6 Atopic dermatitis
INTRODUCTION
Atopic dermatitis is a genetically determined pruritic dermatitis, associated with an immediate (type 1) hypersensitivity to specific environmental allergens. It is one of the most common causes of chronically recurring inflammatory skin disease and involves complex interactions of environmental, microbial, genetic, immunological and pharmacological factors.
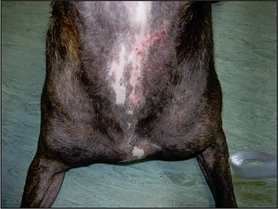
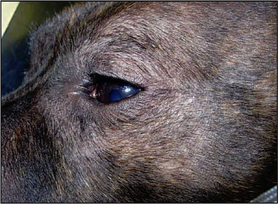
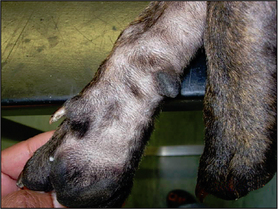
The most common presenting signs are pruritus, erythema and secondary microbial infections. The distribution of the pruritus and the lesions typically involve the face, the ears, the ventral aspects of the abdomen, the perianal areas and the feet. The lesions vary from erythema and salivary staining, to self-induced alopecia, hyperpigmentation, lichenification, scaling, crusting and erosions. Otitis externa is seen in four out of five cases, usually involving the concave aspects of the pinna and the vertical ear canals. Recurring conjunctivitis, periocular dermatitis and sneezing may be evident in some cases.
Secondary microbial infections with Staphylococcus spp. or Malassezia pachydermatis are frequent findings in cases of atopic dermatitis and their importance should not be underestimated. Commonly, one of the first signs of the onset of atopic dermatitis is the development of a cutaneous yeast or bacterial infection, and infection is a major reason for the flare-up of pruritus in apparently well-controlled cases and one of the most common reasons for clients seeking veterinary attention. Clinical signs associated with staphylococcal infections include papules, pustules, epidermal collarettes, scaling and crusting. Malassezia dermatitis tends to cause erythema, greasy secretion and the matting of hair shafts over occluded areas such as the ventral neck, or between the digits.
It is common for the pruritus to be present initially only during the summer months, but with the passing of time it tends to become a year-round problem.
At least three of the following major and minor criteria should be satisfied to make a diagnosis of atopic dermatitis:
CASE HISTORY
The main points of interest in the history were:
In summary, this was a young dog, of a breed predisposed to atopic dermatitis, with a non-seasonal, glucocorticoid-responsive, facial, aural, pedal and ventral pruritus, and flea control was intermittent.
CLINICAL EXAMINATION
A full physical examination should be carried out prior to examining the skin. The dermatological examination should include all of the skin, extending from the tip of the nose to the tip of the tail and from the dorsum to the pads of the feet. One of the biggest pitfalls is to examine only the affected sites, thus missing other clues that could aid the diagnosis.
The early signs of atopic dermatitis can be subtle and there may be no other clinical signs other than pruritus, although, as in this case, many dogs will have erythema, self-induced alopecia, excoriations, papules, hyperpigmentation and other changes associated with secondary microbial overgrowth or infections. Some individuals will show erythema of the concave aspects of the pinna and the vertical ear canals without any history of ear disease.
The significant clinical findings in this case were:
In all cases, unless there is an immediate diagnosis, it is best to formulate a list of differential diagnoses and methodically rule each one in or out. This list is drawn up from a consideration of the history and clinical signs, the latter comprising both the general pattern of disease as well as individual lesion recognition. This case is typical of many cases of pruritus in that the differential diagnosis list was quite extensive.
In this case the lesions consisted of inflammatory and non-inflammatory alopecia, hypotrichosis, papules, epidermal collarettes, acute moist dermatitis and erythema.
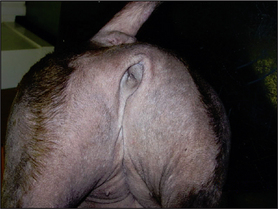
There were also two different types of hair loss. There were areas of self-induced alopecia and inflammation, and other areas of alopecia where the skin was not inflamed. In this case, the non-inflammatory alopecia over the caudal thighs was unlikely to have arisen because of self-trauma or a folliculitis (the common causes of inflammatory alopecia). This presentation of non-inflammatory alopecia is common in Staffordshire bull terriers with pattern alopecia, which is a poorly understood, non-inflammatory alopecia that results in so-called ‘miniaturization’ of hair follicles and in the well-recognized, breed-associated patterns of alopecia.
CASE WORK-UP
Staffordshire bull terriers are genetically predisposed to demodicosis; therefore, skin scrapings and hair plucks should always be performed on this breed, or in any case where there is evidence of papules, pustules, alopecia, crusting or scaling. Primary lesions such as papules are commonly associated with staphylococcal infections and/or sarcoptic mange. Cytology and response to therapy are used to confirm the involvement of secondary pyoderma and Malassezia dermatitis, both of which can contribute significantly to the degree of pruritus.
Diagnostic tests: The following diagnostic tests were performed:
Therapeutic trials: Initially, the following therapeutic trials were performed concurrently:
Re-examination: On re-examination 4 weeks later, the pruritus had persisted but the papular lesions had resolved and no microbial organisms were seen on repeat cytology. The ongoing pruritus confirmed an underlying allergic aetiology.
Dietary trials: To rule out the involvement of an adverse food reaction, a hydrolysed diet was fed for 8 weeks. No other foods and only water to drink were allowed during this period. Weekly bathing with an antimicrobial shampoo was continued. Flea control was continued using imidacloprid every 4 weeks. The pruritus persisted during this period, with intermittent episodes of increased pruritus. In this case the diet trial ruled out an adverse food reaction as a cause of the disease.
DIAGNOSIS
This patient satisfied several of the major and minor clinical signs of atopic dermatitis, and there was no decrease in pruritus in response to the therapeutic and diet trials. Therefore, a clinical diagnosis of atopic dermatitis was made, which was supported by further testing.
PROGNOSIS
Although the condition is not life threatening most atopic dogs require lifelong management. Severe microbial infections and long-term adverse effects of drugs used for the management could influence the patient’s longevity. The owner was advised that there is no known treatment for the pattern alopecia.
AETIOPATHOGENESIS
A full review of the aetiopathogenesis of atopic dermatitis is beyond the scope of this section. However, the aetiopathogenesis is complex and multifactorial, and failure to recognize all the factors involved will probably result in unsuccessful management of the condition.
The primary event is the presentation of the allergen to the immune system that, through a chain of events, results in sensitization and subsequent hypersensitivity. A recent study showed that allergen exposure by either the percutaneous or inhalant route resulted in very similar lesion distribution and clinical signs. Other evidence supports the percutaneous penetration of allergens and the involvement of Langerhan’s cells in the processing of the antigens, which are then presented to T-helper cells. In genetically predisposed individuals, this activates proliferation of TH2 cells that secrete cytokines such as IL-13, IL-5 and IL-4. IL-4 favouring the synthesis of IgE by B cells. This allergen-specific IgE binds, via the high-affinity Fc receptors, to dermal mast cells.
Subsequent re-exposure to allergens results in the cross-linking of adjacent IgE, leading to mast cell de-granulation and the release of both preformed and newly synthesized inflammatory mediators. The release of preformed mediators, such as histamine, tryptase, chymase and heparin, results in the early signs of inflammation, such as erythema and pruritus. The newly formed mediators include the prostaglandins, leucotrienes, cytokines and chemokines. They are associated with the late-phase reaction that occurs about 4–6 hours after the initial event and are responsible for the recruitment of the inflammatory cells (TH2 cells and eosinophils) commonly seen on histological examination of the lesional skin.
One of the primary abnormalities in atopic dermatitis is a defective epidermal barrier function that aids penetration of allergens and irritants. Furthermore, atopic dogs have higher levels of bacterial colonization compared to healthy animals. Both these factors further exacerbate the pruritus which is also affected by the allergen load, which in turn depends on factors such as climate, season, indoor environment and geographical location. House dust mites, storage mites, grass pollens, weed pollens, tree pollens, mould spores, insect antigens and dander are all potential allergens in canine atopic dermatitis.
In animals in which the clinical disease is IgE mediated and is demonstrated by serum or intradermal allergy tests, the condition is referred to as atopic dermatitis. There is a subset of animals with clinical signs that satisfy the clinical diagnostic criteria of atopic dermatitis yet fail to demonstrate any allergen-specific IgE either by serum or intradermal testing; in these cases, the condition is referred to as atopic-like dermatitis.
EPIDEMIOLOGY
The exact incidence of atopic dermatitis is not known, although the disease is increasingly being recognized in general practice. The onset of clinical signs is usually between 1 and 3 years of age, and although there is an increase in the number of dogs being presented above 6 months, it is rarely seen in dogs under that age. Similarly, onset is not usually seen in dogs over 7 years old, unless there has been a change in environmental allergen load in a predisposed animal (e.g. a house move or exposure to novel allergens).
Genetic factors have been implicated in the development of the disease and breed predisposition is seen. Although atopic dermatitis can occur in any dog, certain breeds such as terriers, golden retrievers, Labradors, German shepherd dogs, English bull terriers and bulldogs are particularly predisposed.
The definitive diagnosis of atopic dermatitis is based on major and minor historical and clinical features, and on ruling out all other possible causes of the pruritus. From a practical point of view, at least three or four major features should be present to satisfy the diagnosis and the minor criteria are used to further support the diagnosis.
The purpose of allergy (intradermal or serological) testing is to support the clinical diagnosis, to allow the selection of allergens for immunotherapy and, where possible, targeted avoidance of them. It also allows the introduction of measures to reduce the allergen load in the environment. A small subset of animals will have negative serum and intradermal tests (see ‘Aetiopathogenesis’ section).
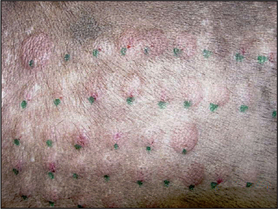
Intradermal testing
Intradermal testing is preferred by dermatologists for determining which allergens the patient is sensitized to. The test is based on an immediate hypersensitivity response (10–25 minutes) to environmental allergens, resulting in an erythematous wheal at the site of a positive reaction. A positive histamine control and a negative control are always included. An objective scoring system may be used but experienced clinicians tend to use a subjective scoring system based on the diameter, erythema and the elevation of the wheal at each test site, comparing them to the reactions at the positive and negative control sites. Anti-inflammatory drug therapy interferes with the sensitivity of the test and therefore it is important to make sure that the timing of the test takes into account the withdrawal times of any medication the animal may have been taking. However, there are a number of other reasons for both false-negative and false-positive reactions, and up to 15% of individuals with typical clinical signs of atopic dermatitis are negative to intradermal (or serum) allergy testing.
False negatives
Common reasons for negative intradermal allergy tests, when a clinical diagnosis of atopic dermatitis has been made, are:
Serum allergy tests
Serum allergy tests measure the levels of allergen-specific IgE and several laboratories in the UK offer the test. They are performed using the high-affinity Fc receptor technology, or using monoclonal or polyclonal antibodies. The Fc receptor, found on the surface of mast cells, binds specifically to IgE with a high affinity. This test is considered the more reliable serological test, because it reduces the false-positive reactions that can be associated with the presence of IgG.
The main advantages of serological testing are convenience to the owner and clinician, because it does not require the patient to be anaesthetized or clipped. In the past it has been suggested that serum IgE levels are unaffected by drugs; however, more recent reports suggest that they do have an effect and therefore withdrawal times, similar to those in intradermal testing, should be followed.
TREATMENT AND MANAGEMENT
The management of atopic dermatitis involves a combination of allergen avoidance, control of flare factors (such as secondary bacterial and yeast infections and ectoparasitism), allergen-specific immunotherapy and anti-inflammatory treatment. Frequently, a combination of treatments is a more effective way of managing this disease, rather than reliance on just one therapy. This approach also helps to minimize the use of those treatments which have severe side-effects.
Once atopic dermatitis is diagnosed, the client needs to understand that:
The clinician needs to be prepared to tailor the treatment to the needs of the patient and the client at any given time.
Allergen-specific immunotherapy: The mechanism of allergen-specific immunotherapy is not known, but by presenting large volumes of allergen to the immune system by subcutaneous injections rather that very small amounts by percutaneous absorption, the aim is to alter the way the immune system responds to the allergen.
The treatment involves the administration of allergen extracts, identified either by intradermal testing or by serological testing, by subcutaneous injections in increasing amounts over several weeks (induction phase) until the maintenance dose is reached. Thereafter the maintenance dose is usually injected monthly (maintenance phase), although this may vary between patients. There are a number of different induction protocols described depending on whether an aqueous or alum precipitated immunotherapy is used. These protocols are usually specified by the laboratory or company providing the therapy. The response may take 3–6 months to become noticeable; however, at least 10 months of treatment is recommended before making a final decision on efficacy. The reported success rates vary between 50% and 70%. In general, a third of patients may be expected to have their symptoms fully controlled, a third will require additional treatment to control symptoms and a third will derive no benefit at all from immunotherapy. It is evident in some patients that although they may require other treatment to control pruritus, the use of immunotherapy allows dosage reductions of these other treatments, which may have important cost and safety implications. Concomitant therapy may be required during the induction phase of treatment. Most cases require lifelong treatment and the majority of cases where immunotherapy is withdrawn because they are apparently cured tend to relapse months or years later. The frequency of injections can be tailored to individual needs. The therapy has few side-effects, is easy to administer and is cost-effective for long-term management.
Factors that influence the outcome of allergen-specific immunotherapy are:
Anaphylaxis is rare; however, if it occurs emergency treatment is required. It is recommended that the patient should be kept at the veterinary surgery for at least 30 minutes following the injections during the induction phase.
Glucocorticoids: Glucocorticoids are highly effective in relieving pruritus and are often required in the early stages of the treatment to break the itch–scratch cycle. However, ideally they should not be used until all microbial infections have been treated. Anti-inflammatory doses of oral methylprednisolone or prednisolone are the drugs of choice and the treatment should be tapered to alternate days or every third day as soon as possible. Adverse effects include polyuria, polydipsia, polyphagia and recurrent pyoderma in some individuals. Most atopic dogs with non-seasonal pruritus require continuous therapy and, when glucocorticoids are used, it is best to inform the client of the potential long-term effects of these drugs and the dose should be adjusted to the lowest dosage that just controls the pruritus.
Ciclosporin: Ciclosporin has been recently licensed for the management of atopic dermatitis in dogs. It is an immunomodulating drug that inhibits the activation of T cells and consequently the cascade of immunological events that lead to clinical disease. It is reported to have a success rate of about 80% and experience has shown it to be a useful drug in the management of atopic dermatitis. It also appears to be a useful drug when used to control pruritus during the induction phase of immunotherapy. However, the drawbacks of this drug include its expense (especially for large dogs), the difficulty in administration to some dogs, vomiting, diarrhoea and abdominal pain. Other adverse effects include gingival hyperplasia, lameness, increased hair growth and papillomatosis. Unlike glucocorticoids, the long-term adverse effects of this drug are not yet known.
Antihistamines: The effect of this group of drugs is variable and is thought to benefit about 15–30% of dogs. They act on H1 receptors and therefore interfere with the action of histamine on blood vessels and histamine-induced pruritus. However, the complex pathogenesis of atopic dermatitis suggests that only a small proportion of the pruritus may be histamine induced. Therefore, antihistamines may be most successful when used in dogs during the early stages of the disease or before mast cell degranulation has occurred. Individual responses to antihistamines vary and therefore no universal drug is recommended (Table 6.1). Those individuals that fail to respond to the antihistamine the clinician is most comfortable using should undergo a trial treatment for 10–14 days with another. A recent review of published studies showed that a combination of chlorpheniramine and hydroxyzine may be the most effective way of using this group of drugs.
Table 6.1 Most commonly used antihistamines in dogs
| Drug | Dose |
|---|---|
| Clemastine | 1 mg/10 kg p.o. every 12 hours |
| Hydroxyzine | 2 mg/kg p.o. every 8–12 hours |
| Chlorpheniramine | 2–12 mg p.o. every 12 hours |
| Diphenhydramine | 1–2 mg/kg p.o. every 8–12 hours |
| Promethazine | 0.2–0.4 mg/kg p.o. every 8 hours |
| Alimemazine | 0.5–2 mg/kg p.o. every 12 hours |
Essential fatty acids (EFAs): Although, on their own, essential fatty acids have little effect in managing the pruritus, they have been shown to have synergistic action with corticosteroids and antihistamines. EFAs restore and maintain the epidermal lipid barrier, which may reduce the allergen penetration and episodes of microbial infections. In addition, they can alter the metabolism of arachidonic acid, resulting in production of anti-inflammatory prostaglandins and leucotrienes. The best response is thought to result from administration of EFA containing ratios of omega-6 fatty acids (evening primrose oil) to omega-3 fatty acids (marine fish oil) of between 5 : 1 and 10 : 1.
Plant extracts: Phytopica® contains an extract obtained from three plants, Rehmannia glutinosa, Paeonia lactiflora and Glycyrrhiza uralensis. It was shown to significantly reduce Canine Atopic Dermatitis Extent and Severity Index (CADESI) scores in approximately 25% of dogs in a study, although there was no significant reduction in owner assessment of pruritus between control and treated groups. The product is available as a powder which is sprinkled on food and is palatable.
TOPICAL TREATMENTS
Topical glucocorticoids: Topical glucocorticoids may be of value in some individuals early in the course of inflammation. There are a number of ointments, creams and gels available for veterinary use in the UK containing a combination of antimicrobial and a glucocorticoid with varying potency depending on the product. Although of benefit, particularly when treating focal lesions, they have their limitations because prolonged use of topical glucocorticoids can lead to atrophy of the skin, systemic absorption, and signs consistent with iatrogenic hyperadrenocorticism. Furthermore, the presence of hair and the area affected also limit their use. However, recently a glucocorticoid spray containing 0.584% m/vol of hydrocortisone aceponate has been licensed for topical use in dogs. Its benefits include the ease of application even on haired skin, lack of systemic absorption and greatly reduced risk of cutaneous atrophy following topical application. At the time of writing the use of this product in clinical practice has been limited to clinical trials.
Tacrolimus: Tacrolimus is used topically in people with moderate to severe atopic eczema and its use has been reported in dogs to decrease erythema and pruritus in localized areas. It does not have the same atrophic effect on the skin as glucocorticoids. However, it is not licensed for veterinary use and necessary precautions should be taken if it is prescribed with written owners’ consent.
Control of flare factors
In general, practical recommendations should be made to reduce allergen load in the pet’s home environment and to reduce its exposure to them when out of doors (Table 6.2). Microbial infections must be treated beyond clinical cure and measures to try to prevent further episodes of infection should be recommended. In addition, either specific therapy to alter the immune response to allergens or symptomatic treatment will be required.
Table 6.2 Measures to reduce allergen load and exposure
| Indoor allergens such as house dust mites and danders | |
| Outdoor allergens such as pollens |
Regular topical therapy in the form of shampoo therapy is recommended as an adjunct to allergen-specific immunotherapy and/or to systemic treatment. The type of shampoo used will need to be tailored to the patient’s needs. For instance, antimicrobial shampoo may be recommended for a dog that is predisposed to developing secondary infections, while a shampoo containing colloidal oatmeal may be prescribed for a dog that suffers from dry scaling and pruritus.
Treatment in this case
In this case the owner was advised that there is no known treatment for the pattern alopecia and that the allergic dermatitis would require long-term management, which in most cases is lifelong. The following treatments and control measures were recommended:
Allergen-specific immunotherapy: The dog was started on an allergen-specific immunotherapy, based on the results of the intradermal test. All the allergens identified from the test were included in the immunotherapy. It is not a licensed treatment in the UK and therefore requires a special treatment certificate for its use.
In this case concurrent treatment was also prescribed:
In addition, ongoing, thorough flea control using monthly applications of a spot-on adulticide was prescribed as well as the environmental treatment to control dust mites. Weekly baths with 2% miconazole/2% chlorhexidine shampoo were advised to help control recurrent microbial infections.
Information on localized cleansing and/or total bathing should be given to owners. At this stage the frequency of the flea control should be adjusted, as the frequency of bathing will have an effect on the duration of the preparation’s action. Routine ear cleaning following bathing and during management of atopic dermatitis is advised, as many dogs have concurrent otic disease. The nurses should be able to show the owners how to flush ears at home without damaging the ear epithelium. Some dogs may require Elizabethan collars to prevent self-induced dermatitis.