10 INTERPRETATION OF THE LEUKOGRAM
1 What is the predominant blood leukocyte during health in the domestic animals?
In cats, dogs, and neonatal ruminants, neutrophils are the most abundant leukocyte, followed by lymphocytes. In mature ruminants, lymphocytes are the predominant leukocyte, with fewer neutrophils. In horses and pigs, numbers of neutrophils and lymphocytes are approximately equal. Eosinophils occur in very low numbers on blood smears from healthy animals, and basophils are almost never observed.
2 What is a left shift?
The term left shift indicates the presence of neutrophil precursors in blood, typically increased numbers of band neutrophils and rarely metamyelocytes and earlier precursors. A left shift occurs when the number of segmented neutrophils in the blood pools and marrow storage pools are depleted by excessive emigration to inflammatory sites. Band neutrophils (and earlier precursors) will prematurely leave the marrow at this point. Although often occurring with bacterial infections, left shifts also result from noninfectious causes of inflammation, such as tissue necrosis and immune-mediated disease.
3 How are left shifts categorized?
Leukograms with an increased concentration of band (or earlier) neutrophils are often categorized as “regenerative” or “degenerative” left shifts. Increased concentrations of both segmented and band neutrophils constitute a regenerative left shift. This is regarded as a somewhat favorable change, since the granulopoietic response of marrow is maintaining an appropriately increased concentration of segmented neutrophils during inflammation. No uniform definition exists for degenerative left shift. Some maintain it is the opposite of a regenerative left shift, with the segmented neutrophil concentration within or below its reference interval and with increased band (or earlier) neutrophils. Others restrict “degenerative” to leukograms with absolute concentrations of band neutrophils that exceed the segmented neutrophils, a rare event. Despite controversy over the definition, it is agreed that a degenerative left shift is of greater concern than a regenerative left shift. With degenerative left shift, marrow granulopoiesis is unable to maintain an appropriate segmented neutrophil concentration because of granulopoietic suppression and/or excessive demand for blood neutrophils at sites of inflammation.
4 Define mature neutrophilia.
The term mature hemophilia describes an increased concentration of segmented neutrophils and no band neutrophils (or earlier precursors) on the leukogram. It is observed in many different conditions, and without knowledge of the degree of neutrophilia, it can be a relatively nonspecific finding. However, mature neutrophilia is a convenient term to describe leukograms with neutrophilia and no left shift.
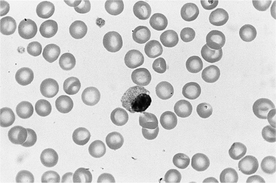
5 What is a right shift?
The term right shift describes increased numbers of hypersegmented neutrophils, with five or more nuclear lobes. Hypersegmented neutrophils are not specifically included in the leukocyte differential count, so right shifts are not quantitated like left shifts. Hypersegmentation of neutrophils usually occurs as an aging change, when neutrophils circulate in blood for a longer time. Right shifts occur most often in animals (especially dogs) on glucocorticoid therapy or with hyperadrenocorticism, since glucocorticoids inhibit the emigration of neutrophils from blood vessels and prolong their circulation. Rarer causes of hypersegmentation are developmental disorders of neutrophils, such as erythroid macrocytosis of poodles, vitamin B12 maladsorptive disorder in giant schnauzers, myelodysplasia in animals with myelogenous leukemia, and benign idiopathic neutrophil hypersegmentation in horses (Figure 10-1).
6 What are the components of a glucocorticoid-induced (“stress”) leukogram?
Stress leukograms occur in all animals but are most common and pronounced in dogs. Endogenous and exogenous glucocorticoids affect the concentration of most leukocytes. A prime feature of the stress leukogram is mature neutrophilia, resulting from increased release of segmented neutrophils from the marrow storage pool, as well as demargination of blood neutrophils to the circulating neutrophil pool. Monocytosis also occurs in dogs because monocytes shift from their marginal pool to the circulating monocyte pool. Another characteristic finding is lymphopenia, the result of diminished recirculation of lymphocytes from lymphoid tissues to venous blood. Chronic glucocorticoid administration also causes lysis of lymphocytes. Eosinopenia may be present, probably because eosinophils are sequestered in marrow or other tissues. The stress leukogram develops over several hours and peaks 6 to 8 hours after administration of exogenous glucocorticoids. These leukocyte changes persist up to several days, depending on the type and dose of glucocorticoid.
7 What is the maximal degree of neutrophilia expected in a stress leukogram?
The segmented neutrophil concentration typically increases two to three times the upper limit of its reference interval in stress leukograms. In dogs, for example, the segmented neutrophil concentration may be as high as 35,000 cells/microliter (μl) because of endogenous/exogenous glucocorticoids. Many inflammatory leukograms with a mature neutrophilia occur in the same range as stress leukograms, making differentiation of stress and inflammatory leukograms difficult without a clinical history and physical examination of the patient.
8 What disease should be suspected when a stress leukogram is absent in a clinically ill animal?
Unchanged or even increased concentrations of lymphocytes and eosinophils (instead of lymphopenia and eosinopenia) during illness suggest a “relaxed” leukogram. These changes are characteristic of dogs and cats with hypoadrenocorticism. The lack of mineralocorticoids produces distinctive electrolyte findings in animals with hypoadrenocorticism, but the lack of glucocorticoids produces the distinctive changes on the leukogram.
9 What is a physiologic leukogram?
The physiologic leukogram typically occurs in young (<12 months of age) animals, although it is least likely to occur in dogs. The leukocyte changes are mediated by epinephrine during the “fight or flight” response that occurs with excitement, fear, or anxiety or with physical exertion, including parturition or convulsions. The distinguishing feature of the physiologic leukogram is lymphocytosis. The lymphocytosis is attributed to increased entry of lymphocytes into blood, presumably from splenic contraction and muscular activity that enhance the lymphatic and thoracic duct flow of lymphocytes into blood. A mature neutrophilia is also present, caused by demargination of blood neutrophils. The increased cardiac output and blood flow apparently sweep neutrophils from the marginal pool into the circulating neutrophil pool. Minimal changes occur in monocyte, eosinophil, and basophil concentrations. The physiologic leukogram is observed infrequently, in part because this leukocytosis resolves within 30 minutes.
10 Describe the components of an inflammatory leukogram.
On the inflammatory leukogram, neutrophil concentrations may vary from decreased to increased during inflammation. The blood concentration depends on the balance between the rate of blood neutrophil emigration to tissues in response to increased chemoattractants, and the rate of marrow granulopoiesis that replaces the blood neutrophils. The monocyte concentration often parallels an increase in neutrophils, since they share a common stem cell (colony-forming unit [CFU]-GM) and respond to granulocyte-macrophage colony-stimulating factor (GM-CSF). Increased numbers of band neutrophils may be released from the marrow storage pool with depletion of segmented neutrophils. Lymphopenia and eosinopenia may also occur with inflammation. Although lymphopenia and eosinopenia are often attributed to concurrent glucocorticoid release, inflammatory mediators can induce these changes independent of glucocorticoids. Depending on the cause of inflammation, eosinophilia may actually occur.
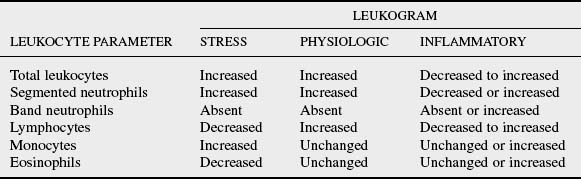
11 Compare the changes in leukocyte concentration that occur in stress, physiologic, and inflammatory leukograms.
12 Rank the domestic animals by their ability to develop maximal degrees of neutrophilia during inflammation.
13 Are there “panic values” for changes on the leukogram?
Severe neutropenia (<500 or <1000 cells/μl) is the only leukocyte change that requires the immediate attention of the clinician. When caused by decreased marrow production, severe neutropenia is especially a concern because improvement of the neutrophil concentration is not likely in the short term. Animals with severe neutropenia are at risk to develop sepsis, so administration of prophylactic antibiotics should be considered to prevent bacterial infection.
14 What changes on serial leukograms would be considered favorable in animals with inflammatory disease?
15 List the general mechanisms (with appropriate examples) that cause neutropenia.
16 Why are cattle more likely to develop neutropenia with acute inflammation?
This common finding in cattle is attributed to a smaller storage pool of segmented neutrophils in marrow or slower replenishment of this storage pool with segmented neutrophils. In cattle with acute inflammation, particularly localized inflammation such as mastitis, neutropenia (often with a left shift) is initially present for 1 to 2 days. For this reason, degenerative left shifts are not unusual in cattle with acute inflammation and do not carry the same concern associated with this leukogram in other animals. Increased marrow granulopoiesis should quickly compensate and lead to “rebound” neutrophilic leukocytosis in the ensuing days.
17 Why does endotoxemia cause neutropenia?
One of the many systemic effects of endotoxin is to enhance the adhesion of blood neutrophils to the endothelial surface by activating adhesion molecules. Redistribution of blood neutrophils occurs as a shift from the circulating to marginal pool, manifested as a severe neutropenia. Because the neutrophils essentially remain in blood but are marginated and are not represented on the leukogram, the change is termed pseudoneutropenia. The neutropenia is an acute but transient change often followed by neutrophilia, since endotoxin causes release of neutrophils from the marrow storage pool.
18 Why does severe neutropenia occur with parvoviral infection?
Canine and feline parvoviruses target cells with a high mitotic rate, including hematopoietic precursors. The blood and marrow pools of segmented neutrophils are quickly depleted because of the short half-life of neutrophils and the lack of replenishment by impaired granulopoiesis. In addition, loss of villous enterocytes results from viral damage to the intestinal crypt cells. The resulting increased enteric absorption of endotoxin further aggravates the neutropenia in parvovirus-infected animals. Neutropenia without thrombocytopenia or anemia are the classic hematologic findings in parvoviral infections.
19 List two general mechanisms (with appropriate examples) that cause neutrophilia.
20 List specific diseases that cause extreme neutrophilia in the dog.
Because dogs can develop a higher degree of neutrophilia than other animals, extremely high concentrations of neutrophils (>50,000/μl) are possible. The leukogram may include a mature neutrophilia or a regenerative left shift. Although studies have demonstrated a lower rate of survival in dogs (and cats) with extreme neutrophilia, the disease causing this leukocytosis is a likely determinant of survivability.
21 What is a leukemoid response?
The term leukemoid response was coined to describe leukograms with neutrophilic leukocytosis, marked left shift with bands and earlier precursors, and reactive lymphocytes. These leukograms were found to be inflammatory in origin, but the initial blood smear was more suggestive of myelogenous leukemia. Unfortunately, this term is incorrectly applied to leukograms with extreme neutrophilia (>50,000/μl) that lack a left shift or have a modest left shift (bands only). The inflammatory origin of these leukograms is quite evident, and myelogenous leukemia is not likely. These leukograms are correctly described as “marked” or “extreme” neutrophilia. The “leukemoid” terminology is intended for the rare leukogram with marked left shift, for which leukemia is a realistic differential diagnosis.
22 What acute change in neutrophil concentration is expected after surgical correction of pyometra in dogs?
Pyometra is a classic example of localized intense neutrophilic inflammation, which induces a significant increase in granulopoiesis. After hysterectomy, the chemotactic signals causing neutrophil emigration to the inflammatory site immediately disappear. However, the neutrophilic precursors in the expanded proliferative and storage pools in marrow continue to mature and release neutrophils, which causes an even greater degree of neutrophilia after surgery. Granulopoiesis will quickly down-regulate, and the neutrophilia will abate after a few days.
23 In which tissues is inflammation not likely to induce an inflammatory leukogram?
Neutrophilic inflammation in the urinary bladder, intestine, epidermis, and central nervous system typically causes minimal or no change in the leukogram. This is attributed to loss of inflammatory mediators (e.g., CSFs) or their seclusion in protected tissue spaces. Therefore, no systemic inflammatory response is reflected in granulopoiesis and the leukogram.
25 List conditions associated with basophilia.
27 List conditions associated with lymphopenia.
Chylous effusions caused by anterior mediastinal mass, congestive heart failure, or thoracic duct leakage
29 Why is monocytosis a favorable indicator for recovery from leukopenia?
After transient suppression of hematopoiesis and development of leukopenia, an increased blood concentration of monocytes typically precedes the recovery of neutrophils and other granulocytic leukocytes. Monocytes have a rapid marrow transit time (2 days for monoblasts to differentiate to monocytes), versus the longer marrow transit time (6 days) for differentiation of segmented neutrophils from myeloblasts. In addition, monocytes rapidly leave the marrow, whereas neutrophils enter a marrow storage pool, which can delay their entry into blood.
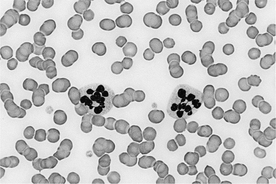
30 What is the significance of mast cells on blood smear?
Mastocytemia, or the presence of mast cells in blood, is often associated with disseminated mast cell neoplasms or mast cell leukemia in dogs and cats. However, low numbers of mast cells are also observed on blood or “buffy coat” smears in several inflammatory diseases of the dog, including immune-mediated hemolytic anemia, parvoviral infection, dermatitis, pleuritis, and peritonitis. The morphology of mast cells with inflammatory diseases may be similar to neoplastic mast cells, with varying degrees of metachromatic granulation evident in both disease types (Figure 10-2).
Feldman BF, Zinkl JG, Jain NC, editors. Schalm’s veterinary hematology, ed 5, Baltimore: Lippincott Williams & Williams, 2000.
Harvey JW. Leukocytes. In: Atlas of veterinary hematology: blood and bone marrow of domestic animals. Philadelphia: Saunders; 2001:45-74.
Latimer KS, Prasse KW. Leukocytes. In: Latimer KS, Mahaffey E, Prasse KW, editors. Duncan and Prasse’s veterinary laboratory medicine: clinical pathology. Ames: Iowa State University Press; 2003:46-79.
Stockham SL, Scott MA. Leukocytes. In: Fundamentals of veterinary clinical pathology. Ames: Iowa State University Press; 2002:49-83.