Chronic Diarrhea
Alfredo Guarino, David Branski, Harland S. Winter
Definition and Epidemiology
Chronic diarrhea is defined as stool volume of more than 10 g/kg/day in toddlers/infants and greater than 200 g/day in older children that lasts for 14 days or more. In practice, this usually means having loose or watery stools more than 3 times a day. Awakening at night to pass stool is often a sign of an organic cause of diarrhea. The epidemiology has 2 distinct patterns. In developing countries, chronic diarrhea is, in many cases, the result of an intestinal infection that persists longer than expected. This syndrome is often defined as protracted diarrhea, but there is no clear distinction between protracted and chronic diarrhea. In countries with higher socioeconomic conditions, chronic diarrhea is less frequent and the etiology often varies with age. The outcome of diarrhea depends on the cause and ranges from benign, self-limited conditions, such as toddler's diarrhea, to severe congenital diseases, such as microvillus inclusion disease, that may lead to progressive intestinal failure.
Pathophysiology
The mechanisms of diarrhea are generally divided into secretory and osmotic, but often diarrhea is a combination of both mechanisms. In addition, inflammation and motility disorders may contribute to diarrhea. Secretory diarrhea is usually associated with large volumes of watery stools and persists when oral feeding is withdrawn. Osmotic diarrhea is dependent on oral feeding, and stool volumes are usually not as massive as in secretory diarrhea (Fig. 341-1).
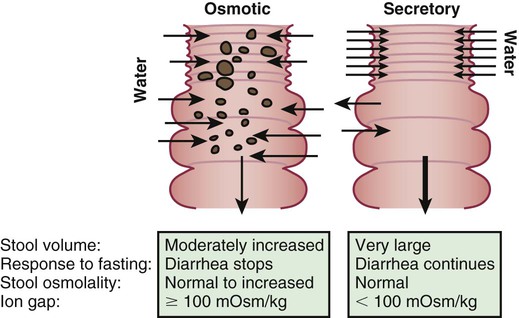
Secretory diarrhea is characterized by active electrolyte and water fluxes toward the intestinal lumen, resulting from either the inhibition of neutral NaCl absorption in villous enterocytes or an increase in electrogenic chloride secretion in secretory crypt cells as a result of the opening of the cystic fibrosis transmembrane regulator (CFTR) chloride channel or both. The result is more secretion from the crypts than absorption in the villous that persists during fasting. The other components of the enterocyte ion secretory machinery are (1) the Na-K 2Cl cotransporter for the electroneutral chloride entrance into the enterocyte; (2) the Na-K pump, which decreases the intracellular Na+ concentration, determining the driving gradient for further Na+ influx; and (3) the K+ selective channel, that enables K+, once it has entered the cell together with Na+, to return to the extracellular fluid.
Electrogenic secretion is induced by an increase of intracellular concentration of cyclic adenosine monophosphate, cyclic guanosine monophosphate, or calcium in response to microbial enterotoxins, or to endogenous endocrine or nonendocrine moieties, including inflammatory cytokines. Another mechanism of secretory diarrhea is the inhibition of the electroneutral NaCl-coupled pathway that involves the Na+/H+ and the Cl−/HCO3− exchangers. Defects in the genes of the Na+/H+ and the Cl−/HCO3− exchangers are responsible for congenital Na+ and Cl− diarrhea, respectively.
Osmotic diarrhea is caused by nonabsorbed nutrients in the intestinal lumen as a result of 1 or more of the following mechanisms: (1) intestinal damage (e.g., enteric infection); (2) reduced absorptive surface area (e.g., active celiac disease); (3) defective digestive enzyme or nutrient carrier (e.g., lactase deficiency); (4) decreased intestinal transit time (e.g., functional diarrhea); and (5) nutrient overload, exceeding the digestive capacity (e.g., overfeeding, sorbitol in fruit juice). Whatever the mechanism, the osmotic force generated by nonabsorbed solutes drives water into the intestinal lumen. A very common example of osmotic diarrhea is lactose intolerance. Lactose, if not absorbed in the small intestine, reaches the colon, where it is fermented to short-chain organic acids, releasing hydrogen that is detected in the lactose breath test, and generating an osmotic overload. In many children chronic diarrhea may be caused by multiple mechanisms.
Etiology
Enteric infections are by far the most frequent cause of chronic diarrhea, both in developing and industrialized countries but, outcomes are often very different. In the former, comorbid conditions, such as HIV/AIDS, malaria, or tuberculosis, result in malnutrition that impairs the child's immune response, thereby potentiating the likelihood of prolonging diarrhea or acquiring another enteric infection. In children with HIV/AIDS, the viral infection itself impairs immune function and may trigger a vicious circle with malnutrition. Sequential infections with the same or different pathogens may also be responsible for chronic diarrhea.
In developing countries, enteroadherent Escherichia coli and Giardia lamblia have been implicated in chronic diarrhea, whereas, in developed countries, chronic infectious diarrhea usually runs a more benign course and the etiology is often viral, with a major role of rotavirus and norovirus (Table 341-1). Opportunistic microorganisms induce diarrhea exclusively, more severely or for more prolonged periods, in specific populations, such as immunocompromised children. Specific agents cause chronic diarrhea or exacerbate diarrhea in many chronic diseases. Clostridium difficile or cytomegalovirus act as opportunistic agents in oncologic patients as well as in patients with inflammatory bowel diseases. Cryptosporidium may induce severe and protracted diarrhea in AIDS patients.
Table 341-1
A Comparative List of Prevalent Agents and Conditions in Children with Persistent Infectious Diarrhea in Industrialized and Developing Countries
| AGENT/DISEASE | |
| INDUSTRIALIZED COUNTRIES | DEVELOPING COUNTRIES |
Rotavirus* | |

In small intestinal bacterial overgrowth, diarrhea may be the result of either a direct interaction between the microorganism and the enterocyte or the consequence of deconjugation and dehydroxylation of bile salts, and hydroxylation of fatty acids due to an increased proliferation of bacteria in the proximal intestine. Postenteritis diarrhea syndrome is a clinicopathologic condition in which small intestinal mucosal damage persists after acute gastroenteritis. Sensitization to food antigens, secondary disaccharidase deficiency, persistent infections, reinfection with an enteric pathogen, or side effects of medication may be responsible for causing postenteritis diarrhea syndrome, thought to be related to perturbations of the intestinal microbiome. Functional diarrhea which may be related to the pathogenesis of irritable bowel syndrome may be caused by complications of an acute gastroenteritis. Noninfectious chronic diarrhea is the manifestation of a broad number of heterogeneous conditions that vary with the age of the patient (Table 341-2; see also Table 336-5).
Table 341-2
Main Etiologies of Noninfectious Chronic Diarrhea in Children Older and Younger Than 2 Yr of Age
| ETIOLOGY | YOUNGER THAN 2 YR | OLDER THAN 2 YR |
| Abnormal digestive processes | Shwachman-Diamond syndrome, isolated pancreatic enzyme deficiency, chronic pancreatitis, Johanson-Blizzard syndrome, Pearson syndrome. Trypsinogen and enterokinase deficiency: chronic cholestasis; use of bile acids sequestrants; primary bile acid malabsorption | Cystic fibrosis, terminal ileum resection |
| Nutrient malabsorption | Congenital sucrase-isomaltase deficiency; congenital lactase deficiency; glucose-galactose malabsorption; fructose malabsorption; congenital short bowel | Hypoalactasia; acquired short bowel |
| Immune/inflammatory | Food allergy; autoimmune enteropathy; primary and secondary immunodeficiencies; IPEX syndrome | Celiac disease; eosinophilic gastroenteritis, inflammatory bowel diseases |
| Structural defects | Microvillus inclusion disease, tufting enteropathy, phenotypic diarrhea, heparan-sulphate deficiency, α2β1 and α6β4 integrin deficiency, lymphangiectasia, enteric anendocrinosis (neurogenin-3 mutation) | Rare |
| Defects of electrolyte and metabolite transport | Congenital chloride diarrhea, congenital sodium diarrhea, acrodermatitis enteropathica, selective folate deficiency, abetalipoproteinemia, activating guanylate cyclase mutation | Late onset chloride diarrhea |
| Motility disorders | Hirschsprung disease, chronic intestinal pseudoobstruction (neurogenic and myopathic) | Thyrotoxicosis |
| Neoplastic diseases | Neuroendocrine hormone-secreting tumors: Apudomas such as VIPoma, Zollinger- Ellison, and mastocytosis | Neuroendocrine hormone-secreting tumors: Apudomas such as VIPoma, Zollinger- Ellison, and mastocytosis |
| Diarrhea associated with exogenous substances | Excessive intake of carbonated fluid, foods or drinks containing sorbitol, mannitol, or xylitol; excessive intake of antacids or laxatives containing lactulose or Mg(OH)2; excessive intake of methylxanthines-containing drinks (cola, tea, coffee) | Excessive intake of carbonated fluid, foods or drinks containing sorbitol, mannitol, or xylitol; excessive intake of antacids or laxatives containing lactulose or Mg(OH)2; excessive intake of methylxanthines-containing drinks (cola, tea, coffee) |
| Chronic nonspecific diarrhea | Functional diarrhea* | Irritable bowel syndrome† |
A reduction of intestinal absorptive surface is responsible for diarrhea in celiac disease, a genetically determined permanent gluten intolerance that affects as many as 1 in 100 individuals, depending on geographic origin. In the genetically susceptible host, gliadin, the major protein of gluten, reacts with the immune system to cause villous atrophy. The reduction of functional absorptive surface area is reversible upon restriction of gluten from the diet. Celiac disease presents with more severe intestinal symptoms in younger children. Allergy to cow's milk protein and other food proteins also may present during infancy with chronic diarrhea. Eosinophilic gastroenteritis is characterized by eosinophilic infiltration of the intestinal wall and is strongly associated with atopy. However, whereas diarrhea in food allergy responds to withdrawal of the responsible food, this does not always occur in eosinophilic gastroenteritis, in which immune suppression may be needed.
Lactose intolerance or carbohydrate malabsorption may be caused by a brush-border enzyme defect in lactase, sucrase-isomaltase, or to a defect in the sodium/glucose cotransporter protein (SGLT1) that is transcribed from the SLC5A1 gene causing congenital glucose-galactose malabsorption. The result of these genetic mutations is chronic diarrhea. More commonly, lactose intolerance is secondary to lactase deficiency caused by intestinal mucosal damage. Depending on ethnicity, a progressive, age-related, loss of lactase activity may begin around age 7 yr and affects approximately 80% of the nonwhite population, and acquired hypolactasia may be responsible for chronic diarrhea in older children receiving cow's milk (adult-type lactase deficiency).
In older children and adolescents, inflammatory bowel diseases, including Crohn disease, ulcerative colitis, and inflammatory bowel disease–undetermined, cause chronic diarrhea that is often associated with abdominal pain, elevated inflammatory markers, and increased concentrations of fecal calprotectin or lactoferrin (see Chapter 336). The age of onset of inflammatory bowel disease is broad, with rare cases described in the 1st few mo of life, but the peak incidence in childhood occurring in adolescence. The severity of the symptoms is highly variable with a pattern characterised by long periods of well-being followed by exacerbations. Growth retardation and delays in sexual maturation may precede the onset of gastrointestinal symptoms by up to 18 mo.
Chronic diarrhea may be the manifestation of maldigestion caused by exocrine pancreatic disorders. In most patients with cystic fibrosis, exocrine pancreatic insufficiency results in steatorrhea and protein malabsorption. In Shwachman-Diamond syndrome, exocrine pancreatic hypoplasia may be associated with neutropenia, bone changes, and intestinal protein-losing enteropathy. Specific isolated pancreatic enzyme defects, such as lipase deficiency, result in fat and/or protein malabsorption. Familial pancreatitis, associated with a mutation in the trypsinogen gene, may be associated with exocrine pancreatic insufficiency and chronic diarrhea. Mutations in CFTR, CTRC, PRSS1, SPINK 1, and SPINK 5 are all associated with hereditary pancreatitis.
Liver disorders may lead to a reduction in the bile salts pool resulting in fat malabsorption. Bile acid loss may be associated with diseases affecting the terminal ileum, such as Crohn disease, or following ileal resection. In primary bile acid malabsorption, neonates and young infants present with chronic diarrhea and fat malabsorption caused by mutations of ileal bile transporter.
The most benign etiology of chronic diarrhea is nonspecific diarrhea that encompasses functional diarrhea (or toddler's diarrhea) in children younger than 4 yr of age and irritable bowel syndrome in those 5 yr of age and older. The diseases fall under the umbrella of functional disorders, in that in older children abdominal pain is often associated with diarrhea alternating with constipation and growth and weight gain are normal.
Diarrhea may be the result from an excessive intake of fluid and carbohydrate. If the child's fluid intake were >150 mL/kg/24 hr, fluid intake should be reduced not to exceed 90 mL/kg/24 hr. The child is often irritable in the 1st days of the fluid restriction; however, persistence results in a decrease in the stool frequency and volume. If the dietary history suggests that the child is ingesting significant amounts of fruit juice, especially apple juice, then the consumption of juice should be decreased. Sorbitol, which is a nonabsorbable sugar, is found in apple, pear, and prune juices, and often causes diarrhea in toddlers. Moreover, apple and pear juices contain higher amounts of fructose than glucose, a feature postulated to cause diarrhea in toddlers. In older children, irritable bowel syndrome is often associated with abdominal pain and may be related to anxiety, depression, and other psychologic disturbances.
The most severe etiology of chronic diarrhea includes a number of heterogeneous congenital conditions leading to syndromes related to intractable diarrhea. This is often the result of a permanent defect in the structure or function of the enterocyte, leading to progressive, potentially irreversible intestinal failure. The main etiologies of intractable diarrhea include structural enterocyte defects, disorders of intestinal motility, immune-based disorders, short gut syndrome, and disorders without demonstrable abnormalities.
Structural enterocyte defects are caused by specific molecular defects responsible for early onset, severe diarrhea. In microvillus inclusion disease, microvilli are sequestered in vacuoles as a consequence of autophagocytosis because of a defect in protein trafficking disrupting enterocyte polarity (Fig. 341-2). Intestinal epithelial dysplasia (or tufting enteropathy) is caused by focal crowding of enterocytes that produce epithelial abnormalities resembling tufts (tears). Abnormal deposition of laminin and heparan sulfate proteoglycan on the basement membrane has been detected in intestinal epithelia. An abnormal intestinal distribution of α2β1 and α6β4 integrins is implicated in tufting enteropathy. These ubiquitous proteins are involved in cell–cell and cell–matrix interactions, and play a crucial role in cell development and differentiation.
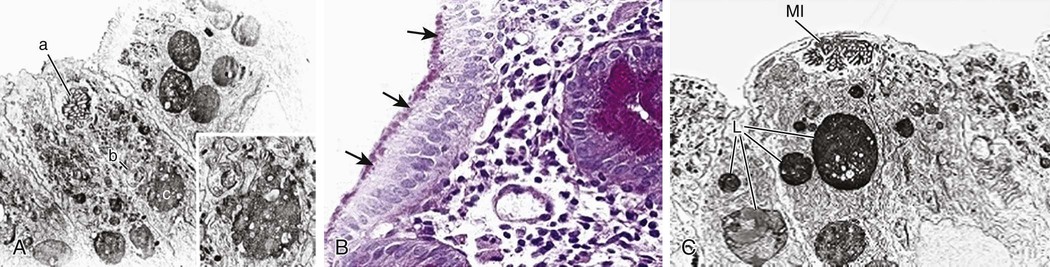
Electrolyte transport defects are a subgroup of structural enterocyte defects that include congenital chloride diarrhea, in which a mutation in the solute carrier family 26 member 3 gene (SLC26A3) leads to severe intestinal Cl− malabsorption from a defect in or absence of the Cl−/HCO3− exchanger. The consequent defect in bicarbonate secretion leads to metabolic alkalosis and acidification of the intestinal content, with further inhibition of Na+/H+ exchanger-dependent Na+ absorption. Patients with congenital sodium diarrhea show similar clinical features, because of a defective Na+/H+ exchanger in the small and large intestine, leading to massive Na+ fecal loss and severe acidosis. Familial diarrhea syndrome caused by a mutation guanylate cyclase-C is characterized by abdominal pain, dysmotility, and inflammation coupled with mild secretory diarrhea.
Disorders of intestinal motility include abnormal development and function of the enteric nervous system, such as in Hirschsprung disease and chronic idiopathic intestinal pseudoobstruction (which encompass both the neurogenic and the myogenic forms). Other motility disorders may be secondary to extraintestinal disorders, such as in hyperthyroidism and scleroderma. Motility disorders are associated with either constipation or diarrhea or both, with the former usually dominating the clinical picture.
Autoimmune processes may target the intestinal epithelium, alone or in association with extraintestinal symptoms. Autoimmune enteropathy is associated with the production of antienterocyte and antigoblet cell antibodies, primarily immunoglobulin A, but also immunoglobulin G, directed against components of the enterocyte brush-border or cytoplasm and by a cell-mediated autoimmune response with mucosal T-cell activation. An X-linked immune-dysregulation, polyendocrinopathy, and enteropathy (IPEX syndrome) is associated with variable gene mutations and phenotypes of chronic diarrhea. Other immunoregulatory defects, found in patients with agammaglobulinemia, isolated immunoglobulin A deficiency, and combined immunodeficiency disorders, may result in persistent infectious diarrhea.
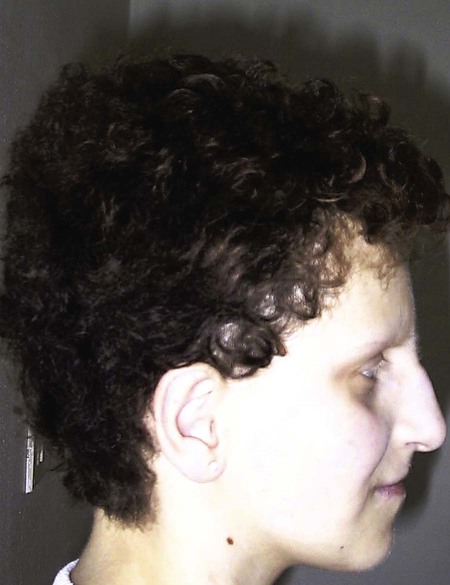
Phenotypic diarrhea, also defined as syndromic diarrhea or trichohepatoenteric syndrome, is a rare disease presenting with facial dysmorphism, woolly hair, severe diarrhea, and malabsorption (Fig. 341-3). Half of the patients have liver disease.
Short bowel syndrome is the single most frequent etiology of chronic diarrhea and intestinal failure (see Chapter 338.7). Many intestinal abnormalities such as stenosis, segmental atresia, and malrotation may require surgical resection, but the most frequent primary cause of short bowel is necrotizing enterocolitis. In these conditions, the residual intestine may be insufficient to carry on its digestive–absorptive functions. Rarely, a child may be born with a congenitally short small bowel resulting in delayed growth. In rare cases of severe chronic diarrhea, the gastrointestinal symptoms may be the initial manifestation of mitochondrial disease, carbohydrate deficient glycoproteins, or a primary immune deficiency. Multiple food protein hypersensitivity also is included in the list of causes of protracted diarrhea syndrome. The disease is believed to be the result of a reaction against specific proteins contained in foods. Diarrhea often resolves with fasting or when an amino-acid–based formula is started. Although most children with food intolerance in infancy are eventually able to resume a regular diet, some require restrictions throughout their lives. When the cause of the diarrhea remains undetermined and the clinical course is inconsistent with organic disorders, factitious disorder by proxy should be considered.
Genetic and Molecular Basis of the Protracted Diarrhea Syndrome
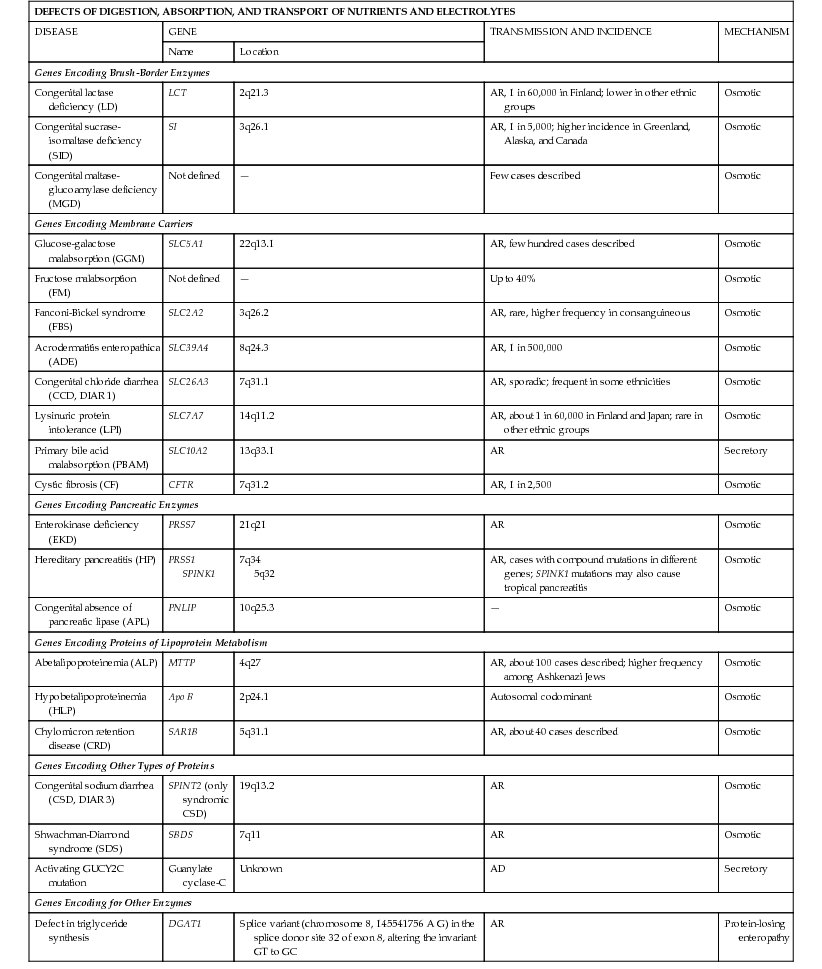
The genetic and molecular basis of many causes of protracted diarrhea have been identified recently and a new classification of congenital diarrheal disorders (CDDs) has been proposed (Table 341-3). CDDs are a group of rare, but severe enteropathies, with a similar clinical presentation despite a different outcome. However, diarrhea is the result of structural and functional abnormalities resulting in either secretory or osmotic diarrhea. Often diarrhea presents at birth or shortly thereafter, but in milder forms diarrhea may go unrecognized for years. CDDs are rare diseases, however in most specific disorders the specific genetic defect and transmission are known. Hereditary fructose intolerance is associated with mutations in the ASDOB gene that encodes for the aldolase B enzyme that is found primarily in the liver and is involved in the metabolism of fructose. Individuals with hereditary fructose intolerance may have nausea, abdominal pain/bloating, vomiting, diarrhea, and hypoglycemia. Continued ingestion of fructose results in hepatomegaly and eventually cirrhosis. The incidence of hereditary fructose intolerance is estimated to be 1 in 20,000-30,000. In contrast, fructose malabsorption is common in Western countries with estimates as high as 40% of the population. These individuals cannot absorb fructose and often develop bloating, abdominal pain, diarrhea, and flatulence. They do not have liver disease.
Table 341-3
Classification of Congenital Diarrheal Disorders Based on Their Molecular Defect and Their Inheritance
| DEFECTS OF DIGESTION, ABSORPTION, AND TRANSPORT OF NUTRIENTS AND ELECTROLYTES | ||||
| DISEASE | GENE | TRANSMISSION AND INCIDENCE | MECHANISM | |
| Name | Location | |||
| Genes Encoding Brush-Border Enzymes | ||||
| Congenital lactase deficiency (LD) | LCT | 2q21.3 | AR, 1 in 60,000 in Finland; lower in other ethnic groups | Osmotic |
| Congenital sucrase-isomaltase deficiency (SID) | SI | 3q26.1 | AR, 1 in 5,000; higher incidence in Greenland, Alaska, and Canada | Osmotic |
| Congenital maltase-glucoamylase deficiency (MGD) | Not defined | — | Few cases described | Osmotic |
| Genes Encoding Membrane Carriers | ||||
| Glucose-galactose malabsorption (GGM) | SLC5A1 | 22q13.1 | AR, few hundred cases described | Osmotic |
| Fructose malabsorption (FM) | Not defined | — | Up to 40% | Osmotic |
| Fanconi-Bickel syndrome (FBS) | SLC2A2 | 3q26.2 | AR, rare, higher frequency in consanguineous | Osmotic |
| Acrodermatitis enteropathica (ADE) | SLC39A4 | 8q24.3 | AR, 1 in 500,000 | Osmotic |
| Congenital chloride diarrhea (CCD, DIAR 1) | SLC26A3 | 7q31.1 | AR, sporadic; frequent in some ethnicities | Osmotic |
| Lysinuric protein intolerance (LPI) | SLC7A7 | 14q11.2 | AR, about 1 in 60,000 in Finland and Japan; rare in other ethnic groups | Osmotic |
| Primary bile acid malabsorption (PBAM) | SLC10A2 | 13q33.1 | AR | Secretory |
| Cystic fibrosis (CF) | CFTR | 7q31.2 | AR, 1 in 2,500 | Osmotic |
| Genes Encoding Pancreatic Enzymes | ||||
| Enterokinase deficiency (EKD) | PRSS7 | 21q21 | AR | Osmotic |
| Hereditary pancreatitis (HP) | PRSS1 SPINK1 | 7q34 5q32 | AR, cases with compound mutations in different genes; SPINK1 mutations may also cause tropical pancreatitis | Osmotic |
| Congenital absence of pancreatic lipase (APL) | PNLIP | 10q25.3 | — | Osmotic |
| Genes Encoding Proteins of Lipoprotein Metabolism | ||||
| Abetalipoproteinemia (ALP) | MTTP | 4q27 | AR, about 100 cases described; higher frequency among Ashkenazi Jews | Osmotic |
| Hypobetalipoproteinemia (HLP) | Apo B | 2p24.1 | Autosomal codominant | Osmotic |
| Chylomicron retention disease (CRD) | SAR1B | 5q31.1 | AR, about 40 cases described | Osmotic |
| Genes Encoding Other Types of Proteins | ||||
| Congenital sodium diarrhea (CSD, DIAR 3) | SPINT2 (only syndromic CSD) | 19q13.2 | AR | Osmotic |
| Shwachman-Diamond syndrome (SDS) | SBDS | 7q11 | AR | Osmotic |
| Activating GUCY2C mutation | Guanylate cyclase-C | Unknown | AD | Secretory |
| Genes Encoding for Other Enzymes | ||||
| Defect in triglyceride synthesis | DGAT1 | Splice variant (chromosome 8, 145541756 A G) in the splice donor site 32 of exon 8, altering the invariant GT to GC | AR | Protein-losing enteropathy |
| DISEASE | OMIM NUMBER | TRANSMISSION AND INCIDENCE | MECHANISM |
| DEFECTS OF ENTEROCYTE DIFFERENTIATION AND POLARIZATION | |||
| Microvillous inclusion disease (MVID, DIAR 2) | 251850 | AR; rare; higher frequency among Navajo | Secretory |
| Congenital tufting enteropathy (CTE, DIAR 5) | 613217 | AR; 1 in 50,000-100,000; higher among Arabians | Secretory |
| Trichohepatoenteric syndrome (THE) | 222470 | AR; 1 in 400,000 | Secretory |
| DEFECTS OF ENTEROENDOCRINE CELL DIFFERENTIATION | |||
| Congenital malabsorptive diarrhea (CMD, DIAR 4) | 610370 | AR; few cases described | Osmotic |
| Proprotein convertase 1/3 deficiency (PCD) | 600955 | AR | Osmotic |
| DEFECTS OF MODULATION OF INTESTINAL IMMUNE RESPONSE | |||
| Autoimmune polyglandular syndrome type 1 (APS1) | 240300 | AR; AD (1 family) | Secretory |
| Immune dysfunction, polyendocrinopathy, X-linked (IPEX) | 601410 | X-linked (autosomal cases described), very rare | Secretory |
| IPEX-like syndrome | — | Not X-linked | Secretory |
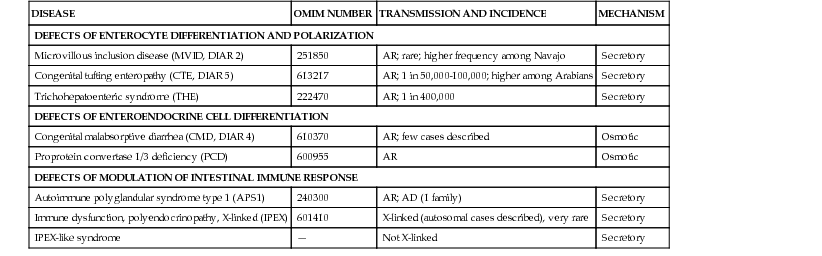
The incidence of other genetic disorders associated with CDD ranges from 1 in 2,500 for cystic fibrosis, 1 in 5,000 for sucrose-isomaltase deficiency, 1 in 60,000 for congenital lactase deficiency, to 1 in 400,000 to trichohepatoenteric syndrome. For most CDDs, such as polyendocrinopathy, X-linked (IPEX) syndrome, or autoimmune polyglandular syndrome type 1, the clinical application of exome sequencing is likely to increase identification of more patients with these rare causes of chronic diarrhea.
Selected CDDs are more frequent in ethnic groups where consanguineous marriages are common, or in some geographic areas because of founder effects. For example, congenital lactase deficiency is more common in Finland; lysinuric protein intolerance has a higher incidence either in Finland and in Japan because of founder effect, and a specific mutation is typically found in each of the 2 ethnic groups. A defect in the DGAT1 gene was identified using whole-exome sequencing in an Ashkenazi Jewish family and associated with the early onset of vomiting and nonbloody diarrhea with protein-losing enteropathy.
Most cases of protracted diarrhea syndrome are not easily treated. The natural history of protracted diarrhea is related to the primary intestinal disease and the specific defect in nutrient absorption. Treatment is more favorable for motility disorders and autoimmune enteropathy than for structural enterocyte defects. Children with motility disorders may have persistent symptoms, but they are rarely fatal; whereas children with structural enterocyte defects have a more severe course, poorer prognosis, and are more likely to be candidates for intestinal transplantation (see Chapter 339). Some late-onset CDDs may be relatively mild and are recognized only later in life.
Evaluation of Patients
Because of the spectrum of etiologies, the medical approach should be based on diagnostic algorithms that begin with assessment for infectious causes, and then consider the age of the child, growth, and clinical and epidemiologic factors. Early onset may suggest a congenital or severe condition. In later infancy and up to 2 yr of age, infections and allergies are more common; inflammatory diseases are more frequent in older children and adolescents. Celiac disease and chronic nonspecific diarrhea should always be considered independently of age because of their relatively high frequency at all ages.
Specific clues in the family and personal history may provide useful indications, suggesting a congenital, allergic or inflammatory etiology. A history of polyhydramnios is consistent with congenital chloride-sodium diarrhea, or cystic fibrosis. An acute onset of diarrhea that runs a protracted course suggests post-enteritis diarrhea or small intestinal overgrowth or the onset of chronic nonspecific diarrhea (toddler's diarrhea). In children, with chronic nonspecific diarrhea there is often a history of an acute gastroenteritis. The association of diarrhea with specific foods may indicate a nutrient basis, such as intolerance to selected nutrients (fructose). Anthropometric evaluation is essential to understand if diarrhea has affected weight gain and growth. The amount of weight loss over time provides an estimate of the severity of diarrhea. Normal weight and growth strongly support functional diarrhea that may respond to simple dietary management.
Initial clinical examination should include the evaluation of general and nutritional status. Dehydration, marasmus, or kwashiorkor require prompt supportive interventions to stabilize the patient. Nutritional evaluation should start with the evaluation of the weight and height curves, and of the weight-for-height index to determine the impact of diarrhea on growth. Weight is generally impaired before height, but with time, linear growth also becomes affected, and both parameters may be equally abnormal in the long-term. Assessment of nutritional status includes a dietary history and biochemical and nutritional investigations. Caloric intake should be quantitatively determined and the relationship between weight modifications and energy intake should be carefully considered.
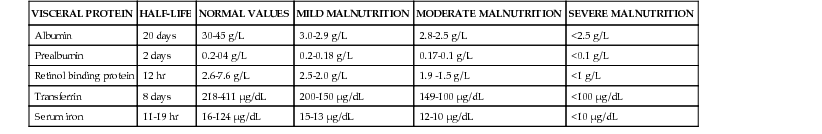
Biochemical markers may assist in grading malnutrition (Table 341-4) as the half-life of serum proteins may distinguish between short- and long-term malnutrition. Assessment of body composition may be performed by measuring mid-arm circumference and triceps skinfold thickness or, more accurately, by bioelectrical impedance analysis or dual-emission x-ray absorptiometry scans. Evaluation of micronutrient concentrations should always be considered. Zinc, magnesium, vitamin A, and folate deficiency are associated with chronic diarrhea and should be provided if needed.
Table 341-4
Degree of Malnutrition as Estimated by Visceral Protein Concentrations in Children with Chronic Diarrhea
| VISCERAL PROTEIN | HALF-LIFE | NORMAL VALUES | MILD MALNUTRITION | MODERATE MALNUTRITION | SEVERE MALNUTRITION |
| Albumin | 20 days | 30-45 g/L | 3.0-2.9 g/L | 2.8-2.5 g/L | <2.5 g/L |
| Prealbumin | 2 days | 0.2-04 g/L | 0.2-0.18 g/L | 0.17-0.1 g/L | <0.1 g/L |
| Retinol binding protein | 12 hr | 2.6-7.6 g/L | 2.5-2.0 g/L | 1.9 -1.5 g/L | <1 g/L |
| Transferrin | 8 days | 218-411 µg/dL | 200-150 µg/dL | 149-100 µg/dL | <100 µg/dL |
| Serum iron | 11-19 hr | 16-124 µg/dL | 15-13 µg/dL | 12-10 µg/dL | <10 µg/dL |
Diagnosis of functional diarrhea is based on clinical assessment using established age-related criteria. It should be noted that a child with functional diarrhea may be inappropriately “treated” with a diluted hypocaloric diet in an effort to reduce the diarrhea, resulting in impaired growth.
The search for an etiology may be based on the relevant causes of diarrhea for the age of the child. Continued diarrhea with fasting or fecal electrolyte concentrations discriminate between secretory and osmotic diarrhea. Associated symptoms and selected investigations provide important diagnostic clues. Signs of general inflammation such as fever, mucoid or bloody stools, and abdominal pain may suggest inflammatory bowel disease. The presence of eczema or asthma is associated with an allergic disorder, whereas specific extraintestinal manifestations (arthritis, diabetes, thrombocytopenia, etc.) may suggest an autoimmune disease. Specific skin lesions may be suggestive of acrodermatitis enteropathica that might respond to zinc replacement. Typical facial abnormalities and woolly hair are associated with phenotypic diarrhea (see Fig. 341-3).
Investigations
Microbiologic investigation should include a thorough list of intestinal bacterial, viral, and protozoan pathogens. Proximal intestinal bacterial overgrowth may be determined using the hydrogen breath test, after an oral glucose or lactulose load, but either substrate may give false results.
Initial investigations of a child with chronic diarrhea should always include an assessment of intestinal inflammation using fecal calprotectin or lactoferrin, and serology for celiac disease (see Chapter 338.2). The role of a mucosal biopsy is determined by the noninvasive diagnostic evaluation in consultation with a pediatric gastroenterologist.
Noninvasive assessment of digestive-absorptive function and of intestinal inflammation plays a key role in the diagnostic work-up (Table 341-5). Abnormalities in the digestive–absorptive function tests suggest small bowel involvement, whereas intestinal inflammation, as demonstrated by increased fecal calprotectin or lactoferrin, supports colitis. Histology is important in establishing mucosal involvement, noting changes in the epithelial cells, or in identifying specific intracellular inclusion bodies caused by pathogens, such as cytomegalovirus, or the presence of parasites. Electron microscopy is essential to detect subcellular structural abnormalities such as microvillous inclusion disease. Immunohistochemistry allows the study of mucosal immunity as well as of other cell types (smooth muscle cells and enteric neuronal cells).
Table 341-5
Noninvasive Tests for Intestinal Digestive–Absorptive Function and Inflammation
| TEST | NORMAL VALUES | IMPLICATION |
| α1-Antitripsin concentration | <0.9 mg/g | Increased intestinal permeability/protein loss |
| Steatocrit | <2.5% (older than 2 yr) fold increase over age-related values (younger than 2 yr) | Fat malabsorption |
| Fecal-reducing substances | Absent | Carbohydrate malabsorption |
| Elastase concentration | >200 µg/g | Pancreatic function |
| Chymotrypsin concentration | >7.5 units/g >375 units/24 hr | Pancreatic function |
| Fecal occult blood | Absent | Blood loss in the stools/inflammation |
| Fecal calprotectin concentration | <100 µg/g (in children to 4 yr of age) <50 µg/g (older than 4 yr) | Intestinal inflammation |
| Fecal leukocytes | <5/microscopic field | Colonic inflammation |
| Nitric oxide in rectal dialysate | <5 µM of NO2−/NO3− | Rectal inflammation |
| Dual sugar (cellobiose/mannitol) absorption test | Urine excretion ratio: 0.010 ± 0.018 | Increased intestinal permeability |
| Xylose oral load | 25 mg/dL | Reduced intestinal surface |
Imaging has a major role in the diagnostic approach. Abdominal ultrasound may help in detecting liver and pancreatic abnormalities or an increase in distal ileal wall thickness that suggests inflammatory bowel disease. A preliminary plain abdominal x-ray is useful for detection of abdominal distention, suggestive of intestinal obstruction, or increased retention of colonic feces. Intramural or portal gas may be seen in necrotizing enterocolitis or intussusception. Structural abnormalities such as diverticula, malrotation, stenosis, blind loop, inflammatory bowel disease, as well as motility disorders, may be investigated through a barium meal and a small bowel follow-through. Capsule endoscopy allows the exploration of the entire intestinal tract searching for structural changes, inflammation or bleeding and the new SmartPill measures pressure, pH, and temperature as it moves through the gastrointestinal tract, assessing motility.
Specific investigations should be carried out for specific diagnostic indications. Prick and patch test may support a diagnosis of food allergy. However, elimination diet with withdrawal of the suspected harmful food from the diet and subsequent challenge is the most reliable strategy by which to establish a diagnosis. Bile malabsorption may be explored by the retention of the bile acid analog 75Se-homocholic acid-taurine (75SeHCAT) in the enterohepatic circulation. A scintigraphic examination, with radio-labeled octreotide is indicated in suspected APUD cell neoplastic proliferation. In other diseases, specific imaging techniques such as computed tomography, or nuclear magnetic resonance endoscopic retrograde cholangiopancreatography and magnetic resonance cholangiopancreatography may have important diagnostic value.
Once infectious agents have been excluded and nutritional assessment performed, a stepwise approach to the child with chronic diarrhea may be applied. The main causes of chronic diarrhea should be investigated, based on the features of the diarrhea and the specific nutrient(s) that is (are) affected. The use of whole-exome sequencing is of benefit in children suspected of having mendelian-related causes of chronic diarrhea. A step-by-step diagnostic approach is important to minimize the unnecessary use of invasive procedures as well as the cost, while optimizing the yield of the diagnostic work-up (Table 341-6).
Table 341-6
Stepwise Diagnostic Approach to Children with Diarrhea
| STEP 1 |
| Intestinal Microbiology |
| Screening Test for Celiac Disease: |
| Serology according to age and level of IgA (including AGA IgA/IgG, EMA IgA/IgG, tTG IgA/IgG) |
| Noninvasive Tests for: |
| Tests for Food Allergy: |
| Prick/patch tests for foods |
| Abdominal Ultrasounds (Scan of Last Ileal Loop) |
| STEP 2 |
| Evaluation of Intestinal Morphology: |
Endoscopy and standard jejunal/colonic histology* |
| STEP 3 |
| Special Investigations: |

Treatment
Chronic diarrhea associated with impaired nutritional status should always be considered a serious disease, and therapy should be started promptly. Treatment includes general supportive measures, nutritional rehabilitation, elimination diet, and medications. The latter include therapies for specific etiologies as well as interventions aimed at counteracting fluid secretion and/or promoting restoration of disrupted intestinal epithelium. Because death in most instances is caused by dehydration, replacement of fluid and electrolyte losses is the most important early intervention.
Nutritional rehabilitation is often essential and is based on clinical and biochemical assessment. Potentially harmful nutrients must be identified and avoided. In moderate to severe malnutrition, caloric intake may be progressively increased to 50% or more above the recommended dietary allowances. The intestinal absorptive capacity should be monitored by digestive function tests. In children with steatorrhea, medium-chain triglycerides may be the main source of lipids. A lactose-free diet should be started in all children with chronic diarrhea and is recommended by the World Health Organization. Lactose is generally replaced by maltodextrin or a combination of complex carbohydrates. A sucrose-free formula is indicated in sucrase-isomaltase deficiency. Semielemental or elemental diets have the dual purpose of overcoming food intolerance, which may be the primary cause of chronic diarrhea, particularly in infancy and early childhood, and facilitating nutrient absorption. The sequence of elimination should begin from less to more restricted diets, that is, cow's milk protein hydrolysate to amino-acid–based formulas, depending on the child's situation. In severely compromised infants, it may be prudent to start with amino-acid–based feeding.
When oral nutrition is not feasible or fails, enteral or parenteral nutrition should be considered. Enteral nutrition may be performed via nasogastric or gastrostomy tube and is indicated in a child who is not able to be fed orally, either because of inability to tolerate nutrient requirements or because of extreme weakness. Continuous enteral nutrition is effective in children with a compromised absorptive capacity, such as short bowel syndrome where the remaining mucosal surface is intact. In extreme wasting, enteral nutrition may not be tolerated and parenteral nutrition is required.
Micronutrient and vitamin supplementation are part of nutritional rehabilitation, especially in malnourished children in developing countries. Zinc supplementation is important in both prevention and therapy of chronic diarrhea, since it promotes ion absorption, restores epithelial proliferation, and stimulates immune response. Nutritional rehabilitation has a general beneficial effect on the patient's general condition, intestinal function, and immune response.
Functional diarrhea in children may benefit from a diet based on the “4 F” principles (reduce fructose and fluids, increase fat and fiber). Probiotics have been used with some success as adjunctive therapy based on the evidence that changes in intestinal microflora might be beneficial in several other intestinal diseases.
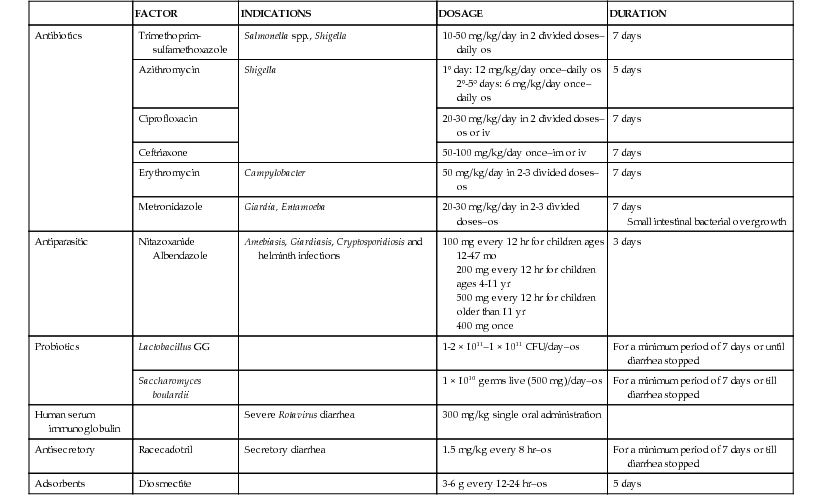
Pharmacologic therapy includes antiinfectious drugs, immune suppression, and drugs that may inhibit fluid loss and promote cell growth. If a bacterial agent is detected, specific antibiotics should be prescribed. Empiric antibiotic therapy may be used in children with either small bowel bacterial overgrowth or with suspected infectious diarrhea. Table 341-7 summarizes the treatment of postinfectious persistent diarrhea. Immune suppression should be considered in selected conditions such as autoimmune enteropathy.
Table 341-7
Treatment of Infectious Persistent Diarrhea
| FACTOR | INDICATIONS | DOSAGE | DURATION | |
| Antibiotics | Trimethoprim-sulfamethoxazole | Salmonella spp., Shigella | 10-50 mg/kg/day in 2 divided doses–daily os | 7 days |
| Azithromycin | Shigella | 1° day: 12 mg/kg/day once–daily os 2°-5° days: 6 mg/kg/day once–daily os | 5 days | |
| Ciprofloxacin | 20-30 mg/kg/day in 2 divided doses–os or iv | 7 days | ||
| Ceftriaxone | 50-100 mg/kg/day once–im or iv | 7 days | ||
| Erythromycin | Campylobacter | 50 mg/kg/day in 2-3 divided doses–os | 7 days | |
| Metronidazole | Giardia, Entamoeba | 20-30 mg/kg/day in 2-3 divided doses–os | 7 days Small intestinal bacterial overgrowth | |
| Antiparasitic | Nitazoxanide Albendazole | Amebiasis, Giardiasis, Cryptosporidiosis and helminth infections | 100 mg every 12 hr for children ages 12-47 mo 200 mg every 12 hr for children ages 4-11 yr 500 mg every 12 hr for children older than 11 yr 400 mg once | 3 days |
| Probiotics | Lactobacillus GG | 1-2 × 1011–1 × 1011 CFU/day–os | For a minimum period of 7 days or until diarrhea stopped | |
| Saccharomyces boulardii | 1 × 1010 germs live (500 mg)/day–os | For a minimum period of 7 days or till diarrhea stopped | ||
| Human serum immunoglobulin | Severe Rotavirus diarrhea | 300 mg/kg single oral administration | ||
| Antisecretory | Racecadotril | Secretory diarrhea | 1.5 mg/kg every 8 hr–os | For a minimum period of 7 days or till diarrhea stopped |
| Adsorbents | Diosmectite | 3-6 g every 12-24 hr–os | 5 days |
Treatment may be also directed at modifying specific pathophysiologic processes. Secretion of ions may be reduced by proabsorptive agents, such as the enkephalinase inhibitor racecadotril. In diarrhea caused by neuroendocrine tumors, microvillus inclusion disease and enterotoxin-induced severe diarrhea, a trial of somatostatin analog octreotide may be considered. Zinc promotes both enterocyte growth and ion absorption and may be effective when intestinal atrophy and ion secretion are associated. However, when therapeutic attempts have failed, the only option to avoid intestinal failure may be parenteral nutrition or eventually intestinal transplantation.
Diarrhea from Neuroendocrine Tumors
Helen Spoudeas, David Branski
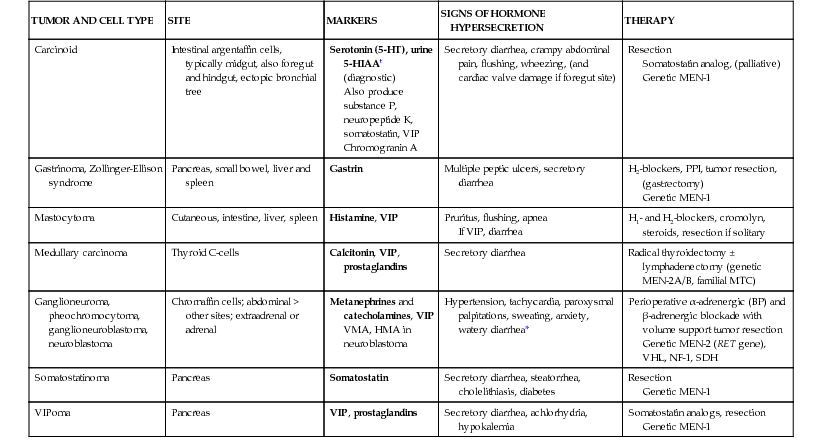
Rare tumors of the neuroendocrine cells of the gastroenteropancreatic axis and adrenal and extraadrenal sites derive from the APUD system. They are characterized by an excessive production of 1 or several peptides, which, when released into the circulation, exert their endocrine effects and can be measured by radioimmunologic methods (in the plasma or as their urinary metabolites) and hence act as tumor markers. In clinically functioning tumors, the hypersecretion causes a recognizable syndrome that can include watery diarrhea. Though rare, neuroendocrine tumor (NET) should be considered a potential cause in patients with a particularly severe or chronic course (resulting in electrolyte and fluid depletion), associated flushing, palpitations, or bronchospasm, or a positive family history of multiple endocrine neoplasia 1 or 2 syndromes (Table 341-8).
Table 341-8
Diarrhea Caused by Neuroendocrine Tumors
| TUMOR AND CELL TYPE | SITE | MARKERS | SIGNS OF HORMONE HYPERSECRETION | THERAPY |
| Carcinoid | Intestinal argentaffin cells, typically midgut, also foregut and hindgut, ectopic bronchial tree | Serotonin (5-HT), urine 5-HIAA† (diagnostic) Also produce substance P, neuropeptide K, somatostatin, VIP Chromogranin A | Secretory diarrhea, crampy abdominal pain, flushing, wheezing, (and cardiac valve damage if foregut site) | Resection Somatostatin analog, (palliative) Genetic MEN-1 |
| Gastrinoma, Zollinger-Ellison syndrome | Pancreas, small bowel, liver and spleen | Gastrin | Multiple peptic ulcers, secretory diarrhea | H2-blockers, PPI, tumor resection, (gastrectomy) Genetic MEN-1 |
| Mastocytoma | Cutaneous, intestine, liver, spleen | Histamine, VIP | Pruritus, flushing, apnea If VIP, diarrhea | H1- and H2-blockers, cromolyn, steroids, resection if solitary |
| Medullary carcinoma | Thyroid C-cells | Calcitonin, VIP, prostaglandins | Secretory diarrhea | Radical thyroidectomy ± lymphadenectomy (genetic MEN-2A/B, familial MTC) |
| Ganglioneuroma, pheochromocytoma, ganglioneuroblastoma, neuroblastoma | Chromaffin cells; abdominal > other sites; extraadrenal or adrenal | Metanephrines and catecholamines, VIP VMA, HMA in neuroblastoma | Hypertension, tachycardia, paroxysmal palpitations, sweating, anxiety, watery diarrhea* | Perioperative α-adrenergic (BP) and β-adrenergic blockade with volume support tumor resection Genetic MEN-2 (RET gene), VHL, NF-1, SDH |
| Somatostatinoma | Pancreas | Somatostatin | Secretory diarrhea, steatorrhea, cholelithiasis, diabetes | Resection Genetic MEN-1 |
| VIPoma | Pancreas | VIP, prostaglandins | Secretory diarrhea, achlorhydria, hypokalemia | Somatostatin analogs, resection Genetic MEN-1 |
Depending on the tumor type, the peptide marker(s) in the plasma and/or the 24-hr urinary metabolite(s) measured (on 2 occasions), form the basis of the biochemical diagnosis, the prognosis (tumor load) and treatment monitoring. Baseline tests should include plasma chromogranin A and urinary 5-hydroxyindoloacetic acid, other specific biochemistry being guided by the suspected syndrome (see Table 341-8). Carcinoid tumors are gastroenteropancreatic NETs, typically of the midgut (rather than fore- or hindgut), which may cause flushing and bronchospasm in addition to diarrhea and which, because of their portal drainage, are the most prone to late presentation and malignancy. Localization of any NET is best achieved with a multimodality approach at a center of excellence. Thus whole-body CT, MRI, and somatostatin receptor scintigraphy may be required (because nearly all NETs express membrane receptors for small peptides, e.g., somatostatin), with gallium-68 positron emission tomography/CT recommended for detecting an unknown primary. Long-acting somatostatin analogs might also have a role in palliation.
Tumor resection is the treatment of choice but is potentially hazardous and can precipitate life-threatening adrenergic crises; it should only be undertaken by an endocrine surgeon with experience under carefully controlled medical and anesthetic conditions and in conjunction with an endocrinologist. Tumor histochemistry will confirm the NET type and classification of NETs should be based on the World Health Organization 2010 Union for International Cancer Control TNM criteria (7th edition). This diagnosis in a child should prompt a genetic referral to exclude a tumor predisposition syndrome in which the child is the index case and tumor registration. Management and follow-up is multidisciplinary and should be undertaken in an age-appropriate setting with access to adult specialists with expertise in these rare conditions.