CHAPTER 6. The neuromuscular system
Chapter contents
The nervous system 183
Skeletal muscle structure 197
Muscle fibres 201
Kinaesthetic sense and proprioception 207
Mechanical characteristics of muscle–tendon units 213
Muscle architecture and function 219
Review questions 223
References 223
The muscular system is the interface between the nervous and skeletal systems. The muscles produce the forces to move the levers of the skeletal system, but the nervous system determines the level and timing of the muscle forces. The nervous system constantly monitors and interprets information from the various senses concerning body position and body movement, including information from muscles and joint-supporting structures, and on the basis of this information sends instructions to the muscles to coordinate body movement in both voluntary and involuntary (reflex) movements. Those parts of the nervous and muscular systems responsible for bringing about coordinated movement are referred to as the neuromuscular system. This chapter describes the structure and function of the neuromuscular system.
The nervous system
The nervous system consists of approximately 13000 million nerve cells called neurones and an equally large number of specialized connective tissue cells called glial cells. Neurones are specialized to conduct electrochemical impulses rapidly throughout the body to coordinate all the essential biological functions. The cells of the nervous system are organized into two functional divisions and two structural divisions (Williams et al 1995). The functional divisions are the cerebrospinal nervous system and the autonomic nervous system.
The cerebrospinal nervous system, also known as the somatic, craniospinal or voluntary nervous system, is under voluntary control except for reflex movements. A reflex movement provides protection by rapidly removing part of the body from a source of danger without conscious effort. The cerebrospinal nervous system includes those parts of the nervous system concerned with consciousness and mental activities, and control of skeletal muscle. The autonomic nervous system, also known as the visceral or involuntary nervous system, is not under voluntary control; it includes those parts of the nervous system that control the visceral muscles, the heart, and the exocrine and endocrine glands.
The two structural divisions of the nervous system are the central nervous system and the peripheral nervous system. The central nervous system consists of the brain and spinal cord. The peripheral nervous system consists of 43 pairs of nerves (bundles of nerve fibres), which arise from the base of the brain and the spinal cord (Figure 6.1). The upper 12 pairs of nerves arise from the base of the brain and are called cranial nerves. The other 31 pairs arise from the spinal cord and are called spinal nerves. The cranial and spinal nerves convey information between the central nervous system and the rest of the body.
Neurones
Neurones differ in size and shape, but they all have three common structural features: a cell body, processes of varying length that extend from the cell body, and specialized sites for communicating with other neurons, with specialized receptors such as pain receptors, and with specialized effectors such as motor end plates in muscle. Neurones are classified by the direction in which they conduct impulses in relation to the brain. Sensory or afferent neurones conduct impulses towards the brain, and motor or efferent neurones conduct impulses away from the brain.
Nerve fibres
The processes that extend from the cell bodies of neurones are called nerve fibres. Nerve fibres vary in length from a few millimetres to more than 1m. There are two types of nerve fibre, dendrites and axons. Dendrites, or afferent fibres, conduct impulses towards the cell body. Axons, or efferent fibres, conduct impulses away from the cell body. In addition to the sensory and motor classification, neurones are classified on the basis of the number of processes arising from the cell body into pseudounipolar, bipolar and multipolar.
In a pseudounipolar neurone, there appears to be one process arising from the cell body that quickly divides into an afferent fibre and an efferent fibre (Figure 6.2A). Sensory neurones in the peripheral nervous system are pseudounipolar neurones. A bipolar neurone has two distinct processes, one afferent and one efferent (see Figure 6.2B). Bipolar neurones are found in the sensory areas of the eye, ear and nose. Multipolar neurones have numerous relatively short dendrites with a single axon that may branch at various points (see Figure 6.2C). Most of the neurones in the brain and spinal cord are multipolar neurones.
Myelinated and non-myelinated nerve fibres
Glial cells provide mechanical and metabolic support to neurones. In the central nervous system there are a variety of glial cells including astrocytes, which provide support for blood vessels, and oligodendrocytes, which provide support for nerve fibres. In the peripheral nervous system, there is only one type of glial cell, Schwann cells (Gamble 1988). All nerve fibres of the peripheral nervous system are enveloped by Schwann cells, which provide the same type of mechanical support as the oligodendrocytes provide for nerve fibres in the central nervous system. The Schwann cells around some fibres produce a fatty substance called myelin, which is deposited around the fibres as a multilayered myelin sheath. The sheath is in the form of a spiral with up to 100 regularly spaced layers of myelin separated by folds of Schwann cell membrane. The outer fold of the Schwann cell membrane is referred to as the neurilemma (Figure 6.3).
The greater the number of layers of myelin in the sheath, the faster the speed of nerve transmission along the nerve fibre. Nerve fibres that have a myelin sheath are referred to as myelinated or medullated nerve fibres and those nerve fibres that do not are referred to as non-myelinated or non-medullated nerve fibres. Each myelinated nerve fibre is enclosed within a continuous chain of Schwann cells and each Schwann cell envelops approximately 1mm of nerve fibre. At the junction between adjacent Schwann cells the myelin sheath is interrupted such that the nerve fibre is only covered by the neurilemma; these regions are referred to as nodes of Ranvier (see Figure 6.3A). The nodes of Ranvier facilitate intercellular exchange between the nerve fibres and the surrounding extracellular fluid, which is important for the nutrition of the nerve fibre and for the transmission of impulses along the nerve fibre. Myelinated nerve fibres in the central nervous system are different from those in the peripheral nervous system in that they are not surrounded by Schwann cells (there are no Schwann cells in the central nervous system) and have no neurilemma. It is thought that oligodendrocytes are responsible for the formation of the myelin sheath around these fibres at the embryonic stage (Williams et al 1995).
Non-myelinated nerve fibres of the peripheral nervous system are also enclosed within continuous chains of Schwann cells. However, in comparison to myelinated fibres, there are no nodes of Ranvier in non-myelinated fibres and there may be as many as nine nerve fibres enveloped within the folds of the Schwann cells in the chain (see Figure 6.3B).
Nerve fibre endings
The dendrites and axons of all types of neurones have a large number of terminal branches or nerve endings devoid of Schwann cells and myelin sheath. There are three types of nerve endings:
1. Sensory: where the nerve ending is in contact with a specialized receptor organ such as a pain receptor.
2. Motor: where the nerve ending is in contact with a specialized effector organ such as a motor end plate in muscle.
3. Synapse: where the nerve ending is in contact with another neurone.
Sensory and motor nerve endings are referred to as end organs. Neurones that only have synapses at their nerve endings are called association neurones or interneurones.
Nerve impulse transmission
The cytoplasm of a neurone and the extracellular fluid surrounding the neurone contain many different ions (electrically charged atoms). These include positively charged inorganic ions such as sodium (Na+) and potassium (K+), negatively charged inorganic ions such as chloride (Cl−) and various organic anions (negatively charged amino acids and proteins [A−]).
Like most membranes in the body, a nerve fibre membrane is semi-permeable, i.e. it has a large number of tiny holes through which ions and small molecules (aggregations of ions) pass from one side of the membrane to the other. The movement of ions through the nerve fibre membrane depends on the permeability of the membrane (the number and size of the holes in the membrane) and the force tending to drive the ions through the membrane. The driving force has electrical, chemical and, in the case of Na+ and K+, mechanical components. The electrical component depends on the polarity of the ions; like charges repel each other and unlike charges attract each other. The chemical component depends on the concentration of ions in different regions; ions move from an area of high concentration to areas of lower concentration. The mechanical component results from specialized regions of the membrane collectively referred to as the Na+−K+ pump. The Na+−K+ pump transports Na+ out of the cytoplasm and into the extracellular fluid and transports K+ in the opposite direction.
Under resting conditions, when the nerve fibre is not transmitting an impulse, the net effect of the membrane permeability and the driving forces on the various ions is that the electrical charge on the outside of the fibre membrane is approximately 70mV (millivolts) higher than on the inside; the potential difference across the membrane is approximately 70mV, with the inside negative with respect to the outside (Figure 6.4A). This resting potential difference is referred to as resting membrane potential (RMP) (Enoka 1994).
The arrival of a stimulus at a nerve fibre in a state of RMP alters the permeability of the fibre membrane to Na+ and K+ so that Na+ flows into the cell and K+ flows out of the cell. The flow of Na+ into the cell is initially greater than the flow of K+ out of the cell so that the potential difference across the membrane decreases. If the decrease in potential difference, which depends on the strength of the stimulus, reaches a critical level of approximately 60mV (with the inside of the membrane negative with respect to the outside), the membrane will be depolarized, i.e. the potential difference across the fibre membrane rapidly changes by approximately 100mV from 70mV, with the inside of the membrane negative with respect to the outside, to approximately 30mV, with the inside of the membrane positive with respect to the outside (see Figure 6.4A).
This change in potential difference constitutes an action potential, which results in the flow of electrical current, called local current, between the depolarized region of the cell membrane and the adjacent unpolarized regions (both sides) (see Figure 6.4B and C). The establishment of local current results in progressive (rapid wave) depolarization of the rest of the cell membrane so that the impulse is transmitted along the whole length of the fibre. After depolarization, the membrane is rapidly depolarized such that the action potential appears as a spike in a graph of the change in membrane potential with time (see Figure 6.4A).
The duration of the action potential spike (depolarization and repolarization) is less than one millisecond (Gamble 1988). Repolarization is due largely to a rapid decrease in the flow of Na+ into the cell (due to reduced permeability of the membrane to Na+ ions) and continued flow of K+ out of the cell. Following repolarization there is usually a period (15–100ms) of hyperpolarization in which the potential difference across the membrane is slightly greater than RMP as RMP is gradually restored (see Figure 6.4A). A stimulus not strong enough to cause depolarization results in a period of hypopolarization prior to restoration of RMP, i.e. a period in which the potential difference across the membrane is slightly less than RMP (see Figure 6.4A).
Synapses
Every branch of an axon terminates in an end bulb (or end foot) that rests on the surface of a neighboring neurone to form a synapse, a specialized region that facilitates one-way communication between the two neurones. Each neurone synapses with hundreds or thousands of other neurones (Williams et al 1995). The most common type of synapse is axodendritic (between an axon and a dendrite), but synapses may also be axosomatic (between an axon and a cell body), and axoaxonic (between two axons).
Speed of nerve impulse transmission
The speed of transmission or conduction of impulses is directly proportional to fibre diameter and the thickness of the myelin sheath; the larger the diameter and the thicker the myelin sheath, the faster the speed of conduction. Non-myelinated fibres do not have a myelin sheath and, thus, have much lower conduction speeds than myelinated fibres. Nerve fibres of the peripheral nervous system are classified on the basis of conduction speed and fibre diameter (Table 6.1). There are five main categories of afferent fibres (Ia, Ib, II, III and IV) and five main categories of efferent fibres (Aα, Aβ, Aγ, B and C). Skeletal muscle is innervated by the fastest afferent fibres (Ia and Ib) and the fastest efferent fibres (Aα, Aβ, Aγ).
Nerve tissue organization in the brain
The brain is the largest and most complex aggregation of neurones in the nervous system. It consists of the cerebrum and the cerebellum (see Figure 6.1). The cerebrum occupies most of the cranium and is bigger than the cerebellum. The region of the cerebrum close to and including its surface is called the cerebral cortex and consists of grey matter, i.e. the cell bodies of neurones and their processes, which are largely non-myelinated, together with their synapses and supporting glial cells. The cerebrum is heavily convoluted with fissures of varying depth. The largest fissure is the longitudinal central fissure that divides the cerebrum in the median plane into right and left cerebral hemispheres.
The cerebellum, which occupies the posterior inferior aspect of the cranium, is separated from the cerebrum by the transverse fissure. Like the cerebrum, the region of the cerebellum close to and including its surface consists of grey matter and is called the cerebellar cortex. The cerebellum is not convoluted but is traversed by numerous small furrows. The convolutions of the cerebrum and furrows of the cerebellum significantly increase their surface areas and, thus, the volume of grey matter. The inner parts of the cerebrum and cerebellum consist of white matter, i.e. largely myelinated nerve fibres organized into groups that link the different parts of the cerebrum and cerebellum with each other and with the spinal cord.
Nerve tissue organization in the spinal cord and spinal nerves
In transverse section, the spinal cord is roughly oval shaped with an anterior median fissure and a posterior medial septum (Figure 6.5). The central area is dominated by a roughly H-shaped mass of grey matter, with the rest of the cord consisting of white matter in which the groups of fibres run parallel with the spinal cord. The posterior projections (or horns) of the grey matter are continuous with afferent fibres that enter the spinal cord via the left and right dorsal (posterior) roots of the corresponding left and right spinal nerves. The cell bodies of the afferent fibres are located in the dorsal root ganglia; a ganglion is an aggregation of cell bodies outside the spinal cord.
The anterior projections of the grey matter are continuous with efferent fibres that leave the spinal cord via the left and right ventral (anterior) roots of the corresponding spinal nerve. A spinal nerve is formed by the aggregation of the afferent and efferent fibres as they pass through the corresponding intervertebral foramen. Each spinal nerve divides into an anterior and posterior branch just outside the intervertebral foramen; the anterior branch is usually much larger than the posterior branch. In each branch, the individual nerve fibres are enveloped in a thin layer of connective tissue called the endoneurium, which supports a blood capillary network. The fibres are grouped together in bundles called fasciculi or funiculi and each fasciculus is sheathed within a layer of connective tissue called the perineurium. The fasciculi are grouped together and sheathed within another layer of connective tissue called the epineurium to form the complete spinal nerve (Figure 6.6). Each spinal nerve consists of a mixture of myelinated and non-myelinated nerve fibres.
In all regions of the spinal cord apart from most of the thoracic region, the spinal nerves link up with each other on each side of the spinal cord to form networks called plexuses (see Figure 6.1). The upper four pairs of cervical nerves form the left and right cervical plexuses that innervate the head and upper part of the neck. The other cervical spinal nerves combine with the first pair of thoracic spinal nerves to form the left and right brachial plexuses that innervate the upper limbs. The twelfth thoracic spinal nerves combine with the lumbar spinal nerves to form the lumbar plexuses that innervate the lower trunk and pelvis. The sacral and coccygeal spinal nerves with branches from the fourth and fifth lumbar spinal nerves combine to form the left and right sacrococcygeal plexuses that innervate the legs. The second to the eleventh thoracic spinal nerves, which innervate the trunk, do not form plexuses.
Voluntary and reflex movements
In general terms, the brain interprets sensory information from the various receptors and on the basis of this information brings about appropriate responses via effectors to ensure normal bodily functioning. Whereas the autonomic nervous system operates largely at a subconscious level, the cerebrospinal nervous system normally operates at the level of consciousness, i.e. the individual consciously processes information, such as visual, auditory and kinaesthetic (sense of movement) information, and consciously brings about appropriate responses. For example, consider a child just about to touch a hot coffeepot resting on a stove (Figure 6.7).
1. As the child’s hand moves close to the pot, the heat radiating from the pot may excite the heat receptors in the skin of her hand.
2. This information is transmitted by sensory neurones in the peripheral nervous system to the spinal cord, where the nerve endings of the axons of the sensory neurones synapse with many other neurones.
3. Some of these neurones relay the information to the brain centre responsible for heat sensation in the hand, and the child experiences the sensation of heat.
4. Having sensed the heat from the coffeepot, the child decides to move her hand away from the pot to avoid the danger.
5. Impulses are sent from the brain to motor neurones in the spinal cord that innervate the muscles of the arm.
6. The impulses are relayed to the muscles of the arm resulting in movement of the hand away from the coffeepot.
 |
| Figure 6.7 |
The above sequence, illustrated in Figure 6.7, is typical of all voluntary movements. If the child had not sensed the danger and had actually touched the hot coffeepot, she would have jerked her hand away from the pot with lightening speed before she was even conscious of the heat. This extremely rapid involuntary reaction is called a reflex action and is one of a host of similar reflexes that result in instant reactions to protect the body from potentially harmful stimuli. In this particular case, the reflex action results in the child’s hand being in contact with the coffeepot for only a fraction of the time it would have taken for the child to take voluntarily (and, therefore, consciously) her hand away from the pot. Consequently, the child may sustain a relatively slight burn rather than a serious burn, which would have resulted from more prolonged contact with the coffeepot.
The increased speed of a reflex action compared to a voluntary action is due to a reduction in the distance over which the impulses travel from receptor to effector. In this particular case, the heat from the coffeepot is sensed by the heat receptors in the hand, which send impulses to the spinal cord. This is basically the same as in a voluntary action (stages 1 and 2 in Figure 6.7). However, in a reflex action the impulses are transmitted directly across the spinal cord to the motor neurones that innervate the muscles of the arm (stage 3A in Figure 6.7). The impulses are relayed to the muscles of the arm and the reflex action is completed. Consequently, a reflex action is faster than a voluntary action because no time is spent in transmitting impulses up the spinal cord to the brain, making a decision and transmitting impulses back down the spinal cord (stages 3, 4 and 5 in Figure 6.7 are omitted). A reflex action is triggered when the intensity of the impulse output from the receptors is above a certain threshold indicating extreme danger to the body. In this particular case, the hotter the coffeepot, the greater the intensity of output from the heat receptors and the greater the likelihood of a reflex action being triggered.
As the impulses are transmitted across the spinal cord (stage 3A in Figure 6.7), the impulses are simultaneously transmitted to the brain as in a voluntary action (stage 3 in Figure 6.7). However, stage 3 (transmission of impulses to the brain and sensation of heat) takes longer than stages 3A and 6 such that it is a fraction of a second after the child jerks her hand away from the coffeepot before she perceive any pain in the hand.
Nerve fibre injuries
Injuries to peripheral nerves usually occur as a result of compression or traction (stretching) or a combination of the two (Kleinrensink et al 1994). Compression and traction may damage any or all of the main structures: connective tissue sheaths, Schwann cells, myelin sheath (when present) and nerve fibres. Compression and traction damage the nerve structures directly due to crushing and tearing, respectively. Prolonged compression also may damage the nerve structures indirectly as a result of ischemia, i.e. a disruption of the local blood supply (due to compression of the local capillary network), which results in a deficiency of oxygen and nutrients to the affected tissues.
Injuries to peripheral nerves are classified on the basis of degree of structural and functional damage into neuropraxia, axonotmesis and neurotmesis (Seddon 1972). Neuropraxia is the lowest level of damage. It involves damage to Schwann cells and myelin sheaths, but little or no damage to nerve fibres or endoneurium. Neuropraxia is characterized by a disruption to impulse transmission, which is often associated with pain and tingling in the areas innervated by the affected nerves. Recovery of a nerve fibre from neuropraxia usually occurs within 10 to 14 days. In axonotmesis, the nerve fibre and Schwann cell covering or myelin sheath are severed, but the endoneurium remains intact. The severed part of the fibre degenerates. Recovery begins when fibre sprouts emerge from the severed end of the part of the fibre still attached to the cell body. One of the sprouts eventually dominates and gradually grows along the tube formed by the endoneurium. Growth occurs at a rate of approximately 2.5cm per month, and function gradually returns to normal as the fibre and myelin sheath (when present) returns to normal. In neurotmesis, the nerve fibre, Schwann cell covering or myelin sheath, and endoneurium are all severed. The severed part of the fibre degenerates and without surgical repair it is unlikely that much recovery will occur. With surgical repair some recovery occurs similarly to the way in which a fibre recovers from axonotmesis, but functional outcome is often less than satisfactory.
Skeletal muscle structure
The composition and basic function of the muscular system are described in chapter 2. This section describes the macrostructure of skeletal muscle.
Origins and insertions
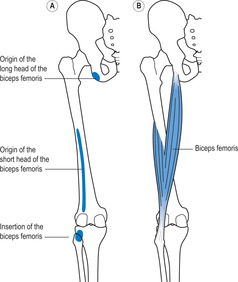
Most of the muscles are attached to the skeletal system by tendons or aponeuroses. However, one or both attachments of some muscles attach directly onto bone without an intervening tendon or aponeurosis. For example, the biceps femoris arises from two sites: a tendinous attachment to the ischial tuberosity and directly from a long narrow area on the lateral aspect of the femur (Figure 6.8A). The muscle is attached by a single tendon to the head of the fibula and adjoining posterior aspect of the lateral tibial condyle. Most of the muscles of the upper and lower limbs are arranged in line with the direction of the long bones. For descriptive purposes, the proximal and distal attachments of each of these muscles are referred to as the origin and insertion of the muscles, respectively. For example, the origin of the biceps femoris is on the ischial tuberosity (long head) and femur (short head) and the insertion is on the fibula and tibia (see Figure 6.8B).The origins and insertions of the muscles of the trunk and the muscles that link the trunk to the limbs tend to be medial and lateral, respectively, but there are exceptions.
Pennate and non-pennate muscles
A skeletal muscle is made up of skeletal muscle cells bound by various layers of connective tissue that support extensive networks of nerves and blood vessels. Muscle cells are long and thin and, as such, they are usually referred to as muscle fibres. Each muscle fibre is approximately 50μm (1μm = one-millionth of a metre) wide. All the fibres in an individual muscle are about the same length. In some muscles the fibres are relatively short; for example, in the muscles that move the eyes, the fibres are 2 to 4mm long. In contrast, the fibres in some other muscles are very long; for example, the fibres of the sartorius are approximately 30cm long. The muscle fibre length in most muscles is between these two extreme values.
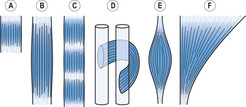
The fibres in all muscles are organized into bundles (as described later), and the fibres in each bundle run parallel with each other. However, the arrangement of the bundles of fibres with respect to the origin and insertion of the muscle is either pennate or non-pennate. In a pennate muscle, the fibres run obliquely with respect to the origin and insertion so that the line of pull of the fibres is oblique to the line of pull of the muscle (Figure 6.9). Pennate muscles have a feather-like appearance and are classified according to the number of groups of fibres into unipennate, bipennate and multipennate. In a unipennate muscle there is one group of fibres that inserts onto the sides of two non-parallel tendons (or one bony attachment and one non-parallel tendon) (see Figure 6.9A). The flexors and extensors of the fingers are unipennate muscles. In a bipennate muscle such as the gastrocnemius there are two groups of fibres that insert onto the opposite sides of a central tendon (see Figure 6.9B). A multipennate muscle such as the deltoid is, in effect, two or more bipennate muscles combined into a single muscle (see Figure 6.9C).
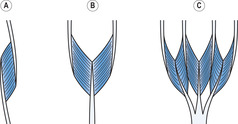 |
| Figure 6.9 |
In a non-pennate muscle, the fibres run in line with the line of pull of the muscle. There are five main types: quadrilateral, strap, spiral, fusiform and fan shaped (Figure 6.10). In a spiral muscle, the muscle curves around other muscles or bones. In a fusiform (or spindle-shaped) muscle, the fibres are gathered at each end to attach onto long, relatively narrow tendons. In a fan-shaped muscle, the fibres converge from a broad origin to a relatively small insertion. The effect of pennate and non-pennate arrangements on muscle function is described later in the chapter.
Fusiform muscle–tendon units
Figure 6.11 shows the structure of a fusiform muscle–tendon unit. Each muscle fibre is enveloped in a layer of areolar tissue called the endomysium, which helps to bind the muscle fibres together and provides a supporting framework for blood capillaries and the terminal branches of nerve fibres. The muscle fibres are grouped together by irregular connective tissue (a mixture of collagenous and elastic) in bundles of up to 200 fibres; each bundle is called a fasciculus (or funiculus) and the connective tissue sheath is called the perimysium. The fasciculi are bound together to form the belly of the muscle by a layer of irregular collagenous connective tissue called the epimysium. The muscle fibres gradually taper at each end. The tapering of the fibres accompanies a gradual thickening in the epimysium and perimysium layers and a change in the composition of the epimysium and perimysium layers from irregular collagenous and irregular elastic connective tissue to regular collagenous connective tissue as the epimysium and perimysium layers merge to form a tendon or aponeurosis.
Each muscle receives one or more nerves (collections of sensory and motor nerve fibres), which usually enter the muscle together with the main blood vessels (arteries enter, veins leave) at a region of the muscle that does not move a great deal during normal movement; this region is referred to as the neurovascular hilus (Williams et al 1995). The blood vessels and nerves branch through the epimysium and perimysium layers down to the endomysium of the individual muscle fibres.
A skeletal muscle is made up of skeletal muscle fibres bound together by various layers of connective tissue that support extensive networks of nerves and blood vessels and form tendons and aponeuroses. The muscle fibres are organized into bundles that have a pennate or non-pennate arrangement
Muscle fibres
A muscle fibre consists of hundreds or thousands of myofibrils embedded in sarcoplasm (muscle cytoplasm) and enclosed by a cell membrane called the sarcolemma (Figure 6.12A and B). Each muscle fibre has a large number of nuclei that lie just beneath the sarcolemma. Each myofibril is approximately 1μm wide; the myofibrils are arranged parallel to each other and run the whole length of the muscle fibre. Each myofibril exhibits a characteristic pattern of alternate light and dark transverse bands due to the way that the components of the myofibril reflect light (under an electron microscope). The light and dark bands are referred to as I (isotropic) and A (anisotropic) bands, respectively. Since the light and dark bands coincide in adjacent myofibrils, the muscle fibre has a striped or striated appearance; thus, skeletal muscle is often referred to as striated muscle.
 |
| Figure 6.12 |
Each myofibril has two types of filaments arranged in a highly ordered way that gives rise to the light and dark bands. One type of filament is thicker than the other. The thicker filaments occupy the A bands and are composed of the protein myosin; these filaments are usually referred to as myosin filaments or A filaments (see Figure 6.12C). The thinner filaments, actin filaments or I filaments, occupy the I bands and are composed largely of the protein actin (Edman 1992). Each I band is divided by a transverse Z disc. The section of a myofibril between two successive Z discs is called a sarcomere, which is the basic structural unit of a muscle fibre. A myofibril consists of a chain of sarcomeres. The actin filaments project from each side of the Z discs and reach into the A bands of the corresponding sarcomeres where they interdigitate with the myosin filaments (see Figure 6.12C). The region between the ends of the two groups of actin filaments in the middle of a sarcomere is referred to as the H zone.
Each actin filament basically consists of two strands of actin molecules wound together longitudinally in a helical manner (see Figure 6.12D). Each myosin filament is composed of myosin molecules. Each myosin molecule is a club-like structure consisting of two adjacent globular heads attached by a relatively short curved neck to a long shaft (see Figure 6.12E). In a myosin filament, the myosin molecules are packed such that the shafts form the main body of the filament with the heads projecting from the main body at regular intervals (see Figure 6.12F). The two halves of each myosin filament are mirror images of each other, i.e. the myosin molecules in one half of the filament are oriented in the opposite direction to the myosin molecules in the other half. This arrangement, significant in the functional interaction between the actin and myosin filaments, is such that no myosin heads project from the central region of each filament; this central region is sometimes referred to as the inert zone, M band or M line. Each myosin filament is surrounded by six actin filaments in a regular hexagonal arrangement (see Figure 6.12G). In each half of a myosin filament, the myosin molecules are arranged in groups of six; a line joining the heads of the molecules in each group forms a spiral around the myosin filament. The corresponding head in each group faces the same actin filament.
Types of muscular contraction
When a muscle fibre contracts, the heads of the myosin molecules attach to special sites on the actin filaments in the regions where the actin and myosin filaments overlap. The attachments are referred to as cross bridges and each cross bridge generates a certain amount of tension. When a muscle fibre relaxes, the myosin heads detach from the actin and no tension is exerted between them.
When a muscle contracts, the tension exerted by the muscle is directly proportional to the number of cross bridges; the larger the number of cross bridges, the greater the tension. When a muscle contracts it tends to shorten, i.e. it tends to pull its origin and insertion closer together. However, while contracting, the muscle may shorten, lengthen or stay the same length depending on the external load on the muscle (the load tending to lengthen the muscle). If the muscle force is greater than the external load, the muscle will shorten; this type of contraction is called a concentric contraction. If the muscle force is less than the external load, the muscle will lengthen; this type of contraction is called an eccentric contraction. Concentric and eccentric contractions are often referred to as isotonic contractions, i.e. contractions that involve a change in the length of the muscle. If the muscle force is equal to the external load, the length of the muscle will not change; this type of contraction is called an isometric contraction.
Sliding filament theory of muscular contraction
During all types of muscular contraction the actin and myosin filaments stay the same length, but in isotonic contractions the degree of interdigitation between the two sets of filaments changes as the length of the muscle fibres changes; the width of the A bands stays the same, but the width of the I bands and H zones varies. As the muscle fibres shorten, the region of interdigitation increases and the width of the I bands and H zones decreases. As the muscle fibres lengthen, the region of interdigitation decreases and the width of the I bands and H zones increases. These observations led to the formulation of the sliding filament theory of muscular contraction (Huxley and Hanson 1954). The essential features of the theory are as follows:
1. When a muscle contracts, force is generated by the formation of cross bridges.
2. Flexion of the necks of the myosin molecules while the heads are in contact with the actin filaments exerts a pulling force on the actin filaments, which causes the actin filaments to slide relative to the myosin filaments, so that the muscle shortens.
4. The coupling and decoupling of different cross bridges occurs at different times so that tension can be maintained while the muscle fibre shortens.
Whereas details of the processes involved have still to be discovered, the sliding filament theory has now gained general acceptance (Edman 1992).
Motor units
The functional unit of skeletal muscle is the motor unit. A motor unit consists of a motor neurone with an Aα axon (sometimes referred to as an alpha motoneurone), together with all the terminal branches of the axon and the muscle fibres that they innervate (Gamble 1988) (Figure 6.13). The number of muscle fibres innervated by a single alpha motoneurone is referred to as the innervation ratio. The innervation ratio of different motor units varies considerably, from approximately 1:4 in the muscles that move the eyes, to approximately 1:2000 in the large back extensor and leg extensor muscles. When an alpha motoneurone transmits an action potential, all of the fibres in the motor unit contract. Consequently, muscles associated with very fine motor control, such as the muscles that move the eyes, have motor units with low innervation ratios so that the amount of force produced can be finely controlled. Muscles associated with forceful movements are made up of motor units with relatively high innervation ratios. The fibres of a single motor unit are usually mixed with the fibres of other motor units, but are grouped within a relatively small area of the muscle.
 |
| Figure 6.13 |
A muscle fibre contracts when an action potential is generated in the sarcolemma at the junction with a motor end plate. The action potential is transmitted along and across the muscle fibre (by a system of tubules continuous with the sarcolemma) resulting in contraction. A single action potential produces a muscle twitch, i.e. a force that rapidly peaks and then equally rapidly dies away. If a series of action potentials is generated at a high enough frequency, the twitches fuse to produce tetanus, a sustained level of force.
Slow and fast twitch muscle fibres
Whereas the basic structure and function of all muscle fibres is the same, muscle fibres and, consequently, motor units vary in relation to the following:
Muscle fibres are classified into slow twitch fibres and fast twitch fibres on the basis of their contraction times. Slow twitch fibres, also referred to as type I and red fibres, have contraction times of 100 to 120ms. Fast twitch fibres, also referred to as type II fibres and white fibres, have contraction times of 40 to 45ms (Gamble 1988; Gregor 1993). The metabolism of slow twitch fibres is basically aerobic and, therefore, they are resistant to fatigue. Fast twitch fibres are subdivided on the basis of their metabolic characteristics into type IIa (aerobic and fatigue resistant) and type IIb (anaerobic and fatigue sensitive). The muscle fibres in a particular motor unit have the same functional characteristics and, as such, motor units can be classified into three categories:
• slow contracting, fatigue resistant (S)
• fast contracting, fatigue resistant (FR)
• fast contracting, fatigable (FF)
Individuals differ in the proportions of the different types of muscle fibres in their muscles. The average person has approximately 50% type I, 25% type IIa and 25% type IIb fibres in their calf muscles. In comparison, elite distance runners have a much higher proportion of type I fibres, and elite sprinters have a much higher proportion of type IIb fibres (Gamble 1988).
Whereas the classification of muscle fibres into types I, IIa and IIb is widely used, the categories represent ranges of metabolic and functional characteristics rather than discrete categories (Sargeant 1994). The metabolic and functional characteristics of muscle fibres appear to be influenced considerably by the type of innervation the fibres receive. In experimental animals, it has been shown that altering the type of innervation results, over time, in a change in the metabolic and functional characteristics of muscle fibres (Noth 1992). Table 6.2 lists the characteristics of slow and fast twitch motor units.
Kinaesthetic sense and proprioception
The central nervous system constantly receives sensory information from a wide variety of sources concerning the different aspects of physiological functioning. Awareness of body position and body movement is provided by a range of sensory organs, in particular, those concerned with the sensations of effort and heaviness, timing of the movement of individual body parts, the position of the body in space, joint positions and joint movements.
The input from these sources contributes to what is referred to as kinaesthetic sense (or kinaesthesia) (Figure 6.14). Some aspects of kinaesthetic sensitivity, such as a sense of effort and heaviness, and a sense of timing of actions, are generated by sensory centres that monitor the motor commands sent to muscles. Other aspects of kinaesthetic sensitivity are generated largely by input from peripheral receptors that monitor the execution of motor commands, i.e. the actual movements. For example, input from the eyes and ears is responsible for generating a sense of the position of the body in space. The sense of the position of a joint and movement of a joint is generated by a group of receptors located in the skin and musculoskeletal tissues. These receptors are called proprioceptors, and the sensation they provide is called proprioception. Proprioceptors are mechanoreceptors; they are activated by physical distortion.
Proprioceptors
The existence of proprioceptors in the skin, muscles, tendons, joint capsules and ligaments is well established, but the precise role of the different proprioceptors and the interrelationships between them is less clear (Grigg 1994). However, it appears that proprioceptors in joint capsules and ligaments are largely responsible for generating a sense of joint position and joint movement in the end ranges of joint movements, where joint capsules and ligaments may become taut. Between the end ranges it seems unlikely that the proprioceptors in joint capsules and ligaments provide much sensory information since they are unlikely to be under sufficient tension to excite them.
Joint and ligament proprioceptors
There are two main types of proprioceptors in joint capsules, Ruffini end organs (or Ruffini corpuscles) and Pacinian corpuscles. Ruffini end organs, which appear to be mainly responsive to tension and Pacinian corpuscles, which appear to be responsive to compression, are widely distributed between the collagenous fibres of the joint capsule and surrounding fascia.
The proprioceptors in ligaments and skin are similar to those found in joint capsules. However, there are few proprioceptors in ligaments and skin compared to the number in joint capsules. In addition, since tension in ligaments and skin may be caused by movement in a number of different directions, it is unlikely that the proprioceptors can provide information on movement in specific directions. For these reasons, it is thought that the contribution of the proprioceptors in ligaments and skin to proprioception is relatively small (Grigg 1994). However, they may make a significant contribution to joint stabilization in reflexive muscular activity.
Muscle and tendon proprioceptors
Whereas proprioceptors in joint capsules and, to a lesser extent, in ligaments, appear to generate information on joint position and joint movement in the end ranges of joint movements, proprioceptors in muscles, called muscle spindles, and tendons, called Golgi tendon organs, seem to be responsible for generating this type of information for movement between end ranges. Golgi tendon organs and muscle spindles are responsive to tension.
Each muscle spindle consists of a number of tiny muscle fibres enclosed within a spindle-shaped connective tissue capsule (Figure 6.15). The muscle spindle fibres are referred to as intrafusal (inside the spindle) to distinguish them from the larger and more numerous extrafusal fibres that make up the vast majority of the muscle. Muscle spindles are embedded between extrafusal fibres. Intrafusal fibres are classified on the arrangement of their nuclei into nuclear bag fibres and nuclear chain fibres. In both types of fibre, the nuclei are located in the central region of the fibre, which is devoid of myofibrils. In nuclear bag fibres, the nuclei cluster in a group, whereas in nuclear chain fibres the nuclei are arranged in a line parallel to the long axis of the fibre. The regions of the fibres on each side of the central region contain a large number of myofibrils and are referred to as the polar regions (Enoka 1994).
 |
| Figure 6.15 |
Nuclear bag fibres and nuclear chain fibres are usually about 8mm and 4mm long, respectively, and in a typical muscle spindle there are two bag fibres and four chain fibres (Roberts 1995). The central regions of both types of fibre are supplied with type Ia and type II sensory nerve endings. The endings of type Ia nerve fibres spiral around the intrafusal fibres and the endings of type II nerve fibres consist of a number of branches with end bulbs; the spirals and end bulbs adhere closely to the sarcolemma of each fibre.
The polar regions of the intrafusal fibres are supplied with motor nerve endings from Aβ and Aγ motor nerve fibres. Stimulation of the intrafusal fibres via these nerves results in contraction of the polar regions, which stretches the central regions and excites the spiral and end bulb nerve endings. This results in sensory discharge via the type Ia and II sensory nerve fibres. The type Ia and II fibres synapse in the spinal cord directly with the Aα motor neurones that supply the extrafusal muscle fibres of the same muscle, resulting in contraction of the extrafusal fibres. The level of contraction of the extrafusal fibres depends on the level of activation, which, in turn, depends on the degree of tension in the intrafusal fibres. Under resting conditions there is always a certain amount of tension in the intrafusal muscle fibres (due to activation by the sensory centers of the brain via the Aβ and Aγ fibres), which, in turn, results in a certain amount of tension in the extrafusal muscle fibres. The resting level of tension in the intrafusal and extrafusal muscle fibres is called muscle tone; the level of muscle tone in the extrafusal fibres is determined by the level of muscle tone in the intrafusal fibres.
Sensory output from muscle spindles can be generated by stimulation via the Aβ and Aγ motor nerve fibres (sometimes referred to as the fusimotor nerves) that innervate the intrafusal fibres. However, sensory output from muscle spindles can also be generated by stretching the muscle as a whole, since this will stretch the muscle spindles and excite the spiral and end bulb nerve endings. Low-velocity stretching of active muscles, eccentric muscle contraction, is an essential feature of normal movements; as a muscle group shortens, its antagonist partner experiences eccentric contraction. It is thought that the sensory information provided by muscle spindles as a result of stretching during eccentric contraction provides a sense of joint position and joint movement, especially during mid-range movements (Gandevia et al 1992). The amount of sensory information generated depends on the muscle tone of the intrafusal fibres; increasing the muscle tone of the intrafusal fibres increases the sensitivity of the spindles to stretch, and vice versa.
Whereas low-level stretch of a muscle seems to be important in generating proprioceptive information concerning joint movement and joint position, rapid stretch results in reflex contraction of the muscle (via the spindle afferent to muscle efferent loop) to prevent subluxation of the associated joints. These stretch reflexes are important in protecting joints from injury. For example, recurrent inversion sprains of the ankle are associated with deficient stretch reflex of the everters and dorsiflexors (Garn & Newton 1988). Excitation of muscle spindles appears to be mainly responsible for initiating reflex muscle contractions. However, there is evidence that proprioceptors in joint capsules and ligaments may also contribute to the initiation of such reflex contractions, especially at the ends of joint ranges of movement (Matthews 1988; Hall et al 1994). In these situations, joint capsules and ligaments can be taut and their proprioceptors most active.
Proprioceptors in joint capsules and ligaments (Ruffini end organs and Pacinian corpuscles) appear to generate information on joint position and joint movement in the end ranges of joint movements. Proprioceptors in muscles (muscle spindles) and tendons (Golgi tendon organs) appear to be responsible for generating this type of information for movement in between end ranges of joint movements
Role of proprioceptors
The precise roles of the various types of proprioceptors are not yet clear. However, there is general agreement that proprioceptive information aids coordination and balance and, in particular, maintains joint congruence (Grigg 1994; Wilkerson & Nitz 1994). It appears that injury to muscles, ligaments and capsules may damage proprioceptors resulting in long-term proprioceptive deficits, which, in turn, contribute to the development of degenerative joint diseases such as osteoarthritis (Freeman & Wyke 1967; Garn & Newton 1988; Hall et al 1995). There is evidence that proprioceptive information from muscle–tendon units can be enhanced by specific exercises that emphasize activation of muscle spindles and, thereby, improve muscle tone. Such enhancement may compensate for proprioceptive deficits in other structures such as joint capsules and ligaments (Skinner et al 1986; Steiner et al 1986; Beard et al 1994).
Mechanical characteristics of muscle–tendon units
The amount of force generated by a muscle–tendon unit depends on the length of the unit at the time of stimulation and the speed with which it changes length in the ensuing contraction. In this regard, an isometric contraction is simply one point on the continuum between maximum velocity of shortening (concentric contraction) and maximum velocity of lengthening (eccentric contraction).
Length–tension relationship in a sarcomere
The amount of tension generated in a sarcomere depends on the number of cross bridges attached between the actin and myosin filaments; the greater the number of cross bridges, the greater the tension. The actual number of cross bridges depends on the degree of interdigitation between the actin and myosin filaments. Too much and too little interdigitation decreases the number of myosin heads that are in a position to attach to form cross bridges.
Figure 6.16 shows the isometric length–tension relationship for a sarcomere; the sarcomere was maximally stimulated, but not allowed to shorten, at a number of different sarcomere lengths and the force was recorded at each length (Gordon et al 1966). When the sarcomere is extended to the point where there is no interdigitation between the actin and myosin filaments, no tension is generated since there are no myosin heads in a position to attach to form cross bridges. This situation is represented by the point L5 in Figure 6.16. As the sarcomere shortens and the degree of interdigitation gradually increases, there is a linear increase in the amount of force generated up to the point L4 where the length–tension relationship levels off. At L4 the maximum number of cross bridges has been formed and, as such, tension is maximum.
The region of the length–tension relationship between L4 and L5 is referred to as the descending limb. At L4 the H zone corresponds to the inert zone of the myosin filaments (see Figure 6.12C). As the sarcomere shortens between L4 and L3 the tension stays the same since no more cross bridges can be formed (the inert zones of the myosin filaments do not have myosin heads to attach cross bridges). The region of the length–tension relationship between L3 and L4 is referred to as the plateau region. At L3 the ends of the actin filaments in each half of the sarcomere come together, i.e. the H zone is zero. As the sarcomere shortens between L3 and L2 the actin filaments progressively overlap each other, resulting in a progressive drop in tension since the overlapping actin filaments interfere with cross bridge formation in the region of overlap. The region of the length–tension relationship between L2 and L3 is referred to as the shallow ascending limb. At L2 the ends of the myosin filaments abut the Z discs. As the sarcomere shortens between L2 and L1 there is a rapid and progressive drop in tension due to progressive overlap of the actin filaments and progressive longitudinal compression of the myosin filaments, both of which reduce the number of myosin heads available to form cross bridges. At L1 no cross bridges can be formed and, consequently, no tension is generated. The region of the length–tension relationship between L1 and L2 is referred to as the steep ascending limb.
Length–tension relationship in a muscle–tendon unit
The length–tension relationship of a muscle–tendon unit is different from that of a sarcomere due to the muscle–tendon unit’s connective tissue, which exerts passive tension when stretched. Consequently, the tension produced by a muscle–tendon unit will be the sum of the tension produced by the contractile (muscle) component and the passive tension exerted by the connective tissue components. Figure 6.17 shows the isometric length–tension relationship (or curve) for a muscle–tendon unit. The contributions of the contractile and connective tissue components to total tension at any particular length are shown in the separate curves. Some of the connective tissue components are parallel with the muscle fibres and some are arranged in series; this has given rise to the terms parallel elastic component and series elastic component (Huijing 1992). The parallel elastic component consists of sarcolemma, endomysia, perimysia and epimysium. The series elastic component consists of tendons and aponeuroses, and strands of the protein titin, which connect the ends of the myosin filaments to the Z discs in each sarcomere. In addition to contributing to passive tension when stretched, the titin strands stabilize the hexagonal arrangement of the actin and myosin filaments (Lieber and Bodine-Fowler 1993).
 |
| Figure 6.17 |
In the absence of stimulation and any external load, the muscle–tendon unit assumes a rest length at which the tension in the unit is zero, with no tension in the contractile component, and no tension (no stretch) in the connective tissue component. Figure 6.17 shows that rest length is associated with that part of the isometric length–tension curve at which tension in the contractile component is maximum.
As in a sarcomere, the shorter the muscle–tendon unit, the lower the tension generated. Tension tends to reduce to zero when the unit shortens to approximately 60% of its rest length (see Figure 6.17). However, this is unlikely to occur in practice due to the arrangement of the muscle–tendon units on the skeleton. When a muscle-tendon unit contracts in a very shortened state and, as such, generates low force, it is said to be in a state of active insufficiency (Elftman 1966). This is more likely to occur with muscle–tendon units that cross more than one joint. For example, the hamstrings extend the hip and flex the knee (see Figure 2.5). However, the muscle fibres in the hamstrings are not long enough to fully extend the hip and flex the knee simultaneously. If you stand on one leg and attempt to fully extend the hip and fully flex the knee of the other leg at the same time, you will be unable to flex the knee much more than 90°; i.e. the hamstrings will be in a state of active insufficiency. In contrast, if you flex the hip and then flex the knee, you will be able to flex the knee approximately 140°. This is possible because the hamstrings operate closer to rest length and are therefore able to exert more force (Elftman 1966).
Figure 6.17 shows that as a muscle–tendon unit lengthens beyond rest length, the isometric tension generated is fairly constant between 100% and 150% of rest length and then increases to a maximum at approximately 175% of rest length. The change in isometric tension between rest length and maximum tension is associated with a gradual decrease in the amount of tension produced by the contractile component and a gradual increase in the amount of passive tension. Tension in the contractile component would be reduced to zero if the muscle–tendon unit were lengthened to approximately 210% of rest length. However, the parallel elastic connective tissue components ensure that this situation does not arise by limiting the maximum length of the muscle–tendon unit to approximately 175% of rest length. In this situation, the muscle–tendon unit is said to be in a state of passive insufficiency (Elftman 1966).
When a muscle–tendon unit lengthens, all of the sarcomeres do not lengthen to the same extent. Theoretically a situation could occur where some of the sarcomeres in a muscle fibre were fully extended, i.e. no interdigitation between the actin and myosin filaments, while other sarcomeres were not fully extended. If the muscle were stimulated to contract in this state, the fully stretched sarcomeres would not be able to contract, whereas the other sarcomeres would be able to contract. This would result in further stretching and, consequently, damage to the already fully stretched sarcomeres. This theoretical condition has been referred to as muscle instability, and may be prevented, at least in part, by passive insufficiency (Alexander 1989). Consequently, the maximum working length range of a muscle–tendon unit is determined by the lengths at which active insufficiency and passive insufficiency occur, i.e. between approximately 75% and 175% of rest length. However, it is likely that the normal working range is between approximately 100% and 130% of rest length. This range incorporates the region of the length–tension curve where contractile tension is maximum and, as such, allows maximum flexibility in tension generation (see Figure 6.17).
Force-velocity relationship in a muscle–tendon unit
The everyday physical tasks that individuals perform are usually well within the strength capability of the muscle–tendon units used. In such movements, the muscle–tendon units generate just enough tension to overcome the external load acting on them so they can move the external load. The external load may simply be the weight of a limb segment such as the forearm in a movement involving elbow flexion. At other times the external load consists of the weight of the limb segments together with any additional load that is being moved such as something held in the hand.
When the amount of force produced by a muscle (muscle–tendon unit) just matches the external load, the muscle contracts isometrically. The maximum load the muscle can sustain isometrically is called the isometric strength of the muscle. When the external load is less than isometric strength, the muscle is able to contract concentrically. The speed of shortening in a concentric contraction depends on how much force the muscle needs to produce to move the external load. The greater the external load, the greater the muscle force needs to be, and the greater the muscle force (as a proportion of isometric strength), the slower the speed of shortening. A muscle can shorten at maximum speed when the external load on the muscle is zero. When the external load on a muscle is greater than the isometric strength of the muscle, it is forced to lengthen (contract eccentrically).
In an eccentric contraction a muscle resists the stretching load. In so doing, the attached cross bridges are themselves stretched, adding to the overall tension such that the force produced by the muscle is greater than the isometric strength of the muscle. The force produced by a muscle during eccentric contraction depends on the speed of lengthening, which depends on the size of the external load. The greater the external load (in relation to the isometric strength of the muscle), the greater the speed of lengthening. The greater the speed of lengthening, the greater the effect of the stretch reflex, and, therefore, the greater the force produced by the muscle. When the external force exceeds the maximum strength of the muscle, the muscle and its tendon will be damaged. The relationship between muscle force and speed of shortening or lengthening is referred to as the force–velocity relationship (Figure 6.18). Figure 6.19 shows the effect of the force–velocity relationship on the length–tension relationship of a muscle–tendon unit. The figure shows that at any particular length, the greater the speed of shortening, the lower the tension, and the greater the speed of lengthening, the higher the tension.
The amount of force generated by a muscle–tendon unit depends on the length of the muscle–tendon unit at the time of stimulation (length–tension relationship) and the speed with which it changes length in the ensuing contraction (force–velocity relationship)
Muscle architecture and function
All muscles are made up of muscle fibres. However, the length and the orientation of the fibres (pennate or non-pennate) have a considerable effect on the function of the muscles. The fundamental relationships between muscle architecture and muscle function are (1) excursion (the distance that the muscle can shorten) and velocity of shortening are proportional to fibre length, and (2) force is proportional to the total physiological cross-sectional area of the muscle fibres (Lieber & Bodine-Fowler 1993).
All muscle fibres are comprised of similar sarcomeres, and the number of sarcomeres determines the length of a muscle fibre. Each sarcomere in a muscle fibre is capable of shortening to the same extent as all the other sarcomeres in the muscle fibre. Consequently, the excursion of the muscle fibre is equal to the sum of the excursions of all the individual sarcomeres; the greater the number of sarcomeres, the longer the muscle fibre, the greater the excursion. Excursion and velocity of shortening are directly related since velocity of shortening is the rate of change of excursion, i.e. the rate of change in length of the muscle. The longer the muscle fibre (in terms of number of sarcomeres), the greater its excursion and velocity of shortening.
Theoretically, the ideal muscle (in terms of force and excursion capabilities) has a large cross-sectional area and very long fibres. However, such a muscle would be bulky and create considerable packing problems due to its girth and areas of attachment to the skeletal system. Since there are no muscles with both of these characteristics, it is reasonable to assume that the architecture of the muscular system has evolved to provide the best compromise between structure and function. The muscles of the body represent a broad range of combinations of force and excursion capability (Lieber 1992), and it is, perhaps, not surprising that most movements of the body involve simultaneous activity in a number of muscles with each muscle performing a particular role.
Figure 6.20A and C shows two muscle–tendon units with the same rest length and the same muscle mass (same volume of myofibrils). One muscle is a non-pennate parallel-fibred muscle (see Figure 6.20A) and the other is a unipennate muscle (see Figure 6.20C). The physiological cross-sectional area of each muscle is the cross-sectional area of all of the muscle fibres perpendicular to their line of pull. Assuming that the length of the muscle fibres in the pennate muscle is only half that of the fibres in the non-pennate muscle, it follows that the physiological cross-sectional area of the pennate muscle is double that of the non-pennate muscle (since the muscle mass is the same in both muscles). Consequently, the pennate muscle can exert double the force of the non-pennate muscle. However, as the fibres in the pennate muscle are oblique to the line of pull of the muscle–tendon unit, not all of the force is in the line of pull of the muscle–tendon unit. In a pennate muscle, the angle of the fibres with respect to the line of pull of the muscle–tendon unit is usually 30° or less (Alexander 1968), such that 90% or more of the force exerted by the muscle is directed in the line of pull of the muscle. Consequently, pennate muscles are normally capable of exerting far more force in the line of pull of the muscle–tendon unit than non-pennate muscles of the same muscle mass.
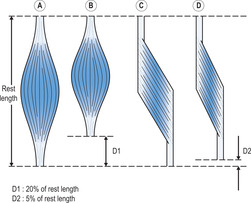 |
| Figure 6.20 |
In contrast to their capacity to generate force, the excursion range of non-pennate muscle–tendon units is usually much greater than that of pennate muscle–tendon units of similar muscle mass. The increased excursion range of non-pennate muscles is due to increased fibre length in non-pennate muscles and the obliquity of the fibres in pennate muscles, which reduces excursion in the line of pull of the muscle. In Figure 6.20B and D, the muscles are shown with their fibres shortened to 70% of their rest length. The corresponding shortening of the muscle–tendon units is 20% and 5% in the non-pennate and pennate muscles, respectively.
Biarticular muscles
Many of the skeletal muscles, especially those in the upper and lower limbs, span more than one joint. These muscles are usually referred to as biarticular muscles; muscles that span more than two joints function in the same way as muscles that span two joints (Lieber 1992). Biarticular muscles are too short to fully flex or fully extend simultaneously all of the joints that they span. For example, the hamstrings are two-joint muscles that can extend the hip and flex the knee, but (as described in a previous section) the hamstrings cannot perform these actions maximally at the same time. Indeed, hip extension is normally associated with knee extension, and hip flexion is normally associated with knee flexion, as in walking and running. In this way, the length of the muscles stay within the normal working range of approximately 100% to 130% of rest length. The functional advantages of biarticular muscles are that tension is produced in one muscle rather than two (or more), which conserves energy, and working within the 100% to 130% range allows maximal flexibility in tension generation (Lieber 1992; Van Ingen Schenau, Pratt & Macpherson 1994).
Review questions
1. Differentiate between the cerebrospinal nervous system and the central nervous system.
2. Differentiate between resting potential and action potential.
3. Describe the general organization of nerve tissue in the spinal cord and spinal nerves.
4. Describe the sequence of events within the nervous system in a typical voluntary movement.
5. Describe the various categories of pennate and non-pennate muscles.
6. Describe the sliding filament theory of muscular contraction.
7. Differentiate between kinaesthetic sense and proprioception.
8. Differentiate between active and passive insufficiency in skeletal muscle.
9. Describe the fundamental relationships between muscle architecture and muscle function.
References
Alexander, RMcN, Animal mechanics. (1968) Sidgwick and Jackson, London .
Alexander, RMcN, Muscles for the job, New Scientist 122 (1660) (1989) 50–53.
Beard, DJ; Dodd, CAF; Trundle, HR; Simpson, AHRW, Proprioception enhancement for anterior cruciate ligament deficiency, Journal of Bone and Joint Surgery 76B (1994) 654; 59.
Edman, KAP, Contractile performance of skeletal muscle fibres, In: (Editor: Komi, PV) Strength and power in sport (1992) Blackwell Scientific, Oxford, pp. 96–114.
Elftman, H, Biomechanics of muscle, Journal of Bone and Joint Surgery 48A (1966) 363; 77.
Enoka, RM, Neuromechanical basis of kinesiology. (1994) Human Kinetics, Champaign, IL .
Freeman, M; Wyke, B, Articular reflexes at the ankle joint: an electromyographic study of normal and abnormal reflexes of ankle joint mechanoreceptors upon reflex activity in the leg muscles, British Journal of Surgery 54 (1967) 990–1001.
Gamble, JG, The musculoskeletal system: physiological basics. (1988) Raven Press, New York .
Gandevia, SC; McClosky, DI; Burke, D, Kinesthetic signals and muscle contraction, Trends in Neuroscience 15 (1992) 62–65.
Garn, S; Newton, R, Kinesthetic awareness in subjects with multiple ankle sprains, Physical Therapy 68 (1988) 1667; 71.
Gordon, AM; Huxley, AF; Julian, FJ, The variation in isometric tension with sarcomere length in vertebrate muscle fibres, Journal of Physiology (London) 184 (1966) 170; 92.
Gregor, RJ, Skeletal muscle mechanics and movement, In: (Editor: Grabiner, MD) Current issues in biomechanics (1993) Human Kinetics, Champaign, IL, pp. 171–211.
Grigg, P, Peripheral neural mechanisms in proprioception, Journal of Sport Rehabilitation 3 (1994) 2–17.
Hall, MG; Ferrell, WR; Baxendale, RH; Hamblen, DL, Knee joint proprioception: threshold detection levels in healthy young subjects, Neuro-Orthopedics 15 (1994) 81–90.
Hall, MG; Ferrell, WR; Sturrock, DL; et al., The effect of the hypermobility syndrome on knee joint proprioception, British Journal of Rheumatology 34 (1995) 121; 25.
Huijing, PA, Mechanical muscle models, In: (Editor: Komi, PV) Strength and power in sport (1992) Blackwell Scientific, Oxford, p. 130; 50.
Huxley, HE; Hanson, J, Changes in the cross striations of muscle during contraction and stretch and their structural interpretation, Nature 173 (1954) 973; 77.
Kleinrensink, GJ; Stoeckart, R; Meulstee, J; et al., Medicine and Science in Sports and Exercise 26 (1994) 877; 83.
Lieber, RL, Skeletal muscle structure and function. (1992) Williams & Wilkins, Baltimore .
Lieber, RL; Bodine-Fowler, SC, Skeletal muscle mechanics: implications for rehabilitation, Physical Therapy 73 (1993) 844; 56.
Matthews, PB, Proprioceptors and their contribution to somatosensory mapping: complex messages require complex processing, Canadian Journal of Physiology and Pharmacology 66 (1988) 430; 38.
Noth, J, Motor units, In: (Editor: Komi, PV) Strength and power in sport (1992) Blackwell Scientific, Oxford, pp. 21–28.
Roberts, TDM, Understanding balance: the mechanics of posture and locomotion. (1995) Chapman and Hall, London .
Sargeant, AJ, Human power output and muscle fatigue, International Journal of Sports Medicine 15 (1994) 116; 21.
Seddon, JH, Surgical disorders of peripheral nerves. (1972) Livingstone, Edinburgh .
Skinner, HB; Wyatt, MP; Stone, ML; et al., Exercise-related knee joint laxity, American Journal of Sports Medicine 14 (1986) 30–34.
Steiner, ME; Grana, WA; Chillag, K; Schelberg-Karnes, E, The effect of exercise on anterior-posterior knee laxity, American Journal of Sports Medicine 14 (1986) 24–29.
Van Ingen Schenau, GJ; Pratt, CA; Macpherson, JM, Differential use and control of mono- and biarticular muscles, Human Movement Science 13 (1994) 495–517.
Wilkerson, GB; Nitz, AJ, Dynamic ankle stability: mechanical and neuromuscular relationships, Journal of Sport Rehabilitation 3 (1994) 43–57.
In: (Editors: Williams, PL; Bannister, LH; Berry, MM; et al.) Gray’s anatomy (1995) Longman, Edinburgh.