CHAPTER 18 Infectious diseases
Clinical aspects
Introduction
The widespread use of FNB for both palpable and impalpable lesions where the clinical and imaging differential diagnosis (DD) includes malignancy and sometimes infection has led to the diagnosis of infectious lesions. But with the increase in the number of immune compromised (IC) patients in developed countries, particularly due to the HIV epidemic and cancer therapy, FNB increasingly is used to assess skin, lymph node, lung and other sites where infection is the main diagnostic expectation.1,2 FNB can make the diagnosis of many specific infections on cytomorphology and special stains, and it also delivers material for the full array of ancillary studies including cultures, PCR and DNA analysis of other infections. The rapid diagnosis of infections, some of which cannot be cultured, can facilitate immediate treatment.
Now, with the gradual spread of FNB into the developing world where infections are the leading cause of morbidity and mortality, there has been a rapid if patchy increase in the use of FNB to make specific infectious diagnoses. FNB is minimally invasive, inexpensive, acceptable to patients, provides a rapid accurate diagnosis and immediate triaging for the most cost-effective selection of special stains and ancillary tests, and provides material for culture and the full range of ancillary tests. FNB is a very powerful diagnostic tool, especially where surgical resources and histopathological and microbiological laboratories are in short supply and grossly underfinanced.3 A cervical lymph node in a young adult that would, in subSaharan Africa, be treated expectantly as TB, can have an FNB that may show tuberculosis, metastatic carcinoma, lymphoma, reactive lymph node or branchial cyst. The FNB empowers the clinician to make the correct, often infectious, diagnosis, provides the best possible outcome for the patient and saves the medical system – which may be severely stressed by the AIDS epidemic, patient numbers and inadequate funding – the cost of surgical biopsy, hospital bed occupancy and inappropriate treatment
Place of FNB in the investigative sequence
The role of FNB is dependent on the body site and the facilities and expertise of personnel at particular institutions. In both the developed world and the four-fifths of the world where medical resources are limited, an inexpensive outpatient FNB can triage and, in the vast majority of cases, make a definitive diagnosis of any palpable lesion. FNB is the most efficient, least invasive and most cost-effective investigative procedure, particularly in cases where an infectious process is expected based on an integration of patient history and examination. This is exemplified by patients with suspected or known immune deficiency due to cancer, therapy or HIV disease.4,5
When dealing with imaging-detected deep lesions and impalpable superficial lesions, percutaneous FNB using ultrasound or CT guidance and endoscopic and endoscopic bronchial ultrasound-directed FNB are the mainstay of tissue diagnosis, supported when necessary by core biopsy. As interventional radiology, especially ultrasound, extends into the developing world, FNB of impalpable lesions will increase.
Specific sites
In the head and neck region after completion of a history and examination, which may include endoscopy of the upper aerodigestive tract, FNB of palpable lumps is the first line of investigation in all patients, especially the IC. The exact site of the lesion suggests the DD. In the preauricular, upper cervical and submental regions, acute or chronic sialadenitis,6,7 salivary gland neoplasms and a full range of lymph node lesions including reactive nodes, metastatic tumor, mycobacterial infection, lymphoma or other specific infection have to be considered. Rapid staining and immediate assessment of FNB slides is very powerful in this setting to direct selection of ancillary testing.
In the mid-cervical region, lymph node lesions including metastatic carcinoma, possible lymphoma, mycobacterial infections and non-specific infections are most common, but paragangliomas and large, often inflamed, tender and even fixed branchial cysts should always be considered.
In the lower cervical and adjacent midline region, lesions of the thyroid, lymph nodes and rarely the parathyroids form the differential diagnosis. Specific infections of the thyroid due to tuberculosis, viruses and, less commonly, Aspergillus spp, Candida spp, Histoplasma capsulatum, Cryptococcus neoformans, Coccidioides, Pneumocystis jirovecii blastomycosis and Pseudallescheria have been reported in increasing numbers.8 In the posterior triangle lymph nodes and schwannomas, and in the suboccipital area lymph nodes with specific infections including toxoplasmosis, and trichilemmal cysts and other skin lesions need to be considered.
In the axilla, femoral and inguinal regions lymph nodes may contain metastatic tumor, suppurative or granulomatous infections, non-specific reactive processes and lymphomas. Lymphogranuloma venereum and Granuloma inguinale should be considered in the groin.
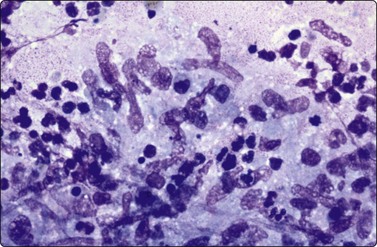
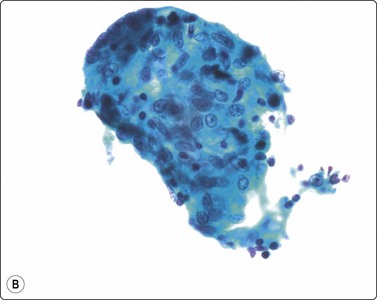
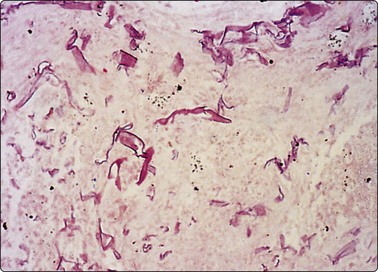
Skin nodules and palpable subcutaneous lesions are easily sampled by FNB. Pyogenic abscesses and infections such as tuberculosis9 are easily differentiated from tumor metastases, and, in the HIV population, from Kaposi’s sarcoma. Both superficial and deep fungal infections can be diagnosed by FNB and, especially in the IC, may represent disseminated fungal infection due to Aspergillus spp, Cryptococcus neoformans, histoplasmosis, blastomycosis,10 sporotrichosis and Candida spp,11 or one of the increasing number of other reported fungal infections producing subcutaneous nodules or mycetomas, including, Phialophera parasiticus, Sporothrix schenkii, Cladosporium spp, Exophiala jeanselmei12 and Microsporum canis.13 Actinomyces producing actinomycetomas, and rhinosporidiosis,14 filariasis,15 myaisis,16 leishmaniasis and leptosporosis all occur in the skin. The tuberculoid, borderline and lepromatous variants of leprosy, as well as neuritic and cystic lesions, can all be diagnosed.17 FNB of cellulitic skin to diagnose surgical wound skin infections18 and Buruli ulcers19 by direct smears and culture have been recommended. Skin nodules due to myaisis are rare but eminent FNB targets (Fig. 18.1).
Dipteran larva in background of pus from 23-gauge FNB of skin abscess (HE, ×600).
(Courtesy Dr Peter Pogany, Budapest, Hungary)
FNB without prior imaging is the first-line diagnostic approach for suspected breast abscesses yielding suppuration and a wide range of common bacteria and other agents including brucellosis20 and can assist in the drainage of the abscess. Cultures are mandatory. Repeat FNB is required if a significant palpable or ultrasound lesion remains after treatment because very occasional high-grade carcinomas can be associated with obscuring necrosis, neutrophilic infiltrate or multinucleated giant cells (MNGC) and a careful assessment of any inflammatory smear is required to ensure these unusual but rare carcinomas are not missed. Breast FNB may produce granulomatous material where mycobacterial and other infections must be excluded by culture.21
Lesions in the testis and scrotum including mycobacterial orchitis and epididymitis can be sampled by FNB to allow differentiation from neoplasms with no risk of a neoplasm seeding in the FNB track and jeopardising possible surgery.22
FNB of bone, soft tissue and skeletal muscle lesions,23 when palpable or under ultrasound direction, is a straightforward procedure and the diagnosis of osteomyelitis due to tuberculosis24 or other infectious agents can be made and confirmed on culture.25
FNB of deep and impalpable lesions found on imaging diagnostic work-up usually involve a DD of malignancy, and FNB is the most rapid, least invasive and inexpensive test available to diagnose specific infections by smears, special stains, cultures and other ancillary tests.
In most developed countries, FNB of central lung lesions, mediastinal, hilar, coeliac, peripancreatic and porta hepatis lymph nodes and pancreatic lesions is now performed under endoscopic ultrasound or endoscopic bronchial ultrasound direction. Percutaneous FNB of peripheral lung, liver and splenic lesions also yield infectious diagnoses, and may be the diagnostic test of choice, for example in paediatric peripheral lung lesions.26
In the liver, amebic and tuberculous abscesses, schistosomiasis27 and pyogenic abscesses due to fungi, staphylococcus, streptococcus, and Gram-negative bacteria including Klebsiella spp and Escherichia coli can be diagnosed. The use of albendazole and steroids as cover for the potential but very low risk of anaphylactic shock due to spillage of hydatid cyst contents during FNB should be considered if hydatid disease is suspected in an FNB of any site.28
In the pancreas, tuberculosis and infections diagnosed by EUS FNB have been increasingly reported in cases suspected of carcinoma.29 EUS-FNB also accesses the adrenal glands, where suspected metastases have been proven to be tuberculosis, cryptococcosis and disseminated histoplasmosis.30
In the lung, FNB is frequently used to distinguish tuberculosis or Gram-negative or -positive bacterial abscesses from carcinomas. Aspergillosis, mucormycosis,31 candidiasis and cryptococcosis, as well as, P.jirovecii in the HIV population, and CMV, coccidioidomycosis32 and parasites including paragonamiasis,33 amebiasis, hydatid disease and microfilariasis have all been reported.
In cases of splenomegaly and investigation of splenic lesions found on imaging, FNB is best performed under ultrasound guidance. Diagnoses include tuberculosis, lymphoma and other lesions but uncommon infections such as leishmaniasis should be considered.34
FNB of cerebral cortex lesions via burr holes can be used to diagnose toxoplasmosis, CMV and fungal infections35 in the DD with metastases and primary brain tumors.36
Choroiditis, uveitis and retinitis can be diagnosed by FNB performed by an experienced ophthalmologist, ideally using 25 or 27-gauge needles, and the fluid can be used for cultures, PCR and viral serology studies.
Technical considerations
Every FNB should be approached on the basis that there may be an infectious cause and that the patient may suffer from a transferable infection. The best guide is a good clinical history and examination. Every FNB operator should have, and teach, a single safe technique to use in all FNBs they perform. Gloves, and in cases of HIV patients double gloving, are recommended to cut down the potential of needle stick injuries. When removing the needle from the syringe to put air into the syringe once the FNB is finished, the hand that is to remove the needle should be brought down from the wrist of the hand holding the syringe holder, to the barrel of the syringe and then to the hub of the needle, rather than bringing this hand up to the needle point. Optical or safety glasses, gowns, uncluttered bench areas for placing needles and syringes during the procedure, and adequate clinic space are required.
When a cystic lesion or abscess is suspected it is recommended that a needle on a syringe should be used, creating a ‘closed’ system to avoid infective fluid spurting from the needle hub, and to assist in obtaining several milliliters of pus for culture. But using the needle by itself gives more sensitivity for FNB of palpable lesions, and can be used for a second pass of any residual mass after draining a cyst, or for solid and small lesions, especially skin nodules. Needles of 22 to 27 gauge are recommended for lymph nodes and skin lesions. If pus or necrosis is found and material is needed for extra slides, stains and cultures, a 22 gauge or 23 gauge will produce more material. The diagnostic yield of the cultures is higher with more material, so repeat needling using all the material for culture is recommended rather than just saline rinses of needle and syringe.37
The macroscopic appearances of the FNB smears may give clues to the infectious nature of the material, with pus having its own pale creamy green color, distinctive slippery feel on smearing and possible purulent odor. However, the best use of FNB in all situations is to have immediate slide smearing, staining and assessment by a cytopathologist or experienced cytotechnician, to confirm adequacy of material, provide a provisional diagnosis and to triage the case immediately for ancillary tests. In many cases, rapid diagnosis of a specific organism or the immediate diagnosis of an organism, such as P. jirovecii, that cannot be cultured, assists patient care, and facilitates the cost-effective selection of ancillary tests.
Ideally, both Giemsa and Papstain should be prepared routinely by specimen splitting of the material on the slide or separate FNB passes. Giemsa is better for bacteria, negative images of mycobacteria and fungal hyphae, yeasts and CMV cytoplasmic inclusions, and Papstain is better for some fungal hyphae and yeasts, and viral intranuclear inclusions. If infection is suspected or the immediate provisional reports suggest infection, extra air-dried slides for the auramine, ZN, Gram and methenamine silver stains (GMS) should be made immediately, and material taken for cultures, PCR, direct immunofluorescence (IF), flow cytometry and gene rearrangement studies (if lymphoma is a DD), and ultimately, in some organisms, phylogenetic analysis based on nuclear small subunit RNA sequence alignments. No single stain or protocol will diagnose all infections and culture. Destained H&E- or Pap-stained slides can be used for GMS and other special stains, and repeat FNB recommended.
A cell block should be prepared in almost all cases from saline rinses of the needle or ideally from separate needling, for H&E, special stains, immunoperoxidase studies (IP) and electron microscopy where required. In TB or HIV endemic areas or where the patient is thought to be IC, direct inoculation into culture bottles can be used.38 In cases such as liver abscesses where anaerobes are expected, pus should be placed directly into appropriate microbiologic containers. If the DD is lymphoma, especially in lymph nodes, lungs or spleen, FNB material should be placed into normal saline, Hanks medium or similar for flow cytometry and cytogenetic and gene rearrangement studies.
Immune compromised patients
FNB is extremely useful in the ever-increasing number of IC patients, including patients on long-term steroids, undergoing treatment for solid and hematologic malignancies, solid organ transplant recipients and HIV-positive persons.4,5 The successful role of FNB has continued into the era of highly active antiretroviral therapy (HAART).39 The typical inflammatory reactions to common infectious agents may be both deficient and atypical, and unusual and opportunistic infections may be present, making culture of FNB material mandatory.4 More than one infection may be present, for example CMV and bacterial infections in transplant patients.
Extra slides routinely should be prepared for Gram, auramine, ZN and GMS, and material taken for cultures for AFB, fungi and bacteria. A cell block should be prepared, and flow cytometry included in the work-up if lymphoma is suspected.
In these patients, suppurative lymphadenitis suggests not only a bacterial infection, but also disseminated fungal infection, most commonly Aspergillus spp, Cryptococcus neoformans or Candida spp infection. Granulomatous lymphadenitis raises the differential diagnosis of tuberculosis or possibly sarcoidosis. In Giemsa-stained smears from IC patients, mycobacterial infection may produce large numbers of plump histiocytes with cross-hatched cytoplasm representing ‘negative image’ bacilli, which may also be seen in the background serum. If neutrophils are admixed with spindle cells and histiocytes, bacillary angiomatosis should be considered, and the causative organism, Bartonella henselae and Bartonella quintana, demonstrated using Warthin-Starry stain.
Cytological findings
The diagnosis of infections by FNB occurs in the setting of FNB of a lesion suspected of malignancy where infection may or may not have been considered, or in the setting where infection is the leading provisional diagnosis based on clinical and imaging findings.
When a FNB yields suppurative or necrotic inflammatory material confirming the clinical and imaging findings, this is diagnostic, especially when special stains reveal a specific organism and this is confirmed on cultures or PCR. However, if a neoplasm is expected and pus or even granulomatous material is found, repeat FNB and further investigation such as core biopsy should be considered, for example an FNB of a mid-cervical mass producing bland squamous debris and suppuration consistent with a branchial cyst, or a FNB of a perihilar lymph node producing granulomatous material consistent with tuberculosis may be diagnostic of a benign infectious process, but FNB of a lung lesion producing necrosis with negative special stains and cultures usually warrants repeat FNB to exclude a necrotic squamous cell or other carcinoma.
Pyogenic infection in FNB is most commonly seen in lymph nodes in the head and neck and other regions in children, teenagers and young adults and in the lung, but suppuration is common in FNB from IC patients from all sites particularly the skin.
Criteria
Problems and differential diagnosis
The overdiagnosis of atypia or malignancy can occur when atypia is seen in epithelial components involved by the infectious process. These epithelial components may be infiltrated by neutrophils and commonly show enlarged hyperchromatic nuclei, which have predictable size, shape, chromatin pattern and nucleoli in each nucleus, rather than nuclear pleomorphism usually associated with malignancy. Attention should be given to the acute inflammatory setting in which the atypical epithelial components are found, as suppuration is not commonly associated with a primary malignancy of lung or breast. On the other hand, the diagnosis of malignancy can be missed in the setting of metastatic squamous cell carcinoma associated with necrosis and suppuration.
Suppuration is usually associated with Streptococcus pyogenes, Staphylococcus aureus, Streptococcus pneumoniae and Gram-negative bacteria including Klebsiella spp and Escherichia coli, as well as fungal infections such as Aspergillus spp, Cryptococcus neoformans and Candida spp, especially in IC patients.
In the mid-cervical region of the neck, a tender hot red mass raises a DD of inflamed lymph node, inflamed branchial cyst or possibly metastatic squamous cell carcinoma. The clinical site of the lesion, age of the patient and other history help to differentiate these lesions and repeat passes (up to 3 or 4) will usually provide an adequate sample. A diagnosis of malignancy should be made only in the presence of preserved squamous nuclei with malignant features, and not on pyknotic nuclei in degenerate squames in a suppurative or granulomatous background. Finally, a trial of antibiotic treatment with a repeat FNB of any residual mass may make the diagnosis. In the breast, squamous metaplasia in inflamed subareolar ducts (subareolar recurring abscess or Suska’s disease) can give rise to cytological atypia and worrying clinical signs of Paget’s disease or carcinoma. Correlation of imaging, clinical and cytological findings is essential.
In IC patients with neutropenia, a thin proteinaceous background may contain scant or no neutrophils and an organism must be sought with special stains and cultures. In AIDS patients, there may be plentiful plump histiocytes with cell debris and neutrophils resembling acute bacterial infections, but plentiful curved bacilli will be seen as negative images in the background and cross-hatched in macrophage cytoplasm (See Fig. 18.5). Culture and drug sensitivity testing is required.
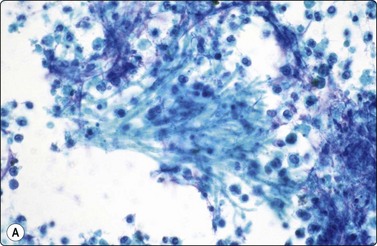
In fungal and cat scratch disease,40 suppurative granulomas with neutrophils interspersed among the epithelioid histiocytes in the granulomas may be present (Fig. 18.2).
Clinical
The commonest cause of granulomatous cytomorphology, and the diagnosis which must always be excluded, is that of mycobacterial infections. These are endemic worldwide and the prevalence of infections including atypical mycobacterial infections has increased with the HIV epidemic in almost all countries, especially India and sub-Saharan Africa. In developed countries, increased intercontinental travel, migration and increased numbers of IC patients of all types have fostered the resurgence of mycobacterial infections.
Criteria
Culture for fungi and mycobacteria with drug sensitivity testing is mandatory.
Problems and differential diagnosis
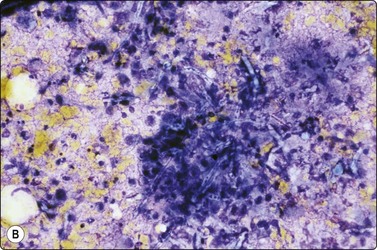
Mycobacterium tuberculosis infection may produce granulomas consisting of epithelioid histiocytes with or without Langhan’s MNGC and caseous necrosis, but in some cases may produce only caseous necrosis (Fig. 18.4). In IC patients, especially AIDS patients, FNB of lymph nodes may show neutrophils, a variable number of lymphocytes, plasmacytoid lymphocytes and plasma cells, depending on the stage of HIV lymphadenopathy. But where TB is endemic and HIV-positive patients present without the very low CD4 counts achieved in AIDS patients in developed countries in the era before HAART, typical granulomas can be seen.
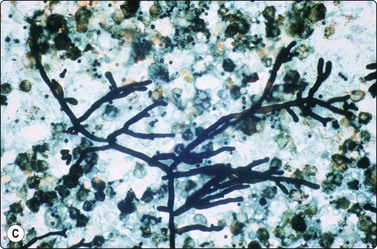
Negative images of mycobacteria may be seen in the background serum and cytoplasm of histiocytes in the Giemsa stain (Fig. 18.5), and the beaded 3–4-micron bacilli stain in the ZN (Fig. 18.6A) and auramine (Fig. 18.6B) stains and autoflouresce in the Papstain.41
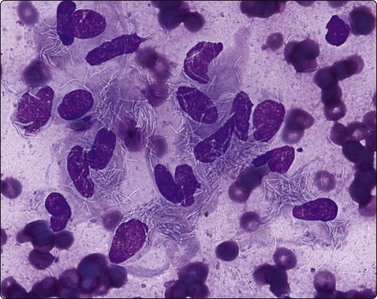
Fig. 18.5 Negative image of curved beaded atypical mycobacteria in histiocyte cytoplasm and serum background; note tendency for several bacilli to attach end to end (HIV-positive patient) (Giemsa, ×1000).
(Courtesy Dr William Geddie, Toronto, Canada)
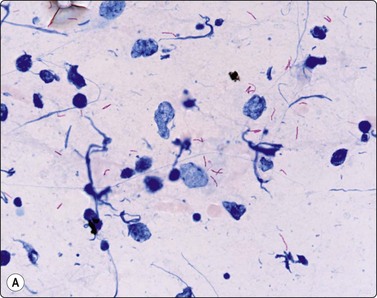
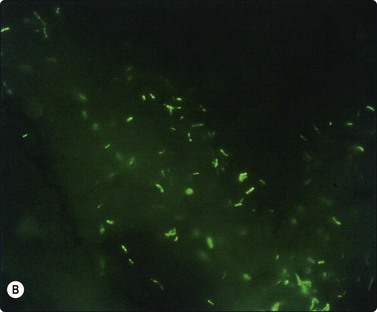
Fig. 18.6 (A) Curved beaded mycobacteria in Ziehl Neelsen stain (ZN, ×1000); (B) Mycobacteria in Auramine stain (Auramine, ×1000).
Caseating necrosis can be extensive in FNBs of lymph nodes and lung and may be the only material on the slides in M. tuberculosis cases, where the DD is necrotizing metastasis from SCC or a cavitating lung primary.
Mycobacteria may be very scant in FNB material, limiting the usefulness of AFB stains on direct smears and cell blocks (see below). The auramine IF stain allows rapid scanning of direct smears and multiple sections of cell block material, and PCR and ELISA stains increase the detection rate.
Sarcoidosis has distinctive rounded epithelioid granulomas infiltrated by lymphocytes without necrosis, but its diagnosis requires negative AFB stains, mycobacterial cultures and PCR with the correct clinical presentation and serology (Fig. 18.7).42
Fungal infections can be associated with granulomas and the yeasts and hyphae may be seen as negative images or variably stained in the Giemsa and Papstain depending on the specific fungus and the degree of degeneration of the hyphae. Cryptococcus may have no granulomas, especially in IC patients, and the 5–15-micron round yeasts may be seen within the cytoplasm of macrophages and MNGC or in the background with narrow-necked budding, a mucicarmine-positive and Giemsa-negative ‘halo-like’ capsule, and variably stained body (Fig. 18.8).43-45 In profoundly IC AIDS patients, there may be no capsule and the distinction from Histoplasma capsulatum relies on culture, as both stain with Giemsa. Histoplasmosis has largely intracellular smaller 2– 5-micron round yeasts showing variable budding in the GMS, and in Papstains appear as ‘holes’ in macrophage cytoplasm (Fig. 18.9).46-48
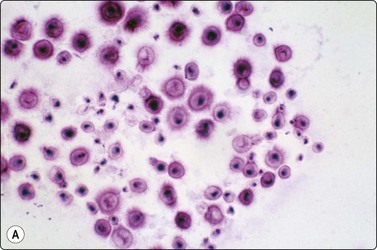
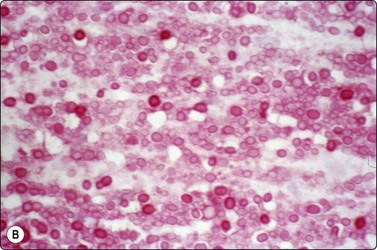
Fig. 18.8 (A) Cryptococcus yeasts with occasional narrow-necked budding and negative-staining capsule delineated by round body and surrounding serum (Giemsa, ×600); (B) Cryptococcus in mucicarmine stain with positive capsule and occasional narrow- necked budding (Muc, ×600).
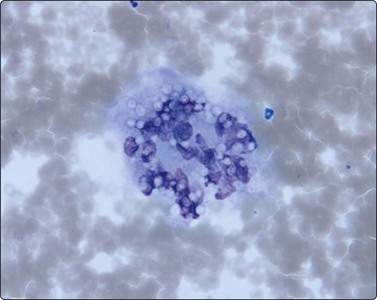
Fig. 18.9 Punched out ‘holes’ in cytoplasm of multinucleated histiocyte represent 3– 5-micron yeasts of Histoplasma capsulatum (Giemsa, ×600).
(Courtesy Dr William Geddie, Toronto, Canada)
In Southeast Asia and China, Penicillium marneffei, a rapidly emerging opportunistic infection in HIV-positive patients, has 2–4-micron yeasts in macrophages and sausage shaped yeasts in the extracellular background, showing septa in the GMS due to their division by binary fission, not budding.49,50 The budding yeasts of Sporothrix schenkii are cigar shaped (Fig. 18.10),51,52 and Paracoccidioides brasiliensis, endemic in Central and South America, has variably sized yeasts with pathognomonic multiple budding.33,53
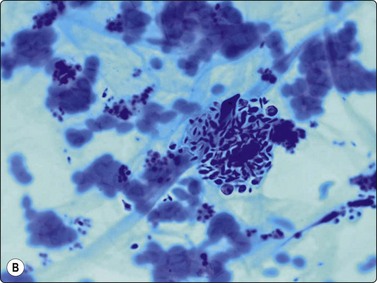
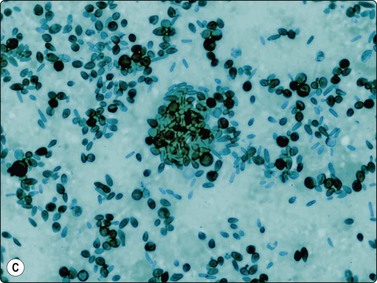
Fig. 18.10 (A, B, C) Sporotrichosis: oval and elongate yeasts in macrophages and in the GMS in the background (Pap, Giemsa, Grocott Methenamine silver, ×1000).
(Courtesy Dr Rene Gerhard, Sao Paolo, Brazil)
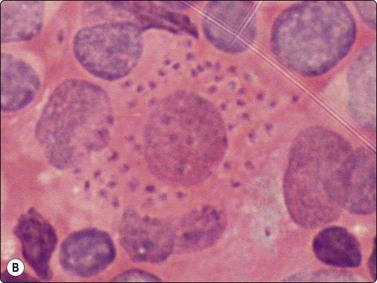
Blastomycosis produces large 8–20-micron yeasts with broad-based budding and a thick double-contoured, weakly birefringent wall featuring inner and outer wall staining (Fig. 18.11).8,10,54 Blastomycosis shares a distribution with coccidioidomycosis which has much larger 20–120-micron refractile, thick-walled spherules, some of which contain 2–4-micron endospores (Fig. 18.12),32 which have to be differentiated from leishmaniasis, whose amistigotes contain nuclei and kinetoplasts (Fig. 18.13), and the crescentic tachyzoites containing dot nuclei of toxoplasmosis (Fig. 18.14A). In lymph node FNBs, toxoplasmosis is associated with follicular hyperplasia and discrete small syncytial epithelioid granulomas (Fig. 18.14B), and the tachyzoites in cysts and the background are rarely seen. Confirmatory diagnosis can be made by serology.
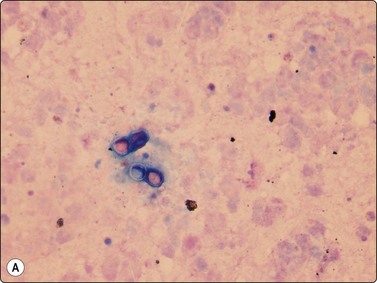
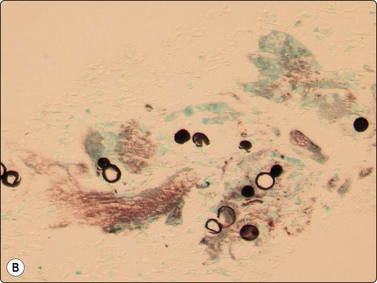
Fig. 18.11 (A, B) Blastomycosis: 10– 12-micron yeasts showing broad based budding (Giemsa and GMS, ×1000).
(Courtesy Dr David Carter, USA)
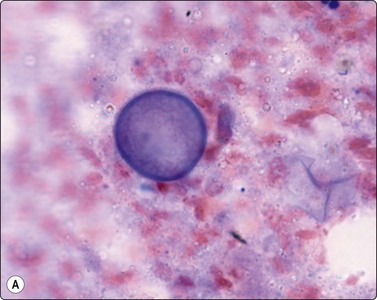
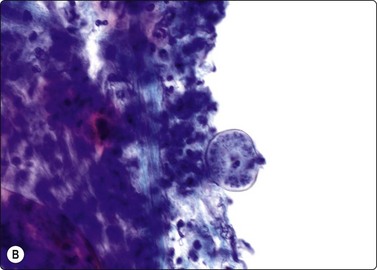
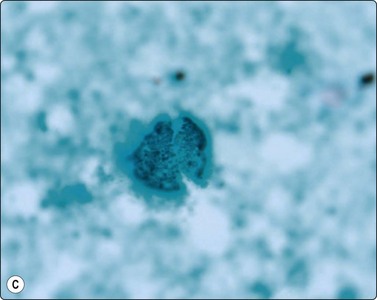
Fig. 18.12 (A, B, C) Coccidiodomycosis: 60-micron spherule with faintly birefringent wall containing 2–4-micron endospores (Giemsa, Pap, GMS, ×1000).
(Figures 12A, 12C Courtesy Dr William Geddie, Toronto, Canada; Figure 12B Courtesy Dr Matthew Zarka, Scottsdale, Arizona)
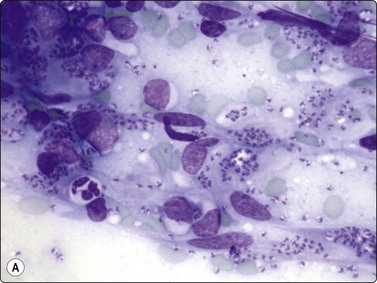
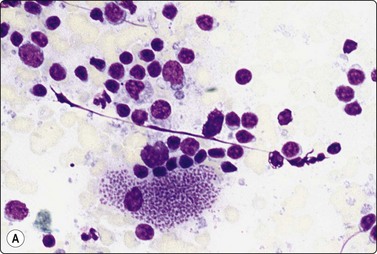
(A) Leishman-Donovan bodies in background and in macrophage cytoplasm (Giemsa, ×400); (B) Leishman-Donovan bodies in macrophage cytoplasm at high power (Pap, ×1000).
(Courtesy Dr Khosrow Daneshbod, Shiraz, Iran)
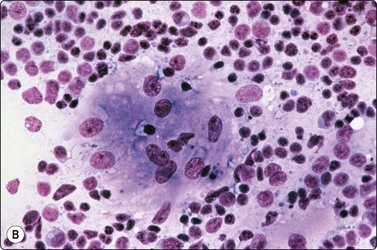
(A) brachyzoites in macrophage cytoplasm (Giemsa, ×600); (B) Syncytial epithelioid minute granuloma in reactive lymphoid background.
Candida spp have thin, branched pseudo-hyphae and 3– micron yeasts,55 while Aspergillus spp have 5–7-micron diameter septate hyphae showing acute angle branching (Fig. 18.15)56,57 and Mucormycosis has thicker, irregular 2– 50-micron, ribbon-like nonseptate hyphae and variable but often 90 degree branching (Fig. 18.16).32 Aspergillosis can be associated with birefringent needle-like calcium oxalate crystals58 and marked metaplastic squamous atypia in the margins of cavitating lung lesions.
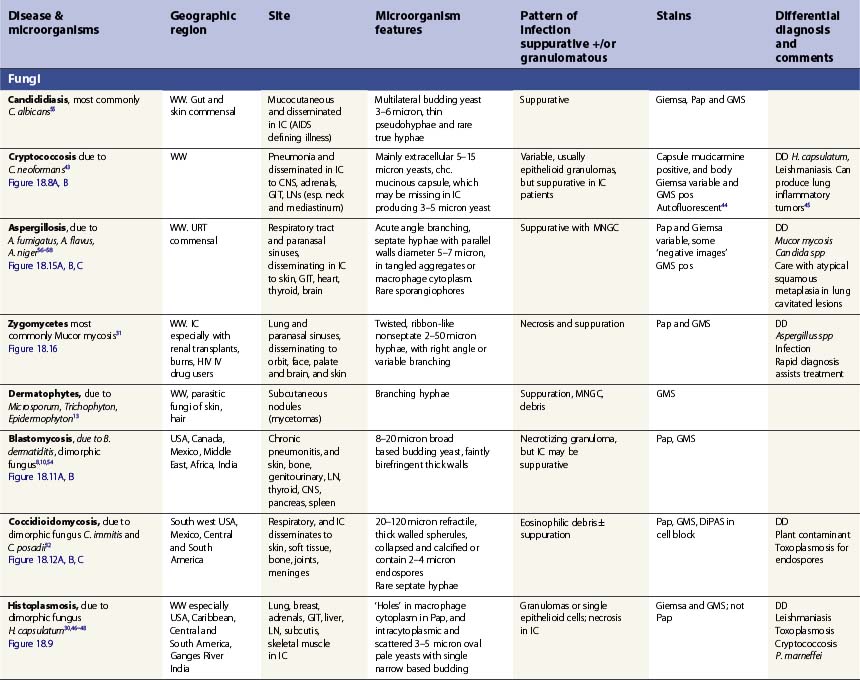
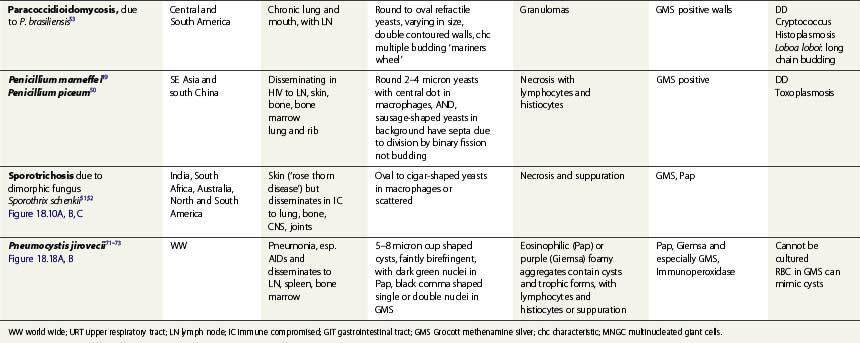
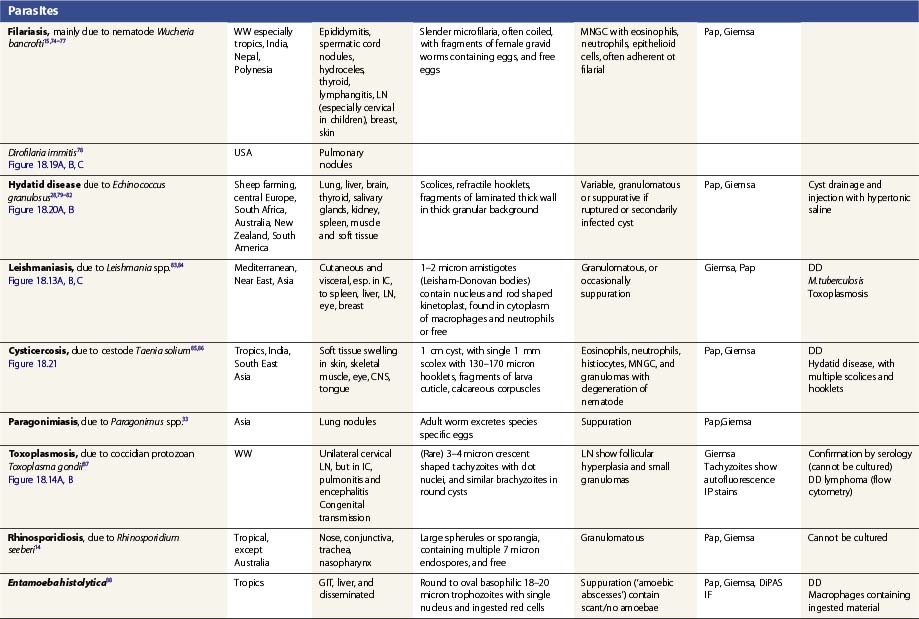
Specific infections are described in Tables 18.1 and 18.2, which emphasize the morphological diagnosis in direct smears and special stains. For a more detailed discussion of their microbiology, please refer to standard texts.59
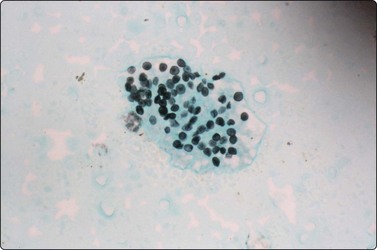
(A) granular foamy aggregate of 5-micron cup-shaped cysts (Pap, ×400); (B) Aggregated cup-shaped cysts with dot nuclei (GMS, ×400).
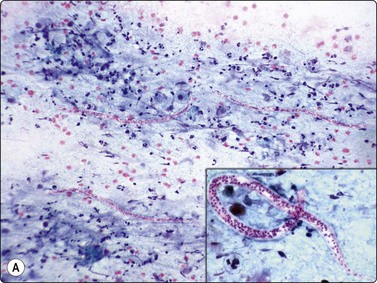
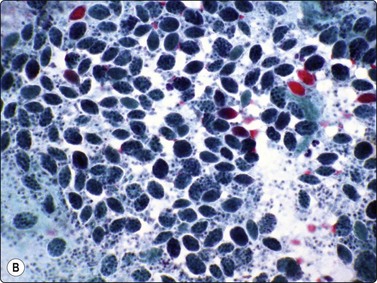
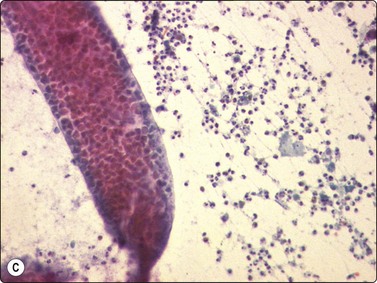
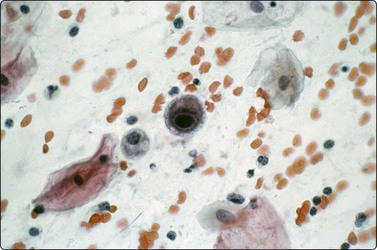
Fig. 18.19 Filariasis due to Dirofilaria repens
(A) With multiple microfilariae in background of neutrophils, with blunt heads and tapered rear ends (insert) (Pap, ×200); (B) Aggregated eggs (Pap, ×400); (C) Adult gravid female in background of eosinophils, from FNB of ‘malignant’ breast lesion (Pap, ×200).
(Figures 19A, 19B Courtesy Dr Khosrow Daneshbod, Shiraz, Iran; Figure 19C Courtesy Dr Roy, Mumbai, India)
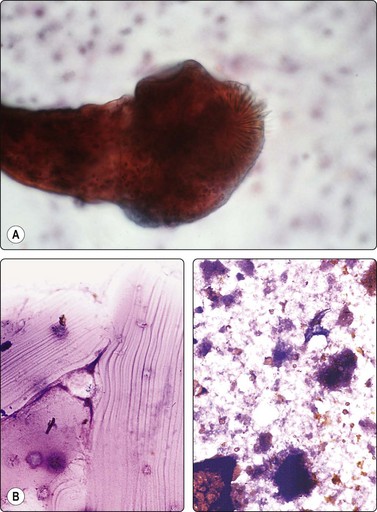
Fig. 18.20 (A) Hydatid scolex, showing hooklets and sucker (Pap, ×1000); (B) Hydatid laminated membrane. (C) Hooklet (Pap, ×400).
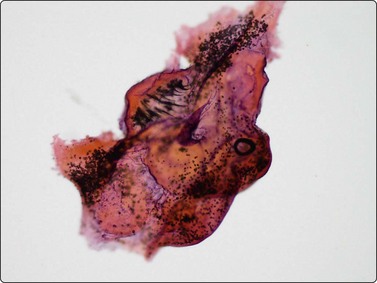
Intact larva including scolex with hooklets (sickle shaped) and attached wall fragment (HE, ×200).
(Courtesy Dr Harsh Mohan, Chandigash, India)
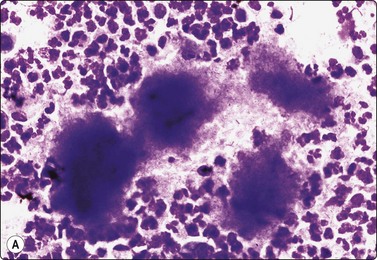
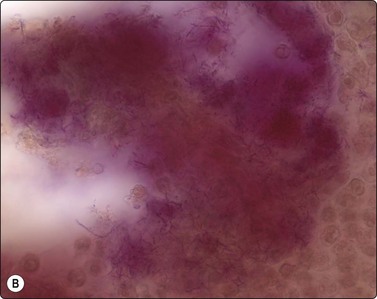
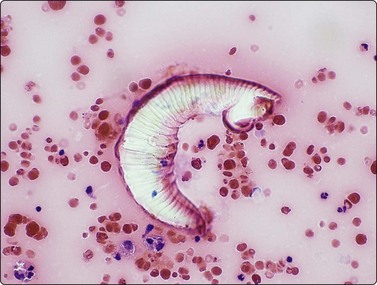
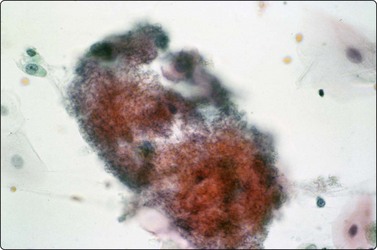
(A) Aggregated filaments which can form macroscopic “sulphur granules” amid neutrophils (Giemsa, x400); (B) Aggregated filaments (Gram, ×1000).
(Figure 22B Courtesy Dr William Geddie, Toronto, Canada)
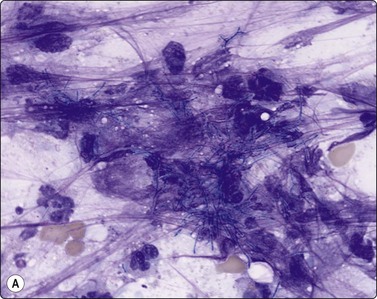
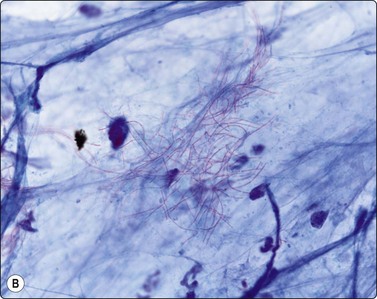
(A) Filaments in background of pus (Giemsa, ×1000); (B) Filaments (ZN, ×1000).
(Courtesy Dr William Geddie, Toronto, Canada)
Mycobacterial infections include M. tuberculosis, M. avium complex, M. leprae and M. ulcerans (Buruli’s ulcer).
Tuberculosis is endemic across most countries, and with the increase in IC patients and the HIV epidemic there has been a resurgence of TB in the developed world and the development of multi-resistant strains.60 FNB of all sites offers a rapid, inexpensive and efficient outpatient clinic diagnostic method based on cytomorphology (see Figs 18.3-18.5) and the Ziehl-Neelsen (see Fig. 18.6A) and auramine (see Fig. 18.6B) stains, and the FNB delivers material for culture in traditional Lowenstein-Jensen or other media or PCR or immunohistochemistry.38,61 This is highlighted by the role of FNB in children with cervical lymphadenopathy.41
Extrapulmonary TB most commonly presents with head and neck lymphadenopathy where the DD is often malignancy – for example, intraparotid lymph nodes can present as a neoplasm – and in the skin,9 bone,24 vertebral and paravertebral regions,23 testes and epididymis22 and pancreas.62 Mycobacterium avium complex is the commonest nontuberculous mycobacterium and the commonest cause of cervical lymphadenopathy in immune competent children, and may disseminate from lungs and gut to involve lymph nodes and skin in HIV-positive patients.63
Positive cytology is usually defined as granulomas, MNGC, caseation and mycobacterial visualization, but there is a wide range of cytomorphological patterns on smears varying from the classic features of epithelioid granulomas with Langhan’s giant cells and caseous necrosis, to mostly or solely caseous necrosis, and to tuberculous abscess with plentiful neutrophils, degenerating epithelioid cells and a watery necrotic background. The cytology pattern and number of AFB on ZN stain depends on the patient’s immune status, and in cases where FNB produces only caseous necrosis the percentage showing AFB on the ZN stain may be as low as 7%.64 M. avium complex especially may yield histiocytes and some neutrophils in a watery background with a variable number of AFB in HIV-positive patients.63 Culture or PCR is mandatory for confirmation, speciation and drug resistance, but these tests are often not available in TB-endemic developing world areas, where there is reliance on demonstrating AFB.
Other techniques have been put forward including the modified bleach method for ZN staining,64 the IF auramine-rhodamine stain, immunostaining of direct cytological smears,65 PCR on DNA scraped and processed from dried unstained cytology smears,66 and the ELISA test,67 most of which can achieve positive rates in the range 70% to 85%.
But the combination of cytomorphology, ZN stain and autofluorescence on Papstain can achieve concordance with culture in around 70% cases,41 and culture remains the mainstay of diagnosis and drug sensitivity testing.61
Extrapulmonary TB in the breast presents as a mass, discharging sinuses, cold abscess or non-healing ulcers, raising a DD of carcinoma. AFB stains and cultures, however, may not be positive, and other causes of granulomatous mastitis such as Brucella melitenses, fungi and organizing fat necrosis should be excluded by cultures and PCR.20
FNB of the skin and other lesions of leprosy due to Mycobacterium leprae, which is endemic across the developing world, are highly diagnostic, especially if skin is blanched by gently lifting and squeezing the site to be aspirated to reduce bleeding and the Fite stain and cultures are used.17 The DD is cutaneous tuberculosis with its epithelioid granulomas and caseous necrosis.9 There is a spectrum of lesions:
The FNB can classify most cases to match the Ridley and Jopling histological classification of leprosy. Soft tissue swellings with nerve abscesses in neuritic leprosy show abundant caseous necrosis and many degenerate neutrophils in a thin proteinaceous background with a negative AFB stain.69
This neoplasm is caused by human herpesvirus-8 and is derived from infected endothelial cells. It is an AIDS-defining illness and one of the commonest neoplasms seen in homosexual AIDS patients, although in the HAART era, its incidence has decreased and been overtaken by lymphomas and squamous mucosal lesions. In lymph nodes, the spindle cell proliferation involves the subcapsular and other sinuses, and the remaining lymph node shows the various stages of HIV infection, from follicular hyperplasia through to involution. Nodal involvement should be considered in patients with tender lymphadenopathy, low CD3 count, associated infections and Kaposi’s sarcoma skin lesions.70
Criteria for diagnosis (Fig. 18.17)
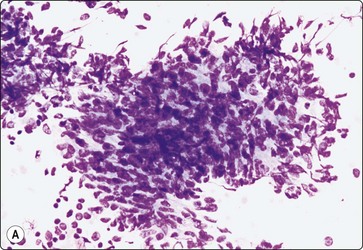
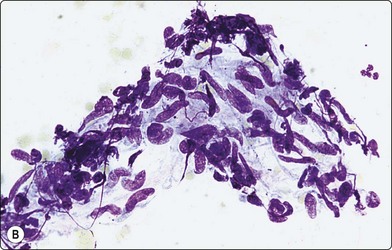
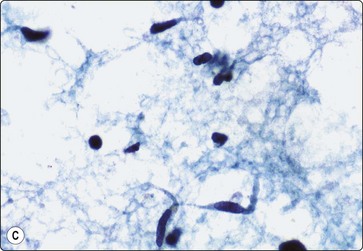
Fig. 18.17 (A) Tissue fragment of Kaposi’s sarcoma, showing crowded, spindle shaped cells with hyperchromatic, blunt-ended nuclei (Giemsa, x200); (B) Tissue fragment of spindle cells, with poorly defined cytoplasm Giemsa, x400); (C) Single spindle cells with cigar-shaped bland nuclei, and granular chromatin (Pap, x400) Kaposi’s sarcoma.
Problems and differential diagnosis
The tissue fragments may resemble granulomas, but the nuclei are more elongated and spindly and usually are hyperchromatic. They lack the ‘sand shoe’ indentations of epithelioid histiocytes, and MNGC are not seen. Mycobacterial infection may involve the same node. Occasionally, Kaposi’s sarcoma produces highly cellular smears with large tissue fragments containing slit-like spaces and hemosiderin. Mitoses are scant.
1 Silverman JF, Gay RM. FNA and Surgical Pathology of Infectious Diseases. Contemporary Issues in Clin Microbiol. 1995;15:251-278.
2 Powers C. Diagnosis of Infectious diseases: a cytopathologist’s perspective. Clin Micro Reviews. 1998;11:341-365.
3 Wright CA, Pienaar JP, Marais BJ. FNAB: diagnostic utility in resource limited settings. Annals of Trop Paed. 2008;28:65-70.
4 Jayaram G, Chew M. Fine needle aspiration cytology of lymph nodes in HIV infected individuals. Acta Cytol. 2000;44:960-966.
5 Vanisri HR, Nandini NM, Sunila R. FNAC findings in HIV lymphadenopathy. Ind J Path Microbiol. 2008;51:481-484.
6 Santiago K, Rivera A, Cabaniss D, et al. FNA of CMV Sialadenitis in a patient with AIDS; pitfalls of Diff-Quik Staining. Diagn Cytopathol. 2000;22:101-103.
7 Michelow P. Infective parotitis Editorial. Acta Cytol. 2006;50:601-602.
8 Wineland A, Siegel E, Francis C, et al. FNA diagnosis of thyroid blastomycosis. Endocr Pract. 2008;14:224-228.
9 Kathuria P, Agarwal K, Koranne RV. The role of FNAC and ZN staining in the diagnosis of cutaneous tuberculosis. Diagn Cytopathol. 2006;34:826-829.
10 Shukla S, Singh S, Jain M, et al. Paediatric cutaneous blastomycosis: a rare case diagnosed on FNAC. Diagn Cytopathol. 2009;37:119-121.
11 Das R, Dey P, Chakrabarti A, et al. FNAB in fungal infections. Diagn Cytopathol. 1997;16:31-34.
12 Gabhane SK, Gangane N, Anshu. Cytodiagnosis of Eumycotic Mycetoma. A case report. Acta Cytol. 2008;52:354-356.
13 Barboza-Quintana O, Garza-Guajardo R, Assad-Morel C, et al. Pseudomycetoma for Microsporum canis. Report of a case diagnosed by FNAB. Acta Cytol. 2007;51:424-428.
14 Deshpande AH, Agarwal S, Kelkar AA. Primary cutaneous rhinosporidiosis diagnosed on FNAC: a case report with review of literature. Diagn Cytopathol. 2009;37:125-127.
15 Kumar B, Karki S, Yadava SK. Role of FNAC in diagnosis of filarial infestation. Diagn Cytopathol. 2010.
16 Pogany P, Szucs E, Lichtenberger G, et al. Diagnosis of myaisis by FNAC. A case report. Acta Cytol. 2008;52:228-230.
17 Mehdi G, Maheshwari V, Ansari HA, et al. Modified FNA technique for diagnosis of granulomatous skin lesions with special reference to leprosy and cutaneous tuberculosis Diagn Cytopathol 2009; 26.
18 Parikh AR, Hamilton S, Sivarajan V, et al. Diagnostic FNA in postoperative wound infections is more accurate at predicting causative organisms than wound swabs. Ann R Coll Surg Engl. 2007;89:166-167.
19 Eddyani M, Fraga AG, Schmitt F, et al. FNA, an efficient sampling technique for bacteriological diagnosis of nonulcerative buruli ulcer. J Clin Microbiology. 2009:1700-1704.
20 Nemenqani D, Yaqoob N, Khoja H. Breast brucellosis in Taif, Saudi Arabia: cluster of six cases with emphasis on FNA evaluation. J Infect Dev Ctries. 2009;3:255-259.
21 Nemenqani D, Yaqoob N, Hafiz M. FNAC of granulomatous mastitis with special emphasis on microbiologic correlation. Acta Cytol. 2009;53:667-671.
22 Sah SP, Bhadani PP, Regmi R, et al. FNAC of tubercular epdidymitis and epididymo-orchitis. Acta Cytol. 2006;50:243-249.
23 Akhtar I, Flowers R, Siddiqi A, et al. FNAB of vertebral and paravertebral lesions. Retrospective study of 124 cases. Acta Cytol. 2006;50:364-371.
24 Handa U, Garg S, Mohan H, et al. Role of FNA in tuberculosis of bone. Diagn Cytopathol. 2009;38:1-4.
25 De Lucas EM, Gonzalez Mandly A, Gutierrez A, et al. CT-guided FNA in vertebral osteomyelitis: true usefulness of a common practice. Clin Rheumatol. 2009;28:315-320.
26 Levison J, Van Asperen P, Wong C, et al. The Value of CT guided FNA in infants with lung abscesses. J Paediatr Child Health. 2004;;40:474-476.
27 Chen HZ, Chuang SC, Hsieh FC, et al. Diagnosis of Schistosomiasis japonica infection coincident with hepatocellular carcinoma by FNA. Diagn Cytopathol. 2007;35:722-724.
28 Langlois S, Le P. FNA of hepatic hydatids and hemangiomas; an overstated hazard. Australian Radiol. 1989;33:144-149.
29 Somsouk M, Shergill AK, Grenert JP, et al. Actinomycosis mimicking a pancreatic head neoplasm diagnosed by EUS-guided FNA. Gastro Endoscopy. 2008;68:186-187.
30 Eloubeidi MA, Luz LP, Crowe DR, et al. Bilateral adrenal gland enlargement secondary to histoplasmosis mimicking adrenal metastases: diagnosis with EUS-guided FNA. Diagn Cytopathol. 2010;38:357-359.
31 Deshpande AH, Munshi MM. Rhinocerebral mucormycosis diagnosed by aspiration cytology. Diagn Cytopathol. 2000;23:97-100.
32 Raab S, Silverman JF, Zimmerman KG. FNAB of pulmonary coccidioidomycosis: spectrum of cytology findings in seventy three patients. Am J Clin Pathol. 99, 1993. 582–558
33 Vijayan VK. Parasitic lung infections. Current Opinion in Pulmonary Medicine. 2009;15:274-282.
34 Kumar PV, Monabati A, Raseki AR, et al. Splenic lesions: FNA findings in 48 cases. Cytopathology. 2007;18:151-156.
35 Amr SS, Al-Tawfiq JA. Aspiration cytology of brain abscess from a fatal case of cerebral phaeohyphomycosis due to Ramichloridium mackenzi. Diagn Cytopathol. 2007;35:695-699.
36 Baviatsis EJ, Kouyialis AT, Stranjalis G, et al. CT guided stereotactic aspiration of brain abscesses. Neurosurg Rev. 2003;26:206-209.
37 Granville LA, Laucirica R, Verstovsek G. Clinical significance of cultures collected from FNAB. Diagn Cytopathol. 2008;36:85-88.
38 Wright CA, Bamford C, Prince Y, et al. Mycobacterial transport medium for routine culture of FNABs. Arch Dis Child. 2010;95:48-50.
39 Lowe SM, Kocjan GI, Edwards SG, et al. Diagnostic yield of FNAC in HIV infected patients with lymphadenopathy in the era of HAART. Int J STD & AIDS. 2008;19:553-556.
40 Stastny JF, Wakely PEJr, Frable WJ. Cytologic features of necrotizing granulomatous inflammation consistent with cat-scratch disease. Diagn Cytopathol. 1996;15:108-115.
41 Wright CA, van der Burg M, Geiger D, et al. Diagnosing mycobacterial lymphadenitis in children using FNAB: Cytomorphology, ZN staining and autofluorescence- making more of less. Diagn Cytopathol. 2008;36:245-251.
42 Tambouret R, Geisinger KR, Powers CN, et al. The clinical application and cost analysis of fine needle aspiration biopsy in the diagnosis of mass lesions in sarcoidosis. Chest. 2000;117:1004-1011.
43 Gustafson KS, Feldman L. Cryptococcal lymphadenitis diagnosed by FNAB. Diagn Cytopathol. 2007;35:103-104.
44 Mathai AM, Rau AR, Kini H. Cryptococcal autofluorescence on FNA of Lymph node. Diagn Cytopathol. 2008;36:689-690.
45 Kushner YB, Brimo F, Schwartzman K, et al. A rare case of pulmonary inflammatory myofibroblastic tumour diagnosed by FNAC. Diagn Cytopathol. 2009:24.
46 Fitzhugh VA, Maniar KP, Kim MK, et al. Adrenal Histoplasmosis. Diagn Cytopathol. 2009;38:188-189.
47 Zafar N. Histoplasmosis in FNAC of axillary lymph nodes in an HIV-positive woman with clinical suspicion of lymphoma. Diagn Cytopathol. 2009;37:584-585.
48 Goel D, Prayaga AK, Rao N, et al. Histoplasmosis as a cause of nodular myositis in an AIDS patient diagnosed on FNAC. Acta Cytol. 2007;51:89-91.
49 Lim D, Lee Y-S, Chang AR. Rapid diagnosis of Penicillium marneffei infection by FNAC. J Clin Pathol. 2006;59:443-447.
50 Santos PE, Piontelli E, Shea YR, et al. Penicillium piceum infection: diagnosis and successful treatment in chronic granulomatous disease. Med Mycol. 2006;44:749-753.
51 Fontes PC, et al. Sporotrichosis in an HIV-positive man with oral lesions. A case report. Acta Cytol. 2007;51:648-650.
52 Gerhard R, de Moscoso PC, Gabbi TV, et al. FNAB of disseminated sporotrichosis: a case report. Diagn Cytopathol. 2008;36:174-177.
53 Gerhard R, Basso MC. Diagnosis of three cases of paracoccidioidomycosis by FNAC. Cytopathology. 2007;18:123-129.
54 Deutsch JC, Burke TL, Nelson TC. Pancreatic and splenic blastomycosis in an immune-competent woman diagnosed by EUS FNA. Endoscopy. 2007;39:E272-E273.
55 Prasad VM, Erickson R, Contreras ED, et al. Spontaneous candida mediastinitis diagnosed by endoscopic ultrasound guided FNA. Am J Gastroenterol. 2000;95:1072-1075.
56 Fischler DF, Hall GS, Gordon, et al. Aspergillus in cytology specimens; a review of 45 specimens from 36 patients. Diagn Cytopathol. 2001;16:26-30. 1997
57 Kumar Behera SK, Patro M, Mishra D, et al. FNA in Aspergilloma of frontal sinus. A case report. Acta Cytol. 2008;52:500-504.
58 Modem RR, Florence RR, Goulart RA, et al. Pulmonary Aspergillus associated calcium oxalate crystals. Diagn Cytopathol. 2006;34:692-693.
59 Chief P, Murray R, editors. Manual of Clinical Microbiology, 9th ed, ASM Press, 2007.
60 Kaufmann SHE, Walker BD. AIDS and TB: a deadly liaison. John Wiley and Sons, 2009.
61 Kishore Reddy VC, Aparna S, Prasad CE, et al. Mycobacterial culture of FNA- A useful tool in diagnosis. Ind J Med Microbiol. 2008;26:259-261.
62 Loya AC, Prayaga AK, Sundaram C, et al. Cytologic diagnosis of pancreatic tubeculosis in immune competent and immune compromised patients. Report of two cases. Acta Cytol. 2005;49:97-100.
63 Fitzhugh VA, McCash SI, Park E, et al. Mycobacterium avium complex infection in a neck abscess: a diagnostic pitfull in FNAB of head and neck lesions. Diagn Cytopathol. 2009;37:527-530.
64 Gangane N, Anshu, Singh R. Role of modified bleach method in staining of acid fast bacilli in lymph node aspirates. Acta Cytol. 2008;52:325-328.
65 Goel M, Budhwar P. Species specific immunocytchemical localization of M. tuberculosis complex in FNA of tuberculous lymphadenitis using antibody to 38kDa immunodominant protein antigen. Acta Cytol. 2008;52:424-433.
66 Purohit MR, Mustafa T, Sviland L. Detection of Mycobacterium tuberculosis by polymerase chain reaction with DNA eluted from aspirate smears of tuberculous lymphadenitis. Diagn Mol Pathol. 2008;17:174-178.
67 Jain A, Verma RK, Tiwari V, Goel MM. Dot ELISA vs PCR of FNA of Tuberculous lymphadenitis. Acta Cytol. 2005;49:17-21.
68 Siddaraju N, Roy SK, Manish MM, et al. FNAC diagnosis of erythema nodosum leprosum. A case report. Acta Cytol. 2007;51:800-802.
69 Siddaraju N, Sistla SC, Singh N, et al. Pure neuritic leprosy with nerve abscess presenting as a cystic soft tissue mass; a report of a case diagnosed by FNBC. Diagn Cytopathol. 2009;37:355-358.
70 Gambarino F, Carrilh C, Ferro J, et al. FNA diagnosis of Kaposi’s sarcoma in a developing country. Diagn Cytopathol. 2000;23:322-325.
71 Abati AD, Opitze L, Brones C, et al. Cytology of extrapulmonary Pneumocystis carinii infection in the AIDS. Diagn Cytopathol. 1991;7:615-617.
72 Ng VL, Yajko DM, Hadley WK. Extrapulmonary pneumocystis. Clin Microbiol Rev. 1997;10:401-418.
73 Anuradha (no initial)Sinha A. Extratpulmonary Pneumocystis carinii infection in an AIDS patient. A case report. Acta Cytol. 2007;51:599-601.
74 Mallick MG, Sengupta S, Bandyopadhyay A, et al. Cytodiagnosis of filarial infections from an endemic area. Acta Cytol. 2007;51:843-849.
75 Roy P, Rekhi B, Chinoy RF. Panorama of cytomorphological findings of filariasis in the contralateral breast, clinically mimicking a carcinoma in a known case: a case report. Diagn Cytopathol. 2008;36:794-796.
76 Kishore B, Khare P, Gupta RJ, et al. Microfilaria of Wucheria bancrofti in cytologic smears: a report of 5 cases with unusual presentations. Acta Cytol. 2008;52:710-712.
77 Dwivedi RC, Gupta P, Dwivedi RC, et al. Lymphadenovarix of the head-neck region – A rare presentation of Bancroftian filariasis. J Trop Pediatr. 2009;55:332-334.
78 Negahban S, et al. Dirofilaria repens diagnosed by presence of Microfilariae in FNA. Acta Cytol. 2007;51:567-570.
79 Das DK, Bhambhani S, Pant CS. Ultrasound guided FNAC; diagnosis of hydatid diseases of the abdomen and thorax. Diagn Cytopathol. 1995;12:173-176.
80 Oztek I, Baloglu H, Demirel D, et al. Cytologic diagnosis of pulmonary unilocular cystic hydatidosis. A study of 131 cases. Acta Cytol. 1997;41:1159-1166.
81 Gupta R, Mathur SR, Agarwala S, et al. Primary soft tissue hydatidosis: FNAC diagnosis in two cases. Diagn Cytopathol. 2008;36:884-886.
82 Daneshbod Y, Khademi B. Hydatid disease of the submandibular gland diagnosed by FNA; a case report. Acta Cytol. 2009;53:454-456.
83 Geramizadeh B, Vasei M. FNA of a breast mass in visceral leishmaniasis. Acta Cytol. 2007;51:499-500.
84 Daneshbod Y, Daneshbod K, Khademi B, et al. New cytologic clues in localized Leishmania lymphadenitis. Acta Cytol. 51, 2007. 699–671
85 Handa U, Garg S, Mohan N. FNA in the diagnosis of subcutaneous cysticercosis. Diagn Cytopathol. 2008;36:183-187.
86 Aggarwal S, Wadhwa N. Swelling on the tongue: a rare presentation of oral cysticercosis. Diagn Cytopathol. 2009;37:236-237.
87 Zaharopoulos P. Demonstration of parasites in Toxoplasma lymphadenitis by FNAC. Report of two cases. Diagn Cytopathol. 2000;22:11-15.
88 Wee A, Nilsson B, Yap I, et al. Aspiration cytology of liver abscesses, with an emphasis on diagnostic pitfalls. Acta Cytol. 1995;39:453-462.
89 Lacoste C, Escande MC, Jammet P, et al. Breast Actinomyces neuii abscess simulating primary malignancy: a case diagnosed by FNA. Diagn Cytopathol. 2009;37:311-312.
90 Fernandez H, D’ Souza CR, Shekar JC, et al. Cytodiagnosis of actinomycetoma. Diagn Cytopathol. 2009;37:506-508.
91 Angeles RM, Lasala RP, Fanning CV. Disseminated subcutaneous nocardiosis caused by Nocardia farcinica diagnosed by FNB biopsy and 16S ribosomal gene sequencing. Diagn Cytopathol. 2008;36:266-269.
92 Mathur VK, Sood R, Aron M, et al. Cytologic diagnosis of pulmonary nocardiosis. A report of 3 cases. Acta Cytol. 2005;49:567-570.
93 Stastny JF, Wakeley PE, Frable WJ. Cytologic features of necrotizing granulomatous inflammation consistent with cat-scratch disease. Diag Cytopathology. 1996;15:108-115.
94 Santiago K, Rivera A, Cabaniss D, et al. FNA of CMV Sialadenitis in a Patient with AIDS: Pitfalls of Diff-Quik Staining. Diagn Cytopathol. 2000;22:101-103.