A summary of the examination of chronic kidney disease and extending the genitourinary examination
Extending the genitourinary examination
INVESTIGATIONS
Kidney disease is investigated by the following:
1. Examination of the urine (e.g. dipstick testing, urine culture for infection) and urinary sediment.
2. Tests of renal function—for example, glomerular function by serum creatinine and estimated glomerular filtration rate (e-GFR, which is derived from the serum creatinine level), and tubular function by measuring electrolytes and urine (pH, specific gravity, glucose, protein excretion).
3. Blood tests to search for the causes of renal dysfunction—for example, looking for renal disease: hepatitis B or C, HIV, complement and immune complexes, tests for autoimmune disease, immunoelectrophoresis.
4. Blood tests to assess the effects of renal disease—for example, electrolyte changes; blood count (for anaemia); serum glucose (diabetes mellitus); calcium, phosphate and parathyroid hormone; uric acid.
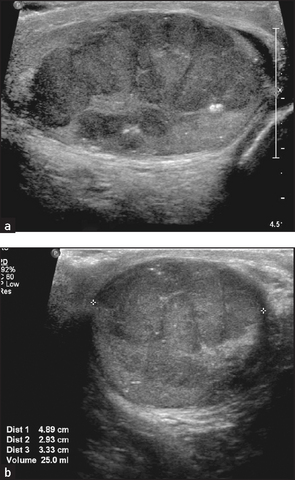
5. Ultrasound to look at renal size and for any renal mass or urinary obstruction (ureters and bladder); and by arterial Doppler to measure renal blood supply. Ultrasound of the scrotum is used to identify scrotal masses. Note the kidneys are usually both small on ultrasound in chronic kidney disease, but the exceptions include diabetes mellitus and amyloid or polycystic kidneys.
6. Renal biopsy—for example, to diagnose glomerulonephritis.
The urine
The ghosts of dead patients that haunt us do not ask why we did not employ the latest fad of clinical investigation; they ask why did you not test my urine?
This valuable fluid must not be discarded in any patient in whom a renal, diabetic, gastrointestinal or other major system disease is suspected.
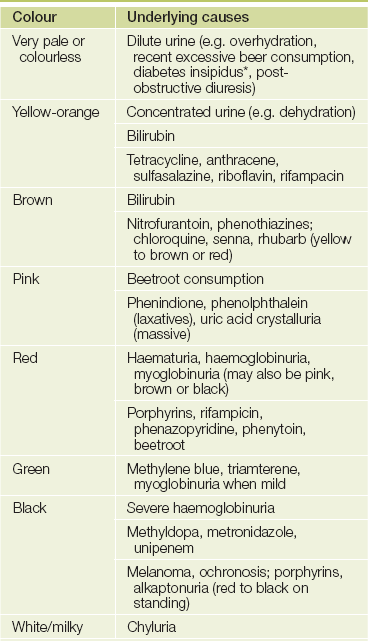
Colour
Look at the colour of the urine (see Table 19.1).
TABLE 19.1
Some causes of urine colour changes
*Diabetes comes from a Greek word meaning ’passing through’ and refers to the large amount of urine passed by people with uncontrolled diabetes mellitus or with diabetes insipidus. Mellitus means ’sweet’ and insipidus means ’tasteless’. We should be grateful that taste testing is no longer a part of the urinary examination.
Transparency
Phosphate or urate deposits can occur normally and produce white (phosphate) or pink (urate) cloudiness. Fainter cloudiness may be due to bacteria. Pus, chyle (lymphatic fluid) or blood can cause a more turbid appearance.
Smell
A mild ammoniacal smell is normal. A UTI causes a fishy smell, and antibiotics can sometimes be smelt in the urine, as can asparagus.
Specific gravity
A urinometer, which is a weighted float with a scale, is used to measure specific gravity. The depth to which the float sinks in the urine indicates the specific gravity, which is read off the scale on the side. The specific gravity can also be estimated by dipstick methods.
Water has a specific gravity of 1, and the presence of solutes (especially heavy solutes such as glucose or an iodine contrast medium) in urine increases the specific gravity. The normal range is 1.002 to 1.025. A consistently low specific gravity suggests CKD (as there is failure of the kidneys to concentrate the urine) or diabetes insipidus (where there is a deficiency of antidiuretic hormone resulting in passage of a large volume of dilute urine). A high specific gravity suggests fluid volume depletion or diabetes mellitus with the presence of large amounts of glucose in the urine.
There is a rough correlation between the specific gravity of the urine and its osmolarity. For example, a specific gravity of 1.002 corresponds to an osmolarity of 100 mOsm/kg, while a specific gravity of 1.030 corresponds to 1200 mOsm/kg.
Chemical analysis
A chemical reagent colour strip allows simultaneous multiple analyses of pH, protein, glucose, ketones, blood, nitrite, specific gravity, the presence of leucocytes, bile and urobilinogen. The strip is dipped in the urine and colour changes are measured after a set period. The colours are compared with a chart provided. It should be noted that the specific gravity by dipstick is pH dependent and insensitive to non-ionised molecules, and therefore correlates poorly with urine osmolality.
pH
Normal urine is acidic, except after meals when for a short time it becomes alkaline (the alkaline tide). Measuring the pH of urine is helpful in a number of critical circumstances. Sometimes the urine has to be made alkaline for therapeutic purposes, such as treating myoglobinuria or recurrent urinary calculi due to uric acid or cystine. Distal renal tubular acidosis should be suspected if the early morning urine is consistently alkaline and cannot be acidified. UTIs with urea-splitting organisms, such as Proteus mirabilis, can also cause an alkaline urine which, in turn, favours renal struvite stone formation.
Protein
The colours are compared with a chart provided. The strip tests give only a semi-quantitative measure of urinary protein (+ to ++++) and, if positive, must be confirmed by other tests. It is very important to note that the dipstick is sensitive to albumin but not to other proteins. A reading of + of proteinuria may be normal, as up to 150 mg of protein a day is lost in the urine. Causes of abnormal amounts of protein in the urine are outlined in Lists 19.1 and 19.2. Chemical dipsticks do not detect the presence of Bence-Jones proteinuriaa (immunoglobulin light chains).
If proteinuria is detected on dipstick testing, this should be quantified and careful urine (phase-contrast) microscopy should be carried out to look for evidence of active renal disease.
Glucose and ketones
A semi-quantitative measurement of glucose and ketones is available. Glycosuria usually indicates diabetes mellitus, but can occur with other diseases (see List 19.3). False-positive or false-negative results can occur with vitamin C (large doses), bacteria, oxidising detergents and hydrochloric acid, tetracyclines or levodopa ingestion.
Ketones in the urine of patients with diabetes mellitus are an important indication of the presence of diabetic ketoacidosis (see List 19.3). The three ketone bodies are acetone, betahydroxybutyric acid and acetoacetic acid. Lack of glucose (starvation) or lack of glucose availability for the cells (diabetes mellitus) causes activation of carnitine acetyltransferase, which accelerates fatty-acid oxidation in the liver. However, the pathway for the conversion of fatty acids becomes saturated, leading to ketone body formation. The strip tests react only to acetoacetic acid. Ketonuria may also be seen associated with fasting, vomiting and strenuous exercise.
Blood
Blood in the urine (haematuria) is abnormal and can be seen with the naked eye if 0.5 mL is present per litre of urine (see List 19.4). Blood may be a contaminant of the urine when women are menstruating. A positive dipstick test is abnormal and suggests haematuria, haemoglobinuria (uncommon) or myoglobinuria (also uncommon). The presence of more than a trace of protein in the urine in addition suggests that the blood is of renal origin. False-positives may occur when there is a high concentration of certain bacteria and false-negative results can occur if vitamin C is being taken.
Nitrite
If positive, this usually indicates infection with bacteria that produce nitrite. More-specific dipstick tests for white cells are now available; a positive test has an LR of 4.2 for a urinary infection and a negative test has an LR of 0.3.1
The urine sediment
Every patient with suspected renal disease should have a midstream urine sample examined (see Good signs guide 19.1). Centrifuge 10 mL of the urine at 2000 rpm for 4 minutes. Remove the supernatant, leaving 0.5 mL; shake well to re-suspend, then place one drop on a slide with a coverslip. Look at the slide using a low-power microscope and at specific formed elements under the high-power field (HPF) for identification. There is a significant false-negative rate when there are low numbers of formed elements in the urine. Look for red blood cells (RBCs), white blood cells (WBCs) and casts.
Red blood cells
These appear as small circular objects without a nucleus. Usually none are seen, although up to 5 RBCs/low-power field (LPF) may be normal in very concentrated urine. If their numbers are increased, try to determine whether the RBCs originate from the glomeruli (more than 80% of the RBCs are dysmorphic—irregular in size and shape) or the renal tract (the RBCs are typically uniform).
White blood cells
These cells have lobulated nuclei. Usually fewer than 6 WBCs/HPF are present, although up to 10 may be normal in very concentrated urine. Tubular epithelial cells have a compact nucleus and are larger. Pyuria indicates urinary tract inflammation. Bacteria may also be seen if there is infection, but bacterial contamination is more likely if squamous epithelial cells (which are larger and have single nuclei) are prominent. Sterile pyuria is characteristic of renal tuberculosis but may also occur in acute or chronic tubulo-interstitial disease. Multistix test strips will often test for the presence of WBCs.
Casts
Casts are cylindrical moulds formed in the lumen of the renal tubules or collecting ducts (see Figure 19.3). They are signs of a damaged glomerular basement membrane or damaged tubules. The size of a cast is determined by the dimension of the lumen of the nephron in which it forms. The presence of casts is a very important abnormality and indicates renal disease.
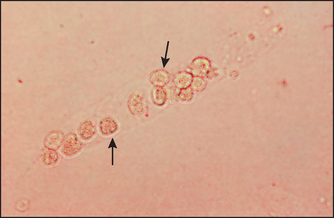
Figure 19.3 Renal tubular cell cast. The cast has numerous renal tubular cells with round nuclei (arrows). These casts are a sign of acute tubular necrosis. (Henry JB. Clinical diagnosis and management by laboratory methods, 20th edn. Saunders, 2001.)
Hyaline casts are long cylindrical structures. One or two RBCs or WBCs may be present in the cast. Normally there are fewer than 1 per LPF. They consist largely of Tamm-Horsfall mucoprotein secreted by the renal tubules.
Granular casts are abnormal cylindrical granular structures that arise from the tubules, usually in patients with proteinuria. They consist of hyaline material containing fragments of serum proteins.
Red cell casts are always abnormal and indicate primary glomerular disease (haematuria of glomerular origin or vasculitis). They contain 10 to 50 red cells, which are well defined.
With white cell casts many WBCs adhere to or inside the cast. These are abnormal, indicating bacterial pyelonephritis or, less commonly, glomerulonephritis, kidney infarction or vasculitis.
Fatty casts (i.e. the presence of fat in casts) are suggestive of the nephrotic syndrome.
IMAGING THE KIDNEYS
Some common renal scan findings are presented in Figures 19.4–19.8.
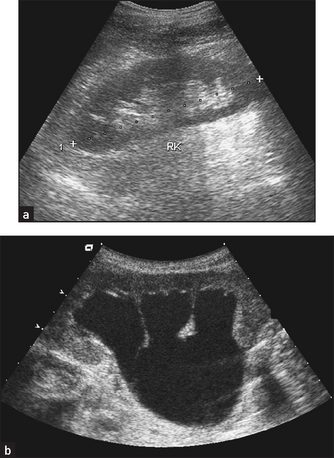
Figure 19.4 Ultrasound of (a) normal kidneys and (b) hydronephrosis with cortical atrophy (From Adam A et al. Grainger & Allison’s diagnostic radiology, 5th edn. Churchill Livingstone, 2008.) Churchill Livingstone
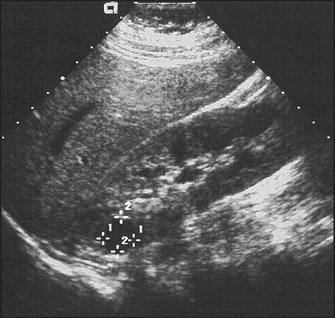
Figure 19.5 Ultrasound of polycystic kidneys. There are multiple discrete cysts, the largest 1.2 × 1.2 centimetres. (From Kaplan BS, Meyers K. Paediatric nephrology and urology: requisites, 1st edn. Mosby, 2004.)
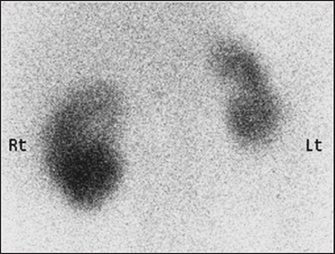
Figure 19.6 Nuclear scan of small scarred left kidney in a patient with hypertension. There is an area of infarction in the upper pole of the right kidney. (From Handbook of systemic autoimmune diseases, Volume 7, 2007.)
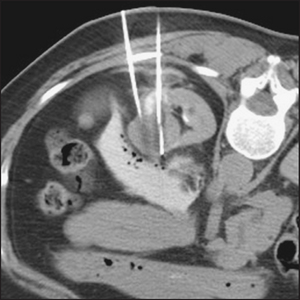
Figure 19.7 CT scan of renal tumour showing needles used for ablation treatment (From Allan PL, Baxter GM. Clinical ultrasound, 3rd edn. Churchill Livingstone, 2011.)
aHenry Bence-Jones (1818–73), physician at St George’s Hospital, London, described this in 1848.