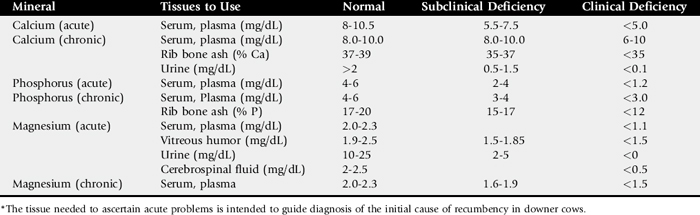
CALCIUM, MAGNESIUM, AND PHOSPHORUS
Adequate blood calcium (Ca), magnesium (Mg), and phosphorus (P) concentrations are vital to normal function of animals. Mechanisms for maintaining blood Ca, Mg, and P concentrations within normal limits perform efficiently most of the time. Occasionally, these homeostatic mechanisms fail, and metabolic diseases, such as milk fever, occur. Inadequate blood Ca, P, Mg, or potassium (K) concentrations (discussed later in chapter) can cause a cow to lose the ability to rise to her feet, as these minerals are necessary for nerve and muscle function. Less severe disturbances in blood concentrations of these minerals can cause reduced feed intake, poor ruminal and intestine motility, poor productivity, and increased susceptibility to other metabolic and infectious disease.
CALCIUM
Ca is the major mineral of bone. The skeleton of a 600-kg cow contains approximately 8.5 kg of Ca. Extracellular Ca is also essential to ensure transmission of nervous tissue impulses, excitation of skeletal and cardiac muscle contraction, and blood clotting and as a component of milk. Intracellular Ca, although 1/10,000 the concentration of extracellular Ca, is involved in the activity of a wide array of enzymes and serves as an important second messenger conveying information from the surface of the cell to the interior of the cell. Low extracellular Ca concentration causes hyperexcitability of the nervous system, causing muscle fasciculations or even tetany. At the neuromuscular junction, the amount of acetylcholine released during an endplate action potential is directly related to the amount of Ca that enters the terminal end of the motor neuron, which is in turn dependent on extracellular Ca concentration. Thus, hypocalcemia reduces the strength of muscle contraction, resulting in paresis. In many species (dog, cat, human, horse) the prevailing effects of hypocalcemia result in tetany. In the cow and sheep, hypocalcemia manifests as paresis, though it is important to point out that both effects of hypocalcemia are occurring at the same time and careful evaluation of a hypocalcemic animal will reveal both signs.
Calcium Homeostasis
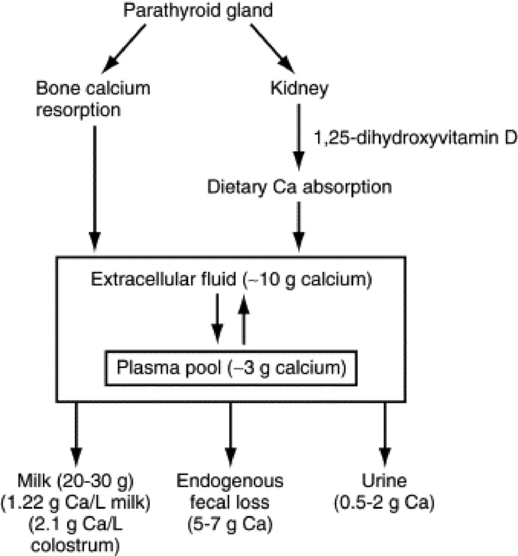
Blood Ca concentration in the adult cow or sheep is maintained at 8.5 to 10 mg/dL and is slightly higher in young animals (Fig. 41-18). There are approximately 3 g of Ca in the plasma pool of a 600-kg cow and 8 to 9 g in the extracellular pool. The Ca concentration in the blood is tightly regulated, primarily by the parathyroid gland, which responds to even a small decrease in Ca concentration by secreting PTH into the blood. PTH first will act on the kidney to increase renal tubular reabsorption of Ca from the glomerular filtrate. However, because only small amounts of Ca are lost in urine (<1 to 2 g/day in the cow), this action of PTH is sufficient to restore normal blood Ca concentration only if the total deficit is small. Larger Ca deficits cause prolonged secretion of PTH (hours to days), which stimulates osteoclastic resorption of bone Ca and stimulates renal production of 1,25-dihydroxyvitamin D. The 1,25-dihydroxyvitamin D stimulates the intestinal epithelial cells to produce Ca binding proteins and Ca pumps so that Ca within the lumen of the gut is efficiently transported across the intestinal epithelial cells into the blood. Should an animal be fed a Ca- or vitamin D—deficient diet, it will generally maintain normal blood Ca concentrations for weeks to months by resorbing bone Ca. However, this ultimately will cause bone disease such as osteoporosis and osteomalacia. An increase in blood Ca concentration above normal shuts off PTH and stimulates release of calcitonin from the thyroid gland C cells. This hormone increases renal clearance of Ca and decreases osteoclast activity so more Ca is retained in bone.
ACUTE HYPOCALCEMIA (MILK FEVER) IN DAIRY COWS
Nearly half of all dairy cows will experience subclinical hypocalcemia (<7.5 mg/dL) within 24 hours of calving. Although this reduces ruminal and abomasal contractility and reduces feed intake, clinically these animals appear normal. Hypocalcemia also contributes to metritis, mastitis, and retained placenta by negatively affecting uterine and teat sphincter contractility and the immunity of the cow. With further reduction in blood Ca concentration the animals can appear ataxic, and some will be slightly bloated as eructation is reduced. Finally, in as many as 5% of U.S. cows, blood Ca will fall below 5 mg/dL, and in a few Ca will be as low as 2 mg/dL. These cows are recumbent and unable to rise and have a condition commonly referred to as milk fever or parturient paresis. They often lay with the neck out in an S-shaped curve, and close examination of muscles in the legs and neck will reveal fasciculation of some muscle groups. The heart muscle does not contract strongly, so on auscultation the heartbeat is muffled. The heart rate is generally increased to try to compensate for the low ventricular ejection volumes. The cow loses the ability to thermoregulate and takes on the ambient temperature of its surroundings. In most cases this causes the skin, especially skin of the ears, to be cold. However, in hot climates or when the cow is recumbent in the hot sun the body temperature may be elevated well above normal. Milk fever typically occurs within a day or two of calving—usually after but occasionally just before calving.
Acute hypocalcemia also occurs with many infectious conditions, such as mastitis or metritis, especially if endotoxins are elaborated. As a rule the blood Ca concentration is 6 to 8 mg/dL. This form of hypocalcemia is a result of redistribution of Ca within organs and will not be discussed further other than to note that not all hypocalcemic cows have the syndrome known as milk fever (Box 41-4).
Treatment of milk fever and hypocalcemia should be done as early as possible, especially if recumbency is present. The pressure exerted by the massive weight of the cow can cause a “crush syndrome” effect on the down side appendages in as little as 4 hours. This causes ischemia of the muscles and nerves and is followed by necrosis of these tissues, resulting in the “downer syndrome” cow.344 The fastest way to restore normal plasma Ca concentration is to administer an intravenous injection of Ca salts (commonly Ca borogluconate). In general, commercial preparations for intravenous use supply from 8.5 to 11.5 g of Ca per 500 mL. They may also contain sources of Mg, P (often as ineffective phosphite), and glucose. The most effective intravenous Ca dose is approximately 2 g of Ca per 100 kg of body weight. A good rule of thumb is to administer the Ca at a rate of 1 g/min. If Ca is administered too rapidly, fatal arrhythmia of the heart and cessation during systole can occur. Intravenous Ca treatments increase blood Ca above normal for approximately 4 hours. Ca salts can also be injected subcutaneously, but absorption is variable because blood flow to the periphery is often compromised. The amount of Ca that can be injected into a single subcutaneous site should be limited to 1 to 1.5 g of Ca (50 to 75 mL of most commercial preparations). Ca preparations designed for intramuscular administration are also available (Ca levulinate or Ca lactate). Most of these preparations must be limited to 0.5 to 1 g of Ca per injection site to avoid tissue necrosis. To get an effective dose of Ca into the clinically hypocalcemic animal might therefore require 6 to 10 injections into widely separated spots. Oral Ca treatments are not recommended as treatments for clinical milk fever cases, although they can be effective aids in prevention of milk fever.
HYPOCALCEMIA IN LATE-GESTATION BEEF COWS AND EWES
For beef cows and ewes, especially those carrying twins, meeting the sudden increase in fetal skeletal demand (8 to 10 g/fetal calf/day) for Ca often presents a greater challenge to Ca homeostasis than does lactation. Estrogen, which increases dramatically in late gestation, decreases osteoclast activity. This reduces the ability to use bone Ca reabsorption to meet fetal skeletal Ca demands. As a result, most primary hypocalcemic disorders of the beef cow and ewe occur in late gestation. They are usually prevented by increasing Ca and/or Mg in the gestation diet; provided the animal is eating. Often the syndrome in beef cows and ewes is complicated by inadequate energy intake and a state of inappetence associated with the pregnancy toxemia syndrome.
Etiology of Milk Fever
Dairy cows producing colostrum (containing 1.7 to 2.3 g of Ca per kilogram) or milk (containing 1.2 g of Ca per kilogram) typically secrete 20 to 30 g of Ca each day in early lactation. Put simply, hypocalcemia and milk fever occur when cattle do not obtain enough Ca from their bones and diet to replace Ca lost to milk. The long-accepted reason this occurs was that high dietary Ca fed to the cow before calving placed the cow in such a state of positive Ca balance that the parathyroid gland atrophied, rendering it too sluggish to adequately respond when onset of lactation placed the cow into negative Ca balance. However, in recent years it has been discovered that high dietary Ca does not have this effect, and in most cases PTH secretion is quite adequate in these animals.
Role of Metabolic Alkalosis
Metabolic alkalosis predisposes cows to milk fever and subclinical hypocalcemia by altering the conformation of the PTH receptor, rendering the tissues less sensitive to PTH (Fig. 41-19).345-347 Lack of PTH responsiveness by bone tissue prevents effective use of bone canaliculi fluid Ca (sometimes referred to as osteocytic osteolysis) and prevents activation of osteoclastic bone resorption. Failure of the kidneys to respond to PTH reduces renal reabsorption of Ca from the glomerular filtrate. More important, the kidneys fail to convert 25-hydroxyvitamin D to 1,25-dihydroxyvitamin D. Therefore enhanced intestinal absorption of dietary Ca, which normally would help restore blood Ca to normal, fails to be instituted.
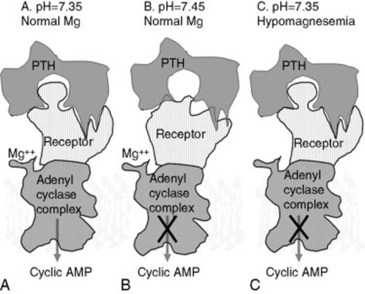
Fig. 41-19 Parathyroid hormone (PTH) effects at the surface of target bone and kidney cells. A, Under normal conditions, PTH released in response to hypocalcemia interacts with its receptor, located on the surface of bone and kidney cells, in a lock-and-key fashion. This stimulates G-proteins and adenylate cyclase (adenylate cyclase complex), resulting in production of cyclic adenosine monophosphate (AMP), which acts as a second messenger within the cytosol of target cells. This initiates mechanisms such as bone calcium (Ca) reabsorption and renal production of 1,25-dihydroxyvitamin D to restore blood Ca concentration to normal levels. B, Alkalotic conditions induced by high-potassium diets induce a change in the shape of the PTH receptor protein so that it is less able to recognize and bind PTH, resulting in failure to activate the cell by producing cyclic AMP. C, Magnesium is required for function of the adenylate cyclase complex. Hypomagnesemia reduces the ability of PTH-stimulated cells to produce cyclic AMP, resulting in failure to activate the cell.
Metabolic alkalosis of dairy cows is caused by high-potassium diets. This is best explained by Stewart’s348 strong ion difference theory of acid-base physiology, which states that the number of moles of positively charged particles (cations) in any given solution (including body fluids) must equal the number of moles of negatively charged particles (anions) in the solution. Put into extremely simplified terms, if positively charged particles are added to a solution, such as the plasma, the number of H+ cations will decrease and the number of OH− anions will increase to maintain the electroneutrality of the solution (the solution becomes more alkaline). Conversely, adding anions to a solution causes an increase in H+ and a decline in OH− to maintain electroneutrality, and the pH decreases (the solution becomes more acidic).
Cations and anions enter the blood from the digestive tract, making the cation-anion difference of the diet the ultimate determinant of blood pH. The major cations present in feeds and the charges they carry are sodium (+1), potassium (+1), Ca (+2), and Mg (+2). The major anions and their charges found in feeds are chloride (−1), sulfate (−2), and phosphate (assumed to be −3). Cations or anions present in the diet will alter the pH of the blood only if they are absorbed into the blood. The difference between the number of cation and anion particles absorbed from the diet determines the pH of the blood.
When formulating dairy cow late-gestation diets, the cation-anion difference of a diet is described in terms of mEq/kg or mEq/100 g of diet, using various equations involving Na, K, Ca, Mg, Cl, sulfur, and P, with and without adjustments for absorbability of each mineral in the diet:
The equations largely become an academic pursuit, as several of the variables in the formulas shown are somewhat fixed constants. A strategy I use is to set dietary Ca at 0.85% to 1% and dietary P and Mg at 0.4%. Dietary sulfur needs to be above 0.22% to ensure adequate substrate for ruminal microbial amino acid synthesis but below 0.4% to avoid possible neurologic problems associated with sulfur toxicity. The key to milk fever prevention is to keep sodium and potassium as close to the requirement of the cow as possible (0.1% for Na and 1% for potassium). The key to reduction of subclinical hypocalcemia is to then add chloride to the ration to counteract the effects of low levels of potassium on blood alkalinity. As a rule of thumb, the amount of dietary chloride needed to acidify the cow’s blood (and urine) is approximately 0.5% less than the concentration of K in the diet. For example, if dietary K can be reduced to only 2% of the diet, Cl would need to be roughly 1.5% to acidify the cow. This level of Cl in the diet is likely to cause a decrease in DMI. Chloride sources differ in their palatability, and because achieving low dietary K can be difficult, it is prudent to use a palatable source of Cl when formulating the diet. Ammonium chloride (or ammonium sulfate) can be particularly unpalatable when included in rations with a high pH. At the higher pH the ammonium cation is converted to ammonia, which is highly irritating when smelled by the cow. Prilling the Cl (and SO4) salts can reduce the unpleasant taste of the salts. In our experience, hydrochloric acid has proved the most palatable source of anions. Hydrochloric acid can be extremely dangerous to handle when it is procured as a liquid concentrate. Several companies now manufacture hydrochloric acid—based anion supplements that are safe to handle.
These are simply guidelines for anion supplementation that I use and are based on inclusion of Ca, Na, S, Mg, and P at the levels outlined previously. Urine pH of the cows provides a cheap and relatively accurate assessment of blood pH and can be a good gauge of the appropriate level of anion supplementation.349 Urine pH on high-cation diets is generally above 8.2. Limiting dietary cations will reduce urine pH only a small amount (pH 7.5 to 7.8). For optimal control of subclinical hypocalcemia the average pH of the urine of Holstein cows should be between 6.2 and 6.8, which essentially requires addition of anions to the ration. In Jersey cows the average urine pH of the close-up cows has to be reduced to between 5.8 and 6.3 for effective control of hypocalcemia. If the average urine pH is between 5.0 and 5.5, excessive anions have induced an uncompensated metabolic acidosis, and the cows will experience a decline in DMI. Urine pH can be checked 48 or more hours after a ration change. Urine samples should be free of feces and made on midstream collections to avoid alkalinity from vaginal secretions. Anion-supplemented diets are generally fed for the last 2 to 3 weeks before calving.
High Dietary Phosphorus
High blood P concentrations inhibit the activity of the renal 25-hydroxyvitamin D 1α-hydroxylase enzyme.350-351 As summarized by Jorgensen,352 the incidence of milk fever increases when dietary P exceeds 90 g/day and little negative effect of dietary P is seen when dietary P is less than 50 g/day. Cows need approximately 35 to 40 g of P each day.353
Use of Low-Calcium Prepartal Diets to Prevent Milk Fever
By feeding late-gestation cows a diet that supplies less Ca than they require, the cows can be placed into negative Ca balance. This causes a minor decline in blood Ca concentration, which stimulates PTH secretion, which in turn stimulates osteoclastic bone resorption and renal production of 1,25-dihydroxyvitamin D. This increases bone Ca release and prepares the intestine to absorb Ca efficiently, should it become available. At parturition, the lactational drain of Ca is more easily replaced because the cow’s bone osteoclasts are already active and in high numbers and the previous stimulation of enterocytes by 1,25-dihydroxyvitamin D allows efficient use of dietary Ca. Unfortunately, a truly low-Ca diet should supply considerably less than 20 g of absorbed Ca per day. This is difficult to actually achieve on most dairy farms, although increasing use of straw in prepartal diets and the use of Ca binders, such as zeolite or vegetable oils, may make this approach more practical.354
Physiologic Milk Fever Risk Factors
The incidence of milk fever rises as a cow ages, especially as she enters her third or greater lactation. As animals age the number of active bone cells is reduced. Heifers, which are still growing, rarely have problems with hypocalcemia. Lower numbers of active osteoblasts mean fewer cells to respond to PTH and mobilize bone Ca. In addition, as animals age the number of receptors for 1,25-dihydroxyvitamin D on target tissues declines.
The incidence of milk fever is higher in Jersey cows compared with Holstein cows. Although Jersey cow colostrum and milk Ca concentration tend to be higher than in Holsteins, this does not appear to be the only factor. Preliminary data from our laboratory suggest that the intestine of Jersey cows possesses approximately 15% fewer receptors for 1,25-dihydroxyvitamin D than does intestine of Holstein cows.
Preventing Milk Fever with Vitamin D
Earlier literature often recommended feeding or injecting massive doses (up to 10 million units) of vitamin D 10 to 14 days before calving to prevent milk fever. This will pharmacologically increase intestinal Ca absorption and can help prevent milk fever. Unfortunately, the dose of vitamin D that effectively prevents milk fever is very close to the level that causes irreversible metastatic calcification of soft tissues, and therefore this strategy cannot be recommended. Lower doses may actually induce milk fever because the high levels of 25(OH)D and 1,25-dihydroxyvitamin D induced by the treatment suppress PTH secretion and directly suppress renal synthesis of endogenous 1,25-dihydroxyvitamin D.355
Treatment with 1,25-dihydroxyvitamin D and its analogues can be much more effective, but problems with timing of administration, withdrawal from treatment, expense, and availability have made these treatments not practical.356,357
Hypomagnesemia as a Cause of Hypocalcemia
Hypomagnesemia affects Ca metabolism in two ways. Moderate hypomagnesemia (<1.6 mg/dL) interferes with PTH action on tissues. When PTH binds its receptor on bone or kidney tissues, it normally initiates activation of adenylate cyclase and phospholipase C, resulting in production of the second messengers, cyclic adenosine monophosphate (AMP) and inositol 1,4,5-triphosphate. Both adenylate cyclase and phospholipase C have an Mg++ binding site that must be occupied by an Mg ion for full activity.358 Hypomagnesemia, from inadequate absorption of dietary Mg, is the second most common cause of milk fever in cows around the time of calving and is the most common cause of midlactation milk fever in dairy cows. More severe hypomagnesemia (<1.2 mg/dL) can inhibit PTH secretion in response to hypocalcemia. This appears to be a factor in the development of some hypomagnesemic tetany syndromes of grazing beef and dairy cattle. In these syndromes blood Mg concentration falls slowly over time until such point that PTH function is impaired. It is at this point that blood Ca concentration declines precipitously and clinical symptoms, such as tetany, become apparent.359
Agronomic Considerations for Producing Low—Dietary Cation-Anion Difference Forages
Reducing K in the ration of the late-gestation cow can present a problem. By restricting K application to the soil, it is possible to avoid luxury consumption of K by legumes and cool season grasses. Producers should also be aware that forages take up Cl from the soil, and it is possible to find hays that are low in K and high (1% to 1.2%) in Cl. Producers should use the lowest DCAD forage possible, not simply the lowest K forage. Forages intended for the close-up dry cow should routinely be analyzed for both K and Cl by a wet chemistry method. Near infrared determinations of forage mineral content are not currently useful.
Oral Calcium Treatments at Calving
Ca is absorbed across the intestinal mucosa by two methods. The first method is by the active transport of Ca across intestinal cells—a process mediated by the hormone 1,25-dihydroxyvitamin D. The second method is by passive diffusion across the tight junctions that hold adjacent mucosal cells together. The concept behind oral Ca supplementation is that the cow’s ability to use active transport of Ca across intestinal cells is inadequate to help her maintain normal blood Ca concentrations. By giving the animal large amounts of very soluble Ca, it is possible to force Ca across the intestinal tract by means of passive diffusion between intestinal epithelial cells. Best results are obtained with doses of Ca between 50 and 125 g. For best control of hypocalcemia, a dose is given at calving and again 24 hours later. Administering 50 g of Ca from CaCl as a drench in 250 mL of water is roughly equivalent to administering 4 g of Ca intravenously.360 Unfortunately, hypocalcemic cows have poor swallowing and gag reflexes, making them vulnerable to aspiration pneumonia. Aspiration of Ca solutions leads to a severe pneumonia. Ca chloride has been used in oral Ca preparations but can be very caustic. Ca propionate is less injurious to tissues and has the added benefit of supplying propionate, a gluconeogenic precursor. Toxic doses of Ca can be delivered orally; approximately 250 g of Ca in a soluble form will kill some cows.
CHRONIC CALCIUM DEFICIENCIES
Nutritional Secondary Hyperparathyroidism
A deficiency of dietary Ca or vitamin D can reduce the amount of Ca that can be absorbed across the intestine into the extracellular fluids. Diets severely deficient in Ca will not supply adequate Ca to replenish Ca lost from extracellular pools. As a result, blood Ca concentration decreases and PTH secretion increases. PTH can act on the kidney to reduce urinary Ca loss and increase synthesis of 1,25-dihydroxyvitamin D to enhance intestinal Ca absorption efficiency. But if dietary Ca is very low, increasing the efficiency of intestinal Ca absorption cannot substantially increase the amount of Ca entering the extracellular pool of Ca. The only action of PTH that can improve blood Ca concentration in this situation is to enhance bone Ca resorption. The continued removal of bone without replacement results in fibrous osteodystrophy. The clinical signs of nutritional secondary hyperparathyroidism include hypertrophy of the parathyroid glands (although they rarely can be palpated) and cessation of growth in young animals. The lack of Ca available for mineralization of bone matrix causes the growth plates to be soft, weak, and swollen. The cortices of the long bones are thin, and minor or major fractures are common.
Vitamin D deficiency prevents the kidneys from being able to synthesize 1,25-dihydroxyvitamin D. Therefore unless dietary Ca is greatly elevated, the reduced efficiency of dietary Ca absorption will prevent adequate Ca from entering the extracellular fluids, and, again, PTH will activate bone resorption mechanisms to attempt to maintain normocalcemia.
Renal Secondary Hyperparathyroidism
Renal secondary hyperparathyroidism is a syndrome caused by renal failure and is occasionally observed in older animals. In cows, pyelonephritis can develop in late gestation through early lactation owing to poor urine flow and retrograde urine flow resulting in destruction of renal tissue. A major function of the kidneys in most species is to remove excess phosphate from the circulation. As renal function is lost, phosphate is retained and hyperphosphatemia develops. This occurs when the remaining amount of functional renal tissue is less than 25% of normal.
Hyperphosphatemia has two effects. The first effect is to reduce the ionized Ca content of the blood. This is because Ca and phosphate ions normally exist in blood at concentrations that are slightly below the levels that would cause saturation of the fluids resulting in precipitation of Ca phosphate salt from the solution. However the greatly elevated phosphate level in the blood of renal failure patients can exceed the equilibrium of Ca and phosphate in solution, causing precipitation of Ca from the blood. More important, as phosphate builds up in the blood it has a second effect on the remaining renal tissue. It blocks the activation of the 1α-hydroxylase that catalyzes conversion of 25-dyhydroxyvitamin D to 1,25-dyhydroxyvitamin D within the kidney. Therefore even though PTH should stimulate the remaining functional renal tissue to produce 1,25-dyhydroxyvitamin D, this action of PTH is blocked by hyperphosphatemia.
As renal function is lost, the amount of renal tissue available for production of 1,25-dyhydroxyvitamin D decreases, and blood concentrations of 1,25-dyhydroxyvitamin D decline. This reduces dietary Ca absorption and further depresses blood Ca concentration. This in turn stimulates increased PTH secretion, and reabsorbing bone Ca reserves again becomes a major means of maintaining normocalcemia. Fibrous osteodystrophy follows the prolonged secretion of PTH.
As bone Ca is reabsorbed, more P is also reabsorbed. However, the loss of renal function prevents PTH from having its usual phosphaturic effect, exacerbating the hyperphosphatemia. A vicious cycle of increasing blood P, increasing PTH, and increased bone resorption ensues. The loss of renal function can rarely be reversed.
Osteoporosis
Three forms of osteoporosis commonly occur. Of these, only lactational osteoporosis is a common syndrome of concern in animals. During lactation in virtually all species there is an obligatory loss of bone mass from the skeleton. In this case the osteoclasts within a bone resorption unit reabsorb bone to a 50-micron depth if dietary Ca is adequate, or to greater depths if the animal is in severe negative Ca balance. However, osteoblast movement into the resorption cavity is transiently inhibited, and no new bone is deposited. This occurs to some extent in all females shortly after parturition, even when they are in positive Ca balance. However, the degree and duration can be magnified greatly by negative Ca balance. Once the animal has undergone the obligatory period of lactational osteoporotic bone loss (4 to 5 weeks in the cow), if the animal is in positive Ca balance the osteoblasts migrate to the resorption cavity and replace the lost bone. Lactational osteoporosis can help the female meet the Ca demands of lactation by uncoupling bone formation from bone resorption. In high-producing dairy cows, dietary Ca intake is inadequate to meet lactational Ca demands for the first 4 to 6 weeks of lactation. These animals can lose as much as 13% of their skeletal mass during this period, which is replaced in later lactation when dietary Ca intake allows the cow to enter a period of positive Ca balance.
MAGNESIUM
Mg is a major intracellular cation serving as a cofactor for enzymatic reactions vital to every major metabolic pathway. Extracellular Mg is vital to normal nerve conduction, muscle function, and bone mineral formation. In a 600-kg cow there is approximately 0.84 g of Mg in the blood, 3 g of Mg in the extracellular fluids, 84 g of Mg inside cells, and 210 g of Mg within bone mineral. Bone is not a significant source of Mg that can be used in times of Mg deficit, as bone reabsorption occurs in response to Ca homeostasis, not Mg status. Maintenance of normal plasma Mg concentration is nearly totally dependent on continuous dietary Mg absorption.
Hypomagnesemia
Hypomagnesemia generally leads to hyperexcitability, tetany, convulsion, and, too often, sudden death. Hypomagnesemia is often accompanied by and complicated by hypocalcemia. Hypomagnesemia is a common problem in ruminants.
Normal plasma Mg concentration is 1.8 to 2.4 mg/dL in cows and 2.2 to 2.8 mg/dL in sheep. The kidneys play a key role in maintaining Mg homeostasis under conditions of hypermagnesemia when they fail to reabsorb Mg from glomerular filtrate, increasing renal Mg excretion to lower blood Mg concentration. The renal threshold for Mg (plasma concentration at which all Mg filtered across the glomerulus is reabsorbed) is 1.8 mg/dL in the cow and 2.2 mg/dL in sheep. Plasma Mg concentrations below these levels indicate that dietary Mg absorption is not sufficient and little or no Mg will be detected in urine.
Moderate hypomagnesemia (1.1 to 1.8 mg/dL) is associated with reduced feed intake, nervousness, and reduced milk fat and total milk production. This can be a chronic problem in some dairy herds that often goes unnoticed. It can also predispose these animals to milk fever as described earlier.
Role of the Rumen in Hypomagnesemia
Mg is well absorbed from the small intestine of young calves and lambs. As the rumen and reticulum develop, these sites become the main, and perhaps the only, sites for net Mg absorption.361 In adult ruminants the small intestine is a site of net secretion of Mg. Mg absorption from the rumen is dependent on the concentration of Mg in solution in the ruminal fluid and the integrity of the Mg transport mechanism, which is an Na-linked active transport process.362
The soluble concentration of Mg in ruminal fluid can be low for several reasons. Chief among these are low Mg content of forages and inadequate dietary supplementation. Mg solubility also declines sharply as ruminal pH rises above 6.5. Grazing animals tend to have higher ruminal pH because of the stimulation of salivary buffer secretion. When high-grain rations are fed, ruminal fluid pH is often below pH 6.5, increasing Mg solubility and thus availability. Forages can also contain organic compounds, such as unsaturated fatty acids, which are converted to tricarballylate, which can form insoluble Mg salts in the rumen.
High dietary K can reduce the rumen’s ability to absorb Mg. High K concentration in the ruminal fluid depolarizes the apical membrane of the ruminal epithelium, reducing the electromotive potential needed to drive Mg across the ruminal wall.362 Feeding ionophores (monensin, lasalocid) can improve activity of the Na-linked Mg transport system in the rumen, increasing Mg absorption efficiency approximately 10%.363 Lush high-moisture pastures increase the rate of passage of material, including Mg, from the rumen so that Mg may leave the rumen before it can be absorbed.
Occurrence of Hypomagnesemic Tetany
Hypomagnesemic tetany occurs most often in beef cows, dairy cows, and ewes in early lactation grazing lush pastures high in potassium and nitrogen and low in Mg and Na. This is often referred to as grass tetany, spring tetany, grass staggers, or lactation tetany. Mg deficiency occurs most often in spring or fall when pastures are growing at maximal rates and is most common in grazing lactating ruminants, as milk production removes 0.15 g of Mg from the blood for each liter of milk produced. Cool weather seems to play a factor as well, probably through its effects on plant Mg uptake, although there is some indication that the physiologic response of the cow to cool weather affects Mg status directly.
When plasma Mg levels fall below 1.1 mg/dL, twitching is sometimes seen in the muscles of the face, shoulder, and flank. As hypomagnesemia progresses, tetanic spasms of the muscles become more common, eventually causing the cow or ewe to stagger and fall. Clonic convulsions quickly follow, with chomping of the jaws and frothy salivation. Affected animals lay with the head arched back and the legs paddling. The heart rate can approach 150 beats/min, and the heartbeat is often audible without a stethoscope. Respiratory rate approaches 60 breaths/min, and rectal temperature can approach 40.5° C (105° F) as a result of the excessive muscular activity. The eyelids flutter, and there is usually marked nystagmus. Animals may get up after several minutes and repeat these convulsive episodes several times before they finally die. Hypomagnesemic tetany in calves is clinically similar to that in adults and is often accompanied by moderate hypocalcemia.
Ewes are generally hypocalcemic and hypomagnesemic. Affected ewes are usually in the second to fourth week of lactation and are usually suckling more than one lamb. Affected ewes are generally depressed, stand with their heads down, and are reluctant to move. As hypomagnesemia and hypocalcemia progress, the animals develop tetany and clonic convulsions just as in cattle. The clinical signs in goats are similar to those observed in cattle.
Cerebrospinal fluid (CSF) Mg concentrations below 1 mg/dL are responsible for the clonic convulsions seen in animals with hypomagnesemic tetany. Blood samples obtained during or shortly after an episode of tetany may have near-normal levels of Mg as a result of muscle damage and leakage of Mg from intracellular pools. CSF Mg concentration will remain low during tetany and also can be a reliable indicator of Mg status for up to 12 hours after death. Vitreous humor Mg concentrations below 1 mg/dL are also found in animals with tetany and can be a reliable indicator for 24 to 48 hours after death, provided that environmental temperatures have not exceeded 23° C.364 Aqueous humor has not proved a reliable sample.
Treatment of Hypomagnesemia
Animals exhibiting hypomagnesemic tetany need immediate treatment. This will require administering 1.5 to 2.25 g of Mg intravenously in the adult cow. Most of the commercially available intravenous solutions used to treat milk fever supply 1.5 to 4 g of Mg, usually as the chloride, borogluconate, or hypophosphite salts of Mg. Response to therapy can be disappointing, and success is related to the interval between onset of tetany and treatment. Cows should not be stimulated to rise for at least 30 minutes after treatment, to avoid initiating tetany and convulsions. Cattle that recover do so approximately an hour after treatment, which is the time it takes CSF Mg concentration to return to normal. Many of these cows will relapse and require further treatment within 12 hours.
The rate of relapse can be reduced using orally administered Mg salts once the animal has regained good swallowing reflexes (to avoid aspiration pneumonia). Administering a slurry of 100 g of Mg oxide in water intraruminally or by drenching can be effective. This provides approximately 50 g of Mg to the animal. Adding 50 g of Ca carbonate, 100 g of dicalcium phosphate, and 50 g of sodium chloride may enhance the effectiveness of the slurry, especially if hypocalcemia and hypophosphatemia accompany the hypomagnesemia. Alternatively, 200 to 400 mL of a 50% Mg sulfate solution can be administered by drench. Mg sulfate is more available for absorption than Mg oxide. If hypomagnesemic tetany has occurred in one cow or ewe in a herd or flock, steps should be taken immediately to increase Mg intake in other members of the herd to prevent further losses. Administering an additional 10 to 15 g of Mg per pregnant cow, 20 g of Mg per lactating beef cow, and 30 g of Mg per lactating dairy cow daily, usually in a grain mix, will often prevent further hypomagnesemic tetany cases. Ewes and does can be treated with ⅛ of these formulas. The problem with prevention is ensuring the extra Mg gets into the animal, especially when working with animals at pasture.
Prevention
USING OTHER TRANSPORT MECHANISMS TO ABSORB DIETARY MAGNESIUM
The active transport mechanism for Mg absorption across the ruminal wall is critical to the survival of the animal when dietary Mg concentration is less than 0.25%. Unfortunately there are several known factors, such as dietary K, and several unknown factors that prevent this pathway from functioning well. A second pathway for absorption of Mg exists and operates only at high ruminal fluid Mg concentrations. At high ruminal Mg concentration the Mg will flow down its concentration gradient into the extracellular fluids of the cow. This passive transport mechanism is not subject to inhibition by K and is subject only to the concentration of Mg in solution in the rumen.362
The concentration of Mg in ruminal fluid required to use concentration gradient—driven absorption of Mg is at least 4 mMol/L. The minimum level of Mg required in the diet to use this pathway in order to prevent negative Mg balance in the face of high K levels in ruminants is approximately 0.35%.365 Mg content of the close-up dry cow ration and the early lactation ration should be between 0.35% and 0.4% as insurance against the possibility that the active transport processes for Mg absorption are impaired.
ASSESSING MAGNESIUM STATUS AT PARTURITION
PTH causes increased renal tubular reabsorption of Mg. The kidneys excrete less Mg, increasing blood Mg in the typical milk fever cow. However, if dietary Mg is insufficient or ruminal absorption of Mg is impaired, the blood Mg concentration will be below the normal renal threshold for Mg (1.85 mg/dL in cattle) and almost no Mg will appear in the urine. The animal should be considered abnormal for Mg. Sampling the blood of several cows within 12 hours of calving is a simple, effective index of Mg status of the periparturient cows. If serum Mg concentration is not at least 2 mg/dL in 9 of 10 cows sampled, it suggests inadequate dietary Mg absorption from either lack of dietary Mg or interferences with absorption. This same test can be used in the first weeks of lactation to see if the lactating cow diet is providing adequate Mg to the animal. This can be important, as hypomagnesemia may limit productivity of lactating cows as well as contributing to hypocalcemia in the herd.
PHOSPHORUS
Traditionally, phosphorus in clinical medicine is abbreviated as P, although it should be understood by the reader that the biologically relevant form of phosphorus is actually inorganic phosphate, not elemental phosphorus. P is a component of phospholipids, phosphoproteins, nucleic acids, and energy-transferring molecules such as ATP. P is an essential component of the acid-base buffer system. It is second only to Ca as a major component of bone mineral, with the skeleton of a 600-kg cow containing approximately 3.9 kg of P.
Phosphorus Homeostasis
Plasma P concentration is normally 4 to 8 mg/dL. Approximately 1 to 2 g of P are present in the plasma inorganic P pool, and 4 to 7 g of P in the extracellular P pool, of a 500-kg cow. Intracellular P concentration is approximately 78 mg/dL, and total body intracellular P content is approximately 155 g, with 5 to 6 g located within erythrocytes. Maintaining the extracellular P pool involves replacing P removed for bone and muscle growth, endogenous fecal loss, urinary P loss, and milk production with P absorbed from the diet or reabsorbed from bone. During late gestation, fetal skeletal development can withdraw up to 10 g of P per day from the maternal P pools. Approximately 0.3 g of P is incorporated into each kilogram of body tissue (muscle) gained during growth of the animal. Production of milk removes from the extracellular pool approximately 0.9 g of P per kilogram of milk produced. Salivary secretions remove between 30 and 90 g of P from the extracellular P pool each day.353 Factors affecting salivary phosphate secretion include the time spent ruminating (chewing activity) and the PTH status of the animal. PTH stimulates parotid salivary P secretion and can increase salivary phosphate concentrations twofold to threefold.366 Salivary phosphate secretions help buffer the rumen and supply ruminal microbes with a readily available source of P necessary for cellulose digestion. Most of the salivary phosphate secreted is recovered by intestinal absorption. However, even on low-P diets, a minimum of 5 g of secreted P per day is not recovered and is lost to feces. Urinary P loss is usually between 2 and 12 g/day. Bones of a 600-kg cow contain approximately 4 kg of P, some of which can be withdrawn and returned to the blood during osteoclastic resorption of bone.
Ruminal microbes are able to digest phytic acid, so most of the phytate-bound P, the form of up to 70% of P in plants, is available for absorption in ruminants. P is primarily absorbed in the small intestine via an active transport process that is responsive to 1,25-dihydroxyvitamin D. Intestinal P absorption efficiency can, in theory, be upregulated during periods of P deficiency, as renal production of 1,25-dihydroxyvitamin D can be directly stimulated by very low plasma P. However, the plasma P level must reach very low levels (<1 to 2 mg/dL) to stimulate increased renal production of 1,25-dihydroxyvitamin D. Plasma P concentrations are generally well correlated with dietary P absorption. P absorbed in excess of needs is excreted in urine and saliva.
PTH, secreted during periods of Ca stress, increases renal and salivary excretion of P, which can be detrimental to maintenance of normal blood P concentrations. This is one reason that hypocalcemic animals tend to become hypophosphatemic. PTH could conceivably increase blood P concentration because it stimulates bone mineral resorption. Also, because PTH stimulates the kidney to produce 1,25-dihydroxyvitamin D, it can increase the efficiency of intestinal phosphate absorption. However, it must be remembered that PTH is secreted primarily in response to hypocalcemia, not hypophosphatemia.
Hypophosphatemia, Pregnancy Toxemia, and Downer Cows
Beef cows and ewes fed a diet marginal in P will have a chronic hypophosphatemia of 2 to 3.5 mg/dL. In late gestation, plasma P can decline precipitously as the growth of the fetus accelerates and removes substantial amounts of P from the maternal circulation. These animals often become recumbent and are unable to rise, though they appear fairly alert and will often eat feed placed in front of them. Cows and ewes carrying twins are most often affected. Plasma P concentration in these recumbent animals is often less than 1 mg/dL. The disease is often referred to as pregnancy toxemia and is usually complicated by concurrent hypocalcemia, hypomagnesemia, and in some cases hypoglycemia. Diets that are marginal in P are generally indicative of diets that are marginal in energy, because grains are usually very good sources of P.
At the onset of lactation in the dairy cow, the production of colostrum and milk draws large amounts of P out of the extracellular P pools. This alone often causes an acute decline in plasma P levels. In addition, if the animal is also developing hypocalcemia, PTH will be secreted in large amounts, increasing urinary and salivary loss of P. In dairy cows, plasma P concentrations routinely fall below the normal range at parturition, and in cows with milk fever plasma P concentrations are often 1 to 2 mg/dL. Plasma P concentrations usually increase rapidly after treatment of the hypocalcemic cow with intravenous Ca solutions. This rapid recovery is caused by reduction in PTH secretion, which reduces urinary and salivary loss of P. Administration of Ca generally causes resumption of gastrointestinal motility, which allows absorption of dietary P and reabsorption of salivary P secretions that were sequestered within the rumen.
Some dairy cows that develop acute hypophosphatemia do not spontaneously recover normal plasma P concentration. This is sometimes the case in cows that are classified as downer cows. This syndrome often begins as milk fever, but unlike the typical milk fever cow, in these cows the plasma P remains low (<1 mg/dL) despite successful treatment of the hypocalcemia. Protracted hypophosphatemia in these cows appears to be an important factor in the inability of these animals to rise to their feet, but why plasma P remains low is unclear. In some cases the inability to absorb the salivary phosphate is secondary to poor ruminal motility, but not in all cases. Excessive cortisol secretion could also drive blood P concentration down, probably by forcing extracellular P inside cells. Treatment of cows with phosphate-containing solutions can effect a recovery in some animals. For oral treatment, the dose is 50 g of P supplied in a 200-g monosodium phosphate drench. Intravenous treatment consists of 6 g of P supplied by 23 g of monosodium phosphate dissolved in 1 L of saline. Oral treatment restores normal blood Pa a little more slowly than intravenous treatment, but the effect lasts much longer.367 If depletion of intracellular P stores is also involved in the downer syndrome, it seems likely that intravenous treatment alone simply does not supply enough P to replenish intracellular stores of P.
The hypophosphatemic downer cow syndrome does not appear to be caused by low P diets, as affected cows are often receiving diets containing 0.4% dietary P. The best preventative measure seems to be to avoid development of hypocalcemia.
Chronic Phosphorus Deficiency
P deficiency is fairly common in grazing ruminants. Plant growth in arid climates or on tropical soils is often limited by poor soil P content or availability. Although P content of the immature plant may be suitable (0.3% P on a DM basis) the mature dry plants often contain less than 0.15% P, which can lead to P deficiency in ruminants forced to subsist on these forages. With the exception of rickets and postparturient hemoglobinuria, the clinical problems associated with P deficiency are general and nonspecific. They can include an unthrifty appearance, reduced feed intake, pica, reduced rate of gain, and reduced milk production.
OCCURRENCE
The most common of the P deficiency syndromes is the unthrifty cow that grows slowly, milks poorly, and is infertile. In arid areas of the world with inferior soil quality, infertility and poor growth resulting from inadequate P intake affects nearly all animals. Beef animals grown in these areas may routinely require 3 or more years to reach market weight. Brood cows often calve only every other year. In more temperate areas, P deficiency can develop in animals grazing overly mature forages or crop residues, such as corn stalks. Cows subsisting on forages that are low in digestibility are at risk for developing P deficiency unless supplemented. Increasing awareness of the environmental problems associated with P content of animal wastes and increasing expense of P supplements may encourage producers to feed diets marginally sufficient in P.
Sheep can, in some instances, be successfully raised on pasture that has been associated with P deficiency in cattle, suggesting that sheep are slightly more resistant to P deficiency syndromes than are cattle. This may simply reflect the higher intake per kilogram of body weight of sheep and their habit of selecting the less mature plants, which are generally higher in P.
Rickets and Osteomalacia
Rickets is a disease of young, growing animals in which the cartilaginous matrix at the growth plate and the osteoid matrix formed during bone remodeling fail to mineralize. In adults (no active growth plates), the term osteomalacia is used to describe the failure of osteoid matrix to mineralize. Ca and phosphate ions come together in a ratio of 10 Ca ions to 6 phosphate ions at the point of mineralization of the bone cartilage or osteoid matrix. Bone ash content varies somewhat with the bone examined but is generally 57% to 62% of bone dry weight, with 36% Ca and 17% P in adult animals. Failure to supply P in the diet will result in low plasma P concentrations that will not support the mineralization process, and the bone matrices will fail to mineralize. Bone ash, as well as total P and Ca content, will be below normal.
Low plasma Ca concentrations (arising from vitamin D deficiency or severe Ca deficiency) can also result in failure to mineralize bone matrices. Bone ash is reduced, and the bones of young animals become “rubbery,” bending without breaking. Joint surfaces are often eroded, contributing to lameness in affected animals.
In principle, Ca deficiency differs from P deficiency in that normal osteoid is formed but fails to mineralize in P deficiency, whereas in Ca deficiency the normal osteoid is either not formed at all (osteoporosis) or replaced by fibrous tissue. In vitamin D deficiency it is common to see mixed lesions (osteomalacia, osteoporosis, and fibrous osteodystrophy) all in the same bone. Vitamin D deficiency also seems to reduce secretion of type X collagen by the chondrocytes within the growth plate and somehow prevents the programmed cell death of chondrocytes within the zone of provisional calcification, a requirement for bone formation.
Chronic Moderate Hypophosphatemia
Animals fed diets containing less P than necessary to meet physiologic needs will develop hypophosphatemia and all the physiologic consequences of failure to grow, inappetence, and unthriftiness. Milk production, but not P content, will decline. Impaired reproduction has often been attributed to “P deficiency.” However, in most cases in which cows develop P deficiency the situation is complicated by concurrent energy deficiency, likely the direct cause of the reproductive failure.
Unfortunately the belief that “marginal” dietary P contributes to reproductive inefficiency has been used as justification for feeding diets that are much higher in P than is required. Wu and Satter368 present convincing evidence that high-producing cows perform well in terms of milk production and fertility when fed diets containing 0.37% to 0.40% P.
Postparturient Hemoglobinuria
Intravascular hemolysis, anemia, and hemoglobinuria are occasionally reported during the first 6 weeks of lactation. Cows that have been treated for ketosis seem at greater risk for developing postparturient hemoglobinuria. Many, but not all, cows that develop this syndrome are hypophosphatemic. Severe hypophosphatemia is postulated to depress the ability of erythrocytes to produce ATP> Glyceraldehyde-3-phosphate dehydrogenase, a key enzyme in glycolysis, requires inorganic phosphate as a cofactor. Without sufficient ATP to power sodium pumps, the intracellular sodium concentration rises, the cells become more rigid, and as a result they rupture as they pass through the capillary beds. However, hypophosphatemia alone is rarely sufficient for increased red blood cell fragility. Often these cows are on diets that are also deficient in selenium, copper, and energy. In light of these observations, it is likely naive to suggest that the cause of postparturient hemolysis is hypophosphatemia alone (Table 41-9).
HYPOKALEMIA SYNDROME IN CATTLE
Based on reports available in the literature, hypokalemia syndrome in cattle can be defined as presence of flaccid paralysis, recumbency, abnormal neck position, and serum potassium concentration below 2.5 mmol/L.
Occurrence
Hypokalemia is a frequent biochemical finding in cows presented with anorexia and gastrointestinal stasis.369 It is often mild (>3 mmol/L) and not associated with obvious clinical signs in ruminants. Hypokalemia syndrome is a rare clinical condition reported in a total of 25 animals in three studies.370-372 Lactating dairy cows appear to be at a higher risk, but the syndrome has also been described in calves and heifers.
History
Lactating dairy cows less than 60 days in milk (DIM) constitute the majority of cases in the literature. Any disease causing significant anorexia for 1 week or more, particularly infectious disease, is a risk factor.371 Repeated administration of isoflupredone acetate is reported in most cases, for treatment of either acetonemia in lactating cows370 or infectious respiratory disease in calves and heifers.371 In one case, repeated off-label intramammary administration of isoflupredone acetate preceded hypokalemia syndrome. Multiple doses of dextrose and insulin are also reported to be risk factors.370 Intravenous fluid administration for 2 days or longer without serum biochemistry followup has been documented in two cases of calves with severe neonatal diarrhea.
In most cases recumbency caused by hypokalemia syndrome was not the initial event but rather occurred during the evolution of another disease such as acetonemia or infectious disease. Moreover, recumbency was not always the presenting complaint.371 Some animals first manifested anorexia, reduced fecal output, kyphosis, and reluctance to move. However, evolution to severe paresis and decubitus occurred rapidly in all these cases.
Clinical Signs
In the early stages of the syndrome, absence of feces, paretic gait, kyphosis, inability to stand for a long time, and tachycardia are observed. Sometimes an abnormal neck posture is evident before recumbency. The typical case presentation of hypokalemia syndrome includes decubitus, S-shaped neck, abnormal feces and ruminal motility, abnormal appetite, and tachycardia with or without arrhythmia. Decubitus is a result of flaccid paralysis: the tail has little to no tonus and is held in an abnormal position, resistance to all manipulations is weak, and the cow is unable to raise her head. It is not rare to observe that the animal eats and drinks willingly if food and water are provided near the mouth. Abnormal appetite therefore may be partly caused by an inability to move the head to reach food and water. The neck is carried in an S-shaped and twisted fashion that differs from that of the hypocalcemic cow. The neck is never fixed in this abnormal position. When moved there is no resistance, and if the head is placed in an anatomically correct position there is an odd feeling of luxation. As soon as the head is not held, the neck resumes the abnormal S-shaped position, deviating to either direction. This neck posture may precede decubitus and persist after decubitus has resolved. Cardiac arrhythmias (Fig. 41-20) also accompany hypokalemia syndrome and may be ventricular (ventricular tachycardia, accelerated escape ventricular rhythm) or supraventricular (atrial fibrillation) in origin.
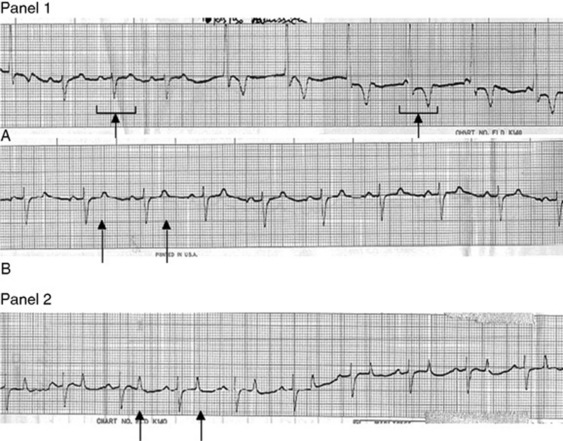
Fig. 41-20 Electrocardiograms. Panel 1, Electrocardiograms of a cow with hypokalemia syndrome before treatment (base-apex, 25 mm/sec, 1 cm = 1 mV). Potassium serum concentration was 2 mmol/L. A, Note the ventricular escape rhythm: normal heartbeat and more than five ventricular QRS complexes. B, Note the flattened T waves and increased intervals. Panel 2, Electrocardiogram of a cow with hypokalemia syndrome after treatment (base-apex, 25 mm/sec, 1 cm = 1 mV). Potassium serum concentration was 4 mmol/L. Note the normal T waves and intervals.
Hypokalemia syndrome is most often the consequence of a concomitant disease or treatments administered. Fever and acetonemia are the most frequently associated problems. The identification of the concomitant disease(s) is essential for accurate prognosis and treatment.
Etiology
Hypokalemia may result from an imbalance of the internal or external potassium balance or both (Fig. 41-21).369,370,373-376 Internal potassium balance is determined by exchanges between extracellular and intracellular compartments, whereas external balance is a result of systemic intake and losses. Currently, except for cases with documented repeated isoflupredone acetate administrations, the exact determinants causing hypokalemia syndrome in cows are not known.
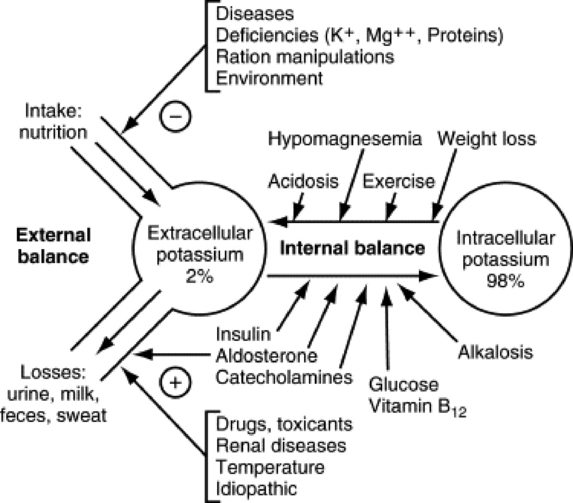
Fig. 41-21 Potassium balances and major determinants in cattle.
Adapted from Brobst D: J Am Vet Med Assoc 188:1019, 1986.
At present, because of the limited data available, it is not possible to determine if hypokalemia syndrome cases are mainly a result of potassium depletion, internal imbalance, or both. The prolonged inappetence observed before hypokalemia, along with the amount and duration of potassium needed for treatment, suggests potassium depletion is significant. Serum potassium concentration is a poor indicator of the potassium status of the organism because 98% of whole body potassium is located in the intracellular compartment.374,377 Determination of intracellular potassium concentration in erythrocytes or muscle cells could be a more accurate way to assess potassium depletion.377,378 At present these data are not available for hypokalemia syndrome cases.
As a result of the potent mineralocorticoid activity of isoflupredone acetate, cases associated with its administration show both internal and external potassium balance abnormalities (see Fig. 41-21).376 Repeated doses can reduce blood potassium concentration by 70%.379 In a small experimental study (N = 9), we were able to reproduce the syndrome by combining food restriction and multiple doses of isoflupredone acetate in lactating dairy cows.380 The use of this steroid could also complicate the treatment of the hypokalemia syndrome by increasing the renal losses of potassium. Based on current knowledge, use of isoflupredone acetate in the anorectic, acetonemic cow that has received dextrose, with or without insulin, should be restricted to one administration at the labeled dosage. Hypokalemia syndrome has also been reported with dexamethasone administration, although it is a less potent mineralocorticoid.372
In noniatrogenic cases the relationship between protracted disease and severe hypokalemia may be explained by a decrease in potassium intake (anorexia) with increased release of catecholamines caused by stress (see Fig. 41-21). Alternatively, renal potassium adaptation381 may be responsible for the potassium loss. Animals chronically fed a high-potassium diet are able to survive the high potassium load due to increased renal losses. These increased losses persist when potassium intake is suddenly reduced. Renal adaptation (reduction of urinary potassium excretion) takes 24 to 48 hours to occur, thus creating a potassium depletion.381 Because of a forage-based diet, lactating dairy cows may eat more than 10 times their daily potassium requirement, causing high urinary potassium excretion.371 Therefore these cows appear to be at risk for potassium depletion because of delayed renal adaptation when potassium intake is abruptly reduced. This model cannot explain why some cows develop the syndrome when exposed to these conditions but others do not.
Other factors known to promote significant hypokalemia have not been documented in cows with hypokalemia syndrome, such as anomaly of the aldosterone system, diuretic (particularly furosemide) abuse, nephropathy (proximal or distal tubular renal acidosis), or severe diarrhea. Diarrhea with protracted reduced potassium intake, increased losses, and internal potassium imbalance resulting from acidosis has been associated with hypokalemia syndrome in calves.371 Error in fluid administration causing increased urinary losses of potassium along with return to a normal internal potassium balance can further exacerbate hypokalemia in diarrheic calves.
Pathophysiology
Potassium is the most important intracellular cation.382 The gradient between the potassium concentrations of the intracellular and extracellular compartments is the determinant of the resting cellular membrane potential and plays a role in the formation and transmission of action potentials.383 Therefore, clinical signs reported in cattle and other species with severe hypokalemia are multisystemic. Moreover, structural consequences in the myocardium373 and the muscle cells have been reported in cattle.370,371
Dysrhythmia and electrocardiographic changes were present in 50% of cases in one study.371 The arrhythmogenic electrical activity changes implicated in hypokalemia include the following: hyperpolarization of the cardiac cell resulting in spontaneous automatic activity, slow conduction caused by increased difference between resting membrane potential and threshold potential, increased action potential duration as a result of slow repolarization, depressed fast responses because of higher membrane potential when a slow repolarizing cell is stimulated, slow responses in fibers normally exhibiting fast responses, and conduction block.384 These electrical events alone or in combination with structural changes such as necrosis can induce the atrial and ventricular arrhythmia observed.
Pathophysiology of cardiac and muscle necrosis caused by potassium depletion includes (1) ischemia caused by lack of vasodilatation during muscle cell contraction, resulting from absence of potassium release into the extracellular space, and (2) glycogen deficiency resulting from impaired glycogen synthesis.376,385 Muscle lesions compatible with hypokalemic myopathy have not been consistently documented.370-372 It is presently not known if the presence of hypokalemic myopathy is necessary to the occurrence of the clinical signs observed in the bovine syndrome or if it is a prognostic determinant.370,371
Diagnosis and Ancillary Test Results
Initial presentation of hypokalemia syndrome (intestinal ileus, abnormal gait) could mimic an intestinal syndrome. History may allow distinction between these conditions. Hypokalemic syndrome often follows or is concomitant with a long disease, in contrast with the usual acute evolution of an intestinal syndrome. According to the results of a small experimental study,380 potassium fractional urinary excretion (FEK) could be a useful tool in the medical management of a cow suspected to be developing bovine hypokalemia syndrome. In this study, FEK was abnormally high in the presence of severe hypokalemia 12 to 24 hours before manifestation of clinical signs.
According to our experience the clinical signs of a twisted, S-shaped neck associated with flaccid paralysis as described earlier are almost pathognomonic. Other conditions that could be included in the differential diagnosis are hypocalcemia, botulism, tick paralysis, and myelopathy (cervical spinal trauma, cervical osteomyelitis). Conditions like listeriosis, vertebral malformations, luxations, fractures, and torticollis can be associated with abnormal neck position but not flaccid paralysis and absence of pain during manipulations. Hypokalemia syndrome, even if rare, should be included in the differential diagnosis of hypocalcemia, particularly when response to calcium administration is not normal.
Confirmation of the clinical diagnosis requires serum biochemical analysis. To date, all published cases of bovine hypokalemia syndrome have been associated with serum K+ concentrations less than 2.5 mmol/L. Aside from hypokalemia, moderately elevated AST and creatine kinase (CK) activity (median value of 171 and 1564 IU/L respectively371), severe metabolic hypochloremic alkalosis (median value [TCO2] = 41.5 mmol/L, [Cl−] = 86.9 mmol/L371), and mild hyperglycemia (median [glucose] = 6.05 mmol/L371) are observed in the majority of published cases.
Lumbar muscle biopsy (between L2 and L5) is a way to differentiate primary hypokalemic myopathy from secondary myopathy caused by decubitus. Determination of the serum creatine kinase MB isoenzyme (CK-MB) activity could help to detect myocardium damages. Detection of myopathy in epaxial muscles and/or myocardium at necropsy is not diagnostic if potassium depletion or hypokalemia is not documented.
Some authors have suggested that electrocardiographic changes could be used to detect a potassium-depleted status, irrespective of the serum potassium concentration.386 A method of determining both the internal and external potassium balances simultaneously in dairy cows has been presented.378 In this study, ingested potassium, milk potassium concentration (LK), and FEK defined the potassium external balance. Serum potassium concentration (SK) and red blood cell potassium concentration (CIEK) defined the potassium internal balance. Significant variations were detected throughout the day for CIEK, LK, and FEK.378 Therefore, in order to monitor potassium balances over time, samples should be collected at the same time each day.
Treatment
Nursing care is of primary importance to prevent complications such as mastitis or myopathy caused by prolonged decubitus; to maintain alimentation by providing food and water near the mouth; and to provide favorable conditions for the cow to get up. The animal should be kept on clean and atraumatic bedding such as pasture, sand, or accumulated litter. It must be regularly milked and turned from one side to the other at least every 8 hours. Use of a flotation tank (Aquacow Rise System) is of great value once the serum potassium concentration has returned to normal range.
All other identified problems must be addressed, including dehydration, acetonemia, and infectious disease. Treatments for arrhythmias, other than potassium, were not given in the reported cases.
Specific treatment is mainly per os potassium chloride supplementation. Based on the published data and our experience, we recommend a total (per os and intravenous) KCl dose per day of 50 g/100 kg of body weight of cow. Intravenous administration should be reserved for dehydrated cows, as overhydration may worsen hypokalemia by increasing potassium renal losses. When potassium chloride is given IV, the rate should not exceed 0.5 mEq of K+/kg/hour, to avoid high risk of cardiac toxicity. Serum potassium should be monitored twice daily to allow adjustment of the treatment regimen. Potassium supplementation is needed for an average of 5 days.371
The abnormal neck position can be the first and last clinical sign in the evolution of the hypokalemia syndrome. Other clinical signs usually disappear on average in 3 days, the same day potassium concentration typically returns to normal. Some degree of paresis may persist for 2 to 3 days after return to normokalemia. We recommend continued potassium supplementation for 1 or 2 days after the return to normal clinical state and appetite. Increased renal potassium losses are promoted by the high quantity of potassium given during the treatment. In our experience it is important to decrease potassium supplementation slowly to avoid potassium depletion caused by delayed renal adaptation to the lower potassium diet. For example, 25 g of KCl per 100 kg of cow may be given the day after return to normokalemia, then 12.5 g of KCl per 100 kg of cow for 2 more days.
Because potassium depletion is a primary cause of myopathy, it is important not to try to raise the cow before hypokalemia has resolved. This emphasizes the importance of good nursing care and regular decubitus side changes. Once serum potassium concentration reaches the normal range, a water flotation tank is the best system to manage the recumbency.
Prognosis
The survival rate was very different in the two series of cases reported: 2 of 8370 versus 11 of 14.371 The prognosis may be affected by the age, concomitant disease, initial potassium depletion degree, presence and severity of hypokalemic myopathy, or treatments received.369 The quality of the supportive care is probably a major determinant of the outcome, as for any downer cow. In the field, because of time and energy needed from the owner, our experience warrants a guarded prognosis. The availability of a flotation tank appears a significant positive prognostic factor.371,372