ENAMEL
After reading this chapter the student will be able to:
• describe the physical features of enamel such as the structure of the enamel rods, incremental lines, lamellae, tufts, and spindles
• discuss how these affect the permeability of enamel
• discuss the surface characteristics and the etching of enamel
OVERVIEW
Enamel, the hard protective substance that covers the crown of the tooth, is the hardest biologic tissue in the body. It consequently is able to resist fractures during the stress of mastication. Enamel provides shape and contour to the crowns of teeth and covers the part of the tooth that is exposed to the oral environment.
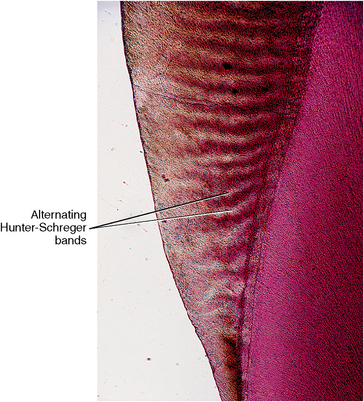
Enamel is composed of interlocking rods that resist masticatory forces. Enamel rods are deposited in a keyhole shape by the formative ameloblastic cells. Groups of ameloblasts migrate peripherally from the dentino-enamel junction as they form these rods. Ameloblasts take variable paths, which produces a bending of the rods. These cells maintain a relationship as they travel in different directions and produce adjacent rods. The enamel rod configuration viewed in incidental light appears as light and dark bands of rod groups termed Hunter-Schreger bands. Because these rods bend in an exaggerated, twisted manner at the cusp tips, they are called gnarled enamel.
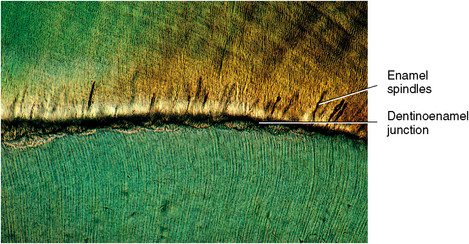
All enamel rods are deposited at a daily appositional rate or increment of 4 μm. Such increments are noticeable, like rings in a cross section of a tree, and appear as dark lines known as striae of Retzius or lines of Retzius. The growth lines become apparent on the surface of enamel as ridges, known as perikymata. Two structures are noticeable at the dentinoenamel junction: spindles, the termination of the dentinal tubules in enamel, and tufts, hypocalcified zones caused by the bending of adjacent groups of rods.
Because enamel is composed of bending rods, which, in turn, are composed of crystals, minute spaces or gaps exist where crystals did not form between rods. This feature causes enamel to be variable in its density and hardness. Therefore, some areas of enamel may be more prone to penetration by small particles. This characteristic leads to tooth destruction by dental caries. After enamel is completely formed, no more enamel can be deposited.
PHYSICAL PROPERTIES
Because enamel is very hard, it is also brittle and subject to fracture. Fracture is especially likely to occur if the underlying dentin is carious and has weakened the enamel’s foundation.
Enamel is about 96% inorganic mineral in the form of hydroxyapatite and 4% water and organic matter. Hydroxyapatite is a crystalline calcium phosphate that is also found in bone, dentin, and cementum. The organic component of enamel is the protein enamelin, which is similar to the protein keratin that is found in the skin. The distribution of enamelin between and on the crystals aids enamel permeability. Enamel is grayish white but appears slightly yellow because it is translucent and the underlying dentin is yellowish. Enamel ranges in thickness from a knifelike edge at its cervical margin to about 2.5 mm maximum thickness over the occlusal incisal surface.
ROD STRUCTURE
Enamel is composed of rods that extend from their site of origin, at the dentinoenamel junction, to the enamel outer surface (Fig. 7-1). Each rod is formed by four ameloblasts. One ameloblast forms the rod head; a part of two ameloblasts forms the neck; and the tail is formed by a fourth ameloblast. Figure 7-2 shows the six-sided design that is the shape of the ameloblast in contact with the forming keyhole- or racquet-shaped rod, which is columnar in its long axis. The head of the enamel rod is the broadest part at 5 μm wide, and the elongated thinner portion, or tail, is about 1 μm wide. The rod, including both head and tail, is 9 μm long. The enamel rod is about the same size as a red blood cell (Fig. 7-3).
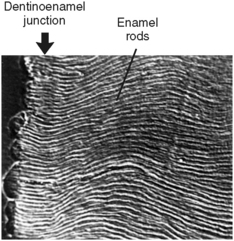
Fig. 7-1 Enamel rods appear wavy in section of enamel as they extend from the dentinoenamel junction on the left to the enamel surface on the right. This figure is possible because the section is etched and viewed with a scanning electron microscope. (From Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
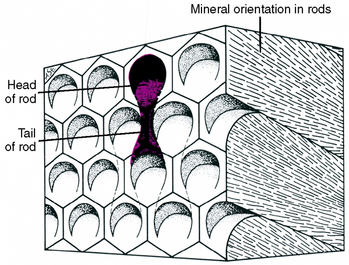
Fig. 7-2 Diagram showing outline of six-sided ameloblasts overlying keyhole-shaped enamel rods. Parts of four cells form each enamel rod. Crystal orientation of three rods can be seen on the right side of the model. (From Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
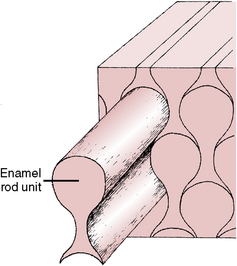
Fig. 7-3 One rod is pulled out to illustrate how individual enamel rods interdigitate with neighboring rods. (From Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
Each rod is filled with crystals. Those in the head follow the long axis of the rod, and those in the tail lie in the cross axis to the head (Figs. 7-4 and 7-5). The upper right rod head of Figure 7-4 indicates how the mineral is oriented during the rod’s development, which forms the rod head and tail as seen on the left side of the figure. The architecture of the mineral orientation is complex, especially when viewed in any direction other than cross section (see Fig. 7-5).
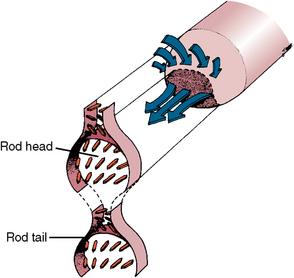
Fig. 7-4 The left side of diagram shows orientation of crystals in the forming rod head and tail. The right part shows how forming crystals pack in the rod from the cell complex (arrows).
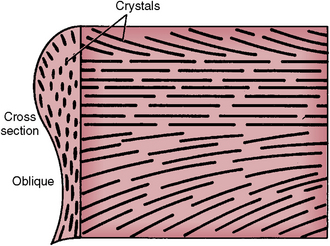
Fig. 7-5 Orientation of crystals in a mature enamel rod as indicated by cross section and side of a cut rod. (Modified from Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
Rods form nearly perpendicular to the dentinoenamel junction and curve slightly toward the cusp tip. This unique rod arrangement also undulates throughout the enamel to the surface. Each rod interdigitates with its neighbor, the head of one rod nestling against the necks of the rods to its left and right (see Fig. 7-3). The rods run almost perpendicular to the enamel surface at the cervical region but are gnarled and intertwined near the cusp tips (Fig. 7-6). The surface of each rod is known as the rod sheath, and the center is the core. The rod sheath contains slightly more organic matter than the rod core (Fig. 7-7).
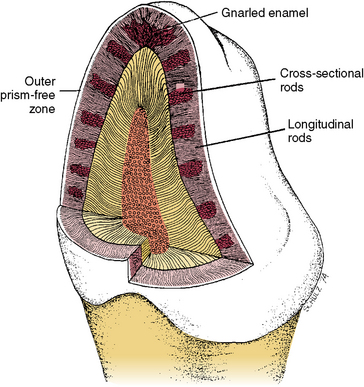
Fig. 7-6 Diagram of enamel rod orientation as shown in both longitudinal and cross section of the crown. Enamel rods are intertwined at the cusp tip; this is called gnarled enamel. Groups of outer enamel rods all run nearly perpendicular to the surface of the enamel, whereas inner groups of enamel rods alternate. Some appear in cross section while adjacent groups appear longitudinal. (Modified from Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
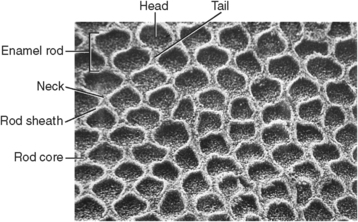
Fig. 7-7 Enamel rods in cross section. Each rod has a sheath and core. The rod sheath surrounds rod head and tail. This enamel sample has been etched to reveal organic matrix.
Groups of rods bend to the right or left at a slightly different angle than do adjacent groups (see Fig. 7-6). It is believed that this feature provides the enamel with strength for mastication and biting. When light is projected at the surface of a thin slab of enamel, light and dark bands appear. These bands are seen because the light transmits along the long axis of one group of rods but not along the adjacent rods, which lie at right angles. This is known as the Hunter-Schreger bands phenomenon (Fig. 7-8). These bands are named after the dental scientist who first noted the Schreger band effect microscopically. The repeating pattern from the cervical area to the incisal or occlusal areas can be seen along the long axis of the tooth. Hunter-Schreger bands extend through one half to two thirds of the thickness of enamel as shown in a diagram (see Fig. 7-6) and a tooth section (see Fig. 7-8).
INCREMENTAL LINES
The incremental lines in enamel are the result of the rhythmic recurrent deposition of the enamel. As the enamel matrix mineralizes, it follows the pattern of matrix deposition and provides the growth lines in enamel (Fig. 7-9). These lines may be accentuated because of a variation in the mineral deposited at the point of enamel hesitation in deposition. In some cases, the incremental lines are not visible. With enamel development, a row of ameloblasts covering the crown hesitates during deposition. These hesitation lines mark the path of amelogenesis. The spaces between the crystals entrap air molecules, accentuating these lines. Dr. Retzius, who first noted these “growth lines,” termed them the striae of Retzius.
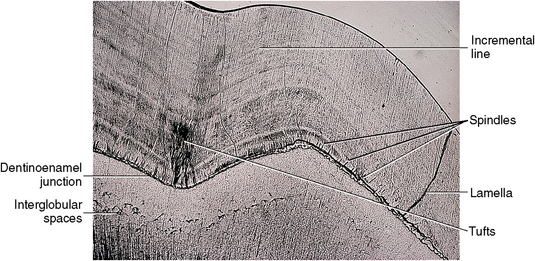
Fig. 7-9 Photomicrograph of dentinoenamel junction showing dentin below and enamel above this junction. Enamel exhibits incremental lines, tufts, spindles, and lamella. Within dentin, a band of primary dentin is just below the dentinoenamel junction. At the lower border of this band of primary dentin is a row of interglobular spaces.
Part of the enamel of most deciduous teeth is formed before birth and part after birth. Because environment and nutrition change abruptly at the time of birth, a notable line of Retzius occurs at that time. This is known as the neonatal line (Fig. 7-10). Although the neonatal line is an accentuated incremental line, it can be seen microscopically that this line is prominent for another reason. The enamel internal to this line is of a different consistency from that external to it because it was formed before birth, and the external was formed after birth. The prenatal enamel has fewer defects than the postnatal. The staining of the postnatal enamel has numerous minute spaces that are stained with pigment.
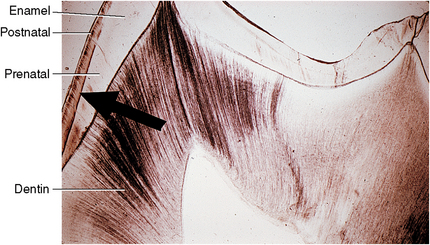
Fig. 7-10 Photomicrograph of section of enamel and dentin of primary tooth by transmitted light. The neonatal line is at the point of the large arrow. Enamel to the left of this line is a darker stain than enamel to the right of it. The enamel formed before birth is less pigmented and has fewer defects than postnatal enamel. Dentin exhibits numerous dead tracts as dark lines. Dead tracts are tubules filled with air; hence they appear black in transmitted light.
ENAMEL LAMELLAE
Enamel lamellae are cracks in the surface of enamel that are visible to the naked eye (see Figs. 7-9 and 7-11). Lamellae extend from the surface of enamel toward the dentinoenamel junction. Some lamellae form during enamel development, creating an organic pathway or tract. Spaces between groups of rods are another example of lamellae and may be caused by stress cracks that occur because of impact or temperature changes. Breathing cold air or drinking hot or cold beverages may cause small checks to occur in enamel, especially enamel weakened by underlying caries. Lamellae are not tubular defects but appear leaflike, extending around the crown (see Fig. 7-11). Lamellae are a possible avenue for dental caries.
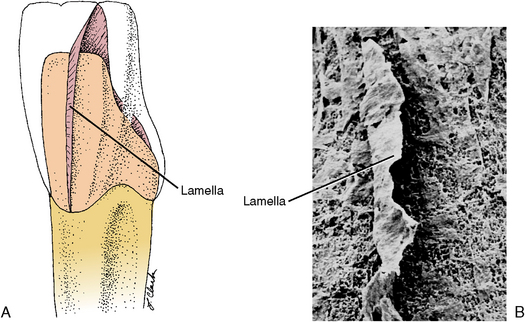
Fig. 7-11 Enamel lamellae. A, Diagram of possible location of leaflike enamel lamellae extending from the cervical to incisal enamel. B, Scanning electron micrograph of lamellae in enamel.(Enamel was decalcified away, and lamellar space was impregnated with resin for its maintenance.) (Modified from Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
ENAMEL TUFTS
Enamel tufts are another defect in enamel filled with organic material. They are located at the dentino-enamel junction and appear at right angles to it. They can extend one fifth to one tenth of the distance from the dentinoenamel junction to the outer surface of the tooth (Figs. 7-12 and 7-13). Tufts form between groups of enamel rods, which are oriented in slightly different directions at the dentinoenamel junction. These spaces are thus developed between adjacent groups of rods, which are filled with organic material termed enamelin. The interface of the junction of dentin and enamel is scalloped, and often tufts arise from these scalloped peaks (see Fig. 7-12).

Fig. 7-12 Transmitted light micrograph of the dentinoenamel junction area showing enamel tufts. In addition to tufts, scalloped dentinoenamel junction and fine enamel rod structure can be seen between tufts. Below the junction are dentinal tubules. (From Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
ENAMEL SPINDLES
Spindles arise at the dentinoenamel junction and extend into enamel. These spindles are extensions of dentinal tubules that pass through the junction into enamel (see Fig. 7-13). Because dentin forms before enamel, the odontoblastic process occasionally penetrates the junction, and enamel forms around this process, forming a tubule. These small tubules may contain a living process of the odontoblast, possibly contributing to the vitality of the dentinoenamel junction. Tubules are found singularly or in groups and are shorter than tufts, only a few millimeters in length. The fingerlike spindles appear quite different than the broader and longer tufts.
SURFACE CHARACTERISTICS
The enamel surface may be smooth or have fine ridges. Such ridges result from the termination of the striae of Retzius on the surface of enamel (Fig. 7-14). These surface manifestations are ridges called perikymata or imbrication lines. Perikymata are produced by the ends of rod groups accentuated by hesitation of ameloblasts before the next group of rods contact the enamel surface (Fig. 7-15). This manifestation is more prominent on the facial surface of the tooth, near the cervical region (see Fig. 7-14). Another feature of outer enamel near its surface is the zone of prismless enamel, which is 20 to 40 μm thick. Throughout this zone, no Schreger band effect is noted. This zone is not accentuated except near the cervical region and in deciduous teeth. The prismless zone of enamel is important because it appears as a structureless microcrystalline environment of enamel rods oriented nearly perpendicular to the enamel surface. This enhances the integrity of the enamel surface and should be recognized when a bevel for restorations is prepared.
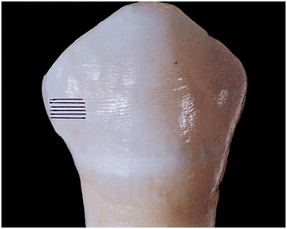
Fig. 7-14 Fine ridges on the enamel surface of the crown are perikymata or imbrication lines. (From Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
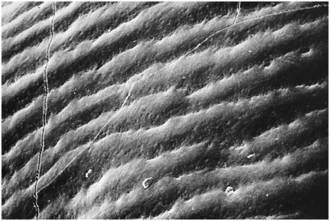
Fig. 7-15 Scanning electron micrograph of perikymata in Figure 7-14 at a much higher magnification, which shows alternating ridges and valleys. (From Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
PERMEABILITY
Enamel permeability is a feature of clinical importance. The passage of fluid, bacteria, and bacterial products through enamel is an important consideration in clinical therapy. Permeability of enamel is caused by several factors, some of which are evident as they relate to leakage around faulty restorations and decomposition of the tooth by dental caries. These latter examples need no further explanation, but fluid and fine particles can also pass through unbroken enamel by way of pathways described previously in this chapter, such as lamellae, cracks, tufts, and spindles. These all contribute to the microporosity of enamel. The minute spaces between or around enamel rods and through crystal spaces within rods are also important and are called microlamellae. Differences in crystal orientation can cause enamel to have minute spaces, which can be seen at high magnification (Fig. 7-16). Also, surface irregularities, such as those found in central fissures and near the cervical region, are important in influencing permeability.

Fig. 7-16 Transmission electron micrograph of a cross section of enamel rods that shows differences in rod sheath and rod core crystal orientation. (From Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical.) Thieme Medical
Enamel and dentin are both composed of hydroxyapatite crystals, although the crystals in enamel are about 30 times larger than those in dentin (Fig. 7-17). Crystal size is a factor in the extreme hardness of enamel in contrast to dentin.

Fig. 7-17 Dentinoenamel junction. Enamel is above and dentin below. Crystallites of enamel and dentin are different in size and orientation. Whereas crystals of human enamel may be 90 nm (900 Å) in width and 0.5 μm in length, those of dentin are only 3 nm (30 Å) in width and 100 nm (1,000 Å) in length. Crystals of dentin are similar in size to bone. (Electron micrograph × 35,000.)
ETCHING
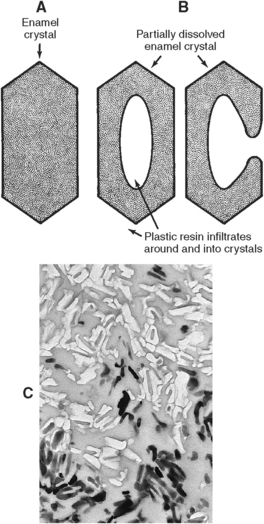
Etching with dilute acids, such as citric acid, may alter the surface of enamel. This dilute acid selectively etches the ends of the enamel rods and provides adherence of a plastic sealant to the surface of enamel rods (Fig. 7-18). The rod sheath resists demineralization to a greater extent than the rod core. The core of the crystal is rich in coronated apatite and is more sensitive to demineralization than the peripheral hydroxyapatite (Fig. 7-19). The purpose of this procedure is to produce an intact surface and thus prevent caries.
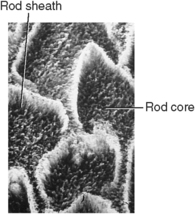
Fig. 7-18 Acid-etched enamel rod core dissolved to greater extent than rod sheath, which provides for attachment of sealant.
SELF-EVALUATION QUESTIONS
1. Describe the shape and size of the enamel rods.
2. Define Hunter-Schreger bands.
3. Define striae of Retzius. What is a synonym?
4. Describe gnarled enamel. Where is it located?
5. What are perikymata and imbrication lines?
6. What are the location and importance of tufts?
7. Define and give the cause of neonatal lines.
9. What is the inorganic component of enamel, dentin, and bone?
 CLINICAL COMMENT
CLINICAL COMMENT Consider the Patient
Consider the Patient