63 Helminths
Intestinal worm infections; filariasis; schistosomiasis; hydatid disease
Key points
• Helminths are multicellular parasitic worms that often have complex life cycles.
• Intestinal nematodes (roundworms) are extremely common, especially in conditions of poor hygiene.
• Heavy infections with hookworm can give rise to severe anaemia; benzimidazoles offer effective therapy.
• Serious infections caused by tissue nematodes include bancroftian filariasis (elephantiasis) and onchocerciasis (river blindness); they are treated with ivermectin or diethylcarbamazine.
• Trematodes (flukes) cause schistosomiasis (bilharzia) and other diseases such as Chinese liver fluke infection.
• Cestodes (tapeworms) are often several metres long, but usually cause little pathology.
• Most trematode and cestode infections respond to praziquantel.
• Hydatid disease is caused by a dog tapeworm. Treatment is by surgery.
Medical helminthology is concerned with the study of parasitic worms. These creatures are responsible for an enormous burden of infection throughout the world and, although few helminthic infections are life-threatening, their impact on human health is incalculable. Most helminths have no independent existence outside the host and are therefore truly parasitic. As they rely on the host for sustenance, it is not in their interest to cause the host harm; consequently they do not usually exhibit great virulence and are characterized more by the novel methods that they have evolved to prevent rejection by the host defences. The pathogenic manifestations of helminthic disease, which can none the less be considerable, are ordinarily due to physical factors related to the location of the worms, their lifestyle or their size (see Ch. 11).
There are two major groups of helminth: nematodes, or roundworms, and platyhelminths, or flatworms. Flatworms are, in their turn, represented by two classes: trematodes (flukes) and cestodes (tapeworms).
Nematodes
The principal nematode parasites of man are conveniently considered under two headings: intestinal nematodes and tissue nematodes.
Intestinal nematodes
Infection with intestinal roundworms (Table 63.1) is generally associated with conditions of poor hygiene. Such infections are extremely common, particularly throughout the tropics and subtropics, although several are also found in temperate regions. Low worm burdens are generally asymptomatic, but heavy infections may cause problems, especially in young children in whom they have been associated with impaired development.
Table 63.1 Principal intestinal nematodes of man
| Species | Common name | Relevant examination |
|---|---|---|
| Ancylostoma duodenale | Hookworm | Stool concentration (ova) |
| Ascaris lumbricoides | Common roundworm | Stool concentration (ova) |
| Enterobius vermicularis | Threadworm | Peri-anal swab (ova), adult worms on stool |
| Necator americanus | Hookworm | Stool concentration (ova) |
| Strongyloides stercoralis | – | Stool concentration (larvae) or culture |
| Toxocara canisa | Dog roundworm | Serology |
| Trichostrongylus spp. | Hookworm | Stool concentration (ova) |
| Trichuris trichiura | Whipworm | Stool concentration (ova) |
a Not an intestinal parasite of man; causes visceral larva migrans (see text).
Ascaris lumbricoides
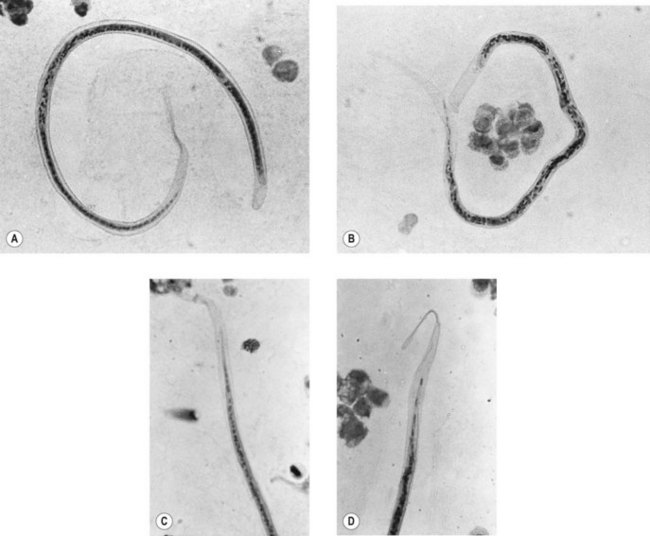
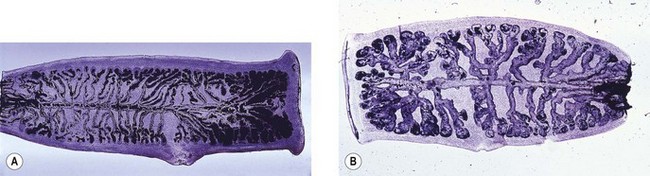
This is the common roundworm, which infects more than a billion people in the world. The adults are large and fleshy and, as with many nematodes (other than the hookworm group), the smaller male can be recognized by his characteristically crooked tail. The eggs (ova) are produced in huge numbers; they are thick-walled, bile-stained and typically exhibit a corrugated albuminous coat (Fig. 63.1A). In the absence of a male worm, the female produces infertile eggs, which are more elongated and irregular than the fertile variety (Fig. 63.1B).
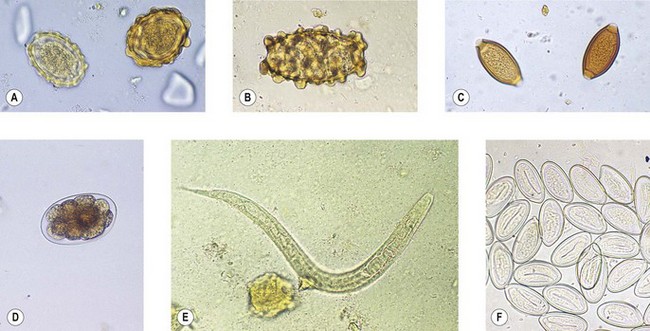
Fig. 63.1 Eggs of intestinal helminths: (A) Ascaris lumbricoides (fertile eggs); (B) A. lumbricoides (infertile egg); (C) Trichuris trichiura; (D) hookworm; (E) Strongyloides stercoralis (larva); (F) Enterobius vermicularis.
In warm, moist conditions, infective larvae develop within fertile eggs, but do not hatch. Such eggs can survive for long periods in soil. If ingested, the eggs hatch in the duodenum and the larvae penetrate the gut mucosa to reach the bloodstream. They are carried to the pulmonary circulation, where they gain access to the lung and undergo two moults before migrating via the trachea to the intestinal tract. Having completed their round trip, they mature in the gut lumen and live for several years.
Ascaris lumbricoides is a well-adapted parasite that is usually not pathogenic in the ordinary sense. However, pneumonic symptoms may accompany the migratory phase, and the adult worms may invade the biliary and pancreatic ducts. Moreover, heavy infection with these large worms can cause intestinal obstruction. Allergy is also sometimes a problem.
The dog ascarid, Toxocara canis, may accidentally infect man. Larvae hatch in the small intestine and penetrate the gut wall, but they are unable to complete their migratory phase. Instead, they find their way to remote parts of the body, a condition known as visceral larva migrans. Occasionally the larvae reach the eye and cause serious retinal lesions. Larvae of several other roundworms, including Angiostrongylus, Gnathostoma and Anisakis species, are occasionally implicated in visceral larva migrans in some parts of the world.
Trichuris trichiura
This is the common whipworm, often found together with ascaris. The adults live with the head (the ‘whip’ end of the worm) embedded in the colonic mucosa. Each female lays thousands of characteristic ‘tea-tray’ eggs (Fig. 63.1C) every day. Like those of ascaris, they develop infective larvae in warm, moist conditions, but the ova do not hatch outside the body. However, after ingestion and hatching, there is no migratory phase and adult worms develop directly in the large intestine.
Infection is usually trivial, although massive infections can cause rectal prolapse in young children, and a form of dysentery is described.
Hookworm
The two human hookworms, Ancylostoma duodenale and Necator americanus, are widely distributed throughout the tropics and subtropics. The two species produce indistinguishable thin-walled eggs (Fig. 63.1D) that hatch in soil. Infection is usually acquired by walking barefoot in soil contaminated with human faeces. The larvae undergo several moults before infective larvae are produced. These are capable of penetrating unbroken skin, and in this way they gain access to the bloodstream to begin a migratory phase similar to that of ascaris. When they reach the gut they attach by their mouthparts to the mucosa of the small intestine. The adult worms, which are about 1-cm long, are similar, but the buccal capsule of A. duodenale bears two pairs of teeth, whereas N. americanus has two so-called ‘cutting plates’. Unlike most nematodes, the tail of male hookworms has a membranous bursa used for attachment to the female during copulation.
Hookworms ingest blood and move from site to site in the gut mucosa, leaving behind small bleeding lesions. These two facts are responsible for the chief pathological manifestation of heavy infection with hookworms: iron deficiency anaemia.
Larvae of animal hookworms, notably the dog hookworm, A. caninum, may penetrate human skin, but do not migrate further. They do, however, cause local irritation by wandering through subcutaneous tissue, a condition called cutaneous larva migrans.
Trichostrongylus species
Various species of Trichostrongylus have been associated with human disease, particularly in the Middle East. Like the hookworms, they are bursate nematodes with similar, but more elongated, eggs.
Strongyloides stercoralis
This parasite is also related to the hookworms but differs in several important respects. There is a distinct free-living phase in the life cycle, during which males and females reproduce. Human infections arise after penetration of infective larvae through skin and there is a migratory phase involving the lungs. However, human infection appears to be restricted to female worms, which attach to the gut mucosa and produce eggs that contain fully developed larvae; these hatch within the intestinal lumen so that larvae, not eggs, are found in faecal samples (Fig. 63.1E). Infection can persist for many years, probably because some larvae can develop sufficiently within the body to initiate a fresh cycle of development and cause auto-infection.
Symptoms are usually benign, but in debilitated individuals larvae may be activated to penetrate the gut wall and invade other organs, a serious condition known as hyperinfection.
Enterobius vermicularis
This is the common threadworm, which infects children throughout the world. It has the simplest life cycle of all intestinal worms. Adults live in the large intestine and are occasionally found in the appendix. Mature, gravid females crawl through the anus at night and lay their eggs in the peri-anal area. The eggs are characteristically flattened on one side (Fig. 63.1F) and usually contain fully developed larvae. Ingestion of these eggs initiates a fresh infection. Symptoms are restricted to itching (pruritus ani) associated with the deposition of eggs.
As eggs are not discharged by the worm into faeces, faecal examination is not appropriate in the laboratory diagnosis of threadworm infection. The diagnosis is established by finding the characteristic ‘threads’ on the surface of formed stools, or by examination of swabs (or Sellotape impressions) of unwashed peri-anal skin for the characteristic ova.
Treatment of intestinal nematode infections
In endemic areas, the need to treat intestinal worm infections has to be balanced against the severity of symptoms (if any), the inevitability of re-infection, and the use of scarce medical resources. In the industrially developed world, where the infections are less common and mostly imported, a more liberal approach to treatment can be adopted.
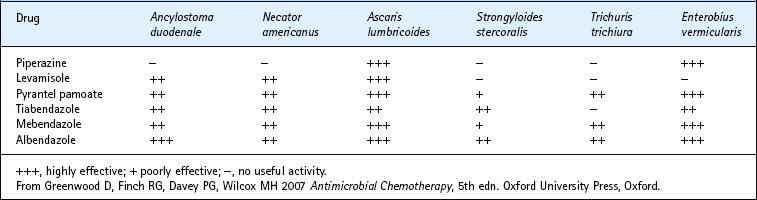
The range of options is listed in Table 63.2. Most effective (and expensive) are the benzimidazole derivatives, especially albendazole and mebendazole.
Tissue nematodes
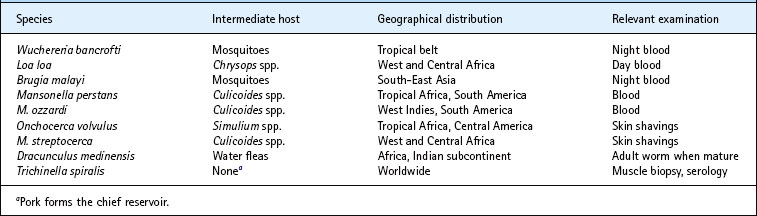
This group includes the filarial worms, the Guinea-worm (Dracunculus medinensis) and Trichinella spiralis (Table 63.3).
Filarial worms
Filarial worms have a complex life cycle involving developmental stages in an insect vector. They vary considerably in their pathogenic effects, but some are responsible for disabling diseases that have a major impact on communities living in endemic areas.
Wuchereria bancrofti
This worm is transmitted by the bite of various species of mosquito throughout the tropical belt of the world. Over 100 million people are thought to be infected. The larvae invade the lymphatics, usually of the lower limbs, where they develop into adult worms. The presence of adult worms causes lymphatic blockage and gross lymphoedema, which sometimes leads to the bizarre deformities associated with bancroftian filariasis, elephantiasis.
Embryonic forms (microfilariae) are liberated into the bloodstream. They retain the elastic egg membrane as a sheath, which covers the whole larva (Fig. 63.2A). Microfilariae remain in the pulmonary circulation during the day, emerging into the peripheral circulation only at night, to coincide with the biting habits of the insect vector. The physiological basis of this nocturnal periodicity is not understood, but it can be reversed by altered sleep patterns in, for example, night-shift workers. Moreover, strains of Wuchereria bancrofti encountered in some Pacific islands do not exhibit a nocturnal periodicity. Aside from these exceptions, blood for examination for W. bancrofti must be taken during the night, optimally between midnight and 2 a.m.
Loa loa
This worm is restricted in distribution to central and western parts of tropical Africa, where it is transmitted by biting flies (Chrysops species; see p. 672). The adult worms live in subcutaneous tissue and wander round the body, provoking localized reactions known as Calabar swellings and sometimes migrating across the front of the eye.
The sheathed microfilariae of Loa loa (Fig. 63.2B) exhibit diurnal periodicity, so that, unlike those of W. bancrofti, they appear in peripheral blood only during the day.
Brugia malayi
This parasite is probably related to W. bancrofti. It is transmitted by mosquitoes in parts of India, the Far East and South-East Asia. Adult worms inhabit the lymphatics and, like W. bancrofti, can cause elephantiasis. Microfilaraemia usually shows a nocturnal periodicity.
Onchocerca volvulus
This filarial worm is common in parts of tropical Africa and Central America. It is transmitted by Simulium damnosum and related species of black-fly (see p. 672). Adult worms develop in subcutaneous and connective tissue, and often become encapsulated in nodules, which form on bony parts of the body such as the hip, elbow and (particularly in Central America) the head. The microfilariae are not found in blood, but live in the superficial layers of the skin causing itching and, in heavy chronic infections, gross thickening of the skin. The eye is commonly invaded by microfilariae, which may cause corneal and retinal lesions that lead to blindness. Because the vector breeds by rivers, the condition is known as river blindness.
If nodules are present, diagnosis can be made by finding macroscopic worms within an excised nodule. Otherwise, superficial slivers of skin, taken from calves, buttocks and shoulders, are suspended in a drop of saline and examined microscopically for motile microfilariae.
Mansonella species
Mansonella perstans is widespread throughout tropical Africa and parts of South America; the related M. ozzardi is restricted to parts of the West Indies and South America. They are transmitted by biting midges (Culicoides species; see p. 671). The unsheathed microfilariae appear in the bloodstream and exhibit no periodicity. They are generally regarded as non-pathogenic.
M. streptocerca causes skin infections similar to those of O. volvulus, although the symptoms are usually milder. It is restricted to parts of western and central Africa.
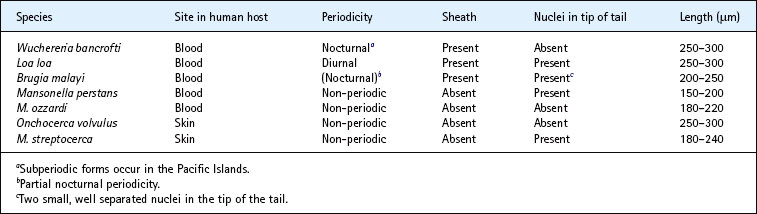
Differential characteristics of microfilariae
The microfilariae of filarial worms can be differentiated in stained preparations of clinical material by various criteria, the most useful of which are the presence or absence of a sheath and the disposition of the somatic nuclei in the tip of the tail (Table 63.4 & Fig. 63.2). Giemsa stain is suitable for the demonstration of somatic nuclei, but hot (60°C) haematoxylin is necessary to stain the sheath.
Treatment of filariasis
Diethylcarbamazine (DEC) has been used for many years for the treatment of all forms of filariasis. It effectively kills microfilariae, but is not reliably lethal to adult worms. It is relatively non-toxic, but death of the microfilariae is often accompanied by a severe allergic reaction (Mazzotti reaction), especially in onchocerciasis. Suramin kills the adult worms, but is much more toxic than DEC.
The treatment of onchocerciasis has been revolutionized by use of the veterinary anthelminthic ivermectin. This drug is effective in a single oral dose and is less likely than DEC to elicit a severe reaction. Periodic administration of ivermectin, together with vector control measures, has had an important impact on onchocerciasis in endemic areas.
Ivermectin and albendazole are effective in other forms of filariasis, although neither drug kills adult worms. As they exhibit activity against intestinal nematodes, including A. lumbricoides, these may be incidentally expelled during treatment. Albendazole is being used, alone or in combination with ivermectin, in campaigns aimed at eradicating lymphatic filariasis.
Surprisingly, tetracyclines also have an effect in filariasis, apparently by inhibiting endosymbiotic bacteria (Wohlbachia species) that are essential for the fertility of the worms.
Dracunculus medinensis
This is the Guinea-worm. The infective larvae develop within water fleas of the genus Cyclops, and human infection is normally acquired through infected drinking water. The larvae penetrate the gut mucosa and grow to maturity in connective tissue, usually of the lower limbs. The male is small and insignificant, but the female may reach a length of 1 m. After fertilization, the female worm incubates the larvae to maturity and, when ready to give birth, emerges to the skin surface to provoke an intensely irritating blister. When the sufferer immerses the blister in water, the uterus of the female worm bursts, liberating up to 1 million larvae, which are ingested by water fleas to continue the cycle.
Attempts can be made to wind out the dead worm over several days, but breakage of the worm often occurs, and pyogenic cocci may be carried into the tissues to cause a cellulitis. Chemotherapy is not usually helpful, other than to treat secondary bacterial infection. Prevention is the best approach; health education campaigns and the provision of safe water have much reduced the prevalence of this disease, and complete eradication is now possible.
Trichinella spiralis
Unlike most parasitic worms, Trichinella spiralis has an extremely wide host range. Human infections are usually acquired by eating undercooked pork products, although other meat, including bear and walrus meat, has been incriminated. The infected larvae lie dormant in skeletal muscle (Fig. 63.3) and are released when the meat is digested. Male and female worms develop to maturity attached to the mucosa of the small intestine. The female is viviparous, producing numerous larvae during a lifespan of only a few weeks. The larvae penetrate the gut wall and migrate to skeletal muscle, where they enter the quiescent phase. Most of the symptoms of trichinosis, which can be severe – even life threatening – are associated with the migration of larvae.
Mebendazole is said to be effective against the adult worms and the larvae, but treatment is unsatisfactory. Symptoms usually develop only during the invasive stage, and measures to control the sequelae of invasion are more important than anthelminthic therapy.
Trematodes
The flukes (Table 63.5) are a diverse group of worms that share a similar life cycle involving a snail host and, often, a second intermediate host that provides the vehicle for the transmission of infection. Most flukes have a restricted geographical distribution that reflects the habitat of the appropriate type of snail.
Most trematodes are hermaphrodite, but the most important human flukes, the schistosomes, are differentiated into separate sexes.
Life cycle
When excreted, trematode eggs often contain a fully developed ciliated organism called a miracidium, although in some species immature eggs are produced that require a period of development before the miracidium is formed. In water, the miracidium escapes, either through a lid-like operculum in the egg shell or (in the case of the schistosomes) by osmotic rupture of the egg. The miracidium penetrates the appropriate species of snail and undergoes several stages of asexual reproduction before emerging as a free-swimming body called a cercaria. The cercariae encyst in the muscle of fish (Clonorchis sinensis), crabs and crayfish (Paragonimus westermani), water chestnuts (Fasciolopsis buski) or vegetation (Fasciola hepatica), and man becomes infected by ingesting the encysted metacercariae. In the case of Schistosoma species, the cercariae remain in water and penetrate unbroken skin to gain access to the body.
Clonorchis sinensis (syn. Opisthorchis sinensis)
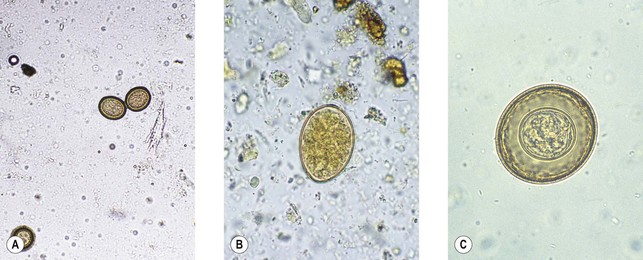
This is the Chinese liver fluke. Infection is acquired from uncooked freshwater fish, notably carp. The metacercariae excyst in the small intestine and pass into the bile ducts, where they mature. Typical small, flask-shaped eggs with a prominent operculum (Fig. 63.4A) are excreted in large numbers into the faeces.
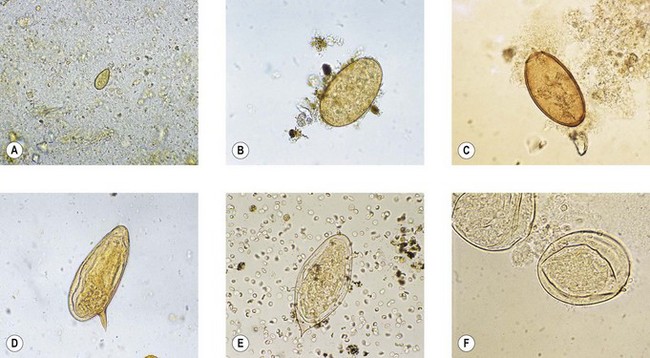
Fig. 63.4 Eggs of trematodes: (A) Clonorchis sinensis; (B) Fasciola hepatica; (C) Paragonimus westermani; (D) Schistosoma mansoni; (E) S. haematobium; (F) S. japonicum.
Infection is commonly asymptomatic, but fibrosis of the bile ducts with impairment of liver function may occur in heavy, chronic infections. As with most fluke infections, praziquantel is the drug of choice for treatment.
A closely related fluke, Opisthorchis felineus, which is a parasite of the cat, has been associated with human disease in parts of eastern Europe.
Fasciola hepatica
This is the cosmopolitan liver fluke of sheep. Human infections have usually been associated with eating wild watercress from infected sheep pastures. The adult worm is larger than C. sinensis, and lighter infections can cause biliary fibrosis and obstructive jaundice. The large, immature eggs with an indistinct operculum (Fig. 63.4B) may be found in faeces, but are usually sparse.
Unlike other trematode infections, fascioliasis does not reliably respond to praziquantel, and treatment with the veterinary anthelminthic triclabendazole or the more toxic chlorophenol derivative bithionol may be required.
Paragonimus westermani
This is the lung fluke, which is found in parts of the Far East. Closely related species have occasionally been implicated in human disease in parts of Africa and South America. Human infection follows ingestion of raw, infected muscle of freshwater crabs and crayfish. The metacercariae penetrate through the gut wall and diaphragm to reach the lung, where they develop to maturity. Occasionally the larvae find their way to the brain. Pulmonary infection usually provokes the production of sputum, in which the characteristic large eggs (Fig. 63.4C) can be found, often associated with flecks of altered blood. Praziquantel is used for treatment.
Intestinal flukes
Several genera of intestinal flukes cause human infection, particularly in the Far East. Fasciolopsis buski is found in restricted foci in China and South-East Asia. Infection is often acquired by the habit of opening water chestnuts with the teeth. The adult flukes live attached to the wall of the small intestine and produce a large number of eggs that resemble those of F. hepatica.
Other intestinal flukes include Gastrodiscoides hominis, Heterophyes heterophyes, Metagonimus yokogawai and various species of Echinostoma. Infection is usually asymptomatic unless the worm burden is large. Such evidence as exists suggests that praziquantel is effective in these cases.
Schistosoma species
The schistosomes, or blood flukes, also known as bilharzia after the discoverer, Theodor Bilharz, are the most important of the pathogenic trematodes. At least 200 million people are infected, principally in Africa, where Schistosoma mansoni and S. haematobium are widespread, and S. intercalatum is encountered in some areas. S. mansoni is also found in parts of the West Indies and South America; S. japonicum and the related S. mekongi are restricted to the Far East.
Human infection follows exposure to cercariae in water harbouring infected snails. The cercariae penetrate the skin, often causing a transient dermatitis, called swimmer’s itch. Once in the bloodstream, the schistosomula migrate to the liver, where they develop into mature male and female worms. The integument of the mature male worm is adapted in the form of two long flaps, the gynaecophoral canal, in which the female is held. The mature worms migrate to the small veins of the rectum (S. mansoni, S. intercalatum, S. japonicum and S. mekongi) or the bladder (S. haematobium). Eggs, which contain a fully developed miracidium, are passed through the rectal mucosa on to the surface of colonic faeces or through the bladder wall into the urine.
The ova of S. mansoni (Fig. 63.4D) are large (about 140 µm long) and possess a characteristic lateral spine, whereas those of S. haematobium (Fig. 63.4E) and S. intercalatum have a terminal spine. The smaller, more rounded, eggs of S. japonicum and S. mekongi do not have a prominent spine, but may exhibit a rudimentary nipple-like appendage (Fig. 63.4F).
Pathogenesis
The adult worms adopt the subterfuge of coating themselves with host antigens to evade attack by host defences; in themselves, the adults are innocuous. Most of the serious manifestations of schistosomiasis arise from the deposition of eggs, with the formation of granulomata and fibrotic lesions of the liver, bladder or other organs. Such effects may herald malignant changes. Heavy infection with S. mansoni may give rise to schistosomal dysentery, whereas S. haematobium infections are commonly accompanied by a marked haematuria (visible in Fig. 63.4E).
Laboratory diagnosis
Ova of rectal schistosomes can be sought on the surface of formed faeces. Blood-stained mucus should be examined, if present. Alternatively, microscopical examination of snips of rectal mucosa teased out in a drop of saline on a microscope slide may reveal viable or calcified eggs.
For the diagnosis of infection with S. haematobium, the last few drops of urine at the end of micturition (terminal urine) are most likely to be rich in ova. Excretion is said to be maximal around midday.
To test for viability of the eggs after treatment, the ova can be hatched in water (the motile miracidia can be seen with the help of a hand-lens) or the eggs can be examined microscopically for the characteristic flickering movement of excretory ‘flame cells’.
Various serodiagnostic tests, including enzyme-linked immunosorbent assay (ELISA), are available, but are no substitute for demonstration of the ova. Antigen detection methods have also been developed.
Treatment
Praziquantel is effective against all the human schistosomes and is the drug of choice. Because of its lack of toxicity and simplicity in administration. praziquantel has been used, together with molluscicides and water purification, in control programmes.
Other compounds are more selective in their action: metrifonate (an organophosphate compound) is active against S. haematobium; oxamniquine is effective in S. mansoni infection.
Cestodes
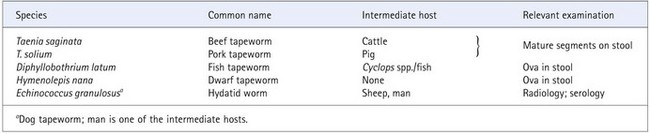
The species of tapeworm most commonly involved in human infection are listed in Table 63.6.
Table 63.6 Principal cestode parasites of man
Taenia species
Taenia saginata, the beef tapeworm, is much more prevalent than the related T. solium, the pork tapeworm. Both have a relatively simple life cycle, alternating between man and the intermediate host. Human infection is acquired by eating raw or undercooked beef or pork containing the encysted larval stage, the cysticercus. The larvae hatch in the small intestine and attach to the mucosal surface by four suckers on the head (scolex) of the worm. The scolex of T. solium additionally carries a crown of hooklets. The worm grows backwards from the head, first producing immature segments (proglottids), which continue to develop as they become more distant from the head. When sexually mature, the proglottids, which exhibit both male and female characteristics, cross-fertilize one another, and eggs start to be produced in the uterine canal. This becomes grossly distended as more eggs are produced, so that the fully gravid segments at the end of the worm become nothing more than bags full of eggs. The complete chain of segments is known as a strobila, and may measure 10 m or more.
Eggs are not laid. They are retained within the proglottids, which become detached from the end of the worm and are passed with the faeces. Animals become infected by ingesting the eggs from pastures contaminated with inadequately treated sewage or by the droppings of birds that scavenge in untreated sewage.
Considering the size of the worm, infection is usually remarkably asymptomatic. However, in the case of T. solium, eggs may hatch in the human host and form cysticerci. When these lodge in the brain, they may cause a serious epileptiform disease, cerebral cysticercosis.
Laboratory diagnosis
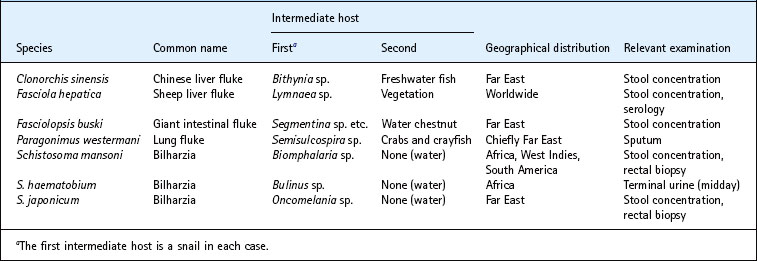
Taenia infection is usually diagnosed by finding the typical segments in faeces. Since eggs are not laid, faecal examination for ova is inappropriate. T. saginata can usually be differentiated from T. solium if the segment is pressed between two microscope slides and examined macroscopically. In the case of T. saginata, numerous branchings of the central uterine canal are evident, whereas there are usually far fewer branchings with T. solium (Fig. 63.5). The eggs (Fig. 63.6A) are thick walled and contain an oncosphere with six hooklets. The eggs of T. saginata and T. solium are indistinguishable.
Treatment
A single dose of praziquantel is usually successful. Niclosamide is also used, but this drug causes the worm to disintegrate, with the consequent theoretical (but unproven) risk of auto-infection in the case of T. solium through the intraluminal release of eggs. Treatment of cerebral cysticercosis is problematical, but albendazole and praziquantel have been used successfully.
Diphyllobothrium latum
This is the fish tapeworm, which is prevalent in lakeland areas where freshwater fish is eaten raw. A few cases caused by the related D. nihonkaiense have been reported, mainly from Japan. The life cycle is reminiscent of that of the trematodes. The mature adult worm, which may attain a length of 10 m, lays numerous operculate eggs within which a ciliated body called a coracidium develops. This hatches in water and is ingested by the water flea (Cyclops species). After a period of development, the larva awaits ingestion by a freshwater fish in which it invades the muscle as an infective plerocercoid larva or sparganum.
Human infection is usually asymptomatic, although a form of pernicious anaemia caused by competition for dietary vitamin B12 has been described.
The characteristic immature eggs have an indistinct operculum (Fig. 63.6B) and are usually present in large numbers in faeces. Occasionally, a length of the worm may break off and be passed in the stool.
Hymenolepis nana
In contrast to the enormous length of Taenia species and D. latum, Hymenolepis nana is only 2–4 cm long, and is consequently known as the dwarf tapeworm. It has a very simple life cycle with no known intermediate host. The characteristic ‘poached egg’ ova (Fig. 63.6C) are directly infective, and it is surprising that infection is not more common.
Infection is usually asymptomatic; heavy infections can be treated with praziquantel or niclosamide.
A slightly larger species, H. diminuta, is occasionally found in man. This is a parasite of small rodents and is transmitted by their fleas.
Echinococcus granulosus
This is the tapeworm of the dog and other canine species, and, unusually, man is an intermediate host. It is a small worm, consisting usually of just four segments and measuring less than a centimetre. Sheep are the usual intermediate hosts. Other animals, including man, may become infected, especially in sheep-farming areas, where the cycle of transmission is maintained between sheep and dogs.
After ingestion of the eggs, which resemble those of Taenia species, larvae hatch in the small intestine, penetrate the gut mucosa and are carried by the bloodstream to various organs (commonly the liver), where they are filtered out. The larva starts to grow, eventually forming a cystic cavity, the hydatid cyst. The inner wall of the cyst contains the germinal layer, from which develop brood capsules that bud off and fall into the cyst cavity. Within these brood capsules new scolices develop, and some of these may initiate the formation of daughter cysts within the main cavity. The young cyst may die and calcify, but it often continues to grow inexorably, eventually seriously compromising the function of the organ in which it is situated.
In certain parts of the world, notably the arctic regions of North America and Siberia, infection with a related canine tapeworm, E. multilocularis, is encountered. The hydatid cyst infiltrates the surrounding tissue, making it difficult to remove surgically.
Laboratory diagnosis
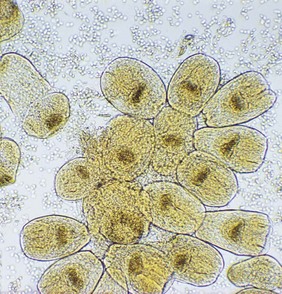
The diagnosis is usually made on clinical and radiological evidence. Examination of the cyst fluid (hydatid sand) reveals the typical invaginated scolices (Fig. 63.7), but diagnostic puncture of cysts is not recommended because of the risk of spillage (see below).
Imaging techniques supported by serological tests offer the best means of diagnosis. ELISA is the preferred laboratory method, but other serodiagnostic tests are also available. A skin test with antigen derived from hydatid fluid (Casoni test) was formerly used, but is unreliable.
Treatment
Cysts of E. granulosus can often be removed surgically, but accidental spillage of viable scolices into body cavities may cause an anaphylactic reaction and, moreover, is likely to lead to the development of fresh cysts. For this reason the hydatid cyst is first injected with a scolicidal agent, such as hypertonic saline or ethanol.
The relative impermeability of the cyst militates against successful chemotherapy, but some success has been obtained with benzimidazole derivatives, notably albendazole, and with praziquantel.
Cook GC, Zumla A. Manson’s Tropical Diseases, ed 22, London: Elsevier: Saunders, 2008.
Crompton DWT. How much helminthiasis is there in the world? Journal of Parasitology. 1999;85:397–403.
Guerrant RL, Walker DH, Weller PF. Tropical Infectious Diseases, ed 3. Philadelphia: Elsevier: Saunders; 2011.
Peters W, Pasvol G. Atlas of Tropical Medicine and Parasitology, ed 6. London: Elsevier: Mosby; 2006.
Taylor MJ, Hoerauf A, Bockarie M. Lymphatic filariasis and onchocerciasis. Lancet. 2010;376:1175–1185.
World Health Organization. Basic Laboratory Methods in Medical Parasitology. Geneva: WHO; 1991.
Wellcome Trust 1998 Schistosomiasis (Topics in International Health series).
Available from Wellcome Trust, London (www.wellcome.ac.uk/tih) and (for developing countries) from TALC (Teaching-aids at Low Cost; www.talcuk.org)
Centers for Disease Control and Prevention. DPDx – CDC Parasitology Diagnostic Website http://www.dpd.cdc.gov/dpdx/
World Health Organization: Basic Laboratory Methods in Medical Parasitology http://whqlibdoc.who.int/publications/9241544104_%28part1%29.pdf