Chapter 23 Miscellaneous conditions
KEY POINTS
 Septic arthritis is a medical emergency requiring urgent referral for joint aspiration and appropriate intravenous antibiotic therapy
Septic arthritis is a medical emergency requiring urgent referral for joint aspiration and appropriate intravenous antibiotic therapy Non-pharmacological management of acute gout and pseudogout includes application of ice-packs to the affected joint and initial rest followed by rapid mobilisation
Non-pharmacological management of acute gout and pseudogout includes application of ice-packs to the affected joint and initial rest followed by rapid mobilisation Lifestyle modification advice regarding weight loss, restriction of alcohol and purine-rich foods is a key component of the management of gout
Lifestyle modification advice regarding weight loss, restriction of alcohol and purine-rich foods is a key component of the management of gout Neuropathic arthropathy is an uncommon complication of diabetes mellitus; a high index of suspicion is necessary to avoid delay in treatment and permanent disability
Neuropathic arthropathy is an uncommon complication of diabetes mellitus; a high index of suspicion is necessary to avoid delay in treatment and permanent disabilityPart 1: Septic arthritis, gout, pyrophosphate arthropathy, sarcoidosis and diabetes mellitus
Septic arthritis is an uncommon but important medical emergency. Although less common than other causes of the acute, hot, swollen joint (Box 23.1); it remains the most important diagnosis to make and treat appropriately. Despite appropriate treatment, septic arthritis is associated with joint destruction and long-term morbidity and functional loss. The incidence of septic arthritis is approximately two to six cases per 100,000 population per year and is highest at extremes of age (Cooper & Cawley 1986, Kaandorp et al 1997a).
AETIOLOGY AND PATHOLOGY
Organisms may enter a joint from several routes. The most frequent modes are haematogenous spread, spread from an infected local focus, e.g. osteomyelitis or cellulitis, and direct penetrating injury (Nade 2003).
The most common causative organisms are staphylococcal and streptococcal species with Staphylococcus aureus being the most common (Gupta et al 2001, Kaandorp et al 1997a, Weston et al 1999). Methicillin-resistant staphylococcus aureus (MRSA) septic arthritis is becoming increasingly prevalent (Nixon et al 2007). Gram-negative organisms are less frequently implicated: Haemophilus influenzae is most frequently seen in children. Less frequently encountered organisms include enterobacteria, Pseudomonas aeruginosa and Mycobacterium tuberculosis.
Risk factors for septic arthritis include underlying joint disease (especially rheumatoid arthritis), prosthetic joints, low socioeconomic class, intravenous drug abuse, alcoholism, diabetes mellitus, previous intra-articular injection and cutaneous ulceration (Mathews et al 2007).
CLINICAL PRESENTATION AND CLINICAL FEATURES
The classical presentation of septic arthritis is with a short history of a painful, hot, swollen, tender single joint with reduced range of movement (Mathews et al 2007). The most frequently affected joint is the knee. However, involvement is polyarticular in 22% of cases. Fever may be present but is absent in approximately 50% of cases.
DIAGNOSIS AND DIFFERENTIAL DIAGNOSIS
The diagnosis of septic arthritis relies upon prompt aspiration of the affected joint prior to commencing antibiotic therapy (Coakley et al 2006). Gram stain, culture and crystal examination of synovial fluid should be performed. A possible infected prosthetic joint requires referral to an orthopaedic surgeon for aspiration in theatre. Blood cultures should always be taken and may identify a causative organism in 33% of cases (Weston et al 1999). Inflammatory markers such as white cell count, erythrocyte sedimentation rate and C-reactive protein should be measured but may not be raised in a significant number of cases (Gupta et al 2001, Weston et al 1999). The important differential diagnoses of septic arthritis include crystal arthropathies, e.g. gout and pseudogout, haemarthrosis, reactive arthritis and a monoarticular presentation of an inflammatory arthritis (Box 23.1).
MANAGEMENT
As soon as the joint has been aspirated, intravenous antibiotics should be commenced. The choice of antibiotics will usually be guided by local microbiologists and should be modified in light of the results of Gram stain and culture. Conventionally, the duration of intravenous antibiotic therapy is 2 weeks followed by 4 weeks of oral therapy (Coakley et al 2006). Regular aspiration to dryness should be performed and arthroscopic aspiration may be required.
PROGNOSIS
Septic arthritis is associated with considerable mortality and morbidity (Gupta et al 2001, Kaandorp et al 1997b, Weston et al 1999). The mortality of septic arthritis is approximately 11%. Adverse functional outcome is seen in 21–24% of cases. Factors predicting poor outcome include older age, pre-existing joint disease and the presence of synthetic material within the joint (Mathews et al 2007).
GOUT
Gout is a crystal deposition disease caused by the formation of monosodium urate (MSU) crystals in and around joints. It is one of the most common inflammatory arthropathies having a prevalence of 0.52–1.39% and incidence of approximately 13 cases per 10,000 patient-years (Harris et al 1995, Lawrence et al 1998, Mikuls et al 2005, Wallace et al 2004). The prevalence and incidence of gout are thought to be rising (Arromdee et al 2002, Wallace et al 2004). Gout is traditionally sub-divided into primary and secondary gout according to clinical features and risk factors (Table 23.1).
Table 23.1 Comparison of the clinical features and risk factors of primary and secondary gout
| PRIMARY GOUT | SECONDARY GOUT | |
|---|---|---|
| Age | Middle-aged | Elderly |
| Gender | Males | Equal gender distribution |
| Acute attacks | Common | Less common May present with tophi alone |
| Distribution | Predominantly lower limb | Equal upper and lower limb |
| Risk factors | Family history of gout | Diuretics |
| Metabolic syndrome | Renal failure | |
| Hypertension | ||
| Obesity | ||
| Hyperlipidaemia | ||
| Insulin resistance | ||
| Excess alcohol consumption | ||
| Purine-rich diet |
AETIOLOGY AND PATHOLOGY
The primary risk factor for the development of gout is an elevated serum urate (uric acid) level (SUA) or hyperuricaemia. Uric acid is the end-product of purine metabolism in humans. Hyperuricaemia arises from overproduction or renal under-excretion of uric acid, or a combination of both. As hyperuricaemia develops, and body tissues become super-saturated with urate, the formation of MSU crystals leads to clinical gout.
Several independent risk factors for the development of gout are recognised (Table 23.1). Primary gout occurs almost exclusively in men and displays a familial tendency: several genetic factors have been identified (Cheng et al 2004, Huang et al 2006, Taniguchi et al 2005, Wang et al 2004). Further independent risk factors include hypertension, obesity and lifestyle factors (excess alcohol consumption, especially beer, and a diet rich in animal purines, for example red meat and seafood) (Choi et al 2004a, 2004b, 2005, Mikuls et al 2005). Gout associates with other features of the metabolic syndrome including hyperlipidaemia, insulin resistance and cardiovascular disease (Abbott et al 1988, Mikuls et al 2005). Diuretic therapy and renal failure are risk factors for secondary gout (Choi et al 2005, Mikuls et al 2005). Osteoarthritis (OA) predisposes to local MSU crystal deposition (Roddy et al 2007).
CLINICAL PRESENTATION AND CLINICAL FEATURES
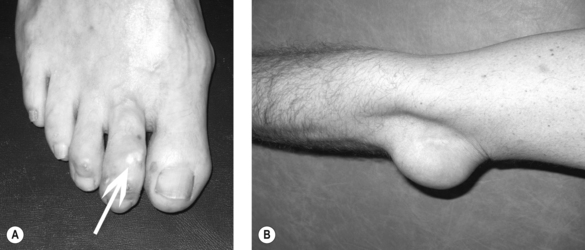
Clinical gout is sub-divided into acute gouty arthritis, interval (intercritical) gout and chronic tophaceous gout. Acute gouty arthritis typically manifests as acute mono-arthritis characterised by severe pain with associated redness, warmth, swelling and tenderness. The first metatarsophalangeal (MTP) joint is affected most frequently followed by the mid-foot, ankle, knee, finger interphalangeal joints, wrist and elbow. Attacks occur rapidly, often overnight, peaking within 24 hours, before resolving completely over a 2–3 week period. A variable period elapses before the next attack occurs (interval gout). With time, attacks become more frequent, more severe and are more often pauci- or polyarticular leading to chronic tophaceous gout characterised by chronic arthropathy and chalky-white subcutaneous deposits of MSU crystals (tophi). Typical sites for tophi include the toes (Fig. 23.1A), Achilles’ tendons, fingers, elbows (Fig. 23.1B) and, less commonly, the helix of the ear.
DIAGNOSIS AND DIFFERENTIAL DIAGNOSIS
Definitive diagnosis of gout requires compensated polarised light microscopy of aspirated synovial fluid or tophaceous material to demonstrate MSU crystals which appear needle-shaped with strong negative birefringence. The differential diagnosis includes septic arthritis, pseudogout, reactive arthritis and palindromic rheumatism (Box 23.1). Crystal arthropathies and septic arthritis can be readily differentiated by crystal examination, Gram stain and culture of synovial fluid.
Measurement of serum urate level (SUA) is an important investigation both to confirm hyperuricaemia and monitor response to treatment. However, the SUA level must be interpreted with caution. Many hyperuricaemic individuals do not develop gout (Campion et al 1987) and SUA is frequently lowered during an attack of acute gout (Schlesinger et al 1997).
MANAGEMENT
Acute gout
Treatment of the acute attack of gout aims to relieve pain by reducing inflammation and intra-articular hypertension. Local application of ice packs four times per day has been shown to reduce pain associated with acute gouty arthritis (Schlesinger et al 2002). Rest and elevation of the affected joint during an acute attack and the use of a bed-cage have been recommended (Jordan et al 2007). Joint aspiration reduces intra-articular hypertension and often provides rapid pain relief. Effective pharmacological therapies for acute gout include non-steroidal anti-inflammatory drugs (NSAIDs), both non-selective and COX-2 selective agents, low-dose colchicine and systemic and intra-articular corticosteroids (Alloway et al 1993, Fernandez et al 1999, Morris et al 2003, Rubin et al 2004, Schumacher et al 2002).
LONG-TERM MANAGEMENT
Gout is potentially curable. The aim of long-term management is lowering of SUA to facilitate crystal dissolution (Zhang et al 2006).
Modification of provoking factors including lifestyle
Risk factor modification is a key component of the management of gout. Advice regarding weight loss, restriction of alcohol (especially beer) and purine-rich foods should be offered to all patients with primary gout (Jordan et al 2007, Zhang et al 2006). In diuretic-induced gout the diuretic should be stopped or the dose reduced wherever possible.
Urate-lowering therapy
The aim of urate-lowering therapy is to reduce SUA below the saturation threshold of urate (Li-Yu et al 2001). The most commonly used drug in the UK is allopurinol. Other options include uricosuric agents such as sulphinpyrazone, probenecid and benzbromarone. New drugs in development include febuxostat and recombinant uricase.
PYROPHOSPHATE ARTHROPATHY
Calcium pyrophosphate dihydrate (CPPD) crystal deposition is a common age-related phenomenon that has three main clinical manifestations: (1) acute synovitis (pseudogout), (2) chronic pyrophosphate arthropathy and (3) the incidental finding of cartilage calcification (chondrocalcinosis) on plain radiographs. Over 40 years of age, the UK community prevalence of pyrophosphate arthropathy is 2.4% and chondrocalcinosis is 4.5% (Neame et al 2003, Zhang et al 2004).
AETIOLOGY
CPPD deposition is most commonly idiopathic being more common in females and with increasing age. In the context of chronic pyrophosphate arthropathy, it associates with OA (Dieppe et al 1982, Zhang et al 2004). CPPD deposition may also occur as a consequence of metabolic disease; most importantly, haemochromatosis, hyperparathyroidism, hypomagnesaemia and hypophosphatasia (Jones et al 1992). Familial CPPD deposition is uncommon although genetic mutations associating with chondrocalcinosis have been identified (Pendleton et al 2002, Williams et al 2003).
CLINICAL PRESENTATION AND CLINICAL FEATURES
Pseudogout is a common cause of acute monoarthritis in the elderly. As occurs with gout, the joint appears swollen, tender, red and warm. Onset is rapid, peaking within 24 hours, with resolution occurring within 2–3 weeks. The most commonly affected site is the knee followed by the wrist, ankle and shoulder. Systemic upset is common.
Chronic pyrophosphate arthopathy presents with features of OA (chronic pain and stiffness, crepitus, bony swelling and restricted movement) with or without superimposed attacks of acute synovitis. The knee is again the most frequently affected site. Chondrocalcinosis is a common asymptomatic incidental radiographic finding in the elderly.
CPPD crystals can be identified by compensated polarised light microscopy of aspirated synovial fluid as rhomboid or rod-shaped crystals with weak positive birefringence. Plain radiographs may show chondrocalcinosis or the structural changes of OA. Chondrocalcinosis mainly affects fibrocartilage and is most commonly seen at the knee (Fig. 23.2), wrist and symphysis pubis. Screening for haemochromatosis, hyperparathyroidism, hypomagnesaemia and hypophosphatasia is indicated under the age of 55 years and when radiographic chondrocalcinosis is particularly striking and widespread (Wright & Doherty 1997).
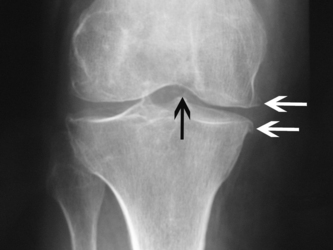
Figure 23.2 X-ray of the knee showing tibiofemoral chondrocalcinosis (black arrow), medial compartment loss of joint space and medial osteophytes (white arrows) - pyrophosphate arthropathy.
The most important differential diagnoses of pseudogout include septic arthritis and gout (Box 23.1). Chronic pyrophosphate arthropathy may be confused for rheumatoid arthritis (RA) or polymyalgia rheumatica depending on the sites involved.
MANAGEMENT
The evidence for non-pharmacological management of pseudogout is sparse but treatment options are similar to those for gout: application of ice-packs and initial rest of the affected joint with rapid mobilisation once improving. Pharmacological options include NSAIDs (with caution in the elderly), low-dose colchicine and systemic and intra-articular corticosteroids.
There is no specific therapy for chronic pyrophosphate arthropathy. Treatment principles are as for OA and may include education, exercise, weight loss, walking aids, analgesics and surgery with superimposed attacks of acute synovitis treated as above.
SARCOIDOSIS
Sarcoidosis is a multisystem disorder of unknown aetiology characterised by non-caseating granulomatous inflammation of affected organs. A recent UK study found the incidence of sarcoidosis to be 5.0 per 100,000 person-years between 1991 and 2003 (Gribbin et al 2006). The incidence is higher in females and Afro-Caribbean individuals and peaks between the ages of 20 and 49 years (Rybicki et al 1997).
CLINICAL PRESENTATION AND CLINICAL FEATURES
Almost any body system can be involved although the lungs, skin, lymph nodes and eyes are most frequently affected (Baughman et al 2001). Musculoskeletal involvement, including arthritis, tenosynovitis, myopathy and bony lesions, occurs in 10–25% of cases (Gumpel et al 1967, Perruquet et al 1984, Rybicki et al 1997). Sarcoid arthritis has acute and chronic forms. Acute sarcoid arthritis presents with acute swelling, warmth, erythema and tenderness characteristically affecting the ankles or knees (Gumpel et al 1967, Visser et al 2002). It is often accompanied by fever, bilateral hilar lymphadenopthy and erythema nodosum (Löfgren’s syndrome) (Mana et al 1999, Visser et al 2002). Chronic sarcoid arthropathy is more common in Afro-Caribbean subjects and targets the same joints as acute sarcoid arthritis manifesting as an oligo- or polyarthritis (Gumpel et al 1967). Affected patients tend to have evidence of chronic disease in other body systems, for example, the skin or interstitial lung disease.
DIAGNOSIS AND DIFFERENTIAL DIAGNOSIS
The diagnosis of acute sarcoidosis may be made on clinical grounds when the presentation is typical, for example, Löfgren’s syndrome (Mana et al 1994, Mana et al 1999). Erythrocyte sedimentation rate and c-reactive protein are often elevated (Visser et al 2002). Hypercalcaemia or hypercalciuria may also be present. Radiography and computed tomography (CT) of the chest may reveal bilateral hilar lymphadenopathy or evidence of interstitial lung disease. In many cases, a biopsy specimen of an affected organ is required to make a tissue diagnosis, for example, transbronchial lung biopsy or lymph node biopsy. Angiotensin-converting enzyme (ACE), which is produced by macrophages within granulomata, is elevated in 40–90% but may be normal in acute sarcoid arthritis.
Given the protean manifestations of sarcoidosis, the differential diagnosis is wide and depends upon which organs are affected. Important diagnoses to consider include tuberculosis, lymphoma and malignancy. The differential diagnosis of acute sarcoid arthritis includes reactive arthritis and gout. Chronic sarcoid arthropathy may mimic RA.
MANAGEMENT
The treatment of acute sarcoid arthritis is largely symptomatic. Symptoms are often responsive to NSAIDs (Visser et al 2002) although corticosteroids may be necessary in a small number of patients. Corticosteroids are the mainstay of treatment for patients with significant symptomatic severe pulmonary and extra-pulmonary disease (Paramothayan et al 2005, Pettersson et al 2000). Steroid-sparing agents such as methotrexate, antimalarials or ciclosporin are often tried although the evidence-base for their use is limited (Paramothayan et al 2006). Response of sarcoidosis to infliximab is reported (Doty et al 2005, Saleh et al 2006, Sweiss et al 2005) although these findings were not replicated in a recent phase II randomised trial (Baughman et al 2006).
PROGNOSIS
Acute sarcoidosis is usually a benign condition (Demirkok et al 2007, Glennas et al 1995, Perruquet et al 1984, Visser et al 2002) with 90% of cases achieving remission within 2 years (Mana et al 1999). A small number of cases progress to develop chronic disease (Gran & Bohmer 1996, Neville 1983). Mortality is infrequent but is usually due to pulmonary involvement (Reich 2002).
DIABETES MELLITUS
Diabetes mellitus is associated with several musculoskeletal conditions including septic arthritis, adhesive capsulitis, Dupuytren’s contracture, palmar flexor tenosynovitis, carpal tunnel syndrome, neuropathic (Charcot) arthropathy and cheiroarthropathy (Cagliero et al 2002, Geoghegan et al 2004, Mathews et al 2007). The latter two are discussed below.
DIABETIC NEUROPATHIC (CHARCOT) ARTHROPATHY
Diabetic neuropathic arthopathy is a destructive arthropathy which occurs almost exclusively in the presence of a dense lower limb sensorimotor peripheral neuropathy. It affects 0.1–0.4% of diabetics (Rajbhandari et al 2002). Peak onset is in the 6th decade with an equal gender distribution. Diabetes has usually been long-standing. An acute neuropathic joint typically presents with local swelling, warmth and erythema that may not be referrable clinically to a particular joint. Although pain may be present, it is usually painless. Diabetic neuropathic arthropathy most commonly affects the foot. Foot pulses are usually present and are often hyperdynamic. There may be accompanying ulceration.
A high index of suspicion is necessary to avoid delay in treatment and permanent disability. The diagnosis of neuropathic arthopathy is made largely on clinical grounds although plain radiographs, isotope bone scans and MRI may help to differentiate the main differential diagnoses which include osteomyelitis, cellulitis and acute gout. Causes other than diabetes mellitus include leprosy and tabes dorsalis (syphilis). Management of the acute neuropathic joint aims to reduce weight-bearing of the affected limb and may require plaster casts or orthoses (Armstrong et al 1997). Bisphosphonates may also be of benefit (Jude et al 2001). Management of the chronic neuropathic joint relies upon adaptation of footwear. Surgical correction of deformity may also play a role.
DIABETIC CHEIROARTHROPATHY
Diabetic cheiroarthropathy is characterised by an inability to fully extend the metacarpophalangeal joints and scleroderma-like skin thickening (Crispin & Cocer-Varela 2003). It is seen more frequently in insulin-dependent disease and associates with disease duration and vascular complications (Frost & Beischer 2001). The characteristic clinical sign is a positive “prayer” sign which describes the inability to flatten the hands together as in prayer (Crispin & Cocer-Varela 2003). Treatment can be challenging and consists of physiotherapy, wax therapy, corticosteroid injection or, in severe cases, surgery.
Part 2: Vasculitis
DEFINITION
Vasculitis literally means inflammation of blood vessels and indicates that the vessel wall itself is the site and target of inflammation. This results in damage (necrosis) of the vessel wall, hence the term sometimes used-necrotizing vasculitis.
The consequence of vasculitis depends on the size, site and number of blood vessels involved and a number of different clinical patterns are recognised (Scott & Watts 1994) (Table 23.2). Symptoms range from benign skin rashes to life threatening diseases involving vital internal organs. The most serious types of vasculitis are those that involve small/medium arteries. It is also important to recognise that vasculitis can occur as a primary event (arising de novo/primary vasculitis) or in association with a number of infections and connective tissue diseases, including rheumatoid arthritis, Sjögren’s syndrome and systemic lupus erythematosus.
Table 23.2 Classification of systemic vasculitis
| DOMINANT VESSELS INVOLVED | PRIMARY | SECONDARY |
|---|---|---|
| Large arteries | Giant cell arteritis | |
| Takayasu arteritis | Aortitis associated with RA | |
| Isolated CNS angiitis | Infection (e.g. syphilis) | |
| Medium arteries | Classical PAN | Infection (e.g. Hepatitis B) |
| Kawasaki disease | Hairy cell leukaemia | |
| Small vessels and medium arteries | Wegener’s granulomatosisa | Vasculitis 2° to RA, SLE, SS |
| Churg-Strauss syndromea | Drugs, malignancy | |
| Microscopic polyangiitisa | Infection (e.g. HIV) | |
| Drugsb | ||
| Small vessels (leukocytoclastic) | Henoch-Schönlein purpura | Infection (e.g.hepatitis B, C) |
| Essential mixed cryoglobulinaemia | ||
| Cutaneous leukocytoclastic angiitis |
CNS, central nervous system; HIV, human immunodeficiency virus; PAN, polyarteritis nodosa; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus; SS, Sjögren’s syndrome.
a Diseases most commonly associated with ANCA (anti-myeloperoxidase and anti-proteinase 3 antibodies), a significant risk of renal involvement, and which are most responsive to immunosuppression with cyclophosphamide.
b e.g. sulphonamides, penicillins, thiazide diuretics, and many others.
Reproduced from Scott & Watts (1994) with permission
EPIDEMIOLOGY/AETIOLOGY
The primary systemic vasculitides are rare. Giant cell arteritis is probably the most common, but the primary ANCA-associated vasculitides (Wegener’s granulomatosis, Churg-Strauss syndrome and microscopic polyangiitis) have an incidence of approximately 20 million/year. If all the vasculitides are lumped together, the incidence is approximately 150 million/year but this may still be an under-estimate as many cases of giant cell arteritis are seen in the community, and mild small vessel skin vasculitis (e.g. drug reaction) may go unrecognised or unreported.
The cause of vasculitis is in most circumstances unknown. Important infections linked to vasculitis include hepatitis B (classic polyarteritis nodosa) and hepatitis C (cryoglobulinaemic vasculitis). A number of drugs may trigger vasculitis, usually small vessel vasculitis. The ANCA-associated vasculitides have only recently come into the group of conditions thought to be autoimmune because of the presence of ANCA – an antibody directed against neutrophil cytoplasmic antigens, where the specificity for proteinase 3 is strongly linked to Wegener’s granulomatosis and myeloperoxidase to microscopic polyangiitis. These antibodies form part of the immunological investigations carried out in cases where vasculitis is suspected.
LARGE VESSEL VASCULITIS
Giant cell arteritis is commonly linked to polymyalgia rheumatica. It is a disease affecting particularly an older population over the age of 50, and usually over the age of 60, presenting commonly with severe temporal headaches, temporal artery tenderness and a high ESR. It is important that this diagnosis is recognised as it is very sensitive to steroid treatment, but untreated can lead to blindness because of involvement of blood vessels affecting the optic nerve (ciliary artery and branches). Takayasu’s arteritis by contrast affects a much younger population, is very rare in the UK and presents with non-specific systemic symptoms which later lead to loss of pulses and claudication, especially of the upper limbs.
MEDIUM VESSEL VASCULITIS
Polyarteritis nodosa is a term that has been around for over 140 years and was loosely applied to many cases of systemic vasculitis. Recent reviews and the introduction of testing for the ANCA antibodies has led to a reclassification of this as a disease restricted to medium sized vessels which is now quite rare. It is characterised by the formation of aneurysms in medium sized arteries. Patients can present with systemic symptoms, rash (particularly livido reticularis), nerve damage (mononeuritis multiplex), hypertension and internal organ infarction. The disease when triggered by hepatitis B is best treated with anti-viral treatment plus plasma exchange.
Kawasaki disease (mucocutaneous lymph node syndrome) is an acute inflammatory disease affecting children, usually under the age of 5 years, presenting with fever, rash, swollen lymph glands and palmoplantar erythema. In about a quarter of untreated children the coronary arteries can be involved, leading to a small but significant mortality (approx 2%) and sometimes later in life myocardial ischaemia and infarction.
MEDIUM AND SMALL VESSEL VASCULITIDES
This group is also sometimes known as the primary systemic vasculitides. They are separated from pure small vessel vasculitis because they can involve larger vessels and be associated with aneurysms, they are also particularly associated with ANCA and are not infrequently associated with significant renal impairment due to an inflammatory glomerulonephritis. Although they occur at any age, the peak incidence is 60–70 years.
Wegener’s granulomatosis is the most common of the three diseases in the UK and northern Europe (microscopic polyangiitis is commoner in southern Europe and Japan). It is characterised by granuloma formation involving the upper and lower airways and glomerulonephritis. The lungs are involved in about 45% with nodules which can cavitate, leading to haemoptysis. Upper airway involvement, particularly in the naso-pharynx, leads to destruction of the sinuses and presents often with epistaxis, crusting and also complications such as deafness. The prognosis of Wegener’s granulomatosis in terms of mortality relates to kidney involvement, but the upper airway involvement can lead to very significant morbidity and be difficult to treat.
Microscopic polyangiitis (MPA) is characterised by vasculitis that usually presents with kidney involvement, but sometimes presents with lung haemorrhage due to small vessel bleeding from vasculitis. The kidney changes are identical to Wegener’s-glomerulonephritis with few immune deposits (‘pauci immune’ glomerulo-nephritis). Churg-Strauss syndrome is characterised by allergy, particularly late onset asthma followed by systemic vasculitis with flitting pulmonary shadowing (presenting like pneumonia) with increased numbers of eosinophils in the circulation and in tissues involved with vasculitis. Asthma can pre-date the vasculitis by many years. Kidney involvement is rarer than (but identical to that seen) in Wegener’s and MPA, but particular features more common in Churg-Strauss syndrome are heart problems (peri/myocarditis) and neuropathy.
SMALL VESSEL VASCULITIS
This type of vasculitis is usually confined to the skin. It is important to recognise that if a patient presents with vasculitic skin rash this can be part of the other diseases outlined above. There are a number of very specific patterns of small vessel vasculitis known as Henoch-Schönlein purpura (HSP), cutaneous leukocytoclastic vasculitis and cryoglobulinaemia. Henoch-Schönlein purpura is mainly seen in children who present with a purpuric rash, arthritis (usually involving large joints), abdominal pain, and haematuria. The kidney disease is usually mild in comparison with the ANCA-associated vasculitides. Immunoglobulin A is detected in involved tissue, particularly the kidney. When HSP involves adults the kidney involvement can be more severe, and it may be difficult sometimes to differentiate from microscopic polyangiitis.
Cryoglobulins are proteins which precipitate in the cold. Cryoglobulinaemic vasculitis usually presents with a skin rash, but also digital ischaemia and ulcers, arthralgia and mild neuropathy. It is strongly linked to hepatitis C infection.
INVESTIGATIONS OF VASCULITIS
These can be split into different sections as follows:
DIFFERENTIAL DIAGNOSIS
The important diagnoses to exclude in a patient with suspected vasculitis include infection, particularly bacterial endocarditis (urgent echocardiography is therefore essential in all cases), and cholesterol embolism in the elderly can mimic vasculitis with skin infarction, as can antiphospholipid syndrome (clotting in blood vessels due to antiphospholipid antibodies). A very rare condition is atrial myxoma which is also detected by echocardiography.
The presence of protein or blood on urine dipstix is probably the most important investigation because kidney involvement can be sub-clinical and kidney damage can occur without any very specific symptoms. This in the presence of a systemic illness needs to be treated as an emergency. The white cell count can be useful when eosinophils are present, suggesting allergies such as Churg-Strauss syndrome. A low white count is usually suggestive of infection or a disease such as SLE. Very abnormal liver function tests raise the possibility of hepatitis (B or C).
Although ANCA is useful in aiding diagnosis (PR3 ANCA is highly specific (greater than 90%) for Wegener’s granulomatosis), tissue biopsy is still important to confirm the diagnosis to target the organs clinically involved. Renal biopsy is perhaps the most useful because of the link between kidney involvement and prognosis (see below). In patients that are difficult to diagnose, then angiography can be useful to show aneurysms. Finally, in addition to echocardiography for investigating the differential diagnosis of vasculitis, blood cultures and viral serology are essential because of the important differential diagnosis of infection.
PROGNOSIS
The natural history of primary systemic vasculitis untreated is of a progressive, usually fatal disease. Survival improved with the introduction of steroids for some conditions, but the most dramatic improvement followed the introduction of cyclophosphamide in the late 1960s and early 1970s. For example, the median survival of Wegener’s granulomatosis with no treatment was 5 months for those with renal involvement, 12.5 months with steroids alone but now the survival rate at 5 years is in excess of 80% in most series.
Small vessel vasculitis is usually associated with a good prognosis, as is large vessel vasculitis, except for specific problems such as blindness as outlined above.
TREATMENT
Treatment is dependent on the type of vasculitis. Small vessel vasculitis can often be treated with low dose corticosteroids and the large vessel vasculitides usually respond well to higher doses (oral prednisolone 40–60 mg per day). The duration of treatment is usually in excess of one year. Once symptoms settle the steroid dose can be rapidly reduced, titrated to clinical features and inflammatory markers such as the ESR/CRP.
Cyclophosphamide is almost always used to treat the primary systemic/ANCA-associated vasculitides combined with steroids. Cyclophosphamide can be given as continuous low dose oral therapy or intermittent pulse intravenous therapy. Both treatments are equally effective but pulse therapy is associated with a lower total cyclophosphamide dose and less toxicity. Particularly concerning problems with long-term oral cyclophosphamide are the risk of haemorrhagic cystitis and bladder tumours, but also infertility and infections. With intravenous cyclophosphamide bladder protection is enhanced by the addition of mesna and patients are often given trimethoprim-sulfamethoxazole to prevent infections with pneumocystis. Fertile males should be offered sperm storage before being given cyclophosphamide and ovarian function is often lost in females given cyclophosphamide, especially if over the age of 30–35. Corticosteroids are usually given at a dose of 1 mg/kg with rapid reduction after remission has been obtained within 2–3 months.
After remission has been induced, usually with cyclophosphamide and steroids, patients are switched to either azathioprine or methotrexate. Long-term treatment is usually advised because of a significant risk of relapse, higher in Wegener’s granulomatosis than in microscopic polyangiitis. Plasmapheresis is sometimes given to patients with severe renal disease or pulmonary haemorrhage, intravenous immunoglobulin is effective treatment for Kawasaki disease and the role of the new biologic agents is currently being evaluated, but the results of treatment with rituximab in particular appear to be encouraging.
REHABILITATION
Even with effective medical treatment, e.g. low dose corticosteroids, patients with vasculitis may have difficulty with walking, impaired muscle strength, limitation of joint movement, reduced proprioception and limitation in activities of daily living (Seo 2006). Monitoring of joint stiffness and pain can determine when referral back to the Rheumatologist is required. In polymyalgia rheumatica, this is more common in the shoulder and pelvic girdle, and neck. Stiffness can be worse on wakening and on exertion. Muscle strength can be difficult to determine in patients with polymyalgia rheumatica because of muscle tenderness and pain (Paget & Spiera 2006). Tenderness can lead to muscle atrophy although in the absence of pain, muscle strength should be normal (Paget & Spiera 2006). Dynamic exercises need to be tailored to the individual to maintain physical functioning and core stability and are often targeted to the shoulder girdle and hips. In severe cases and in the elderly referral to occupational therapy is warranted.
 Familiarise yourself with on-line patient resources suggested below:
Familiarise yourself with on-line patient resources suggested below:
http://www.rheumatology.org/public/factsheets/diseases_and_conditions/gout.asp?aud=pat
Pseudogout and calcium crystal diseases-accessed February 2009
What is sarcoidosis? - accessed February 2009
http://www.nhlbi.nih.gov/health/dci/Diseases/sarc/sar_whatis.html
Long-term complications - Musculoskeletal conditions-accessed February 2009
http://www.diabetes.org.uk/Guide-to-diabetes/Complications/Long_term_complications/Musculoskeletal_Conditions/
CASE STUDY 23.1
Mr Addisson is a 51-year-old bricklayer. One day after being admitted to hospital with a myocardial infarction, he wakes with an acutely painful, swollen, red left knee. He reports several episodes of acute gout affecting his big toes in the past, which his GP has treated with non-steroid anti-inflammatory drugs. He has also recently been commenced on allopurinol 100 mg daily. He is overweight and drinks four pints of beer every day.
Investigation
An aspiration of the left knee is performed. Examination of synovial fluid using compensated polarised light microscopy identifies monosodium urate crystals confirming the diagnosis of gout. No organisms are identified on Gram stain and culture. Serum urate is 480 micromol/L (normal range 200–400).
Management
Ice-packs are applied locally to the joint. An intra-articular injection of corticosteroid into the left knee is performed providing rapid relief of his pain and swelling. The joint is rested initially and he is then mobilised as his cardiovascular status allows. He is advised to lose weight and to restrict his alcohol intake. Once he is discharged, follow-up is arranged to increase the dose of allopurinol with the aim of lowering the serum urate into the lower half of the normal range.
References and further reading
Abbott R.D., Brand F.N., Kannel W.B., et al. Gout and coronary heart disease: the Framingham Study. J. Clin. Epidemiol.. 1988;41:237-242.
Alloway J.A., Moriarty M.J., Hoogland Y.T., et al. Comparison of triamcinolone acetonide with indomethacin in the treatment of acute gouty arthritis. J. Rheumatol.. 1993;20:111-113.
Armstrong D.G., Todd W.F., Lavery L.A., et al. The natural history of acute Charcot’s arthropathy in a diabetic foot specialty clinic. Diabet. Med.. 1997;14:357-363.
Arromdee E., Michet C.J., Crowson C.S., et al. Epidemiology of gout: is the incidence rising? J. Rheumatol.. 2002;29:2403-2406.
Baughman R.P., Drent M., Kavuru M., et al. Infliximab therapy in patients with chronic sarcoidosis and pulmonary involvement. Am. J. Respir. Crit. Care Med.. 2006;174:795-802.
Baughman R.P., Teirstein A.S., Judson M.A., et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am. J. Respir. Crit. Care Med.. 2001;164:1885-1889.
Cagliero E., Apruzzese W., Perlmutter G.S., et al. Musculoskeletal disorders of the hand and shoulder in patients with diabetes mellitus. Am. J. Med.. 2002;112:487-490.
Campion E.W., Glynn R.J., DeLabry L.O. Asymptomatic hyperuricemia, risks and consequences in the normative aging study. Am. J. Med.. 1987;82:421-426.
Cheng L.S., Chiang S.L., Tu H.P., et al. Genomewide scan for gout in Taiwanese Aborigines reveals linkage to chromosome 4q25. Am. J. Human Genet.. 2004;75:498-503.
Choi H.K., Atkinson K., Karlson E.W., et al. Obesity, weight change, hypertension, diuretic use, and risk of gout in men: the health professionals follow-up study. Arch. Intern. Med.. 2005;165:742-748.
Choi H.K., Atkinson K., Karlson E.W., et al. Alcohol intake and risk of incident gout in men: a prospective study. Lancet. 2004;363:1277-1281.
Choi H.K., Atkinson K., Karlson E.W., et al. Purine-rich foods, dairy and protein intake, and the risk of gout in men. N. Engl. J. Med.. 2004;350:1093-1103.
Coakley G., Mathews C., Field M., et al. BSR & BHPR, BOA, RCGP and BSAC guidelines for management of the hot swollen joint in adults. Rheumatology. 2006;45:1039-1041.
Cooper C., Cawley M.I. Bacterial arthritis in an English health district: a 10 year review. Ann. Rheum. Dis.. 1986;45:458-463.
Crispin J.C., Cocer-Varela J. Rheumatologic manifestations of diabetes mellitus. Am. J. Med.. 2003;114:753-757.
Demirkok S.S., Basaranoglu M., Akinci E.D., et al. Analysis of 275 patients with sarcoidosis over a 38 year period; a single-institution experience. Respir. Med.. 2007;101:1147-1154.
Dieppe P.A., Alexander G.J., Jones H.E., et al. Pyrophosphate arthropathy: a clinical and radiological study of 105 cases. Ann. Rheum. Dis.. 1982;41:371-376.
Doty J.D., Mazur J.E., Judson M.A. Treatment of sarcoidosis with infliximab. Chest. 2005;127:1064-1071.
Fernandez C., Noguera R., Gonzalez J.A., et al. Treatment of acute attacks of gout with a small dose of intraarticular triamcinolone acetonide. J. Rheumatol.. 1999;26:2285-2286.
Frost D., Beischer W. Limited joint mobility in type 1 diabetic patients: associations with microangiopathy and subclinical macroangiopathy are different in men and women. Diab. Care. 2001;24:95-99.
Geoghegan J.M., Clark D.I., Bainbridge L.C., et al. Risk factors in carpal tunnel syndrome. J. Hand Surg. (British). 2004;29:315-320.
Glennas A., Kvien T.K., Melby K., et al. Acute sarcoid arthritis: occurrence, seasonal onset, clinical features and outcome. Br. J. Rheumatol.. 1995;34:45-50.
Gran J.T., Bohmer E. Acute sarcoid arthritis: a favourable outcome? A retrospective survey of 49 patients with review of the literature. Scand. J. Rheumatol.. 1996;25:70-73.
Gribbin J., Hubbard R.B., Le J.I., et al. Incidence and mortality of idiopathic pulmonary fibrosis and sarcoidosis in the UK. Thorax. 2006;61:980-985.
Gumpel J.M., Johns C.J., Shulman L.E. The joint disease of sarcoidosis. Ann. Rheum. Dis.. 1967;26:194-205.
Gupta M.N., Sturrock R.D., Field M. A prospective 2-year study of 75 patients with adult-onset septic arthritis. Rheumatology. 2001;40:24-30.
Harris C.M., Lloyd D.C., Lewis J. The prevalence and prophylaxis of gout in England. J. Clin. Epidemiol.. 1995;48:1153-1158.
Huang C.M., Lo S.F., Lin H.C., et al. Correlation of oestrogen receptor gene polymorphism with gouty arthritis. Ann. Rheum. Dis.. 2006;65:1673-1674.
Jones A.C., Chuck A.J., Arie E.A., et al. Diseases associated with calcium pyrophosphate deposition disease. Semin. Arthrit. Rheum.. 1992;22:188-202.
Jordan K.M., Cameron J.S., Snaith M., et al. British Society for Rheumatology and British Health Professionals in Rheumatology guideline for the management of Gout. Rheumatology. 2007;46:1372-1374.
Jude E.B., Selby P.L., Burgess J., et al. Bisphosphonates in the treatment of Charcot neuroarthropathy: a double-blind randomised controlled trial. Diabetologia. 2001;44:2032-2037.
Kaandorp C.J., Dinant H.J., van de Laar M.A., et al. Incidence and sources of native and prosthetic joint infection: a community based prospective survey. Ann. Rheum. Dis.. 1997;56:470-475.
Kaandorp C.J., Krijnen P., Moens H.J., et al. The outcome of bacterial arthritis: a prospective community-based study. Arthrit. Rheum.. 1997;40:884-892.
Lawrence R.C., Helmick C.G., Arnett F.C., et al. Estimates of the prevalence of arthritis and selected musculoskeletal disorders in the United States. Arthrit. Rheum.. 1998;41:778-799.
Li-Yu J., Clayburne G., Sieck M., et al. Treatment of chronic gout. Can we determine when urate stores are depleted enough to prevent attacks of gout? J. Rheumatol.. 2001;28:577-580.
Mana J., Gomez-Vaquero C., Montero A., et al. Löfgren’s syndrome revisited: a study of 186 patients. Am. J. Med.. 1999;107:240-245.
Mana J., Salazar A., Manresa F. Clinical factors predicting persistence of activity in sarcoidosis: a multivariate analysis of 193 cases. Respiration. 1994;61:219-225.
Mathews C.J., Kingsley G., Field M., et al. Management of septic arthritis: a systematic review. Ann. Rheum. Dis.. 2007;66:440-445.
Mikuls T.R., Farrar J.T., Bilker W.B., et al. Gout epidemiology: results from the UK General Practice Research Database, 1990–1999. Ann. Rheum. Dis.. 2005;64:267-272.
Morris I., Varughese G., Mattingly P. Colchicine in acute gout. Br. Med. J.. 2003;327:1275-1276.
Mukhtyar, C., Guillevin, L., Cid, M.C., et al., 2008. EULAR recommendations for the management of large vessel vasculitis. Ann. Rheum. Dis. published online 15 Apr.
Mukhtyar, C., Guillevin, L., Cid, M.C., et al., 2008. EULAR recommendations for the management of primary small and medium vessel vasculitis. Ann. Rheum. Dis. Published online 15 Apr.
Nade S. Septic arthritis. Bailliéres Best Prac. Res. Clin. Rheumatol.. 2003;17:183-200.
Neame R.L., Carr A.J., Muir K., et al. UK community prevalence of knee chondrocalcinosis: evidence that correlation with osteoarthritis is through a shared association with osteophyte. Ann. Rheum. Dis.. 2003;62:513-518.
Neville E., Walker A.N., James D.G. Prognostic factors predicting the outcome of sarcoidosis: an analysis of 818 patients. Quart. J. Med.. 1983;52:525-533.
Nixon J.C., Mortimer K., Holden S., et al. Clinical features of septic arthritis in patients admitted to hospital in Nottingham 1997–2001. Rheumatol.. 2007;46(Suppl 1):i145.
Paget, S.A., Spiera, R.F.P., 2006. Polymyalgia Rheumatica. In: Clinical Care in the Rheumatic Diseases, third ed. Association of Rheumatology Health Professionals, pp. 153–156., third ed.
Paramothayan, N.S., Lasserson, T.J., Jones, P.W., 2005. Corticosteroids for pulmonary sarcoidosis. Coch. Database. Syst. Rev. CD001114-.
Paramothayan, S., Lasserson, T.J., Walters, E.H., 2006. Immunosuppressive and cytotoxic therapy for pulmonary sarcoidosis. Coch. Database. Syst. Rev. 3 CD003536-.
Pendleton A., Johnson M.D., Hughes A., et al. Mutations in ANKH cause chondrocalcinosis. Am. J. Human Genet.. 2002;71:933-940.
Perruquet J.L., Harrington T.M., Davis D.E., et al. Sarcoid arthritis in a North American Caucasian population. J. Rheumatol.. 1984;11:521-525.
Pettersson T. Sarcoid and erythema nodosum arthropathies. Bailliéres Best Pract. Res. Clin. Rheumatol.. 2000;14:461-476.
Rajbhandari S.M., Jenkins R.C., Davies C., et al. Charcot neuroarthropathy in diabetes mellitus. Diabetologia. 2002;45:1085-1096.
Reich J.M. Mortality of intrathoracic sarcoidosis in referral vs population-based settings: influence of stage, ethnicity, and corticosteroid therapy. Chest. 2002;121:32-39.
Roddy, E., Zhang, W., Doherty, M., 2007. Are joints affected by gout also affected by osteoarthritis? Ann. Rheum. Dis. Published Online First: 6 February 2007. doi:10.1136/ard.2006.063768.
Rubin B.R., Burton R., Navarra S., et al. Efficacy and safety profile of treatment with etoricoxib 120 mg once daily compared with indomethacin 50 mg three times daily in acute gout: a randomized controlled trial. Arthrit. Rheum.. 2004;50:598-606.
Rybicki B.A., Major M., Popovich J., et al. Racial differences in sarcoidosis incidence: a 5-year study in a health maintenance organization. Am. J. Epidemiol.. 1997;145:234-241.
Saleh S., Ghodsian S., Yakimova V., et al. Effectiveness of infliximab in treating selected patients with sarcoidosis. Resp. Med.. 2006;100:2053-2059.
Schlesinger N.Jr., Baker D.G., Schumacher H.R. Serum urate during bouts of acute gouty arthritis. J. Rheumatol.. 1997;24:2265-2266.
Schlesinger N., Detry M.A., Holland B.K., et al. Local ice therapy during bouts of acute gouty arthritis. J. Rheumatol.. 2002;29:331-334.
Schumacher H.R.Jr., Boice J.A., Daikh D.I., et al. Randomised double blind trial of etoricoxib and indometacin in treatment of acute gouty arthritis. Br. Med. J.. 2002;324:1488-1492.
Scott D.G.I., Watts R.A. Classification and epidemiology of systemic vasculitis. Br. J. Rheumatol.. 1994;33:897-899.
Seo, P., 2006. Vasculitis. In: Clinical Care in the Rheumatic Diseases, third ed. Association of Rheumatology Health Professionals (third ed), pp. 199–202.
Sweiss N.J., Welsch M.J., Curran J.J., et al. Tumor necrosis factor inhibition as a novel treatment for refractory sarcoidosis. Arthrit. Rheum.. 2005;53:788-791.
Taniguchi A., Urano W., Yamanaka M., et al. A common mutation in an organic anion transporter gene, SLC22A12, is a suppressing factor for the development of gout. Arthrit. Rheum.. 2005;52:2576-2577.
Visser H., Vos K., Zanelli E., et al. Sarcoid arthritis: clinical characteristics, diagnostic aspects, and risk factors. Ann. Rheum. Dis.. 2002;61:499-504.
Wallace K.L., Riedel A.A., Joseph-Ridge N., et al. Increasing prevalence of gout and hyperuricemia over 10 years among older adults in a managed care population. J. Rheumatol.. 2004;31:1582-1587.
Wang W.H., Chang S.J., Wang T.N., et al. Complex segregation and linkage analysis of familial gout in Taiwanese aborigines. Arthrit. Rheum.. 2004;50:242-246.
Watts R.A., Scott D.G., Pusey C.D., et al. Vasculitis – aims of therapy. A review. Rheumatology. 2000;39:229-232.
Weston V.C., Jones A.C., Bradbury N., et al. Clinical features and outcome of septic arthritis in a single UK Health District 1982–1991. Ann. Rheum. Dis.. 1999;58:214-219.
Williams C.J., Pendleton A., Bonavita G., et al. Mutations in the amino terminus of ANKH in two US families with calcium pyrophosphate dihydrate crystal deposition disease. Arthrit. Rheum.. 2003;48:2627-2631.
Wise C.M. Crystal-associated arthritis in the elderly. Rheum. Dis. Clin. N. Am.. 2007;33:33-55.
Wright G.D., Doherty M. Calcium pyrophosphate crystal deposition is not always ‘wear and tear’ or aging. Ann. Rheum. Dis.. 1997;56:586-588.
Zhang W., Doherty X., Bardin T., et al. EULAR evidence based recommendations for gout. Ann. Rheum. Dis.. 2006;65:1301-1311. Part I: Diagnosis. Report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT)
Zhang W., Doherty M., Bardin T., et al. EULAR evidence based recommendations for gout. Part II: Management. Ann. Rheum. Dis.. 2006;65:1312-1324. Part II: Management. Report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT)
Zhang W., Neame R., Doherty S., et al. Relative risk of knee chondrocalcinosis in siblings of index cases with pyrophosphate arthropathy. Ann. Rheum. Dis.. 2004;63:969-973.
Further reading
Nade S. Septic arthritis. Bailliéres best practice in research. Clin. Rheumatol.. 2003;17:183-200.
Jordan K.M., Cameron J.S., Snaith M., et al. BSR and BHPR guideline for the management of gout. Rheumatology. 2007;46:1372-1374.
Zhang W., Doherty M., Bardin T., et al. EULAR evidence based recommendations for gout. Part I: Diagnosis. Ann. Rheum. Dis.. 2006;65:1301-1311. Report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT)
Zhang W., Doherty M., Bardin T., et al. EULAR evidence based recommendations for gout. Part II: Management. Ann. Rheum. Dis.. 2006;65:1312-1324. Report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT)
Wise C.M. Crystal-associated arthritis in the elderly. Rheum. Dis. Clin. N. Am.. 2007;33:33-55.
Pettersson T. Sarcoid and erythema nodosum arthropathies. Bailliéres best practice in research. Clin. Rheumatol.. 2000;14:461-476.
Crispin J.C., Cocer-Varela J. Rheumatologic Manifestations of diabetes mellitus. Am. J. Med.. 2003;114:753-757.
Mukhtyar, C., Guillevin, L. & Cid, M.C., et al., 2008. EULAR recommendations for the management of primary small and medium vessel vasculitis. Ann. Rheum. Dis. published online 15 Apr.
Mukhtyar, C., Guillevin, L. & Cid, M.C., et al., 2008. EULAR recommendations for the management of large vessel vasculitis. Ann. Rheum. Dis. published online 15 Apr.