CHAPTER 3 Imaging methods for guidance of aspiration cytology
Percutaneous biopsy is a well-established and routine practice in imaging departments,1,2 and is a frequently performed interventional radiographic procedure, either as an inpatient or outpatient examination, by most trained radiologists. The technique, indications and complications are extensively described in basic imaging textbooks.3 Development of expertise often occurs by ‘apprenticeship’ during undergraduate training for radiologists and for many other clinicians. The procedure is safe, inexpensive and minimally invasive. An understanding of the imaging modalities, the lesion to be biopsied and overall experience of the proceduralist contribute to the success of the procedure.
Fine needle aspiration biopsy (FNAB) typically yields a small sample for cytological assessment, with limited or no architectural information. The decision to proceed to larger-diameter needles (core biopsy) may be determined either by the results of the fine needle aspirate or, in some instances, due to the cytologist being unavailable to interpret the findings at the time of the biopsy.
Fine needle aspiration biopsy and core biopsy often complement each other in facilitating and assisting in the diagnostic process.
There have been continuous improvements in needles, biopsy guides and mechanical biopsy devices, together with technological advances in the major imaging methods of computed tomography (CT) and ultrasonography (US). The use of magnetic resonance imaging (MRI), and the development of stereotactic guidance, particularly for brain and breast biopsies, is now more readily available. Previously inaccessible lesions can be safely sampled and many more areas of the body are now routinely biopsied under guidance. Radiological guidance has allowed the development of more invasive procedures such as catheter drainage, villus biopsy, fetal blood and tissue sampling and core biopsy.4 This leads to a reduction in open biopsy and two-stage surgical procedures by providing a definitive diagnosis prior to primary surgical treatment.
More than one imaging modality may be required, first to localize the lesion and then to obtain biopsy material. The radiologist performing the procedure should determine the method used and will be influenced by availability of equipment, difficulty in scheduling, urgency of the procedure and perhaps operator preference and experience. Ultrasound guidance offers flexibility and speed, whereas CT often provides safer access for deeper tissue biopsy. Ultimately, the imaging modality which offers the best lesion visualization and safest route will dictate the biopsy pathway. Fluoroscopy is an alternative which may be utilized for pulmonary and bone lesions, although not as widely used as CT guidance.
All the imaging techniques have advantages and disadvantages in various parts of the body.
The portability, ease of use and relative speed of ultrasound make it a favourable modality for guided biopsy procedures, particularly for superficial and moderately deep lesions. The utility of ultrasound guided biopsy has also been recognized by specialties outside of radiology, particularly for intraoperative guided lesion assessments and biopsy.
Where practical, ultrasound is the preferred biopsy option, particularly given there is no ionizing radiation. In many instances, however, the nature and position of a lesion may mandate the use of CT guidance. Modern CT equipment allows for real-time assessment of the needle position using CT fluoroscopy, streamlining the biopsy process. This pathway involves ionizing radiation, exposing both the patient and the interventional team. The increasing body mass index of the patient population, particularly within the Western world, has led to a progressive increase in the imaging dose required to visualize relevant structures and organs. The end result is an increased radiation dose to the patient and to the interventional team members. An elevated awareness of this issue has motivated the implementation of dose minimization strategies where practicable.
Ultimately, however, the modality used will be dictated by the availability of equipment, staff and site of the lesion.
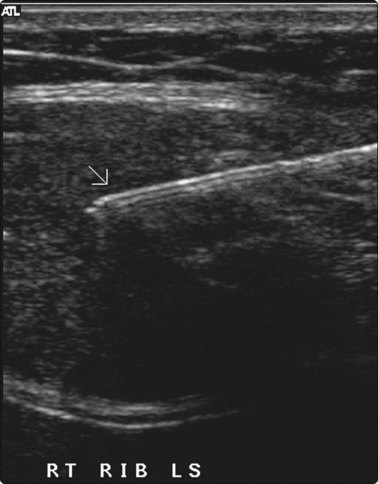
The presence of a pathologist at the biopsy generally facilitates a more efficient process. Optimal results can only be obtained by meticulous localization before biopsy and this may occupy most of the procedural time; fortunately, this can be performed prior to the arrival of the pathologist (Fig. 3.1). The pathologist may direct the radiologist to a different area of the lesion, for example to the viable periphery rather than the central necrotic area of a solid lesion, and may request additional tissue for ancillary tests, such as special stains, electron microscopy or culture, or to determine the need for a core biopsy.5
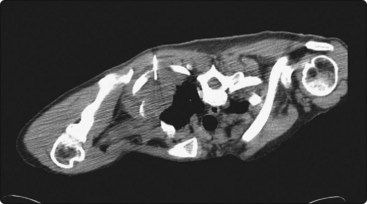
Fig. 3.1 Pancoast tumour biopsy
Care is required in positioning the patient for maximum comfort and access to the lesion to avoid ribs and scapula.
It matters little whether the radiologist or pathologist performs the actual aspiration. In many cases, however, the radiologist is more skilled in interpreting the images and is better able to manipulate the needle in three dimensions while viewing a two-dimensional image.
Fluoroscopy
Fluoroscopy is the traditional method for guidance of biopsies to most parts of the body. With the advent of highly technical guidance methods such as CT and US, it is used less and is often considered less accurate. However, it provides a quick alternative for those radiologists not experienced in US guidance and is most useful in guidance for small, very mobile lesions, such as focal lower zone lung lesions. In this instance, the real-time fluoroscopy may be the only method of accurately placing the needle tip within a small lesion. It also offers efficient sampling options for cortical bony lesions. Fluoroscopy is a reasonable biopsy alternative because of its low cost and general availability.
Although fluoroscopic guided biopsy may be easier with a biplane system, a single-plane system with tube tilting to provide a stereotactic type of view of the lesion and needle can also be used.
Ultrasound
Ultrasound (US) is the only real-time guidance which allows imaging in any plane and is the only suitable guidance for biopsy of fetal tissues. Its use is limited in certain areas, as ultrasound is not transmitted through air or bone. Some parts of the body,6 such as the chest wall and musculoskeletal system, though neglected in the past, have undergone an increase in interest for both diagnostic and interventional studies. Developments such as operative probes and vaginal and rectal transducers are now combined with portable, handheld (as compared to mobile) US units, for use in intensive care areas and operating theaters, wards and clinics, and to regional and remote areas.7
Real-time monitoring is a major advantage as the exact location of the needle tip can be seen during biopsy and adjustments to its position can be performed to increase the accuracy of sampling. Visibility of needles can be a problem and needles should be tested for echogenicity prior to use. Many manufacturers also provide fine needle aspiration biopsy needles with etched tips to aid in ultrasound visualization. Stylets within needles, and particularly movement of the stylet within the needle, will improve visibility. The gauge of the needle does not necessarily relate to echogenicity and in many instances a fine needle may be more highly echogenic than a subsequent core biopsy needle. Using color Doppler may enhance visibility during movement of the needle.
The choice of transducer and frequency is dictated by the area of the body to be biopsied and depth of the lesion. Intracavitary probes and developments with intravascular and intraluminal transducers allow biopsy and intervention into virtually every part of the body.
While freehand guidance is usually preferred, if a biopsy attachment is used there should be easy separation of the needle to reduce the risk of tearing tissues, particularly in areas of the body where respiratory movement may occur, for example liver and kidney. The shortest puncture route is normally chosen, though as with hepatic biopsies it is advisable that the needle traverses at least a rim of normal parenchyma to reduce the risk of hemorrhage.
While sterile water can be used as a coupling medium, there appears to be no real risk of reaction from use of sterile coupling medium, but it must not contaminate the aspirate. Sterilization of the transducer and attachment is routine, including universal precautions against infection.
Technique of ultrasound guided biopsy
The longest part of the examination is preparation – identifying the lesion, positioning the patient, and identifying the site of puncture and direction of the needle. The depth of the lesion from the skin surface is measured to determine the length of the needle required; the biopsy route is typically oblique and thus measurements need to reflect this. Continuous real-time ultrasound is used to visualize the tip of the needle entering the lesion. Scanning continues during aspiration to ensure that the needle stays within the lesion, and the aspirate may be seen moving within the needle lumen.
Maintaining sterility
Sterility of the transducer can be achieved by wiping the transducer with skin preparation, placing a layer of gel onto the end of the transducer, and carefully placing it into a sterile bag (sterilized plastic wrap, specially manufactured covers held there by a sterile rubber band), maintaining sterility at all times. Care must be taken with the transducer cord to prevent it draping over the sterile area. If coupling gel is used it should be sterile, but an alternative to gel is to use sterile saline or skin disinfectant, although these require occasional replenishment as the alcohol or water rapidly dries on the skin. The tip of the transducer merely needs to be dipped into the fluid, and enough will usually adhere to provide adequate transmission of sound waves. Coupling agents such as betadine or chlorhexidine solution may be preferred over sterile gel in some instances, as this may contaminate samples and compromise sample quality.
Biopsy procedure
The transducer is ideally held in the optimal longitudinal position to visualize the lesion. Prior to insertion of the needle, it is imperative that the alignment of the transducer is checked, so that the image on the screen is aligned with the correct orientation. Optimally, this is done by tilting the transducer slightly up and down, and assessing the areas brought into view, but should be confirmed by pressing lightly on the point of proposed skin puncture by the needle, so that the visible disturbance of the soft tissues confirms correct orientation.
Some operators have an assistant holding the transducer, maintaining its position exactly parallel to the long axis of the lesion, and ensuring that it does not slip away from the puncture site. This allows the operator to concentrate on introducing the needle exactly parallel to the transducer, and is a useful technique particularly in complex biopsy cases. Some prefer to hold both and relinquish the transducer once the needle has punctured the lesion. Only practice will determine which method is more comfortable for each individual.
Under real-time guidance the needle is introduced through the skin, 1–2 cm proximal to the transducer, and at the midpoint of the narrow side of the transducer. The needle is then advanced along the line of the transducer. The length of the needle should become visible, and the tip is seen to puncture the front wall of the lesion, then inserted a few more millimeters. If the needle does not become visible on the screen, the transducer can be used to find it, or alternatively the needle alignment can be adjusted. The angle of the needle should be changed to lie parallel to the transducer, as it is only when the lesion and the needle are in line that a successful puncture can occur.
Some operators prefer to use the transducer in an axis transverse to the lesion, once again located just distal to the proposed point of entry. This is less accurate, and does not necessarily locate the tip of the needle, merely showing a portion of the shaft.
CT scanning
There are very few areas of the body which cannot be biopsied under CT control, and extremely small lesions can be sampled. Focal masses of several millimeters within the lung and skull base (Fig. 3.2) can be biopsied and retroperitoneal biopsies are limited only by availability of needles long enough to traverse the abdomen of large patients. Traversing with fine needles offers fewer risks compared with the larger-caliber needles.7 CT gantry tilt also further facilitates lesion access where appropriate.
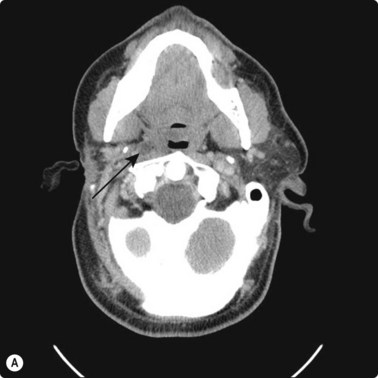
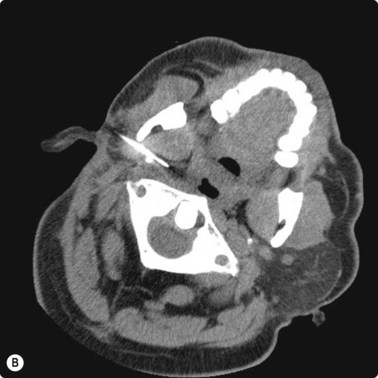
Fig. 3.2 (A) Circular low dense lesion right retropharyngeal node in a patient post right parotidectomy and radiotherapy for parotid squamous cell carcinoma. (B) Coaxial fine needle aspiration biopsy technique under CT control confirmed nodal recurrence. The utility of CT biopsy techniques and fine needle aspiration biopsy helped direct this patient’s management.
Localization of the needle tip within a lesion is very accurate with CT (Fig. 3.3). It provides detailed cross-sectional images of the body which are not limited by the same physical properties as are ultrasound images, such as interference from bowel gas and bone.
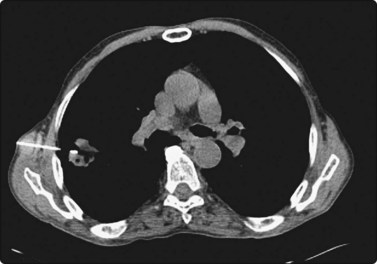
Fig. 3.3 Inflammatory lesion of the lung
Tissue is obtained for culture and other appropriate tests. Local anaesthetic is visible in the subcutaneous tissues.
Many of the CT scanners in modern imaging departments have biopsy software packages, allowing for real-time and relatively fast visualization of the target lesion and its relationship to the biopsy needle.
Successful biopsy of lung lesions is often dependent on the coordination of patient breath holding, CT fluoroscopic imaging and needle positioning. Confirmation of the location of the needle tip should be obtained prior to sampling. Extrapleural approaches to medially situated lesions, particularly lesions in the anterior mediastinal, subcarinal or paraspinal regions, avoids the traversing of aerated lung, thereby negating the potential for pneumothorax or air leak after transthoracic needle biopsy.8 When a paraspinal extrapleural approach is used, successive 10-ml aliquots of a mixture of equal saline and 1% lidocaine (lignocaine) are injected and intermittent scanning is performed to assess the needle route. Once a safe extrapleural route to the lesion has developed, a coaxial needle system or biopsy gun is advanced into the lesion for sampling.
The CT scans allow cross-sectional localization of needle placement.
The needle tip should be localized as accurately as possible to a position a few millimeters short of the area to be biopsied and the needle advanced the last few millimeters only during the biopsy. This prevents blood from accumulating around the needle tip and degrading the cytology specimen during the time required for scanning. Various techniques are available, including the use of guide or tandem needles and also stereotaxis. Artifacts from metallic needles and respiratory movement are rarely significant, particularly with the latest generation of CT scanners.
Magnetic resonance imaging (MRI)
It was initially predicted that MRI would never be suitable for guidance of biopsy and interventional procedures. However, the sensitivity of this new imaging technique is generally greater than that of other imaging methods and shows lesions which would not otherwise be detected. This is particularly evident in the brain, liver and breast.9 Biopsy needles with low ferromagnetic properties have been developed to allow biopsy under MRI control.
MRI guided biopsy has offered particular utility in the evaluation of breast lesions, particularly where they are not well visualized by other imaging techniques, or if the biopsy is technically difficult. It has extended the evaluation options for the ill-defined and subcentimeter mass, as well as those not well demonstrated by mammography or sonography.10 While MRI guided biopsy offers some logistical challenges, progress in devices and techniques are positioning this option more in the realm of mainstream lesion biopsy algorithms. Very fast scan times are also available, overcoming one of the earlier problems with MRI.
Dedicated interventional scanners are available, with easy access to the patient and lower field strengths to overcome anaesthetic and needle problems, yet still providing adequate resolution. Unfortunately, the expense of such dedicated scanners and their complex installation needs to restrict its availability to major clinical and research centers.
Breast biopsy and carbon marking for localization of clinically occult lesions
Mammographic screening has led to developments in biopsy and localisation of the small, clinically occult lesions detected by these programs. The most useful and common methods are ultrasound with and without a transducer attachment, a stereotactic attachment for upright mammographic X-ray units (Fig. 3.4) or a dedicated prone stereotactic mammographic biopsy table. When combined with FNAB and localization, either with a hookwire or preferably with carbon marking of the track,11 these techniques efficiently provide maximal information for the clinician with the minimum of inconvenience to the patient (Table 3.1).11 There is then the option to proceed to core biopsy if indicated by the FNAB result.
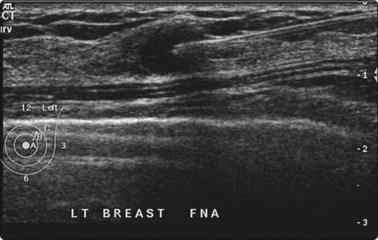
Fig. 3.4 Fine needle biopsy of a suspicious tumour mass in a small breast
Continuous visualization of the needle tip prevents the risk of pneumothorax.
Table 3.1 Advantages of carbon marking compared to hookwire localization
| Carbon marking | Hookwire |
|---|---|
| Accurate | Position less accurate |
| Permanent | May pull out or migrate |
| Comfortable for patient | Less comfortable for patient |
| Safe | Known risks, e.g. pneumothorax |
| Can be inserted at any time | Must be inserted immediately prior to surgery |
| Results of FNAB/core available prior to surgery | |
| Reduces two-stage surgical procedures | |
| Easily visible | Surgeons more familiar with technique |
| Radiologists easily trained | Surgeons require familiarization |
| Very inexpensive | Significant expense for hookwires |
Digital stereotactic localization, with either the upright or prone units, shortens the time of the procedure in comparison with a film-screen radiographic technique and gives rapid accurate localization. There are disadvantages to the prone units, particularly in cost and inability to utilize them for other mammographic purposes, and there is some restriction in positioning and access to deep lesions in the breast. These are overcome with some modern upright mammographic units which allow gantry tilt and, with a digital stereotactic attachment, can localize a breast lesion from any projection, including from the inferior aspect, with the patient lying on her side. This mitigates the problem of vasovagal attacks, one of the criticisms of the upright biopsy method. There is also much greater access to the breast with this method.
Many operators prefer US guidance for its speed, flexibility and real-time facilities. The patient is also able to lie comfortably during the procedure. The method is limited by the type of lesion; many microcalcifications are not visible on ultrasound examination. There is also the slight risk of pleural puncture or pneumothorax if used by inexperienced operators. The needle should run parallel to the chest wall and never be introduced perpendicular to the ribcage (see Fig. 3.4). By positioning the patient and compressing the breast, the depth of the lesion can be reduced. Breast lesions often require multiple sampling, typically best achieved using ultrasound guidance (Fig. 3.5).
If core biopsy is performed for a suspected malignant lesion, it is recommended that the needle track be resected during subsequent surgery to avoid the potential of tumor seeding the track. This surgery can best be guided by injection of carbon along a line parallel to and a few millimeters from the core biopsy track. The surgeon is then able to follow this to the lesion, without further localization being required.
Carbon localization of nonpalpable breast lesions
The introduction of breast screening programs in many parts of the world has been an impetus for more extensive use of aspiration biopsy, and the expertise and experience gained has increased the use of aspiration biopsy in all areas of the body. Until the introduction of carbon localization of occult breast lesions, hookwire localization under either mammographic (grid or stereotactic) or ultrasound localization was required. The use of carbon marking of the track from skin to lesion has many advantages11 (see Table 3.1) and and can almost always replace hookwire localization (Fig. 3.6).
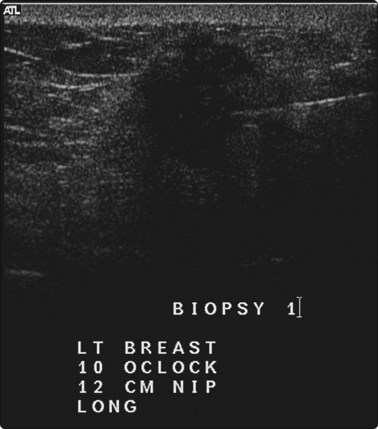
Fig. 3.6 Nonpalpable primary breast carcinoma
Ultrasound-guided fine needle biopsy and carbon marking of the needle track.
Carbon marking consists of injecting 1–2 mL of a sterile solution of 4% medical grade charcoal in normal saline through a 20G or 22G needle from the lesion to the skin surface, after the needle tip is localized exactly at the lesion, while gradually withdrawing the needle. A small skin tattoo remains to indicate to the surgeon the site for resection. The carbon solution is a suspension, not a solution, and occasionally the needle may block during injection due to a large particle of carbon. It is useful to draw the solution into a syringe at the start of the procedure, then leave the syringe lying flat, so that large particles will precipitate to the bottom of the barrel. The syringe should lie in that position during transport and injection, without agitation – sterile tubing allows this to occur for any localizing route. Heating or refrigeration of the suspension does not decrease the chance of a needle blocking with large particles of carbon, nor does limited or excessive agitation, although regular users of the technique may advocate these. A major advantage of carbon localization is that it can be performed routinely at the same time as aspiration biopsy, if there is any likelihood of surgical biopsy being required. The track of the biopsy is then marked for resection, decreasing the risk of implantation of malignant cells. The results of fine needle biopsy (and, if indicated, core biopsy), are known prior to a decision on surgery, and if the lesion is considered benign the carbon track can remain indefinitely, unlike hookwire localization where surgery is required to remove the hookwire. When core biopsy is performed, the carbon track is placed several millimeters to the side of the biopsy track, so that it persists after the hematoma due to the biopsy has subsided. If surgery is performed soon after a core biopsy, the surgeon may not need to visualise the carbon track, as extensive hemorrhage caused by a core biopsy will guide the resection. If the decision is made that surgery is not required, the skin tattoo and track marking can remain permanently with no ill effects.
One of the many advantages of carbon localization is that lesions which are only visible in the cranio-caudal view on mammography, but which lie in the inferior part of the breast, can be marked by carbon injection from the lesion to the inferior skin (‘push through technique’), avoiding a wire traversing the bulk of the breast, and preventing a long dissection or a cut-down to the wire (Fig. 3.7)
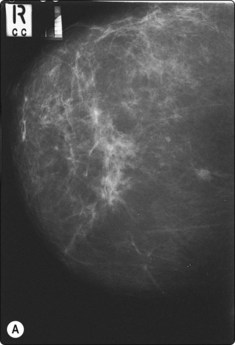
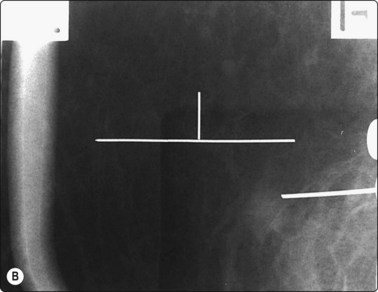
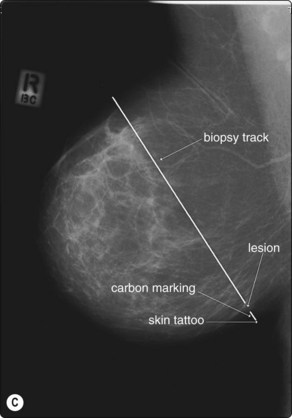
Fig. 3.7 ‘Push through technique’ of carbon localization of a clinically occult breast lesion. (A) C–C view of an inferior breast lesion (proven to be a primary carcinoma, < 1 cm), biopsied and carbon localized by the ‘push through technique’. (B) Stereotactic confirmation of the tip of the needle adjacent to the lesion is essential prior to aspiration and carbon track marking. (C) Diagrammatic representation of ‘push through technique’. Stereotactic biopsy in a C–C projection is performed, then a track is marked from the lesion to the inferior skin surface, producing a skin tattoo marking the site for surgical resection.
Use of guide needles
A guide needle may range from a short 2–3-cm needle to 12-cm needle, through which a fine-gauge needle is passed in a coaxial method (Fig. 3.8). The gauges of the needle combinations are usually 18/22 or 22/26. Use of these needles has been advocated during guided techniques for the following reasons:6
Risks and complications
Complications depend on the site of the lesion.
The main complications for CT guided lung biopsy include hemoptysis and pneumothorax.6,12 In particular, the rate of pneumothorax following lung biopsy with CT guidance reported in the literature ranges from 10% to 60%.2,12,13 The various items which may influence this rate include patient factors (age, sex, lung function, and presence of emphysema), lesion variables (size, depth, location, and pleural contact), and procedure related factors (experience of the operator, degree of difficulty, and type of needle used) (Fig. 3.9). Where possible, an extrapleural route for biopsy is preferred. The reported incidence depends on the method used to detect such a pneumothorax. Radiographs are routinely taken 4 hours after the procedure with expiratory films if there is any doubt. Patients with chronic lung disease are at much greater risk of pneumothorax complications.
The procedure can be safely performed on outpatients, but there should be adequate counseling to return if pain, dyspnea or significant hemoptysis occurs.
Soft tissue and solid organ biopsy risks may include hemorrhage, infection,7 tumor implantation,14 and occasionally major disability or death. In addition, liver interventions may develop a bile leak, and contribute to a bile peritonitis, typically mild. Skull base biopsy procedures may cause neuropraxia or vascular trauma. Nevertheless, the benefits of fine needle biopsy, and the low risk compared to other diagnostic investigations including interventional imaging procedures and surgery, make it an invaluable tool in the investigation of any indeterminate lesion (Fig. 3.9).
There is always a risk of false-positive results (< 1%) and false-negatives (> 10% in lung lesions).3 This is especially important in benign lesions where the tissues may be non-specific and the biopsy result nondiagnostic. In these patients it is imperative radiographically to confirm that the needle tip is in the lesion during the actual aspiration. There can then be appropriate assessment of the cells obtained during discussion with the treating clinician. The importance of imaging and recording the position of the tip of the needle at aspiration is critical in certain situations, such as during adrenal mass biopsy, as the pathologist is unable to distinguish between cells from a cortical adenoma and normal adrenal cortical cells. This may also apply to other benign neoplasms which are of small size and composed of cells similar to the normal tissue of origin.
Seriously unwell patients have greater morbidity and mortality from biopsy, and decreased ability to cope with complications.3 However, the alternatives to making a diagnosis often pose a greater risk. Communication with the clinicians is imperative to determine the need, relative risk and type of investigation required.
Fine needle biopsies of liver lesions, including hydatids and hemangiomata, are safe provided the needle passes through normal liver tissue to act as a seal (Fig. 3.10).
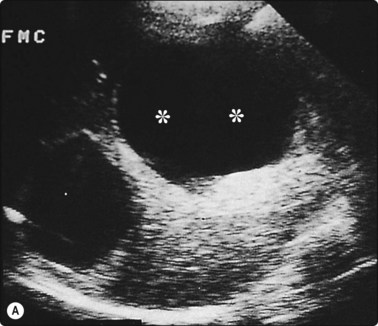
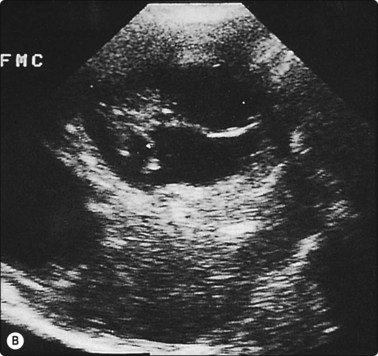
Fig. 3.10 Aspiration biopsy of hydatid disease without complication
(A) Two clearly defined anechoic cysts within the liver. The more anterior cyst (**) was completely aspirated without complication. (B) Five months later the patient was rescanned and multiple loculi within the cyst provided the diagnosis of hydatid disease. This was confirmed at surgery.
Fine needle biopsies in patients with very low platelets or poor bleeding and clotting studies are relatively safe, but are best done under real-time ultrasound control, preferably with minimal number of passes, and front wall puncture only, allowing for focal compression after the biopsy. The length of observation required after biopsy to determine possible complications varies with the site of biopsy, the patient’s clinical condition, prior risk factors (for example bleeding and clotting studies) and whether a responsible adult will be available at home to assist in return to the hospital should a complication occur. This should also be considered in rural and remote areas where the distance to hospital can be significant. All factors should be considered before determining if the procedure is safely performed as an outpatient examination, and a period of observation of 1–4 hours is usually adequate after most internal organ biopsies. These procedures should always be scheduled during the morning so that if complications arise, staff are available to treat them, and the period of observation is during normal working hours.
The overall mortality and morbidity related to FNAB have been estimated in many studies and the risk of death is approximately 1 : 15 000. This compares favorably with the more invasive studies which the technique replaces. Experience and the use of certain guidelines will reduce the risk of hemorrhage and spread of infection or tumor, especially in liver biopsies.
Ultrasound has considerable advantage in the upper abdomen because of the ability to guide biopsies in oblique planes. CT guidance is usually used in the axial plane to image the needle perpendicular to that plane, gantry tilt provides a practical and simple means of avoiding bone, pleura and other organs. It is particularly useful for spine and disc aspirates and in the upper abdomen.
Various contraindications to FNAB have been given in the literature. These include the risk of biopsy of pheochromocytomas, hydatid cysts (Fig. 3.11)15 and hemangiomas and of biopsy in the presence of ascites. The risks are considerably less than previously stated15 and ascites does not affect the risk of biopsy.
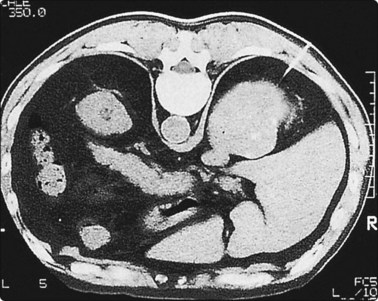
Fig. 3.11 Adrenal mass biopsy in a hemophiliac patient
The patient was a severe haemophiliac with only 2% of normal levels of factor VIII. As he was experiencing discomfort in the right upper quadrant, a cytological diagnosis was sought for the mass within the right adrenal. Biopsy was performed during several days of infusion of factor VIII. No complications occurred and the cytological diagnosis indicated old organising haemorrhage.
Regular audit of the results of FNAB will indicate the risk from all procedures performed in a department,2 and will indicate individual operators who may require review of their technique or indications in relation to excessive complications or nondiagnostic yield. This is an important part of quality assurance.
Pitfalls in aspiration biopsy technique
Occasional errors may include:
1 Nordenstrom B. A new technique for transthoracic biopsy of lung changes. Br J Radiol. 1965;38:550-553.
2 Cardella JF, Curtis WB, Bertino RE, et al. Quality improvement guidelines for image-guided percutaneous biopsy in adults. J Vasc Interv Radiol. 2003;14:S227-S230.
3 Grainger RG, Allison DJ, editors. Diagnostic Radiology – a textbook of medical imaging, 4th ed, vol. 2. London: Churchill Livingstone, 2001;1272.
4 Langlois S, Le P, Henderson DW, et al. Antenatal diagnosis of lamellar ichthyosis by ultrasonically guided needle biopsy of foetal skin. In: Gill RW, Dadd MJ, editors. Proceedings of 4th Meeting of the World Federation for Ultrasound in Medicine and Biology. Oxford: Pergamon Press; 1985:305.
5 VanSonnenberg E, Goodacre BW, Wittich GR, et al. Image-guided 25-gauge needle biopsy for thoracic lesions: diagnostic feasibility and safety. Radiology. 2003;227:414-418.
6 Ho LM, Thomas J, Fine SA, et al. Usefulness of sonographic guidance during percutaneous biopsy of mesenteric masses. AJR. 2003;180:1563-1566.
7 Langlois S, Le P. Portable ultrasound on deployment. ADF Health. 2003;4(2):77-80.
8 Langen Hi, Jochims M, Schneider W, et al. Distension of extrapleural spaces with contrast medium or air: value in creating safe percutaneous access to the mediastinumin cadavers. AJR. 1995;164:843-849.
9 Morris EA, Liberman L, Ballon DJ, et al. MR of occult breast carcinoma in a high-risk population. AJR. 2003;181:619-626.
10 Han B-K, Schnall MD, Orel SG, et al. Outcome of MRI-guided breast biopsy. AJR. 2008;191:1798-1804.
11 Langlois S, Le P, Carter ML. Carbon localisation of impalpable mammographic abnormalities. Australasian Radiol. 1991;35(3):237-241.
12 Geraghty PR, Kee ST, McFarlane G, et al. CT-guided transthoracic needle aspiration biopsy of pulmonary nodules: needle size and pneumothorax rate. Radiology. 2003;229(2):475-481.
13 Saji H, Nakamura H, Tsuchida T, et al. The incidence and risk of pneumothorax and chest tube placement after percutaneous CT-guided lung biopsy: the angle of the needle trajectory is a novel predictor. Chest. 2002;121:1521-1526.
14 Logan PM, Connell DG, O’Connell JX, et al. Image guided percutaneous biopsy of musculoskeletal tumours:an algorithm for selection of specific biopsy techniques. AJR. 1996;166:137-141.
15 Langlois S, Le P. Fine-needle biopsy of hepatic hydatids and haemangiomas: an overstated hazard. Australasian Radiol. 1989;33:144-149.
16 Moulton JS, Moore PT. Coaxial percutaneous biopsy technique with automated biopsy devices:value in improving accuracy and negative predictive value. Radiology. 1993;186:515-522.
17 Titton RL, Gervais DA, Boland GW, et al. Sonography and sonographically guided fine-needle aspiration biopsy of the thyroid gland: indications and techniques, pearls and pitfalls. AJR. 2003;181:267-271.