17 Pemphigus foliaceus in a cat
INTRODUCTION
Pemphigus foliaceus is an uncommon, sterile pustular, autoimmune skin disease that is recognized in both dogs and cats. The pustules quickly rupture, resulting in the formation of extensive crusting of affected areas. The degree of crusting is one of the diagnostic features of this disease. The definitive diagnosis is confirmed by cytology and histopathological examination.
CASE HISTORY
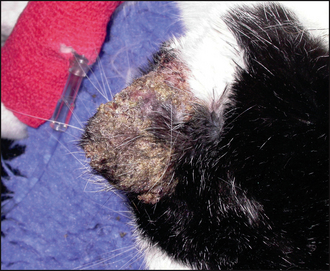
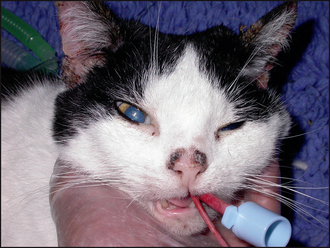
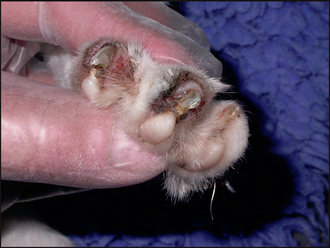
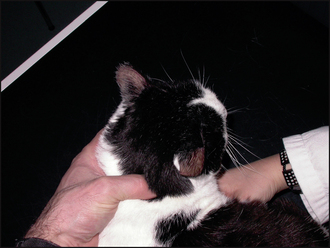
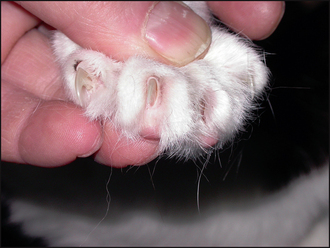
Typically, pemphigus foliaceus presents with a history of crusting skin lesions that most commonly start over the pinnae and face, with later involvement of the trunk and limbs. Paronychia is a common finding in cats. Pruritus may be absent to severe, and many cases show intermittent depression, pyrexia and anorexia. Quite frequently, when questioned, owners may have recognized a waxing and waning of the symptoms over time.
The relevant history in this case was:
CLINICAL EXAMINATION
Crusting is caused by leakage of pus, serum or blood onto the skin surface. In this case the colour of the crust was not consistent with haemorrhage but suggested a purulent discharge. One possible origin of such crusts is from a pustular skin disease.
The combined presence of focal crusting lesions affecting the pinnae and bridge of the nose, along with a purulent paronychia, was highly suggestive of pemphigus foliaceus.
Alternative, but less likely, crusting and erosive skin diseases included:
CASE WORK-UP
Cytology is an essential diagnostic tool where there is an exudative discharge. Fungal culture was also indicated to rule out any involvement of dermatophytosis.
The following diagnostic tests were performed:
DIAGNOSIS
The history, clinical signs, cytological and histopathological examinations, and the negative fungal culture, were consistent with a diagnosis of pemphigus foliaceus.
PROGNOSIS
The prognosis for feline and canine pemphigus foliaceus is very variable. Mildly affected cases with localized lesions may respond to topical medications. At the other end of the spectrum, there are very severe cases that may be refractory to therapy or that may develop life-threatening side-effects to treatment. In young animals, permanent resolution may follow successful medical therapy; in other cases, ongoing therapy is required to prevent recrudescence.
AETIOLOGICAL AND PATHOPHYSIOLOGICAL ASPECTS OF PEMPHIGUS FOLIACEUS
Pemphigus foliaceus is currently considered a heterogeneous autoimmune skin disease, resulting from the formation of predominantly IgG1 and IgG4 autoantibodies that target the intercellular spaces of the stratum spinosum and stratum granulosum of the epidermis. The precise target(s) (autoantigens) of these antibodies remain(s) unclear. There have been no studies reporting the nature of the specific autoantigen in cats, but a considerable amount of work has been done in looking for the autoantigen in canine pemphigus foliaceus. It was originally thought that the major autoantigen was desmoglein-1, a component of the desmosomes, which are responsible for keratinocyte–keratinocyte adhesion in the epidermis of the skin and hair follicles, but recent work has shown that this is an antigen of minor importance in canine pemphigus foliaceus, highlighting the heterogeneous nature of this disease.
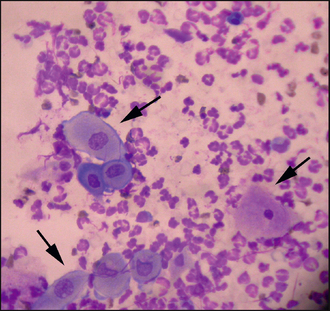
Pustule formation: The exact mechanism of pustule formation in association with antibody formation is complex and incompletely elucidated. However, antibody binding to desmosomal structures leads to the release of plasminogen by keratinocytes that results in activation of plasmin, a protease that destroys desmosomal structures, leading to acantholysis and the separation of keratinocytes in the stratum spinosum. The clefts thus formed are filled by neutrophils migrating into the epidermis from the circulation to form pustules. Acantholytic keratinocytes, cells that have become detached from the stratum spinosum as a result of the acantholytic process, are also present within the pustules. These cells are nucleated, as opposed to the fully differentiated cells of the stratum corneum that have lost their nuclei. They tend to have a rounded appearance and have basophilic staining characteristics. The cytological finding of acantholytic keratinocytes combined with a neutrophilic infiltrate is consistent with pemphigus foliaceus, but may also be found in cases of bacterial pyoderma and also dermatophytosis, particularly in cases of infection with Trichophyton spp.
Trigger factors
Drug induced: Most cases of pemphigus foliaceus in both cats and dogs appear spontaneously. However, a subset of cases may be drug induced and, in these cases, the disease should resolve when the drug is discontinued. Drug-related pemphigus foliaceus has been reported following administration of cefalexin and trimethoprim-potentiated sulphonamides in dogs, and ampicillin and methimazole in cats.
Chronic skin disease: Anecdotally, dermatologists report cases of canine pemphigus foliaceus that occur in association with a history of chronic skin disease. Typically, these cases have a history of chronic inflammatory, pruritic skin disease and then suddenly develop a more severe disease associated with pustules and crusting, diagnosed as pemphigus foliaceus. Whether there is a cause and effect association with the chronic skin disease is not clear. There is the possibility that some of these cases may be a cutaneous drug reaction associated with drugs used to treat the chronic skin disease.
Sunlight: There is some evidence that sunlight exposure may be a triggering factor for pemphigus foliaceus, particularly in lesions affecting the face. Some experimental work has found that UVB exposure of non-lesional skin from a dog with facial pemphigus foliaceus led to acantholysis.
EPIDEMIOLOGY
There is little epidemiological information on the incidence of either canine or feline pemphigus foliaceus; however, this is an uncommon condition, with a reported incidence in one referral hospital population of only 0.3% of patients referred for skin disease. It does, however, account for around one-third of canine autoimmune skin diseases, and in the authors’ experience is the most common autoimmune skin disease in cats.
No definitive breed predisposition has been reported in the cat. Some studies have shown a familial incidence of canine pemphigus foliaceus in the Japanese akita, Dobermann, Finnish spitz, schipperke, bearded collie, Newfoundland, dachshund, English cocker spaniel, chow-chow and Shar Pei. There appears to be no age or sex predisposition in either cats or dogs.
TREATMENT OPTIONS
Glucocorticoids
Glucocorticoids are the mainstay of treatment for pemphigus foliaceus. Mild focal lesions may be responsive to topical therapy, but more severely affected cases require immunosuppressive doses of corticosteroids to achieve remission.
General guidelines: It is beneficial to use the highest recommended dosage until clinical signs abate, because this usually results in the quickest clinical response and, ultimately, a lower total cumulative glucocorticoid dosage. Short-acting glucocorticoids such as prednisolone or methylprednisolone are the drugs of choice. Cats generally require higher dosages than dogs to achieve the same clinical effect; one reason for this is they have fewer glucocorticoid receptors. Treatment is administered until complete remission of clinical signs and then the dosage is very gradually tapered down over a period of months to maintenance levels. If there is a poor response to prednisolone or methylprednisolone, then triamcinolone, betamethasone or dexamethasone may be administered.
Immunosuppressive glucocorticoid dosages for induction and maintenance:
Adverse effects of glucocorticoids: Glucocorticoids, particularly at these dosages, will commonly result in adverse effects. In general, cats are more resistant to the side-effects of glucocorticoids than dogs. Short-term side-effects of glucocorticoids in dogs and cats can include polyuria, polydipsia, polyphagia, panting, mood alteration including aggression, and diarrhoea. Long-term side-effects include the signs of iatrogenic hyperadrenocorticism: weight gain, redistribution of body fat and muscle atrophy leading to a pot-bellied appearance, hepatomegaly, alopecia, comedone formation, calcinosis cutis (dogs only), pyoderma and demodicosis. Urinary tract infections, diabetes mellitus, pancreatitis and gastrointestinal ulceration are other potential complications. Skin fragility is a rare but serious side-effect in cats treated with glucocorticoids. Recently, congestive cardiac failure has been reported in cats, particularly following long-acting glucocorticoid injections. Sudden withdrawal of glucocorticoids, following prolonged administration, can lead to an Addisonian crisis.
Chlorambucil
The alkylating agent chlorambucil is commonly used, in addition to systemic glucocorticoids, at a dosage of 0.1–0.2 mg/kg q24 h/48 h for the management of pemphigus foliaceus in small dogs and cats. A 2-mg tablet is the smallest tablet size available in the UK, and to avoid the necessity of the owner having to break the tablets, a 2-mg tablet is generally administered on alternate days to treat cats weighing between 4 and 6 kg. Chlorambucil is not licensed for use in animals in the UK. Alkylating agents are slow acting and a case treated solely with chlorambucil could take up to 8 weeks to respond, which is why these drugs are generally administered with glucocorticoids. However, in the longer term, their use can reduce the cumulative dose of glucocorticoids required to achieve and maintain remission of the disease.
The adverse effects of chlorambucil include gradual bone marrow suppression, including neutropenia and thrombocytopenia, alopecia and gastrointestinal disturbance. Cerebellar toxicity has been reported when administered at very high doses.
Azathioprine
The purine synthesis inhibitor, azathioprine, is another treatment option that may be considered in addition to glucocorticoid therapy for the treatment of canine pemphigus foliaceus. However, there is a high risk of irreversible bone marrow suppression with the use of azathioprine in cats and the drug should not be used in this species.
Azathioprine is metabolized in the liver to 6-mercaptopurine, the active metabolite, which affects the formation of nucleic acids. The primary effect of the drug is inhibition of T cell lymphocyte function and T cell-dependent antibody synthesis with little effect on B cells. Azathioprine has a slow onset of action and frequently takes 4–6 weeks to be effective. In view of the slow onset, it is usually initially administered along with glucocorticoids. Ultimately, in long-term control of the disease, azathioprine may have a glucocorticoid-sparing effect or it may be possible to withdraw glucocorticoid therapy altogether. The dosage in dogs is usually given at 2 mg/kg s.i.d. (1.2–2.5 mg/kg). If there is no clinical response and no evidence of lymphopenia after 4–6 weeks, the dosage may be increased. Once remission is achieved, it may be possible to reduce to alternate-day therapy.
There are two main pathways for metabolism of 6-mercaptopurine. Firstly, thiopurine methyl transferase (TPMT) activity results in methylation and oxidation and, secondly, xanthine oxidase, present in the liver, metabolizes azathioprine to 6-thiouric acid, an inactive compound. Low levels of erythrocyte TPMT activity are associated with increased drug toxicity in man and low TPMT has also been demonstrated in some dogs.
The principal toxic effect of azathioprine is bone marrow suppression with thrombocytopenia and anaemia. In general, this develops gradually. Lymphopenia is a frequent finding and appears to correlate with a favourable therapeutic response. Other adverse effects include diarrhoea, which may be haemorrhagic; hepatotoxicity; and acute pancreatitis – possibly due to decreased pancreatic secretion.
Close patient monitoring is advisable when using azathioprine and should initially include haematology (including platelets) and biochemistry every 2 weeks. After the disease has been controlled and dosages are being tapered, monitoring may be reduced to every 2–3 months.
Ciclosporin and tacrolimus
Both ciclosporin and tacrolimus are immunosuppressive agents that have the potential to treat autoimmune disease. These drugs block regulatory proteins that up-regulate activation genes of T helper lymphocytes, particularly interleukin-2, a cytokine responsible for lymphocyte proliferation. However, up to the time of writing, there has been no published evidence as to the efficacy of ciclosporin in the treatment of pemphigus foliaceus in cats. A small-scale study using ciclosporin to treat canine pemphigus foliaceus showed a very poor response rate, although this may have been a reflection of inadequate dosage. Tacrolimus was reported to be helpful in the treatment of canine pemphigus erythematosus, but this is a localized disease, and tacrolimus has the potential for severe toxicity if administered systemically and so was not an option for the treatment of the generalized disease in this cat.
Chrysotherapy
Gold has immune-modulating effects, but its exact mode of action is unknown. Aurothioglucose was reported to be effective for the treatment of pemphigus in cats, but it is no longer available. The only preparation now available is sodium aurothiomalate, but there are no reports on its use for the treatment of feline pemphigus foliaceus. Toxic reactions to gold therapy in cats and dogs include bone marrow suppression, oral ulceration and glomerulonephropathy. One drawback to using gold injections is that they have to be given by painful deep intramuscular injection. Routine monitoring when using gold should include a complete blood count every 2–3 weeks, and full serum biochemical examination and urinalysis every 4–6 weeks for the first few months of therapy. These tests should be repeated every 3–6 months thereafter.
Treatment in this case
The extent and severity of the lesions in this case were an indication to use a treatment which was going to be both effective and produce a rapid therapeutic effect.
The decision was made to start treatment with a combination of prednisolone at a dosage of 4 mg/kg divided b.i.d. and chlorambucil (2 mg on alternate days). The owner understood that there was a significant risk of inducing diabetes mellitus with this treatment.
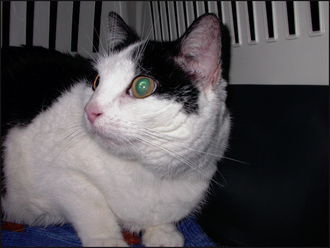
Within 3 weeks, there had been an improvement in the appearance of the lesions with resolution of pinnal crusting and paronychia, although the dorsal crusting lesions persisted, but after 6 weeks of treatment all crusting lesions had resolved (Figs 17.5, 17.6 and 17.7).
Glycosuria and hyperglycaemia were evident on repeat haematological and biochemical examinations after 2 weeks of glucocorticoid therapy, and so insulin therapy was initiated.
The treatment of an autoimmune skin disease is a balance between giving sufficiently high dosages of medication to achieve remission and yet not inducing serious adverse effects. On occasions, this can be a difficult balance to achieve. Drug dosages vary from patient to patient and depend on the response to treatment, the severity of adverse effects monitored clinically, and on the results of haematological and biochemical parameters. Once remission has been achieved, it is important to taper drug dosages as slowly as possible; rapid dose reductions can result in earlier and more severe recrudescence of disease. Drug dosages should be tapered to the level at which the disease is just observed to recur. This is the only way to establish the lowest drug dosages required for long-term control of disease.
In severe cases of pemphigus foliaceus in both cats and dogs, gentle bathing to remove the crust will speed resolution of the disease and be soothing to the animal. A whirlpool bath is the best way to accomplish this, but soaks in a warm bath or gentle use of a shower head will have a similar beneficial effect. Plain water should be used rather than shampoos or detergent-based products.
Nurses should be familiar with the side-effects of glucocorticoid and other immunosuppressive therapies.
FOLLOW-UP
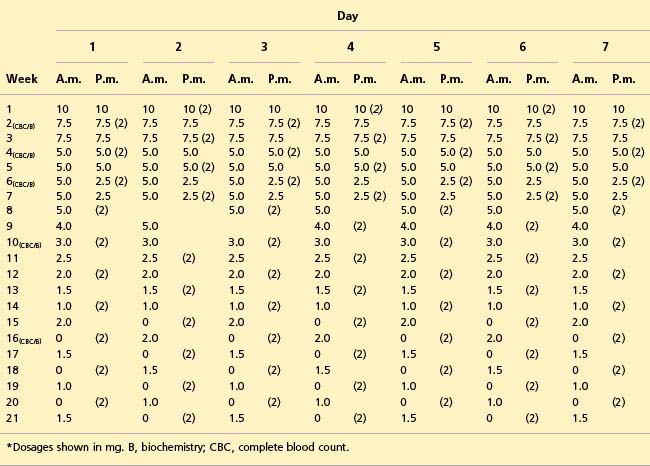
Over a 4-month period, the dosage of prednisolone was gradually reduced (Table 17.1). Routine haematology and biochemistry were initially performed fortnightly and urinalysis weekly, as shown in Table 17.1. Insulin therapy was required until the prednisolone dosage was reduced to 1 mg daily, at which point it could be discontinued without glycosuria or hyperglycaemia. Alternate-day chlorambucil therapy was maintained throughout the treatment course. The owner noted recurrence of erythema and scaling over the pinnae when the alternate-day prednisolone dosage was reduced to 1 mg on alternate days, which resolved when increased to 1.5 mg on alternate days. At the time of writing, the cat has been maintained on prednisolone (1.5 mg) alternating with chlorambucil (2 mg) for the past 8 months without recurrence of the skin lesions.
Table 17.1 Example of prednisolone and chlorambucil (values in parentheses) drug dosage reductions for treatment of pemphigus foliaceus*
The diagnosis of pemphigus foliaceus is based on a combination of clinical signs, and cytological and histopathological examinations.
Clinical signs in cats
In contrast to canine pemphigus foliaceus, feline lesions tend to be milder, and feline pustules are very fragile and quickly rupture and form crusts. A purulent, sometimes caseous, paronychia affecting the majority of the digits is a frequent finding in cats with pemphigus foliaceus.
Clinical signs in dogs
Intact pustules are more likely to be seen in canine pemphigus foliaceus; they tend to be large and may span multiple hair follicles.
Lesion distribution
Although the disease can be generalized in both cats and dogs, predilection sites include the medial aspects of the pinnae, the bridge of the nose, footpads and the nasal planum. The presence of pustules or focal crusted macules over the medial aspects of the pinnae is strongly suggestive of pemphigus foliaceus.
Sampling
The diagnosis of pemphigus foliaceus is based on cytology and histopathology. An intact pustule is the best lesion to sample both for cytology and for histopathology, but they are fragile transient lesions and rapidly rupture to form crusts. Furthermore, the disease waxes and wanes and pustules may be formed for only a few hours to days, followed by a long period of crusting. Thus, in many cases, there may be no pustules when the animal is presented and, in this situation, direct impressions for cytological evaluation may be made from the underside of crust. Similarly, crusted erosions may be sampled for histopathological examination. In these situations, the diagnosis of pemphigus foliaceus is made by examination of the crust and it is important to include the crust in the submission, particularly if it becomes detached from the underlying skin during the biopsy process.
Antibacterials
The diagnosis of pemphigus foliaceus should not be based solely on finding neutrophils and acantholytic keratinocytes on cytology, because the latter cells may be seen in cases of pyoderma and dermatophytosis. Thus, it is advisable to perform fungal cultures and start systemic antibacterial therapy while waiting on the results of the histopathological examination. Some of these cases of apparent pemphigus foliaceus respond completely to antibacterials.