Enzootic bovine leukosis (bovine lymphosarcoma)
Etiology
Bovine leukemia virus (BLV), the causative agent of enzootic bovine leucosis (EBL), is an exogenous C-type oncovirus in the Retroviridae family
Epidemiology
Occurs worldwide, prevalence of infection varies between countries. Persistent infection is most common, followed by persistent lymphocytosis (PL) in 30% of infected animals, and less than 5% of infected animals develop lymphosarcoma. Clinical disease most common in mature cattle. Infected animal is only source of virus which is transmitted horizontally by infected lymphocytes in blood from parturition, contaminated surgical instruments, rectal palpation, blood-sucking insects. Congenital infection in 4–8% of calves born to infected cows. Genetic makeup of animal determines if becomes infected and develop disease. Economic losses due to loss in milk production traits and premature culling Is not a zoonosis
Signs
No clinical signs during stage of infection and persistent lymphocytosis. Lymphosarcomas characterized by loss of body weight, inappetence, pallor, weakness, and loss of milk production. Enlargement of all lymph nodes. Abomasal ulceration. Congestive heart failure. Paresis and paralyis due to neural involvement. Stertor due to enlargement of retropharyngeal lymph nodes. Eventually weak and recumbent. Sporadic bovine leukosis consists of juvenile lymphosarcoma, thymic lymphosarcoma, and cutaneous forms of tumors which may resemble enzootic bovine leukosis but are BLV-negative serologically
Lesions
Multicentric lymphoid tumors affecting all body systems especially heart, digestive tract, nervous system, reproductive tract
Differential diagnosis list
Congestive heart failure due to traumatic pericarditis
Control
Test and slaughter seropositive animals in herds and areas with low prevalence of infection. Use bulk tank milk ELISA as screening test. Establish virus free herds and certify by retesting. Control disease in herds and countries with high prevalence of infection by limitation of spread within herds and prevent introduction of infection
ETIOLOGY
Enzootic bovine leukosis and persistent lymphocytosis
The causative agent is bovine leukemia virus (BLV), an exogenous C-type oncovirus in the Retroviridae family. The complete genomic sequence of BLV strain from a cow has been described.1 Infection occurs by iatrogenic transfer of infected lymphocytes from one individual to another and is followed by a permanent antibody response and, less frequently, development of persistent lymphocytosis or lymphosarcoma.2 The virus causes a chronic B-cell proliferative disease in cattle and is an important model for human T-cell leukemia virus type 1 infection because of many shared molecular and biological features. However, EBL is not a zoonosis. It has leukemogenic activity, can be grown in tissue culture and produces specific antibodies in calves2 and sheep.
Sporadic bovine leukosis
Sporadic bovine leukosis affects animals under 3 years of age including:
• Juvenile form in calves less than 6 months old, characterized by multiple lymph node enlargement
• Thymic form in yearlings less than 2 years old, characterized by a swelling in the neck causing bloat and edema
• Cutaneous form in cattle 1–3 years old, characterized by the development of nodes and plaques in the skin
• Cutaneous T-cell lymphoma in two Friesian cows in Azores has been described.3
The bovine leukemia virus cannot be cultured from nor antibodies to the virus be detected in animals affected with sporadic bovine leukosis. There is no evidence that these forms of sporadic bovine leukosis are associated with an infectious agent.4
EPIDEMIOLOGY
Prevalence of infection and occurrence of clinical disease
Leukosis in cattle was originally described in Germany in 1871.2 Reports of the disease in cattle became common following World War II and most countries which raise cattle have reported the occurrence of the disease. The infection is now common in cattle in Canada, the United States, and many countries in Europe and South America.2
Serological surveys in cattle in the United States indicate prevalence rates within herds ranging from 0–100%. The disease does not spread rapidly and the number of herds containing positive reactors to the agar gel immunodiffusion (AGID) test is usually small. However, in infected herds the number of seropositive animals may be as high as 80%. Infection with the virus is estimated to be at least 20% in the adult dairy cow population of the United States, 6–11% in Canada, 27% in France, 37% in Venezuela; in the United Kingdom the prevalence of infection is low. In New Zealand, it is estimated that about 6.5% of the dairy herds have infected cattle, with an estimated within herd prevalence of 3.7%.5 The prevalence of infection in beef cattle in Australia is 0.22%. In a national survey in Canada, 40% of the herds contained BLV-infected cows. In Prince Edward Island in Canada, 49.2% of the herds tested had at least one positive reactor, and 5.5% of all the cows tested were positive. In maritime Canadian dairy cattle, the individual cow prevalence was 21% and the herd prevalence 70%.6
The seroprevalence of BLV infection in breeding beed bulls under 2 years of age offered for sale in Kansas was 8.5%.7 This indicates that young bulls purchased for entry into recipient herds could be infected with the virus.
In Argentina the individual seroprevalence is 33%, while the percentage of infected herds with one or more infected animals is 84%.8 The infection occurs in water buffalo in Brazil9 and in draught animals in Cambodia.10
An outbreak of enzootic bovine leukosis in Egypt was associated with the importation of Holstein Friesian heifers and bulls from Minnesota in 1989 to form a closed dairy herd in Upper Egypt.11 In 1996, clinical evidence of enzootic leukosis occurred and ELISA testing revealed a BLV seroprevalence of 37.7% in cattle under 2 years of age and 72.8% in animals over 2 years of age.
The occurrence of clinical lymphosarcoma in countries where the infection occurs is about 1 per 1000 per annum and in infection-free countries, 1 per 50 000 per annum. All of these data are subject to serious error because of the selective nature of the surveys, and until much larger random surveys are conducted, it is not possible to give accurate figures. Even in countries or areas where the infection and the disease are common, there are many herds that remain uninfected. Dairy cattle are much more commonly infected than beef cattle, and have a much higher incidence of lymphosarcoma. In severely affected dairy herds, an annual mortality rate of 2% is unremarkable and it may be as high as 5%.
All breeds of cattle are susceptible to BLV infection. It occurs rarely in animals less than 2 years of age and increases in incidence with increasing age.2 The prevalence of infection is higher in large herds than in smaller herds. The higher prevalence in dairy herds compared to beef herds is probably due to their closer confinement and the higher average age of the herds.
Methods of transmission
Direct contact
Horizontal transmission is the usual method by which the virus is spread under natural conditions. It appears that close physical contact and exchange of contaminated biological materials are required for transmission. The virus is present mostly in lymphocytes and can be found in the blood, milk and tumor masses. Most susceptible cattle become infected by exposure to infected lymphocytes, and not by cell-free virus.2 Either 10 μl (45 240 lymphocytes) or 1 μl (4524 lymphocytes) of whole blood from a BLV-seropositive cow when injected into calves resulted in infection and seroconversion. It is likely that a threshold number of approximately 100 BLV-infected cells is required to establish infection in the recipient.3 Therefore, any means by which BLV-infected lymphocytes can be transmitted from one cow to another is a potential means of transmission. Natural transmission occurs mostly in cattle more than 1.5 years of age, usually during the summer months between in-contact animals and possibly by insect or bat transmission of infected lymphocytes in whole blood. Some observations found an increased risk of infection in dairy cattle during the periparturient period; the crude odds ratio ranged from 4.7–6.0. This suggests that vaginal secretions, exudates and placentas from cows, as well as contaminated calving instruments may serve as sources of infected blood cells. The virus has been found in the nasal secretions of infected cattle for 2–4 years but there is no evidence that transmission to other animals occurred.
Semen, artificial insemination and embryo technology
Most workers have failed to find the virus in semen, and artificial insemination (AI) is not a method of spread, nor is embryo transplantation using zona pellucida intact embryos. Fertilized embryos from donors infected with BVL virus have been transferred without infection of the fetus. It is possible to produce transferable stage in vitro fertilized embryos which are free of in the integrated BLV provirus, from ooctyes which had been exposed to BLV during maturation.12 However, the virus has been found in semen collected by massage of the donor’s urethra and accessory glands per rectum, a procedure which is associated with contamination of semen with blood.2 While transmission by AI has not been demonstrated, it is possible that semen containing infected lymphocytes transmission could serve as a source of the virus. Thus bulls at AI centers will be required to be negative serologically to BVL virus. Properly collected semen from BLV seropositive bulls will not contribute to dissemination of viral infection.13
Transmission experiments suggest that the virus is not present in saliva but it does appear intermittently in urine.2 It is present in nasal and tracheal washings but only in cells, not as a free virus.
Iatrogenic transmission
Transmission can occur via infected blood which contaminates surgical instruments, such as dehorning gouges, ear tattooing pliers and hypodermic needles used on infected and then susceptible animals without disinfection. Transmission can also occur during blood transfusions and vaccines containing blood such as those for babesiosis and anaplasmosis.2 Amounts of blood as small as 0.1 μL are capable of transmitting the infection. Thus the infection can be transmitted via the tuberculin intradermal test. However, while some studies have found that use of common needles for blood sampling infected and non-infected cows at the same time poses a great risk of transmission of the virus to non-infected cows, other studies suggest that the quantities of infective blood passed during injection with common needles is too small to induce infection. The routine practice of brucellosis vaccination, ear-tagging and tattooing in dairy herds did not seem to be associated with the spread of the disease but infection could be reduced from 80% to 4% in heifers between the time of weaning to calving by altering dehorning methods. Transmission via infective milk is possible by the passage of infected lymphocytes through intestinal mucosal epithelium during the first few hours of life. However, infection via this route appears to occur very rarely, if at all, possibly because of the presence of maternal antibodies in the milk.
Rectal palpation
The virus can be transmitted by rectal inoculation of infected blood into cattle and sheep. Using blood-contaminated sleeves from palpating seropositive heifers to palpate seronegative cows resulted in transmission of infection as evidenced by antibody formation.14 This poses the possibility that the virus can be transmitted by rectal examination of cattle, particularly in dairy herds, when a single rectal palpation sleeve is used repeatedly during reproductive tract examinations as part of a health management program when many animals are palpated. Field studies to examine whether using the same sleeve for more than one animal or an individual sleeve for each animal, indicate that rectal transmission is a potential route of spread of BLV, but that it is related to frequency of palpation and age of cattle. Controlled studies of rectal palpation of cows in a dairy herd over a period of 22 months, using a single sleeve per animal or not changing the sleeve between an infected animal and seronegative animals resulted in a 2.8-fold increase in the risk of BLV infection.15 Thus rectal examination without a change of sleeve may be a risk factor in some herds.
Insects
Blood-sucking insects may be involved in transmission of the virus. Evidence implicating arthropod vectors in BLV transmission is indirect, involving experiments in which virus-carrying arthropods or parts of them were transferred to uninfected cattle. In several experiments, infected tabanids, other biting flies, and ticks were placed by hand on cattle and sheep. Minced mouthparts or hematophagus insects previously fed on BLV-infected cattle also were injected into hosts. In some countries there is empirical evidence that the incidence of seroconversion is higher after the tabanid fly season.2 A space–time study found a significant positive geographical correlation between the rate of incidence of BLV infection and the density of the horsefly population. Seasonal variations in the incidence rates also occur; the highest rates are generally observed during summer, and the lowest during winter, spring and early summer. There is also a time link between the rate of seroconversion and the variations in activity of the horsefly population. Experimentally, the virus has been transmitted by horse flies, Tabanus fuscicostatus, from a seropositive cow to recipient calves and goats. Horse flies take relatively large blood meals, have a painful bite, and are often interrupted in feeding and must finish feeding on other animals. This behavior and the large number of flies, and the low volume of blood and small number of lymphocytes required to transmit BLV, make tabanid flies candidates for mechanical vectors of the virus. The stable fly, Stomoxys calcitrans, has an insufficient mouth part volume to carry enough blood lymphocytes to transmit the virus.
Congenital infection
Congenital infection occurs in 4–8% of calves born from BLV-seropositive cows in naturally infected herds. These probably occur as a result of transplacental exposure to the virus during gestation. Calves born from clinically healthy cows naturally infected with BLV are negative for BLV before receiving colostrum, and in utero transmission of BLV may occur but is infrequent.16 Calves born from seropositive cows acquire colostral antibodies if they ingest colostrum and the antibody levels decline during the first 6–7 months of life. In one study the minimum and maximum duration of colostral antibodies were 14 and 147 d, respectively with a half-life of 36). The decay of colostral antibodies and the age at which a calf can be expected to become seronegative is a function of the quantity of BLV antibodies absorbed by the calf and the infection status of the calf.
Prevention of in utero transmission can be done using embryo transfer.
Interspecies transmission
Cattle are the only species infected naturally, although sheep and goats can be infected experimentally. The infection does not spread from cattle to commingled sheep, nor between experimentally infected and non-infected sheep. However, horizontal transmission of a naturally occurring lymphosarcoma in sheep is associated with an antigenically similar virus to the BVL virus. It is assumed that horizontal spread of the BVL virus from cattle to sheep will not occur. The experimental transfer of infection from cattle to sheep is effected so readily that it has become a preferred technique for testing for the presence of a virus.
Source of infection
In cattle, infection with the virus is permanent, spontaneous recovery has not been demonstrated and the virus is maintained in the cattle population. The virus is located in lymphocytes in a covert non-productive state, resulting in an inability of antibodies to arrest the infection, and multiplication of the virus is not necessary for survival or transmission. The virus is also capable of periodic antigenic change and circumventing control by immune mechanisms, thus the infected animal remains a source of infection for long periods, probably for life, regardless of the simultaneous presence of specific antibodies. This virus–host system is the same as that of other retroviruses, especially equine infectious anemia (EIA) and visna–maedi of sheep. In most circumstances, infection occurs when animals are in close physical contact and are more than 12 months old. Infection is established readily by SC and ID injection and by intratracheal infusion, but it does not occur after oral administration.2
Experimental transmission of the infection using tumor material, infected blood or tissue culture virus can be achieved in cattle, sheep, goats and with some doubts to chimpanzees, but the tumors are produced only in the three ruminants. A sheep bioassay can be used to determine the presence of the virus in infected cattle.17
Risk factors
Animal risk factors
The prevalence of infection based on seroprevalence is positively associated with increasing age in both dairy and beef cattle.2 The prevalence of infection in dairy cattle under 17–24 months of age is much lower than in adult cattle and increases sharply after 24 months of age when heifers join the milking herd and are in close contact with older cattle.2 The rate of spread may also be associated with the prevalence of infection; in herds with a prevalence of 13–22% when first tested, the spread was slow; in a herd with a prevalence of 42%, the spread was much more rapid.
Genetic resistance and susceptibility.
Infection with the BLV is not synonymous with clinical disease. Most animals which become infected do not develop neoplastic disease. Once infection has occurred, the subsequent development of only an antibody response, or antibody plus persistent lymphocytosis (PL) or antibody plus lymphosarcoma, with or without PL, is determined by the host’s genetic makeup. Lymphosarcoma, the terminal stage of BLV infection involving the clonal transformation of infected B-cells, occurs in about 1% of BLV-infected cattle and is under genetic control of the host.18
A complex relationship exists among genetic merit, milk production, BoLA genotype, and susceptibility to PL. Cows with high genetic potentials for milk and fat yields are more susceptible to PL than cows with lower genetic potentials, but cows with PL do not produce yields of milk or fat according to their predicted genetic values. Early attempts to quantify the economic impact of subclinical infection emphasized differences in milk production between seropositive and seronegative cattle. However, seropositive cattle may be in different stages of the disease complex. Antibodies to BLV may be present in recently infected cows with no other abnormality, in cows over 3 years of age with PL, and in animals older than 6 years of age with tumors.18 In addition, the genetic potential was not considered. It is now known that genetic merit is correlated with susceptibility to BLV infection and PL and thus inconsistent results are not surprising. When seropositive cows are divided into PL and non-PL categories and the genetic potential for various measures of milk production for each animal are taken into account, the results are much more clear.18
The phenotype frequencies of two BoLA-A alleles are associated with resistance and susceptibility to PL.18 Genetic resistance to PL maps closely to the class II genes of the bovine major histocompatibility complex, BoLA). The frequency of the PL resistance-associated BoLA-A14 allele in any age group was higher in BLV-infected non-PL cattle than in BLV-infected PL cattle in the same age group and increased from 30% in 3-year-old cows to 52% and 59% in 7- and 8-year-old cows which were retained in the herd. In contrast, the frequency of BoLA-A14 in cows with PL decreased in frequency from 7% in 3- to 5-year-old cows to 0% in cows older than 6 years. The relationship between BoLA-A allele frequencies and BLV infection over time, and between BLV infection and milk yield, imply an association of BoLA-A alleles with the full expression of milk and fat production potentials under conditions where BLV infection is prevalent. This suggests that genetic resistance to PL is associated with longevity in the herd, where there is a high prevalence of BLV infection.
Susceptibility to other diseases.
A highly significant correlation was shown between BLV infection and the persistence of Trichophyton verrucosum infection, which suggests that the immune system may be impaired in BLV-infected cows. Observations in Sweden indicate many significant associations between BLV infection status and measures of incidence, reproduction and production, but most were of low magnitude. The risk for other infectious diseases seemed to be greater among BLV-infected herds, while the risk for non-infectious diseases did not differ.
Immune mechanisms
Bovine leukemia virus is a type C retrovirus infecting B-cells and causing enzootic bovine leucosis.19 The disease is divided into three stages: serologically positive, but negative for lymphocytosis; serologically positive and positive for persistent lymphocytosis; and leukemia. Both humoral and cell-mediated immunity are induced in natural BLV infection. The cytokine profiles change as the infection progresses and interleukin-2 contributes to the development of BLV-induced persistent lymphocytosis. Tumor necrosis factor (TNF-α) are involved in the control of B-cell death, the virus-induced B-cell proliferation and the leukemogenesis of B-1 cells.
Following infection, a persistent antibody response occurs primarily to the envelope glycoprotein gp51 and the major core protein p24 of the BLV virions. The time from infection to development of antibodies can be as long as 14 weeks. Experimental infection of calves with the virus results in seroconversion which can be demonstrated with the ELISA in 4–5 weeks after infection.20 Acute lymphocytosis occurs at about the same time after infection.
Environmental and management risk factors
Lack of biosecurity
The introduction of infected animals into a herd has a significant positive effect on the subsequent prevalence of infection and clinical disease.21 The appearance of new outbreaks of leukosis is almost always the consequence of the introduction of BLV-infected animals in farms or areas previously free of the infection. Some outbreaks have followed restocking after brucellosis eradication. Others have occurred following the enlargement of the size of a dairy herd by purchasing animals from a variety of sources. In dairy herds in the Canadian Maritimes, those herds which had routine vaccination practices for other infectious diseases, the seroprevalence for BLV was lower than in herds which did not.22 The infection was introduced into an accredited BLV-free dairy herd following the introduction of 75 pregnant heifers 2.5 years before a clinical case of lymphosarcoma occurred in the herd and recognized at slaughter.23 Some of the heifers orginated from a BLV-infected herd including the animal with lymphosarcoma.
Calf management
The level of calf management in dairy herds is also a major risk factor. Any environmental factor or management practice which allows newborn calves access to infective blood will increase the level of infection in the calves, including:
1. Prolonged close contact between the cow and calf immediately after parturition
Some observations have found positive associations between BLV status of dairy herds and weaning age, housing pre-weaned calves in hutches or separate calf housing, and contact between young-stock and older animals during the winter housing period.24 In Ontario, dairy herds with at least one seropositive cow were more likely to have calves raised in calf hutches in winter, more likely to have calved in separate pens in the winter, and less likely to have cows calved in separate pens during the summer.25 However, the calves housed in hutches were not the same animals sampled for BLV and the results indicate only that those farms using this management procedure had a higher prevalence of BLV. BLV-seropositive cows had a slight, significant increase in calving interval compared to BLV-negative cows.
Pathogen risk factors
The BLV is an exogenous C-type retrovirus closely related to the human T-lymphocyte virus types I and II.2 It is highly cell associated and persists in a subpopulation of peripheral B-lymphocytes which proliferate as a result of the infection. Free virus is rarely or never found in the blood of infected cattle, and therefore not highly contagious. Once an animal is infected, the infection persists for life in the chromosomes of the infected host. The virus can be experimentally transmitted to a variety of animal species such as sheep, goats, pigs, rabbits, rhesus monkeys, chimpanzees, and buffalo.
Economic importance
General comments
Enzootic bovine leukosis causes significant economic losses associated with the costs of control and eradication programs. In Europe, the losses have been significant to the extent that an eradication campaign supported by the European Community has been in place for many years. Denmark has had an established national program for the control of the disease since 195926 and because of its importance, Sweden introduced a control program in 1990 with the aim of complete eradication of BLV from the Swedish cattle population.3
The nature and extent of the economic losses associated with enzootic bovine leukosis (EBL) have been controversial because the evidence has been conflicting. The obvious economic losses include the culling of cattle with lymphosarcoma, shortening of lifespan and loss of production potential, and restrictions on export of cattle and semen to importing countries. In Canada, BLV-seropositive bulls are barred entry into artificial insemination units.
In a spreadsheet analysis of dairy herds in the Maritimes in Canada, total annual costs for an average, infected 50 cow herd were $806.00 for enzootic bovine leukosis, compared to $2472.00 for Johne’s disease. $2412.00 for BVD, and $2304.00 for neosporosis.27 The association between EBL infection and annual value of production on dairy herds in the United States, as part of the National Animal Health Monitoring System’s 1996 dairy herd study, found that compared to herds with no test-positive cows, herds with test-positive cows produced 218 kg less milk per cow.28 The average reduction in average value of production was $59.00 per cow relative to test-negative herds. Most of the economic loss was due to reduced milk production in test-positive herds.
The effects of subclinical BLV infection on milk production, reproductive performance, longevity and culling rate are variable. In some observations, a BLV-seropositive cow had a shorter lifespan than both its seronegative counterpart and the entire milk cow population. Among older dairy cows, BLV-seropositive cows were culled prematurely, compared with uninfected cows.29 The culling rate was higher and milk production was lower in BLV-infected herds compared to BLV-free herds. The effect on reproduction was minor. In other observations, milk production, somatic cell count, age at disposal and culling were not influenced by seropositivity. A comparison of culling rates among dairy cows grouped on the basis of serologic status for BLV did not find any association between culling rate and serological status.30
Economic effects on milk production traits
When the effects of infection were examined according to genetic potential for milk and fat production in dairy cows, the results were surprising. The genetic potential for milk production was significantly greater in seropositive cows with PL and in seropositive hematologically normal cows than in seronegative herdmates. At the individual cow level, infected cows had greater milk production than uninfected cows based on seropositivity to BLV and 305-day mature equivalent fat-corrected milk production.31 Among seropositive cows, those with PL were culled at a younger age and had reduced production in the last lactation relative to other groups. Cows with PL do not produce butterfat according to their potential.
Using data collected over a 6-year period, milk and fat yields in BLV-infected cows with PL declined significantly relative to their BLV-infected non-persistently-infected herdmates.18 The estimated annual loss in milk yield is 366 kg for cows which had PL for 2 years, and 1204 kg for cows with PL for 3 years.18 The economic losses to the dairy industry in the United States have been estimated on the basis of the total number of cows with PL, price of milk, average milk yield, net income per cow, and loss in milk yield in cows with PL. Assuming 70% of cows are infected and 20% of infected cows develop PL, it is estimated that economic loss is about 0.25% of the gross income of milk production18 and the percent annual loss in net profit to the dairy industry due to PL is 3%.
The estimated costs of bovine leukemia infection, including costs of clinical disease and subclinical infection, in a dairy herd representative of the mid-Atlantic region in the US are $412.00 for a case of lymphosarcoma; for a herd with a 50% prevalence of infection, annual incidence of lymphosarcoma was 0.66.32 The mean annual cost of subclinical infection at 50% prevalence of infection was $6406.00. Mean annual cost of a test-and-manage control program was $1765.00. The cost of clinical disease and subclinical infection varied substantially with the prevalence of infection, whereas the cost of control varied with herd size. A basic BLV control program is considered economical in herds in which the prevalence of infection is ≤12.5%.
Economic effects of clinical disease
On an industry basis, the economic losses from lymphosarcoma are not large because only 0.1–5% of seropositive cows and 10–50% of cows with persistent lymphocytosis develop lymphosarcoma. However, for individual farms, a high incidence of the disease can be a major cause of economic loss, particularly in high producing elite dairy herds where pedigreed livestock are sold. In these pedigreed herds, individual animals are kept to a much older age than in the average commercial herd and, because of the increased prevalence of lymphosarcoma in cows over 5 years of age, the death losses are likely to be very severe in exactly the group of cows which is critical to the success of herd. In addition, there is the severe downgrading effect on the salability of stock from a herd known to have a disease in which genetic susceptibility is an important causative factor.
Loss may also result from lymphosarcoma by way of reduced production during the developmental stages of the disease. The course of the disease is usually sufficiently brief to make this a relatively unimportant consideration. Similarly, the immunosuppressive effect of infection with the virus appears not to have influence on the prevalence of other diseases.
Trade restrictions
A major economic effect of the disease lies in import restrictions placed by some countries on infected cattle and on semen either from infected bulls or from non-infected bulls from a positive herd. It is the practice, particularly in countries that do not have the disease, to require proof of freedom from infection with the virus from animals about to be imported into the country. This trend has been increased by the introduction of the infection into the United Kingdom in cattle imported from Canada. This is a matter of major importance when the cattle are purebred and are sold at high prices as breeding animals. Some countries are already demanding a negative blood test for all cattle and meat to be imported, and this could represent a loss of export markets for some countries.
Zoonotic implications
The possibility of transmission of the virus from cattle to humans is a real one; the virus is commonly present in the milk of infected cows and the disease has been transmitted to chimpanzees in this way. However, in spite of exhaustive, but certainly not complete, studies there is no evidence to support the notion that transmission occurs from cattle to humans.2 A case–control study failed to show any relationships between human acute lymphoid leukemia and exposure to dairy cattle and drinking raw milk nor with residence in the general area where dairy cattle are raised.2 The measurement of occurrence of disease in persons living on farms is the critical measurement because short-term pasteurization procedures destroy the infective agent in milk; farm dwellers who take their milk from the supply before the pasteurization points are thus exposed. All the evidence suggests that humans are at minimal risk of acquiring BVL infection and the diseases clinically associated with the virus.2
Using an immunoblot test, a serological survey of 257 humans in California found at least one antibody isotype reactive with BLV in 74% of the sera tested.33 This does not necessarily mean that humans are actually infected with BLV. Only 9% of the subjects indicated any direct contact with cattle or their biological products. The antibodies could be a response to heat-denatured BLV antigens from consumed milk or meat.
Other species
Lymphosarcoma occurs sporadically in all species, but natural infection with the BVL virus has been demonstrated only in sheep and capybaras.
Although there is no evidence of a relationship between bovine viral leukosis and any disease of pigs, there is a record of enzootic leukosis in that species which is inherited.
PATHOGENESIS
Virus and lesion
The virus establishes a persistent infection in a subpopulation of B-lymphocytes by integrating proviral DNA into the host cellular DNA.
The four possible outcomes after exposure of cattle to BVL virus are outlined in Fig. 21.1, including:
1. Failure of the animal to become infected, probably because of genetic resistance
2. Establishment of a permanent infection and the development of detectable levels of antibodies. (These animals are latent carriers of infection)
3. Establishment of a permanent infection; the animal becomes seropositive and also develops a persistent lymphocytosis, a benign lymphoproliferative process. It is not a preclinical stage of lymphosarcoma
4. Infected, seropositive animals that may or may not have been through a stage of persistent lymphocytosis and which develop neoplastic malignant tumors – lymphosarcoma.
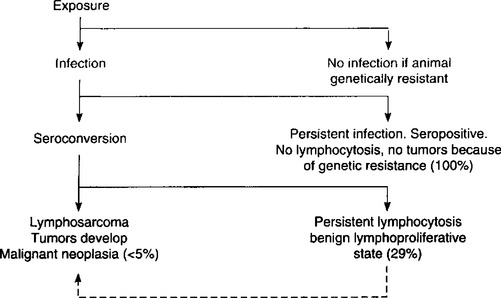
Fig. 21.1 Possible pathways after exposure to BVL virus (percentage figures indicate proportion of seroconverted animals that develop the particular form referred to2).
Whether or not the animal becomes infected or develops any of the other forms of the disease depends on the recipient’s genetic constitution (see Animal risk factors above). The outcome may also be influenced by the animal’s immune status and the size of the infective dose of virus. About 80% of animals with the adult form of the disease have a marked depression of IgM globulins. The immunological responsiveness of leukotic cattle to administered antigens is significantly depressed overall, especially to IgM, resulting from a deficiency in its production in the spleen and lymph nodes.
Lymphomatosis is a neoplasm of the lymphoreticular system. It is never benign and the lesions develop at varying rates in different animals so that the course may be quite short or protracted over several months.
The effects of BLV infection on milk production may not be related solely to overall animal health but may also be mediated directly at a cellular level.34 An in vitro system revealed that the casein production and mRNA synthesis by mammary epithelial cells from a BLV-infected cow were reduced compared with control cell lines without BLV.
Lesions and clinical disease
In adult cattle, almost any organ may be the site of lesions, but the abomasum, heart, and visceral and peripheral lymph nodes are most commonly affected. In calves, the visceral lymph nodes and spleen and liver are the common sites. Depending on the organ which is most involved, several clinical syndromes occur. Involvement of the abomasal wall results in impaired digestion and persistent diarrhea. When the atrial wall is affected, congestive heart failure occurs. In nervous tissue, the primary lesion is in the roots of peripheral nerves and spreads along the nerve to involve meninges and cord. Involvement of the spinal meninges and nerves results in the gradual onset of posterior paralysis. The skin, reproductive tract, and periorbital tissues are commonly affected. In the cutaneous form, intradermal thickenings develop which persist but do not cause discontinuity of the epithelium. They are composed of aggregations of neoplastic lymphocytes. Invasion of periorbital tissues commonly results in exophthalmos. Esophageal obstruction may result from mediastinal lymph node involvement in calves.
The exact nature of the tumor is unclear. The tumors consist of aggregations of neoplastic lymphocytes, but in many cases they may be more accurately described as reticulosarcoma. They are highly malignant and metastasize widely. The hemogram is variable and, although there may be an accompanying lymphocytosis, the presence of large numbers of immature lymphocytes in the blood smear is a more reliable indication of the presence of the disease. Some degree of anemia is common.
CLINICAL FINDINGS
Enzootic (adult) bovine viral leukosis (bovine lymphosarcoma)
This disease is characterized by the occurrence of multiple cases of adult multicentric lymphosarcoma, with tumors developing rapidly in many sites with an accompanying great variation in clinical signs and syndromes. An approximate indication of the frequency with which individual signs appear is set out in Fig. 21.2.35
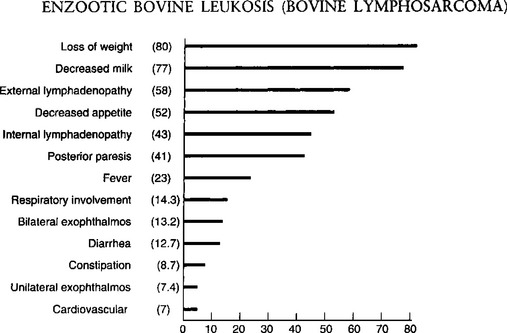
Fig. 21.2 Clinical diagnosis: frequency of predominant signs of bovine leukemia – 1100 field cases.
(By courtesy of Canadian Veterinary Journal34.)
The usual incubation period is 4–5 years with most occurring 4–5 years after the original case was introduced or a blood transfusion from an outside herd was given. This form is rarely seen in animals under 2 years of age and is most common in the 4–8 years age group. Persistent lymphocytosis without clinical signs occurs earlier but rarely before 2 years of age. Many cows remain in the preclinical stage for years, often for their complete productive lifetime without any apparent reduction in performance, but clinical disease appears in a proportion of these cows. The clinical signs and the duration of the illness vary with the number and importance of the sites involved and the speed with which the tumor masses grow.
In 5–10% of clinical cases the course is peracute and the affected animals often die suddenly or unexpectedly without any prior evidence of illness. Involvement of the adrenal glands, rupture of an abomasal ulcer or an affected spleen followed by acute internal hemorrhage are known causes. These animals are often in good bodily condition.
In most cases the course is subacute (up to 7 d) to chronic (several months) and initiated by an unexplainable loss of body condition and appetite, pallor and muscular weakness. The heart rate is not increased unless the myocardium is involved and the temperature is normal unless tumor growth is rapid and extensive, when it rises to 39.5–40°C (103–104°F). Although the following specific forms of the disease are described separately, in any one animal any combination of them may occur. In many cases, clinical illness sufficient to warrant the attention of the veterinarian is not observed until extensive involvement has occurred and the possibility of slaughter of the animal for beef purposes cannot be considered. On the other hand, many cases are examined at a time when diagnostic clinical signs are not yet evident. Once signs of clinical illness and tumor development are detectable the course is rapid and death occurs in 2–3 weeks.
Enlargement of the superficial lymph nodes
Enlargement of the superficial lymph nodes occurs in 75–90% of cases and is often an early clinical finding. This is usually accompanied by small (1 cm in diameter) SC lesions, often in the flanks and on the perineum. The skin lesions are probably enlarged hemolymph nodes and are of no diagnostic significance, often occurring in the absence of other signs of the disease. In many cases with advanced visceral involvement, peripheral lesions may be completely absent. Enlargement of visceral lymph nodes is common, but these are usually subclinical unless they compress other organs such as intestine or nerves. They may be palpable on rectal examination and special attention should be given to the deep inguinal and iliac nodes. In advanced cases, extensive spread to the peritoneum and pelvic viscera occurs and the tumor masses are easily palpable.
Although the enlargement of lymph nodes is often generalized, in many cows only a proportion of their nodes are involved. The enlargements may be confined to the pelvic nodes or to one or more SC nodes. Involvement of the nodes of the head is sometimes observed. The affected nodes are smooth and resilient and in dairy cows are easily seen and their presence may be marked by local edema. Occasionally, the entire body surface is covered with tumor masses 5–11 cm in diameter in the SC tissue.
Digestive tract lesions
Digestive tract lesions are common. Involvement of the abomasal wall results in a variable appetite, persistent diarrhea, not unlike that of Johne’s disease and occasionally, melena due to bleeding of an abomasal ulcer. Tumors of the mediastinal nodes may cause chronic, moderate bloating.
Cardiac lesions
Lesions in the heart usually invade the right atrial wall primarily, causing right-side congestive heart failure. There is hydropericardium with muffling of the heart sounds, hydrothorax with resulting dyspnea, engorgement of the jugular veins and edema of the brisket and sometimes of the intermandibular space. The heart sounds are commonly muffled and other cardiac abnormalities may be obvious. Tachycardia due to insufficiency and arrhthymia due to heart block are common. A systolic murmur is also common along with an abnormal jugular pulse. The liver may be enlarged and palpable caudal to right costal arch, and passive congestion of the liver and visceral edema result in persistent diarrhea.
Nervous system involvement
Neural lymphomatosis is usually manifested by the gradual onset over several weeks of posterior paralysis. Knuckling of the fetlocks of the hind legs while walking is common and one leg may be more affected than the other. This is followed by difficulty in rising, and finally clinical recumbency and inability to stand. At this stage, sensation is retained, but movement is limited or absent. There may be a zone of hyperesthesia at the site of the lesion, which is usually at the last lumbar or first sacral vertebra. Appetite and other functions, apart from the effects of recumbency, are usually normal. Metastases in the cranial meninges produce signs of space-occupying lesions with localizing signs referable to the site of the lesion.
Less common lesions
These include enlargement of the retropharyngeal lymph nodes which may cause stertor and dyspnea. Sometimes clinically detectable lesions occur in the periorbital tissues, causing protrusion of the eyeball (exophthalmos), and in limb muscles, ureter and kidney,36 and genitalia. Involvement of the uterus may be detectable as multiple nodular enlargement on rectal examination.37 Severe bilateral exopthalmos may occur along with generalized lymphadenopathy.38 Periureteral lesions may lead to hydronephrosis with diffuse enlargement of the kidneys while tumors in renal tissue cause nodular enlargements. In either case terminal uremia develops.
BLV particles have been detected by electron microscopy around lymphocytes in the mammary tissue of BLV antibody positive cows affected by subclinical mastitis.39 Whether the virus is a causative agent or an immunosuppressent in bovine mastitis is unknown.
Sporadic bovine leukosis
The calf, thymic and cutaneous forms are designated sporadic bovine leukosis.
Juvenile or calf lymphosarcoma
This occurs in calves at 2 weeks to 6 months of age and is manifested by gradual loss of weight and the sudden enlargement of all lymph nodes, and depression and weakness. Fever, tachycardia and posterior paresis are less constant signs. Death occurs in 2–8 weeks after the onset of signs. Signs of pressure on internal organs, including bloat and congestive heart failure may occur. Diffuse infiltration of major nerves of a limb may also occur.40 Unusually the disease may be fully developed in utero, so that the newborn calf is affected with tumors, or be delayed until 2 years of age. Lymphosarcoma of the pharyngeal region causing retropharyngeal swelling and dyspnea in a 7-month-old beef steer has been recorded.41
Bone and bone marrow necrosis associated with the calf form of sporadic bovine leukosis has been recorded in calves 3 weeks to 8 months of age. Clinical findings included unthriftiness and inactivity, posterior ataxia, superficial lymph node enlargement, lameness and respiratory distress. Involvement of the tibio-tarsal joint, ribs, and spinal canal may also occur resulting in ataxia and paresis.42 Multiple bone infarcts and bone marrow necrosis were present at necropsy. Lymphosarcoma of the mandible of a 2-year-old heifer has also been recorded.
Thymic lymphosarcoma of young cattle
Infiltration of the thymus is a common finding in animals 1–2 years of age and is characterized by massive thymic enlargement and lesions in bone marrow and regional lymph nodes. Jugular vein engorgement and marked brisket edema extending to the submandibular region are common. Moderate bloat due to inability to eructate because of compression of the esophagus may occur.25 The thymic mass is usually not palpable. This form is more common in beef than in dairy cattle. An atypical lymphosarcoma in a mature cow negative for BLV and similar to the thymic form has been reported.43 Metastatic thymic lymphosarcoma in a calf has been recorded36 including a case with metastases causing spinal cord compression. Cases of both the systemic and local forms of sporadic bovine leukosis have been described.25 A large number of the thymic lymphosarcoma occurred in calves in five regions in France over a period of 5 months. Most of the calves had been sired by the same bull, which suggests that the disease had an inherited basis.
Cutaneous lymphoma
This is most common in cattle less than 3 years of age. It is rare and manifested by cutaneous plaques (1–30 cm diameter) appearing on the neck, back, croup and thighs. The plaques become covered with a thick, gray-white scab and the hair is shed; then the center becomes depressed and the nodule commences to shrink. The surface of some placques may become ulcerated and have a serosanguinous exudate. Some of the lesions have a cauliflower-like appearance, appear black, and are ulcerated and foul-smelling. After a period of weeks or months hair grows again and the nodules disappear as does the enlargement of the peripheral lymph nodes. Spontaneous regression of bovine cutaneous leukosis has been recorded. Relapse may occur in 1–2 years with reappearance of cutaneous lesions and signs of involvement of internal organs as in the enzootic form of the disease. In one series of 10 heifers, all animals had lymphadenopathy.44 Some had leukoctyosis and some had lymphocytosis. The body condition may vary from normal to thin and underdeveloped. Some affected animals may have a fever.
Cutaneous T-cell lymphoma in two Friesian cows in the Azores has been reported.4 The lesions consisted of raised pink plaques, with no pruritus or signs of associated pain, which were extensively distributed over both lateral and ventral body regions. Immunocytochemistry found the tumor cells positive for CD3, confirming the T-cell origin of the cells which involved both skin and regional lymph nodes.
Other species
Outbreaks of lymphosarcoma in sheep have been observed with clinical, epidemiological, hematological and necropsy findings similar to those of enzootic bovine leukosis. B-cell leukemia has been described in sheep.45
Infection of other species with BVL virus has not been demonstrated, but epidemic occurrences of lymphosarcoma have been observed in pigs, but only sporadic cases in horses.
In pigs, non-specific emaciation, limb weakness and anorexia are most commonly observed. Sporadic cases in this species are unlikely to be recorded, and although outbreaks have occurred, they and the enzootic form are not commonly encountered. In one herd with the enzootic disease all cases encountered were in pigs less than 6 months of age. There was stunting of growth, development of a pot belly, enlargement of peripheral lymph nodes, and a lymphocytosis, including the presence of immature cells.
In horses, the disease occurs most commonly in animals over 6 years of age. The common clinical manifestations are:
• Subcutaneous enlargements which may ulcerate
• Enlargement of internal and external lymph nodes
The course varies from acute to chronic, but most affected horses die within a month of first showing signs. Diffuse alimentary lymphoma of the small intestine in adult horses is characterized by:
Malignant lymphoma with ulcerative pharyngitis in horses has been recorded.
CLINICAL PATHOLOGY
A definitive antemortem diagnosis depends on the clinicopathological examination of the animal. Several diagnostic techniques are available and it is important to make the appropriate selection for the particular stage of the disease that is being considered, thus:
• Diagnosis of the viral infection is made by serological or virological techniques
• Persistent lymphocytosis is identified by hematology
• Neoplastic tumors are identified by histological examination of a biopsy specimen.
Because of the increasing economic impact of BLV infection in the cattle industry, the availability of a highly sensitive and specific assay for the identification of BLV-infected animals is of critical importance. Such an assay is needed for the selection of BLV-free cattle for commercial sale, prepurchase testing of breeding animals, and import or export testing, and for control and eradication programs. Ideally, the assay should be practical, inexpensive, and able to be adapted for large-scale use.
Diagnosis of the presence of infection with BVL virus
Serological tests
Virtually all cattle infected with BLV will continuously have antibodies against the major internal (p24) and envelope (gp51) virion proteins in their serum, and serological tests are commonly used for the diagnosis of BLV infection in cattle over 6 months of age.
Radioimmunoprecipitation assay (RIP).
The RIP using gp51 or p24 as antigen, is a highly sensitive and specific method for serologic diagnosis of BLV infection.46 The RIP assay has been used as the criterion-referenced standard to critically evaluate the performance of other diagnostic tests for BLV infection. Detailed comparisons of various BLV assays in a large number of cattle of various origins and ages found that the RIP assay is the most sensitive and specific test. However, its major disadvantage is that it requires a gamma counter and radioistopes, which are expensive.
This is a good screening test to determine the presence of infection in an individual animal or herd.2 The estimated specificity of 99.8% and the sensitivity of 98.5% indicate that the test is a reliable and accurate method to detect BLV infection. False-positive and false-negative results do occur and may be associated with some variability of the immune system or from human error. The AGID test is the official reference test of the Office International des Epizooties and the European Community and is the test recognized by most governments as the official test for purposes of testing imported animals. There is however, a lack of standardization between the BLV-AGID kits used in North America and Europe.47
This is suitable for individual cow testing because of its accuracy. There are several versions of this and the one using the virion gp antigen is preferred.2 It is one of the most sensitive tests and is useful for the detection of BLV antibodies in cattle exposed no longer than 2 weeks, in milk samples, and in serum samples from periparturient dams.
In more recent years, ELISA-based testing has replaced the AGID in eradication programs in several countries. It is more sensitive than other serological tests and can be used on milk.2 The superior sensitivity of the ELISA for pooled serum samples allows detection of antibodies in herds with a prevalence of less than 1%, whereas the AGID test detected only 50% of the herds detected by the ELISA.2 Two commercially available ELISAs and the polymerase chain (PCR) were evaluated and compared with the AGID to detect antibodies to BLV or its nucleic acid.5 The ELISA tests detected about 10% more reactors than the AGID and the elecrophoretic immunoblotting results. Some ELISA positive animals were not detected by the PCR.
Four commercially available BLV-ELISA kits from Europe or the United States were compared to the AGID test officially approved by the Canadian Food Inspection Agency. The ELISA tests were more sensitive than the AGID test kits and the gp51 BLV-ELISA is now recognized as an official test method for the serodiagnosis of bovine leukosis in Canada.48
A highly sensitive and specific blocking ELISA comparable to the radioimmunoprecipitation assay for the detection of BLV antibodies in serum and milk samples has been developed.46
The chronology of seroconversion is important in the serological diagnosis. Calves from infected dams have a 20% chance of being infected in utero and seropositive at birth. If they are serologically negative at birth they seroconvert at their first ingestion of colostrum from seropositive cows and this passively acquired immunity persists for 2–7 months. These calves, and calves from uninfected mothers become positive at varying ages depending on when they come into contact with infection, usually when they are placed into the infected adult herd. This can be as early as 9 months of age, but as a general rule positive reactions are uncommon in cattle which are less than 2 years of age. Seroconversion usually takes place 3–4 months after the negative animals are placed in the infective group, although the interval is longer in the winter than in the summer. Infected animals are seropositive and infected for long periods, usually for life.
Using the ELISA, experimental infection of calves at 3–4 months of age results in seroconversion to the virus at 4–5 weeks after infection.20
In a control and eradication program, early detection of infected calves is difficult because colostral antibodies to BLV cannot be differentiated from antibodies resulting from natural infection. By using measures of colostral antibody concentration, calves infected in utero could be identified by 80 d of age. Calves over 6–8 months of age with a positive immunodiffusion test will likely be infected permanently. Calves which have ingested colostrum from seropositive cows usually have maternal antibodies and polymerase chain reaction tests are necessary to detect the virus and distinguish between infected and virus-free calves.49
This is much more sensitive than the AGID and has been adopted for testing milk from individual cows and pooled milk samples.46,50 A comparison of the ELISA and AGID tests for the detection of BLV antibodies in bovine serum and milk found a high level of agreement.50 The bulk tank milk ELISA is useful for identification of herds which are negative for BLV infection.51 The ELISA identified 80% of herds as positive for BLV and had an apparent sensitivity and specificity of 0.97 and 0.62, respectively.51 But after accounting for the sensitivity and specificity of the AGID test in individual animals, the specificity of the ELISA test for milk was 0.44. With the moderately low specificity, herds identified as positive by the ELISA would require further testing at the individual or herd level to definitively establish their BLV status.
An indirect ELISA to detect antibodies to BLV in bulk-milk samples in Sweden is being used to assist in the eradication of infection from Swedish herds.52 The antibody level in milk is lower than in serum but the sensitivity of the ELISA is as effective as for sera. Testing of bulk milk is a useful and practical method for large-scale epidemiological studies and initial eradication programs. Heifers, bulls, and dry cows are not included when bulk milk is tested and all animals over 1 year of age need to be sampled individually before a herd is declared free of the virus. The sensitivity and specificity of the milk ELISA is estimated to be adequate until the prevalence of BLV-infected individuals in the country is less than 1%.
Detection of virus
Polymerase chain reaction (PCR).
The PCR is a sensitive and specific assay for direct diagnosis of BLV infection in peripheral blood lymphocytes.53 The test is useful for the early detection of BLV infection even before antibodies are present. It is more sensitive than the AGID test or the ELISA in detecting infected cattle where the prevalence of infection is less than 5%.54 The test can identify proviral DNA of BLV in the lymphocytes of calves at birth which are born to infected cows.55 All calves found to be infected at birth were born to BLV-positive cows with persistent lymphocytosis. At birth, the presence of a titer can be due to colostral antibodies or perinatal infection and the PCR test can differentiate uninfected newborn calves with colostral antibodies from BLV-infected calves and detect the presence of the virus in the presence of antibodies.47 The PCR has a practical application in the identification of BLV-infected calves, regardless of colostral antibody, which allows immediate removal of the source from the herd. In a dairy herd with a high prevalence of BLV, a positive PCR assay result provided definitive evidence that a cow was infected with BLV.56 However, sensitivity and specificity were 0.672 and 1.00, respectively. Predictive value of a positive test was 1.00, and predictive value of a negative test was 0.421. Thus PCR assay alone is unreliable for routine detection of BLV in herds with a high prevalence of BLV infection.
The PCR can also be used to ensure that cattle used in the production of a whole blood vaccine for tick-borne disease are free from BLV infection.17 The sheep bioassay, currently in use, requires 4 months of serological testing to insure that donor animals are not infected. Replacement of the sheep bioassay with the PCR could result in considerable saving of time and effort. Use of the PCR requires stringent precautions to prevent false-positive results due to contamination of samples with PCR product.17
A nested PCR identified 98% of BLV seropositive cows from blood and 65% from milk, whereas real-time PCR detected 94% of BLV seropositive cows from blood and 59% from milk.57 BLV was also detected in 10% of seronegative cows most likely because of early detection before seroconversion.
Differentiation between enzootic and sporadic bovine leukosis.
The role of BLV in some cases of sporadic bovine lymphomas needs to be re-examined. The findings of persistently seronegative PCR-positive and seropositive PCR-negative cattle indicates that the BLV cannot be excluded as a causative agent in sporadic bovine leukosis. Enzootic bovine leukosis cannot be distinguished from sporadic bovine leukosis on histopathological examination. The ELISA is recommended as a method of choice to differentiate between EBL and sporadic bovine leukosis (SBL) because it is a rapid, reliable and sensitive test which is inexpensive and easy to perform. In cases where no blood or other fluids are obtained, the PCR test is the most useful method for the direct detection of BLV.
Diagnosis of persistent lymphocytosis (PL)
Approximately 30% of animals infected with the BLV develop PL, which is defined as an increase in the absolute lymphocyte count of three or more standard deviations above the normal mean as determined for that respective breed and age group of animals in leukosis-free herds. The PL is an increase in peripheral B-lymphocytes. It has been suggested that one additional criterion for PL should be that it persists for more than 3 months. When PL was first recognized in herds which experienced clinical lymphosarcoma, it was considered a subclinical expression of the tumor stage of the disease. It became an important diagnostic criterion in control and eradication programs until serological tests became available to more accurately identify infected animals. The majority of cells involved in PL are normal lymphocytes but atypical and abnormal forms have been described and are considered as indicative of preleukemic condition. The total count increases from a normal of 6000 to as high as 15 000/μL. The percentage of lymphocytes in the total white blood cell count increases from the normal of 50–65% is considered a positive result. The presence of 25% or more of the total lymphocyte count as atypical immature cells is also considered a significant aberration. The PL may subside in animals which subsequently develop lymphosarcoma.
The association between the strength of serologic recognition of BLV by the use of ELISA and lymphocyte count in bovine leukosis virus-infected cows has been examined.52 The sample-to-positive ratio, which is the ratio between the test sample and a positive control sample, was compared among lymphocytotic and nonlymphocytotic cows. The sample-to-positive ratio and lymphocyte count were related but cows with high sample-to-positive ratio were not always lymphocytotic. Culling cows on the basis of sample-to-positive ratio will reduce culling of ELISA-positive cows, however, culling on the basis of lymphocyte count will eliminate a greater proportion of the reservoir of infection.
Diagnosis of lymphosarcoma
This can only be done by histopathological examination of a section of tumor material obtained by biopsy or necropsy. A needle aspirate of an enlarged peripheral lymph node may provide a rapid and inexpensive diagnosis. Enlarged lymph nodes or hemolymph nodes are the usual sources, but when the genital tract is involved an exploratory laparotomy is usually performed so that a sample can be obtained. The lymphocyte count may increase to 20 000–30 000 μL, and in some cases may reach values of 50 000–100 000 μL, and even 400 000– 500 000 μL. Conversely, in some cases, the lymphocyte count decreases. Chromosomal changes may be detectable in cells from lymph nodes or in leukocytes from peripheral blood of affected animals. When there is myocardial involvement there may be obvious changes in the electrocardiogram but these are unlikely to be of value in differential diagnosis.
NECROPSY FINDINGS
In cattle, firm white tumor masses may be found in any organ although two rather different patterns of distribution are apparent. In newborn and young animals, the common sites are: kidneys, thymus, liver, spleen and peripheral and internal lymph nodes. This may or may not be a characteristic of the ‘sporadic’ form of the disease. In adults, the heart, abomasum and spinal cord are often involved. In the heart, the tumor masses invade particularly the right atrium, though they may occur generally throughout the myocardium and extend to the pericardium. The frequency of early changes in the subepicardial tissue of the right atrium suggests that this is an area from which tissues should be selected in latent or doubtful cases. The abomasal wall, when involved, shows a gross, uneven thickening with tumor material in the submucosa, particularly in the pyloric region. Similar lesions occur commonly in the intestinal wall. Deep ulcerations in the affected area are not uncommon. Involvement of the nervous system usually includes thickening of the peripheral nerves coming from the last lumbar or first sacral cord segment or more rarely in a cranial cervical site. This may be associated with one or more circumscribed thickenings in the spinal meninges. Affected lymph nodes may be enormously enlarged and be composed of both normal and neoplastic tissue. The latter is firmer and whiter than normal lymphoid tissue and often surrounds foci of bright yellow necrosis. Less common sites include the:
• Ureters (usually near the renal pelvis)
• Uterus (either as nodular masses or diffuse infiltration)
• Mediastinal, sternal, mesenteric and other internal lymph nodes
When performing the necropsy, it is important to remember that lymphosarcoma can appear not only as discrete nodular masses but as a diffuse tissue infiltrate. The latter pattern results in an enlarged pale organ which is easily misinterpreted as a degenerative change rather than as a neoplastic process.
Histologically, the tumor masses are composed of densely packed, monomorphic lymphocytic cells. Attempts to better characterize the nature of these cell populations have been published.58,59 The cleaved variant of the diffuse large cell type with a high mitotic index is characteristic of enzootic lymphoma and this high-grade type of B-cell tumor may be a consequence of the viral etiology of this form of the disease.31 It is possible to confirm viral infection in some cases by a PCR test but such testing is rarely justified. Immunohistochemical staining of formalin-fixed, paraffin-embedded tissue sections of tumors can be used to con-firm that neoplastic lymphocytes are of thymic origin when thoracic masses are examined.60
Samples for confirmation of diagnosis
• Histology – formalin-fixed samples of gross lesions, plus enlarged lymph nodes, bone marrow, liver, spleen, thymus, right atrium, abomasum, uterus (LM, IHC)
Because of the very wide range of clinical findings, a definitive diagnosis of BVL is often difficult. Enlargement of peripheral lymph nodes without fever or lymphangitis is unusual in other diseases, with the exception of tuberculosis, which can be differentiated by the tuberculin test. In the absence of these enlargements, the digestive form may easily be confused with Johne’s disease. The cardiac form closely resembles traumatic pericarditis and endocarditis, but there is absence of fever and toxemia, and the characteristic neutrophilia of these two diseases is usually absent. Involvement of the spinal nerves of meninges may be confused with spinal cord abscess or with the dumb form of rabies. An examination of cerebrospinal fluid may be of value in determining the presence of an abscess and rabies has a much shorter course and other diagnostic signs. Multiple lymph node enlargements in the abdominal cavity, and nodular lesions in the uterine wall may be confused with fat necrosis, but the nature of the lesion can usually be determined by careful rectal palpation. Stertor caused by enlargement of the retropharyngeal lymph nodes is also commonly caused by tuberculosis and actinobacillosis.
Cases of atypical lymphosarcoma43 and thymic lymphosarcoma41 which are BLV-negative may resemble lymphosarcoma of enzootic bovine leucosis.61
Echocardiography is now being used to detect intracardiac masses which may be compatible with EBL.62
CONTROL
The disease can be eradicated from a herd and even a country, or controlled at a low level. The option chosen depends initially on the prevalence of infection in the herd, the value of the animals in the herd, and whether a governmental indemnity offered for seropositive cows which are culled and sent to slaughter.
History of Compulsory Eradication Program in Denmark
Control and eradication programs have been in effect on a nationwide basis in several western European countries.2 Denmark began an eradication program in 1959 based on the occurrence of clinical lymphosarcoma, and the identification of cattle with PL using the Bendixen hematological key for classifying cattle as normal, suspect or lymphocytic. Leukosis was declared a reportable disease, and all adult cattle from herds in which cases of leukosis originated were subjected to a hematological examination. Affected herds were quarantined, and indemnity was offered to induce owners to have their entire herd slaughtered. This herd-slaughter policy was continued until 1982. When the AGID test became available, the Bendixen key was discontinued and only the AGID test was used in the official program between 1979 and 1982. Routine testing was discontinued in 1982. Surveillance involved testing random sera collected from every sixth adult cow to be slaughtered. According to the official Danish control program, the incidence of tumors in adult cattle at the start of the eradication program was at least 10 times greater than 10 years later. The hematological test was less sensitive than subsequent serological tests but the specificity was fairly high and only a few herds were erroneously classified as leukosis herds.61 When the serological tests were introduced, some herds which were classified as leukosis-free based on the hematological key, were found to be infected.
Voluntary eradication programs using the AGID test have been effective in other member countries of the European Community in the last two decades and have been successful in reducing the prevalence of infection and disease.2 In the Federal Republic of Germany, eradication was achieved in 5 years. These programs have been successful in part because of the low prevalence of infection and the economic losses from culling seropositive cows has not been large.
In Britain, EBL is a reportable disease but is uncommon.41 A national testing program was begun in 1992. All blood samples collected for routine periodic testing for brucellosis have also been tested for BLV and milk samples are collected every 3 months from dairy herds for BLV testing. The prevalence of infection has been low and the source of infection undetermined. Some of the animals had been imported from Canada but in other cases there was no association with importation.
Considering the animal-health aspects and possible consumer reactions against having a widespread retrovirus infection in food-producing animals, and the requirements for exporting cattle and semen, Sweden introduced a national program for the eradication of BLV in 1990.52 An ELISA test was evaluated for detection of antibodies to BLV in individual and bulk milk and serum samples. It is proposed that eradication can be based on using the ELISA on milk samples in combination with other diagnostic tests and the prompt removal of infected animals.
In Canada and the United States, it is considered cost prohibitive to cull and slaughter all seropositive cattle because of the high prevalence of infection. Many seropositive cows are valuable pedigreed animals, and there are no indemnity programs available. Thus all control and eradication programs in these countries are herd based and strictly voluntary. Livestock producers are willing to adopt control programs because of the economic losses associated with export restrictions if their cattle are infected, and the losses due to the occasional clustering of cases of lymphosarcoma.
Enzootic bovine leukosis was eradicated from Finland in 1996.63 The disease was first recognized in 1966 and it required 30 years using the key principle of test and slaughter policy to achieve eradication. The infection status was monitored at meat inspection, and hematologically between 1970 and 1977, serologically between 1978 and 1989. Annual surveys including all dairy herds and samples from beef animals were conducted in 1990–2001. Bulk tank samples represented the dairy herds in the surveys; beef animals were sampled individually at slaughter. The maximum positive herd-level percentage in the survey was 0.03%. The herd level prevalence of infection never exceeded 5%.
Eradication programs
Enzootic bovine leukosis can be eradicated only by:
• Test and slaughter of cattle infected with the virus. Programs based on the culling of seropositive cows are effective
• The maintenance of a closed herd which permits the entry of only those animals free of infection.
The efficiency of such a program depends on the accuracy of the test used to identify the infected animals, and the repetition of the test at an appropriate interval so that animals that were in the incubation stages of the disease at the time of the first test will have had time to seroconvert. The recommended procedure is:
1. Identify infected animals using the AGID test2
2. Cull and slaughter seropositive animals immediately
3. Retest the herd 30–60 d later
4. Use the PCR assay to test young calves and as a complementary test for clarifying doubtful test results in herds with a low prevalence of infection.54
Testing is repeated until the herd has a negative test. When the herd is negative, testing is repeated every 6 months and the herd declared free when there have been no positive reactors for 2 years. Future introductions into the herd are managed most safely by artificial insemination or fertilized ovum transfer, or importations of animals which have been tested and are seronegative on two tests carried out 30 and 60 days prior to arrival.
In herds where the prevalence is high, a two-herd scheme can be successful. Newborn calves are removed from seropositive cows immediately after birth, fed colostrum from seronegative cows and raised in isolation. All animals over 6 months of age are tested periodically and seropositive animals culled. The parent herd is eventually disposed of as negative replacement animals become available. Only those bulls which are seronegative may be used and they must be tested every 3 months.
Although eradication is biologically feasible, it is unlikely that area eradication programs will be implemented on an extensive scale because losses from the disease are not sufficiently high, and there is a high risk of insect vectors reintroducing it which poses a real threat to maintenance of a BVL-free herd. The cost-effectiveness of an eradication program on a national basis would be a major consideration. For an individual herd, it is feasible provided some steps were taken to increase genetic resistance of the residual stock and to reduce the chances of in-contact infection occurring.
Limitation of spread of infection
In herds with a high prevalence of infection, the test and slaughter method of eradication is not economically viable if the animals have a high economic value because of superior genetic potential. Control of infection in these herds is possible using embryo transfer from infected dams to negative recipients and isolation of newborn calves but these are not practical on a country-wide basis. An alternative method is segregation of BLV-infected and non-infected animals based on the AGID test. This is known as the test and segregation method, which is based on the evidence that the spread of infection between animals is relatively slow and that the virus is spread by movement of infected animals from one herd to another and within a herd. Following the initial testing of the herd, the herd is divided into two groups, BLV-positive and BLV-negative, and kept at least 200 m apart. A third separate location is used for quarantine of replacement animals. Replacement animals must be found negative in two consecutive AGID tests, the first within 30 d before purchase and the second after 60 d of isolation, prior to being moved into the negative group. The AGID tests are conducted every 3 months and the reactors removed to the positive group location until the remaining animals in the herd have attained BLV-negative status by the test. Thereafter, the tests are done every 6 months and continued until at least four consecutive negative tests are obtained for each herd. Variations of this method of test and segregation with subsequent removal of seropositive animals in the routine culling program have been successful. The colostrum and milk fed to calves in the BLV-negative group must be from seronegative cows or be pasteurized to inactivate the virus.
In Canada, cattle owners may enrol in the Canada Health Accredited Herd program to declare freedom from EBL. All reactors must be removed from the herd. If a large number of reactors are detected, two herds on two separate farms can be established: one herd comprised of the reactors and the other of cattle which are seronegative. Calves from the reactor herd can be added to the accredited herd in accordance with strict isolation and testing procedures. To qualify for accredited certification, a herd must have two consecutive negative herd tests, at least 4 and less than 12 months apart. The tests must include all cattle in the herd. The first annual renewal test must occur no more than 12 months following the second qualifying test for certification, and must include all cattle in the herd. Subsequent renewal tests must occur within the same 12-month interval. Only cattle 24 months of age and older must be tested but a herd inventory and audit must be performed on the whole herd. In herds with reactors, the two qualifying tests do not begin until at least 4 months after the removal of the last reactor uncovered during any test. Herd additions can be made during the qualifying test period or after certification has been achieved. Each animal must be accompanied by a health certificate and depending on the enzootic bovine leukosis status of the originating herd, certain testing and isolation procedures could apply. Owners wishing to have their animals attend exhibitions can do so providing they adhere to certain conditions. Properly processed semen and embryos can be introduced without restrictions. Owners are encouraged to follow preventive health management practices to augment the eradication of enzootic bovine leukosis from their herds. These include all areas where blood transfer could occur (needles, dehorning, castrating, extra teat removal, ear tagging, tattooing, hoof trimming, rectal palpations, drenching) and other procedures which transfer leukocytes, and routine insect control.
Prevention of infection in calves and young stock
Several management techniques can be used to prevent infection in calves from birth until they become herd replacements. The feeding of newborn calves with colostrum and milk from seronegative cows has been widely accepted as effective in preventing infection in calves. Postnatal infection in calves can also be minimized by feeding milk replacer, and/or whole milk from non-infected cows. The use of colostrum and milk from non-infected cows permits early serological detection of infected calves. However, feeding colostrum from seropositive cows to newborn calves can provide significant protection from infection during the first 3 months of life. Field studies indicate that colostrum-derived BLV antibodies may prevent as much as 50% of the infection during the first 3 months compared to calves which did not receive colostrum with BLV antibodies. Further reduction in the risk of infection via colostrum can be achieved by pasteurization of the colostrum – 63°C for 30 min.64 The colostrum-derived BLV antibodies will however delay early detection of infection in calves. The replacement of whole milk feeding with high-quality milk replacer may also be considered.
Transmission to newborn calves can also be reduced by avoiding exposure to maternal blood at the time of parturition, housing calves in individual hutches with individual feeders and waterers, and management techniques to avoid iatrogenic transmission.65 When handling a group of calves, the youngest ones should be handled first and the older and sick calves last. Equipment which could act as fomites in transferring blood should be disinfected with chlorhexidine when used between calves. These instruments include:
Dehorning calves using the electrocautery method before 2 months of age can reduce the prevalence of infection compared to gouge dehorning, which allows the transfer of infected blood between calves. Handling facilities which become contaminated with blood should be cleaned between calves. Fly control should be instituted as necessary. Single needles should be used for vaccination and calves should be tested serologically for BLV infection at about 6 months of age.
A marked reduction in the prevalence of infection within the heifer age groups of a dairy herd with a high prevalence can be achieved by:
Biosecurity
Prevention of entry of infection into herd can be achieved by insuring that all imports into the herd have been tested at least 30 d prior to arrival and are seronegative. Control of insect vectors is highly desirable. Blood transfusions and vaccines containing blood, such as those used for babesiosis and anaplasmosis are particularly potent means of spreading the disease and donors must be carefully screened to insure that they are free of the disease. In the future the selection of cattle with inherent resistance to BLV may be a possibility. Embryo transfer from valuable pedigreed seropositive cows may aid in reducing prenatal infection. Insemination is not a method of transmission so that artificial breeding programs are not disrupted.
Vaccine
The possibility of a vaccine for BLV has been explored.2 However, the prospects are not good thus far. A BLV vaccine would have to be non-infectious, non-oncogenic, and should not interfere with the serological tests commonly used to detect infection.
1 Dube S, et al. Virol. 2000;277:379.
2 Johnson R, Kaneene JB. Vet Bull. 1992;62:287.
3 Peleteiro MC, et al. Vet Dermatol. 2000;11:299.
4 Klintevall K. Bovine leukemia virus: course of infection and means of detection. Dissertation Department of Veterinary Microbiology Faculty of Veterinary Medicine Swedish University of Agricultural Sciences and The National Veterinary Institute Department of Virology Uppsala Sweden, 1995.
5 Reichel MP, et al. New Z Vet J. 1998;46:140.
6 VanLeeuwen JA, et al. Can Vet J. 2001;42:193.
7 Gnad DP, et al. Intern J Appl Res Vet Med. 2004;2:215.
8 Trono KG, et al. Vet Microbiol. 2001;83:235.
9 Molnar E, et al. Vet Rec. 2000;146:705.
10 Meas S, et al. J Vet Med Sci. 2000;62:779.
11 Zaghawa A, et al. J Vet Med B Infect Dis Vet Public Health. 2002;49:123.
12 Bielanski A, et al. Vet Rec. 2000;146:255.
13 Choi KY, et al. J Vet Diagn Invest. 2002;14:403.
14 Wentink GH, et al. Vet Rec. 1993;132:135.
15 Divers TJ, et al. Prev Vet Med. 1995;23:133.
16 Meas S, et al. Vet Microbiol. 2000;84:275.
17 Eaves FW, et al. Vet Microbiol. 1994;39:313.
18 Da Y, et al. Proc Natl Acad Sci. 1993;90:6538.
19 Kabeya H, et al. J Vet Med Sci. 2001;63:703.
20 Klintevall K, et al. Comp Immun Microbiol Infect Dis. 1997;20:119.
21 Casal J, et al. Prev Vet Med. 1990;10:47.
22 Chi J, et al. Prev Vet Med. 2002;55:57.
23 Garazi S, et al. Israel J Vet Med. 2001;56:125.
24 Sargeant JM, et al. Prev Vet Med. 1997;31:211.
25 Hendrick SH. Can Vet J. 2002;43:617.
26 Goetschau A, et al. Am J Epidemiol. 1990;131:356.
27 Chi J, et al. Prev Vet Med. 2002;55:137.
28 Ott SL, et al. Prev Vet Med. 2003;61:249.
29 Pollari FL, et al. Am J Vet Res. 1993;54:1400.
30 Rhodes JK, et al. J Am Vet Med Assoc. 2003;223:229.
31 Pollari FL, et al. Can J Vet Res. 1992;56:289.
32 Rhodes JK. J Am Vet Med Assoc. 2003;223:346.
33 Buehring GC, et al. Aids Res Human Retro. 2003;19:1105.
34 Motton DD, Buehring GC. J Dairy Sci. 2003;86:2826.
35 Reed VI. Can Vet J. 1981;22:95.
36 Sparling AM. Can Vet J. 2000;41:315.
37 Alijarrah AH, et al. J Am Vet Med Assoc. 2004;224:1591.
38 Malatestinic A. Can Vet J. 2003;44:664.
39 Wellenberg GJ, et al. Vet Microbiol. 2002;88:27.
40 Dubreuil P, et al. Can Vet J. 1998;39:431.
41 Ivany JM, et al. Can Vet J. 2000;41:486.
42 Oliver-Espinosa O, et al. Can Vet J. 1994;35:777.
43 Southwood LL, et al. Vet Rec. 1996;138:260.
44 Schweizer G, et al. Vet Rec. 2003;153:525.
45 Valentine BA, McDonough SP. Vet Pathol. 2003;40:117.
46 Gutierrez SE, et al. Am J Vet Res. 2001;62:1571.
47 Simard C, et al. Can J Vet Res. 2000;64:96.
48 Simard C, et al. Can J Vet Res. 2000;64:101.
49 Klintevall K, et al. Vet Microbiol. 1994;42:191.
50 Brenner J, et al. Israel J Vet Med. 1994;49:165.
51 Sargeant JM, et al. Prev Vet Med. 1997;31:223.
52 Nagy DW, et al. J Am Vet Med Assoc. 2002;220:1681.
53 Klintevall K, et al. Vet Microbiol. 1994;42:191.
54 Fechner H, et al. J Vet Med Series B. 1996;43:621.
55 Agresti A, et al. Am J Vet Res. 1993;54:373.
56 Nagy DW, et al. J Am Vet Med Assoc. 2003;222:963.
57 Kuckleburg CL, et al. J Vet Diag Invest. 2003;15:72.
58 Wernau W, et al. Vet Pathol. 1997;34:222.
59 Vernau W, et al. Vet Pathol. 1992;29:183.
60 Alexander AN, et al. J Vet Int Med. 1996;10:275.
61 Gibson LAS. Vet Rec. 1995;136:156.
62 Schmity DG, Seahorn TL. J Am Vet Med Assoc. 1990;205:159.
63 Nuotio L, et al. Prev Vet Med. 2003;59:43.