Chapter 26 Vitamins and minerals
Vitamins
Introduction
To some extent this chapter is distinct from the rest of the book in that vitamins are not, in most cases, really drugs and as a group their chemistry is diverse. Thus they do not have common modes of biological action and there is no clear progression in their development as therapeutic agents. However, vitamin and mineral supplements, after pain killers and skin treatments, are the third highest selling over-the-counter medicines. For certain vitamins, e.g. vitamin C and vitamin E, there is an interest in the efficacy in disease prevention. The chemistry and biochemistry of vitamins, in its own right, is very interesting and has some bearing on the actions of a number of drugs.
The word vitamin stems from the term ‘vital amine’ which arose from the discovery of one of the first vitamins, the amine thiamine (vitamin B1). Thiamine is an amine, which is not true of all vitamins, but the name vitamin was generally adopted. A vitamin can be defined as an organic compound present in the diet in small amounts which is essential for the normal physiological functioning of the organism but which cannot be synthesised by the organism. Absence of the vitamin leads to clear symptoms of deficiency. A balanced diet should provide enough vitamins but there is some support for the view that certain vitamins can be used in amounts larger than the recommended daily amount (RDA) as therapeutic agents or for prophylaxis, e.g. large doses of vitamin C for treating colds or vitamin D for prevention of osteoporosis.
Since a balanced diet should provide for most vitamin requirements, it is debatable whether vitamin supplements are required other than in situations where there is likely to be deficiency such as vitamin B12 to supplement a vegan diet or folate during pregnancy. However, there are indications that in some cases vitamins can be used as medicine. The other issue that arises in these cases is whether or not large doses of vitamins are toxic, and in some cases this is the case. It is highly unlikely that moderate supplementation of the diet with multivitamins will do any harm and may be quite beneficial, certainly in the case of antioxidant vitamins such as C and E. But, as with any medicine, it would be preferable not to take large doses for long periods. Treatment of an acute condition, such as the use of large doses of vitamin C to treat a cold after its onset, for a short period, is the best use of these supplements.
Drug treatment can cause depletion of certain vitamins and minerals and Table 26.1 summarises the effects of various drugs on normal levels of nutrients. Thus vitamin supplementation may be recommended when someone is taking a vitamin-depleting drug for a long period.
Table 26.1 Effects of drugs on vitamins and minerals in the body
| Drug | Effects |
|---|---|
| Antacids (aluminium hydroxide, sodium bicarbonate) | Reduces calcium, copper and folic acid absorption |
| Gentamicin | Interferes with potassium and magnesium function |
| Tetracycline | Binds to calcium |
| Phenobarbital, phenytoin, primidone | Affects vitamin D and vitamin K function |
| Aspirin | Folate, iron and vitamin C function |
| Corticosteroids | Affects calcium function. |
| Colchicine | Vitamin B12 absorption |
| ACE inhibitors | Bind zinc and this may be responsible for affecting the sense of smell and taste |
| Furosemide | Depletes calcium, magnesium and potassium |
| Cimetidine and ranitidine | Affect vitamin B12 absorption |
| Chlorpromazine | Affects riboflavin absorption |
| Alcohol | Depletes vitamin C and thiamine |
Vitamin A
Dietary sources (% RDA per 100 g in brackets)
There are relatively few rich dietary sources of vitamin A and it can be obtained mainly from green and yellow vegetables and liver. Sources include: beef liver (4000%), red peppers (3000%), carrots (1500%), butter (500%), eggs (300%), pumpkin (200%), peach (150%) and beans (25%).
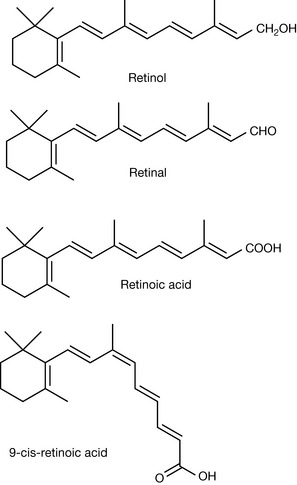
It is absorbed directly in the form of retinol esters from liver, butter and eggs and the esters are converted to free retinol by esterases in the body. Plant foodstuffs such as carrots and spinach contain vitamin A in the form of carotene (provitamin A) and this is converted in the body to retinol via the action of beta carotene dioxygenase which produces two molecules of retinal from each molecule of beta carotene. It exists in a number of different forms (vitamers; Fig. 26.1).
Chemistry and biological functions
Role in the visual process
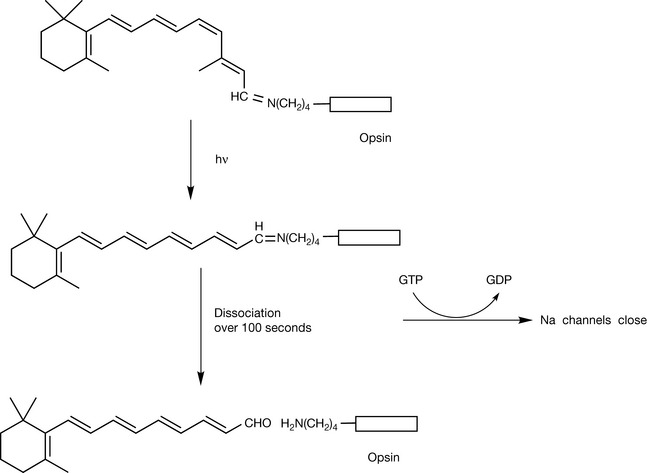
Vitamin A is crucial for the functioning of the visual process. It is bound to the visual protein rhodopsin and its interconversion between its cis and trans forms triggers the visual process (Fig. 26.2). Vitamin A in the form of retinal forms a Schiff’s base with a lysine residue in opsin, the visual pigment protein, and at the same time undergoes isomerisation from the all trans form to yield a cis configuration at the 11 position (Fig. 26.2). The vitamin A protein complex absorbs light in the 400–600 nm region and this causes it to revert to the all trans form; thus the light energy is converted to molecular motion. The all trans retinal protein complex is unstable and slowly dissociates.
Retinoids
Dietary vitamin A gives rise to a number of important biologically active metabolites known as the retinoids. The most important forms of these are the all trans form and the 9-cis form of retinoic acid (Fig. 26.1). Retinoids have a wide range of biological activities and have effects on embryonic development, spermatogenesis, regulation of immune function, bone metabolism and differentiation of epithelia. They mediate these effects via specific retinoid receptors which are similar to the nuclear steroid binding receptors in that they directly affect DNA transcription. Since retinoids have been found to exert effects on cell differentiation they have been tested as anticancer drugs along with synthetic analogues. Retinoids show some promise in a number of types of cancer but their use is limited by high levels of toxicity. Vitamin A has also been used in the treatment of skin diseases such as acne.
Symptoms of deficiency
Vitamin A is stored effectively by the body, particularly the liver, because of its high degree of lipophilicity. This means that symptoms of deficiency are slow to manifest. The only unequivocal signs of deficiency are xerophthalmia (drying of the conjunctiva and the eyeball) and nyctalopia (night blindness). Other symptoms are very generalised, including drying of the skin and mucous membranes. In some parts of the world, particularly India and Africa, vitamin A deficiency does occur and it is estimated that 250 000 to 500 000 children a year go blind as a result of vitamin A deficiency caused by malnourishment.
Toxicity
Vitamin A deficiency is unlikely where a balanced diet is consumed. There is probably greater risk from toxicity due to taking too much vitamin A. Persistent large excesses of vitamin A, >1000 × RDA, have to be taken for toxicity to manifest, although the threshold for toxicity is given as ca. 4 × RDA. This is the narrowest margin between RDA and toxicity for any vitamin. Symptoms of toxicity include: the skin flaking, alopecia, conjunctivitis, nausea, dizziness, loss of muscular coordination and brittle bones. Extreme intoxication produces liver failure and death.
Vitamin B1 (thiamine)
Dietary sources (% RDA per 100 g in brackets)
Most foods contain a low concentration of thiamine. The most important dietary sources are: Brewers yeast (1500%), pork (120%), whole wheat and other whole grains (70%) pork liver (60%), oatmeal (70%), peas (30%), asparagus (25%) and salmon (25%) (Fig. 26.3).
Biological functions
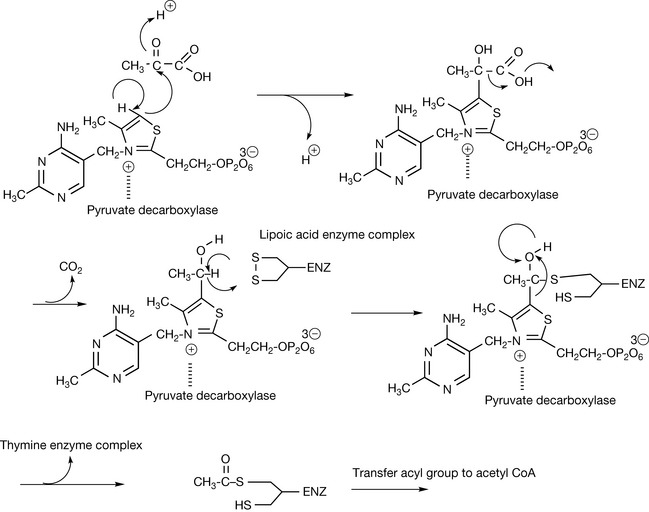
Vitamin B1 gave rise to the science of nutrition when it was discovered that populations consuming a diet of polished rice where the outer husk of the rice had been removed suffered from the disease beriberi, which could be reversed when diet was improved. Vitamin B1 was eventually isolated from yeast extract in 1932 and characterised. Like the rest of the B vitamins, it is very water soluble; thus excess thiamine is not retained to any great extent by the body although the thiamine required for physiological function is retained by binding to proteins. It has an essential role in metabolism of glucose, catalysing the decarboxylation of pyruvic (Fig. 26.4) and α-ketoglutaric acid plus a number of other substrates. Following decarboxylation of pyruvate the remaining acetate group is transferred to lipoic acid and then to acetyl CoA. Thiamine may have a role in neurotransmission although the biological nature of that role is not entirely clear. It has been found in synaptosomal membranes and nerve stimulation has been found to result in a release of thiamine.
Symptoms of deficiency
Symptoms of deficiency include: anorexia, cardiac enlargement, muscular weakness (symptoms of beriberi) and neurodegeneration. Excessive alcohol consumption appears to have an effect on thiamine status, contributing to its damaging effects, and thiamine deficiency resulting from alcohol abuse can give rise to psychosis. Since thiamine is very water soluble, diuretics may also produce thiamine deficiency, and haemodialysis may produce deficiency. Some foodstuffs including tea, coffee and raw fish contain factors that antagonise the activity of thiamine.
Possible therapeutic indications
It has been observed that high thiamine intake is associated with a lower likelihood of developing cataract. Trials assessing the benefit of thiamine supplementation in arresting the progression of Alzheimer’s disease indicated that there was no benefit. There was no strong evidence for thiamine being beneficial in treating heart disease or cancer. There have been reports that thiamine can increase mental acuity. Thiamine supplementation at 25–50 mg per day has been reported to be effective in reducing mosquito bites, although 2 weeks of supplementation are required to produce an effect. Another study found thiamine to be ineffective as an insect repellent.
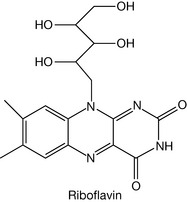
Vitamin B2 (riboflavin)
Dietary sources (% RDA per 100 g in brackets)
Meats, dairy products and green vegetables are the most important sources. Bioavailability is greater from animal products. Good sources include: beef liver (300%), cheese (35%), eggs (25%), pork (25%), lamb (25%) broccoli (20%) and asparagus (20%) (Fig. 26.5).
Biological function
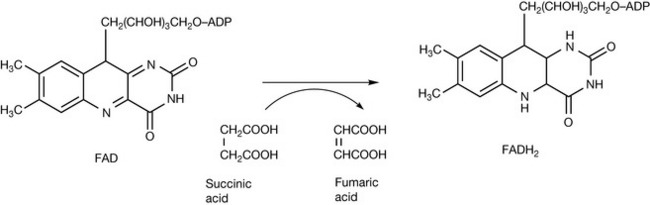
Riboflavin is another highly water-soluble vitamin and was isolated from yeast extract in 1933. It has a bright yellow colour and excess vitamin is rapidly excreted in the urine, turning it bright yellow. Riboflavin is incorporated into flavin adenine dinucleotide (FAD) which is an essential co-factor of flavoproteins, which are involved in biological oxidation and reduction and are required for the metabolism of carbohydrates, amino acids and lipids. Figure 26.6 shows the oxidation of succinic acid to fumaric acid which uses FAD as a co-factor. The FADH2 formed then enters the terminal respiratory chain where eventually the 2H atoms are converted into H2O.
Symptoms of deficiency
Many tissues are affected by riboflavin deficiency (ariboflavinosis). Riboflavin deficiency requires 3–4 months of deprivation to manifest and symptoms include lesions on the lips, inflammation of the tongue, lowered levels of white and red blood cells, excessive sensitivity to pain and vascularisation of the cornea. Deficiency also results in decreased conversion of tryptophan into niacin. A test for deficiency is to measure glutathione reductase levels, which are depressed when riboflavin levels are low as are xanthine oxidase levels. There is a link between riboflavin deficiency in pregnancy and the development of pre-eclampsia. However, supplementation with riboflavin in a trial indicated that there was no effect on the prevention of pre-eclampsia. Alcoholics are at increased risk of riboflavin deficiency as are anorexics and lactose intolerant subjects who may not consume dairy products, which are good sources of riboflavin. Physically active people such as athletes may have an extra requirement for riboflavin.
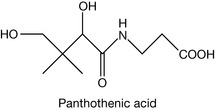
Vitamin B5 (pantothenic acid)
Dietary sources (% RDA per 100 g in brackets)
The most important dietary sources are meats and some vegetables: pork liver (150%), kidney (80%), eggs (60%), peanuts (60%), wheat bran (60%), mushroom (40%), lentils (30%), broccoli (25%) and avocado (25%) (Fig. 26.7).
Biological functions
Vitamin B5 is a component of co-enzyme A (CoA) which is required for the biosynthesis of fatty acids and lipids; the synthesis of cholesterol and metabolism involving acetylation.
Symptoms of deficiency
Deficiency is only observed in severe malnourishment and would be difficult to distinguish from the general debility produced by malnutrition.
Therapeutic indications
Administration of pantothenic acid orally and application of pantothenol ointment to the skin have been shown to promote wound healing in animals. However, few data exist in humans to support these findings. A pantothenic acid derivative called pantethine has been reported to have a cholesterol lowering effect.
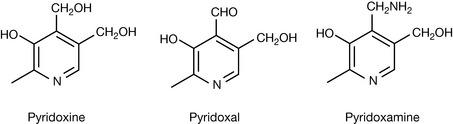
Vitamin B6
Dietary sources (% RDA per 100 g in brackets)
Vitamin B6 is widely distributed in foodstuffs, occurring mainly in meats and whole grains. Pyridoxine is more bioavailable from meat. Sources include: liver (80%), walnuts (70%), chicken (30–60%), tuna (40%), beef (30%), spinach (25%), whole wheat (25%), potatoes (20%), cauliflower (20%) and eggs (15%) (Fig. 26.8).
Biological functions
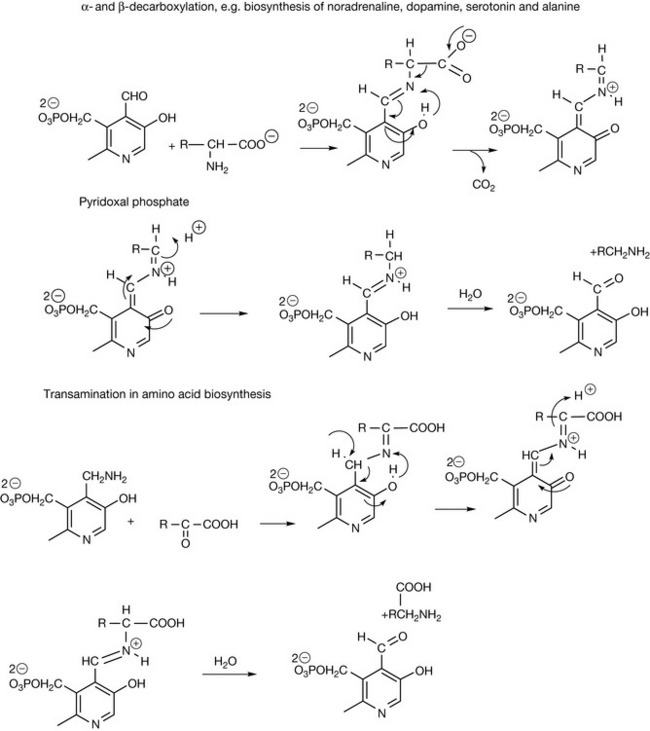
Pyridoxine is the most key of all co-enzymes. It is involved in decarboxylation reactions which are required in the synthesis of neurotransmitters such as serotonin, noradrenaline, dopamine and γ-aminobutyric acid (GABA) from amino acids. It is involved in transamination reactions which are required for synthesis of amino acids from ketoacids and in elimination where, for instance, an amino group is removed from an amino acid. It is also involved in racemisation where the stereochemistry at a chiral centre is reversed. Figure 26.9 shows reaction mechanisms for decarboxylation with vitamin B6 as the co-factor. The vitamin is activated by conversion to its phosphate ester. It has been said that nature created pyridoxine for the delight of organic chemists who enjoy pushing electrons around. This is amply illustrated in Figure 26.9 and these are only two of pyridoxine’s reactions! Various explanations exist as to how it exerts its co-enzyme activity, and some mechanisms protonate the pyridine nitrogen. This is unlikely at physiological pH since its pKa value is <5.0. The phenolic group is quite acidic and the most stable intermediates are as shown in Figure 26.9, where a linear conjugated system is formed yielding a reactive ortho quinone methide intermediate. The ortho quinone methide of pyridoxine has been observed to be formed in aqueous solution by photochemical reaction. This possible reaction might give some concern with regard to liver toxicity if large doses of pyridoxine are taken as a vitamin supplement.
Pyridoxine is involved as a co-factor coenzyme in about 100 enzyme systems. Thus, in addition to the reactions mentioned above, it is required for: glycogen phosphorylase, which catalyses the release of glucose from stored glycogen, haemoglobin biosynthesis, the generation of glucose from amino acids (gluconeogenesis), the biosynthesis of niacin from tryptophan and nucleic acid biosynthesis.
Symptoms of deficiency
Severe deficiency of pyridoxine is uncommon. Alcoholics are thought to be most at risk of vitamin B6 deficiency due to low dietary intake and impaired metabolism of the vitamin. General symptoms of deficiency include sleeplessness, nervous disorders, dermatological symptoms, depression and anaemia. Abnormal electroencephalogram patterns have been noted in some studies of vitamin B6 deficiency.
Possible therapeutic indications
Pyridoxine has been proposed a treatment for elevated levels of homocysteine, which is believed to be involved in the aetiology of coronary artery disease. There is some evidence that it promotes turnover of homocysteine. There is evidence that it can improve immune function in the elderly when administered at levels about 2–3 × RDA. There is some evidence that pyridoxine supplementation may improve cognitive function in the elderly although not mood. There is some evidence that pyridoxine may reduce the risk of kidney stone formation. Pyridoxine has been used for many years in the treatment of morning sickness and there is some evidence that it is effective when used at levels of 25 mg every 8 hours. It has been used in treating premenstrual syndrome and there is some evidence that it is effective. It is recommended that pyridoxine supplementation is used with drugs such as isoniazid and penicillamine that deplete its levels. Large doses (0.1–1 g) have been used in the treatment of schizophrenia and it has been found that pyridoxine can interfere with the actions of LDOPA and phenytoin.
Toxicity
Pyridoxine is not particularly toxic, and even where massive doses (>2 g/day) were taken there was merely some evidence of lack of muscular coordination (ataxia) and loss of motor control (muscle spasm/twitch). There is some evidence that pyridoxine can cause sensory neuropathy and for this reason it is recommended that it is not taken in doses >100 mg per day.
Vitamin B12 (cyanocobalamin)
Dietary sources (% RDA per 100 g in brackets)
Vitamin B12 is synthesised by bacteria in the guts of animals; thus animal tissues are the main source. Dietary sources include: beef liver (8000%), beef kidney (2500%), trout (500%), herring (300%), eggs (80%), cheese (50–100%), chicken (30%) and milk (30%). Grains and fruits do not contain vitamin B12.
Biological functions
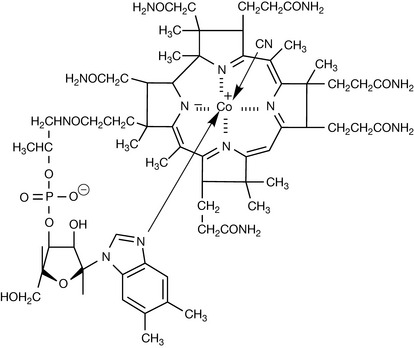
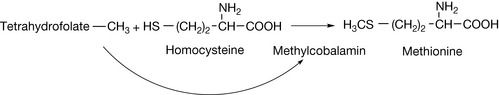
There are a number of closely related forms of vitamin B12 of which cyanocobalamin is one (Fig. 26.10). Cyanocobalamin is the synthetic form of the vitamin which is given in supplements; however, in the body the active form of the vitamin either has a methyl group in place of the CN ion (methylcobalamin) or deoxyadenosine (5′-deoxyadenosylcobalamin). Vitamin B12 is essential for the correct functioning of the brain and nervous system and for haemoglobin synthesis. Some of the effects of B12 deficiency can be removed by supplementation with folic acid since vitamin B12 deficiency results in loss of folate due to it accumulating as methyl tetrahydrofolate. Methylcobalamin is required for the function of the folate-dependent enzyme, methionine synthase, which is required for the synthesis of methionine, from homocysteine (Fig. 26.11). Methionine in turn is required for the synthesis of S-adenosylmethionine, a methyl group donor used in many biological methylation reactions including methylation of purines and pyrimidines.
Reduced levels of methionine synthase can lead to an accumulation of homocysteine which has been linked to cardiovascular disease.
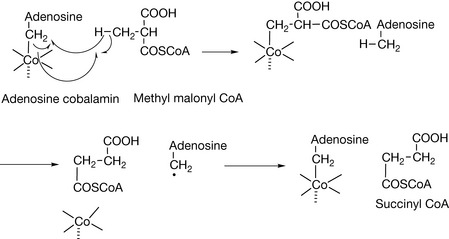
Vitamin B12 is also required by the enzyme that catalyses the conversion of L-methylmalonyl-CoA to succinyl-CoA. This biochemical reaction plays an important role in the production of energy from fats and proteins, particularly the metabolism of branch chain amino acids such as leucine. This reaction requires the deoxyadenosine form of B12. The reaction is shown in Figure 26.12, where the adenosine cobalamin allows a rearrangement of the methylmalonyl CoA to succinyl CoA. Succinyl CoA is also required for the synthesis of haemoglobin. Vitamin B12 deficiency is characterised by elevated levels of methylmalonyl CoA in blood, although this is not a definitive test for it.
Symptoms of deficiency
Since B12 only occurs in meat, deficiencies are likely to result from a strict vegetarian diet containing no animal products. Vitamin B12 deficiency is also estimated to affect 10–15% of individuals over the age of 60. Vitamin B12 is absorbed from the small intestine as a complex with a protein called intrinsic factor (IF). The most common cause of vitamin B12 deficiency is pernicious anaemia which is an autoimmune disease where the cells of the stomach become inflamed and do not secrete the required amounts of acid and enzymes to release vitamin B12 from food. In addition, antibodies to IF further prevent B12 absorption. The condition is treated with high doses of vitamin B12 supplements or by intramuscular injection of vitamin B12. A similar condition occurs in the elderly where there is malabsorption of vitamin B12 from food due to decreased secretion of stomach acid. This condition is easier to treat with supplementation since IF levels are still normal. Symptoms include megaloblastic anaemia, neuropathy, memory loss and abnormalities of lipid metabolism.
Therapeutic indications
With folic acid vitamin B12 has been shown to reduce levels of the cardiac risk factor homocysteine in plasma. It is not clear if a reduction homocysteine does lower the risk of heart disease. There is some indication that vitamin B12 can decrease the risk of cancer but it is difficult to differentiate its effects from the effects of folate metabolism. It is important that vitamin B12 intake is adequate during pregnancy to reduce the risk of the neural tube defects developing in the unborn child. Thus vitamin B12 is an important supplement for vegans and the infants of vegan mothers where they are breast fed. Extensive research has been conducted into whether or not vitamin B12 supplementation can prevent the development of Alzheimer’s disease; however, the trials thus far are inconclusive.
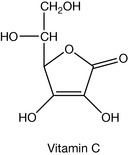
Vitamin C (ascorbic acid)
Dietary sources (% RDA per 100 g in brackets)
Vitamin C is lost on storage and lost to a great extent in cooking where it dissolves in the cooking water; thus it is provided most effectively by raw fruits and vegetables and their juices. Sources include: rosehips (2500%), green pepper (250%), blackcurrant (250%), broccoli (200%), Brussels sprouts (200%), watercress (150%), strawberry (120%), oranges/lemons (100%), apple (15%).
Biological functions
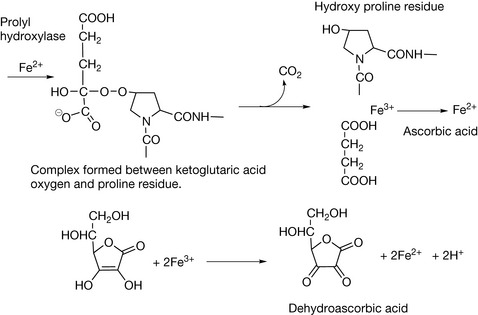
Vitamin C (Fig. 26.13) deserves a book in itself since so much research has been conducted into it over the years. Most living organisms produce vitamin C and thus do not require it as a vitamin; however, primates, guinea pigs, bats and birds require vitamin C as a vitamin. It has many biological functions. It is essential for the biosynthesis of the protein of connective tissue, collagen, since it is a co-factor for the hydroxylation of proline residues of procollagen in order to form the connective tissue protein collagen (Fig. 26.14). It is not possible to write a completely satisfactory mechanism for this reaction but it is known that Fe2+, ketoglutaric acid and molecular oxygen are involved in the initial reaction which is catalysed by proline hydroxylase. Ketoglutaric acid is decarboxylated to succinic acid and this results at the same time in the hydroxylation of proline and the oxidation of Fe2+ to Fe3+. The evidence seems to point to a role for ascorbic acid after the event in reducing Fe3+ back to Fe2+. Figure 26.14 shows the equation for the reduction of Fe3+ to Fe2+ but even this is a simplification. The hydroxylation of procollagen to collagen results in the H-bonding of the collagen chains to each other, thus producing a material which on a weight-for-weight ratio is stronger than steel. Vitamin C is involved in a series of reactions which require hydroxylation including: the biosynthesis of noradrenaline, serotonin and carnitine, the metabolism of cholesterol to bile acids and oxidative metabolism of many drugs.
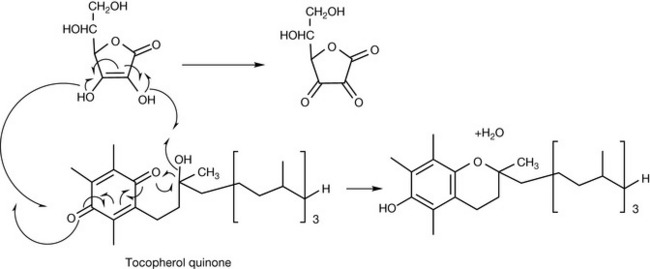
Other roles of vitamin C include: the oxidative degradation of the amino acid tyrosine, enhancing the absorption of iron from foods, antihistamine effects through increasing histamine degradation rates, immunostimulating effects through increasing neutrophil activation, stimulating IgG and IgM formation and protecting essential proteins against damage by neutrophils. A very important biological role for vitamin C is in the reduction of the oxidised form of vitamin E so that vitamin E can preserve its role in protection of biological membranes from oxidation (Fig. 26.15).
Symptoms of deficiency
The most obvious symptom of vitamin C deficiency is scurvy, which is due to defects in collagen formation causing impaired wound healing, haemorrhage and weakening of the gums and cartilage. Mild vitamin C deficiency manifests as fatigue and anorexia, and may be associated with hypercholesterolaemia and increased risk of heart disease. Smoking and alcohol consumption reduce vitamin C levels in plasma.
Possible therapeutic indications
Vitamin C is the most widely studied vitamin in terms of therapeutic activity. Despite the fact that doses of vitamin C of 100–150 mg/day result in tissue saturation, higher intakes can result in increases in concentrations in extracellular fluids such as plasma with associated pharmacologic activity. The main problem with most evidence for vitamin C efficacy in disease prevention is that many studies report positive results based on dietary consumption of vitamin C where other compounds absorbed from fresh fruit and vegetables may also have a preventative role.
Extensive studies have been made of the role of vitamin C in preventing heart disease. A very large pooling of data indicated that when vitamin C was taken as a supplement at >700 mg/day there was a 25% reduction in the chance of getting coronary heart disease (CHD). Treatment with high doses of vitamin has consistently proved that vitamin C enhances blood vessel dilation in patients with atherosclerosis.
The role of vitamin C in the prevention and treatment of cancer has been studied for many years. Very early work was conducted by Linus Pauling at Vale of Leven hospital in Scotland in which positive claims that were made could never be reproduced. The theory behind cancer prevention is that high levels of the vitamin enhance collagen biosynthesis which helps to contain tumours; however, there is no reason to suppose the high levels of vitamin C lead to higher levels of collagen synthesis. There is some evidence for the prevention of cancer by vitamin C, particularly in the case of stomach cancer.
The role of vitamin C in prevention and treatment of the common cold has been extensively studied. In this case, doses of 1–5 g per day may be taken. Controlled studies appear to indicate only small effects although a study involving people at peak fitness including athletes and skiers found that vitamin C supplementation reduced the incidence of colds by 50%. Also, a long-term trial over 5 years in Japan found that supplementation at 500 mg daily reduced the incidence of colds by 66%. The means of administration may be critical, and localised administration via lozenges and hence buccal absorption may be most effective.
Toxicity
The toxicity of vitamin C is very low and doses of 10 g/day appear to be quite safe. The major concern at high doses is the risk of the formation of calcium oxalate kidney stones and it might be wise not to take calcium supplements with vitamin C. The increase of oxalate levels, even at high doses, only increases by about 50%. The evidence for rebound effects where, following withdrawal of high-dose therapy, vitamin C levels fall and for in vivo mutagenicity is not conclusive. The worst effects of vitamin C at high dose appear to be GI disturbances and diarrhoea.
Vitamin D (ergocalciferol)
Dietary sources (% RDA per 100 g in brackets)
Vitamin D is found largely in fish and to a lesser extent in other animals and very little in plants. Often, foods contain added vitamin D. Sources include: cod liver oil (250%), sardines (40%), herring (10%), salmon (10%), chicken (2%), butter (1%) and spinach (0.01%) (Fig. 26.16).
Biological functions
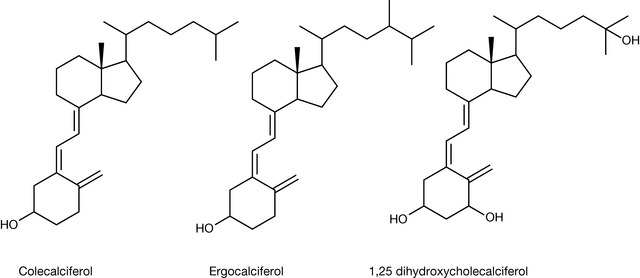
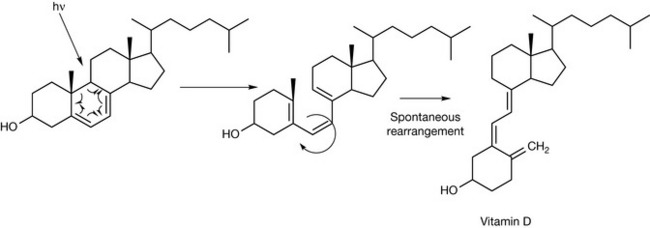
Vitamin D is not strictly a vitamin because it can be made by the body if sufficient sunlight is available. If sunlight is of only low intensity, vitamin D deficiency can occur. The action of sunlight is required to convert 7-dehydrocholesterol into vitamin D via a photochemical reaction (Fig. 26.17). Thus, for people in far northern or far southern latitudes, absorption of vitamin D from the diet is necessary.
Vitamin D is composed of a number of related structures but the two most prominent members of the family are colecalciferol and ergocalciferol, which is formed from ergosterol rather than 7-dehydrocholesterol. Colecalciferol is transformed into the most active form of the vitamin, 1,25-dihydroxycholecalciferol, by hydroxylation at the 25 position in the liver and finally at the 1 position in the kidneys.
1,25-Dihydroxycholecalciferol, together with the peptide hormones calcitonin and parathyroid hormone, functions to regulate calcium and phosphate homeostasis. Vitamin D acts like a steroid hormone binding to receptor proteins and then to DNA, triggering the transcription of calcium-binding proteins. These binding proteins are involved in uptake of Ca2+ from the intestine, its resorption by the kidney and its binding by bone. These proteins are also involved in phosphate absorption from the intestine.
Symptoms of deficiency
In the absence of the formation of vitamin D via the action of sunlight, it has to be absorbed from restricted dietary sources; thus deficiency can occur. Even with mild vitamin D deficiency, loss of calcium from the bone can occur to compensate for reduced calcium absorption from the diet. This increases the risk of osteoporosis. Severe vitamin D deficiency in children results in rickets, where the bones of the growing child become bowed. Despite addition of vitamin D to foodstuffs, rickets is still reported from cities around the world. Vitamin D deficiency in adults results in a proneness to fractures in adults (osteomalacia) and also manifests as muscle weakness. There are a number of risk factors for vitamin D deficiency: infants who are exclusively breast fed and are not exposed to sunlight, people with dark skin, the elderly, people with Crohn’s disease and obesity.
Therapeutic indications
There is some evidence that vitamin D in the form of colecalciferol taken at a level >800 IU is effective in reducing the risk of bone fractures in the elderly when taken with calcium supplementation at >1 g per day. Vitamin D can promote cell differentiation and there is some evidence from epidemiological studies that vitamin D is effective in reducing the incidence of some cancers, including gastrointestinal, prostate and breast cancer. Vitamin D has been shown to modulate T-cell response and thus may have an effect on autoimmune diseases, and there is some evidence to support this.
25-(OH)-vitamin D has shown some potential for the treatment of psoriasis.
Toxicity
At high levels (>100 × RDA) vitamin D causes hypercalcaemia, which can result in deposition of calcium phosphate in soft tissues such as heart and kidney. Certain patients are at higher risk of suffering from hypercalcaemia, including those suffering from hyperparathyroidism, sarcoidosis, tuberculosis and lymphoma. Although vitamin D is potentially toxic it can be regarded as being safe at levels <500 μg per day. Certain drugs including phenytoin, carbemazepam and ketoconazole can reduce vitamin D levels. Patients taking digitalis are more sensitive to the effects of hypercalcaemia.
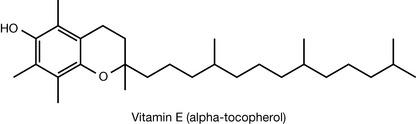
Vitamin E (tocopherols)
Dietary sources (% RDA per 100 g in brackets)
The main dietary sources are vegetable oils and, to a lesser extent, grains and nuts.
Sources include: wheat germ oil (1000%), peanut oil (100%), olive oil (80%), coconut oil (10%), maize (10%), whole wheat (10%) and oats (8%) (Fig. 26.18).
Biological functions
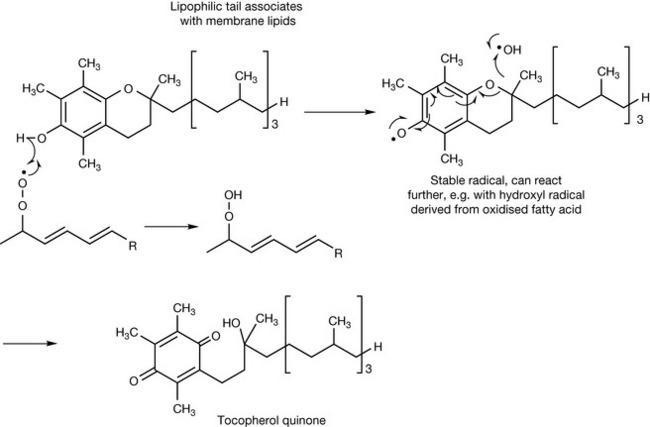
Vitamin E exists as eight different closely related structures, four tocopherols and four tocophertrienols. Alpha-tocopherol is regarded as being the most important in humans. Whereas vitamin C functions as an antioxidant in an aqueous environment, vitamin E acts to protect lipophilic structures such as the phospholipids which make up cell membranes from oxidation. Fatty acids containing double bonds are prone to damage via peroxidation. Figure 26.19 shows the action of vitamin E in terminating free radical-generated peroxidation of an unsaturated fatty acid. If the radical were not quenched, it would go on to initiate a chain reaction, damaging the membrane. The radical formed by vitamin E is quite stable and unreactive but can go on to react with a second radical such as hydroxyl (the most reactive of all radicals) to form tocopherol quinone. Vitamin E can be regenerated from tocopherol quinone by vitamin C as shown in Figure 26.15 or by another reducing compound such as glutathione. Many reactive oxygen species (ROS) are generated in the body and vitamin E plays an important role in protecting the body against them. A number of other roles have been identified for vitamin E including immune modulation and inhibition of platelet aggregation.
Symptoms of deficiency
Since vitamin E is lipophilic and thus efficiently stored by the body, acute deficiency is rarely observed. In conditions where there is malabsorption of fat, vitamin E deficiency may be observed. Deficiency of vitamin E is associated with loss of membrane function and this may be associated with: impaired balance, peripheral neuropathy, muscle weakness and damage to the retina.
Possible therapeutic indications
Several large studies have shown that adequate vitamin E intake or supplementation may reduce the risk of developing heart disease. However, there was no evidence that the vitamin reduced the likelihood of those with heart disease dying. There is some evidence that vitamin E can boost immune response in the elderly. There is no strong link between vitamin E supplementation and reduction of breast or lung cancer; however, there is strong evidence that vitamin E can reduce the incidence of prostate cancer. There is some evidence for effectiveness in treatment of diabetes and in reducing risk of developing dementia in the elderly. A number of studies have examined the effects of vitamin E on Parkinson’s disease and there is evidence that it can play a preventative role. Since vitamin E may be oxidised to a quinine, which can act as an antagonist of vitamin K, in a manner similar to warfarin, people taking anticoagulants should not take high doses of vitamin E.
Toxicity
Vitamin E is one of the least toxic vitamins and is safe up to at least 100 × RDA (LD50 values in rats are 2 g/kg). The greatest risk in susceptible individuals is of haemorrhage due to vitamin K antagonist effects. However, at very high doses, vitamin E can antagonise other fat-soluble vitamins such as A and D.
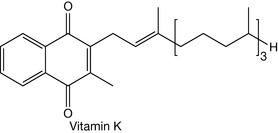
Vitamin K
Dietary sources (% RDA per 100 g in brackets)
Green leafy vegetables are the major sources, while fruits are poor sources. Spinach (300%), cauliflower (250%), broccoli (200%), cabbage (200%), lettuce (150%), beef liver (100%), wheat bran (100%) and oats (100%) (Fig. 26.20).
Biological functions
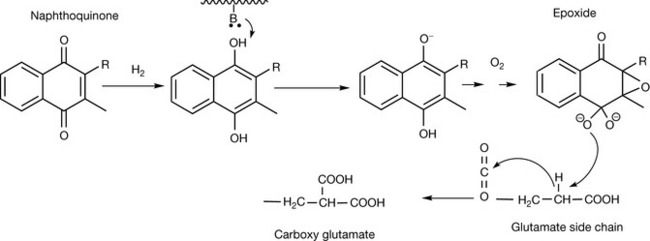
Vitamin K is a co-factor for glutamyl carboxylase, which is responsible for the post-translational carboxylation of glutamyl residues in the clotting factors II, VII, IX and X (Fig. 26.21). The introduction of carboxyl groups into these proteins allows them to bind calcium, which is part of the clotting process. The anticoagulant drug warfarin interferes with the recycling of vitamin K epoxide back into its naphthoquinone form, resulting in a reduction in vitamin K levels and thus reduced tendency for clot formation. Paradoxically, vitamin K is also required for the anticlotting proteins, protein C and protein S. Vitamin K is also required for the activation of the calcium-binding proteins matrix gla protein and osteocalcin which are required for bone mineralisation. Like many vitamins, there are several structurally related forms of vitamin K.
Symptoms of deficiency
The symptom of vitamin K deficiency is increased risk of haemorrhage, ease of bruising, nose bleeding and blood in the urine. In infants, vitamin K deficiency may result in intracranial bleeding. Vitamin K deficiency is rare in adults although vitamin K status is assessed in the newborn and in the USA a routine injection of vitamin K is recommended for newborn babies, particularly premature babies where the vitamin K cycle may not be fully established. Human breast milk is relatively low in vitamin K. There has been some controversy over whether or not childhood leukaemia can be linked to injection of newborns with vitamin K, but a large retrospective study found no link. Certain drugs such as warfarin, sulphonamides and cephalosporins can affect vitamin K function.
Therapeutic indications
There is no strong evidence for vitamin K supplementation reducing the incidence of bone fracture in the elderly although there is some evidence that long-term therapy with anticoagulants such as warfarin can reduce bone density. There some evidence that vitamin K deficiency may promote calcification of atherosclerotic plaques through reducing the protective role afforded by carboxylated matrix gla protein.
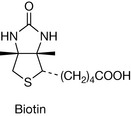
Biotin
Dietary sources (% RDA per 100 g in brackets)
Liver, nuts and eggs are the most important food sources for humans. Bioavailability is very variable. Sources include: molasses (150%), brewers yeast (100%), soybeans (100%), wheat bran (50%), walnuts (50%), peanuts (50%), oats (30%), eggs (15%) and cauliflower (15%) (Fig. 26.22).
Biological functions
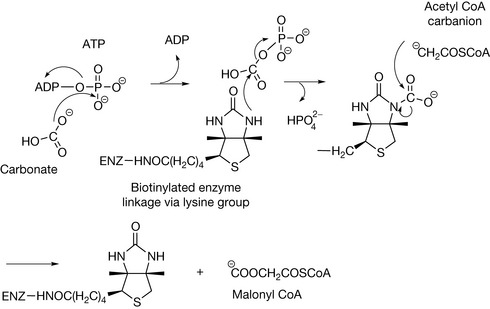
Biotin is a di-alkyl urea and is a co-factor in four key carboxylase enzymes which are involved in fatty acid biosynthesis and amino acid metabolism. In order to function as a co-factor, biotin has to be linked covalently to the enzyme via a lysine group. It then functions as a carrier for a carboxyl group which is attached to a weakly basic urea nitrogen in the co-factor via reaction with carbonyl phosphate. Figure 26.23 shows the carboxylation of acetyl CoA by acetyl CoA carboxylase to form malonyl CoA; this is a crucial step in fatty acid biosynthesis. The other carboxylations which biotin participates in are the carboxylations of pyruvyl CoA (required in the formation of glucose from amino acids and fatty acids), propionyl CoA (required in the catabolism of amino acids and cholesterol) and methyl crotonyl CoA (required in the catabolism of leucine).
Biotin is also used in the modulation of histone activity. Histones are a family of proteins responsible for packaging DNA in order to form chromosomes. It is believed that attachment of biotin (biotinylation) helps reduce the binding of histones to DNA, thereby allowing DNA replication to occur.
In vitro biotinylation of amine and sulphhydryl groups within proteins is used as a method for tagging proteins in biochemical studies.
Symptoms of deficiency
Few cases of biotin deficiency have been reported in humans. There are some rare congenital disorders of biotin metabolism. However, it has been shown to occur following extended consumption of raw eggs since egg white contains a protein avidin which binds to biotin and prevents its absorption. Cooking of eggs denatures the avidin. Symptoms of deficiency include: hair loss, a red rash on the face, unusual fat distribution and general depression and lethargy.
Therapeutic indications
Two rare hereditary disorders of biotin metabolism have been identified; these can both be treated by supplementation with high-dose biotin. There may be an increased requirement for biotin during pregnancy where cells are rapidly dividing and there is an increased requirement for DNA replication. Anticonvulsant medication used in treating epilepsy may deplete biotin. There is some evidence that biotin stimulates glycogen synthesis and thus may be useful as a supplement for diabetics. A number of trials indicated that biotin might be useful as a treatment for brittle nails. Biotin has been used for treating seborrhoeic dermatitis in infants.
Folate
RDA 0.2 mg (0.4 mg in pregnancy).
Dietary sources (% RDA per 100 g in brackets)
Folate is widely distributed in foodstuffs but is unstable to cooking and has reduced bioavailability in fruits and vegetables. Sources include: brewers yeast (800%), beef liver (100–500%), broccoli (100%), spinach (50%), Brussels sprouts (50%), peas (50%), wheat bran (40%), bananas (25%), oranges (15%) and tomatoes (15%) (Fig. 26.24).
Biological functions
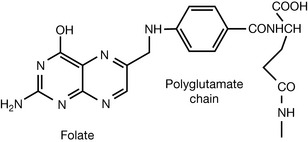
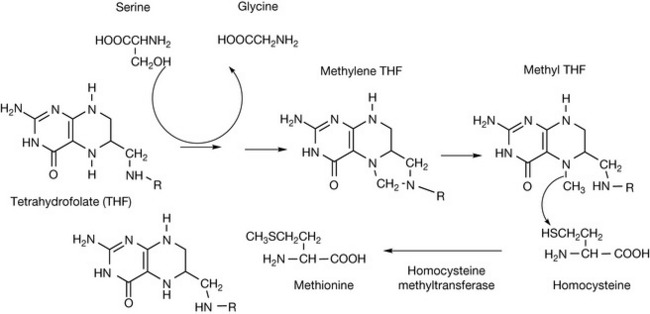
Folic acid is important for the production of new cells and thus is particularly important during pregnancy. Its importance stems from its role as and acceptor/donor of one carbon unit which is a biochemical process required during DNA and RNA synthesis. In order to accept one carbon unit, folic acid is transported into the cell and trapped there by polyglutamylation. It is then reduced to tetrahydrofolate which can accept one carbon unit from several sources. Figure 26.25 shows the donation of a methyl group from methyltetrahydrofolate to homocysteine; the methyl unit is derived from serine, which is the most commonly used primary source of methyl groups. Tetrahydrofolate is involved in the methylation of some of the purines and pyrimidines used in DNA biosynthesis, including thymidine and all of the purines.
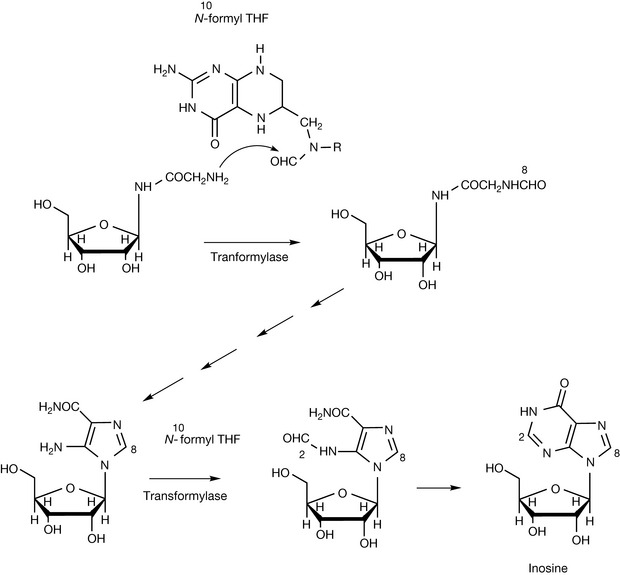
Figure 26.26 shows the role of formyl THF in the biosynthesis of inosine monophosphate which is subsequently elaborated to yield the purine bases used in DNA and RNA biosynthesis. Formyl THF, also known as leucovorin, is used to mitigate the myelosuppressive effects of methotrexate used in cancer chemotherapy which inhibits dihydrofolate reductase, thus reducing the levels of the various single carbon transfer forms of tetrahydrofolate.
Symptoms of deficiency
Folate deficiency can occur in alcoholics, pregnancy and dietary insufficiency. Deficiency results in changes in the red and white blood cells. Changes in the red blood cells lead to the same symptoms as pernicious anaemia which are observed in vitamin B12 deficiency. Increased levels of folate are required during pregnancy, lactation and rapid growth.
Therapeutic indications
Folate supplementation is often given in pregnancy where its deficiency can lead to birth defects resulting from defects in neural tube development such as spina bifida, anencephaly and encephalocele. The critical point where adequate folic acid levels are required is between 21 and 27 days post-conception where most women do not realise they are pregnant. Fortification of foodstuffs with folic acid has reduced the incidence of spina bifida greatly and it is recommended that women of childbearing age should take supplements of folic acid at 400 μg per day. There is no strong evidence that folic acid can reduce the risk of developing heart disease although it can reduce the levels of the risk factor homocysteine in plasma. Folate may reduce the risk of developing colorectal cancer and breast cancer although supplementation with high levels of folic acid may actually accelerate tumour growth. Supplementation may also be important in the elderly who exhibit depressed levels of folate, and adequate levels of folate may be important in reducing the risk of developing Alzheimer’s disease.
Toxicity
The toxicity of the vitamin is low and it is safe up to at least 10 × RDA even in pregnant women. There is a risk that taking folic acid supplements may mask some of the effects of vitamin B12 deficiency without solving the underlying problem, thus resulting in neurological damage. Some drugs and xenobiotics reduce folate utilisation including ethanol, aspirin, ibuprofen, trimethoprim, pyrimethamine, phenytoin and phenobarbital.
Niacin
Dietary sources (% RDA per 100 g in brackets)
Niacin is widely distributed in foodstuffs and can also be derived from tryptophan and thus the tryptophan content of food is also an important source. Sources include: brewers yeast (300%), peanuts (100%), tuna (75%), wheat bran (50–200%), chicken (30–80%), mushrooms (25%), brown rice (25%) and peppers (20%) (Fig. 26.27).
Biological functions
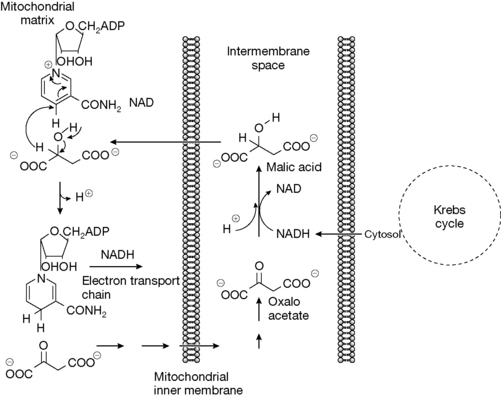
Niacin in the form of nicotinamide functions as an essential component of the enzyme co-factors NAD(H) and NADP(H) which are involved in approximately 200 enzyme reactions. NADH is involved extensively in energy metabolism where glucose in metabolised to produce ATP, the main energy storage molecule in the body. NAD and NADH function as a redox pair, where in some cases NADH acts as a reducing agent and in other cases NAD acts as an oxidising agent. This is illustrated in Figure 26.28 which shows the maleate–aspartate shuttle which is required to effectively transport NADH through the inner membrane of the mitochondrion, which is impermeable to NADH, into the mitochondrial matrix where it can enter the electron transport chain which results in the production of ATP. To achieve this, NADH produced in the cytosol by the Krebs cycle is used to reduce oxaloacetate to malic acid in the intermembrane space which is then transported into the mitochondrion where it is then oxidised by NAD to produce NADH.
Although NADPH only differs from NADH by addition of a phosphate group, it is used for different purposes, being used purely for reduction, not oxidation, and thus it is used in anabolic processes such as synthesis of fatty acids. In addition to redox reactions, NAD fulfils another important function which is in the transfer of the ADP-ribose portion of the molecule to a protein substrate, releasing nicotinamide as a side product. The ADP-ribose is attached to glutamate, aspartate or arginine side chains. This process has an important role in cell signalling. A number of bacterial toxins are ADP-ribosyl tranferases; for instance, cholera toxin ADP-ribosylates G-proteins, which provokes high levels of fluid secretion in the gut and thus life-threatening diarrhoea.
Symptoms of deficiency
The most marked indication of niacin deficiency is pellagra, where the skin is reddened as if sunburned on exposed surfaces along with cracking and shedding of the skin, inflammation of the tongue and diarrhoea. Pellagra was quite common in previous centuries in areas where the staple cereal was maize because although niacin is present in corn it is not readly bioavailable from this source. Niacin can also be synthesised by the body from dietary tryptophan although the conversion is not that efficient.
Therapeutic indications
There is some evidence that niacin can reduce the incidence of cancers of the mouth and throat. Niacin has been used to protect β-cells in the pancreas in insulin-dependent diabetics and has been reported to increase insulin sensitivity. The effects of niacin in lowering cholesterol and low density lipoprotein have been studied extensively. High doses up to 1 g per day are required but the evidence for effectiveness is not wholly conclusive. There is some evidence that high doses of niacin improve survival rates in HIV infection. In conjunction with tryptophan, niacin has been used to treat depression.
Niacin has been used to stimulate tooth eruption and gastric motility.
Essential elements
Introduction
The major elements used in bulk by organisms are: hydrogen, sodium, potassium, magnesium, calcium, carbon, nitrogen, oxygen, phosphorus, sulphur and chlorine.
In addition, there are a number of essential trace elements which include: iron, copper, zinc, nickel, manganese, chromium, molybdenum, boron, silicon, selenium, iodine and fluorine. For some organisms vanadium is required. It is possible that tungsten, arsenic, tin and bromine may also possibly be essential trace elements.
Sodium
Biological functions
Osmotic control and maintenance of blood volume, conduction of electrical impulses, stability of DNA and membranes. Potassium is more much more abundant within cells and sodium is confined to extracellular fluids.
Symptoms of deficiency
Sodium deficiency is rare under normal circumstances but where excessive loss of sodium chloride through perspiration occurs salt intake may have to be increased. It also has to be replaced when electrolytes are lost through diarrhoea and vomiting.
Therapeutic indications
Sodium deficiency is corrected by infusions in severe cases or using oral rehydration salts to replace sodium lost by vomiting or perspiration. Extensive studies have been made on the effect of salt intake on blood pressure; however, the actual effects of a reduction in the diet remain controversial. Excessive sodium levels have been linked to hypertension but this is probably only in a subpopulation of susceptible individuals.
Potassium
Symptoms of deficiency
An abnormally low plasma potassium concentration is referred to as hypokalaemia. Hypokalaemia results most commonly from loss of potassium as a result of prolonged vomiting, the use of some diuretics, some forms of kidney disease and metabolic disturbances. Symptoms of hypokalaemia include fatigue, muscle weakness and cramps, and intestinal paralysis. Acute hypokalaemia may result in cardiac arrhythmias that can be fatal.
Therapeutic indications
High dietary potassium has been linked to lower blood pressure, and dietary supplementation with moderate amounts of potassium has been found to lower blood pressure. Many drugs have effects in either raising or lowering potassium levels in the body. Levels are raised by digoxin, trimethoprim-sulfamethoxazole, ibuprofen, ACE inhibitors, angiotensin receptor blockers, spironolactone and heparin. Levels of potassium are lowered by diuretics, corticosteroids pseudoephedrine, some penicillins, carbenoxolone, caffeine and theophylline.
Calcium
RDA 800 mg (children 400–600 mg).
Dietary sources
Milk, cheese, broccoli, kale, sweet potatoes, canned salmon with bones, sardines, calcium fortified orange juice.
Biological function
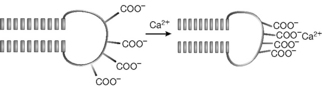
Calcium is the positively charged equivalent of phosphate, producing mechanical effects within organisms. Thus potassium causes vasoconstriction and vasodilation. When a muscle fibre receives a nerve impulse, ion channels in the muscle cell open, allowing calcium ions to enter the cell, which results in displacement of calcium ions stored within the cell, and finally binding of the calcium ions to proteins in the cell that cause the cell to contract and thus produce muscle movement. Calcium binds to negatively charged groups on proteins, particularly glutamate, causing the conformation of the protein to change as shown in Figure 26.29. The effectiveness of calcium in this type of activity relates to its high charge density and relatively large atomic radius so that it can interact with several carboxylate groups. The ability of calcium to bind to proteins and change their shape also is important in other protein systems apart from muscle cells. Thus calcium binds to enzymes which are involved in phosphorylation and de-phosphorylation, not actually at the active site but influencing the effect of the active site on the substrate (allosteric binding). The role of calcium in blood clotting and bone mineralisation has been discussed under vitamin K and vitamin D, respectively.
Symptoms of deficiency
Low blood levels of calcium are often associated with malfunction of the parathyroid gland but this is rarely due to calcium deficiency in the tissues since the skeleton contains large amounts of reserve calcium. Other causes include alcoholism, low magnesium intake and vitamin D deficiency. Low calcium levels cause muscular twitching and in the extreme case tetany. Calcium intake may be of concern in the elderly if not enough dairy products are being consumed, and also in vegans. High-fibre diets can reduce calcium absorption.
Magnesium
Dietary sources
Brown rice, avocados, spinach, haddock, oatmeal, baked potatoes, broccoli, yoghurt, bananas.
Biological roles
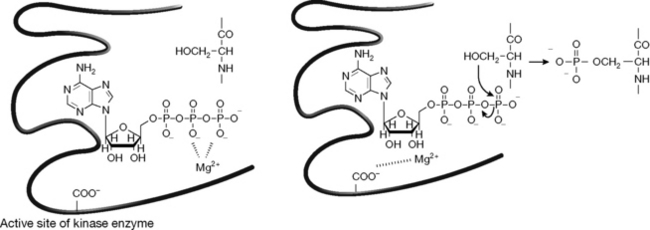
The magnesium ion has the highest charge density of any element used in biology (charge/atomic radius). Magnesium is strongly associated with di- and triphosphates which are highly negatively charged and can balance the high positive charge on the magnesium. This makes magnesium the most important cation at the active site of enzyme reactions involving phosphorylation/de-phosphorylation. Thus Mg2+ has an important role in stabilising and energising biomolecules containing phosphate groups such as ATP, GTP, DNA, RNA, phospholipids and polysaccharides such as phosphoinositols. For example, in the active site of a kinase (phosphorylating enzyme) the transfer of the phosphate group is assisted by Mg2+ moving away from ATP, thus destabilising it and facilitating transfer of a phosphate group to the substrate (Fig. 26.30) Thus it has a role in the energetic processes of the cell. It also has a role in cell division through its effects on DNA and RNA replication and it is involved in cholesterol biosynthesis though its association with pyrophosphate in isopentenyl pyrophosphate.
Symptoms of deficiency
Low magnesium levels can increase muscular cramp and muscle weakness and cause changes in heart muscle. Magnesium deficiency can occur in alcoholism, in association with gastrointestinal disorders, after treatment with diuretics and as a result of severe renal disease.
Therapeutic indications
There is some evidence that low serum magnesium levels are associated with the development of hypertension. There is a clear therapeutic use for high-dose magnesium infusions in the treatment of the life-threatening condition pre-eclampsia, characterised by seizures, which can occur during pregnancy. This has been the treatment of choice for many years and it is believed that the Mg2+ relieves cerebral blood vessel spasm. This is possibly the reason why there is some evidence that Mg2+ may be an effective treatment for migraine headaches. It also has been used to prevent ventricular arrhythmia. High levels of calcium intake can result in magnesium depletion.
Iron
Dietary sources
Beef, baked potatoes, soybeans. Iron is only absorbed efficiently from meat in its haem form. Plants generally contain organic substances which complex with iron, reducing its bioavailability.
Biological functions
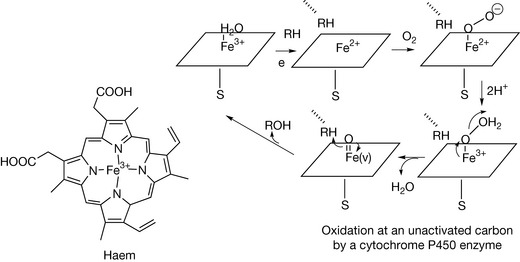
The main role of iron is as part of the haem molecule which becomes incorporated into a variety of haem proteins which are involved in oxygen carrying, oxidation and peroxidation reactions and in the transfer of electrons. The haem unit is bound into the active site of the enzyme by a sulphur atom attached to either a methionine or cysteine residue. Figure 26.31 shows the action of the haem unit within a cytochrome P450 (CyP450) enzyme. CyP450 enzymes are the most important enzymes in phase I metabolism of drugs and xenobiotics and also in the synthesis of hormones such as the steroid hormones.
Symptoms of deficiency
Iron deficiency is the most common nutrient deficiency in the world. Iron deficiency manifests as hypochromic anaemia where the red blood cells are pale in colour and this manifests as pale skin, tiredness, headache and palpitations. People most at risk of iron deficiency include: babies between the ages of 6 months and 4 years; adolescents, particularly adolescent girls who have commenced menstruation; pregnant women; people with gluten intolerance; vegetarians because of the low levels of iron in vegetables compared with meat; and athletes.
Zinc
Biological functions
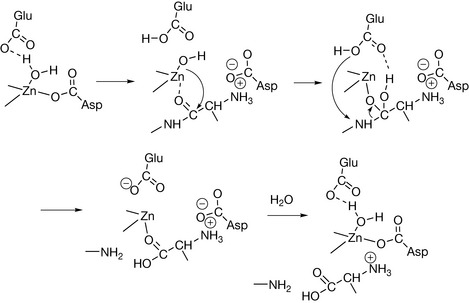
Zinc (Zn2+) is the mineral equivalent of vitamin C in that it appears to have many functions and can almost be regarded as a hormone. Its role in biology is due to a combination of the fact that it binds readily to a variety of ligands and is capable of rapid off/on ligand exchange. Zinc is present at the active sites of hydrolytic enzymes: proteases, nucleotidases, collagenase. Figure 26.32 shows the proposed mechanism for zinc at the active site of a peptidase enzyme, e.g. carboxypeptidase, which hydrolyses terminal amino acids in proteins. At the active site of the enzyme zinc binds to water, making the water more basic so that it can attack the peptide bond. This type of mechanism is used in all the hydrolytic activities of zinc enzymes. Zinc is also involved in many of the enzymes associated with DNA synthesis: RNA polymerase, reverse transcriptase, tRNA synthetase. It is also involved with other key enzymes: carbonic anhydrase, alcohol dehydrogenase and phospholipase C.
Zinc cross-links proteins in a manner similar to S–S bridges. For example, insulin may be formulated as its zinc complex in order to produce a slow-release form of the drug. In the body, this cross-linking results in the formation of zinc fingers in receptor proteins which are important binding sites for hormones such as corticosteroids.
Symptoms of deficiency
These include impaired immunity, weight loss, loss of taste and smell, loss of appetite, skin rashes and depression. Some people may be borderline zinc deficient. Accelerated rates of wound healing and improved appetite and taste acuity occurred when these people were supplemented with zinc. Zinc deficiency may occur in pregnant women, young children and strict vegetarians.
Therapeutic indications
Zinc supplementation has been proposed to be useful in accelerating development in premature babies. Zinc is important for male fertility. Zinc has effects on the immune system and it has been shown that zinc in combination with oral rehydration therapy decreases duration and severity of acute childhood diarrhoea which affects over 3 million children in developing countries each year. Zinc lozenges have been used in treating the common cold but there is no strong evidence that they are effective; combining zinc with vitamin C may be more effective. Zinc may have some effect in promotion of wound repair. Its oxide, of course, has been used for many years in creams for treating nappy rash and in calamine lotion where it shows antipruritic properties.
Toxicity
Large amounts of zinc (2000 mg) cause vomiting. Even taking >30 mg daily can interfere with copper absorption. Taking supplements of >15 mg per day for prolonged periods would not generally be advisable. Zinc nasal sprays for treating colds should be avoided since they can cause a loss of the sense of smell.
Copper
Biological functions
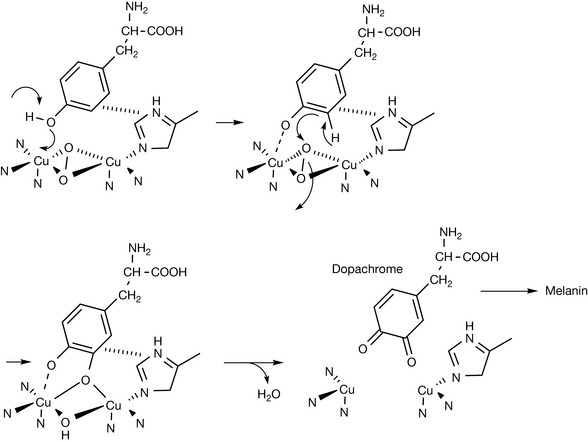
In some senses enzymes with copper at their active sites act in an opposite manner to those with zinc at their active sites. Copper-based enzymes are responsible for carrying out oxidation reactions which lead to formation of organic structural polymers such a collagen and melanin (responsible for protection against UV radiation). Copper-based enzymes have two copper atoms in the Cu(I) oxidation state at their active site to which molecular oxygen becomes bound. Figure 26.33 shows the action of the copper-based enzyme tyrosinase which oxidises the amino acid tyrosine to produce dopachrome, the precursor of the melanin pigments.
Copper is also present in amine oxidase which terminates the action of many nitrogenous drugs and neurotransmitters such as adrenaline by converting them to aldehydes and also in enzymes which reduce O2 to H2O, thus protecting cells against oxidation.
Symptoms of deficiency
Copper-deficient animals have weakened hearts and blood vessels and bone defects similar to those observed in osteoporosis.
Manganese
Dietary sources
Pineapple juice, wheat bran, wheat germ, whole grains, seeds, nuts, cocoa, shellfish, tea.
Biological functions
Manganese is involved in carbohydrate, lipid and protein metabolism and is required for the synthesis of glycoproteins and mucopolysaccharides such as hyaluronic acid. It is also involved in glucose metabolism. In plants, it is involved in the enzyme that splits water into oxygen and hydrogen.
Symptoms of deficiency
Animals which are manganese deficient have malformed bones which are similar to those observed in osteoporosis. They also have problems with their tendons.
Cobalt
Chromium
Biological functions
Chromium is involved in glucose metabolism and it appears to enhance the action of insulin. It is also involved in the action of a number of enzymes.
Symptoms of deficiency
Deficiency may be associated with glucose intolerance, which is associated with elevated cholesterol and glucose levels in plasma.
Therapeutic indications
Chromium is not common even in a balanced diet and thus supplements may be of value. It may benefit newly diagnosed diabetics who have mild glucose intolerance. It may also benefit those with low blood sugar levels since it promotes the action of insulin. Chromium is best absorbed in the form of its nicotinate or picolinate salts.
Selenium
Biological functions
Selenium is involved in the protection of cells and membranes against oxidative damage and its action appears to be linked to the action of vitamin E. Selenium appears to have antiviral effects, for example on the AIDS virus. It is present in a number of proteins including glutathione peroxidase and in the form of the amino acid selenocystine.
Therapeutic indications
Environmental damage tends to reduce selenium levels in the soil and thus there is less selenium in the food chain. Selenium functions as an immunostimulant and antioxidant. It also binds toxic elements such as arsenic, cadmium and mercury. Supplements are best taken in the form of selenomethionine or selenocystine rather than selenite.
Molybdenum
Biological functions
Molybdenum is present in some enzymes, especially those required for purine metabolism, such as xanthine oxidase and metabolism of sulphur-containing compounds.
Symptoms of deficiency
Molybdenum deficiency is difficult to induce even in animals and no deficiency syndrome has been recognised in humans to date.
Fluoride
Biological function
Cannot be regarded as essential although its incorporation into tooth enamel protects teeth from bacterial attack. Fluoride is also taken up by bone tissue, increasing its strength, although trials using it to treat osteoporosis have had mixed results.