SARCOCYSTOSIS (SARCOSPORIDIOSIS)
Etiology Sarcocystis species. There are a number of species, with a number of different carnivore species as their final host but generally a specific intermediate host.
Epidemiology High prevalence of infection in most areas. Source of infection is feces of carnivore, primarily farm dogs and cats fed raw meat, or other carnivores if they have access to ruminant carcasses.
Clinical findings Severity of disease dose-dependent. The vast majority of infections are sub-clinical. Abortion, depressed growth rate. Neurological disease and ataxia in sheep. Severe infection with some species results in carcass condemnation.
Clinical pathology Anemia and elevated blood concentrations of enzymes associated with tissue damage in acute disease.
Lesions Non-suppurative encephalitis in sheep with neurological signs. Non-suppurative encephalitis, myocarditis, and hepatitis in aborted fetus. Cysts in carcass in chronic cases.
Diagnostic confirmation Identification of parasite microscopically in biopsy or postmortem material.
Treatment and control No effective treatment, amprolium or salinomycin may aid in control. Proper disposal of carcasses. Raw meat not to be fed to farm dogs and cats. Coyote control.
ETIOLOGY
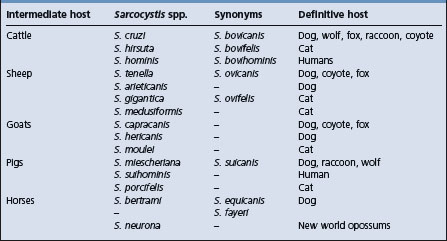
Sarcocystis species are obligate two-host sporozoan parasites in the phylum Apicomplexa. There are a number of species, each with definitive omnivorous or carnivorous species as final hosts. One system of naming the species identifies the intermediate and definitive host in the name, i.e. S. bovifelis, and has been commonly used in the literature. However, currently the organisms are identified by their original names. Table 26.5 shows the currently accepted name of Sarcocystis species of importance in agricultural animals and their definitive hosts.
EPIDEMIOLOGY
Occurrence
In all countries where there have been surveys, the prevalence of infection in cattle, sheep, and horses approaches 100% with a lower, but significant infection rate in swine.1 Clinical disease is rare.
Source of infection
Sarcocystis spp. have an obligatory prey– predator live-host life cycle in which the definitive host is a predator or scavenger.1,2 The carnivorous definitive host becomes infected by ingesting tissue of the intermediate host that contains mature sarcocysts. Following ingestion, bradyzoites are released from the sarcocyst in the stomach and intestine, and they transform into micro- and macrogamonts. The male microgamonts mature to release microgametes, which fertilize the macrogamont to form an oocyst. The oocyst subsequently sporulates to release sporocysts which are passed in the feces and contain infective sporozoite.
The prepatent period is variable, approximately 14 days, and there is no illness in the carnivore host in association with this cycle. However, the replicative cycle of the parasite in the intestine results in the presence of large numbers of sporocysts in the feces and the infection is patent for a long period. Intermediate hosts become infected by ingesting sporocysts in the food or water.1,2
Risk factors
Climate
Sporocysts are not dependent on weather conditions for maturation, and are quite resistant to environmental influences. Under experimental conditions they can survive freezing, but they are susceptible to drying.2 Consequently they can probably overwinter in the environment. Field studies have shown a lower herd prevalence of sarcocystosis in cattle in arid and semi-arid environments compared to cattle from temperate and tropical areas, which is probably a consequence of the relative aridity as well as the lower density of the definitive and intermediate hosts in arid areas.3,4
Species of Sarcocystis
Individual species vary in their pathogenicity and in their ability to produce clinical disease in the intermediate host. In cattle, for example, S. cruzi is considerably more pathogenic than S. hominis.1,2
S. tenella is the most pathogenic species for sheep, and S. capricanis for goats and naturally occurring clinical disease in sheep is not observed with S. gigantea or S. medusiformis.5 There is a strong correlation between the number of sporocysts ingested and the severity of disease. The size of the sarcocyst that occurs in the tissues of the intermediate host also varies with the infecting species. Those from cats and occurring in sheep (S. gigantea, S. medusiformis) or cattle (S. hirsuta) are of particular economic importance, as they produce macroscopically visible sarcocysts that can result in meat condemnation. S. cruzi produces microscopic sarcocysts in muscle and will escape gross detection at meat inspection.
Farm dogs
There is a positive association between herds infected with Sarcocystis and the presence of working dogs on the farm, the practice of leaving carcasses in the field and the feeding of dogs with raw meat,4 and virtually all of the reported clinical cases of sarcocystosis in cattle in the literature record that the dogs on the farm were fed offal or uncooked beef. Housing of dogs and cattle in the same shed or area can have increased risk for infection and clinical disease,1,6 and cattle pastured close to farm buildings where there are dogs are at greater risk. The presence of foxes on farms is also strongly associated with Sarcocystis infection in those herds that leave carcasses on the field.4
Cats
The main risk for cat-associated sarcocystosis is the farm cat that is fed raw meat. Farm cats use hay barns as dens and can contaminate hay and other feedstuffs.7 Feral cats have the potential to distribute sporocysts widely in the grazing environment; however, the presence of feral cats on a farm may not increase the risk for Sarcocystis infection of cattle,4 as scavenged sheep or cattle carcasses are a relatively unimportant part of the diet of feral cats.7
Stocking density
Risk for infection with Sarcocystis is higher with higher stocking densities,4 which may reflect a more intense contamination of pastures by working dogs. Cattle on farms that graze sheep and cattle on the same pastures are less likely to be infected.
PATHOGENESIS
In the intermediate host, sporozoites are released from ingested sporocysts in the small intestine where they penetrate the mucosa and enter the endothelial cells of blood vessels. The stages of schizogony and the distribution of merozoites vary according to the species, but in cattle endothelial infection is followed by parasitemia, with merozoites subsequently localizing in striated muscle and nervous tissue where they develop into sarcocysts. Immature sarcocysts can be found in muscle 45–60 days following ingestion of sporocysts and are infective at about 70 days.2
Schizogony in the endothelial cells of the arterioles and capillaries results in widespread hemorrhage and anemia. Fever is associated with the parasitemia, and in the experimental disease coincides with the time of maturation of the first- and second-generation schizonts.1 The vascular lesion appears to be an essential part of the disease’s pathogenesis. It is proposed that the parasite produces growth retardation as a result of changes in plasma concentrations of somatostatin and growth hormone, and changes in cytokine interactions with the endocrine system.10
The severity of the illness and the degree of infection of tissues at postmortem examination in experimentally induced cases increase with the size of the infective dose. The number of asymptomatic infections probably reflects the early ingestion of a few sporocysts that provoke a strong immunity to later challenge. When groups of animals that have not been exposed to infection previously are suddenly brought into contact with heavy contamination, especially from dogs and cats, outbreaks of clinical disease are likely to occur.
CLINICAL FINDINGS
Infection and disease can occur at all ages. Clinical disease may be more severe where there is intercurrent nutritional stress, and copper deficiency may be an exacerbating factor. Monensin is suspected of being able to potentiate recent infections to cause a severe myositis.11
Cattle
Acute illness is recorded with experimental infections but is rarely seen, or recognized, in the field. Illness commences with a rise in temperature and heart rate, followed by anorexia, anemia, weight loss, a fall in milk production, nervousness, muscle twitching, hypersalivation, lameness, abortion, and, in heavy infections, death. The agent is an occasional cause of non-suppurative encephalomyelitis in cattle and manifest with ataxia and recumbency.
Chronic disease in cattle is manifest by poor weight gains, loss of hair of the neck, rump and the switch of the tail (‘rat-tail’), anemia, and abortion.
Sheep
In sheep, naturally occurring sarcocytosis has been associated with S. tenella and S. arieticanis and presents primarily as a neurological disorder, with muscle weakness, trembling, ataxia of varying severity, followed by hind limb paresis or flaccid paralysis and lateral recumbency. All ages of sheep can be affected, although lambs under 6 months are most susceptible. Attack rates in a susceptible group can be as high as 75% with a high case fatality rate.12-16
Infection may also be manifest with depressed growth, reduced wool growth, and anemia.8,13 Less common manifestations include signs of congestive heart failure associated with endocardial and myocardial infection.17 Infestation of the muscle of the esophagus in sheep is believed a cause of esophageal dysfunction and regurgitation in sheep.18
Swine
Natural clinical disease is not recognized. Sarcocystosis produced experimentally in pigs is manifested by cutaneous purpura on the snout, ears, and buttocks, and dyspnea, tremor and weakness or recumbency.19 There is evidence that breed of pig affects the severity of clinical disease with experimental infections and also the subsequent severity of the parasite load.20
CLINICAL PATHOLOGY
Characteristic laboratory findings in the systemic disease include a responsive anemia, a prolonged prothrombin time, and high titers of antibody to Sarcocystis. Blood creatine phosphokinase, lactic dehydrogenase, and aspartate aminotransferase are significantly elevated.19 An indirect hemagglutination test (IHA) and an ELISA test are available for serological surveys. Titers of antibodies are not high at the time of an acute illness, but are at diagnostic levels 1 week to 3 months afterwards.1,14 An ELISA based on antigens from merozoites has high sensitivity and specificity for detection of infection in individual animals, and a 100% sensitivity for detecting herd infection with small sample size.21 Most animals have been exposed to Sarcocystis spp., and serological examination cannot differentiate clinical disease from asymptomatic infection.
NECROPSY FINDINGS
Emaciation, lymphadenopathy, laminitis, anemia and ascites are present, but the most obvious feature is the petechial and ecchymotic hemorrhages throughout the body.22 There are also erosions and ulcerations in the oral cavity and esophagus, likely as a result of microvascular damage. Microscopically, schizonts are found in endothelial cells throughout the body, and hemorrhages, lymphocytic infiltration, and edema are seen in heart, brain, liver, lung, kidney, and striated muscle.22 Death is probably a result of the severe necrotizing myocarditis that occurs. There is an association between eosinophilic myositis and sarcosporidiosis, but this relationship is not proven in all cases.1
In sheep presenting with neurological disease there may be no findings at gross postmortem, but a non-suppurative encephalomyelitis on histological examination.12,14,15 Aborted bovine fetuses show non-suppurative encephalitis, myocarditis, and hepatitis.5
A number of options are available to achieve a definitive diagnosis of the species present, including animal transmission studies, immunohistochemistry, electron microscopy and PCR. Such techniques are seldom required for routine diagnostic cases.
Samples for confirmation of diagnosis
• Histology – formalin-fixed heart, skeletal muscle (several sites, including tongue, masseter muscle) (LM).
Diagnosis in clinical cases is difficult because of the non-specific signs observed and the widespread prevalence of infection. Sarcosporidiosis is a consideration in the examination of problems of fever and anemia of undetermined origin in cattle and of ill-thrift in cattle and sheep.
Muscle biopsy may aid in the determination of the presence of infection, but still begs the question of its significance to the clinical disease.
The differential diagnoses for abortion in cattle are covered under brucellosis, and sheep under brucellosis. Causes of encephalitis and ataxia in sheep are listed under those headings.
TREATMENT
No approved treatment is available, but amprolium or salinomycin may relieve the signs. Amprolium 100 mg/kg given daily from the time of inoculation reduces the severity of infection in experimentally infected calves and sheep,13,23 and has been used to control an outbreak in sheep.12 Treatment of experimentally infected calves with salinomycin (4 mg/kg BW daily in divided doses for 30 days) reduced the severity of the illness.24 Monensin may have a similar ameliorating effect,9,25 but is also suspected of exacerbating muscle lesions.10 Oxytetracycline, at very high dose rates, and halofuginone may be effective in acute infections.13
CONTROL
Control is difficult as it involves the separation of carnivores and stock, which is not possible on most farms. However, infection in farm dogs and cats could be avoided if all meat fed to them was thoroughly cooked. Freezing will not destroy the infectivity. Coyotes and wild dogs should be controlled and livestock carcasses not left on fields. Prior exposure to small numbers of pathogenic sarcocysts produces a strong immunity, but there is no vaccine available.
Dubey JP, Speer CA, Fayer R. Sarcocystosis of animals and man. Boca Raton, Florida: CRC Press, 1989.
Dubey JP. A review of Neospora caninum and Neospora-like infections in animals. J Protozool Res. 1992;2:40-52.
Anderson ML, Barr B, Conrad PA. Protozoal causes of reproductive failure in domestic animals. Vet Clin North Am Food Anim Pract. 1994;103:439-461.
1 Fayer R, Dubey JP. Sarcocystosis of animals and man. Boca Raton, Florida: CRC Press, 1989.
2 Fayer R, Dubey JP. Comp Cont Educ. 1986;8:F180.
3 Savini G, et al. Epidemiol Infect. 1992;108:107.
4 Savini G, et al. Prev Vet Med. 1994;19:137.
5 Anderson ML, et al. Vet Clin North Am Food Anim Pract. 1994;103:439.
6 Carrigan MJ. Aust Vet J. 1986;63:22.
7 Langham NPE, Charleston WAG. Am J Agric Res. 1990;33:429.
8 Munday BL. Vet Parasitol. 1986;21:21.
9 Dubey JP, et al. J Parasitol. 1989;75(422):980.
10 Fayer R, Elsasser TH. Parasitol Today. 1991;7:250.
11 Jeffrey M, et al. Vet Rec. 1989;124:422.
12 Fitzgerald SD, et al. J Vet Diag Invest. 1993;5:291.
13 Jeffrey M. In Practice. 1993;151:2.
14 Henderson JM, et al. Can Vet J. 1997;38:168.
15 Sargison ND, et al. Vet Rec. 2000;146:225.
16 Caldow GL, et al. Vet Rec. 2000;146:7.
17 Scott PR, Sargison ND. Vet Rec. 2001;149:240.
18 Braun U, et al. Can Vet J. 1990;31:391.
19 Braun U, et al. Can Vet J. 1990;31:392.
20 Reiner G, et al. Vet Parasitol. 2002;106:99.
21 Savini G, et al. Prev Vet Med. 1997;32:35.
22 Johnson AJ, et al. Am J Vet Res. 1975;36:995.
23 Fayer R, Johnson AJ. J Parasitol. 1975;61:932.