38 Leptospira
Leptospirosis; Weil’s disease
Key points
• The term leptospirosis is used to describe all infections in man and animals, regardless of the clinical presentation or strain of Leptospira involved.
• The incidence rate is low in Europe (fewer than 1 case/100 000 inhabitants) and associated predominantly with occupational or recreational exposure.
• Antibiotics offer some benefit if started within 4 days of the onset of illness, and preferably within 24–48 h.
• Animals that acquire infection may not develop discernible disease, but become long-term carriers, so-called maintenance hosts.
• More than 200 pathogenic serovars are known and each is able to infect a range of animals that may become long-term carriers capable of infecting others. The organisms can also survive for long periods in the environment. Together, these features make control of leptospirosis a substantial challenge.
The recognition of human leptospirosis as a distinct clinical entity is usually attributed to Adolf Weil of the University of Heidelberg in 1886, although the disease had been described in animals since the mid 19th century. The term Weil’s disease acknowledges Weil’s observations in differentiating what was later proven to be a leptospiral infection from other forms of infective jaundice.
In 1914, Ryokichi Inada and his colleagues in Kyushu, Japan, observed spiral organisms in the livers of guinea-pigs inoculated with blood taken from Japanese miners with infectious jaundice, presumed to be Weil’s disease. They named the organisms Spirochaeta icterohaemorrhagiae, reflecting their spiral shape and the fact that human infections were associated with jaundice (icterus) and haemorrhage. In Europe, similar organisms were demonstrated in some cases of jaundice in German soldiers involved in the First World War. In 1917, another Japanese scientist, Hideyo Noguchi, recognized that the organisms associated with Weil’s disease differed from other known spirochaetes and proposed the genus name Leptospira, meaning a ‘slender coil’. Today, more than 300 serovars have then been isolated from the environment, animals and man.
Leptospirosis is a zoonosis and has one of the widest geographical distributions of any zoonotic disease. The highest incidence is found in tropical and subtropical parts of the world. Probably every mammal has the potential to become a carrier of some serovar of Leptospira. These carriers harbour leptospires in their kidneys and excrete the bacteria into the environment when they urinate. This enables spread among their own kind and to other species, including human beings, who may directly or indirectly come into contact with their urine.
In man, the disease varies in severity from a mild self-limiting illness to the fulminating and potentially fatal disease described by Weil. Fortunately, full recovery without long-term morbidity is the most frequent outcome.
Description
Classification
The family Leptospiraceae belongs to the order Spirochaetales and can be subdivided into three genera: Leptospira, Leptonema and Turneriella. Only Leptospira spp. are considered to be pathogenic for animals and man.
The genus Leptospira was originally divided into two groups:
These two groups differ in their nutritional requirements and other phenotypic properties. For example, the growth of pathogenic strains is inhibited by the purine analogue 8-azaguanine, whereas saprophytic strains grow normally in the presence of this compound. Similarly, unlike L interrogans, L. biflexa can grow at low ambient temperature (11–13°C).
More recently, genotypic classification has defined 20 species of Leptospira:
• 9 pathogenic species (L. interrogans, L. kirschneri, L. borgpetersenii, L. santarosai, L. noguchii, L. weilii, L. alexanderi, L. alstoni, and L. kmetyi)
• 6 saprophytic species (L. biflexa, L. wolbachii, L. meyeri, L. vanthielii, L. terpstrae and L. yanagawae)
• 5 ‘intermediate’ species (L. inadai, L. broomii, L. fainei, L. wolffii and L. licerasiae), which are of unclear pathogenicity.
Leptospires are serologically classified in serovars, defined on the basis of structural heterogeneity in the carbohydrate component of the lipopolysaccharide (LPS). More than 200 different pathogenic serovars are currently recognized.
The genetic classification of Leptospira does not correlate with the phenotypic classification because serovars of the same serogroup may be distributed between different species. However, the serological classification is still widely used as it provides useful information for clinical or epidemiological investigations. The accepted nomenclature is generic name, followed by species name, followed by serovar, followed by strain (if appropriate). For example:
• Leptospira (generic name) interrogans (species name) serovar Icterohaemorrhagiae
• Leptospira (generic name) interrogans (species name) serovar Hardjo strain Hardjoprajitno.
The complete DNA sequence of strains belonging to two pathogenic species, L. interrogans and L. borgpetersenii, and one saprophytic species, L. biflexa, have been determined and this should provide insight into the molecular mechanisms of the survival and persistence of Leptospira in host and environment.
The organism
Leptospires range between about 6 and 20 µm in length, but are only about 0.1 µm in diameter, which allows them to pass through filters that retain most other bacteria. They are Gram-negative, but take up conventional stains poorly. They can be visualized by Giemsa staining, silver deposition, fluorescent antibody methods or electron microscopy. These bacteria are so thin that they are best viewed by dark-field microscopy. They have a helical-cell shape with one or both ends appearing hooked and they rotate rapidly around their long axis (Fig. 38.1).
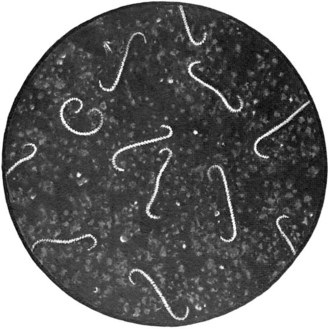
Fig. 38.1 Appearance of living leptospires as seen by dark-field microscopy. Note the very fine coils and characteristic hooked ends.
(From an original painting by Dr Cranston Low. In Low RC, Dodds TC 1947 Atlas of Bacteriology. Livingstone, Edinburgh.)
Leptospira spp. possess a double-membrane structure composed of a cytoplasmic membrane, the periplasm and the outer membrane that contains the LPS and many membrane-associated lipoproteins which are the main targets for the host immune response. Two endoflagella with their free ends towards the middle of the bacteria lie in the periplasmic space between the cell wall and the outer envelope, and are wrapped around the cell wall. Each flagellum is attached to a basal body located at either end of the cell. The flagella are similar in structure to those of other bacteria and are responsible for motility, but the mechanism involved in their rapid movement is incompletely understood.
Leptospires are killed rapidly by desiccation, extremes of pH (e.g. gastric acid) and antibacterial substances that occur naturally in human and bovine milks. They are susceptible to low concentrations of chlorine and are killed by temperatures above 40°C (after about 10 min at 50°C and within 10 s at 60°C).
Metabolism
Leptospires require aerobic or micro-aerophilic conditions for growth. Adequate sources of nitrogen, phosphate, calcium, magnesium and iron (as a haem compound or ferric ions) are essential. They can use fatty acids as their major energy source, but are unable to synthesize long-chain fatty acids with 15 or more carbon atoms. Pathogenic species require the presence of unsaturated fatty acids to utilize saturated fatty acids. Vitamins B1 (thiamin) and B12 (cyanocobalamin) are also essential and the addition of biotin is needed for the growth of some strains. These components are provided in Ellinghausen–McCullough–Johnson–Harris (EMJH) medium.
Optimal growth of pathogenic species in culture takes place at 28–30°C at pH 7.2–7.6. They are slow growing with a generation time of about 20 h: colonies are visible after 3–4 weeks on solid medium, whereas saprophytes grow more rapidly (colonies are visible after 1 week). Culture media do not generally contain selective agents as leptospires may be sensitive to them, so great care must be taken to avoid bacterial or fungal contamination at the time of inoculation and during the prolonged incubation period.
Pathogenesis
The pathogenesis of leptospirosis is incompletely understood, but a vasculitis resulting in damage to the endothelial cells of small blood vessels is probably the main underlying pathology.
Infection is acquired by direct or indirect contact with infected urine, tissues or secretions. Ingestion or inhalation of leptospires is not thought to pose a risk and human-to-human spread is very rare. Leptospires generally gain entry through small areas of damage on the skin or via mucous membranes. It is possible that they may also pass through waterlogged skin, although this is probably not a major route of infection.
The term ‘leptospirosis’ is used to describe all infections in both man and animals, regardless of the clinical presentation or strain of Leptospira involved. There are no serovar-specific disease patterns, although some serovars tend to cause more severe disease than others. In the past many names (epidemic pulmonary haemorrhagic fever, cane cutter’s disease, Fort Bragg fever, Weil’s disease, autumnal fever, etc.) were used to describe the particular clinical presentation or to reflect occupational, geographical, seasonal or other epidemiological features of leptospiral disease. Because of this, the full range of disease presentations was not appreciated and, even now, leptospiral infection may not be suspected unless the patient has the classically severe disease involving the liver and kidneys described originally by Weil. Some reports suggest that human infection with some serovars can, in rare cases, cause abortion.
Clinical features
Typically, acute symptoms develop 5–14 days after infection, although rarely the incubation period can be as short as 2–3 days or as long as 30 days. The infection presents with an influenza-like illness characterized by the sudden onset of headache, muscular pain, especially in the muscles of the lower back and calf, fever and occasionally rigors. Conjunctival suffusion and a skin rash may be seen in some cases.
During a bacteraemic phase lasting 7–8 days after the onset of symptoms, the leptospires spread via the blood to many tissues, including the brain. In severe cases the illness often follows a biphasic course: the bacteraemic phase is followed by an ‘immune’ phase, with the appearance of antibody and the disappearance of recoverable leptospires from the blood. In this phase patients may show signs of recovery for a couple of days before the fever, rigors, severe headaches and meningism return. Bleeding may occur, together with signs and symptoms of jaundice and renal impairment. Typically, bilirubin concentrations are markedly raised, but other liver function test results may be only moderately increased.
In some cases of leptospirosis, pulmonary manifestations of infection are predominant. Patients can present with cough, shortness of breath or haemoptysis. In severe cases, adult respiratory distress syndrome and pulmonary haemorrhage can supervene and lead to death.
In severe fulminating disease the patient may die within the first few days of illness, but with appropriate treatment the prognosis is usually good. Many deaths throughout the world are due to the failure to provide adequate supportive management, especially in relation to the maintenance of renal function. Generally, patients are well within 2–6 weeks but some require up to 3 months to recover fully. In a few patients, symptoms persist for many months, but neither long-term carriage of leptospires nor chronic disease has been conclusively demonstrated in man.
After infection, immunity develops against the infecting strain, but may not fully protect against infection with unrelated strains.
Laboratory diagnosis
The initial diagnosis must rely on the medical history and clinical findings backed up with details of possible occupational or recreational exposure.
Serology
Antibodies can usually be demonstrated by the sixth day after symptoms have developed (Fig. 38.2), although their detection may be delayed if antibiotics were administered early in the course of the illness.
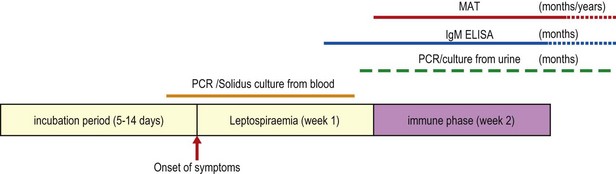
Fig. 38.2 Disease kinetics of leptospirosis. Infection produces leptospiraemia in the first few days after exposure. Leptospires are then cleared from the bloodstream as the titres of serum agglutinating antibodies increase (immune phase). Leptospires are also transiently shed in urine for long periods. ELISA, enzyme-linked immunosorbent assay; MAT, microscopic agglutination test; PCR, polymerase chain reaction.
The microscopic agglutination test (MAT), which can indicate the likely infecting serogroup or serovar, is generally accepted as the ‘gold standard’. Doubling dilutions of patient’s serum are titrated against pools of reference serovars representing the most common serogroups. After incubation, tests are read by dark-field microscopy; 50% agglutination of the leptospires by the patient’s or control serum represents a positive result. Sera collected soon after the onset of symptoms often show cross-reactivity to different serogroups in the microscopic agglutination test. In contrast, sera obtained during the convalescent phase of the illness generally show a significantly higher titre to the infecting serogroup or serovar.
Most other tests give no indication of the infecting serovar. Several enzyme-linked immunosorbent assay (ELISA) kits offer ease of use together with relatively good sensitivity and specificity. Most detect IgM antibodies, which, are detectable in the acute phase of the illness and may remain for several months after infection (Fig. 38.2).
Examination of blood and urine
The detection of leptospires in blood offers the earliest confirmation of infection. In theory, leptospirosis can be diagnosed by dark-field microscopy of blood taken during the first week of illness or, much less reliably, in urine during the second week. Dark-field microscopy of blood is technically demanding as Brownian movement of collagen fibrils, red blood cell membranes and other artefacts can resemble viable leptospires. Examination of urine is seldom worthwhile as a method of early diagnosis of infection in man.
Culture of blood may be useful in severe fulminating disease. Identification of infecting serovars requires specialized techniques that are available only in national reference laboratories. In recent years, PCR-based methods have been described and these assays are now used in many diagnostic and reference laboratories for the detection of leptospires in biological fluids of patients. Molecular diagnostic techniques offer the potential for more rapid diagnosis of leptospirosis than the currently available serological methods. This is of particular value in the critically ill patient. Reference laboratories can advise on the examination of cerebrospinal fluid or other tissue, including those taken at post mortem.
Treatment
Antibiotics offer some benefit if started within 4 days of the onset of illness, and preferably within 24–48 h. In severe illness, intravenous benzylpenicillin is the drug of choice. For milder infections a 7–10-day course of oral amoxicillin is appropriate. Patients allergic to penicillins can be treated with erythromycin.
The value of antibiotic treatment is probably overestimated and few trials have been conducted. However, supportive management to maintain tissue and organ function, such as the temporary maintenance of renal function by dialysis, may be life-saving.
Epidemiology
Animals that acquire infection may not develop discernible disease, but become long-term carriers – so-called maintenance hosts. Many rodents fall into this category. For example, rats acquiring inapparent infection with pathogenic strains may carry the bacteria in the convoluted tubules of the kidney (possibly life-long), resulting in chronic excretion of viable leptospires in their urine. Similarly, cattle may become a maintenance host for serovar Hardjo, dogs for serovar Canicola, and pigs for serovar Pomona or Bratislava. The reasons for this tolerance are unclear, as infection with other serovars may cause illness of varying severity followed by the transient shedding of the leptospires in the urine for only a few weeks. Moreover, an animal may become a long-term maintenance host for one serovar and yet develop disease and transient carriage after infection with another.
Changes in industrial, agricultural and social practices may result in the rapid change of both the density and type of animal populations in an area, with subsequent change in the predominant serovars of Leptospira causing disease in people and animals.
Viable leptospires are present in the semen of infected animals; in rodents a significant increase in the carriage of leptospires is seen once sexual maturity has been reached. Spread across the placenta occurs in several animal species, leading to infection and possibly death of the fetus.
Outside the animal host, leptospiral survival is favoured by warm, moist conditions at neutral or slightly alkaline pH. This no doubt contributes to the seasonal pattern of human infections, which peak in the summer months in both hemispheres. Even small reductions below pH 7.0 markedly reduce the survival of leptospires. The anaerobic conditions and low pH of raw sewage explains their short survival time compared with that in aerated sewage. Salt water is also relatively toxic to leptospires. They do not survive well in undiluted cow’s milk and therefore drinking unpasteurized milk poses minimal risk. However, they will survive in water at pH 7.0 or in damp soil for up to 1 month. If the soil is saturated with urine they may survive for up to 6 months, indicating the potential for long-term exposure to an infection risk even if the reservoir host has been removed for some time.
Leptospirosis in man is an emerging disease with more 500 000 severe cases occurring annually; case fatality rates exceed 10%. The disease is expected to become more important due to predicted global climate changes and rapid urbanization in developing countries where slum settlements have produced the conditions for epidemic rat-borne transmission of the disease.
Exposure to virulent leptospires may be direct, through contact with the urine or tissues of infected animals. Direct exposure is generally associated with particular occupations that bring human beings into contact with animals (e.g. butchers, veterinary surgeons, animal breeders, hunters or pet owners). Indirect exposure, through contact with freshwater or humid environment contaminated with the urine of an infected animal, is more common. Such indirect exposure is associated with particular occupations (e.g. sewer workers, rice-field workers) or situations (e.g. triathlon participants, military manoeuvres). In slum communities in developing countries, indirect exposure from rats is thought to be the main source of infection.
Infections related to exposure to surface waters have shown a significant rise in industrialized countries. The increase is almost certainly due to the greater recreational use of surface waters for activities such as canoeing, rafting, fishing, and the use of rivers for the swimming section of triathlon competitions. There has also been an increase in cases of leptospirosis acquired abroad, particularly among travellers on adventure holidays with water contact. In England and Wales between 1990 and 2004, leptospirosis was acquired by 111 travellers, 60 of whom had been to South-East Asia, compared with 194 indigenous cases.
Morbidity and mortality from leptospirosis have declined markedly because of improved hygiene levels in industrialized countries. In countries with limited facilities for medical care death may occur in 25% or more cases.
Control
With more than 200 known pathogenic serovars, each able to infect a wide range of animals that may become long-term carriers capable of infecting others, together with the organisms’ ability to survive for long periods in the environment, the complete prevention or eradication of leptospirosis is impossible.
Mass immunization of domestic livestock will prevent clinical disease in the animals and reduce the risk of human acquisition of infection.
To be fully effective, a vaccine should not only protect against disease in the animal but also prevent the establishment of the carrier state and the shedding of viable leptospires in the urine. It is also important that the vaccine contains antigens representing circulating serovars, as protection will be optimal only against the vaccine components. Current vaccines protect for only 1–2 years and the economics of farming may influence a farmer’s decision as to whether or not to immunize cattle. Cuba and China have used vaccines for mass prevention campaigns in human populations and France has used a human vaccine containing only serovar Icterohaemorrhagiae since 1981.
Awareness of leptospirosis through the education of doctors, employers and the general public has helped to develop safer practices or procedures in the workplace and during recreational pursuits. This awareness should include consideration of leptospirosis in the differential diagnosis of fever in the returning tourist. Measures to reduce rodent populations in the vicinity of human activity, such as removing rubbish, especially waste food, and prevention of the access of rats into buildings is most important. Simple measures to reduce the risks of acquiring infection also include covering cuts and abrasions with waterproof plasters and wearing protective footwear before exposure to surface waters.
In parts of the world where the prevalence of human infection in certain groups is high, selective human immunization schemes may be of benefit if a suitable vaccine is available. Antimicrobial prophylaxis with doxycycline may be of value in high-risk exposure situations in which prompt medical help is unavailable.
Faine S, Adler B, Bolin C, Perolat P. Leptospira and Leptospirosis, ed 2. Melbourne: MediSci; 1999.
Kmety E, Dikken H. Classification of the Species Leptospira interrogans and History of its Serovars. Groningen: University Press; 1993.
Ko AI, Goarant C, Picardeau M. Leptospira: the dawn of the molecular genetics era for an emerging zoonotic pathogen. Nature Reviews Microbiology. 2009;7:736–747.
Levett PN. Leptospirosis. Clinical Microbiology Reviews. 2001;14:296–326.
eMedicine. Leptospirosis in humans. http://www.emedicine.medscape.com/article/220563-overview.
Centers for Disease Control and Prevention. Leptospirosis. http://www.cdc.gov/ncidod/dbmd/diseaseinfo/leptospirosis_g.htm.
International Leptospirosis Society (ILS). http://www.med.monash.edu.au/microbiology/staff/adler/ils.html.