Chapter 45 Principles of Antimicrobial Therapy
PRINCIPLES OF ANTIMICROBIAL THERAPY
“Rational therapeutics” is the scientific account of the management and care of a patient for the purpose of combating a disease or a disorder based on knowledge of the disease and the action of the remedies used. It requires clinical judgment, overall medical knowledge, information about a specific patient,1 selection of the proper drug, and the formulation of a dosage regimen appropriate to the patient after appraisal of potential benefits and risks of that therapy.2 That process is not simplified by the vast number of antimicrobial drugs available. It is therefore essential that basic principles be applied so that antimicrobial drugs may be appropriately selected and used in patients. The purpose of this chapter is to explain general principles of rational antimicrobial therapy (Box 45-1) rather than to discuss individualized treatment of specific infectious conditions. Guidelines for prudent use of antimicrobial drugs have been published by several professional associations and organizations. Guidelines are by nature inherently dynamic and can be general or very specific, but in either case they should follow a principled approach.3
The ultimate expectation of an antimicrobial drug is to inflict an insult on an infectious organism that is sufficient to kill the organism or to render it susceptible to lethal effects of natural host defenses or the microenvironment around it, without adversely affecting the patient.4,5 To accomplish this goal, antimicrobial drugs must be selectively toxic to the infectious organism.6,7 Different structures, biochemical activity, virulence factors, mechanisms of resistance, generation times, and nutritional requirements of infectious organisms (i.e., bacteria, viruses, fungi, and parasites) form the mechanistic basis for selective toxicity of antimicrobial drugs. No individual drug is sufficient to meet all therapeutic needs. It is irrational to treat a viral infection with antibacterial drugs or a bacterial infection with antiparasitic drugs. However, principles of selection and use of antimicrobial drugs are similar regardless of the infectious agent.
PRINCIPLE 1: CONSIDER THE PATIENT
The patient occupies the paramount position with regard to treatment and recovery from infection. Although the organism is the target of the drug’s action, it is the patient with an infection that receives the drug. Before the patient can be considered adequately recovered, infectious agents must be controlled and removed from the site of infection, debris must be removed from the site, and the damaged tissue must be repaired or replaced.8 Inactivation of microbes is the only role played by antimicrobial drugs; the patient must participate in that aspect and perform the others. Most infections are prevented by efficient nonspecific host defense mechanisms.9 When infections occur, many patients recover without treatment because of nonspecific and specific host defense mechanisms. Ideally, if the host’s defenses are functional and a pathogen’s virulence can be weakened by antimicrobial drugs, the host should be able to kill and remove the microbe.
Many host-related factors must be considered during the formulation of a therapeutic plan.10,11 The animal’s age, sex, breed, use, residence, contacts with other animals, travel, diet, exposure to inclement weather, vaccination status, and medical experiences may influence natural defenses in the animal and prevalence of certain infections. Prevalence of an infectious disease may vary with year, susceptible population, and geography. Medical history of the herd may also be of value because virulence of some infectious agents may change after “passage” through several animals.
Clinical signs must be interpreted carefully because identification of the agent is frequently not possible on the basis of clinical signs alone.10 Clinical manifestations of infection result from direct effects of microbial pathogens, their toxins, or the inflammatory response elicited in the host. Similar clinical signs may be caused by different microbial agents, and one species of microbe may produce a variety of clinical signs.12 Signs of noninfectious conditions can mimic those of infectious conditions.
Inadequacies in host defense mechanisms are as important as the organism’s virulence in determining whether infection will result from contact with an infectious agent and whether recovery will result if infection develops.9 This is exemplified by infections associated with combined immunodeficiency syndrome or failure of passive transfer. It is irrational to expect antimicrobial drugs to resolve those conditions because the primary problem is not an “antimicrobial deficiency.” Reasonable attempts should be made to assess weaknesses in a host’s defenses that may contribute to the disease. Natural defenses of note include skin, mucous membranes, mucociliary escalator activity, cough reflex, transit time through the gastrointestinal tract, blood supply at portals of entry of infectious agents, humoral and cellular immunity, and resident microflora.13 It is not possible to assess many of these objectively in most clinical situations. The attending veterinarian must have astute clinical acumen to subjectively evaluate contributory factors. Attempts should be made to replenish temporarily inadequate host defenses. If the host’s defensive response contributes to clinical signs (e.g., if inflammatory edema compromises breathing), the response should be modified to enhance convalescence.
Antimicrobial drugs can alter the response of the host’s immune system or the stimulatory antigen.14 The outcome of complex interactions among antimicrobial drugs, bacteria, and phagocytic cells depends on the organism, the drug, its concentration, and the duration and timing of exposure to the phagocyte and microbe.15 Antimicrobial therapy of proven efficacy should not be withheld from a patient to potentially enhance that animal’s immunity. Effects of antimicrobial drugs on chemotaxis, phagocytosis, metabolism of neutrophils, and the complement system are demonstrable in vitro but have inconclusive or insignificant clinical importance.14 Inhibitory effects of chloramphenicol or rifampin on cellular and humoral immunity have been studied most extensively, but their immunomodulatory effects are not sufficient to warrant restricting their clinical use. Clinical consequences of effects of antimicrobial drugs on immunity are limited but should be conservatively considered. For example, florfenicol did not interfere with responses of cattle vaccinated against bovine herpesvirus type 1.16 Tilmicosin had neutrophilic antiinflammatory activity in cattle that were experimentally infected with Mannheimia (Pasteurella) hemolytica.17 Some antimicrobial drugs accumulate in phagocytes, but increased bactericidal activity is not equally correlated to the degree of that accumulation; likewise, no relationship has been demonstrated to clinical response.18,19
Special Considerations
MATURATIONAL STATUS
An animal’s responses to pharmacologic agents are significantly influenced by the age or maturity of the animal.10,20,21 Physiologic differences that alter disposition of drugs in vivo are primarily responsible for that. Pharmacologic effects of xenobiotics (chemicals that are foreign to the biologic system) administered to brood animals before conception, during gestation, or at parturition or to neonates are not universally predictable. Drugs may distribute into gonads at sufficient concentrations to adversely affect gametogenesis. Drugs in luminal fluids of the oviducts or uterus may be teratogenic. Distribution of drugs into the placenta and fetus is affected by factors such as placental circulation, placental maturation, placental and fetal biotransformation of drugs, and fetal circulation. Among these variables, species-related differences may occur. Therefore drugs should be administered conservatively to brood animals, pregnant animals, and neonates and with adequate explanation of pertinent facts to the client. Potentiated sulfonamides administered to gestating mares for treatment of equine protozoal myelitis were shown by Fenger and colleagues to be associated with increased incidence of anemia, fever, anorexia, depression, and abortion.22 Stallions treated with potentiated sulfonamides and another diaminopyrimidine (pyrimethamine) were shown to develop signs of neuromuscular weakness and ejaculatory dysfunction.23 It is not clear how each of these signs could be attributed to these drugs. Indirect effects such as folate deficiency may account for some of the adverse signs that have been observed. Some adverse findings with one species should be interpreted with care when dealing with another species. Anemia is a good example. An aspirate or biopsy of bone marrow is necessary to adequately evaluate anemia in horses, whereas peripheral blood may be sufficient with other species.
Biotransformation of drugs in neonates can be altered by administration of medication to the gestating dam. Phenylbutazone administered to gestating mares during the final days of gestation resulted in substantial amounts of phenylbutazone and of oxyphenylbutazone in plasma of their foals.24 Although phenylbutazone is not an antimicrobial drug and results of similar studies with antimicrobial drugs are not known to us, this example serves to emphasize the importance of considering peripheral effects of medication administered to gestating animals.
Fluoroquinolones are known to cause cartilaginous arthropathies in weight-bearing joints of young animals of several species. The horse may be unique in that adult horses have also developed the same syndrome, which was inconsistently related to dose and duration of treatment with enrofloxacin.25,26 Tissues peripheral to plasma contained higher concentrations of fluoroquinolones than did plasma; therefore accumulation from repeated dosage regimens should be considered when developing those regimens.25,27-29 Although arthropathic syndrome has now been recognized and acknowledged as a risk associated with use of fluoroquinolones, nothing is known about the healing and recovery of joints that are injured. Therefore the overall assessment of risks of treatment with those drugs remains speculative; anecdotal reports suggest that the injured cartilage may repair and not carry prolonged clinical consequences, but that needs to be confirmed.
LACTATION
There are at least three aspects to consider regarding distribution of drugs into milk: the lactating animal, the suckling animal, and the milk-consuming public. Distribution of drugs into milk is influenced by the same factors that influence distribution of drugs into other tissues.10,21,30 Some antimicrobial drugs are concentrated in milk, others equilibrate between milk and circulating blood, and still others reach insignificant concentrations in milk (Table 45-1).31-39 A ratio greater than 1 indicates accumulation of the drug in milk relative to plasma. Results often vary with pH of milk. Concentrations of tetracycline may be high as a result of chelation with calcium but remain bioinactive.
Table 45-1 Ratio of the Concentration of Drug in Ultrafiltrate of Milk to That in Plasma After Systemic Administration of the Drug31-39
| Drug | Ratio (Range) (Milk:Plasma) | Reference |
|---|---|---|
| Ampicillin | 0.24–0.30 | 28 |
| Benzyl penicillin | 0.13–0.26 | 28 |
| Cephaloridine | 0.24–0.28 | 28 |
| Erythromycin | 6.00–7.30 | 29 |
| Gentamicin | 0.20–0.50 | 30 |
| Kanamycin | 0.60–0.80 | 30 |
| Lincomycin | 2.50–6.25 | 31 |
| Spectinomycin | 0.37–1.12 | 32 |
| Sulfacetamide | 0.08–0.11 | 33 |
| Sulfadiazine | 0.16–0.19 | 33 |
| Sulfadimethoxine | 0.13–0.24 | 34 |
| Sulfadimidine | 0.59–0.62 | 33 |
| Sulfanilamide | 0.97–1.04 | 33 |
| Sulfathiazole | 0.37 | 33 |
| Tetracycline | 1.22–1.91 | 35 |
| Trimethoprim | 3.00–4.90 | 36 |
| Tylosin | 1.00–5.35 | 32 |
Therapeutic implications of the distribution of drugs into milk are clear, but inflammation of the mammary gland may alter distribution of drugs. Mastitic milk may resemble an exudate rather than “milk,” thus providing a different microenvironment from that of milk. Nearly all drugs administered to a lactating animal will be detectable in her milk and can thereby expose the suckling animal. However, the total amount of most drugs received by the suckling animal via milk does not pose significant concerns.20
Antimicrobial drugs in milk may render that milk unsuitable for use as food.30 Subtherapeutic concentrations of drugs in milk may cause no adverse effect, may cause the milk to be condemned as adulterated, may interfere with production of cheese or other dairy products, or may induce reactions in an allergic person who consumes the milk. For these reasons, compliance with and adherence to withdrawal times should be considered seriously.
WITHDRAWAL TIMES
Withdrawal times are intended to allow adequate time for elimination of the drug from the animal to reduce the risk of violative residues of drugs inadvertently entering the food supply. Withdrawal times are established for drugs that are licensed for use in animals intended for use as food, and the withdrawal time applies only when the drug is used according to the approved labeling.13 Because of this, it is important before treatment is initiated to discuss the patient’s condition with the client, to determine if the animal (regardless of species) is intended for use as food, and to ascertain if extralabel use of the drug is needed. Without such a discussion the client cannot make a wise decision regarding the risks and benefits of therapy, and precautions to prevent adulteration of food may be inadvertently omitted.
Withdrawal times are not established for drugs that are used in an extralabel manner; therefore the most conservative time should be allowed when drugs are used in a manner inconsistent with their labeling.40,41 Aids to determine useful withdrawal times include screening tests for drugs in milk* or urine.† Results may not match those of methods used by regulatory agencies assigned the task of detection of residues, and those agencies may test different tissues.
EXTRALABEL USE OF DRUGS
Veterinarians often encounter infectious conditions for which the drug of choice is not approved by the U.S. Food and Drug Administration’s Center for Veterinary Medicine (FDA-CVM).13 In these situations the veterinarian is not exempt from professional and ethical obligations. Treatment must be based on experience, established practices, and scientifically substantiated facts. Drugs that are used in any manner other than that described on the drug’s labeling (e.g., the treatment of diseases, with doses, routes of administration, duration of treatment, or species not specified on the approved labeling) are said to be used in an extralabel manner. The Animal Medicinal Drug Use Clarification Act (AMDUCA)40 of 1994 amended the Federal Food, Drug, and Cosmetic Act so that a particular use or intended use of a drug shall not be deemed unsafe if such use or intended use is (1) by or on the lawful written or oral order of a licensed veterinarian within the context of a veterinarian-client-patient relationship, and (2) in compliance with regulations promulgated by the Secretary of the U.S. Department of Health and Human Services. In short, the amended act contains statutory allowance of extralabel use of drugs, and regulatory authority is retained by the FDA-CVM. For veterinarians to discharge their professional duties within the public trust and in an ethical manner, a professional approach to extralabel use of drugs is necessary. Some guidelines include the following:
Regulations specify contents of acceptable labeling. Consultation with a representative of the FDA-CVM is advised before treatment is initiated when particularly perplexing therapeutic problems exist and no alternatives to extralabel use of drugs are apparent.
HERD TREATMENT
Each member of a herd may have the same disorder, but one animal cannot receive treatment that is intended for another (there are no surrogate patients), and treatment of an animal that does not need it is wasted treatment. Treatment must be implemented for each affected animal in the herd. The method of treatment must be practical and efficacious, or compliance by the client will be difficult. Unfortunately, requests by some owners and veterinarians for “herd treatment” are really requests to treat each animal identically and easily rather than according to its individual needs. Each animal may indeed require the same treatment, and treating all animals in question may be acceptable, but each animal deserves a reasonable assessment of its condition before treatment is instituted. Metaphylactic or therapeutic medication of all animals has benefits and risks, but the cost must be offset by savings in areas such as labor of handling of the animals, reduced needs for therapeutic medication, or improved production.45,46
COST OF THERAPY
The cost of appropriate therapy is an important consideration for the client. The client (or his or her financial advisor) is the only one who can decide whether the value of treatment is “worth” the “cost.” The cost of medical care is not static, and the veterinarian should keep the client informed of the financial commitment associated with treatment. Naturally, cost-effective therapy should be sought. This is not to say that the least expensive therapy is the best therapy or that ineffective drugs should be used just because they are inexpensive. Similarly, expensive drugs are not “better” just because they cost more. Clients require adequate information to make financial and medical decisions regarding their animals.
PRINCIPLE 2: DOCUMENT THE INFECTION
Antimicrobial therapy is predicated on the premise that the disease is caused by an infectious agent and that the patient will be unable to effectively eliminate the infection without antimicrobial treatment.1,4 The primary purpose of documentation of infection is to help the veterinarian determine the necessity of this treatment. Clinical experience and diagnostic and interpretive skills are particularly important. Use of antimicrobial drugs for relatively trivial infections exerts selective pressure for resistant organisms.5 Contamination or colonization by microorganisms (i.e., contamination of the surface of granulation tissue) does not necessarily constitute infection and is not always a sufficient reason for antimicrobial treatment. Inoculum size, microbial virulence, concurrent infection, site of infection, and resident flora contribute to the significance of infection. Death resulting from some bacterial diseases such as botulism (except toxicoinfectious botulism), enteric colibacillosis, or enterocolitis caused by salmonellae is not directly caused by vegetative bacterial invasion, so antimicrobial drugs are a secondary component of treatment. Without evidence of involvement of a susceptible causative agent(s), use of an antimicrobial drug is irrational and exposes the patient to unnecessary risks.1 The more information that can be discerned about the infecting agent, the more reasonable will be the treatment.1,11
Sequential steps necessary to document an infection are as follows1,47:
There are nonspecific and specific methods of documenting infection.10 Nonspecific methods include medical history, clinical signs, hematologic changes, and characteristics of lesions. The drug(s) used for initial therapy will most often be selected on the basis of the veterinarian’s clinical judgment and medical knowledge, as well as on nonspecific indications of infection.2,11,47 This is not a trial-and-error approach.48 Potential for a particular infectious cause, most probable causative agent, status of the patient’s natural defense mechanisms, and site of infection must be considered when interpreting clinical signs.49 “Response to treatment” is an inconsistent means of revealing the cause of infection. Although nonspecific methods are the weakest form of documentation, that information, coupled with astute clinical judgment, is often successfully used in practice. For example, a young horse with fever, nasal discharge, and submandibular lymphadenitis can be assumed (with a relatively high degree of certainty) to have strangles, and penicillin could be chosen for therapy. However, the possibilities for misdiagnosis are numerous in this and many other disease conditions, and specific methods of documenting infection should be used. An aspirate obtained from involved lymph nodes of the patient in the example may reveal chains of gram-positive cocci, and Streptococcus zooepidemicus may be isolated. The immediate treatment may not change, but the management of the rest of the animals in the herd would differ greatly from that indicated for strangles.
Different organisms may be susceptible to and successfully treated with one antimicrobial drug, making treatment choice easy. However, a specific causative diagnosis may be of considerable relevance for the development of herd-health programs.
Antimicrobial therapy cannot be consistently successful if formulated on the basis of nonspecific diagnostic methods and historical probabilities alone.10 Treatment formulated in that manner may cause more harm than good by interfering with the pursuit of a specific diagnosis, by allowing the development of superinfection, or by inducing reactions to the drugs.
Specific diagnostic methods should be attempted before initial treatment is instituted. Specific methods of documenting infection require proper collection and submission of appropriate samples, reliable laboratory procedures, and accurate interpretation of results. Because laboratory facilities and personnel can work only with materials submitted to them, the veterinarian must properly collect and submit appropriate samples to the laboratory and interpret results relative to the condition of the individual patient. Procurement of tissues or bodily fluids for cytologic, histologic, or microbiologic evaluation remains the cornerstone of accurate documentation of a specific infection.10 Inadequate or improper sampling and improper submission of samples to the diagnostic laboratory are the most frequent and often unrecognized reasons for failure of documentation of causative agents of infectious diseases. The sample must be representative of the site of infection and may be bodily fluid (blood, peritoneal fluid, pleural fluid, percutaneous transtracheal aspirate, material draining from a site of infection) or tissue (biopsy, scraping, curettage, aspirate). Collection and submission procedures differ for aerobic and anaerobic bacteria, mycoplasma, protoplasts, viruses, and parasites. Special media or particular constraints of time and temperature must often be observed to transport fastidious organisms. Because each laboratory may use different techniques, it is wise to contact laboratory personnel in advance to learn preferred collection and submission equipment and procedures. The veterinarian and laboratory personnel can assist each other in reducing inappropriate collection and submission practices.
The choice of site to be sampled is critical. Isolates from draining tracts are unreliable and should be evaluated in the context of other related information.50 Organisms isolated from wounds are of questionable significance unless they are present in pure culture or are clearly predominant. In these instances susceptibility testing is probably warranted. Any microorganism isolated from bodily fluids that are normally sterile (blood, cerebrospinal fluid (CSF), pleural fluid, or synovial fluid) in the presence of clinical evidence of infection should be evaluated for its antimicrobial susceptibility in vitro. If dissemination of infection is suspected, isolation of the agent from samples obtained from sites distant from the primary site of infection will strengthen confidence in the diagnosis.10 Cultures of blood samples should be performed if dissemination is suspected because of systemic signs or if no primary site of infection is apparent. It is important to properly prepare the site of venipuncture so that the sample does not become contaminated. Results of cultures may be difficult to interpret if such details are ignored. The type of sample to be submitted (biopsy, feces, fluid, aspirate, blood) is selected on the basis of the disease. Recommended sites and materials to be sampled are presented in discussions of specific diseases elsewhere in this text.
If the patient has received antimicrobial drugs, the collection procedure should be delayed until the drugs are adequately eliminated from the animal’s body so that residual drugs will not interfere with bacterial growth. Because approximately 99% of a drug will be eliminated from the body within seven half-lives of disappearance, an appropriate delay between treatment and sampling can be estimated if pharmacokinetics of the drug are known. However, the delay calculated by this method may not be appropriate for severely ill patients. A fairly reliable rule of thumb is to wait 18 to 36 hours after the last dose of a drug. If a repository form of the drug is used, a longer delay is indicated. If the patient’s condition does not permit a delay, the probability of isolating organisms from bodily fluids can be increased if samples are passed through a device designed to remove antimicrobial drugs from the sample.* Directions for use of such a device should be closely followed. If samples are obtained for culture at the time of a necropsy, the treatment history may be important for interpretation of results of subsequent antibiograms.
Evaluation of stained smears of samples remains the most rapid and useful method of early recognition of some infectious agents.1,10 A direct smear should be made from a part of the sample, stained, and evaluated microscopically for bacteria and to characterize the cytologic response. Gram stain for bacteria, Wright stain for cytologic examination, acid-fast stain for mycobacteria, methylene blue and potassium hydroxide preparations for fungi, India-ink preparations for cryptococci, darkfield microscopic examination for spirochetes, fresh wet mounts for motile organisms (trichomonads), phase-contrast microscopy, immunofluorescence, and electron microscopy are methods of identification of infectious agents and inflammatory cells by direct examination. Techniques of DNA typing and polymerase chain reaction (PCR) are finding their place as modern diagnostic procedures for identifying some organisms.
Knowledge of morphologic and staining characteristics of bacteria is important to predict the identity of the microorganisms observed in the smear (Box 45-2).1 Gram or Wright stain will demonstrate the presence of most microorganisms. If only one stain is used, Wright stain* is preferred because its cytologic staining is superior to that of Gram stain. Characteristics of the microorganisms (fungal, bacillary, or coccoid bacteria), the nature of the inflammatory cells, and suspicions of which microorganism(s) might be present at the site of infection are especially beneficial for deciding initial treatment. With this information, treatment can be rationally initiated at least 24 hours before results of cultures are known and within minutes of the time that diagnostic samples are obtained. Although the list of those organisms is shrinking, some microorganisms respond predictably to certain antimicrobial drugs. Other organisms respond unpredictably because of genetically related resistance, which may arise by mutation, induction, or acquisition of a plasmid. If gram-negative bacilli are seen in samples, at least one member of the unpredictable class of microorganisms should be suspected. If one or more unpredictable species are present, isolation and antimicrobial susceptibility procedures must be performed.
Box 45-2 Morphologic Description of Microorganisms1
* Corynebacteria characteristically are pleomorphic; they align in V shapes after division and often appear as “Chinese letters.”
Serologic methods of diagnosing infections may be beneficial.10 However, because most patients have recovered from infections by the time a convalescent sample is obtained, serologic diagnoses may not be available in time to influence treatment. However, serologic data may be beneficial for the development of preventive strategies. The class of immunoglobulin and the timing of its production are determined by the previous antigenic experience of the host. High concentrations of specific immunoglobulin M (IgM) are useful in diagnosing certain viral infections and toxoplasmosis. Acute and convalescent sera demonstrating a fourfold rise in the concentration of a specific IgG indicate the presence of infection. Samples of serum should be obtained at an interval of 2 to 3 weeks to permit an adequate time lapse for the formation of significant amounts of IgG. Although the presence of IgG in a single serum sample, regardless of concentration, indicates exposure to the agent, it is of little assistance in diagnosing a current infection. There is no consensus about what constitutes adequately protective concentrations of immunoglobulins. Because serologic response is used to diagnose some conditions, is the presence of immunoglobulins an indication of infection, protection, vaccination, or passive transfer?
Documentation of infection entails more than merely listing isolated microorganisms on a laboratory report. The veterinarian must decide if the isolated organisms could be responsible for the condition in the animal, if they are commensal, resident flora, or if they are merely contaminants resulting from improper sampling technique. For example, Escherichia coli isolated from a sample of feces from a calf does not indicate that the calf’s diarrhea was caused by that organism. However, if that isolate was shown to possess the K99 pilus antigen and the ability to produce enterotoxin and if the signalment and clinical signs are compatible with coliform enteritis, the veterinarian can establish a cause-effect relationship from the bacterial presence.
PRINCIPLE 3: DETERMINE MICROBIAL SUSCEPTIBILITY IN VITRO
Quantitative assays of a microbe’s susceptibility to antimicrobial drugs in vitro are necessary for patients with severe or complicated infectious processes and for those with infections caused by organisms with unpredictable susceptibility patterns.1,10,51,52 It is important to remember when formulating a therapeutic strategy that “the bug denotes the drug.”4 A microbe is considered sensitive to a drug if the concentration of the drug that inhibits growth of the organism in the testing system in vitro can be achieved in vivo after administration of the drug by methods customarily used in clinical situations. Two commonly used quantitative assays of susceptibility are broth dilution and disc diffusion tests.6,10,50-54 Reliability and reproducibility of these procedures depend on the organism examined, standardization of the inoculum, medium used, conditions of incubation, and concentrations of the drug. The development and promotion of performance standards, as well as interpretive criteria, for in vitro antimicrobial susceptibility testing of bacteria isolated from animals is the mission of the Subcommittee on Veterinary Antimicrobial Susceptibility Testing of the Clinical and Laboratory Standards Institute (CLSI).* Results of these procedures can be coupled with knowledge of the clinical pharmacology of potentially useful drugs and of pathophysiologic changes in the patient to design an individualized dosage regimen for the patient.
Broth dilution procedures involve inoculation of a known number of organisms into tubes or wells holding a volume of broth that contains specific concentrations of antimicrobial drugs. These inoculated tubes or wells are incubated under standardized microenvironmental conditions. The lowest concentration of antimicrobial drug that inhibits visible bacterial growth is defined as the minimum inhibitory concentration (MIC) of that drug for that organism. The lowest concentration of antimicrobial drug that prevents bacterial growth, when an aliquot of the inoculated broth that contains no visible growth is subcultured onto drug-free agar or into broth, is the minimum bactericidal concentration (MBC). Therefore the MIC is a measure of the bacteriostatic concentration of the drug, and the MBC is a measure of the bactericidal concentration of the drug. Drugs that are by some conventions classified as bactericidal have an MBC within one or two twofold dilutions of the MIC. The adjectives bacteriostatic and bactericidal should be used to describe concentrations of drugs rather than to classify drugs. The disk diffusion susceptibility test involves application of antimicrobial drug-impregnated paper disks onto inoculated agar plates. The antimicrobial drug diffuses from the disk into the agar, and a progressively decreasing gradient of concentrations of the drug is developed centrifugally around the disk. If the drug is active against the organism, a growth-free zone will surround the disk. The size of the growth-free zone can be correlated with the MIC determined by dilution assays. Because multiple factors affect diffusion of drugs in the medium, the size of the growth-free zone induced by one drug cannot be equated to a zone of the same size induced by another drug.
The microdilution method provides information that may be more clinically relevant than that offered by the agar dilution method for determining therapy for bacterial infections of animals. The quantitative endpoint of susceptibility can be correlated with concentrations of the drug in bodily fluids or tissues. As a result, a more informed choice of treatment can be made than is usually possible when using agar dilution methods.
Standards for susceptibility in vitro do not necessarily apply to activity against pathogens that can be found within phagocytes in the patient. Results of intracellular antimicrobial activity were not predictable based on data from standard susceptibility procedures and have generated additional questions about the wisdom of generalizing susceptibility information in vitro or as predictable of clinical response.18,19
Laboratory reports of susceptibility of microorganisms to antimicrobial drugs provide only part of the information needed to formulate appropriate therapeutic actions. Interpretation and application of that information remain the responsibility of the veterinarian. It is reasonable to assume that if the same conditions are present at the site of infection as in vitro, the results obtained in vitro could also be expected in vivo. However, standardized microenvironmental conditions of a system in vitro are seldom present in the patient, and those conditions in the patient are usually unknown to the attending veterinarian. Temperature, humidity, partial pressures of oxygen and carbon dioxide, pH, osmotic pressure, presence of debris from damaged tissue, inactivating substances, and nutritional substrates at the site of infection are as important to therapeutic success as is the concentration of the drug. Limitations of susceptibility procedures and difficulties encountered with interpretation and application of results are eclipsed by the benefit provided. During recent years the clinical relevance of categorizing organisms as “susceptible,” “intermediate,” or “resistant” has been questioned. Although it is generally agreed that patients with “susceptible” organisms should respond successfully to those antimicrobials, classification of organisms as “intermediate” or “resistant” is not necessarily associated with clinical failure. Retrospective and prospective studies, as well as studies with models—computerized and laboratory animal—have been undertaken to resolve the dilemma, but accurate predictors of clinical outcome remain elusive.55 Niederman states that the clinical relevance of resistance is overemphasized.3 Other investigators emphasize that the outcome of microbe, antimicrobial, and host defenses is dependent on time and concentration of medication to which the microbe is exposed, regardless of medication evaluated.18 Those times and concentrations may be clinically relevant but beyond those used by standard procedures in vitro. Several reports demonstrate that being a member of a subpopulation of patients, categorized by disease, organism, and concurrent health issues, has significant influence on final clinical outcome.3,56,57 Metlay conveys concerns that changing treatment patterns based on perceptions of clinical relevance of resistance rather than on scientific evidence severely limits the ability to continue to monitor effectiveness of available treatments; yet clinical effectiveness continues to be the primary factor driving selection of antimicrobial medication.
Although the previously mentioned evidence germinated in human medicine to form the basis for development of treatment protocols, similar data for veterinary patients are severely lacking.
PRINCIPLE 4: USE AN APPROPRIATE DOSAGE REGIMEN
In most instances there are insignificant differences in clinical response after bactericidal or bacteriostatic therapeutic protocols.53,58 Bacterial meningitis, endocarditis, and gram-negative bacillary infections in neutropenic human patients are conditions in which bactericidal concentrations of active drugs at the site of infection were correlated with improved response. Confirmatory data with animal patients are lacking, but extrapolation may be acceptable when similar causative organisms are present. Because of complex interactions among microbes, subtherapeutic concentrations of drugs, postantimicrobial effects, host defenses, and microenvironment at the site of infection, dogmatic statements about the requirement for bactericidal protocols should be avoided.
The drug selected for treatment must reach the site of infection, at an adequate concentration of the active form(s), for a sufficient time that its selectively toxic effect can be inflicted on the infectious agent. Astute application of clinical judgment and knowledge of pathophysiology and clinical pharmacology are important for the development of an appropriate dosage regimen so that therapeutic success may be expected.
A dosage regimen has six components: formulation of the drug to be used, dose of drug to be administered, route of administration, site of administration, dosing interval, and duration of treatment.10,59 When the appropriate drug has been selected, dose, route, site, and interval of administration can be formulated using pharmacokinetic values of the drug in the targeted species. The drug’s bioavailability, its distribution to the site of infection, the duration of therapeutic concentrations at the site of infection, and its clearance from the body must be considered. The following are affected by drug- and host-related factors: solubility of the drug and its formulation in water and lipids, formation of a concentration gradient, blood flow at the site of absorption and at the site of infection, ionization of the drug, binding of the drug to proteins, biotransformation of the drug, chemical characteristics of the drug, presence of an inflammatory response, and microenvironment at the site of infection.
Postantimicrobial effects of some drugs may be significant. These effects can be considered with pharmacokinetic values and mathematically factored into the calculation of a dosage regimen.60 Few patients have conditions that require detailed mathematic calculation of a dosage regimen. Patients that may require such attention are those with compromised function of organs involved with biotransformation or elimination (primarily liver or kidneys), those that need treatment with drugs that are potentially toxic, and those that receive two or more drugs that interact in a potentially dangerous manner or that alter biotransformation or elimination of themselves or other drugs. Diligent monitoring of the high-risk patient for evidence of insult or toxic damage to organs is recommended (e.g., routine urinalysis, blood urea nitrogen, creatinine, sorbitol dehydrogenase).
The ability of an antimicrobial drug to distribute to an infected site depends on circulating concentrations of the drug, molecular size, binding of the drug to proteins in plasma and in tissue, water and lipid solubility of the drug, ionization of the drug, inflammation, active transport mechanisms, affinity for the particular tissue, and rate of elimination.10,18,53,61 Increased blood flow and capillary permeability associated with inflammation at the site of infection allow passage of drugs into areas that might otherwise be inaccessible. The blood-brain barrier poses such a barrier to drug passage. Distribution of penicillins and cephalosporins into CSF and other tissues is inversely proportional to the degree of plasma protein binding of the drug and directly varies with the degree of inflammation present. Aminoglycosides enter the CSF poorly, regardless of the presence of inflammation. However, no drug completely equilibrates across the blood-brain barrier; therefore high systemic concentrations of drugs are necessary to achieve adequate concentrations in the CSF, unless the intrathecal route of administration is used.
For reasons and by mechanisms that aren’t totally understood, some drugs, particularly macrolides, concentrate in tissues and remain in those tissues for expended periods.3,19,55,57 Those characteristics are at risk of being misinterpreted because specific contributions to clinical outcome are not clear. Also, the time course of those drugs in plasma, as described by classical pharmacokinetic methods and as used typically for pharmacokinetic and pharmacodynamic (PK/PD) modeling, do not adequately apply to the time course in the target tissue. Therefore for macrolides, typical PK/PD modeling does not adequately serve as a predictor of clinical outcome.
Drugs may be concentrated along routes of elimination and used advantageously to treat infections along those routes. However, when usual pathways of excretion are impaired, therapeutic success may fall, and drug-induced toxicity becomes more likely. Drugs that concentrate in urine can be used to treat urinary tract infections, even though concentrations of those drugs in blood may be ineffective against infection elsewhere (e.g., benzathine penicillin G or ampicillin trihydrate in the horse). This illustrates the necessity for relating the susceptibility of the organism to the concentration of drug at the site of infection. Penicillins, cephalosporins, trimethoprim-sulfonamide, and other drugs that attain high concentrations in urine may be effective against organisms that are otherwise considered resistant. Similarly, drugs that are eliminated by the biliary route may be used to treat biliary or hepatic infections. Drugs that undergo significant biliary elimination or reach beneficial concentrations in the liver include erythromycin, chloramphenicol, tylosin, and tetracyclines.
Duration of treatment varies with the disease and individual patient.3 Activity of the host’s defenses and ability of the organism to resist those defenses, mechanism(s) by which the organism develops resistance to the drug, location of the infection, and primary activity of the drug influence decisions about the duration of therapy. If antimicrobial drugs are truly necessary for the patient, one dose of the drug(s) is seldom sufficient unless the product is designed for prolonged exposure of the pathogen to adequate concentrations. Usually, treatment continues beyond resolution of the patient’s clinical condition. Duration of treatment remains a judgment call of the attending veterinarian.
Microenvironmental conditions at the site of infection must be compatible with the selected drug. Supernate of fluid from abscesses is acidic, hyperosmotic, and hyperionic, with relatively low concentrations of sodium and chloride and high concentrations of potassium and phosphate.62 Relative to the respective concentrations in serum, concentrations of calcium are lower, those of magnesium are higher, and those of albumin and protein are lower in abscesses. Regarding drugs with intracellular sites of action, only that part of the drug that is nonionized, unbound to proteins or other constituents, and escapes inactivation by enzymes or competitive compounds crosses the bacterial plasma membrane to reach the site of action to be therapeutically active. Penicillins and cephalosporins inhibit synthesis of the bacterial cell wall, but death of bacteria occurs when the bacteria rupture as a result of the relatively hyperosmolar interior of the bacteria. If the extracellular microenvironment is isosmolar relative to the interior of the organism, a cell-wall variant (protoplast or spheroplast) may form and continue to survive.6,63
Aminoglycosides are ineffective in an anaerobic microenvironment because the oxygen-dependent transport system that is necessary for intracellular uptake of the drug by susceptible bacteria is nonfunctional in an anaerobic environment.6,53,62 Drugs that are extensively bound to proteins in plasma are also extensively bound to proteins in pus.62 In addition to binding to proteins, aminoglycosides and polymyxins bind to constituents in sediment of pus from human patients. Binding of gentamicin is reversible and does not inactivate the drug. The antimicrobial activity of gentamicin may be inhibited as much as 16- to 32-fold by the acidic pH, high ionic content, and osmolality of pus. Activity of microbial β-lactamase at the site of polymicrobial, anaerobic infection can be sufficient to reduce the concentration of effective β-lactam antimicrobial drugs. Debris from tissue may provide adequate substrates for bacteria to circumvent effects of trimethoprim or sulfonamides.6,64
Size and purity of the inoculum also influence antimicrobial activity. A certain number of molecules of aminoglycosides is needed to kill a single bacterium; therefore antimicrobial activity is influenced by the amount of active drug relative to the size of the inoculum.53 Many infections are polymicrobic, and antimicrobial activity at the site of infection is influenced by complex interactions among microbes, the host, and antimicrobial drugs that are not influential in susceptibility test systems in vitro.62
The effect of most antimicrobial drugs is best when the pathogen is actively growing and dividing, because at that time the organism is most susceptible.6 When infections mature, bacterial growth rates are reduced, and bacterial population density increases.53 Mature infections caused by gram-positive cocci respond poorly to delayed treatment. Effects of delayed treatment against Bacteroides fragilis have been demonstrated with metronidazole, clindamycin, cefoxitin, and moxalactam. Metronidazole was the least affected by delay. A group of β-lactam antimicrobial drugs, the penems, have good activity against slowly growing bacteria.65 They have been used in animal patients, but their applicability in veterinary medicine will be better defined by results of well-designed studies.
A drug formulation administered to animals of different species or to different animals of the same species may have different pharmacokinetic characteristics. Pharmacokinetic characteristics may be altered by disease and can vary from animal to animal. Pharmacokinetic studies are usually performed with healthy animals; pharmacokinetics of drugs in diseased animals may be substantially different. Pharmacokinetic values are used to describe the time course of a drug in the body and to predict what will occur if the drug is administered to another animal. If predicted values do not accurately describe the time course in diseased animals, those predicted values are not acceptable, and data from appropriately designed studies with diseased animals should supersede previous data. A calf with hypovolemic shock associated with gram-negative bacteremia and endotoxemia may have inadequate peripheral perfusion with subsequent inadequate absorption of drugs administered intramuscularly. Because of individual variation, this principle can be remembered as “the horse directs the course”4 or “the cow dictates how.”
A good example of this is found in the combination of trimethoprim and sulfadiazine. The pharmacokinetics of both drugs are relatively similar in both horses and cows. However, in cattle the time course and circulating concentrations of each drug differ significantly.66-68 Absorption of trimethoprim appears to be the limiting factor affecting circulating concentrations of that drug when it is administered extravascularly. However, this should not be considered as an overall impediment to the efficacy of this combination of drugs. Efficacy has been demonstrated experimentally against salmonellosis in calves68; its efficacy against urinary tract infections cannot be adequately judged by circulating concentrations of these drugs.
PRINCIPLE 5: MONITOR RESULTS OF THERAPY
It is surprising to note that optimal therapy (drug, dose, route, dosing interval, duration, and ancillary treatment) against pulmonary infections in people is not inflexible or established.3,57,58 The same is probably true for infections in animals. It is extremely difficult to determine the minimally effective dosage regimen of a drug; therefore the veterinarian must monitor, by appropriate means, results of treatment.
Without monitoring the therapeutic response, the veterinarian is unable to assess success or failure of treatment. Assessment should continue throughout the treatment period.3,10 Nonspecific and specific methods used to document infection can also be used to monitor therapeutic response. Intervals at which these procedures should be performed depend on the type, severity, and site of the infection. Clinical signs, morbidity, mortality, hematologic changes, radiologic signs, microbial reduction, and monitored concentrations of drugs are useful in assessing success of therapy; but each has inherent limitations and none is clearly superior.3,56,57 Measurement of concentrations of some antimicrobial drugs in serum or plasma is an aid to assessing adequacy of the dosage regimen and reducing risks of toxicity.50 Therapeutic drug monitoring is most applicable when patients are receiving aminoglycosides, have impaired function of organs of elimination or biotransformation of the drugs, or receive more than one drug, which may result in adverse interactions.50,69 Therapeutic success against bacterial endocarditis is more likely when the patient’s serum (with drug) will inhibit bacterial growth in a 1:8 or higher dilution and will kill the organisms in a 1:4 or higher dilution.50,52 Critical concentrations of antimicrobial drug in serum for other infections have not been established but are generally targeted at concentrations that are severalfold higher than the MIC.
Studies are needed that evaluate the relationship between circulating concentrations of the drug and response to therapy. As with documentation of infection, interpretation of these data is important. Fever, other signs of inflammation, and even hematologic abnormalities can result from the therapy (drug-induced fever, leukocytosis) and nonspecific factors and may not be caused by infection.10,70 Monitoring is the only means of determining clinical applicability of results of susceptibility tests in vitro and efficacy of the treatment used. Disparity between results in vitro and those in vivo is not uncommon.3,52,53,56,57,64
PRINCIPLE 6: INVESTIGATE CAUSES OF THERAPEUTIC FAILURE
Failure of treatment can result from any of several factors (Box 45-3). When response to treatment is not as expected, the cause should be sought, and the problem should be corrected. Potential for therapeutic failure can be decreased by applying principles outlined in this chapter, by considering the status of the host’s defenses, by initiating appropriate treatment before the condition becomes irreversible, and by providing appropriate adjunctive therapy with drugs or by lavage, drainage, or removal of foreign bodies from the site of infection. Conditions caused by bacteria will not respond to antiparasitic drugs; those caused by viruses, helminths, or fungi will not respond to antibacterial drugs; those caused by helminths will not respond to antifungal drugs. These examples serve to demonstrate inappropriate choice of drug, but equally inappropriate is the use of drugs to which the organisms are resistant, that are ineffective in the microenvironment, or that do not distribute to sites of infection. Occasionally an infection that does not respond may be overcome by changing the dose of drug being used, the formulation, the route of administration, or the administration interval or by prolonging the duration of treatment. Compliance with the selected dosage regimen is necessary before therapeutic success can be expected. Studies have revealed that inadequate compliance is commonly encountered.3,57,71 Disease can alter host-related factors such as function of organs, perfusion, or inflammatory response, which affect absorption, distribution, biotransformation, and/or elimination of the drug. Because antimicrobial drugs act in concert with the host’s defense mechanisms, any defect in these defenses can reduce the efficacy of antimicrobial drugs. A microenvironment that is inappropriate for activity of the drug, organisms that are resistant to the selected drug, or superinfection may also result in therapeutic failure. Interactions of drugs in vivo or in comixtures can alter pharmacokinetic variables of drugs or render them chemically ineffective. Direct toxicity of antimicrobial drugs can be detrimental to the patient, as well as cause therapeutic failure. If the patient’s condition has advanced to a point of irreversibility, any amount of appropriate therapy may be ineffective. Therapeutic failure is too frequently blamed on an inactive or “bad” drug. This is probably the least common cause of therapeutic failure if dates of expiration and conditions of storage of the drug are properly observed and if the product is appropriately used as prescribed.
PRINCIPLE 7: RESTRICT CONCOMITANT USE OF ANTIMICROBIAL DRUGS
Fixed-drug combinations or concomitant use of two or more antimicrobial drugs is occasionally appropriate, as shown in Box 45-4.10,52,53 However, in most instances one drug with a specific antimicrobial spectrum will provide adequate therapy and will reduce the potential of adverse effects in the patient or the potential for selection of resistant organisms. There are very few, if any, situations in which some fixed-drug combinations are superior to individual drugs. When used concomitantly against a specific organism or organisms, two or more antimicrobial drugs may be synergistic, additive, antagonistic, or indifferent in their effect. Selection of two or more antimicrobial drugs for concomitant use should not be undertaken without considering their cumulative effects and the legitimate need for concomitant use of drugs. Too frequently, antimicrobial drugs are combined or used concomitantly to provide “broad-spectrum coverage” because of the attending veterinarian’s diagnostic insecurity or to replace diagnostic procedures. Concomitant use of antimicrobial drugs should be limited to the following: to provide synergy against infecting organisms, to prevent bacterial resistance to antimicrobial drugs, to extend the antimicrobial spectrum as part of the initial therapy against life-threatening conditions, or to treat mixed-bacterial infections. Concomitant use of antimicrobial drugs beyond these situations is not justified, unnecessarily exposes the patient to risks of adverse reactions to drugs, and increases pressure for development of resistance to drugs by the infecting organism or others.
Box 45-4 Concomitant Use of Antimicrobial Drugs
* Potentially lethal complications may develop in horses or sheep that receive lincomycin.
Synergy results when antimicrobial activity of two drugs in combination is greater than would be expected by the sum of the activity of the individual drugs. Synergy can result if the drugs act at different sites in the same metabolic pathway (e.g., trimethoprim + sulfonamide) or at different sites (e.g., 30S and 50S ribosomal subunits) in the organism. Activity of one drug may improve the entry of another drug into the organism (e.g., penicillin or cephalosporin + aminoglycoside) or prevent the degradation of another drug by the organism (e.g., amoxicillin + clavulanic acid). Synergistic combinations of antimicrobial drugs may be essential in some severe infections that are difficult to eradicate or infections in patients with temporarily impaired defense mechanisms. However, concomitant use of antimicrobial drugs is not necessary in routine treatment of most infections. Synergy does not imply that a “better” clinical response will result. Despite evidence of synergy between two drugs in vitro, convincing evidence that such synergistic combinations are superior to single drugs in vivo when treating defined infections is sadly lacking in veterinary medicine. Controlled, prospective clinical investigations designed to evaluate synergistic responses in vivo are desperately needed.
Antagonism has been demonstrated between penicillin and tetracycline.10 In some instances, ampicillin and chloramphenicol have been antagonistic, whereas in other instances they have been therapeutically beneficial. Antimicrobial drugs that have the same or an anatomically proximate site of action (phenicols, macrolides, and lincosamides) should not be used concurrently because of antagonistic competition for those sites in bacteria.
If bacterial resistance to drugs occurs by mutation and with separate frequencies for two different antimicrobial drugs, the statistical probability that mutational resistance against both drugs will develop is inversely related to the product of the individual frequencies. Following this rationale, concomitant use of antimicrobial drugs has been most successfully applied in the treatment of infections caused by organisms in which resistance to single drugs develops rapidly. Resistance against multiple drugs that is encoded by plasmids should not be expected to follow this pattern. Data fail to support the popular belief that such a combination of drugs is necessary to avoid the development of resistance when there is evidence that resistance forms in face of this concomitant use. The popularity of combination treatments does not necessarily mean they are more efficacious than single-drug treatments.
It is justifiable and recommended to use more than one antimicrobial drug as initial treatment of life-threatening infections before results of bacterial cultures are known. This treatment should be based on the presumption that most, if not all, organisms that cause the infection will be susceptible to selected drugs and that withholding treatment will most likely result in death of the patient. Because such therapy is initiated before the causative agent and its susceptibility can be ascertained, it is imperative that all appropriate diagnostic samples be obtained before initiation of such treatment. It is important that historical or statistical data be taken into account when selecting the most probable causative agent(s). After the infectious agent and its susceptibility are known, treatment should be appropriately adjusted. Extended antimicrobial spectrum is too often abused as a substitute for collection of diagnostic data, because of the veterinarian’s diagnostic insecurity or because of the client’s insistence on treatment of the patient.
PRINCIPLE 8: APPROPRIATELY ATTEND TO ADVERSE REACTIONS TO DRUGS
Adverse reactions to drugs can develop in patients as a result of drug-related or host-related factors.10,70,72 Most can be classified as side effects of the drug, hypersensitivity to the drug or its metabolites, interactions of two or more drugs in vivo or incompatibilities in an admixture before administration, alterations of indigenous microflora by the drug, and direct toxic effects of the drug on the host’s tissues or as idiosyncratic or idiopathic reactions that defy elucidation and classification.
Adverse effects are not always predictable and may not be side effects of the drug. Side effects of a drug are usually predictable because they result from the pharmacologic activity of the drug. For example, tetracycline may be selected primarily for its activity against a respiratory pathogen in a foal. Discolored dental enamel on permanent teeth may be an acceptable side effect, but altered intestinal microflora and fatal diarrhea are adverse effects.
All types of hypersensitivity reactions can occur in response to drugs. Clinical signs demonstrated by animals with hypersensitivity reactions to drugs are typical of those caused by other allergens. Because the molecular weight of most drugs is too small for the parent drug to serve as an allergen, polymers of the drug or its metabolites chemically combine with amino acids, polypeptides, or carbohydrates to form allergenic complexes. Polymers alone may be large enough to be allergenic. Figures regarding the frequency and type of hypersensitivity reactions in animal patients may not be representative because reports of such reactions are probably not complete. In the United States, adverse reactions to a drug in animal patients should be reported to the manufacturer of the product and to the FDA-CVM. It is my opinion that the manufacturer should be the first to be contacted. Manufacturers are the primary resource for data related to their products and are usually equipped and staffed to respond to emergency clinical situations that may result from adverse reactions.
Interactions of drugs in vivo and incompatibilities of drugs in admixtures are possible when more than one drug is used.72,73 Some interactions and incompatibilities are well known, and references to these data should be available. Pharmacists have considerable reference material to consult, and their input is extremely valuable in these matters. Before drugs are mixed or administered concurrently, interactions and compatibilities should be determined (Table 45-2). In this context, polyionic fluids, vitamins, minerals, and other substances should all be considered as “drugs.”
Table 45-2 Specific Drugs and Additives That Are Incompatible When Comixed Before Administration72,73


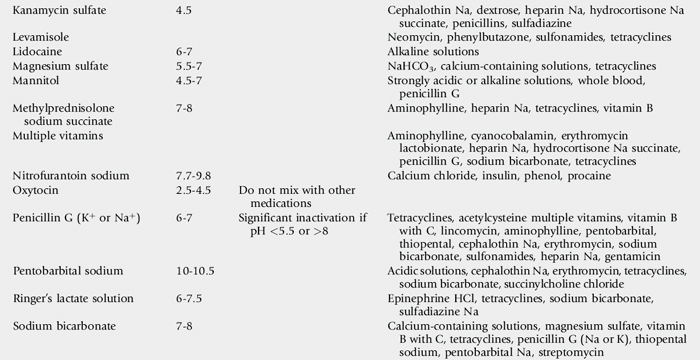
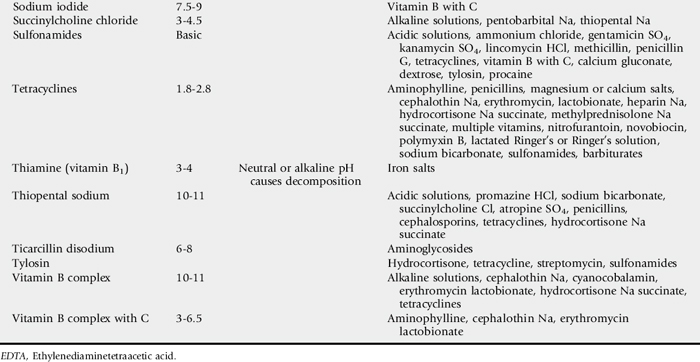
Alterations in indigenous microflora can reduce the effectiveness of natural host defenses, allow superinfection, or alter normal function of some bodily systems. Fibrinonecrotic colitis in horses that receive clindamycin or lincomycin is an example. This problem is serious enough that the use of these drugs is contraindicated in horses. Tetracyclines (especially doxycycline), macrolides, or potentiated sulfonamides can also alter the intestinal bacterial flora and thereby may precipitate potentially lethal diarrhea.74,75 These drugs may be drugs of choice for treatment of some patients, and the adverse effects considered in context while a strategy for treatment is formulated.
Toxic effects of antimicrobial drugs may not be predictable. Some drugs such as aminoglycosides or amphotericin B have predictable, dose-related toxicities. The ratio of toxic-to-therapeutic concentrations (therapeutic index) of drugs varies with the drug’s properties and host-related factors that alter the patient’s susceptibility to toxicity of the drug. If a drug with a low toxic-to-therapeutic ratio is chosen to treat an infection, its toxic and therapeutic effects should be closely monitored. If toxicity of a drug is predictable by its circulating concentrations, total amount of the drug administered, or duration of therapy, careful attention should be afforded these values. The greatest difficulty in predicting either efficacy or toxicity is knowing the minimum concentration of the drug that predictably causes each of these effects in the individual patient. Monitoring circulating concentrations of aminoglycosides is an essential aspect of therapy with these drugs because of their potential for causing toxicity and erratic pharmacokinetics in patients.69,76 Amphotericin B is also predictably toxic, and careful monitoring of its effects is necessary. Toxicity of drugs that require biotransformation for activation or inactivation can be enhanced or reduced by alterations in biotransformation resulting from disease or concomitantly administered drugs. Chondrotoxic effects of fluoroquinolones in adult and young horses is supported by results of some investigations25,26 and refuted by others.27-29 Perhaps an equally important question to be answered while trying to resolve discussions of occurrence of that arthrotoxicity is how well the insulted articular cartilage heals. Just as with disease, drug-injured tissue may regenerate and regain healthy function resulting in indistinguishable effects at a later date.
Frequently the true pathogenesis of an adverse effect is not known. It may not be clearly related to the drug or may not have been observed previously. Because drug-related reactions in animals are iatrogenic, reasonable suspicion that a reaction could be drug related should be maintained until proven otherwise. Observations and thoroughness of investigation by the attending clinician remain the best means of establishing the cause-effect relationship of drug-induced reactions. That diagnosis is often arrived at by a process of elimination of other potential causes.
PROPHYLACTIC OR METAPHYLACTIC USE OF ANTIMICROBIAL DRUGS
The medical definition of prophylaxis is the prevention of disease.77 The word is derived from the Greek prefix, pro, which signifies before or in front of, and the word phylaxis, which means a guarding or protection from infection. Prophylactic use of antimicrobial drugs implies that the medication is administered before exposure to an infectious organism in order to prevent infection. The term metaphylaxis is also of Greek origin, with the prefix meta meaning after, beyond, or over. The prefix indicates change, transformation, exchange, after, or next. Metaphylactic use of antimicrobial drugs implies timely administration of the medication after exposure to an infectious organism, during the transformation or development of clinical signs of the disease, before clinical disease is apparent. In previous editions of this text, no distinction between prophylactic use and metaphylactic use of antimicrobial drugs was made. That has been corrected in this edition.
Despite apparently widespread administration of antimicrobial drugs to prevent disease, prophylactic use of antimicrobial drugs remains controversial and of unproven value in many instances.77a Far too frequently veterinarians and lay personnel use antimicrobial drugs prophylactically because of concern over diagnostic accuracy. Although indiscriminate use of antimicrobial drugs to prevent disease should be condemned, prophylaxis can be useful in certain circumstances.78 Unfortunately these circumstances have not been well defined in veterinary medicine, partly because of difficulties in assessing response to microbial prophylaxis.79 If a beneficial effect results from prophylactic use of drugs, the therapist is encouraged to repeat their administration when later presented with a similar situation. For example, antimicrobial drugs may be administered to horses with viral upper respiratory disease to prevent secondary bacterial pneumonia. If the horse does not develop pneumonia, it is assumed that the antimicrobial drug prevented bacterial pneumonia. Therefore the next horse that demonstrates signs of upper respiratory disease will receive antimicrobial drugs prophylactically. This is not sound reasoning, nor is it justification for prophylactic use of antimicrobial drugs. That horse may not have developed pneumonia if the antimicrobial drugs had been withheld. Seldom is a determination made about whether a result is “because of” or “in spite of” the prophylactic use of medication.
In some instances, antimicrobial drugs are used prophylactically with guidance by clinical impressions. Unfortunately, objective data to support such uses are not always available. Controlled, blinded, prospective clinical investigations are of paramount importance for obtaining that information.79 Proper performance of such studies can be difficult. The multitude of factors involved with such studies (e.g., large numbers of patients, criteria for selection of patients, confirmatory diagnostic procedures, choice of “appropriate controls,” variables to be evaluated, dosage regimens, and ancillary measures to be used) result in a complexity that discourages most veterinarians who initially desire to embark on such studies. An additional factor that complicates studies of the efficacy of antimicrobial drugs perioperatively is that historical incidences of infection may not be reliable controls because surgical techniques vary among surgeons and have changed because of advances in surgical materials and procedures. Therefore prophylactic use of antimicrobial drugs in many situations remains a matter of opinion, risk, and concern based on conditions pertaining to the individual patient rather than a matter of fact that is universally applicable. Consequently, recommendations for prophylactic use of antimicrobial drugs should not be dogmatic.
However, principles for prophylactic use of antimicrobial drugs in each situation are the same. Antimicrobial drugs are occasionally used in animals to prevent coccidiosis, clostridial infection of wounds, bacterial pneumonia, and bacterial infections after orthopedic or intraabdominal surgical procedures. Morbidity and mortality associated with coccidiosis was reduced in susceptible calves by preexposure administration of monensin.80 The incidence of clostridial infections in untreated wounds is not accurately known. Specific immunity in the host, complicating factors, use of biologics, and ancillary procedures influence the incidence and severity of such infections. Because clostridial organisms produce severe and often lethal infections, the risk of fatal vegetative infection warrants the prophylactic use of antimicrobial drugs in high-risk patients. However, antimicrobial drugs should not be considered a substitute for active immunization provided by biologic products or for proper local treatment of the wound.
Antimicrobial drugs used prophylactically are perhaps used most to prevent or control bacterial pneumonia. It has been stated that viral respiratory diseases predispose the respiratory tract to secondary bacterial infections. Concerns that viral infections predispose the entire respiratory tract to bacterial infection are based on the fact that the former can destroy ciliated epithelium and inhibit mucociliary clearance mechanisms, reduce production of surfactant, promote virulence of bacteria, and inhibit phagocytic activity against some bacteria.81-84 But the efficacy of antibacterial mechanisms in preventing bacterial superinfections appears to depend on the bacteria and the virus(es) involved.
All viral pneumonias do not result in bacterial superinfections; conditions must be appropriate for that to occur.82 One investigator proposed that the secondary bacterial infection of the upper respiratory tract was probably “beneficial in the total infection experience” of the young horse entering training or racing.85 In spite of these data the true incidence and risk-related factors surrounding secondary bacterial infections are not known. Several controlled prospective studies have evaluated metaphylactic use of antimicrobial drugs against respiratory disease in cattle. Similar, but limited, studies performed with horses have shown that some transported horses may benefit from prophylactic use of antimicrobial drugs.86
Control of bovine respiratory disease complex (BRDC) with antimicrobial drugs in cattle at high risk for developing the disease has been studied intensely for several years.87-98 Nevertheless, effects of metaphylactic use of antimicrobial drugs in BRDC and recommendations for such use are far from universal. Many factors contribute to the incidence of BRDC: vaccination history of the animals, environment, diet, duration of shipment, time of initial medication after arrival, number of calves per group, commingling of animals from different sites of origin, condition of the calves, method of administration of medication (by injection, in water, or in feed), duration of administration, and sequence of administration if more than one antimicrobial drug was used.
Many factors can complicate assessment of metaphylactic use of antimicrobial drugs against BRDC, but efficacy of control of BRDC can be evaluated by several variables that include morbidity, mortality, epidemiologic pattern of morbidity and mortality, days of treatment, amount of medical attention required (labor and medication), weight gain, relapses, and/or economic return.87,89,91,93-96,99
Metaphylactic use of antimicrobial drugs can be beneficial, but it is unrealistic to expect total elimination of disease. Control or management of a disease and its effects would be a more realistic expectation. The effect desired from prophylactic or metaphylactic use of drugs must be specifically understood. Reduced morbidity or mortality, reduced severity of clinical signs, reduced duration of therapy, improved growth rate, improved feed efficiency, and improved or maintained production may be benefits of metaphylactic use of antimicrobial drugs, but false confidence in prevention of disease should be avoided. Volatility of costs associated with production and the market probably influences profit more than does the cost of metaphylactic use of antimicrobial drugs.
Ambiguity of antimicrobial prophylaxis or metaphylaxis in equine medicine is supported in part by a review of reports of experimentally induced as well as naturally occurring equine rhinopneumonitis and equine influenza. Equine herpesvirus 1 produces viral bronchopneumonia100 and interstitial pneumonia.101 Serous to mucopurulent rhinitis was observed commonly and was similar with equine influenza.81,85,100-106 Secondary bacterial infections were confined to the upper respiratory tract (i.e., rhinitis, lymphadenopathy) and did not include bacterial pneumonia.99,102,104,105
Perioperative use of antimicrobial drugs prophylactically is a common practice. In human patients, such use has proven to be of benefit in prosthetic cardiac valvular operations, gynecologic surgical procedures, and gastrointestinal operations.79 The drugs were administered orally, systemically, or topically as indicated by the procedure. A study with 122 dogs and seven cats undergoing elective, clean surgical procedures revealed no significant difference in incidence of infection between a group that received ampicillin and a group that received a placebo.107 Similar studies with horses are not known to the author at this time. Principles for prophylactic use of antimicrobial drugs perioperatively are similar to those outlined here. Principles for timely metaphylactic use of antimicrobial drugs have been proposed and are similar to those listed for prophylaxis.87
When antimicrobial drugs are used prophylactically, several principles should be followed.77a-79,108-111
Decisions about prophylactic use of antimicrobial drugs are not easily made and are usually based on logic and analogous situations.78 Classification of clinical circumstances and diseases of human patients has been proposed as an aid to such decisions for that species. That classification considers the risk of developing infection based on function of the patient’s natural defenses, duration of exposure to pathogens, and potential for infection by either one or multiple organisms. Direct application of that classification system to animal patients may be difficult.
Assessment of some defense mechanisms in domestic animal patients is complicated and beyond routine use in many clinical situations. Some pathogens are noted for their ability to temporarily compromise the host’s defenses, yet effects of others are unknown. Methods that are applicable (e.g., determination of concentrations of immunoglobulins, numbers and function of phagocytes and of lymphocytes) should be used when indicated and when appropriate medical management is determined. However, numbers of cells do not equate with function, and concentrations of immunoglobulins do not equate with specific activity. Animal patients must convalesce in an environment that is less hygienic than that of human patients. Animals are constantly exposed to many primary and opportunistic pathogens (bacteria, viruses, fungi, parasites) and intermittently exposed to individual pathogens that cause diseases of epizootic proportions. Therefore criteria for classification by risk of infection and indications for prophylactic use of antimicrobial drugs in animals need to be developed from epidemiologic studies of diseases of animals and controlled clinical investigations. At this time, that information is sadly lacking.
Except in specific instances with specific goals in mind, the use of antimicrobial agents to prevent infection has not been as valuable as the therapeutic use of antimicrobials. Prophylactic use of antimicrobial drugs must be tailored to the specific needs of each individual patient, as determined by the attending veterinarian and tempered by application of the principles outlined here.
EXTRALABEL USE OF MEDICATIONS IN FOOD ANIMALS
In 1994 the Animal Medicinal Drug Use Clarification Act (AMDUCA) was signed into law, amending the Food, Drug, and Cosmetic Act and legalizing most instances of extralabel use of drugs (ELUD) by veterinarians.114 For food animal practitioners, with this privilege comes the weighty responsibility of protecting the health of consumers of animal products. Veterinarians must meet very specific conditions before they may legally use or prescribe drugs in an extralabel fashion. Extralabel drug uses include prescribing or administering a human or veterinary drug at a higher dose, frequency, or duration, by a different route of administration, in a different species, or for a different indication than as described on the product’s label. ELUD is limited to cases in which the health of the animal is threatened or when suffering or death may result from a lack of treatment. ELUD may not be used to enhance production or for reproductive cycle manipulation in normal animals. ELUD may be considered in food-producing animals only when no approved drug is available that contains the same active ingredient in the required dose, form, and concentration or when the veterinarian finds that there is no approved drug that is clinically effective for the intended use. Extralabel drug use in or on animal feed is also expressly prohibited. Veterinarians’ obligations regarding extralabel use are listed in Box 45-5. Veterinarians violating state or federal laws regulating the transport, sale, or use of drugs may face a variety of sanctions including warning letters, fines, temporary or permanent revocation of license, and incarceration.
Box 45-5 Conditions Necessary for Extralabel Use of Drugs
DRUGS PROHIBITED FROM EXTRALABEL USE IN FOOD ANIMALS
Certain drugs may not be prescribed or used even under the auspices of AMDUCA.115 Under statutory authority provided in AMDUCA, the FDA‘s Center for Veterinary Medicine (CVM) has prohibited approximately a dozen drugs or drug classes, making their extralabel use in food animals illegal. Other compounds not specifically listed in AMDUCA are prohibited by virtue of the fact that no approved animal or human products are commercially available. Lastly, extralabel use of treatments is also regulated by the Pasteurized Milk Ordinance (PMO). The extralabel use in food animals of the compounds listed in Box 45-6 represents one of the FDA’s highest priorities for regulatory attention.
Box 45-6 Drugs or Drug Classes Prohibited from Extralabel Use of Drugs*
* Extralabel use of some drugs or drug classes (not specifically addressed in the Animal Medicinal Drug Use Clarification Act) are prohibited because these drugs are not available as approved animal or human products, by U.S. Food and Drug Administration (FDA) policy or in the Pasteurized Milk Ordinance (see text).
† Defined by FDA as dairy cattle (lactating or dry) older than 20 months of age.
Diethylstilbestrol
From the 1940s to the 1970s U.S. physicians prescribed diethylstilbestrol (DES, a potent nonsteroidal synthetic estrogen) to pregnant women to prevent miscarriage and other reproductive diseases. In 1971 a link between in utero exposure to DES and a rare vaginal cancer (clear cell adenocarcinoma) was established. In the same year the FDA published an alert advising doctors against the use of DES during pregnancy. The U.S. Department of Agriculture (USDA) banned the use of DES in food animals in 1979.
Chloramphenicol
An estimated one in 10,000 to 50,000 people exposed to chloramphenicol will develop a non—dose-related aplastic anemia. Because of concerns that this idiosyncratic and frequently fatal complication could be triggered by residues, chloramphenicol use in food animals was prohibited in 1984. The prohibition extends to all formulations of chloramphenicol including ophthalmic ointments. Florfenicol, a synthetic member of the chloramphenicol family, lacks the p-NO2 group thought to be responsible for inducing the aplastic anemia. Florfenicol may be used extralabel in food-producing species.
Nitroimidazoles
Historically, dimetridazole and ipronidazole have been approved for the treatment of histomoniasis (infectious enterohepatitis, blackhead) in turkeys. Ipronidazole and the human drug metronidazole have been used off-label to eliminate the carrier state of trichomoniasis in bulls. Laboratory studies of members of this drug class demonstrated mutagenicity and carcinogenicity, leading to the prohibition of their use in food animals. Because there are no approved veterinary nitroimidazole labels, the use of any member of this drug class in food animals is illegal.
Sulfonamide in Adult Dairy Cattle
The observed carcinogenicity of sulfonamides in laboratory animals, coupled with frequent finding of sulfonamide residues in milk, led to the prohibition of extralabel use of these compounds in adult dairy cattle. The FDA defines a lactating cow as any dairy cow (milking or dry) older than 20 months of age. Only one sulfonamide has a label for dairy cattle, sulfadimethoxine (SDM). The use of any sulfonamide other than SDM in adult dairy cattle is illegal. In addition, extralabel use of SDM in lactating dairy cattle is prohibited. This would include use of a higher dose of SDM or using slow-release formulation SDM boluses.
Nitrofurans
In the past, nitrofurazone and furazolidone were approved for a variety of protozoal and bacterial infections in poultry and swine. Topical formulations (sprays and “puffer” products) have also historically been marketed for wounds and ocular infections in livestock (“pinkeye”). Based on laboratory evidence of carcinogenicity and the absence of a reliable detection method, the FDA withdrew approval for systemic animal nitrofuran products in 1991 and prohibited all extralabel treatment (including topical use) in 2002. Because nitrofurans have no approved food animal uses, the use of any member of this drug class in food animals is illegal.
Clenbuterol
Marketed in the United States as an equine bronchodilator, this synthetic sympathomimetic has been used illicitly to increase weight gain and lean body mass in food animals, particularly show animals. Because muscle depletion and fat redeposition commences after drug withdrawal, producers may be tempted to market animals with little or no withdrawal interval. Cooking temperature only minimally denatures the compound, and toxicity from residues has resulted in hundreds of emergency hospitalizations in European consumers.
Fluoroquinolones
Uncertainty exists related to the magnitude and significance of human pathogen resistance resulting from antibiotic use in animals. Fluoroquinolones are mainstay treatments of antibiotic-resistant Salmonella and anthrax infections in humans. Concern that fluoroquinolone use in food animals was promoting human pathogen resistance prompted CVM in 1997 to prohibit extralabel use of these compounds. Any deviation from a fluoroquinolone product label (altering species use, dosage, route of administration, or disease indication) is illegal. In the case of the approved beef cattle formulation of enrofloxacin and danofloxacin, this prohibition extends to use in all non—beef-production animals including lactating and nonlactating dairy cows, heifer replacements, bulls, and veal calves.
Glycopeptides
Vancomycin, the only glycopeptide antibiotic available in the United States, is often the therapy of last resort for methicillin-resistant Staphylococcus aureus (MRSA) infections in humans. Demonstration of vancomycin-resistant Enterococcus in the feces of poultry and swine fed the glycopeptide avoparcin led CVM in 1997 to prohibit extralabel use of glycopeptides in food animals.
Phenylbutazone in Adult Dairy Cattle
The use of phenylbutazone, a nonsteroidal antiinflammatory agent in humans, has been associated with a variety of adverse drug reactions including fatal blood dyscrasias. After USDA reports of a high incidence of phenylbutazone tissue residues in cull dairy cows, CVM prohibited its use in female dairy cattle older then 20 months of age. Antiinflammatory therapy for lactating dairy cattle is available as flunixin meglumine. Flunixin should be used only on-label (intravenously) because intramuscular or intravenous administration can greatly extend withdrawal times.116
Antiviral Drugs in Poultry
Concern exists that use of antiviral drugs in poultry could promote drug resistance in zoonotic pathogens, particularly avian influenza H5N1 (“bird flu”). These concerns led CVM in 2006 to prohibit extralabel use of two drug classes used in treating human influenza. Adamantidine and neuraminidase inhibitors may not be used in chickens, turkeys, and ducks.
Dipyrone
Aside from compounds specifically prohibited through AMDUCA, there are several compounds that CVM has reminded veterinarians cannot be legally obtained as finished animal or human products and therefore have no legal extralabel uses.
Dipyrone, used historically as an antipyretic and antiinflammatory, has been associated with teratogenicity and agranulocytosis in humans and cannot be used in food animals.
Estradiol Cypionate
Before 2003 estradiol cypionate (ECP, a synthetic estrogen) was commercially marketed as reproductive therapy in food animals. ECP had not undergone a formal approval process, however, and discretionary marketing was discontinued for lack of food safety and efficacy data. Use in or compounding of ECP for food animals remains illegal.
Hormone Implants in Veal Calves
Although growth-promoting hormone implants have been marketed for ruminating cattle, these products never underwent regulatory approval for nonruminating veal calves. Concern existed in CVM that nonruminating calves may eliminate the hormone contained in the implants differently then ruminants. In 2005 a new warning statement was added to the label of all growth-promoting hormone implants reminding producers and veterinarians that their use in veal calves is illegal.
TREATMENT OF COMPANION OR PACK ANIMALS WITH PROHIBITED SUBSTANCES
The prohibitions listed previously pertain to food-producing animals only and not companion species such as dogs and cats. Veterinarians occasionally treat companion animals belonging to a food-producing species (horses, llamas, pygmy goats, pot-bellied pigs). As long as these animals are never offered for slaughter, CVM does not normally consider these to be food animals. Practitioners who have used a prohibited substance in a companion or pack animal that subsequently enters the food supply are subject to enforcement actions under the Food Drug and Cosmetic Act. In choosing to use a prohibited drug in a companion or pack animal, the practitioner is accepting a certain amount of liability because the ultimate fate of such treated animals is often beyond their control. Practitioners may choose to have owners sign an agreement (entered into the medical record) not to introduce the animal into the human chain.
EXTRALABEL USE OF MEDICATED FEEDS IN MINOR SPECIES
AMDUCA also prohibits the extralabel use of medications in feed. Generated by concerns of antibiotic resistance, AMDUCA’s Section 530.11 specifically prohibits the “extra-label use of an approved new animal drug or human drug in or on an animal feed.” As a matter of enforcement discretion, CVM generally has not objected to mixing a drug with an individual animal’s feed, but extralabel mass medication in feed is prohibited “without limitation or exception.”
Veterinarians may, however, be called on to treat minor food animal species such as farmed exotic ruminants, fish, or game birds. CVM has found that for many of these species there exist very few approved drugs. In addition CVM recognizes that some minor food animal species (such as fish and game birds) cannot be practically medicated in any way other than through the use of medicated feeds. In such situations, a veterinarian may determine that extralabel use of medicated feeds (approved for use in other species) can prevent suffering and death in these minor species.
Although AMDUCA prohibits ELUD in feed, in the previously described circumstances CVM “ordinarily will not consider regulatory action” against the veterinarian or animal producer provided that certain criteria listed in Box 45-7 are met.117 The CVM policy being implemented in these cases is akin to that of extralabel drug use by veterinarians before the 1994 passing of AMDUCA. The drug use is still illegal, but CVM will apply regulatory discretion relative to taking enforcement action.
Box 45-7 Conditions Necessary for Extralabel Use of Medicated Feed in Minor Species
A critical aspect of CVM’s regulatory discretion in this practice is that labeled treatment alternatives are not available. The best resource to aid practitioners in determining what treatments are approved for minor food animal species is the Minor Species Drug Approval site (www.nrsp-7.org).
COMPOUNDING IN VETERINARY PRACTICE
Compounding is defined as a manipulation to produce a dosage form of a drug other than that provided for in the labeling, such as reconstitution. CVM recognizes the need for compounding within certain areas of veterinary practice, such as mixing or dilution. Because of the absence of safety, efficacy, and food safety data for compounded drugs, potential exists for adverse reactions in treated animals or consumers of their products. Under AMDUCA, it is legal for veterinarians (or pharmacists on the order of a veterinarian) to compound from U.S.-approved animal and human drugs if the conditions listed in Box 45-8 are met.114,118
ANTIDOTES FOR FOOD ANIMALS
For both regulatory and economic reasons, there are relatively few antidotes available to treat cases of toxicity in food-producing species. In most circumstances practitioners treating toxicity in food animals are compelled to use products in an extralabel manner or to compound antidotes from bulk sources.119 From a regulatory standpoint veterinary antidotes can be placed in one of three categories, discussed in the following paragraphs.
Unapproved Commercially Marketed Veterinary Antidotes
CVM has applied regulatory discretion to allow commercial manufacture and marketing of several compounds that can be used as antidotes, even through they have never completed formal regulatory approval. These are atropine sulfate, epinephrine, and vitamin K.
Antidotes Labeled for Humans Only
There are additional human-label antidotes that can be used off-label under AMDUCA. These include pralidoxime chloride (2-PAM) and dimercaprol (British anti-lewisite or BAL).
Antidotes Compounded from Bulk Drug
Several chemicals commonly recommended as food animal antidotes are available only through compounding from bulk drug. As described in the previous section, compounding veterinary medications from bulk drugs is illegal. However, analogous to its policy concerning ELUD in feed for minor species, CVM will not normally consider regulatory action against veterinarians compounding certain antidotes from bulk sources if certain conditions are met, essentially the same as those listed in Boxes 45-5 and 45-8. CVM has listed nine chemical antidotes against which the FDA and CVM would presently not ordinarily object. These include ammonium molybdate, ammonium, tetrathiomolybdate, ferric ferrocyanide, methylene blue, picrotoxin, pilocarpine, sodium nitrite, sodium thiosulfate, and tannic acid.117,118 This list should not be construed as containing the only drugs for which the FDA would extend regulatory discretion for compounding, and inquiries about additional compounds can directed to the FDA/CVM, Division of Compliance, 301-827-1168. It is critical that veterinarians recognize that in toxicity cases withdrawal periods will need to be established not only for the antidotes but the toxicants as well. For this reason practitioners are advised to consult the Food Animal Residue Avoidance Databank (FARAD) when dealing with cases of toxicosis in food animals.
FOOD ANIMAL RESIDUE AVOIDANCE DATABANK SUPPORT FOR VETERINARIANS
Consistent regulatory requirements for ELUD (using approved drugs, feed, or compounding) are that approved, effective veterinary drugs are not available and that the attending veterinarian prescribe scientifically valid withdrawal periods before marketing of milk, meat, eggs, or other edible products. Commonly the most efficient way for a food animal practitioner to meet these requirements is to consult FARAD. Veterinarians, producers, students, or regulatory personnel may obtain free consultation with specialists by contacting 1-888-US-FARAD or accessing the FARAD website at www.farad.org. FARAD maintains a database of all food animal products approved in the United States and more then 9000 pharmacokinetic citations describing depletion of more then 2000 chemicals in various species. The FARAD website contains a database of approved food animal drugs searchable by drug, trade name, and species as well as downloadable copies of past published summary recommendations.
1 Hirsh DC, Ruehl WW. A rational approach to the selection of an antimicrobial agent. J Am Vet Med Assoc. 1984;185:1058.
2 Davis LE. Rational therapeutics. In: Mansmann RA, McAllister ES, Pratt PW, editors. Equine medicine and surgery. ed 3. Santa Barbara: American Veterinary Publications; 1982:131.
3 Niederman MS. Principles of appropriate antibiotic use. Int J Antimicrob Agents. 2005;26(Suppl 3):S170.
4 Brumbaugh GW. Rational selection of antimicrobial drugs for treatment of infections of horses. Vet Clin North Am Equine Pract. 1987;3:191.
5 Knifton A. Criteria for selection of antibiotics. Vet Rec. 1984;114:357.
6 Edwards DI. Principles of antimicrobial drug action. In: Edwards DI, editor. Antimicrobial drug action. Baltimore: University Park Press; 1980:8.
7 Jawetz E. Antimicrobial drugs: mechanisms and factors influencing their action. In: Kagan BM, editor. Antimicrobial therapy. ed 3. Philadelphia: Saunders; 1980:3.
8 Jones TC, Hunt RD. Healing and regeneration. In: Jones TC, Hunt RD, editors. Veterinary pathology. ed 5. Philadelphia: Lea & Febiger; 1983:211.
9 Smith H. The chemotherapeutic potential of inhibition or circumvention of the determinants of microbial pathogenicity. Greenwood D, O’Grady R, editors. The scientific basis of antimicrobial chemotherapy. Thirty-Eighth Symposium of the Society for General Microbiology, New York. Press Syndicate of the University of Cambridge; 1985:367.
10 Root RK, Hierholzer WJ. Infectious disease. In Clinical pharmacology: basic principles of therapeutics, ed 2, New York: Macmillan; 1978:709.
11 Weinstein AJ. Common sense (clinical judgment) in the diagnosis and antibiotic therapy of etiologically undefined infections. In: Kagan BM, editor. Antimicrobial therapy. ed 3. Philadelphia: Saunders; 1980:193.
12 Evans AS. Epidemiological concepts and methods. In: Viral infections of humans: epidemiology and control. New York: Plenum; 1982:3.
13 Mercer HD. Residue avoidance: withdrawal times for drugs not labeled for food animals. In: Proceedings of the Tenth Annual Food Animal Medicine Conference: The Use of Drugs in Food Animal Medicine. Columbus: Ohio State University Press; 1985:7.
14 Coleman DL, Ryan JL. Modulation of the immune response by antimicrobial agents. Root RK, Sande MA, editors. New dimensions in antimicrobial therapy: contemporary issues in infectious diseases. New York: Churchill Livingstone; 1984;vol 1:277.
15 Yourtee EL, Root RK. Effect of antibiotics on phagocyte-microbe interactions. In: Root RK, Sancle MA, editors. New dimensions in antimicrobial therapy: contemporary issues in infectious diseases, vol 1. New York: Churchill Livingstone; 1984:243.
16 Hunsaker B. Immune function of calves vaccinated intradermally against bovine herpesvirus type 1 [dissertation]. Texas A&M University, 1999.
17 Chin AC, Morck DW, Merrill JK, et al. Anti-inflammatory benefits of tilmicosin in calves with Pasturella haemolytica–infected lungs. Am J Vet Res. 1998;59:765.
18 Barcia-Macay M, Seral C, Mingeot-Leclercq M-P, et al. Pharmacodynamic evaluation of the intracellular activities of antibiotics against Staphylococcus aureus in a model of THP-1 macrophages. Antimicrob Agents Chemother. 2006;50:841.
19 Brumbaugh GW, Herman JD, Clancy JS, et al. Effect of tilmicosin on chemotactic, phagocytic, and bactericidal activities of bovine and porcine alveolar macrophages. Am J Vet Res. 2002;63:36.
20 Mirkin BL. Pharmacodynamics and drug disposition in pregnant women, in neonates, and in children. In: Melmon KL, Morrelli HF, editors. Clinical pharmacology: basic principles in therapeutics. ed 2. New York: MacMillan; 1978:127.
21 Papich MG, Davis LE. Drug therapy during pregnancy and in the neonate. Vet Clin North Am Small Anim Pract. 1986;16:525.
22 Fenger CK, et al. Equine protozoal myeloencephalitis: findings from a retrospective study. Proceedings of the Forty-Second Annual Convention of the American Association of Equine Practitioners. 1996:80.
23 Bedford SJ, McDonnell SM. Semen, testicular volume, sperm production efficiency, and sexual behavior of stallions treated with trimethoprim-sulfamethoxazole and pyrimethamine. Proceedings of the Forty-Fourth Annual Convention of the American Association of Equine Practitioners. 1998:1.
24 Crisman MV, Wilcke JR, Sams RA, Gerken DF. Concentrations of phenylbutazone and oxyphenbutazone in postparturient mares and their neonatal foals. J Vet Pharmacol Ther. 1991;14:330.
25 Langston VC, Sedrish S, Boothe DM. Disposition of single-dose oral enrofloxacin in the horse. J Vet Pharmacol Ther. 1996;19:316.
26 Beluche LA, Bertone AL, Anderson DE, et al. In vitro dose-dependent effects of enrofloxacin on equine articular cartilage. Am J Vet Res. 1999;60:577.
27 Giguere S, Sweeney RW, Belanger M. Pharmacokinetics of enrofloxacin in adult horses and concentration of the drug in serum, body fluids, and endometrial tissues after repeated intragastrically administered doses. Am J Vet Res. 1996;57:1025.
28 Kaartinen L, Panu S, Pyorala S. Pharmacokinetics of enrofloxacin in horses after single intravenous and intramuscular administration. Equine Vet J. 1997;29:378.
29 Dowling PM, Wilson RC, Tyler JW, Duran SH. Pharmacokinetics of ciprofloxacin in ponies. J Vet Pharmacol Ther. 1995;18:7.
30 Gingerich DA. Pharmacokinetics of drugs used for therapy of the mammary gland. Proceedings of the Tenth Annual Food Animal Medicine Conference: The Use of Drugs in Food Animal Medicine. Columbus: Ohio State University Press; 1985:117.
31 Ziv G, Shani J, Sulman FG. Pharmacokinetic evaluation of penicillin and cephalosporin derivatives in serum and milk of lactating cows and ewes. Am J Vet Res. 1973;34:1561.
32 Rasmussen F. Mammary excretion of benzylpenicillin, erythromycin, and penethamate hydroiodide. Acta Pharmacol Toxicol. 1959;16:194.
33 Ziv G, Sulman FG. Distribution of aminoglycoside antibiotics in blood and milk. Res Vet Sci. 1974;17:68.
34 Ziv G, Sulman FG. Penetration of lincomycin and clindamycin into milk in ewes. Br Vet J. 1973;129:83.
35 Ziv G, Sulman FG. Serum and milk concentrations of spectinomycin and tylosin in cows and ewes. Am J Vet Res. 1973;34:329.
36 Rasmussen F. Mammary excretion of sulphonamides. Acta Pharmacol Toxicol. 1958;15:139.
37 Stowe CM, Sisodia GS. The pharmacologic properties of sulfadimethoxine in dairy cattle. Am J Vet Res. 1963;24:525.
38 Sisodia CS, Stowe CM. The mechanism of drug secretion into bovine milk. Ann N Y Acad Sci. 1964;111:559.
39 Rasmussen F. Renal and mammary excretion of trimethoprim in goats. Vet Rec. 1970;87:14.
40 Available at www.fda.gov/cvm/fda/infores/AMDUCA/5340.html
41 AVMA backs up FDA’s defense of animal-drug regulation policies. J Am Vet Med Assoc. 1986;188:465.
42 Martinez MN. Article IV: clinical application of pharmacokinetics. Special series: use of pharmacokinetics in veterinary medicine. J Am Vet Med Assoc. 1998;213:1418.
43 Deckar WM, Peters PF, editors. Congressional committee alleges policies violate Food, Drug, and Cosmetic Act and encourage illegal use of drugs by veterinarians. Veterinary Medical News from Washington, July 31. American Veterinary Medicine Association; 1985:1.
44 FDA spells out extra-label use guidelines. J Am Vet Med Assoc. 1984;185:950.
45 Lofgreen GP. Mass medication in reducing shipping fever—bovine respiratory complex in highly stressed calves. J Anim Sci. 1983;56:529.
46 Walton JR. Principles of treatment of respiratory disease including herd medication. Pig Vet Soc Proc. 1982;9:71.
47 Paulson JA, Gordon IB, Mortimer EA. Prophylactic antibiotics. In: Kagan BM, editor. Antimicrobial therapy. ed 3. Philadelphia: Saunders; 1980:468.
48 Wilkowske CJ, Hermans PE. General principles of antimicrobial therapy. Mayo Clin Proc. 1983;58:6.
49 Orsini JA. Principles of antimicrobial selection for horses. Mod Vet Pract. 1986;67:719.
50 Rosenblatt JE. Laboratory tests used to guide antimicrobial therapy. Mayo Clin Proc. 1983;58:14.
51 Cox HU, Luther DG, Newman SS, Roy AF. Comparison of antibiograms determined by disk diffusion and microdilution methods for selected gram-negative bacilli. Am J Vet Res. 1981;42:546.
52 Washington JA. Susceptibility tests. In: Kagan BM, editor. Antimicrobial therapy. ed 3. Philadelphia: Saunders; 1980:11.
53 Ernst JD, Sande MA. In vitro susceptibility testing and the outcome of treatment of infection. Root RK, Sande MA, editors. New dimensions in antimicrobial therapy and contemporary issues in infectious diseases, vol 1. New York: Churchill Livingstone, 1984.
54 Hewitt WM. The agar diffusion assay. In: Microbiological assay: an introduction to quantitative principles and evaluation. New York: Academic; 1977:17.
55 McKellar QA, Sanchez Bruni SF, Jones DG. Pharmacokinetic/pharmacodynamic relationships of antimicrobial drugs used in veterinary medicine. J Vet Pharmacol Ther. 2004;27:503.
56 Micozzi A, Bucaneve G. Prophylaxis and treatment of bacterial infections: do we need new strategies. Rev Clin Exp Hematol. 2005;92:1.
57 Metlay JP. Antibacterial drug resistance: implications for the treatment of patients with community acquired pneumonia. Infect Dis Clin North Am. 2004;18:777.
58 Neu HC. Optimal antibiotic therapy in bronchopulmonary infections. Infection. 1980;1(Suppl):62.
59 Baggot JD, Love DN, Rose RJ, Raus R. Selection of an aminoglycoside antibiotic for administration to horses. Equine Vet J. 1985;17:30.
60 Brown SA. Minimum inhibitory concentrations and postantimicrobial effects as factors in dosage of antimicrobial drugs. J Am Vet Med Assoc. 1987;191:871.
61 Evans NA. Tulathromycin: an overview of a new triamilide antimicrobial for livestock respiratory disease. Vet Ther. 2005;6:83.
62 Bryant RE. Effect of the suppurative environment on antibiotic activity. Root RK, Sande MA, editors. New dimensions in antimicrobial therapy: contemporary issues in infectious diseases. New York: Churchill Livingstone; 1984;vol 1:313.
63 Sears PM, Fettinger M, Marsh-Salin J. Isolation of L-form variants after antibiotic treatment in Staphylococcus aureus bovine mastitis. J Am Vet Med Assoc. 1987;191:681.
64 Wormser GP, Keusch GT. Trimethoprim-sulfamethoxazole in the United States. Ann Intern Med. 1979;91:420.
65 Tuomanen E. Antibiotics which kill nongrowing bacteria. TIPS. 1987;8:121.
66 Guard CL, Schwark WS, Friedman DS, et al. Age-related alterations in trimethoprim-sulfadiazine disposition following oral or parenteral administration in calves. Can J Vet Res. 1986;50:342.
67 Shoaf SE, Schwark WS, Guard CL. The effect of age and diet on sulfadiazine-trimethoprim disposition following oral and subcutaneous administration to calves. J Vet Pharmacol Ther. 1987;10:331.
68 White G, Piercy DWT, Gibbs HA. Use of a calf salmonellosis model to evaluate the therapeutic properties of trimethoprim and sulfadiazine and their mutual potentiation in vivo. Res Vet Sci. 1981;31:27.
69 Sojka JE, Brown SA. Pharmacokinetic adjustment of gentamicin dosing in horses with sepsis. J Am Vet Med Assoc. 1986;189:784.
70 Davis LE: Adverse drug reactions in the horse, Vet Clin North Am Equine Pract 3:153, 987
71 Sawant AA, Sordillo LM, Jayarao BM. A survey on antibiotic usage in dairy herds in Pennsylvania. J Dairy Sci. 2005;88:2991.
72 Paul JW. Drug interactions and incompatibilities. Vet Clin North Am Equine Pract. 1987;3:145.
73 Davis LE. Pharmacology. In Veterinary values, ed 2, New York: Ag Resources; 1985:35.
74 Owen RH, Fullerton J, Barnum DA. Effects of transportation, surgery, and antibiotic therapy in ponies infected with Salmonella. Am J Vet Res. 1983;44:46.
75 White G, Prior SD. Comparative effects of oral administration of trimethoprim-sulfadiazine or oxytetracycline on the faecal flora of horses. Vet Rec. 1982;111:316.
76 Martin-Jimenez T, Papich MG, Riviere JE. Population pharmacokinetics of gentamicin in horses. Am J Vet Res. 1998;59:1589.
77 Root RK, Hierholzer WJ. Infectious disease. In: Melmon KL, Morrell HF, editors. Clinical pharmacology: basic principles of therapeutics. ed 2. New York: Macmillan; 1978:709.
78 Paulson JA, Gordon IB, Mortimer EA. Prophylactic antibiotics. In: Kagan BM, editor. Antimicrobial therapy. ed 3. Philadelphia: Saunders; 1980:468.
79 Van Scoy RE, Wilkowske CJ. Prophylactic use of antimicrobial agents. Mayo Clin Proc. 1983;58:241.
80 Watkins LE, et al. The prophylactic effects of monensin fed to cattle inoculated with Coccidia oocysts. Agri-Practice. 1986;7:18.
81 Bryans JT. Application of management procedures and prophylactic immunization to the control of equine rhinopneumonitis. Proceedings of the Twenty-Seventh Annual Convention of the American Association of Equine Practitioners. 1981:259.
82 Jakab GJ. Mechanisms of virus-induced bacterial superinfections of the lung. Clin Chest Med. 1981;2:59.
83 Sweet C, Smith H. Pathogenicity of influenza virus. Microbiol Rev. 1980;44:303.
84 Austin SM, Foreman JH, Hungerford LL. Case-control study of risk factors for development of pleuropneumonia in horses. J Am Vet Med Assoc. 1995;207:325.
85 Doll ER. Immunization against viral rhinopneumonitis of horses with live virus propagated in hamsters. J Am Vet Med Assoc. 1961;139:1324.
86 Foreman JH. Practical aspects of the use of ceftiofur sodium in the treatment of equine respiratory infections. Proceedings of the Thirty-Ninth Annual Convention of the American Association of Equine Practitioners. 1993:307.
87 Young C. Antimicrobial metaphylaxis for undifferentiated bovine respiratory disease. Comp Cont Educ (Pract Vet). 1995;17:133.
88 Camp TH, Stevens DG, Stermer RA, Anthony JP. Transit factors afflicting shrink, shipping fever and subsequent performance of feeder calves. J Anim Sci. 1981;52:1219.
89 Lofgreen GP. Mass medication in reducing shipping fever—bovine respiratory complex in highly stressed calves. J Anim Sci. 1983;56:529.
90 Lofgreen GP, et al. Energy level in starting rations for calves subjected to marketing and shipping stress. J Anim Sci. 1975;41:1256.
91 Martin SW. A mail survey of the efficacy of prophylactic medication in feed and/or water of feedlot calves. Can J Comp Med. 1985;49:15.
92 Perry TW, Beeson WM, Mohler MT, Harrington RB. Value of chlortetracycline and sulfamethazine for conditioning cattle after transit. J Anim Sci. 1971;32:137.
93 Perry TW, Riley JG, Mohler MT, Pope RV. Use of chlortetracycline for treatment of new feedlot cattle. J Anim Sci. 1986;62:1215.
94 Bennett BW, Rupp GP, McCormick RM. Pre-shipment preventive medication of calves with liquamycin at weaning. Agri-Practice. 1983;4:6.
95 Morck DW, Merrill JK, Thorlakson BE, et al. Prophylactic efficacy of tilmicosin for bovine respiratory tract disease. J Am Vet Med Assoc. 1993;202:273.
96 Booker CW, Schunicht OC, Guichon PT, et al. An evaluation of the metaphylactic effect of ceftiofur crystalline free acid in feedlot cattle. Vet Ther. 2006;7:257.
97 Kilgore WR, Spensley MS, Sun F, et al: Clinical effectiveness of tulathromycin, a novel triamilide antimicrobial, for the control of respiratory disease in cattle at high risk for developing bovine respiratory disease, Vet Ther 6:136
98 Rooney KA, Nutsch RG, Skogerboe TL, et al: Efficacy of tulathromycin compared with tilmicosin and florfenicol for the control of respiratory disease in cattle at high risk of developing bovine respiratory disease, Vet Ther 6:154
99 Lofgreen GP, Stinocher LH, Kiesling HE. Effects of dietary energy, free choice alfalfa hay, and mass medication on calves subjected to marketing and shipping stress. J Anim Sci. 1980;50:590.
100 Allen GP, Bryans JT. Molecular epizootiology, pathogenesis, and prophylaxis of equine herpesvirus-I infections. Prog Vet Microbiol Immunol. 1986;2:78.
101 Bryans JT, et al. Neonatal foal diseases associated with perinatal infection by equine herpesvirus I. J Equine Med Surg. 1977;1:20.
102 Bryans JT. Herpesviral diseases affecting reproduction in the horse. Vet Clin North Am (Large Anim Pract). 1980;2:303.
103 Coggins L. Viral respiratory disease. Vet Clin North Am (Large Anim Pract). 1979;1:59.
104 Doll ER, Wallace ME, Richards MG. Thermal, hematological, and serological responses of weanling horses following inoculation with equine abortion virus: its similarity to equine influenza. Cornell Vet. 1954;44:181.
105 Doll ER, Bryans JT. Immunization of young horses against viral rhinopneumonitis. Cornell Vet. 1963;53:24.
106 Doll ER, Bryans JT. Epizootiology of equine viral rhinopneumonitis. J Am Vet Med Assoc. 1963;142:31.
107 Vasseur PB, Paul HA, Enos LR, Hirsh DC. Infection rates in clean surgical procedures: a comparison of ampicillin prophylaxis vs. a placebo. J Am Vet Med Assoc. 1985;187:825.
108 Danziger L, Hassan E. Antimicrobial prophylaxis of gastrointestinal surgical procedures and treatment of intraabdominal infections. Drug Intell Clin Pharm. 1987;21:406.
109 Wilkowske CJ, Hermans PE. General principles of antimicrobial therapy. Mayo Clin Proc. 1983;58:6.
110 Riviere JE, Kaufman GM, Bright RM. Prophylactic use of systemic antimicrobial drugs in surgery. Compend Cont Educ (Pract Vet). 1981;3:345.
111 van den Bogaard AEJr, Weidema WF. Antimicrobial prophylaxis in veterinary surgery. J Am Vet Med Assoc. 1985;186:990.
112 Brumbuagh GW. Antimicrobial therapy of adult horses with emergency conditions. Vet Clin North Am (Equine Pract). 1994;10:527.
113 Sanitz EM, Jang SS, Hirsh DC. In vitro susceptibilities of selected obligate anaerobic bacteria obtained from bovine and equine sources to ceftiofur. J Vet Diagn Invest. 1996;8:121.