Chapter 4 The measurement of patient-reported outcome in the rheumatic diseases
KEY POINTS
 The measurement and communication of disease burden and the consequence of healthcare are essential components of healthcare. Patients have an important contribution to make to this process.
The measurement and communication of disease burden and the consequence of healthcare are essential components of healthcare. Patients have an important contribution to make to this process. Well-developed patient-reported outcome measures (PROMs) can provide a reliable, valid and clinically relevant resource for including the patient perspective in healthcare
Well-developed patient-reported outcome measures (PROMs) can provide a reliable, valid and clinically relevant resource for including the patient perspective in healthcare The effective incorporation of PROMs into routine practice requires training in the use and interpretation of PROMs and ongoing support to ensure the appropriate use of PROMs-related data. Further rigorous evaluation of the contribution of PROMs to routine practice is required
The effective incorporation of PROMs into routine practice requires training in the use and interpretation of PROMs and ongoing support to ensure the appropriate use of PROMs-related data. Further rigorous evaluation of the contribution of PROMs to routine practice is required Advances in technology, such as electronic data capture, may enhance the feasibility of PROM application, whilst the timely provision of scores may enhance the utility to routine practice. Demonstrating the cost effectiveness of incorporating PROMs into routine practice is essential to good practice and to attracting appropriate resources to support data collection
Advances in technology, such as electronic data capture, may enhance the feasibility of PROM application, whilst the timely provision of scores may enhance the utility to routine practice. Demonstrating the cost effectiveness of incorporating PROMs into routine practice is essential to good practice and to attracting appropriate resources to support data collectionINTRODUCTION
Most modern healthcare systems embrace patient-centred care, entailing care that is responsive to the needs, preferences and values of the individual (Hibbard 2003). However, the experience of health and illness, and outcomes of healthcare desired by patients are multifaceted and often uniquely individual (Quest et al 2003, Ramsey 2002). Facilitating the patient to effectively communicate these values is an important part of patient-centred care. Although ‘what matters’ as an outcome of healthcare may differ between members of the healthcare team, the patient has a significant role to play (Ganz 2002, Haywood 2006, Hibbard 2003). Failure to include the patients’ perspective may inadequately inform clinical decision-making, and provides a limited evaluation of healthcare. Whilst rheumatic diseases can affect survival, there is growing evidence demonstrating the significant negative impact on an individual’s quality of life (e.g. Chorus et al 2003, Finlay & Coles 1995). A wide range of health care interventions in rheumatology are concerned with reducing the burden of disease and improving the patients’ experience of ill-health, for example, alleviating pain and improving function, well-being or quality of life. Identifying the most important outcomes of care and the most appropriate methods of assessment is central to effective care delivery. Understanding patient-reported outcome, and the appropriate methods of measuring these outcomes, clearly has an important role to play in modern healthcare.
Across the rheumatic diseases, all therapeutic interventions require rigorous evaluation to provide an evidence-base for policy decision-making and clinical practice. This is of critical relevance to chronic conditions where treatment effects are often subtle and difficult to detect (Hobart et al 2004). Evaluation of healthcare can be used in several ways, for example:
This chapter will introduce the role of patient-reported outcome measures (PROMs) as an important mechanism to enhance patient participation in healthcare management and evaluation, with a particular focus on routine practice. Challenges to the use of PROMs are discussed. The chapter concludes by highlighting key areas for future debate and research into the effective use of PROMs of relevance to the rheumatology healthcare team.
MEASURING HEALTH
Health is a complex construct, the measurement of which should include issues of relevance to patients, healthcare professionals and providers. Measures of health outcome are often categorised as clinical or disease-focused, such as laboratory-based or radiographic assessment; or humanistic or patient-reported, such as health status and health-related quality of life (HRQL) (Ganz 2002, McHorney 2002). These two broad approaches to measurement provide wide ranging and complementary information, often of relevance to both patients and clinicians (Ganz 2002, McHorney 2002). Two classification frameworks are helpful for exploring the different concepts that can be measured: 1) the International Classification of Functioning, Disability and Health (ICF) (WHO 2001); and 2) Donabedian’s Structure, Process and Outcome (Donabedian 1966).
INTERNATIONAL CLASSIFICATION OF FUNCTIONING, DISABILITY AND HEALTH
The revised International Classification of Functioning, Disability and Health (ICF) (Stucki et al 2007, WHO 2001) provides a useful framework for understanding disease impact and measuring the outcomes of healthcare. The central concept of ‘functioning’ has been broadened to embrace a biopsychosocial model of health, describing health in terms of biological, psychological, social and personal factors (Dagfinrud et al 2005). Moreover, personal and environmental factors, acting as contextual facilitators or barriers, may further influence the impact of disease and its assessment (Dagfinrud et al 2005, Liang 2004).
The framework provides a dynamic and interactive model (Fig. 4.1), which complements traditional measures of ‘body structure’ and ‘body function’ (abnormalities or changes in bodily structure or function) with the broader assessment of ‘activity’ (abnormalities, changes or restrictions in an individual’s interaction with the physical context or environment) and ‘participation’ (changes, limitations or restrictions in an individual’s social context; involvement in a life situation, viewed from a societal perspective) (Wade & de Jong 2000).
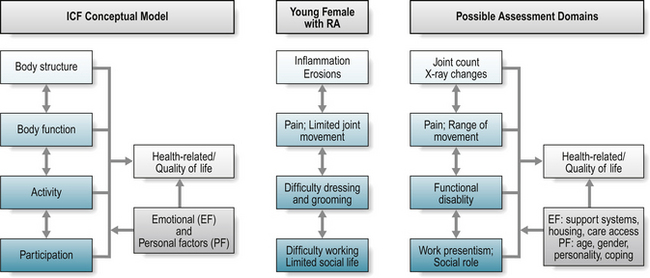
Figure 4.1 The International Classification of Function (ICF) Conceptual Model: illustrated with example of possible assessment domains for a woman with rheumatoid arthritis (RA).
Applying the relative burden of rheumatoid arthritis (RA) for a young female to the model (Fig. 4.1): RA may cause inflammation and joint damage in the small joints of the hand that can be detected by radiographic evaluation (body structure) and limitation in joint range of movement (body function). These changes may also further impact on an individual’s ability to partake in particular activities, for example, dressing and personal grooming, or playing a musical instrument (activity), and subsequent ability to maintain their desired work role or to socialise with friends (participation). Patients experience the impact of disease as problems relating to their health status and quality of life, and it is usual for a patient to present at a clinic with problems associated with limitation in specific activities or difficulties with participation, as outlined above. The combination of constructs reflects the person in his or her world (Van Echteld et al 2006), providing a systematic and comprehensive, more socially driven, approach to understanding the impact of ill-health (Wade & de Jong 2000).
The impact of ill-health, any associated assessment, and extent of information divulged by patients, may be further influenced by both environmental and/or personal factors, including the individual’s relationship with members of the healthcare team. Health professionals are important facilitators in enhancing environmental factors for people with a rheumatic disease (Van Echteld et al 2006) through, for example, the provision of appliances to facilitate an individual’s capacity for independence in dressing and grooming. Similarly, patients with ankylosing spondylitis (AS) identified family support as helpful (an environmental facilitator), whereas adverse weather was unhelpful (Van Echteld et al 2006). Personal factors include age, gender, coping mechanisms and personality, and are influenced by environmental factors, the level of social support, and individual factors including level of motivation, expectations for health and healthcare (Liang 2004).
The overall impact of these limitations may be captured within the broader concept of life quality – what do these limitations mean in terms of an individual’s quality of life? Within the proposed framework, quality of life, or health-related quality of life, forms a separate assessment domain (Wade & de Jong 2000). Standardisation of the ICF framework has supported the identification of key areas relevant to the wider understanding of ‘function’ (Stucki et al 2007, Van Echteld et al 2006), facilitated a better understanding and analysis of patient problems, and fostered communication between health professionals (Wade & de Jong 2000). In summary, the ICF provides a useful, conceptual model to guide appropriate assessment of what should be measured to capture the breadth of disease impact; it does not, however, direct how these health constructs, or domains, should be measured (Box 4.1).
BOX 4.1 Study activity: osteoporosis and the ICF framework
 What are the presenting complaints of this patient when you review them in a clinic setting? How do these presenting features fit within the revised ICF framework?
What are the presenting complaints of this patient when you review them in a clinic setting? How do these presenting features fit within the revised ICF framework?STRUCTURE, PROCESS AND OUTCOME
Traditionally, the quality of health care has been assessed in terms of Donabedian’s classic structure-process-outcome framework (Campbell et al 2000, Donabedian 1966, Mitchell & Lang 2004). Structure embraces the operational characteristics of a service, and includes patient, healthcare professional and organisational variables (Pringle & Doran 2003, Wade & de Jong 2000). Structure also considers the accessibility and relative quality of the many components of health care, for example, how accessible was care for an individual with a rheumatic disease? Process explores how the health care services work in relation to the nature and extent of the patients problem (Wade & de Jong 2000), and includes assessment of the appropriateness of care, location and timing. For example, did an individual with rheumatic disease receive care that was appropriate to their needs, at the right time, and in a suitable location? At the patient level, process measures may also include measures of disease process, such as radiographic imaging, histological and biomedical markers (Bellamy 2005), and patient-reported measures of health status.
Measures of outcome reflect the overall aim of the healthcare service, and may include functional and clinical outcomes, or clinical targets. Although important, global outcome measures such as mortality and morbidity rates are generally irrelevant to a patient’s experience and are generally unhelpful in communicating the broader impact of disease and success of healthcare (Clancy & Lawrence 2002). A necessary change of emphasis in assessment recognises the importance of measuring the quality of survival and the wider impact on an individual’s health-related quality of life. From an individual perspective, important outcomes may assess if, for example, healthcare has minimised their experience of pain and fatigue, assisted the individual in maximising their participation in his/her chosen social setting or work environment, or reduced the distress and pain experienced by the patient’s family and carers (Wade & de Jong 2000). Assessment that embraces the patient perspective has an important role to play in communicating their relative experience of disease and healthcare.
MEASURING HEALTH: THE PATIENTS’ PERSPECTIVE
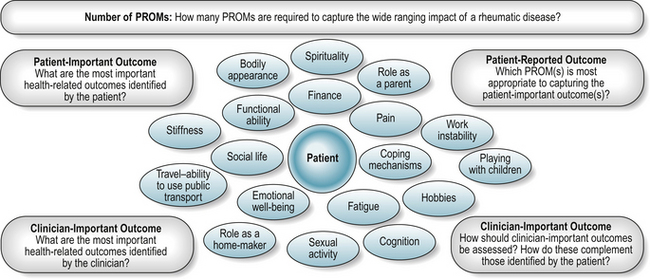
Challenges of including the patient’s voice in the assessment process include identifying the most important outcomes and the most appropriate and acceptable methods of assessment (Fig. 4.2, Box 4.2).
PATIENT-REPORTED OUTCOME AND PROMS: DEFINITION
A patient reported outcome (PRO) has recently been defined as ‘any report coming from patients about a health condition and its treatment’ (Burke et al 2006). PRO assessment is therefore broad ranging, and includes health-related quality of life, needs assessment, treatment satisfaction and treatment adherence.
Well-developed, relevant and scientifically rigorous patient-reported measures or patient-reported outcome measures (PROMs) provide a measure of an individual’s experiences and concerns in relation to their health, health care and quality of life (Fitzpatrick et al 1998, Ganz 2002). PROMs are largely self-completed questionnaires containing several questions, or items, to reflect the broad nature of health-related concerns. They are available to measure a wide range of health-related concepts such as pain, physical disability, treatment side-effects, satisfaction or experience of health care, health care costs, and quality of life (McDowell 2006). Although variously referred to as measures of quality of life, health-related quality of life (HRQL), or health status, measures of health status often have a narrower focus, for example, the assessment of pain, fatigue and functional disability, than measures of HRQL, which may include a broader range of health-related domains, for example, emotional well-being and work disability. Measures of quality of life should capture all aspects of life, including non-health related issues.
PROM development has traditionally been dominated by clinical experts who, although knowledgeable, cannot communicate the impact of ill-health and treatment in the same way as a patient (Fitzpatrick et al 1998). Furthermore, evidence of discrepant views between patients and health care providers in their assessment of important health outcomes (Hewlett 2003, Kessler & Ramsey 2002, Kvien & Heiberg 2003, Liang 2004) highlights the irrefutable need for simple and effective methods to enhance patient involvement in PROM development and evaluation. Application of the revised ICF framework has supported the comparative evaluation of available PROMs, and highlighted the often limited patient contribution to PROM development, the limited content of widely used PROMs, a lack of precision relating to the concepts assessed, and hence a limited ability to capture the broader concepts of patient participation (Van Echteld et al 2006). Where PROMs seek to communicate the patient voice, methods to ensure that patients reported outcomes embrace patient important outcomes are essential.
Once questions, or items, are generated to reflect a particular construct, such as fatigue, a measurement scale or set of response options are described for each item (Streiner & Norman 2003), such as a series of graded descriptive, or categorical, responses (i.e. no fatigue, mild, moderate, severe, very severe fatigue) with associated numerical values, or a horizontal visual analogue scale (VAS) with descriptive and numerical anchors (i.e. 0 ‘no fatigue’ to 100 ‘extreme fatigue’). The associated numerical scores may then be combined, usually summed, to give a final score. The addition of numerical values does not guarantee that the distance between successive categories is the same, that is, continuous or interval level data, and the majority of measurement response scales produce categorical data on an ordinal level of measurement. However, under most circumstances, and unless score distribution is severely abnormal, data can be analysed as if continuous or interval level data (McDowell 2006, Streiner & Norman 2003).
Proxy completion of questionnaires where relatives or clinicians complete the measure on behalf of the individual can be an important source of information particularly in the case of children (Varni et al 2005), chronically debilitated or cognitively impaired patients (Neumann et al 2000). Discrepancies between a proxy measure of health and that reported by an individual have been observed. Discrepancies are often greater for non-observable constructs such as pain and depression, and less for more observable constructs such as physical ability (Ball et al 2001).
PATIENT-REPORTED OUTCOME MEASURES: TYPOLOGY
Garratt et al (2002) describe four broad categories of PROM: generic, domain-specific, condition or population-specific, and individualised.
Generic
Generic measures contain multiple concepts of health of relevance to both patients and the general population, supporting comparison of health between different patient groups, and between patient groups and the general population (Haywood 2006). Population-based normal values can be calculated for generic instruments which supports data interpretation from disease-specific groups (Ware 1997).
Two distinct classes of generic instrument can be described: health profiles and utility measures. Scores on different domains of health covered by a single health profile are presented separately to support data interpretation, therefore reflecting a clinical perspective. Sometimes an index or summary score may be generated, but proponents of profiles argue that measurement is most meaningful within separate domains (McDowell & Newell 1996). The Short-Form 36-item Health Survey (SF-36) (Ware 1997, Ware et al 2002) (www.sf-36.org) is a widely used example of a generic health profile both generally and in patients with rheumatological or musculoskeletal conditions (Garratt et al 2002) (Table 4.1). The comparison of generic health status between members of the general population, and patients with RA or AS (matched for important factors such as age, gender, demographic, work and disease status) demonstrated that both patient groups had worse levels of physical (PCS) and mental (MCS) health when compared to the general population (Chorus et al 2003). When the two patient groups were compared, patients with RA reported worse levels of physical health, but better levels of mental health than patients with AS.
Table 4.1 Selected generic and specific patient-reported outcome measures (PROMs) of relevance to the rheumatic diseases
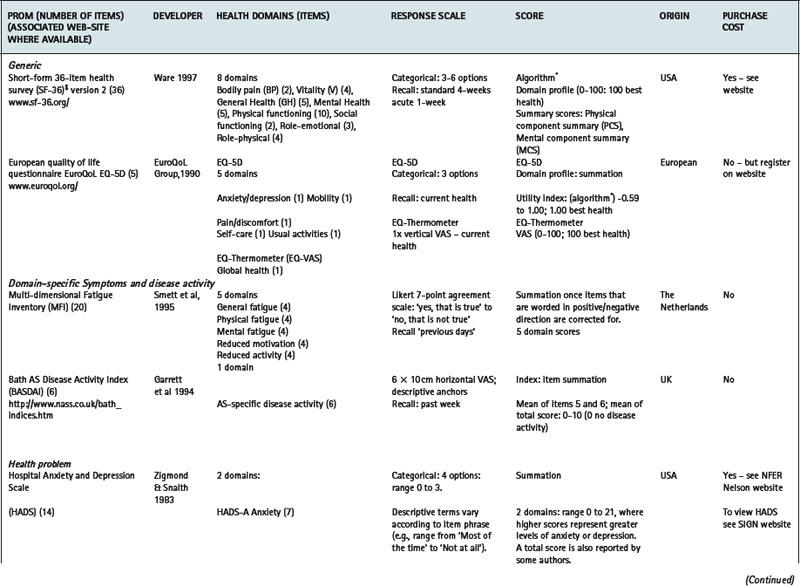
Utility measures incorporate the values and preferences for health outcome generated by the patient (direct weighting), reflecting the individual perspective, or the general population (indirect weighting), reflecting the societal perspective (Fitzpatrick et al 1998). Although utility measures usually include several domains of health, by providing numerical values for health states that use states of perfect health and death as reference values, utility measures can be used to generate single index values that combine health status with survival data – for example, to produce quality adjusted life years (QALYs). The EQ-5D is an example of a utility measure that incorporates indirect valuations of health states (EuroQolGroup 1990) (www.euroqol.org) (Table 4.1). Although widely used in the rheumatic diseases (Brazier et al 2004, Haywood et al 2002, Marra et al 2005), the EQ-5D has been criticised for having both limited item content and response options, and hence may be limited in detecting small, but important, changes in health in people with chronic ill-health (McDowell & Newell 1996), such as rheumatoid arthritis (Marra et al 2005) and ankylosing spondylitis (Haywood et al 2002).
Domain-specific PROMs
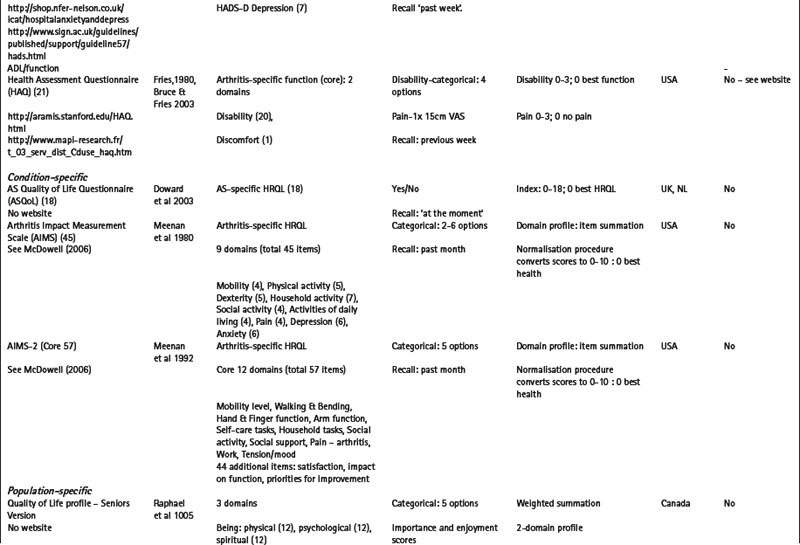
Measures may be specific to a health domain, such as fatigue, for example, the multi-dimensional fatigue inventory (MFI) (Smets et al 1995), or disease activity, for example, the Bath AS disease activity index (BASDAI) (Garrett et al 1994). Alternatively, they may be specific to a health problem, for example, the hospital anxiety and depression scale (HADS) (Zigmond & Snaith 1983) which is specific to depression and anxiety (Table 4.1), or a described function, for example, activities of daily living (McDowell 2006), such as the health assessment questionnaire (HAQ) (Bruce & Fries 2003, Fries et al 1980) (Table 4.1).
Condition or population-specific PROMs
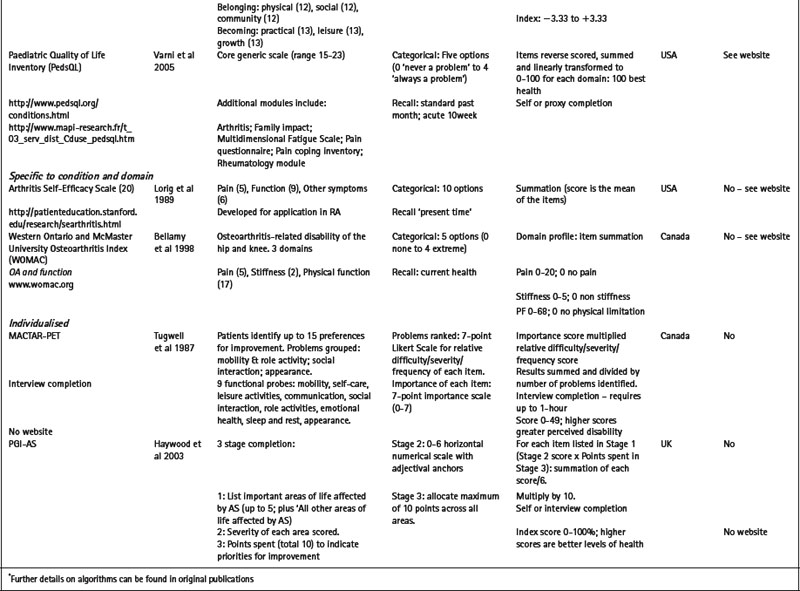
Alternatively, measures may be specific to a particular condition or disease, for example, the arthritis impact measurement scale version 2 (AIMS2) is specific to people with arthritis and assesses physical, emotional and social well-being (Meenan et al 1992); or to a patient population, for example, the quality of life profile – seniors version (QLP-SV) (Raphael et al 1995) is specific to the assessment of quality of life in older people. Some measures may be specific to a population with additional modules that provide further disease or symptom specific information: for example, the paediatric quality of life inventory (PedsQL) (Varni et al 2005) has a core generic scale for the assessment of paediatric quality of life, with additional modules for fatigue (PedsQL multidimensional fatigue scale) and rheumatology-related problems (PedsQL rheumatology module pain and hurt scale) (Varni et al 2007). Numerous measures are specific to a condition and a health domain. For example, the arthritis self-efficacy scale (Lorig et al 1989) is specific to the assessment of self-efficacy in patients with rheumatoid arthritis, and the Western Ontario and McMaster university osteoarthritis index (WOMAC) is specific to the assessment of functional ability in lower limb osteoarthritis (Bellamy et al 1988) (Table 4.1).
Individualised
The majority of conventional PROMs are highly standardized questionnaires with a pre-determined set of items and response options. Although standardized questionnaires often have good measurement properties (Fitzpatrick et al 1998), they may omit issues of importance to individual patients, while containing items of little relevance to others. Measures that adopt a more individualised approach, such as the schedule for the evaluation of individual quality of life (SEIQoL) (McGee et al 1991) and the patient generated index (Martin et al 2007, Ruta et al 1994), support the incorporation an individual’s problems and priorities in the assessment process. Evidence suggests that these measures have enhanced content validity in comparison to more standardized measures (Haywood et al 2003), and that patients’ view them as more valid assessments of health (Neudert et al 2001).
Several individualised measures have encouraging evidence of required measurement properties following completion by patients with a range of rheumatic diseases including the disease repercussion profile (DRP) (RA) (Carr & Thompson 1994), the MACTAR-patient elicitation technique (MACTAR-PET) (RA) (Tugwell et al 1987), the personal impact health assessment questionnaire (PI HAQ) (RA) (Hewlett et al 2002) and the PGI (AS) (Haywood et al 2003) (Table 4.1).
The respondent burden associated with individualised measures is often greater than that observed for more standardized approaches, and several measures require interview administration. As a consequence, the application of individualised measures in clinical research is limited (Patel et al 2003). However, the enhanced content validity and relevance to patients suggests that individualised measures may have an important role to play in routine practice (Greenhalgh et al 2005, Marshall et al 2006) but to date there are no published trials that rigorously evaluate the role of individualised measures in this setting (Marshall et al 2006).
Condition-specific, scientifically rigorous PROMs that have involved patients in item generation have greater clinical relevance, are more acceptable to patients, and more responsive to change in health than generic measures (Wiebe et al 2003). However, the broad content of generic measures supports the identification of co-morbid features and unexpected treatment side-effects that may not be identified by specific measures. Furthermore, certain generic measures, such as the EQ-5D (EuroQoL Group 1990) and the SF-6D generated from the SF-36 version 2 (Ware et al 2002, Marra et al 2004, 2005), can inform economic evaluations of service delivery. The combination of generic and specific measures has been recommended in health outcome assessment (McDowell & Newell 1996), and together are an essential element in the development of evidence-based healthcare. Combined, the evidence supports determination of the absolute and comparative effectiveness of interventions, their marginal relative benefits, and the economic implications of long-term therapies. However, the optimal combination of measures has not been determined in a way that makes it easy to implement this recommendation across diverse patient groups. A fundamental consideration is the appropriateness of each measure to the proposed application.
PROMs: QUALITY ASSESSMENT
The measurement of patient-reported outcome in healthcare has enjoyed increasing attention over the last 25 years and as a result several hundred measures are now available. For many of the rheumatic diseases there is often a choice (Garratt et al 2002). However, the quality of PROMs varies widely, and guidance or consensus on selection is often lacking. The appropriate selection of an outcome measure should be guided by a wide range of measurement (reliability, validity, responsiveness to clinically important change, precision, interpretation) and practical issues (appropriateness, acceptability, feasibility). These concepts have been detailed by several authors (Fitzpatrick et al 1998, Haywood 2008, McDowell 2006), and are summarised in Tables 4.2 and 4.3.
Table 4.2 Criteria for selecting patient-reported outcome measures (PROMs): Measurement properties
| CRITERION* | DEFINITION | EVALUATION |
|---|---|---|
| Reliability (OMERACT filter ‘discrimination;) | ||
| Temporal stability-are the results stable over time? | Test-retest reliability assesses score temporal stability (correlation coefficient); scores should remain unchanged in stable patients | |
| Are the scores internally consistent? (multi-item measures only) | Internal consistency reliability evaluates the ability of items to measure a single underlying health domain (Cronbachs alpha). | |
| Reliability estimates > 0.70 and 0.90 recommended for group and individual assessment respectively | ||
| Validity (OMERACT filter ‘truth’) | ||
| Does the PROM measure what it claims to measure? | ||
| Content validity – how well do items cover the important parts of health to be assessed? | Qualitative appraisal of item content. | |
| Face validity – what do the items appear to measure? | How were items generated? Role of clinicians, patients etc? | |
| Qualitative appraisal of item content. | ||
| How were items generated? Role of clinicians, patients etc? | ||
| Criterion validity – how well does the measure perform against a gold-standard measure of the same construct? | Quantitative evaluation-comparative evaluation (if gold-standard available) | |
| Construct validity | ||
| – does the PROM show convergence/divergence with appropriate variables? | Quantitative comparison with other variables (health, clinical, socio-demographic, service use) – e.g. what are the hypothesised and actual levels of correlation with other variables? | |
| – does the PROM show divergence between groups of patients? | Quantitative evidence of discriminative ability between defined patient (extreme) groups. For example, pain levels between active and non-active disease groups/general population. | |
| – is there evidence to support the hypothesised domain structure? | Internal construct validity (dimensionality)-statistical methods such as factor analysis | |
| Responsiveness (OMERACT filter ‘discrimination;) | ||
| Does the PROM detect change over time that matters to patients? | Change in health following an intervention or over time. | |
| (sometimes referred to as ‘sensitivity to change’) | Distribution-based assessment-relates score change to some measure of variability; e.g. Effect Size statistics. | |
| Anchor-based assessment-relationship between score change and external variable: e.g. patient’s perception of change | ||
| Precision | ||
| How precisely do the PROM numerical values relate to the underlying spectrum of health, and discriminate between respondents in relation to their health? | Data quality and precision influenced by item coverage and response categories; where more than 20% of respondents have maximum bad or good health scores, score distribution indicates floor or ceiling effects respectively, and hence a lack of precision | |
| (sometimes referred to as ‘sensitivity’) | ||
| Interpretability (included in OMERACT filter ‘feasibility’) | ||
| How interpretable are the scores? | Distribution-based – describe change over time and group differences that are likely to be clinically meaningful. | |
| How do the scores relate to meaningful or worthwhile change in health? | Anchor-based-relate score change or differences between groups to external variables to estimate ‘Minimal Important Difference’: e.g. patient or clinician perception of change | |
OMERACT headings (Bellamy 1999)
* Criterion headings informed by Fitzpatrick et al (1998) and Haywood (2007).
Table 4.3 Criteria for selecting patient-reported outcome measures (PROMs): practical properties
| CRITERION * | DEFINITION | EVALUATION |
|---|---|---|
| Appropriateness | ||
| How well does the intended purpose of the PROM meet the intended application? Is it relevant? | For example, if the primary intent of the intervention is to reduce pain, pain should be an important component of a primary outcome measure | |
| If the patient has indicated that their ability to engage in paid employment is their most important outcome, how appropriate is the selected PROM to communicate this outcome? | ||
| Acceptability | ||
| Is the PROM acceptable to patients? How willing or capable of completing the PROM are the patients? Is it relevant? |
Most readily assessed through completion/response rates, and missing values. Difficult to evaluate directly. Focus groups with patients are useful for exploring acceptability. Extensive involvement of patients during PROM development should improve acceptability. | |
| Feasibility (OMERACT filter ‘feasibility’) | ||
| What time and resources are needed to collect, process and analyse the PROM? | Specific to the requirements of clinicians, researchers, and other staff. | |
| Is the PROM self-completed or does it require interview administration? How long does completion require? Is completion appropriate to the available setting? | ||
| Are instructions for completion and scoring provided? Is special equipment, for example, computer completion, required or available for completion and scoring? How accessible is such equipment? | ||
| How quickly can scores be generated? How are results presented to clinicians and/or patients? – for example, graphic representation of scores. | ||
| If the scores are required for face-to-face discussion during the patient consultation, how quickly can the PROM be scored, analysed and fed-back to the healthcare professional and the patient? | ||
* Criterion headings taken from Fitzpatrick et al (1998) and Haywood (2007)
The field of rheumatology benefits greatly from the work of Outcome measures in rheumatology and clinical trials (OMERACT: www.omeract.org). OMERACT recommend the application of a slightly different set of criteria, or ‘filter’, when assessing the appropriateness of PROM for application in defined settings (Bellamy 1999) (Tables 4.2, 4.3). The filter summarises measurement and practical properties under three headings: 1) Truth: is the measure truthful, does it measure what it purports to measure? Is the result unbiased and relevant? This concept relates to issues of face, content, construct and criterion validity; 2) Discrimination: does the measure discriminate between health states or situations that are of interest? The situations can be states at one time (for classification or prognosis) or states at different times (to measure change). The concept is related to issues of measurement reliability and responsiveness; and 3) Feasibility: can the measure be applied easily, given constraints of time, money, and interpretability? The concept of feasibility is an essential element in measurement selection.
It is essential that users are aware of these key properties to support appropriate selection, interpretation and communication of data generated. In selecting a PROM, the user must decide what exactly is required in terms of the proposed application, appropriateness to the patient (population) and setting, and the feasibility of application and scoring. In short, an appropriate balance between required detail, accuracy of assessment and the burden of collecting the information must be sought (McDowell 2006), as summarised in Table 4.4.
Table 4.4 Assessing the appropriateness of a patient-reported outcome measure (PROM) to the proposed application
| QUESTION | WHAT SHOULD BE CONSIDERED? |
|---|---|
| What is the proposed application for the PROM? | |
| In what setting is the PROM to be applied?-routine practice; clinical research; health policy; quality of care evaluation | |
| Will the data to be used to assess current disease state? | |
| Will the data be used to assess disease progress over time? | |
| Will the data be used to evaluate a programme of care? Are group level or individual level data required? | |
| Will the data be used to communicate the impact of healthcare to interested stakeholders? What outcomes are viewed as important indicators of care? | |
| Will the PROM be used to enhance patient involvement in the consultation process? | |
| What patient characteristics will influence PROM selection? | |
| Disease-specificity and co-morbidity | |
| Age – ability to self-complete; need for assistance; need for proxy completion. | |
| Level of disability – ability to self-complete; need for assistance; need for proxy completion | |
| Tolerance for completion – the relative length of PROM and associated respondent burden | |
| What is the assessment time frame? | |
| Will the assessment be cross-sectional, or longitudinal with multiple assessment points? | |
| What is the PROM recall period and how does this relate to the proposed application? | |
| Will the PROM be used to assess acute symptoms? This requires measurement that is responsive to clinically important change over relatively short time-frames | |
| Will the PROM be used to assess the long-term impact of a condition? The longer-term effects of disease impact requires measurement that is sensitive to small changes over relatively long time-frames | |
| How complex/simple should the assessment be? | |
| What detail is required? How broad ranging is the assessment provided? | |
| To whom will the results of the assessment be communicated? What is the important outcome to be communicated? | |
| How feasible is it to include the assessment of Body Structure, Body Function, Activity and Participation? | |
| What is the relevance of the proposed assessment to the clinical question? | |
| Is the data easily interpreted? For example, does the PROM produce a single index scores and/ or a profile score over several domains. | |
| What facilities are required to support the assessment? | |
| In what setting is the PROM to be applied? Clinic-based; trial-based completion; patient self-completion | |
| What support is required for PROM completion and scoring? For example, paper-based completion; electronic data capture and scoring; computer-based support etc. | |
| What is the associated cost of PROM completion, scoring, data feedback and administration support; plus computer support where appropriate. | |
| What facilities are required/available to support the real-time feedback of scores to inform shared clinical decision-making. | |
GUIDANCE FOR PROM SELECTION IN THE RHEUMATIC DISEASES
Working groups
Several resources now exist to support healthcare professionals in the selection and application of PROMs. OMERACT consists of several international working groups dedicated to improving measurement across the rheumatic diseases. In recent years a wider range of health professionals and patients have participated in the process, with a significant impact on the identification of patient-important outcomes. Using a data-driven, iterative consensus approach, core assessment domains have been recommended across a range of conditions including RA (Tugwell & Boers 1993), AS (van der Heijde et al 1997,1999), osteoarthritis (Bellamy et al 1997), osteoporosis (Sambrook 1997), systemic lupus erythematosus (Smolen et al 1999), systemic sclerosis (Merkel et al 2003) and fibromyalgia (Mease et al 2007) (Table 4.5).
Table 4.5 OMERACT recommended core domains for rheumatoid arthritis, ankylosing spondylitis and osteoarthritis
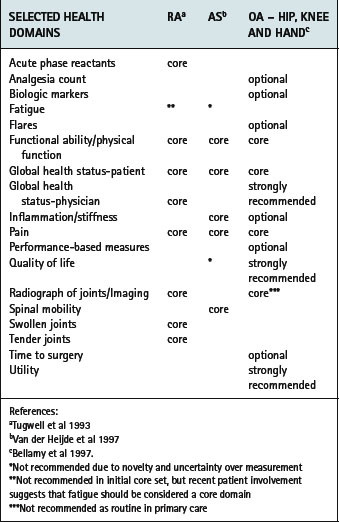
For some groups associated outcome measures (for example, AS (van der Heijde et al 1997, van der Heijde et al 1999)) and guidance for the interpretation of improvement have been recommended (for example, AS (Anderson et al 2001, Van Tubergen et al 2003)). However, the majority of the initial recommendations did not involve patients in the identification of core domains. Recent patient involvement has raised the importance of including fatigue as a core patient-centred assessment domain (Kirwan & Hewlett 2007, Kirwan et al 2007). Moreover, significant developments in the measurement of health outcome in recent years suggest that core domains should be revisited, for example, the assessment of fatigue and quality of life in AS. Application of the revised ICF framework (Stucki et al 2007) and accessing the views of patients will be invaluable to further exploring the core concept of what should be measured in the rheumatic diseases.
Special interest groups within OMERACT have been established, including disease-specific groups such as the Assessment in AS (ASAS) group (www.asas-group.org), the group for research and assessment of psoriasis and psoriatic arthritis (GRAPPA) (www.grappanetwork.org/) (Gladman et al 2007), the OMERACT/OARSI initiative for osteoarthritis (www.oarsi.org/) (Pham et al 2003) and the fibromyalgia research and outcomes group (Mease et al 2007). Other special interest or task groups are listed on the OMERACT web-site and associated publications. There are additional task groups exploring concepts such as work productivity (Escorpizo et al 2007), assessing single joints (Giles et al 2007), biomarkers (Keeling et al 2007), imaging-related concepts, and the Effective Musculoskeletal Consumer (Kristjansson et al 2007). A focus is also provided for measurement specific concepts including assessment of the minimal clinical importance (MCI) and computerised adaptive testing (Chakravarty et al 2007). These groups often run assessment workshops linked to main rheumatology conferences.
Structured reviews
Clearly, selecting an appropriate PROM can be a time-consuming process. The growing availability of structured reviews of PROM performance in different patient populations of relevance to the rheumatic diseases, for example, low back pain (Bombardier 2000), knee-specific PROMs (Garratt et al 2004), ankylosing spondylitis (Haywood et al 2005), shoulder disability (Bot et al 2004) and the assessment of fatigue in RA (Hewlett et al 2007) can be useful assets to health practitioners seeking guidance for measurement selection.
Web-resources
In addition to the OMERACT related sites, a range of on-line resources exist to support the identification and selection of PROMs. For example, the patient-reported health instruments database hosted by the national centre for health outcomes development (NCHOD, PHI programme), Oxford University (http://phi.uhce.ox.ac.uk/), the patient-reported outcome and quality of life instruments database (http://www.qolid.org/), the MAPI research institute (http://www.mapi-research.fr/index.htm) and the international society for quality of life research (http://www.isoqol.org/).
At the core of the PHI programme is a freely accessible website with a range of facilities, including a searchable bibliography, designed to support the identification, selection and application of PROMs. The website also provides summaries of published reviews of PROMs and evidence of expert consensus opinion across a growing range of disease and population-specific areas, including those of relevance to musculoskeletal practitioners. The PROQoLID and MAPI websites provide limited free access to a range of published PROMs, providing details of the developers, purpose, number of questions, and administration. Additional information can be accessed for a fee. Additional, selected websites of relevance to PROMs assessment are are available from the ‘Links’ page of the PHI Programme website (http://phi.uhce.ox.ac.uk/)
Individual PROMs
Many of the widely available PROMs have dedicated websites, as listed in Table 4.1. An up-dated list of these resources is available from the ‘links’ page of the PHI programme website (http://phi.uhce.ox.ac.uk/) (Box 4.3).
BOX 4.3 Study activity: identifying and selecting PROMs for a patient with osteoarthritis
A fifty-year old male long-distance lorry driver with moderate OA of the knee complains of pain and stiffness in his knees, and a difficulty getting in/out of the cab of his HGV. This is affecting his work: his knee pain increases during long drives and is finding it difficult to assist in loading/unloading the lorry. He is worried about the impact on his effectiveness at work.
The patient indicates that the most important outcomes of care are to reduce his pain and stiffness, particularly when driving for long periods, to improve his physical function so that he can get in/ out of his cab more readily. Ultimately, he hopes to improve his effectiveness at work.
PROMs – APPLICATIONS
PROMs have been applied in research settings, including population-based health surveys and clinical trials, for several decades (Haywood 2006). However, over recent years there has been growing interest in the contribution to be made to routine practice settings. Application in routine practice will provide the focus of this section.
ROUTINE PRACTICE
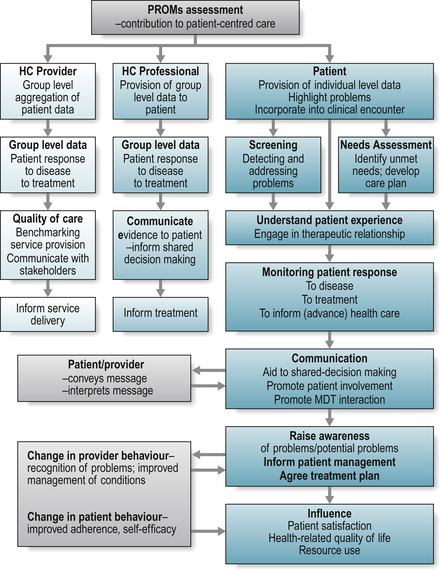
Enquiring about a patients’ perception of health is a familiar part of the clinical encounter for most health professionals. Although medical care has historically concentrated on treating impairments to body structure and function, a patient’s complaint is usually expressed in terms of their functional limitation and the wider impact on their ability to participate in society (Fig. 4.1). Increasingly, most clinicians recognise the importance of treating the patient as a whole, and not just treating the disease. Therefore, enquiring about the broader impact of disease, and identifying patient-important outcomes of care, is essential to patient-centred care (Fig. 4.2). Most simply, PROMs enable patients to report the impact of health problems and report the course and outcome of interventions in accessible and meaningful ways.
Historically, clinicians have adopted a non-standardized approach to assessment, and although often helpful in developing a rapport between patient and clinician, it is unlikely that the information elicited is of benefit beyond the personal encounter (Morris et al 1998). Moreover, such non-standardized approaches have limited value in communicating the impact of health and associated healthcare, and in comparing health status between individuals. Members of the multidisciplinary healthcare team often establish individual relationships with patients, which provide the potential to elicit aspects of a patient’s experience that may not necessarily be reported to other members of the team (Zebrack 2007). Standardized PROMs may provide a common language to facilitate communication between team members.
Three distinct applications of PROMs-related data of relevance to routine practice can be described (Fig. 4.3):
Health care quality and service delivery
The assessment of health care quality has historically focused on the perspectives of the care-provider or healthcare organization, such as cost, length of stay and patient mortality, and within a chronic disease context few assessments have included the patient’s experience of care (Groves & Wagner 2005). With the increasing culture of accountability, clinicians and patients are concerned that health care is both efficient and of high quality. At a managerial and policy level, measures of health outcome support the comparison of costs and benefits of competing health care programmes, where managers will seek to provide the best health care for the best price (Fig. 4.3). Rationing of healthcare is the inevitable consequence of limited resources, and the use of well-developed, responsive PROMs may provide beneficial information to support the distribution of healthcare resources (Haywood 2006, Department of Health 2008). Generic measures may be strategically important in health policy, particularly those that consider consumer needs and preferences, by supporting the comparison of health status and economic evaluation across population groups. For example, generic measures may support an evaluation of the relative health burden of people with OA on a waiting list for a total hip replacement versus those with RA waiting for access to hydrotherapy services.
Moreover, PROMs provide complementary evidence to more traditional measures of process and outcome, providing a more holistic assessment of the impact of service delivery. For example, aggregated data may assist the health care provider in identifying a common patient concern that requires, or suggests, a more systematic approach to health care provision (Fig. 4.3). Although limited empirical evidence for the performance of PROMs in quality of care evaluation exists (Appleby & Devlin 2004, Mitchell & Lang 2004), well-developed PROMs have the potential to provide reliable and valid group level data supporting the routine monitoring of patients health status for the purposes of quality assessment and, where appropriate, the comparative benchmarking of clinic performance (Appleby & Devlin 2004, Department of Health 2008) (Fig. 4.3). The simple format of most measures contributes to the acceptability and feasibility required for routine data collection, for example, measures may be self-completed in a clinic or ward setting (Haywood 2006).
Communicating the impact of disease and healthcare from group level data
With the advent of novel methods of service delivery, for example, consultant therapists, and new treatments for the rheumatic diseases with potentially toxic side-effects, patient acceptability and experience are an important determinant of overall effectiveness (Haywood 2006). Well-developed PROMs provide a powerful, quantifiable and standardized research tool, for use alongside more traditional methods of assessment, against which the effectiveness of health care interventions can be judged. Such measures have the potential to facilitate better research and to increase scientific knowledge about patient outcomes, and are increasingly accepted as valid measures of treatment effectiveness (Fitzpatrick et al 1998). Regulatory bodies, including the National Institute for Health and Clinical Excellence (NICE 2004) and the Federal Drug Administration (FDA) (Burke et al 2006), have provided guidance on the application of PROMs in support of treatment effectiveness. When applied in population-based surveys PROMs provide a important mechanism for assessing population and community health status, illustrating the wide ranging burden of disease and the associated unmet needs. Guidance to support PROM selection for clinical research has been detailed by several authors (Burke et al 2006, Fitzpatrick et al 1998, Haywood 2006).
Where measured outcomes have increased relevance to routine practice, with demonstrated impact on patient outcome, this may facilitate the translation of knowledge into routine practice. Healthcare professionals may utilise group level PROM data from research to provide evidence of other patients’ experience of the disease and associated healthcare. These data are expected to enable patients to develop a better understanding of the expected disease and healthcare impact in relation to the patient-reported variables, and hence to support shared decision-making (Fig. 4.3). Patient completion of PROMs in a routine practice setting may enhance their understanding and familiarity with such data.
Patient completion of PROMs in routine practice
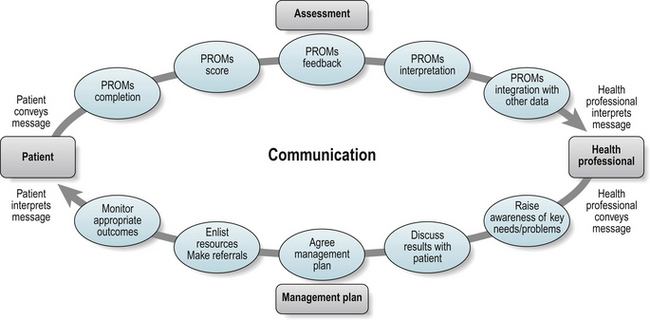
Well-developed, standardized PROMs provide a systematic approach towards the elicitation, recording and monitoring of patient-generated information in routine practice (Figs. 4.3,4.4). Such data may inform health screening (Gilbody et al 2003, Higginson & Carr 2001, McHorney 2002), the identification of patient needs and preferences (Higginson & Carr 2001, McHorney 2002), contribute to shared decision-making, and the regular monitoring of disease impact (Higginson & Carr 2001, McHorney 2002, Skevington et al 2005), and ultimately inform and advance healthcare. Moreover, there is growing evidence that PROMs may further enhance communication between health professionals and individual patients (Fig. 4.4), and even influence a patients sense of well-being (Haywood et al 2006, Marshall et al 2006, Velikova et al 2004) (Fig. 4.3). The growing evidence in support of the role of PROMs suggests that they should be viewed as integral to routine practice, adding to and complementing the broad range of information captured from other sources, including traditional biomedical assessment, other members of the healthcare team, and family members.
Support for the potential contribution of PROMs to the consultation process stems from arguments that such measures play a diverse role in changing how health problems are viewed and managed by patients and their healthcare providers (Figs 4.3, 4.4). The introduction of PROMs may encourage patients to share health-related problems that concern them in addition to ‘symptoms’ elicited in traditional consultations. PROMs may also act as a trigger so that potential problems or concerns are raised earlier in the consultation process. Similarly, health professionals are stimulated to think outside of conventional limitations in identifying problems and selecting solutions jointly with patients. Especially in the context of chronic disease, PROMs may have a powerful role to play over time in facilitating shared, and timely, identification of goals and priorities between health professionals and patients faced with complex, evolving and multi-faceted problems (Marshall et al 2006).
However, as recently argued by several authors (Greenhalgh et al 2005, Haywood et al 2006, Marshall et al 2006), the evaluation of PROMs in routine practice settings needs to be far more thoughtfully designed to take account of the likely real world contribution of measures designed to enhance patient involvement. Until more appropriate evaluative studies have been conducted it is premature to reach definitive conclusions on the advantages of PROMs in routine practice. Highlighting the utility of PROMs in identifying the health needs, priorities and preferences of patients, whilst demonstrating the potential cost advantages from PROMs application by facilitating the provision of timely and appropriate interventions, may ultimately provide the driving force for incorporating PROMs into routine clinical practice (Haywood et al 2008, Varni et al 2005).
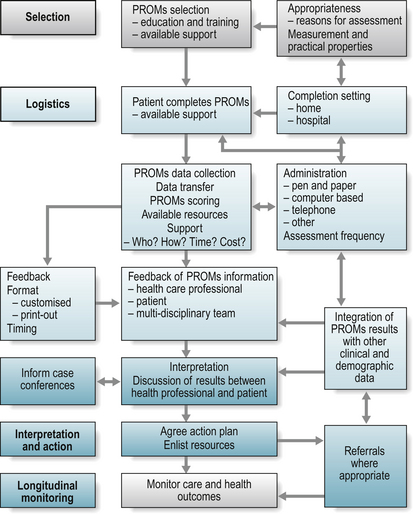
Four key phases describing the decision-making process required when including PROMs application in routine practice can be described (Fig 4.5): 1) PROM selection; 2) the logistics of PROM application, scoring and feedback of data; 3) interpretation of results and associated action plan; and 4) longitudinal monitoring of care.
Selecting PROMs for use in routine practice
The clinical and patient perceived relevance of a PROM is central to PROM selection and application (Figs. 4.2, 4.5, Tables 4.2-4.4). PROMs should beneficially influence the care process by informing diagnosis and/or care planning. Selection therefore requires that they are appropriate to the primary clinical question. Similarly, the patient value placed on the outcome is central to understanding how ill-health or the outcomes of care affects their life: just because a PROM is completed by the patient, does not mean that it measures what is important to the patient. A wide range of constructs are necessary to explore the whole extent of a patient’s experience of a chronic rheumatic disease (Fig. 4.2), and there are many available PROMs to address these constructs (Garratt et al 2002). Therefore, selecting those most appropriate to the routine practice setting is crucial. Where the PROM supports the identification of ‘what matters’ to the patient it should contribute to the care process.
LOGISTICS OF APPLYING PROMS IN ROUTINE PRACTICE
Once the decision to include PROMs in routine practice has been made, attention to the logistics of collecting the required information is necessary to ensure that the application will serve the intended purpose. Two key issues should be considered (Fig. 4.5) (Bezjak 2007):
METHOD OF PROM ADMINISTRATION
Multiple methods for the administration and feedback of results from PROMs exist, including traditional pen and paper completion, and a growing range of methods supporting electronic data capture (Table 4.6). The choice of method should be informed by the reasons for PROM application, the rapidity with which results are required, the frequency with which assessments will occur, and available resources to support application, analysis and feedback of data. For example, if the application demands the immediate availability of scores to inform individual decision-making, some form of computer-based assessment is likely to be most appropriate. Similarly, if it is preferable for PROM results to be linked to other clinical and demographic data, a more sophisticated computer-based application may be preferred. Computer-based completion will also support integration of scores into the electronic record, more rapid aggregation of data, and customised feedback of data. Although pen and paper completion of PROMs is cheaper initially, such completion may be associated with increased financial and time costs in relation to scoring and feedback of data. The delayed and often limited presentation of results may also reduce data utility. Although the electronic capture of PROM information has start-up costs associated with the initial investment in soft and hard-ware, methods for data entry, data analysis and data feedback are integrated into the system and hence relative costs are reduced. However, continued technical support is essential.
Table 4.6 Modes of patient-reported outcome measure (PROM) administration
| ADMINISTRATION MODE | POSITIVES | NEGATIVES |
|---|---|---|
| Non-computer based completion | ||
| Pen and paper | Mobile | Potential high costs for data entry |
| Simple | Potential delay in data feedback | |
| Cheap to copy and integrate into practice | Limited presentation of scores | |
| Familiar | ||
| Computer-based completion | ||
| Computer touch-screen | Independent completion | Initial purchase cost |
| Low administration demands | Ongoing support essential – human resource cost | |
| Potential for cost-saving data collection | Subject to regular hard-ware changes | |
| Increasing familiarity | ||
| Potential for rapid data transfer and scoring – supporting real time provision of scores | ||
| Potential for integration with other data and with electronic record | ||
| Attractive output features | ||
| Palm-pilot/ Personal digital assistant | As above | As above: Plus small display screen and small pen for entering response |
| Tablet-personal computer | As above: Plus large display screen |
As above |
| Smart pen technology | As above: Plus easy to use – built-in scanner |
As above |
| Voice-interactive technology | As above: Plus voice-interactivity reduces potential difficulties with pens or touch-screen data entry |
As above |
| Telephone completion | Touch-pad completion Interview completion |
|
| Internet completion | ||
| Home-based computer – internet access | Independent completion | Data protection |
| Real-time collaboration | ||
| Low administration demands Increasing familiarity | ||
| Supports self-management | ||
| Other | ||
| Computer adaptive testing (CAT) | Reduced respondent burden. Increased precision of assessment | Not yet widely available |
The needs and disabilities of patients will inevitably inform administration modes. For example, patients with advanced RA may find it difficult to hold a fine pen, causing difficulties with self-completion of pen and paper formats or use of a palm-pilot/hand-held computer. Assistance may be required if pen and paper completion is the only available format. Alternatively, touch-screen technology may be more appropriate. It is rare for one system to accommodate the needs of all patients within routine practice and greater degrees of freedom in relation to the selected modes of administration are possible than within a research setting. It may be appropriate to combine modes to find the most feasible and most efficient for the encounter, to reduce burden and increase efficiency for patient and clinician. When pen-and-paper completion and computer-based completion are compared within the same setting, anecdotal evidence suggests that there is very little difference in terms of completion rates and level of acceptance, but scoring, analysis and feedback is easier following computer-based completion (Bezjak 2007).
The increasing availability of home computers supports the self-monitoring of disease via internet completion of PROMs, the results of which can be linked with disease self-management programmes (Van Stel et al 2007). The results may also be fed-back to members of the multidisciplinary team via email or dedicated web-pages, providing an efficient way to monitor active treatment. The real time feedback of information suggests that patients’ needs can be addressed as and when they occur rather than at a pre-arranged clinic visit which may not coincide with, or be well-suited to, responding to symptoms and other quality of life issues (Donaldson 2007).
Support for data collection, scoring and feedback of data
The successful application of PROMs into routine practice requires appropriate support for both health professionals and patients. This support may relate to systems level support and the logistics of administration, or the personal support for both health professionals and patients.
Clinical practice varies widely, presenting many challenges for the application of PROMs. At a systems level, it is essential to explore how data collection fits in with the wider healthcare setting. That is, who supports it and how? Different modes of completion have different demands in relation to scoring and data feedback, and adequate investment is essential. For example, if the data are collected using pen and paper format, support to ensure that PROMs are scored and fed back in a timely manner to clinicians is required; without this support PROMs data may be left unused. If data are captured electronically, support to ensure that system failures are rapidly addressed is required.
The ability, knowledge and willingness of health professionals to incorporate PROMs into routine practice is critical to the relative success of PROMs as an intervention to enhance care (Haywood et al 2008, Jacobsen et al 2002). Familiar barriers to the incorporation of PROMs into routine practice include concern over the availability and appropriateness of questionnaires, patient disabilities, limited resources or time, limited knowledge, and limited guidance (Haywood et al 2008, Skevington et al 2005). There may also be an unwillingness to discuss certain items raised in PROMs, often because of the perceived lack of support or resources to address issues raised.
Although evidence from the oncology field suggests that the assessment of quality of life using PROMs does not add time to the clinical encounter (Velikova et al 2004), to reduce the perception of increased burden, the introduction of PROMs assessment may require some ‘trade-off’ with more traditional approaches to assessment being removed from assessment (Haywood et al 2008). Many clinicians lack knowledge and confidence in the use of PROMs, and to jettison familiar methods of assessment in favour of something new, irrespective of the evidence-base, requires significant support and guidance (Haywood 2006). Specific education provided via web-based facilities, support by the professional bodies and training courses have been highlighted as the most preferable routes to receive training and information (Haywood et al 2008). The clinical relevance of selected measures, supported by training and support to encourage a positive attitude to the usefulness of formal assessment should facilitate staff acceptance. Where more formal assessment becomes a regular part of assessment practice, supported by knowledgeable clinicians, patient acceptance is enhanced.
At the patient level, the concept of completing PROMs should be clearly introduced to support adherence. For example, explaining why they are being used, how they contribute to the therapeutic relationship, the frequency with which they will be completed, and how the results will be used. The logistics of, and available support for, PROM completion should also be discussed. The relevance, simplicity and appeal of the PROM will contribute to completion. Moreover, in order to accept the incorporation of more standardized PROM assessment into the care process, patients and clinicians need to experience any potential enhancement to their interactions (Haywood et al 2006).
Interpretation and action
For PROM-related information to successfully add to the armamentarium of information collected in routine practice, this information must be actionable. Although well-developed condition-specific or individualised measures are likely to have enhanced clinical relevance than generic measures, providing information that is more actionable, appropriate education and training in the application and interpretation of PROMs is required.
This perceived need for further training in the use of PROMs in routine practice has recently been confirmed in surveys of UK-based continence specialists (Haywood et al 2008) and GPs (Skevington et al 2005). Although guidance for score interpretation at a group level exists for some rheumatic diseases (for example, in AS (Anderson et al 2001, Van Tubergen et al 2003)), specific guidance for the interpretation of PROMs in rheumatology routine practice does not currently exist.
The presentation of PROM data provides a visual profile that raises key problems and highlights areas for concern. Rather than focusing on the aggregate score, clinicians should also explore the range of problems raised. The integration of clinical expertise with an understanding of PROM scores supports interpretation. Clinical intuition will often indicate when patients are not doing well, and well-developed PROMs may pick this up sooner than traditional measures (Fallowfield 2007). Information about PROM scores in particular clinical settings, and the ‘normal pattern’ of scores informed by condition or population-based values, should signal further action or referral. For example, various cut-points have been suggested for the Hospital Anxiety and Depression Score (HADS) to indicate levels of anxiety or depression (McDowell 2006). For each domain, a cut-point of 8/9 suggests mild anxiety or depression and 11/12 suggests severe anxiety or depression. The longitudinal monitoring of health supports the comparison of scores over time. Relating scores to additional clinical variables may further assist in decision-making. Where specific guidance for the interpretation of PROM score change is not available, evidence suggests that an acceptable starting point for the interpretation of a clinically important change in health is a score change of 10% or 10-point change on a 0-100 scale (Sloan et al 2005). Specific management guidelines related to score interpretation (Meadows et al 1998, Van Stel et al 2007, Varni et al 2005, Velikova et al 2004) and illustrations through the use of case vignettes, for example, of stable patients and patients whose health has deteriorated, may further assist this process (Velikova et al 2004) (Boxes 4.4, 4.5).
BOX 4.4 Interpretation of PROM scores – profile of an ‘unwell’ patient
Subjective history:
PROM assessment (score presented graphically with cut-points highlighted):
 Disease activity: scores on the BASDAI (7/10) and pain VAS (9/10) suggest high levels of disease activity and pain respectively.
Disease activity: scores on the BASDAI (7/10) and pain VAS (9/10) suggest high levels of disease activity and pain respectively.BOX 4.5 Interpretation of PROM scores – profile of a ‘well’ patient
PROM assessment (score presented graphically with cut-points highlighted):
 Disease activity: low scores on the BASDAI (2/10) and pain VAS (1/10) suggest low levels of disease activity and pain respectively.
Disease activity: low scores on the BASDAI (2/10) and pain VAS (1/10) suggest low levels of disease activity and pain respectively. General health status-profile: SF-36. High scores on all domains suggesting good general health. All scores are above the population mean.
General health status-profile: SF-36. High scores on all domains suggesting good general health. All scores are above the population mean.However, as with all methods of assessment, it is important to retain a healthy scepticism in relation to specific PROM scores: scores can be influenced by a range of contextual factors, including gender, age, race, ethnicity, completion format, setting, relationship with provider, and there is potential for wide variation at individual level. Application of PROMs at an individual level requires a trade-off between scientific precision and clinical usefulness, and critical thinking must be applied to all sources of information informing the health status of the individual. Exploring the performance of PROMs alongside other clinical variables is central to this.
Longitudinal monitoring
The routine application of PROMs recognises that not all health professionals will ask all patients all of the appropriate questions, all of the time, or in a way that patients will be able to answer (Ganiats 2007). Moreover, there is evidence that, without the use of PROMs, clinicians do not access information that is important to patients as well as they think they do (Fallowfield et al 2001). The routine application of PROMs supports the longitudinal monitoring of care and health outcomes (Figs. 4.3-4.5), providing information that is essential to the process of care. The longitudinal collection of individual data supports data interpretation over time.
The following box provides an illustration of the ‘ideal’ application of PROMs in a routine practice setting (Box 4.6).
BOX 4.6 Application of PROMs in routine practice: an illustration
PROM feedback
CONCLUSION
The measurement of patient-reported outcome in the rheumatic diseases continues to evolve. Well-developed PROMs provide a major source of evidence of the patient experience to inform value judgements of both patients and members of the multi-disciplinary healthcare team (Patrick & Chiang 2000). They can inform routine practice, service delivery, and healthcare policy, while enhancing communication between patient and provider, leading to common understanding of the health problem, and agreed management solutions and outcomes; greater patient adherence and treatment satisfaction. Improved health status may then be achieved with advancing technology, electronic data capture of PROMs may enhance the feasibility of application, supporting the timely provision of scores and the clinical utility to routine practice. Moreover, information can be readily distributed between members of the multidisciplinary team, fostering information flow between different care providers and supporting the continuity of care. Demonstrating the cost effectiveness of incorporating PROM assessment into routine practice is essential to good practice, and to attracting the necessary resources to support more efficient data capture (Haywood et al 2008).
Members of the multidisciplinary team should be engaged in the development, selection and use of PROMs in routine practice. This will require training and support to foster the necessary change in assessment culture. It is essential that opportunities for further development of consensus around core sets, responder criteria, PROM development and selection are identified, and that patients and members of the wider multidisciplinary team contribute fully to this process. Further evaluation of the contribution of PROMs to routine practice is required that takes into account the roles of different members of the multidisciplinary team.
In summary patient-centred care and patient participation in decisions about health and healthcare are important goals of healthcare. Patient-reported outcome measures have the potential to enhance patient participation and patient-provider collaboration, providing information that is reliable, valid and of relevance to a range of stakeholders, thus supporting patient choice. However, challenges exist with regards to the selection, application and performance of PROMs, particularly in a routine practice setting. These challenges must be addressed if PROMs are to be more widely accepted as key sources of evidence of the impact of ill-health and treatment effectiveness, and a mechanism to enhance patient participation in routine practice.
References and further reading
Anderson J., Baron G., van der Heijde D., et al. Ankylosing Spondylitis Assessment Group preliminary definition of short-term improvement in Ankylosing Spondylitis. Arthritis Rheum.. 2001;44:1876-1886.
Appleby J., Devlin N. Measuring Success in the NHS. Using Patient-Assessed Health Outcomes to Manage the Performance of Healthcare Providers. London: King’s Fund; 2004.
Ball A.E., Russell E.M., Seymour D.G., et al. Problems in using health survey questionnaires in older patients with physical disabilities. Can proxies be used to complete the SF-36? Gerontology. 2001;47(6):334-340.
Bellamy N., Kirwan J., Boers M., et al. Recommendations for a core set of outcome measures for future phase III clinical trials in knee, hip, and hand osteoarthritis.Consensus development at OMERACT III. J. Rheumatol.. 1997;24:799-802.
Bellamy N. Science of assessment. Ann. Rheum. Dis.. 2005;64(Suppl 2):ii42-ii45.
Bellamy N., Buchanan W., Goldsmith C., et al. Validation Study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J. Rheumatol.. 1988;15:1833-1840.
Bellamy N. Clinimetric concepts in outcome assessment: the OMERACT filter. J. Rheumatol.. 1999;26(4):948-950.
Bezjak, A., 2007. Logistics of Collecting Patient-Reported Outcomes (PROS) in Clinical Practice: an overview. Conference on Patient Reported Outcomes in Clinical Practice. International Society for Quality of Life Research. Budapest, Hungary, Proceedings, p13-14. 2007
Bombardier C. Outcome assessments in the evaluation of treatment of spinal disorders. Spine. 2000;25:3097-3103.
Bot S.D.M., Terwee C.B., Van der Windt D.A.W.M., et al. Clinimetric evaluation of shoulder disability questionnaires: a systematic review of the literature. Ann. Rheum. Dis.. 2004;63:335-341.
Brazier J., Roberts J., Tsuchiya A., et al. A comparison of the EQ-5D and SF-6D across seven patient groups. Health Econ.. 2004;13:873-884.
Bruce B., Fries J.F. The Stanford Health Assessment Questionnaire: a review of its history, issues, progress, and documentation. J. Rheumatol.. 2003;30:167-178.
Burke L., Stifano T., Dawisha S. Guidance for Industry – Patient-Reported Outcome Measures : Use in Medical Product Development to Support Labelling Claims. Rockville, MD: U.S Department of Health and Human Sciences; 2006. Food and Drug Administration
Campbell S.M., Roland M.O., Buetow S.A. Defining quality of care. Soc. Sci. Med.. 2000;51(11):1611-1625.
Carr A.J., Thompson P.W. Towards a measure of patient-perceived handicap in rheumatoid arthritis. Br. J. Rheumatol.. 1994;33:378-382.
Chakravarty E.F., Bjorner J.B., Fries J.F. Improving patient reported outcomes using item response theory and computerized adaptive testing. J. Rheumatol.. 2007;34(6):1426-1431.
Chorus A.M., Miedema H.S., Boonen A., et al. Quality of life and work in patients with rheumatoid arthritis and ankylosing spondylitis of working age. Ann. Rheum. Dis.. 2003;62:1178-1184.
Clancy C.M., Lawrence W. Is outcomes research on cancer ready for prime time? Med. Care. 2002;40:III92-III100.
Dagfinrud H., Kjeken I., Mowinckel P., et al. Impact of functional impairment in ankylosing spondylitis: impairment, activity limitation, and participation restrictions. J. Rheumatol.. 2005;32(3):516-523.
Donabedian A. Evaluating the quality of medical care. Milbank Q.. 1966;44(Suppl.):166-206.
Donaldson, M., 2007. A non-visit approach to use of PROS in Oncology practice. Conference on Patient Reported Outcomes in Clinical Practice. International Society for Quality of Life Research. Budapest, Hungary, Proceedings, p14-15.
EuroQol-Group. EuroQol – A New Facility For The Measurement Of Health-Related Quality Of Life. The EuroQol Group. Health Policy. 1990;16:199-208.
Escorpizo R., Bombardier C., Boonen, et al. Worker productivity outcome measures in arthritis. J. Rheumatol.. 2007;34(6):1372-1380.
Fallowfield L., Ratcliffe D., Jenkins V., et al. Psychiatric morbidity and its recognition by doctors in patients with cancer. Brit. J. Cancer 20. 2001;84(8):1011-1015.
Fallowfield, L., 2007. Training healthcare professionals in communication about PROS in clinical practice. Conference on Patient Reported Outcomes in Clinical Practice. International Society for Quality of Life Research. Budapest, Hungary, Proceedings, p15-16.
Finlay A.Y., Coles E.C. The effect of severe psoriasis on the quality of life of 369 patients. Brit. J. Dermatol.. 1995;132(2):236-244.
Fitzpatrick R., Davey C., Buxton M.J., et al. Evaluating Patient-Based Outcome Measures for use in Clinical Trials. Health Technol. Assess.. 1998;2:I-Iv. 1-74
Fries J.F., Spitz P., Kraines R.G., et al. Measurement of patient outcome in arthritis. Arthritis Rheum.. 1980;23:137-145.
Ganz P.A. What Outcomes Matter To Patients: A Physician-Researcher Point Of View. Med. Care. 2002;40(III):I11-I119.
Ganiats, T., 2007. Content for PROS used in Clinical Practice: the clinician perspective. Conference on Patient Reported Outcomes in Clinical Practice. International Society for Quality of Life Research. Budapest, Hungary, Proceedings, p12-13.
Garratt A., Schmidt L., Mackintosh A., et al. Quality of life measurement: bibliographic study of patient assessed health outcome measures. Brit. Med. J.. 2002;324:1417-1421.
Garratt A.M., Brealey S., Gillespie W.J., et al. Patient-assessed health instruments for the knee: a structured review. Rheumatology (Oxford). 2004;43(11):1414-1423.
Garrett S., Jenkinson T., Kennedy L., et al. A new approach to defining disease status in ankylosing spondylitis: The Bath ankylosing spondylitis disease activity index. J. Rheumatol.. 1994;21:2286-2291.
Gilbody S.M., Whitty P.M., Grimshaw J.M., et al. Improving the detection and management of depression in primary care. Qual. Saf. Health Care. 2003;12(2):149-155.
Giles J.T., Mease P., Boers M., et al. Assessing single joints in arthritis clinical trials. J. Rheumatol.. 2007;34(3):641-647.
Gladman D.D., Mease P.J., Strand V., et al. Consensus on a core set of domains for psoriatic arthritis. J. Rheumatol.. 2007;34(5):1167-1170.
Greenhalgh J., Long A.F., Flynn R. The use of patient reported outcome measures in routine clinical practice: Lack of impact or lack of theory? Soc. Sci. Med.. 2005;60:833-843.
Haywood K.L., Garratt A.M., Carrivick S.E., et al. Continence specialists use of quality of life information in routine practice: a national survey of practitioners. Quality of Life Research. 2009;18(4):423-433. May
Haywood K.L., Garratt A.M., Dziedzic K., Dawes P.T. Generic measures of health-related quality of life in ankylosing spondylitis: reliability, validity and responsiveness. Rheumatology (Oxford). 2002;41(12):1380-1387.
Haywood K.L., Garratt A.M., Dziedzic K., et al. Patient centered assessment of ankylosing spondylitis-specific health related quality of life: evaluation of the Patient Generated Index. J. Rheumatol.. 2003;30(4):764-773.
Haywood K.L., Garratt A.M., Dawes P.T. Patient-assessed health in ankylosing spondylitis: a structured review. Rheumatology (Oxford). 2005;44(5):577-586.
Haywood K.L. Patient-reported outcome I: measuring what matters in musculoskeletal care. Musculoskeletal Care. 2006;4(4):187-203.
Haywood K., Marshall S., Fitzpatrick R. Patient participation in the consultation process: a structured review of intervention strategies. Patient Educ. Couns.. 2006;63(1-2):12-23.
Haywood, K.L., Garratt, A.M., Carrivick, S., et al., 2008. Continence specialists use of quality of life information in routine practice: a national survey of practitioners. Qual. Life Res. in press.
Hewlett S., Smith A.P., Kirwan J.R. Measuring the meaning of disability in rheumatoid arthritis: the Personal Impact Health Assessment Questionnaire (PI HAQ). Ann. Rheum. Dis.. 2002;61(11):986-993.
Hewlett S.A. Patients and clinicians have different perspectives on outcomes in arthritis. J. Rheumatol.. 2003;30:877-879.
Hibbard J.H. Engaging healthcare consumers to improve the quality of care. Med. Care. 2003;41:I-61-I-70.
Higginson I.J., Carr A.J. Measuring quality of life: Using quality of life measures in the clinical setting. Brit. Med. J.. 2001;322:1297-1300.
Hobart J.C., Riazi A., Lamping D.L., et al. Improving the evaluation of therapeutic interventions in multiple sclerosis: development of a patient-based measure of outcome. Health Technol. Assess.. 2004;8(9):1-48.
Jacobsen P.B., Davis K., Cella D. Assessing quality of life in research and clinical practice. Oncology (Williston Park). 2002;16(9 Suppl 10):133-139.
Keeling S.O., Landewe R., van der Heijde D., et al. Testing of the preliminary OMERACT validation criteria for a biomarker to be regarded as reflecting structural damage endpoints in rheumatoid arthritis clinical trials: the example of C-reactive protein. J. Rheumatol.. 2007;34(3):623-633.
Kessler L., Ramsey S.D. The outcomes of the cancer outcomes research symposium: a commentary. Med. Care. 2002;40(III):104-108.
Kirwan J.R., Hewlett S. Patient perspective: reasons and methods for measuring fatigue in rheumatoid arthritis. J. Rheumatol.. 2007;34(5):1171-1173.
Kirwan J.R., Minnock P., Adebajo A., et al. Patient perspective: fatigue as a recommended patient centered outcome measure in rheumatoid arthritis. J. Rheumatol.. 2007;34(5):1174-1177.
Kristjansson E., Tugwell P.S., Wilson A.J., et al. Development of the effective musculoskeletal consumer scale. J. Rheumatol.. 2007;34(6):1392-1400.
Kvien T.K., Heiberg T. Patient perspective in outcome assessments–perceptions or something more? J. Rheumatol.. 2003;30:873-876.
Liang M.H. Pushing the limits of patient-oriented outcome measurements in the search for disease modifying treatments for osteoarthritis. J. Rheumatol.. 2004;70:61-65.
Lorig K., Chastain R.L., Ung E., et al. Development and evaluation of a scale to measure perceived self-efficacy in people with arthritis. Arthritis Rheum.. 1989;32(1):37-44.
Marra C.A., Esdaile J.M., Guh D., et al. A comparison of four indirect methods of assessing utility values in rheumatoid arthritis. Med. Care. 2004;42(11):1125-1131.
Marra C.A., Woolcott J.C., Kopec J.A., et al. A comparison of generic, indirect utility measures (the HUI2, HUI3, SF-6D, and the EQ-5D) and disease-specific instruments (the RAQoL and the HAQ) in rheumatoid arthritis. Soc. Sci. Med.. 2005;60(7):1571-1582.
Marshall S., Haywood K., Fitzpatrick R. Impact of patient-reported outcome measures on routine practice: A structured review. J. Eval. Clin. Pract.. 2006;12(5):559-568.
Martin F., Camfield L., Rodham K., et al. Twelve years’ experience with the Patient Generated Index (PGI) of quality of life: a graded structured review. Qual. Life Res.. 2007;16(4):705-715.
McDowell I. Measuring Health: A Guide to Rating Scales and Questionnaires, third ed. New York: Oxford University Press, 2006.
McDowell I., Newell C. Measuring Health: A Guide to Rating Scales and Questionnaires, second ed. New York: Oxford University Press, 1996.
McGee H.M., O’Boyle C.A., Hickey A., et al. Assessing the quality of life of the individual: the SEIQoL with a healthy and a gastroenterology unit population. Psychol. Med.. 1991;21(3):749-759.
McHorney C.A. The potential clinical value of quality-of-life information response to Martin. Med. Care. 2002;40(III):56-62.
Meadows K.A., Rogers D., Greene T. Attitudes to the use of health outcome questionnaires in the routine care of patients with diabetes: a survey of general practitioners and practice nurses. Brit. J. Gen. Pract.. 1998;48(434):1555-1559.
Mease P., Arnold L.M., Bennett R., et al. Fibromyalgia syndrome. J. Rheumatol.. 2007;34(6):1415-1425.
Meenan R.F., Mason J.H., Anderson J.J., et al. AIMS2. The content and properties of a revised and expanded arthritis impact measurement scales health status questionnaire. Arthritis Rheum.. 1992;35(1):1-10.
Merkel P.A., Clements P.J., Reveille J.D., et al. Current status of outcome measure development for clinical trials in systemic sclerosis. Report from OMERACT 6. J. Rheumatol., 30. 2003;7:1630-1647.
Mitchell P.H., Lang N.M. Framing the problem of measuring and improving healthcare quality: has the quality health outcomes model been useful? Med. Care. 2004;42(II):4-11.
Morris J., Perez D., McNoe B. The use of quality of life data in clinical practice. Qual. Life Res.. 1998;7:85-91.
Neudert C., Wasner M., Borasio G.D. Patients’ assessment of quality of life instruments: a randomised study of SIP, SF-36 and SEIQoL-DW in patients with amyotrophic lateral sclerosis. J. Neurol. Sci.. 2001;191:103-109.
Neumann P.J., Araki S.S., Gutterman E.M. The use of proxy respondents in studies of older adults: lessons, challenges, and opportunities. J. Am. Geriatr. Soc.. 2000;48(12):1646-1654.
NICE. Guide to the Methods of Technological Appraisal. London: National Institute of Health and Clinical Excellence; 2004.
Patel K., Veenstra D., Patrick D. A review of selected patient-generated outcome measures and their application in clinical trials. Value Health. 2003;6:595-603.
Patrick D.L., Chiang Y.P. Measurement of health outcomes in treatment effectiveness evaluations: conceptual and methodological challenges. Med. Care. 2000;38(III):4-25.
Pham T., Van Der Heijde D., Lassere M., et al. Outcome variables for osteoarthritis clinical trials: The OMERACT-OARSI set of responder criteria. J. Rheumatol.. 2003;30(7):1648-1654.
Pringle D., Doran D. Patient Outcomes as an Accountability. In: Doran D.M., editor. Nursing-Sensitive Outcomes. State of the Science. London: Jones and Bartlett Publishers; 2003:1-26.
Quest E., Aanerud G.J., Kaarud S., et al. Patients’ perspective. J. Rheumatol.. 2003;30(4):884-885.
Ramsey S. What is cancer outcomes research? A diversity of perspectives for an emerging field. Med. Care. 2002;40(Suppl. 6):III-1-III-2.
Raphael D., Smith T.F., Brown I., et al. Development and properties of the short and brief versions of the Quality of Life Profile - Seniors Version. International Journal of Health Sciences. 1995;6:161-168.
Rose M., Bezjak A., et al. Logistics of collecting patient-reported outcomes (PROS) in clinical practice. An overview and practical examples. Qual. Life Res. Feb.. 2009;18(1):125-136. Epub 2009 Jan 20
Ruta D., Garratt A.M., Leng M., et al. A new approach to the measurement of quality of life. The Patient-Generated Index. Med. Care. 1994;32(11):1109-1126.
Sambrook P. Guidelines for osteoporosis trials. J. Rheumatol.. 1997;24:1234-1236.
Skevington S.M., Day R., Chisholm A., et al. How much do doctors use quality of life information in primary care? Testing the trans-theoretical model of behaviour change. Qual. Life Res.. 2005;14:911-922.
Sloan J.A., Cella D., Hays R.D. Clinical significance of patient-reported questionnaire data: another step toward consensus. J. Clin. Epidemiol.. 2005;58(12):1217-1219.
Smets E.M., Garssen M., et al. The Multidimensional Fatigue Inventory (MFI) psychometric qualities of an instrument to assess fatigue. J. Psychosom. Res.. 1995;39(3):315-325.
Smolen J., Strand V., Cardiel M., et al. Randomized clinical trials and longitudinal observational studies in systemic lupus erythematosus: consensus on a preliminary core set of outcome domains. J. Rheumatol.. 1999;26(2):504-507.
Streiner D., Norman G. Health Measurement Scales: A Practical Guide To Their Development And Use, 3rd edn. Oxford: Oxford Medical Publications. 2003.
Stucki G., Boonen A., Tugwell P., et al. The World Health Organisation International Classification of Functioning, Disability and Health: a conceptual model and interface for the OMERACT process. J. Rheumatol.. 2007;34(3):600-606.
Tugwell P., Bombardier C., Buchanan W.W., et al. The MACTAR patient preference disability questionnaire -an individualized functional priority approach for assessing improvement in physical disability in clinical trials in rheumatoid arthritis. J. Rheumatol.. 1987;14:446-451.
Tugwell P., Boers M. Developing consensus on preliminary core efficacy endpoints for rheumatoid arthritis clinical trials. OMERACT committee. J. Rheumatol.. 1993;20(3):555-556.
Van der Heijde D., Bellamy N., Calin A., et al. Preliminary core sets for endpoints in ankylosing spondylitis. Assessments in ankylosing spondylitis working group. J. Rheumatol.. 1997;24(11):2225-2229.
Van der Heijde D., Van der Linden S., Dougados M., et al. Ankylosing spondylitis: plenary discussion and results of voting on selection of domains and some specific instruments. J. Rheumatol.. 1999;26(4):1003-1005.
Van Echteld I., Cieza A., Boonen A., et al. Identification of the most common problems by patients with ankylosing spondylitis using the international classification of functioning, disability and health. J. Rheumatol.. 2006;33(12):2475-2483.
Van Stel, H.F., Van der Meer, V., Bakker, M.J., et al., 2007. Internet based self-monitoring in adults with asthma using the asthma control questionnaire. Conference on patient reported outcomes in clinical practice. International Society for Quality of Life Research. Budapest, Hungary. Proceedings: Abstract 133.
van Tubergen A., van der Heijde D., Anderson J., et al. Comparison of statistically derived ASAS improvement criteria for ankylosing spondylitis with clinically relevant improvement according to an expert panel. Ann. Rheum. Dis.. 2003;62(3):215-221.
Varni J.W., Burwinkle T.M., Lane M.M. Health-related quality of life measurement in pediatric clinical practice: an appraisal and precept for future research and application. Health Qual. Life Outcomes. 2005;16(3):34.
Varni J.W., Burwinkle T.M., Limbers C.A., et al. The PedsQL as a patient-reported outcome in children and adolescents with fibromyalgia: an analysis of OMERACT domains. Health Qual. Life Outcomes. 2007;12(5):9.
Velikova G., Booth L., Smith A.B., et al. Measuring Quality of Life in routine oncology practice improves communication and patient well-being: A Randomized Controlled Trial. J. Clin. Oncol.. 2004;22:714-724.
Wade D.T., de Jong B.A. Recent advances in rehabilitation. Brit. Med. J.. 2000;320(7246):1385-1388.
Ware J. SF-36 Health Survey. Manual Interpretation Guide. Minrod Press; 1997.
Ware J.E., Kosinski M., Dewey J.E. How to Score Version Two of the SF-36 Health Survey (Standard and Acute Forms). Lincoln, RI: Quality Metric Inc; 2002.
WHO. International Classification of functioning, disability and health. ICF. Geneva: World Health Organisation; 2001.
WHOQol Group. The Development Of The World Health Organisation Quality of Life Assessment Instrument (The WHOQol). In: Orley J., Kuyken W., editors. Quality of Life Assessment: International Perspectives. Berlin: Springer-Verlag; 1994:41-47.
Wiebe S., Guyatt G., Weaver B., et al. Comparative responsiveness of generic and specific quality-of-life instruments. J. Clin. Epidemiol.. 2003;56:50-52.
Zebrack, B., 2007. The use and utility of patient-reported outcomes: reflections from a cancer survivor and research scientist. Conference on Patient Reported Outcomes in Clinical Practice. International Society for Quality of Life Research. Budapest, Hungary. Proceedings: p10-11.
Zigmond A.S., Snaith R.P. The Hospital Anxiety and Depression Scale. Acta Psychiat. Scand.. 1983;67:361-370.