Positioning Principles
Evaluation Criteria
The goal of every technologist should be to take not just a “passable” radiograph but rather an optimal one that can be evaluated by a definable standard, as described under evaluation criteria.
An example of a four-part radiographic image evaluation as used in this text for a lateral forearm is shown on the right. The positioning photo and the resulting optimal radiograph (Figs. 1-99 and 1-100) are shown for this lateral forearm, as described in Chapter 4.
Evaluation Criteria Format
The technologist should review and compare radiographs using this standard to determine how close to an optimal image was achieved. A systematic method of learning how to critique radiographs is to break the evaluation down into these four parts.
1. Anatomy demonstrated: Describes precisely what anatomic parts and structures should be clearly visualized on that image (radiograph).
2. Position: Generally evaluates four issues: (1) placement of body part in relationship to the IR, (2) positioning factors that are important for the projection, (3) correct centering of anatomy, and (4) collimation
3. Exposure: Describes how exposure factors or technique (kilovoltage [kV], milliamperage [mA], and time) can be evaluated for optimum exposure for that body part. No motion is a first priority, and a description of how the presence or absence of motion can be determined is listed. (Motion is included with exposure criteria because exposure time is the primary controlling factor for motion.)
4. Image markers: A fourth area of evaluation involves image markers. Anatomic side markers, “Right” or “Left,” patient position, or time markers must be placed correctly before exposure so that they are not superimposed over essential anatomy.
Image Markers and Patient Identification
A minimum of two types of markers should be imprinted on every radiographic image. These are (1) patient identification and date and (2) anatomic side markers.
Patient Identification and Date (Film-Screen Cassette [ANALOG] Systems)
Generally, this patient information, which includes data such as name, date, case number, and institution, is provided on an index card and is photoflashed on the film in the space provided by a lead block in the film cassette. Each cassette or film holder should have a marker on the exterior indicating this area where the patient ID, including the date, will be flashed (Fig. 1-101).
Throughout this text, the preferred location of this patient ID marker is shown in relation to the body part. A general rule for chests and abdomens is to place the patient ID information at the top margin of the IR on chests and on the lower margin on abdomens (see arrows on Fig. 1-102). The patient ID marker must always be placed where it is least likely to cover essential anatomy. The anatomic side markers should always be placed in a manner on the IR so that they are legible and esthetically correct. It must be within the collimation field so that it provides a permanent indicator of correct side of the body or anatomic part.
Digital systems
With storage phosphor cassette–based systems, often a bar-code system imprints the patient information before or after exposure. Care must be taken so that this area does not obscure the essential anatomy that is being demonstrated. With flat panel detector with thin film transistor (FPD-TFT) systems and charged couple device (CCD) systems, patient identification is typically entered before exposure.
Anatomic Side Marker
A right or left marker must also appear on every radiographic image correctly indicating the patient's right or left side or which limb is being radiographed, the right or the left. This may be provided as the word “Right” or “Left” or just the initials “R” or “L.” This side marker preferably should be placed directly on the IR inside the lateral portion of the collimated border of the side being identified, with the placement such that the marker will not be superimposed over essential anatomy.
These radiopaque markers must be placed just within the collimation field so that they will be exposed by the x-ray beam and included on the image.
The two markers, the patient ID and the anatomic side marker, must be placed correctly on all radiographic images. Generally, it is an unacceptable practice to write or annotate digitally this information on the image after it is processed because of legal and liability problems caused by potential mismarkings. A radiograph taken without these two markers may have to be repeated, which results in unnecessary radiation to the patient, making this a serious error. In the case of digital images, annotating the image to indicate side markers is an unacceptable practice. The exposure should be repeated to ensure correct anatomy was imaged.
Additional Markers or Identification
Certain other markers or identifiers also may be used, such as technologist initials, which generally are placed on the R or L marker to identify the specific technologist responsible for the examination. Sometimes the examination room number is also included.
Time indicators are also commonly used; these note the minutes of elapsed time in a series, such as the 1-minute, 5-minute, 15-minute, and 20-minute series of radiographs taken in an intravenous urogram (IVU) procedure.
Another important marker on all decubitus positions is a decubitus marker or some type of indicator such as an arrow identifying which side is up. An “upright” or “erect” marker must also be used to identify erect chest or abdomen positions compared with recumbent, in addition to an arrow indicating which side is up.
Inspiration (INSP) and expiration (EXP) markers are used for special comparison PA projections of the chest. Internal (INT) and external (EXT) markers may be used for rotation projections, such as for the proximal humerus and shoulder. Sample markers are shown in Fig. 1-103.
Professional Ethics and Patient Care
The radiologic technologist is an important member of the health care team who is responsible in general for radiologic examination of patients. This includes being responsible for one's actions under a specific code of ethics.
Code of ethics describes the rules of acceptable conduct toward patients and other health care team members as well as personal actions and behaviors as defined within the profession. The ARRT code of ethics is provided in the box on this page.
Essential Projections
Routine Projections
Certain basic projections are listed and described in this text for each radiographic examination or procedure commonly performed throughout the United States and Canada. Routine projections are defined as projections commonly taken on patients who can cooperate fully. This varies depending on radiologist and department preference and on geographic differences.
Special Projections
In addition to routine projections, certain special projections are included for each examination or procedure described in this text. These are defined as projections most commonly taken to demonstrate better specific anatomic parts or certain pathologic conditions or projections that may be necessary for patients who cannot cooperate fully.
The authors recommend (on the basis of recent survey results) that all students learn and demonstrate proficiency for all essential projections as listed in this text. This includes all routine projections as well as all special projections as listed and described in each chapter. Examples of these routine projections and special projection boxes for Chapter 2 are shown. Becoming competent in these projections ensures that students are prepared to function as imaging technologists in any part of the United States.
General Principles for Determining Positioning Routines
Two general rules or principles are helpful for remembering and understanding the reasons that certain minimum projections are performed for various radiographic examinations.
Minimum of Two Projections (90° From Each Other)
The first general rule in diagnostic radiology suggests that a minimum of two projections taken as near to 90° from each other as possible are required for most radiographic procedures. Exceptions include an AP mobile (portable) chest, a single AP abdomen (called a KUB—kidneys, ureter, and bladder), and an AP of the pelvis, in which only one projection usually provides adequate information.
Three reasons for this general rule of a minimum of two projections are as follows:
2 Localization of lesions or foreign bodies
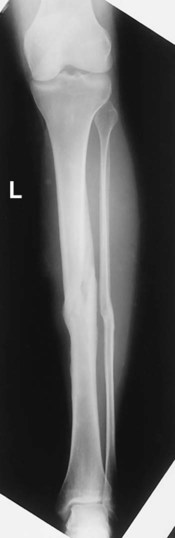
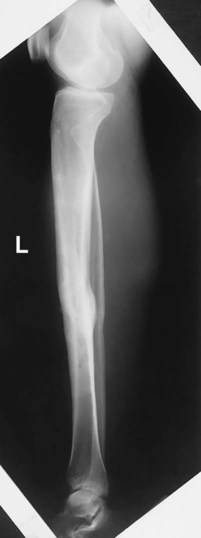
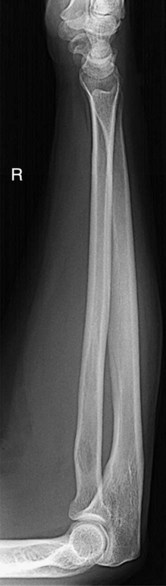
A minimum of two projections, taken at 90° or as near right angles from each other as possible, are essential in determining the location of any lesion or foreign body (Fig. 1-104).
3 Determination of alignment of fractures
All fractures require a minimum of two projections, taken at 90° or as near right angles as possible, both to visualize fully the fracture site and to determine alignment of the fractured parts (Figs. 1-105 and 1-106).
Minimum of Three Projections When Joints Are in Area of Interest
This second general rule or principle suggests that all radiographic procedures of the skeletal system involving joints require a minimum of three projections rather than only two. These are AP or PA, lateral, and oblique projections.
The reason for this rule is that more information is needed than can be provided on only two projections. For example, with multiple surfaces and angles of the bones making up the joint, a small oblique chip fracture or other abnormality within the joint space may not be visualized on either frontal or lateral views but may be well demonstrated in the oblique position.
Following are examples of examinations that generally require three projections as routine (joint is in prime interest area):
Examples of examinations that require two projections as routine include the following:
Exceptions to Rules
• Postreduction upper and lower limbs generally require only two projections for checking fracture alignment.
• A pelvis study requires only a single AP projection unless a hip injury is suspected.
Palpation of Topographic Positioning Landmarks
Radiographic positioning requires the location of specific structures or organs within the body, many of which are not visible to the eye from the exterior. Therefore, the technologist must rely on bony landmarks to indicate their location. These bony structures are referred to as topographic landmarks. Fig. 1-110 shows examples of topographic landmarks of the pelvis. Topographic landmarks can be located by a process referred to as palpation.
Palpation refers to the process of applying light pressure with the fingertips directly on the patient to locate positioning landmarks. This must be done gently because the area being palpated may be painful or sensitive for the patient. Also, the patient should always be informed of the purpose of this palpation before this process is begun, and patient permission should be obtained.
NOTE: Palpation of certain of these landmarks, such as the ischial tuberosity or the symphysis pubis, may be embarrassing for the patient and may not be permitted by institutional policy. Technologists should use other related landmarks as described in later chapters.
Viewing Radiographic Images
The manner in which PA and AP projection radiographic images are placed for viewing depends on the radiologist's preference and the most common practice in that part of the United States. However, in the United States and Canada, a common and accepted way to place radiographic images for viewing is to display them so that the patient is facing the viewer, with the patient in the anatomic position. This always places the patient's left to the viewer's right. This is true for either AP or PA projections.
Lateral positions are marked R or L by the side of the patient closest to the IR. Placement of lateral radiographic images for viewing varies depending on the radiologist's preference. One common method is to place the image so that the viewer is seeing the image from the same perspective as the x-ray tube. If the left marker is placed anteriorly to the patient, the L would be on the viewer's right (Fig. 1-114). However, some radiologists prefer to view laterals turned 90° and with the anteriorly placed L marker on the viewer's left. Technologists should determine the preferred method for viewing laterals in their department.
PA or AP oblique projections are placed for viewing the same way that a PA or AP projection is placed, with the patient's right to the viewer's left.
Decubitus chest and abdomen projections are generally viewed the way the x-ray tube “sees” them, placed crosswise with the upside of the patient also on the upper part of the view box (Fig. 1-114).
Upper and lower limb projections are viewed as projected by the x-ray beam onto the IR; the R or L lead marker appears right-side-up if it has been placed on the IR correctly.
Images that include the digits (hands and feet) generally are placed with the digits up. However, other images of the limbs are viewed in the anatomic position with the limbs hanging down (Fig. 1-116).
Viewing CT or MRI Images
The generally accepted way of viewing all CT and MRI axial images is similar to that used for conventional radiographs, even though the image represents a thin “slice” or sectional view of anatomic structures. In general, these images are placed so the patient's right is to the viewer's left (Fig. 1-117).


































































 to 5 inches (11 to 13 cm) proximal or superior to the symphysis pubis extending 3 to
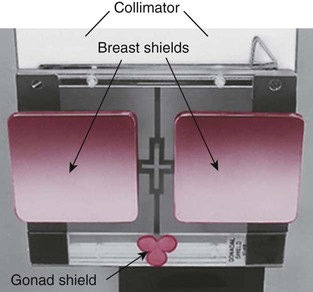
to 5 inches (11 to 13 cm) proximal or superior to the symphysis pubis extending 3 to  inches (8 to 9 cm) each way from the pelvic midline. The lower border of the shield should be at or slightly above the symphysis pubis, with the upper border extending just above the level of the anterior superior iliac spines (ASIS) (
inches (8 to 9 cm) each way from the pelvic midline. The lower border of the shield should be at or slightly above the symphysis pubis, with the upper border extending just above the level of the anterior superior iliac spines (ASIS) (