Chapter 11 Non-invasive ventilation
INTRODUCTION
The application of non-invasive ventilatory support to improve ventilation is not a new idea. The tank ventilator or ‘iron lung’, which provides negative pressure to the chest wall, was first developed in the 19th century (Woollam 1976). Further developments and modifications occurred, but it was not until the poliomyelitis outbreaks of the 1940s and 1950s that such devices became widely used. Continuous positive airway pressure (CPAP) through a facemask for patients with pulmonary oedema and other forms of acute respiratory failure was extensively described in the 1930s (Barach et al 1938). However, with the development of positive pressure ventilators and the introduction of the endotracheal tube in the 1960s, use of non-invasive forms of ventilatory support for acute respiratory failure declined. Negative pressure devices continued to be used in patients with severe respiratory muscle impairment following poliomyelitis and in other patient groups presenting with chronic respiratory failure where long-term ventilatory support in the home was required (Garay et al 1981, Weirs et al 1977).
Since the mid-1980s, interest in non-invasive ventilatory support has again flourished, specifically the use of positive airway pressure devices and facemask interfaces. Although this interest had its genesis in the area of sleep-disordered breathing and chronic respiratory failure, clinicians have rapidly recognized the value of this therapy in acute medical and surgical conditions where respiratory failure develops, in weaning from conventional ventilatory support and as an adjunct to established respiratory care programmes. In this chapter we will outline the mechanisms by which abnormal sleep-breathing contributes to the development of awake respiratory failure and the place nocturnal ventilatory support plays in reversing this. We will also look at the potential application of this technique in a broadening range of clinical conditions.
BREATHING, SLEEP AND RESPIRATORY FAILURE
It has been recognized for many years that significant changes in breathing and ventilation can occur during sleep (Gastaut et al 1966). The contribution abnormal breathing, during sleep, can play in the development of awake hypercapnia is now more fully appreciated. Our understanding of what happens to breathing during sleep has been greatly enhanced by three major developments in technology:
 The routine use of accurate oximeters (Trask & Cree 1962), which have allowed the continuous monitoring of arterial oxygenation over prolonged periods of time.
The routine use of accurate oximeters (Trask & Cree 1962), which have allowed the continuous monitoring of arterial oxygenation over prolonged periods of time. The development of a comfortable and acceptable nasal mask interface (Sullivan et al 1981) has provided a simple yet effective means by which abnormalities of breathing can be reversed.
The development of a comfortable and acceptable nasal mask interface (Sullivan et al 1981) has provided a simple yet effective means by which abnormalities of breathing can be reversed.These developments made it possible to continuously monitor changes in breathing associated with sleep state and to provide patients with a treatment intervention that is both effective and acceptable on a long-term basis.
Changes in breathing during sleep
Sleep is associated with a number of normal physiological events that have little effect on individuals with normal respiratory drive and mechanics. However, in patients with a range of respiratory abnormalities, sleep can lead to worsening respiratory function and gas exchange.
The awake state itself is associated with an additional stimulus to breathe, over and above that determined by the metabolic control system. This is known as the wakefulness drive to breathe and is lost with the onset of sleep. General postural muscle tone is also reduced at sleep onset, resulting in increases in upper airway resistance and reductions in ventilatory drive. At the same time, ventilatory responses to both hypoxia and hypercapnia are reduced so that there is an attenuated response to changes in gas exchange compared with wakefulness. As a result, a small fall in ventilation occurs with sleep in the range of 10–15% (Douglas et al 1982).
Although reduced, ventilation during non-rapid eye movement (NREM) sleep is steady, particularly during periods of slow-wave sleep. However, even in normal subjects there is substantial variation in breathing during rapid eye movement (REM) sleep, most pronounced during periods of phasic eye movements. During these episodes, alveolar ventilation may fall by as much as 40% (Douglas et al 1982). REM sleep is also associated with alterations in respiratory control, caused by descending inhibition of alpha and gamma motor neurons. This produces hypotonia of postural muscles, including the intercostal and accessory respiratory muscles and a reduction in the rib cage contribution to ventilation. As a result, ventilation during REM sleep becomes heavily reliant on diaphragmatic activity.
In patients with severely compromised lung function or significant inspiratory muscle weakness, recruitment of other inspiratory and accessory muscles, including the abdominals, may occur to augment breathing. By this compensatory mechanism, individuals are usually able to maintain adequate ventilation during wakefulness and NREM sleep for prolonged periods. In those with significant lung disease, recruitment of the intercostal muscles occurs not only to augment ventilation but to maintain end-expiratory lung volume, thereby preventing small airway closure. With the transition into REM sleep, this postural muscle activity will be lost, resulting in a reduction in minute ventilation, worsening ventilation–perfusion relationships and a deterioration in gas exchange. Falls in saturation will be more severe in those patients with awake saturation values already near the steep portion of the oxyhaemoglobin dissociation curve. The degree of abnormal breathing which then occurs will depend upon the patient’s arousal response. Arousal causes a change in state from sleep to transient wakefulness, permitting the re-emergence of accessory muscle activity and restoration of ventilation, albeit briefly. In this way, arousal acts as a defensive mechanism, limiting the degree of gas exchange abnormality that is permitted to occur. However, this response also leads to sleep fragmentation, which in itself can alter respiratory drive and arousal thresholds, so that eventually more extreme blood gas derangement must occur before the arousal response is activated.
The role of sleep in the development of awake hypercapnic respiratory failure
It is now well recognized that disturbed breathing first becomes apparent in REM sleep (Bye et al 1990). However, as REM sleep takes up only a relatively small proportion of total sleep time, patients with REM hypoventilation, even if severe, may remain clinically stable for months or even years before significant daytime hypercapnia becomes apparent. Initially, ventilation and sleep between periods of REM hypoventilation are usually normal, often through the recruitment of accessory respiratory muscles. In addition, the arousal mechanism operates to defend ventilation by limiting the amount of time spent in REM sleep and therefore the degree of abnormal gas exchange that occurs. Characteristically, awake blood gases remain normal during this initial stage.
Progression of abnormal breathing into NREM sleep heralds the second stage in the evolution of sleep-induced respiratory failure (Piper & Sullivan 1994a). Mechanisms responsible for this progression include not only a deterioration of the underlying disease itself but also the appearance of other factors that may load breathing such as ageing, weight gain, upper airway dysfunction or the development of an intercurrent illness such as a chest infection. Sleep fragmentation from abnormal breathing events has the capacity to further alter respiratory control and depress arousal. These factors allow more severe sleep-disordered breathing to occur, with less arousal between events. This begins a vicious cycle whereby resetting the sensitivity of the ventilatory control system occurs so that higher levels of carbon dioxide and lower levels of oxygen are tolerated without stimulating a change in respiration, not only asleep but during wakefulness as well. During this stage, daytime CO2 retention becomes apparent (Fig. 11.1).
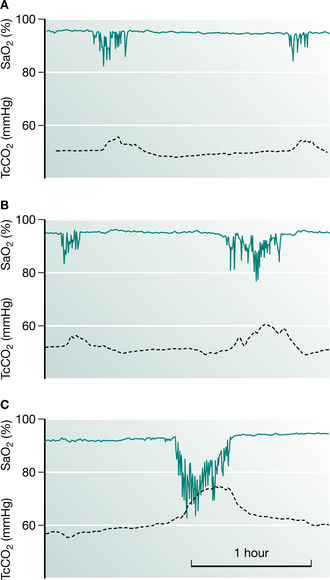
Figure 11.1 Serial recordings of oxygen saturation (SaO2) (solid green line) and transcutaneous carbon dioxide (TcCO2) (dotted line) from a patient with Duchenne muscular dystrophy showing progressive nocturnal respiratory failure. (A) Mild sleep-disordered breathing, with modest falls in SaO2. (B) Eight months later more substantial oxygen desaturation was apparent during REM sleep, with rises in carbon dioxide. (C) Severe REM desaturation occurs, with failure of SaO2 to return to baseline values between periods of abnormal breathing. This was accompanied by large rises in CO2. Over the same period, awake CO2 had risen from 40 to 45 mmHg (5.3 to 6.0 kPa), with no change in inspiratory muscle pressures.
The final stage in the development of sleep-induced hypercapnia is characterized by unstable respiratory failure both awake and asleep. During this stage, changes in blood gases during sleep are extreme and sleep architecture may be profoundly disturbed. By this stage, the clinical condition of the patient may deteriorate considerably, which can be mistaken for a progression of the underlying disease process. It is crucial that clinicians recognize patients with nocturnal hypoventilation early and treat appropriately, in order to prevent the complications associated with an acute respiratory crisis (Box 11.1).
NON-INVASIVE VENTILATION
Rationale and methods
Non-invasive ventilation (NIV) is a technique whereby positive pressure is applied to the airways and lungs without the need for an endotracheal or tracheostomy tube. Whether used for the management of acute or chronic conditions, during awake or sleep states, the aim of this therapy is the same: to improve gas exchange by assisting inspiratory efforts and reducing the work of breathing.
In patients presenting with acute respiratory failure there is a deterioration in gas exchange accompanied by changes in pulmonary mechanics with increased respiratory loads. As a consequence a shallow, rapid breathing pattern develops with shortening of inspiratory time and reductions in tidal volume. Non-invasive ventilatory support in this setting aims to augment the patient’s tidal volume while reducing the amount of effort or work performed. In addition, maintaining a low level of positive pressure during expiration can subsequently reduce inspiratory effort by counterbalancing intrinsic PEEP (the end-expiratory recoil pressure of the respiratory system due to incomplete expiration). The improved breathing pattern with NIV should improve alveolar ventilation while preventing respiratory muscle fatigue. Ventilators that provide a range of settings and features can be desirable in the acute situation in order to ensure the machine has the capability of addressing the individual’s breathing needs.
The mechanism by which NIV improves awake spontaneous breathing in patients with chronic respiratory failure is a little less clear. Three possible hypotheses have been put forward (Hill 1993). The first relates to chronic muscle fatigue and proposes that the use of NIV permits intermittent rest of fatigued muscles, restoring function. The second hypothesis proposes that NIV increases lung volumes and compliance. Finally, the respiratory centre sensitivity to CO2 may be blunted during the development of chronic respiratory failure. Non-invasive ventilation, by preventing hypoventilation during sleep, may work by restoring ventilatory sensitivity to carbon dioxide and hence improve awake breathing. In a study designed to determine the relative importance of each of these mechanisms, Nickol et al (2005) found there was no change in measures of inspiratory muscle strength, lung function or respiratory system compliance with the commencement of NIV in patients with restrictive thoracic disease despite significant improvements in awake blood gases. However, they did find an increase in ventilatory sensitivity to CO2 supporting this as a principle mechanism for the improvements in breathing seen in patients with chronic respiratory failure who had been started on NIV.
Ventilator systems
Primarily two types of ventilator systems are available for mask ventilation: volume-preset and pressure-preset devices. Each type of support has its own advantages and limitations. A successful outcome using mask ventilation will depend upon the clinician’s under-standing of the underlying pathological processes that have contributed to the patient’s respiratory deterioration, and choosing a machine and mode of ventilatory support which best meet the respiratory needs of the patient.
Volume-preset machines such as the PLV®-102 (Respironics, Inc, Murrysville, PA, USA), the PV501 (BreasTM, Molnycke, Sweden) or the BromptonPAC (PneuPAC Ltd, Luton, Beds, UK) operate as time-cycled flow generators and deliver a fixed tidal volume irrespective of the airway pressure generated, provided that leaks from the system are minimized. Pressure-preset systems include bilevel positive pressure devices, the most widely recognized being the BiPAP® machines (Respironics, Inc, Murrysville, PA, USA). Other pressure-preset devices include the Vivo30® (BreasTM, Molnycke, Sweden), VS IntegraTM (ResMed, Bella Vista, NSW, Australia), and the VPAPTM III device (ResMed, Bella Vista, NSW, Australia) (Fig 11.2A). With these devices, tidal volume will vary according to the preset inspiratory pressure, the inspiratory–expiratory pressure difference and the chest wall and lung compliance of the patient. Bilevel devices are reported to compensate better for mild to moderate leaks than volume-preset devices (Mehta et al 2001). However, leaks are common during mask ventilation, particularly during sleep, and this may adversely affect the quality of ventilation and sleep architecture even with bilevel positive pressure devices (Piper & Willson 1996, Teschler et al 1999).
Figure 11.2 Examples of two types of ventilatory support systems: (A) VPAPTM III ST-A (ResMed), a bilevel positive pressure device; (B) PLV® ContinuumTM ventilator able to deliver both volume and pressure ventilation.
(Image A used with the permission of ResMed Limited, North Ryde, Australia; image B used with permission of Respironics, Inc, Murrysville, PA, USA)
In studies comparing the efficacy of these two systems, little difference has been found either in acute (Girault et al 1997) or chronic respiratory failure (Tuggey & Elliott 2005), although many patients find pressure preset devices easier to tolerate. Poor tolerance to volume-preset devices may be related to an increase in airway resistance causing an elevation of inspiratory pressure in the mask, which may be uncomfortable or may cause leaks, thus limiting the effectiveness of ventilation. On the other hand, in patients with low chest wall compliance, higher airway pressures may be needed to maintain optimal ventilation. In these patients, volume-preset ventilators can prove more reliable and effective in delivering a stable tidal volume despite changing chest wall mechanics or airway resistance. A change to a volume-preset device should always be considered if hypoventilation persists on bilevel ventilatory support (Schonhofer et al 1997).
Criteria for choosing mode of ventilatory support
A number of criteria need to be considered when choosing a machine and mode of ventilatory support. These include:
Sleep study data are useful in patients requiring long-term ventilation in identifying any degree of upper airway dysfunction that may be present as well as determining the patient’s respiratory drive during sleep. Understanding the features and limitations of the various machines available and the modes of ventilatory support in which they can operate will assist in selecting the appropriate system to meet the patient’s needs. A recent development is the availability of portable ventilators that are able to deliver both pressure and volume ventilation, allowing flexibility with regard to changing the mode of ventilatory sup-port with changes in the patient’s condition (e.g. PLV® ContinuumTM (Fig. 11.2B), Respironics, Inc, Murrysville, PA, USA; PV403, BreasTM, Molnycke, Sweden; VS UltraTM, ResMed, Bella Vista, NSW, Australia).
Settings
The mode of support needs to be set so that the breaths delivered will be either machine-triggered or patient-triggered. With bilevel devices, a spontaneous mode of support is available, where the machine cycles into inspiration in response to the patient’s spontaneous inspiratory effort. The volume-preset and a number of the bilevel devices can also be set to deliver a preset respiratory rate should the patient fail to trigger the device. Titration of the inspiratory positive airway pressure (IPAP) for a patient on a bilevel device or the tidal volume for a patient using a volume-preset device is made on the basis of patient tolerance and the effect such a setting has on ventilation and gas exchange. However, when setting pressures or volumes it should be borne in mind that excessively high inspiratory pressures will promote leakage of air from the mouth, reducing the effectiveness of ventilatory support. Excessive hyperventilation can also occur, which may induce upper airway obstruction and the appearance of central apnoea. The use of expiratory positive airway pressure (EPAP) may be advantageous in a number of clinical conditions, including controlling upper airway closure, recruiting collapsed alveoli or to overcome intrinsic end-expiratory pressure. Where inspiratory time or flow can be set, this will be based on the patient’s own respiratory pattern, taking into account the effect short inspiratory times can have on gas exchange.
In patients where the compliance of the alveoli is heterogeneous, delivered tidal volume may be directed towards those alveoli with short filling times, producing overdistension of already inflated units and therefore not contributing to improved gas exchange. Prolonging the inspiratory time allows the recruitment of alveoli with slower filling times, so that increased ventilation can contribute to improved gas exchange.
Timing of intervention
There is extensive evidence to support the use of NIV as first-line therapy in patients with acute exacerbations of COPD (Lightowler et al 2003). However, the technique is not universally successful, with a proportion of patients continuing to deteriorate despite appropriate support. The likelihood of failure increases as acidosis worsens (Ambrosino et al 1995, Plant et al 2001). Instituting NIV in mild exacerbations has not been shown to be beneficial and may be poorly tolerated (Keenan et al 2003). Therefore, identifying and intervening during this window of therapeutic opportunity is important to achieve the best clinical outcomes with NIV. Generally speaking, patients with a pH in the range of 7.25–7.35 are the best candidates for the procedure.
Determining when to start NIV in patients with chronic respiratory failure is less clear cut. Most would agree that the presence of daytime hypercapnia (PaCO2 >6 kPa (45 mmHg) or daytime symptoms of nocturnal hypoventilation such as daytime sleepiness, early morning headaches, severe orthopnoea or alterations in cognitive function would be clear indications for assessment and appropriate intervention. However, for many patients the onset of nocturnal respiratory failure occurs over an extended period of time, in some cases even years. With such an insidious onset, the signs and symptoms of chronic hypoventilation may be overlooked or incorrectly attributed to the ongoing progression of the primary disease process.
One study looked at the ‘preventative’ use of NIV in patients with Duchenne muscular dystrophy (DMD), free of daytime respiratory failure (Raphael et al 1994). No benefit was found from early intervention with this technique, with the treated group showing a similar rate of deterioration in blood gases and pulmonary function as the control group. However, nocturnal monitoring was not performed either at baseline or during therapy in these patients. A more recent study looking at patients with neuromuscular disease, including patients with DMD, found that those with nocturnal hypoventilation despite daytime normocapnia were likely to deteriorate and develop daytime respiratory failure and/or progressive symptoms within 2 years if NIV was not introduced (Ward et al 2005). This suggests that the timing of NIV intervention is important, with identification of those patients at risk of developing nocturnal hypoventilation, monitoring of nocturnal gas exchange in these individuals and intervention with NIV before the development of awake respiratory failure ensues. In this way, the chances of the patient presenting with an acute respiratory crisis may be minimized.
INDICATIONS FOR NON-INVASIVE VENTILATION IN CHRONIC RESPIRATORY FAILURE
Assessment of chronic hypoventilation
Although a number of investigators have tried to use daytime pulmonary function tests as a predictor of the degree of abnormal breathing occurring during sleep, no strong correlation has been found (Bye et al 1990). However, it has been shown that a low vital capacity, a significant fall in vital capacity from erect to supine or a maximum inspiratory pressure of less than 30 cmH2O are all indicators that sleep-disordered breathing and hypoventilation may be present (Bye et al 1990, Ragette et al 2002). Each of these tests can be easily carried out at the bedside as part of the overall assessment of a patient presenting in respiratory failure. Strong use of the accessory respiratory muscles at rest, including the sternomastoid and the abdominal muscles, should raise the possibility that respiratory function may worsen during sleep.
In general, if there is awake hypercapnia then there will be substantial sleep-linked worsening of respiratory failure (Piper & Sullivan 1994a), although the converse does not necessarily hold true. Many subjects with awake CO2 within the normal range will have significant sleep-linked respiratory failure. The failure of clinicians to recognize this and intervene appropriately puts a patient at risk of developing daytime respiratory failure within the following 12–24 months (Ward et al 2005).
The limitations of daytime indices as predictors of nocturnal hypoventilation mean that more detailed sleep investigations may be required in order to accurately assess the severity and nature of the breathing disorder. While hypoventilation and desaturation are likely to occur predominantly in REM sleep, upper airway obstruction may also be present and needs to be identified and appropriately managed in order for NIV to be maximally effective. In patients with suspected nocturnal hypoventilation, monitoring of carbon dioxide is important looking at evening to morning changes in arterial values or continuous monitoring with transcutaneous measures (Ward et al 2005). Regardless of the investigations used, a high index of suspicion in patients with diagnoses known to be associated with nocturnal hypoventilation is warranted. In these individuals, baseline and serial testing of respiratory function should be performed with the view to identifying any deterioration early so that more specific investigation can be undertaken.
Conditions
Kyphoscoliosis
The final stages of severe kyphoscoliosis have been characterized by progressive respiratory failure associated with severe nocturnal hypoventilation (Ellis et al 1988). REM hypoventilation is probably caused by a combination of a very high work of breathing due to low chest wall compliance for a diaphragm that is at a significant mechanical disadvantage. In some patients sleep-disordered breathing is also complicated by upper airway obstruction. Mask ventilation is particularly suitable for these patients as other methods of assisted ventilation are very difficult. Tracheostomy can be problematic because of the loss of the extrathoracic trachea and the fitting of a cuirass is made exceptionally difficult by the chest wall deformity. Non-invasive ventilation can be readily achieved with a mask in this group despite the stiffness of the chest wall (Ellis et al 1988).
Cystic fibrosis
Although low-flow oxygen therapy has been the mainstay of treatment for patients with cystic fibrosis (CF) who develop respiratory failure, several reports have shown that, at least acutely during sleep, oxygen therapy can promote CO2 retention (Gozal 1997, Milross et al 2001). The beneficial effects of nocturnal non-invasive ventilation for patients with end-stage CF are recognized. Non-invasive ventilation has been shown to be of value during periods of acute deterioration, where marked pulmonary deterioration occurs despite maximum conventional therapy (Piper et al 1992). Use of mask ventilatory support in this setting can correct hypoxaemia without inducing additional CO2 retention. In addition, this technique may also be used to stabilize the patient in the short term while donor organs become available (Hodson et al 1991) or on a longer-term basis, allowing the patient to return home (Piper et al 1992). Although in initial reports volume preset machines were used, bilevel pressure devices are now being increasingly used with similar outcomes (Gozal 1997, Milross et al 2001).
Some patients report improved sputum clearance after initiation of nasal ventilatory support, possibly related to better tolerance of longer chest physiotherapy sessions (Piper et al 1992). Improved lung expansion and chest wall excursion while on the machine may also play a part. Studies have shown that use of nasal ventilatory support during chest physiotherapy was able to ameliorate adverse effects such as reduced respiratory muscle performance and oxygen desaturation (Fauroux et al 1999, Holland et al 2003).
Duchenne muscular dystrophy
Ventilatory support is often reluctantly prescribed for patients with progressive neuromuscular disease, owing to a perceived lack of quality of life for these patients. However, health professionals often underestimate quality of life in such patients. The use of long-term non-invasive ventilation has been shown to stabilize pulmonary function and prolong life expectancy while improving quality of life in patients with Duchenne muscular dystrophy (DMD) and awake hypercapnia (Simonds et al 1998). Non-invasive ventilation has also been initiated early in this disorder in an attempt to prevent the decline in lung function that occurs with increasing respiratory muscle weakness. However, no benefits from such a strategy could be identified (Raphael et al 1994). Therefore, NIV is an effective long-term therapy in this group of patients once nocturnal hypoventilation occurs, but may not have a prophylactic role.
Chronic obstructive pulmonary disease
Nocturnal nasal ventilation has been used effectively in selected patients with stable chronic obstructive pulmonary disease (COPD). However, this form of therapy is not tolerated as well as in other diagnostic groups and longer-term outcomes are not as favourable as in patients with neuromuscular and chest wall disorders (Simonds & Elliott 1995). Those patients most likely to benefit from nocturnal ventilatory support appear to be those with significant daytime hypercapnia, who have symptomatic sleep problems and in whom nocturnal hypercapnia can be successfully reduced by overnight ventilation. Meecham Jones et al (1995) reported a randomized crossover study of nasal pressure support ventilation plus oxygen therapy compared with domiciliary oxygen therapy alone in 18 hypercapnic patients with COPD. Improvements in daytime arterial blood gas tensions, overnight transcutaneous carbon dioxide (TcCO2), total sleep time and sleep efficiency were seen during non-invasive ventilation and oxygen therapy compared with oxygen therapy alone, suggesting that control of hypoventilation with non-invasive ventilation can be achieved. Importantly, these authors found that those who showed the greatest reduction in nocturnal hypercapnia with ventilation were likely to gain the greatest benefit from the treatment. However, a number of other randomized trials in this population have failed to find a clinically significant benefit of NIV (Casanova et al 2000, Clini et al 2002). These discrepant results may be due to patient selection or the way in which NIV was delivered and monitored. It would seem that patients with high daytime CO2, with a higher likelihood of nocturnal hypoventilation, are more likely to respond to this therapy than patients with near normal awake CO2 levels. In addition, data from Meecham Jones et al (1995) as well as uncontrolled trials (Windisch et al 2005) highlight the importance of using sufficiently high inspiratory pressures to control CO2 during sleep in order to achieve improvements in daytime ventilation and symptoms.
Motor neuron disease
Respiratory insufficiency usually occurs as a late manifestation of this disorder, when global peripheral and respiratory muscle weakness has occurred. However, in a small number of patients, presentation with hypercapnia, severe orthopnoea and sleep fragmentation may be seen. Although nasal ventilatory support has been shown to be effective in relieving these symptoms in this group, its use also raises some ethical and clinical concerns that need to be discussed with the patient and their caregiver (Polkey et al 1999). There has been reluctance to initiate such therapy for a condition that is known to be relatively rapidly progressive and where many will experience involvement of the bulbar muscles and swallowing difficulties (Meyer & Hill 1994). However, a recent randomized controlled trial has shown that NIV improves survival while maintaining and even improving quality of life in these patients (Bourke et al 2006). In those with severe bulbar impairment there was no survival benefit with NIV, but quality of life related to symptoms did improve.
Non-invasive ventilation appears to have a place as a management alternative in motivated patients with appropriate home supports, where established respiratory failure is present or where quality of life is impaired by sleep disruption or severe orthopnoea (Polkey et al 1999). However, before undertaking such therapy in this group, frank discussion with the patient and caregiver needs to occur. Potential benefits of NIV in palliating symptoms should be discussed as well as its limitations in the face of progressively worsening respiratory and general muscle strength and disability.
Obesity hypoventilation syndrome
Another group that responds rapidly and positively to non-invasive ventilation are those patients with obesity hypoventilation syndrome (Perez de Llano et al 2005, Piper & Sullivan 1994b). This syndrome is characterized by extreme obesity, excessive daytime sleepiness and severe derangement of awake blood gases. Patients frequently present grossly decompensated with right heart failure, lower limb oedema and hypercapnia. Use of non-invasive ventilatory support in these patients may result in improved awake blood gases and clinical condition within days of starting therapy, improving quality of life and daily function. The aim of therapy in these patients is to maintain upper airway stability while improving alveolar ventilation. Bilevel ventilatory support is usually required initially, particularly in those with severely deranged nocturnal and awake blood gases. However, after a short period of nocturnal ventilatory support, a proportion of patients can be transferred to the more simple CPAP therapy for longer-term domiciliary use (Perez de Llano et al 2005, Piper & Sullivan 1994b).
INDICATIONS FOR NON-INVASIVE VENTILATION IN ACUTE RESPIRATORY FAILURE
In order to reduce the problems associated with endotracheal intubation and ventilation, an increasing number of centres are now using non-invasive ventila tion as a treatment alternative for patients with acute respiratory failure. It avoids the complications of endotracheal intubation, is more comfortable for the patient, allowing speech and swallowing, and avoids the need for sedation and immobilization. Treatment does not have to be instituted in the intensive care or emergency department environment and is increasingly started on general medical or surgical wards (Bott et al 1993, Lightowler et al 2003, Piper & Willson 1996, Plant et al 2000).
Assessment of acute respiratory failure
Appropriate patient selection is essential for a success-ful treatment outcome. Non-invasive ventilation should be seen as a therapy to prevent the need for intubation rather than an alternative to it. Therefore, when undertaking this therapy it is important to be able to identify those patients who are unlikely to respond well, in order that a delay in mandatory intubation does not occur (Box 11.2). The ideal patient should be cooperative enough to tolerate a mask and to follow simple instructions. A successful outcome depends to a large degree on the ability to rapidly correct acidosis, decrease CO2 and reduce respiratory rate (Lightowler et al 2003). This in turn will be influenced by the ability of the patient and the therapist to minimize mouth/mask leaks and to coordinate breathing with the ventilator. A common reason for failure of this therapy is the inability of the patient to effectively remove secretions. Identification of those patients with secretion retention and prompt intervention by the physiotherapist to aid in clearing the airways can be pivotal in increasing the likelihood of therapy being successful. However, if hypercapnia and acidosis fail to improve within the first few hours of treatment, longer-term success is unlikely (Ambrosino et al 1995, Anton et al 2000, Lightowler et al 2003).
Box 11.2 Characteristics of patients with acute respiratory failure unlikely to do well on non-invasive ventilation
(Ambrosino et al 1995, Brochard et al 1995, Kramer et al 1995)
In patients who are hypoxaemic but retain carbon dioxide, the use of non-invasive ventilation permits higher levels of inspired oxygen to be introduced without unduly worsening hypercapnia. Under these circumstances, the use of non-invasive ventilation supports patients until their acute deterioration can be reversed (Conway et al 1993).
Conditions
Chronic obstructive pulmonary disease
The majority of studies reported to date have involved patients with chronic obstructive pulmonary disease (COPD) during an acute exacerbation. Evidence from a number of randomized trials have shown that NIV is clearly beneficial, with significant reductions in mortality, endotracheal intubation, complication rates and shorter length of hospital stay compared with standard medical therapy (Lightowler et al 2003). It appears that the type of ventilator (volume preset or bilevel pressure support) or the type of interface chosen (nose or full face mask) is not pivotal in determining the success of treatment. However, results will be influenced by the patient’s tolerance and adaptation to the machine and some patients may find the bilevel pressure support devices easier to adapt to. Very dyspnoeic patients tend to be mouth breathers and where it is not possible for the patient to maintain lip closure, a full facemask needs to be used to ensure machine–patient synchronization and delivery of an effective tidal volume. The technique has been used with equal success both in high dependency (Brochard et al 1995) and general ward situations (Bott et al 1993, Plant et al 2000). However, when considering location of care, the degree of acidosis, the therapeutic goals of NIV for the individual and the experience and level of staffing must be taken into account. While NIV may be a highly effective technique in patients with COPD, failure of therapy still occurs and arrangements for escalation of management or institution of a more palliative treatment approach need to made at the time NIV is first initiated (British Thoracic Society 2002).
Pulmonary oedema
Cardiogenic pulmonary oedema has also been shown to respond well to mask positive pressure therapy, either in the form of continuous positive airway pressure (CPAP) or bilevel ventilatory support (Masip et al 2005, Nava et al 2003, Peter et al 2006). Both therapies have been shown to reduce the need for intubation and reduce mortality compared to standard medical care, including oxygen supplementation, although the evidence for CPAP is stronger (Masip et al 2005, Peter et al 2006). A recent meta-analysis suggested a trend towards a higher occurrence of new cases of myocardial infarction with NIV compared with CPAP (Peter et al 2006). Therefore, current guidelines suggest CPAP should be used as first-line therapy in patients presenting with acute pulmonary oedema (British Thoracic Society 2002). Bilevel ventilatory support should be reserved for those in whom CPAP is unsuccessful (British Thoracic Society 2002, Peter et al 2006), or for the subgroup of patients who are hypercapnic (Nava et al 2003).
Hypoxaemic respiratory failure
Although the majority of initial work with NIV has been carried out in patients with hypercapnic respiratory failure, a number of recent randomized controlled trials have provided evidence that NIV can provide similar benefits in selected patients presenting with hypoxaemic respiratory failure (Antonelli et al 2000, Ferrer et al 2003), including those with community-acquired pneumonia (Confalonieri et al 1999). However, given the variability in pathophysiology leading to hypoxaemia, it is not surprising that the benefits of using NIV in patients with hypoxaemic respiratory failure have been mixed. Despite some promising preliminary results, the role of NIV in acute asthma remains controversial (Ram et al 2005) and requires further investigation. The current evidence suggests that NIV is most likely to be effective in patients with an underlying diagnosis of COPD (Confalonieri et al 1999) or in those who are immunosuppressed (Hilbert et al 2001). In patients developing acute respiratory failure following solid organ transplantation (Antonelli et al 2000) or after lung resection (Auriant et al 2001), use of NIV has been found to reduce the need for intubation and improve survival. However, as therapy failure is higher in patients with hypoxaemic respiratory failure treated with NIV compared to patients with hypercapnia (Delclaux et al 2000), treatment is best carried out and monitored in a high-dependency area rather than on a general ward.
Obesity hypoventilation syndrome
Although some patients with obesity hypoventilation syndrome (OHS) will present with stable chronic respiratory failure, the diagnosis is frequently overlooked until the patient presents with acute respiratory failure. Despite significant reductions in chest wall compliance and, in most cases, increased upper airway loading secondary to the presence of upper airway obstruction, these patients can be effectively managed with NIV in the acute setting, avoiding the need for intubation and its subsequent complications (Perez de Llano et al 2005). Even patients with simple obesity are at risk of developing postoperative respiratory failure following major surgery. Many of these patients probably have pre-existing sleep-disordered breathing. The affects of anaesthesia and analgesia may worsen an already compromised upper airway, producing apnoea and its sequelae such as hypoxaemia and blood pressure fluctuations. In addition, diaphragm inhibition after upper abdominal surgery can exacerbate REM hypoventilation. These patients generally respond well and rapidly to positive pressure, improving gas exchange and pulmonary function.
‘Do-not-intubate’ patients
While NIV is generally applied with the aim of avoiding the need for intubation rather than a substitution for it, for patients in whom intubation and ICU is considered inappropriate or futile, NIV may be used as a ceiling therapy (British Thoracic Society 2002). However, when applied in this way, higher failure rates have to be expected, with data suggesting a hospital mortality of 37% in COPD patients, 39% in acute cardiogenic pulmonary oedema, 68% in non-COPD hypercapnic respiratory failure and 86% in hypoxaemic failure (Schettino et al 2005). If applying NIV in this situation, it is important to bear in mind that the therapy and monitoring take time to set up and treatment is not without some possible discomfort to the patient. These factors need to be considered carefully if thinking about using NIV in patients with a likely terminal condition.
PRACTICAL ISSUES IN THE APPLICATION OF NON-INVASIVE VENTILATION
Interfaces
Both nasal and full facemasks may be used to deliver ventilatory support (Figs 11.3A & B, 11.4) and advances in design of both types have meant a greater degree of comfort and choice for the patient. Evidence from the literature suggests that successful outcomes in acute respiratory failure can be achieved with either type of interface (Kwok et al 2003), although the full facemask is often preferred in the acute setting to better control mouth leaks and ensure the effective delivery of ventilatory support (Kwok et al 2003, Navalesi et al 2000). In a study looking at various interfaces in patients with chronic hypercapnic respiratory failure, the nasal mask was the best tolerated although the oronasal and nasal plug interfaces were associated with significantly lower PaCO2 levels (Navalesi et al 2000). Chinstraps may be useful in those cases where the patient prefers a nasal mask but where mouth leaks remain problematic (Fig. 11.5).

Figure 11.3 Interfaces used for NIV. (A) HC407 nasal mask (Fisher and Paykel Healthcare Ltd, Auckland, New Zealand); (B) Ultra MirageTM II nasal mask (ResMed, North Ryde, Australia); (C) SwiftTM nasal pillows (ResMed, North Ryde, Australia); (D) ComfortLiteTM nasal pillows.
(Respironics, Inc, Murrysville, PA, USA). (Use of the Ultra MirageTM II and SwiftTM nasal pillow images made with the permission of ResMed Limited. Use of the ComfortLiteTM image made with the permission of Respironics, Inc, Murrysville, PA, USA)
Figure 11.4 Examples of facemasks. (A) Ultra MirageTM II full facemask (ResMed, North Ryde, Australia); (B) Total face- maskTM.
(Image used with the permission of Respironics, Inc, Murrysville, PA, USA.)
Figure 11.5 Nasal mask (ProfileLiteTM, Respironics, Inc, Murrysville, PA, USA) with chinstrap (Seatec Aquasuits, Australia).
Often overlooked is the impact that mask design can have on the effectiveness of ventilatory support. Ventilators designed specifically for non-invasive applications generally operate using a single circuit with an expiratory port placed either within the mask itself or close to the mask at the end of the ventilator circuit. In a lung model, Schettino and colleagues (2003) demonstrated that the use of an oronasal mask with the exhalation port within the mask generated less CO2 rebreathing than when the exhalation was placed in the ventilator circuit itself. In a later study, also using a modified lung model, Saatci and co-workers (2004) found that the use of facemasks with expiratory ports over the nasal bridge produced beneficial flow characteristics within the facemask and nasal cavity, resulting in a decrease in dead space. Expiratory ports placed at other sites such as at the cheeks or at the mask–circuit connection were not as effective. In addition, having some level of constant pressure throughout the expiratory phase was benefi cial in reducing the total dynamic dead space close to or even below physiological dead space. While these factors may not be of great clinical significance for most patients using NIV, in those with a borderline response to NIV, a change in mask to one with lower dynamic dead space or more favourable positioning of the expiratory ports may be the difference between success and failure of the technique.
Humidification, nebulization and oxygen therapy
In some patients, the high flows of cold dry air across the nasal passages can cause distressing nasal symptoms that may affect compliance with therapy, or increase nasal resistance (Richards et al 1996). Patients may report sneezing, nasal stuffiness or rhinorrhoea and erroneously believe they are developing a head cold. The use of an in-line humidifier such as an HC–150 (Fisher & Paykel Healthcare Ltd, Auckland, New Zealand), that can both warm and moisten the air, will largely improve these symptoms. Nasal symptoms frequently point to the presence of significant mouth leaks and this should be rectified, as leaks may reduce the effectiveness of ventilation. In patients with bronchial hypersecretion, such as cystic fibrosis or bronchiectasis, the addition of in-line humidification while using nasal ventilatory support may be useful in ensuring secretions are well hydrated. Patients with acute respiratory failure may become dehydrated and can also benefit from additional humidification of the airways to improve secretion clearance (Wood et al 2000). Heated humidifiers are preferable to heat moisture exchangers as the latter have been shown to increase dead space, resulting in a higher PaCO2 despite significant increases in minute ventilation (Jaber et al 2002).
In patients who require ventilatory support on a continuous basis, nebulized bronchodilators and normal saline can be given during mask ventilatory support by adding the nebulizer chamber in-line close to the mask interface. Bilevel ventilatory support devices have been used to deliver β2-agonists in the emergency department for patients with bronchospasm with a greater increase in peak expiratory flow rates compared with aerosols delivered by small-volume nebulizers alone (Pollack et al 1995). Fauroux and colleagues (2000) have shown that delivery of a nebulized aerosol by NIV enhances total lung aerosol deposition without increasing particle impaction in the proximal airways in patients with cystic fibrosis.
Generally, supplemental oxygen is not required in those patients with chronic respiratory failure from neuromuscular or chest wall disorders. However, in patients with parenchymal disease or those with acute respiratory failure, additional oxygen is likely to be needed and can be added either into the ventilator tubing or into a port on the mask itself. In some portable ventilator systems, it may be possible to dial up an exact FiO2. However, most simple bilevel devices do not have this feature and the concentration of oxygen being delivered can only be estimated. Factors such as where along the circuit the oxygen is injected, the ventilator settings, the type of leak port and the oxygen flow will all influence the delivered oxygen concentration. In practice, the flow rate of oxygen used is determined by the oxygen saturation achieved in the patient.
Monitoring and initiating therapy
When commencing mask ventilation in the acute situation, careful monitoring is mandatory in order to gauge the effectiveness of ventilatory support. Continuous monitoring of oxygen saturation should be carried out for at least the first 24 hours, with transcutaneous carbon dioxide monitoring if available (British Thoracic Society 2002) (Fig. 11.6). Direct measurements of arterial blood gases should be taken before starting ventilation, then again at 1 hour, 6 hours, 24 hours and as needed thereafter depending on the patient’s clinical condition. In addition, heart rate, respiratory rate and FiO2 should be recorded hourly for the first few hours until the patient is stable. Blood pressure measurements may also be necessary, particularly if there is any question of the patient’s haemodynamic stability. Monitoring in the acute situation is crucial to identify early any deterioration in the patient’s clinical situation or to uncover any technical problems with NIV so that prompt and appropriate action can be taken.
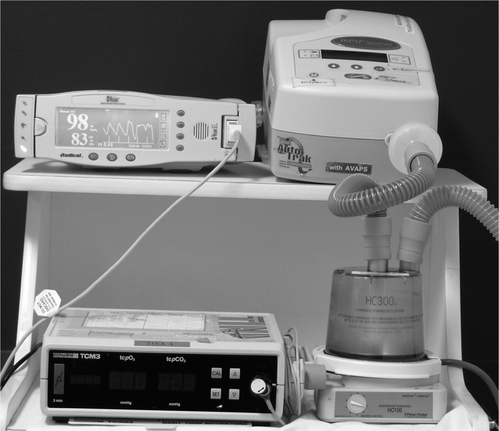
Figure 11.6 Monitoring set up for NIV trials. Oximeter and transcutaneous carbon dioxide monitor (lower left of picture) used to measure the physiological response to nasal ventilatory support. An active humidifier has been placed in the circuit between the bilevel ventilator and the patient (bottom right-hand corner).
Initial reports found that NIV in the acute care setting was a time-consuming procedure (Chevrolet et al 1991). While more recent experience suggests that this is not necessarily the case (Kramer et al 1995, Nava et al 1997), it does need to be appreciated that a time commitment from a member of the NIV team is necessary to initially coach the patient, check the efficacy of the technique and make adjustments to the machine settings. Nevertheless, it has been shown that the use of this technique can be transferred to the general ward environment without a reduction in efficacy (Plant et al 2000), while achieving significant cost savings (Plant et al 2003). This, however, requires adequate training and the opportunity for staff to develop sufficient skills and knowledge to perform the technique effectively and safely (British Thoracic Society 2002). Non-invasive ventilation in the acute setting is usually continued until blood gases have stabilized for several hours, then trial periods off the mask are started. The patient’s response to spontaneous ventilation is monitored and mask ventilatory support re-instituted if breathing deteriorates. In some cases, almost continuous use of the mask during the first day or two may be necessary. There is then a gradual withdrawal of awake ventilatory support to nocturnal use only. Prior to hospital discharge, investigation into the need for domiciliary therapy should be undertaken.
In patients with chronic respiratory failure, acclimatization to the mask and machine can be carried out during the day, with monitoring of oxygen saturation and preferably CO2, either end-tidal or transcutaneous. Low pressures, commonly IPAP 9–10 cmH2O and EPAP 4 cmH2O, are trialled initially, with increases in the inspiratory pressure based on patient tolerance and the impact on saturation (Box 11.3). Once the patient has a broad understanding of the sensations to expect when using mask ventilation, adjustments to other settings, such as pressurization times or inspiratory trigger levels, can be undertaken to improve comfort and synchronization.
Box 11.3 Steps in initiating NIV therapy for home ventilatory support
 Ensure the mask fits comfortably and that the patient can experience the mask on their face without the ventilator connected
Ensure the mask fits comfortably and that the patient can experience the mask on their face without the ventilator connected Allow the patient the opportunity to feel the operation of the machine through the mask on their hand or cheek before applying it over their nose or mouth
Allow the patient the opportunity to feel the operation of the machine through the mask on their hand or cheek before applying it over their nose or mouth Allow the patient the opportunity to practise breathing with the ventilator, either holding the mask in place or allowing them to hold it in place before applying the straps
Allow the patient the opportunity to practise breathing with the ventilator, either holding the mask in place or allowing them to hold it in place before applying the straps Adjust settings initially for comfort and establish whether the patient can relax comfortably in a sleeping posture
Adjust settings initially for comfort and establish whether the patient can relax comfortably in a sleeping posture Provide opportunities for the patient to feed back any discomfort or uncertainty with regard to the use of the equipment
Provide opportunities for the patient to feed back any discomfort or uncertainty with regard to the use of the equipmentFrequently the patient will fall asleep during these initial trials and problems such as mouth leaks or the development of upper airway obstruction may be identified at this time, and alterations to machine settings made. Once the patient is able to sleep for a number of hours on the machine, a sleep study or nocturnal respiratory monitoring should be performed. Although some centres base machine settings on ventilation achieved during wakefulness, such settings may not be adequate during sleep for patients with chronic respiratory failure. This may relate to changes in the behaviour of the glottis, mouth leaks, alteration in respiratory drive or compliance of the respiratory system associated with changes in sleep state. Depending on the monitoring system used, a number of respiratory variables can be recorded, including mask pressure, leak, oxygen saturation, chest wall motion, diaphragmatic and other respiratory muscle electromyograms or inspiratory/expiratory tidal volumes. This information is useful in determining the efficacy of ventilatory support and in troubleshooting. If ventilation is not being adequately supported, a change in the mode or type of ventilator can be made.
Studies have shown that NIV will be effective in improving arterial blood gases and offloading the inspiratory muscles irrespective of whether settings are determined on the basis of the patient’s comfort and tolerance or by invasive monitoring of respiratory mechanics (Fanfulla et al 2005, Vitacca et al 2000).
Adverse effects
A number of complications and adverse effects can arise during attempts to establish patients on non-invasive ventilation. Mouth leaks during the inspiratory phase of the ventilator cycle are probably the most common problem. This leak can reduce effective ventilation and may only be obvious during certain sleep stages. Leaks may be seen in the presence of upper airway obstruction, if asynchrony between the patient and the ventilator develops or if the lips and palate fail to provide a seal. If the leak is significant it can usually be remedied by the use of a chinstrap which should cradle the chin and hold the lower jaw up (Fig. 11.5). The chinstrap is designed to have elastic sections on the sides so that patients can still move their jaw comfortably and call out and breathe should their nose become blocked or the ventilator fail. Other solutions for this problem include repositioning of the neck, taping the lips, mouth guards and full facemasks. Full facemasks are usually preferred once the patient’s confidence has been established. The presence of leaks will not only reduce the degree of effective ventilation reaching the lungs but also may cause sleep fragmentation (Teschler et al 1999). Upper airway obstruction can occur particularly if the cycling pressure is allowed to drop below the closing pressure of the upper airway. It is very difficult to establish effective ventilation when this occurs, although it can, in some very mild cases, be reduced by positioning the patient’s head so that the neck is slightly more extended. Sometimes a chinstrap alone is effective in lifting the jaw and thereby opening the upper airway. An increase in end-expiratory pressure usually ensures adequate ventilation.
Mask leaks commonly occur on either side of the bridge of the nose and can cause significant irritation to the eyes. If the leak is small it can be compensated for by the machine and this is preferable to pulling the mask too tightly onto the face. Patients usually learn to eliminate the leaks by repositioning the mask or by adjusting the strap alignment. Elastic straps of the head harnesses usually need regular replacement to ensure effective mask pressures. Other solutions include custom-built masks and a change in sleeping posture.
Mask pressure can cause pressure sores or pressure marks on the bridge of the nose or across the top lip in particular. These are best prevented by careful selection of mask for size and skin sensitivity. The areas respond well to standard pressure care including gentle massage, being left clean, dry and open and getting a regular amount of sunshine. The bridge of the nose often becomes thick and tough with time, although some people have recurring problems. For these patients the bridge of the nose can be protected with special pressure-absorbing materials that are commonly used with prostheses. Alternatively the patient may need to use a mouthpiece or nasal pillows (e.g. SwiftTM, ResMed, North Ryde, Australia; ComfortLiteTM, Respironics, Inc, Murrysville, PA, USA) (Fig. 11.3C & D) which fit securely into the nares without pressure on the nasal bridge, permitting pressure areas to heal. Head harness or strap pressure can cause abrasions over the back of the neck or over the ears. This can be simply relieved by re-designing the head harness to realign the straps or to include cotton wadding or a pad over the tender parts. If patients require NIV for extended periods of time, alternating between two types of interface such as a nasal mask and mouthpiece can help avoid sustained pressure on the skin.
Abdominal distension can be caused by air in the stomach, particularly when high cycling pressures are required for effective ventilation. This problem is less frequent now but would be more likely with volume-cycled ventilators. It appears that air can track through the stomach to the bowel and cause considerable discomfort. Every effort should be made to lower the cycling pressure without compromising effective ventilation. Some patients find relief from lying on their left side at night, some from having an empty stomach and others resort to medications including charcoal and acidophilus tablets. Pulmonary barotrauma is another potential but rarely encountered complication of positive pressure therapy. In some disorders managed by NIV, such as cystic fibrosis and COPD, subpleural cysts may be present, increasing the possibility of rupture due to or coincident with the use of positive pressure therapy. Patients with Duchenne muscular dystrophy have also been reported to have an increased risk of spontaneous pneumothorax. Although the risk is small it is potentially life threatening for a patient with limited respiratory reserve. Clinicians and patients should be aware of the potential for this occurring, with prompt investigation of any sudden shortness of breath and chest pain the patient experiences (Simonds 2004).
Home management
Once the patient is tolerating the machine during sleep and ventilator settings are finalized, patients should be encouraged to manage the equipment themselves and solve any problems that may arise prior to home discharge. Alternatively, if individuals remain dependent on some assistance the home carers should be brought in for at least part of the night to develop skills in setting up and troubleshooting.
Most people adapt well to ongoing ventilatory support in the home. However, acceptance of therapy may differ depending on the underlying pathology, the patient’s response to therapy and relief of symptoms. Initial experience with mask ventilation may also influence outcome. A number of hospitals start ventilation on an inpatient basis to provide the patient with maximum support while minimizing problems (Meecham Jones et al 1995, Piper & Willson 1996).
It is important to provide full information to patients and families at the time of considering ongoing ventilation to ensure that an informed choice is made, especially in patients with progressive disorders. Some patients may need time to adapt to the idea of assisted ventilation, and will initially reject ongoing therapy. Others may find it beyond their resources and capabilities to acquire and manage the technology or may fear it will weaken their muscles further, causing them to become ventilator-dependent. Each of these beliefs needs to be explored and discussed without judgement. They can be resolved in a number of ways. Patients need to be allowed to make their own choice and when the symptoms of respiratory failure or sleep deprivation become severe enough they may then seek relief and ask to try therapy again. Alternatively patients can be counselled, often with the help of other patients, that the benefits outweigh any real or perceived detriments. For most patients, compliance is usually dependent on relief of symptoms, and once they are sleeping on the ventilator overnight with control of blood gases, the improvement in daytime function often sustains continuing use of ventilation.
Ongoing review of the patient once discharged home on ventilatory support is essential to ensure therapy remains effective and appropriate. This is especially important for patients with progressive disorders and for children, as their circumstances and needs will change with time. Monitoring of basic lung function such as spirometry and arterial blood gases can identify any deterioration in the patient’s condition and signal the need for more extensive investigation. In many of the newer home ventilators, objective compliance with therapy can be monitored using data stored within the device itself. This is important when trying to determine whether return of symptoms or deterioration in awake blood gases are a consequence of inadequate nocturnal use or progression of the underlying disorder. Other issues such as weight control, enrolment in a rehabilitation programme or referral to other services may also need to be discussed during these reviews. For patients with progressive neuromuscular disorders, review of swallowing and nutritional status is also necessary, and these patients will require extensive input from a number of specialties as their muscle weakness increases. Equipment needs to be inspected and reviewed regularly, and worn masks, filters and accessories replaced.
While patients are not ventilator-dependent, many express considerable anxiety about the risk of being without the ventilator even for one night. They may be anxious about the symptoms of sleep deprivation and hypercapnia. Those who are geographically isolated or live alone are particularly vulnerable if there is equipment failure. Back-up systems and emergency plans are valuable and need to be worked out with each indivi-dual. Patients should be encouraged to enter into a regular maintenance agreement with the companies or hospitals supplying the equipment. All relevant instructions for cleaning and maintenance should be provided in writing and in their preferred language. In a recent study looking at the performance of ventilation equipment in the patient’s home, considerable differences between the settings prescribed, the values set on the control panel of the machines and the actual ventilation settings delivered were found (Farre et al 2006). This highlights the importance of both the patient and the prescribing centre developing regular equipment review procedures to ensure therapy is both safe and effective.
SPECIAL APPLICATIONS OF NON-INVASIVE VENTILATION
Children and infants
Although non-invasive ventilatory support is now seen as a first-line therapy for adults with hypercapnic respiratory failure, this technique has, to date, been less commonly used by paediatric centres. To some extent this is due to a lack of randomized controlled trials of NIV in children, although an increasing number of published reports would suggest use of the technique in children is increasing (Teague 2005). In addition, some reports have suggested that infants and young children may not be tolerant of mask therapy (Heckmatt et al 1990), limiting the application of this technique. However, one of the earliest reports of mask ventilation use involved a 6-year-old-child with congenital central hypoventilation syndrome (Ellis et al 1987a). Since that time, a number of studies have appeared in the literature describing the successful use of nasal masks for both CPAP and NIV in more than 300 children. Many of those described were under 6 years of age, yet tolerated therapy well both in the short and long term (Fortenberry et al 1995, Simonds et al 2000, Waters et al 1995).
Reports of nasal ventilation use in paediatric patients have included children with upper airway obstruction, cystic fibrosis, congenital central hypoventilation syndrome and neuromuscular disorders (Ellis et al 1987b, Fauroux et al 2005, Simonds et al 2000). Prior to the introduction of mask ventilation, children with chronic respiratory failure from these disorders were managed with tracheostomy in order to deliver positive pressure to the lungs. Although effective, the tracheostomy tube can also interfere with speech development and may predispose the child to chest infections. While mask ventilation can also be associated with side effects, most commonly skin breakdown and leak, these problems are generally minor or manageable. In the largest study of its kind to date, Simonds and colleagues (2000) reported the use of domiciliary mask ventilation in 40 children with respiratory failure secondary to congenital neuromuscular and skeletal disorders. The youngest child commenced on therapy was 9 months of age. Thirty-eight tolerated mask ventilatory support in the long term, resulting in reversal of nocturnal hypoventilation and significant improvements with daytime spontaneous CO2 and O2 levels.
Mask ventilation has also been used in children presenting with acute hypoxaemic or hypercapnic respiratory failure (Fortenberry et al 1995, Padman et al 1998). As with adults, therapy has been instituted to improve gas exchange, reduce the work of breathing and avoid intubation. It has also been used successfully to wean patients who failed attempted extubation or to facilitate extubation in those likely to have difficulty resuming spontaneous ventilation (Brinkrant et al 1997). In circumstances where there are ethical or medical concerns about the use of invasive ventilation techniques in children with severe neurological dysfunction or terminal diseases, mask ventilation offers a realistic active treatment alternative (Teague 2005). Although randomized trials are lacking, mounting clinical evidence suggests mask ventilation in children with acute respiratory failure is both feasible and safe.
Increasingly, bilevel devices rather than volume preset are used in this population due to simplicity, portability and cost. These devices are flow-initiated and therefore can be easier to trigger and more comfortable than volume-preset devices. In addition, the availability of setting EPAP with bilevel devices can be valuable in patients requiring stabilization of the upper airway as well as ventilatory support. The principles of adjusting settings for NIV in children are no different from those used with adults. Initial IPAP and EPAP pressures are set to achieve both patient comfort and the goals of ventilatory support, and are altered later depending on clinical response and patient acceptance. Many of the modern bilevel devices now have internal alarms and these should be set to alert carers to the loss of airway pressure associated with mask removal or machine malfunction. External alarm systems may need to be added to those devices without internal alarms.
Generally, children tolerate masks well, although initially extra time and effort may be needed to encourage the child to keep the mask in place. Imagination and patience, from both the clinician and the child, help. Compared with adult masks, there is a limited range for children and even fewer for neonatal use. Most ‘children’s’ masks are simply scaled-down versions of the adult model. Although this is not much of a problem for the older, larger child, it can make fitting a mask for the younger patient (<2 years) a little more challenging. Medium-sized adult nasal masks can be used as full facemasks for the smaller child (Simonds et al 2000). Care needs to be taken when choosing a mask to ensure dead space is minimized and sufficient carbon dioxide washout is occurring. However, as the technique becomes more widely accepted for this population, a larger choice of mask should become available.
A specific problem that can arise in children using long-term mask therapy is that of altered facial skeletal development resulting from the application of tightly fitting headgear and nasal mask. Although only mild effects have been reported (Fauroux et al 2005, Simonds et al 2000), the impact of mask therapy on craniofacial development should be taken into consideration when choosing and fitting mask equipment. Simonds and colleagues (2000) reported four cases of mild mid-facial hypoplasia in their series of 40 children treated long-term with mask ventilation. Fauroux and colleagues (2005) found that global facial flattening was present in 68% of children using home mask ventilation. Strategies to minimize these side effects include using different masks rotated on a weekly basis to reduce and vary the pressure over the maxillary region and reducing the amount of time NIV is used daily (Fauroux et al 2005, Simonds et al 2000). Regular maxillofacial evaluation and follow-up is essential in this population (Fauroux et al 2005). In those patients requiring long-term therapy, frequent review is necessary to ensure the ventilator settings and the mask size remain adequate as the child grows and develops. In addition, the child’s parents/ carers need to be trained to supervise therapy and solve problems as they arise.
There is no doubt that as clinicians become more experienced and confident with the technique, more paediatric centres will come to accept mask ventilation as a suitable modality for children. As a consequence, increasingly more children will be offered mask ventilation as first-line therapy for the management of respiratory failure, in hospital as well as in the home. The success of this therapy relies heavily on initiation of treatment by skilled therapists and training both the child and family in its use.
Weaning and early extubation
Most patients can be weaned from mechanical ventilation without incident. However, a small number will require a prolonged weaning period, particularly when there is a history of underlying lung, chest wall or neuromuscular disease. Although various weaning strategies have been developed to facilitate the resumption of spontaneous breathing, some patients will not tolerate removal of ventilatory support without developing unacceptably high levels of carbon dioxide retention. Non-invasive ventilatory support can be a useful tool in the weaning of such patients from conventional mechanical ventilation, permitting the earlier removal of the endotracheal tube than with conventional invasive pressure support techniques (Girault et al 1999, Nava et al 1998). Data from three randomized controlled trials have shown that early extubation and application of NIV results in shorter length of stay, reduces the incidence of complications and improves survival, especially in patients with underlying COPD (Ferrer et al 2003, Girault et al 1999, Nava et al 1998). However, the application of NIV to unselected patients in the post-extubation period either prophylactically (Jiang et al 1999) or after respiratory failure develops within 48 hours of extubation (Esteban et al 2004, Keenan et al 2002) and does not appear to be of benefit. In contrast, in patients with a high risk of post-extubation failure, the use of NIV applied immediately following extubation has been shown to reduce the need for reintubation (Ferrer et al 2006, Nava et al 2005) and to improve survival in those with hypercapnia at extubation (Nava et al 2005). Therefore, based on current evidence, there does not appear to be a place for the routine use of NIV in all patients following extubation (Jiang et al 1999). However, NIV should be considered in any patient considered at risk of developing post-extubation respiratory difficulties, with therapy initiated immediately post-extubation rather than waiting for signs of respiratory distress to develop (Ferrer et al 2006, Nava et al 2005).
In patients already tracheostomized and on partial ventilatory support, nasal mask ventilation may be substituted for tracheal support (Restrick et al 1993). This is usually started on a continuous basis, with the patient removing the mask for short periods for eating, speaking and coughing. Periods of spontaneous breathing are then interspersed with periods on the nasal mask, the balance being determined by patient tolerance and clinical response. Once nasal ventilatory support has been shown to be acceptable and to effectively support ventilation, the tracheostomy tube is removed. Non-invasive ventilation is then used nocturnally and for any rest/sleep period during the day as required. Although many patients may be weaned entirely from the mask, some will have an underlying process which features sleep-disordered breathing. Therefore, investigation into the presence of nocturnal breathing abnormalities and discharge home on nocturnal ventilatory support should be considered.
Physiotherapy intervention during non-invasive ventilation
Physiotherapists have long been involved in the application of positive pressure therapy to patients with respiratory problems with the aim of improving gas exchange, mobilizing secretions or reducing dyspnoea. Through until the mid-1970s physiotherapists routinely administered respiratory medications by intermittent positive pressure breathing (IPPB) through a mask or mouthpiece (Bennett et al 1976), or used IPPB to increase ventilation in patients with severe pain or with poor ventilatory control. The mid-1980s saw the introduction of nocturnal mask ventilation as a feasible and effective method of supporting breathing during sleep, resulting in improved awake gas exchange, daytime function and quality of life. Physiotherapists were central in introducing and applying this technique in a wide range of respiratory disorders (Ellis et al 1987a, 1987b, 1988, Holland et al 2003, Keilty et al 1994, Milross et al 2001, Piper & Sullivan 1994b, Piper & Willson 1996, Piper et al 1992). When the potential for this technique in the management of acute respiratory failure was realized, physiotherapists were again amongst the forerunners in applying and evaluating therapy (Bott et al 1993, Conway et al 1993).
In daily practice, physiotherapists may become involved with the application of this technique in a number of different ways (Box 11.4). Data from a survey looking at physiotherapy practice in European intensive care units found that almost half the respondents reported physiotherapy involvement in implementing and supervising NIV (Norrenberg & Vincent 2000). In a survey of physiotherapy involvement in non-invasive ventilation hospital services in the British Isles, more than 90% of respondents reported physiotherapy involvement in some aspect of the management of patients using NIV, with around half contributing to implementation of the technique (Moran et al 2005). The skills and knowledge base of physiotherapists regarding respiratory disease and its management place them in a good position to be key members contributing to any non-invasive ventilation service.
Box 11.4 Role of the physiotherapist in home ventilation services
Even where therapists are not directly involved in implementing NIV, the widespread use of this technique both in high dependency and ward areas means that at some point most will be involved in the care of patients using this therapy (Moran et al 2005). In the acute setting one of the most common causes of NIV failure is the inability to manage secretion clearance effectively (Carlucci et al 2000). While there is currently limited published data confirming the efficacy of various airway clearance techniques during acute NIV use, the practice is widespread (Moran et al 2005). In a small randomized trial in patients with acute hypercapnic respiratory failure, Inal-Ince and colleagues (2004) found that the addition of the active cycle of breathing techniques (ACBT) to NIV resulted in a small reduction in the length of time NIV was required (5 vs 6.7 days, p = 0.03). However, this did not impact on any other outcome measure such as length of ICU stay. In patients with acute exacerbations of COPD and large amounts of sputum, the addition of positive expiratory pressure (PEP) and assisted coughing to routine NIV management resulted in greater amounts of sputum removed and a shorter period of NIV use (Bellone et al 2002). These interesting, preliminary results illustrate the potential benefits of traditional airway clearance techniques in conjunction with NIV in the management of patients with acute respiratory failure and require further exploration.
It has also been shown that NIV may have a place in selected patients as a stand-alone method to aid in secretion removal (Fauroux et al 1999, Holland et al 2003). For the breathless patient who is unable to lie flat, use of NIV may permit the use of postural drainage positions that would otherwise not be tolerated (Piper et al 1992). Anecdotally, patients report being able to tolerate longer physiotherapy sessions when using ventilatory support, which is important in patients who tire easily but who have retained or copious secretions. In patients with severe muscle weakness and poor cough, mask ventilation may be used to assist deep breathing and mobilization of secretions. In these circumstances techniques such as overpressures, breath stacking and mechanical in-exsufflation may be used. The tidal volume or inspiratory pressure of the device may be increased during physiotherapy sessions to aid chest wall expansion and assist the mobilization of secretions (Piper & Moran 2006).
Exercise
By the time patients with chronic respiratory failure present for nocturnal ventilatory support they are usually severely debilitated. Their presentation is usually characterized by severe shortness of breath on exertion, excessive daytime sleepiness, fatigue, prolonged illness and regular hospitalization. Consequently significant peripheral deconditioning has likely occurred which will limit their tolerance to daily activities and exercise performance. After a period of nocturnal ventilatory support, patients are able to perform a great deal more work without fatigue (Fuschillo et al 2003, Schonhofer et al 2001). Survey data has shown that exercise is commonly used as part of the physiotherapy management for patients using NIV (Moran et al 2005). However, fewer centres use the technique routinely during exercise training. The beneficial effects of positive pressure during exercise in patients with severe lung disease have been reported (Keilty et al 1994, van’t Hul et al 2002). Acute benefits include reduced breathlessness, increased exercise time and improved oxygen saturation. However, the long-term benefits of routine application of this technique during exercise training remain unclear and await further investigation.
CONCLUSION
Non-invasive ventilation is a technique which can improve gas exchange and reduce the work of breathing and is becoming increasingly used to manage both chronic and acute respiratory failure. Evidence for the effectiveness of this technique in a wide range of clinical situations has seen it become an integral part of the respiratory management for patients with respiratory failure. Physiotherapists working in respiratory care require knowledge of and skills with the equipment and its applications in order to continue to provide holistic and effective treatment programmes to manage these patients.
Acknowledgements
We would like to thank the following companies for their kind permission to use images of their products: Fisher and Paykel Healthcare Ltd, Auckland, New Zealand; ResMed Limited, North Ryde, Australia and Respironics, Inc, Murrysville, PA. ResMed Limited owns the copyright of the ResMed images and ResMed Limited reserves all rights.
Ambrosino N, Foglio K, Rubini F, et al. Noninvasive mechanical ventilation in acute respiratory failure due to chronic obstructive pulmonary disease: correlates for success. Thorax. 1995;50:755-757.
Anton A, Guell R, Gomez J, et al. Predicting the result of noninvasive ventilation in severe acute exacerbations of patients with chronic airflow limitation. Chest. 2000;117:828-833.
Antonelli M, Conti G, Bufi M, et al. Noninvasive ventilation for treatment of acute respiratory failure in patients undergoing solid organ transplantation. Journal of the American Medical Association. 2000;283:235-241.
Auriant I, Jallot A, Herve P, et al. Noninvasive ventilation reduces mortality in acute respiratory failure following lung resection. American Journal of Respiratory and Critical Care Medicine. 2001;164:1231-1235.
Barach AL, Martin J, Eckman M. Positive-pressure respiration and its application to the treatment of acute pulmonary edema. Annals of Internal Medicine. 1938;12:754-795.
Bellone A, Spagnolatti L, Massobrio M, et al. Short-term effects of expiration under positive pressure in patients with acute exacerbations of chronic obstructive pulmonary disease and mild acidosis requiring non-invasive positive pressure ventilation. Intensive Care Medicine. 2002;28:581-585.
Bennett L, Heath J, Mitchell R. An inpatient observation and comparison of the Bennett’s IPPB and aerosol methods of administering salbutamol. Australian Journal of Physiotherapy. 1976;23:111-113.
Bott J, Carroll MP, Conway JH, et al. Randomized controlled trial of nasal ventilation in acute ventilatory failure due to chronic obstructive airways disease. Lancet. 1993;341:1555-1557.
Bourke SC, Tomlinson M, Williams TL, et al. Effects of non-invasive ventilation on survival and quality of life in patients with amyotrophic lateral sclerosis: a randomized controlled trial. Lancet Neurology. 2006;5:140-147.
Brinkrant DJ, Pope JF, Eiban RM. Pediatric noninvasive nasal ventilation. Journal of Child Neurology. 1997;12:231-236.
British Thoracic Society Standards of Care Committee. Non-invasive ventilation in acute respiratory failure. Thorax. 2002;57:192-211.
Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. New England Journal of Medicine. 1995;333:817-822.
Bye PTP, Ellis ER, Issa FG, Donnelly PD, Sullivan CE. The role of sleep in the development of respiratory failure in patients with neuromuscular disease. Thorax. 1990;45:241-247.
Carlucci A, Richard JC, Wysocki M, Lepage E, Brochard L. Noninvasive versus conventional mechanical ventilation. An epidemiologic survey. American Journal of Respiratory and Critical Care Medicine. 2000;163:874-880.
Casanova C, Celli BR, Tost L, et al. Long-term controlled trial of nocturnal nasal positive pressure ventilation in patients with severe COPD. Chest. 2000;118:1582-1590.
Chevrolet JC, Jolliet P, Abajo B, Toussi A, Louis M. Nasal positive pressure ventilation in patients with acute respiratory failure. Difficult and time-consuming procedure for nurses. Chest. 1991;100:775-782.
Clini E, Sturani C, Rossi A, et al. The Italian multicentre study noninvasive ventilation in chronic obstructive pulmonary disease patients. European Respiratory Journal. 2002;20:529-538.
Confalonieri M, Potena A, Carbone G, et al. Acute respiratory failure in patients with severe community acquired pneumonia: a prospective randomized evaluation of non-invasive ventilation. American Journal of Respiratory and Critical Care Medicine. 1999;160:1585-1591.
Conway JH, Hitchcock RA, Godfrey RC, Carroll MP. Nasal intermittent positive pressure ventilation in acute exacerbations of chronic obstructive pulmonary disease - a preliminary study. Respiratory Medicine. 1993;87:387-394.
Delclaux C, L’Her E, Alberti C, et al. Treatment of acute hypoxemic nonhypercapnic respiratory insufficiency with continuous positive airway pressure delivered by a face mask. A randomized controlled trial. Journal of the American Medical Association. 2000;284:2352-2360.
Douglas NJ, White DP, Pickett CK, Weil JV, Zwillich CW. Respiration during sleep in normal man. Thorax. 1982;37:840-844.
Ellis ER, McCauley VB, Mellis C, Sullivan CE. Treatment of alveolar hypoventilation in a six-year-old girl with intermittent positive pressure ventilation through a nose mask. American Review of Respiratory Disease. 1987;136:188-191.
Ellis ER, Bye PTP, Bruderer JW, Sullivan CE. Treatment of respiratory failure in patients with neuromuscular disease. American Review of Respiratory Disease. 1987;135:148-152.
Ellis ER, Grunstein RR, Chan CS, Bye PTP, Sullivan CE. Treatment of nocturnal respiratory failure in kyphoscoliosis. Chest. 1988;94:811-815.
Esteban A, Frutos-Vivar F, Ferguson ND, et al. Noninvasive positive-pressure ventilation for respiratory failure after extubation. New England Journal of Medicine. 2004;350:2452-2460.
Fanfulla F, Delmastro M, Berardinelli A, Lupo ND, Nava S. Effects of different ventilator settings on sleep and inspiratory effort in patients with neuromuscular disease. American Journal of Respiratory and Critical Care Medicine. 2005;172:619-624.
Farre R, Navajas D, Prats E, et al. Performance of mechanical ventilators at the patient’s home: a multicenter quality control study. Thorax. 2006;61:400-404.
Fauroux B, Boule M, Lofaso F, et al. Chest physiotherapy in cystic fibrosis: improved tolerance with nasal pressure support ventilation. Pediatrics. 1999;103:E32.
Fauroux B, Itti E, Pigeot J, Isabey D, et al. Optimization of aerosol deposition by pressure support in children with cystic fibrosis: an experimental and clinical study. American Journal of Respiratory and Critical Care Medicine. 2000;162:2265-2271.
Fauroux B, Lavis J-F, Nicot F, Picard A, Boelle P-Y, Clement A, Vazquez M-P. Facial side effects during noninvasive positive pressure ventilation in children. Intensive Care Medicine. 2005;31:965-969.
Ferrer M, Esquinas A, Leon M, et al. Non-invasive ventilation in severe hypoxemic respiratory failure. A randomized clinical trial. American Journal of Respiratory and Critical Care Medicine. 2003;168:1438-1444.
Ferrer M, Valencia M, Nicolas JM, et al. Early noninvasive ventilation averts extubation failure in patients at risk. American Journal of Respiratory and Critical Care Medicine. 2006;173:164-170.
Fortenberry JD, Del Toro J, Jefferson LS, Evey L, Haase D. Management of pediatric acute hypoxemic respiratory failure with bilevel positive pressure (BiPAP) nasal mask ventilation. Chest. 1995;108:1059-1064.
Fuschillo S, De Felice A, Gaudiosi C, Balzano G. Nocturnal mechanical ventilation improves exercise capacity in kyphoscoliotic patients with respiratory impairment. Monaldi Archives for Chest Disease. 2003;59:281-286.
Garay SM, Turino GM, Goldring RM. Sustained reversal of chronic hypercapnia in patients with alveolar hypoventilation syndromes: long-term maintenance with noninvasive mechanical ventilation. American Journal of Medicine. 1981;70:269-274.
Gastaut H, Tassinari CA, Duron B. Polygraphic study of the episodic diurnal and nocturnal manifestations of the Pickwick syndrome. Brain Research. 1966;1:167-186.
Girault C, Daudenthun I, Chevron V, et al. Noninvasive ventilation as a systematic extubation and weaning technique in acute-on-chronic respiratory failure. American Journal of Respiratory and Critical Care Medicine. 1999;160:88-92.
Girault C, Richard JC, Chevron V, et al. Comparative physiologic effects of noninvasive assist-control and pressure support ventilation in acute hypercapnic respiratory failure. Chest. 1997;111:1639-1648.
Gozal D. Nocturnal ventilatory support in patients with cystic fibrosis: comparison with supplemental oxygen. European Respiratory Journal. 1997;10:1999-2003.
Heckmatt JZ, Loh L, Dubowitz V. Night-time nasal ventilation in neuromuscular disease. Lancet. 1990;335:579-582.
Hilbert G, Gruson D, Vargas F, et al. Noninvasive ventilation in immunosuppressed patients with pulmonary infiltrates, fever, and acute respiratory failure. New England Journal of Medicine. 2001;344:481-487.
Hill N. Noninvasive ventilation. Does it work, for whom, and how? American Review of Respiratory Disease. 1993;147:1050-1055.
Hodson ME, Madden BP, Steven MH, Tsang VT, Yacoub MH. Non-invasive mechanical ventilation for cystic fibrosis patients - a potential bridge to transplantation. European Respiratory Journal. 1991;4:524-527.
Holland AE, Denehy L, Ntoumenopoulos G, Naughton MT, Wilson JW. Non-invasive ventilation assists chest physiotherapy in adults with acute exacerbations of cystic fibrosis. Thorax. 2003;58:880-884.
Inal-Ince D, Savci S, Topeli A, Arikan H. Active cycle of breathing techniques in non-invasive ventilation for acute hypercapnic respiratory failure. Australian Journal of Physiotherapy. 2004;50:67-73.
Jaber S, Chanques G, Matecki S, et al. Comparison of the effects of heat and moisture exchangers and heated humidifiers on ventilation and gas exchange during non-invasive ventilation. Intensive Care Medicine. 2002;28:1590-1594.
Jiang JS, Kao SJ, Wang SN. Effect of early application of biphasic positive airway pressure on the outcome of extubation in ventilator weaning. Respirology. 1999;4:161-165.
Keenan SP, Powers C, McCormack DG, Block G. Noninvasive positive-pressure ventilation for postextubation respiratory distress. A randomized controlled trial. Journal of the American Medical Association. 2002;287:3238-3244.
Keenan SP, Sinuff T, Cook DJ, Hill NS. Which patients with acute exacerbation of chronic obstructive pulmonary disease benefit from noninvasive positive-pressure ventilation? A systematic review of the literature. Annals of Internal Medicine. 2003;138:861-870.
Keilty SEJ, Ponte J, Fleming TA, Moxham J. Effect of inspiratory pressure support on exercise tolerance and breathlessness in patients with severe stable chronic obstructive pulmonary disease. Thorax. 1994;49:990-994.
Kramer N, Meyer TJ, Mehang J, Cece RD, Hill NS. Randomized, prospective trial of noninvasive positive pressure ventilation in acute respiratory failure. American Journal of Respiratory and Critical Care Medicine. 1995;151:1799-1806.
Kwok H, McCormack J, Cece R, Houtchens J, Hill NS. Controlled trial of oronasal versus nasal mask ventilation in the treatment of acute respiratory failure. Critical Care Medicine. 2003;31:468-473.
Lightowler JV, Wedzicha JA, Elliott MW, Ram FSF. Non-invasive positive pressure ventilation to treat respiratory failure resulting from exacerbations of chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis. British Medical Journal. 2003;326:185-189.
Masip J, Roque M, Sanchez B, et al. Noninvasive ventilation in acute cardiogenic pulmonary edema. Systematic review and meta-analysis. Journal of the American Medical Association. 2005;294:3124-3130.
Meecham Jones DJ, Paul EA, Jones PW, Wedzicha JA. Nasal pressure support ventilation plus oxygen compared with oxygen therapy alone in hypercapnic COPD. American Journal of Respiratory and Critical Care Medicine. 1995;152:538-544.
Mehta S, McCool FD, Hill NS. Leak compensation in positive pressure ventilators: a lung model. European Respiratory Journal. 2001;17:259-267.
Meyer TJ, Hill NS. Noninvasive positive pressure ventilation to treat respiratory failure. Annals of Internal Medicine. 1994;120:760-770.
Milross MA, Piper AJ, Norman M, et al. Low-flow oxygen and bilevel ventilatory support. Effects on ventilation during sleep in cystic fibrosis. American Journal of Respiratory and Critical Care Medicine. 2001;163:129-134.
Moran FM, Bradley JM, Elborn JS, Piper AJ. Physiotherapy involvement in non-invasive ventilation hospital services: a British Isles survey. Int J Clin Pract. 2005;59:453-456.
Nava S, Ambrosino N, Clini E, et al. Noninvasive mechanical ventilation in the weaning of patients with respiratory failure due to chronic obstructive pulmonary disease. Annals of Internal Medicine. 1998;128:721-728.
Nava S, Carbone G, DiBattista N, et al. Noninvasive ventilation in cardiogenic pulmonary edema. A multicenter randomized trial. American Journal of Respiratory and Critical Care Medicine. 2003;168:1432-1437.
Nava S, Evangelisti I, Rampulla C, et al. Human and financial costs of noninvasive mechanical ventilation in patients affected by COPD and acute respiratory failure. Chest. 1997;111:1631-1638.
Nava S, Gregoretti C, Fanfulla F, et al. Noninvasive ventilation to prevent respiratory failure after extubation in high-risk patients. Critical Care Medicine. 2005;33:2465-2470.
Navalesi P, Fanfulla F, Frigerio P, Gregoretti C, Nava S. Physiologic evaluation of noninvasive mechanical ventilation delivered with three types of masks in patients with chronic hypercapnic respiratory failure. Critical Care Medicine. 2000;28:1785-1790.
Nickol AH, Hart N, Hopkinson NS, et al. Mechanisms of improvement of respiratory failure in patients with restrictive thoracic disease treated with non-invasive ventilation. Thorax. 2005;60:754-760.
Norrenberg M, Vincent JL. A profile of European intensive care unit physiotherapists. Intensive Care Medicine. 2000;26:988-994.
Padman R, Lawless ST, Kettrick RG. Noninvasive ventilation via bilevel positive airway pressure support in pediatric practice. Critical Care Medicine. 1998;26:169-173.
Perez de Llano LA, Golpe R, Ortiz Piquer M, et al. Short-term and long-term effects of nasal intermittent positive pressure ventilation in patients with obesity-hypoventilation syndrome. Chest. 2005;128:587-594.
Peter JV, Moran JL, Phillips-Hughes J, Graham P, Bersten AD. Effect of non-invasive positive pressure ventilation (NIPPV) on mortality inpatients with acute cardiogenic pulmonary oedema: a meta-analysis. Lancet. 2006;367:1155-1163.
Piper AJ, Moran FM. Non-invasive ventilation and the physiotherapist: current state and future trends. Phys Ther Rev. 2006;11:37-43.
Piper AJ, Sullivan CE. Sleep breathing in neuromuscular disease. In: Saunders N, Sullivan CE, editors. Sleep and breathing. 2nd edn. New York: Marcel Dekker; 1994:761-821.
Piper AJ, Sullivan CE. Effects of short-term NIPPV in the treatment of patients with severe obstructive sleep apnea and hypercapnia. Chest. 1994;105:434-440.
Piper AJ, Parker S, Torzillo PJ, Sullivan CE, Bye PTP. Nocturnal nasal IPPV stabilizes patients with cystic fibrosis and hypercapnic respiratory failure. Chest. 1992;102:846-850.
Piper AJ, Willson G. Nocturnal nasal ventilatory support in the management of daytime hypercapnic respiratory failure. Australian Journal of Physiotherapy. 1996;42(1):17-29.
Plant PK, Owen JL, Elliott MW. Early use of non-invasive ventilation for acute exacerbations of chronic obstructive pulmonary disease on general respiratory wards: a multicentre randomized controlled trial. Lancet. 2000;355:1931-1935.
Plant PK, Owen JL, Elliott MW. Non-invasive ventilation in acute exacerbations of chronic obstructive pulmonary disease: long term survival and predictors of in-hospital outcome. Thorax. 2001;56:708-712.
Plant PK, Owen JL, Parrott S, Elliott MW. Cost effectiveness of ward based non-invasive ventilation for acute exacerbations of chronic obstructive pulmonary disease: economic analysis of randomized controlled trial. British Medical Journal. 2003;326:956-960.
Polkey MI, Lyall RA, Davidson AC, Leigh PN, Moxham J. Ethical and clinical issues in the use of home non-invasive mechanical ventilation for the palliation of breathlessness in motor neurone disease. Thorax. 1999;54:367-371.
Pollack CV, Fleisch KB, Dowsey K. Treatment of acute bronchospasm with beta-adrenergic agonist aerosols delivered by a nasal bilevel positive airway pressure circuit. Annals of Emergency Medicine. 1995;26:552-557.
Raphael JC, Chevret S, Chastang C, Bouvet F. Randomized trial of preventive nasal ventilation in Duchenne muscular dystrophy. Lancet. 1994;343:1600-1603.
Ragette R, Mellies U, Schwake C, Teschler H. Patterns and predictors of sleep disordered breathing in primary myopathies. Thorax. 2002;57:724-728.
Ram FS, Wellington S, Towe B, Wedzicha JA. Non-invasive positive pressure ventilation for treatment of respiratory failure due to severe acute exacerbations of asthma. Cochrane Database of Systematic Reviews. 3, 2005. : CD004360
Restrick LJ, Scott AD, Ward EM, et al. Nasal intermittent positive-pressure ventilation in weaning intubated patients with chronic respiratory failure from assisted intermittent, positive-pressure ventilation. Respiratory Medicine. 1993;87:199-204.
Richards GN, Cistulli PA, Ungar G, Berthon-Jones M, Sullivan CE. Mouth leak with nasal continuous positive airway pressure increases nasal airway resistance. American Journal of Respiratory and Critical Care Medicine. 1996;154:182-186.
Saatci E, Miller D, Stell IM, Lee KC, Moxham J. Dynamic dead space in face masks used with noninvasive ventilators: a lung model study. European Respiratory Journal. 2004;23:129-135.
Schettino G, Altobelli N, Kacmarek RM. Noninvasive positive pressure ventilation reverses acute respiratory failure in select ‘do-not-intubate’ patients. Critical Care Medicine. 2005;33:1976-1982.
Schettino GPP, Chatmongkolchart S, Hess DR, Kacmarek RM. Position of exhalation port and mask design affect CO2 rebreathing during noninvasive positive pressure ventilation. Critical Care Medicine. 2003;31:2178-2182.
Schonhofer B, Sonneborn M, Haidl P, Bohrer H, Kohler D. Comparison of two different modes for noninvasive mechanical ventilation: volume vs pressure controlled device. European Respiratory Journal. 1997;10:184-191.
Schonhofer B, Wallstein S, Wiese C, Kohler D. Non-invasive mechanical ventilation improves endurance performance in patients with chronic respiratory failure due to thoracic restriction. Chest. 2001;119:1371-1378.
Simonds AK. Pneumothorax: an important complication of non-invasive ventilation in neuromuscular disease. Neuromuscular Disorders. 2004;14:351-352.
Simonds AK, Elliott MW. Outcome of domiciliary nasal intermittent positive pressure ventilation in restrictive and obstructive disorders. Thorax. 1995;50:604-609.
Simonds AK, Muntoni F, Heather S, Fielding S. Impact of nasal ventilation on survival in hypercapnic Duchenne muscular dystrophy. Thorax. 1998;53:949-952.
Simonds AK, Ward S, Heather S, Bush A, Muntoni F. Outcome of paediatric domiciliary mask ventilation in neuromuscular and skeletal disease. European Respiratory Journal. 2000;16:476-481.
Sullivan CE, Berthon-Jones M, Issa FG, Eves L. Reversal of obstructive sleep apnea by continuous positive airway pressure applied through the nose. Lancet. 1981;1:862-865.
Teague WG. Non-invasive positive pressure ventilation: current status in paediatric patients. Paediatric Respiratory Review. 2005;6:52-60.
Teschler H, Stampa J, Ragette R, Konietzko N, Berthon-Jones M. Effect of mouth leak on effectiveness of nasal bilevel ventilatory assistance and sleep architecture. European Respiratory Journal. 1999;14:1251-1257.
Trask CH, Cree EM. Oximeter studies on patients with chronic obstructive emphysema, awake and during sleep. New England Journal of Medicine. 1962;266:639-642.
Tuggey JM, Elliott MW. Randomised crossover study of pressure and volume non-invasive ventilation in chest wall deformity. Thorax. 2005;60:859-864.
van’t Hul A, Kwakkel G, Gosselink R. The acute effect of non-invasive ventilatory support during exercise on exercise endurance and dyspnea in patients with chronic obstructive pulmonary disease. A systematic review. J Cardiopulm Rehab. 2002;23:307-313.
Vitacca M, Nava S, Confalonieri M, et al. The appropriate setting of noninvasive pressure support ventilation in stable COPD patients. Chest. 2000;118:1286-1293.
Ward S, Chatwin M, Heather S, Simonds AK. Randomised controlled trial of non-invasive ventilation (NIV) for nocturnal hypoventilation in neuromuscular and chest wall disease patients with daytime normocapnia. Thorax. 2005;60:1019-1024.
Waters KA, Everett FM, Bruderer JW, et al. Obstructive sleep apnea: the use of nasal CPAP in 80 children. American Journal of Respiratory and Critical Care Medicine. 1995;152:780-785.
Weirs PWJ, LeCoultre R, Dallinga OT, et al. Cuirass respirator treatment of chronic respiratory failure in scoliotic patients. Thorax. 1977;32:221-228.
Windisch W, Kostic S, Dreher M, Virchow JC, Sorichter S. Outcome of patients with stable COPD receiving controlled noninvasive positive pressure ventilation aimed at a maximal reduction of PaCO2. Chest. 2005;128:657-662.
Wood KE, Flaten AL, Backes WJ. Inspissated secretions: a life-threatening complication of prolonged non-invasive ventilation. Respiratory Care. 2000;45:491-493.
Woollam CHM. The development of apparatus for intermittent negative pressure respiration. Anaesthesia. 1976;31:537-547.