Chapter 7 Principles and Applications of Vacuum-Assisted Closure (VAC)
1 What is vacuum-assisted closure (VAC)?
Vacuum-assisted closure (VAC) is a system that promotes the healing of wounds. VAC is based on the principal that negative pressure applied to the wound will promote an improved environment for wound healing.
The concept of a vacuum was first presented by Evangelista Torriculli in the early seventeenth century. In 1993, Fleischmann described his technique of porous polyvinyl alcohol foam wrapped around suction drains, which was introduced into a wound sealed with a polyurethane drape and attached to a suction apparatus at 600 mm Hg. His description in German presented 15 patients. In 1997, Drs. Louis Argenta and Michael Morykwas presented their experience using the V.A.C. device, which was licensed by Kinetic Concepts of San Antonio, Texas. They presented their experience at Wake Forest University in North Carolina with 175 chronic wounds, 94 subacute wounds, and 31 acute wounds over a 9-year period. In a second paper published at the same time, they presented their animal study experience over the same 9-year period.
2 How is the VAC applied and managed while treating a wound?
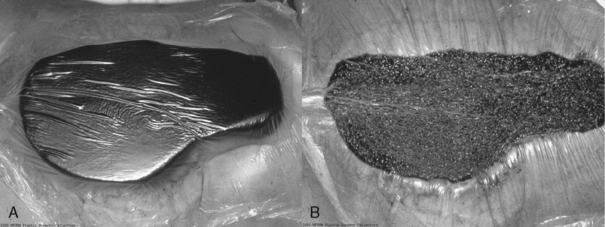
VAC requires placement of a sponge material on or into the wound. The sponge is made of reticulated polyurethane ether foam that is cut to fit the wound. An occlusive drape then is placed over the sponge and onto the surrounding skin. An opening is made in the drape, and tubing is fixed to the exposed sponge with an occlusive seal. The suction tube is attached to a collection canister, which is attached to an adjustable vacuum pump (Fig. 7-1).
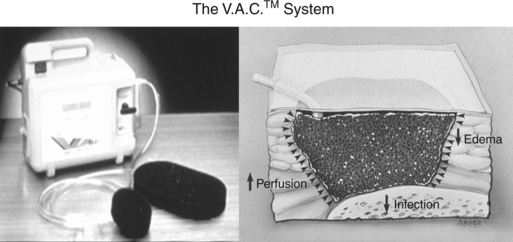
Figure 7-1 The V.A.C. system suction machine and schematic of the sponge filling the dead space of a wound.
(From WFUSM Plastic Surgery Collection.)
Foam made of polyvinyl alcohol (white foam) also can be used in VAC. The vacuum pump can be used as continuous or intermittent vacuum using pressures of 75 to 125 mm Hg when using the polyurethane foam and as higher continuous pressures of 125 to 175 mm Hg when using the polyvinyl alcohol foam. The foam dressing is changed every 48 hours. At the time of foam change, other traditional wound healing modalities, such as pulse lavage, can be used. Silver-impregnated foam sponge has been introduced recently to provide a bacteriocidal sponge in patients with colonized wounds.
3 How does the VAC work?
Although clinical and laboratory experience to date has been exhaustive, the exact mechanism explaining why VAC management is so effective in treating wounds is not fully understood. It is evident that suction on the wound results in the following:
It is also postulated that tissue and cellular deformation creates a steady-state stress on the cell walls, which in turn stimulates growth factor pathways.
4 What have laboratory studies shown?
Morykwas demonstrated in a pig model an increase in blood flow, increased granulation tissue formation, increased bacterial clearance, and increased length of survival in random pattern flaps subjected to negative pressure. He showed an increase in blood flow in both subcutaneous tissue and muscle with a peak blood flow of four times baseline values when 125 mm Hg was applied to the vacuum. The increase in local blood flow declined after 5 to 7 minutes of continuous subatmospheric pressure and finally returned to baseline. With pressures of 400 mm Hg and greater, a fall in blood flow to below that seen in baseline values when applying intermittent subatmospheric pressure was noted (Fig. 7-2). An increase in local blood flow was observed when negative pressure was applied, with a return to baseline when vacuum application was turned off. Wounds treated with VAC were noted to fill with granulation tissue at a significantly increased rate compared with control wounds treated with saline dressing changes. This increase in granulation tissue was noted with continuous suction and was even more significant with intermittent suction. Bacterial clearance studies showed a significant drop in organisms per gram of tissue between the fourth and fifth days, with bacterial levels remaining low throughout the duration of VAC treatment. Bacterial levels in the control wounds peaked at day 5 and dropped after a mean of 11 days.
5 What are the indications for use of the VAC?
VAC is indicated in any patient who would benefit from this device, which promotes wound healing. VAC is indicated for chronic open wounds, diabetic ulcers, stasis ulcers, acute and traumatic wounds, and dehisced wounds. It has been shown to be of immense benefit in stabilizing acute skin grafts. It is beneficial in the treatment of venomous bites and extravasation injuries. It is useful in soft tissue decompression in extremities after fasciotomy.
VAC treatment is advantageous compared with traditional dressing changes for a number of reasons. The sponge must be changed every 2 to 3 days compared with traditional dressing, which must be changed daily at a minimum and often two or three times each day. VAC sponges need to be changed only every 2 to 3 days because a closed, moist environment is maintained during the course of vacuum application. The wounds heal more rapidly, resulting in either a more expeditiously healed wound or a shorter time lag between initial injury or débridement and definitive surgical wound closure.
6 What are contraindications to VAC?
The VAC should not be used under several circumstances. It is contraindicated if hemostasis is not adequate. If a wound is bleeding or oozing, the negative pressure may continue removing blood into the collection system. VAC also is not indicated if necrotic or sloughing tissue is present in the wound. The wounds must be surgically débrided prior to application of the sponge, and VAC treatment does not preclude the need for sound surgical principles of adequate débridement and hemostasis. The sponge should not be placed directly over major exposed blood vessels and organs. If vasculature or major organs are exposed, a petroleum-impregnated gauze should be interposed between the vital structure and sponge. If the vital structure is compromised with any question of viability of a vascular wall, VAC treatment probably is not indicated. Instances of exsanguination in situations where the VAC was inappropriately applied to compromised vasculature have been reported. The system should not be used in nonenteric fistulae that are unexplored without knowing what may lie at the base of the fistula.
7 What are the complications of vacuum-assisted therapy?
The skin surrounding the treated wound may develop sensitivity and irritation from the sponge or overlying occlusive drape, yeast infection due to prolonged moisture under or adjacent to the drape may develop, or pressure necrosis of the skin, which is most commonly caused by poorly placed tubing compressed too tightly against the skin, may occur. Hematoma and hemorrhage may occur in wounds with inadequate hemostasis. Wound infection may develop in inadequately débrided wounds. Intermittent débridement, pulse lavage, and standard wound management modalities must be incorporated into the VAC management protocol. Toxic shock when the negative pressure was not maintained has been reported. If the wound fails to respond to VAC management, then other modalities should be instituted. Patients may experience pain. Lowering the suction pressure will often alleviate the pain, but clearly if pain cannot be controlled with analgesia, VAC management may have to be abandoned.
8 What role does the VAC have in the management of chronic nonhealing wounds?
Chronic wounds are those that fail to heal in the orderly phases of inflammation, proliferation, and maturation. Common chronic wounds include pressure ulcers, diabetic ulcers, venous stasis ulcers, vasculitic ulcers, and chronic nonhealing wounds resulting from trauma or dehisced surgical wounds. Systemic reasons such as chronic debilitation, malnourishment, diabetes, and sepsis may be present, but in all cases a local phenomenon inhibits or fails to stimulate the wound healing cascade. The chronic wound develops progressive edema, compromise of perfusion, and protease imbalances. These wounds have elevated levels of proteolytic enzymes and cytokines, which inhibit granulation tissue formation and epithelialization. This unfavorable environment is ideal for bacterial colonization and development of a vicious cycle with inability of the wound to heal.
The fluid that is drawn from the wound by the VAC system is rich in cytokines, acute phase proteins, and proteolytic enzymes, which suggests that inhibiting factors are removed from the wound. As the wound develops increased blood flow resulting from removal of interstitial fluid and a less favorable environment for bacterial proliferation, chronic nonhealing wounds begin to behave more like acute wounds with rapid healing. Wounds that previously were stagnant for weeks, months, and in some instances years usually demonstrate a steady progression to a healed state.
9 What is the role of the VAC in the management of acute wounds?
The VAC provides an ideal wound dressing and can be used either for healing the acute wound or as a bridging and preparatory modality until definitive wound closure is accomplished. The VAC maintains a moist stable wound while minimizing edema formation and maximizing arterial inflow. Prior to application of the VAC the acute wound must be appropriately managed with débridement of nonviable tissues, irrigation, and hemostasis. Because VAC sponges are changed only every 2 to 3 days, patients experience less pain with fewer dressing changes and, in the case of massive wounds where dressings must be changed intraoperatively, operative visits and frequent administrations of anesthesia are minimized.
10 Is the VAC system efficacious in the management of wounds in children?
The VAC device enables earlier coverage with local tissue and skin grafts in children with complex tissue injuries as it does in adult patients. This minimizes the need for microvascular tissue transfers and other local flaps with the attendant donor site scarring and morbidity. Children are more sensitive to the pain and discomfort associated with VAC changes and are more likely to require anesthesia or heavy sedation for VAC changes. Therefore, children are less likely to be good candidates for outpatient VAC. However, the VAC does minimize the need for daily dressing changes and expedites the time from injury to satisfactory wound healing or definitive wound closure.
11 How is the VAC used to treat acute wounds with exposure of bone, tendon, and vital structures?
The VAC is extremely useful for wounds in which bone is exposed, especially when the periosteum is intact. The VAC system promotes granulation tissue, either bridging the time for definitive wound closure or enabling a simpler surgical option such as Integra and a split-thickness skin graft. If healthy tendon is exposed in the wound, a nonadherent dressing beneath the foam may minimize desiccation and trauma to the tendon.
12 How is the VAC used to salvage exposed orthopedic hardware?
If hardware is exposed in the wound, VAC therapy often promotes healthy granulation tissue, enabling secondary wound closure or flap coverage without the need to remove the hardware. Sound surgical principles must be followed, and if loose screws, large plate exposure, or infection is present, hardware must be removed and alternative fixation established.
13 How is the VAC used to manage the open abdominal wound and abdominal compartment syndrome?
If an open abdominal wound is exposed to the fascial level, the VAC system is applied as with any soft tissue wound. If the fascia is open following a fascial dehiscence, care must be taken to avoid injury to the underlying bowel and vital structures.
Exposure of intraabdominal contents will be seen in the abdominal compartment syndrome. Bowel edema due to abdominal trauma from lengthy intraabdominal operations can be significant such that the abdominal wall cannot be closed.
A fenestrated nonstick dressing should be placed over the bowel to act as an interface between bowel and overlying sponges. The VAC will remove intraabdominal fluid, with resolution of bowel edema while aiding skin and fascial approximation. The patient can be returned to the operating room for reexploration as needed. As bowel edema resolves and the defect narrows, the wound will be amenable to secondary closure. If the wound cannot be closed secondarily, for example, following loss of abdominal wall soft tissues in a traumatic injury, usually sufficient granulation tissue will be present on the bowel to enable performance of a temporizing skin graft.
14 What is the role of the VAC in the management of sternal wounds?
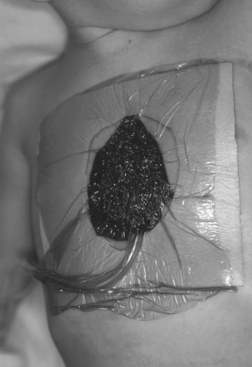
Cardiac surgeons have accepted the VAC as an effective method for managing a sternal wound dehiscence. When the vacuum pump draws air from the porous sponge, the sponge collapses, pulling the edges of the sternum toward the midline (Fig. 7-3). This stabilizes the chest wall, minimizing the need for ventilator support in acutely ill patients. Once the wound is adequately débrided, definitive closure of the sternum or defect can be accomplished. Patients with open sternal wounds are at risk for right ventricular rupture as well as leakage of fresh vascular grafts. Care must be taken to interface the heart and overlying sponge with petroleum gauze.
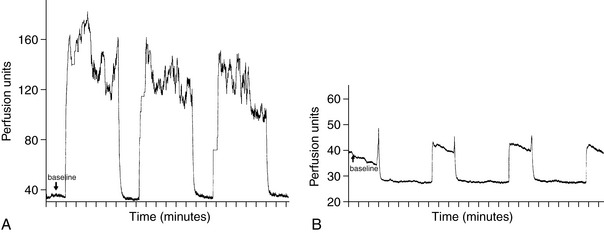
Figure 7-3 A, Recording from laser Doppler needle flow probe placed into subcutaneous tissue at edge of wound. Flow (perfusion units) measured over time (minutes) increased from baseline on cyclical application of 125 mm Hg and returned to baseline during off cycle (5 minutes on, 2 minutes off cycle). B, Recording from laser Doppler needle flow probe placed into subcutaneous tissue at edge of wound. Flow (perfusion units) measured over time (minutes) decreased from baseline levels on cyclical application of 400 mm Hg and returned to baseline during off cycle (5 minutes on, 2 minutes off cycle).
(Reprinted with permission from Morykwas MJ, Argenta LC, Shelton-Brown EI, McGuirt W: Vacuum-assisted closure: A new method for wound control and treatment: Animal studies and basic foundations. Ann Plast Surg 38:553–562, 1997.)
The VAC is efficacious in the management of superficial sternal infections and partial dehiscences. In many instances, appropriate débridement, removal of wires, and VAC management will lead to a healed situation.
15 What is the role of the VAC in extravasation injuries and toxic bites?
We know that the efflux in VAC collecting systems is rich in wound fluids, including cytokines and other wound healing factors. Morykwas showed the efflux to be rich in myoglobin in patients with crush injuries and myoglobinuria. Morykwas also demonstrated that early application of the VAC to a site of doxorubicin injection in a pig model prevented ulcer formation. It is evident from these experiences that the VAC withdraws fluids from the wound environment. The vacuum system applied to extravasation of toxic medications and venomous bites is an effective means to aspirate toxic materials that remain in the wound environment after débridement. Our group has used this method after extravasation injuries and brown recluse spider bites, with excellent response.
16 How does the VAC benefit patients requiring decompression fasciotomy?
VAC dressings are an ideal way to manage wounds following a decompression fasciotomy. With VAC treatment the edematous muscle and tissue are decompressed rapidly over a period of 2 to 3 days. The interval between fasciotomy and wound closure is lessened. In most instances the fasciotomy wound can be closed secondarily rather than requiring a skin graft for wound coverage, which has been the traditional method for management of fasciotomy wounds treated with daily saline dressing changes.
17 What role can the VAC play in skin grafting?
The VAC is an ideal method with which to stent a skin graft. When the air is withdrawn from the sponge, the firm sponge serves as a stent that holds the graft in place for revascularization. The negative pressure also seems to stimulate more rapid neovascularization, providing a better bed for full-thickness skin grafts, dermal substitutes, and grafts on the diploic layer of bone with little granulation tissue.
18 How is the VAC helpful in managing wounds with artificial dermal substitutes such as Integra?
An advantage of using the VAC in skin grafting is that the sponge conforms to the wound, providing an optimal splint of the underlying skin. This is also true in the case of dermal substitutes such as Integra. Molnar showed both experimentally and clinically that the VAC promotes more rapid vascularization of Integra. Traditionally, Integra is not ready for a secondary skin graft for 2 to 3 weeks, but when managed with the VAC a secondary skin graft can be applied in 1 week or less with 93% skin graft take (Fig. 7-4).
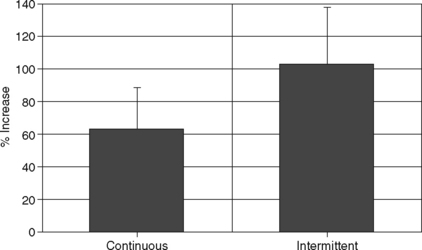
Figure 7-4 Percent increase (mean ± SD) in rate of granulation tissue formation of acute wounds in pigs compared to conventional wet to moist saline gauze dressing changes (control). Both continuous (N = 10) and intermittent (N = 5) application of subatmospheric pressure to the wounds resulted in a significant increase (P ≤.01) in the rate of granulation tissue formation.
(Reprinted with permission from Morykwas MJ, Argenta LC, Shelton-Brown EI, McGuirt W: Vacuum-assisted closure: A new method for wound control and treatment: Animal studies and basic foundations. Ann Plast Surg 38:553–562, 1997.)
19 How is the VAC used in the management of acute burns?
Morykwas showed in a swine model that the VAC decreases burn wound progression (Figs. 7-5 and 7-6). This is likely due to removal of edema fluid allowing improved blood flow into the burn wound environment. This in turn minimizes tissues in the zone of stasis progressing to the zone of coagulation with resultant tissue necrosis. When applied to hand burns, the VAC results in more rapid reduction of hand edema, allowing improved physical therapy and hand mobility.
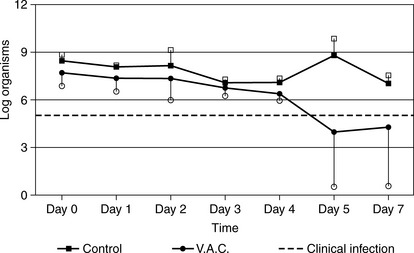
Figure 7-5 Daily quantitative bacterial loads (mean ± SD) (log number of organisms per gram of tissue) of deliberately infected wound tissues. Wounds treated with vacuum-assisted closure (VAC) exhibited a significant decrease in the number of microorganisms after 4 days of treatment (N = 5).
(Reprinted with permission from Morykwas MJ, Argenta LC, Shelton-Brown EI, McGuirt W: Vacuum-assisted closure: A new method for wound control and treatment: Animal studies and basic foundations. Ann Plast Surg 38:553–562, 1997.)
20 Can the VAC be placed over a fresh wound closure or fresh flap?
The collapsed VAC sponge does not traumatize a fresh wound closure or a fresh flap. During the first 24 hours after wound closure the wound is not yet sealed, and the VAC is beneficial in drawing serous ooze from between the suture line. It can be applied over a fresh flap and does not result in any compromise to the underlying flap. Indeed, the negative pressure likely enhances blood flow to the distal aspects of the flap.
21 Does the VAC require prolonged hospitalization and how is it used in outpatient management?
If the patient’s general medical condition lends itself to outpatient care, the patient can be managed with the VAC on an outpatient basis. The Freedom VAC is a small battery-operated vacuum pump that enables the patient to be mobile at home. Nurses who are trained in VAC management only need to visit the home every 2 to 3 days for VAC dressing changes rather than daily or even twice daily for saline dressing changes. This minimizes the need for lengthy hospitalizations of patients who otherwise can be managed at home, with a reduction in the cost of wound management.
22 Is VAC management of a wound cost-effective?
VAC management of a wound has higher costs for the materials, which include the sponges, occlusive drapes, and the vacuum machine, which must be rented per diem. However, because the VAC system promotes more rapid healing, the duration of dressing changes until final wound healing or definitive wound closure is shorter, which more than compensates for the costs of the materials. As physicians, outpatient nurses, and home care specialists become more experienced and adept at managing the VAC, patients are discharged to home VAC treatment, which reduces the need for prolonged inpatient care and these inherent costs.
Argenta L.C., Morykwas M.J. Vacuum-assisted closure: A new method for wound control and treatment: Clinical experience. Ann Plast Surg. 1997;38:563-576.
Banwell Banwell P., Teot L. Topical negative pressure (TNP): The evolution of a novel wound therapy. J Wound Care. 2003;12:23-26.
Barker D.E., Kaufman H.J., Smith L.A., Ciraulo D.L., Richart C.L., Burns R.P. Vacuum pack experience of temporary abdominal closure: A 7-year experience with 112 patients. J Trauma. 2000;48:201-206.
DeFranzo A.J., Argenta L.C., Marks M.W., et al. The use of vacuum-assisted closure therapy for the treatment of lower extremity wounds with exposed bone. Plast Reconstr Surg. 2001;108:1184-1191.
Fleischmann W., Becker U., Bischoff M., Hoekstra H. Vacuum sealing: Indication technique and results. Eur J Orthop Surg Trauma. 1995;5:37-40.
Joseph E., Hamori C.A., Bergman S., et al. A prospective randomized trial of vacuum-assisted closure versus standard therapy of chronic non healing wounds. Wounds. 2000;12:60-67.
Molnar J.A., DeFranzo A.J., Hadaegh A., et al. Acceleration of Integra incorporation in complex tissue defects with subatmospheric pressure. Plast Reconstr Surg. 2004;113:1339-1346.
Mooney J.F.3rd, Argenta L.C., Marks M.W., et al. Treatment of soft tissue defects in pediatric patients using V.A.C. system. Clin Orthop Relat Res. 2000;376:26-31.
Morykwas M.J., Argenta L.C., Shelton-Brown E.I., McGuirt W. Vacuum-assisted closure: A new method for wound control and treatment: Animal studies and basic foundations. Ann Plast Surg. 1997;38:553-562.
Morykwas M.J., David L.R., Schneider A.M., et al. Use of sub-atmospheric pressure to prevent progression of partial thickness burns in a swine model. J Burn Care Rehabil. 1999;20(1 Pt 1):15-21.
Morykwas M.J., Kennedy A.C., Argenta J.P., Argenta L.C. Use of sub-atmospheric pressure to prevent doxorubicin extravasation ulcers in a swine model. J Surg Oncol. 1999;72:14-17.
Scherer L.A., Shiver S., Chang M., et al. The vacuum assisted closure device: A method of securing skin grafts and improving graft survival. Arch Surg. 2002;137(8):930-933.
Skagen K., Henriksen O. Changes in subcutaneous blood flow during locally applied negative pressure to the skin. Acta Physiol Scand. 1983;117(3):411-414.
Tang A.T., Okri S.K., Haw M.P. Vacuum-assisted closure to treat deep sternal wound infection following cardiac surgery. J Wound Care. 2000;9:229-230.