30 TESTS FOR EVALUATION OF LIVER DISEASE
1 What are the major categories of enzymes used to evaluate liver disease?
There are two major categories of liver enzymes: leakage enzymes and cholestatic enzymes. Leakage enzymes are enzymes that leak into the plasma when hepatocyte injury or death occurs. Therefore, high activities in serum are an indication of hepatocellular injury. Commonly measured leakage enzymes include the following:
Cholestatic enzymes are enzymes for which synthesis is increased as a result of bile retention or administration of drugs. Bile retention usually results from intrahepatic or extrahepatic bile duct obstruction. Commonly measured cholestatic enzymes include the following:
2 What are isoenzymes?
Isoenzymes are molecules with similar function but variable molecular structure. In general, these isoenzymes are produced in different tissues. For example, LDH (also LD) has five isoenzymes. LD1 and LD2 are primarily located in cardiac muscle, whereas LD5 is primarily located in liver and skeletal muscle. By determining the specific isoenzyme increased in serum, the source of the cellular injury can be identified.
3 What determines the level of a liver enzyme in plasma?
Multiple factors are involved in determining the activity of an enzyme in plasma and include the following:
4 Do liver enzyme tests evaluate liver function?
Enzyme tests provide information about hepatocellular injury or cholestasis but do not define how much functional liver is present. Therefore, specific liver function tests are needed to assess liver function.
5 What are the most clinically useful leakage enzymes?
Clinically useful leakage enzymes vary with the species tested. In dogs and cats, ALT is used as a liver-specific leakage enzyme. Horses, ruminants, and pigs have very little ALT in their liver, so serum ALT activity does not increase with liver disease; therefore SDH is used as a liver-specific leakage enzyme in these species. AST is used as a nonspecific indicator of tissue injury in all the species just listed.
6 How long do leakage enzymes remain elevated in blood of dogs and cats after transient hepatocellular injury?
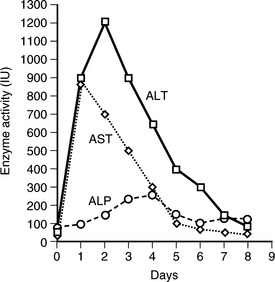
ALT remains elevated for 1 to 3 weeks after a toxic insult in dogs and cats. AST remains elevated for 5 to 7 days (Figure 30-1).
7 How long do leakage enzymes remain elevated in blood of horses and ruminants after transient hepatocellular injury?
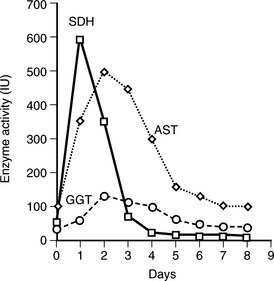
SDH remains high for only 3 to 4 days. AST stays elevated for 1 to 2 weeks (Figure 30-2).
8 Does the magnitude of increase in leakage enzymes predict the number of hepatocytes injured?
In general, the higher the activity in the serum, the greater is the number of hepatocytes injured.
9 Do leakage enzymes predict whether liver injury is reversible?
Leakage enzymes do not predict reversible liver injury. Acute reversible toxic or traumatic hepatocellular injury can result in very high serum activities of leakage enzymes, whereas chronic fibrotic liver disease, which is irreversible, may result in only slight increases in leakage enzymes.
10 Are there other factors in addition to hepatocellular injury that increase serum enzyme activities?
A mild to moderate increase in ALT can result from enzyme induction associated with administration of drugs such as anticonvulsants, thiacetarsemide, and glucocorticoids. AST is present in most tissue and increases with muscle injury as well as hepatocelluar injury. AST is also present in kidney, pancreas, and erythrocytes. Artifactual hemolysis of blood samples may result in increased serum ALT activity.
11 Is alkaline phosphatase a liver-specific cholestatic enzyme?
ALP is not liver specific. ALP isoenzymes are present in all tissues with high activity in liver, bone, kidney, intestine, and placenta. The renal, intestinal, and placental isoenzymes have a very short half-life in the plasma (<6 minutes) and thus do not result in a significant increase in plasma activity. The bone isoenzyme is produced by osteoblasts. Young growing animals may have up to three times the normal adult serum activity. Serum activity may increase two to four times as a result of diseases that increase osteoblastic activity. Anticonvulsant may increase serum activity by two to six times. Corticosteroids induce a marked increase in a specific hepatic isoenzyme in dogs. The steroid isoenzyme increases within 6 days after exposure to endogenous or exogenous corticosteroids and can reach very high levels by 3 to 4 weeks. Serum ALP activity decreases slowly over weeks to months after cessation of exposure to corticosteroids.
12 How can the steroid-induced isoenzyme in the dog be differentiated from the liver isoenzyme?
The liver isoenzyme activity is greater than 90% inhibited by addition of levamisole, whereas the steroid-induced isoenzyme is relatively resistant to addition of levamisole. GGT is also present on some chemistry profiles. Because GGT is more liver specific than ALP, an increase in GGT in addition to ALP is consistent with cholestatic liver disease.
13 Is γ-glutamyltransferase only present in the liver?
GGT is present in most tissue, but the greatest activity is in the canalicular surface of hepatocytes, in bile duct epithelium, and in renal tubules. Serum activity of GGT is primarily of hepatic origin.
14 Is γ-glutamyltransferase a better cholestatic enzyme than alkaline phosphatase?
Whether GGT or ALP is the better cholestatic enzyme depends on the species. Dog liver contains about three times more ALP than cat liver, and cat ALP has a shorter serum half-life. Cholestatic liver disease thus results in much lower serum ALP activity in cats than in dogs. Therefore, ALP is a more sensitive test for cholestatic liver disease than GGT in the dog, whereas ALP and GGT have approximately equal sensitivity in the cat. The cow, sheep, pig, and horse have greater liver and serum GGT activity than the dog and cat. Also, ALP has a broad reference range in cattle, pigs, sheep, and horses, so GGT is a better test for liver disease in these species. In horses, cattle, and sheep, GGT may increase in acute hepatocellular injury as well as in cholestatic liver disease.
15 Do liver enzyme tests predict pathologic changes in the liver?
Enzyme tests predict liver changes only in very general terms. Serum leakage and cholestatic enzyme tests are sensitive indicators of hepatocellular injury and cholestatic disease, respectively. However, serum activities in a variety of diseases overlap and cannot be differentiated. In most cases, acute hepatocellular injury or necrosis can be differentiated from chronic fibrotic disease. In acute hepatocellular injury in dogs and cats, ALT and AST may increase 50 to 100 times the mean reference interval. A slight to moderate increase in ALP and little to no increase in GGT occurs. In acute diffuse hepatocellular injury in ruminant and horses, a marked increase in SDH and AST and a variable (mild to marked) increase in GGT occur. Alternatively, in extrahepatic bile duct obstruction or in chronic fibrotic liver disease, ALP and GGT are greatly increased, and leakage enzymes are only mildly increased. However, responses vary among the species. In cholestatic liver disease the magnitude of increase in ALP is marked for dogs, significantly less for cats and horses, and variable in ruminants. In my experience, attempting to predict other types of liver pathology solely on the serum enzyme activities is unreliable.
16 What defines a test of liver function?
A liver function test is a test that evaluates the capacity of the liver to perform a single function or multiple functions that are unique to the liver.
17 Can liver dysfunction be reversible?
Liver dysfunction can be reversed, particularly in acute hepatic disease. In acute toxic or traumatic liver disease, swelling or altered cellular metabolism may result in transient hepatocellular dysfunction. The liver also has the capacity to regenerate destroyed hepatocytes and thus regain functional mass. In chronic hepatitis, liver failure results from a combination of the opposing effects of hepatocellular destruction and liver regeneration. By the time liver dysfunction is detected, the destructive effects have overcome the regenerative effects. This condition is usually irreversible.
18 What are common liver function tests?
Because the liver has a very large number of unique metabolic functions, multiple liver function tests have been described. These tests can be subclassified as follows:
19 Do liver function tests vary in sensitivity?
Liver function tests vary greatly in their sensitivity in detecting liver dysfunction. The exact sensitivity of liver function tests has not been well defined and may vary among species. The most sensitive liver function test appears to be bile acids, which can detect 40% to 50% liver dysfunction. The BSP and ammonia tolerance tests are somewhat less sensitive. Liver function test included in serum chemistry profiles (e.g., total bilirubin, albumin) are very insensitive liver function tests, requiring 80% to 90% liver dysfunction before results are beyond reference limits.
20 What is the best liver function test for routine use?
Serum albumin and total bilirubin are the least expensive and easiest tests to perform but have significant limitations. Both are very insensitive tests and are not liver specific. Of the other tests, the serum bile acids test is the easiest to perform, the most sensitive, and liver specific. Therefore the bile acids test has largely replaced BSP and ammonia tolerance tests.
21 How is the serum bile acids test performed?
In dogs and cats, fasting (8-12 hours) and/or 2-hour postprandial blood samples are drawn. During fasting, serum concentrations are lower because bile acids are removed from the blood and stored in the gallbladder. Feeding induces gallbladder contraction, which releases bile acids into the intestine. As bile acids are reabsorbed through the portal circulation, most are removed by the liver, but some enter the peripheral circulation. The 2-hour postprandial bile acids test increases the sensitivity of the test. It is particularly useful in detecting portosystemic venous shunts because fasting serum concentrations may be normal.
22 How does hepatocellular dysfunction result in increased bile acids in serum?
Reduced functional liver mass or shunting of portal blood away from the liver increases the number of bile acids that bypass the liver and enter the peripheral circulation. Also, intrahepatic bile duct obstruction, which is associated with liver disease, causes regurgitation of bile acids into the peripheral blood.
23 How do BSP and ammonia tolerance compare to bile acids as tests of liver function?
Both the BSP test and the ammonia tolerance test are more difficult to perform and have equal or lesser sensitivity for detection of liver dysfunction than the bile acids test. Therefore the bile acids test has largely replaced BSP and ammonia tolerance tests.
24 Is hyperbilirubinemia (icterus) a specific indicator of liver disease?
Hyperbilirubinemia can be caused by prehepatic, hepatic, or posthepatic causes; icterus is not a specific indicator for liver disease. Prehepatic causes are associated with hemolytic anemias. In animals with prehepatic icterus, hyperbilirubinemia is usually associated with a moderate to severe anemia, whereas the anemia associated with chronic liver disease is usually mild. Hepatic icterus is associated with diffuse hepatocellular swelling, severe acute hepatitis, and end-stage chronic hepatitis. Icterus is frequently accompanied by hepatic enzymopathy and low serum albumin. Posthepatic icterus results from extrahepatic bile duct obstruction.
25 In addition to determination of total bilirubin, is determination of direct (conjugated) and indirect (unconjugated) bilirubin useful in differentiating the cause of icterus?
In hemolytic anemias, unconjugated bilirubin is formed from the breakdown of heme. As a result, bilirubin in the blood is expected to be primarily unconjugated. In liver disease or extrahepatic bile duct obstruction, unconjugated bilirubin is conjugated by the remaining functional hepatocytes, but the conjugated bilirubin is regurgitated into the blood as a result of bile duct obstruction. Therefore the increase in serum bilirubin with liver disease and bile duct obstruction would be expected to be primarily conjugated. However, the relative concentrations of conjugated and unconjugated bilirubin in prehepatic and hepatic icterus frequently do not fit the expected patterns. Because of a high incidence of confusing results and the availability of other tests to differentiate hemolytic disease reliably from hepatic disease, conjugated and unconjugated bilirubin determinations are infrequently performed.
26 Are there species differences in bilirubin metabolism?
Dogs have a low renal threshold for bilirubin, and bilirubinuria frequently occurs without hyperbilirubinemia. Horses frequently become icteric, and bilirubin is primarily unconjugated despite the cause of hyperbilirubinemia. Unconjugated bilirubin is predominant in horses probably because uptake of bilirubin by the liver is the limiting step in bilirubin metabolism. With anorexia, uptake is reduced further, which can result in marked increases (5-10 mg/dl) in serum bilirubin concentration. Hyperbilirubinemia occurs infrequently in ruminants. When present, the magnitude of increase is slight and the cause more frequently related to hemolytic than hepatic disease.
27 What type of changes in serum proteins is expected in liver disease?
Hypoalbuminemia is an extremely late change in chronic liver failure. Hyperglobulinemia is a frequent finding in chronic liver disease and may result from increased numbers of enteric antigens bypassing the liver and entering the peripheral blood. A chronic immune response to these antigens results in a polyclonal gammopathy.