Chapter 9 Companion animal health and welfare
This chapter introduces the general principles of animal health and welfare, and then continues to apply these to dogs and cats specifically. The impacts of the five needs for the average pet owner are considered to promote responsible pet ownership. Routine prophylactic measures that should be employed in companion animal species are described and the pathogens they often serve to prevent are discussed.
BUYING AND CHOOSING A PET
For many people buying a pet happens as a result of a ‘spur of the moment’ decision. Pets can become ideal companions or your worst nightmare. Pet owners have a legal responsibility to their pet to keep it in good health and in suitable conditions; in the UK this is governed by the Animal Welfare Act 2006.
The Animal Welfare Act 2006 states that all animal owners have a legal ‘duty of care’ to ensure the welfare of their pet. It identifies the five freedoms or key needs of every animal:
While many pet owners already provide for these needs, anyone who fails to do so could be liable for a fine or even a prison sentence. The duty of care can be applied to the owner of the animal, any person in charge of the animal in lieu of the owner, e.g. veterinary surgeon or boarding kennels, and a parent or guardian if the owner is under 16 years of age and legally not an adult.
The five key needs provide legal definitions which quantify animal health and welfare not just for companion animal species but for all farm animals and wild animals kept in captivity. The achievement of some of the key needs, for example the need for a proper diet, can appear relatively clear to the pet owner whilst others require a more thorough understanding of the animal species, for example the need to express normal behaviour. The definition of the key needs has provided a forum for animal welfare organisations to police pet owners and animal keepers to enable animals to be seized and prosecution to occur if intent or neglect is intended without the need for suffering to have actually taken place. Tables 9.1 and 9.2 identify some of the factors prospective pet owners should consider when choosing a suitable pet.
Table 9.1 Factors to consider when choosing a pet
| Who is the pet for? | |
| Does the owner work? | |
| Is the owner disabled? | |
| Will there be contact with children? What are their ages? | |
| Who is going to be responsible for the pet? | |
| How much time will be dedicated to looking after the pet? | |
| How often do the owners go on holiday? Will the pet go with them? | |
| Where do the owners live? What type of house? | |
| What type of pet would be considered? |
Table 9.2 Factors to consider when assessing suitability of animals as family pets
| Family | |
| Working owners | |
| Financial implications | |
| Dogs | |
| Cats | |
| Rabbits | |
| Exotic species |
SPREAD OF INFECTION
Disease in animal species is the result of the infiltration of pathogens into the body systems. Pathogens are disease-causing microorganisms; the most common encountered in companion animal species are bacteria, viruses, fungi and protozoa. The animal body is adapted to fight infection from pathogens via a range of strategic defences (Box 9.1).
Box 9.1
DEFINITIONS
Carrier status
Animals may occasionally come into contact with a microorganism and not exhibit any clinical signs of the disease – these animals are termed carriers.
Carriers are important in veterinary medicine because even though they show no clinical signs they may still shed/excrete the microorganism and infect other animals. There are two types of carrier: healthy carriers and convalescent carriers. A healthy carrier is an animal that has been exposed to an infectious disease but has never shown any clinical signs. Healthy carriers will carry the microorganism and shed it into the environment, posing a potential health risk, e.g. Haemobartonella felis and Campylobactor. Convalescent carriers are animals that have recovered from a clinical disease; these animals may shed large quantities of microorganism for variable time periods after recovery, e.g. leptospirosis.
Routes of transmission
Animals do not magically become infected by microorganisms. The microorganism has to find a way into the animal’s body and a multitude of methods are utilised. When considering how a microorganism has passed to an animal you have to establish:
Routes by which a microorganism may leave an animal include the following:
Routes of transmission from one animal to another include:
Direct contact involves actual physical contact from an infected animal with an uninfected one, usually via body secretions or parasites. Indirect contact involves spread from one animal to another one via other objects, known as fomites, e.g. food bowls and bedding, or spread from one animal to another through other animals, known as vectors, e.g. mice, fleas and sheep.
Routes of entry into a new host include:
Ingestion
The infectious microorganism is taken into the body. The level of clinical disease depends on the quantity of microorganism ingested and the health status of the animal.
Inhalation
The number of air changes in animal accommodation can also affect the numbers of pathogens present in the environment.
Through the skin
The skin is one of the body’s defence mechanisms to prevent pathogenic infections penetrating the skin.
Methods used to control infection
The study of animal diseases has provided clues on how to prevent them spreading. These include:
Routine hygiene
The use of safe but effective disinfectant products combined with strict hygiene controls is essential to reduce the risk of infection. Schedules of work for any animal establishment should detail what is to be cleaned, the order of cleaning (cleanest to dirtiest to prevent contamination) and a record of cleaning. The disinfection of housing, bedding, feeding utensils and equipment, use of disinfectants at the correct dilution for the appropriate contact times, good practice between animals by staff washing their hands or using alcohol gels to reduce spread of infections and isolation protocols can reduce risk.
Isolation/quarantine
The availability of an isolation facility can aid in the control of infectious disease outbreaks and provides a relatively safe environment for the assessment of animals whose previous infection status is not known. Quarantine enables the isolation of potential disease-harbouring animals, thus reducing the risk of introducing a disease into a population. Strict controls are required to manage the facility, including:
What happens when an animal is exposed to a disease?
The animal body has a range of non-specific defence mechanisms that aim to prevent infection or combat the initial invasion. These include:
These are complemented by specific defence mechanisms; these are mechanisms that are activated within the body in response to the entry of a pathogen. They are collectively known as the immune system and involve the lymphatic and circulatory systems. There are different types of immune response and these vary depending on:
An antigen is a foreign protein that initiates an immune response. Antibodies are complex proteins produced by B lymphocytes. Specific antibodies are produced for specific diseases and their role is to destroy or inactivate specific antigens.
T lymphocytes play an active role in immunity and have the ability to destroy virus-infected cells and tumours without antibodies. The immune system begins to act the instant a foreign pathogen invades the body but it takes time for the levels of T and B lymphocytes to build up to sufficient numbers to be effective. During this time the animal will feel unwell and demonstrate the clinical signs of the disease.
The body has special memory cells that remember which diseases the body has encountered in the past and if that disease invades again the antibody response is triggered by these memory cells. Memory cells can remember some diseases for a lifetime, e.g. smallpox, but others need to be regularly updated by booster vaccination.
Vaccination
Vaccination introduces specially treated non-infective versions of diseases to initiate an immune response to achieve antibodies and give memory cells an encounter with the disease. Vaccination does not begin in very young animals because there are two types of immunity. Active immunity occurs due to stimulation of T and B lymphocytes whereas passive immunity is a short-lived immunity which can be transferred from dam to offspring or by an injection of antibodies. There are many different brands of vaccine available and each has a specific protocol that should be adhered to.
PROMOTION OF HEALTH
A number of generic factors can be considered by the informed observer regardless of the animal species; refer to Tables 9.3 and 9.4. The evaluation of the specific animal and the observer’s responses can provide essential clues to animal health, particularly in wild species housed in captivity where comprehensive health checks are prohibitive for safety reasons. The physical and clinical parameters for individual animals can be considered to provide a picture of the animal’s health status, but the environment the animal has had access to may directly influence health. Good husbandry is essential to health and knowledge of species and breed requirements is required by handlers and keepers. The five key needs must be met by the animal accommodation but their implementation will vary depending on the ultimate destination of the animal; for example, an intensively farmed pig would experience a very different environment than a free-range pig.
| Type | Description |
|---|---|
| Active | Active response by immune system |
| Naturally acquired | Active response due to natural exposure, e.g. infection |
| Artificially acquired | Active response due to artificial exposure, e.g. vaccination |
| Passive artificial | Ready-made antibodies, e.g. tetanus antitoxin injected |
| Passive natural | Antibodies cross the placenta or are present in milk and transmitted from dam to offspring |
| Innate | Certain species are immune to certain diseases |
Table 9.4 Factors that indicate health status
| Factor | Good health | Poor health |
|---|---|---|
| Appetite | Normal | |
| Skin/coat condition | ||
| Mobility | Normal gait | |
| Activity levels | Normal | |
| Urinary output | Normal | |
| Faecal output | Normal | |
| Behaviour | Normal interactions observed | |
| Injury | No signs | |
| Temperature | Normal range considering animal’s activity, e.g. if temperature is slightly increased after exercise, this would be considered normal | Outside normal range |
| Pulse rate | Normal range | |
| Heart rate | Normal range and heart sounds | Abnormal range, rhythm or heart sounds |
| Respiratory rate | Normal range | Abnormal range, pattern or noise |
| Inflammation/localised heat | None | Presence suggests injury or infection |
| Sleep patterns | Normal for activity level, environment | Abnormal – prolonged or shortened |
| Social interaction | Normal for species | |
| Discharges | None observed | Presence of discharges, e.g. ocular, aural |
| Environment | ||
| Vocalisation | Normal | |
| Capillary refill time | 1–2 seconds | 3+ seconds |
| Mucous membrane colour | Salmon pink | Brick red – poisoning, cyanotic (blue), grey, white – all may be due to reduced oxygen Petechia – pinpoint haemorrhage |
All handlers and keepers should be trained in systems to ensure consistency of practice; this can be achieved by utilising schedules of work for common tasks, staff training, mentor schemes and monitoring the environment. Careful consideration of construction materials, the order of work, products used for disinfection, equipment employed, sharing of utensils and food bowls can improve or reduce the probability of cross-contamination. Establishing strict hygiene controls in association with a quarantine or isolation protocol if disease is suspected and for new arrivals should reduce the risk of infection spreading. Prophylactic measures should be employed via vaccination, routine parasite control and regular health checks to promote health.
Disinfection
Disinfection can be defined as the destruction or reduction of microorganisms that are pathogenic, not including bacterial spores. Transient bacteria numbers will be affected. Disinfectants are classified into different compound groups, each of which is developed for a specific use:
Using a disinfectant can still be ineffectual if you do not follow the core steps listed below or ignore the manufacturer’s instructions.
Inactivation of disinfectants
Disinfectant compounds vary in the extent their efficiency can be compromised by external factors. The presence of organic material, mixing products, the addition of detergent, hard water, dilution rate, contact time, temperature and presence of bacteria can all inactivate specific products and it is essential that the manufacturer’s instructions and datasheet should be meticulously followed to ensure the product used is effective.
How can you categorise the risk?
Any indidivual who is responsible for cleaning and reducing the risks of cross-contamination and infection within the animal environment should be able to identify and categorise risk areas within the business and select suitable products and suitable cleaning regimes.
INFECTIOUS DISEASES OF COMPANION ANIMAL SPECIES
An infectious disease is defined as a disease that is capable of being passed from one animal (it may be restricted to one species) to another and that can be described as contagious (Box 9.2).
Box 9.2
DEFINITIONS
Feline infectious diseases
There are numerous feline infectious diseases that should be considered, including:
Feline infectious respiratory disease
This is a viral respiratory disease, of which there are two types: feline calicivirus and feline rhinotracheitis virus or feline herpesvirus (FHV). The condition can also be known as feline upper respiratory tract disease, feline influenza or cat flu.
Feline rhinotracheitis
Aetiology
Pathogenesis and pathology
Feline panleukopenia
This is also known as feline infectious enteritis and also affects cats, mink, ferrets and racoons. It is characterised by a decrease in white blood cells or a panleukopenia and the destruction of intestinal mucosa or enteritis.
Feline coronavirus
This virus predominantly affects young cats. It has intrauterine transmittal and has a variable incubation period. There are two types:
Pathogenesis
Feline leukaemia
This disease is often seen in the younger members of multicat households and in cats that have direct contact with other felines as it is spread by direct contact.
Pathogenesis
Feline immunodeficiency virus
This disease is most common in entire cats aged between 5 and 9 years which are allowed to roam.
Canine infectious diseases
There are a number of canine infectious diseases that should be considered, including:
Canine parvovirus
Pathogenesis
Canine distemper
Pathogenesis
Clinical signs
Canine leptospirosis
Aetiology
Pathogenesis
Infectious canine hepatitis
Pathogenesis
Canine infectious tracheobronchitis (kennel cough)
Leishmaniasis
Pathogenesis
Giardia
Pathogenesis
Equine infectious diseases
Equine herpesvirus (EHV)
Aetiology
Equine influenza
Aetiology
Pathogenesis
Clinical signs
Equine viral arteritis (EVA)
Pathogenesis
Tetanus
Clinical signs
Streptococcus equi (strangles)
Strangles is the most common bacterial disease in horses; it is highly contagious and can spread through stables rapidly. Horses present depressed and dull, and stop eating. They exhibit pyrexia and the lymph nodes around the throat swell, forming abscesses. The horse can have difficulty breathing and swallowing (hence the name ‘strangles’). A nasal discharge is at first clear and then becomes purulent after the abscesses have ruptured in the nasal passages; abscesses may be lanced. Abscesses that rupture shed highly infective pus into the environment, which can infect other horses. In some outbreaks and in up to 10% of cases, these abscesses spread to other parts of the body (a condition known as ‘bastard strangles’), which is nearly always fatal. Diagnosis is via nasopharyngeal swab. Treatment is supportive therapy and strict isolation and hygiene controls in the yard.
In the USA and Canada a number of other equine infectious diseases are considered, including:
Rabbit infectious diseases
Respiratory disease
Pathogenesis
Viral haemorrhagic disease (VHD)
Pathogenesis
All species: infectious diseases
Salmonellosis
Pathogenesis
Rabies
Pathogenesis
Parasites
A parasite is defined as a plant or an animal that lives on or in another, usually larger, host organism in a way that harms or is of no advantage to the host.
Ectoparasites are parasites that live on or in the body surface, i.e. the skin. There are two categories:
Endoparasites are parasites that live inside the body, usually in the gastrointestinal tract, respiratory tract or heart.
Parasiticides are products that kill parasites; care should be taken as they will only kill if administered at the correct dose rate, and if the manufacturer’s instructions are followed.
Ectoparasites
Most ectoparasites are of the phylum Arthropoda. They include fleas, lice, mites, ticks, chiggers and flies. There are many treatments available and control can be challenging, as the animal itself, its environment and the animals it has interacted with require consideration. It is recommended to consult a veterinary surgeon before choosing a suitable product as many commercial variations are available.
Fleas: Ctenophalides canis, C. felis
Both dogs and cats are commonly infested with cat fleas, Ctenophalides felis (Figure 9.1). Fleas are not host-specific and prefer to inhabit a warm and humid environment akin to many human households. Fleas feed on blood, injecting an anticoagulant into the animal to prevent clotting occuring during feeding, and it is this substance which causes pruritus and allergic responses in animals, e.g. miliary dermatitis in cats and flea-allergic dermatitis in dogs. Signs of infestation include pruritus, loss of coat condition and flea dirt in the coat. In young and immunosuppressed animals a heavy flea burden can result in severe anaemia due to excessive blood loss. Flea dirt is flea faeces and a simple test is to use a fine-toothed comb to collect coat debris then place it on damp paper and the dirt will make the paper a red/brown colour as it comprises digested blood.
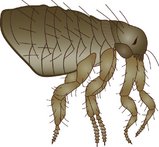
(Reproduced from Aspinall V 2006 The Complete Textbook of Veterinary Nursing. Butterworth Heinemann, London, with permission.)
The flea lifecycle can be completed in favourable conditions in as little as 12–16 days; adult fleas lay eggs in the fur of animals; they are not sticky so fall off the coat into the environment. In houses with carpeted surfaces these provide an ideal environment for the flea eggs to hatch. The eggs hatch into larval stages which feed on adult flea dirt, skin flakes and organic debris in their environment; they undergo two moults before they pupate and then emerge as adults. Fleas act as an intermediate host for numerous diseases and parasites, including Dipyldium caninum. Affected animals should be treated with insecticide and their environment should be treated concurrently, placing special attention on moving furniture and not forgetting all areas they inhabit, e.g. the car. Flea eggs can remain dormant for up to 2 years in the environment until movement vibrations from animals or humans stimulate them to hatch.
Ticks: Ixodes ricinus, I. hexagonas, Dermacentor reticulatus, Rhipicephalus sanguineus
There are many different species of tick and all species feed on blood from a host animal. The larval and adult ticks feed until engorged and can then drop off into the environment; females lay their eggs on the ground in a moist location before they die. A heavy infestation can result in anaemia and incorrect removal may leave the mouthparts in situ, and these mouthparts could become infected. Ticks are also important vectors for babesiosis. Treatment is insecticide and ticks themselves should be removed using a proprietary tick removal device.
Lice
Lice are host-specific, possess claws to attach themselves to their host and feed on blood. Lice can be described as biting or sucking depending on whether their mouthparts have evolved to chew the skin to feed or to pierce the skin to enable them to suck. Lice eggs are known as nits and are sticky, enabling them to attach to the coat hairs and complete their lifecycle on the host.
The biting louse of the dog is Trichodectes canis and in the cat is Felicola subrostratus. Dogs also have a sucking louse, Linognathus setosus. In horses the sucking louse is Haematopinus asini and the biting louse Damalinia equi.
Animals with a lice infestation are described as having a pediculosis and exhibit pruritus; their coat condition is poor and nits or lice may be observed. Anaemia can result and young, old and debilitated animals are most affected. Treatment is a suitable insecticide and, as the nits are not usually killed, treatment should be repeated 10 days after the first application.
Mites: subsurface and surface mites
Subsurface mites live in the dermis and present with short legs whilst surface mites live on the epidermis and tend to have longer legs.
Ear mites:
Otodectes cynotis
Otodectes cynotis are surface mites that possess suckers and hairs to grasp the walls of the ear canal (Figure 9.2). They present on examination as off-white dots on ear wax; infected animals will shake, scratch and rub their head and their ears will be excessively waxy and often smell. They are spread by direct contact and treatment by acaricidal product is required.
Chorioptes equi
A similar surface mite to Otodectes which infects horses, this mite infects the skin, particularly the feathers, in heavy breeds of horse.
Psoroptes equi/P. cuniculi
These are two closely related surface mites which look identical and have suckers on stalks; in the rabbit P. cuniculi causes ear infections and in horses P. equi produces a pruritic dermatitis.
Cheyletiella spp.
These surface mites affect dogs, cats and rabbits; each has its own specific mite. They are relatively large and can just be seen with the naked eye. They cause a scurfy, flaky coat and are known as ‘walking dandruff’ as the mites are often found under skin debris. They are capable of surviving away from the host so both the animal and its environment should be treated.
Harvest mites:
Neotrombiculus autumnalis
Another surface mite, this is prevalent in late summer/early autumn and will feed then drop off infected animals. They are an orange/red colour and may result in pruritus and dermatitis.
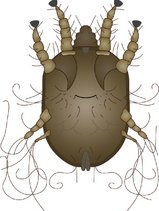
Sarcoptes scabeii
Sarcoptes scabeii (Figure 9.3) is a subsurface mite that is zoonotic, although it only survives for up to 3 weeks in humans. It affects dog, foxes and sometimes horses, cats and rabbits and is commonly known as mange. The mite lays its eggs in burrows it creates in the skin and it causes an erythematous, alopecic dermatitis characteristically of the pinna, hock and elbow and which expands to cover the animal. It is highly pruritic. Treatment is via application of insecticide.
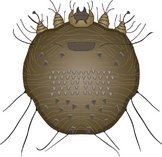
Demodex spp.
Demodex (Figure 9.4) is a characteristically cigar-shaped subsurface mite which is thought to be normally present with no ill effects in healthy animals. It can overpopulate at times of stress or immunosuppression to produce a widespread alopecic dermatitis with secondary bacterial infections common. Treatment is by acaricide.
Endoparasites
Endoparasites can be broadly divided into four main groups:
Nematodes
Nematodes are known as roundworms as they are cylindrical in cross-section and look a little like spaghetti. Ascarids are large roundworms found in the intestines of the horse, dog and cat. Ascarids include:
Toxocara canis
Eggs from infected animals are passed in the faeces and are ingested from the environment or via eating animals which are already infected. The larvae migrate through the liver and lungs to the gastrointestinal tract to mature to adult worms (Figure 9.5); some larvae remain in somatic cells and are stimulated during pregnancy to migrate transplacentally to the developing fetuses. They can also migrate in a transmammary route after birth, which is is why it is important to treat puppies regularly with an appropriate endoparasiticide. Heavy burdens can result in loss of condition and an enlarged abdomen or ‘worm belly’ and may result in intussusception or intestinal obstruction. T. canis are zoonotic and in humans develop in the eye and viscera, which can result in ocular and liver disease.
Toxocara cati
The lifecycle is similar to that of T. canis but no prenatal migration occurs in cats, with infection via the transmammary route in kittens. Again there is zoonotic potential.
Toxocara leonina
This may affect dogs and cats and is zoonotic; infection occurs by ingesting eggs or paratenic hosts (intermediate hosts that are not needed in the lifecycle).
Parascaris equorum
These are usually only problematic for foals and younger horses. Eggs can remain viable in the soil for many years and horses usually ingest them as they graze. The eggs hatch in the gastrointestinal tract and the larvae migrate into the veins, then move to the lungs where they are coughed up, reswallowed and passed into the small intestine. Once in the small intestine the larvae develop into adult roundworms. The adults, which can grow up to about 50 cm long, lay eggs which are passed out in the faeces. Prevention by suitable anthelminthic is required as well as pasture management.
Hookworms
Hookworms are named after the appearance of their mouthparts. They are parasites of the small intestine with numerous species affecting a range of animals. Eggs hatch in the environment and develop into larvae, which are ingested or may enter through the skin.
Uncinaria stenocephala
This is endemic in the UK in dogs. Larvae are either ingested or can penetrate the skin: animals living in a kennel environment are most susceptible. Dogs present with diarrhoea.
Ancyclostoma caninum
This is endemic in the EU and occasionally in the UK. The larvae thrive in warm environments and infection is by ingestion or skin penetration; transmammary infection can occur. Infected animals often exhibit melaena due to the presence of undigested blood in the faeces. It also occurs in foxes, coyotes, wolves and raccoons.
Whipworms
These are so called as their anterior end is thin and whip-like. The adults inhabit the intestine.
Trichuris vulpis
Canine, fox and coyote whipworm eggs are passed into the environment in faeces and require a temperate climate to develop; larvae are enclosed in a shell and may remain viable for up to a year until favourable conditions present. Heavy burdens can result in diarrhoea and metabolic disturbances.
Strongyloides spp.
Infections are common in young animals, with Strongyloides westeri affecting foals through transmammary migration of larvae or ingested from the environment. Larvae mature to adult worms in the small intestine. Infection can occur by eating infective larvae or by penetration through the skin. If the larvae enter the horse’s system through its skin, they move to the lungs, then up the windpipe where they are coughed up and swallowed. They mature in the small intestine, where adult females lay the eggs that are passed out in the manure.
If the larvae enter through the skin the next stop is the lungs, where they can cause bleeding and respiratory problems. Inflammation and rashes can develop where the larvae penetrated the skin. The worst damage occurs in untreated foals. Infected through their mother’s milk, they can suffer diarrhoea, weakness, weight loss and failure to thrive and grow at a normal rate. Dogs may also present with diarrhoea from Strongyloides infestation.
Strongyles
Equids can potentially be infected by a range of nematodes. Control by pasture management and regular anthelminthics is required.
Small redworms (cyathostomins)
These worms are the most common horse parasites. Adults living in the large intestine lay eggs that are passed out in the faeces. The worm eggs hatch and develop through three stages, with the third being the infective stage. If a grazing horse eats the infective stage the larvae will migrate to the intestinal lining, where they can remain dormant for long periods of time. Vast numbers of larvae can potentially build up in this area, then suddenly reactivate and erupt out of the gut wall, which can cause extensive damage to the gut wall. Larvae emerge into the large intestine and develop into adults which lay eggs that are passed out in the droppings, completing the cycle.
Large redworm (Strongylus vulgari, S. equinus and S. edentatus)
The large redworm differs from small redworms in size and lifecycle. Horses become infected by ingesting larvae as they are grazing. The larvae of large redworms migrate through the blood vessels to the arteries of the intestinal tract where they can cause severe damage. After about six months they return to the intestine as egg-laying adult worms. Adult worms vary in size between 1.5 and 5 cm and, in large numbers, can cause several different disease problems including clot formation in mesenteric arteries resulting in necrosis of the intestine and ulceration of intestinal tissue.
Pinworm (Oxyuris equi)
Pinworms are so called as they look like carpet tacks; they are not thought to be harmful but can provoke irritation around the tail. Pinworms inhabit the large and small colon and have a relatively simple lifecycle. The females lay their eggs around the anus of the horse, using a sticky substance which is irritating to the horse. The eggs are dislodged as droppings are passed and fall on to the pasture where they are eaten by horses.
Lungworms
Dictyocaulus arnfieldi
The equine lungworm is long and slender; the larvae go through the walls of the intestine and into the circulatory system. They are carried in the circulatory system into the lungs, where they mature. The eggs pass through the horse’s system through the manure. Female lungworms lay eggs containing larvae. The horse eats the eggs of the lungworm off damp grasses. If there are large numbers of the larvae present, the lungs may become irritated, causing the horse to have a severe cough, difficulty breathing and loss of appetite.
Infection is usually light in older horses because they develop resistance to the parasite and usually have no signs. If foals are infected, they could die from a lungworm infection because they have less immunity.
Heartworms
Heartworms are nematodes that colonise the heart. There are three canine heartworms of note: Angiostrongylus vasorum (which can also affect foxes), Dirofilaria immitis and Dipetalonema reconditum. D. immitis and D. reconditum are not endemic in the UK but are endemic in the USA and some parts of Europe. Prevention is essential for animals inhabiting or travelling in affected areas as surgical removal and anthelminthics can be used as treatment but both pose risks.
Cestodes
Cestodes are commonly known as tapeworms. They are flat in appearance and have a head or scolex and a chain of segments or proglottids. The tapeworm attaches to the intestinal wall and produces gravid segments full of eggs which move out of the animal via the anus. They resemble grains of rice and are sticky and mobile. Most have intermediate hosts and control is via an anthelminthic with cestodial action.
Anoplocephala perfoliata
This is an equine tapeworm which can grow to about 8 cm long and about 1.5 cm wide. It lives in the midpart of the gut at a junction between the small and large intestine, known as the ileocaecal junction. Infected horses pass eggs on to the pasture. These eggs are eaten by tiny oribatid mites which are present in their thousands in every square metre of grass. Once inside the mite, the eggs hatch and develop into an intermediate infective stage. Grazing horses inadvertently eat mites with almost every mouthful. The adults attach in clusters to the lining of the gut at the ileocaecal junction and release eggs, thereby completing their lifecycle; recent research implicates tapeworm infestation with certain types of colic.
Dipylidium caninum
This tapeworm affects both dogs and cats, with intermediate hosts being fleas and other biting lice. Infection usually occurs when an infected intermediate host is ingested and there are few signs of infestation unless present in large numbers.
Taenia spp.
There are a number of Taenia spp. that affect animals:
Infections occur when infected intermediate hosts are ingested.
Echinococcus granulosus
This is a very small tapeworm that affects dogs and foxes and huge numbers must be present before signs of infestation are observed. It occurs worldwide and it is zoonotic; if humans ingest eggs a hydrated cyst can develop in the lungs, requiring surgery or anthelminthic treatment. Infection in dogs usually occurs by eating raw offal of infected animals (sheep is the intermediate host).
Trematodes
Flukes or trematodes are found in the intestine, bile ducts, blood and lungs of domestic animals.
Common liver fluke: Fasciola hepatica
Adult flukes in the liver lay eggs in the bile, which carries them into the intestine. They leave in the host’s faeces. After hatching, the immature fluke must penetrate a snail for the lifecycle to continue. Multiplication occurs within particular species of snail. After leaving the snail the flukes encyst on grass where they are eaten by horses. The young parasites penetrate the gut and pass to the liver. The snails live in swampy, wet areas. F. hepatica occurs worldwide in wet areas where Lymnaea snails may exist. Animals experience anaemia and decreased growth. Liver damage results in organ condemnation at slaughter.
Protozoa
These are unicellular organisms which can be parasitic.
Coccidiosis
Isopora spp. are parasites of the gastrointestinal tract in dogs and cats; some animals tolerate them well whereas others will present with diarrhoea. Horses and rabbits may be infected by Eimeria spp., resulting in diarrhoea. Sarcocystis neurona produces a severe neural disease in horses – equine protozoal myeloencephalitis.
Appleby M. What should we do about animal welfare. Oxford: Blackwell Science; 1999.
Appleby M C, Hughes B O. Animal welfare. Oxford: CABI Publishing; 1997.
Aspinall V. The complete textbook of veterinary nursing. London: Butterworth Heinemann; 2006.
Blood D C, Studdert V P. Bailliere’s comprehensive veterinary dictionary. London: Baillière & Tindall; 1998.
Chandler E A, Gaskell C J, Gaskell R M. Feline medicine and therapeutics, 2nd edn. Oxford: Blackwell Sciences/BSAVA; 1994.
Dawkins M S. Animal suffering. The science of animal welfare. London: Chapman & Hall; 1980.
Garner R. Animals, politics and morality. Manchester: Manchester University Press; 1993.
Gaskell R M, Bennett M. Feline and canine infectious disease. Oxford: Blackwell Science; 1996.
Gorman N. Canine medicine and therapeutics, 4th edn. Oxford: Blackwell Science/BSAVA; 1998.
Lane D R, Cooper B, Turner L. BSAVA Textbook of Veterinary Nursing. Oxford: BSAVA; 2007.
Meredith A, Redrobe S. Manual of exotic pets, 4th edn. Gloucester: BSAVA; 2002.
Ramsey I, Tennant B. Manual of canine and feline infectious diseases. Oxford: BSAVA; 2001.
1998, Sainsbury D. 2nd edn. Oxford: Blackwell Science, 1998.
2000, Spedding C. London: Earthscan, 2000.
Torrance A G, Mooney C T. Manual of small animal, 2nd edn. Oxford: BSAVA; 1998.
Warren D M. Small animal care and management. New York: Delmar; 1995.