Infective Endocarditis
Daniel Bernstein
Infective endocarditis includes acute and subacute bacterial endocarditis, as well as nonbacterial endocarditis caused by viruses, fungi, and other microbiologic agents. It is a significant cause of morbidity and mortality in children and adolescents despite advances in the management and prophylaxis of the disease with antimicrobial agents. The inability to eradicate infective endocarditis by prevention or early treatment stems from several factors. The disease represents a complex interplay between a pathogen and host factors such as endothelial disruption and immune function that is still not completely understood; the nature of the infecting organism has changed over time; diagnosis may be difficult during early stages and is thus often delayed until a more serious infection has set in; and special risk groups have emerged, including intravenous drug users; survivors of cardiac surgery, especially those with mechanical prosthesis; patients taking immunosuppressant medications; and patients who require chronic intravascular catheters. Some patients get endocarditis on what was thought to be a previously healthy native valve, although on surgical inspection is found to have mild structural abnormalities.
Etiology
Viridans-type streptococci (α-hemolytic streptococci) and Staphylococcus aureus remain the leading causative agents for endocarditis in pediatric patients. Other organisms cause endocarditis less frequently and, in ≈6% of cases, blood cultures are negative for any organisms (Table 437-1). No relationship exists between the infecting organism and the type of congenital defect, the duration of illness, or the age of the child. Staphylococcal endocarditis is more common in patients with no underlying heart disease; viridans group streptococcal infection is more common after dental procedures; group D enterococci are seen more often after lower bowel or genitourinary manipulation; Pseudomonas aeruginosa or Serratia marcescens is seen more frequently in intravenous drug users; and fungal organisms are encountered after open heart surgery. Coagulase-negative staphylococci are common in the presence of an indwelling central venous catheter.
Table 437-1
Bacterial Agents in Pediatric Infective Endocarditis
| COMMON: NATIVE VALVE OR OTHER CARDIAC LESIONS |
| UNCOMMON: NATIVE VALVE OR OTHER CARDIAC LESIONS |
| PROSTHETIC VALVE |

Epidemiology
Infective endocarditis is often a complication of congenital or rheumatic heart disease but can also occur in children without any abnormal valves or cardiac malformations. In developed countries, congenital heart disease is the overwhelming predisposing factor. Endocarditis is rare in infancy; in this age group, it usually follows open heart surgery or is associated with a central venous line.
Patients with congenital heart lesions where there is turbulent blood flow because of a hole or stenotic orifice, especially if there is a high-pressure gradient across the defect, are most susceptible to endocarditis. This turbulent flow traumatizes the vascular endothelium, creating a substrate for deposition of fibrin and platelets, leading to the formation of a nonbacterial thrombotic embolus (NBTE) that is thought to be the initiating lesion for infective endocarditis. Biofilms form on the surface of implanted mechanical devices such as valves, catheters, or pacemaker wires that also serve as the adhesive substrate for infection. The development of transient bacteremia then colonizes this NBTE or biofilm, leading to proliferation of bacteria within the lesion. Bacterial surface proteins, such as the FimA antigen in viridans streptococci, act as adhesion factors to the NBTE or biofilm, after which bacteria can rapidly proliferate within the vegetation. Given the heavy colonization of mucosal surfaces (the oropharynx, or gastrointestinal, vaginal, or urinary tracts) by potentially pathogenic bacteria, these surfaces are thought to be the origin of this transient bacteremia. There is controversy over the extent to which daily activities (such as brushing or flossing the teeth) versus invasive procedures (such as dental cleanings or surgery) contribute to this bacteremia. Transient bacteremia is reported to occur in 20-68% of patients after tooth brushing and flossing, and even in 7-51% of patients after chewing food. The magnitude of this bacteremia is also similar to that resulting from dental procedures. Maintenance of good oral hygiene may be a more important factor in decreasing the frequency and magnitude of bacteremia.
Children at highest risk of adverse outcome after infective endocarditis include those with prosthetic cardiac valves or other prosthetic material used for cardiac valve repair, unrepaired cyanotic congenital heart disease (including those palliated with shunts and conduits), completely repaired defects with prosthetic material or device during the 1st 6 mo after repair, repaired congenital heart disease with residual defects at or adjacent to the site of a prosthetic patch or device, valve stenosis or insufficiency occurring after heart transplantation, permanent valve disease from rheumatic fever (mitral stenosis, aortic regurgitation), and previous infective endocarditis. Patients with high velocity blood flow lesions such as ventricular septal defects and aortic stenosis are also at high risk. In older patients, congenital bicuspid aortic valves and mitral valve prolapse with regurgitation pose additional risks for endocarditis. Surgical correction of congenital heart disease may reduce but does not eliminate the risk of endocarditis, with the exception of repair of a simple atrial septal defect or patent ductus arteriosus without prosthetic material.
In ≈30% of patients with infective endocarditis, a predisposing factor is presumably recognized. Although a preceding dental procedure may be identified in 10-20% of patients, the time of the procedure may range from 1 to 6 mo prior to the onset of symptoms, hence the continued controversy over the absolute risk of infective endocarditis after dental procedures. Primary bacteremia with S. aureus is thought to be another risk for endocarditis. The occurrence of endocarditis directly after most routine heart surgery is relatively low, but it can be an antecedent event, especially if prosthetic material is utilized.
Clinical Manifestations
Table 437-2 outlines the manifestations of infective endocarditis.
Table 437-2
Manifestations of Infective Endocarditis
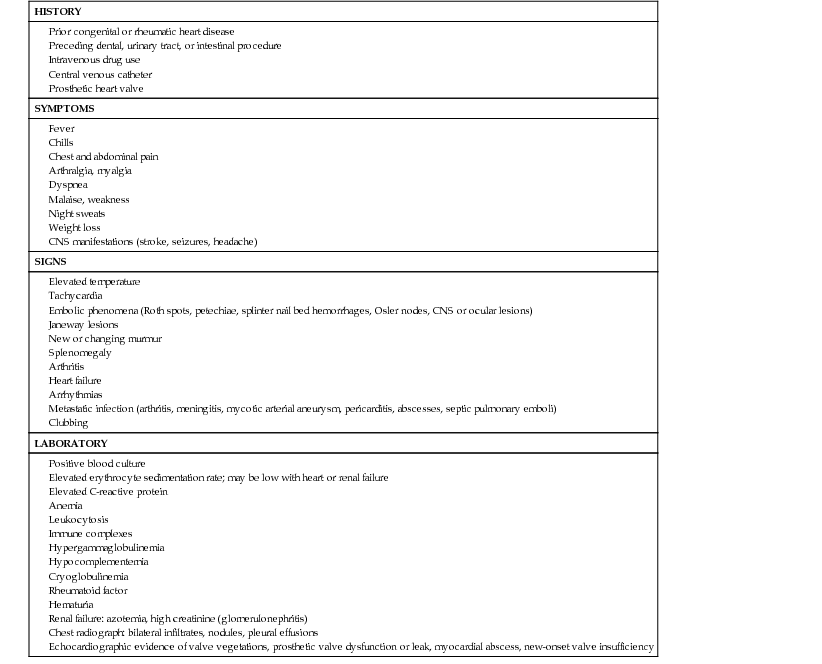
Early manifestations are usually mild, especially when viridans group streptococci are the infecting organisms. Prolonged fever without other manifestations (except, occasionally, weight loss) that persists for as long as several months may be the only symptom. Alternatively, the onset may be acute and severe, with high, intermittent fever and prostration. Usually, the onset and course vary between these 2 extremes. The symptoms are often nonspecific and consist of low-grade fever with afternoon elevations, fatigue, myalgia, arthralgia, headache, and, at times, chills, nausea, and vomiting. New or changing heart murmurs are common, particularly with associated heart failure. Splenomegaly and petechiae are relatively common. Serious neurologic complications such as embolic strokes, cerebral abscesses, mycotic aneurysms, and hemorrhage are most often associated with staphylococcal disease and may be late manifestations. Meningismus, increased intracranial pressure, altered sensorium, and focal neurologic signs are manifestations of these complications. Myocardial abscesses may occur with staphylococcal disease and may damage the cardiac conducting system, causing heart block, or may rupture into the pericardium and produce purulent pericarditis. Pulmonary and other systemic emboli are infrequent, except with fungal disease. Many of the classic skin findings develop late in the course of the disease; they are seldom seen in appropriately treated patients. Such manifestations include Osler nodes (tender, pea-size intradermal nodules in the pads of the fingers and toes), Janeway lesions (painless small erythematous or hemorrhagic lesions on the palms and soles), and splinter hemorrhages (linear lesions beneath the nails). These lesions may represent vasculitis produced by circulating antigen–antibody complexes.
Identification of infective endocarditis is most often based on a high index of suspicion during evaluation of an infection in a child with an underlying risk factor.
Diagnosis
The critical information for appropriate treatment of infective endocarditis is obtained from blood cultures. All other laboratory data are secondary in importance (see Table 437-2). Blood specimens for culture should be obtained as promptly as possible, even if the child feels well and has no other physical findings. Three to 5 separate blood collections should be obtained after careful preparation of the phlebotomy site. Contamination presents a special problem inasmuch as bacteria found on the skin may themselves cause infective endocarditis. The timing of collections is not important because bacteremia can be expected to be relatively constant. In 90% of cases of endocarditis, the causative agent is recovered from the first 2 blood cultures. Bacteremia is low grade in 80% (<100 colony-forming units [CFU]/mL or blood). The laboratory should be notified that endocarditis is suspected so that, if necessary, the blood can be cultured on enriched media for longer than usual (>7 days) to detect nutritionally deficient and fastidious bacteria or fungi. Although bacteremia may occur in the absence of endocarditis, bacteremia secondary to Streptococcus mutans, Streptococcus bovis I, Streptococcus mitior, Streptococcus sanguis, and S. aureus (in the absence of focal musculoskeletal infection) is highly concerning for endocarditis. Antimicrobial pretreatment of the patient reduces the yield of blood cultures to 50-60%. The microbiology laboratory should be notified if the patient has received antibiotics so that more sophisticated methods can be used to recover the offending agent. Other specimens that may be cultured include scrapings from cutaneous lesions, urine, synovial fluid, abscesses, and, in the presence of manifestations of meningitis, cerebrospinal fluid. Serologic diagnosis or polymerase chain reaction of resected valve tissues is necessary in patients with unusual or fastidious microorganisms (Table 437-3; Fig. 437-1).
Table 437-3
Diagnostic Approach to Uncommon Pathogens Causing Endocarditis
| PATHOGEN | DIAGNOSTIC PROCEDURE |
| Brucella spp. | Blood cultures; serology; culture, immunohistology, and PCR of surgical material |
| Coxiella burnetii | Serology (IgG phase I >1 in 800); tissue culture, immunohistology, and PCR of surgical material |
| Bartonella spp. | Blood cultures; serology; culture, immunohistology, and PCR of surgical material |
| Chlamydia spp. | Serology; culture, immunohistology, and PCR of surgical material |
| Mycoplasma spp. | Serology; culture, immunohistology, and PCR of surgical material |
| Legionella spp. | Blood cultures; serology; culture, immunohistology, and PCR of surgical material |
| Tropheryma whipplei | Histology and PCR of surgical material |
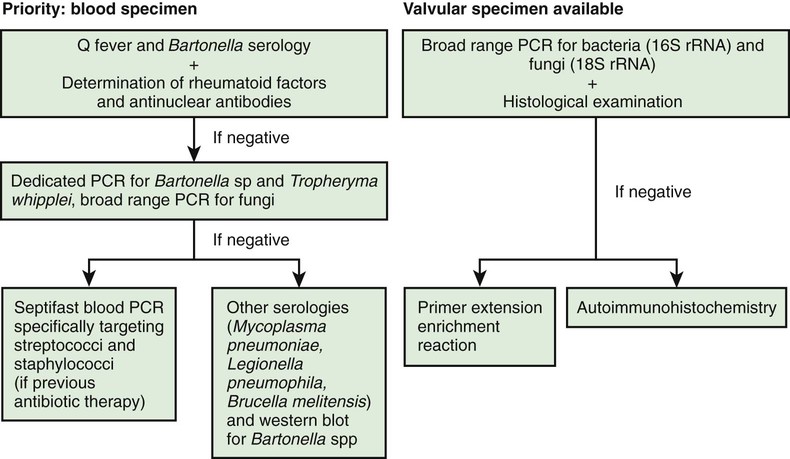
The index of suspicion should be high when evaluating infection in a child with an underlying contributing factor. The combination of transthoracic and transesophageal echocardiography enhances the ability to diagnose endocarditis. Two-dimensional echocardiography can identify the size, shape, location, and mobility of the lesion; when combined with Doppler studies, the presence of valve dysfunction (regurgitation, obstruction) can be determined and its effect on left ventricular performance quantified. Echocardiography may also be helpful in predicting embolic complications, given that lesions >1 cm and fungating masses are at greatest risk for embolization. The absence of vegetations does not exclude endocarditis, and vegetations are often not visualized in the early phases of the disease or in patients with complex congenital heart lesions.
The Duke criteria help in the diagnosis of endocarditis. Major criteria include (1) positive blood cultures (2 separate cultures for a usual pathogen, 2 or more for less-typical pathogens), and (2) evidence of endocarditis on echocardiography (intracardiac mass on a valve or other site, regurgitant flow near a prosthesis, abscess, partial dehiscence of prosthetic valves, or new valve regurgitant flow). Minor criteria include predisposing conditions, fever, embolic-vascular signs, immune complex phenomena (glomerulonephritis, arthritis, rheumatoid factor, Osler nodes, Roth spots), a single, positive blood culture or serologic evidence of infection, and echocardiographic signs not meeting the major criteria. Two major criteria, 1 major and 3 minor, or 5 minor criteria suggest definite endocarditis. A modification of the Duke criteria may increase sensitivity while maintaining specificity. The following minor criteria are added to those already listed: the presence of newly diagnosed clubbing, splenomegaly, splinter hemorrhages, and petechiae; a high erythrocyte sedimentation rate; a high C-reactive protein level; and the presence of central nonfeeding lines, peripheral lines, and microscopic hematuria.
Prognosis and Complications
Despite the use of antibiotic agents, mortality remains high, in the range of 20-25%. Serious morbidity occurs in 50-60% of children with documented infective endocarditis; the most common is heart failure caused by vegetations involving the aortic or mitral valve. Myocardial abscesses and toxic myocarditis may also lead to heart failure without characteristic changes in auscultatory findings and, occasionally, to life-threatening arrhythmias. Systemic emboli, often with central nervous system manifestations, are a major threat. Pulmonary emboli may occur in children with ventricular septal defect or the tetralogy of Fallot, although massive life-threatening pulmonary embolization is rare. Other complications include mycotic aneurysms, rupture of a sinus of Valsalva, obstruction of a valve secondary to large vegetations, acquired ventricular septal defect, and heart block as a result of involvement (abscess) of the conduction system. Additional complications include meningitis, osteomyelitis, arthritis, renal abscess, purulent pericarditis, and immune complex-mediated glomerulonephritis.
Treatment
Antibiotic therapy should be instituted immediately once a definitive diagnosis is made. When virulent organisms are responsible, small delays may result in progressive endocardial damage and are associated with a greater likelihood of severe complications. The choice of antibiotics, method of administration, and length of treatment should be coordinated with consultants from both cardiology and infectious diseases (Tables 437-4 and 437-5). Empirical therapy before the identifiable agent is recovered may be initiated with vancomycin plus gentamicin in patients without a prosthetic valve and when there is a high risk of S. aureus, enterococcus, or viridans streptococci (the 3 most common organisms). High serum bactericidal levels must be maintained long enough to eradicate organisms that are growing in relatively inaccessible avascular vegetations. Between 5 and 20 times the minimal in vitro inhibiting concentration must be produced at the site of infection to destroy bacteria growing at the core of these lesions. Several weeks are required for a vegetation to organize completely; therapy must be continued through this period so that recrudescence can be avoided. A total of 4-6 wk of treatment is usually recommended. Depending on the clinical and laboratory responses, antibiotic therapy may require modification and, in some instances, more prolonged treatment is required. With highly sensitive viridans group streptococcal infections, shortened regimens that include oral penicillin for some portion have been recommended. In nonstaphylococcal disease, bacteremia usually resolves in 24-48 hr, whereas fever resolves in 5-6 days with appropriate antibiotic therapy. Resolution with staphylococcal disease takes longer.
Table 437-4
Therapy of Native Valve Endocarditis Caused by Highly Penicillin-Susceptible Viridans Group Streptococci and Streptococcus bovis
| REGIMEN | DOSAGE* AND ROUTE | DURATION, WK | COMMENTS |
| Aqueous crystalline penicillin G sodium | 12-18 million U/24 hr IV either continuously or in 4 or 6 equally divided doses | 4 | Preferred in patients with impairment of 8th cranial nerve function or renal function |
| or | |||
| Ceftriaxone sodium | 2 g/24 hr IV/IM in 1 dose | 4 | |
| Pediatric dose†: penicillin 200,000 U/kg per 24 hr IV in 4-6 equally divided doses; ceftriaxone 100 mg/kg per 24 hr IV/IM in 1 dose | |||
| Aqueous crystalline penicillin G sodium | 12-18 million U/24 hr IV either continuously or in 6 equally divided doses | 2 | 2 wk regimen not intended for patients with known cardiac or extracardiac abscess or for those with creatinine clearance of <20 mL/min, impaired 8th cranial nerve function, or Abiotrophia, Granulicatella, or Gemella spp. infection; gentamicin dosage should be adjusted to achieve peak serum concentration of 3-4 µg/mL and trough serum concentration of <1 µg/mL when 3 divided doses are used; nomogram used for single daily dosing |
| or | |||
| Ceftriaxone sodium | 2 g/24 hr IV/IM in 1 dose | 2 | |
| plus | |||
| Gentamicin sulfate‡ | 3 mg/kg per 24 hr IV/IM in 1 dose, or 3 equally divided doses Pediatric dose: penicillin 200,000 U/kg per 24 hr IV in 4-6 equally divided doses; ceftriaxone 100 mg/kg per 24 hr IV/IM in 1 dose; gentamicin 3 mg/kg per 24 hr IV/IM in 1 dose or 3 equally divided doses§ | 2 | |
| Vancomycin hydrochloride¶ | 30 mg/kg per 24 hr IV in 2 equally divided doses not to exceed 2 g/24 hr unless concentrations in serum are inappropriately low Pediatric dose: 40 mg/kg per 24 hr IV in 2-3 equally divided doses | 4 | Vancomycin therapy recommended only for patients unable to tolerate penicillin or ceftriaxone; vancomycin dosage should be adjusted to obtain peak (1 hr after infusion completed) serum concentration of 30-45 µg/mL and a trough concentration range of 10-15 µg/mL |
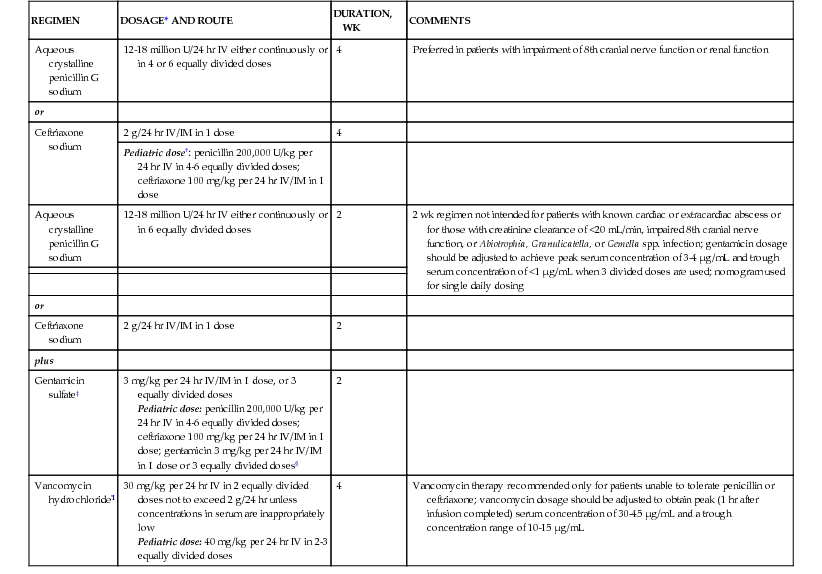
Table 437-5
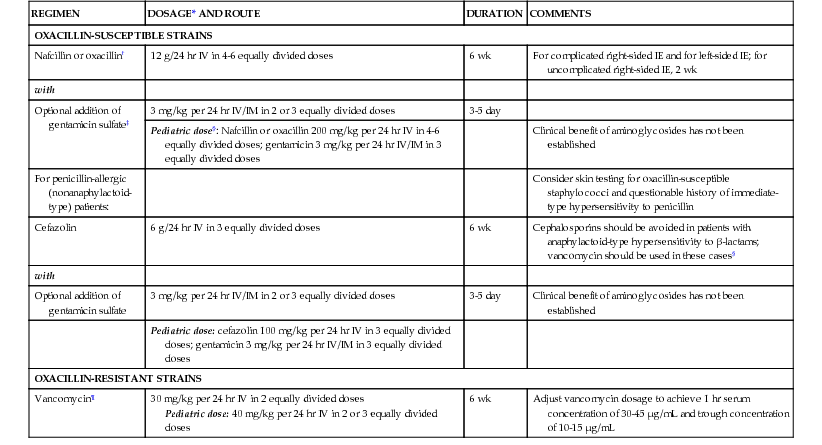
Therapy for Endocarditis Caused by Staphylococci in the Absence of Prosthetic Materials
| REGIMEN | DOSAGE* AND ROUTE | DURATION | COMMENTS |
| OXACILLIN-SUSCEPTIBLE STRAINS | |||
| Nafcillin or oxacillin† | 12 g/24 hr IV in 4-6 equally divided doses | 6 wk | For complicated right-sided IE and for left-sided IE; for uncomplicated right-sided IE, 2 wk |
| with | |||
| Optional addition of gentamicin sulfate‡ | 3 mg/kg per 24 hr IV/IM in 2 or 3 equally divided doses | 3-5 day | |
| Pediatric dose§: Nafcillin or oxacillin 200 mg/kg per 24 hr IV in 4-6 equally divided doses; gentamicin 3 mg/kg per 24 hr IV/IM in 3 equally divided doses | Clinical benefit of aminoglycosides has not been established | ||
| For penicillin-allergic (nonanaphylactoid-type) patients: | Consider skin testing for oxacillin-susceptible staphylococci and questionable history of immediate-type hypersensitivity to penicillin | ||
| Cefazolin | 6 g/24 hr IV in 3 equally divided doses | 6 wk | Cephalosporins should be avoided in patients with anaphylactoid-type hypersensitivity to β-lactams; vancomycin should be used in these cases§ |
| with | |||
| Optional addition of gentamicin sulfate | 3 mg/kg per 24 hr IV/IM in 2 or 3 equally divided doses | 3-5 day | Clinical benefit of aminoglycosides has not been established |
| Pediatric dose: cefazolin 100 mg/kg per 24 hr IV in 3 equally divided doses; gentamicin 3 mg/kg per 24 hr IV/IM in 3 equally divided doses | |||
| OXACILLIN-RESISTANT STRAINS | |||
| Vancomycin¶ | 30 mg/kg per 24 hr IV in 2 equally divided doses Pediatric dose: 40 mg/kg per 24 hr IV in 2 or 3 equally divided doses | 6 wk | Adjust vancomycin dosage to achieve 1 hr serum concentration of 30-45 µg/mL and trough concentration of 10-15 µg/mL |
If the infection occurs on a valve and induces or increases symptoms and signs of heart failure, appropriate therapy should be instituted, including diuretics, afterload reducing agents, and in some cases, digitalis. Surgical intervention for infective endocarditis is indicated for severe aortic, mitral or prosthetic valve involvement with intractable heart failure. Severe heart failure may be associated with acute valve regurgitation, obstruction, or fistula formation. Rarely, a mycotic aneurysm, rupture of an aortic sinus, intraseptal abscess causing complete heart block, or dehiscence of an intracardiac patch requires an emergency operation. Other surgical indications include failure to sterilize the blood despite adequate antibiotic levels in 7-10 days in the absence of extracardiac infection, myocardial abscess, recurrent emboli, and increasing size of vegetations while receiving therapy. Vegetations (aortic, mitral, prosthetic valve) >10-15 mm are at high risk of embolism. Although antibiotic therapy should be administered for as long as possible before surgical intervention, active infection is not a contraindication if the patient is critically ill as a result of severe hemodynamic deterioration from infective endocarditis. Removal of vegetations and, in some instances, valve replacement may be lifesaving, and sustained antibiotic administration will most often prevent reinfection. Replacement of infected prosthetic valves carries a higher risk.
Fungal endocarditis is difficult to manage and has a poorer prognosis. It has been encountered after cardiac surgery, in severely debilitated or immunosuppressed patients, and in patients on prolonged courses of antibiotics. The drugs of choice are amphotericin B (liposomal or standard preparation) and 5-fluorocytosine. Surgery to excise infected tissue is occasionally attempted, though often with limited success. Recombinant tissue plasminogen activation may help lyse intracardiac vegetations and avoid surgery in some high-risk patients.
Prevention
Recommendations by the American Heart Association for antimicrobial prophylaxis before dental and other surgical procedures received a major revision in 2007. A substantial reduction in the number of patients who require prophylactic treatment and the procedures requiring coverage was recommended. The primary reasons for these revised recommendations were that (1) infective endocarditis is much more likely to result from exposure to the more frequent random bacteremias associated with daily activities than from a dental or surgical procedure; (2) routine prophylaxis may prevent “an exceedingly small” number of cases; and (3) the risk of antibiotic-related adverse events exceeds the benefits of prophylactic therapy. Improving general dental hygiene was felt to be a more important factor in reducing the risk of infective endocarditis resulting from routine daily bacteremias. The current recommendations limit the use of prophylaxis to those patients with cardiac conditions associated with the greatest risk of an adverse outcome from infective endocarditis (Table 437-6). Patients with permanently damaged valves from rheumatic heart disease should also be considered for prophylaxis. Prophylaxis for these patients is recommended for “all dental procedures that involve manipulation of gingival tissue or the periapical region of teeth or perforation of the oral mucosa.” Furthermore, “placement of removable prosthodontic or endodontic appliances, adjustment of orthodontic appliances, placement of orthodontic brackets, shedding of deciduous teeth and bleeding from trauma to the lips or oral mucosa” are not indications for prophylaxis. Given that many invasive respiratory tract procedures do cause bacteremia, prophylaxis for many of these procedures is considered reasonable. In contrast to prior recommendations, prophylaxis for gastrointestinal or genitourinary procedures is no longer recommended in the majority of cases. Prophylaxis for patients undergoing cardiac surgery with placement of prosthetic material is still recommended. Given the highly individual nature of these recommendations and the continued concern amongst some cardiologists over their adoption, direct consultation with the child's cardiologist is still the best method for determining a specific patient's ongoing need for prophylaxis (Table 437-7).
Table 437-6
2007 Statement of the American Heart Association (AHA): Cardiac Conditions Associated with the Highest Risk of an Adverse Outcome from Infective Endocarditis for Which Prophylaxis with Dental Procedures Is Reasonable
| CONGENITAL HEART DISEASE (CHD)* |
Unrepaired cyanotic CHD, including palliative shunts and conduits Completely repaired CHD with prosthetic material or device, whether placed by surgery or catheter intervention, during the 1st 6 mo after the procedure† Repaired CHD with residual defects at the site or adjacent to the site of a prosthetic patch, or prosthetic device (which inhibit endothelialization) |
| Cardiac transplantation recipients who develop cardiac valvulopathy |

Table 437-7
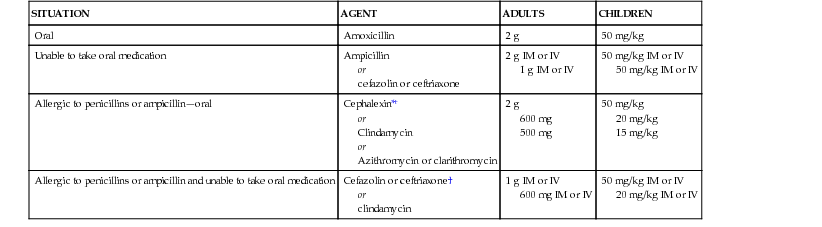
2007 Statement of the American Heart Association (AHA): Prophylactic Antibiotic Regimens for a Dental Procedure
| SITUATION | AGENT | ADULTS | CHILDREN |
| Oral | Amoxicillin | 2 g | 50 mg/kg |
| Unable to take oral medication | Ampicillin or cefazolin or ceftriaxone | 2 g IM or IV 1 g IM or IV | 50 mg/kg IM or IV 50 mg/kg IM or IV |
| Allergic to penicillins or ampicillin—oral | Cephalexin*† or Clindamycin or Azithromycin or clarithromycin | 2 g 600 mg 500 mg | 50 mg/kg 20 mg/kg 15 mg/kg |
| Allergic to penicillins or ampicillin and unable to take oral medication | Cefazolin or ceftriaxone† or clindamycin | 1 g IM or IV 600 mg IM or IV | 50 mg/kg IM or IV 20 mg/kg IM or IV |
Continuing education regarding both oral hygiene and, in appropriate cases, the need for prophylaxis is important, especially in teenagers and young adults. Vigorous treatment of sepsis and local infections and careful asepsis during heart surgery and catheterization reduce the incidence of infective endocarditis.