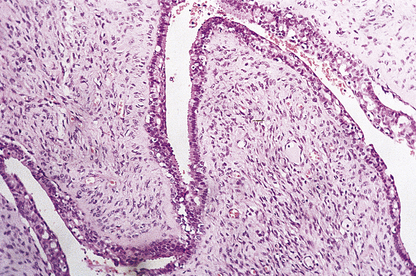
Fig. 18.30 Phyllodes tumour. The stroma is cellular and it is forming club-like fingers covered with epithelium.
Recurrence is a major problem with phyllodes tumours. The risk of recurrence is less if the tumours are small, with a low mitotic rate and minimal cellular atypia, and have a rounded rather than an infiltrative edge. With each recurrence, the stroma of the tumour tends to become more atypical with a higher mitotic rate. The chance of metastasis then increases, and this is usually via the blood stream to lung and bones; lymph node involvement is rare. In one series of cases, recurrence occurred in 30% of cases and 16% died from metastatic disease; however, these patients were a pre-selected group whose original tumours had a more aggressive-looking stroma.
Angiosarcomas
Angiosarcomas are rare tumours that can occur at any time from adolescence to old age but are commoner in young women. Although most cases occur spontaneously, angiosarcomas can arise in irradiated mastectomy scars and in lymphoedematous arms after radical mastectomy for breast cancer (Stewart–Treves syndrome).
Angiosarcomas can present as a lump, or cause a diffuse enlargement of the breast. Discoloration of the overlying skin can be seen in some cases. Macroscopically, they can be haemorrhagic or appear as ill-defined areas of induration (Fig. 18.31).
Histologically, the tumours consist of numerous vascular channels that infiltrate into fat and around normal breast structures. The channels are lined by endothelial cells which have hyperchromatic nuclei. Papillary areas can be present and, in the more undifferentiated tumours, there can be sheets of large, pleomorphic endothelial cells with little evidence of vascular channels.
The clinical outcome tends to parallel the histological appearances. Those tumours with well-formed vascular spaces and little atypia of the endothelium are less aggressive. Metastasis is by the blood stream to lungs, bone, liver and brain.
Other sarcomas
Fibrosarcoma, liposarcoma and leiomyosarcoma can all occur in the breast but are rare.
Lymphomas
Lymphomas may be primary, but are more usually secondary to disease elsewhere in the body.
Commonly confused conditions and entities relating to breast pathology
| Commonly confused | Distinction and explanation |
|---|---|
| Fibroadenoma and fibroadenosis | Fibroadenoma is a localised circumscribed benign neoplasm comprising epithelial cells and specialised fibrous tissue. Fibroadenosis is an obsolete name for fibrocystic change, a hyperplastic lesion. |
| Fibroadenoma and phyllodes tumour | Fibroadenoma and phyllodes tumour both comprise neoplastic epithelial and fibrous tissue components. However, in phyllodes tumours the fibrous tissue component is more cellular and abundant, and the lesion has less well defined margins; borderline and malignant variants occur. |
| Ductal epithelial hyperplasia and ductal carcinoma in situ | Ductal epithelial hyperplasia is a benign proliferation of duct epithelium, whereas ductal carcinoma in situ has undergone neoplastic transformation, although it is not yet invasive. These lesions can have morphological similarities. A proportion share genetic alterations. |
| Radial scar and complex sclerosing lesion | Radial scars and complex sclerosing lesions differ only in size: the latter are >10 mm in diameter. Both mimic carcinomas radiologically and histologically, but they are benign non-neoplastic lesions. |
| Medullary carcinoma of the breast and of the thyroid | The term medullary refers only to the soft consistency (resembling the medulla of the brain). There is no other relationship between these lesions. |
| Paget’s disease of the nipple and of bone | Both lesions were described by Sir James Paget (1814–1899). There is no other relationship between these lesions. |
Dixon J.M.. ABC of breast diseases, 2nd edn. London: BMJ; 2000.
Elston C.W., Ellis I.O.. The breast. Edinburgh: Churchill Livingstone; 1998.
Harris J.R., Morrow M., Lippman M.E. Diseases of the breast. 3rd edn 2004. Lippincott, Williams & Philadelphia: Wilkins
Nathanson K.L., Wooster R., Weber B.L.. Breast cancer genetics: what we know and what we need. Nature Medicine. 2000;7:552-556.
Rosen P.P. Rosen’s breast pathology. 2nd edn 2001. Philadelphia: Lippincott-Raven.
Sloane J.P.. Biopsy pathology of the breast, 2nd edn. London: Arnold; 2001.
World Health Organization. WHO classification of tumours. Pathology and genetics of the breast and female genital tract. Lyons: IARC; 2003.
http://www.cancerresearchuk.org