7 Spreading infection
ASSUMED KNOWLEDGE
It is assumed that at this stage you will have knowledge/competencies in the following areas:
If you think that you are not competent in these areas, revise them before reading this chapter or cross-check with relevant texts as you read.
INTENDED LEARNING OUTCOMES
At the end of this chapter you should be able to:
CLINICAL FEATURES OF INFECTION
Local features
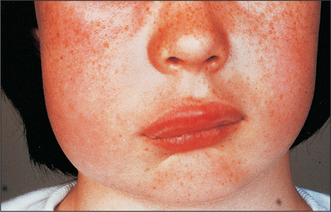
Many signs of infection (Fig. 7.1) are those of inflammation (pain, swelling, redness, heat), but not all inflammation is in response to infection: all these signs can be seen in rheumatoid arthritis. In infection you may also find suppuration (pus formation), an obvious cause and a greater systemic response.
The pain tends to be throbbing or aching or tenderness. Its severity depends upon the pressure of fluid within the tissue and changes with time.
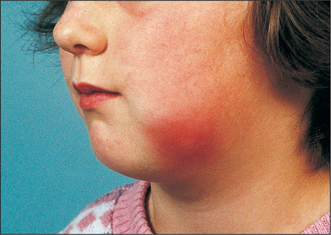
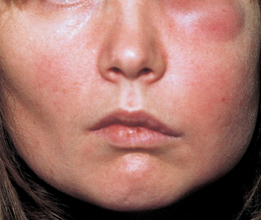
Where swelling is largely due to oedema it is relatively soft. It tends to move within the tissues and accumulates at sites least constrained by fascia, as for instance, lips and eyelids (Fig. 7.2).
Some swelling is due to the cellular infiltrate of inflammation. This is more firm and is described as ‘indurated’ (hard). This induration is not due to fibrosis, but nevertheless may take days or weeks to resolve in infections in which it is a prominent part.
For swelling due to oedema or to cellular infiltrate it is difficult to define the precise margin of a swollen area: there is a gradual change at the edge towards normality.
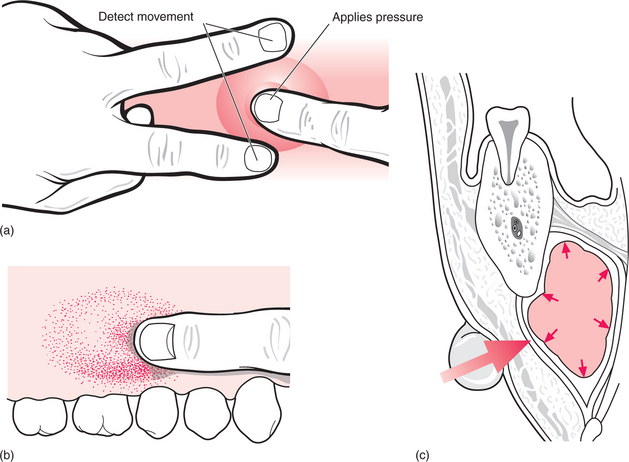
Many infections form pus; this adds to the swelling. A collection of pus is called an abscess. When close to the surface it may cause a yellowish discolouration of the overlying mucosa but, when deeper, all that will be seen is the redness of inflammation. Swelling due to pus has a very different feel to it from that due to inflammatory exudates. It is described as ‘fluctuant’, but that encompasses several different sensations detected by the examining fingers (Fig. 7.3). Classically, fluctuance is determined by placing two fingers at the sides of a swelling and detecting fluid movement caused by a third finger on the centre. That is not easy inside the mouth, where it may be possible to detect fluid movement only by running one finger along the swelling. For deeply placed abscesses in the neck, the feeling is more like tense springiness.
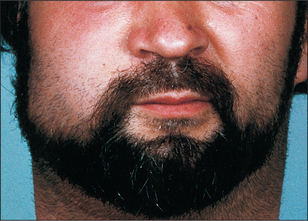
The redness (and local heat) of inflammation is due to increased blood flow. There is no local increase above body core temperature and thus for intraoral locations there may be no local ‘heat’.
Bacterial infections of dental origin have a characteristic natural history. The time scale is typically hours to days, from the first symptoms to the first request for medical or dental assistance. If infection is initially periapical there may be considerable pain, while exudate and pus are under pressure within bone, followed by a reduction in pain and rapidly increasing facial or neck swelling as the infection escapes bone and pressure reduces. At this stage the external swelling is largely due to oedema, and therefore soft. Over a period of 1–5 days pus may form centrally within this swelling: this localization is associated with developing pain, local tenderness and fluctuance. Oedema and pus may spread inwards towards the pharynx as readily as outwards towards the face.
When infection shows no significant localization of pus and has a greater tendency to spread it is called cellulitis. Where the predominant feature is pus formation it is called an abscess. However, almost all infections show elements of both and any infection starting as a cellulitis tends to localize over a period of days.
Spread of infection
Pus tends to move under influences such as pressure, gravity, local heat or muscle layers towards surfaces. When it reaches a surface (internal or external) it bursts out or discharges, but often with large abscesses it takes days to drain and spontaneous drainage is unreliable. Pus is an effective defence against spreading infection.
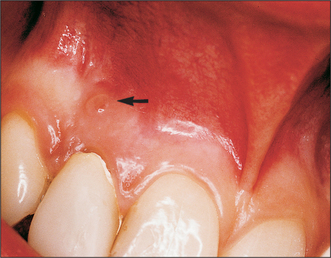
Most suppurative dental infections discharge into the mouth via a sinus, sometimes without obvious acute infection (Fig. 7.4), and usually onto the labiobuccal aspect of the alveolus. Apical infection from maxillary lateral incisors is more likely to drain palatally and from any tooth may point lingually, palatally or even onto the skin (Fig. 7.5). The commonest site of discharge onto skin is the point of the chin, arising from infection at the apex of a mandibular incisor. However, it is when, rarely, the infection tracks beyond the alveolus but does not readily escape onto a surface that the infections described in this chapter develop. The interlinked planes and spaces to which dental infections may spread have few absolute boundaries but can be summarized by considering the example of the third molar.
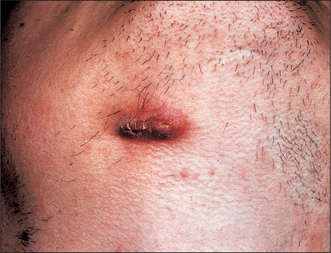
Fig. 7.5 An extraoral sinus (beneath the mandible on the right side, related to apical infection on a lower molar).
The crown of the part-erupted mandibular third molar, particularly if distoangular, may be below the attachment of buccinator/superior constrictor, allowing infection to escape laterally to the buccal space (Fig. 7.6), posteriorly to the masticator space or posteromedially to the lateral pharyngeal space. The masticator space is the potential space surrounding the ascending ramus and the elevator muscles of the mandible. Infection (whether or not pus has formed) makes these muscles resistant to lengthening, resulting in limited mouth opening, called trismus. Trismus in odontogenic infection indicates involvement of masticatory muscles.
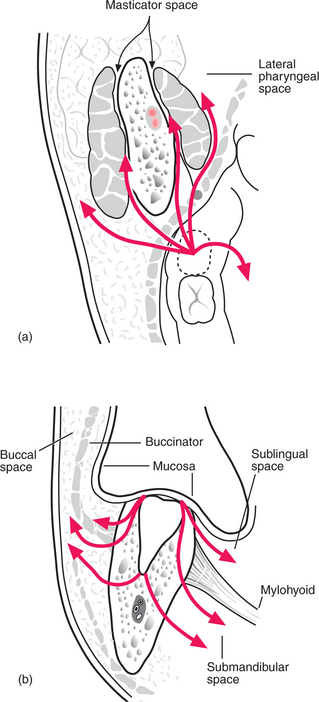
Fig. 7.6 Routes of spread of infection from a lower third molar.
Apical infection from the lower wisdom tooth may escape laterally to the buccal space, producing swelling of the cheek above the lower border of the mandible. As the apex is below the attachment of mylohyoid, infection tracking medially enters the submandibular space, producing swelling in the neck, but sometimes upward bulging of the floor of the mouth too.
Spaces into which infections typically track from the teeth
Infections involving the lateral pharyngeal or retropharyngeal spaces are of particular concern, because of the risk of respiratory obstruction and because they may track downwards directly into the mediastinum, resulting in life-threatening mediastinal infections.
Occasionally infection arising from a maxillary canine or premolar may spread upwards and backwards to involve the orbit. If there develops a thrombophlebitis of the ophthalmic veins or the deep facial vein, such infections may spread to the cavernous sinus.
Recognizing these clinical features should enable you to describe an infection in terms of its spread (i.e. the spaces involved) and its tendency to localization or further spread, then with the duration thus far and the level of systemic upset, make an estimate of the severity of the infection. For all infections of dental origin, there should also be an identifiable cause: a part-erupted third molar; a non-vital tooth with its apex beyond muscle attachments; a site of injection; a fracture; a foreign body.
Systemic features
A raised body core temperature is common in infections of all types. The normal temperature varies widely according to the metabolic rate and the time of day. The upper limit of the normal range is 37.0°C but this may actually be a raised temperature for some individuals, and a ‘normal’ temperature may be higher than this such as at the time of ovulation in women. Therefore take temperature only as a guide and watch for changes over time. Temperature may be measured sublingually, provided that the mouth will open satisfactorily and it is not too painful. Endaural (within the ear) measurement can also be accurate and convenient if the equipment is available. Alternatively, take the axillary temperature, allowing for it being about 1°C below core temperature.
A substantial abscess may cause temperature ‘spikes’ (Fig. 7.7) on a daily basis. A single temperature reading taken at a trough between such spikes will be misleading. The pulse and respiratory rates rise with or slightly ahead of the temperature.
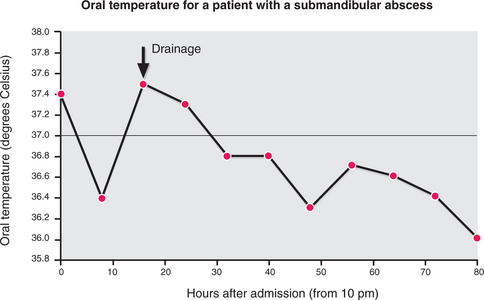
Fig. 7.7 A spiking temperature (oral measurement) in a patient with a submandibular abscess of dental origin.
The malaise (feeling unwell) that is standard with infections such as influenza is often not a prominent feature of bacterial odontogenic infection. If the infection is severe, a greyish pallor of the face may be evident, but again this is relatively unusual and less than that with viral infections.
Regional lymph nodes are usually enlarged and tender, although if there is much neck swelling individual groups of nodes may not be distinguishable on palpation. Almost all cervicofacial infections drain to the jugulodigastric node in the upper part of the deep cervical chain, but mandibular infections tend to go first to the submandibular nodes (or anteriorly, to the submental nodes). Facial skin infections may drain to the facial node.
PATTERNS OF PRESENTATION
Alveolar abscess
This infection is largely confined to the mouth, with swelling centred around the alveolus near the cause. Usually within 2 days of appearance of first symptoms pus forms and becomes evident as a fluctuant swelling on the labiobuccal aspect of the alveolus. The degree of systemic disturbance is often slight.
Cellulitis
The overlying skin is swollen and oedematous (pitting occurs in some cases), with particular swelling of lips and eyelids. There is usually no true fluctuance (unlike the abscess, although most infections do form some localized fluid collection) and the development tends to be more open-ended, with a progressive spread to involve adjacent spaces, cross the midline and eventually down the neck. Often the systemic upset is more severe than with an abscess.
Cervicofacial space abscess
There is less oedema, and the infection seems more deeply placed than that of a cellulitis because there is less skin inflammation, but the clinical signs and the symptoms depend upon the spaces involved. Both masticator and lateral pharyngeal space infections are associated with severe trismus. In either case the abscess cavities may be inaccessible to the examining finger, preventing identification of fluctuance. Lateral pharyngeal abscesses and sublingual space infection may cause severe pain on swallowing. Sublingual space infection also causes raising of the floor of mouth and the tongue.
DISTINGUISHING INFECTIVE FROM NEOPLASTIC DISORDERS
There is usually no difficulty in distinguishing infective from other disorders. However, confusion can arise in the slower, lower-grade infection and the superficial infected tumour. Secondary malignancies are less common in the mouth than primaries, and by arising within bone may cause confusion.
Generally, infection develops over a few days, but responds to removal of the cause and/or drainage of pus. Malignancies develop over weeks to months and do not respond to treatments suited to infections. Induration is common in long-standing infection, and may persist for days to weeks after treatment, but should show signs of improvement with treatment. By the time tumours are evidently infected, they are usually obviously ulcerated, which would be rare for an infection of dental origin.
Lymph node involvement may also reveal differences between tumours and infection. Usually, dental infections cause lymphadenopathy in the upper part of the cervical chain and submandibular nodes. Infected lymph nodes are likely to be enlarged, firm or rubbery in consistency, tender, usually mobile, while neoplastic lymph nodes are often enlarged, hard in consistency, non-tender, fixed, especially in advanced disease. A lesion associated with enlarged nodes lower in the neck, or showing spread upwards or backwards in the face or neck, should arouse suspicion.
The rule must be: if infection is responding poorly to what should be satisfactory treatment, neoplasia should be considered.
INVESTIGATION
Microbiology
The identity and antibiotic sensitivity of the causative microorganisms is commonly determined from pus samples. To sample with a swab (Fig. 7.8), soak it in pus from the main abscess cavity and not from the skin or mucosal incision, to avoid contamination by surface organisms. In samples left open to the air, oxygen kills the anaerobes and drying kills most other bacteria; therefore swabs must be sent for culture within 1 hour, in an appropriate transport medium, to the microbiology laboratory. Aspirates of pus taken with a syringe and needle are more readily protected from the air and may be more reliable, but still require rapid attention. For a spreading infection, without pus, the organisms can often be grown from a blood sample. This procedure is best performed in hospital. On occasion organisms might be sought in tissue washings or biopsies.
For most minor infections of dental origin, culture of microorganisms adds little because, by the time sensitivity results are known (2–3 days), the local treatment and antibiotics have substantially resolved the infection. This is not a safe approach with extensive infections, which have a low, but real, incidence of serious outcomes.
Response to infection
Reduced resistance should be considered in those with severe infection. The normal response to acute bacterial infection includes a considerable increase in circulating blood white cells, particularly the neutrophil polymorphs. (An increase in lymphocytes is associated with viral infections.) A full blood count will also demonstrate anaemia (if present), or a reduced white cell count. A blood film will identify abnormalities of red or white cell morphology. If the infective nature of the condition is doubted, C-reactive protein (CRP) may be a helpful guide to the severity of inflammation. Urinalysis or a fasting blood sugar estimation may detect previously undiagnosed diabetes, but remember that severe infection itself tends to raise the blood sugar level. Also consider the recent use of corticosteroids, alcohol or drug abuse or HIV infection.
SURGICAL TREATMENT OF INFECTION
Early removal of the ‘cause’, such as by tooth extraction, is important in management of dental infections. If the severity or spread of infection makes local anaesthesia, access for extraction or induction of general anaesthesia impracticable or dangerous, this treatment may be delayed.
Drainage of pus is an essential part of the treatment of suppurative infections. Sometimes it may be appropriate to encourage spontaneous drainage, particularly in small, localized, superficial abscesses, but usually active surgical intervention is required.
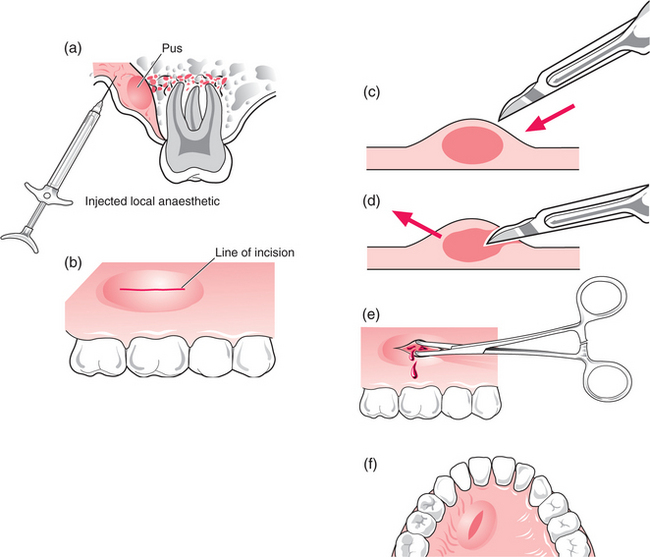
Drainage of intraoral abscesses (Fig. 7.9) may be performed using local anaesthetic injected close to the site of incision (for large, deeply placed abscesses, general anaesthesia may be required). Topical ethyl chloride anaesthetizes only to a very shallow depth. The blade of the scalpel is inserted parallel to the gingival margin, directly into the abscess to the full depth in its long axis, then used to cut outwards towards the surface. This should be followed immediately by a flow of pus. Gently opening the cavity allows the pus to drain. If a pus sample is to be collected, it may be taken at any time up till now. The base of the abscess cavity is usually bare bone. If no discharge of pus occurs, it is likely either that there is no pus in the lesion (yet) or that the incision is not deep enough.
For small abscesses a drain is not usually necessary.
Anaesthesia
The author prefers to use local anaesthetic injected close to the abscess or, if this is not practicable, general anaesthesia. Some prefer to relieve pain with a topical spray of ethyl chloride (to lower the mucosal temperature below 4°C); however, this provides little pain relief. Topical local anaesthetics work to a depth of several millimetres and can be satisfactory for very superficial abscesses.
Drains
Larger and deeper abscesses tend to seal off shortly after drainage, leaving pus inside or still forming: something must be done to hold the cavity open. In the mouth a corrugated rubber or tubular plastic drain (Fig. 7.10)—or, in desperation, the finger of a sterile rubber glove—may be used. These must be sutured in for at least 24 hours. For palatal abscesses, it is more convenient and successful to excise an ellipse of mucosa from the centre of the abscess so that when the mucosa is pushed flat by the tongue the wound cannot seal.
Larger abscesses
Cervicofacial space abscesses require a more vigorous approach to drainage, and even the buccal space abscesses, which can theoretically be incised intraorally, are better approached from outside the mouth. Usually this will be under general anaesthesia on an inpatient basis.
The incision should be placed in a neck crease to leave the least evident scar (Fig. 7.11). To approach a submandibular abscess, make the incision at least two fingers’ breadth below the angle of the mandible to avoid the marginal mandibular branch of the facial nerve. The nerve may also be displaced downwards by the swelling of tissue above, and in inflamed tissue its identification is near impossible.
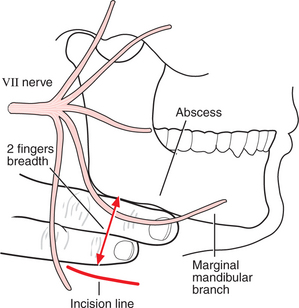
Fig. 7.11 Landmarks for drainage of a submandibular abscess. The marginal mandibular branch of the facial nerve dips below the lower border of the mandible. The incision line should be at least two fingers’ breadth below the lower border.
As pus tends to track downwards under gravity, it is usual to make the incision at the lowest (most dependent) part of the expected cavity. For buccal space abscesses the incision may also be placed in the neck to hide it in a skin crease.
Once the skin incision is made, the abscess is approached by blunt dissection using scissors or a curved haemostat. This involves pushing the end of the instrument into the wound with the tips together, then forcibly opening the instrument to develop a plane of dissection. This is repeated until the abscess cavity is reached. Blunt dissection minimizes the risk of injury to nerves and vessels. A sample of pus should be taken by aspiration at this time, reducing the likelihood of skin contamination.
When the cavity is entered, the access should be enlarged to enable the little finger to be inserted to explore the cavity and gently disrupt any fibrous septae between locules of pus. Some operators irrigate the cavity with saline at this stage to reduce residual contamination.
A drain should be cut that will extend from the deepest part of the cavity beyond the skin edge. This is sutured in place with a material which can be found easily for removal (such as black silk), but the wound is not closed. A non-adherent dressing is placed over the wound and an absorbent dressing placed over that. Some surgeons prefer to seal a stoma bag over the wound to collect the pus. The drain is usually removed after 24–72 hours, depending upon the size of the abscess and its tendency to continue draining. Some surgeons will shorten the drain daily, to allow the deeper part of the wound to fill with inflammatory tissue first. The wound often continues to discharge for a week or more after the drain is removed and the dressings need to be changed daily (or sooner if soaked) until the wound dries.
MEDICAL AND SUPPORTIVE TREATMENT
Antibacterial chemotherapy is central to the treatment of bacterial infection. However, antibiotics may be over-prescribed and there are certainly circumstances when antibiotics are unnecessary.
Antibacterial drugs should be used when:
Antibacterials should not be used to control pus (indeed they will not), but on occasion will prevent spread of infection while awaiting localization. If antibiotics are used to control an abscess, the abscess may go ‘quiet’, but it will become painful and obviously infected again within days of ceasing the antibiotics. Such a persistently swollen, tender, indurated mass is sometimes called an ‘antibioma’, but should not be confused with a tumour, from either its name or its appearance.
Choice of antibiotics
The initial choice of antimicrobial drug is empirical. Most infections of dental origin are caused by a mixture of organisms and both aerobes and anaerobes can often be cultured. In mixed infections, eliminating one organism can be effective in treating the infection, because of synergism between the organisms.
Factors that determine the choice of antimicrobial drug include:
A common first choice is metronidazole (note: this is active only against anaerobes). For mild infection it may be given orally at a dose of 200–400 mg three times a day for 5–7 days. Metronidazole should be taken with or after meals as it is irritant to the stomach. It produces an unpleasant reaction with alcohol and patients should therefore be advised to avoid alcohol while they are taking the drug. Compliance is likely to be poor in patients who drink a lot of alcohol.
The broad-spectrum penicillin amoxicillin covers a range of organisms wider than that of the basic penicillin, penicillin V, but is still well tolerated orally (and with less tendency to cause diarrhoea than oral ampicillin). It is given at a dose of 500 mg three times daily.
The cephalosporins, such as cefradine (250–500 mg four times daily), also have a wider range of activity against oral organisms than penicillin V, and there is some evidence that they may be more effective clinically than either amoxicillin or metronidazole. However, the differences appear to be small and, although cephradine is not an expensive drug it does cost more than penicillin V and metronidazole.
Where it is necessary to ensure a high and consistent blood level of an antibiotic, it is usual now to administer the drug intravenously. This implies hospital admission.
Because the initial choice is empirical, there is a tendency to use two antibiotics in combination when an infection is severe and there is a risk of serious outcome. Again there are arguments for and against a variety of combinations of drugs. The author’s choice for intravenous use is metronidazole 500 mg 12-hourly and ampicillin 500 mg 6-hourly.
Alternative drugs are needed in cases of adverse reaction or if the organisms isolated are not sensitive to the first-choice drugs. Consideration may be given to macrolide antibiotics such as erythromycin (for oral use) or clarithromycin (parenteral), tetracyclines, lincosamides and occasionally the aminoglycosides. Reference may be made to texts on microbiology and therapeutics, and consultant microbiologists will advise in cases of difficulty.
Failure to control an infection with antibiotics may be due to:
Supportive care
The role of supportive care is more difficult to prove. There is little evidence that bed rest affects the outcome of dental infections. However, it is unlikely that taking vigorous exercise is beneficial.
Fluid intake, on the other hand, is of great importance. A patient with a painful mouth and face, especially if it is painful to swallow, often eats nothing and drinks too little, resulting in dehydration over a period of days. Fluid requirements are increased if the temperature is raised, so rehydration is essential. The average adult requires about 2.5 L of fluid per day, but if pyrexial that may rise to 3 or 3.5 L. If adequate fluid cannot be taken by mouth, it must be given intravenously, which implies hospital admission.
Patients with dental infections rarely become dangerously pyrexial (temperature exceeding 40°C), but if they do it is necessary to reduce the temperature with aspirin or paracetamol, or by sponging with tepid water and circulating air over the body.
CASES REQUIRING INPATIENT MANAGEMENT
Localized dental infections in fit individuals are usually managed in dental practice. There are certain cases, however, in which outpatient management is impracticable or unnecessarily risky.
Good reasons for hospital admission include:
Usually on admission the following will be done:
Each case is treated individually and reviewed to ensure earliest recognition of problems. The patient is discharged when the infection is under control, any drainage has been performed and risk of relapse is small.
SPREADING CELLULITIS IN THE FLOOR OF THE MOUTH (LUDWIG’S ANGINA)
A cellulitis starting in the floor of the mouth (Fig. 7.12), often arising from a mandibular molar and not readily localizing, has the potential to threaten life by obstructing the airway.
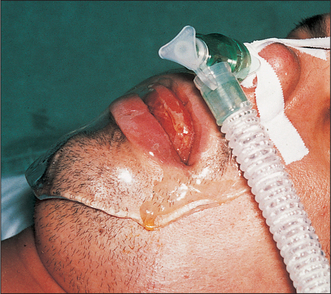
Fig. 7.12 A severe cellulitis of the floor of mouth and submandibular space (Ludwig’s angina).
The tongue protrudes from the mouth and there is extensive oedema below the chin. The patient has been intubated to protect the airway. An inert jelly covers the mouth to prevent drying.
Corticosteroids have been advocated to reduce swelling in these cases, but they reduce resistance to infection and the available evidence on their efficacy is not conclusive.
OSTEOMYELITIS
Osteomyelitis is defined as the spreading infection of bone marrow. Although the clinical features of osteomyelitis are different from those of soft-tissue infections, the disorder may start in the same way and it is valuable to distinguish this infection early. The cancellous bone of the jaw (usually the mandible) seems well protected from apical infection, which usually moves rapidly out into soft tissue. However, sometimes this protection fails, allowing intraosseous spread.
This may simultaneously cause thrombosis of veins in the marrow and stripping of periosteum by pus (Fig. 7.13). The blood supply to the mandible is substantially from the inferior alveolar artery in young people, but becomes progressively dependent on the periosteum and muscle attachments with age. Spreading infection with thrombosis and periosteal stripping thereby causes loss of blood supply to the infected area, resulting in bone necrosis.
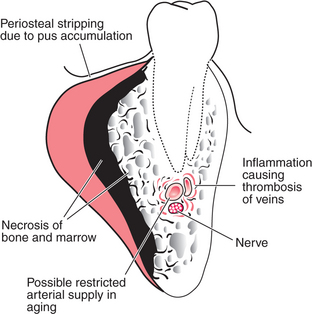
Fig. 7.13 The pathogenesis of osteomyelitis of the mandible.
Inflammation causes thrombosis of vessels in the marrow; periosteal stripping by pus (or surgery) causes loss of periosteal blood supply, with consequent necrosis of bone. This encourages continuance of infection as well as bone resorption.
Osteomyelitis sometimes occurs as an acute infection; it is more likely to do so in the maxilla in children. In that case the infection is particularly severe and probably of different pathogenesis from the chronic mandibular disease. Occasionally osteomyelitis may involve the periosteal surface of the bone exclusively, or appear solely as a sclerotic reaction of the marrow.
Clinical features
The clinical features are thus those of infection in general, plus tenderness of more teeth than would be expected, possibly mobility of teeth, loss of function of the inferior alveolar nerve and numbness of the lower lip. Because of periosteal inflammation and stripping there is a firm or woody expansion of the affected bone area, with no clearly defined junction between the swelling and normal bone. The overlying skin may be mobile, provided there is little acute infection. If the infection is not rapidly resolved, bone resorption becomes evident on radiographs (Fig. 7.14a), necrotic bone may separate as a sequestrum, and new bone may form on the inner aspect of the stripped periosteum and may become visible on tangential radiographs (Fig. 7.14b). Eventually the weakened mandible may fracture.
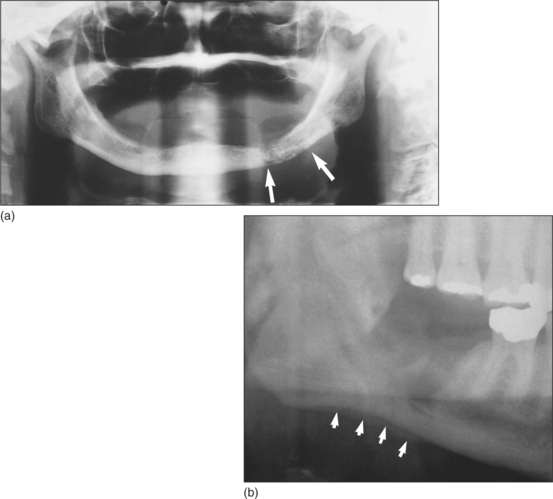
Fig. 7.14 (a) Typical radiological appearance of osteomyelitis. Patchy bone resorption is highlighted by arrows. (b) A thin layer of subperiosteal bone (arrows) has formed at the lower border of the mandible in a patient with osteomyelitis.
Time scale is important in the diagnosis of osteomyelitis. It often takes weeks from the first symptoms till a clear diagnosis is made, even though the condition is obviously infective early on.
Predisposing factors
Factors predisposing to osteomyelitis include those which make any infection more likely (e.g. use of corticosteroids or alcohol dependency) and anything which tends to open up the marrow space widely (fracture or surgical removal of a tooth), or increased density of bone, such as in Paget’s disease or osteopetrosis.
Treatment
The investigation and management of osteomyelitis are best performed within a hospital setting.
Treatment relies upon antibiotics, maintained for 6–8 weeks. The initial choice of antibiotic is usually empirical. Penicillin V is safe and well tolerated, with a reasonable spectrum of activity against oral microorganisms and appears to be effective in this condition. The tetracyclines are bound in bone by chelation of calcium, but are inactive in that form; they have, however, been very successfully used in osteomyelitis. The lincosamides achieve high bone concentrations, but there is a small risk of pseudomem-branous colitis on long-term use. The cephalosporins and penicillins also have their advocates. The initial choice may not be that critical, but the causative organism should be identified and antibiotic treatment modified accordingly.
Any controllable predisposing factor (such as anaemia) should be dealt with early in the management of osteomyelitis.
Surgery is necessary if a substantial sequestrum forms. Occasionally, in resistant cases, it is necessary to remove the lateral cortical plate of bone to allow access for granulation tissue to the remaining bone. Pathological fractures require immobilization and often bone grafting once the infection is settled.
ACTINOMYCOSIS
Actinomycosis is a specific infection caused by Actinomyces species, often arising from a dental source. It differs from many infections of dental origin in being much slower in onset and more chronic in its course. Microbiological diagnosis can be difficult and the clinical signs vary, making this a diagnosis about which one may be uncertain.
If the infection has followed a specific event such as a fracture of the mandible, the time scale is usually a few weeks. There is often low-grade swelling, tenderness and induration (hardening) of the skin of the face or neck. Sometimes this is localized to an area as small as 3 cm, but it can be much more extensive. Often, then, over a short period pain increases, a fluctuant abscess forms superficially and the abscess discharges, only to build up again over days to weeks. The classical actinomycosis (Fig. 7.15) with multiple discharging sinuses and pus containing yellow ‘sulphur granules’ is relatively rare but is easily identified when seen.
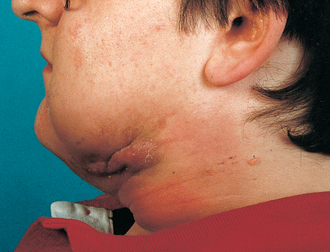
Fig. 7.15 Actinomycosis, with multiple submandibular sinuses, in a patient who did not seek treatment for a fractured mandible 3 months previously.
Treatment is with oral penicillin for a period of about 3 months, with surgical drainage of pus as appropriate. If there is a dental cause, it should be treated early.
NECROTIZING INFECTIONS
Severe necrotizing infections are rare now in Europe, but are still common in parts of the world where poor nutrition is widespread. The mildest form seen is acute necrotizing ulcerative gingivitis, which is well covered in periodontal texts.
Cancrum oris or ‘noma’ is extremely destructive of facial soft tissue, especially around the mouth, and is recognized largely by that feature.
Treatment is with antibiotics (metronidazole and penicillin) in the first instance to control the infection, surgical removal of non-vital tissue (debridement) and closure of the mucosa to the skin surfaces of the wound. Reconstruction is delayed until the general health is stabilized.
Rarely, necrotizing infections caused by a mixed growth of Staphylococcus aureus and a β-haemolytic Streptococcus may start from minor skin abrasions. This has been called ‘synergistic gangrene’. Like cancrum oris, it is usually an indication of a severe underlying reduction in infection resistance. The prognosis is extremely grave.
MRSA
Infections of the head and neck, particularly hospital-acquired wound infections, may be caused by methicillin-resistant S. aureus (MRSA). This organism is remarkably resistant to a range of antibiotics and is prevalent in hospitals because of the widespread use of antibiotics. Infections of this type are a particular risk to the elderly or debilitated patient in hospital. Efforts must be maintained to minimize spread of such infection by strict hygiene measures, by identifying and isolating those with the infection and by careful wound care to encourage rapid healing where infection has occurred.
Adekeye E.O., Cornah J. Osteomyelitis of the jaws: a review of 141 cases. British Journal of Oral and Maxillofacial Surgery. 1985;23:24-35.
Calhoun K.H., Shapiro R.D., Stiernberg C.M., Calhoun J.H., Mader J.T. Osteomyelitis of the mandible. Archives of Otolaryngology. 1988;114:1157-1162.
Fazakerley M.W., McGowan P., Hardy P., Martin M.V. A comparative study of cephradine, amoxycillin and phenoxymethyl penicillin in the treatment of acute dentoalveolar infection. British Dental Journal. 1993;174:359-363.
Har-El G., Aroesty J.H., Shaha A., Lucente F.E. Changing trends in deep neck abscess. Oral Surgery. 1994;77:446-450.
Lewis M.A.O., MacFarlane T.W., McGowan D.A. A microbiological and clinical review of the acute dentoalveolar abscess. British Journal of Oral and Maxillofacial Surgery. 1990;28:359-366.
Lindner H.H. The anatomy of the fasciae of the face and neck with particular reference to the spread and treatment of intraoral infections (Ludwig’s) that have progressed into adjacent fascial spaces. Annals of Surgery. 1986;204:705-714.
Wannfors K., Gazelius B. Blood flow in jaw bones affected by chronic osteomyelitis. British Journal of Oral and Maxillofacial Surgery. 1991;29:147-153.
Young P., Smith S.P., Caesar H. Airway management in Ludwig’s angina. British Journal of Hospital Medicine. 1995;54:239.
SELF-ASSESSMENT
Answers on page 264.