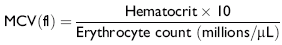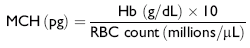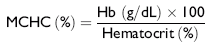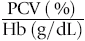Chapter 24 Alterations in the Erythron
The erythron is composed of all data pertaining to erythrocytes in the peripheral blood. In most instances the routine complete blood count (CBC), which includes microscopic evaluation of a blood smear, provides the data discussed in this chapter. After evaluation of the CBC, additional data on the erythroid compartment may be necessary, such as can be obtained from staining for Heinz bodies, the Coombs’ test, or the erythrocyte fragility test. All pertinent tests associated with erythroid diseases are discussed in this chapter except bone marrow analysis.
Any description of hematologic alterations in large animals would be incomplete without a brief discussion of the unique features of the equine erythron. To correctly interpret hematologic data, the practitioner must appreciate the characteristics that distinguish the horse from other domestic animals.
ANEMIA
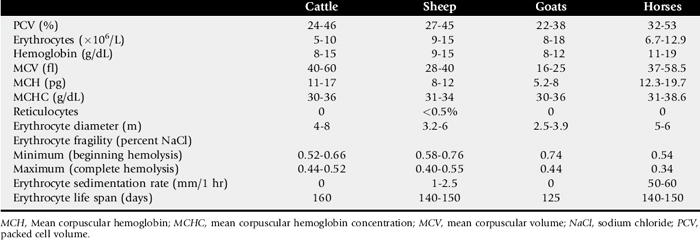
Anemia is functionally defined as decreased oxygen-carrying capacity of the blood. The most accurate laboratory indication of anemia is a drop in the PCV or hematocrit below the normal range. The PCV must always be interpreted in light of the animal’s hydration status and level of excitement, especially in horses. Because goats have very small erythrocytes (Table 24-1), the PCV in these animals must be determined by microhematocrit centrifugation (12,000 g for 5 minutes) to prevent plasma trapping in the erythrocyte column.1 This is the most accurate method of PCV determination in all species.
The three pathophysiologic mechanisms for the development of anemia are blood loss, increased erythrocyte destruction (hemolysis), and inadequate erythrocyte production. In the first two instances the bone marrow is normal and responds by increased erythropoiesis (regenerative or responsive anemias). In ruminants, regenerative anemias are attended by the appearance of immature erythrocytes in the peripheral blood. Inadequate erythrocyte production is caused by a bone marrow abnormality, and by definition the anemia is nonregenerative. Often anemia in large animals is caused by a combination of pathophysiologic mechanisms (Boxes 24-1 and 24-2).
Alteration in Mean Corpuscular Volume
The mean corpuscular volume (MCV) is a reflection of mean erythrocyte size, as expressed in the following equation:
An increased MCV (macrocytosis) indicates a regenerative anemia, because immature erythrocytes are larger than mature ones. Iron deficiency results in a decreased MCV (microcytosis), because cells undergo an extra division as a result of inadequate hemoglobin concentration. Inadequate spinning of blood causes a spurious elevationof the MCV by trapped plasma. This is most commonly a problem in goats.
Alteration in Mean Corpuscular Hemoglobin
The mean corpuscular hemoglobin (MCH) is an estimation of the amount of hemoglobin (Hb) in the blood per erythrocyte. It is calculated according to the following equation:
An increased MCH may indicate (1) the presence of reticulocytes (immature erythrocytes) in the peripheral blood or (2) hemolysis, either in vivo or in vitro. Iron deficiency results in a decreased MCH.
Alteration in Mean Corpuscular Hemoglobin Concentration
The MCH concentration (MCHC) is the most accurate of erythrocytic indices. It can be expressed as a percentage or in grams per deciliter:
Reticulocytosis (erythroid regeneration) or iron deficiency results in a decreased MCHC; hemolysis (in vivo or in vitro) causes an increased MCHC. Inadequate spinning of blood produces a spurious reduction in MCHC.
Anisocytosis
Variation in the size of erythrocytes is caused by the presence of macrocytes or microcytes (or both) among normal cells. Slight to moderate anisocytosis is normal in cattle, but marked anisocytosis in ruminants is a sign of regenerative anemia. Macrocytic erythrocytes may be seen at the peak of erythrocyte release during equine regenerative anemia, but in most horses effective regeneration occurs without macrocytosis.2
Polychromasia
Variation in color among the cells (using Romanovsky stains) is caused by the presence of reticulocytes that stain bluish because of residual DNA. In ruminants, polychromasia is a sign of regenerative anemia. Reticulocytosis can be quantitated most accurately by using new methylene blue stain, which causes cytoplasmic DNA to appear as blue granules. Insufficient cellular hemoglobin caused by iron deficiency results in decreased staining intensity and increased central pallor of erythrocytes (hypochromia). Hypochromia is difficult to recognize in large animals because their erythrocytes are small.
Poikilocytosis
The presence of abnormally shaped erythrocytes indicates increased erythrocyte fragility or diseases characterized by erythrocyte fragmentation. In rare cases, poikilocytosis may accompany iron deficiency or disseminated coagulopathy in large animals.
Basophilic Stippling
Tiny blue granules occasionally are observed in Romanovsky-stained erythrocytes containing residual DNA. This is a normal feature of regenerative anemia in cattle and sheep. In cattle, basophilic stippling also may indicate chronic lead poisoning.
Howell-Jolly Bodies
Basophilic nuclear remnants commonly are seen in immature erythrocytes during responsive anemia in ruminants. In healthy horses a few Howell-Jolly bodies occur normally.
Nucleated Erythrocytes
Nucleated erythrocytes and metarubricytes occasionally appear in the peripheral blood during the responsive phase of severe anemia in ruminants.
Heinz Bodies
Oxidative stress to erythrocytes causes denaturation of hemoglobin, which precipitates as aggregates, called Heinz bodies. Heinz bodies appear as round structures projecting from one edge of the red cell and are most easily visible on new methylene blue preparations. Erythrocytes containing Heinz bodies are susceptible to intravascular hemolysis and removal in the mononuclear phagocyte system (MPS). Heinz body hemolytic anemia is seen in cattle that have ingested toxic amounts of onions or plants of the Brassica genus. Horses develop Heinz body anemia in association with toxicoses caused by phenothiazine, red maple leaves, and, in rare cases, onions.
Autoagglutination
Aggregation of erythrocytes may be observed grossly or microscopically during immune-mediated anemia in horses or cattle. Marked rouleau formation, which occurs normally in horses, may be differentiated from agglutination by diluting the blood sample 1:4 with 0.9% saline. Both rouleau formation and autoagglutination, rarely induced by severe inflammation, are dispersed by saline dilution.
Increased Erythrocyte Fragility
The erythrocyte fragility test is a measure of the susceptibility of erythrocytes to hemolysis in a range of hypotonic saline concentrations (see Table 24-1). An increase in osmotic fragility is indirectly suggestive of immune-mediated anemia; the Coombs’ test is more specific.
Positive Direct Antiglobulin (Coombs’) Test Result
A positive Coombs’ test result indicates the presence of antibodies on the surface of erythrocytes. A positive test result may be found in idiopathic autoimmune hemolytic anemia in any species and in horses with neonatal isoerythrolysis or equine infectious anemia. The Coombs’ reagent is a mixture of antibodies directed against immunoglobulins and complement of a certain species. Because the endpoint of this test is agglutination, it cannot be performed on blood that is autoagglutinating. There are many false-negative results.
Erythrocytic Parasites
During the acute stages of bovine anaplasmosis and babesiosis in horses, cattle, sheep, and goats, intraerythrocytic parasites can be found. Anaplasma marginale is seen as a round, basophilic inclusion at the edge of cells, present in highest numbers before a hemolytic crisis. Babesia trophozoites occur in erythrocytes as round, bizarre, rod-shaped, or typical piriform (teardrop-shaped) structures. Absence of intraerythrocytic parasites does not rule out anaplasmosis or babesiosis.
Clinical Signs of Anemia
The major clinical signs of anemia (e.g., tachycardia, tachypnea, reduced exercise tolerance, and depression) reflect physiologic adjustments to inadequate oxygen transport to body tissues. The PCV level at which clinical signs occur depends on the rate of development, the severity of the anemia, and the physical demands placed on the animal. Other clinical signs depend on the cause and mechanism of anemia development. Anemia is accompanied by mucosal pallor except when it is caused by hemolysis, which results in icterus. Red urine (hemoglobinuria) indicates intravascular hemolysis, which may be accompanied by fever. Melena, hematuria, and petechial hemorrhages may indicate chronic blood loss.
Although diagnosing anemia is easy, determining the cause, which dictates proper treatment, may be complex. The practitioner first must determine the pathophysiologic classification of the anemia and then consider possible causes.
Approach to Diagnosis of Anemia in Horses
The ratio of PCV/HB less than 3 suggests intravascular hemolysis (e.g., PCV=15; Hb=6; 15/6 is <3).
Approach to Diagnosis of Anemia in Ruminants
ERYTHROCYTOSIS (POLYCYTHEMIA)
Erythrocytosis is defined as an increase in the PCV, erythrocyte count, and hemoglobin concentration above the normal range. Erythrocytosis may be absolute or relative (apparent), caused by hemoconcentration (dehydration, shock) or splenic contraction (Box 24-3). Absolute erythrocytosis (primary or secondary) is caused by increased erythropoiesis that creates a greater total circulating erythrocyte mass.
Primary absolute erythrocytosis (polycythemia vera) is an idiopathic, myeloproliferative disorder associated with a normal partial pressure of oxygen (PO2) and reduced erythropoietin levels. Secondary absolute erythrocytosis is caused by an increase in erythropoietin. Chronic tissue hypoxia, which may accompany residence at high altitudes, chronic pulmonary disease, and heart defects that produce arteriovenous shunting induce a physiologic or compensatory increase in serum erythropoietin that results in absolute secondary erythrocytosis. Inappropriate elaboration of erythropoietin (normal PO2) and secondary erythrocytosis rarely occur in chronic renal, hepatic, or endocrine disorders, especially those caused by neoplasia.
In domestic animals, absolute erythrocytosis usually occurs secondary to chronic diseases that produce tissue hypoxia. Primary absolute erythrocytosis and inappropriate secondary erythrocytosis caused by hepatocellular carcinoma have been described in horses.4 Familial erythrocytosis has been described in cattle, and the source of the increase in erythropoietin has not been identified.5 Clinical signs of erythrocytosis are vague; they include lethargy, weight loss, mucosal hyperemia, and signs of underlying disease.
Diagnosis of erythrocytosis is based on persistent elevation of the PCV, hemoglobin, and erythrocyte count in the absence of clinical evidence of shock or dehydration and without response to intravenous fluid therapy. Chronic hypoxia can be ruled out by determining the arterial oxygen concentration. Thoracic radiographs and echocardiography can delineate cardiorespiratory function more thoroughly. Examination of the bone marrow is indicated, although erythroid hyperplasia is not specific for primary or secondary erythrocytosis. In the absence of hypoxemia and a demonstrable disease that could lead to appropriate secondary erythrocytosis, polycythemia vera and inappropriate secondary erythrocytosis must be considered. Renal and hepatic disease should be excluded by determining the serum creatinine, hepatic enzyme, and bile acid levels. The only way to clearly differentiate primary from secondary absolute erythrocytosis is to determine the serum erythropoietin. This is a bioassay that is not routinely available, and it is relatively insensitive to minor changes in erythropoietin concentrations.
1 Jain NC. Schalm’s veterinary hematology, ed 4. Philadelphia: Lea & Febiger, 1986.
2 Radin MJ, Eubank MC, Weiser MG. Electronic measurement of erythrocyte volume and volume heterogeneity in horses during erythrocyte regeneration associated with experimental anemias. Vet Pathol. 1986;23:656.
3 Duncan JR, Prasse KW, Mahaffey EA. Veterinary laboratory medicine: clinical pathology, ed 3. Ames, Iowa: Iowa State University Press, 1994.
4 Beech J, Bloom JC, Hodge TG. Erythrocytosis in a horse. J Am Vet Med Assoc. 1984;184:986.
5 Tennant B, et al. Familial erythrocytosis in cattle. J Am Vet Med Assoc. 1967;150:1493.