Chapter 106 Principles of Fascia and Fasciocutaneous Flaps
1 What exactly is a fasciocutaneous flap?
According to Tolhurst, any vascularized flap that contains fascia for the intent to augment the overall vascularity is a fasciocutaneous flap. Lamberty disagrees with such a simplistic viewpoint, arguing that a “true” fascial flap must include a specific known “septocutaneous” perforator that discretely supplies the fascia. A broader and very reasonable definition by Nahai states that fasciocutaneous flaps are skin flaps made more reliable by inclusion of the deep fascia, a maneuver that usually ensures preservation of circulation to the skin by whatever means.
BASIC ANATOMY
2 Describe the vascular contributions to the “fascial plexus.”
The fascial plexus is not a discrete structure per se but represents a confluence of multiple, adjacent vascular networks and their branches that have emanated from the perforators of the deep fascia or “fascial feeders.” Intercommunications among these networks exist at the subfascial, fascial, suprafascial, subcutaneous, and subdermal levels.
3 Where are “fascial feeders” found?
These are branches of the source vessels to a given angiosome that do not perforate the deep fascia. Instead, they terminate within the subfascial plexus.
4 What are the six patterns of perforators of the deep fascia that can each supply a distinct type of fasciocutaneous flap?
See Fig. 106-1.
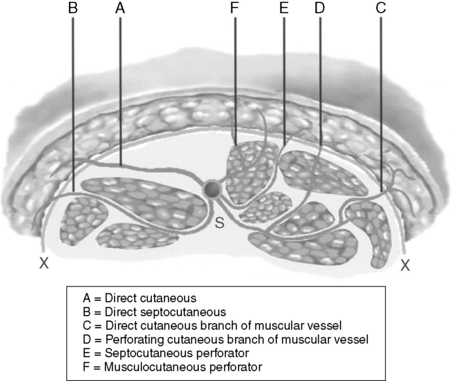
Figure 106-1 Pathways of the various known cutaneous perforators that pierce the deep fascia to supply the fascial plexus. S, Source vessel; X, deep fascia.
(Modified with permission from Hallock GG: Direct and indirect perforator flaps: The history and the controversy. Plast Reconstr Surg 111:855–866, 2003.)
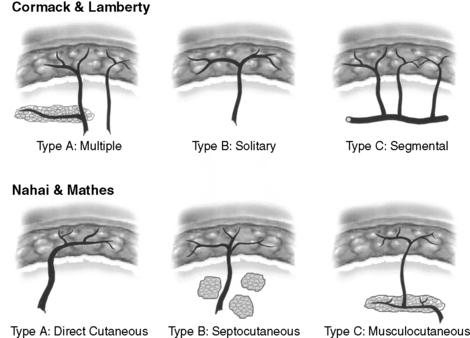
5 Are direct septocutaneous vessels and septocutaneous perforators actually different?
Indeed they are. Both traverse an intermuscular septum, but direct septocutaneous vessels are of relatively large caliber and can nourish the fascial plexus of a large cutaneous territory alone, for example, the circumflex scapular vessels whose cutaneous branch supplies almost the entire dorsal thoracic fascia. Septocutaneous perforators are diminutive and tend to be found as a sequential and close-knit array of branches from the same source vessel, for example, those found nourishing the radial forearm flap.
6 The dorsal thoracic fascia is synonymous with the territory of what fasciocutaneous flaps?
The dorsal thoracic fascia is equivalent to the upper back fascia. It can almost in toto be supplied by the cutaneous branch of the circumflex scapular vessels, which historically has been used to base the scapular and parascapular flaps and their variants. Of course, there are other contributions, such as from intercostal and musculocutaneous perforators.
7 Is a “muscle” perforator flap just a type of fasciocutaneous flap?
This is an extremely controversial point, yet a “muscle” perforator flap relies on the large perforating musculocutaneous branches of the source pedicle to a muscle. These are identical to the “perforating cutaneous branches of a muscular vessel” according to Nakajima et al., and the latter are the basis of one of their types of fasciocutaneous flaps.
8 Simplify the stratification of the types of deep fascial perforators as being either “direct” or “indirect” perforators
The system of nomenclature for skin flaps has become incredibly chaotic due to use of terms such as “axial flaps,” “random flaps,” and “septocutaneous flaps.” This can be simplified by considering that all perforators contributing to the fascial plexus do so either directly or indirectly. Using the terms introduced by Nakajima et al., direct perforators (e.g., axial, septocutaneous, or direct cutaneous branch of a muscular vessel) course from the source vessel of the given angiosome to the skin without first supplying any other deep structure. Indirect perforators (e.g., musculocutaneous perforators) first pass through some intermediary structure before reaching the subdermal plexus. The corresponding flaps would then be either direct or indirect fasciocutaneous flaps.
9 What role does the deep fascia have in most fasciocutaneous flaps?
Although the deep fascia does have an intrinsic microcirculation, for all practical purposes it is avascular. Its real value, when included as part of a fasciocutaneous flap, may be to prevent inadvertent injury to the suprafascial portion of the fascial plexus, thereby increasing the reliability of the flap.
10 Can a fasciocutaneous flap be neither fascial nor cutaneous?
Because the deep fascia itself adds little to the overall circulation to a fasciocutaneous flap, it can be excluded totally as long as the vasculature within the overlying tissues is kept intact. Typically, this maneuver still will allow survival of what then would be a non-fascia fasciocutaneous flap. The skin is an end organ relying on the subdermal plexus fed by the fascial perforators, and it too is superfluous because it does not contribute to vascularization of the fascial plexus per se but is, in fact, a parasite. Thus it also could be excluded without affecting the viability of the rest of the fasciocutaneous flap.
11 Describe the composition of the subcutaneous flap and the adipofascial flap
If both the skin and deep fascia are excluded from a fasciocutaneous flap, what is left is a subcutaneous flap. Similarly, retention of the deep fascia without the skin component creates an adipofascial flap. Because the major portion of the fascial plexus usually is found within the subcutaneous tissues, both can be most simply considered just variations of fasciocutaneous flaps that differ only in their composition.
12 Define the three subtypes of fasciocutaneous flaps using either the Cormack-Lamberty or Nahai-Mathes schema
These two major classification schemas categorize fasciocutaneous flaps into three subdivisions according to the pathway of origin of their fascial perforators (Fig. 106-2).
13 In what body regions do direct fascial perforators predominate when compared with musculocutaneous perforators?
Because most direct perforators of the deep fascia arise within intercompartmental or intermuscular septa, they are more prevalent wherever long, slender muscles are found, such as in the extremities. In contrast, musculocutaneous perforators are more numerous over the broad, flatter muscles associated with the trunk, where muscular septa are few and far apart.
BASIC PHYSIOLOGY
14 Explain how the axis determines the proper orientation for designing a fasciocutaneous flap
The predominant direction of blood flow within a given fascial plexus determines its axis. For the most part, the vector summation of the contributions to the fascial plexus by all perforators is longitudinal in the extremities and somewhat oblique or horizontal in the torso. The design of a fasciocutaneous flap oriented along this axis or direction of flow will maximize its potential length by best capturing adjacent perforator territories.
15 How can the maximum potential length of a fasciocutaneous flap be estimated?
Safe limits for the design of a fasciocutaneous flap have been determined only by trial and error, for example, a length-to-width ratio of 2:1 for the lower extremity (compared with 1:1 for random flaps). There can be no set rules because deep fascia perforators are so frequently anomalous in caliber and location not only among individuals but also in opposite sides of the same individual.
16 What is the point of rotation of a fasciocutaneous flap?
Applicable to local flaps, this corresponds to the site where the flap is tethered by its vascular pedicle.
17 What is the arc of rotation of a fasciocutaneous flap?
The distance from the point of rotation to the most distant safe length of the given flap determines the range of coverage or arc of rotation through which a local fasciocutaneous flap can be transposed. Whereas the expected range in flap size for a given body region has been learned through the experience of other surgeons, the maximal arc of rotation really remains conjectural at this time.
18 Who is Pontén, and what are his “superflaps”?
Bengt Pontén of Sweden is generally credited for reintroducing the concept of the fasciocutaneous flap. He observed that undelayed cutaneous flaps from the lower leg, if oriented along a longitudinal axis with retention of the deep fascia, had extraordinary viability up to even a 3:1 length-to-width ratio. Historically, only 1:1 flaps (without fascia) previously had been considered safe in this region. His flaps were proximally based and sensate, with no discrete perforator ever identified (i.e., Cormack-Lamberty type A). Some authorities consider these “superflaps” to be identical to the neurocutaneous flaps of the lower extremity, which also have a unique robustness; and in both cases may be more than just a coincidence.
19 Define a distal-based fasciocutaneous flap. Why is it more dependable than its muscle flap counterpart?
For muscle flaps, distal-based implies that the vascular pedicle chosen to sustain the muscle enters that border of the flap either farthest away from the heart or its dominant vascular supply, which by definition would be a minor or secondary blood supply. Usually for distal-based fasciocutaneous flaps, the pedicle is found at that boundary farthest from the heart. They are more dependable because flow in the fascial plexus typically is multidirectional, which would be equivalent if the chosen distal fascial perforator had characteristics comparable to the proximal perforator. It is the quantity of flow via the chosen perforator (usually proportional to caliber) rather than the pedicle orientation per se that determines the extent of viability of a fasciocutaneous flap.
20 State the primary advantage of a distal-based fasciocutaneous flap
Proximal skin territories known to be reliable can be transposed on a distal pedicle for potential coverage of acral defects. This is especially valuable in the extremities where otherwise a free flap might be the only acceptable alternative. In this regard, the distal-based sural flap has become a workhorse flap. The skin of the calf, relying on a distal perforator of the peroneal artery, can be transferred to cover the foot and ankle, primarily in lieu of a microsurgical tissue transfer.
21 Are distal-based fasciocutaneous flaps and retrograde flow-flaps the same entity?
Sometimes they can be. Retrograde-flow flaps usually are distal-based flaps where both arterial inflow to and venous outflow from a more proximal skin territory is in a reverse direction from the normal. A good example is the distal-based radial forearm flap where the radial artery is perfused in a reverse fashion from the ulnar artery via an intact superficial palmer arch. However, a distal perforator sustaining a distal-based flap, if appropriately designed, still could maintain an orthograde pattern of flow. This is true, for example, if the radial recurrent artery is chosen to supply a distal-based lateral arm flap. Suffice it to say that a distal-based flap may be the same as, but is not necessarily synonymous with, a retrograde-flow flap. Furthermore, reverse flow does not necessarily mean retrograde flow.
22 How does venous regurgitation occur in a retrograde flow flap?
Normally, the reversal of venous outflow is obstructed by valves. Two hypotheses currently explain the clinical observation of venous regurgitation in retrograde-flow flaps despite the presence of valves:
The next time you dissect a major limb artery, carefully check the two venae comitantes and note the numerous communicating branches that cross over the artery to reach each other. Usually just a mere nuisance that hinders dissection of that artery, these branches may be an important avenue for circumventing the valves in that segment of veins (Box 106-1).
23 How is Allen’s test relevant to the Chinese flap?
Because the radial forearm flap initially was developed by the Chinese, it is sometimes referred to as the “Chinese flap.” If the radial artery is included with the flap, the ulnar artery must maintain sufficient collateral circulation to the hand. Compression of both arteries at the wrist followed by release of only the ulnar artery must demonstrate complete hand perfusion (i.e., a negative Allen’s test). Otherwise, sacrifice of the radial artery with this flap would be contraindicated.
24 What is the superficial ulnar artery trap?
The superficial ulnar artery trap is an excellent example of the potentially disastrous complications that plague the fasciocutaneous system of flaps due to the frequency of anatomic anomalies. Normally, the ulnar artery lies deep within the medial intermuscular septum of the forearm. However, in approximately 9% of individuals it lies superficial to the deep fascia. Thus the unsuspected inclusion of all suprafascial structures within a radial forearm flap would totally devascularize the hand!
APPLIED ANATOMY
25 Why has the radial forearm flap fallen into disrepute in some quarters of the world?
Although the flap itself has numerous attractive attributes, donor site morbidity potentially can be disastrous. This includes hand ischemia, dysesthesias, dysfunction from tendon adhesions, osteomyelitis, and a nonesthetic appearance if a skin graft was used. Other cutaneous flaps now can offer the same advantages without the risks.
26 What is the Becker flap?
The territory of the Becker flap (named after the noted anatomist) corresponds to the dorsal ulnar border of the forearm. Becker described a fairly constant dorsal branch of the ulnar artery that supplies this region. Because the point where it pierces the deep fascia is only a few centimeters proximal to the pisiform, it can be used as a distal-based island flap (with orthograde perfusion) to take the proximal ulnar forearm skin for provision of coverage of the hand without sacrificing the ulnar artery itself.
27 Why has the groin flap fallen into disfavor?
Although a cutaneous flap from the groin still can be useful as a pedicled or free flap, the dissection can be hampered by the high frequency of vascular anomalies. The groin flap is a notorious example of a not so uncommon and major problem with using fasciocutaneous flaps as a group. The lateral groin is nourished medially by both the superficial circumflex iliac artery (SCIA) and superficial inferior epigastric artery (SIEA) and laterally by contributions from the deep circumflex iliac artery. A reciprocal relationship in the size or variation even in the presence of these vessels is the norm. In 48% of cases, the SIEA and SCIA share an origin from the common femoral artery. They may be inversely related or equal in diameter, or one may be altogether missing.
28. The importance of the triangular space of the thorax is because what direct fascial perforator emanates through it?
The largest branch of the subscapular artery usually is the circumflex scapular, which in turn passes through the triangular space to give off a cutaneous perforator to the dorsal thoracic fascia. This in turn terminates in branches that radiate like the spokes of a wheel to supply many named upper back fasciocutaneous flaps, such as the ascending scapular (ascending branch), scapular (transverse branch), parascapular (descending branch), and inframammary extended circumflex scapular flap (unnamed branch).
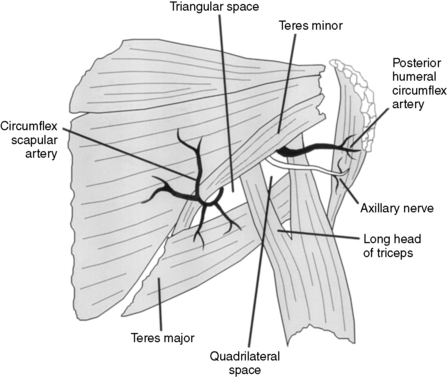
29 Name the muscles that define the boundaries of the triangular space
The teres minor superiorly, the teres major inferiorly, and the long head of the triceps muscle laterally form a potential triangular shaped opening located just superior to the posterior axilla (Fig. 106-3).
30 What important structures pass through the quadrilateral space to form the neurovascular pedicle for a sensate upper arm fasciocutaneous flap?
The posterior circumflex humeral (PCH) vessels and axillary nerve traverse the quadrilateral space. A cutaneous branch of the PCH and the lateral brachial cutaneous nerve from the axillary nerve supply the deltoid flap, which is a potentially sensate flap from the upper outer arm.
31 Name the structures that define the boundaries of the quadrilateral space
The medial side of the quadrilateral space is formed by the long head of the triceps, the inferior side by the teres major, and superior border by the teres minor muscle. The humerus defines the lateral border (see Fig. 106-3).
32 Perhaps the most notorious liability of the fasciocutaneous flap is the risk of morbidity at the donor site, especially if a skin graft has been required for closure. Describe at least three ways in which this specific risk can be minimized
Any variants of the fasciocutaneous flap that exclude the cutaneous component (e.g., subcutaneous, fascial, or adipofascial flap) leave behind the original skin with its intact subdermal plexus. This can be used to close the donor site directly. In patients with enough local skin redundancy, primary closure may also be possible even with a fasciocutaneous flap if the desired flap is small or if pretransfer or posttransfer tissue expansion has been performed. Finally, certain geometric designs of local flaps can be used with the intent not just to close the defect but also to provide simultaneous donor site closure, such as a V-Y advancement or bilobed fasciocutaneous flap.
33 Name some advantages of fasciocutaneous flaps when compared with muscle flaps
Probably the most valuable asset is that no functioning muscle is expended. Fasciocutaneous flaps are readily accessible because they are near the skin surface. As a corollary, deep underlying neurovascular structures can be avoided and risk of their injury minimized. If a cutaneous nerve can be incorporated into the flap design, true sensate flaps are possible (Table 106-1).
Table 106-1 Attributes and Liabilities of Fasciocutaneous versus Muscle Flaps
| Fasciocutaneous Flaps | Muscle Flaps | |
|---|---|---|
| Accessibility | + | − |
| Anatomic anomalies | − | + |
| Availability | + | − |
| Composite flaps | = | = |
| Use in infected or irradiated wound | ± | + |
| Donor site morbidity | − | + |
| Dynamic transfer | − | + |
| Expendable | + | − |
| Malleability | − | + |
| Microsurgical tissue transfer | = | = |
| Reliability | = | = |
| Sensate | + | − |
| Size | + | ± |
| Thinness | − | + |
+, Asset; −, detriment; =, no significant difference; ±, variable.
34 Identify the source vessel and type of perforator in these 10 commonly used fascia flaps
See Table 106-2 and Fig. 106-4.
Table 106-2 Source Vessel and Flap Subtype Perforator
| Fascia Flap | Source Vessel | Subtype Perforator |
|---|---|---|
| 1. Temporoparietal | Superficial temporal | Axial (direct) |
| 2. Lateral arm | Posterior radial collateral | Septocutaneous (direct) |
| 3. Radial forearm | Radial | Septocutaneous (direct) |
| 4. Groin | Superficial circumflex iliac or inferior epigastric | Axial (direct) |
| 5. Anterolateral thigh | Lateral circumflex femoral descending branch or perforating branch of muscular vessel | Septocutaneous (direct) Musculocutaneous (indirect) |
| 6. Saphenous | Descending geniculate | Septocutaneous (direct) |
| 7. Peroneal | Peroneal or perforating branch of muscular vessel | Musculocutaneous (indirect) |
| 8. Dorsal thoracic | Circumflex scapular | Septocutaneous (direct) |
| 9. Posterior interosseous | Posterior interosseous | Septocutaneous (direct) |
| 10. Gluteal thigh | Inferior gluteal descending branch | Septocutaneous (direct) |
Becker C., Gilbert A. The ulnar flap: Description and applications. Eur J Plast Surg. 1988;11:79-82.
Chuang D.C.C., Colony L.H., Chen H.C., Wei F.C. Groin flap design and versatility. Plast Reconstr Surg. 1989;84:100-107.
Cormack G.C., Lamberty B.G.H. A classification of fasciocutaneous flaps according to their patterns of vascularization. Br J Plast Surg. 1984;37:80-87.
Cormack G.C., Lamberty B.G.H. The Fasciocutaneous System of Vessels: The Arterial Anatomy of Skin Flaps, 2nd ed, Edinburgh: Churchill Livingston; 1994:105-129.
del Pinal F., Taylor G.I. The deep venous system and reverse flow flaps. Br J Plast Surg. 1993;46:652-664.
Devansh D. Superficial ulnar artery trap. Plast Reconstr Surg. 1996;97:420-426.
Donski P.K., Fodgestam I. Distally based fasciocutaneous flap from the sural region. A preliminary report. Scand J Plast Reconstr Surg. 1983;17:191-196.
Hahn S.M., Kim N.H., Yang I.H. Deltoid sensory flap. J Reconstr Microsurg. 1990;6:21-28.
Hallock G.G. Clinical scrutiny of the de facto superiority of proximally versus distally based fasciocutaneous flaps. Plast Reconstr Surg. 1997;100:1428-1433.
Hallock G.G. Direct and indirect perforator flaps: The history and the controversy. Plast Reconstr Surg. 2003;111:855-866.
Jones B.M., O’Brien C.J. Acute ischaemia of the hand resulting from elevation of a radial forearm flap. Br J Plast Surg. 1985;38:396-397.
Kim P.S., Gottlieb J.R., Harris G.H., Nagle D.J., Lewis V.L. The dorsal thoracic fascia: Anatomic significance with clinical applications in reconstructive microsurgery. Plast Reconstr Surg. 1987;79:72-80.
Lin S.D., Lai C.S., Chiu C.C. Venous drainage in the reverse forearm flap. Plast Reconstr Surg. 1984;74:508-512.
Mathes S.J., Nahai F. The reconstructive triangle: A paradigm for surgical decision making. In: Reconstructive Surgery: Principles, Anatomy, & Technique. New York: Churchill Livingstone; 1997:9-36.
Nahai F. Surgical indications for fasciocutaneous flaps (invited comment). Ann Plast Surg. 1984;13:502-503.
Nakajima H., Fujino T., Adachi S. A new concept of vascular supply to the skin and classification of skin flaps according to their vascularization. Ann Plast Surg. 1986;16:1-17.
Nakajima H., Minabe T., Imanishi N. Three-dimensional analysis and classification of arteries in the skin and subcutaneous adipofascial tissue by computer graphics imaging. Plast Reconstr Surg. 1998;102:748-760.
Niranjan N.S., Price R.D., Govilkar P. Fascial feeder and perforator-based V-Y advancement flaps in the reconstruction of lower limb defects. Br J Plast Surg. 2000;53:679-689.
Ohsaki M., Maruyama Y. Anatomical investigations of the cutaneous branches of the circumflex scapular artery and their communications. Br J Plast Surg. 1993;46:160-163.
Ozkan O., Coskunfirat O.K., Dikici M.D., Ozgentas H.E. A rare and serious complication of the radial forearm flap donor site: Osteomyelitis of the radius. J Reconstr Microsurg. 2005;21:293-296.
Pontén B. The fasciocutaneous flap: Its use in soft tissue defects of the lower leg. Br J Plast Surg. 1981;34:215-220.
Richardson D., Fisher S.E., Vaughan E.D., Brown J.S. Radial forearm flap donor-site complications and morbidity: A prospective study. Plast Reconstr Surg. 1997;99:109-115.
Timmons M.J., Missotten F.E.M., Poole M.D., Davies D.M. Complications of radial forearm flap donor sites. Br J Plast Surg. 1986;39:176-178.
Tolhurst D.E.L. Fasciocutaneous flaps and their use in reconstructive surgery. Perspect Plast Surg. 1990;4:129-145.
Touam C., Rostoucher P., Bhatia A., Oberlin C. Comparative study of two series of distally-based fasciocutaneous flaps for coverage of the lower one-fourth of the leg, the ankle, and the foot. Plast Reconstr Surg. 2001;107:383-392.
Wei F.C., Jain V., Celik N., Chen H.C., Chuang D.C.C., Lin C.H. Have we found an ideal soft-tissue flap? An experience with 672 anterolateral thigh flaps. Plast Reconstr Surg. 2002;109:2219-2226.
Wei F.C., Jain V., Suominen S., Chen H.C. Confusion among perforator flaps: What is a true perforator flap? Plast Reconstr Surg. 2001;107:874-876.