45 Papillomaviruses and polyomaviruses
Warts: warts and cancers; polyomavirus associated nephropathy; progressive multifocal leuco-encephalopathy
Key points
Papillomaviruses
• Papillomaviruses are ubiquitous, found in most mammalian species as well as in birds and reptiles. More than 100 types of HPV are recognized.
• Some HPVs are associated with skin lesions such as common hand warts and plantar verrucas.
• More than 30 HPV types infect the genital mucosa and are associated with a variety of lesions ranging from benign genital warts to invasive cervical cancers.
• E6 and E7 are transforming genes of HPV whose protein products bind to cellular tumour suppressors p53 and Rb respectively.
• HPV types can be grouped into high-risk and low-risk types, dependent on their oncogenic potential.
• HPV-16 is the most common high-risk HPV worldwide, and is associated with more than 50% of cervical cancers.
• HPV vaccines based on L1 VLPs have been shown to be highly efficacious, over a period of at least 7 years, against infection and disease associated with the HPV types present in the vaccine.
Polyomaviruses
• Merkel cell polyomavirus is the only human polyomavirus so far that is clearly associated with malignant tumours in humans.
• BKV and JCV are associated with diseases in immunosuppressed individuals.
• Polyomavirus-associated nephropathy (PVAN) caused by BKV is a significant cause of allograft dysfunction and graft loss after renal transplantation.
• Progressive multifocal leuco-encephalopathy (PML) caused by JCV is found mostly in patients with advanced HIV disease, but recently is also associated with the use of immune modulating therapeutic monoclonal antibodies.
Papillomaviruses and polyomaviruses are both characterized by having double-stranded DNA genomes, and were, for a long time, classified within the same virus family, the papovaviridae. While there are some notable homologies between Papillomaviruses and Polyoma viruses, more recent evidence showed significant dissimilar, genome level differences and in 2004, they were reclassified into 2 distinct virus families; the papillomaviridae and the polyomaviridae. For this reason, this chapter is divided into 2 distinct sections.
General properties
Papillomaviruses are very common, ancient viruses, thought to have existed when humans initially became a species. PVs are species-specific viruses that can infect an array of vertebrates, with the species name prefacing PV, e.g. bovine papillomavirus (BPV), canine papillomavirus (CPV) and human papillomavirus (HPV). Papillomaviruses are viruses that infect squamous epithelia and mucous membranes, and cause skin cells to proliferate. Infection with PVs can lead to benign or malignant consequences depending on a number of viral, host and environmental factors.
The malignant nature of PVs has been known for a long time. Eighty years ago, it was found that 25% of benign warts caused by cottontail rabbit PV underwent malignant change within a year, whereas in domestic rabbits up to 75% of lesions showed malignant change in the presence of tar which contains carcinogens. Similarly, cattle fed a diet containing bracken fern which contains the carcinogen quercetin, show malignant transformation of tumours of the alimentary canal (BPV type 4) and bladder (BPV type 2). Different types of HPV cause non-malignant warts and cancers seen in humans.
• an icosahedral capsid composed of 72 capsomeres (Fig. 45.1)
• a supercoiled double-stranded DNA genome, with a single coding strand, a molecular weight of approximately 5 × 106 Da and consisting of about 7900 base pairs
All papillomaviruses are dependent on the natural epithelial cell maturation or differentiation process of the cell for completion of their life cycle. Although viral DNA can be found in stem cells in the basal epithelial layer, whole virions are found only in terminally differentiated skin cells.
Human papillomaviruses
Genome organization
The HPV genome is divided into an early region with two large (E1 and E2) and several smaller (E4–E7) open reading frames and a late region with two large genes (L1 and L2). Separating the two is a non-coding region which contains regulatory elements. The E region largely encodes the proteins responsible for pathogenicity, while the L region encodes the structural proteins. The coding sequences are distributed in all three open reading frames with considerable overlap along a single strand of DNA. Cumulatively, HPV encodes 8 proteins. Unravelling the functions of these proteins has contributed significantly to our understanding of the pathogenicity of different HPV types and particularly to the transforming ability of some types.
Life cycle
HPV requires breaks in the surface layers, sometimes referred to as micro wounds to infect basal epithelial cells. In most PVs, the L1 capsid protein binds to heparan sulphate proteoglycans on the basement membrane which facilitates a conformational change to the virus. This in turn exposes L2 which then facilitates transfer to a second, unknown epithelial receptor. The virus enters the cell in an endosome. Genome is uncoated and reaches the nucleus approximately 24h after initial attachment of virus. Recent evidence suggests this is through nuclear membrane pores following breakdown of the membrane during mitosis.
HPV does not encode its own polymerase, so in order to produce new virus particles, it must make use of the dynamic cellular resources available in a differentiating cell. Like other viruses, the productive life cycle for HPV has several stages. The first stages of uncoating and viral genome maintenance expression with a small a mount of genome amplification occur in the basal layer; high levels of genome amplification and particle assembly take place in the spinous and granular (suprabasal) layers while particle release is confined to the uppermost cornified layer. HPV completes these stages through a tightly regulated sequence of gene expression (Table 45.1) and maintains a stable extra-chromosomal state referred to as an episome throughout.
Table 45.1 Papillomavirus gene functions
| Gene | Function | Comments |
|---|---|---|
| E1 | Episomal maintenance | Frequently disrupted by integration |
| E2 | Regulation of transcription and replication. DNA-binding protein; very similar in BPV and HPV | Regulates transcription and viral replication in association with E1. Frequently disrupted by integration |
| E4 | Virion maturation, disrupts cytoskeleton | May facilitate release of virions from differentiated keratinocytes |
| E5 | Transforming function – alters signalling from growth factor receptors | Enhances transforming capabilities of E6 and E7 |
| E6 | Transforming function – binds to p53 and other pro-apoptotic proteins | Co-operates with E7 to stimulate cells into S phase, retard cell differentiation and increase efficiency of transformation |
| E7 | Major transforming gene – binds to members of pocket protein family such as Rb | Induces proliferation. Works cooperatively with E6 but is capable of transforming cells independently of E6 |
| L1 | Production of major capsid proteins, facilitates attachment and entry | Group- and type-specific determinants (for classification) |
| L2 | Production of minor capsid proteins, facilitates attachment and entry | Type-specific determinants (for classification) |
| URR (upstream regulatory region) | Regulation of gene function and initiator of viral replication | Non coding region. Contains both positive and negative transcriptional control elements. |
However HPV does not always complete a productive life cycle and where this organized pattern of events is lost, significant abnormal changes to the cell called transformation can occur. The usual life cycle is ‘aborted’.
Transformation
Transformation is an unlikely and unfortunate consequence of HPV infection. Transformation depends on several interlinking factors including HPV type, location and duration of infection, extent of immune response and environmental and constitutive risk factors. E6 and E7 are considered the primary transforming viral proteins (oncoproteins), although E5 plays a supporting role in enhancing the actions of both.
It is thought that integration of the viral genome into the host chromosome releases E6 and E7 from transcriptional control, leading to the de-regulated high-level protein production that is necessary for cell transformation. Most HPV-associated tumours therefore show integrated rather than episomal virus. E6 and E7 work cooperatively: E7 induces proliferation of suprabasal cells which avoid apoptosis through the actions of E6. These oncoproteins target and disable pRb and p53, two key proteins involved in cell cycling that normally identify abnormal growth features and prevent proliferation. E6 and E7 can also act directly on centrosomes leading to genomic instability and abnormal mitosis which would normally trigger p53-mediated apoptosis. For these reasons p53 is often referred to as ‘the guardian of the cell’. These viral–cellular interactions lead to unchecked growth of genetically unstable cells, vulnerable to secondary mutations and susceptible to malignant progression. The properties and affinities of E6 and E7 oncoproteins associated with high-risk HPV types are quite different to the properties of these proteins in low-risk HPV types.
Classification of HPV
Due to their complex lifecycle, HPVs are difficult to culture in the laboratory so their characterisation has been performed through genome sequence analysis. The formal, systematic classification system defines an HPV type as a complete HPV genome whose L1 nucleotide sequence differs from that of any other genome by at least 10%, with HPV types distinguished by a number according to when they were discovered. Currently, well over 100 types of HPV are recognized and these are organized into separate genera (Fig. 45.2). Within the genera or ‘major branches’ of the phylogenetic tree, minor branches contain HPV species which unite types that are genomically distinct but similar in terms of their biological activity. Further subdivisions occur including HPV subtypes that differ by between 2–10% and HPV variants that differ by <2%.
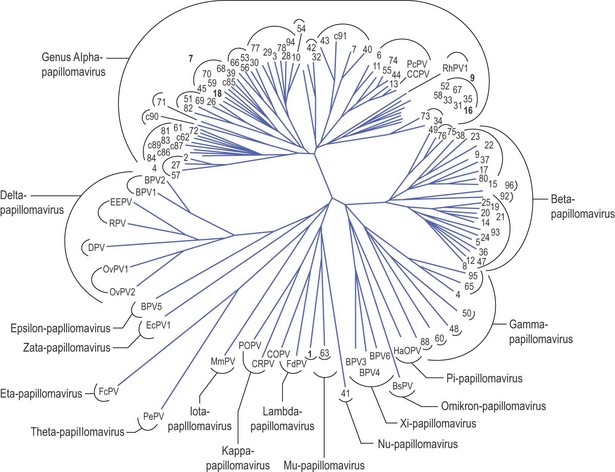
Fig. 45.2 HPV phylogenetic tree. The majority of oncogenic HPVs are classified within the alpha genus; alpha species groups 9 and 7 contain types HPV 16 and 18 respectively (highlighted), which are responsible for the majority of cervical cancers.
Adapted from de Villiers et al (2004) Virology 324:17–27.
Different HPVs have evolved to fill different biological niches and HPVs are frequently grouped according to the skin type and/or anatomical area with which they are associated. Consequently, the descriptive terms cutaneous HPVs, mucosal HPVs and genital HPVs are sometimes used, and these link to different branches of the phylogenetic tree, e.g. cutaneous types form one clear branch and genital types with greatest malignant potential form a second branch.
Another way of stratifying HPV is as ‘low-’ or ‘high-risk’ (LR or HR) according to their potential association with cancer. Evidence for risk status comes from studies where the types detected in cancer cases are compared to the types detected in non-cancerous control tissue. A recent study of over 30 000 cervical cancers revealed the 13 most common HR-HPV types to be -16, -18, -58, -33, -45, -31, -52, -35, -59, -39, -51 and -56, with HPV-16 and/or HPV-18 accounting for 70–76% of invasive cervical cancer in all world regions. Table 45.2 shows the clinical associations of HPV types.
Table 45.2 Classification and clinical manifestations of HPV types
| Genus | Type | Clinical Associations |
|---|---|---|
| Alpha | HPV-2, -27, -57 | Common skin warts and sometimes genital warts in children |
| HPV-6, -11 | Benign mucosal lesions – genital warts, laryngeal papillomas | |
| Species 9: HPV-16, -31, -33, -35, -51, -52, -56, -58, -59 | Pre-invasive squamous and glandular lesions and cancers (high-risk types) | |
| Species 7: HPV-18, -45, -59 | Pre-invasive squamous and glandular lesions and cancers (high-risk types) | |
| Beta | HPV-5, -8 | Cutaneous benign and malignant lesions in immunosuppressed patients |
| Gamma | HPV-4, -65 | Cutaneous benign lesions |
| Mu | HPV-1, -63 | Cutaneous benign lesions, frequently on feet |
Adapted from Bernard SU 2005 The clinical importance of the nomenclature, evolution and taxonomy of human papillomaviruses. Journal of Clinical Virology 32S: S1–S6.
Transmission and epidemiology
HPV requires close skin to skin contact for transmission and most people will acquire an HPV infection at some point in their life. HPV is also a relatively stable virus and has been found in fomites such as damp surfaces and towels. Wet skin is more vulnerable to surface breaks hence the prevalence of plantar warts (verrucas; HPV-1) associated with swimming pool environments. Sexual contact/intimacy is the main route for transmission of HPV to the genital area, although transmission does not require penetrative sex. In males, HPV has been detected on several areas of the penis, the scrotum, the area surrounding the anus and the buttocks. Similarly in females, HPV can be found at all genital sites. Occasionally HPV can be transmitted to the genital areas by non-sexual routes; a mother with HPV can infect her child intra-partum and family studies have shown evidence of horizontal transmission between carer and child.
The mean duration of infection varies according to HPV type and age of the infected individual. Genital carriage of HPV decreases dramatically with age in females with highest prevalence around 40% in the 20–25 year group. Although there is less epidemiological data in males, recent evidence suggests infection levels are more consistent over the adult years. Most HPV infections clear within 1–2 years, depending on other influences including smoking, persistent oral contraceptive use, co-infection with other STIs and early age of first intercourse.
About 90% of genital warts are associated with the low-risk HPV-6 and HPV-11. HR-HPV such as HPV-16 and HPV-18 are more likely to be associated with subclinical or latent infections which can be detected by looking for HPV DNA. From many international studies, the median prevalence of high-risk HPV has been estimated to be 15% among women with normal cervical cytology. The peak prevalence is found in young women aged under 25 years, and declines with age. Persistent infection with HR-HPV carries a much greater risk of progression to cancer.
Clinical features and pathogenesis of HPV infections
Cutaneous warts
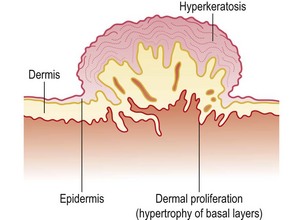
Cutaneous warts commonly infect the keratinized epithelium of the hands and feet, producing typical warts frequently seen in young children and adolescents (Fig. 45.3A). Typically, HPV-1 and HPV-4 are commonly found on the feet, whereas HPV-3 and HPV-10 are associated with flat warts and HPV types 2, 4 and 7 with common warts. HPV-7 can be particularly troublesome in butchers and fishmongers where frequent exposure to water causes maceration of the skin and facilitates virus entry and spread. Histologically, the lesions are benign with hypertrophy of all layers of the dermis and hyperkeratosis of the horny layer (Fig. 45.4). They usually disappear spontaneously but occasionally may be resistant to treatment. Regrowth of the lesions after treatment is probably due to persistence of the virus in the skin surrounding the original wart.
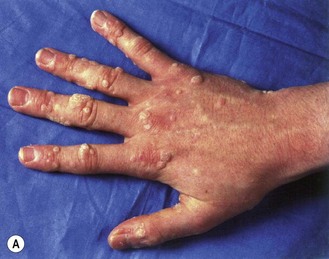
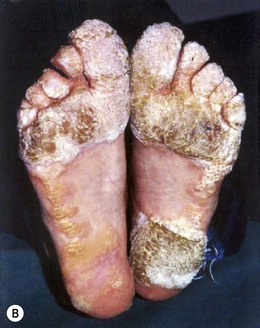
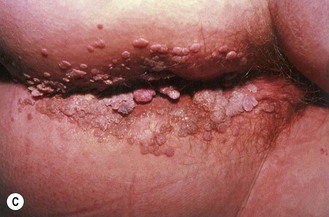
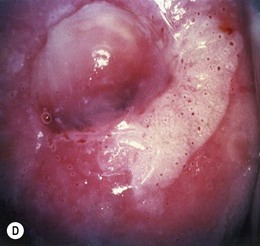
Fig. 45.3 Typical clinical presentations of HPV. (A) Common hand warts on child. (B) Extensive plantar warts in renal transplant patient. (C) Genital warts (condylomata acuminata). (D) Cervical flat wart after application of acetic acid.
Courtesy of Dr MH Bunney and Dr EC Benton.
Host factors contribute to the control and outcome of HPV infections. Both humoral and cell-mediated immune responses develop, with regression largely resulting from T helper (TH) type 1-driven cytotoxic T cells and protection from subsequent infection with the same HPV type from TH2 stimulation of B cells. Patients with primary or secondary cell mediated immunodeficiency have an increased risk of developing warts which can be extensive (Fig. 45.3B), persistent or recurrent. For example, in people suffering from the rare genetic skin disorder epidermodysplasia verruciformis (EV), in which there is a selective depletion of specific T cell clones, large plane warts associated with virus types such as HPV-5 and HPV-8 develop and persist for life. Similarly, up to 40% of renal allograft recipients develop cutaneous warts within a year of receiving the graft, rising to more than 90% in those with graft survival for longer than 15 years. EV-associated HPV types are found in a variety of lesions, including a high proportion of psoriatic skin lesions but also in normal skin and hair follicles in people with no immune defects, suggesting that the viruses are widespread and that development of visible lesions is well controlled by the immune system of healthy individuals.
Anogenital warts
Genital HPV infections are very common in the general population and a huge increase in incidence has been reported in recent years. Anogenital warts (also known as condylomata acuminata; Fig. 45.3C) are found predominantly in sexually active adults and are the most common clinical manifestation.
In women, vulvar and vaginal warts are usually plainly visible, but on the cervix they may be indistinguishable from the normal mucosa without the aid of a colposcope to magnify the cervical epithelium. Subclinical lesions may proliferate and become clinically apparent if the immune response is disturbed, as in pregnancy. The application of 5% acetic acid causes whitening of epithelium in which there is a high concentration of nuclear material and reveals subclinical lesions as areas of densely white whorled epithelium known as flat or noncondylomatous warts (Fig. 45.3D). In men, the most common sites for lesions are the shaft of the penis, perianal skin and the anal canal. Subclinical infection on the penis can be detected using a colposcope and 5% acetic acid as in women.
Orolaryngeal lesions
Recurrent respiratory papillomatosis (RRP)
This is a rare condition characterized by the presence of benign squamous papillomata on the mucosa of the respiratory tract, most commonly on the larynx. It has a bimodal distribution with peaks of incidence in children under 5 and adults over 15 years of age, and is caused by infection with HPV types 6 and 11. Children acquire the disease by passage through an infected birth canal, whereas adults acquire the disease from orogenital contact with an infected sexual partner. The transmission rate is low. The disease presents with an abnormal cry in children and/or hoarseness of voice. As the lesions grow they may cause life-threatening upper airway obstruction that requires emergency surgery. Recurrence after treatment is common. Malignant conversion of laryngeal papillomata has been described in the past, usually after radiotherapy to treat the initial lesion.
Oral papillomatosis
A variety of papillomata and benign lesions occur on the oral mucosa and tongue and are associated with a range of HPV types. The occurrence of multiple lesions on the buccal mucosa is known as oral florid papillomatosis. Subclinical lesions can be detected on the oral mucosa of normal adults after the application of acetic acid. The virus types here are those found more commonly in the genital tract, and infection is acquired during orogenital contact with an infected sexual partner.
HPV and cancer
Premalignant lesions of the genital tract
Malignant disease of the cervix is preceded by neoplastic change in the surface epithelium, a condition known as cervical intra-epithelial neoplasia (CIN). The initial transforming event takes place in the deepest layer of the epithelium, the germinal layer, and abnormal cells spread through the surface layers. This condition increases in severity from:
• CIN-I (low-grade squamous intra-epithelial lesions, LSIL in American classification) which can be associated with both low (including HPV-6 and HPV-11) and high-risk types and is very unlikely to progress
• CIN-II (high-grade or HSIL) in which abnormal cells including mitotic figures can be found up to the mid-epithelium with loss of stratification and differentiation. CIN-II often regresses spontaneously
• CIN-III (also called high-grade or HSIL, but with more extensive disturbance and mitotic figures including superficial layers of epithelium) which, if untreated, can progress to invasive cancer in a large proportion of affected individuals (Fig. 45.5).
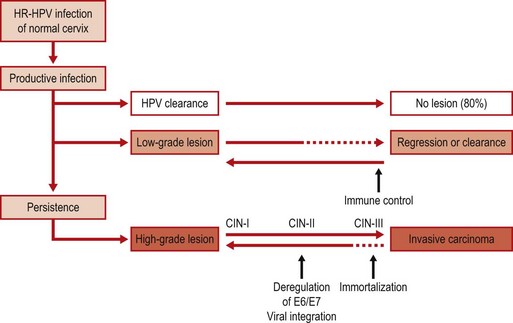
Fig. 45.5 HPV infection and pathogenesis of cervical cancer. CIN, cervical intra-epithelial neoplasia; HR, high risk.
Infection with multiple types is common but is not associated with an increased risk of progression. The development of intraepithelial neoplasia at other sites is similar (VIN for vulva; VAIN for vagina and AIN for anus) and HPV DNA can be detected in all grades of the premalignant lesions of the female and male genital tract.
Invasive cancers
The association of HPV with invasive cancers of the skin and aero-digestive tract as well as the genital tract is well documented. Malignant conversion of skin warts occurs in about one-third of patients with EV at a relatively young age on skin exposed to sunlight, with HPV types 5 or 8 in more than 90% of squamous cancers. Malignant conversion of cutaneous warts also occurs in renal allograft recipients.
It is now recognized that a subset of oropharyngeal squamous cell cancers (OSCC), particularly those in the lingual and palatine tonsils, have an HR-HPV aetiology. The incidence of HPV-associated OSCC has risen in the last decade, particularly in individuals <50 years old who have been less exposed to the ‘traditional’ risk factors of drinking and smoking. Interestingly, HPV-associated OSCC has a better prognosis compared to HPV negative OSCC.
The most common association with invasive cancer, however, is with tumours of the anogenital tract. More than 99% of invasive cervical cancers contain HPV DNA. The proportion of cancers due to different HPV types varies around the world, and the incidence shows wide demographic, ethnic and socio-economic variation. Key facts include:
• Throughout the world, HPV-16 is by far the most common HR-HPV.
• HPV-16 is the cause of more than 50% of invasive cervical cancers worldwide.
• Cervical cancer is the second most common cancer of women worldwide with a mean incidence of 35 per 100 000.
HPV-18 and the related HPV-45 are found more frequently in the more aggressive adenocarcinomas. In-situ DNA hybridization of tumour biopsies has shown that HPV DNA is unevenly distributed throughout the tumour, suggesting that these cancers are polyclonal in origin.
In most animal cancers associated with papillomavirus there is a co-factor. In man the co-factors associated with genital tract malignancies are similar to those noted above for HPV infection with smoking, seminal fluid factors, immune status, long-term use of oral contraceptives and genetic background being most significant. In the development of HPV-related carcinomas at other sites, exposure to ultraviolet or ionizing radiation and chewing of betel-quid are additional risk factors. Thus:
• Women who smoke are at increased risk of CIN and cancer of the cervix.
• Women who are immunosuppressed, whether due to drug treatment (transplant recipients) or secondary to haematological malignancies (leukaemias) or to infection with human immunodeficiency virus (HIV), are also at increased risk of cervical cancer.
The incidence of anal cancer in men who have sex with men and in HIV-infected men is similar to that of cervical cancer. It is thought that the presence of a cellular transformation zone makes these sites more susceptible to the oncogenic effects of high-risk HPV infection. In contrast, vaginal, vulval and penile cancers are rare, and these areas lack a transformation zone.
Laboratory diagnosis
Morphological identification
HPV infection may be readily diagnosed when there are typical, visible clinical lesions. In cervical smears, HPV infection can be recognized morphologically by the presence of vacuolated cells with enlarged hyperchromatic nuclei described as koilocytes. However, koilocytes are not always present, are not sufficiently specific for HPV, and cannot differentiate between low- and high-risk HPV types. Histological examination of a biopsy taken from a lesion identified at colposcopy will show more specific features of HPV infection, including papillomatosis, hyperkeratinization of the surface layer, hypertrophy of the basal layers and disorganization of the epidermal structure (Fig. 45.4).
Serology
The lack of native antigen hampered the development of serological assays for several years. However serological assays for HPV now exist using virus-like particles (VLPs) as a source of antigen. These assays are useful for natural history studies where past infection is assessed and also for monitoring vaccine induced immunity. However, as HPV has no viraemic phase, is non-lytic and creates little inflammation, natural infection induces a low-level antibody response with a <50% seroconversion rate. Consequently, serology tests lack sufficient sensitivity to be useful for clinical management.
Molecular detection
Nucleic acid based detection of HPV DNA and RNA has been developed extensively for research, epidemiological and clinical use. Clinical applications often focus on the detection of DNA from a group of clinically relevant HR-HPV types and most are based on the detection of HPV-L1 DNA. Sensitivity is high but because infection is so common it is difficult to distinguish between clinically significant persistent infections and transient infections. RNA tests which detect transcription of E6 and E7 oncogenes have the potential to be more specific for disease. Work is also progressing on the detection of secondary markers known to be triggered by high level oncogenic expression including the cellular marker, p16INK4a. In research and surveillance, specialized genotype-specific assays are more relevant. These are often based on PCR amplification using consensus or degenerate primers followed by hybridization to type specific probes and colorimetric detection of the hybrids in nylon strips.
In cervical screening, HR-HPV DNA tests which give a presence/absence result are most frequently used and are extremely sensitive for the detection of high-grade cervical lesions (between 95–100%). Women who test negative are very unlikely to have significant disease. For effectiveness, cytology screening needs to be repeated regularly (every 3–5 years in the UK between ages 20–65) as it is only around 60% sensitive for the detection of significant lesions. Nevertheless, the lower specificity of HPV screening tests, particularly in young women (<30 years) who have high prevalence of infection means that tests should be performed only in appropriate contexts, where the result will be meaningful and help to inform patient management.
Cervical disease management systems incorporating HPV testing into the repertoire include:
Triage of low-grade (cytological defined) abnormalities
While most low-grade abnormalities will clear, a proportion (15–20%) will harbour significant lesions. HPV testing is use to stratify risk, with HPV-negative women subjected to a less intensive clinical follow up than high-risk HPV-positive women.
Post-treatment follow-up (test of cure)
After treatment of high-grade lesions, women are monitored to ensure their treatment has been successful. HPV testing can help to identify potential residual disease and reduce the intensity of follow-up in women who test HPV negative.
HPV primary screening followed by cytology triage
Several European trials, together recruiting over 250 000 women, have shown the ability to detect high-grade disease more quickly with HPV screening than with cytology. New clinical testing algorithms based on HPV primary testing, followed by cytology of HPV-positive cases, are being introduced and will allow the screening interval to be increased to 5–6 years or longer in women over 30 years of age.
Treatment and control
In most immunocompetent people, warts are a cosmetic nuisance and will eventually disappear spontaneously. Warts may be destroyed by cryotherapy with dry ice or liquid nitrogen, but simple topical treatment with salicylic acid at home is often successful. Genital warts are more problematic. They are the most common sexually transmitted infection and represent a significant burden to healthcare providers. In pregnant women, vaginal warts may occasionally grow to such a size that the birth canal is obstructed and surgical removal is required. Prevention of spread of wart viruses can be achieved by avoiding contact with affected individuals. Thus, the use of condoms will diminish the risk of spread of genital warts. Interferon, photodynamic therapy and indole-3-carbinol have been used for treatment of recurrent laryngeal warts after the reduction of tumour load by cautery or excision.
Many treatments can be used for genital warts including:
• antiproliferative agents such as podophyllin or 5-fluorouracil, although close monitoring is required
• destructive therapies such as trichloracetic acid, liquid nitrogen or surgical excision
• immunomodulators such as imiquimod, which activates monocytes/macrophages and causes direct release of interferon-α (self-applied topical treatment).
All of these treatments will remove the lesions but do not always eradicate the virus from surrounding normal epithelium. Incomplete treatment is the most common cause of recurrence of warts; all of a patient’s warts must have disappeared with restoration of normal skin texture before a cure is considered. This is also true for treatment of CIN. Long-term follow-up to ensure clearance is a significant cost to health care and a significant anxiety for women.
Vaccines
Vaccines are available which can prevent the development of papillomata and cancers in cattle and rabbits, consisting principally of virus-like particles of L1, or L1 with L2 proteins. The development and introduction of prophylactic vaccines for HPV based on genetically engineered virus-like particles has provided a unique opportunity to limit the spread of HPV infection and the global burden of HPV related cancers and its precursors. Two vaccines are currently licensed, one containing L1 of HPV-16 and HPV-18 (bivalent vaccine) and the other L1 of HPV-6, -11, -16 and -18 (quadrivalent vaccine). Both proved to be safe and highly efficacious in clinical trials, providing almost complete protection from high grade cervical disease associated with HPV-16 and HPV-18 (both vaccines) and from genital warts (quadrivalent vaccine). In 2008, the UK introduced a national schoolgirl immunization programme for 12-13 year olds which has achieved record coverage of 80–90%. Vaccine is available in most countries, but is very expensive. For maximum effectiveness, population-based programmes with immunization before sexual debut should be implemented. While protection from infection is now possible, it will be many years before the ability of vaccine to prevent or halt cervical cancer development can be detected and even then only by implementing robust programmes of surveillance.
Therapeutic vaccines for the control of existing HPV-associated disease focus on HPV oncogenes and aim to generate potent E6 and E7-specific T cell-mediated immune responses. Modified DNA vaccines with improved immunogenicity provide a potentially promising approach and have reached the stage of clinical trials.
Polyomaviruses
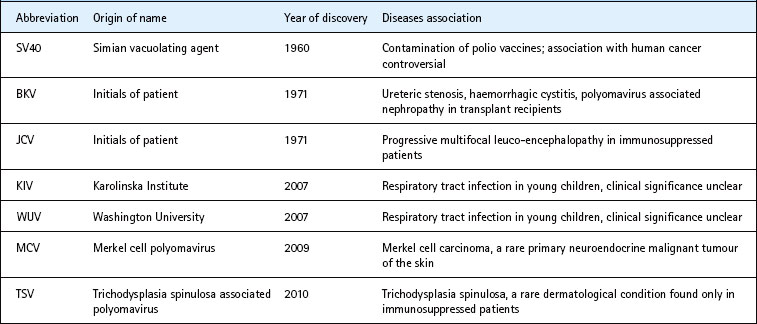
The term polyomaviruses literally stands for many (poly) tumour (oma) viruses, so called due to their ability to induce various tumours in experimental animals. So far, seven human polyomaviruses have been identified (Table 45.3). Initially, only murine and hamster polyomaviruses appeared to be oncogenic in their natural hosts. However, the more recently discovered Merkel cell polyomavirus has a clear association with Merkel cell carcinoma, a rare human neuro-endocrine cutaneous tumour found in the immunosuppressed or those exposed to sunlight. The older members BK virus and JC virus have no clear association with malignancies and both were named after the initials of the patients from whom they were first isolated.
The virions are 42–45 nm in size with a 72-capsomere icosahedral capsid. The genome consists of a double-stranded super-coiled loop of DNA, approximately 5100 bp in length, with both strands coding for virus proteins. Capsids contain three structural proteins, VP1, VP2 and VP3.
Replication and transformation
There are three non-capsid regulatory proteins: large T (T Ag), small t and agnoprotein. Murine polyomaviruses have an additional middle T Ag. These are the first antigens to appear after infection; they accumulate in the nucleus, stimulate cellular growth and are important for replication. This is followed by a switch to transcription of the late region of the genome and production of the three structural capsid proteins. The growth cycle in culture is 36–44 h, and the release of mature virus particles follows lysis of the cell. The structural proteins determine host range and infectivity. BKV can be grown with difficulty in human embryonic kidney cells, whereas JCV, which is not only species but also tissue specific, replicates only in human embryo glial cell cultures. Recently, the 5-hydroxytryptamine 2A (5HT2A) receptor for serotonin on the surface of glial cells was identified as the receptor protein for entry of JCV.
The early proteins are associated with immortalization and transformation of host cells. Large T antigen binds to both Rb and p53, and prevents the induction of cell death. Polyomaviruses do not have a viral DNA polymerase. Instead, they utilize the host DNA polymerase α/primase complex to which T Ag binds. T Ag also binds to double stranded (ds) DNA and has helicase activity to help to unwind the viral dsDNA for DNA replication.
Clinical features and pathogenesis
Primary BKV and JCV infections are probably mostly asymptomatic. Rarely, upper respiratory tract symptoms, cystitis and encephalitis have been described. SV40 is primarily a simian virus. Its association with human infection is related to the inadvertent use of SV40 contaminated poliovirus vaccines in the 1950s and 1960s. The role, if any, of SV40 in human diseases is controversial. WUV and KIV are found in respiratory samples. Their role in human respiratory tract infection is at present unclear.
Reactivation of BKV and JCV is common, particularly in organ transplant patients, with up to 40% of renal allograft recipients and haematopoietic stem cell transplant (HSCT) patients excreting a polyomavirus in urine in the early months after transplantation. BKV reactivation is associated with haemorrhagic cystitis (HC) in HSCT patients and polyomavirus associated nephropathy (PVAN) in renal transplant patients. JCV is associated with progressive multifocal leuco-encephalopathy (PML), seen in immunosuppressed patients, most commonly patients with advanced HIV infection.
Haemorrhagic cystitis (HC)
This is a common complication after HSCT. Early onset HC (<2 weeks) is probably not related to viral infection. However, late onset HC is often related to BK reactivation. BK virus excretors are 4 times more likely to develop HC than non-excretors and there is a strong temporal relationship between BK excretion and onset of HC. Pathogenesis may be related to immune reconstitution as its onset often coincides with engraftment.
Polyomavirus-associated nephropathy (PVAN)
The association of BKV reactivation in the renal allografts of transplant recipients with graft dysfunction was made only in the last 10 years. Its discovery coincided with the introduction of newer more potent immunosuppressive agents such as tacrolimus and mycophenolate mofetil. So far, no specific immunsuppressive agents have been clearly associated with PVAN. Rather, the overall level of immunosuppression is thought to be responsible.
PVAN is mostly reported in patients receiving renal allografts and only rarely after other transplantations. Onset of PVAN occurs at a median time of about 6 months after transplantation and up to 1–5% of renal allografts are affected. Men are more likely to develop PVAN than females in most case series. The only presenting clinical feature is that of deterioration in graft function. Hence, the major differential diagnosis is allograft rejection. Renal biopsy is the gold standard for differentiation of these two conditions. However, non-invasive methods of diagnosis of PVAN are also available. Since the management of graft rejection is to increase immunosuppression whereas that of PVAN is to decrease it, an accurate diagnosis is necessary to inform management.
Progressive multifocal leuco-encephalopathy (PML)
This condition, associated with the reactivation of JCV in the brain, was first described in 1958 in patients with Hodgkin’s lymphoma and chronic lymphocytic leukaemia. Over the past 10 years, PML has been found almost exclusively in patients with acquired immune deficiency syndrome (AIDS). More recently, PML has also been described in patients receiving monoclonal antibody therapy such as rituximab and natalizumab.
Patients with PML have multiple foci of demyelination, usually affecting white matter in the cerebral hemispheres, but occasionally elsewhere in the central nervous system. Affected oligodendrocytes become swollen, with hyperchromatic nuclei and occasional basophilic inclusions pathognomonic of PML. Replication of the virus occurs in the nucleus, causing cell destruction and breakdown of the myelin sheath.
The clinical features depend on the areas affected. Symptoms include visual, mental and speech impairment, hemiplegia, loss of memory, personality change and dementia. Death usually occurs within 6 months of the first signs of the disease. Other causes of similar symptoms such as cerebral lymphoma, toxoplasmosis and tuberculosis need to be excluded.
Laboratory diagnosis
In HC, polyomaviruses can be detected in large quantity in urine. The presence of BKV in the urine can be confirmed by BKV specific PCR. Patients with uncomplicated HC do not normally have significant BK viraemia (i.e. virus detectable in perpipheral blood).
The gold standard of PVAN diagnosis is histological examination of renal biopsy and identification of viral inclusion bodies in the renal tubular cells. The presence of polyomavirus can be confirmed by immunohistological staining using monoclonal antibodies against polyomavirus T Ag. Histology can also exclude or detect concomitant presence of graft rejection. Non-invasive methods such as urine cytology detection of polyomavirus infected uroepithelial cells (decoy cells) or detection of polyomavirus in urine by electron microscopy, though sensitive, are non-specific and have poor positive predictive values. The best non-invasive diagnostic method, with the highest positive predictive value, is quantitative plasma BK viral load. Serial plasma viral load can also be used to monitor the progress of the disease.
The ‘gold standard’ for diagnosis of PML depends on a combination of neuroimaging and biopsy. JCV DNA may not be detectable in the cerebrospinal fluid (CSF) even with the use of highly sensitive nested PCR. Intrathecal antibody detection may be useful if JCV DNA is negative in CSF.
Antibodies to polyomaviruses can be measured using haemagglutination inhibition (HAI) and enzyme-linked immunosorbent assay (ELISA). HAI uses whole virions and gives a species-specific result, whereas in ELISA disrupted virions or purified VP1 can be used to give a more sensitive and type-specific result. Rising titres and the presence of immunoglobulin M are diagnostic of recent infection. Failure to detect antibody to JCV nearly always excludes a diagnosis of PML.
Transmission and epidemiology
Very little is known about the transmission of polyomaviruses. Both BKV and JCV are frequently detected in urine. Respiratory, sexual and fecal-oral routes of transmission are all possible. In human tissues, BKV DNA may be found in tonsils, lung, lymph nodes and spleen; and JCV DNA in lung, liver, spleen, lymph nodes and leucocytes.
Serological studies show that BKV infection is a common event in early childhood. By 3 years of age 50–60% of children have antibody, rising to almost 100% by the age of 10 years. JCV circulates independently of BKV in the community, and acquisition of antibodies is slower and increases steadily with age to about 60–70% in adulthood.
Treatment
Cidofovir is active against polyomaviruses in vitro. As polyomaviruses do not carry viral DNA polymerase, the action of cidofovir is likely to be targeted against other proteins such as host polymerase or viral T Ag. Its clinical use in HC or PVAN has not been particularly successful. In addition, cidofovir is nephrotoxic and can only be used in very small doses in renal patients. Leflunomide is a mild immunosuppressive agent licensed for the treatment of rheumatoid arthritis. It also has anti-viral properties and has been used as a treatment for PVAN. It has significant haematological toxicity which can be restrictive and dose limiting. The main management of PVAN is pre-emptive reduction of immunosuppression. In order to achieve this, non-invasive surveillance methods for PVAN such as quantitative plasma viral load measurement need to be available.
There is no established treatment for PML. In HIV patients, the use of combination anti-retroviral therapy (cART) may prolong survival, but this could paradoxically also increase the damage through immune reconstitution inflammatory syndrome (IRIS). Several chemotherapeutic agents, including cytosine arabinoside (ARA-C, cytarabine), cidofovir and mefloquine, have been tried. With the recent discovery of JCV utilizing the serotonin receptor to enter into glial cells, there has been interest in investigating the use of 5HT2a antagonists.
Bosch FX. Prevention of Cervical Cancer: Progress and Challenges on HPV Vaccination and Screening. Vaccine. 2008;26:K1–K94.
Dörries K. The Human Polyomaviruses. Zuckerman AJ, Banatvala JE, Schoub BD, et al. Principles and Practice of Clinical Virology, ed 6, Chichester, UK: John Wiley & Sons Ltd, 2009.
Khalili K, Stoner GL. Human Polyomaviruses. Molecular and Clinical Perspectives. New York: Wiley-Liss, 2001.
Li N, Franchesci S, Howell-Jones R, et al. HPV type distribution in 30,848 invasive cervical cancers worldwide: variation by geographical region, histological type and year of publication. International Journal of Cancer. 2011;128:927–935.
Marur S, D’Souza G, Westra WH, Forastiere AA. HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncology. 2010;11:781–789.
McCance D. Papillomaviruses. In: Zuckerman AJ, Banatvala J, Schoub BD, et al. Principles and Practice of Clinical Virology. ed 6. Oxford: Wiley-Blackwell; 2009:807–822.
Moody CA, Laimins LA. Human papillomavirus oncoproteins: pathways to transformation. Nature Reviews in Cancer. 2010;10:550–560.