58 Rhabdoviruses
Key points
• Rabies has the highest case-fatality (100%) rate of any infectious disease once clinical symptoms are observed. There are at least 55 000 cases annually worldwide.
• Over 99% of human rabies infections are due to bites from a rabid dog. Most of these occur in developing countries and the majority in children under the age of 15.
• Pre- and post-exposure vaccination is available and both are extremely effective.
• A paradigm shift is required to focus rabies control efforts towards the elimination of rabies in domestic dogs.
• Reducing the risk of rabies transmission to humans means implementation of vaccination schemes for both domestic animals and wildlife.
• The number of reported human rabies cases due to bat bites is on the increase worldwide.
• Vaccination of people in high-risk groups (laboratory workers, animal handlers and those travelling to rabies endemic areas) is recommended.
The rhabdoviruses belong to the order Mononegavirales, family Rhabdoviridae, and as such each of the viruses contains a single strand of non-segmented, negative sense RNA as its genome. This diverse group of over 150 viruses includes those that infect mammals, reptiles, birds, fish, insects and plants. Within this virus family there are six different genera including: the vesiculoviruses, ephemeroviruses and lyssaviruses that infect mammals; the cyto- and nucleorhabdoviruses that infect plants; and the novirhabdoviruses that infect fish. Only the lyssaviruses and the vesiculoviruses are recognised as viral agents able to infect both animals and humans and cause clinical disease. Of these two genera the lyssaviruses include one of the most notable viral infections known to man: rabies. First described in Mesopotamia in 2300 BC, rabies has been recognized in humans and animals for many centuries as an almost invariably fatal disease once clinical signs have been observed. Disease usually occurs with the onset of an acute encephalomyelitis, often preceded by periods of excitement or agitation, and quickly followed by coma and death. Hypersalivation, hydrophobia and aerophobia are also prominent features.
Virus structure and lifecycle
Virion morphology
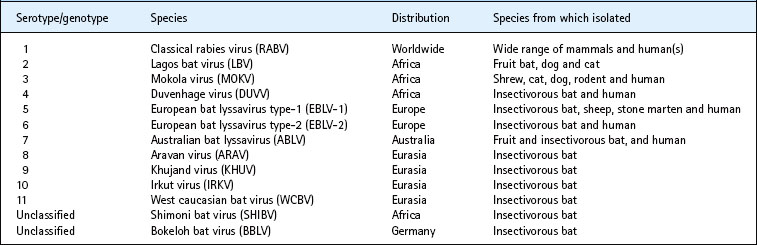
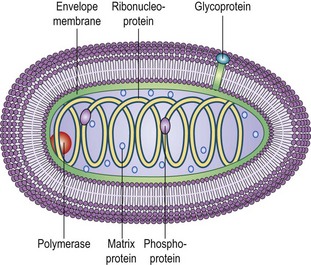
The lyssavirus genus includes rabies virus and ten other defined species differentiated according to their genomic sequence. Two further isolates, Shimoni bat virus (SHIBV) and Bokeloh bat virus (BBLV), are awaiting official classification (Table 58.1). First established by Louis Pasteur as a transmissible agent, the rabies virion is ‘bullet-shaped’ in appearance, with an average diameter of approximately 75 nm (60–110 nm), an average length of 180 nm (130–200 nm), and has helical symmetry (Fig. 58.1).
Virus genome structure
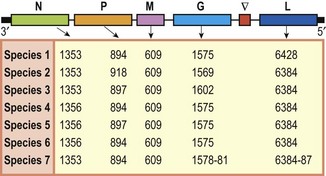
The rabies virus genome is composed of approximately 12 000 nucleotides, although variations in length exist between rabies virus strains (Fig. 58.2). The genomic RNA contains all the information necessary to produce the five different viral proteins: nucleoprotein (N), phosphoprotein (P), matrix protein (M), glycoprotein (G) and the large polymerase protein (L). The viral ribonucleoprotein (RNP) core consists of the viral RNA encapsidated by N proteins and associated with the P and L proteins. This RNP is the minimal replicative unit for these viruses. The other viral proteins, M and G are involved in virion structure and attachment to cellular receptors, respectively. The intergenic region (G-L) is approximately 450 nucleotides in length and does not appear to encode any polypeptides. Studies on the G-L region of a wide range of RABV isolates have shown that these viruses do not have the first termination signal identified in the Pasteur virus (PV) strain but instead have a second termination signal, conserved in all RABV genomes, and closer to the beginning of the L-gene.
Virus adsorption and cell entry
Infection is initiated as soon as the virus envelope glycoprotein (G) attaches to the host cell. This process of adsorption involves the interaction of the glycoprotein spikes, the major determinants for virus neuropathogenicity, with cell surface receptors. The mechanisms by which rabies virus targets cells for infection, the nature of the cell receptor(s) and the determinants of cell fusion remain unclear. Neuron-specific receptors for the virus including p75 neurotrophin receptor (p75NTR), the nicotinic acetylcholine receptor (nAChr) and the neuron adhesion molecule (NCAM), may promote efficient entry into neurons. However, it is unlikely that these are the only receptors utilised by the virus, as efficient viral replication may occur in a number of different cell lines including chick or duck embryo cells, baby hamster kidney cells, mouse neuroblastoma cells, human diploid lung fibroblasts, chick embryo fibroblasts, and Vero cells, although with minimal cytopathic effects. Therefore, cell receptors must exist on a number of different cell types that lyssaviruses can exploit and use for cell entry and replication.
Virus penetration and uncoating
After the virion has bound to its cellular receptor, viral entry (internalization) usually proceeds by fusion of the viral envelope with the cellular membrane. The virus may also enter the cell through coated pits and uncoated vesicles (viropexis or pinocytosis), which often incorporate several virions at once. Following internalization, the viral glycoprotein mediates fusion of the viral envelope with the endosomal membrane and releases the viral RNP in an endosomal vesicle. The endosome then fuses with a lysosome where the enzymes allow the ribonucleocapsid to discharge into the cytoplasm.
Transcription
The RNP complex contains all of the components required to activate viral transcription. Complete uncoating of the nucleocapsid is not necessary as the viral RNA polymerase is capable of copying the virus RNA while it is still in the RNP form. This provides the genomic RNA with protection from cellular ribonucleases. Each of the five viral proteins is encoded on individual monocistronic messenger RNAs (mRNA). These mRNAs are capped, methylated and polyadenylated by the enzymes packaged in the virion. Transcription proceeds following attachment of the polymerase complex to the genome promoter at the terminal 3′ end of the genome. The transcriptase complex then produces transcripts of each virus gene with a transcriptional gradient being produced through polymerase ‘fall-off’ at each gene boundary where intergenic gene start and stop signals exist. This results in a much greater expression of 3′ proximal genes. This transcriptional gradient is seen in many non-segmented negative strand viruses and is thought to be an evolutionary mechanism to ensure appropriate expression of each gene.
Translation
Following transcription, the individual viral mRNAs are processed using the host cell translation system. Free polyribosomes, present in the cytoplasm, translate the N, P, M and L mRNAs, and the G protein is constructed as a transmembrane molecule in the rough endoplasmic reticulum (RER), followed by transportation via the Golgi apparatus to the plasma membrane of the cell. In neuronal cells, visible changes are sometimes apparent and large accumulations of viral protein occur with the continuation of the transcription translation process. These dense areas of protein are referred to as ‘Negri bodies’.
Replication, assembly and budding
Once the mRNAs have been translated into proteins a change in composition of the viral polymerase complex occurs that leads to a switch from transcription to replication. This switch is thought to be associated with the accumulation of N protein in the cell but the exact mechanism remains unclear. Once the polymerase is in a replicative mode, the complex initially drives the production of a full-length anti-genome (positive-) sense strand of RNA, with the viral polymerase ignoring transcriptional signals present at each of the gene boundaries. The anti-genome strand serves as an intermediate for the formation of nascent genome (negative-) sense RNA.
The negative sense genomic RNA is then encapsulated by the M protein which forms a matrix surrounding the RNP core. Interactions between M and the RNP are thought to enable migration of the RNP to areas of the plasma membrane in which the glycoprotein is embedded. Condensation and coiling of the RNPs is then initiated by the matrix protein. Once associated with the glycoprotein, the condensed M-RNP forms a complete virion which buds from the plasma membrane and is released from the cell.
Epidemiology
Rabies virus
Rabies infection in man is generally acquired from the bite of an infected animal. The domestic dog (Canis familiaris) is the most important vector, although bat rabies continues to cause human infection across much of Central and South America. Extensive vaccination campaigns in dog and terrestrial wildlife populations have reduced the incidence of rabies across the globe. However the virus still remains endemic across much of the developing world, where the majority (99%) of human deaths due to rabies occur, mainly in Africa and Asia. The World Health Organisation (WHO) estimates an annual toll of 55 000 deaths following human infection with rabies virus, although this is likely to be a gross underestimate. Despite having a devastating impact on human life, rabies infection is preventable either through pre-immunisation, or in the event of exposure, post exposure prophylaxis (PEP) involving vaccination and the administration of rabies immunoglobulin. Importantly, a high proportion of infections occur in children under the age of 16, often in poor rural areas where vaccine and PEP are not readily available.
The UK is currently classified as ‘rabies free’, with 25 human cases of rabies reported between 1946 and 2010. The majority of these cases were acquired in countries other than the UK. However, the death of a bat worker in Scotland in 2002 due to infection with an indigenous bat rabies virus, European bat lyssavirus type-2 (EBLV-2), was a notable exception. Of the human cases reported in the UK, 19 had not received any PEP, two had started PEP, and three had no known history of pre- or post-exposure vaccination.
In endemic areas, urban rabies occurs among scavenger dogs and cats living in close proximity to human populations, and poses a more immediate threat. Sylvatic rabies, which refers to the spread of disease amongst wildlife, poses less of an immediate threat to the human population. The main focus of sylvatic spread is confined to different species depending on the region in which an outbreak occurs. Examples include: fox rabies in continental Europe, Canada and northern parts of the USA; rabid racoons along the US eastern seaboard; rabies in skunk populations in the US mid-western states; rabies in coyotes in the southern states of the USA; and rabies in the mongoose populations of the West Indies and in Africa. As there is limited contact between these wild species and humans, spread is mostly to other susceptible wildlife.
Control of the spread of rabies in dogs has been achieved using surveillance projects and the initiation of vaccination programmes. These approaches have been successful in the USA and many European countries, resulting in a concomitant reduction in the number of rabies cases in humans in these countries. The red fox (Vulpes vulpes) was the principal reservoir of sylvatic rabies virus in many of these countries but oral vaccination campaigns have eliminated the virus across Western Europe and the USA. However, reintroduction from areas of endemicity in Eastern Europe and the Americas has occurred; in France in 2008 after importation of a rabid dog, and in Italy in 2008 when rabies was isolated from red foxes. Both Italy and France had been classified as rabies free since 1997 and 1998, respectively, and remained so up until these recent re-introductions.
Despite elimination of terrestrial rabies, lyssaviruses present in bat populations continue to pose a further, albeit low risk, threat to human life. In 1916, in Brazil, a number of cattle and also humans were diagnosed with rabies following bites from infected haematophagus vampire bats. Further infection was then reported in many Central and South American countries, and also in the West Indies, with considerable mortality in cattle. Numerous reports have shown that many species of insectivorous and fruit-eating bats may harbour rabies virus, with excretion and transmission occurring at different rates. Exposure appears to occur readily in some bat colonies from the close contact among large numbers congregating together, evidenced by the presence of rabies virus neutralising antibodies in bat populations.
While rabies virus has a worldwide distribution, other species appear to be restricted to particular geographic regions and hosts. Lagos bat virus (LBV), first isolated from a fruit bat colony on the island of Lagos in Nigeria, is distributed throughout Africa and has been isolated from domestic cats and dogs, but not from humans. Mokola virus, distributed across Africa, has been isolated from shrews and humans, but not from bats. Duvenhage virus (DUVV), first isolated in South Africa, has been shown to cause disease in both bats and humans.
In Europe, four related lyssaviruses exist within bat populations. European bat lyssavirus type-1 (EBLV-1) is present in Europe as two distinct genetic lineages, EBLV-1a and EBLV-1b. EBLV-1a is thought to be the most recently introduced strain from North Africa via southern Spain, and it exhibits an east–west European division. The distribution of EBLV-1b follows a north-south division. EBLV-1a and EBLV-1b have only been reported together in France and the Netherlands. Most of the EBLV-1 cases in European bats have been identified in one bat species, Eptesicus serotinus, which habituates both the UK and mainland Europe, and should be regarded as the host species for EBLV-1. European bat lyssavirus type-2 (EBLV-2) was first isolated in 1985 from a Swiss bat biologist who had been working with bats in Finland, Switzerland and Malaysia. A year later this virus was isolated from bats in Denmark (Myotis daubentonii and M. dasycneme) and from M. daubentonii in Germany, the only known natural wild hosts of this virus (apart from a single case in N. noctula). In total, there are only 19 records of this virus (17 in bats, two in humans) from Denmark, Finland, Germany, the Netherlands, Ukraine, Switzerland and the UK. Both EBLV-1 and EBLV-2 have been shown to cause disease in mammals including companion animals, wildlife and domestic livestock. EBLV-1 has been shown to cause disease in sheep, foxes, a stone marten, ferrets, cats and dogs. In contrast, EBLV-2 has not been detected naturally in mammals other than bats and humans.
West caucasian bat virus (WCBV) was isolated in 2002 from Miniopterus schreibersii bats in southeastern Europe. This is the only isolation of this virus. Antigenically this isolate represents the most divergent member of the lyssaviruses, and shows a lack of serological cross reactivity to the other lyssavirus species. Bokeloh bat virus (BBLV) a new ‘putative’ bat lyssavirus isolated in Bokeloh, Germany, in 2010 from a Myotis nattereri bat, has yet to be classified.
Virus transmission
The inability of rabies virus to penetrate intact skin means that it has to gain entry to the body through broken skin (often as a result of a biting incident) or through the mucous membranes (eyes, nose and mouth). In many developing countries, where canine immunisation programs are minimal or non-existent, the predominant source of human infection is from dogs (99% of cases worldwide). In regions of the world where canine rabies has been controlled by vaccination, wild animal bites represent the main source of human infection. Bat bites are also a source of concern throughout the world, as these bites are often small and may go unnoticed.
There have also been reports of rabies infection due to aerosolised virus, either following potential exposure to virus in caves containing large numbers of potentially infected bats, or following accidental laboratory exposure. Experimental aerosol transmission has been reported using a murine model. Transfer of infection via infected corneal transplants has been reported on at least eight occasions in five different countries. A number of organ recipients have acquired rabies having received organs from infected donors. In 2004 in the USA, transplantation of kidneys, liver and an arterial segment from a donor who died from unexplained encephalitis, resulted in the deaths of the transplant recipients from rabies virus. A history of bat bite was subsequently identified in the donor. A similar situation occurred in Germany in 2005. Even though virus has been shown to be excreted in both saliva and conjunctival exudates, direct transmission of virus from person-to-person spread is extremely rare, although possible transmission from mother-to-child has been reported.
Laboratory diagnosis
It can often be difficult to diagnose a case of unidentified encephalitis as rabies infection, especially if there is no recorded history of animal bite or travel to a rabies endemic area. A number of diagnostic techniques can be used to test for the presence of virus neutralising antibodies, virus genome or live virus. Tests on CSF, saliva and on skin biopsy from the nape of the neck (nuchal skin biopsy) are the only reliable assays that can be used ante-mortem. Post-mortem diagnosis of rabies virus infection in brain samples is reliable and is used as a confirmatory test.
Diagnostic methods
Rabies virus antigen detection techniques
The ‘gold standard’ Office International des Epizooties (OIE) and WHO approved diagnostic test is the fluorescent antibody test (FAT), which detects virus antigen in brain samples (hippocampus, brainstem or cerebellum) using fluorescently labelled anti-rabies antibodies. This is reliable and can provide results within 2 h. Sensitivity, however, is dependent upon the quality of the sample received.
Rabies virus nucleocapsid can also be detected using the enzyme-linked immunosorbent assay (ELISA), which has the added advantage of being able to detect virus in autolysed samples. A number of variations of this technique are available.
Rabies virus isolation
The mouse inoculation test (MIT) involving inoculation of sample into mouse brain and observation of the animal for a period of 28 days or until clinical signs developed, was the gold standard for rabies virus isolation. This has now been superseded and replaced by tissue culture methods.
The rabies tissue culture isolation test (RTCIT) is used to isolate lyssaviruses in a range of clinical specimens from a number of host species, including dogs, cats, bats, wild carnivores and humans. Samples are inoculated onto a neuroblastoma monolayer. Following a 4-day incubation period, the plates are fixed and stained using fluorescein-labelled monoclonal-antibodies (mAbs) and the presence or absence of virus is determined.
Histopathological examination
Histopathological analysis of tissue samples is often used post-mortem as a confirmatory test. The sample is fixed, blocked and stained with eosin and haemotoxylin before application of a rabies specific antibody, which detects the presence of rabies virus nucleoprotein.
In situ hybridisation can also be used to detect both messenger and genomic viral RNA. This assay uses a digoxigenin labelled riboprobe to detect lyssavirus RNA in brain samples.
Serological assays
The presence of rabies virus neutralising antibodies can be detected using the fluorescent antibody virus neutralisation assay (FAVN), a rapid fluorescent focus inhibition test (RFFIT) or an ELISA-based detection system. The FAVN is the current method of choice when assessing both human and animal antibody titres following pre-exposure vaccination. The basic premise for both the RFFIT and FAVN is that a known quantity of virus is mixed with different dilutions of the test serum and the neutralising limit dilution is determined. The non-neutralised virus is then detected using a fluorescence-conjugated virus specific antibody. The results are then calibrated from standard reference sera and expressed as international units (IU). Due to the use of live virus in these assays each test must be undertaken in high containment facilities.
More recently, a virus neutralisation assay using a rabies virus-pseudotyped lentivirus has been developed. This approach uses lentiviral expression of the viral glycoprotein as a surrogate for live lyssavirus. The pseudotype assay has a number of advantages over serological assays based on the use of live virus. Although it requires tissue culture facilities, it does not need to be performed in a high containment facility and is therefore more feasible in rabies endemic areas that may lack such facilities; also the assay requires lower volumes of serum, which is of importance where only small volumes of serum are available.
Molecular methods for the detection of rabies virus RNA
The use of molecular based assays to detect the presence of virus nucleic acid is becoming more commonplace as these techniques become more widely available and accessible. These techniques are not currently accepted as prescribed diagnostic techniques by the OIE or WHO, but are useful as screening tools and for the generation of sequence data for phylogenetic analysis of samples.
The reverse transcriptase polymerase chain reaction (RT-PCR) is one of the most widely used tools in rabies virus detection and research. The hemi-nested RT-PCR and various other similar techniques are utilised routinely in laboratories worldwide for rabies virus detection. Other assays that have recently been developed include the Taqman PCR, a real time assay that can distinguish between lyssavirus species, and also a PCR ELISA.
The detection of rabies virus ante-mortem is more difficult, as the virus is only excreted intermittently in saliva samples and antibodies are not always detectable in CSF. The use of nucleic acid sequence based amplification (NASBA) is more sensitive that RT-PCR when assessing ante-mortem samples. This technique amplifies viral RNA directly in an isothermal reaction, and therefore does not require expensive thermocycling equipment.
Other techniques are currently being investigated. Microarray utilises oligonucleotide probes designed to bind to the different lyssavirus species. While this technique currently has limited sensitivity when using clinical samples, in the future it could prove a useful tool in the diagnosis of viral diseases of the CNS, for which rabies virus detection will form part of the differential diagnosis
Treatment
Vaccination
It was the French scientist Louis Pasteur who first introduced vaccination following rabies virus exposure. The original vaccine was a crude extract of infected rabbit spinal cord, which had been desiccated and passaged several times to produce a ‘fixed’ strain of the virus. The success of this vaccination regime in cats and dogs was announced at the Academies des Sciences in 1885. The successful treatment of a young boy who experienced multiple bites from a rabid animal subsequently established this approach. The Semple vaccine, a modification of Pasteur’s original vaccine, was a suspension of 4% inoculated rabbit brain attenuated with 5% phenol. This was used in the UK from 1919–1966, but due to the high rate of allergic encephalitis following its administration, was replaced by a duck embryo-derived vaccine in 1966. This was not produced in neuronal cell-lines and there was a decrease in the incidence of adverse reactions following its introduction. This type of vaccine has since been replaced by cell culture-derived inactivated vaccines, several types of which are currently available, including: human diploid cell vaccine (HDCV), purified chick embryo cell vaccine (PCEV) and Vero cell vaccine. While each of these current vaccines has proven to be both safe and immunogenic, they are not necessarily available in developing countries, and they do not protect against all lyssavirus species.
Pre-exposure prophylaxis
In the UK, the HDCV and PCEV vaccines are licensed and available. These are normally administered to those who require pre-exposure vaccination, e.g. laboratory staff working directly with the virus. For those people travelling to rabies-endemic countries, pre-exposure vaccination may be recommended depending on the local incidence of rabies in the country to be visited, the availability of appropriate anti-rabies biologicals, and the intended activity and duration of stay of the traveller. The protocol requires administration of three intramuscular doses of vaccine at days 0, 7 and 28, following which the levels of neutralising antibody are assessed using a certified test. Response to these vaccines can persist for at least two years post-vaccination. These vaccines are safe and well tolerated, with adverse reactions being extremely rare.
There are a number of different animal vaccines available, mostly live-attenuated preparations that would not be acceptable for use in humans. The animal is generally inoculated intramuscularly, with boosters required every 1–3 years depending on the vaccine type.
Post-exposure prophylaxis
Wound cleansing and immunizations, undertaken as soon as possible after suspected contact with an animal and following WHO recommendations, can prevent the onset of rabies in virtually 100% of exposures, although some exceptions to this have been recorded. The wound should be washed immediately with copious amounts of water and preferably cleansed with soap. Alcohol, quaternary ammonium compounds, iodine or povidone should be used, if available. The level of treatment or care required depends on the category of the contact as outlined by the WHO (category 1, 2 or 3, see Table 58.2). Post-exposure anti-rabies vaccination should always include administration of both passive antibody (human rabies immunoglobulin, HRIG) and vaccine in patients without any history of rabies pre-vaccination, for both bite and non-bite exposures, regardless of the interval between exposure and initiation of treatment. HRIG provides immediate availability of neutralising antibodies at the site of exposure before the patient elicits an endogenous antibody response following vaccination.
Table 58.2 WHO categories of rabies exposure
| Category | Exposure | Treatment |
|---|---|---|
| 1 | Touching, feeding of animals or licks on unbroken skin | No treatment if history is reliable |
| 2 | Minor scratches or abrasions without bleeding or licks on broken skin and nibbling of uncovered skin | Treat with vaccine |
| 3 | Single or multiple transdermal bites, scratches or contamination of mucous membrane with saliva (i.e. licks) | Treat with immunoglobulin and vaccine |
Depending on vaccine type, the post-exposure schedule requires four to five intramuscular doses over four weeks. According to the WHO, two intramuscular doses of a cell-derived vaccine separated by three days are sufficient for rabies-exposed patients who have previously undergone complete pre-vaccination or PEP with cell-derived rabies vaccines. Rabies immune globulin treatment is not necessary in such cases.
There have been seven reports of recovery from rabies once clinical signs have developed, five of whom had received pre-exposure vaccination, and two of whom had no pre-exposure vaccination, but were treated according to variations of the Milwaukee protocol. The first was a 15-year-old girl who developed clinical rabies one month after she had been bitten by a bat. A ‘therapeutic’ coma was induced allowing an immune response to be elicited. A cocktail of drugs including ketamine, midazolam, ribavirin and amantadine was then used as part of the treatment regimen. Lumbar puncture after 8 days showed a raised level of anti-rabies antibodies and the girl was brought slowly out of the coma. Signs of paralysis and sensory denervation were observed. After nearly 5 months following initial hospitalisation she was alert and communicative but with involuntary movements, difficulty speaking and an unsteady gait. The second was reportedly in Brazil in 2008; a young boy was treated following a similar protocol and survived. Similar regimens have been attempted for at least 12 other rabies patients without success.
Control
Control of the spread of rabies would require elimination of infection from all reservoir species throughout the world, which is unlikely due to its broad host range, and its urban as well as sylvatic spread. Methods such as shooting and gassing animals have short-term effects. Therefore the principal aim of current strategies is to eliminate rabies in domestic dogs worldwide, thus breaking the transmission cycle and reducing the risk of human infection acquired from dogs.
A number of countries, mainly islands, have been classified as terrestrial rabies free including Australia, New Zealand, Hawaii, Ireland and Great Britain since the early 1900s, and Switzerland, Belgium, Finland, Luxemburg and the Netherlands since 2001. This has been achieved by a combination of strict quarantine laws and vaccination programmes. Although achieving a rabies-free status is more difficult in countries that share borders with rabies endemic regions, the control of sylvatic (wildlife) rabies throughout Eastern Europe and North America is underway. This has largely been possible through the vaccination of domestic animals (dogs and cats) likely to come into contact with these species, as well as oral bait vaccination schemes for wild animals. As most cases of rabies in humans are due to contact with a rabid dog or cat, these vaccination programmes, together with PEP for those individuals exposed in specific incidents to suspected rabid animals, have notably reduced the number of human fatalities due to rabies virus infection in the developed world. This approach, together with vaccination of specific individuals who may come into contact with rabies virus, or rabid animals during the course of their work (e.g. laboratory workers, those individuals handling animals at quarantine centres, veterinarians, animal health inspectors, bat handlers and travellers to areas where rabies is still endemic) has reduced the number of incidents in developed countries who have adopted this scheme.
Baer GM. Rabies – an historical perspective. Infectious Agents and Disease. 1994;3:168–180.
Department of Health. Memorandum on Rabies: Prevention and Control. London: Department of Health; 2000.
Jackson AC, Wunner WH. Rabies, ed 2. London: Elsevier: Academic Press; 2007.
Rupprecht CE, Barrett J, Briggs D, et al. Can rabies be eradicated? Developmental Biology (Basel). 2008;131:95–121.
Rupprecht CE, Dietzschold B, Koprowski H. Lyssaviruses. Geneva: Springer-Verlag, 1994.
Schnell MJ, McGettigan JP, Wirblich C, Papaneri A. The cell biology of rabies virus: using stealth to reach the brain. Nature Reviews Microbiology. 2010;8:51–61.
Tordo N, Marianneau MP. Viruses and bats: rabies and Lyssavirus. Bulletin et Memoires de l Academie Royale de Medecine de Belgique (Brussels). 2009;164:7–15.
Wunner WH, Briggs DJ. Rabies in the 21st Century. PLoS Neglected Tropical Diseases. 2010;4:e591.
Department of Health. Memorandum on Rabies: Prevention and Control. http://www.dh.gov.uk/prod_consum_dh/groups/dh_digitalassets/@dh/@en/documents/digitalasset/dh_4080657.pdf.
Department for Environment, Food and Rural Affairs. Disease factsheet: Rabies. http://archive.defra.gov.uk/foodfarm/farmanimal/diseases/atoz/rabies/.
Eurosurveillance. http://www.eurosurveillance.org/.
Global alliance for rabies control (GARC). http://www.rabiescontrol.net/.
OIE – World Organisation for Animal Health. http://www.oie.int/.
World Health Organization. Human and animal rabies. http://www.who.int/rabies/.
World Health Organization. Rabies Bulletin Europe. http://www.who-rabies-bulletin.org/.
World Health Organization. Rabnet. http://www.who.int/rabies/rabnet/.
World Health Organization. Immunization, vaccines and biologicals. http://www.who.int/immunization/topics/rabies/en/index.html.