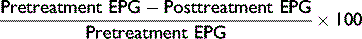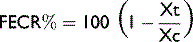Chapter 49 Parasite Control Programs
The traditional approach to parasite control programs has focused on using the appropriate anthelmintic at appropriate intervals. Parasitic disease in domestic animals is assumed to be the result of not dosing the animals often enough with anthelmintics. Scant attention has been paid to the interaction of the parasite with the host and the environment because of the reliance on anthelmintics. These drugs have been placed directly into the hands of the producer, as the expertise of a veterinarian did not seem necessary in the control of parasites. However, reports of resistance to anthelmintics and emergence of new manifestations of parasitism are surfacing throughout the world. It has become increasingly apparent over the past 20 years that this approach to parasite control is no longer sustainable.
Secondary to the notion that parasitism is under control is the decrease in research to develop new anthelmintics. Currently there is little in pharmacologic development other than variations on the current anthelmintics. Research programs in parasitology of domestic animals are facing funding reductions as research priorities are shifted to other diseases. As producers and owners struggle to deal with the realities of anthelmintic resistance, veterinary medicine must reassess traditional approaches to parasite control programs. Veterinarians will need to reeducate themselves away from the traditional tools of deworming, anthelmintic rotation, and pasture rotation. Integrated management strategies incorporating selective use of anthelmintic agents, enhancement of host immunity to parasitic infection, and grazing and environmental management have become increasingly important in the design of sustainable parasite control programs.
The impact of parasitic infection varies widely with geographic area and management system. General guidelines may be suggested for parasitic control, but it is inadvisable to adhere to any rigid anthelmintic schedules or even management recommendations. The best parasite control programs are those designed with the goals of the producer in mind, as well as the costs and returns of treatment. Other factors that must be considered include the animal’s environment, climatic variations, and geographic location. Although many producers and owners would like a “cookbook” approach to parasite control, these are rarely effective across the various management conditions. It is unfortunate that an epidemiologically and economically sound parasite control program designed for animals in one geographic area may be neither efficient nor effective in another location.
The most important concept in the design of sound parasite control programs is the interaction of the parasite with the host and the environment. An understanding of the life-cycle and epidemiology will suggest the most effective methods for parasite control. In this chapter parasite factors, host factors, and environmental factors affecting transmission and disease expression are discussed for each major class of parasites in each host species (horses, cattle, small ruminants). The methods of monitoring parasite infections and anthelmintic resistance are presented in detail. The classes of anthelmintics and their modes of action are discussed, and finally, coccidiosis in cattle and small ruminants is summarized at the end of the chapter.
EQUINE PARASITIC DISEASE
From the perspective of parasite control, horses should be divided into two age groups: adults and young horses under the age of 18 months. Small strongyles are, epidemiologically speaking, the principal parasite of the adult horse.1 The fecundity, rapid generation time, and emergence of anthelmintic resistance make control of small strongyles the primary focus of adult equine anthelmintic strategies. Parasite control in adult horses is also tailored to include tapeworms (Anoplocephala species), bots (Gastrophilus species), large strongyles (Strongylus species), and the equine pinworm (Oxyuris equi). In addition to the parasites that affect older horses, horses under 18 months of age are susceptible to the equine roundworm, Parascaris equorum. Horses under 6 months of age are also sporadically affected by the equine threadworm Strongyloides westeri.
Small Strongyles
The small strongyles consist of more than 40 species of nematode parasites primarily of the genus Cyathostoma. Small strongyles have a direct and completely enteral life-cycle in which adults produce strongyle type eggs that are indistinguishable from those of large strongyles.2,3 Eggs passed in the feces develop at a critical temperature range of 7.2° to 29.4° C (45° F to 85° F) to first-stage larvae (L1), which hatch and undergo continued development on pasture, becoming second-stage larvae (L2) then infective third-stage larvae (L3). Transmission is almost totally limited to pasture, with little infection thought to originate from stalls or dry lots. The rate of development is directly proportional to the environmental temperature; development takes as little as 3 days to several weeks at lower temperatures. L1 die quickly at higher temperatures, and freezing generally kills strongyle eggs. Resilience of the infective L3 is dramatically different owing to retention of the L2 cuticle, which protects from desiccation but also prevents continued feeding. Warm weather leads to rapid death of L3 as energy stores are depleted by activity in the absence of intake. In contrast, at very low temperatures energy depletion does not occur, and L3 remain viable in freezing conditions. Accordingly, L3 disappear quickly in hot, dry climates but remain viable in the winter.
L3 are ingested with herbage and exsheath in the small intestine, cecum, and ventral colon, where the majority enter a period of dormancy as early L3 (EL3) in the crypts and epithelial cells of the cecum and colon. Continued development through late L3 to fourth-stage larvae (L4) occurs within the cyst. Development to fifth-stage larvae (L5), the sexually mature, egg-producing stage, occurs in the gastrointestinal (GI) lumen. At any given time EL3 constitute the greatest proportion of cyathostomes.4,5 Cyst formation within the wall of the large intestine conveys some degree of protection from the immune response and from anthelmintic therapy.6 Practically speaking, limitations to drug diffusion may also be a factor in the generation of anthelmintic resistance for drugs with larvicidal activity. In contrast, anthelmintics that lack larvicidal activity should not be expected to exert resistance pressure in encysted forms. Therefore encysted stages remain effectively in refugia when nonlarvicidal anthelmintic therapies are used.
Encysted L3 are an important clinical entity for several reasons. There is evidence that seasonal signals as well as signals from lumen-dwelling mature forms delay development of encysted larvae, creating an important reserve for reinfection of the lumen. When environmental conditions are not favorable to larval development, large numbers of EL3 can remain dormant.4,5 As seasonal conditions become favorable, large numbers of hypobiotic larvae are signaled to exit and complete their life-cycle.7 In addition, lumen-dwelling adult small strongyles remain in balance with encysted forms such that anthelmintic killing of luminal parasites triggers a reemergence and repopulation of the lumen from the intramural reserve.8 Studies of small strongyle—parasitized ponies moved to a parasite-free environment indicate that the encysted stages can serve to reseed the lumen for at least 30 months of confinement under parasite-free conditions. Therefore it is obvious that under practical conditions of access to contaminated pastures, viable encysted larvae are present in the colon for years after ingestion, even in the absence of continued exposure. This would indicate that it is relatively difficult to “empty” horses of small strongyles.
Clinical syndromes associated with cyathostome infections have been extensively reviewed.1 The larval challenge dose, age, prior cyathostome exposure, and immunity of the host interact to determine the clinical picture. Most infections are asymptomatic with little response to encysted larvae. Clinical signs result from larval penetration into or emergence from the large intestinal mucosa.9 Despite an emphasis on diarrhea in most reports, weight loss is the primary clinical finding in horses with clinical cyathostomiasis. During an initial high-exposure infection, local irritation can result in decreased feed efficiency, anorexia, anemia, weight loss, and diarrhea as a result of local inflammatory reactions in the large intestine. Blood biochemistry and hematology may demonstrate neutrophilia, hypoalbuminemia, and hyperglobulinemia from 2 to 9 weeks after infection.9-11 Prior exposure tends to hasten the onset of laboratory abnormalities.
Larval cyathostomiasis is a potentially fatal disease syndrome that results from a synchronous emergence of encysted larvae that disrupt the mucosal barrier of the cecum and ventral colon.2,12 Fluid and protein transudation into the GI lumen and leakage of bacterial toxins into the vasculature are facilitated. All animals experience some degree of larval reemergence during winter or spring, but larval cyathostomiasis generally affects animals less than 6 years of age, when they have not yet acquired significant resistance to infection.12-14 Classic clinical signs include colic, which may be severe, impaired GI motility, sudden-onset diarrhea, and weight loss. Weight loss, fever, and dependent edema have also been reported in the absence of diarrhea.15 Some affected individuals may die acutely with few signs, whereas others may become emaciated and die over a period of 2 to 3 weeks.2,12
Signs consistent with larval cyathostomiasis have also been observed 7 to 10 days after administration of anthelmintics.16 This is presumed to result from a synchronous reactivation of hypobiotic larvae that is triggered by removing adult parasites from the GI lumen.8 It is conceivable that anthelmintic therapy during the period of seasonal cyathostome emergence superimposes signals for emergence on seasonal signals such that clinical disease is worse than either signal alone.
Cyathostome infection may also cause recurrent diarrhea in adult animals,17 a severe weight loss syndrome with associated edema and pyrexia in young horses,15 chronic weight loss leading to diarrhea,18,19 cecocolic intussusception,20,21 nonstrangulating infarction,19,22 cecal tympany.19
Controlling small strongyle infection is the primary goal of anthelmintic therapy in adult horses. Anthelmintic resistance in small strongyles is a growing problem, and parasite control strategies must be tailored to minimize anthelmintic use and maximize the generation of natural immunity.
Large Strongyles
Three species of large strongyles—Strongylus vulgaris, Strongylus edentatus, and Strongylus equinus—parasitize the horse. All three species have similar preparasitic phases. However, the migratory route of S. vulgaris makes it, on a per-worm basis, the most pathogenic of the enteric parasites of the adult horse. Both the life-cycle of and pathology caused by S. vulgaris have been comprehensively reviewed.70 Infective L3 are ingested from pasture. S. vulgaris L3 penetrate the mucosa of the small intestine, molt to L4 over 7 days, and then begin their arterial migration by penetrating the submucosal arteries. L4 travel via the cecal and colic arteries (by 14 days postinfection), reaching the root of the cranial mesenteric artery and its main branches by day 21 after infection. Larvae mature over a period of 3 to 4 months, then return primarily to the cecum and colon via the arteries. After a short period of maturation within the wall of the cecum and colon, the young adult parasites are released into the lumen of the intestine, where they mature in another 6 to 8 weeks. The prepatent period is 6 to 7 months.
Pathology and accordingly clinical signs of S. vulgaris infection result from the extensive larval migrations through the mesenteric arterial system.23-25 Larval migrations result in marked cellular infiltration and damage the endothelium of the arteries, leading to thrombus formation. The walls of all branches of the ileocecal colic artery are affected, and with chronicity the vessels become thickened and dilated with aneurysm formation. Disease results from thrombus showering of the bowel, leading to multifocal avascular necrosis in areas of the intestine that are supplied by the occluded arteries. Clinically affected horses show varying degrees of pain depending on the nature of the infarcts. Fever and serosanguineous peritoneal fluid with elevated protein and red and white blood cell counts are common.
The life-cycle of S. equinus was characterized by Slocombe.26S. equinus also invades the wall of the small intestine, cecum, and colon, causing formation of small cystic and hemorrhagic nodules where the organisms molt to L4. Twelve to 14 days after infection larvae exit the nodules, traversing the peritoneal cavity to the liver by 19 to 20 days after infection. Larvae remain in the liver for at least 12 weeks and then return to the large intestine by traversing the abdominal cavity directly or passing through the pancreas and then the abdominal cavity. By 15 weeks postinfection mature L5penetrate the lumen of the large intestine to complete the sexual phase of their life-cycle. The prepatent period of S. equinus is approximately 9 months.
S. edentatus L3 invade the walls of the terminal small intestine, cecum, and right ventral colon, where they penetrate the vasculature and then migrate to the liver via the hepatic portal vein.27-30 Larvae remain in the liver for approximately 40 days, during which time they molt to L4.28 Larvae then migrate via the hepatic ligament to the parietal peritoneum of the right flank and molt to immature adults between 13 and 15 weeks after infection.27S. edentatus organisms then return to the large intestine via the mesentery between 3 and 5 months after infection, migrating through the walls of the cecum and colon and causing the formation of parasitic nodules. Adults emerge into the large intestine to complete the sexual phase of their life-cycle. The prepatent period is generally acknowledged to be approximately 11 months, although some authors suggest it may be as short as 6 months.29
The pathology of S. edentatus and S. equinus is fairly restricted to hemorrhagic nodules and parasitic cysts of the large intestine.26-28 Such lesions are generally regarded as not severe enough to account for recognizable signs of colic. However, these lesions are evidence of poor deworming and a reasonable indicator that other large and small strongyle burdens are likely. Larval migrations through the liver may produce nodules and formation of fibrous tissue but are not considered clinically significant.
Although large strongyle infections are sporadically identified, they have been virtually eliminated in most areas through the widespread use of macrocyclic lactones and larvicidal fenbendazole regimens (10 mg/kg sid × five treatments) that kill both adults and migrating larvae.31 This reflects the protracted prepatent period (>5 months) of the large strongyles, coupled with limited survival of larvae in the environment. Simultaneous treatment of all horses on a premises with larvicidal anthelmintic regimens eradicates the parasite because despite continued ingestion of infective larvae from the pasture, the larvae never reach sexual maturity to produce eggs that can recontaminate the pasture. Therefore, given a maximal survival of large strongyle larvae of 12 months on pasture, larvicidal anthelmintics administered at 5-month intervals will effectively eliminate large strongyles from a premises within 18 months.
Tapeworms
During the last two decades the equine tapeworm, Anoplocephala perfoliata, has risen from clinical obscurity to be recognized as a significant potentiating factor in certain types of abdominal pain. The tapeworm life-cycle is indirect, cycling through oribatid mites, which horses swallow while grazing. Oribatid mites feed on organic material in feces on pasture, ingesting tapeworm eggs. Eggs develop into infective cysticercoids within the mite over a 2- to 4-month period. Tapeworms are hermaphrodites in that each infective form, the cysticercoid, contains the sexual organs of the male and female. After ingestion of cysticercoids by the horse, eggs develop in saclike body segments, termed proglottids, within 6 to 10 weeks.32 Proglottids then break away and pass in the manure. Detection of tapeworm eggs in manure is extremely insensitive for identification of tapeworm infestation, and this is thought to reflect an uneven distribution of the eggs within equine feces, associated with patchy distribution of the disintegrating proglottids.33-35 Accordingly, serologic diagnostic methods have proven advantages for detection of tapeworm infection.35
Three species of tapeworms have been identified in North America.36,37A. perfoliata, the most commonly identified, inhabits the region of the ileocecal valve. Anoplocephala mamilliana is the smallest and Anoplocephala magna the largest of the tapeworms that inhabit the small intestine and stomach. Postmortem investigations in Kentucky have indicated that the prevalence of tapeworm infections exceeds 50% of the population.38,39 Serologic evaluation of horses from 19 U.S. states representing 10 geographic regions chosen to encompass the entire United States indicated an overall prevalence of 54%, with a geographic distribution that was lower in the western United States, ranging from 1.3% in California to near 100% in Minnesota.40 Lower prevalence of tapeworm seropositive status in young animals and stallions was attributed to reduced access to pasture in these individuals.
Tapeworms infect horses of all ages, although a peak in worm burden has been identified in animals between 6 months and 2 years of age and in animals over the age of 15 years.41 Susceptibility to tapeworms appears to mimic susceptibility to small strongyles in that some horses develop immunity, others maintain small burdens, and a small percentage (10%) of horses maintain large tapeworm loads regardless of deworming.36,39,42-47Anoplocephala organisms cause severe inflammation, ulceration of the mucosa, edema, and scarring at their attachment sites, which can in some cases partially obstruct the lumen.48-50 This inflammation, in coordination with large quantities of acetylcholine found within A. perfoliata, is believed to interfere with normal peristalsis. Tapeworm burden increases the risk of spasmodic colic eightfold, and the risk of ileocecal impaction twenty-eight-fold.51-54 Tapeworms also increase the risk of ileocecal intussusception and cecal rupture.55-58
Effective recommendations for the control of equine tapeworms in the United States are compromised because epidemiologic studies have been confined to Kentucky and investigations of oribatid mites are lacking. Tapeworm infections occur year-round throughout the world.39,47,59-62 However, investigations in Spain, Switzerland, Sweden, and Kentucky have identified significant seasonal increases in A. perfoliata infection from late summer through early spring.30,59-61,63-65 Comprehensive examination of 372 horses from Spanish abattoirs also demonstrated that A. magna infections peaked in fall.60 Of importance, gravid A. perfoliata organisms were not evident in summer and displayed an increasing seasonal prevalence peaking in spring, suggesting that strategic use of cestodicidal drugs before spring may be especially effective in interrupting the life-cycle.
Praziquantel (1 mg/kg) and double to triple doses of pyrantel pamoate (13.2 to 19.8 mg/kg) are efficacious in eliminating tapeworms.66,67 Limited evidence suggests that pyrantel tartrate (2.64 mg/kg) for 30 consecutive days is also efficacious.68,69 In the United States praziquantel is available only in combination with moxidectin or ivermectin. Annual treatment with an efficacious product is recommended for horses beginning at weaning. Available epidemiologic findings suggest that annual treatments might be of greatest benefit in late fall to minimize burdens and prevent the development of gravid forms. However, on farms with a confirmed tapeworm burden, based on fecal counts, serologic evaluation of the herd, or a history of tapeworm-related colics, treatments two to three times per year may be justified, especially during the late summer through early spring.
Bots
Two primary species of bot larvae infect horses in North America.39,66,70,71Gasterophilus nasalis lays eggs in the intermandibular region, whereas Gasterophilus intestinalis eggs are found attached to the forelegs. By day 5, larvae hatch and migrate to the mouth in the case of G. nasalis or enter the mouth as the horse rubs and scratches the eggs with its muzzle and teeth in the case of G. intestinalis. First-stage G. intestinalis larvae embed deeply in the tongue and migrate to the interdental spaces of the upper molars, where they molt to L2, whereas G. nasalis larvae reside in the interdental spaces. L2 are swallowed and come to reside in species-dependent predilection sites, where they molt to L3. G. intestinalis organisms attach to the nonglandular lining of stomach in the region of the margo plicatus, and G. nasalis organisms attach to the most proximal portions of the duodenum, where they remain for up to 12 months. In late spring, larvae pass in the feces, pupate to adult flies over 3 to 9 weeks, and begin laying eggs again until fall. Fly activity ceases with the onset of cold weather.
Moderate Gasterophilus species infestations are asymptomatic.71 Large larval burdens in the tongue can cause inflammation and perhaps difficulty swallowing.63-65 However, this association is not well recognized. Heavy loads in the stomach may be associated with gastric ulceration, abscessation, and rarely stomach rupture and peritonitis.75,76
Gasterophilus infection is easily controlled through once-yearly administration of boticidal drugs, ivermectin, or moxidectin.77,78 Highest numbers of larvae are found in the stomach from winter through early spring.71 Accordingly, timing of treatment should be in late autumn or early winter. Daily grooming to remove bot eggs, which are yellow and easily visible, minimizes infection.
Pinworms
The parasitic phase of O. equi, the equine pinworm, begins with ingestion of an embryonated egg containing infective L3. Larvae hatch in the small intestine and come to inhabit the mucosal crypts of the cecum and colon. Maturation to gravid adults includes a lengthy maturation phase that ranges from 139 to 156 days, creating a prepatent period that approximates 5 months. Gravid females lay their eggs in clumps in a yellowish-grey gelatinous material around the anus. After completing their egg laying, the females pass out of the anus and die.
The primary clinical sign of O. equi infection is intense pruritus of the tail head that is referable to drying and cracking of the egg masses in the region of the anus. Tail rubbing facilitates deposition of the eggs in the environment. In severe infections, mild colic can result from inflammation of the cecum and colonic mucosa.
Benzimidazoles (BZDs), pyrantel pamoate, pyrantel tartrate, and the macrocyclic lactones are all efficacious against pinworms. Owing to the prolonged prepatent period, treatment regimens tailored to control ascarid or small strongyle infections will control O. equi infections. Frequent cleansing of the perineum will also limit spread.
Roundworms
Infection with the equine roundworm, P. equorum, is limited to young horses owing to the development of acquired lifelong immunity by 18 to 24 months of age.79 The high prevalence, size, fecundity, and persistence in the environment make P. equorum the most pathogenic parasite of the young horse. The life-cycle of P. equorum is direct, with a hepatotracheal migration and a prepatent period of 10 to 12 weeks.80,81 Mature ascarids range in length from 1 to 14 inches, and the females are extremely prolific, producing hundreds of thousands of eggs per day.80 These eggs are very sticky, enabling them to adhere to pasture and surfaces in the environment from which they are ingested. Ascarid eggs are also extremely resistant to chemicals and the environment, remaining viable for over 10 years and being capable of withstanding bleach, iodine, cresol, quaternary ammonium compounds, and steam cleaning.82 Over a period of weeks eggs mature to an infective stage in the environment characterized by the presence of L3, which is visible within the egg. After ingestion, larvae emerge from the eggs in the small intestine, penetrate the intestinal wall, and travel via blood or lymphatics to the liver. Larvae migrate within the liver for approximately a week; they molt to L4 before being carried, via the vasculature, to the lungs. Larvae break into the alveoli, ascend the bronchial tree, are swallowed and then mature to L5 and reproduce.
Clinical signs referable to adult ascarid infection include weight loss, stunted growth, rough hair coat, a pendulous pot-bellied appearance, lethargy, depression, and abdominal pain.83-85 Migratory stages of the parasite are commonly associated with respiratory signs including fever, coughing, and nasal discharge, which may be partially responsive to antimicrobial therapy but recur when therapy is discontinued.81,84 The most immediately life-threatening effect of ascarid infection is the ascarid impaction which is an obstructive mass of dead worms occurring shortly after deworming in the face of high ascarid burdens.86 Affected animals display signs of severe abdominal pain referable to small intestinal obstruction.
High fecundity and egg resistance make elimination of Parascaris from an infected premises virtually impossible. Ascarid burdens in weanlings often reach several hundred, yielding fecal egg counts (FECs) in the millions of eggs per gram feces (epg).80 Pasture is the primary route of exposure to ascarids, and accordingly clean pastures (see the discussion under Clean Pasture) should be prioritized for young animals and mares with foals. Young animals are also commonly exposed when they lick the infected stall environment. Despite the chemical and environmental resistance of ascarid eggs, efforts should be made to decrease the environmental load by frequently removing manure from stalls and pasture (before development of infective stages); feeding horses off the ground in feeders that can be cleaned; washing stall surfaces with detergent and phenol-based disinfectant (especially in stalls that have held foals and weanlings); and bathing the mare, including her udder, before foaling. Anthelmintic protocols tailored to the control of Parascaris will also adequately control most other intestinal parasites of the young horse with the exception of tapeworms.
Threadworms
S. westeri is a sporadic cause of diarrhea in foals before weaning. S. westeri is acquired primarily through lactogenic transmission, with infections being acquired during nursing in the first few days postpartum.87 Less commonly infection can be acquired from a wet environment.88 The life-cycle is completely enteral, with the parasite infecting the small intestine and producing eggs within 6 to 14 days of infection.87-89 Sources have reported acquired immunity to be complete by 6 to 12 months of age.88-90S. westeri transmission is significantly reduced, although not eliminated, by treating the mare with ivermectin at foaling.90 Foals can be treated empirically with either ivermectin or oxibendazole (15 mg/kg) within the first 2 weeks of life or based on positive fecal examination results.90-92
A New Paradigm for Equine Parasite Control
For at least two decades administration of anthelmintics has focused on the practice of “rotation” or deworming of all horses with drugs from different anthelmintic classes, each separated by a predictable interval. These regimens are not in the patients’ best interest for several reasons. First, rotation does not slow the progression of resistance and in fact actually selects for resistance to all drugs in the rotation.93-97 Rotational regimens also provide no venue to detect anthelmintic resistance, and, because an alternate class of dewormer is used sequentially, resistance may be masked by the regular substitution of efficacious products in the deworming schedule. Equally, the concept of “slow rotation,” in which a single anthelmintic is applied for an entire year, is not prudent because of the high prevalence of resistance among small strongyles.6 Slow rotation, without monitoring the effectiveness of the anthelmintic by assessing for a reduction in FECs, allows resistant parasites to propagate unchecked for a prolonged period. During this time, parasites that are sensitive to the anthelmintic are selectively killed, allowing the resistant parasites to dominate the population. Computer modeling has demonstrated that the most effective method of delaying resistance is the simultaneous use of two effective and chemically distinct anthelmintics.
Investigations focused on anthelmintic resistance have identified a phenomenon of individual variability in parasite susceptibility in many species including the horse. Certain individuals that are “more susceptible” or permissive to strongyle infection maintain higher quantitative FECs then their herdmates, despite identical exposures, and account for the majority of pasture contamination.98,99 Other individuals can be identified that limit the infection and pass few to no eggs in their feces. Calendar-driven deworming ultimately translates to anthelmintic overuse in animals whose immune response can limit the infection, whereas suboptimal parasite control may result when the same treatment regimen is applied to susceptible individuals. Anthelmintic overuse hastens development of anthelmintic resistance.100,101 Therefore the time-honored principle of simultaneous anthelmintic treatment of all herdmates is being rewritten to be simultaneous reevaluation of all herdmates and treatment of horses with moderate and high strongyle susceptibility as documented by elevated FEC.
The last two decades of anthelmintic research have determined that preserving a population of parasites termed refugia or said to be in refugia is critical to controlling anthelmintic resistance. The most basic biologic meaning of refugia is an isolated population of once widespread species. Refugia should be viewed as “wild-type” parasites that have not been selected for anthelmintic resistance. Accordingly, the genetics of parasites in refugia are critical to preventing dominance of anthelmintic-resistant strains. Refugia are protected during anthelmintic treatment of the host only when they are on pasture and, in the case of nonlarvicidal anthelmintics, when they are encysted. Because refugia are anthelmintic sensitive, a central tenant to their preservation is decreasing the frequency of anthelmintic use, especially when parasite numbers are low in the environment. Balancing a desire for decreased anthelmintic treatments with maintaining the health of the horse can be achieved only through the rational application of routine FECs to differentiate horses that need treatment from those that do not.6
Factors Influencing Parasite Control Strategies for Adult Horses
The primary goal of parasite control in adult horses is to minimize infective small strongyle larvae on pasture. A thorough understanding of three factors is critical to the proper timing and selection of anthelmintics. These factors are the load of infective larvae in the environment, the residual capacity of the anthelmintic agent, and the horse’s ability to limit egg excretion via an effective immune response.
Climactic conditions of a geographic region directly affect the lifespan of infective larvae and accordingly the load of infective larvae in the environment. This is exploited when developing a parasite control strategy by timing anthelmintic administration for the times of peak fecal egg production.102 Ultimately such strategic timing will minimize the number of anthelmintic treatments, which limits resistance by decreasing selection pressure. In contrast, anthelmintic treatment should be avoided when pasture refugia are diminished, because such treatments place the greatest selection pressure on the population.103 Pasture refugia are at their lowest numbers when climactic conditions limit the survival of infective larvae on pasture.
In warm temperate and subtropical or tropical climates such as the southern United States, refugia are at their lowest during the summer because larvae cannot survive the extreme heat of the southern climate, providing a period of grazing in the summer that is relatively free of exposure to small strongyles.103,104 In this region, peak fecal egg production occurs from autumn (September) through spring (April). It is important to recognize that infective larvae are present on pastures in the warm temperate, subtropical, and tropical regions of the southern United States throughout the winter months. In the northern cool temperate regions, refugia are at their lowest during the winter, whereas larval development is favored during spring, summer, and fall.103,105 Northern winters (November through March) do not support hatching of eggs nor larval development, although L3 that have already developed sufficiently to be competent for infection do persist during these months.105 This is important to recognize because rested pastures in northern climates remain infective until early summer, when rising temperatures cause the demise of L3. Despite the fact that infective larvae are present on pasture in northern climates during winter, management practices, in which the horses are stabled and their manure is removed, limit winter exposure to infective larvae. This reflects the requirement for a moist environment in order for strongyle larvae to develop to infective L3, conditions that are not achieved in the stall. Furthermore, ammonia from a dirty, wet stall environment is toxic to nematode larvae.106 Together these factors create a winter period in northern climates that is relatively free from exposure to infective strongyle larvae.
In addition to climactic factors, the residual ability of a given class of anthelmintics to suppress egg excretion must be considered. This characteristic of each anthelmintic is reflected in the egg reappearance period (ERP), which is the time after treatment that a horse’s feces will remain negative for strongyle eggs. The ERP has generally been reported to be on the order of 8 weeks for ivermectin107-110 versus 12 weeks for moxidectin.109 However, shortening of the ERP for ivermectin to 6 weeks has been demonstrated on some farms, raising concerns regarding reduction in efficacy and emerging resistance to macrocyclic lactones.97,110,112 For pyrantel the ERP is on the order of 4 to 6 weeks.108,110,112 ERP after BZD administration is on the order of 4 weeks, but periods as short as 2 weeks have been reported.112,113 Longer ERPs reflect the residual ability of an anthelmintic to prevent emergence and sexual reproduction of encysted small strongyle larvae. The clinician must recognize that BZD resistance is widespread in small strongyle populations, making monitoring of FEC reduction with these products especially important.6,114 From the standpoint of formulating an anthelmintic treatment regimen, the ERP is a useful interval for reevaluating FECs after anthelmintic therapy in order to determine if an individual horse requires subsequent anthelmintic therapy. Shortening of the ERP may also be an early indicator of reduction in anthelmintic efficiency.
The ability of a horse’s immune response to limit strongyle infections and egg production directly influences the intervals between anthelmintic administration. Strongyle resistance is reflected in an index termed the Strongyle Contamination Potential (SCP), which is defined as the FEC 4 weeks after the ERP of the previous anthelmintic. At this time residual anthelmintic effects are exhausted, and the ability to limit fecal egg production is reflective of the immunity of the host. SCP has been categorized as low (<150 epg), medium (150 to 500 epg), and high (>500 epg), corresponding to approximately 40%, 25%, and 35% of the population, respectively.112 FECs at the beginning of the parasite season (September in the south, April in the north) are also reflective of the relative immunity of the individual to small strongyles, because infective larvae are in low numbers on pasture at these times. Accordingly, FECs at these times are proportional to the individual’s tendency to “permit” the development and sexual reproduction of the few larvae that are ingested, or more significantly, hypobiotic strongyle larvae that are emerging to complete their life-cycle.
Parasite Control Strategy for Adult Horses
To be effective, parasite control strategies must be multifactorial and take into consideration the husbandry and dynamics of the premises or owner, ages of the patients, and epidemiology of the parasites. Anthelmintic resistance issues have highlighted the need to employ all measures that minimize pasture contamination. Strongyles develop from eggs to infective L3 outside the host, and simply removing feces from the environment before eggs become infective has been shown to provide parasite control that is superior to that of anthelmintic administration.115 Horses maintained in environments with fecal removal had lower FECs and grazing area was increased by 50% when compared with cohorts that were given anthelmintics. Pasture vacuuming and manual removal of feces from pasture may not always be feasible, and such measures have been dismissed in deworming schemes of the past. However, with anthelmintic resistance rising and with the need to minimize reliance on anthelmintics in order to protect refugia, the ability of manure removal to virtually eliminate anthelmintic use should cause responsible veterinarians to strongly encourage some degree of environmental management.
When targeted deworming is applied, the deworming season begins in September in warm temperate, subtropical, and tropical climates of the southern United States and in April in the cool temperate climates found in the northern regions of North America. Quantitative FECs are performed and reflect the small strongyle susceptibility of the individual horses because environmental loads are minimal at these times. Anthelmintic treatments are administered according to treatment thresholds, which range from 100 to 500 strongyle type epg (generally 150 to 200 epg). Evidence-based medicine indicates that these thresholds are efficacious in decreasing anthelmintic administration while preserving health.97,99,111,116 Lowering the treatment threshold imposes greater selection pressure for resistance and serves to limit immunity to small strongyles. After the initial evaluation and treatment at the beginning of the parasite season, FECs are repeated at intervals according to the ERP, with subsequent anthelmintic treatments being administered to only those individuals whose FECs exceed the treatment threshold. Within this program, the fall deworming should be with a macrocyclic lactone and praziquantel combination to target Gasterophilus species and Anoplocephala species. The cost of repeated fecal examinations may be concerning initially to owners employing targeted deworming. However, such regimens have been shown to be economically viable because they decrease anthelmintic use as much as 78%.97 Repeated evaluations of FECs indicate that individuals exceeding the treatment threshold at the beginning of the parasite season (or 4 weeks after the ERP of the previous anthelmintic) are likely to maintain elevated FEC and require repeated deworming at intervals consistent with the ERP. Animals with FEC below the treatment threshold should have FECs monitored according to the appropriate ERP and be dewormed when the treatment threshold is reached.
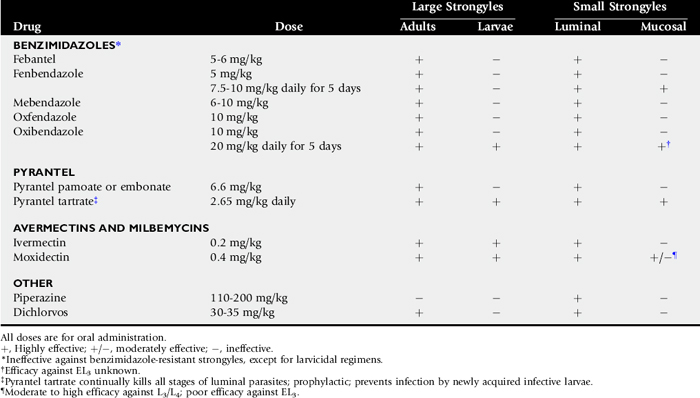
It is proposed that horses whose FEC exceeds 500 after the ERP or in the beginning of the parasite season (i.e., those with high SCP) should be singled out for larvicidal anthelmintic therapy.112 See Table 49-1. This reflects the fact that these animals are permissive to the infection, allowing both pasture-derived larvae and emerging encysted larvae to complete their life-cycle and produce eggs that contaminate the pasture. These horses are a reservoir for contaminating the environment, harboring large numbers of hypobiotic larvae that repopulate the GI lumen when adults are removed by deworming. Both moxidectin and “larvicidal dose” fenbendazole (10 mg/kg for 5 days) have larvicidal activity against encysted small strongyles, making these agents especially useful in managing individuals that are highly permissive to strongyle infections.117-119 However, it is important to recognize that neither moxidectin nor larvicidal dose regimens of fenbendazole are 100% efficacious. A report of small strongyle resistance to larvicidal fenbendazole doses in Kentucky yearlings should prompt caution in the use of such protocols because they exert extreme resistance pressure.120
A cornerstone of every deworming program is to identify anthelmintic resistance. Anthelmintic resistance is of tremendous concern in small strongyles, in which resistance to all commonly used anthelmintics, with the exception of the macrocyclic lactones, has been identified.6 Shortening of the ERP is an important indicator of anthelmintic resistance in the horse.121 However, the gold standard for detecting resistance has been the FEC reduction test (FECRT). FEC reduction (FECR) is determined by comparing FEC before and 10 to 14 days after anthelmintic administration. The percentage of reduction is calculated using the following formula:
The FECRT has several problems. First, the action levels to identify anthelmintic resistance have not been determined for equine parasites, so values are extrapolated from other species. Second, the FECRT is relatively insensitive, meaning that when anthelmintic resistance is identifiable by FECRT, resistance genes are widely disseminated within the parasite population. At this time egg reductions in excess of 90% are considered evidence of BZD and tetrahydropyrimidine efficacy. Values of 80% to 90% raise suspicion of resistance, and FECRs less than 80% indicate that resistance is present. In the case of the macrocyclic lactones ivermectin and moxidectin, egg reductions less than 98% are cause for concern.6,122
The primary concern with new additions to the herd is their ability to introduce resistant strongyles to a previously sensitive population.6 In this respect it is especially important that farms without anthelmintic resistance take precautions to prevent the introduction of resistant strongyles. FECRT should be performed in conjunction with the initial dewormings, and larvicidal treatment regimens should be selected to kill encysted parasites, which might bear resistance genes. Both moxidectin and larvicidal regimens of fenbendazole may be used. However, the widespread prevalence of BZD resistance in small strongyles must be considered because larvicidal doses of fenbendazole may be inferior to moxidectin to prevent the introduction of resistant worms in new additions. Accordingly, single-dose administration of a macrocyclic lactone has been advocated for new arrivals after fenbendazole larvicidal regimens to remove remaining luminal worms. New additions should be quarantined until appropriate response to anthelmintic therapy, as supported by a reduction in FECs, is available to substantiate that resistant parasites will not be introduced to the herd. On the other hand, horses staying less than 6 weeks may be efficaciously treated with ivermectin because ivermectin resistance is extremely uncommon and the ERP for ivermectin is 6 to 8 weeks.
PARASITE CONTROL STRATEGIES FOR YOUNG HORSES
S. westeri is the earliest maturing nematode of foals, passing eggs by 10 to 14 days, followed by small strongyle eggs which can be detected at 6 weeks.3,87-89 However, by virtue of its prevalence, size, fecundity, and pathogenicity in young horses, management of P. equorum is the primary focus of deworming strategies for horses under 18 months of age. Unfortunately, studies to identify treatment thresholds based on FEC, expected ERPs, and FECRT thresholds to identify resistance are not available. Anthelmintic strategies for controlling P. equorum should be expected to change as such data become available. In the absence of these data, current recommendations focus on interval administration designed to prevent egg production, which is known to exert significant anthelmintic resistance pressure in nematodes. Accordingly, it is prudent to document the presence of P. equorum on a premises by examining the feces for characteristic eggs. Four- to 6-month-old foals have been shown to have the highest P. equorum FECs, making them excellent sentinels.123 Parasite control programs in young horses on farms that lack P. equorum should focus on small strongyles.
Several published reports show poor reductions in ascarid FEC after both ivermectin and moxidectin, indicating macrocyclic lactone resistance.110,123,124 Daily exposure to low doses of pyrantel tartrate and monthly deworming with ivermectin are being scrutinized for their role in perpetuating anthelmintic resistance in equine parasites.6,122 Daily administration of pyrantel tartrate has been shown to interfere with the development of acquired immunity to small strongyles.125 Although the effect of daily pyrantel administration on immunity to ascarids is not known, inhibition of invasion and migration that are characteristic of this therapy could also limit immunity to ascarids. This is especially concerning in horses that have been maintained on daily dewormer and then introduced to heavily contaminated premises. In such horses the lower innate immunity to the parasite could increase morbidity associated with infection.
Monthly deworming with ivermectin has been advocated to decrease lung pathology in young horses caused by ascarid migration. However, ascarid resistance to ivermectin raises concern regarding the selection pressures imposed by monthly ivermectin administration. Accordingly, practitioners should monitor vigilantly for evidence of anthelmintic resistance in young horses where repeated ineffective deworming could yield burdens capable of causing an ascarid impaction on subsequent deworming with an efficacious product. A link between emerging ivermectin resistance in P. equorum populations and surgical ascarid impactions has been postulated.126Resistance should be considered a possibility with any anthelmintic agent, particularly when a single drug is used repeatedly. Guidelines for FECRT identification of P. equorum resistance are not available. However, FECR for both BZDs and pyrantel have been shown to reach 100% in ivermectin-resistant P. equorum populations, suggesting that values previously described for small strongyles are reasonable choices.124
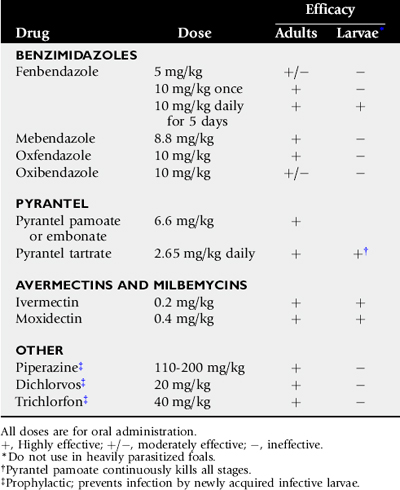
BZDs, tetrahydropyrimidines (pyrantel pamoate and pyrantel tartrate), and the macrocyclic lactones (ivermectin and moxidectin) are generally considered efficacious against ascarids. Moxidectin products are not approved for use in foals under 6 months of age. Recognize that doses of BZDs required to effectively kill ascarids are double the doses recommended to kill parasites of adult horses. This highlights the need for accurate weights to ensure proper administration, which is a problem in young horses, in which body weight is often estimated. This problem can be alleviated by use of weight tapes.
Although the timing of the initial anthelmintic treatment is influenced by the environmental load of P. equorum, treatment on endemic farms should begin at 8 to 10 weeks of age, which is the minimal prepatent period after initial exposure.80,81,127 Recognize that in addition to ascarid control, ivermectin administration should be incorporated at 5-month intervals if large strongyles are present on the farm. One of these treatments should be an ivermectin and praziquantel combination in the fall to kill Gasterophilus and Anoplocephala species. Both BZDs and pyrantel pamoate have a slightly shorter duration of ascarid egg suppression than the macrocyclic lactones because they do not kill migrating ascarids. The treatment interval with these compounds is 56 days, which is the minimum time required for migrating larvae not killed by BZD or pyrantel treatment to reach the intestine and produce eggs. More frequent administration of BZD or pyrantel maximizes resistance pressure without altering egg production unless shortening of the ERP occurs.
Macrocyclic lactones (restricted to ivermectin administration in foals) and larvicidal fenbendazole regimens (10 mg/kg × 5 days) have the advantage of killing all ascarids in the intestine as well as migrating larvae outside of the intestine.128-129 See Table 49-2. Therefore treatment intervals shorter than 56 days with larvicidal drugs are still efficacious in the control of ascarid infections but maximize resistance pressure. On farms with a high P. equorum burden, ivermectin may be administered as early as 45 days of age. Treatment intervals as short as 30 to 45 days with larvicidal drugs have been recommended by some practitioners on farms with high ascarid loads to minimize lung pathology. These recommendations should be critically evaluated in light of the resistance pressure imposed by the practice. Treatment intervals after administration of larvicidal agents should approximate 70 days because the foal is essentially cleared of all ascarids and begins the cycle of infection anew after treatment. Commonly the recommended ivermectin interval is shortened to 60 days for brevity in communication. Regardless, larvicidal drugs should be followed by retreatment before 70 days, and nonlarvicidal agents require retreatment in 56 days. Treatments are repeated until the horse develops solid acquired immunity as supported by negative ascarid fecal examinations, which generally occurs by 18 months of age. Failure to remain within these intervals risks egg contamination of the pasture that persists for years.
Ascarid eggs are extremely resilient, and infection occurs from both pasture and fomites in the stall environment. Emerging anthelmintic resistance in P. equorum highlights the need to institute management practices that prevent ingestion of parasite eggs. Clean pastures (see below) should be provided for young animals and mares with foals. Manure should be removed at least twice weekly from stalls and pasture, before infective larvae can develop. Horses should be fed off of the ground in feeders that can be cleaned. Although ascarid eggs already in the environment cannot be eliminated, washing stall surfaces, especially those that have held foals and weanlings, and bathing the mare including her udder before foaling help to minimize egg contamination.
A word of caution: if high ascarid burdens are suspected based on a prior poor deworming history or high FECs, deworming with highly efficacious anthelmintics is contraindicated because of the risk of ascarid impaction. In such cases young horses should be initially dewormed with an anthelmintic of lower efficacy, such as fenbendazole at a dose of 5 mg/kg, followed in 1 to 2 weeks with proper doses of an anthelmintic known to be efficacious on the premises.
CLEAN PASTURE
Creating a pasture that is entirely devoid of parasites is impossible. However, as part of a comprehensive parasite control program the following measures will reduce the parasite burden on pasture.130 Pastures that have been vacant for at least 2 months during the warm season, fields that have recently produced hay, and pastures grazed by alternate livestock species can be viewed as having a reduced parasite burden. Dragging and harrowing disperse parasite larvae, which is advantageous only in the summer and only if pastures can be left unoccupied for 2 weeks in the south or 4 weeks in the northern climates. Pastures should not be harrowed after October 1 in the United States because parasite larvae dispersed by harrowing will not undergo the climate extremes required to kill them. Similarly, manure should not be spread on pasture. Reducing the stocking density of a pasture will decrease parasite exposure because the horses are not forced to graze as close to their feces in order to meet their forage demands. Often, decreasing the number of horses on a pasture is not practical, but even in such cases useful pasture can be maximized and larval burdens minimized by removing feces from the pasture every few days. This interval exploits the nematode parasite’s requirement for a period of development outside of the horse in order to become infective. Removal of feces before this maturation prevents infection.
Less Common Equine Parasites
STOMACH WORMS
Four species of nematode parasites inhabit the equine stomach.131-135 These are Habronema muscae, Habronema microstoma, Draschia megastoma, and Trichostrongylus axei. The first three are collectively termed spiruroid species in that they belong to the nematode superfamily Spiruroidea. Habronema and Draschia cycle through intermediate hosts, the flies Musca domestica and Stomoxys calcitrans. L1 exit from embryonated eggs of infected horses during transit through the intestinal tract and are ingested by maggots in the environment. Larvae mature within the pupating fly; the life-cycle is completed within 2 weeks, and the adult flies emerge. L3 migrate to the proboscis, and horses become infected when the flies are swallowed or the larvae are released on or around the muzzle, eyes, sheath, or open wound as the flies feed. The prepatent period for D. megastoma and Habronema species is approximately 2 months. Fourth-stage larvae and adults localize in the glandular stomach, predominantly in the region of the margo plicatus.
The most common pathology caused by spiruroid nematodes is a result of larvae that do not access the GI tract but are instead deposited in wounds or moist areas of the skin, where the parasite is therefore unable to complete its life-cycle.136,137 During their migrations within the skin the larvae elicit tremendous eosinophilic responses characterized by the formation of eosinophilic granulomas. These granulomatous lesions, known as cutaneous habronemiasis or summer sore, consist of rapidly proliferating granulation tissue that is refractory to treatment and tremendously pruritic. Accordingly, lesions are generally ulcerated. Within the GI tract, D. megastoma, the most pathogenic of the species, causes submucosal eosinophilic granulomas near the margo plicatus that coalesce and later develop into large fibrous masses with purulent cystic cavities.131Habronema species stimulate the secretion of large amounts of thick tenacious mucus on the glandular part of the stomach, close to the margo plicatus, with adult worms embedded in the mucus.
The prevalence of spiruroid nematodes in horses has not been examined in the United States since 1985 to 1986, when a tremendous decline in prevalence was identified and attributed to the introduction of ivermectin in the United States in 1983.133 Before ivermectin’s introduction D. megastoma was the most prevalent of the spiruroid nematodes with a regional prevalence ranging from 24% to 60%.134,135,138,139 However, after ivermectin introduction D. megastoma prevalence rapidly declined to less than 5% of the population of horses examined in Kentucky.133 At this time spiruroid parasite infections are sporadic because of widespread use of avermectins.
Collectively the stomach parasites are controlled with the administration of macrocyclic lactones.131 Spiruroid transmission wanes in the cold, when the intermediate host is no longer active. Therapy in the fall, which also coincides with the timing for treatment of bots, will interrupt the life-cycle. Elimination of spiruroid parasites from granulomatous summer sore lesions is more challenging owing to limited diffusion of the anthelmintic into the fibrous capsule of the lesions.136 Accordingly, topical preparations consisting of organophosphates (OPs) and corticosteroids are commonly used in association with oral avermectin administration. Repeated treatments are commonly required.
Equine infection with the stomach worm T. axei is generally associated with shared grazing between horses and ruminants. T. axei also infects pigs. The life-cycle of T. axei closely resembles that of the strongyles. Exposure begins in the spring when infective T. axei L3 that survive winter are ingested during grazing. Pasture contamination wanes during the summer. L3 develop into adults in the lumen of the mucosal crypts or deeply in the mucosa of the stomach. The prepatent period is approximately 3 weeks. Light infections with T. axei are generally asymptomatic, but heavy infections lead to a hyperplastic reaction of the glandular tissue, predominantly in the fundus, and production of abundant mucus.132 Raised plaques enlarge, reaching several centimeters in diameter, and become eroded in the center as the disease progresses, appearing as reddened ulcers surrounded by hypertrophied gastric mucosa. In large numbers T. axei can trigger a severe watery diarrhea.
The epidemiology of T. axei transmission mimics that of strongyles in that infective L3 die off during hot, dry weather, effectively eliminating transmission during the summer months, but larvae that reach pasture in late summer can be infective or overwinter to infect in the spring. Accordingly, T. axei infections can be controlled by several methods depending on the husbandry of the farm. Infected horses can be treated with macrocyclic anthelmintics before their introduction to pastures that have not been grazed by ruminants or just before they are introduced to ruminant-grazed pastures after the summer drop in L3 pasture contamination. Horses that cannot be removed from infected pasture will require frequent deworming to minimize pasture infection during the spring, fall, and winter, as the prepatent period for T. axei is 3 weeks.
LUNGWORMS
See the discussion of lungworm infection in large animals.
ONCHOCERCA CERVICALIS
Unlike the previously discussed parasites that inhabit the GI tract, Onchocerca cervicalis is a filarid nematode whose adult organisms are found woven within the nuchal ligament.140 This predilection site, which has little blood supply, makes it impossible to eliminate the adult parasite.141 Microfilariae resulting from sexual reproduction tend to congregate in certain regions of the body including the ventral midline and face where they are ingested when Culicoides feed in these regions.142 From this intermediate host, microfilariae complete their life-cycle when they are transferred to another horse. O. cervicalis microfilariae cause pruritus, which may be severe in certain individuals and mild in others.141 Such pruritus does not appear to be as dependent on microfilarial burden as on the reactivity of the individual.
Onchocerca microfilariae are effectively eliminated by the macrocytic lactones.143,144 Complete resolution of signs may require 30 days, and recurrence of signs after treatment is not uncommon. Recurrence has been attributed to death of the microfilariae. In the absence of regular administration of macrocytic lactones, recurrence of signs is predictable owing to continued microfilaria production by the adult parasite.
GASTROINTESTINAL NEMATODE INFECTIONS IN CATTLE
Cattle are hosts to numerous species of nematodes.145-147 Of these, nematodes in the genera Ostertagia, Haemonchus, Trichostrongylus, and Cooperia are most prevalent and usually are considered to be the most important of the nematode species. Mixed infections are the rule and even though some genera, such as Nematodirus, Oesophagostomum, and Trichuris, comprise a smaller portion of the total nematode population, their presence contributes to the overall assault on the animal’s health and well-being. The various nematodes do differ somewhat with respect to their site of infection and pathologic effects, but their general life-cycle patterns are quite similar.
Life-Cycle
Adult female nematodes produce eggs that are passed out of the host with the feces. Under optimal conditions in the external environment, first-stage larvae (L1) can develop and hatch from eggs within 24 hours. L1 grow and develop to second-stage larvae (L2), which in turn grow and develop into third-stage larvae (L3). In general, the third stage is the infective larval stage. After ingestion, L3 develop into fourth-stage larvae (L4), which then develop into immature adults. Sexually mature adult nematodes develop within 2 to 4 weeks after ingestion of the L3 unless arrested development occurs. The life-cycle of Nematodirus is the same except that development to infective L3 occurs within the egg before hatching. For Trichuris, development to the infective L1 occurs in the egg. However, rather than hatching in the external environment, L1 hatch after ingestion of the eggs by the animal, and approximately 8 weeks are required before sexually mature adults are present.147,148
Climate and management of pastures and animals are among the numerous factors that influence the level and extent of parasitism. Although temperature is considered to be the driving force behind larval development, larval development can proceed only in the presence of adequate moisture. Larvae of all stages can be killed by extremely low temperatures, desiccation, and/or exposure to direct sunlight. Larval development and transmission tends to occur in predictable seasonal patterns based in part on regional climatic differences.146,147,149 In the southern United States, infective L3 persist longest when conditions are cool and wet (October to May) but die off quickly during the summer after rain-induced liberation from the fecal pat.150,151 Nematodes acquired by grazing cattle during the summer months came from eggs recently deposited on pasture. In the northern United States, infective L3 may be on pasture year-round. Significant numbers of Cooperia and Nematodirus may be acquired for at least 12 months after deposition of eggs on pasture, with acquisition of fewer numbers for up to 24 months. Acquisition of low levels of Ostertagia can occur for at least 14 months after deposition of eggs. In subtropical climates, seasonality may be much less marked, and pasture infectivity may follow the rainfall pattern. In arid climates, large numbers of larvae may be present on the pasture whenever local conditions permit lush grass growth.147,152-154
Not only can larvae survive on pasture, but some species can arrest development within the host. This usually occurs during the season when adverse environmental conditions would decrease larval survival in the external environment. Best known for this phenomenon is Ostertagia ostertagi. In northern temperate climates, pasture larval populations peak in the summer and early fall, and L4 tend to overwinter in the host, resuming development in the spring. In warmer climates with hot, dry summers, the highest numbers of infective larvae may be found in the late spring to early summer, and L4 tend to oversummer in the host, resuming development in the fall.147,155,156
Pathophysiology
Of all the cattle nematodes, O. ostertagi has long been considered to be the most pathogenic nematode in temperate regions. The pathophysiology of ostertagiasis centers around the development of larvae in the gastric glands of the abomasum. As the larvae develop within the glands, they cause gland hyperplasia and intense eosinophilic infiltration. Mucosal glandular cells lose their differentiation, and cell junctions are weakened. Albumin is lost into the lumen. Parietal cells cease to function, causing a decrease in HCl production. The change in pH stimulates overproduction of gastrin, which initiates cell proliferation and hyperplasia. Alkalinity also decreases the bactericidal activity of the abomasum, resulting in bacterial overflow from the rumen into the intestine. In addition a pH greater than 5 prevents the conversion of pepsinogen to pepsin. As a result, pepsinogen is released into the blood through permeable cell junctions. Hyperplasia and loss of cell differentiation become widespread and create the typical “Moroccan leather” appearance of the abomasum. In experimental models, Ostertagia infection in calves is associated with elevated peripheral eosinophil counts and decreased lymphocyte counts. Emergence of the larvae may complete the destruction of the glands. If the infection is severe, the proliferated cells and abomasal mucosa may slough, producing a diphtheritic membrane.149,157
Populations at Risk
Although exposure to some GI nematodes (GINs) readily induces immune responses that limit future populations of nematodes within the gut, cattle remain susceptible to O. ostertagi for many months. Protective immunity is usually not evident without prolonged exposure and may not occur until the animals are 2 years of age or older.149 Consequently, clinical type I ostertagiasis occurs primarily in young cattle (up to approximately 18 months of age) during their first grazing season, with type II disease present in older animals (2 to 4 years of age). After their initial exposure and induction of immunity, adult cattle rarely show signs of nematode infection or require anthelmintic treatment. Although mature cattle ingest infective larvae, fewer larvae establish infections, so parasite burdens and the magnitude of fecal egg shedding generally are decreased. Most preventive and treatment strategies therefore are directed at young grazing stock, primarily beef calves and dairy replacement heifers.
Clinical Manifestations
In young animals, GINs may simply cause poor growth and ill thrift, or they may cause serious clinical illness and even death. Inappetence, a common feature of parasitic gastroenteritis, can result in reduced weight gain, growth, and onset of puberty.
The synchronous development and maturation of inhibited larval O. ostertagi can result in severe clinical disease, called type II ostertagiasis. Usually seen in cattle 2 to 4 years of age, it occurs months after ingestion of infective larvae. Anorexia, ill thrift, and hypoproteinemia are consistent signs. The animals may also show fever, diarrhea, anemia, and submandibular edema. The prognosis for recovery is guarded owing to the widespread destruction of abomasal glands. Conversely, type I ostertagiasis results from the rapid acquisition of large numbers of larvae that complete development to the adult stage within the usual 3-week time frame. The primary physiologic change is appetite suppression, which accounts for the reduction in weight gains in calves during their first grazing season. Although the underlying mechanism for the two types is the same, the seasonal occurrence of each type varies in accordance with the epidemiologic patterns of the area.145,146,158
Compounding the effects of O. ostertagi is the presence of other GINs. Larval and adult Haemonchus organisms are blood feeders, producing anemia. T. axei produces local and systemic changes similar to those produced by O. ostertagi, resulting in similar clinical signs. Infection with Oesophagostomum radiatum produces structural and functional changes including anemia, hypoproteinemia, diarrhea, anorexia, and weight loss.146,159
Control of Gastrointestinal Nematodes
ANTHELMINTICS
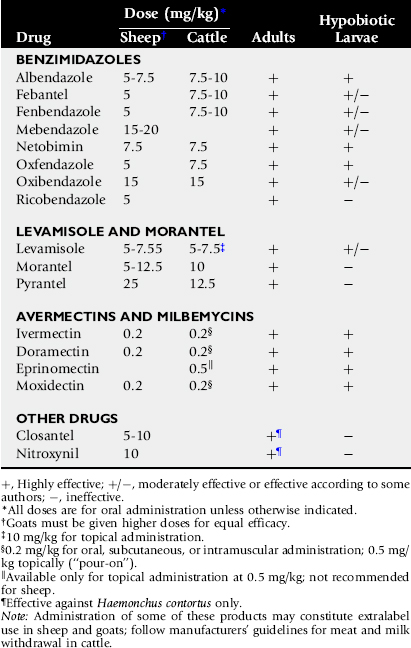
Adult Ostertagia and other GINs are susceptible to most of the commonly used anthelmintics. Drugs and doses are listed in Table 49-3. Drug withdrawal times must be considered when selecting anthelmintics for beef cattle and lactating dairy cows, and the manufacturers’ recommendations followed. Eprinomectin and moxidectin, in topical formulations, have no withdrawal period for either meat or milk.
The newer macrocyclic lactones are particularly effective against both adult and larval stages of the various GINs in cattle, including inhibited L4.160-162 In addition, their residual effect helps minimize pasture infectivity during the grazing season (see later).163-166 Intraruminal sustained-release devices (SRDs) containing a BZD, levamisole, morantel, or ivermectin can also be highly effective at limiting both clinical disease and pasture infectivity.167-171 Use of highly efficacious drugs or SRDs can attenuate the immune response to GINs during the first grazing season in calves. However, in most cases immunity is sufficient to prevent clinical disease during the following grazing season, and weight gains by the end of the second season are similar to those of immune animals.168,169,171
Although the macrocyclic lactones are all highly effective, some differences between products in both efficacy and duration of effect are apparent. Doramectin, eprinomectin, and moxidectin are more effective and/or have a longer residual effect than ivermectin against Ostertagia and Cooperia.172-178 Doramectin, eprinomectin, and moxidectin have a residual effect against Ostertagia for approximately 5 weeks, whereas ivermectin is effective for only 2 to 3 weeks.165,178 As the prepatent period for Ostertagia is approximately 3 weeks, the treatment intervals in most management situations are 8 weeks for doramectin, eprinomectin, and moxidectin and 5 to 6 weeks for ivermectin. Duration of effect also varies somewhat with the parasite and the level of infection. Persistence of effect against Cooperia appears to be 1 to 2 weeks shorter than for Ostertagia, regardless of the product used.164,165,178-180 The residual effect may be shortened by a week or so at high infection levels.178,179
Historically, anthelmintic resistance by GINs has been far less of a problem in cattle than in sheep or goats. However, this situation appears to be changing. Reports of anthelmintic-resistant nematodes of cattle are on the increase in the United States and elsewhere around the world.149,181-187 Resistance has been reported in the most frequently used anthelmintic classes including the BZDs, macrocyclic lactones, and imidazothiazoles. Species of Cooperia are most commonly associated with these reports, although resistant species of Ostertagia and Haemonchus have also been documented.
Treatment Intervals
The choice of drug and treatment interval should be made on the basis of an individual herd or farm, as part of an overall control program. Factors to consider include the geographic location, time of year, and grazing management. Grazing management is discussed elsewhere.188 There are several options for preventing clinical disease and maximizing gains in first-season grazing calves using strategic anthelmintic treatments:
 Two treatments with an avermectin or moxidectin early in the grazing season. The first treatment can be given either at turnout or weaning or 3 weeks into the grazing period. Depending on the product used, the second dose is given 6 weeks (ivermectin) or 8 weeks (doramectin, eprinomectin, moxidectin) later. This strategy can prevent clinical disease, keep FECs low, and increase weight gains during the first grazing season.189,190 An alternative when using ivermectin in situations in which pasture infectivity is high is to treat calves at 3, 8, and 13 weeks after turnout or weaning.191
Two treatments with an avermectin or moxidectin early in the grazing season. The first treatment can be given either at turnout or weaning or 3 weeks into the grazing period. Depending on the product used, the second dose is given 6 weeks (ivermectin) or 8 weeks (doramectin, eprinomectin, moxidectin) later. This strategy can prevent clinical disease, keep FECs low, and increase weight gains during the first grazing season.189,190 An alternative when using ivermectin in situations in which pasture infectivity is high is to treat calves at 3, 8, and 13 weeks after turnout or weaning.191 Use of an intraruminal SRD, where available, at turnout or weaning. This strategy may be most cost-effective on farms where pasture infectivity is high.
Use of an intraruminal SRD, where available, at turnout or weaning. This strategy may be most cost-effective on farms where pasture infectivity is high. Treatment during peak pasture infectivity (e.g., summer and early autumn in temperate climates). Treatment interval depends on the product being used: every 3 weeks for nonivermectin drugs, every 5 weeks for ivermectin. For example, ivermectin can be given at 10, 15, and 20 weeks after turnout or weaning. This strategy prevents clinical disease in the majority of calves while allowing some level of infection, which stimulates an immune response in first-season calves. However, treatment at the start of the grazing season has been shown to result in better weight gains than tactical treatments given during the grazing season.167
Treatment during peak pasture infectivity (e.g., summer and early autumn in temperate climates). Treatment interval depends on the product being used: every 3 weeks for nonivermectin drugs, every 5 weeks for ivermectin. For example, ivermectin can be given at 10, 15, and 20 weeks after turnout or weaning. This strategy prevents clinical disease in the majority of calves while allowing some level of infection, which stimulates an immune response in first-season calves. However, treatment at the start of the grazing season has been shown to result in better weight gains than tactical treatments given during the grazing season.167 “Dose and move” strategy. This strategy consists of treating calves with a single dose of anthelmintic, then moving them to a clean pasture just before the anticipated peak in pasture infectivity (e.g., early to mid summer in temperate climates). This strategy minimizes the number of anthelmintic treatments during the grazing season. However, it is effective only on farms where a clean pasture is available. Furthermore, any residual nematodes left behind will likely possess resistant genes; therefore contamination of the clean pasture will be with resistant nematodes. This must be taken into account when planning for the future use of the pasture (see later).
“Dose and move” strategy. This strategy consists of treating calves with a single dose of anthelmintic, then moving them to a clean pasture just before the anticipated peak in pasture infectivity (e.g., early to mid summer in temperate climates). This strategy minimizes the number of anthelmintic treatments during the grazing season. However, it is effective only on farms where a clean pasture is available. Furthermore, any residual nematodes left behind will likely possess resistant genes; therefore contamination of the clean pasture will be with resistant nematodes. This must be taken into account when planning for the future use of the pasture (see later).Integrating effective pasture management can reduce the number of anthelmintic treatments necessary192; however, there currently is no realistic alternative to the continued use of available compounds. Therefore it is imperative that the efficacy of these compounds be maintained for as long as possible. Recommendations designed to promote this in cattle include the following: (1) do not treat second-year and adult cattle to maintain a population of unexposed nematodes (refugia) on the farm; (2) do not graze first-year calves on the same pasture each year (avoids exposure to larvae produced from resistant nematodes); and (3) do not use the same family of anthelmintic year after year in calves.184
The use of complimentary classes of drugs may be necessary in the situation where a producer is not satisfied with the response to treatment. The combination of a macrocyclic lactone with a BZD or levamisole has been shown to be effective against all important nematodes, including anthelmintic-resistant forms. Similar combinations may also be used in feedlots to help provide maximal productivity and enhance response to vaccination programs. However, care must be exercised when using this recommendation because heavy, indiscriminate use of anthelmintics is a strong selector for resistant genotypes and will hasten the development and spread of drug-resistant parasite populations.149
Adult Cattle
The cattle most at risk for clinical disease and production losses are beef calves and dairy replacement heifers in their first season at pasture. Strategic treatment early in the grazing season is effective in most management situations. Development of immunity should protect the animals during their second and subsequent grazing seasons. Treatment of adult cattle generally is unnecessary, unless immunity is inadequate or pasture infectivity is high. Anthelmintic treatment is most likely to be warranted in first-calf heifers and newly acquired cows that may not have been pastured as heifers. In some situations it may be beneficial to treat beef cows after spring calving.
EVALUATION OF ANTHELMINTIC PROGRAMS
Because of the importance of arrested larvae in the pathophysiology of ostertagiasis, FECs can be misleading. The parasite does the most damage to the host as it enters the gastric gland and as it leaves; however, the infection is unlikely to be patent during these periods. Consequently, animals with from type I or type II ostertagiasis may have low FECs. Therefore FECs are most useful as a tool to evaluate pasture contamination and the success of control programs. Detection of anthelmintic resistance currently depends on the FECRT. Unfortunately, this will only detect clinical resistance, which usually occurs only when the frequency of resistant alleles in the population reaches 25%.196 Consequently, this test generally does not detect the presence of resistant nematodes within a population until clinical resistance occurs—that is, a less than expected response to the treatment is noticed. The FECRT estimates anthelmintic efficacy by comparing pretreatment and posttreatment FECs. Fecal samples are collected immediately before treatment and then 10 to 14 days later; for cattle, waiting the full 14 days before collection of the second sample is currently recommended.149
Clinical Management
DIAGNOSIS
The most reliable method of diagnosis is necropsy. The “Moroccan leather” abomasal lesion is pathognomonic for Ostertagia. It may be difficult to identify parasites with a casual macroscopic examination because both larvae in gastric glands and adult worms are small and easily overlooked. Histologic section and abomasal wall digestion techniques can be used to identify the presence of larvae. Antemortem, the history and clinical signs are most useful in suggesting a diagnosis of ostertagiasis. FECs are not specific for the disease.
TREATMENT
Animals should be treated with an avermectin or moxidectin and moved to a less contaminated environment at the first indication of clinical signs. History of the animals, time of year, and clinical judgment will determine whether all animals in the group should be treated. Animals with type I ostertagiasis can be expected to respond well; however, the prognosis for animals with type II ostertagiasis is less encouraging. Even though the larvae may be killed, the damage to the abomasal mucosa may limit complete recovery. Animals with profound hypoproteinemia and dehydration respond poorly compared with those showing only mild diarrhea and slight hypoalbuminemia. Severely affected animals may need treatment with fluids, plasma transfusions, and supportive therapy to survive. Recovered animals often fail to thrive.
GASTROINTESTINAL NEMATODE INFECTIONS IN SHEEP AND GOATS
GIN infections in sheep and goats are responsible for severe clinical syndromes and profound production losses. Young animals, periparturient ewes and does, and animals on substandard planes of nutrition are most susceptible to outbreaks of parasitic disease. The GIN of small ruminants includes Haemonchus contortus, Teladorsagia circumcincta, Trichostrongylus axei, Nematodirus species, and Cooperia species.197 The proportions of each of these nematodes in small ruminant populations vary according to geographic location. H. contortus usually is the most significant pathogen in wet, temperate climates. T. circumcincta may be the predominant infection in northern or arid climates. H. contortus and T. circumcincta represent the majority of parasite burdens seen in small ruminants, with H. contortus being present in highest numbers. Anthelmintic resistance is present in all these parasites, but the prevalence is highest for H. contortus, making it the most economically important GIN of sheep and goats.195
The problem of anthelmintic resistance in GIN of small ruminants has been reported in South Africa, Australia, New Zealand, Malaysia, Spain, France, Denmark, the United Kingdom, Brazil, and the United States.196,197 In the United States, resistance to all classes of anthelmintics has been documented.197,198 Recently resistance to two and three classes of anthelmintics was found on 14 of 15 farms and 6 of 18 farms, respectively, in a survey of 18 goat flocks in Georgia and South Carolina.199 In 2005, the first report of total anthelmintic failure was made on a meat goat farm in Arkansas.200 It is no longer acceptable to plan parasite control programs based solely on the use of anthelmintics. Veterinarians and producers must customize programs to control exposure to infection and reduce the use of anthelmintics. A thorough knowledge of the biology of GINs is necessary to plan effective control programs.
Life-Cycle201,202
The life-cycle of GINs is direct and consists of a host phase and a free-living phase. Worms mate in the host, and females lay eggs that pass in the feces. Eggs hatch and develop to infective larvae while remaining in the fecal mass. Infective larvae then move from the fecal mass onto the surrounding forage, where they can be consumed during grazing, thus completing the cycle. The time from ingestion of infective larvae to egg-laying adults, the prepatent period, is approximately 3 weeks. The time for development from egg to infective larvae can be as short as 4 to 10 days (especially during the summer months); therefore transmission (reinfection) and continual pasture contamination can be rapid. During the colder months, however, development is delayed, and organisms may take up to a month or two to reach the infective larvae stage; therefore pasture contamination and reinfection are minimized. The infective larvae have a protective sheath that makes them relatively resistant to adverse environmental conditions; they can therefore survive for months, thus extending transmission potential. As long as the temperature and moisture conditions remain favorable, development and survival continue; conditions that are too hot, too cold, and/or too dry threaten parasite survival.
Epizootiology201,202
The life-cycle of GINs has four phases.
PHASE 1—SYMBIOTIC OR PARASITIC PHASE
Phase 1 is the interaction between host and parasite. During this phase the parasite has to develop and survive in the host. After ingestion, infective larvae lose their protective sheath and invade the mucosa of the abomasum, small intestine, or large intestine depending on the GIN species involved. While in the mucosa larvae develop to the next larval stage and then return to the surface of the gut mucosa, where they become adult worms. The major host defense mechanism is immunity. When an infectious agent enters the body, the immune system reacts to mobilize various components (antibodies, killer cells, and so on) to inhibit or kill the invaders. These components act on larval stages in the mucosa and the adults in the lumen. How strong the immune response is depends on the age of the host, nutritional status, and concurrent stressors. The immune system matures with age; therefore young animals are relatively more susceptible to infection and become more resistant with age. As a result, young animals usually harbor the highest GIN populations and experience the most severe consequences. Adult animals have developed stronger immunity and harbor lower infection levels. Under poor nutrition and/or stressful conditions, the immune system is compromised and cannot respond adequately. Therefore regardless of the age of the animal, the effects of infection will be worse. The prepatent period of most worms is approximately 3 weeks, but this period can be extended for worms that have the capability to enter a period of delayed development called hypobiosis. This occurs during the season of the year when the environmental conditions are unfavorable for development and survival of the free-living larval stages. Depending on the worm, this happens during either summer or winter.
PHASE 2—CONTAMINATION PHASE
Phase 2 results from eggs passed in the feces. The magnitude of this phase is affected by stocking rate, age of the animals, season of the year, and hypobiosis. The higher the stocking rate, the more feces are deposited on the grazing area, and therefore more eggs, and the lower the stocking rate, the lower the number of eggs deposited. More eggs are also passed from young versus older animals. Most worms have a definite seasonality, so during their “season,” more eggs are produced and passed. Of particular note is a phenomenon called the periparturient rise (PPR) in FEC. This occurs at or around parturition and extends through most of the lactation period. Because parturition and lactation are stressful conditions, the dam’s immune system is compromised. This allows the existing female worms to increase the number of eggs deposited in the feces. If a worm species undergoes hypobiosis, the development time to the adult stage is extended. This will result in fewer adult worms over time and fewer eggs deposited in feces. However, when these hypobiotic larvae resume development, massive numbers become mature adults over a short period of time, and resultant egg production and deposition in the feces can be very high.
PHASE 3—FREE-LIVING PHASE
Phase 3 involves larval development and survival, which depend on prevailing environmental and nutritional conditions. Initially, development and survival from egg to first-stage, then to second-stage, and finally to third-stage (infective) larvae occur within the fecal mass. The first- and second-stage larvae are unprotected and require oxygen and energy (feeding on nutrients and microorganisms) to grow. The infective larvae are enclosed in a protective sheath and do not feed. Temperatures conducive to normal development and survival are 65° F to 85° F. With lower or higher temperatures, development and survival are reduced. Moisture is also crucial for development and survival. Because the initial development and survival occur within the fecal mass, moisture is usually adequate to allow development to the infective stage; however, if the fecal mass dries out quickly owing to high temperatures and/or physical disruption of the fecal mass, the first- and second-stage larvae are susceptible to desiccation and will die. Generally, infective larvae can survive very low temperatures, but sustained temperatures above 95° F are usually lethal. When infective larvae migrate out of the fecal mass, they are relatively resistant to environmental conditions encountered, because of their protective sheath. Temperature is usually the only factor that may adversely affect infective larvae. Again, sustained temperatures above 95° F can be lethal. The moisture conditions at ground level under forage cover usually are adequate for infective larvae to move around and survive. Because they do not feed, length of survival depends on how fast they use energy reserves. The hotter it is, the faster they move and use energy stores, and the shorter their survival time. Eventually, infective larvae move up and down the forage when there is a moisture medium (e.g., advancing and receding dew). Rain also provides a moisture medium for larval movement on forage. For the most part, infective larvae do not move much past 30 to 50 cm from the fecal mass or 5 to 6 cm up the forage. Therefore the lower the animals graze and the closer to the fecal mass, the greater the consumption of infective larvae, and the higher they graze, the lower their exposure to larvae.
PHASE 4—INFECTION PHASE
Phase 4 occurs when available infective larvae are consumed during grazing. This phase is affected again by stocking rate in two ways. If the same (phase 2) animals remain on the same pasture, the stocking rate determines how many eggs initially contaminated the pasture and consequently how many infective larvae will be available to consume. If the initial contaminating animals are removed and replaced by new animals, the new stocking rate will determine the level of exposure of each animal to infective larvae during grazing—that is, the higher the stocking rate, the more chance of exposure, and the lower the stocking rate, the less chance of exposure. It is well known that grazing animals usually do not graze close to fecal masses, so the greater the distance between masses, the less the exposure. Eventually fecal masses disintegrate, forage grows well with the fertilization, and animals will graze over areas where exposure can be high. Natural sources of water, such as streams, ponds, or lakes, provide moisture along the banks where forage can grow readily. When animals congregate to drink and consume the attractive forage, defecation in these areas usually leads to increased contamination and eventually more infective larvae. Essentially a high stocking rate has been artificially created in a relatively small area. The same can be said for areas where supplements, especially hay, are fed if conditions are right for development and survival of the free-living stages. Once infective larvae are consumed, phase 1 is repeated.
Pathophysiology of Gastrointestinal Nematode Parasites
The damage done by H. contortus is the result of the blood sucking by L4 and adult parasites. The pathogenesis of ostertagiasis in sheep is similar to that described in cattle and is a result of destruction of abomasal mucosa. T. axei causes abomasitis, whereas Trichostrongylus colubriformis penetrates beneath the intestinal mucosa and causes blood loss and enteritis.
Clinical Signs of Gastrointestinal Nematode Parasites
Most animals acquire mixed infections of nematodes. Clinical signs may therefore reflect the effects of more than one species of parasite. There appears to be some synergism between O. circumcincta and T. colubriformis, which makes the effect of the combined infection more severe than that of either alone. Although mixed infections with GINs are assumed, the actual population of parasites in small ruminants will vary depending on the geographic location. In general, the majority of clinical illnesses result from H. contortus infections. Most parasitic infections in small ruminants are associated with altered gut function, anorexia, ill thrift, weight loss, and hypoproteinemia. Diarrhea is a variable sign. Animals may die suddenly without overt clinical signs or may exhibit chronic wasting. Clinical signs of H. contortus infection can vary from peracute to chronic and result from decreased nutrient use and anemia or hypoproteinemia. The most common signs are failure to thrive, weight loss, and decreased appetite. Weakness, bottle jaw, pale mucous membranes, poor capillary refill time, and possibly diarrhea develop with more severe or long-term infections. After sudden exposure to large numbers of infective larvae, animals can die acutely, even before the infection is patent. Other diseases such as pneumonia and heat stress may result secondarily.
Anemic Crisis and Patient Management
The anemia created by H. contortus is due to blood consumed by the parasite and usually is chronic. Parasitized patients presented with lethargy and weight loss often have hematocrits <10%, yet many are still capable of rising and walking. Addressing the anemia by blood transfusion must be weighed carefully against stress to the patient caused by restraint, jugular catheterization, and blood administration as well as by the hemolysis of these same cells several days later. Obviously, in life-threatening circumstances (the patient that is unable to stand), administration of whole blood can be critical. In the noncrisis situation (patient rising and/or eating) treatment with an effective anthelmintic should eliminate the parasite and halt blood loss. Because H. contortus consumes blood, the affected animal actually loses substrates essential to erythrocyte production (iron, cobalt, copper). Providing supplemental iron and B-complex vitamins should speed reversal of the anemia. Assuming blood loss has been halted, a 1- to 1.5-point rise in the hematocrit per day is expected when the bone marrow is maximally stimulated. In most parasitized patients a rise of one half this value is a more realistic expectation.203
If blood is to be given, it is essential to be certain that the donor has a normal packed cell volume. An animal in the same environment may be as anemic as the patient, but the individual may be more capable of dealing with the anemia. In lambs weighing less than 45 kg, 1 unit of whole blood (approximately 450 mL) is often sufficient to survive a crisis, provided H. contortus has been controlled.
Populations at Risk
Young animals are most susceptible to infection and clinical manifestations of disease. Lambs and kids may become heavily infected with parasites and shed large numbers of nematode eggs. In sheep, some degree of immunity develops as the animal approaches 1 year of age. Adult animals typically have complete immunity against Nematodirus and variable resistance to Trichostrongylus species. Some immunity to Haemonchus and Ostertagia species develops with age; compared with lambs or kids, adults are more resistant to infection with these species. However, even mature animals may succumb to parasitic infection when malnourished or challenged with heavily contaminated pasture. A PPR in fecal egg production is seen in ewes and does.
Goats are more susceptible to GIN infections than are sheep. The difference lies in part in the host’s immunologic responses to nematode antigens. Goats prefer to browse on brush and trees, and they graze grass only when forced to by management. Under natural conditions, goats would have a low level of exposure to infective larvae, and this might explain their lower level of natural immunity compared with sheep.204,205
Refugia
Biologically, refugia refers to isolated populations of once widespread animal or plant, or in this case parasite, species. Most parasitologists now consider levels of refugia as the single most important factor contributing to selection for anthelmintic-resistant parasites.206,207 Worms in refugia provide a pool of genes susceptible to anthelmintics, thus diluting the frequency of resistant genes. For many years parasitologists and veterinarians have recommended that all animals should be treated with an anthelmintic at the same time. However, this strategy has proven problematic, and a selective approach is now recommended. Only those animals in danger of severe parasitism should actually receive medication. This selective approach is highly compatible with host-parasite dynamics, as 20% to 30% of animals harbor approximately 80% of the parasites.208 Animals with low worm burdens are an important source of refugia, are not in danger of the negative effects of parasitism, and should not be treated.
Management of Gastrointestinal Nematodes in Small Ruminants
The traditional approaches of deworming often, rotating classes of anthelmintics, and moving to clean pastures have allowed anthelmintic resistance to all classes of drugs to develop and the resistant parasites to disseminate over a wide area. It is obvious that drastic changes in the management of GINs in sheep and goats are long overdue.
FAMACHA OR SMART DRENCHING METHOD.209
In order to maintain adequate levels of refugia, it is necessary to leave a portion of the herd or flock untreated. FAMACHA is a selective approach that targets the portion of the herd or flock with high worm burdens, including those animals that are poorly resilient to worm infections.210 This approach will successfully control parasites in the entire group while significantly reducing drug costs and delaying the development of anthelmintic resistance. The use of this system is not without risks, as individuals are not routinely treated with anthelmintics. Failure to observe animals closely for clinical signs may result in the death of some individuals. It is necessary to know which anthelmintics are effective before beginning this system. The differences among farms in overall quality of management, stocking rates, breeds of animals, preexisting levels and spectrum of anthelmintic resistance, presence of nematode species other than H. contortus, and production targets need to be considered while FAMACHA is implemented on individual farms.
FAMACHA is a novel system developed in South Africa for identifying sheep that are anemic; the system has been extended to use in goats.211 In this method the ocular mucous membranes of sheep and goats are categorized by comparison with a laminated color chart bearing pictures of sheep conjunctivae classified into five categories ranging from red (A/1; normal) to practically white (E/5; severe anemia). Because anemia is the primary pathologic effect of infection with H. contortus, this system can be an effective tool for identifying animals that require treatment (but only for H. contortus). FAMACHA has been tested extensively and validated in both sheep and goats in South Africa and in the southern United States.212,213 In all studies the number of false-negative results was very low, suggesting that when FAMACHA is used according to recommended guidelines, death from anemia would be rare.
Based on the results of recent studies in the United States and numerous studies performed in South Africa, guidelines for using FAMACHA have been developed, and it is suggested that these guidelines be read in their entirety before FAMACHA is practiced.209 It is recommended that treatment be withheld until animals score 4 or 5 as long as animals are in good body condition and good overall general health, animals are examined frequently (e.g., every 2 weeks), and good husbandry is used to identify animals in need of treatment (e.g., unthrifty, anorexic, lagging behind, bottle jaw) between FAMACHA examinations. When this approach is used, the number of anthelmintic treatments administered will be reduced greatly, resulting in significantly diminished selection pressure for resistance and therefore a reduction in drug costs. Owing to the increased handling of animals, labor costs will be increased. Only adult animals should be managed with this system. Lambs and kids have comparatively small blood volumes, poor immunity, and poor resilience and can progress rapidly from moderate to severe anemia. This precaution should be extended also to ewes and does during the periparturient and early lactation period, because these animals have decreased immunity to GINs.209,214-216 These and other animals that may be stressed by disease or in poor body condition should always be treated if scored as 3. Alternatively, in the northern parts of the country where H. contortus is an important problem but resistance prevalence is much lower, it may be reasonable to be more liberal when making treatment decisions (e.g., treat all animals with scores of 3, 4, and 5). Many more treatments are given when all animals with scores of 3 and higher are dewormed, but a significant number of animals will remain untreated to supply refugia. These refugia combined with a relatively short transmission and treatment period are likely to produce a very slow evolution of resistance, but the more intensive treatment protocols will improve animal productivity.
On farms where low to moderate levels of resistance to one or more drugs have been diagnosed (60% to 95% reduction in FEC), a useful strategy to help gain the full benefits of both treatment and resistance prevention could be to use these “less-effective” drugs either singly or in combination on all animals scored as 3. Using drugs that are less effective in this group is unlikely to lead to clinical problems because the few 3 scores that are moderately anemic and in need of treatment should receive a sufficient reprieve from infection until the next FAMACHA examination, and the majority of the animals with scores of 3 that are not anemic do not need to be treated. This strategy will help preserve the efficacy of the drugs that are still fully effective by saving them for only the animals in the 4 and 5 categories and also will help to decrease egg contamination of pastures.
Training of producers is critical in the use of this method. It is the responsibility of veterinarians and other animal health professionals to ensure that standards of training are maintained. When FAMACHA is used, it is extremely important that efficacy of anthelmintics is known because animals are not treated until they become anemic. Treating anemic animals with a drug that has moderate to poor efficacy owing to worm resistance may result in animal deaths. Other important precautions for using FAMACHA include but are not limited to the following: (1) the card is an aid in the control of Haemonchus species only; (2) the system should be used by producers only where technical assistance is available from a veterinarian or other animal health professional; (3) other management-based worm control practices must be maintained; (4) smart drenching principles should be used; (5) paleness or reddening of the conjunctivae may have other causes; (6) animals should always be scored with the help of the chart, not from memory; (7) animals are examined at least every 2 to 3 weeks at the beginning of the expected period of Haemonchus species challenge in climates where a seasonal incidence of infection occurs, and during critical periods weekly examinations may be needed; (8) the card is protected from light when not in use and replaced after 1 year of use.197,217
Maintaining treatment records that are included with the FAMACHA kits gives the owner the ability to rate the genetic merits of individuals on the premises. Host resistance to infection with H. contortus measured on the basis of FEC and packed cell volume (PCV) is a moderately heritable trait, and it has been demonstrated that the same animals tend to exhibit the highest FEC and lowest PCV on each occasion they are measured.218-220 Of importance, data from recent investigations examining the heritability of resistance and resilience of merino sheep to infection with H. contortus indicate a high heritability for the clinical estimates of FAMACHA scores.212 Removing the most susceptible animals from the breeding pool each year will have the long-term effect of improving the overall innate genetic resistance and/or resilience of the herd or flock to H. contortus.
GRAZING STRATEGIES
The goal of pasture management is to provide safe pastures for grazing by reducing the exposure of susceptible hosts to infective larvae.224,225 A safe pasture is one that has had no sheep or goats grazed on it for 6 months during cool and cold weather or 3 months during hot, dry weather. Weaning sheep and goats at 2 months of age and rotating them through pastures ahead of the adults while forage is longer will minimize the exposure of susceptible animals to infective larvae. Pastures should be subdivided into smaller lots to allow longer rest periods between grazings. Pastures that are overgrown provide a good environment for larval survival, as ultraviolet light and dry conditions are effective in killing larvae. Keeping pastures clipped will assist in weed and parasite control. Short-duration grazing carries pasture rotation to a level that maximizes forage production and harvesting by controlled animal grazing. It is management intensive but can be effective in controlling parasite burdens. Tilling and reseeding a heavily contaminated pasture will convert it to a safe pasture and gives an opportunity to improve the forage quality. Taking a cutting of hay from a pasture assists in reduction of infective larvae, but one report indicates GINs and tapeworms developing in “worm-free” lambs after they were fed hay from heavily infected pastures. During the most dangerous part of the grazing season it may be necessary to drylot the flock and feed hay and grain from elevated feeders.
Stocking rate is an important consideration in parasite control, as it affects the exposure to infective larvae and the contamination of the pasture. It is impossible to make a general recommendation on stocking rate, as this will vary according to the type of pasture, the time of year, the current weather conditions, and the type of animal being grazed. Rules of thumb are that five to seven goats or five sheep are the equivalent of one cow and suggest five to seven goats per acre. Goats prefer to browse brush and trees, whereas sheep prefer to graze near the ground. Pasture management must include monitoring the condition of herbage to ensure that overgrazing does not occur and to maintain a productive pasture.
In the early spring or at the onset of the rainy season, reduced pasture contamination is the most important aspect of control. The ewe or doe in the periparturient relaxation of resistance, even if she has the genetic capacity for resistance, will be a source of eggs for the environment. Strategic deworming to remove arrested or recently emerged larvae before they contaminate the pasture will have a great impact on pasture contamination. Treatment 2 weeks after a rain that removes recently acquired worms before they can begin passing eggs will also decrease pasture contamination. Providing sufficient dietary protein is vital during the periparturient period and during rapid growth so that animals will tolerate the worm burden better as their immunity is strengthened.223-225 A strong link between nutrition and parasitism has been illustrated between protein intake and resistance to GIN infection. The most dramatic has been the abolishment of the periparturient egg rise in lambing ewes by providing protein at 130% of requirements. Immunity is closely related to protein repletion. GINs increase the demand for amino acids by the sheep. Lambs will voluntarily select a higher protein diet when infected with GINs compared with uninfected lambs. There is conflicting documentation that sheep will decrease feed intake when initially infected with GINs. Some authors hypothesize that the decrease in intake may be caused by stimulation of the immune system or that the host is becoming selective in its diet.
Pastures may be used for hay cropping and grazed during the last half of the grazing season to effectively reduce GIN challenge. When plants high in condensed tannins are grazed, there is evidence that the incoming larvae are adversely affected by condensed tannins, which also provide bypass protein for the host.226-228 There is growing evidence in work from New Zealand and Europe that grazing or feeding of plants containing condensed tannins can reduce FEC, larval development in feces, and adult worm numbers in the abomasum and small intestine. Preliminary tests with sericea lespedeza (Lespedeza cuneata), a perennial warm-season legume, have shown positive effects of reduced FECs in grazing goats, and in sheep and goats in confinement when the forage was fed as hay. In addition, an effect on reducing worm burden has also been reported. Similar results have been observed using quebracho extract for small intestinal worms but not abomasal worms. In addition to its potential use in controlling worms, sericea lespedeza is a useful crop for limited resource producers in the southern United States. It is adapted to hot climatic conditions and drought and acidic, infertile soils unsuitable for crop production or growth of high-input forages, such as alfalfa. It can be overseeded on existing pasture or grown in pure stands for grazing or hay. In addition to hay, sericea lespedeza is being evaluated in the form of meal, pellets, and cubes to be fed as a supplement to grazing animals or as a deworming method under temporary short-term confinement. The physical structure of some plants may challenge larvae to ascend vegetation or may provide protection from adverse pasture conditions. If animals are allowed to browse, their chances of acquiring larvae diminish as the distance from the ground increases. Most infective larvae are found within 2 inches (50 mm) of the soil surface.
Alternate grazing or co-grazing with other species of livestock may harvest Haemonchus larvae from the pasture. Small ruminants can graze after cattle and this is considered to be a safe pasture, assuming adequate parasite control in the cattle. For the most part, each livestock species harbors its own parasite fauna, with the exception of overlap between sheep and goats. Only T. axei, a minor abomasal worm, is found in all livestock species. In general, the Haemonchus organisms in sheep and goats do not do well in cattle, and vice versa. However, some populations of H. contortus may thrive in calves. If practical, cattle and small ruminants can be grazed together where each consumes the parasites of the other, which reduces available infective larvae for the preferred host species.
Anthelmintic administration should be coordinated with the weather. During hot, dry weather, there will be little or no exposure to infective larvae. As soon as there is significant rainfall (½ to 1 inch) larvae exposure goes up exponentially, as previously inactive larvae become active and new larvae are hatched. The producers should be trained to plan deworming within 3 weeks of significant rain after a dry spell. Similar strategies can be used during cool weather. Once ambient temperatures drop below 50° F, the flock can be dewormed and no further treatments are necessary until temperatures become favorable to larval development and activity.
ALTERNATIVE THERAPIES
ANTHELMINTIC USE
Discussion of anthelmintics has deliberately been left until the end of this section because drugs cannot be considered the most important aspect of a parasite control program. Guidelines for drug doses are contained in Table 49-3. At this time, only oral administration of anthelmintics is recommended in small ruminants. In general, goats require 1.5 to 2 times the doses of sheep, with the exception of levamisole, owing to more rapid GI transit times. It is critical that producers accurately assess weights of individual animals and determine the dose appropriately. If an approximate dose is going to be used in all animals, that dose should be for the lightest animal in the group.
The challenge of anthelmintic resistance is life-threatening in small ruminants. There is no point in repeatedly deworming with expensive drugs if the drugs are no longer effective. See the following section on monitoring parasite control programs for information on detecting anthelmintic resistance. On farms where low to moderate levels of resistance to one or more drugs have been diagnosed (60% to 95% reduction in FEC), a useful strategy to help gain the full benefits of both treatment and resistance prevention could be to use these “less-effective” drugs either singly or in combination. Drugs in the BZD family may be effective if multiple-day administration regimens are used.
LUNGWORM INFECTION IN LARGE ANIMALS
Lungworms traditionally are those nematode parasites that reside in the lung as adults. In ruminant and equine species, most clinical cases of lungworm involves the genus Dictyocaulus. Species of Dictyocaulus tend to be host specific with Dictyocaulus viviparus, Dictyocaulus arnfieldi, and Dictyocaulus filaria found in the bronchi of cattle, equids (horses, ponies, donkeys), and small ruminants, respectively. Sheep and goats may also be infected with Protostrongylus rufescens and Muellerius capillaris.239 This section focuses on Dictyocaulus infection, with a brief discussion of other lungworm parasites found in small ruminants.
Because verminous pneumonia requires specific treatment, it is important to distinguish it from diseases caused by other infectious agents. Often the role of lungworm in the cause of respiratory disease may be obscured by a parasite-induced hypersensitivity response or by superimposed secondary bacterial infections.
Life-Cycle
The life-cycle of D. viviparus is direct, with no intermediate host involved. Adults produce embryonated eggs that hatch shortly after oviposition. L1 are passed in the feces, where they develop to L2 and L3 within a week under optimal environmental conditions. After the infective L3 are ingested, they migrate through the wall of the intestine to the mesenteric lymph nodes and through lymphatics into the bloodstream and finally arrive, as L4, in the lung as early as 7 days after ingestion. Larvae may then develop to sexually mature adult worms or they may arrest development as late L4 or immature adults. Arrested development can prolong the normal prepatent period from 3 to 4 weeks to approximately 5 months.239
Most problems with cattle lungworms in North America occur in areas of moist climates where larvae on pasture are protected from desiccation and larval migration away from the fecal pat onto herbage is facilitated. Under favorable conditions, infective L3 can survive on pasture for approximately 11 months.240,241 These larvae plus the larvae produced from carrier animals harboring small numbers of adults or adults developing from arrested larvae can result in high levels of contamination on spring pastures. In subtropical climates, larvae are virtually absent during the hot summers; larval contamination of the pasture peaks in the autumn as a result of carrier animals.242
The life-cycle of D. filaria is similar to that of D. viviparus including the ability of the larvae to arrest development. The prepatent period is approximately 26 days, with peak larval output occurring 39 to 57 days postinfection. As with D. viviparus, D. filaria is more prevalent in moist climates and larvae can survive on pasture throughout the winter. Spring pastures contaminated with overwintering larvae and larvae produced from adults developing from arrested larvae in carrier animals are sources of infection for susceptible lambs or kids.239,243-245
The life-cycle of D. arnfieldi is similar to that of other species of Dictyocaulus except that, in addition to L1, larvated eggs may be passed with the feces. The prepatent period is 2 months.246
Unlike species of Dictyocaulus, M. capillaris and P. rufescens have indirect life-cycles that involve intermediate molluscan hosts. Adults, occurring in the parenchyma (M. capillaris) or small bronchioles (P. rufescens), produce eggs that develop and hatch. L1 are passed with the feces; once on pasture, larvae invade the foot of a susceptible species of snail or slug and develop to infective L3. Sheep and goats become infected when inadvertently ingesting the mollusk while grazing. The larvae penetrate the intestine and migrate to the lung in much the same manner as Dictyocaulus. Early L4, developing L4, and adult M. capillaris become embedded within a fibrous nodule within the parenchyma. Protostrongylus larvae mature in the alveoli and enter the bronchioles as adults. The prepatent period is 5 to 6 weeks, although arrested development may prolong the prepatent period of M. capillaris.239,243-247
Pathophysiology
Although infections with few worms may be inapparent, D. viviparus often results in parasitic pneumonia and bronchitis. The disease process is divided into four phases: penetration, prepatent, patent, and postpatent. In the penetration phase (days 1 to 7), no clinical sign or significant respiratory pathology occurs as the larvae migrate to the lungs. During the prepatent phase (days 7 to 25), clinical signs and lesions become evident. An eosinophilic infiltrate blocks bronchioles; excess mucous production and alveolar collapse occurs. The patent phase (days 25 to 55) is associated with egg-laying adults in the bronchi and trachea. Bronchial and tracheal epithelial damage occurs, air passages are blocked by exudate, and consolidation of lobules results from aspiration of eggs and L1 into bronchioles and alveoli. Bronchitis, tracheitis and pneumonia follow. During the postpatent phase (after day approximately day 50), surviving animals recover and adult worms die off. Peribronchial fibrosis and epithelialization of alveoli may remain. Secondary bacterial infections resulting from weakened pulmonary defenses may also occur.239
Goats tend to be more susceptible to infection with D. filaria and more severely affected than sheep, although individual susceptibility to infections does occur. Heavy infections in both species produce bronchitis, pulmonary edema, atelectasis, and emphysema, although death is uncommon.239,243,244 As with D. viviparus, secondary bacterial infections can occur.
In donkeys, whether D. arnfieldi causes pathologic lesions on its own is controversial because of the presence of other pathologic agents; however, in healthy infected individuals, pathologic changes include discrete circular areas of overinflation surrounding affected bronchi. Histologically these areas contain a small bronchus packed with nematodes. The small airways are often occluded with exudate. Small amounts of mucus surround adult worms in the main bronchi with little cellular reaction. L1 are surrounded by an intense mucopurulent reaction. In addition to these types of lesions in horses, many immature nematodes are present in smaller bronchi, accompanied by bronchial epithelial hyperplasia with large amounts of mucus and pus. Many areas of alveolar hemorrhage and edema are also present.246
Although morbidity with M. capillaris may be high in most flocks, massive infections with M. capillaris or P. rufescens are not common. Pathologic changes caused by P. rufescens are similar to chronic, low-grade Dictyocaulus infection. Heavy infections with M. capillaris infection in goats can result in interstitial pneumonia, bronchopneumonia, or fibrinous pleuritis. In sheep and goats, distinct subpleural nodules containing adults and L1 are common. Secondary bacterial infections can also occur.239,248-251
Populations at Risk
Clinical disease occurs most frequently in pastured calves, lambs, and kids in the first year of life. The disease is also reported in housed cattle, related either to heavily contaminated bedding material or to synchronous maturation of large numbers of hypobiotic larvae. Clinical lungworm infestation is more of a problem in cattle than in sheep, possibly because lambs are treated for GINs more frequently than are calves under most management systems.
Immunity to lungworm infection occurs after first exposure and can develop before the end of the first grazing season. However, it is variable in both degree and duration. In cattle, exposed animals may be immune for 7 to 12 months after infection. In subsequent infections most larvae are either killed before reaching the lung or are inhibited from maturing into adults. Usually the immunity developed during the first grazing season is boosted by exposure in subsequent years and prevents manifestations of disease. However, without reexposure to infective larvae, immunity wanes and disease can occur in adult cattle.239
Occasionally, outbreaks of acute lungworm disease are seen in adult cattle on pasture whose immunity is overwhelmed by exposure to very large numbers of larvae. This condition is called the reinfection syndrome. Although most ingested larvae are killed or fail to mature, some reach the lung and incite an acute, immune-mediated reaction.
Both sheep and goats develop a strong immunity to D. filaria; disease is more common in young animals. As with cattle, continued immunity in sheep depends on reexposure.239,252
Individual horse susceptibility to infection with D. arnfieldi is evident. Infections can occur during any time in the life of the horse. Donkeys apparently do not develop an immunity to infection, with larval excretion increasing with age.246,253
Although immunity to M. capillaris appears to be incomplete (adult nematodes can live for up to 4.5 years), responses to subsequent exposures do limit the numbers of adults that develop and the numbers of larvae shed in the feces. However, reexposure may be cumulative, resulting in older animals harboring larger numbers of worms.239
Clinical Manifestations
Clinical disease (“husk”) caused by lungworm infection in cattle can occur within 1 or 2 weeks of the introduction of susceptible animals to contaminated pasture. However, clinical disease most often occurs 2 to 4 months into the grazing season. Affected animals develop either an acute or a subacute form of the disease, depending on the number of larvae ingested and the animal’s level of immunity. With acute verminous pneumonia in calves, dyspnea and cough are prominent signs. Auscultation of the lungs initially reveals vesicular murmurs and bronchial tones that progress to moist rales as fluid accumulates. The animal may expectorate froth. Occasionally, emphysema occurs and crackling noises are heard on auscultation. Fever may be as high as 40.5° C (105° F). Mortality rates are high, and animals may die before a patent infection has been established.239 The similarity between this presentation and that of bacterial pneumonia or shipping fever should be noted.
The subacute or chronic manifestation of disease is more common. In all species the primary signs are coughing, dyspnea, and loss of condition. The animals show elevated respiratory rates but initially are afebrile. Bronchial irritation caused by adult worms and fluid accumulations produces paroxysmal coughing. Animals with the subacute form of lungworm disease are susceptible to bacterial pneumonia. In fact, the subacute form is easily confused with enzootic pneumonia in calves. Signs of reinfection syndrome in adult cattle include severe cough, tachypnea, harsh respiratory sounds, and, for dairy cattle, a sudden drop in milk production approximately 2 weeks after exposure to heavily contaminated pastures.239
In horses, lungworm infection typically causes a syndrome similar to chronic obstructive pulmonary disease, although asymptomatic infections may also occur. Clinical signs include persistent coughing, nasal discharge, and respiratory distress; death is uncommon. Donkeys, the source of infection in most cases of lungworm disease in horses, tend to remain asymptomatic, although harsh lung sounds may be auscultated.246,253
Infections with M. capillaris are generally asymptomatic; however, impaired pulmonary gas exchange, reduced weights and breeding performance, and increased mortality have been reported.248,249,254
Control of Lungworm Infection
Management strategies designed to decrease exposure to infective larvae are most effective in preventing dictyocaulosis. Young animals should not be overstocked or allowed to graze in moist, low-lying pastures. It is often helpful to rotate pastures so that successive calf crops do not graze the same area. Young stock should not be grazed with older, clinically immune animals that may serve as a source of infective larvae. Horses should not be grazed with donkeys. If they are, then donkeys should be monitored for L1 and treated to reduce larval output and pasture contamination. Limiting contact with intermediate hosts and provision of mollusk-free housing will decrease infections with parasites that require these intermediate hosts.
ANTHELMINTICS
Several anthelmintics are effective against lungworm. Drugs for the treatment and control of lungworm are presented in Table 49-4.239,246 The avermectins and milbemycins are particularly effective against both adult and larval stages and have prolonged residual activity that prevents appearance of larvae in the feces for at least 60 days.255,262 Strategic use of these products, such as treatment at turnout and again 8 weeks later, can substantially reduce pasture infectivity with lungworm larvae. Concerns that such highly efficacious anthelmintics would prevent induction of immunity in first-season grazing animals appear to be largely unfounded, with the possible exception of sustained-release intraruminal devices (see later). The immune response may be attenuated, but exposed animals still develop some degree of immunity to lungworm infection despite prophylactic treatment with these drugs.256,257,263-265
Table 49-4 Drugs Used to Control Lungworm
| Drug | Dose |
|---|---|
| AVERMECTINS AND MILBEMYCINS | |
| Ivermectin, abamectin, doramectin, eprinomectin, moxidectin | 0.2 mg/kg PO, SC, IM*; 0.5 mg/kg topically (“pour-on”); intraruminal sustained-release device (ivermectin) |
| OTHERS | |
| Albendazole | 5 mg/kg PO (O, C); 7.5–10 mg/kg PO (B) |
| Fenbendazole | 5 mg/kg PO (O, C); 5–10 mg/kg PO (B); 30 mg/kg PO (E); intraruminal sustained-release device (B) |
| Mebendazole | 15–20 mg/kg PO (O, C) |
| Netobimin | 7.5 mg/kg PO (B, O, C) |
| Oxfendazole | 4.5–7.5 mg/kg PO (B, O, C) |
| Levamisole | 7.5–10 mg/kg PO, SC; 10 mg/kg topically (“pour-on”) (B, O, C) |
| Diethylcarbamazine | 22 mg/kg IM daily for 3 days or 44 mg/kg IM once (B) |
B, Bovine; C, caprine; E, equine; IM, intramuscularly; O, ovine; PO, by mouth; SC, subcutaneously.
* Route depends on the product and species; follow manufacturers’ recommendations.
Young, susceptible stock may be given a prophylactic treatment in mid-spring or early summer to prevent or limit clinical disease. Housed animals, in particular 1- to 2-year-olds with subclinical but potentially patent infections, should be treated before being turned out to pasture in the spring. All early clinical cases should be treated in an effort to limit disease severity and decrease pasture contamination.
Intraruminal SRDs containing ivermectin, levamisole, or a BZD can prevent clinical disease for several weeks or months during the grazing season.265-271 However, some studies have shown that these devices impaired the development of immunity in treated calves.265,269 SRDs may interfere with development of natural immunity by killing ingested larvae before they can penetrate the intestine and thus stimulate an immune response. However, other studies using these devices have shown that treated calves can develop immunity during the grazing season.272
VACCINES
An effective vaccine has been developed against D. viviparus in cattle and is available in the United Kingdom and parts of western Europe. Two doses of irradiated larvae are administered by mouth 4 weeks apart. The larvae migrate to mesenteric lymph nodes and provoke an immune response but die before they reach the lung. Management considerations associated with the use of the vaccine include the following: (1) calves must be at least 2 months old; (2) vaccinated calves should not be placed on pasture for at least 2 weeks after the second dose; (3) vaccinated calves must not be exposed to heavily infected pastures, nor should they be mixed with animals showing signs of lungworm disease or with unvaccinated calves; (4) immunity is not long-lasting, so animals must continue to be exposed to low levels of infective larvae to maintain immunity; and (5) the vaccine has a short shelf-life and it is relatively expensive. Farms with contaminated grazing areas and a history of lungworm disease benefit most from vaccination, and it is best used in young nursing calves before exposure to pasture.273
Evaluation of Preventive Programs
The efficacy of preventive programs is best assessed by evaluating the number of susceptible animals that demonstrate signs of infection. Because total eradication of the parasite is difficult and low numbers of parasites produce minimum problems, a useful goal is the control of clinical signs such as cough and loss of condition. Fecal larval counts are an unreliable means of evaluating the severity of lungworm infection in an individual animal.
Clinical Management
DIAGNOSIS
As with many parasitic diseases, lungworm is often diagnosed on the basis of farm history, seasonal prevalence, clinical presentation, and response to treatment.239,246,253 Verminous pneumonia may mimic respiratory diseases caused by other agents that require specific treatment. Although definitive diagnosis is difficult, it is important.
To document the presence of lungworm infection, it is necessary to demonstrate larvae, adult worms, or eggs. In cattle and small ruminants, the presence of L1 in fresh feces indicates lungworm infection.239 In horses, L1 may be present in feces; however, patent infections in horses do not always occur, and antemortem diagnosis may best be achieved by examining donkeys co-grazing with the horses.246,253 The Baermann technique is the technique of choice to detect L1 in the feces. However, in acute outbreaks animals may succumb before infections are patent. Transtracheal wash may demonstrate a large number of eosinophils, which is supportive of verminous pneumonia; eggs or larvae are occasionally seen with patent infections. This procedure can also help rule out bacterial pneumonia. Finding of nematodes at postmortem examination is definitive. At necropsy, adult Dictyocaulus organisms are usually apparent in the bronchi or bronchioles, whereas adult P. rufescens organisms tend to be in the bronchioles and adult M. capillaris organisms are found in subpleural nodules on the dorsal aspect of the diaphragmatic lobes. Nematodes and the eggs may also be found in histologic section of the lung.
TREATMENT
In subacute or chronic forms of the disease, removal of the adult parasites with anthelmintics may result in recovery. However, if the infection is heavy and lung damage is severe, anthelmintic treatment is unlikely to result in complete recovery. In some animals, anthelmintic treatment worsens the signs, and some heavily infected animals die. In severely affected individuals, antihistamines and antibiotics should be included in the therapeutic regimen.
EVALUATION OF PARASITE CONTROL PROGRAMS
The efficacy of a given parasite control program should not be assumed; it should be assessed on a regular basis. The routine performance of quantitative tests for parasite burdens allows veterinarians to identify the development of anthelmintic resistance. Although fecal floatation is useful in identifying the presence of parasites, the use of the McMaster technique allows an estimation of the numbers of parasites. Both procedures are relatively simple and can easily be performed in the practice setting. Other specialized laboratory procedures are also discussed. Once resistance has been documented it is important to identify the species involved through larval identification.
FEC and FEC Reduction Testss_body;274–276 (Box 49-1)
Determining numbers of nematode eggs in feces is the simplest and least invasive way to evaluate a parasite control program. Fecal examination may be of limited value in an individual animal because animals dying of parasitic infection may have no eggs in the feces, whereas animals with high FECs may be clinically normal. Nevertheless, FECs can provide information on the level of infection present in an individual animal, particularly when egg counts are repeated over the course of 2 to 3 weeks. Herd average FECs are more useful and provide an accurate reflection of the degree of environmental contamination and rate of infection. Pretreatment and posttreatment FECs can also be used to establish the efficacy of an anthelmintic in a particular group of animals. A quantitative McMaster or Stolley technique, rather than simple flotation, is crucial for accurate monitoring.
Box 49-1 Modified McMaster Technique (Fecal Egg Count)
This is one method for performing a McMaster fecal egg count. Similar protocols are used routinely in many laboratories, so you may see a slightly different procedure recommended elsewhere. The important point is to use the same procedure each time.
The first step is to collect freshly passed feces uncontaminated by soil or bedding. The best way is to wear a rubber glove and extract feces directly from the rectum. Alternatively, feces can be picked up off the ground if done soon after deposition. The collection container should be labeled with the identification of the animal and the date of collection. Fresh samples work best, but accurate results can be obtained if the sample is refrigerated during the interim. If samples are not refrigerated, the eggs will hatch within 12 to 24 hours. Once hatched, eggs cannot be counted.
PROCEDURE
NOTES
This is a dilution technique, and theoretically this ratio of feces to flotation solution will not detect infections with less than 50 eggs per gram of feces (one egg seen on slide), so it is not very accurate for samples with low numbers of eggs. On a practical level this is not important, because from a clinical standpoint slight differences in results when egg counts are low do not matter.
Fairly soon after counting is complete, thoroughly rinse out the McMaster chamber with warm running water. Doing so will keep the chamber clean and ready it to be used again. If fecal solution dries in the chamber do not soak in soapy water for long periods, as this will cause the chamber to become cloudy. If the chamber gets dirty, soak for only a few minutes in water containing dish soap and then rinse completely with tap water.
* Chalex Corporation, Issaquah, WA, chalexcorp@att.net; www.vetslides.com
It is suggested that guidelines published by the World Association for the Advancement of Veterinary Parasitology (WAAVP) be used to perform and evaluate data from an FECRT test, applying practical modifications to fit the situation on the farm.276 Briefly, groups of 15 animals that have not been treated within the past 8 weeks are randomly allocated to treatment groups, and FECs are performed using a modified McMaster technique 10 to 14 days after treatment. An untreated control group must be included. If fewer than 12 to 15 animals per group are available, then treatment groups should be balanced by performing a pretreatment FEC. Animals are then stratified by FEC from highest to lowest, blocked by number of treatment groups, and then within each block are randomly assigned to treatment. Alternatively, where Haemonchus is the primary parasite, animals may be scored using the FAMACHA method as they come through the chute, blocked by FAMACHA score, and then randomly assigned to treatment within blocks. This approach is a bit more complicated but will result in -groups that are balanced, which will result in a more accurate test. Calculations are performed using the following formula:
where Xt and Xc are the arithmetic mean number of eggs per gram (epg) in the treated (t) and untreated control (c) groups, respectively. Software is available for free that performs all calculations and gives data interpretation.277 If the RESO calculator is used, the assignment of resistance status is based both on percent reduction and the 95% confidence intervals. If the RESO calculator is not used, the following guidelines can be applied: reductions of greater than 95% indicate sensitivity, reductions of 90% to 95% indicate low or suspected resistance, and reductions of <90% indicates resistance. FECRT yields reliable data only if FECs are sufficiently high to properly measure a reduction from treatment. If the control group’s mean FECs are below 150 epg, then objective assessment of resistance will not be reliable. Group mean FECs of less than 150 epg can be common in adult sheep; therefore when FECRT is performed on a sheep farm it is preferable to use weaned lambs if available. With goats, low FECs are usually not a problem.
Larval Identification
Ideally, if there is <90% egg count reduction, the eggs should be hatched and the larvae species identified. In the majority of cases these will be H. contortus, but other species, especially Trichostrongylus species, readily develop resistance as well. One can sometimes be fooled into an improper interpretation of egg count reduction results if mixed species are present and only one is resistant.
Egg Hatch Assays and Larval Development Tests276
Eggs from feces are incubated with concentrations of the anthelmintic to be tested, and the eggs hatched. A dose-response curve is generated (DrenchRite test from Horizon Technology).275 The advantage of this test is that a single fecal sample can be tested for all available classes of anthelmintics simultaneously. The cost of this test has recently increased and may discourage owners from pursuing this diagnostic. However, when compared with the cost of using anthelmintics that are ineffective, it is easy to justify the use of this test. It is important to read instructions for submitting this test and scheduling a testing date before the samples are collected.
Larval Culture
Larval cultures can be used to distinguish between large and small strongyles in horses and to identify the various nematode species in ruminants. Most parasitology laboratories can perform this examination. It requires submission of 200 to 400 g of fresh feces. Pooled samples from several herd members are often used.
Pasture Larval Counts
Counts of parasitic larvae on herbage are useful to indicate the level of exposure experienced by the grazing animal. This examination is somewhat tedious but can be performed by many university laboratories. A 2-kg sample of forage is gathered for submission to the laboratory. The grass is sampled by walking a V pattern across the acreage and stopping every three paces to sample grass. The samples are subsequently washed and passed through screening to isolate and identify larvae.
Necropsy Evaluation
The nature and magnitude of parasitic infections can be established by necropsy examination. Gross examination and an estimate of adult worm population in the gut lumen are often sufficient. Many worms detach from the mucosa as the carcass cools; however, the damage done by the parasites can be seen on gross or histologic examination. Occasionally it is necessary to use digestion techniques or histologic examination to document the presence of hypobiotic larvae.
ANTHELMINTIC USE
Anthelmintic drugs are administered to treat, control, and prevent parasitic infections and to minimize the economic losses associated with parasitic infection. Anthelmintics are also used for the treatment of an individual animal exhibiting clinical signs of parasitic disease.
Drug Action
To use an anthelmintic properly, it is necessary to consider its mode of action, spectrum of activity, and duration of effect. Efficacy for a given drug may be defined as its ability to kill adult or larval parasites, suppress parasitic egg production, or promote the expulsion of worms from the GI tract.
Because of the emergence of drug-resistant strains of nematodes, it is difficult to predict how a particular drug will perform in a specific animal or herd. Package inserts should be regarded as guidelines rather than gospel. It is prudent to run periodic pretreatment and posttreatment fecal examinations to assess the performance of commonly used drugs on a given farm. Readers should refer to the sections on horses, cattle, and small ruminants for more specific recommendations.
ANTHELMINTIC DRUGS
Avermectins and Milbemycins
The avermectins and milbemycins are macrocyclic lactones. They act by increasing the permeability of parasite cell membranes to chloride ions, which results in nonspastic paralysis and death of the parasite. These drugs may also act by potentiating presynaptic release of γ-aminobutyric acid (GABA), an inhibitory neurotransmitter, although this theory has been challenged.
These products have a high level and broad spectrum of activity against adult and larval nematodes. They are also effective against various ectoparasites such as mites, lice, ticks, bots, and cattle grubs; however, they are ineffective against flukes and tapeworms. These drugs suppress nematode egg production for longer than other anthelmintics. Because of its long duration of effect, moxidectin suppresses fecal egg counts and protects against reinfection for longer than ivermectin. Concern has been expressed about the environmental impact of these long-acting anthelmintics in grazing animals.
Several products have been developed for use in animals. The avermectins include ivermectin, abamectin, doramectin, and eprinomectin. The milbemycins include nemadectin and moxidectin. Oral (drench, sustained-release intraruminal device), topical (“pour-on”), and injectable formulations are available, depending on the drug. Ivermectin is safe to use during pregnancy, but currently it is not approved for use in lactating dairy animals or females of breeding age. Similarly, doramectin and moxidectin are not approved for use in female dairy cattle of breeding age. Eprinomectin is unique in that it has no withholding period for milk or meat, so there are no restrictions on its use in cattle. At labeled doses, moxidectin is a safe drug; however, accidental overdosage has caused neurologic signs in foals and miniature horses.
Benzimidazoles
BZDs comprise a large class of anthelmintics that interfere with parasitic carbohydrate metabolism by inhibiting the enzyme fumarate reductase. Many BZDs have been developed and marketed. They include albendazole, fenbendazole, mebendazole, oxfendazole, oxibendazole, parbendazole, ricobendazole, thiabendazole, triclabendazole, and the probenzimidazole drugs febantel and netobimin.
BZDs are widely used in horses and ruminants. In general they exhibit a high degree of safety and a broad spectrum of activity against GINs and lungworms. Some members of this class (e.g., albendazole) are also active against liver flukes and certain cestodes in ruminants. Albendazole and netobimin can cause teratogenicity and embryo toxicity in sheep when given during early pregnancy. Fenbendazole, oxfendazole, and oxibendazole are considered to be safe for use in pregnant animals.
Resistance to BZDs has been documented in certain equine, ovine, and caprine parasites. In general a strain of parasite resistant to one BZD drug quickly develops resistance to other BZDs or pro-BZDs, a phenomenon known as side resistance.
Levamisole
Levamisole acts by causing neuromuscular depolarization and paralysis of the parasite. It has been widely used in ruminants to treat GIN and lungworm infections. However, levamisole resistance has become a problem in many areas.
The dose of levamisole should be calculated carefully because toxic doses are only one to two times therapeutic doses. Signs of toxicity may mimic those of OP toxicity, including muscle fasciculations around the lips and eyelids, hypersalivation, spastic movements, depression, and diarrhea. In ruminants, muzzle foam may develop after oral administration of the drug but usually disappears within a few hours after administration. In horses, transitory excitement has been seen after treatment. Levamisole is not recommended for use as an anthelmintic in horses. Concurrent administration of morantel, pyrantel, diethylcarbamazine, or OPs could enhance the toxic effects of levamisole.
Morantel and Pyrantel
The tetrahydropyrimidines morantel and pyrantel are cholinergic agonists that exert their anthelmintic effect by depolarizing neuromuscular junctions and causing irreversible paralysis of susceptible parasites. Morantel is slower in its onset of action but much more potent than pyrantel. These products are effective against many species of adult nematodes but do not appear to be active against larval stages. Pyrantel is also effective against tapeworms in horses when given at twice the recommended dose. The margin of safety is relatively wide, and there is no contraindication to use with other cholinergic drugs. However, it is recommended that morantel and pyrantel not be used concurrently and that neither be given with levamisole. Piperazine antagonizes the effects of morantel and pyrantel, so it should not be used with either of these drugs. Resistance to morantel and pyrantel has been documented in strains of H. contortus and in some cyathostomes (equine small strongyles).
Organophosphates
OPs block neurotransmission by inhibiting acetylcholinesterase. Various formulations of OP drugs are available for treating GI nematodiasis. Commonly used OPs include haloxon, coumaphos, trichlorfon, and dichlorvos. Toxicity occurs with these products in a dose-related manner, so dosages should be calculated with care. In addition, the potential danger to humans administering these products should not be overlooked. Atropine is recommended in cases of overdose in livestock.
Phenothiazine
The mode of action of phenothiazine (PTH) has not been clarified; it is thought to interfere with anaerobic metabolism of nematodes. The various formulations of PTH differ in purity and particle size. The purified product (99% PTH) with small particle size (2 μm) is the most effective.
Although PTH is effective against a wide spectrum of GINs, resistant strains of parasites have emerged in several species. The drug is synergistic with piperazine; combinations of these drugs have effective activity against PTH-resistant nematodes. PTH used in combination with piperazine can be administered at a much lower dose.
PTH toxicity has been reported. Toxic reactions include corneal inflammation, abortion, ataxia, hemolytic anemia, photosensitization, and nephrotoxicity. The drug should not be administered to debilitated or anemic animals or to animals in the last month of pregnancy.
Piperazine
Piperazine salts block neuromuscular transmission, resulting in paralysis of susceptible GINs. The worms are then passively removed from the GI tract by intestinal peristalsis. Piperazines have low toxicity and are safe in young or pregnant animals. However, their spectrum of activity is limited, in practical terms, to ascarids. Piperazine must be used with caution in horses heavily infested with ascarids because the paralyzed ascarids can cause an impaction that may culminate in bowel rupture. Diethylcarbamazine is a piperazine derivative that has been used to control lungworm infection in sheep and cattle.
Praziquantel
Praziquantel is a cesticidal drug that causes spastic paralysis, decreased glucose uptake, and disruption of the tapeworm’s tegument. Although not approved for this use, praziquantel is effective for treating tapeworm infestation in horses. It has also been used to control various cestodes in small ruminants.
COCCIDIOSIS IN FOOD ANIMALS
Coccidiosis causes serious economic losses in a variety of food animal species. Regular infections with a mixture of species generally occur throughout the life of the animal. Infections tend to be asymptomatic and self-limiting unless management or other factors allow the abnormal concentration of oocysts in the environment or when host defenses are compromised. Kids are especially susceptible to coccidiosis and may develop chronic diarrhea as a consequence. In many production operations, coccidiosis is an annual event.
Life-Cycle
Coccidiosis in ruminants is caused by intracellular parasites of the genus Eimeria. Essentially all of the species of Eimeria occurring in ruminants are rigidly host-specific. The life-cycle is complex, involving both sexual and asexual phases within the same host. Single-cell oocysts are passed in the feces and subsequently sporulate in the external environment to become infective. The sporulated oocysts are ingested by the host with contaminated food or water. Digestive enzymes activate the sporozoites within the oocysts, which excyst in the intestine and enter intestinal cells. The asexual phase of development, in which two or more cycles of merogony occurs, is then initiated. Asexual fission results in the production of merozoites. As the meronts mature, they rupture, releasing the merozoites, which enter other cells and repeat the cycle or progress to the sexual phase of development (gamegony). In this phase, merozoites enter new cells and produce macrogametes and microgametes. Microgametes are released by cell rupture and fertilize a macrogamete to form a zygote. A cyst wall then forms around the zygote, resulting in the next generation of oocysts. Once again the host cell ruptures, releasing oocysts into the lumen of the intestine, where they are then passed with the feces. The prepatent period is approximately 2 to 3 weeks and varies with the particular species of Eimeria.278,279
Pathophysiology
Most intestinal Eimeria organisms develop in the epithelial cells of the lamina propria, although a few exceptions (e.g., Eimeria bovis) do exist. As a result of the developmental cycle, particularly the sexual phase, infected host cells are destroyed. However, the degree of damage depends on the species of Eimeria involved, the number of oocysts ingested, and various host factors including age, physical condition, genetic susceptibility, and immunity from previous exposure.278,279 If the number of oocysts ingested is low, nonimmune healthy animals may tolerate infection and show no signs of disease. Intestinal cells are destroyed, but because they normally are replaced at a rapid rate the damage done to the gut is minimal. However, if nonimmune animals are exposed to many oocysts, widespread rupture and exfoliation of intestinal cells alters gut function; causes loss of blood, fluid, albumin, and electrolytes into the gut; and allows secondary bacterial invasion. Sections of sloughed intestinal mucosa and fibrin casts may be seen in the feces, and the feces may be blood tinged.
Neurologic signs have been reported during coccidiosis outbreaks caused by Eimeria zuernii in calves and weaned beef cattle.279,280 The pathophysiology of this manifestation has not been definitively established, and conflicting theories have been proposed, including copper imbalance, plasma electrolyte imbalances, a labile neurotoxin, and a combination of stressors in which coccidia are only one factor.279,281-283
Populations at Risk
Infections with mixtures of pathogenic and nonpathogenic species of Eimeria occur regularly throughout the life of mature animals; hence they provide the source of oocyst contamination leading to infections in young animals. After an initial period of infection the host acquires species-specific immunity. The degree of immunity acquired depends on the quantity of oocysts ingested and is boosted by continuous exposure to oocysts. It is possible that when exposed to few or moderate numbers of oocysts, a nonprotective immune response may occur; however, initial exposure is generally sufficient and animals are usually protected against clinical disease on reexposure to the same species.278,279 Clinical coccidiosis is therefore primarily a disease of young, nonimmune animals crowded together in areas such as feedlots, small pastures, shady creek bottoms during hot summer weather, or areas near feed or water tanks or salt licks. Under these conditions the environment is highly contaminated with oocysts from the immune animals, which pose a significant threat to other young, immunologically naive animals in the same areas. Stress, such as from shipping, weaning, dietary changes, and/or adverse weather (e.g., blizzards), appears to facilitate outbreaks.278,279 Corticosteroid treatment or concurrent illness can also precipitate a peracute form of coccidiosis.
Although outbreaks have been reported in range animals, coccidiosis is chiefly a disease of confinement. Clinical disease usually occurs in cattle less than 1 year of age. In dairy cattle, coccidiosis is most common in calves when they are taken from hutches into group calf pens or mini—free-stall barns. In beef cattle the disease is most prevalent in feedlot calves. In sheep, disease is usually limited to lambs less than 6 months old. Coccidiosis is most common in intensively reared lambs, although suckling lambs on pasture in constant use at high stocking rates are also at risk. Young kids appear particularly susceptible to coccidiosis, with clinical disease especially prevalent 2 to 3 weeks after weaning.284 Previously exposed animals may show clinical signs under conditions of stress and heavy infections. Occasionally, adult animals develop coccidiosis when moved into a new herd and exposed to different species of the parasite to which they are not yet immune.
Clinical Manifestations
The destruction of epithelial cells and subsequent loss of blood, albumin, fluid, and electrolytes typically cause a profuse, sometimes bloody, catarrhalic diarrhea. Dehydration may occur, but most animals continue to drink water and can meet their fluid requirements. Despite the blood loss, anemia is not usually apparent. Typical of infections in dairy cattle, light infections tend to cause watery feces, poor condition, and reduced weight gain. Severe infections cause projectile, bloody diarrhea with mucus, rectal tenesmus, inappetence, dehydration, and weight loss. Clinical signs last approximately 1 week. In lambs, clinical signs are similar to those of cattle except blood and tenesmus do not usually occur. In kids, clinical signs include pasty, watery diarrhea and dehydration.284 Sudden death can occur in both lambs and kids, but the case fatality rate is usually low in most outbreaks. After recovery from the disease the gut does not return to normal function for several weeks, and appetite may be suppressed concurrently, leading to poor growth and/or stunting. Animals that develop “nervous” coccidiosis may exhibit muscle tremors, hyperesthesia, convulsions, nystagmus, and blindness. The mortality rate is high.
Control of Coccidiosis
The spread of coccidiosis depends on the prevalence of oocysts in the environment. The level of infection is directly related to the level of fecal contamination. Houses and pens used for sequential groups of young animals often become highly contaminated and serve as the source of infection for subsequent groups. Therefore minimizing exposure of susceptible animals to infective oocysts depends on management techniques that focus on either sanitation to avoid the buildup of fecal contamination or the elimination of environmental conditions conducive to oocyst survival or both. Decreased stocking rates, proper manure disposal, and elevated feed bunks will help reduce contamination and exposure to oocysts. Sunlight, low humidity, and treatment with formaldehyde, ammonia, or methylbromide will kill oocysts. Reduction of average temperatures in barns to 15° C and of humidity to a maximum of 80% has significantly decreased clinical coccidiosis in some areas.278 Prophylactic use of coccidiostats is inevitable if conditions of animal husbandry cannot or do not improve.
DRUGS
The various drugs that have been used to prevent or treat coccidiosis in ruminants are listed in Table 49-5.285-290 These drugs are most often administered in the feed or water. Therapeutic anticoccidials are used in acute outbreaks; however, the gamonts are the main target of these drugs. Because the life-cycle of the parasite is essentially complete and most of the intestinal damage is done by the time anticoccidial therapy is administered, it is of limited value. In acute outbreaks, scouring calves should be removed from the group. Supportive therapy, including electrolytes, glucose, and antidiarrheals, may help improve survival.278,284
Table 49-5 Drugs Used for Treatment and Prevention of Coccidiosis in Ruminants
| Drug | Treatment* | Prevention* |
|---|---|---|
| Amprolium | 10 mg/kg bwt for 5 days (B) | 5 mg/kg bwt for 21 days (B) |
| 25–40 mg/kg bwt for 5 days (O, C) | 50 mg/kg bwt for 21 days (O) | |
| 65 mg/kg bwt once | ||
| Decoquinate | 0.5 mg/kg bwt for 28–30 days | |
| Diclazuril | 1 mg/kg bwt once; a second dose can be given 14 days later | |
| Lasalocid | 1 mg/kg bwt continuously (B) | |
| 25–100 mg/kg feed, continuously | ||
| 80 mg/kg milk replacer (neonatal calves) | ||
| Free choice in salt at 0.75% total salt mixture | ||
| Monensin | 2 mg/kg bwt for 20 days (O) | 1 mg/kg bwt for 28 days |
| 10–20 ppm in feed, continuously | ||
| 16–33 g/ton of feed, continuously | ||
| Intraruminal controlled-release device (weaned beef calves) | ||
| Nitrofurazone 10 | 15 mg/kg bwt for 5–7 days | 33 mg/kg bwt for 14 days |
| In feed at 0.04% or in water at 0.0133% for 7 days | In feed at 0.04% for 21 days | |
| Salinomycin | 10 ppm in feed (O) | |
| Sulfadimethoxine | 50 mg/kg bwt for 5 days (O) | |
| Sulfamethazine | 110 mg/kg bwt for 5 days | 35 mg/kg bwt for 15 days (B) |
| 140 mg/kg bwt for 3 days | 25 mg/kg bwt for 7 days (O) | |
| 140 mg/kg bwt once, then 70 mg/kg bwt for 5–7 days | 55 g/ton of feed (C) | |
| Sulfaquinoxaline | 10–20 mg/kg bwt for 5–7 days | |
| Toltrazuril | 20 mg/kg bwt | 20 mg/kg bwt once, 10 days after turnout onto pasture |
B, Bovine; bwt, body weight; C, caprine; O, ovine.
* Oral administration; dose or regimen may vary among references.Note: Coccidiostats used in poultry feeds may be toxic to ruminants.
To prevent losses from coccidiosis, it is best to treat exposed animals prophylactically or metaphylactically rather than therapeutically. Often outbreaks of coccidiosis can be predicted based on farm history. Administration of coccidiostats during this time does not completely prevent infections; rather, they prevent clinical infections without interfering with the production of protective immunity. Drugs in this category tend to target merogony, preventing multiplication and subsequent mucosal damage.278,279
OTHER STRATEGIES FOR MINIMIZING CLINICAL INFECTIONS
Use of growth implants containing estradiol and progesterone can attenuate the effects of high oocyst challenge in dairy calves.291 Attempts at developing a conventional vaccine against coccidiosis have been disappointing. Peptide or recombinant vaccines may prove to be efficacious in the future.292 Immunization with a “trickle dose” of oocysts 2 weeks before turnout onto infected pasture may attenuate the effects of natural challenge in calves.293 Oocysts can survive the hay-making process, so pastures known to be infected with coccidian oocysts should not be used to make hay for susceptible animals.294
Evaluation of Preventive Programs
Oocysts can be found using standard flotation techniques.284 However, the presence or absence of oocysts does not necessarily correlate with the presence of disease. In any given herd or group a baseline level of oocyst shedding is normal. Because immunity is incomplete, many immune animals shed oocysts. On the other hand, nonimmune animals with low-level infections may shed oocysts but have no clinical signs or need for treatment. Furthermore, because intestinal damage occurs during the prepatent period, animals with clinical coccidiosis may not yet be shedding oocysts. This variation complicates both evaluation of preventive measures and the diagnosis of coccidiosis in clinically affected animals. The presence of oocysts in healthy or sick animals does not necessarily indicate the need for anticoccidial treatment.
Nevertheless, microscopic examination of diarrheic or bloody feces from young animals still remains the most direct and cost-effective method of diagnosis.278,279 Examination of fecal samples from several animals is needed to obtain a true estimate of the presence of coccidia within a group. Because pathogenicity of the various species of Eimeria varies considerably and the clinical picture is not specific, it is imperative that species identifications be determined.277 This information is then combined with a good history and thorough knowledge of the herd management on the farm in question to guide interpretation of clinical observations and diagnostics.
Herds that require preventive strategies have a history of prior disease and a large proportion of herd members shedding high numbers of oocysts. Efficacy is evaluated on the basis of a decreased prevalence of clinically ill animals.
Clinical Management
DIAGNOSIS
Definitive diagnosis may be made at necropsy. Antemortem, young, diarrheic animals passing large numbers of oocysts are not difficult to diagnose. However, as discussed earlier, making a diagnosis on the basis of oocyst counts alone can be misleading.
TREATMENT
Drugs used in the treatment of coccidiosis are listed in Table 49-5. Supportive therapy, when indicated, is primarily directed at replacing fluid losses and supporting the animal until the gut epithelium regenerates.
1 Love S, Murphy D, Mellor D. Pathogenicity of cyathostome infection. Vet Parasitol. 1999;85:113.
2 Ogbourne CP. Pathogenesis of cyathostome (Trichonema) infection of the horse. Slough, UK: Commonwealth Institute of Helminthology, Commonwealth Agricultural Bureaux, 1978;1. A review, Miscellaneous publication no. 5, Farnham Royal,
3 Reinemeyer CR. Small strongyles. Recent advances. Vet Clin North Am Equine Pract. 1986;2:281.
4 Klei T, French D, Monahan CM, et al. Epidemiology of equine gastrointestinal parasites in southern Louisiana. Proceedings of the 39th Annual Meeting of the American Association of Veterinary Parasitologists, San Francisco. 1994:48.
5 Eysker M, Jansen J, Mirck MH. Inhibited development of Cyathostominae in the horse in the early third stage. Res Vet Sci. 1984;37:355.
6 Kaplan RM. Anthelmintic resistance in nematodes of horses. Vet Res. 2002;33:491.
7 Lyons ET, Drudge JH, Tolliver SC. Larval cyathostomiasis. Vet Clin North Am Equine Pract. 2000;16:501.
8 Gibson TE. The effect of repeated anthelmintic treatment with phenothiazine on the faecal egg counts of housed horses, with some observations on the life cycle of Trichonema spp in the horse. J Helminthol. 1953;27:29.
9 Love S, Murphy D. The pathogenic effects of experimental cyathostome infections in ponies. Vet Parasitol. 1997;70:99.
10 Love S, Escala J, Duncan J, et al. Studies on the pathogenic effects of experimental cyathostome infections in ponies. Proceedings of the 6th International Conference on Equine Infectious Diseases, Newmarket. R & W Publications; 1991:149.
11 Round M. The development of strongyles in horses and the associated serum protein changes. Proceedings of the 2nd International Conference of Equine Infectious Diseases, Basel, Karger. 1970:290.
12 Giles C, Urquhart K, Longstaffe J. Larval cyathostomiasis (immature Trichonema-induced enteropathy): a report of 15 clinical cases. Equine Vet J. 1985;17:196.
13 Reinemeyer C, Smith S, Gabel A, et al. Observations on the population dynamics of five cyathostome nematode species of horses in northern USA. Equine Vet J. 1986;18:121.
14 Klei TR, Chapman MR. Immunity in equine cyathostome infections. Vet Parasitol. 1999;85:123.
15 Mair T. Outbreak of larval cyathostomiasis among a group of yearling and 2-year-old horses. Vet Rec. 1994;135:598.
16 Steinbach T, Bauer C, Sasse H. Small strongyle infection: Consequences of larvicidal treatment of horses with fenbendazole and moxidectin. Vet Parasitol. 2006;139:115.
17 Mair T, Cripps P, Rickets S. Diagnostic and prognostic value of serum protein electrophoresis in horses with chronic diarrhoea. Equine Vet J. 1993;25:324.
18 Reilly B, Cassidy J, Taylor S. Two cases of diarrhea in horses associated with larvae of the small strongyles. Vet Rec. 1993;132:67.
19 Murphy D, Keane M, Chandler K, et al. Cyathostome-associated disease in the horse: investigation and management of four cases. Equine Vet Educ. 1997;9:247.
20 Mair T, Sutton D, Love S. Caecocaecal and caecocolic intussusceptions associated with larval cyathostomosis in four young horses. Equine Vet J Suppl. 2000;32:77.
21 Lyons E, Swercaek T, Tolliver S, et al. A study of natural infections of encysted small-strongyles in a horse herd in Kentucky. Vet Med. 1994;89:1146.
22 Mair T, Pearson G. Multifocal non-strangulating intestinal infarction associated with larval cyathostomiasis in a pony. Equine Vet J. 1995;27:154.
23 Ogbourne C, Duncan J. Strongylus vulgaris in the horse: its biology and veterinary importance. Slough, UK: Commonwealth Institute of Helminthology, Commonwealth Agricultural Bureaux, 1985. 2, Farnham Royal,
24 Drudge JH, Lyons ET. Large strongyles. Recent advances. Vet Clin North Am Equine Pract. 1986;2:263.
25 Duncan J. The life cycle, pathogenesis and epidemiology of S. vulgaris in the horse. Equine Vet J. 1973;5:20.
26 McCraw B, Slocombe J. Strongylus equinus: development and pathological effects in the equine host. Can J Comp Med. 1985;49:372.
27 McCraw B, Slocombe J. Strongylus edentatus: development and lesions from ten weeks postinfection to patency. Can J Comp Med. 1978;42:340.
28 McCraw B, Slocombe J. Early development of and pathology associated with Strongylus edentatus. Can J Comp Med. 1974;38:124.
29 Wetzel R. Die Entwicklungsdauer (Prepatentperiode) von Strongylus edentatus im Pferde. Dtsch Tierarztl Wochenschr. 1952;59:129.
30 Wetzel R, Kersten W. Die Leberphase der Entwicklund von Strongylus edentatus. Wien Tierarztl Monatsschr. 1956;43:664.
31 Herd R, Coles GC. Slowing the spread of anthelmintic resistant nematodes of horses in the United Kingdom. Vet Res. 1995;136:481.
32 Proudman CJ, Trees AJ. Tapeworms as a cause of intestinal disease in horses. Parasitol Today. 1999;15:156.
33 Williamson R, Beveridge I, Gasser R. Coprological methods for the diagnosis of Anoplocephala perfoliata infection of the horse. Aust Vet J. 1998;76:618.
34 Lyons E, Tolliver S, Drudge J, et al. Parasites in Kentucky thoroughbreds at necropsy: emphasis on stomach worms and tapeworms. Am J Vet Res. 1993;44:839.
35 Kania S, Reinemeyer C. Anoplocephala perfoliata coproantigen detection: a preliminary study. Vet Parasitol. 2005;127:115.
36 Tolliver SC, Lyons ET, Drudge JH. Prevalence of internal parasites in horses in critical tests of activity of parasiticides over a 28-year period (1956–1983) in Kentucky. Vet Parasitol. 1987;23:273.
37 Chapman MR, French DD, Klei TR. Gastrointestinal helminths of ponies in Louisiana: a comparison of species currently prevalent with those present 20 years ago. J Parasitol. 2002;88:1130.
38 Lyons ET, Tolliver SC, Collins SS. Prevalence of large endoparasites at necropsy in horses infected with Population B small strongyles in a herd established in Kentucky in 1966. Parasitol Res. 2006;99:114.
39 Lyons ET, Swerczek TW, Tolliver SC, et al. Prevalence of selected species of internal parasites in equids at necropsy in central Kentucky (1995–1999). Vet Parasitol. 2000;92:51.
40 Reinemeyer C, Farley A, Kania S, et al. A prevalence survey of antibodies to Anoplocephala perfoliata in horses from the United States. Proceedings of the 48th Annual Meeting of the American Association of Veterinary Parasitologists, Denver. 2003. July
41 Proudman C. Immunoepidemiology of the equine tapeworm Anoplocephala perfoliata: age-intensity profile and age-dependency of antibody subtype responses. Parasitology. 1997;114:89.
42 Marchiondo A, White G, Smith L, et al. Clinical field efficacy and safety of pyrantel pamoate paste (19.13% w/w pyrantel base) against Anoplocephala spp in naturally infected horses. Vet Parasitol. 2006;137:94.
43 Lyons ET, Tolliver SC, Stamper S, et al. Activity of praziquantel (0.5 mg kg-1) against Anoplocephala perfoliata (Cestoda) in equids. Vet Parasitol. 1995;56:255.
44 Rehbein S, Holste JE, Doucet MY, et al. Field efficacy of ivermectin plus praziquantel oral paste against naturally acquired gastrointestinal nematodes and cestodes of horses in North America and Europe. Vet Ther. 2003;4:220.
45 Grubbs ST, Amodie D, Rulli D, et al. Field evaluation of moxidectin/praziquantel oral gel in horses. Vet Ther. 2003;4:249.
46 Hoglund J, Nilsson O, Ljungstrom BL, et al. Epidemiology of Anoplocephala perfoliata infection in foals on a stud farm in south-western Sweden. Vet Parasitol. 1998;75:71.
47 Nilsson O, Ljungstrom BL, Hoglund J, et al. Anoplocephala perfoliata in horses in Sweden: prevalence, infection levels and intestinal lesions. Acta Vet Scand. 1995;36:319.
48 Williamson RM, Gasser RB, Middleton D, Beveridge I. The distribution of Anoplocephala perfoliata in the intestine of the horse and associated pathological changes. Vet Parasitol. 1997;73:225.
49 Rodriguez-Bertos A, Corchero J, Castano M, et al. Pathological alterations caused by Anoplocephala perfoliata infection in the ileocaecal junction of equids. Zentralbl Veterinarmed A. 1999;46:261.
50 Pearson GR, Davies LW, White AL, O’Brien JK. Pathological lesions associated with Anoplocephala perfoliata at the ileo-caecal junction of horses. Vet Rec. 1993;132:179.
51 Proudman CJ, French NP, Trees AJ. Tapeworm infection is a significant risk factor for spasmodic colic and ileal impaction colic in the horse. Equine Vet J. 1998;30:194.
52 Proudman CJ, Holdstock NB. Investigation of an outbreak of tapeworm-associated colic in a training yard. Equine Vet J Suppl. 2000;32:37.
53 Roberts CT, Slone DE. Caecal impactions managed surgically by typhlotomy in 10 cases (1988–1998). Equine Vet J Suppl. 2000;32:74.
54 Little D, Blikslager AT. Factors associated with development of ileal impaction in horses with surgical colic: 78 cases (1986–2000). Equine Vet J. 2002;34:464.
55 Cosgrove J, Sheeran J, Sainty T. Intussusception associated with infection with Anoplocephala perfoliata in a two year old thoroughbred. Ir Vet J. 1986;40:35.
56 Barclay W, Philips T, Foerner J. Intussusception associated with Anoplocephala perfoliata infection in five horses. J Am Vet Med Assoc. 1982;180:752.
57 Owen R, Jagger D, Quan-Taylor R. Caecal intussusceptions in horses and the significance of Anoplocephala perfoliata. Vet Rec. 1989;124:34.
58 Beroza G, Barclay W, Philips T, et al. Caecal perforation and peritonitis associated with Anoplocephala perfoliata infection in three horses. J Am Vet Med Assoc. 1983;183:04.
59 Roelfstra L, Betschart B, Pfister K. A study on the seasonal epidemiology of Anoplocephala spp infection in horses and the appropriate treatment using a praziquantel gel (Droncit 9% oral gel). Berl Munch Tierarztl Wochenschr. 2006;119:312.
60 Meana A, Pato NF, Martin R, et al. Epidemiological studies on equine cestodes in central Spain: infection pattern and population dynamics. Vet Parasitol. 2005;130:233.
61 Chapman MR, French DD, Klei TR. Seasonal transmission of gastrointestinal parasites of equids in southern Louisiana. J Parasitol. 2001;87:1371.
62 Bucknell DG, Gasser RB, Beveridge I. The prevalence and epidemiology of gastrointestinal parasites of horses in Victoria, Australia. Int J Parasitol. 1995;25:711.
63 Lyons ET, Tolliver SC, Stamper S, et al. Transmission of some species of internal parasites in horses born in 1990, 1991, and 1992 in the same pasture on a farm in central Kentucky. Vet Parasitol. 1994;52:257.
64 Benton RE, Lyons ET. Survey in central Kentucky for prevalence of Anoplocephala perfoliata in horses at necropsy in 1992. Vet Parasitol. 1994;55:81.
65 Lyons ET, Tolliver SC, Collins SS, Drudge JH. Transmission of endoparasites in horse foals born on the same pasture on a farm in central Kentucky (1996–1999). Vet Parasitol. 2001;97:113.
66 Slocombe J. A modified critical test and its use in two dose titration trials to assess efficacy of praziquantel for Anoplocephala perfoliata in equids. Vet Parasitol. 2006;136:127.
67 Reinemeyer C, Hutchens D, Eckblad W, et al. Dose-confirmation studies of the cesticidal activity of pyrantel pamoate paste in horses. Vet Parasitol. 2006;138:234.
68 Greiner E, Lane T. Effects of the daily feeding of pyrantel tartrate on Anoplocephala infections in three horses: a pilot study. J Equine Vet Sci. 1994;14:43.
69 Kivipelto J, Nicklin C, Asquith R. Comparison of pyrantel tartrate (Strongid C) and pyrantel pamoate (3x Strongid) for the control of equine tapeworms. AAEP Proc. 1998;44:236.
70 Blagburn B, Linsay D, Hendrix C, Schumacher J. Pathogenesis, treatment and control of gastric parasites in horses. Compend Cont Educ (Pract Vet). 1991;13:850.
71 Price RE, Stromberg PC. Seasonal occurrence and distribution of Gasterophilus intestinalis and Gasterophilus nasalis in the stomachs of equids in Texas. Am J Vet Res. 1987;48:1225.
72 Griss R, Simhofer H. First-time endoscopic detection of larvae of Gasterophilus spp in the oral cavity in 14 warmblood horses. Berl Munch Tierarztl Wochenschr. 2006;119:416.
73 Tolliver S, Lyons E, Drudge J. Observations on the specific location of Gasterophilus spp. larvae in the mouth of the horse. J Parasitol. 1974;60:891.
74 Cogley T. Effects of migrating Gasterophilus intestinalis larvae (Diptera: Gasterophilidae) on the mouth of the horse. Vet Parasitol. 1989;31:317.
75 Cogley T, Cogley M. Inter-relationship between Gasterophilus larvae and the horse’s gastric and duodenal wall with special reference to penetration. Vet Parasitol. 1999;86:127.
76 Dart A, Hutchins D, Begg A. Suppurative splenitis and peritonitis in a horse after gastric ulceration caused by larvae of Gasterophilus intestinalis. Aust Vet J. 1987;64:155.
77 Bello T. Efficacy of ivermectin against experimental and natural infections of Gasterophilus spp in ponies. Am J Vet Res. 1989;50:2120.
78 Reinemeyer CR, Scholl PJ, Andrews FM, Rock DW. Efficacy of moxidectin equine oral gel against endoscopically-confirmed Gasterophilus nasalis and Gasterophilus intestinalis (Diptera: Oestridae) infections in horses. Vet Parasitol. 2000;88:287.
79 Clayton HM, Duncan JL. The development of immunity to Parascaris equorum infection in the foal. Res Vet Sci. 1979;26:383.
80 Clayton H, Duncan J. The migration and development of Parascaris equorum. Int J Parasitol. 1979;9:285.
81 Srihakim D, Swerczek T. Pathologic changes and pathogenesis of Parascaris equorum infection in parasite-free pony foals. Am J Vet Res. 1978;39:1155.
82 Fairbairn D. The biochemistry of Ascaris. Exp Parasitol. 1957;6:491.
83 Clayton H. Experimental Parascaris equorum infection in the foal [PhD thesis]. Glasgow, Scotland: University of Glasgow, 1978.
84 Clayton H. Clinical signs associated with infection in worm free pony foals and yearlings. Vet Parasitol. 1978;4:69.
85 Clayton H. Ascarids recent advances. Vet Clin North Am Equine Pract. 1986;2:313.
86 Southwood L, Baxter G, Bennett D, Ragle C. Ascarid impaction in young horses. Compend Cont Educ (Pract Vet). 1998;20:100.
87 Lyons ET, Drudge JH, Tolliver SC. On the life cycle of Strongyloides westeri in the equine. J Parasitol. 1973;59:780.
88 Greer GJ, Bello TR, Amborski GF. Experimental infection of Strongyloides westeri in parasite-free ponies. J Parasitol. 1974;60:466.
89 Mirck M, Franken P. Strongyloides westeri Ihle, 1917 [Nematoda: Strongyloididae. II. Parasitological and haematological features of experimental infection (author’s translation)]. Tijdschr Diergeneeskd. 1978;103:355.
90 Ludwig KG, Craig TM, Bowen JM, et al. Efficacy of ivermectin in controlling Strongyloides westeri infections in foals. Am J Vet Res. 1983;44:314.
91 Drudge JH, Lyons ET, Tolliver SC, Kubis JE. Critical tests and clinical trials on oxibendazole in horses with special reference to removal of Parascaris equorum. Am J Vet Res. 1979;40:758.
92 Ryan WG, Best PJ. Efficacy of ivermectin paste against Strongyloides westeri in foals. Vet Rec. 1985;117:169.
93 Michael JF. Strategies for the use of anthelmintics in livestock and their implications for the development of drug resistance. Parasitology. 1985;90:621.
94 Barnes EH, Dobson RJ. Population dynamics of Trichostrongylus columbriformis in sheep: computer model to stimulate grazing systems and the evaluation of anthelmintic resistance. Int J Parasitol. 1990;20:823.
95 Uhlinger C, Kristula M. Effects of alternation of drug classes on the development of oxibendazole resistance in a herd of horses. J Am Vet Med Assoc. 1992;201:51.
96 Reinemeyer R, Henton JE. Observations on equine strongyle control in southern temperate USA. Equine Vet J. 1987;19:505.
97 Little D, Flowers JR, Hammerberg BH, Gardner SY. Management of drug-resistant cyathostominosis on a breeding farm in central North Carolina. Equine Vet J. 2003;35:246.
98 Nielsen MK, Haaning N, Olsen SN. Strongyle egg shedding consistency in horses on farms using selective therapy in Denmark. Vet Parasitol. 2006;35:333.
99 Uhlinger CA. Uses of fecal egg count data in equine practice. Compend Cont Educ (Pract Vet). 1993;13:863.
100 Kettle P. Drenching policy can help breed resistant worms. N Z J Agric. 1980;141:63.
101 Webb R. Epidemiological factors contributing to a high incidence of anthelmintic resistance in field populations in Haemonchus contortus. In: WA Geering, Roe RT, Chapman LA, editors. Proceedings of the 2nd International Symposium on Veterinary Epidemiology and Economics. Canberra: Australia Government Publishing; 1980:220. May 7–11, 1979
102 Herd RP, Willardson KL, Gabel AA. Epidemiological approach to the control of horse strongyles. Equine Vet J. 1985;17:202.
103 Nielsen MK, Kaplan RM, Thamsborg SM, et al. Climatic influences on development and survival of free-living stages of equine strongyles: implications for worm control strategies and managing anthelmintic resistance. Vet J. 2007;174:23.
104 Baudena MA, Chapman MR, French DD, Klei TR. Seasonal development and survival of equine cyathostome larvae on pasture in south Louisiana. Vet Parasitol. 2000;88:51.
105 Kuzmina TA, Kuzmin YI, Kharchenko VA. Field study on the survival, migration and overwintering of infective larvae of horse strongyles on pasture in central Ukraine. Vet Parasitol. 2006;141:264.
106 Helle O, Velle W, Tharaldsen J. Effect of ovine urine and some of its components on viability of nematode eggs and larvae in sheep faeces. Vet Parasitol. 1989;32:349.
107 Boersema JH, Eysker M, van der Aar WM. The reappearance of strongyle eggs in the faeces of horses after treatment with moxidectin. Vet Q. 1998;20:15.
108 Boersema JH, Eysker M, Maas J, van der Aar WM. Comparison of the reappearance of strongyle eggs on foals, yearlings and adult horses after treatment with ivermectin or pyrantel. Vet Q. 1996;18:7.
109 Lind EO, Kuzmina T, Uggla A, et al. A field study on the effect of some anthelmintics on cyathostomins of horses in Sweden. Vet Res Commun. 2007;31:53.
110 von Samson-Himmelstjerna G, Fritzen B, Demeler J, et al. Cases of reduced cyathostomin egg-reappearance period and failure of Parascaris equorum egg count reduction following ivermectin treatment as well as survey on pyrantel efficacy on German horse farms. Vet Parasitol. 2007;144:74.
111 Mercier P, Chick B, Alves-Branco F, White CR. Comparative efficacy, persistent effect, and treatment intervals of anthelmintic pastes in naturally infected horses. Vet Parasitol. 2001;99:29.
112 Briggs K, Reinemeyer C, French D, Kaplan R. Control programs for mature horses, getting your veterinarian involved. Horse. 2004:52. December
113 Rolfe PF, Dawson KL, Holm-Martin M. Efficacy of moxidectin and other anthelmintics against small strongyles in horses. Aust Vet J. 1998;76:332.
114 Kaplan RM, Klei TR, Lyons ET, et al. Prevalence of anthelmintic resistant cyathostomes on horse farms. J Am Vet Med Assoc. 2004;225:903.
115 Herd RP. Epidemiology and control of equine strongylosis at Newmarket. Equine Vet J. 1986;18:447.
116 Matthee S, McGeoch MA. Helminths in horses: use of selective treatment for the control of strongyles. J S Afr Vet Assoc. 2004;75:129.
117 Duncan JL, Bairden K, Abbott EM. Elimination of mucosal cyathostome larvae by five daily treatments with fenbendazole. Vet Rec. 1998;142:268.
118 Xiao L, Herd RP, Majewski GA. Comparative efficacy of moxidectin and ivermectin against hypobiotic and encysted cyathostomes and other equine parasites. Vet Parasitol. 1994;53:83.
119 Eysker M, Boersema JH, Grinwis GC, et al. Controlled dose confirmation study of a 2% moxidectin equine gel against equine internal parasites in The Netherlands. Vet Parasitol. 1997;70:165.
120 Lyons ET, Tolliver SC. Field test data on small strongyles in evaluation of activity of fenbendazole given once a day for 5 consecutive days to thoroughbred yearlings on two farms in Kentucky in 2002 and 2003. Parasitol Res. 2003;91:312.
121 Coles GC, Jackson F, Pomroy WE, et al. The detection of anthelmintic resistance in nematodes of veterinary importance. Vet Parasitol. 2006;136:167.
122 Tarigo-Martinie JL, Wyatt AR, Kaplan RM. Prevalence and clinical implications of anthelmintic resistance in cyathostomes of horses. J Am Vet Med Assoc. 2001;218:1957.
123 Hearn FP, Peregrine AS. Identification of foals infected with Parascaris equorum apparently resistant to ivermectin. J Am Vet Med Assoc. 2003;223:482.
124 Slocombe JO, de Gannes RV, Lake MC. Macrocyclic lactone—resistant Parascaris equorum on stud farms in Canada and effectiveness of fenbendazole and pyrantel pamoate. Vet Parasitol. 2006;145:371.
125 Monahan CM, Chapman MR, Taylor HW, et al. Foals raised on pasture with or without daily pyrantel tartrate feed additive: comparison of parasite burdens and host responses following experimental challenge with large and small strongyle larvae. Vet Parasitol. 1997;73:277.
126 Cribb NC, Cote NM, Boure LP, Peregrine AS. Acute small intestinal obstruction associated with Parascaris equorum infection in young horses: 25 cases (1985–2004). NZ Vet J. 2006;54:338.
127 Briggs K, Reinemeyer C, French D, Kaplan R. Age related parasites: foals and young horses. Horse. 2004. November
128 French DD, Klei TR, Taylor HW, Chapman MR. Efficacy of ivermectin in oral drench and paste formulation against migrating larvae of experimentally inoculated Parascaris equorum. Am J Vet Res. 1989;50:1071.
129 Vandermyde CR, DiPietro JA, Todd KSJr, Lock TF. Evaluation of fenbendazole for larvicidal effect in experimentally induced Parascaris equorum infections in pony foals. J Am Vet Med Assoc. 1987;190:1548.
130 Briggs K, Reinemeyer C, French D, Kaplan R. Dewormer adjuncts: control without or along with chemicals. Horse. 2004. October
131 Blagburn B, Lindsay D, Hendrix C, Schumacher J. Pathogenesis, treatment and control of gastric parasites in horses. Compend Cont Educ Pract Vet. 1991;13:850.
132 Collobert-Laugier C, Lamidey C, Brisseau N, et al. Prevalence of stomach nematodes (Habronema spp, Draschia megastoma and Trichostrongylus axei) in horses examined post mortem in Normandy. Rev Med Vet. 2000;151:151.
133 Lyons E, Tolliver S, Drudge J, et al. Common internal parasites found in the stomach, large intestine, and cranial mesenteric artery of thoroughbreds in Kentucky at necropsy (1985 to 1986). Am J Vet Res. 1987;48:268.
134 Lyons E, Tolliver S, Drudge J, et al. Parasites in Kentucky thoroughbreds at necropsy: emphasis on stomach worms and tapeworms. Am J Vet Res. 1983;44:839.
135 Scialdo R. A survey of stomach parasites of horses from the southwestern, southern and central states. Southwest Vet. 1977;30:155.
136 Vasey JR. Equine cutaneous habronemiasis. Compend Cont Educ (Pract Vet). 1981;8:290.
137 Scott DW. Habronemiasis. In: Scott DW, editor. Large animal dermatology. Philadelphia: Saunders; 1988:251.
138 Lyons E, Tolliver S, Drudge J, et al. Prevalence of Anoplocephala perfoliata and lesions of Draschia megastoma in thoroughbreds in Kentucky at necropsy. Am J Vet Res. 1984;45:996.
139 Reinemeyer C, Smith S, Gabel A, Herd R. The prevalence and intensity of internal parasites of horses in USA. Vet Parasitol. 1984;15:75.
140 Mellor P. Studies on Onchocerca cervicalis Railliet and Henry 1910: II. Pathology in the horse. J Helminthol. 1973;47:111.
141 Lyons ET, Drudge JH, Tolliver SC. Verification of ineffectual activity of ivermectin against adult Onchocerca spp in the ligamentum nuchae of horses. Am J Vet Res. 1988;49:983.
142 Rabalais F, Votava C. Cutaneous distribution of microfilariae of Onchocerca cervicalis in horses. Am J Vet Res. 1974;35:1369.
143 French D, Klei T, Foil C, et al. Efficacy of ivermectin in paste and injectable formulations against microfilariae of Onchocerca cervicalis and resolution of associated dermatitis in horses. Am J Vet Res. 1988;49:1550.
144 Monahan C, Chapman M, French D, Klei T. Efficacy of moxidectin oral gel against Onchocerca cervicalis microfilariae. J Parasitol. 1995;81:117.
145 Gibbs HC, Herd RP. Nematodiasis in cattle. Vet Clin N Am Food Anim Pract. 1986;2:211.
146 Ballweber LR. Endoparasite control. Vet Clin North Am Food Anim Pract. 2006;22:451.
147 Yazwinski TA, Tucker CA. A sampling of factors relative to the epidemiology of gastrointestinal nematodosis of cattle in the United States. Vet Clin North Am Food Anim Pract. 2006;22:501.
148 Reinemeyer CR. Parasitisms of dairy and beef cattle in the United States. J Am Vet Med Assoc. 1994;205:670.
149 Stromberg BE, Gasbarre LC. Gastrointestinal nematode control programs with an emphasis on cattle. Vet Clin North Am Food Anim Pract. 2006;22:543.
150 Williams JC, Bilkovich FR. Development and survival of infective larvae of the cattle nematode. Ostertagia ostertagi, J Parasitol. 1971;57:327.
151 Williams JC, Bilkovich FR. Distribution of Ostertagia ostertagi infective larvae on pasture herbage. Am J Vet Res. 1973;34:1337.
152 Slocombe JOD. Overwintering of bovine gastrointestinal nematodes in southwestern Ontario. Can J Comp Med. 1974;38:90.
153 Gibbs HC. Relative importance of winter survival of larval nematodes in pasture and infected carrier calves in a study of parasitic gastroenteritis in calves. Am J Vet Res. 1979;40:227.
154 Gibbs HC. Persistence in pasture of the infective larvae of nematodes parasitizing Maine dairy cattle. Am J Vet Res. 1980;41:1694.
155 Frank GR, Herd RP, Marbury KS, Williams JC. Effects of transfer of Ostertagia ostertagi between northern and southern U.S.A. on the pattern and frequency of hypobiosis. Int J Parasitol. 1986;16:391.
156 Frank GR, Herd RP, Marbury KS, et al. Additional investigations on hypobiosis of Ostertagia ostertagi after transfer between northern and southern U.S.A. Int J Parasitol. 1988;18:171.
157 Simpson HV. Pathophysiology of abomasal parasitism: is the host or parasite responsible. Vet J. 2000;160:177.
158 Fox MT. Pathophysiology of infection with gastrointestinal nematodes in domestic ruminants: recent developments. Vet Parasitol. 1997;72:285.
159 Elek P, Durie PH. The histopathology of the reactions of calves to experimental infection with the nodular worm, Oesophagostomum radiatum (Rudolphi, 1803) II. Reaction of the susceptible host to infection with a single dose of larvae. Aust J Agric Res. 1966;18:549.
160 Williams JC, DeRosa A, Nakamura Y, Loyacano AF. Comparative efficacy of ivermectin pour-on, albendazole, oxfendazole and fenbendazole against Ostertagia ostertagi inhibited larvae, other gastrointestinal nematodes and lungworm of cattle. Vet Parasitol. 1997;73:73.
161 Williams JC, Stuedemann JA, Bairden K, et al. Efficacy of a pour-on formulation of eprinomectin (MK-397) against nematode parasites of cattle, with emphasis on inhibited early fourth-stage larvae of Ostertagia spp. Am J Vet Res. 1997;58:379.
162 Yazwinski TA, Tucker C, Copeland S, et al. Dose confirmation of moxidectin pour-on against natural nematode infections in lactating dairy cows. Vet Parasitol. 1999;86:223.
163 Jacobs DE, Fisher MA, Hutchinson MJ, et al. An evaluation of abamectin given at turnout and six weeks after turnout for the control of nematode infections in calves. Vet Rec. 1995;136:386.
164 Eysker M, Boersema JH, Cornelissen JB, et al. Residual effect of injectable moxidectin against lungworm and gastrointestinal nematodes in calves exposed to high pasture infectivity levels in the Netherlands. Vet Parasitol. 1996;61:61.
165 Barth D, Ericsson GF, Kunkle BN, et al. Evaluation of the persistence of the effect of ivermectin and abamectin against gastrointestinal and pulmonary nematodes in cattle. Vet Rec. 1997;140:278.
166 Ballweber LR, Smith LL, Stuedemann JA, et al. The effectiveness of a single treatment with doramectin or ivermectin in the control of gastrointestinal nematodes in grazing yearling stocker cattle. Vet Parasitol. 1997;72:53.
167 Taylor SM, Mallon TR, Kenny J, Edgar H. A comparison of early and mid grazing season suppressive anthelmintic treatments for first-year grazing calves and their effects on natural and experimental infection during the second year. Vet Parasitol. 1995;56:75.
168 Kerboeuf D, Hoste H, Hubert J, Le Stang JP. Response of cattle treated with a fenbendazole slow release bolus to challenge from nematodes the following season. Vet Parasitol. 1996;62:107.
169 Schnieder T, Epe C, Von Samson-Himmelstjerna G, Kohlmetz C. The development of protective immunity against gastrointestinal nematode and lungworm infections after use of an ivermectin bolus in first-year grazing calves. Vet Parasitol. 1996;64:239.
170 Bauer C, Holtemoller H, Schmid K. Field evaluation of a fenbendazole slow release bolus in the control of nematode infections in first-season cattle. Vet Rec. 1997;140:395.
171 Claerebout E, Dorny P, Vercruysse J, et al. Effects of preventive anthelmintic treatment on acquired resistance to gastrointestinal nematodes in naturally infected cattle. Vet Parasitol. 1998;76:287.
172 Eddi C, Muniz RA, Caracostantogolo J, et al. Comparative persistent efficacy of doramectin, ivermectin and fenbendazole against natural nematode infections in cattle. Vet Parasitol. 1997;72:33.
173 Hooke FG, Clement P, Dell’Osa D, et al. Therapeutic and protective efficacy of doramectin injectable against gastrointestinal nematodes in cattle in New Zealand: a comparison with moxidectin and ivermectin pour-on formulations. Vet Parasitol. 1997;72:43.
174 Williams JC, Loyacano AF, Broussard SD, et al. Duration of anthelmintic efficacy of doramectin and ivermectin injectable solutions against naturally acquired nematode infections of cattle. Vet Parasitol. 1997;72:15.
175 Williams JC, Loyacano AF, DeRosa A, et al. A comparison of the efficacy of two treatments of doramectin injectable, ivermectin injectable and ivermectin pour-on against naturally acquired gastrointestinal nematode infections of cattle during a winter-spring grazing season. Vet Parasitol. 1997;72:69.
176 Williams JC, Loyacano AF, DeRosa A, et al. A comparison of persistent anthelmintic efficacy of topical formulations of doramectin, ivermectin, eprinomectin and moxidectin against naturally acquired nematode infections of beef calves. Vet Parasitol. 1999;85:277.
177 Ranjan S, Trudeau C, Prichard RK, et al. Nematode reinfection following treatment of cattle with doramectin and ivermectin. Vet Parasitol. 1997;72:25.
178 Vercruysse J, Dorny P, Claerebout E, et al. Evaluation of the persistent efficacy of doramectin and ivermectin injectable against Ostertagia ostertagi and Cooperia oncophora in cattle. Vet Parasitol. 2000;89:63.
179 Vercruysse J, Claerebout E, Dorny P, et al. Persistence of the efficacy of doramectin against Ostertagia ostertagi and Cooperia oncophora in cattle. Vet Rec. 1998;143:443.
180 Molento MB, Trudeau C, Prichard RK, et al. Persistent efficacy of doramectin pour-on against artificially induced infections of nematodes in cattle. Vet Parasitol. 1999;82:297.
181 McKenna PB. Anthelmintic resistance in cattle nematodes in New Zealand: is it increasing? NZ Vet J. 1996;44:76.
182 Yadav CL, Verma SP. Morantel resistance by Haemonchus placei in cattle. Vet Rec. 1997;141:499.
183 Anziani OS, Zimmermann G, Guglielmone AA, et al. Avermectin resistance in Cooperia pectinata in cattle in Argentina. Vet Rec. 2001;149:58.
184 Familton AS, Mason P, Coles GC. Anthelmintic-resistant Cooperia species in cattle. Vet Rec. 2001;149:719.
185 Hoque MN, Begum N, Nooruddin M. Albendazole resistance in gastrointestinal nematode parasites of cattle in Bangladesh. Trop Anim Health Prod. 2003;35:219.
186 Mejia ME, Fernandez Igartua BM, Schmidt EE, Cabaret J. Multispecies and multiple anthelmintic resistance on cattle nematodes in a farm in Argentina: the beginning of high resistance? Vet Res. 2003;34:461.
187 Fashanu SO, Fagbemi BO. A preliminary investigation of resistance to anthelmintics in strongyles of cattle in Shaki, Nigeria. Afr J Biomed Res. 2003;6:111.
188 Stromberg BE, Averbeck GA. The role of parasite epidemiology in the management of grazing cattle. Int J Parasitol. 1999;29:33.
189 Vercruysse J, Hilderson H, Claerebout E, Roelants B. Control of gastrointestinal nematodes in first-season grazing calves by two strategic treatments with doramectin. Vet Parasitol. 1995;58:27.
190 Dorny P, Demeulenaere D, Smets K, Vercruysse J. Control of gastrointestinal nematodes in first season grazing calves by two strategic treatments with eprinomectin. Vet Parasitol. 2000;89:277.
191 Fisher MA, Jacobs DE, Hutchinson MJ, Simon AJ. Evaluation of doramectin in a programme for season-long control of parasitic gastroenteritis in calves. Vet Rec. 1995;137:281.
192 Stuedemann JA, Kaplan RM, Ciordia H, et al. Bermudagrass management in the Southern Piedmont USA. V: Gastrointestinal parasite control in cattle. Vet Parasitol. 2004;126:375.
193 Sangster NC, Dobson RJ. Anthelmintic resistance. In: Lee DL, editor. The biology of nematodes. New York: Taylor & Francis; 1992:531.
194 Radostits OM, Gay CC, et al. Veterinary medicine, ed 9, Philadelphia: Balliére Tindall; 2000:1340.
195 Uhlinger C, Fleming S, Moncol D. Survey for drug-resistant gastrointestinal nematodes in 13 commercial sheep flocks. J Am Vet Med Assoc. 1992;201:77.
196 Young KE, Jensen JM, et al. Verification of DrenchRite for use in North America by comparison with FERT (fecal egg reduction tests). Proc Am Assoc Vet Parasitol. 1997;42:65.
197 Mortensen LL, Williamson LH, Terrill TH, et al. Evaluation of prevalence and clinical implications of anthelmintic resistance in gastrointestinal nematodes of goats. J Am Vet Med Assoc. 2003;223:495.
198 Zajac AM, Gipson TA. Multiple anthelmintic resistance in a goat herd. Vet Parasitol. 2000;87:163.
199 Conway DP. Variance in the effectiveness of thiabendazole against Haemonchus contortus in sheep. Am J Vet Res. 1964;25:844.
200 Kaplan RM, Burke JM, et al. Total anthelmintic failure on a meat goat farm in Arkansas. Proceedings of the American Association of Veterinary Parasitologists 50th Annual Meeting, Minneapolis. 2005. July
201 Bowman DD. Georgi’s parasitology for veterinarians, ed 8. St Louis: Elsevier; 2003.
202 Urquhart GM, Armour JL, Duncan JL, Jennings FW. Veterinary parasitology, ed 2. Cambridge: Blackwell; 1996.
203 Morris DD. Diseases of the hemopoietic and hemolymphatic systems. In Smith BP, editor: Large animal internal medicine, ed 4, St Louis: Mosby, 2009.
204 Kassai T. Phylum: Nemathelminthes—roundworms. In: Veterinary helminthology. Oxford: Butterworth Heinemann; 1999:75.
205 Hazelby CA, Probert AJ, Rowlands DAT. Anthelmintic resistance in nematodes causing parasitic gastroenteritis of sheep in the UK. J Vet Pharmacol Ther. 1994;17:245.
206 Van Wyk JA. Refugia—overlooked as perhaps the most potent factor concerning the development of anthelmintic resistance. Onderstepoort J Vet Res. 2001;68:55.
207 van Wyk JA, Coles GC, Krecek RC. Can we slow the development of anthelmintic resistance? An electronic debate. Trends Parasitol. 2002;18:336.
208 Sréter T, Molnár V, Kassai T. The distribution of nematode egg counts and larval counts in grazing sheep and their implications for parasite control. Int J Parasitol. 1994;24:103.
209 Kaplan RM, Miller JE. FAMACHA Information Guide, Southern Consortium for Small Ruminant Parasite Control. 2004. Available at www.scsrpc.org
210 Barger IA. The statistical distribution of trichostrongylid nematodes in grazing lambs. Int J Parasitol. 1985;15:645.
211 Bath GF, Malan FS, et al. The FAMACHA Ovine Anemia Guide to assist with the control of haemonchosis. Proceedings of the 7th Annual Congress of the Livestock Health and Production Group of the South African Veterinary Association, Port Elizabeth, South Africa, June 5–7. 1996.
212 Van Wyk JA, Bath GF. The FAMACHA system for managing haemonchosis in sheep and goats by clinically identifying individual animals for treatment. Vet Res. 2002;33:509.
213 Kaplan RM, Burke JM, Terrill TH, et al. Validation of the FAMACHA eye color chart for detecting clinical anemia in sheep and goats on farms in the southern United States. Vet Parasitol. 2004;123:105.
214 Herd RP, Streitel RH, McClure KE, Parker CF. Control of periparturient rise in worm egg counts of lambing ewes. J Am Vet Med Assoc. 1983;182:375.
215 Courtney CH, Parker CF, McClure KE, Herd RP. A comparison of the periparturient rise in fecal egg counts of exotic and domestic ewes. Int J Parasitol. 1984;14:377.
216 Miller JE, Bahirathan M, Lemarie SL, et al. Epidemiology of gastrointestinal nematode parasitism in Suffolk and Gulf Coast native sheep with special emphasis on relative susceptibility to Haemonchus contortus infection. Vet Parasitol. 1998;74:55.
217 Hennessy DR. Physiology, pharmacology and parasitology. Int J Parasitol. 1997;27:145.
218 van Wyk JA, Bath GF. The FAMACHA system for managing haemonchosis in sheep and goats by clinically identifying individual animals for treatment. Vet Res. 2002;33:509.
219 Albers GA, Gray GD, Piper LR, et al. The genetics of resistance and resilience to Haemonchus contortus infection in young merino sheep. Int J Parasitol. 1987;17:1355.
220 Barger IA, Dash KM. Repeatability of ovine fecal egg counts and blood packed cell volumes in Haemonchus contortus infections. Int J Parasitol. 1987;17:977.
221 Barger IA. The role of epidemiological knowledge and grazing management for helminth control in small ruminants. Int J Parasitol. 1999;29:41.
222 Vlassoff A, Leathwick DM, Heath ACG. The epidemiology of nematode infections of sheep. N Z Vet J. 2001;49:213.
223 Coop RL, Kyriazakis I. Nutrition-parasite interaction. Vet Parasitol. 1999;84:187.
224 Coop RL, Kyriazakis I. Influence of host nutrition on the development and consequences of nematode parasitism in ruminants. Trends Parasitol. 2001;17:325.
225 Sykes AR, Coop RL. Interaction between nutrition and gastrointestinal parasitism in sheep. N Z Vet J. 2001;49:222.
226 Athanasiadou S, Kyriazakis I, Jackson F, Coop RL. Consequences of long-term feeding with condensed tannins on sheep parasitised with Trichostrongylus colubriformis. Int J Parasitol. 2000;30:1025.
227 Athanasiadou S, Kyriazakis I, Jackson F, Coop RL. Direct anthelmintic effects of condensed tannins towards different gastrointestinal nematodes of sheep: in vitro and in vivo studies. Vet Parasitol. 2001;99:205.
228 Niezen JH, Waghorn GC, Graham T, et al. The effect of diet fed to lambs on subsequent development of Trichostrongylus colubriformis larvae in vitro and on pasture. Vet Parasitol. 2002;105:269.
229 Burke JM, Miller JE, Olcott DD, et al. Effect of copper oxide wire particles dosage and feed supplement level on Haemonchus contortus infections in lambs. Vet Parasitol. 2004;123:235.
230 Waller PJ, Faedo M, Ellis K. The potential of nematophagous fungi to control the free-living stages of nematode parasites of sheep: towards the development of a fungal controlled release device. Vet Parasitol. 2001;102:299.
231 Knox MR, Faedo M. Biological control of field infections of nematode parasites of young sheep with Duddingtonia flagrans and effects of spore intake on efficacy. Vet Parasitol. 2001;101:155.
232 Terrill TH, Larsen M, Samples O, et al. Capability of the nematode-trapping fungus Duddingtonia flagrans to reduce infective larvae of gastrointestinal nematodes in goat feces in the southeastern United States: dose titration and does time interval studies. Vet Parasitol. 2004;120:285.
233 Kabagambe EK, Barras SR, Li Y, et al. Attempts to control haemonchosis in grazing ewes by vaccination with gut membrane proteins of the parasite. Vet Parasitol. 2000;92:15.
234 Knox DP, Redmond DL, Newlands GF, et al. The nature and prospects for gut membrane proteins as vaccine candidates for Haemonchus contortus and other ruminant trichostrongyloids. Int J Parasitol. 2003;33:1129.
235 Smith WD, van Wyk JA, van Strijp MF. Preliminary observations on the potential of gut membrane proteins of Haemonchus contortus as candidate vaccine antigens on naturally infected pasture. Vet Parasitol. 2001;98:285.
236 Gasbarre LC, Miller JE. Genetics of helminth resistance. In: Axford RFE, Bishop SC, Nicholas FW, Owen JB, editors. Breeding for disease resistance in farm animals. New York: CABI Publishing; 1999:129.
237 Stear MJ, Bishop SC, Mallard BA, Raadsma H. The sustainability, feasibility and desirability of breeding livestock for disease resistance. Res Vet Sci. 2001;71:1.
238 Bisset SA, Morris CA, McEwan JC, Vlassoff A. Breeding sheep in New Zealand that are less reliant on anthelmintics to maintain health and productivity. N Z Vet J. 2001;49:236.
239 Panuska C. Lungworms of ruminants. Vet Clin North Am Food Anim Pract. 2006;22:583.
240 Lyons ET, Hemken RD, Button FSJr. Overwintering of larvae of the cattle lungworm (Dictyocaulus viviparus) on pasture in Kentucky. J Am Vet Med Assoc. 1981;179:456.
241 Winters JB, Worley DE. Distribution and seasonal prevalence of bovine lungworm in selected areas in western Montana. Am J Vet Res. 1975;36:327.
242 Eddi CS, Williams JC, Swalley RA. Epidemiology of Dictyocaulus viviparus in Louisiana (U.S.A.). Vet Parasitol. 1989;31:37.
243 Rose JH. Lungworms of the domestic pig and sheep. Dawes B, editor. Advances in parasitology. London: Academic; 1973;vol 11:559.
244 Goldberg A. Experimental infection of sheep and goats with the nematode lungworm Dictyocaulus filaria. Am J Vet Res. 1952;13:531.
245 Ayalew L, Fréchette JL, Malo R, Beauregard C. Seasonal fluctuation and inhibited development of populations of Dictyocaulus filaria in ewes and lambs. Can J Comp Med. 1974;38:448.
246 Matthews JB. Parasitic airway disease. In: Lekeux P, editor. Equine respiratory diseases. Ithaca: International Veterinary Information Service, 2002.
247 Sauerländer R. Experimental infection of sheep and goats with Muellerius capillaris (Protostrongylidae, Nematoda). J Vet Med B Infect Dis Vet Public Health. 1988;35:525.
248 Berrag B, Cabaret J. Impaired pulmonary gas exchange in ewes naturally infected by small lungworms. Int J Parasitol. 1996;26:1397.
249 Berrag B, Cabaret J. Gastrointestinal and pulmonary nematode infections decrease goat productivity in Moroccan semi-arid conditions. J Helminthol. 1998;72:15.
250 Berrag B, Rhalem A, Sahibi H, et al. Bronchoalveolar cellular responses of goats following infections with Muellerius capillaris (Protostrongylidae, Nematoda). Vet Immunol Immunopathol. 1997;58:77.
251 Beresford-Jones WP. Observations on Muellerius capillaris (Müller, 1889) Cameron, 1927 III. Experimental infection of sheep. Res Vet Sci. 1967;8:272.
252 Wilson GI. The strength and duration of immunity to Dictyocaulus filaria infection in sheep and goats. Res Vet Sci. 1970;11:7.
253 Burks BS. Parasitic pneumonitis in horses. Compend Cont Educ (Pract Vet). 1998;20:378.
254 Rose JH. Some observations on the transmission of lungworm infection in a flock of sheep at pasture. Res Vet Sci. 1965;6:189.
255 Jacobs DE, Fisher MA, Hutchinson MJ, et al. An evaluation of abamectin given at turnout and six weeks after turnout for the control of nematode infections in calves. Vet Rec. 1995;136:386.
256 Eysker M, Boersema JH, Cornelissen JB, et al. Residual effect of injectable moxidectin against lungworm and gastrointestinal nematodes in calves exposed to high pasture infectivity levels in the Netherlands. Vet Parasitol. 1996;61:61.
257 Eysker M, Boersema JH, Kooyman FN, et al. Comparison between fenbendazole and moxidectin applied in a dose and move system for the control of Dictyocaulus viviparus infections in calves. Vet Parasitol. 1996;64:187.
258 Barth D, Ericsson GF, Kunkle BN, et al. Evaluation of the persistence of the effect of ivermectin and abamectin against gastrointestinal and pulmonary nematodes in cattle. Vet Rec. 1997;140:278.
259 Hertzberg H, Ochs H, Kohler L, et al. Prevention of gastrointestinal and lungworm infections in alpine calves: use of doramectin pour-on before and after the alpine grazing season. Schweiz Arch Tierheilkd. 1998;140:419.
260 Vercruysse J, Dorny P, Claerebout E, Weatherley A. Field evaluation of a topical doramectin formulation for the chemoprophylaxis of parasitic bronchitis in calves. Vet Parasitol. 1998;75:169.
261 Epe C, Woidtke S, Pape M, et al. Strategic control of gastrointestinal nematode and lungworm infections with eprinomectin at turnout and eight weeks later. Vet Rec. 1999;144:380.
262 Stromberg BE, Averbeck GA, Anderson JF, et al. Comparison of the persistent efficacy of the injectable and pour-on formulations of doramectin against artificially induced infection with Dictyocaulus viviparus in cattle. Vet Parasitol. 1999;87:45.
263 Taylor SM, Kenny J, Edgar HW, et al. Induction of protective immunity to Dictyocaulus viviparus in calves while under treatment with endectocides. Vet Parasitol. 2000;88:219.
264 Eysker M, Boersema JH, Cornelissen JB, Kooyman FN. Efficacy of Michel’s “dose and move” system against Dictyocaulus viviparus infections in cattle using moxidectin as anthelmintic. Vet Parasitol. 1995;58:49.
265 Taylor SM, Kenny J, Edgar HW, Whyte M. Protection against Dictyocaulus viviparus in second-year cattle after first-year treatment with doramectin or an ivermectin bolus. Vet Rec. 1997;141:593.
266 Grimshaw WT, Hong C, Webster R, Hunt KR. Development of immunity to lungworm in vaccinated calves treated with an ivermectin sustained-release bolus or an oxfendazole pulse-release bolus at turnout. Vet Parasitol. 1996;62:119.
267 Hertzberg H, Durgiai B, Schnieder T, et al. Prophylaxis of bovine trichostrongylidosis and dictyocaulosis in the alpine region: comparison of an early and late administration of the oxfendazole pulse release bolus to first-year grazing calves. Vet Parasitol. 1996;66:181.
268 Jacobs DE, Hutchinson MJ, Abbott EM. Evaluation of the effect of the fenbendazole sustained-release intraruminal device on the immunity of calves to lungworm. Vet Rec. 1996;139:60.
269 Borgsteede FH, van der Linden JN, Cornelissen JB, et al. Effect of three sustained-release devices on parasitic bronchitis in first-year calves. Vet Rec. 1998;142:696.
270 Rehbein S, Batty AF, Barth D, et al. Efficacy of an ivermectin controlled-release capsule against nematode and arthropod endoparasites in sheep. Vet Rec. 1998;142:331.
271 Kerboeuf D, Hoste H, Hubert J, Le Stang JP. Response of cattle treated with a fenbendazole slow release bolus to challenge from nematodes the following season. Vet Parasitol. 1996;62:107.
272 Schnieder T, Epe C, Von Samson-Himmelstjerna G, Kohlmetz C. The development of protective immunity against gastrointestinal nematode and lungworm infections after use of an ivermectin bolus in first-year grazing calves. Vet Parasitol. 1996;64:239.
273 McKeand JB. Vaccine development and diagnostics of Dictyocaulus viviparus. Parasitology. 2000;120:S17.
274 Mortensen LL, Williamson LH, et al. Evaluation of prevalence and clinical implications of anthelmintic resistance in gastrointestinal nematodes of goats. J Am Vet Med Assoc. 2003;23(4):495-500.
275 Coles G, et al. World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P.) methods for the detection of anthelmintic resistance in nematodes of veterinary importance. Vet Parasitol. 2003;23:35-44. 1992; 44(1–2)
276 Kaplan RM, Miller JE. FAMACHA Information Guide, Southern Consortium for Small Ruminant Parasite Control. 2004;23. www.scsrpc.org
277 Cameron, A: RESO fecal egg count reduction analysis spreadsheet. AusVet Animal Health Services, University of Sydney, Sydney, Australia 2000. (Based on calculations developed by Martin PJ, Wursthorn L, 1991. RESO faecal egg count reduction test calculator v4.0, CSIRO, Animal Health, Melbourne, Australia). Available at http://www.vetsci.usyd.edu.au/sheepwormcontrol/index.html under Site Map
278 Daugschies A, Majdrowski M. Eimeriosis in cattle: current understanding. J Vet Med B Infect Dis Vet Public Health. 2005;52:417.
279 Jolley WR, Bardsley KD. Ruminant coccidiosis. Vet Clin North Am Food Anim Pract. 2006;22:613.
280 Fanelli HH. Observations on nervous coccidiosis in calves. Bovine Pract. 1983;18:50.
281 Isler CM, Bellamy JE, Wobeser GA. Labile neurotoxin in serum of calves with “nervous” coccidiosis. Can J Vet Res. 1987;51:253.
282 Isler CM, Bellamy JE, Wobeser GA. Characteristics of the labile neurotoxin associated with nervous coccidiosis. Can J Vet Res. 1987;51:271.
283 Isler CM, Bellamy JE, Wobeser GA. Pathogenesis of neurological signs associated with bovine enteric coccidiosis: a prospective study and review. Can J Vet Res. 1987;51:261.
284 Ballweber LR. Veterinary parasitology. Woburn: Butterworth-Heinemann, 2001.
285 Blood DC, Radostits OM, Gay CC. Diseases caused by protozoa. In Veterinary medicine, ed 8, London: Baillière Tindall; 1994:1181.
286 Rebhun WC. Infectious diseases of the gastrointestinal tract. In: Diseases of dairy cattle. Baltimore: Williams & Wilkins; 1995:177.
287 Clarkson MJ, Winter AC. Thin scouring lambs. In A handbook for the sheep clinician, ed 5, Liverpool: Liverpool University Press; 1997:107.
288 Quigley JD3rd, Drewry JJ, Murray LM, Ivey SJ. Effects of lasalocid in milk replacer or calf starter on health and performance of calves challenged with Eimeria species. J Dairy Sci. 1997;80:2972.
289 McAllister TA, Annett CB, Olson ME, et al. Effect of salinomycin on giardiasis and coccidiosis in growing lambs. J Anim Sci. 1996;74:2896.
290 Alzieu JP, Mage C, Maes L, de Mûelenaere C. Economic benefits of prophylaxis with diclazuril against subclinical coccidiosis in lambs reared indoors. Vet Rec. 1999;144:442.
291 Heath HL, Blagburn BL, Elsasser TH, et al. Hormonal modulation of the physiologic responses of calves infected with Eimeria bovis. Am J Vet Res. 1997;58:891.
292 Cox FE. Control of coccidiosis: lessons from other sporozoa. Int J Parasitol. 1998;28:165.
293 Svensson C, Olofsson H, Uggla A. Immunization of calves against Eimeria alabamensis coccidiosis. Appl Parasitol. 1996;37:209.
294 Svensson C. The survival and transmission of oocysts of Eimeria alabamensis in hay. Vet Parasitol. 1997;69:211.
Little D, Flowers JR, Hammerberg BH, et al. Management of drug-resistant cyathostominosis on a breeding farm in central North Carolina. Equine Vet J. 2003;35:246.
Love S. Treatment and prevention of intestinal parasite-associated disease. Vet Clin North Am Equine Pract. 2003;19:791.
Waller PJ. Control strategies to prevent resistance. Vet Parasitol. 1993;46:133.
Coles GC. Sustainable use of anthelmintics in grazing animals. Vet Rec. 2002;151:165.
Fleming SA, Craig T, Kaplan RM, et al. ACVIM consensus statement on anthelmintic resistance in gastrointestinal parasites in small ruminants. J Vet Intern Med. 2006;20:435.
Southern Consortium for Small Ruminant Parasite Control. Available at: www.scsrpc.org
Southern Consortium for Small Ruminant Parasite Control. Available at: www.scsrpc.org
Coles GC, Jackson F, Pomroy WE, et al. The detection of anthelmintic resistance in nematodes of veterinary importance. Vet Parasitol. 2006;136:167.
Fleming SA, Craig T, Kaplan RM, et al. ACVIM consensus statement on anthelmintic resistance in gastrointestinal parasites in small ruminants. J Vet Intern Med. 2006;20:435.
Kassai T. Antihelmintic therapy and control. In: Veterinary helminthology. Oxford: Butterworth Heinemann; 1999:147.
Plumb DC. Veterinary drug handbook, ed 4. Iowa: Iowa State University Press, 2005.