Avitaminoses
A vitamin is usually defined as an organic substance not made by the body, which is soluble in either fat or water and is ordinarily needed in only minute quantities to act as a cofactor in a variety of metabolic reactions. The word ‘vitamin’ has reference to the fact that the substance it designates is essential to life. The term, therefore, is functional and not chemically descriptive.
It is useful to consider the vitamins together, for they share certain features. They are present and active in amounts that are minute in contrast to the considerable quantities of the ordinary nutrients. They differ from other nutrients in that many of them are inactivated by heat and oxidation. Some of the vitamins occur in natural sources in a physiologically inactive form. These are called provitamins. They become active only after conversion within the animal. For example, vitamin A exists in plants as carotene, which is activated in the liver. As will be seen with vitamin D, and to a certain extent, with vitamin A, recent evidence points to a hormonal rather than a coenzyme role for certain vitamins. Because of historical convention and the lack of conclusive evidence that vitamins have a hormonal activity, these compounds will be discussed with the remainder of the vitamins.
Although the avitaminoses are as assorted group of diseases, and as unrelated to each other as the chemical constituents of the various vitamins, they too share enough common characteristics to justify their inclusion as a single group of diseases. The avitaminoses are due to the absence of minute amounts of biologically important materials rather than to the presence of minute amounts of biologically active materials (infectious agents). They cause disease not in a positive but in a negative way. The deficiency is the disease. Another characteristic of the deficiency diseases is that they may be present in varying degrees. There may be latent infection, but not a partial infection. A malignant growth is present or it may not. Deficiency diseases; however, may occur in partial form, i.e. they may occur to a mild degree and in their incipient forms the lesions and symptoms might be difficult to recognize. They may also occur in more severe forms, but they are seldom so serious as to be the immediate cause of death.
Fat-Soluble Vitamins
Vitamin A
The therapeutic usefulness of vitamin A has been known since the time of the Egyptian pharaohs. The Ebers papyrus (circa 1500 BC) recommends liver as a cure for night blindness. However, the isolation, synthesis, and recognition of the metabolic functions of vitamin A were not discovered until the 20th century. The classic works of McCollum and Davis, Drummond, and Steenbock and coworkers provided a foundation upon which more recent vitamin A research is based. Moore published an excellent treatise on vitamin A in 1957. There are over 600 carotenoids in nature and approximately 50 of these can be metabolized to vitamin A. β-carotene is the most prevalent carotenoid in the diet that has provitamin A activity. Approximately 80% of preformed vitamin A is absorbed from the diet and the absorption partially depends on adequate bile concentration.
The best known and most intensively studied role for vitamin A is that in vision. George Wald was awarded the Nobel Prize for medicine in 1967 for his discovery of the role of vitamin A in vision, and he has published an excellent review of the subject. Briefly, rhodopsin (visual purple) is formed by the union of vitamin A (11-cis retinal) and a protein, opsin, in the rods of the retina. When stimulated by light, the 11-cis retinal is isomerized to the all-trans retinal form and split form the protein moiety. The electrical potential generated during this process is transmitted to the brain via the optic nerve, resulting in visual sensation. In the dark, the all-trans form is enzymatically isomerized back to the 11-cis form and subsequently binds to opsin, thus completing the cycle. A continuous supply of vitamin A is therefore necessary for rod (low-light) vision, and the first manifestation of vitamin A deficiency is an impaired, low-light vision, i.e. night blindness.
Current research indicates that in addition to its role in vision and lysosomal stability, vitamin A may have a hormonal function in the regulation of epithelial differentiation. Intracellular receptors have been identified and may transport vitamin A molecules to the cell nucleus, where they interact with DNA to direct cellular differentiation.
The classic work of Wolbach and Howe on the dental changes in vitamin A deficiency of the rat and guinea pig was confirmed and elaborated by Schour and his coworkers. Excellent reviews have been written by Frandsen and Jolly. Most of our knowledge of the dental effects of vitamin A deficiency is based on findings in the continuously developing and erupting incisor tooth of the rat.
It is well established that vitamin A is concerned primarily with the process of differentiation of epithelial cells. In vitamin A deficiency the epithelial cells fail to differentiate. This means that the cells in the basal layer lose their specificity and tend to form a stratified squamous epithelium with keratin production, independent of the type of cell previously formed by the basal cells. Thus one of the basic changes is a keratinizing metaplasia of epithelial cells. This occurs throughout the body, including the mucous membranes of the trachea, conjunctiva, and ureter, and the salivary and other glands (Fig. 15-7).
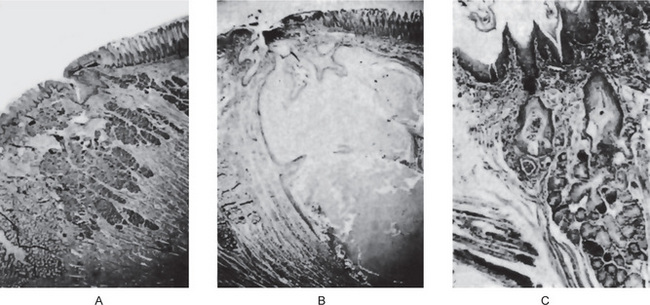
Figure 15-7 Vitamin A deficiency.
Photomicrographs of the tongue of a normal rat (A) and of vitamin A-deficient rat (B, C). There is squamous metaplasia in the mucous glands of the tongue and a large cyst filled with keratinaceous material (B).
In the developing tooth of the rat that is deficient in vitamin A, the odontogenic epithelium fails to undergo normal histodifferentiation and morphodifferentiation, and the result is an increased rate of cell proliferation. Therefore, epithelial invasion of pulpal tissue is characteristic in vitamin A deficiency.
In young rats whose mothers are maintained on a diet deficient in vitamin A for five months preceding their birth, changes are more severe, resulting in a distortion of the shape of both the incisors and the molars. Since the enamel forming cells are disturbed, enamel matrix is arrested and/or poorly defined so that calcification is disturbed and enamel hypoplasia results. The dentin, too, is atypical in structure, lacking the normal tubular arrangement and containing cellular and vascular inclusions. Harris and Navia reported an increase in caries susceptibility of the rat molars of pups nursed by vitamin A-deficient dams, indicating a pre-eruptive role for vitamin A in tooth development. Post-eruptive vitamin A deficiency has been reported to result in higher caries scores. However, Salley and coworkers posited that this increase in caries may be due to changes in salivary gland function rather than to dental changes per se.
The teeth of animals on a vitamin A deficient diet contain less total ash than the teeth of normal animals. Eruption rate is retarded, and in prolonged deficiencies eruption ceases completely. The alveolar bone is retarded in its rate of formation. The gingival epithelium becomes hyperplastic and in prolonged deficiencies shows keratinization. This tissue is easily invaded by bacteria that may cause periodontal disease and microabscess formation. The major and minor salivary glands undergo the typical keratinizing metaplasia. This is characteristic, of course, of all the epithelial cells in vitamin A deficiency. Most of the changes described are reversible with the feeding of vitamin A to deficient animals.
Requirements
The recommended daily dietary allowance for vitamin A ranges from 420 mcg to 800–1000 mcg of retinol equivalents (RE) for adolescent and adult females and males (1 RE = 1 mcg retinal or 6 mcg β-carotene). Pregnant and lactating females should increase their daily intake by 200 and 400 mcg RE, respectively.
Clinical Features of Vitamin A Deficiency
If the deficiency is mild, the manifestations in man are night blindness, xerophthalmia, and keratomalacia. Hyperkeratotic changes in the oral epithelium of adults have also been noted. Follicular keratotic changes have been described in naturally occurring vitamin A deficiency by Frazier and Hu and by Sweet and K’ang. Hume and Krebs, and Steffens and coworkers studied controlled vitamin A deficiency in humans and were able to produce cutaneous manifestations in only one patient.
As it progresses, keratinizing metaplasia appears in the trachea and bronchi, kidney, pelvis, conjunctiva, cornea, salivary glands, and genitourinary tract. Documented autopsy studies have been published by Wilson and DuBois and by Blackfan and Wolbach. If vitamin A deficiency were to cause changes in the human tooth bud, the deficiency state would have to occur before the sixth year of life, since by that time the crowns of all the teeth except the third molars are completely formed. The only cases of changes in human tooth buds attributable to vitamin A deficiency are those described by Boyle and by Dinnerman. Their findings were similar to those described in the rat incisor tooth in vitamin A deficiency. An excellent symposium on vitamin A deficiency and its clinical implications may be found in the Federation Proceedings for 1958.
Measurement of serum retinol (normal range: 30–65 mg/ dl), tests of dark adaptation, impression cytology of conjunctiva, and measurement of body storage pools either by liver biopsy or by isotopic dilution are the various investigations for vitamin A deficiency.
Hypervitaminosis A
Cases of hypervitaminosis A in children are reported with increasing frequency. Gradual loss of hair and dryness of skin, lips, and oral mucosa are the common findings. If it persists, pigmentation, erythema, follicular keratosis, and purpura develop. The syndrome in children is characterized by anorexia, low-grade fever, hepatomegaly, sparse hair, and increased vitamin A serum levels. Radiographs of the long bones show fragmentation of the distal fibular epiphyses and pronounced periosteal thickening. Furman has reported a case of adult hypervitaminosis A.
Vitamin D
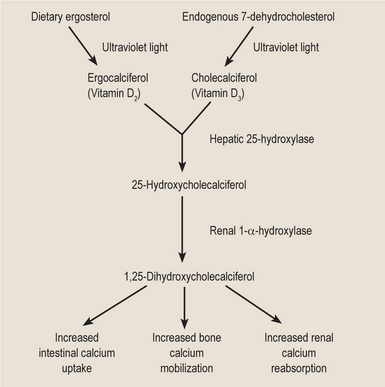
Vitamin D (1,25-dihydroxycholecalciferol) is one of a number of compounds that are grouped together as the hydroxylated cholecalciferols. Vitamin D is commonly referred to as the antirachitic vitamin, although a variety of biochemical analogs have similar activity, e.g. vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol). Mellanby demonstrated in 1919 that rickets could be produced experimentally and prevented by cod liver oil administration. Shortly thereafter, McCollum and coworkers distinguished the antirachitic factor from the previously discovered vitamin A in cod liver oil. Finally Steenbock reported in 1924 that antirachitic activity could be produced in food and animals by exposing them to ultraviolet radiation.
The metabolism and action of vitamin D have been widely described and will not be repeated in detail here. A schematic representation is provided in Figure 15-8. An excellent review of this subject has been published by Haussler and McCain.
Vitamin D has always been classified as a vitamin; however, it is probably best thought of as a hormone. Unlike a true vitamin, the hydroxylated cholecalciferols are not essential nutrients. Vitamin D3 is formed from 7-dehydrocholesterol, which is an intermediate compound in the synthesis of cholesterol. 7-Dehydrocholesterol is ultimately formed from acetyl-CoA, which is never in short supply. The hydroxylated cholecalciferols have the same basic biochemical structure as the steroid hormones, and they control calcium ion concentration in a manner similar to sodium and potassium ion concentration regulation by the mineralocorticoids. Also, vitamin D is not required in many cell cultures. Finally, vitamin D exerts its major influence by combining with nonhistone proteins in the nuclei of intestinal epithelial cells. This combination, in turn, exposes a portion of the genetic material for transcription of a specific protein, calcium-binding protein.
Relationship to Calcium and Phosphorus Homeostasis
A discussion of vitamin D is incomplete without mentioning its relationship to calcium and phosphorus homeostasis. In its role as an activator of calcium-binding protein, vitamin D has protean manifestations in parathyroid function, which subsequently affect calcium and phosphorus levels in the body. Hypervitaminosis D, as seen in overzealous food faddists, results in hypercalcemia with irreversible renal and vascular damage. Hypovitaminosis D, although now uncommon because of dietary fortification, can and does result in secondary hyperparathyroidism. Parathyroid hormone levels are elevated, and serum calcium levels are maintained at the expense of bone calcium. Serum phosphate levels are decreased as a result of the effect of parathyroid hormone on renal excretion of phosphate. Serum alkaline phosphatase levels are increased due to the bones’ attempt at reformation. Dietary calcium forms insoluble calcium phosphates in the intestines because of its increased concentration.
Requirements
The recommended daily dietary allowance of vitamin D from infancy through puberty is 10 mcg of cholecalciferol (400 IU of vitamin D). Rickets can be prevented and growth will proceed at a normal rate with significantly less vitamin D (2–5 mcg of cholecalciferol), provided adequate amounts of calcium and phosphorus are present in the diet. Calcium uptake will be reduced slightly (25–30% compared with 35–40%) with decreased vitamin D intake. The recommended daily dietary intake tapers off to 7.5 mcg in young adulthood and should be maintained at 5 mcg after the age of 25. Pregnant and lactating females should increase their daily intake by 5 mcg.
Vitamin D-deficient Rickets
In common parlance, rickets refers to any disorder in the vitamin D-calcium-phosphorus axis which results in hypomineralized bone matrix, i.e. a failure of endochondral calcification. It should be realized that such a defect may result from a number of etiologies; thus there are a variety of forms of ‘rickets’. A comprehensive review of rickets has been published by Pitt and Haussler.
Historically, vitamin D-deficient rickets developed in urban areas that were deprived of adequate sunlight. When air pollution filters out the ultraviolet portion of the spectrum, cholecalciferol formation is blocked. Infants rapidly develop the characteristic bony deformities. Identical lesions are seen in sun-rich areas where the diet is high in phytate, which binds the available dietary calcium. Social customs, e.g. the use of the purdah, may also result in rickets. The age of onset of the deficiency is important in the eventual morbidity, with premature infants being at highest risk. Although the incidence of rickets in Western societies has been drastically decreased because of food fortification with irradiated ergosterol, e.g. ergocalciferol in milk, Richards and coworkers have reported an incidence of radiographic changes consistent with rickets in 9% of young children in Glasgow, Scotland.
Clinical Features
Rat is the laboratory animal commonly used for the experimental investigation of rickets. The effects of rickets are reflected only in the bones and teeth of the afflicted animal. The changes in the bones are found in the epiphyseal plate, the metaphysis, and the shaft. Since the degree of change encountered depends on the rate of growth of the bones at the time of the deficiency, young animals are more seriously affected than older animals.
In young rats placed on rachitogenic diets, the first change seen is the cessation of calcification of their epiphyseal disks. Since the intercellular ground substance does not become calcified, the cartilage cells are not denied nutrition. Therefore, they do not die, and their continued growth and multiplication lead to an increase in the width of the disk. The disk thickens irregularly because some focal areas usually calcify. The osteoblasts continue to lay down osteoid around the bone and cartilage spicules in the metaphysis, as well as beneath the periosteum in the region of the metaphysis and other areas of the shaft. The changes in the ribs and long bones of children with rickets are essentially the same as those described for the rat. Since undermineralized bone is not as capable of supporting weight as normal bone, children with rickets show bowing of the legs.
Oral Manifestations
Mellanby was the first to report the effects of rickets on the teeth, which included developmental abnormalities of dentin and enamel, delayed eruption, and misalignment of the teeth in the jaws. Her later work showed that affected teeth had a higher caries index than those of controls. In human rachitic teeth there is an abnormally wide predentin zone and much interglobular dentin. Although many reports are found in the literature linking rickets with enamel hypoplasia, infantile rickets does not always result in hypoplastic enamel. The eruption rate of the deciduous and permanent teeth; however, is retarded in rickets.
Osteomalacia: (Adult rickets)
Osteomalacia is the adult equivalent of juvenile (vitamin D-deficient) rickets. Unlike juvenile rickets, only the flat bones and the diaphyses of the long bones are affected. The disease is most commonly seen in postmenopausal females with a history of low dietary calcium intake and little exposure to ultraviolet light. This disorder is endemic in certain areas of India, Japan, and China. Malabsorption is also a commonly reported etiology.
Clinical Features
Essentially there is a remodeling of bone in the absence of adequate calcium, which results in a softening and distortion of the skeleton and an increased tendency towards fracture. Pelvic deformities are commonly seen in affected multiparous females.
Oral Manifestations
Taylor and Day have reported a 50% incidence of severe periodontitis in a series of 22 Indian women with osteomalacia. These data are questionable in view of the prevalence of endemic periodontal disease in this population group.
Radiographic Features
Radiologically there are severe asymmetric deformities of all stress-bearing bones, e.g. the pelvis, spine, and long bones of the legs. Longitudinal hairline fractures are seen in the long bones.
Histologic Features
The histologic findings in osteomalacia, like those in rickets, are nonspecific. There is an attempt at bone remodeling with inadequate calcification of bone matrix. The cortical bone is thin and osteoid borders are found on the trabeculae.
Treatment and Prognosis
The treatment (and for that matter prevention) of osteomalacia consists of dietary enrichment of vitamin D, usually in the form of milk, and the certainty of adequate dietary calcium. Hormonal therapy and fluoride administration have also been reported to be useful in the treatment of the disease. If the osteomalacia is secondary to malabsorption, the daily dietary fat intake must be severely restricted. While the mortality associated with osteomalacia is negligible, the morbidity is prominent and related to the extent of the disease at the time of initial diagnosis. Complications may arise from long bone fractures and compression of the spinal vertebrae.
Vitamin D-resistant Rickets: (Familial hypophosphatemia, refractory rickets, phosphate diabetes)
A number of isolated renal tubular defects, associated with an inability to reabsorb certain metabolites such as water, phosphate, calcium, and potassium have been recognized. Some defects in reabsorption may lead to rickets or osteomalacia. Albright and coworkers first described a case of vitamin D-resistant rickets in 1937. Shortly thereafter, Christensen described a familial pattern of occurrence. Twenty years after its initial description, Winters and colleagues and Graham and coworkers proposed that the disorder was an X-linked dominant defect in renal phosphate metabolism. A large series of cases has been investigated by Stickler and associates.
The disease is now recognized as a specific disorder characterized by:
• Hypophosphatemia and hyperphosphaturia associated with decreased renal tubular reabsorption of inorganic phosphates.
• Familial occurrence, being inherited as an X-linked dominant trait.
• Rickets or osteomalacia which does not respond to the usual doses of vitamin D.
• Normocalcemia with high-normal parathyroid hormone levels.
• Diminished intestinal calcium and phosphate absorption.
• Decreased growth with short stature.
This definition excludes conditions such as sporadic, nonfamilial vitamin D-resistant rickets and familial vitamin D-resistant rickets associated with normal or high serum concentration of inorganic phosphate.
Clinical Features
The mildest form of this disease is a simple hypophosphatemia without clinical manifestation other than a slight decrease in the height of the patient as compared with a normophosphatemic sibling. In hypophosphatemic adults the varying degrees of deformities due to rickets in childhood constitute more serious disturbances, such as bowing of the legs, shortening of stature, continuing osteomalacia, and the presence of pseudofractures.
In children affected with this form of resistant rickets, the disease is usually first recognized when the child begins to walk. The history or X-ray examination, however, might reveal abnormalities such as skull deformities; retardation of eruption of teeth and ‘sitting’ deformities of the legs. Such children have usually received prophylactic doses of vitamin D but have failed to respond. Permanent deformities and short stature are often present.
Among family members with hypophosphatemia, females show considerably less bone disease than males. Few patients have the muscular weakness and atony which are so prominent and frequent in vitamin D-deficient rickets.
Oral Manifestations
Vitamin D-resistant rickets has marked effects on the teeth and supporting structures. These have been discussed in detail by many workers including Marks and his associates, Archard and Witkop, Tracy and his associates, Vasilakis and coworkers, Ainley, and Cohen and Becker.
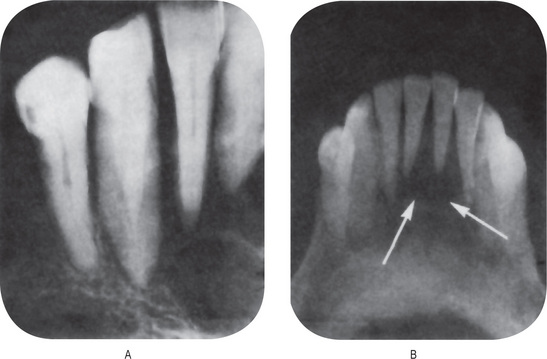
Characteristically, there is histologic evidence of widespread formation of globular, hypocalcified dentin, with clefts and tubular defects occurring in the region of the pulp horns. In addition, these pulp horns are elongated and extend high, often reaching nearly to the dentinoenamel junction. This may even be evident on the radiograph (Fig. 15-9). Because of these defects, there is commonly invasion of the pulp by microorganisms without demonstrable destruction of the tubular matrix. Following this, there is often periapical involvement of grossly normal-appearing deciduous or permanent teeth, followed by the development of multiple gingival fistulas. In addition to abnormal cementum, the lamina dura around the teeth is also reported to be frequently absent or poorly defined on the radiograph, and the alveolar bone pattern is often abnormal.
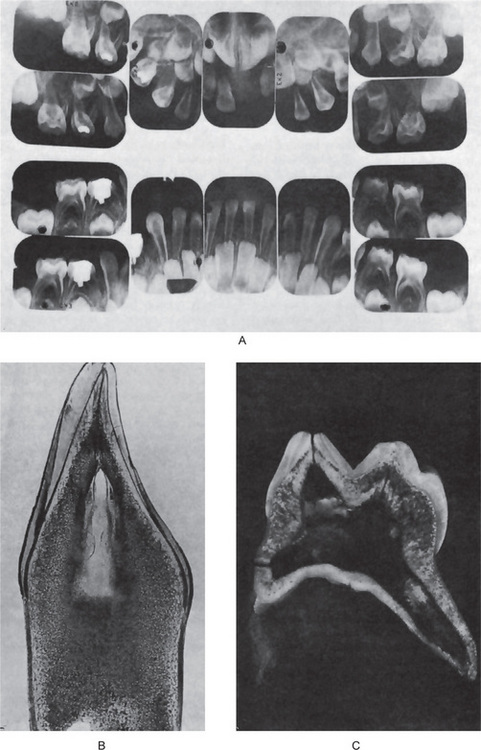
Figure 15-9 Vitamin D-resistant rickets in a boy six years of age.
The full mouth radiographs (A) show the wide root canals and pulp chambers. A ground section of an incisor tooth (B) shows the interglobular nature of the dentin. The deciduous molar (C) when split shows the relatively small quantity of dentin as well as the poor quality of the dentin. Note the connection between the pulp chamber and the occlusal surface of the tooth, a common finding in this disease, accounting for the frequent pulp infection and periapical involvement without the presence of a carious lesion. Courtesy of Dr SS Arnim
Histologic Features
Alterations are found primarily in the cartilage plate and shaft of the long bones and are characterized by a failure of bone salts to be deposited in the cartilage matrix between the rows of hypertrophic cells, so that these cells are not invaded and destroyed by capillaries. The histologic picture is characterized by a broad zone between the multiplying cartilage cells and the shaft, the so-called rachitic metaphysis. This is composed of tongues of cartilage which extend down toward the shaft and are separated from one another by collections of capillaries. This zone contains trabeculae made up of uncalcified cartilage matrix upon which osteoid has been deposited. Since osteoblastic activity is not affected, osteoid is found deposited on pre-existing bony trabeculae. The calcification is interfered with, so the osteoid does not calcify and is not remodeled.
Treatment and Prognosis
The treatment of vitamin D-resistant rickets is highly individualized. Massive doses of vitamin D frequently result in repair, but the risk of hypervitaminosis D in such cases is considerable. Success has been reported using 25-hydroxycholecalciferol in lower dosages than conventional vitamin D (10,000–25,000 IU per day of 25-hydroxycholecalciferol, as opposed to 50,000–100,000 IU per day of vitamin D). Healing of the rickets can be initiated by measures other than prescribing massive doses of vitamin D. Such methods include immobilization and administration of large amounts of phosphate. Decreased dosages of vitamin D (15,000–50,000 IU per day) combined with supplemental oral phosphate have been used successfully.
Renal Rickets: (Renal osteodystrophy)
Painful, crippling bone disease is a common finding in patients with chronic renal disease. Renal rickets results from the inability of diseased kidneys to synthesize 1-α-hydroxylase and convert 25-hydroxycholecalciferol to the active form of vitamin D. Calcium absorption in the intestines is impaired, with a dramatic increase in fecal calcium excretion and negative calcium balance. Secondary hyperparathyroidism may lead to a superimposed osteitis fibrosis cystica.
Treatment and Prognosis
Renal osteodystrophy is refractory to physiologic doses of vitamin D. Kaye and Sagar have reported success in treating renal rickets with dihydrotachysterol, a vitamin D analog. Catto and coworkers have administered 1-α-hydroxycholecalciferol and reported good treatment success. The prognosis for the bone disease is guarded because of the inability to cure the underlying renal disease. Renal transplant patients function adequately after an initial post-transplantation hypercalcemia.
Hypophosphatasia: (Hypophosphatasemia)
Hypophosphatasia, a hereditary disease first recognized as an entity by Rathbun in 1948, is transmitted as a recessive autosomal characteristic. Since then many cases have been reported and several reviews of the disease presented. One such excellent review, that of Bruckner and his associates, stressed the dental findings in this condition as observed in a series of cases. Ritchie, Haupt and associates, Kjellmann and coworkers, Beumer and colleagues, Brittain and coworkers, and Witkop and Rao have discussed in detail the oral manifestations of hypophosphatasia.
The basic disorder is a deficiency of the enzyme alkaline phosphatase in serum or tissues and excretion of phosphoethanolamine in the urine. The severity of disease is not directly related to serum alkaline phosphatase levels. There is an interesting similarity of many aspects of this disease to the condition known as ‘vitamin D-resistant rickets with familial hypophosphatemia’.
Clinical Features
On the basis of clinical manifestations and chronology of the appearance of bone disease, hypophosphatasia is divided into three clinical forms: infantile, childhood, and adult. The infantile form is manifested by severe rickets, hypercalcemia, bone abnormalities, and failure to thrive. Most of these cases are lethal. Hypophosphatasia of childhood is characterized by premature exfoliation of deciduous teeth, increased infection, growth retardation and rachitic-like deformities, including deformed extremities, costochondral junction enlargement (rachitic rosary), and failure of the calvarium to calcify. Pulmonary, gastrointestinal, and renal disorders are also present. The adult form includes spontaneous fractures, prior history of rickets and osseous radiolucencies.
Oral Manifestations
The earliest manifestation of the disease may be loosening and premature loss of deciduous teeth, chiefly the incisors. There are varying reports of gingivitis; however it does not appear to be a consistent feature of the disease.
Radiographic Features
The metaphyses of long bones have been described as showing ‘spotty’, ‘streaky’, or ‘irregular ossification’. Dental radiographs generally reveal hypocalcification of teeth and the presence of large pulp chambers, as well as alveolar bone loss; however, these findings have not been consistently reported.
Histologic Features
The long bones characteristically exhibit an increased width of proliferating cartilage with widening of the hypertrophic cell zone, irregularity of cell columns, irregular penetration of the cartilage by marrow with persistence of numerous cartilage islands in the marrow, and formation of large amounts of osteoid which is inadequately calcified. These findings are indistinguishable from those in true rickets.
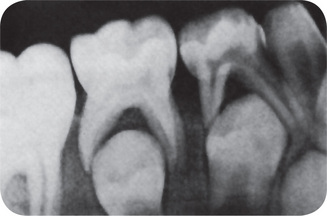
The teeth present a unique appearance characterized by the absence of cementum, presumably as a result of failure of cementogenesis, so that there is no sound functional attachment of the tooth to bone by periodontal ligament (Fig. 15-10). This lack of attachment is thought to account for the early spontaneous exfoliation of the deciduous teeth. Occasional foci of poorly formed cementum may be found on some teeth.
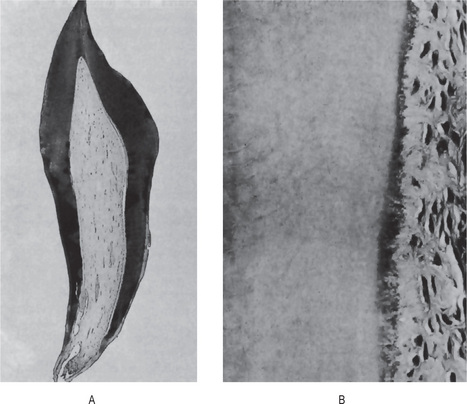
Figure 15-10 Hypophosphatasia.
A maxillary deciduous incisor of a patient with hypophosphatasia which was exfoliated at 15 months of age (A). The tooth root showed only a poor attempt at cementogenesis indicated by the granular, basophilic material between the dentin on the left and the periodontal fibers on the right (B) Courtesy of Dr Robert J Bruckner.
Treatment
Therapeutic measures are generally unsuccessful. Vitamin D in high doses has resulted in partial improvement in some cases, but this may lead to deposition of calcium in many tissues, including the kidney. Bongiovanni and coworkers have reported that administration of high oral doses of phosphate results in moderate improvement in bone calcification as judged radiologically.
Pseudohypophosphatasia
A disease resembling classic hypophosphatasia but with a normal serum alkaline phosphatase level has been reported by Scriver and Cameron. Patients afflicted by pseudohypophosphatasia exhibit osteopathy of the long bones and skull, premature loss of deciduous teeth, hypotonia, hypercalcemia, and phosphoethanolaminuria. Only the alkaline phosphatase level remains normal. This disease also appears to be hereditary. Méhes and coworkers have reported the appearance of hypophosphatasia and pseudohypophosphatasia in the same kindred. This suggests that the two diseases may represent variations of a basic metabolic effect.
Vitamin E
Sixty years ago Evans and Bishop noted that a fat-soluble factor prevented fetal resorption in animals. This factor was named vitamin E and given the generic name of tocopherol, which means ‘the alcohol which brings forth offspring’. Olcott and Emerson soon recognized the antioxidant properties of vitamin E. The main function of vitamin E is to prevent peroxidation of polyunsaturated fatty acids. Vitamin E consists of eight naturally occurring tocopherols of which alpha-tocopherol is the most active.
Vitamin E deficiency in experimental animals results in multisystem disorders, including decreased male fertility, impaired fetal-maternal vascular relationships, nutritional muscular dystrophy, and encephalomalacia, increased vascular disruption, and hemolysis. All of these disorders can be attributed in part to the increased peroxidation of unsaturated fatty acids in vitamin E-deficient animals. Irving has described a loss of pigment and atrophic, degenerative changes in the enamel organ of vitamin E-deficient rats.
Dietary vitamin E deficiency does not occur. It is seen only in severe and chronic diseases like celiac disease or after the resection of small intestine or in children with cystic fibrosis. Infants are born with low levels of vitamin E and are particularly susceptible to vitamin E deficiency, especially if they are fed diets high in polyunsaturated fatty acids. Hassan and coworkers have described this syndrome, which consists of edema, desquamating erythematous papular dermatitis, thrombocytosis, and anemia. Chronic steatorrhea, for example, as it occurs with cystic fibrosis, results in hypovitaminosis E and is manifested by muscular dystrophy-type symptoms, with elevated serum creatinine phosphokinase activity and creatinuria. This secondary vitamin E deficiency has been discussed by Nitowsky and coworkers.
Requirements
The recommended daily dietary allowance for vitamin E ranges from 3 mg of d-α-tocopherol for infants to 10 mg for adult males. Increased intake in pregnant and lactating women is suggested, especially in view of the low perinatal levels of vitamin E in the infant. The average intake of vitamin E in the United States is 15 mg per day; therefore, deficiency states are rare in the absence of underlying steatorrhea and malabsorption.
Vitamin E has gained a great deal of public and scientific attention in the past decade because of its role as a polyunsaturated fatty acid antioxidant. One of the prevailing theories of aging states that aging is, in part, a progressive accumulation of cellular damage resulting from free radicals. As an antioxidant, vitamin E may play a role in the prevention of free radical damage. This subject has been reviewed by Pryor. There are interesting but inconclusive animal studies to support this particular aging hypothesis. Unfortunately, publication of these results prompted megadose consumption of vitamin E by ill-advised members of the lay public. Based on the lack of toxic symptoms in nutrition faddists, vitamin E is thought by Farrel and Bieri to be one of the least toxic of the vitamins.
Water-Soluble Vitamins
Vitamin K
In 1929, Dam noticed a peculiar hemorrhagic diathesis in chicks fed a fat-extracted diet. This clotting defect was not due to a deficiency of vitamin A, D, or E, which had previously been discovered. The new substance was named vitamin K or ‘Koagulation vitamin’. Like other fat-soluble vitamins, vitamin K is absorbed from the gut and is transported to the liver via lymph chylomicrons.
Dam and his coworkers later provided evidence that vitamin K was intimately involved in both the extrinsic and intrinsic systems of coagulation, particularly with prothrombin (factor II) synthesis. Other investigators have since shown a role for vitamin K in the regulation of levels of factors VII, IX, and X (proconvertin, Christmas factor, and Stuart-Prower factor, respectively). Prior to Dam’s discovery of vitamin K, Schofield had described a hemorrhagic disease in cattle, which had consumed spoiled clover. Campbell and coworkers later described this vitamin K antagonist and identified it as dicumarol. A coumarin analog, warfarin is commonly used as an anticoagulant in both humans and animals.
There are two natural forms vitamin K, namely vitamin K1, also known as phylloquinone, derived from vegetable and animal sources and vitamin K2 or menaquinone, synthesized by bacterial flora and found in hepatic tissue.
Vitamin K3 or menadione is a chemically synthesized provitamin and is water soluble. This is converted into menaquinone by the liver. For this reason vitamin K is discussed under water-soluble vitamins.
Vitamin K is necessary for the post-transitional carboxylation of glutamic acid necessary for calcium binding to gamma carboxylated proteins such as prothrombin, factors VII, IX, X, protein C, protein S, and proteins found in the bone.
Vitamin K is found in green leafy vegetables, butter, margarine, liver, milk, and also in vegetable oils.
Primary vitamin K deficiency is rare in humans; however, newborns are particularly susceptible to vitamin K deficiency, and hypoprothrombinemia due to poor placental lipid transmission and a lack of vitamin K-synthesizing gastrointestinal flora may ensue. Secondary hypovitaminosis K may occur in adults with impaired fat absorption, which may accompany obstructive jaundice, sprue, ulcerative colitis, and surgical bowel resection. Iatrogenic deficiency of vitamin K may occur secondary to antibiotic sterilization of the gut.
The most common oral manifestation of vitamin K deficiency is gingival bleeding. Prothrombin levels below 35% will result in bleeding after toothbrushing; however, when prothrombin levels fall below 20%, spontaneous gingival hemorrhages will occur.
Requirements
The minimum daily dietary requirement of vitamin K is estimated to be between 1–2 mcg/kg, depending on the amount of gut bacterial production of the vitamin. The ‘normal mixed diet’ in the United States is estimated to contain 300–500 mcg of vitamin K, which is more than enough to meet minimum daily requirements.
The diagnosis of vitamin K deficiency is usually made on the basis of an elevated prothrombin time or reduced clotting factors. It is usually treated using a parenteral dose of 10 mg.
Menadione, the water-soluble form of vitamin K, has been reported to cause hemolytic anemia and hypobilirubinemia in infants when given parenterally in large doses. Toxicity from dietary vitamin K derivatives has not been reported.
Vitamin C
Vitamin C has been the object of intensive research for many years. Scurvy, which results from vitamin C deficiency, has been known since the time of the Ebers Papyrus in Egypt (1500 BC). The effect on history, through the occurrence of scurvy in military troops, is notable. British sailors in the 19th century were referred to as ‘limeys’ because of their consumption of citrus fruits to prevent scurvy while on long voyages. Hodges and coworkers have described the changes seen in experimental scurvy in man, and an excellent review has been written by Lloyd and Sinclair.
Svirbely and Szent-Gyorgyi isolated hexuronic acid (ascorbic acid) in 1928 and reported the results in 1932. A similar isolation procedure was reported by King and Waugh in 1932. Within two years the structure of vitamin C was determined and synthesized. Interestingly, most animals are capable of synthesizing their own vitamin C. Burns has postulated that humans, monkeys, and guinea pigs are incapable of endogenous vitamin C production owing to an inability to convert L-gulonolactone (a glucose metabolite) to L-ascorbic acid. Because of this inherent defect, guinea pigs are the animal model of choice in studying scurvy. It also aids in the promotion of nonhem iron absorption, carnitine biosynthesis, and the conversion of dopamine to norepinephrine. It is richly present in citrus fruits, green vegetables, tomatoes, and potatoes.
Vitamin C is necessary for a number of metabolic processes, including hydrogen ion transfers and maintenance of intracellular oxidation reduction potentials. It also acts as an antioxidant, facilitates iron uptake in the intestinal tract, and is involved in the formation of folinic acid (the active form of the folic acid). Standinger and associates and Goldberg have reported that ascorbic acid is critical in hydroxylation reactions which require reduced iron or copper. Its role in the hydroxylation of proline in collagen synthesis has been described by Peterkovsky and Udenfriend. Tryptophan, norepinephrine, and tyrosine metabolism all require vitamin C.
In general, the action of vitamin C appears to be to further the normal development of intercellular ground substances in bone, dentin and other connective tissues, since all signs of the deficiency of ascorbic acid are associated with disturbances in these tissues.
The dental changes in scorbutic guinea pigs are so consistent and characteristic that Hojer and Crampton devised biologic assay methods for vitamin C by grading the histologic changes in the mandibular incisor. The characteristic change in the teeth of scorbutic guinea pigs is the atrophy and disorganization of the odontoblasts, resulting early in the deficiency state in the production of irregularly laid down dentin with few, irregularly arranged tubules. Eventually dentin formation ceases, and the predentin becomes hypercalcified, producing a heavy, basophilic staining line between dentin and pulp. The odontoblasts finally become indistinguishable from other pulpal cells (Fig. 15-11).
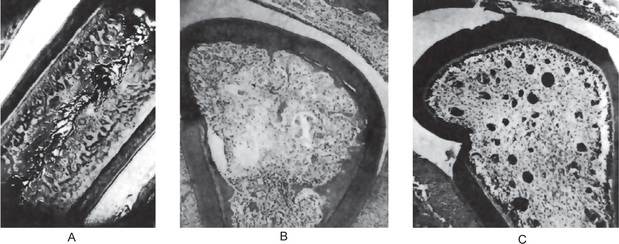
Figure 15-11 Vitamin C deficiency.
Photomicrographs of incisor teeth of guinea pigs with incomplete or early vitamin C deficiency showing the abnormal irregular dentin, (A) longitudinal and (B) cross-section. The odontoblasts eventually fail to lay down dentin (C).
In scorbutic monkeys, hypertrophy of the gingiva covering in the entire crowns of the teeth was reported by Goldman. In some cases subperiosteal hemorrhages lifted the gingiva from the underlying bone. Focal areas of necrosis of the free margin of the gingiva also occurred. The alveolar bone showed atrophic changes, and the marrow spaces were replaced by fibroblasts growing in an edematous space.
Requirements
The recommended dietary intake for vitamin C ranges from 35 mg in infants to 60 mg in adults. Pregnant and lactating women should increase their daily intake by 20 mg and 40 mg, respectively.
Clinical Features of Scurvy
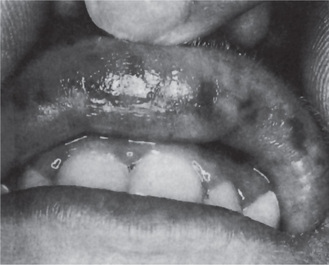
The oral effects of vitamin C deficiency in humans occur chiefly in the gingival and periodontal tissues. The interdental and marginal gingiva is bright red with a swollen, smooth, shiny surface. In fully developed scurvy the gingiva becomes boggy, ulcerates and bleeds. The color changes to a violaceous red. In infants the enlarged tissue may cover the clinical crowns of the teeth (Fig. 15-12). In almost all cases of acute or chronic scurvy the gingival ulcers show the typical organisms, and the patients have the typical foul breath of persons with fusospirochetal stomatitis. In the severe chronic cases of scurvy, hemorrhages into and swelling of the periodontal membranes occur, followed by loss of bone and loosening of the teeth, which eventually exfoliate.
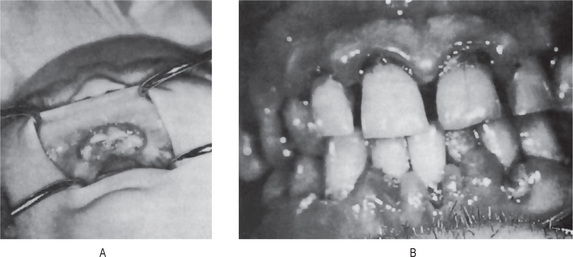
Figure 15-12 Vitamin C deficiency or scurvy in an infant (A) and an adult (B). A, Courtesy of Dr EV Zegarelli; B, Courtesy of Dr ER Costich.
Boyle studied the deciduous and permanent tooth germs of scorbutic infants and found only small cysts and minute hemorrhages in some specimens.
Vitamin C is a threshold substance and is excreted primarily through the kidney. The degree of tissue saturation is the factor which determines the amount excreted. If intake has been normal, a slight increase in intake above normal will be excreted. If, on the other hand, the tissues are undersaturated through low intake or through excess metabolism of vitamin C, even high doses may be largely retained.
The role of ascorbic acid in collagen formation has been extensively studied from many aspects. It has been found that wounds produced in scorbutic guinea pigs fail to heal properly. Although there is fibroblastic proliferation in the wound area, the fibroblasts appear immature and fail to produce collagen. They do form a fluid-like material around themselves, representing an ineffectual attempt at collagen formation.
Histologic Features
The bone changes in scurvy were well reviewed by Follis in his book on the Pathology of Nutritional Disease. He pointed out that in scurvy the osteoblasts fail to form osteoid. The cartilage cells of the epiphyseal plate continue to proliferate in normal fashion, and salts are deposited in the matrix between the columns of cartilage cells. But the osteoblasts fail to lay down osteoid on the spicules of calcified cartilage matrix. In addition, the calcified matrix material is not destroyed, so that a wide zone of calcified but nonossified matrix, called the scorbutic lattice, develops in the metaphysis. The spicules are nonresistant to weightbearing and motion stresses, and they are therefore liable to fracture. The changes which accompany the fractures lead to the characteristic lesions of the skeleton in scurvy.
As the ‘lattice’ increases in width, a more and more fragile zone develops, so that eventually complete fracture of the spicules occurs with separation and deformity of the cartilageshaft junction. This fracturing of the calcified matrix material leads to the classic picture of scurvy, the so-called Trümmerfeldzone or region of complete disintegration. About the fractures and clefts there are pink-staining hyaline material, immature-looking fibroblasts and macrophages containing hemosiderin. The area beneath the Trümmerfeldzone is free of hematopoietic cells and is made up of connective tissue cells, the so-called Gerüstmark. The reason for the migration of marrow cells out of the area, leaving only connective tissue elements, is not clear. In addition, subperiosteal hemorrhages are frequent in scorbutic animals.
Symptoms of scurvy respond well within few days to few weeks to administration of vitamin C. Food rich in vitamin C may lower the incidence of certain cancers like gastric or esophageal by preventing the conversion of nitrites and secondary amines to nitrosamines.
Since vitamin C may be metabolized to oxalate, any higher doses of vitamin C supplementation could result in an increased prevalence of kidney stones.
Vitamin B Complex
Unlike the oral manifestations of vitamin A deficiency and the other vitamin deficiencies heretofore described, the oral signs of deficiencies of the B vitamins occur primarily in the oral soft tissues: the tongue, mucous membranes, gingiva, and lips. Since much of our knowledge of the avitaminoses B is derived from clinical observation, the mechanism of action, and the histologic details of the oral lesions associated with the various vitamin B deficiencies still remain to be elucidated.
At present the vitamin B group contains 11 well-characterized vitamins: thiamin, riboflavin, niacin, pyridoxine, pantothenic acid, biotin, folic acid, vitamin B12, inositol, para-aminobenzoic acid, and choline. Nearly every one of these vitamins forms part of a coenzyme essential for the metabolism of proteins, carbohydrates, or fats.
The B-complex vitamins are needed by all living cells, but with the exception of nicotinic acid and choline, animal tissues are incapable of synthesizing them. The B vitamins must therefore be absorbed from the intestinal tract either from ingested food or from the products of the intestinal flora, or from both.
Most B-complex vitamins occur in nature in bound form within the cells of vegetable or animal tissues. These cellular structures must therefore be broken down by the digestion for the liberation of the vitamin and its eventual absorption from the gut. With the possible exception of vitamin B12, the vitamins of the B complex are not stored in any appreciable amount in the tissues of the body, so if the intake exceeds the requirement, the excess is excreted in the urine.
Although the functions of individual vitamins, whether fator water-soluble, vary greatly, vitamins tend to occur together in nature to some extent. It should be remembered, therefore, that though a lesion induced by the elimination of single vitamin from an experimental diet may occur in experimental animals and may even be induced in human subjects, lesions occurring naturally are probably associated with a deficiency of many of the essential nutrients. We are seeing only the most prominent clinical symptom and not the entire patient when we observe an angular cheilosis and assume that it is due to riboflavin deficiency. We must also remember that, though the most frequent cause of a nutritional deficiency is decreased intake of the essential nutrient, impaired absorption from the alimentary canal, failure of utilization by the tissues, inadequate storage, increased metabolism due to rapid growth, fever, pregnancy, and other factors all contribute to clinical deficiency states.
Pathologic conditions other than deficiency states may impose special demands for vitamins. Adequate nutrition is obviously important in the treatment of disease, but the diet must be governed by the nature of the disturbance. The indiscriminate use of the B vitamins is of no value in the treatment of general ill health.
Thiamin (vitamin B1) is a colorless basic organic compound composed of a sulfated pyrimidine ring. It is readily absorbed from both the small and large intestines. It is phosphorylated mainly by the liver and to a lesser extent by the kidney. In tissues, thiamin is found as thiamin pyrophosphate (cocarboxylase), rarely as free thiamin. The main sources of thiamin are yeast, pork, legumes, whole grains, and nuts.
Thiamin pyrophosphate is required for carbohydrate and branched chain amino acid metabolism. In addition, it acts as coenzyme for transketolase reaction that mediates the conversion of hexose and pentose phosphates. It also plays a role in peripheral nerve conduction but the exact mechanism is unknown.
Clinical Features of Thiamin Deficiency
In man, thiamin deficiency leads to beriberi, which is generally insidious in onset, chronic in course and sudden death may occur. Beriberi may be of two types: wet and dry. In either form, patients may complain of pain and paresthesia. Wet beriberi manifests with cardiovascular symptoms due to impaired myocardial energy metabolism, dysautonomia, cardiomegaly, highoutput cardiac failure, peripheral edema, and peripheral neuritis. In dry beriberi, same symptoms occur but for the edema.
Alcoholic patients with chronic thiamin deficiency are having CNS manifestations known as Wernicke’s encephalopathy, which consists of horizontal nystagmus, ophthalmoplegia, cerebral ataxia, and mental impairment. Along with the abovementioned symptoms, if there is loss of memory and confabulatory psychosis, it is known as Wernicke-Korsakoff syndrome.
Requirements
The recommended daily dietary allowance for thiamin ranges from 0.3 mg for infants to 1.5 mg for young adults. Pregnant and lactating women should increase their daily intake by 0.4 mg and 0.5 mg, respectively.
There is no convincing evidence that thiamin exerts an influence on oral tissues. There are reported cases of oral manifestations of thiamin deficiency, but they are not supported by the experience of volunteer human subjects who lived on diets containing very low levels of thiamin for six months and showed no oral lesions.
Riboflavin
Riboflavin (vitamin B2) is a fully dialyzable, intensely yellow water-soluble pigment which is decomposed by light. It fluoresces green under ultraviolet illumination, is readily absorbed from the intestinal tract and is phosphorylated in the walls of the intestine as well as in other tissues of the body.
Riboflavin is a constituent of two different groups of coenzymes, riboflavin 5′-phosphate (flavin mononucleotide or FMN) and flavin adenine dinucleotide (FAD). These coenzymes are essential to the oxidative enzyme systems utilizing the electron transport system. It is essential for carbohydrate, fat, and protein metabolism reflecting its role as respiratory coenzyme and electron donor.
The riboflavin deficiencies are almost always due to dietary deficiency. Its requirement is increased during pregnancy, lactation, and heavy exercise.
Requirements
The recommended daily dietary allowance for riboflavin ranges from 0.4 mg for infants to 1.7 mg for young adults. Pregnant and lactating women should increase their daily dietary intake by 0.3 mg and 0.5 mg, respectively.
Clinical Features of Riboflavin Deficiency
Riboflavin deficiency is particularly common among children who do not drink milk. In endemic areas, the incidence is greater during the spring and summer months than in other seasons.
A long period of vague, nondescript symptoms usually precedes the appearance of diagnostic lesions. The diagnostic lesions of ariboflavinosis are usually limited to the mouth and perioral regions. The oral manifestations of the disease are well recognized, since they have been experimentally produced by Sebrell and Butler in 18 healthy women placed on a riboflavindeficient diet. Although the exact mechanism involved in the production of the oral lesion is not understood, the clinical stages have been clearly defined.
In the mild deficiency state there is a glossitis which begins with soreness of the tip and/or the lateral margins of the tongue (Fig. 15-13). The filiform papillae become atrophic, while the fungiform papillae remain normal or become engorged and mushroom shaped, giving the tongue surface a reddened, coarsely granular appearance. The lesions extend backward over the dorsum of the tongue. In severe cases the tongue may become glazed and smooth, owing to complete atrophy of all papillae. In many cases the tongue has a magenta color which can be easily distinguished from cyanosis.
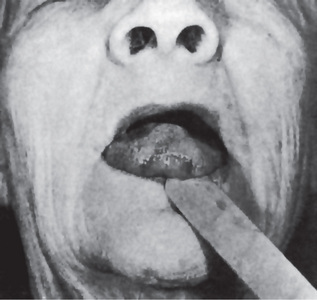
Figure 15-13 Riboflavin deficiency.
The atrophy of the filiform papillae gives the tip of the tongue a smooth, almost ulcerated appearance.
Paleness of the lips, especially at the angles of the mouth, but not involving the moist areas of the buccal mucosa, is the earliest sign of the deficiency disease. The pallor, which usually continues for days, is followed by cheilosis, which is evidenced by maceration and fissuring at the angles of the mouth. The fissures may be single or multiple. Later the macerated lesions develop a dry yellow crust which can be removed without causing bleeding. The lips become unusually red and shiny because of a desquamation of the epithelium. As the disease progresses, the angular cheilosis spreads to the cheek. The fissures become deeper, bleed easily and are painful when secondarily infected with oral and/or skin microorganisms. Deep lesions leave scars on healing. The gingival tissues are not involved.
Riboflavin deficiency also affects the nasolabial folds and the alae nasi, which exhibit a scaly, greasy dermatitis. A fine scaly dermatitis may also occur on the hands, vulva, anus, and perineum. Ocular changes, consisting of corneal vascularization, photophobia, and a superficial and interstitial keratitis, have also been described. Considering that flavoproteins are widely distributed throughout the body, it is surprising that the lesions are so well localized.
In the differential diagnosis of ariboflavinosis, it is important to remember that bilateral angular cheilosis is a nonspecific lesion. Older people with greatly decreased vertical dimension, either through faulty dentures or through attrition of the natural dentition, frequently show the nonspecific angular cheilosis.
Niacin
In the living organism, ingested niacin is transformed into nicotinic acid amide, which is utilized to form coenzyme I (nicotinamide-adenine dinucleotide, or NAD) and coenzyme II (nicotinamide-adenine dinucleotide phosphate, or NADP). A deficiency of this vitamin leads to the classic symptoms of pellagra in human beings and to black tongue in dogs.
Pellagra as a widespread problem in the southeastern United States has largely disappeared. Spies and Butt formulated a working hypothesis of the pathogenesis of the disease as follows.
When the available nicotinic acid amide or compounds with similar functions are not adequate to supply the needs of the body for reasons of decreased supply, inadequate assimilation, increased demand, or increased loss, a disorder in respiratory enzyme systems occurs. As a result a state of generalized reduction in normal cellular respiration supervenes. When this biochemical lesion is severe enough, or has existed long enough, it is translated into functional disturbances in various organ systems of the body. Vasomotor instability in the skin, functional disorders of the alimentary canal, the nervous system, and the circulatory system may occur. It is probable that the most readily affected systems are those weakened by hereditary predisposition or trauma in the wear and tear of everyday life. This may explain the infinite variety of the clinical picture. Finally severe or persisting alterations in physiology lead to structural changes in various tissues which ultimately present the diagnostic lesions of pellagra.
A metabolic interrelationship between the amino acid tryptophan and nicotinic acid has been demonstrated in a number of mammalian species including man. Pyridoxal-5-phosphate is required for the conversion of tryptophan into nicotinic acid in the tissues. The accepted conversion ration (niacin equivalents) is 60 mg tryptophan to 1 mg. nicotinic acid. It is important in pentose, steroid, and fatty acid biosynthesis, glycolysis, protein metabolism and oxidation of lactate, pyruvate, and alcohol.
Clinical Features of Pellagra
The mucous membrane lesions affecting the tongue, oral cavity, and vagina are usually the earliest lesions diagnostic of the disease. Other lesions common in pellagra are the typical dermal lesions of bilaterally symmetric, sharply outlined, roughened, keratotic areas (Fig. 15-14). (The word ‘pellagra’ means rough skin). Mental symptoms and weight loss also occur.
In the prodromal stage of nicotinic acid deficiency, the patient may complain of loss of appetite and vague gastrointestinal symptoms. General weakness, lassitude, mental confusion, forgetfulness, and other ill-defined symptoms develop. The patient then usually complains of a burning sensation in the tongue, which becomes swollen and presses against the teeth, causing indentations. The tip and lateral margins of the tongue become red.
In the acute stages of pellagra, the entire oral mucosa becomes fiery red and painful. The mouth feels as though it had been scalded. Salivation is profuse. The epithelium of the entire tongue desquamates. Tenderness, pain, redness, and ulcerations begin at the interdental gingival papillae and spread rapidly. Superimposed necrotizing ulcerative gingivostomatitis or Vincent’s infection involving the gingiva, tongue, and oral mucosa is a common sequel.
Epithelial changes followed by the characteristic skin rash particularly in the areas exposed to sunlight especially in the neck region are called Casal’s necklace. Vaginitis and esophagitis may also occur.
Pantothenic Acid
The role of pantothenic acid in metabolic processes is not at all clear. It is a constituent of coenzyme A and is widely distributed in foods. Since no evidence of human pantothenic acid deficiency has been recorded, the human requirement for this vitamin is unknown. 5–10 mg per day is considered adequate for children and adults.
Pyridoxine
Pyridoxine (vitamin B6) is actually a complex of three related substances: pyridoxine, pyridoxal, and pyridoxamine. Pyridoxine is the most active compound when ingested.
Pyridoxine plays an important role in protein metabolism, since pyridoxine-deficient animals placed on a high protein diet exhibit the characteristic lesions sooner and die more quickly than animals whose pyridoxine-deficient diets contain smaller amounts of protein. This vitamin has also been shown to be involved in tryptophan metabolism. If young dogs deficient in pyridoxine are fed tryptophan, both kynurenine and xanthurenine acid are excreted in the urine. If pyridoxine is fed to the animals, xanthurenic acid is not excreted, but kynurenine and kynurenic acid are found in the urine. Thus, pyridoxine apparently determines whether xanthurenic acid or kynurenic acid will be excreted after tryptophan feeding. The finding of xanthurenic acid in the urine has been suggested as a biologic test for vitamin B6 deficiency.
Hawkins and Barsky reported that mental depression, mental confusion, albuminuria, and leukopenia occurred in normal people placed on a pyridoxine-deficient diet. The oral lesions of experimentally induced pyridoxine deficiency bear a striking resemblance to pellagrous stomatitis.
In some people with angular cheilosis, pyridoxine administration will effect a cure when riboflavin and nicotinic acid will not.
The minimum daily dietary allowance for pyridoxine is 2.0 mg for adults. Pregnant and lactating women should increase their daily intake by 2.5 mg.
Choline
Choline is an important constituent of lecithin, certain sphingomyelins, and acetylcholine. Little is known of choline requirements, since the need for choline is dependent on their sources of methyl groups in the diet, especially methionine.
Choline deficiency per se probably does not occur. It is possible; however, in cases in which general dietary protein is low to postulate a deficiency of choline and its precursor, methionine. Diets high in choline, methionine, and proteins are used in the treatment of fatty liver and cirrhosis, especially in chronic alcoholics; however, the results have not been promising.
No oral lesions have been ascribed to choline deficiency in man.
Biotin
It is unlikely that biotin deficiency ever develops spontaneously in man. In animals, biotin deficiency is characterized by a scaly, greasy dermatitis, and eventual alopecia. No dental changes are described in biotin-deficient animals.
Inositol
Although inositol has been shown to be necessary for growth in experimental animals, no histologic studies have been reported on animals depleted of inositol. Little is known of its role in animal or human nutrition.
Folic Acid
Various macrocytic anemias, sprue, addisonian (pernicious) anemia, and macrocytic anemia of infancy respond well to folic acid. Folic acid is essential for the growth of many animal species and is also essential in man. The primary function of folic acid is the transfer of one-carbon moieties in a number of metabolic reactions. Folic acid is also necessary for purine synthesis, the conversion of homocysteine to methionine, and the conversion of uridylate to thymidylate. The synthesis of DNA is impossible in the absence of folic acid.
Clinical Features of Folic Acid Deficiency
Folic acid deficiency in man is characterized by glossitis, diarrhea, and macrocytic anemia. The glossitis appears initially as a swelling and redness of the tip and lateral margins of the dorsum. The filiform papillae are the first to disappear, the fungiform papillae remain as prominent spots. In advanced cases, the fungiform papillae are lost and the tongue becomes slick, smooth, and either pallid or fiery red in color. These are the toxic symptoms following aminopterin therapy for leukemia. Aminopterin interferes with the conversion of folic acid to folinic acid. Administration of folic acid in aminopterin toxicity quickly alleviates the glossitis and reverses the symptoms of gastrointestinal disturbances.
The minimum daily dietary allowance for folic acid ranges from 30 mcg in infants to 400 mcg in adults. Pregnant women should double their daily intake, while nursing mothers should increase their intake by 25%.
Vitamin B12
This vitamin includes a group of closely related compounds, the most common form being cyanocobalamin. It is the antipernicious anemia factor, and it has also been used in trigeminal neuralgia with some success. Massive doses, 1000 mcg daily, must be used for the treatment of trigeminal neuralgia.
The minimum daily dietary allowance for vitamin B12 ranged from 0.5 mcg in infants to 3.0 mcg in adults. Pregnant and lactating women should increase their intake by 30%.
Disturbances in Hormone Metabolism
No tissue in the mammalian body is exempt from some sort of hormonal influence, either in the course of its development and growth or in its functional activities. Yet the chemical structures of most hormonal substances are either unknown or only partially defined. Physiologic investigations of the hormones have been centered on their more specific actions, but it is becoming evident that the spheres of action of the hormones are extremely broad and reach far beyond the limits implied by the tissue of origin and its known interrelations with other organs and tissues. As Pincus points out, the expected action of ovarian estrogen as a promoter of female reproductive tract growth and of estrous behavior is accompanied by many activities outside of the reproductive sphere. Estrogens are hair-and bone-growth regulators; they are thymolytic, mitogenetic in the epidermis, enzyme-inhibitory in the adrenal cortex, phagocyte-stimulating, alkalosis-inducing, tumorigenic, antigoitrogenic, and antihyperglycemic. Similar multiplicities of action may be listed for most of the known hormones.
We can readily note that the hormones vary tremendously in chemical composition and in biologic activity. They are united only by their definition as internal secretions.
Over 50 biologically active substances circulate continuously in the blood of mammals as hormones; yet, with few exceptions, these substances are not essential for life. In the rat, for example, neither thyroidectomy, gonadectomy nor hypophysectomy is fatal. Yet, after such operations, the rates of certain processes are reduced to a minimum and cannot be speeded up if the need arises. Although animals with an inadequate hormone balance may live, their mental and physical vigor, their adaptability and drive, are gone or reduced. The mental dullness of the hypothyroid person is a good example of the influence of hormonal defect on the optimal rate of living.
Much experimental work has been done on the symptom complex production as a result of the removal of one or more endocrine glands. Studies after the injection of the active principle of one or more of the endocrine glands, either into an intact animal or into an animal from which an endocrine gland or glands had previously been removed, have added tremendously to the literature on the mode of action of the hormones. In addition, the treatment of human symptoms indicating a deficiency of a particular hormone with the hormone preparation has added much to our knowledge of endocrinology.
With the accelerating increase of literature on the physiology and biochemistry of the hormones, any attempt to review the field would be overwhelming for both the writer and the reader. We will, therefore, restrict our observations to the oral aspects of the disturbances in hormone metabolism.
Pituitary Group of Hormones
The pituitary is considered the master gland of the body. Harvey Cushing’s admirable words adequately describe its function. He stated, “Here in this well-concealed spot, almost to be covered by a thumb nail, lies the very mainspring of primitive existence, vegetative, emotional, and reproductive”.
The pituitary lies in the sella turcica of sphenoid bone beneath the middle cranial fossa. It is bounded anteriorly by the sphenoid sinus, posteriorly by the dorsum sellae, and superiorly by the diaphragma sellae. The gland is surrounded by a rich blood supply. The cavernous sinuses, which form the lateral boundary, drain pituitary hormones through the hypophyseal vein. The pituitary consists of an anterior lobe and a posterior lobe. The cells of the anterior pituitary are divided based on the staining reactions into agranular chromophobes and granular chromophils. The chromophils are further divided into acidophils and basophils.
The anterior lobe is derived from Rathke’s pouch and is therefore epithelial in origin. The posterior lobe develops from the floor of the third ventricle and is composed of nervous tissue. The anterior lobe is glandular in structure and is the active part of the organ. To date, at least six hormones have been identified as coming from the anterior pituitary, namely, somatotropic, thyrotropic, adrenocorticotropic, two gonadotropic, and lactogenic hormones. In addition, the anterior lobe is said to have ketogenic, anti-insulin, diabetogenic, parathyrogenic, and pancreatotropic activity. Removal of the pituitary gland brings the entire internal secretory system into discordance because of a progressive atrophy of all the endocrine glands except, possibly, the parathyroids.
Although the physiologic activity of the posterior lobe has never been proved, extracts of this lobe have a remarkably high pharmacologic potency. Three types of activity, vasoconstrictive, oxytocic, and antidiuretic, have been reported.
Experiments in which the pituitary is removed or in which crude extracts of the gland are injected can give little information about which particular hormone is responsible for the effects observed. Precise information can be obtained only by studying the response of an animal to purified hormones. The evidence indicates that the growth hormone is mainly responsible for the effect of pituitary extracts on teeth, but that the thyrotropic hormone also plays a role. The entire spectrum of human growth hormone has been reviewed by Root.
A few workers have studied the relation of the pituitary gland to dental development, notably Schour and Van Dyke and Baume, Becks and associates. Working with rats, they found that after hypophysectomy there was a progressive retardation of eruption of the incisor tooth, which eventually ceased to erupt. The tooth attained only about two-thirds normal size and showed a distortion of form, especially at the basal end. When an extract of the anterior lobe of the pituitary was injected into the hypophysectomized rats, the eruption rate of the incisor tooth returned to normal.
Becks and his associates pointed out that the only constant pathognomonic sign of hypophysectomy in the rat was a thickening of the dentinal walls at the expense of the pulp chamber (Fig. 15-15). Baume and his associates reported that amelogenesis, and particularly the activities of the odontogenic epithelium, depended directly on the secretion of the anterior pituitary, whereas dentinogenesis and cementogenesis were able to proceed at a depressed rate without the pituitary hormones. They also pointed out some interesting interrelations. They suggested that the histologic changes in the enamel organ of the incisors of hypophysectomized rats were comparable to those of thyroidectomized animals of an equal postoperative interval. They also called attention to the similarity of the folding of the apical third of the incisor tooth of hypophysectomized animals to the changes in the teeth of magnesium-deficient animals, and they suggested that the changes in hypophysectomy may be related to salt and mineral metabolism, thus implicating the adrenal gland and its mineralocorticoids.
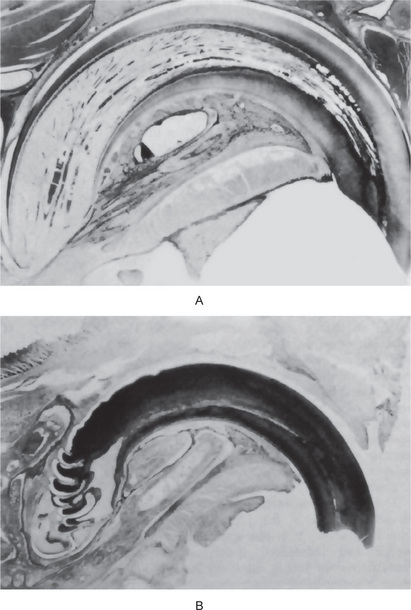
Figure 15-15 Hypophysectomy.
Photomicrographs of a maxillary incisor tooth of a normal rat (A) and of a hypophysectomized rat (B). Courtesy of Dr Herman Becks.
Collins and coworkers showed that the chronic administration of pure growth hormone to hypophysectomized animals allowed the incisors to erupt, but at only half the normal rate. The ameloblasts showed evidence of atrophy, but the dentin formed at a rate of 10μ instead of the normal 16μ per day. Baume and his associates injected thyroxin into hypophysectomized animals, either alone or with purified growth hormone. Their findings led them to the following explanation. The pituitary gland influences eruption not only with its thyrotropin, but also with its growth hormone. The effects of thyroxin on dental growth and development are quantitatively and qualitatively different from those of the pituitary growth hormone. Quantitatively, thyroxin is the factor which stimulates eruptive movement and tooth size, but it has little influence on alveolar growth. Growth hormone, on the other hand, spurs dental as well as alveolar growth, but has little effect on eruption rate. It is also possible that other endocrine organs, by virtue of their effects on metabolic interrelations, also affect tooth development and eruption.
Hypopituitarism
In man, some indication of the role played by the pituitary in the development of the oral tissue can be gained from studies of hypopituitarism as well as hyperpituitarism. Hypopituitarism is caused by compression or atrophy of anterior pituitary cells or defect in the hypothalamic control of hormonal secretion. Before puberty, the hypofunctioning leads to dwarfism, which mainly manifests with features of growth hormone deficiency. After puberty, it affects other endocrine glands also. Some of the common causes of hypopituitarism, which occur after puberty, are pituitary adenoma, Simmonds’ disease or hypophyseal cachexia, and Sheehan’s syndrome (pituitary infarction in the postpartum woman). Hypofunction of posterior lobe leads to deficiency of vasopressin, resulting in diabetes insipidus.
Clinical Features
The typical evidences of hypopituitarism resulting in pituitary dwarfism are a diminutive but wellproportioned body, fine, silky, sparse hair on the head and other hairy regions, wrinkled atrophic skin, and often, hypogonadism. The deficiency may be congenital, or it may be due to a destructive disease of the pituitary, such as an infarct occurring before puberty. There is no distinctive pattern to the basal metabolism in this disease.
In pituitary dwarfs the eruption rate and the shedding time of the teeth are delayed, as is the growth of the body in general. The clinical crowns appear smaller than normal because, even though eruption does occur, it is not complete. The dental arch is smaller than normal and therefore cannot accommodate all the teeth, so that a malocclusion develops. The anatomic crowns of the teeth in pituitary dwarfism are not noticeably smaller than normal, contrary to what might be expected in light of the animal experiments. There are no reports of a careful statistical study of crown size in dwarfism. The roots of the teeth are shorter than normal in dwarfism, and the supporting structures are retarded in growth. The osseous development of the maxilla is not as retarded as that of the mandible.
Hypopituitarism in the adult is usually due to an infarction of the pituitary called Simmonds’ diseases. It is characterized by loss of weight and diminished sexual function. The basal metabolic rate is markedly lowered, and since Simmonds’ disease represents a panhypopituitarism, there is a decrease in the activity of the many hormones of the pituitary gland and of those glands that are under pituitary regulation. In this disease, the skin shows atrophic alterations. Changes in the head include thin eyebrows, loss of eyelashes, sharp features, thin lips, and an immobile expression. There will be a decreased salivary flow due to hypofunctioning of salivary glands which leads to increased caries activity and periodontal disease.
Diagnosis
Radiograph and CT scan are used to diagnose structural abnormalities. Growth hormone assay may also be performed. Regular and early evaluation is required to correct skeletal and dental malocclusions. Fluoride treatment should be initiated and supplementary corticosteroids should be administered during minor oral surgical procedures.
Hyperpituitarism
An increase in the number of granules in the acidophilic cells or an adenoma of the anterior lobe of the pituitary is associated with gigantism or acromegaly. If the increase occurs before the epiphyses of the long bones are closed, gigantism results; if the increase occurs later in life, i.e. after epiphyseal closure, acromegaly develops. The clinical manifestations depend upon the type of cell involved. Somatotrophic adenoma or hyperplasia is also created with elevated levels of growth hormone. It exerts its effect by stimulating hepatocyte, chondrocytes, myoblasts, kidney, and GIT to secrete somatomedin-C, which is the primary promoter of growth.
Clinical Features
Gigantism is characterized by a general symmetric overgrowth of the body, some persons with this disturbance attaining a height of over 8 feet (Fig. 15-16). Later in life such people usually show genital underdevelopment and excessive perspiration, and they complain of headache, lassitude, fatigue, muscle and joint pains, and hot flashes. It is also characterized by the presence of broad, enlarged nose, thick and furrowed oily skin. Organomegaly and hypertension are common findings. Skeletal changes include frontal bossing and prognathic mandible. Increased glove, ring, and shoe size indicates the changes in the hands and feet. Patient may develop class III malocclusion with interdental spacing. Hypercementosis is a common finding in the intraoral radiographs.

Figure 15-16 Pituitary gigantism.
This patient was 7 feet 9 inches tall and weighed over 400 pounds Courtesy of Dr Rohini Sivapathasundharam, Ambattur, Chennai.
The teeth in gigantism are proportional to the size of the jaws and the rest of the body. The roots may be longer than normal.
Acromegaly is a relatively rare disease in which there is hypersecretion by the anterior lobe, the influence being effected after ossification is complete. The following symptoms occur in acromegaly: temporal headaches, photophobia, and reduction in vision. The terminal phalanges of the hands and feet become large. The ribs also increase in size.
The lips become thick and Negroid. The tongue also becomes enlarged and shows indentations on the sides from pressure against the teeth. Microscopically, the surface epithelium and the connective tissues are hyperplastic.
The mandible, because of accelerated condylar growth, becomes large. The resulting prognathism may be extreme, giving the head a typical acromegalic appearance (Fig. 15-17). The teeth in the mandible are usually tipped to the buccal or labial side, owing to the enlargement of the tongue.
Thyroid Hormone
The thyroid gland is situated in the middle of neck, and has two lobes connected by an isthmus. The functional unit of thyroid gland, the thyroid follicle secretes thyroxine and triiodothyronine.
Administration of the thyroid gland or its derivatives, including thyroxine, causes an increased uptake of oxygen by the body as a whole. The precise cellular and enzymatic mechanism for its effect is not known. It is probably not due to increased glycolysis. In addition to increasing oxygenation, the thyroid hormone influences a variety of other actions which affect almost every other function and tissue of the mammalian body. It therefore plays an essential role in differentiation, growth, maturation, water balance, electrolyte balance, protein storage, carbohydrate and lipid metabolism, and other physiologic functions.
Calcitonin, a hypoglycemic polypeptide secreted by the ‘C’ cells of the ultimobranchial elements of the thyroid gland, has gained increasing attention in recent years. It has been isolated, identified, sequenced, and more recently, synthesized. Calcitonin is responsive to hypercalcemia and acts to lower the plasma calcium level. Another of its actions is to inhibit resorption of bone mineral. Its use in human disease is still under active investigation. Excellent reviews of this hormone have been published by Foster, Hirsch and Munson and by Rasmussen and Pechet.
Hypothyroidism
A failure of thyrotropic function on the part of the pituitary gland or an atrophy or destruction of the thyroid gland per se leads to an inability of the thyroid to produce sufficient hormone to meet the requirements of the body. If this failure occurs in infancy, cretinism results. If it occurs in the child, juvenile myxedema occurs; if in the adult, myxedema results. Myxedema is not a rare disease. Hospital records show that 4–8 of every 10,000 admissions enter with myxedema.
Clinical Features
Congenital hypothyroidism, or cretinism, leads to mental defects, retarded somatic growth, generalized edema and other changes, depending on the severity of the deficiency of thyroid hormone. The dentofacial changes in cretinism are also related to the degree of thyroid deficiency. Usually, the base of the skull is shortened, leading to a retraction of the bridge of the nose with flaring. The face is wide and fails to develop in a longitudinal direction. The mandible is underdeveloped, and the maxilla is overdeveloped. The hair is sparse and brittle; the fingernails are brittle, and the sweat glands are atrophic.
The dental changes in juvenile hypothyroidism have been reviewed by Hinrichs, who also presented 36 cases. He indicated that the longer the time between the onset of the disease and the institution of treatment, the greater is the likelihood that the developing dentition will be affected. However, with a few exceptions, he found no striking morphologic changes in the teeth of the patients in his series.
Characteristically, the tongue is enlarged by edema fluid. It may protrude continuously, and such protrusion may lead to malocclusion. The eruption rate of the teeth is delayed, and the deciduous teeth are retained beyond the normal shedding time. Myxedema, the disease produced by thyroid deficiency in adults or children, is usually caused by atrophy of the thyroid gland of unknown etiology. The metabolic rate is lowered, although this finding should not be used as a diagnostic test for myxedema. Concentration of serum protein-bound iodine and radioactive iodine uptake or excretion studies are the diagnostic tests of value.
The myxedematous swelling is probably an extravascular, extracellular accumulation of water and protein in the tissues. The protein has a greater osmotic effect than the serum proteins, accounting for the increased blood protein concentration and decreased plasma volume which are found in myxedema.
The clinical orofacial findings in myxedematous patients are apparently limited to the soft tissues of the face and mouth. The lips, nose, eyelids, and suborbital tissues are edematous and swollen. The tongue is large and edematous, frequently interfering with speech.
Hyperthyroidism
There is apparently some debate as to whether hyperthyroidism should be considered a single disease entity or should be subdivided into several types. Boothby and Plummer described two fundamentally different types of hyperthyroidism:
• Exophthalmic goiter, characterized by diffuse hyperplasia of the thyroid and by eye signs.
• Toxic adenoma, in which hyperfunction originates in a benign tumor of the thyroid gland.
In either case, we are concerned with the manifestations of excess circulating thyroid hormone.
Most of the symptoms of hyperthyroidism are due to an increased metabolic activity of the tissues of the body. This is usually manifested as an increased basal metabolic rate. The serum protein-bound iodine concentration is elevated. The urinary iodine excretion is reduced because of the increased iodine uptake by the thyroid gland.
Clinical Features
Patient may exhibit tremor, tachycardia, sweating, weight loss, nervousness, muscle weakness, heat intolerance, and exopthalmic goiter.
Alveolar atrophy occurs in advanced cases. In children, shedding of the deciduous teeth occurs earlier than normal, and eruption of the permanent teeth is greatly accelerated.
Patients suffering from hyperthyroidism usually present a facial expression of surprise or excitement, with wide-eyed staring. Such patients are nervous and highly emotional; they have increased sensitivity to epinephrine and are usually hypertensive. Thoma warns that they make very poor dental patients.
Gonadal Hormones
Little is known of the relation of disturbances of metabolism of the sex hormones to oral pathology. Some evidence suggests that certain imbalances of estrogenic hormones might be reflected in the oral mucosa. In some women, gingivitis occurs periodically with abnormal or difficult menstruation. During menopause, the oral epithelium is said to become thinner than normal. In some patients a burning sensation of the tongue occurs during or after the menopause, while in other patients a ‘dry feeling’ of the mouth is observed with or without an actual diminution of saliva.
Shafer and Muhler demonstrated a diminution in size and in number of granular tubules in the submaxillary glands of rats after either gonadectomy or the administering of estrogenic substances. Much further work is necessary to clarify the effects of the sex hormones on the oral and dental tissues.
Parathyroid Hormone
Parathyroid glands are four small glands which produce and release parathormone, that maintains plasma ionized calcium level. Ionized calcium is essential for bone and tooth development, neuromuscular excitability, membrane fluidity and integrity, cell communication, cell adhesion, and blood clotting.
Two well-defined clinical entities are associated with the parathyroid glands: hyperparathyroidism, which manifests its symptoms primarily in the bones and the kidneys, and hypoparathyroid tetany.
Primary Hyperparathyroidism: (Osteitis fibrosa cystica)
Primary hyperparathyroidism is a disease in which the parathyroid glands elaborate an excessive quantity of parathyroid hormones. This increased activity is usually due to an adenoma of one or more of the four parathyroid glands, to a hyperplasia of the parathyroid tissue, or rarely, to a functional carcinoma of the parathyroid. The role of the parathyroid tumor in primary hyperparathyroidism has been discussed by Lloyd.
The bone disturbances in hyperparathyroidism vary from vague to radiographically characteristic lesions and even gross clinical evidence of bone lesions. Hypercalcemia may be manifested by poor muscle tone and decreased neuromuscular excitability.
Clinical Features
Hyperparathyroidism is a relatively rare disease which is said to be three times as common in women as in men. It usually affects people of middle age, but it may occur in childhood or in later life. In contrast to these statistics, Silverman and his coworkers, who reviewed 42 consecutive dentulous patients with hyperparathyroidism, found no correlation between gender and age and any aspect of the disease. Pathologic fracture may be the first symptom of the disease, although bone pain and joint stiffness are frequently early symptoms. In Silverman’s group the most common significant early clinical finding was urinary tract stone, which occurred in 33 of the 42 patients. Occasionally the first sign of the disease may be a giant cell tumor or a ‘cyst’ of the jaw. The effects of the disease on bone are of special interest to dentists. Almost all patients with hyperparathyroidism have skeletal lesions, some of which may occur in the skull or jaws. The loss of phosphorus and calcium in this disturbance results in a generalized osteoporosis with abortive attempts at bone repair and new bone formation. The new bone may be resorbed, and the resorption may lead eventually to pseudocyst formation, the extent of which depends on the duration and intensity of the disease. According to Schour and Massler, malocclusion caused by a sudden drifting with definite spacing of the teeth may be one of the first signs of the disease. An extensive review and discussion of this disease was published by Teng and Nathan.
Radiographic Features
The radiographic findings in this disease are of particular importance. The bones of affected persons show a general radiolucency as compared with those of normal people. Later, sharply defined round or oval radiolucent areas develop, which may be lobulated (Fig. 15-18). If such a lobulated lesion develops in the mandible, it must be carefully differentiated from ameloblastoma, which frequently has the same appearance.
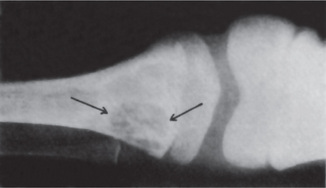
Figure 15-18 Primary hyperparathyroidism.
The circumscribed radiolucent defect in the proximal end of the tibia was accompanied by multiple lesions of other bones, including the jaws.
Small cystic areas may be seen in the calvarium, and large and/or small sharply defined radiolucencies may be present in the maxilla and/or mandible (Fig. 15-19). These small cystic areas must be differentiated from the lesions of multiple myeloma and eosinophilic granuloma. In the jaws, the bone radiograph in hyperparathyroidism has been described as having a ‘ground-glass’ appearance. The lamina dura around the teeth may be partially lost (Fig. 15-20). Twenty of Silverman’s 42 patients had normal dental radiographs, and 17 showed intact lamina dura but abnormal appearing alveolar bone. None showed complete loss of lamina dura, and only five showed partial loss.
Histologic Features
Histologic findings in the bone lesions of hyperparathyroidism are not pathognomonic of the disease, but are of considerable assistance in making the diagnosis. The most characteristic change in the bone is an osteoclastic resorption of the trabeculae of the spongiosa and along the blood vessels in the Haversian system of the cortex. In the areas of resorption one also finds many plump osteoblasts lining islands of osteoid. Fibrosis, especially of the marrow spaces, is marked. The fibroblasts replace resorbed trabeculae, and in the fibrotic islands there is recent and old hemorrhage, and much hemosiderin in evidence. As the disease progresses, ‘osteoclastomas’ develop, characterized by masses of fibroblasts growing in a loose syncytium, among which are numerous capillaries and endothelium-lined blood spaces, red blood cells, many areas of yellow or brown hemosiderin, and innumerable multinucleated giant cells. These latter lesions are indistinguishable microscopically from the central giant cell granuloma of bone. Therefore, any patient who has a lesion diagnosed as a central giant cell lesion should be evaluated medically to rule out the possibility of hyperparathyroidism. This is most easily accomplished by a serum calcium level determination. If hyperparathyroidism is present, the serum calcium will be elevated above the normal level of 9–12 mg/dl.
Diagnosis
It is confirmed by blood investigation which shows hypercalcemia, hypophosphatemia, and elevated serum parathormone level along with hypercalciuria, and hyperphosphaturia. The serum alkaline phosphates level is increased in osteolytic lesions.
Treatment and Prognosis
Excision of the parathyroid tumor will cure the patient. Careful examination of all parathyroid glands at the time of surgery should be carried out since multiple tumors occur with some frequency. Furthermore, since multiple tumors may develop over an extended period of time, patients who have had one parathyroid tumor should be followed for life.
Secondary hyperparathyroidism
Hyperparathyroidism can also occur secondary to other disorders, the most common being end-stage renal disease. Massry and coworkers reported an incidence of hyperparathyroidism in patients with chronic renal failure, ranging from 18% after one year on dialysis to 92% after more than two years. Giant cell lesions were not reported until 1963; however, by Fordham and Williams. Thirty patients undergoing chronic hemodialysis were studied by Spolnik and his associates, and 22 (73%) of these were found to have radiographic evidence of bone disease involving the jaws, including seven brown tumors (four in one patient) with loss of lamina dura also a prominent finding.
Hypoparathyroidism
Elimination of the parathyroid glands—by surgical removal, by destruction due to thrombosis of the blood vessels or disease of the glands, or in rare cases by congenital absence—leads to hypoparathyroidism. The disease is characterized metabolically by a decreased excretion of calcium. The blood chemistry shows a low concentration of serum calcium and a high concentration of serum phosphorus. If the calcium level of the serum falls to 7–8 mg/dl, there is increased neuromuscular excitability, which must be elicited, since it is not manifest. When the serum calcium level falls to 5–6 mg/dl, tetany and the characteristic carpopedal spasms are apparent.
Albright and Strock observed aplasia or hypoplasia of the teeth when hypoparathyroidism developed before the teeth were entirely formed. Similar changes are reported by Frensilli and his associates (Fig. 15-21).
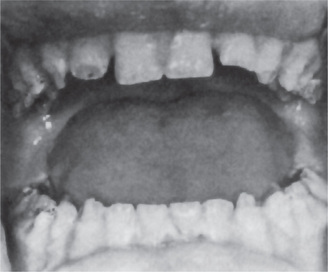
Figure 15-21 Hypoparathyroidism.
The patient, demonstrating enamel hypoplasia, suffered from hypoparathyroidism in infancy Courtesy of Dr EV Zegarelli.
Patients will have increased neuromuscular excitability, resulting in muscle spasms, stiffness, cramping, and tetany. Specific oral manifestations include altered tooth eruption pattern, short, blunted roots, enamel hypoplasia and dentin dysplasia, impacted teeth, and partial anodontia. Circumoral paresthesia is often one of the first symptoms of hypoparathyroidism.
Chronic candidosis, which is refractory to antifungal therapy such as nystatin, is sometimes seen in cases of idiopathic hypoparathyroidism. Such a case has been reported and discussed by Greenberg and his associates. The candidosis usually develops very early in life and precedes the hypocalcemia. The exact relationship between these two occurrences is not clear but it has been suggested that the candida infection may cause the hypoparathyroidism by inducing and ‘immune response’. Enamel hypoplasia frequently accompanies this ‘syndrome’.
Pseudohypoparathyroidism is a condition resembling idiopathic hypoparathyroidism in all respects, including the hypocalcemia and hyperphosphatemia, the drugrefractory candidosis and the enamel hypoplasia, except that parathyroid extract has little or no effect in correcting the hypocalcemia. This disease has been discussed by Croft and his coworkers.
Adrenal Hormones
Adrenal glands are small, paired endocrine glands, situated at the posterosuperior poles of the kidneys. They have an outer cortex and inner medulla, which secrete various hormones.
The adrenal cortex is divided into:
1. Zona glomerulosa, which secretes mineralocorticoids (e.g. aldosterone)
2. Zona fasciculata, which secretes glucocorticoids and cortisol
A little over a 100 years ago (1855), Addison published his classic report on chronic adrenal cortical insufficiency, which is still called Addison’s disease. Only in the last 25 years has it been recognized that the adrenal cortex, as such, plays the essential role in maintaining life. In that period; however, many substances have been isolated from the adrenal cortex, and intensive studies of the physiologic activity of adrenal cortical steroids are still being made. Much has been learned about the adrenal cortex, but much is still unknown. When the final story is written, we shall probably find that most of the metabolic interrelations in human physiology are mediated through the adrenals.
The action of the medullary portion of the adrenal gland is ascribed to epinephrine and norepinephrine. Norepinephrine may be a precursor of epinephrine, and for practical purposes one may consider an action of the hormones of the adrenal medulla to be that of epinephrine. Its effect on the various tissues of the body is similar to that of sympathetic nervous system stimulation. Increased amounts of epinephrine in the body lead to an elevated basal metabolism, mediated through its effect on the liver and on carbohydrate metabolism rather than on the thyroid gland.
The effects of epinephrine and norepinephrine on the circulatory system have been extensively studied. Epinephrine in physiologic doses (1 mcg/kg) causes a constriction of the arterioles and capillaries of the skin, mucous membranes, and abdominal viscera, but a dilatation of the vessels of skeletal and heart muscle. The net result is a rise in blood pressure due to a sufficient vasoconstriction of the end capillaries and small arterioles of the skin and other organs. Epinephrine also relaxes the smooth muscles of the stomach, intestine, bronchioles, and wall of the urinary bladder, while it excites the muscles of the gall bladder, ureter, and sphincters of the intestine. Recent studies have shown that, under controlled conditions, epinephrine acts as an overall vasodilator drug and a powerful cardiac stimulant, while norepinephrine in comparable doses acts as an overall vasoconstrictor substance.
The formation and liberation of the adrenal cortical steroids appear to be dependent upon the action of the pituitary adrenocorticotropic hormone (ACTH). At least six adrenal cortical steroids have been identified, and several unknown factors have been observed. Those identified are corticosterone, 17-hydroxycorticosterone, 17-hydroxyprogesterone, 11-dehydrocorticosterone, 11-desoxycorticosterone, and 17-hydroxy-11-dehydrocorticosterone (cortisone). These steroids are intimately concerned with carbohydrate metabolism, mineral metabolism, protein and fat metabolism, and fluid and electrolyte balance.
So much work is being done at this time on the metabolic influences of the adrenal cortex that almost anything written now will soon be obsolete. What little we know about the cortical hormones of the adrenal gland as they relate to oral pathology is summarized.
Acute Insufficiency of the Adrenal Cortex
This is a term used to describe inadequate glucocorticosteroid or mineralocorticosteroid production. It may be acute or chronic and primary or secondary.
Acute adrenal cortical insufficiency is relatively rare. It usually occurs in connection with an acute septicemia and is called the Waterhouse-Friderichsen syndrome. This disease occurs primarily in children, but can also occur in adults. It is characterized by a rapidly fulminating septic course, a pronounced purpura and death within 48–72 hours. Meningococci, streptococci and pneumococci are the organisms most often responsible for the disease. At autopsy the conspicuous change seen is bilateral adrenal hemorrhage (Fig. 15-22). Apart from that, it can develop also in patients who take large doses of steroids for more than two weeks and abruptly stop.
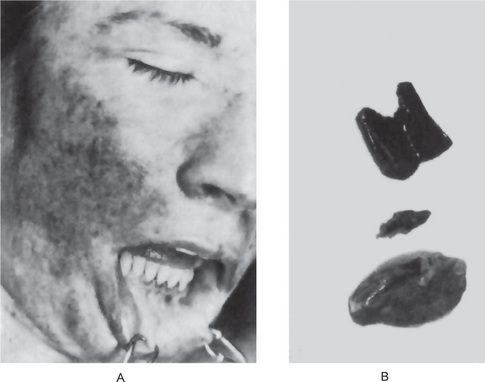
Figure 15-22 Waterhouse-Friderichsen syndrome.
There are petechial hemorrhages and purpura in the skin and oral mucosa, (A). The adrenal glands are hemorrhagic, (B).
The use of antibiotics and cortisone has changed the course of the disease from its usual fatal termination to recovery in some cases.
Chronic Insufficiency of the Adrenal Cortex: Addison’s Disease
Modern medicine can add little to the description of the disease reported by Addison in 1855. It usually develops following the autoimmune destruction of adrenal glands and frequently occurs in conjunction with other autoimmune disorders.
Clinical Features
Early manifestations include lethargy, fatigue, and muscular weakness that may persist for months. The patient feels tired after minimal exertion but feels well on walking. Feeble heart action, general debility, vomiting, diarrhea, and severe anemia are also seen. Females develop irregular menstruation and loss of body hair.
Hypoglycemia, dehydration, hypertension, elevated serum potassium, and postural dizziness develop following low blood levels of cortisol.
Due to an increased level of ACTH in the blood, the secretion of melanocyte stimulating hormone increases, which induces the deposition of melanin in the skin and mucous membrane (Fig. 15-23). This pattern is nonspecific and may consist of multiple focal dark spots or generalized diffuse streaks of hyperpigmentation which may be macular, flat, brown, and variable in shape. In the oral mucosa, a pale brown to deep chocolate pigmentation spreading over the buccal mucosa from the angles of the mouth and/or developing on the gingiva, tongue and lips, may be the first evidence of the disease.
Diagnosis
The diagnosis of Addison’s disease is based on the clinical signs as well as on characteristic changes in the blood sodium and chloride levels. Biopsy of the oral lesions shows acanthosis with silver-positive granules in the cells of the stratum germinativum. Low diurnal plasma cortisol and ACTH levels, along with an elevated level of blood urea nitrogen confirms the diagnosis.
Hyperfunction of Adrenal Gland
Adrenogenital Syndrome
A condition known as the adrenogenital syndrome results when hyperplasia or tumors of the adrenal cortex occur. Depending on the age of onset and the gender of the person affected, the clinical signs are pseudohermaphroditism, sexual precocity and virilism in women or feminization in men. If the disease begins early, premature eruption of the teeth may occur.
Cushing’s syndrome
This syndrome is a result of hormonal excess resulting from any of the following:
• Hyperplastic adrenal cortices without any other clinically evident endocrine lesion.
• Adrenal cortical adenoma or carcinoma.
• Ectopically located adrenal-like tumor, for example, of an ovary.
• ACTH-secreting tumor of the anterior pituitary associated with adrenal cortical hyperplasia.
• Nonpituitary carcinoma, for example, of a lung or the pancreas, with secretion of an ACTH-like material that induces adrenal cortical hyperplasia.
When the syndrome is associated with spontaneous bilateral adrenal hyperplasia, it is referred to as Cushing’s disease. In adults, it is recognized that Cushing’s disease represents approximately 75% of the cases of Cushing’s syndrome. While Cushing’s disease is uncommon in children, McArthur and his associates have reported a series of 13 cases in patients under the age of 15 years. The pathogenesis of this disease has been reviewed by Hunder.
It is characterized by a rapidly acquired adiposity about the upper portion of the body, mooning of the face, a tendency to become round-shouldered and develop a buffalo hump at the base of the neck, alteration in hair distribution, a dusky plethoric appearance with formation of purple striae, muscular weakness, vascular hypertension, glycosuria not controlled by insulin, and albuminuria.
The oral pathologist’s primary concern with this peculiar disease state lies in the bone changes. In children there may be osteoporosis and premature cessation of epiphyseal growth, while in adults there is a severe osteoporosis.
The mechanisms for the bone changes is not well understood. Apparently, 11-desoxycorticosterone is relatively unimportant in the pathogenesis of Cushing’s syndrome. Albright’s explanation for the pathogenesis of the disease is based on the S-F-N (sugar-fat-nitrogen) hormone group of steroids in which the ‘N’ hormone is considered an anabolic one, stimulating osteogenesis and causing closure of the epiphysis, and the ‘S’ hormone is considered an antianabolic one. The mechanism of osteoporosis is then explained on the basis of an excess of ‘S’ hormone, leading to a retardation of osteoblastic activity and reduction in matrix formation.
We appreciate the fact that a number of complex interrelations are concerned in the normal and abnormal control of bone growth and maturation. Considerable interest is now centering on these interrelations and on the precise metabolic or endocrine pathways by which particular hormones influence skeletal growth. Little definite evidence on these topics exists as yet, but some information is available on the effects of cortisone on bone growth. For example, Follis showed that cortisone injections in rats produced retardation and arrest of and interference with resorption of bone. In other species, only retardation of bone growth was found.
Fraser and Fainstat demonstrated that in certain strains of mice the injection of cortisone into pregnant females produced a high percentage of cleft palates in the offspring. This effect was not due primarily to the inhibition of growth, since cleft palate was produced even when the cortisone was administered after the palate had already closed. Doig and Coltman reported several cases of cleft palate in children born of mothers who conceived while receiving injections of cortisone or who received injections of cortisone during the first three months of pregnancy. Obviously, more work is needed in this field before definite conclusions can be drawn.
Stress and the ‘Adaptation Syndrome’
The extensive studies of Hans Selye have done much to stimulate thinking and research in the area of ‘stress’ and the adrenal gland. He formulated a theory of response to prolonged stress as a part of the individual’s adaptive mechanism which may lead to clinical signs and symptoms called the general adaptation syndrome. This theory is a controversial one, and much research is being done to clarify the points of controversy.
Any wasting disease produces atrophy of the adrenal cortex and loss of adrenal lipid. The mechanism for this finding is not known. Selye states that the adrenal changes are due to prolonged stress, with the mobilization of lipids and ultimate exhaustion atrophy of the cortical cells. Apparently, the hormones of the adrenal cortex are necessary for cellular enzymes to catalyze the energy-producing processes of cells. All ‘stressor’ agents, such as cold, heat and trauma, increase the metabolic demands of the organism and stimulate adrenocortical function through stimulation of the pituitary to secrete ACTH. If the stress is continued, the pituitary and the adrenal cortex produce excessive amounts of hormones to increase resistance. Eventually pathologic changes occur in those tissues which respond to the hormonal stimulation, and the diseases of adaptation (hypertension, periarteritis nodosa, and others) results.
Since there is a considerable amount of evidence against Selye’s theory, and since the entire field of adrenal mechanisms in pathologic processes is in a state of flux, only a brief resume of Selye’s theory will be presented. The reader is directed to Selye’s original papers (1946, 1948) and to the excellent critical review by Sayers (1950) for a more comprehensive coverage of the subject.
Selye states that the ‘stressed’ person passes through a succession of stages. The first is the ‘alarm reaction’, which consists of a shock phase and then a countershock phase. The next is the ‘adaptation stage’, in which his resistance to the original stressor is greater, but his resistance to other stressor agents is lowered. If the stressor is continued, he eventually enters a stage of exhaustion and dies. If the stressor is removed, he enters a stage of convalescence and recovers. Figure 15-24 shows diagrammatically the control of adrenocortical activity.
Figure 15-24 The control of adrenocortical activity.
Stress may act on the pituitary or through the hypothalamus to stimulate the secretion of ACTH at the expense of other pituitary hormones. ACTH stimulates the zona fasciculata and zona reticularis, though possibly not the zona glomerulosa, which may secrete DOC independently. The level of adrenocortical hormones in the blood controls the rate of production of ACTH. Rapid utilization of cortical hormones by the tissues lowers the blood level of cortical hormones and thus stimulates ACTH production. Adrenaline (epinephrine) may also stimulate ACTH production, and adenosine triphosphate or potassium excess may stimulate the adrenal cortex From PMF Bishop: Cameron’s Recent Advances in Endocrinology, 7th ed. London. J & A Churchill, Ltd, 1954).
Many people are receiving large doses of cortisone for the treatment of various diseases. We must remember that cortisone interferes with the formation of granulation tissue, proliferation of fibroblasts, and production of ground substance. Since these tissues and cellular products are essential to wound healing, it is important to recognize that surgery is hazardous in hyperadrenocorticism.
Pancreatic Hormone: Insulin
Diabetes Mellitus
Diabetes is a biochemical lesion, and though no complete correlation exists between the occurrence of the disease and histologically demonstrable changes in the pancreas, the role of insulin in the control of the disease and historical considerations make it legitimate to discuss diabetes in the section on the pancreas. Because recent investigations have shown that other endocrine organs play a role in its production, many writers consider diabetes mellitus more generally a disease of metabolism.
Diabetes is a disorder of carbohydrate metabolism characterized by hyperglycemia and glycosuria, reflecting a distortion in the equilibrium between utilization of glucose by the tissues, liberation of glucose by the liver and productionliberation of pancreatic, anterior pituitary and adrenocortical hormones. This metabolic disorder lowers tissue resistance to infection.
It is a disorder caused by relative or absolute lack of insulin, and in the later stages of disease it provides multiple systemic complications. Recent evidences indicate that it is a multifactorial disease with genetic predisposition and destruction of the islet of Langerhans cells.
It is classified into two types:
• Insulin dependant diabetes mellitus (IDDM) (Juvenile onset or type I or brittle or ketosis prone or labile diabetes). It occurs as a result of immunologically mediated destruction of pancreatic beta cells.
• Non-insulin dependant diabetes mellitus (NIDDM) (Adult onset or type II or maturity onset diabetes).
Insulin dependant diabetes mellitus is the less common form of diabetes, characterised by onset before age 20, with a thin body build, extreme thirst, hunger, constant urination, and weight loss. As there is no insulin secreted in these patients, daily injections of insulin are required to control the blood glucose level and also to prevent ketoacidosis. There is a positive family history for the disease with a weaker genetic tendency than type II diabetes mellitus.
Clinical Features
Patients manifest with glycosuria, polyuria, polydipsia, weakness, and weight loss. Abnormal and accelerated metabolism of amino acids and fats results in ketoacidosis.
The complications are microangiopathy and macroangiopathy which include atherosclerosis, retinopathy, neuropathy, renal failure, autonomic insufficiency, and susceptibility to infections.
Oral Manifestations
The oral manifestations are mainly due to inflammation and infection because of the abnormal neutrophil function, microangiopathy, and altered oral flora. Most of the patients present with a dry mouth, persistent gingivitis, multiple carious lesions, periodontal disease, and candidiasis.
Diabetic patients show an increased tendency towards delayed wound healing and dry socket formation.
Because of the lowered tissue resistance, patients with untreated or inadequately controlled diabetes sometimes exhibit a fulminating periodontitis with periodontal abscess formation and inflamed, painful and even hemorrhagic gingival papillae. Bernick and coworkers studied a series of 50 diabetic children and found that gingivitis was increased; however, the rate of caries formation was not related to the duration of the disease. Lin and colleagues noted a significant thickening of the basement membranes of gingival vessels and proposed that gingival biopsies may be useful as an adjunct in the diagnosis of diabetes. Because of excessive fluid loss, diabetic patients commonly complain of dry mouth. Even minor oral surgery is contraindicated in uncontrolled diabetic patients. Vascular changes in the dental pulp, gingiva, and periodontal ligament have been reported in diabetic patients by Russell.
Controlled diabetic patients should undergo dental operations only after consultation with the physician who is treating the patient. There are no oral manifestations of controlled diabetes mellitus.
Progeria: (Hutchinson-Gilford syndrome)
Progeria is a very rare disease originally described by Hutchinson in 1886. It is of unknown etiology and is characterized by dwarfism and premature senility. It is thought to be transmitted as an autosomal recessive trait. The term itself means prematurely old. Progeria has been discussed in an article by DeBusk.
Clinical Features
Affected infants appear normal at birth, but the typical clinical features become manifested within the first few years. The patients all have an amazing resemblance to each other, exhibiting alopecia, pigmented areas of the trunk, atrophic skin, prominent veins, and loss of subcutaneous fat. The individuals have a high-pitched, squeaky voice, a beak-like nose, and a hypoplastic mandible. Coxa valga is also a constant feature, as is severe atherosclerosis. Exophthalmos may be present, as may muscular atrophy and joint deformities. The intelligence of individuals with this disease is generally either normal or above normal. Even at a very early age, the patient resembles a wizened little old person.
Oral Manifestations
The oral findings in progeria have been described by Gardner and Majka. These basically consist of the accelerated formation of irregular secondary dentin, apparently a manifestation of the premature aging process. Delayed eruption of teeth has also been reported by Wesley and colleagues.
References
Ainley, J.E. Manifestations of familial hypophosphatemia. J Endodont. 1978; 4:26.
Albright, F. Cushing’s syndrome: its pathological physiology, its relationship to the adrenogenital syndrome and its connection with the problem of the reaction of the body to injurious agents (‘alarm reaction of Selye’. Harvey Lect Series. 38(123), 1942-43.
Albright, F., Strock, M.S. Association of a calcification of dentin with hypoparathyroidism in rats and cure of same with parathormone, with some correlated observations in man. J Clin Invest. 1933; 12:974.
Albright, F., Butler, A.M., Bloomberg, E. Rickets resistant to vitamin D therapy. Am J Dis Child. 1937; 54:529.
Alfin-Slater, R., Kritchevsky, D. Human Nutrition: A Comprehensive Treatise. Plenum Press, New York. 1979.
Amstutz, H.C., Carey, E.J. Skeletal manifestations and treatment of Gaucher’s disease: review of twenty cases. J Bone Joint Surg. 1966; 48A:670.
Aponte-Merced, L., Navia, J.M. Pre-eruptive proteinenergy malnutrition and acid solubility of rat molar enamel surfaces. Arch Oral Biol. 1980; 25:701.
Archard, H.O., Witkop, C.J., Jr. Hereditary hypophosphatemia (vitamin D-resistant rickets) presenting primary dental manifestations. Oral Surg. 1966; 22:184.
Arcomano, J.P., Barnett, J.C., Wunderlich, H.O. Histiocytosis X. Am J Roentgenol Radium Ther Nucl Med. 1961; 85:663.
Attanasio, A. Multiple Endocrine Diseases: Growth Hormone Action: Inter sexuality. CTS Karger Publishers, Farmington. 1992.
Avioli, L.V., Krane, S.M. Metabolic Bone Disease and Clinically Related disorders. WB Saunders, Philadelphia. 1990.
Avioli, L.V., Lasersohn, J.T., Lopresti, J.M. Histiocytosis X (Schüller-Christian disease): A clinicopathological survey, review of ten patients and the results of prednisone therapy. Medicine. 1963; 42:119.
Baratieri, A., Miani, C., Sacchi, A. A histochemical study of gingival hyperplasia in atypical mucopolysaccharidosis. Parodont Acad Rev. 1968; 4:163.
Batson, R., Shapiro, J., Christie, A., Riley, H.D., Jr. Acute nonlipid disseminated reticuloendotheliosis. Am J Dis Child. 1955; 90:323.
Baume, L.J., Becks, H., Evans, H.M. Hormonal control of tooth eruption III. The response of the incisors of hypophysectomized rats to growth hormone, thyroxin or the combination of both. J Dent Res. 1954; 33:104.
Baume, L.J., Becks, H., Ray, J.D., Evans, H.M. Hormonal control of tooth eruption II: the effects of hypophysectomy on the upper rat incisor following progressively longer intervals. J Dent Res. 1954; 33:91.
Bavetta, L.A., Bernick, S. Lysine deficiency and dental structures. J Am Dent Assoc. 1955; 50:427.
Bavetta, L.A., Bernick, S., Geiger, D., Bergren, W. The effect of tryptophane deficiency on the jaws of rats. J Dent Res. 1954; 33:309.
Becks, H., Furuta, W.J. Effects of magnesium deficient diets on oral and dental structures I: changes in the enamel epithelium. J Am Dent Assoc, 26: 883, 1939 II Changes in the enamel structure. J Am Dent Assoc, 28: 1083, 1941 III Changes in dentine and pulp tissue. Am J Orthod Oral Surg, 28: 1, 1942 IV Changes in paradental bone structure. J Dent Res. 1943; 22:215.
Becks, H., Collins, D.A., Simpson, M.E., Evans, H.M. Changes in the central incisors of hypophysectomized female rats after different postoperative periods. Arch Pathol. 1946; 41:457.
Bendich, A., Olson, J.A. Biological actions of carotenoids. FASEB J. 1989; 3:1927.
Bender, I.B. Dental observations in Gaucher’s disease. J Dent Res. 1938; 17:359.
Bercu, B.B. Basic and Clinical Aspects of Growth Hormone. Plenum Publishing, New York. 1988.
Bernick, S., Bavetta, L.A., Baker, R. Histochemical and electron microscopy studies on tryptophane deficient dentin. J Dent Res. 1955; 34:671.
Bernick, S.M., Cohen, D.W., Daker, L., Laster, L. Dental disease in children with diabetes mellitus. J Periodontol. 1975; 46:241.
Beumer, J., Trowbridge, H.O., Silverman, S., Eisenberg, E. Childhood hypophosphatasia and the premature loss of teeth. Oral Surg. 1973; 35:631.
Bhatia, S., Nesbit Jr, M., Egeler, R.M., et al. Epidemiological study of Langerhans cell histiocytosis in children. J Pediatr. 1997; 130:774.
Bilezikian, J.P., et al. The Parathyroid: Basic and Clinical Concepts. Raven Press, New York. 1994.
Blackfan, K.D., Wolbach, S.B. Vitamin A deficiency in infants: a clinical and pathological study. J Pediatr. 1933; 3:679.
Blomhoff, R., et al. Vitamin A metabolism: new perspectives on absorption, transport, and storage. Physiol Rev. 1991; 71:951.
Bodansky, M., Bodansky, O. Biochemistry of Disease, 2, New York: Macmillan, 1959.
Bohme, M., Wahlgren, C.F. Lipoid proteinosis in three children. Acta Paediatr. 1996; 85:1003.
Bondy, P.K., Rosenberg, L.E. Diseases of Metabolism, 8, Philadelphia: WB Saunders, 1980.
Bongiovanni, A.M., Album, M.M., Root, A.W., Hope, J.W., et al. Studies on hypophosphatasia and response to high phosphate intake. Am J Med Sci. 1968; 225:163.
Boothby, W.M., Plummer, W.A. Diseases of the thyroid gland. Oxford Med. 1936; 3:389.
Bourne, G.H., Kidder, G.W. Biochemistry and Physiology of Nutrition. Academic Press, New York. 1953.
Boyle, P.E. Manifestations of vitamin A deficiency in a human tooth germ. J Dent Res. 1933; 13:39.
Boyle, PE. Kronfeld’s Histopathology of the TeethTheir Surrounding Structures, 3, Philadelphia: Lea and Febiger, 1949.
Boyle, P.E., Bessey, O.A., Wolbach, S.B. Experimental alveolar bone atrophy produced by ascorbic acid deficiency and its relation to pyorrhea alveolaris. Proc Soc Exp Biol Med. 1937; 36:733.
Boyle, P.E., Wolbach, S.B., Bessey, O.A. Histopathology of teeth of guinea pigs in acute and chronic vitamin C deficiency. J Dent Res. 1936; 15:331.
Brady, R.O. The sphingolipidoses. New Engl J Med. 1966; 275:312.
Brady, R.O., Pentchev, P.G., Gal, A.E., Hibbert, S.R., Dekeban, A.S. Replacement therapy for inherited enzyme deficiency: use of purified glucocerebrosidase in Gaucher’s disease. New Engl J Med. 1974; 291:989.
Brittain, J.M., Oldenberg, T.R., Burkes, E.J. Odontohypophosphatasia: report of two cases. J Dent Child. 1976; 43:38.
Bronner, F., Coburn, J.W. Disorders of Mineral Metabolism Vol 1. Trace minerals. Academic Press Inc, (A subsidiary of Harcourt Brace Jovanovich, Publishers), New York. 1981.
Brownstein, M.H., Helwig, E.B. The cutaneous amyloidoses I: localized forms. Arch Dermatol. 1970; 102:8.
Bruckner, R.J., Rickles, N.H., Porter, D.R. Hypophosphatasia with premature shedding of teeth and aplasia of cementum. Oral Surg. 1962; 15:1351.
Brunner, H. Eosinophilic granuloma of mouth, pharynx and nasal passages. Oral Surg. 1951; 4:623.
Buckle, R.M., Care, A.D., Cooper, C.W., Gitelman, H.J. The influence of plasma magnesium concentration on parathyroid hormone secretion. J Endocrinol. 1968; 42:529.
Burk Jr, R.F., Pearson, W.N., Wood, R.P., Viteri, F. Blood selenium levels and in vitro red blood cell uptake of 75-Se in kwashiorkor. Am J Clin Nutr. 1967; 20:723.
Burns, JJ. Biosynthesis of L-ascorbic acid; basic defect in scurvy. Am J Med. 1959; 26:740.
Campbell, H.A., Smith, W.K., Roberts, W.L., Link, K.P. Studies on the hemorrhagic sweet clover disease II The bioassay of hemorrhagic concentrates by following the prothrombin level in the plasma of rabbit blood. J Biol Chem. 1941; 138:1.
Catto, G.R.D., MacLeod, M., Pelc, B., Kodicek, E. 1-a-Hydroxycholecalciferol: a treatment for renal bone disease. Br Med J. 1975; 1:12.
Chambers, R.A., Pratt, RTC. Idiosyncrasy to fructose. Lancet. 1956; 2:340.
Chan, I., El-Zurghany, A., Zendah, B., Benghazil, M., et al. Molecular basis of lipoid proteinosis in a Libyan family. Clin Exp Dermatol. 2003; 28:545.
Chase, D.C., Eversole, L.R., Hale, H.D. Histiocytosis-X with jaw involvement. J Oral Surg. 1974; 32:494.
Chatterjee, M.N., Shinde, R. Text book of medical biochemistry, 6, India: Jaypee Publisher, 2005.
Chawla, T.N., Glickman, I. Protein deprivation and the periodontal structures of the albino rat. Oral Surg. 1951; 4:578.
Cheyne, C. Histiocytosis X. J Bone Joint Surg, 53–B: 366. 1971.
Christensen, J.F. Three familial cases of atypical late rickets. Acta Paediatr Scand. 28(247), 1940-41.
Cline, M.J., Golde, D.W. A review and reevaluation of the histiocytic disorders. Am J Med. 1973; 55:49.
Cohen, S., Becker, G.L. Origin, diagnosis and treatment of the dental manifestations of vitamin-D resistant rickets: review of the literature and report of case. J Am Dent Assoc. 1976; 92:120.
Cohen, R.D., et al. The Metabolic and Molecular Basis of acquired Disease. WB Saunders, Philadelphia. 1990.
Collins, D.A., Becks, H., Asling, C.W., Simpson, M.E., et al. The growth of hypophysectomized female rats following chronic treatment with pure pituitary growth hormone V Skeletal changes: skull and dentition Growth 1949;13(207).
Comar, C.L., Bronner, F. Mineral Metabolism. Academic Press, New York. 1960.
Cornblath, M., Rosenthal, I.M., Reisner, S.H., Wybregt, S.H., et al. Hereditary fructose intolerance. New Eng J Med. 1963; 269:1271.
Cotter, F.E., Pritchard, J. Clonality in Langerhans’ cell histiocytosis. Br Med J. 1995; 310:74.
Crampton, E.W. The growth of the odontoblasts of the incisor teeth as a criterion of the vitamin C intake of the guinea pig. J Nutr. 1947; 33:491.
Croft, L.K., Witkop Jr, C.J., Glas, J.E. Pseudohypoparathyroidism. Oral Surg. 1965; 20:758.
Cushing, H. The Pituitary Body and Its Disorders. JB Lippincott, Philadelphia. 1912.
Dam, H. Cholesterinstoffwechsel in Hühnereiern und Hühnehan Biochem Z. 1929; 215:475.
Dam, H., Schønheyder, F., Tage-Hansen, E. Studies on the mode of action of vitamin K. Biochem J. 1936; 30:1075.
Daneshbod, K., Kissane, J.M. Idiopathic differentiated histiocytosis. Am J Clin Pathol. 1978; 7:381.
Darrow, D.C. Body-fluid physiology: the role of potassium in clinical disturbances of body water and electrolyte. N Eng J Med. 1950; 242(978):1014.
DeBusk, F.L. The Hutchinson-Gilford Progeria syndrome. J Pediatr. 1972; 80:697.
Dinnerman, M. Vitamin A deficiency in unerupted teeth of infants. Oral Surg. 1951; 4:1024.
Di, Orio LP., Miller, S.A., Navia, J.M. The separate effects of protein and calorie malnutrition on the development and growth of rat bones and teeth. J Nutr. 1973; 103:856.
Doig, R.K., Coltman, O.M. Cleft palate following cortisone therapy in early pregnancy. Lancet. 1956; 2:730.
Drummond, J.C. LIX: The nomenclature of the so-called accessory food factors (vitamins. Biochem J. 1920; 14:660.
Duncan, G.G. Diseases of Metabolism, 4, Philadelphia: WB Saunders, 1959.
Egeler, R.M., Favara, B.E., van Meurs, M., et al. Differential in situ cytokine profiles of Langerhans-like cells and T cells in Langerhans cell histiocytosis: abundant expression of cytokines relevant to disease and treatment. Blood, 94:4195. 1999.
Elder, G.H., Gray, C.H., Nicholson, D.C. The porphyrias: a review. J Clin Pathol. 1972; 25:1013.
Enriquez, P., Dahlin, D.C., Hayles, A.B., Henderson, E.D. Histiocytosis X: a clinical study. Mayo Clin Proc. 1967; 42:88.
Epstein, C.J., Brady, R.O., Schneider, E.L., Bradley, R.M., et al. In utero diagnosis of Niemann-Pick disease. Am J Hum Genet. 1971; 23:533.
Evans, H.M., Bishop, K.S. On the existence of a hitherto unrecognized dietary factory essential for reproduction. Science. 1922; 56:650.
Farrell, P.M., Bieri, J.G. Megavitamin E Supplementation in man. Am J Clin Nutr. 1975; 28:1381.
Federation Proceedings Conference: Hypovitaminosis. A Fed Proc. 1958; 17:103.
Fish, E.W., Harris, L.J. The effects of vitamin C deficiency on tooth structure in guinea pigs. Br Dent J. 1935; 58:31.
Fletcher, P.D., Scopp, I.W., Hersh, R.A. Oral manifestations of secondary hyperparathyroidism related to long-term hemodialysis therapy. Oral Surg. 1977; 43:218.
Follis, R.H., Jr. The Pathology of Nutritional Disease Springfield, Ill, Charles C Thoma. 1948;
Idem: Effect of cortisone on growing bones of the rat. Proc Soc Exp Biol Med. 1951; 76:722.
Idem: Non-effect of cortisone on growing bones of mice guinea pigsrabbits. Proc Soc Exp Biol Med. 1951; 78:723.
Idem: Deficiency Disease Springfield Ill Charles C Thoma. 1958;
FoodNutrition, Board., National, Academy of Sciences., National, Research Council. Recomended Dietary Allowances (9th rev ed), Washington D. 1980;
Fordham, C.C., Williams, T.F. Brown tumor and secondary hyperparathyroidism. New Engl J Med. 1963; 269:129.
Foster, G.V. Calcitonin (thyrocalcitonin. New Engl J Med. 1968; 279:349.
Frandsen, A.M. Experimental investigations of socket healing and periodontal disease in rats. Acta Odont Scand, 21 (Suppl 37): 53. 1963.
Frandsen, A.M., Becks, H., Nelson, M.M., Evans, H. The effects of various levels of dietary protein on the periodontal tissues of young rats. J Periodontol. 1953; 24:135.
Franklin, E.C. Amyloidosis Bull Rheum Dis. 1975; 26:832.
Fraser, F.C., Fainstat, T.D. Production of congenital defects in the offspring of pregnant mice treated with cortisone. Pediatrics. 1951; 8:527.
Frazier, C.N., Hu, C.K. Cutaneous lesions associated with deficiency in vitamin A in man. Arch Int Med. 1931; 48:507.
Frazier, C.N., Hu, C.K. Nature and distribution according to age of cutaneous manifestations of vitamin A deficiency: study of 207 cases. Arch Derm Syph. 1936; 33:825.
Frensilli, J.A., Hinrichs, E.H. Dental changes of idiopathic hypoparathyroidism: report of three cases. J Oral Surg. 1971; 29:727.
Furman, K.I. Acute hypervitaminosis A in an adult. Am J Clin Nutr. 1973; 26:575.
Galili, D., Yatziv, S., Russell, A. Massive gingival hyperplasia preceding dental eruption in I: cell disease. Oral Surg. 1974; 37:533.
Gardner, D.G. Metachromatic cells in the gingiva in Hurler’s syndrome. Oral Surg. 1968; 26:782.
Gardner, D.G. The oral manifestations of Hurler’s syndrome. Oral Surg. 1971; 32:46.
Gardner, D.G. The dental manifestations of the Morquio syndrome Mucopolysaccharidosis type IV. Am J Dis Child. 1975; 129:1445.
Gardner, D.G., Majka, M. The early formation of irregular secondary dentine in progeria. Oral Surg. 1969; 28:877.
Gardner, D.G., Zeman, W. Biopsy of the dental pulp in the diagnosis of metachromatic leucodystrophy. Dev Med Child Neurol. 1965; 7:620.
Gildenhorn, H.L., Amromin, G.D. Report of a case with Niemann-Pick disease: correlation of roentgenography and autopsy findings. Am J Roentgenol Radium Ther Nucl Med. 1961; 85:680.
Glickman, I. Acute vitamin C deficiency and the periodontal tissues II: the effect of acute vitamin C deficiency upon the response of the periodontal tissues of the guinea pig to artificially induced inflammation. J Dent Res. 1948; 27:201.
Goldberg, A. The enzymatic formation of haem by the incorporation of iron in protoporphyrin; importance of ascorbic acid, ergothioneine and glutathione. Br J Haematol. 1959; 5:150.
Goldman, H. Experimental hyperthyroidism in guinea pigs. Am J Orthod Oral Surg. 1943; 29:665.
Idem: Report of histopathologic study of jaws of diet-deficiency in monkeys its relation to Vincent’s infection. Am J Orthod Oral Surg. 1943; 29:480.
Goodhart, R.S., Shils, M.E. Modern Nutrition in Health and Disease, 6, Philadelphia: Lea and Febiger, 1980.
Goodman, D.S. Vitamin A and retinoids: recent advances. Fed Proc. 1979; 38:2501.
Idem: Vitamin A metabolism. Fed Proc. 1980; 39:2716.
Gorlin, R.J. Genetic disorders affecting mucous membranes. Oral Surg. 1969; 28:512.
Graham, J.B., McFalls, V.W., Winters, R.W. Familial hypophosphatemia with vitamin D resistant rickets II: three additional kindreds of sex-linked dominant type with a genetic analysis of four such families. Am J Hum Genet. 1959; 1:311.
Green, D.E. Currents of Biochemical Research. Interscience Publishers, New York. 1956.
Greenspan, F.S., Baxter, J.D.Redding MA., Appleton, Lange, eds. Basic and Clinical Endocrinology. 1994;4.
Greenberg, D.M. Metabolic Pathways Vol I: Carbohydrates, Lipids, and Related Compounds (3rd ed). Academic Press, New York. 1967.
Idem: Metabolic Pathways Vol II: Amino Acids Nucleic Acids Porphyrins, Vitamins,Coenzymes (3rd ed.) Academic Press, New Yor. 1968;
Idem: Metabolic Pathways Vol III: Amino AcidsTetrapyrroles (3rd ed.) Academic Press, New Yor. 1969;
Idem: Metabolic Pathways Vol IV: Nucleic Acids Protein SynthesisCoenzymes (3rd ed.) Academic Press, New Yor. 1970;
Greenberg, M.S., Brightman, V.J., Lynch, M.A., Ship, I.I. Idiopathic hypoparathyroidism, chronic candidiasis, and dental hypoplasia. Oral Surg. 1969; 28:42.
Gÿorgy, P., Pearson, W.N., The Vitamins. Chemistry, Physiology, Pathology, Methods. 2. Academic Press, New York, 1967.
Hansen, A.E. Serum lipids in eczema and in other pathologic conditions. Am J Dis Child. 1937; 53:933.
Hamada, T., Lipoid, proteinosis. Clin Exp Dermatol. 2002; 27:624.
Hare, J.W. Signs and Symptoms in Endocrine and Metabolic Disorders. JB Lippincott, Philadelphia. 1986.
Hamada, T., Wessagowit, V., South, A.P., Ashton, G.H., et al. Extracellular Matrix Protein 1 Gene (ECM1) Mutations in Lipoid Proteinosis and Genotype-Phenotype Correlation. J Invest Dermatol. 2003; 120:345.
Harper, J.I., Filipe, M.I., Staughton, RCD. Lipoid protenosis: variations in histochemical characteristics. Clin Exp Dermatol. 1983; 8:135.
Harris, S.S., Navia, J.M. Vitamin A deficiency and caries susceptibility of rat molars. Arch Oral Biol. 1980; 25:415.
Hartman, K.S. Histiocytosis-X: a review of 114 cases with oral involvement. Oral Surg. 49(38), 1980.
Hassan, H., Hashim, S.A., Van Itallie, T.B., Sebrell, W.H. Syndrome in premature infants associated with low plasma vitamin E levels and high polyunsaturated fatty acid diet. Am J Clin Nutr. 1966; 19:147.
Haussler, M.R., McCain, T.A. Basic and clinical concepts related to vitamin D metabolism and action I. New Engl J Med. 1977; 297:974.
Idem: Basicclinical concepts related to vitamin D metabolism action II. New Engl J Med. 1977; 297:1041.
Hawkins, W.W., Barsky, J. An experiment on human vitamin B6 deprivation. Science. 1948; 108:284.
Hill, T.J. A Textbook of Oral Pathology, 4, Philadelphia: Lea and Febiger, 1949.
Hinrichs, E.H. Dental changes in juvenile hypothyroidism. J Dent Child. 1966; 23:167.
Hirsch, P.F., Munson, P.L. Thyrocalcitonin Physiol Rev. 1969; 49:548.
Hodges, R.E., Baker, E.M., Hood, J., Sauberlich, H.E., et al. Experimental scurvy in man. Am J Clin Nutr. 1969; 22:535.
Hofer, P.A., Bergenholtz, A. Oral manifestations in Urbach-Wiethe disease (lipoglycoproteinosis; lipoid proteinosis; hyalinosis cutis et mucosae. Odont Revy. 1975; 26:39.
Hojer, A. Method for determining the antiscorbutic value of a food stuff by means of histological examination of the teeth of young guinea pigs. Br J Exp Pathol. 1926; 7:356.
Hokin, L.E. Metabolic Pathways: Metabolic Transport Vol VI (3rd ed. Academic Press, New York. 1972.
Houpt, M.I., Kenny, F.M., Listgarten, M. Hypophosphatasia: case reports. J Dent Child. 1970; 37:126.
Hume, E.M., Krebs, H.A. Vitamin A requirement of human adults: an experimental study of vitamin A deprivation in man: a report of the Vitamin A subcommittee of the Accessory Food Factors Committee. Med Res Coun Spec Rep, Ser no 264, London, HMSO. 1949.
Hunder, G.G. Pathogenesis of Cushing’s disease. Mayo Clin Proc. 1966; 41:29.
Irving, J.T. Enamel organ of the rat’s incisor tooth in vitamin E deficiency. Nature. 1942; 150:122.
Idem: The effects of avitaminosishypervitaminosis A upon the incisor teeth incisal alveolar bone of rat. J Physiol. 1949; 108:92.
Jaffe, H.L., Lichtenstein, L. Eosinophilic granuloma of bone. Arch Pathol. 1944; 37:99.
Jardon, O.M., Burney, D.W., Fink, R.L. Hypophosphatasia in an adult. J Bone Joint Surg, 52–A: 1477. 1970.
Johnson, R.H. A unique case of pathologic calcification? Oral Surg. 1971; 32:66.
Jolly, M. Vitamin A deficiency: a review I. J Oral Therapeut Pharmacol. 1971; 3:364.
Idem: Vitamin A deficiency: a review II. J Oral Therapeut Pharmacol. 1967; 3:439.
Jones, J.C., Lilly, G.E., Marlette, R.H. Histiocytosis X. J Oral Surg. 1970; 28:461.
Kaye, M., Sagar, S. Effect of dihydrotachysterol on calcium absorption in uremia. Metabolism. 1972; 21:815.
King, C.G. Protein malnutrition: a major international problem. News Report Natl Acad Sci Natl Res Council. 1962; 12:37.
King, C.G., Waugh, W.A. The chemical nature of vitamin C. Science. 1932; 75:357.
Kjellman, M., Oldfelt, V., Nordenram, A., Olow, Nordenram M. Five cases of hypophosphatasia with dental findings. Int J Oral Surg. 1973; 2:152.
Klein, H., Orent, E., McCollum, E.V. The effects of magnesium deficiency on the teeth and their supporting structures in rats. Am J Physiol. 1935; 112:256.
Knudson, A.G., Jr. Inborn errors of sphingolipid metabolism. Am J Clin Nutr. 1961; 9:55.
Kosowicz, J., Rzymski, K. Abnormalities of tooth development in pituitary dwarfism. Oral Surg. 1977; 44:853.
Kruger, G.O., Prickman, L.E., Pugh, D.G. So-called eosinophilic granuloma of ribs and jaws associated with visceral (pulmonary) involvement characteristic of xanthomatosis. Oral Surg. 1949; 2:770.
Kumar V, Cotran RS, Robbins SL. Basic pathology (6th ed). WB Saunders, Philadelphia
Kyle, R.A., Bayrd, E.D. Amyloidosis: review of 236 cases. Medicine. 1975; 54:271.
Leahy, M.A., Krejci, S.M., Friednash, M., et al. Human Herpes virus 6 is present in lesions of Langerhans cell histiocytosis. J Invest Dermatol. 1993; 101:642.
Lahey, M.E. Prognosis in reticuloendotheliosis in children. J Pediatr. 1962; 60:664.
Lee, M., Stanmeyer, W., Wright, A. Nutrition and dental health, Part I, Assessment of human nutrition requirements. Carrboro NC. Health Sciences Consortium661. 1982.
Levin, B. Gaucher’s disease Clinical and roentgenologic manifestations. Am J Roentgenol, Radium Ther Nucl Med. 1961; 85:685.
Levin, B., Oberholzer, V.G., Snodgrass, GJAI., Stimmler, L., et al. Fructosemia: an inborn error of fructose metabolism. Arch Dis Child. 1963; 38:220.
Levin, B., Snodgrass, GJAI., Oberholzer, V.G., Burgess, E.A., et al. Fructosemia: Observation on cases. Am J Med. 1968; 45:826.
Levin, L.S., Jorgenson, R.J., Salinas, C.F. Oral findings in the Morquio syndrome (mucopolysaccharidosis IV. Oral Surg. 1975; 39:390.
Levy, B.M. Effects of pantothenic acid deficiency on the mandibular joints and periodontal structures of mice. J Am Dent Assoc. 1949; 38:215.
Idem: The effect of pyridoxine deficiency on the jaws of mice. J Dent Res. 1950; 29:349.
Idem: The effect of riboflavin deficiency on the growth of the mandibular condyle of mice. Oral Surg. 1949; 2:89.
Levy, B.M., Silberbert, R. Effect of riboflavin deficiency on endochondral ossification of mice. Proc Soc Exp Biod Med. 1946; 63:355.
Lichtenstein, L. Histiocytosis X Integrationof eosinophilic granuloma of bone, ‘Letterer-Siwe disease,’ and ‘Schüller-Christian disease’ as related manifestations of a single nosologic entity. Arch Pathol. 1953; 56:84.
Lichtenstein, L., Jaffe, H.L. Eosinophilic granuloma of bone. Am J Pathol. 1940; 16:595.
Lieberman, P.H., Jones, C.R., Dargeon, H.W.K., Begg, C.F. A reappraisal of eosinophilic granuloma of bone, Hand-Schüller-Christian syndrome and Letterer-Siwe syndrome. Medicine. 1969; 48:375.
Lin, J.H., Duffy, J.L., Roginsky, M.S. Microcirculation in diabetes mellitus. Hum Pathol. 1975; 6:77.
Livolsi, V.A., DeLellis, R.A. Pathology of the Parathyroid and Thyroid Glands. Williams and Wilkins, Baltimore. 1993.
Lloyd, H.M. Primary hyperparathyroidism: an analysis of the role of the parathroid tumor. Medicine. 1968; 47:53.
Loe, H. Periodontal changes in pregnancy. J Periodontal. 1965; 36:209.
Loeb, L. The Biological Basis of Individuality Springfield, Ill, Charles C Thoma. 1945;
Lotz, M., Zisman, E., Bartter, F.C. Evidence for a phosphorus-depletion syndrome in man. New Eng J Med. 1968; 278:409.
Lovestedt, S.A. Oral manifestations of histiocytosis-X. Dent Radiog Photog. 1977; 50:21.
Lovett, D.W., Cross, K.R., Van Allen, M. The prevalence of amyloids in gingival tissues. Oral Surg. 1965; 20:444.
Lucaya, J. Histiocytosis X. Am J Dis Child. 1971; 121:289.
Manouchehr-Pour, M., Bissada, N.F. Periodontal disease in juvenile and adult diabetic patients: a review of the literature. J Am Dent Assoc. 1983; 107:766.
Margolin, F.R., Steinbach, H.L. Progeria Hutchinson-Gilford syndrome. Am J Roentgenol, Radium Ther Med. 1968; 103:173.
Marks, S.C., Lindahl, R.L., Bawden, J.W. Dental and cephalometric findings in vitamin D resistant rickets. J Dent Child. 1965; 32:259.
Marthaler, T.M., Froesch, E.R. Hereditary fructose intolerance dental status of eight patients. Br Dent J. 1967; 123:597.
Massry, S.G., Coburn, J.W., Popovtzer, M.M., Shenaberger, J.H., et al. Secondary hyperparathyroidism in chronic renal failure. Arch Intern Med. 1969; 124:431.
McArthur, R.G., Cloutier, M.D., Hayles, A.B., Sprague, R.G. Cushing’s disease in children Findings in 13 cases. Mayo Clin Proc. 1972; 47:318.
McClain, K., Jin, H., Gresik, V., et al. Langerhans cell histiocytosis: lack of a viral etiology. Am J Hematol. 1994; 47:16.
McClendon, J.F. Fluorine is necessary in the diet of the rat. Fed Proc. 1944; 3:94.
McCollum, E.V., Davis, M. The nature of the dietary deficiencies of rice. J Biol Chem. 1915; 23:181.
McCollum, E.V., Simonds, N., Becker, J.E., Shipley, P.G. Studies on experimental rickets XXI. An experimental demonstration of the existence of a vitamin which promotes calicum deposition. J Biol Chem. 1922; 53:293.
McGavran, M.H., Spady, H.A. Eosinophilic granuloma of bone: a study of twentyeight cases. J Bone Joint Surg, 42–A: 979. 1960.
McKusick, V.A. Heritable Diseases of Connective Tissue, 4, St Louis: CV Mosby, 1972.
McKusick, V.A., Kaplan, D., Wise, D., Hanley, W.B., et al. The genetic mucopolysaccharidoses Medicine. 1965; 44:445.
Méhes, K., Klujber, L., Lassu, G., Kajtár, P. Hypophosphatasia: screening and family investigators in an endogamous Hungarian village. Clin Genet. 1972; 3:60.
Mellanby, E. A further demonstration of the part played by accessory food factors in the aetiology of rickets. J Physiol. 1919; 52:53.
Mellanby, E. An experimental investigation on rickets. Lancet. 1919; 1:407.
Mellanby, M. The influence of diet on teeth formation. Lancet. 1918; 2:767.
Mellanby, M.T. Diet and the teeth; an experimental study. Med Res Coun Spec Rep Ser no 191, London, HMSO. 1934.
Menaker, L., Navia, J.M. Effect of undernutrition during the perinatal period on caries development in the rat II: caries susceptibility in underfed rats supplemented with protein or calorie additions during the suckling period. J Dent Res. 1973; 52:680.
Idem: Effect of undernutrition during the perinatal period on caries development in the rat: III Effects of undernutrition on biochemical parameters in the developing submandibular salivary gland. J Dent Res. 1973; 52:688.
Idem: Effect of undernutrition during the perinatal period on caries development in the rat: IV Effects of differential tooth eruption exposure to a cariogenic diet on subsequent dental caries incidence. J Dent Res. 1974; 52:692.
Idem: Effect of undernutrition during the perinatal period on caries development in the rat: V Change in whole saliva volume protein content. J Dent Res. 1974; 53:592.
Mertz, W. The essential trace elements. Science. 1981; 213:1332.
Moch, W.S. Gaucher’s disease with mandibular bone lesions. Oral Surg. 1953; 6:1250.
Moy, L.S., Moy, R.L., Matsuoka, L.Y., Ohta, A., et al. Lipoid proteinosis: ultrastructural and biochemical studies. J Am Acad Dermatol. 1987; 16:1193.
Munroe, C.O. The dental patient and diabetes mellitus. Dent Clin North Am. 1983; 27:329.
Nanda, A., Alsaleh, Q.A., Al-Sabah, H., Ali, A.M., et al. Lipoid proteinosis: report of four siblings and brief review of the literature. Pediatr Dermatol. 2001; 18:21.
Navia, J.M. Evaluation of nutritional and dietary factors that modify animal caries. J Dent Res. 1970; 49:1091.
Navia, J.M., Di, Orio LP., Menaker, L., Miller, S. Effect of undernutrition during the perinatal period on caries development in the rat. J Dent Res. 1970; 49:1091.
Neufeld, E.F., Fratantoni, J.C. Inborn errors of mucopolysaccharide metabolism Faulty degradative mechanisms are implicated in this group of human disease. Science. 1970; 169:141.
Newbrun, E., Hoover, C., Mettraux, G., Graf, H. Comparison of dietary habits and dental health of subjects with hereditary fructose intolerance and control subjects. J Am Dent Assoc. 1980; 101:619.
Newton, J.A., Rasbridge, S., Temple, A., Pope, F.M., et al. Lipoid proteinosis-new immunopathological observations. Clin Exp Dermatol. 1991; 16:350.
Nitowsky, H.M., Cornblath, M., Gordon, H.H. Studies of tocopherol deficiency in infants and children II Plasma tocophrol and erythrocyte hemolysis in hydrogen peroxide. Am J Dis Child. 1956; 92:164.
Nitowsky, H.M., Tildon, J.T., Levin, S., Gordon, H.H. Studies of tocopherol deficiency in infants and children VII. The effect of tocopherol in urinary, plasma and muscle creatinine. Am J Clin Nutr. 1962; 10:368.
Nizel, A. Nutrition in clinical dentistry, 3, Philadelphia: WB Saunders, 1989.
Nordin, BEC. Pathogenesis of osteoporosis. Lancet. 1961; 1:1011.
Oezarmagan, G., Baykal, C., Gursoy, E.O., Yilmazer, S., et al. Lipoid proteinosis in 2 sisters Hautarzt. 1993; 44:315.
Olcott, H.S., Emerson, O.H. Antioxidants and the autoxidation of fats IX: the antioxidant properties of the tocopherols. J Am Chem Soc. 1937; 59:1008.
Otani, S. A discussion of eosinophilic granuloma of bone, Letterer-Siwe disease and Schüller-Christian disease. Mt Sinai J Med. 1957; 24:1079.
Otani, S., Ehrlich, J.C. Solitary granuloma of bone simulating primary neoplasm. Am J Pathol. 1940; 16:479.
Paige, D.M. Clinical Nutrition. CV Mosby, St Louis. 1988.
Peterkofsky, B., Udenfriend, S. Enzymatic hydroxylation of proline in microsomal polypeptide leading to formation of collagen. Proc Natl Acad Sci, USA. 1965; 53:335.
Peters, S.P., Van Slyke, D.D. Quantitative Clinical Chemistry Vol 1. Williams and Wilkins, Baltimore. 1931.
Pitt, M.J., Haussler, M.R. Vitamin D: biochemistry and clinical applications. Skeletal Radiol. 1977; 1:198.
Plowman, P.N. Endocrinology and Metabolic Diseases. Medical Examination, New York. 1987.
Prasad, A.S., Halsted, J.A., Nadimi, M. Syndrome of iron deficiency anemia, hepatosplenomegaly, hypogonadism, dwarfism and geophagia. Am J Med. 1961; 31:532.
Prasad, A.S., Miale Jr, A., Farid, Z., Sandstead, H.H., et al. Biochemical studies on dwarfism, hypogonadism and anemia. Arch Intern Med. 1963; 111:65.
Prasad, A.S., Schulert, A.R., Miale Jr, A., Farid, Z., et al. Zinc and iron deficiency in male subjects with dwarfism and hypogonadism but without ancylostomiasis, schistosomiasis, or severe anemia. Am J Clin Nutr. 1963; 12:437.
Pryor, W.A. Free radical pathology. Chem, Eng, News. 1971; 49:34.
Rao, G.S. Dietary intake and bioavailability of fluoride. Annu Rev Nutr. 1984; 4:115.
Ramsay, I., Bayliss, R. A Synopsis of Endocrinology and Metabolism. Bristol, England, Wright. 1986.
Rapidis, A.D., Longdon, J.D., Harvey, P.W., Patel, M.F. Histiocytosis-X: an analysis of 50 cases. Int J Oral Surg. 1978; 7:76.
Rasmussen, H., Pechet, M.M. Calcitonin. Sci Am, 223: 42, Octo. 1970.
Rathbun, J.C. Hypophosphatasia. Am J Dis Child. 1948; 75:822.
Reich, C., Seife, M., Kessler, B.J. Gaucher’s disease: a review and discussion of twenty cases. Medicine. 1951; 30:1.
Reiner, A. Oral implication of diabetes. Ann Dent. 1977; 36:46.
Richards, I.D.G., Sweet, E.M., Arneil, G.C. Infantile rickets persists in Glasgow. Lancet. 1968; 1:803.
Rigdon, R.H. Occurrence and association of amyloid with diseases in birds and mammals including man: a review. Tex Rep Biol Med. 1974; 32:665.
Ritchie, GMacL. Hypophosphatasia: a metabolic disease with important dental manifestations. Arch Dis Child. 1964; 39:584.
Rogers, D.R. Screening for amyloid with the thioflavin-T fluorescent method. Am J Clin Pathol. 1965; 44:59.
Root, A. Growth hormone, Pediatrics. 1965; 36:940.
Rose, W.C., Haines, W.J., Johnson, J.E. The role of the amino acids in human nutrition. J Biol Chem. 1942; 146:683.
Rotruck, J.T., Pope, A.L., Ganther, H.E., Swanson, A.B., et al. Selenium: biochemical role as a component of glutathione peroxidase. Science. 1973; 179:588.
Russell, B.G. Gingival changes in diabetes mellitus. Acta Pathol Microbiol Scand. 1966; 68:161.
Idem: The dental pulp in diabetes mellitus. Acta Pathol Microbiol Scand, 70: 319, 1967. Idem: The periodontal membrane in diabetes mellitus. Acta Pathol Microbiol Scand. 1967; 70:318.
Salley, J.J., Bryson, W.F., Eshleman, J.R. The effect of chronic vitamin A: deficiency on dental caries in the Syrian hamster. J Dent Res. 1959; 38:1038.
Sayers, G. The adrenal cortex and homeostasis. Physiol Rev. 1950; 30:241.
Sbarbaro, J.L., Francis, K.C. Eosinophilic granuloma of bone. J Am Med Assoc. 1961; 178:706.
Schofield, F.W. A bried account of a disease of cattle simulating hemorrhagic septicemia due to feeding of sweet clover. Can Vet J. 1922; 3:74.
Schour, I., Massler, M. Endocrines and dentistry. J Am Dent Assoc. 30(597, 763, 943), 1943.
Idem: The effects of dietry deficiencies upon the oral structures. Physiol Rev. 1945;25(442).
Schour, I., Van Dyke, H.B. Changes in the teeth following hypophysectomy I: changes in the incisor of the white rat. Am J Anat. 1932; 50:397.
Schour, I., Hoffman, M.M., Smith, M.C. Changes in the incisor teeth of albino rats with vitamin A deficiency and the effects of replacement therapy. Am J Pathol. 1941; 17:529.
Schroff, J. Eosinophilic granuloma of bone. Oral Surg. 1948; 1:256.
Scriver, C.R., Cameron, D. Pseudohypophosphatasia. New Engl J Med. 1969; 281:604.
Sebrell, W.H., Butler, R.E. Riboflavin deficiency in man (ariboflavinosis. Public Health Rep. 1939; 54:2121.
Sebrell, W.H., Harris, R.S. The Vitamins. Academic Press, New York. 1967.
Sedano, H.O., Cernea, P., Hosxe, G., Gorlin, R.J. Histiocytosis X: clinical, radiologic, and histologic findings with special attention to oral manifestations. Oral Surg. 1969; 27:760.
Selye, H. General adaptation syndrome and diseases of adaptation. J Clin Endocr. 1946; 6:117.
Idem: The alarm reaction the diseases of adaptation. Ann Intern Med. 1948; 26:403.
Selye, H., et al. Stress Montreal. Acta Inc. 1950, 1951, 1952, 1953, 1954; 1955–1956.
Sethuraman, G., Tejasvi, T., Khaitan, B.K., Handa, K.K., et al. Lipoid Proteinosis in Two Siblings: A Report from India. J Dermatol 30: 562. 2003.
Shafer, W.G., Muhler, J.C. Effect of gonadectomy and sex hormones on the structure of the rat salivary glands. J Dent Res. 1953; 32:262.
Shklar, G., McCarthy, P.L. The Oral Manifestations of Systemic Disease. Butterworths, Boston. 1976.
Sherman, H.C. Chemistry of Food and Nutrition. Macmillan Company, New York. 1947.
Shils, M.E. Experimental human magnesium depletion. Medicine. 1969; 48:61.
Silverman, S., Gordon, G., Grant, T., Steinbach, H., et al. The dental structures in hyperparathyroidism. Oral Surg. 1962; 15:426.
Silverman Jr, S., Ware, W.H., Gillooly, C., Jr. Dental aspects of hyperparathyroidism. Oral Surg. 26(184), 1968.
Sleeper, E.L. Eosinophilic granuloma of bone: its relationship to Hand-SchüllerChristian and Letterer-Siwe’s diseases, with emphasis upon oral symptoms and findings. Oral Surg. 1951; 4:896.
Sonis, S.T., Fazio, R.C., Fang, L. Principles and Practice of Oral Medicine. WB Saunders, Philadelphia. 1984.
Somjen, G., Hilmy, M., Stephens, C.R. Failure to anesthetize human subjects by intravenous administration of magnesium sulfate. J Pharmacol Exp Ther. 1966; 154:652.
Spolnik, K.J., Patterson, S.S., Maxwell, D.R., Kleit, S.A., et al. Dental radiographic manifestations of end-stage renal disease. Dent Radiogr Photogr. 1981; 54:21.
Stahl, S.S., Robertson, H.C. Oral lesions in Hand-Schüller-Christian disease: report of case. Oral Surg. 1955; 8:319.
Stanback, J.S., Peagler, F.D. Primary amyloidosis: review of the literature and report of a case. Oral Surg. 1968; 26:774.
Stanbury, J.B., Wyngaarden, J.B., Fredrickson, D.S. The Metabolic Basis of Inherited Disease, 4, New York: McGraw-Hill, 1978.
Standinger, H.J., Krisch, K., Leonhauser, S. Role of ascorbic acid in microsomal electron transport and the possible relationship to hydroxylation reactions. Ann New York Acad Sci. 1961; 92:195.
Steenbock, H. The induction of growth promoting and calcifying properties in a ration by exposure to light. Science. 1924; 60:224.
Steenbock, H., Black, A. Fat soluble vitamins; the induction of growth promoting and calcifying properties in a ration by exposure to ultra-violet light. J Biol Chem. 1924; 61:405.
Steenbock, H., Buotwell, P.W. Fat soluble vitamine V: the stability of the fat soluble vitamine in plant materials. J Biol Chem. 1920; 41:163.
Steenbock, H., Sell, M.T. Fat soluble vitamine X: further observations on the occurrence of the fat soluble vitamine with yellow plant pigments. J Biol Chem. 1922; 51:63.
Steenbock, H., Hart, E.B., Jones, J.H. Fat soluble vitamine XVIII: sunlight in its relation to pork production on certain restricted rations. J Biol Chem. 1924; 61:405.
Steffens, L.F., Bair, H.L., Sheard, C. Dark adaptation and dietary deficiency in vitamin A. Am J Ophthalmol. 1940; 23:1325.
Stickler, G.B., Beabout, J.W., Riggs, B.L. Vitamin D resistant rickets: clinical experience with 41 typical familial hypophosphatemic patients and 2 atypical nonfamilial cases. Mayo Clin Proc. 1970; 45:197.
Svirbely, J.L., Szent-Györgyi, A. CV: the chemical nature of vitamin C. Biochem J. 1932; 26:865.
Sweet, L.K., K’ang, H.J. Clinical and anatomic study of avitaminosis A: among Chinese. Am J Dis Child. 1935; 50:699.
Taylor, G.F., Day, DCM. Osteomalacia and dental caries. Br Med J. 1940; 2:221.
Teng, C.T., Nathan, M.H. Primary hyperparathyroidism. Am J Roentgenol Radium Ther Nucl Med. 1960; 83:716.
Thoma, K.H. Oral Pathology, 4, St Louis: CV Mosby, 1954.
Thompson, SW II., Gell, R.G., Yamanaka, H.S. A histochemical study of the protein nature of amyloid. Am J Pathol. 1961; 38:737.
Tracy, W.E., Steen, J.C., Steiner, J.E., Buist, NRM. Analysis of dentine pathogenesis in vitamin D-resistant rickets. Oral Surg. 1971; 32:38.
Ulmansky, M. Primary amyloidosis of oral structures and pharynx: report of a case. Oral Surg. 1962; 15:800.
Underwood, E.J. Trace Elements in Human and Animal Nutrition. Academic Press, New York. 1977.
Vallee, B.L., Wacker, W.E.C., Ulmer, D.D. Magnesium deficiency tetany syndrome in man. New Engl J Med. 1960; 262:155.
van, der Waal I., Fehmers, M.C.O., Kraal, E.R. Amyloidosis: its significance in oral surgery. Oral Surg. 1973; 36:469.
Van, Wyk CW. The oral mucosa in kwashiorkor: a clinicocytological study. J Dent Assoc S Afr. 1965; 20:298.
Vasilakis, G.J., Nygaard, V.K., DiPalma, D.M. Vitamin D resistant rickets A: review and case report of an adolescent boy with a history of dental problems. J Oral Med. 1980; 35:19.
Van, Dis ML., Allen, C.M., Neville, B.W. Erythematous gingival enlargement in diabetic patients: a report of four cases. J Oral Maxillofac Surg. 1988; 46:794.
Vogel, H.J. Metabolic Pathways: Metabolic Regulation Vol V (3rd ed. Academic Press, New York. 1971.
Wald, G. Molecular basis of visual excitation. Science. 1968; 162:230.
Warkany, J., Nelson, R.C. Skeletal abnormalities induced in rats by maternal nutritional deficiency. Arch Pathol. 1942; 34:375.
Webman, M.S., Hirsch, S.A., Webman, H. Stanley HR: obliterated pulp cavities in the San Filippo syndrome (mucopolysaccharidosis III. Oral Surg. 1977; 43:734.
Weinfeld, A., Stern, M.H., Marx, L.H. Amyloid lesions of bone. Am J Roentgenol Radium Ther Nucl Med. 1970; 108:799.
Wesley, RK Delaney JR., Litt, R. Progeria: clinical considerations for an isolated case. J Dent Child. 1979; 46:1.
Whedon, G.D. Effects of high calcium intakes on bones, blood and tissue: relationship of calcium intake to balance in osteoporosis. Fed Proc. 1959; 18:1112.
Williams, R.F. Lipoid proteinosis: report of a case. Oral Surg. 1971; 31:624.
Willman, C.L., McClain, K.L. An update on clonality, cytokines, and viral etiology in Langerhans cell histiocytosis. Hematol Oncol Clin North Am. 1998; 12:408.
Wilson, D.R., York, S.E., Jaworski, Z.F., Yendt, E.R. Studies in hypophosphatemic vitamin D: refractory osteomalacia in adults. Medicine. 1965; 44:99.
Wilson, J.R., DuBois, R.O. Report of a fatal case of keratomalacia in an infant with postmortem examination. Am J Dis Child. 1923; 26:431.
Winkelmann, R.K. The skin in histiocytosis X. Mayo Clin Proc. 1969; 44:535.
Winters, R.W., Graham, J.B., Williams, T.F., McFalls, V.W., et al. A genetic study of familial hypophosphatemia and vitamin D: resistant rickets. Trans Assoc Am Physicians. 1957; 70:234.
Idem: A genetic study of familial hypophosphatemiavitamin D: resistant rickets with a review of the literature. Medicine. 1958; 37:97.
Witkop, C.J., Roa, S. Inherited defects in tooth structure. Birth Defects, 7: 153, 1971. Wolbach SB, Bessey OA. Tissue changes in vitamin deficiencies. Physiol Rev. 1942; 22:233.
Wolbach, S.B., Howe, P.R. The incisor teeth of albino rats and guinea pigs in vitamin deficiency and repair. Am J Pathol. 1933; 9:275.
Ziskin, D.E., Applebaum, E. Effects of thyroidectomy and thyroid stimulation on growing permanent dentition of rhesus monkeys. J Dent Res. 20(21), 1941.
Ziskin, D.E., Applebaum, E., Gorlin, R.J. The effect of hypophysectomy upon the permanent dentition of rhesus monkeys. J Dent Res. 1949; 28:48.
Ziskin, D.E., Stein, G., Gross, P., Runne, E. Oral gingival and periodontal pathology induced in rats on low pantothenic acid diet by toxic doses of zinc carbonate. Am J Orthod Oral Surg. 1947; 33:407.