Clinical Evaluation of the Child with Hematuria
Cynthia G. Pan, Ellis D. Avner
Hematuria is defined as the presence of at least 5 red blood cells (RBCs) per microliter of urine and occurs with a prevalence of 0.5-2.0% among school-age children. Quantitative studies demonstrate that normal children can excrete more than 500,000 RBCs per 12 hr period; this increases with fever and/or exercise. In the clinical setting, qualitative estimates are provided by a urinary dipstick that uses a very sensitive peroxidase chemical reaction between hemoglobin (or myoglobin) and a colorimetric chemical indicator impregnated on the dipstick. Chemstrip (Boehringer Mannheim), a common commercially available dipstick, is capable of detecting 3-5 RBCs/µL of unspun urine. The presence of 10-50 RBCs/µL may suggest underlying pathology, but significant hematuria is generally considered as >50 RBCs/µL. False-negative results can occur in the presence of formalin (used as a urine preservative) or high urinary concentrations of ascorbic acid (i.e., in patients with vitamin C intake >2000 mg/day). False-positive results may be seen in a child with an alkaline urine (pH > 8), or more commonly following contamination with oxidizing agents such as hydrogen peroxide used to clean the perineum before obtaining a specimen. Microscopic analysis of 10-15 mL of freshly voided and centrifuged urine is essential in confirming the presence of RBCs suggested by >10 RBCs/µL, or a 1+ positive urinary dipstick reading.
Red urine without RBCs is seen in a number of conditions (Table 509-1). Clinically significant heme-positive urine without RBCs may be caused by the presence of either hemoglobin or myoglobin. Hemoglobinuria without hematuria can occur in the presence of acute or chronic hemolysis. Myoglobinuria without hematuria occurs in the presence of rhabdomyolysis resulting from skeletal muscle injury and is generally associated with a 5-fold increase in the plasma concentration of creatinine kinase. Rhabdomyolysis is always clinically significant as it may lead to acute renal injury. It can occur secondary to viral myositis, crush injury, severe electrolyte abnormalities (hypernatremia, hypophosphatemia), hypotension, disseminated intravascular coagulation, toxins (drugs, venom), metabolic disorders of muscles, and prolonged seizures. Clinically innocuous heme-negative urine can appear red, cola colored, or burgundy, owing to ingestion of various drugs, foods (blackberries, beets), or dyes used in food and candy, whereas dark brown (or black) urine can result from various urinary metabolites.
Table 509-1
Other Causes of Red Urine
| HEME POSITIVE |
| HEME NEGATIVE |
| Drugs |
| Dyes (Vegetable/Fruit) |
| Metabolites |

Evaluation of the child with hematuria begins with a careful history, physical examination, and urinalysis. This information is used to determine the level of hematuria (upper vs lower urinary tract) and to determine the urgency of the evaluation based on symptomatology. Special consideration needs to be given to family history, identification of anatomic abnormalities and malformation syndromes, presence of gross hematuria, and manifestations of hypertension, edema, or heart failure.
Table 509-2 lists causes of hematuria. Upper urinary tract sources of hematuria originate within the nephron (glomerulus, tubular system, or interstitium). Lower urinary tract sources of hematuria originate from the pelvocaliceal system, ureter, bladder, or urethra. Hematuria from within the glomerulus is often associated with brown, cola- or tea-colored, or burgundy urine, proteinuria >100 mg/dL via dipstick, urinary microscopic findings of RBC casts, and deformed urinary RBCs (particularly acanthocytes). Hematuria originating within the tubular system may be associated with the presence of leukocytes or renal tubular casts. Lower urinary tract sources of hematuria may be associated with gross hematuria that is bright red or pink, terminal hematuria (gross hematuria occurring at the end of the urine stream), blood clots, normal urinary RBC morphology, and minimal proteinuria on dipstick (<100 mg/dL).
Table 509-2
Causes of Hematuria in Children
| UPPER URINARY TRACT DISEASE |
| Isolated renal disease |
| Multisystem disease |
Systemic lupus erythematosus nephritis* Henoch-Schönlein purpura nephritis Granulomatosis with polyangiitis (formerly Wegener granulomatosis) |
| Tubulointerstitial disease |
| Vascular |
| Anatomic |
| LOWER URINARY TRACT DISEASE |
Inflammation (infectious and noninfectious) Factitious syndrome, factitious syndrome by proxy† |

Patients with hematuria can present with a number of symptoms suggesting specific disorders. Tea- or cola-colored urine, facial or body edema, hypertension, and oliguria are classic symptoms of glomerulonephritis. Diseases commonly manifesting as glomerulonephritis include postinfectious glomerulonephritis, immunoglobulin A (IgA) nephropathy, membranoproliferative glomerulonephritis, Henoch-Schönlein purpura (HSP) nephritis, systemic lupus erythematosus (SLE) nephritis, granulomatosis with polyangiitis (formerly Wegener granulomatosis), microscopic polyarteritis nodosa, Goodpasture syndrome, and hemolytic-uremic syndrome. A history of recent upper respiratory, skin, or gastrointestinal infection suggests postinfectious glomerulonephritis, hemolytic-uremic syndrome, or HSP nephritis. Rash and joint complaints suggest HSP or SLE nephritis.
Hematuria associated with glomerulonephritis is typically painless, but can be associated with flank pain when acute or unusually severe. Frequency, dysuria, and unexplained fevers suggest a urinary tract infection, whereas renal colic suggests nephrolithiasis. A flank mass can suggest hydronephrosis, renal cystic diseases, renal vein thrombosis, or tumor. Hematuria associated with headache, mental status changes, visual changes (diplopia), epistaxis, or heart failure suggests significant hypertension. Patients with hematuria and a history of trauma require immediate evaluation (see Chapter 72). Child abuse must always be suspected in the child presenting with unexplained perineal bruising and hematuria.
A careful family history is critical in the initial assessment of the child with hematuria given the numerous genetic causes of renal disorders. Hereditary glomerular diseases include hereditary nephritis (Alport syndrome), thin glomerular basement membrane disease, SLE nephritis, and IgA nephropathy (Berger disease). Other hematuric renal disorders with a hereditary component include both autosomal recessive and autosomal dominant polycystic kidney diseases, atypical hemolytic-uremic syndrome, urolithiasis, and sickle cell disease/trait.
A complete physical examination is critical to assess the cause of hematuria. Hypertension, edema, or signs of heart failure suggest acute glomerulonephritis. Several malformation syndromes are associated with renal disease including VATER (vertebral body anomalies, anal atresia, tracheoesophageal fistula, and renal dysplasia) syndrome. Abdominal masses may be caused by bladder distention in posterior urethral valves, hydronephrosis in ureteropelvic junction obstruction, polycystic kidney disease, or Wilms tumor. Hematuria seen in patients with neurologic or cutaneous abnormalities may be the result of a number of syndromic renal disorders including tuberous sclerosis, von Hippel-Lindau syndrome, and Zellweger (cerebrohepatorenal) syndrome. Anatomic abnormalities of the external genitalia may be associated with hematuria and/or renal disease.
Patients with gross hematuria present additional challenges because of the associated parental anxiety. The most common cause of gross hematuria is bacterial urinary tract infection. Urethrorrhagia, which is urethral bleeding in the absence of urine, is associated with dysuria and blood spots on underwear after voiding. This condition, which often occurs in prepubertal boys at intervals several months apart, has a benign self-limited course. Less than 10% of patients have evidence of glomerulonephritis. Recurrent episodes of gross hematuria suggest IgA nephropathy, Alport syndrome, or thin glomerular basement membrane disease. Dysuria and abdominal or flank pain are symptoms of idiopathic hypercalciuria, or urolithiasis. Table 509-3 lists common causes of gross hematuria; Figure 509-1 outlines a general approach to the laboratory and radiologic evaluation of the patient with glomerular or extraglomerular hematuria. Asymptomatic patients with isolated microscopic hematuria should not undergo extensive diagnostic evaluation, because such hematuria is often transient and benign.
Table 509-3
Common Causes of Gross Hematuria

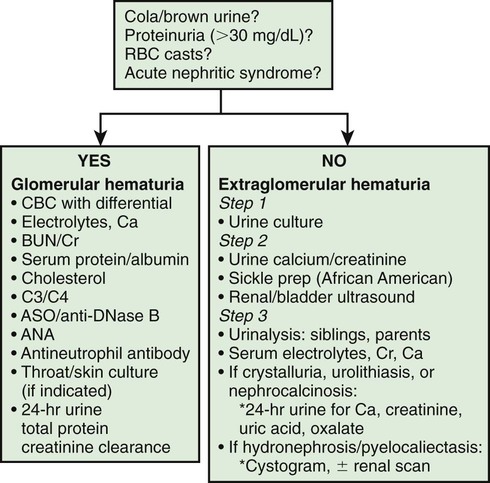
The child with completely asymptomatic isolated microscopic hematuria that persists on at least 3 urinalyses observed over a minimum of a 2 wk period poses a dilemma in regard to the degree of further diagnostic testing that should be performed. Significant disease of the urinary tract is uncommon with this clinical presentation. The initial evaluation of these children should include a urine culture followed by a spot urine for hypercalciuria with a calcium : creatinine ratio in culture-negative patients. In African-American patients, a sickle cell screen should be included. If these studies are normal, urinalysis of all first-degree relatives is indicated. Renal and bladder ultrasonography should be considered to rule out structural lesions such as tumor, cystic disease, hydronephrosis, or urolithiasis. Ultrasonography of the urinary tract is most informative in patients presenting with gross hematuria, abdominal pain, flank pain, or trauma. If these initial studies are normal, assessment of serum creatinine and electrolytes is recommended.
The finding of certain hematologic abnormalities can narrow the differential diagnosis. Anemia in this setting may be caused by hypervolemia with dilution associated with acute renal failure; decreased RBC production in chronic renal failure; hemolysis from hemolytic-uremic syndrome, a chronic hemolytic anemia, or SLE; blood loss from pulmonary hemorrhage, as seen in Goodpasture syndrome; or melena in patients with HSP or hemolytic-uremic syndrome. Inspection of the peripheral blood smear might reveal a microangiopathic process consistent with the hemolytic-uremic syndrome. The presence of autoantibodies in SLE can result in a positive Coombs test, the presence of antinuclear antibody, leukopenia, and multisystem disease. Thrombocytopenia can result from decreased platelet production (malignancies) or increased platelet consumption (SLE, idiopathic thrombocytopenic purpura, hemolytic-uremic syndrome, renal vein thrombosis, or congenital hepatic fibrosis with portal hypertension secondary to autosomal recessive polycystic kidney disease). Although urinary RBC morphology may be normal with lower tract bleeding and dysmorphic from glomerular bleeding, it is not sensitive enough to unequivocally delineate the site of hematuria. A bleeding diathesis is an unusual cause of hematuria. Coagulation studies are not routinely obtained unless personal or family history suggests a bleeding tendency.
A voiding cystourethrogram is only required in patients with a urinary tract infection, renal scarring, hydroureter, or pyelocaliectasis. Cystoscopy is an unnecessary and costly procedure in most pediatric patients with hematuria, and carries the associated risks of anesthesia. The diagnosis of “possible urethral stenosis” as an indication for cystoscopy should be viewed with a high degree of suspicion, because true urethral stenosis is quite rare. This procedure should be reserved for evaluating the rare child with a bladder mass noted on ultrasound, urethral abnormalities caused by trauma, posterior urethral valves, or tumor. The finding of unilateral gross hematuria localized by cystoscopy is rare, but it can indicate a vascular malformation or another anatomic abnormality.
Children with persistent asymptomatic isolated hematuria and a completely normal evaluation should have their blood pressure and urine checked every 3 mo until the hematuria resolves. Referral to a pediatric nephrologist should be considered for patients with persistent asymptomatic hematuria greater than 1 yr duration and is recommended for patients with nephritis (glomerulonephritis, tubulointerstitial nephritis), hypertension, renal insufficiency, urolithiasis or nephrocalcinosis, or a family history of renal disease such as polycystic kidney disease or hereditary nephritis. Renal biopsy is indicated for some children with persistent microscopic hematuria, and most children with recurrent gross hematuria associated with decreased renal function, proteinuria, or hypertension.