8 Immunological principles
Antigens and antigen recognition
Key points
• The immune system recognizes molecules known as antigens. Recognition provokes a response that may be immediate and relatively non-specific (innate immunity) or may develop over time and become progressively more specific (acquired immunity).
• Antigens generally comprise multiple epitopes, each eliciting separate but specific immune responses that, in acquired immunity, may be mediated by antibodies (humoral immunity) or by T lymphocytes, which are responsible for cell-mediated immunity.
• Antibodies (also called immunoglobulins) are glycoproteins with a heterodimeric structure based on heavy (H) and light (L) chains. By genetic recombinations in the cells responsible for antibody synthesis (B lymphocytes), highly polymorphic (variable) regions are generated within H and L chains. H and L chains are brought together such that binding specificity for an enormous range of epitopes is achieved.
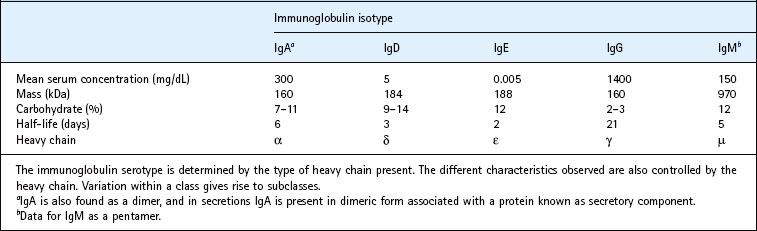
• Five main heavy chain types give rise to the five different immunoglobulin classes: IgG, IgA, IgM, IgE and IgD (in descending order of their relative abundance in human serum). The different classes serve different functions.
• Molecules that elicit innate immune responses display pathogen-associated molecular patterns that are recognized by pattern recognition receptors.
• In acquired immunity, antigens that elicit a humoral response are recognized by immunoglobulin molecules on B lymphocytes.
• Antigen recognition leading to cell-mediated immunity depends on T lymphocyte receptors. These are activated by epitopes resulting from intracellular processing of antigens. These epitopes are presented to T cells in combination with major histocompatibility complex (MHC) molecules on the surface of host cells.
• The two major effectors of cell-mediated immunity are T lymphocyte subsets termed CD4 and CD8 cells; these are, respectively, stimulated by epitopes presented in the context of MHC class II and class I molecules.
An antigen is any substance capable of provoking the lymphoid tissues of an animal to respond by generating an immune reaction directed specifically at the inducing substance and not at other unrelated substances. The response is not to the entire molecule but to individual chemical groups within it that have a specific three-dimensional shape. The specificity of the response to these antigenic determinants or epitopes is an important characteristic of immune responses. The reaction of an animal to contact with antigen, called the acquired immune response, takes two forms: first, the humoral or circulating antibody response and, second, the cell-mediated response, and their characteristics are described in Chapter 9. Most of the information available on the specificity of the immune response comes from studies of the interaction of circulating antibody with antigen. An antibody directed against an epitope of a particular molecule will react only with this determinant or other very similar structures. Even minor chemical changes in the conformation of the epitope markedly reduce the ability of the original antibody to react with the altered material.
The term ‘antigen’, referring to substances that either act as stimulants of the immune response or react with antibody, is used rather loosely by immunologists. Use is made of the functional classification of antigens into:
• substances that are able to generate an immune response by themselves, which are termed immunogens
• molecules that are able to react with antibodies but are unable to stimulate their production directly.
The latter substances are often low molecular weight chemicals, termed haptens, that react with preformed antibodies but become immunogenic only when attached to large molecules, called carriers. The hapten forms an epitope on the carrier molecule that is recognized by the immune system and stimulates the production of antibody. In other words, the ability of a chemical grouping to interact with an antibody is not sufficient to stimulate an immune response. As we will see later, when discussing the sites on molecules recognized by cells of the immune system, all antigens can be considered to be composed of haptens on larger carrier structures. By convention, the term ‘immune response’ was used to refer to the acquired immune system and innate immunity was considered to be a rather non-specific, although relatively effective, defence against infection. In the past few years it has become evident that components of the innate immune system recognize a set of molecular signatures that have been termed pathogen-associated molecular patterns through pattern recognition receptors on the surface of cells and on secreted molecules (see Ch. 9).
General properties of antigens
A substance that acts as an antigen in one species of animal may not do so in another if it is represented in the tissues or fluids of the second species. This underlines the requirement that an antigen must be a foreign substance to elicit an immune response. For example, egg albumen, although an excellent antigen in rabbits, fails to induce an antibody response in fowl. The more foreign and evolutionarily distant a substance is from a particular species, the more likely it is to be a powerful antigen.
A widely recognized requirement for a substance to be antigenic in its own right, without having to be attached to a carrier molecule, is that it should have a molecular weight in excess of 5000 Da. It is, however, possible to induce an immune response to substances of lower molecular weight. For example, glucagon (molecular weight of 3800 Da) can stimulate antibody production, but only if special measures are taken such as the use of an adjuvant which gives an additional stimulus to the immune system. Very large proteins, such as the crustacean respiratory pigment haemocyanin, are very powerful antigens and are used widely in experimental immunology. Polysaccharides vary in antigenicity; for example, dextran with a molecular weight of 600 000 Da is a good antigen, whereas dextran with a molecular weight of 100 000 Da is not.
Some low molecular weight chemical substances appear to contradict the requirement that an antigen be large. Among these are picryl chloride, formaldehyde and drugs such as aspirin, penicillin and sulphonamides. These substances are highly antigenic, particularly when applied to the skin. The reason for this appears to be that such materials form complexes by means of covalent bonds with tissue proteins. The complex of such a substance, acting as a hapten, with a tissue protein acting as a carrier, forms a complete antigen. This phenomenon has important implications in the development of certain types of hypersensitivity (see Ch. 9).
Antigenic determinants
The immune system does not recognize an infectious agent or foreign molecule as a whole, but reacts to structurally distinct areas: antigenic determinants or epitopes. Thus, exposure to a micro-organism will generate an immune response to many different epitopes. The antiserum produced will contain different antibodies reactive with each determinant. This will ensure that an individual is protected from the micro-organism by producing a response to at least a few of the possible determinants. If the host reacted only to the organism as a whole, then failure to react to this one site would have dire consequences: it would not be able to eliminate the pathogen. Certain antibodies may react with an epitope composed of residues that can also be part of two other epitopes recognized by different antibodies (Fig. 8.1).
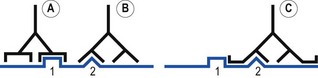
Fig. 8.1 Overlapping epitopes. Two epitopes (1 and 2) on an antigen induce the formation of three antibodies (A, B and C).
A response to antigen involves the specific interaction of components of the immune system, antibodies and lymphocytes, with epitopes on the antigen. The lymphocytes have receptors on their surface that function as the recognition units; on B lymphocytes surface-bound immunoglobulin is the receptor, and on T lymphocytes the recognition unit is known as the T cell receptor. The interaction between an antibody (or cell-bound receptor) and antigen is governed by the complementarity of the electron cloud surrounding the determinants. The overall configuration of the outer electrons, not the chemical nature of the constituent residues, determines the shape of the epitope and its complementary paratope (the part of the antibody or T cell receptor that interacts with the epitope). The better the fit between the epitope and the paratope, the stronger the non-covalent bonds formed and consequently the higher the affinity of the interaction.
Antigenic determinants have to be topographical, that is, composed of structures on the surface of molecules, and can be constructed in two ways. They may be contained within a single segment of primary sequence or assembled from residues far apart in the primary sequence but brought together on the surface by the folding of the molecule into its native conformation. The former are known as sequential epitopes, and those formed from distant residues are conformational epitopes. The majority of antigenic structures recognized by antibodies depend on the tertiary configuration of the immunogen (conformational), whereas T cell epitopes are defined by the primary structure (sequential).
Antigenic specificity
Foreignness of a substance to an animal can depend on the presence of chemical groupings that are not normally found in the animal’s body. Arsenic acid, for example, can be chemically introduced into a protein molecule and, as a hapten, acts as a determinant of antigenic specificity of the molecule. There are many examples in which antibodies are able to distinguish subtle chemical differences between molecules. Thus, antisera can distinguish between glucose and galactose, which differ only by the interchange of a hydrogen atom and a hydroxyl group on one carbon atom.
The ability of antibody (or T cell receptors) to form a high-affinity interaction with an antigen depends on intermolecular forces, which act strongly only when the two molecules come together in a very precise manner. The better the fit, the stronger the bond. An antibody molecule directed against a particularly shaped antigenic determinant might be able to react with another similar but not quite identical determinant, as shown in Figure 8.2. This type of cross-reaction does occur, but the strength of the bond between the two molecules is diminished in the case of the less well fitting determinant.
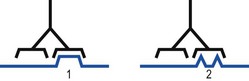
Fig. 8.2 Specificity and cross-reactions. Antibody produced in response to an antigen that contains epitope 1 will also combine with epitope 2.
A common source of confusion concerning the specificity of antibodies arises when an antibody to a particular antigen is found to be capable of combining with an apparently unrelated antigen. For example, glucose residues are present in many different types of molecule, and an antibody that binds to a glucose determinant in antigen X-glucose would be likely to react with the glucose group in antigen Y-glucose, provided the two determinants were equally accessible. The antibody directed against the glucose determinant is not a non-specific type of antibody but is simply reacting with an identical chemical determinant in another antigen molecule.
In laboratory practice, cross-reactivity is often found between antisera to certain bacterial antigens and antigens present on cells such as erythrocytes. Antigens shared in this way are known as heterophile antigens. The best known of the heterophile antigens is the Forssman antigen, which is present on the red cells of many species as well as in bacteria such as pneumococci and salmonellae. Another heterophile antigen is found in Escherichia coli and human red cells of blood group B individuals. These cross-reactivities are probably responsible for the generation of antibodies found in individuals of a certain blood group that bind to the red blood cells of individuals of a different blood group. These antibodies are known as isohaemagglutinins because they are able to bind the red blood cells and clump them together (i.e. cause agglutination).
Immunoglobulins
Towards the end of the nineteenth century, von Behring and Kitasato in Berlin found that the serum of an appropriately immunized animal contained specific neutralizing substances, or antitoxins. This was the first demonstration of the activity of what are now known as antibodies or immunoglobulins. Antibodies are:
• present in the serum and body fluids
• induced when immunogenic molecules are introduced into the host’s lymphoid system
• reactive with, and bind specifically to, the antigen that induced their formation.
The liquid collected from blood that has been allowed to clot is known as serum. It contains many different molecules but no cells or clotting factors. If serum is prepared from an animal that has been exposed to an antigen, it is known as an antiserum as it will contain antibodies reactive to the inducing antigen.
There are five distinct classes or isotypes of immunoglobulins: IgG, IgA, IgM, IgD and IgE. They differ from one another in terms of size, charge, carbohydrate content and, of course, amino acid composition (Table 8.1). Within certain classes there are subclasses that vary slightly in structure and function. These classes and subclasses can be differentiated from one another serologically, using antibody. If antibody from one species is injected into another species they will induce the formation of antibodies that can be used to differentiate between different isotypes.
Antibody structure
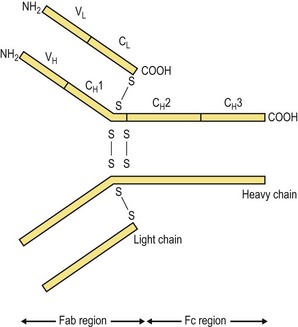
All antibody molecules have the same basic four-chain structure composed of two light chains and two heavy chains (Fig. 8.3). The light chains (molecular weight 25 000 Da) are one of two types designated κ and λ, and only one type is found in any one antibody molecule. The heavy chains vary in molecular weight from 50 000 to 70 000 Da, and it is these chains that determine the isotype. They are designated α, δ, ε, γ and µ for the respective classes of immunoglobulin (Table 8.1). The individual chains are held together by disulphide bridges and non-covalent interactions.
When individual light chains are studied, they are found to comprise two distinct areas or domains of approximately 110 amino acids. One end of the chain is identical in all members of the same isotype, and is termed the constant region of the light chain, CL. The other end shows considerable sequence variation, and is known as the variable region, VL. The heavy chains are also split into domains of approximately the same size, the number varying between the five types of heavy chain. One of these domains will show considerable sequence variation (VH), whereas each of the others (CH) are identical for the same domain of the same isotype. The tertiary structure generated by the combination of the VL and VH regions determines the shape of the antigen-combining site or paratope. As the two light and two heavy chains of any one antibody molecule are identical, each antibody unit has two identical paratopes, situated at the amino (N)-terminal end of the molecule, that recognizes the antigen. The carboxyl (C)-terminal end of the antibody is the same for all members of the same class or subclass, and is involved in the biological activities of the molecule. The area of the heavy chains between the CH1 and CH2 domains contains a varying number of interchain disulphide bonds and is known as the hinge region. A number of enzymes cleave immunoglobulins at distinct points to generate different peptide fragments. Using these enzymes, antibodies can be divided into a Fab region (‘fragment antigen binding’) containing the paratope and an Fc region (‘fragment crystallizable’) that is similar for all antibodies of the same isotype.
Despite the differences between the various isotypes, as shown in Table 8.1, all antibody molecules are composed of the same basic unit structure, with the Fab portion containing the antigen-recognizing paratope and the Fc region carrying out the activities that protect the host (i.e. effector functions). The differences seen in the Fc region of the various heavy chains are responsible for the different biological activities of the antibody isotypes.
IgG
This is the major immunoglobulin of serum, making up 75% of the total and having a molecular weight of 150 000 Da in man. Four subclasses are found in man – IgG1, IgG2, IgG3 and IgG4 – that differ in their relative concentrations, amino acid composition, number and position of interchain disulphide bonds, and biological function. IgG is the major antibody of the secondary response (see Ch. 9) and is found in both the serum and tissue fluids.
IgA
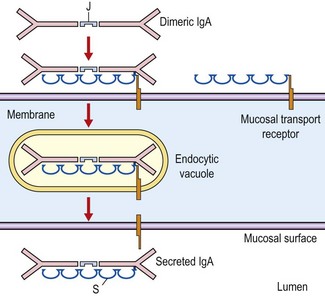
In man, most of the serum IgA occurs as a monomer, but in many other mammals it is found mostly as a dimer. The dimer is held together by a J chain, which is produced by the antibody-producing plasma cells. IgA is the predominant antibody class in seromucous secretions such as saliva, tears, colostrum, and at mucosal–epithelial surfaces in the respiratory, gastrointestinal and genitourinary secretions. This secretory IgA (sIgA) is always in the dimeric form and is composed of two basic four-chain units (two light chains and two heavy chains), a J chain and the secretory component. The secretory component is part of the molecule that transports the dimer produced by a submucosal plasma cell to the mucosal surface (Fig. 8.4). It facilitates passage through the epithelial cells and protects the secreted molecule from proteolytic digestion. There are two subclasses of IgA: IgA1 and IgA2.
IgM
IgM is a pentamer of the basic unit with µ heavy chains and a single J chain. Because of its large size, this isotype is confined mainly to the intravascular pool, and is the first antibody type to be produced during an immune response.
Antigen binding
The variability in amino acid sequence in the variable domains of light and heavy chains is not found over their entire length but is restricted to short segments. These segments show considerable variation, and are termed hypervariable regions. Hypervariable regions contain the residues that make direct contact with the antigen, and are referred to as complementarity determining regions. Although the remaining framework residues do not come into direct contact with the antigen, they are essential for the formation of the correct tertiary structure of the variable domain and maintenance of the integrity of the binding site. In both light and heavy chains there are three complementarity determining regions that, in combination, form the paratope.
The antigen and antibody are held together by various individually weak non-covalent interactions. However, the formation of a large number of hydrogen bonds and electrostatic, van der Waals’ and hydrophobic interactions leads to a considerable binding energy. These attractive forces are active only over extremely short distances, and therefore the epitope and paratope must have complementary structures to enable them to combine. If the electron clouds overlap or residues of similar charge are brought together, repulsive forces will come into play. The balance of attraction against repulsion will dictate the strength of the interaction between an antibody and a particular antigen, that is, the affinity of the antibody for the antigen.
Antibody diversity
It is now known that an antigen selects from the available antibodies those that can combine with its epitopes. It therefore follows that an individual must have the capacity to produce an extremely large number of different antibodies to cope with the vast array of different antigens present in the environment.
Immunoglobulin variability
The paratope is produced by the complementarity determining regions of the light and heavy chains generating a specific three-dimensional shape. Any light chain can join with any heavy chain to produce a different paratope. Thus, theoretically, with 104 different light chains and 104 different heavy chains, 108 different specificities could be generated.
The germline DNA is the structure of the gene as it is inherited. All cells in the body contain all the inherited genes, but different genes become active in different cells at different times. The functional immunoglobulin genes within B lymphocytes, the cells that differentiate to antibody-producing plasma cells, are formed by gene rearrangements and recombinations. These events give rise to the production of different variable domains in each B lymphocyte. Once a functional gene has been constructed, no other rearrangements are allowed to take place within this cell. This dictates that one particular cell will produce antibodies with an identical antigen-combining site, and is known as allelic exclusion. There is evidence that the gene segments for the variable region of immunoglobulins are particularly susceptible to mutations. This can lead to subtle changes in specificity and/or affinity that are important as an immune response develops (see Ch. 9).
When a B lymphocyte is first stimulated by antigen it produces IgM. As the immune response develops, the class of antibody changes. However, the immunoglobulin produced will have the same variable domain and therefore bind to the same antigen. All that is altered, or switched, is the heavy-chain constant region. Thus the progeny of a single B cell will produce different immunoglobulin isotypes as the response to a particular antigen develops, but each will have the same paratope.
Secreted and membrane immunoglobulins
At different stages in its development a B cell produces immunoglobulins that have to be inserted into the membrane or secreted. The membrane-bound immunoglobulin is used as the antigen receptor of the B cell, and a cell that binds antigen through this molecule will then secrete immunoglobulin of the same specificity. The only difference between the two types of antibody is to be found at the C terminus, where the membrane form has an additional part, the transmembrane portion.
Antibody function
Knowledge gained from the structural studies discussed above has gone some way towards an understanding of the biological activities of the immunoglobulin molecule.
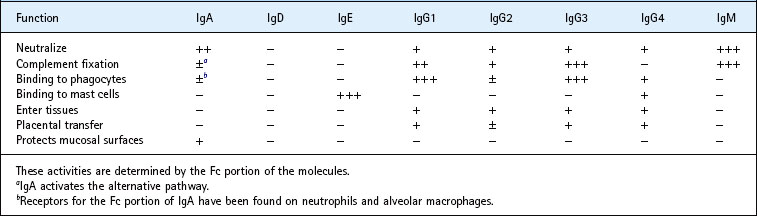
The primary function of an antibody is to bind the antigen that induced its formation. Apart from cases where this results in direct neutralization (e.g. inhibition of toxin activity or of microbial attachment), other effector functions must be generated. The binding of antigen is mediated by the Fab portion, and the Fc region controls the biological defence mechanisms. For every antibody the paratope is different, and different epitopes will therefore be recognized. However, for every antibody of the same isotype, the heavy-chain constant domains are the same, and they therefore all perform the same functions (Table 8.2).
Neutralization
Because antibodies are at least divalent, they can form a complex with multivalent antigens. Depending on the physical nature of the antigen these immune complexes exist in various forms (Fig. 8.5). If the antibody is directed against surface antigens of particulate material such as microorganisms or erythrocytes, agglutination will occur. This results in a clump or aggregate that isolates the potential pathogen, stops its dissemination and stimulates its removal by other mechanisms. If the antigen is soluble, the size of the complex will determine its physical state. Small complexes remain soluble, whereas large complexes form precipitates.
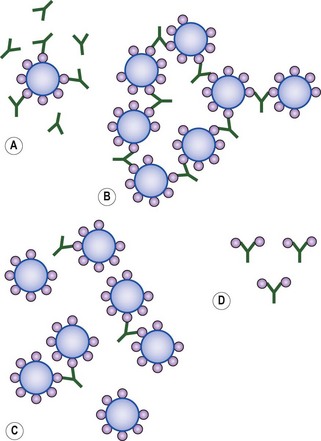
Fig. 8.5 Immune complex formation: (A) Antibody excess; (B) Equivalence; (C) Antigen excess; (D) Monovalent antigen. At antigen or antibody excess, small complexes are formed. Between these two extremes, large complexes are formed that are maximal at equivalence. Antibody cannot agglutinate an antigen with a single epitope.
As might be expected from knowledge of the structure of IgM, its ten combining sites make it a very efficient agglutinating antibody molecule. Rabbit IgM has been shown to be more than 20 times as active as IgG in bringing about bacterial agglutination. Because of its size, IgM is confined largely to the bloodstream and probably plays an important role in protecting against blood invasion by micro-organisms. Certain sites on micro-organisms are critical to the establishment of an infection. Antibody bound to these sites interferes with attachment processes and can, therefore, stop infection by the microbe. The binding of an antibody to functionally important residues in toxins neutralizes their harmful effects.
Complement activation
Activation of the complement system is one of the most important antibody effector mechanisms. The complement cascade is a complex group of serum proteins that mediate inflammatory reactions and cell lysis, and is discussed more fully in Chapter 9. The Fc portion of certain isotypes (see Table 8.2), once antigen has been bound, will activate complement; this requires that C1q, a subunit of the first complement component, cross-links two antibody Fc portions. For this to happen the two regions must be in close proximity. It has been calculated that a single IgM molecule is 1000 times more efficient than IgG. This is because two IgG molecules must be close together for complement activation. A large number of IgG molecules would be required for this to occur if the epitopes were spread. Not all isotypes activate complement, presumably because they do not have the required amino acid sequence, and therefore tertiary structure, in the Fc portion. C1q binds to residues in the CH3 domain of IgM and the CH2 domain of IgG. Some isotypes, when interacting with antigen, can activate the alternative pathway of complement that does not use C1 but gives rise to the same biological activities.
Cell binding and opsonization
The Fc portion of certain immunoglobulin isotypes is able to interact with various cell types (see Table 8.2). Antibodies specific for particular antigens, such as bacteria, play a valuable role by binding to the surface and making the antigen more susceptible to phagocytosis and subsequent elimination. This process is known as opsonization, and again is mediated by the Fc portion of the antibody. A specific conformation on the Fc region of certain isotypes is recognized by Fc receptors on the surface of the phagocyte. The important residues are in the CH2 domain near the hinge region. Individually, the interactions are not strong enough to signal the uptake of the antibody molecule; therefore, free immunoglobulin is not internalized. However, when an antigen is coated by many antibody molecules, summation of all the interactions stimulates phagocytosis or other effector mechanisms.
Certain phagocytic cells have receptors for activated complement components – complement receptors. If the binding of antibody to the antigen activates the complement cascade, various complement components are deposited on the antigen–antibody complex. Phagocytic cells that have receptors for these complement components then ingest the complexes.
The above-mentioned processes require that the antibody is first complexed with antigen. However, certain cell types can bind free antibody. Mast cells and basophils have Fc receptors that are specific for IgE. These cells perform a protective function but are also involved in the hypersensitivity reactions described in Chapter 9. In man, IgG has the ability to cross the placenta and reach the fetal circulation. This is a passive process involving specific Fc receptors. This route is limited to primates, whereas, in ruminants, immunoglobulin from colostrum is absorbed through the intestinal epithelium. Another Fc-mediated mechanism, already described, is the selective transport of IgA into mucosal secretions.
Antigen recognition
The immune system has evolved to protect us from potentially harmful material but it must not respond to self molecules. Two separate recognition systems are present:
Antibody is the recognition molecule of humoral immunity. This glycoprotein is produced by plasma cells and circulates in the blood and other body fluids. Antibody is also present on the surface of B lymphocytes. The interaction of this surface immunoglobulin with its specific antigen is responsible for the differentiation of these cells into antibody-secreting plasma cells. Antibody molecules, whether free or on the surface of a B cell, recognize free native antigen.
This contrasts dramatically with the situation in cell-mediated immunity; the T lymphocyte antigen receptor binds only to fragments of antigen that are associated with products of the major histocompatibility complex (MHC). T cell recognition of antigen is said to be MHC restricted. These MHC products are present on the surface of cells; therefore T cells recognize only cell-associated antigens. This MHC-restricted recognition mechanism has evolved because of the functions carried out by T lymphocytes. Some T cells produce immunoregulatory molecules, lymphokines, some of which influence the activities of host cells and others directly kill infected or foreign cells. Therefore, it would be inefficient or dangerous to produce these effects in response to either free antigen or antigen sitting idly on some cell membrane. The joint recognition of MHC molecules and antigen ensures that the T cell makes contact with antigen on the surface of the appropriate target cell.
B cell receptor
Antibody is found free in body fluids and as a transmembrane protein on the surface of B lymphocytes (i.e. surface immunoglobulin), where it acts as the B cell antigen receptor. The antibody present on the surface of the B cell is exactly the same molecule as is secreted when the cell develops into a plasma cell, except for the extreme C-terminal end as described above. It should be noted that the molecules present on the cell surface are present as monomers even though they are secreted in a polymeric form.
T cell receptor
The complex on T lymphocytes that is involved in antigen recognition is composed of a number of glycoprotein structures. Some of these molecules have been named systematically by CD (cluster of differentiation) nomenclature using antibodies. These generic names are used in preference to other symbols sometimes found in the literature.
The T cell antigen receptor is a heterodimer composed of an α and β or a γ and δ chain. Each chain contains a variable and constant domain, transmembrane portion and cytoplasmic tail. The variable domain folds to form a paratope that interacts with antigenic peptides associated with MHC molecules on the cell surface. The majority of T cells use the α–β heterodimer in antigen recognition. The role of cells that possess the γ–δ molecules is unknown, but they may be involved in the immune response to particular types of antigens at specific anatomical sites. The T cell receptor is the molecule that is responsible for the recognition of specific MHC–antigen complexes, and is different for every T cell. Genetic rearrangements of germline genes, similar to those seen in B cells, produce functional T cell receptors.
CD3 is present on all T cells and is non-covalently linked to the T cell receptor. The CD3 complex is thought to be involved in signal transduction, leading to cell activation, when a ligand binds to the T cell receptor. CD4 and CD8 are mutually exclusive molecules. They are present on T cells that are restricted in their recognition of antigen by MHC class II and class I molecules, respectively. Owing to their almost exclusive correlation with a specific MHC class, it is thought that these molecules bind to non-polymorphic determinants on the MHC molecules.
Major histocompatibility complex
The MHC is the part of the genome that codes for molecules that are important in immune recognition, including interactions between lymphoid cells and other cell types. It is also involved in the rejection of allografts. The MHCs of a number of species have been studied, although most is known about those of the mouse and man.
The gene complex contains a large number of individual genes that can be grouped into three classes on the basis of the structure and function of their products. The molecules coded for by the genes are sometimes referred to as MHC antigens because they were first defined by serological analysis (i.e. using antibodies).
The MHC of man is known as human leucocyte group A (HLA), and in mice it is referred to as histocompatibility 2 (H2).
Gene organization
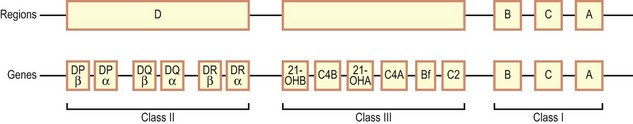
The genes that code for the HLA molecules are found on the short arm of chromosome 6. They are arranged over a region of between 2000 and 4000 kilobases in size, containing sufficient DNA for more than 200 genes. The MHC genes are contained within regions known as A, B, C and D (Fig. 8.6).
MHC class I molecules consist of two non-covalently associated polypeptide chains. A single gene that codes for the larger chain is present in the A, B and C regions, whereas the smaller chain, known as β2-microglobulin, is coded for elsewhere in the genome.
MHC class II molecules are composed of two chains, both of which are coded for within the D region. There are three class II molecules, DP, DQ and DR.
The class III genes are grouped together in a region between D and B. These genes code for a number of complement components and cytokines, but most have nothing to do with the immune system.
MHC antigen structure and distribution
The MHC class I molecule is a dimer composed of a glycosylated transmembrane protein, of molecular weight 45 000 Da, coded for within the MHC, linked to a smaller protein, β2-microglobulin (Fig. 8.7A). The globular protein formed by these two peptides is present on the surface of all human nucleated cells, except neurones. β2-Microglobulin is required for the processing and expression of MHC-encoded molecules on the cell membrane. The MHC-encoded class I glycoprotein folds into three globular domains (α1, α2 and α3) held in place by disulphide bonds and non-covalent interactions. These globular domains are found on the outer surface of the cell. There is a short cytoplasmic tail and a transmembrane portion. β2-Microglobulin is non-covalently associated with the α3 domain.
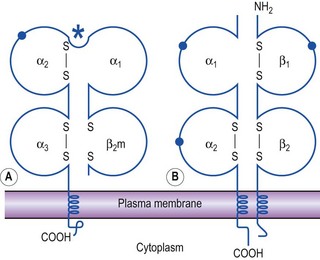
Fig. 8.7 Structure of MHC class I and class II molecules. Schematic representation of (A) class I and (B) class II molecules as found in the plasma membrane. β2m, β2-microglobulin; •, carbohydrate moieties; *, antigen-binding cleft.
The MHC class II molecules consist of two poly-peptide chains (α and β) held together by non-covalent interactions (Fig. 8.7B). They have a much more limited cellular distribution, being limited to the surface of certain cells of the immune system. In man, they are normally found on dendritic cells, B lymphocytes, macrophages, monocytes and activated T lymphocytes. Each chain is composed of two extracellular domains, a transmembrane portion and a cytoplasmic tail.
These two types of molecule are folded into domains of a similar overall structure to immunoglobulin and, along with other molecules of the immune system involved in recognition processes, are thought to have evolved from a common ancestral molecule. A number of members of this immunoglobulin supergene family are depicted in Figure 8.8. MHC class II molecules, some interleukin receptors and Fc receptors are also included in the family.
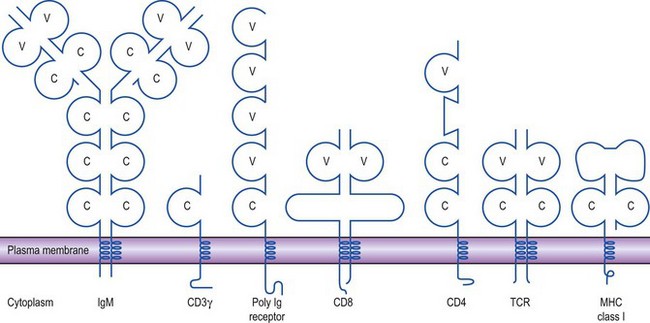
Fig. 8.8 The immunoglobulin supergene family. A number of molecules involved in the immune system display striking similarities in overall structure. Regions similar to immunoglobulin domains are shown as circles; those related to variable and constant domains are designated V and C, respectively.
The MHC antigens of each class have a similar basic structure. However, fine structural differences can be detected in the α1 and α2 domains of class I molecules and in α1 and β1 domains of class II molecules. These domains form a cleft on the outermost part of the molecules in which antigen fragments are found. The variations found are due to differences in the amino acid sequence and can be detected serologically. The variable residues give rise to different three-dimensional shapes on the MHC molecules. This, in turn, influences the selection of which antigen fragments can bind to a particular MHC molecule.
There are, therefore, many different forms of these molecules that can be identified in a population – they are highly polymorphic. Thus, it is highly unlikely that two individuals will have exactly the same MHC antigens. The MHC molecules of a particular individual can be given a designation using tissue-typing reagents. So, each chromosome of an individual will contain the genes that code for an A, B, C, DP, DQ and DR molecule. As the MHC genes are co-dominant, the products of both alleles are expressed on the cell surface. All of the nucleated cells in the body therefore express multiple copies of two HLA-A, two HLA-B and two HLA-C molecules. On certain cell types there will also be HLA-DP, -DQ and -DR molecules that were inherited from both parents.
Function
The MHC class I and II molecules are essential for immune recognition by T lymphocytes which can bind to antigens only when associated with these molecules. The different classes of molecule are involved in antigen recognition by different T cell types or subsets:
• T lymphocytes that have CD4 molecules on their surface recognize antigen in association with MHC class II molecules.
• T lymphocytes that have CD8 molecules are restricted by MHC class I molecules.
The T lymphocyte subsets perform different functions, but the division is not absolute. The one thing that they have in common is that they recognize, through their T cell receptor complex (CD3, CD4 or CD8, TCR), antigen fragments in association with MHC molecules (Fig. 8.9). In general terms:
• CD4-positive (CD4+) cells produce molecules, lymphokines, that stimulate and support the production of immune system cells.
• CD8+ cells are involved in the destruction of virally infected cells (see Ch. 9) and the destruction of tissue grafts from MHC-incompatible donors.
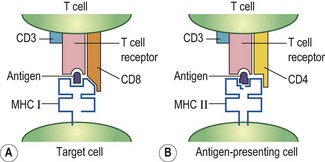
Fig. 8.9 Molecules involved in T cell recognition. (A) Antigen fragments that associate with class I molecules are recognized by T cells that have the CD8 molecule. (B) Antigen fragments that associate with MHC class II are recognized by T cells that have the CD4 molecule on their surface.
CD4+ cells produce molecules that stimulate the growth and differentiation of cells. These molecules are most effective over short distances, as they will be more concentrated. This happens when the two cells involved are actually joined together or in close proximity. The stimulation of CD4+ T cells by antigen fragments on the surface of a responsive cell, or on a cell in the vicinity of a responsive cell, greatly increases the effectiveness of the messenger molecules produced by the T cell. In cell-mediated cytotoxicity the CD8+ T cell has to bind to the infected cell so that the correct cell is killed. Therefore, the correct functioning of T lymphocytes requires direct contact with other cells. This interaction is mediated through T cell receptor recognition of antigen bound to MHC molecules on the host cell surface.