Chapter 27 Treatment decision making in the medical encounter
the case of shared decision making
INTRODUCTION
Over the last two decades, there has been increasing interest among health researchers, clinicians and ethicists in the general topic of treatment decision making between patients and physicians and, more recently, in shared treatment decision making in particular. In this chapter we describe some of the reasons for this interest, the meaning of shared decision making, physician attitudes towards shared decision making and the development and use of decision aids to promote shared decision making. In doing so, we draw heavily on our own conceptual and empirical research on the topic of shared treatment decision making conducted over a period of more than 10 years (Charles et al 1997, 1999a).
THE DEVELOPMENT OF THE SHARED DECISION MAKING APPROACH
Prior to the 1980s, the most prevalent approach to treatment decision making in North America was paternalistic, with the physician assuming the dominant role in the medical encounter (Levine et al 1992). Underlying this deference to professional authority were a number of assumptions (Charles et al 1999a). The first was that for most illnesses, a single best treatment existed and that clinical expertise and experience provided the basis for making the ‘right’ decision. Second, physicians were assumed to consistently and uniformly apply this clinical judgement when selecting treatments for their patients. Third, because of their expertise, physicians were assumed to be in the best position to evaluate treatment benefits and risks for the patient. Finally, professional ethics enjoined physicians to put the patient’s welfare first – a kind of ‘doctor knows best’ mentality.
After 1980, these assumptions began to break down. It became apparent that for an increasing number of illnesses there was no one best treatment, and a more complex decisional context emerged wherein different treatments (including the ‘do nothing’ option) had different types of trade-off between benefits and risks. Because the patient had to live with the consequences, the assumption that physicians were in the best position to evaluate these trade-offs for the patient was increasingly challenged (Eddy 1990, Levine et al 1992, Lomas & Lavis 1996). Moreover, the burgeoning literature in North America on small area variations in medical practice was beginning to show consistent evidence that physician treatments for the same disease often varied considerably across small geographic areas, and that these variations were unrelated to differences in the health status of the respective populations (Chassin et al 1986, 1987; Roos et al 1988; Wennberg et al 1987). These findings called into question the precision of medical practice, including the assumption that physicians uniformly provided the best treatment to patients with a similar disease.
Two other system level trends also cast a negative light on the autonomy of physicians in clinical practice. The first was concern over rising healthcare costs which raised the issue of accountability of physicians to patients, governments and, in the case of the US, to third party payers for clinical decisions (Katz et al 1997). The second and even more direct influence was the rise of consumerism and consumer/patient sovereignty (Charles & DeMaio 1993; Haug & Lavin 1981, 1983) in particular, as manifested in new government legislation safeguarding the rights of patients to be informed about all available treatment options (Nayfield et al 1994) and in the growing interest among many individuals and groups (e.g. physicians, patients and ethicists) to develop and advocate new approaches to treatment decision making which would incorporate a greater role for patients in this process (Gafni et al 1998).
As a result of these and other trends, the appropriateness of the paternalistic model of treatment decision making began to be questioned, and other models, such as the informed and shared approaches, were identified and advocated as potentially preferred options for treatment decision making (Charles et al 1997, 1999a; Gafni et al 1998). One major problem with this emerging literature, however, was that these concepts themselves were not clearly defined; the same words (for example ‘shared decision making’) were used to mean different things, and different labels (such as ‘informed’, ‘shared’) were used without clear distinctions in their application. Thus, while more patient involvement in treatment decision making was being advocated, it was not clear exactly what this meant or how it could be implemented. To shed light on these issues we wrote two papers in the late 1990s (Charles et al 1997, 1999a) attempting to clarify the meaning of shared decision making, to define the key components of this approach and to compare them with those of the informed and paternalistic models of treatment decision making.
THE MEANING OF SHARED DECISION MAKING
Both the informed and the shared decision making models were developed to compensate for alleged flaws in the paternalistic approach. These three models are the most widely discussed in the literature on treatment decision making. The different stages of the treatment decision making process in general are identified in Table 27.1. These stages are: information exchange, deliberation about treatment options and deciding on the treatment to implement (Charles et al 1999a). We have identified these as distinct stages, although in reality they may occur together or in an iterative process. Table 27.1 identifies the ‘ideal type’ roles that both physicians and patients play at each decision-making stage and how these differ by decision-making approach.
Table 27.1 Treatment decision making approaches (Charles et al 1999b)
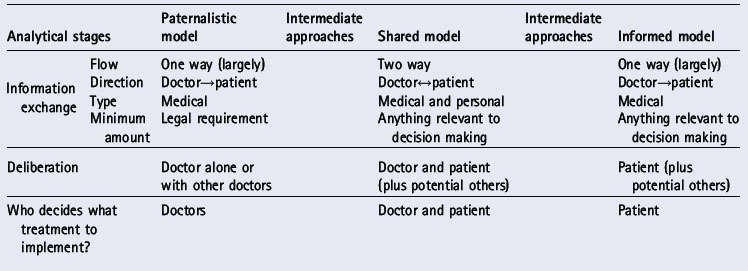
INFORMATION EXCHANGE
Information exchange refers to the type and amount of information exchanged between physician and patient and whether information flow is one way or two way. In the paternalistic model, the exchange is largely one way and the direction is from physician to patient. At a minimum, the physician must provide the patient with legally required information on treatment options and obtain informed consent to the treatment recommended. The patient is depicted in this model as a passive recipient of whatever amount and type of information the physician chooses to reveal. In general, this model assumes that the physician knows best and will make the best decision for the patient, without necessarily requiring any patient input.
In an informed model, information exchange is one way, from physician to patient. The physician is assumed to be the primary source of information for the patient on medical/scientific information about the disease and the treatment options. Beyond information transfer, the physician has no further role in the decision-making process. The tasks of deliberation and decision making are the patient’s alone.
In a shared decision-making process, information sharing is a two way process. At a minimum, the physician must inform the patient of all the relevant information about available treatment options, the benefits and risks of each and the potential effects of these on the patient. However, for a meaningful deliberation and agreement on the treatment of choice, the physician should describe his/her preferences, values and beliefs. The patient needs to provide the physician with information on her/his values, preferences, lifestyle and social context, beliefs and knowledge about the illness and its treatment. It is assumed in this model that both sets of information (technical/scientific) and subjective (values/preferences) are necessary to make the best treatment decision for any given patient.
DELIBERATION
The deliberation stage of decision making refers to the process of expressing and discussing treatment preferences. The minimum requirement as to which person/s are involved in the process varies across the three decision-making approaches. In the paternalistic approach, the physician weighs the benefits and risks of each option alone or in consultation with other physicians while the patient passively listens. In the extreme case of this model, the physician may verbally communicate to the patient only the ultimate treatment decision(s) selected, without soliciting patient input or describing the rationale for that decision.
In the informed model, the physician’s role is limited to information transfer, that is, providing the patient with information about the relevant treatment information and the risks and benefits of each. The patient alone or with input from friends and family undertakes the deliberation process to arrive at an informed decision reflecting personal values and preferences. Underlying this approach are two key assumptions. The first is that information is both necessary and sufficient to enable the patient to make the best decision. The second is that the physician should not have an investment in the decision-making process or the decision made. In other words, patient sovereignty reigns in this approach, with the physician providing technical input only, in the form of relevant scientific information.
In a shared approach both patient and physician deliberate about treatment options in an interactive process where it is assumed that both parties have a legitimate investment in the treatment process and outcome. This emphasis on interaction ensures patient input, but also makes the process potentially more cumbersome and time-consuming than the other approaches. In a shared process both parties need to be willing to engage with each other, exchanging both information and treatment preferences. The physician can legitimately give a treatment recommendation to patients and try to persuade them to accept the recommendation. However, physicians using this approach would also have to listen to patients and try to understand why they might prefer a different option. If no agreement can be reached, several possibilities can occur. The physician would need to decide whether to endorse a particular patient’s choice as part of a negotiated agreement in which patients’ views count, or whether the strength of the physician’s own views precluded agreement with any other treatment option. The patient would need to decide whether to stay with this physician or to seek advice elsewhere.
DECIDING ON THE TREATMENT TO IMPLEMENT
The final stage in the treatment decision-making process is choosing a treatment to implement. In the paternalistic and informed models, the decision maker is one person; in the first case it is the physician and in the second, the patient. However, neither party is totally autonomous because each faces constraints in implementing the decision. The physician must have the patient’s informed consent prior to giving the treatment, and the patient needs authorization from a physician to receive the preferred treatment.
In the shared approach both parties, through the process of deliberation, work towards reaching an agreement that both can live with. As noted above, if agreement cannot be reached the process may terminate at this point unless one party can be persuaded to adopt the other’s preferred option.
The different approaches described above are ‘ideal types’ in the sense that the role depictions for physician and patient in each model are defined as invariant, predictable and distinct from one another. In reality the boundaries around the role behaviour of physicians and patients in each model are rarely so clear-cut. There are various ‘in-between’ approaches to treatment decision making which do not conform precisely to one of the ideal types but rather lie somewhere in-between and may be characterized as shades of grey. For example, starting with the paternalistic model, the more that each stage moves from a physician-dominated encounter to one where the patient’s input is recognized, nourished and valued, the more the model evolves into a shared approach. In fact the majority of physician–patient treatment decision-making processes are likely to reflect some form of in-between approach rather than a pure type.
Even in a single interaction, the decision-making approach used at the beginning of the discussion may evolve into one of the other approaches as the consultation progresses. It should be noted that we have described only the most simple type of interaction, that between one patient and one physician. We have done this to keep our analysis as clear as possible, but we recognize that many decision-making processes involve multiple participants and can take place over time, greatly complicating the process and allowing for the development of coalitions around treatment preferences. Nonetheless, the framework provides an analytic tool for articulating the different stages in treatment decision making, identifying the defining characteristics of the paternalistic, informed and shared approaches to undertaking this task and clarifying the differences between them.
PHYSICIANS’ ATTITUDES TOWARDS SHARED TREATMENT DECISION MAKING
The conceptual model of shared decision making referred to here was developed over several years (Charles et al 1997, 1999a). Since then, a citation analysis undertaken by Makoul & Clayman in 2006 suggests widespread dissemination of and references to this model in the international treatment decision-making literature. Our goal in developing this model was to focus on treatment decision making for serious (potentially life-threatening) illnesses, where several treatment options exist with different possible outcomes and substantial uncertainty, where there is often no right or wrong answer, and where treatments vary in their impact on the patient’s physical and psychological well-being (Charles et al 1999b).
Early on, we also wanted to know how well this model resonated with practising physicians and in particular the extent to which the role expectations that we defined for patients and physicians in a shared approach coincided with those that physicians themselves would define as generic to each approach. As a first empirical exploration we decided to undertake a cross-sectional survey of all Ontario-based medical and radiation oncologists and surgeons treating women with early stage breast cancer as an example of a decision-making context to which our model could be applied. Our goal was to assess the degree of congruence in the meaning of shared decision making as defined in our conceptual model and as perceived by these physicians (Charles et al 2004).
Of 322 eligible surgeons, 232 (72.0%) completed and returned our questionnaire. One hundred and two (78.5%) of the 130 eligible oncologists responded. In the questionnaire we included four clinical treatment decision-making examples or scenarios in which the roles of the patient and the physician were systematically varied. We then asked physicians to read each scenario (see Box 27.1) and identify which one(s) they thought reflected a shared approach to treatment decision making.
Box 27.1 Decision-making examples
Example 1
After looking at your medical records and examining you the doctor presents a treatment that he/she thinks is best for you. The doctor gives you information about the treatment including the risks and benefits. You accept the treatment that the doctor recommends.
Example 2
After looking at your medical records and examining you the doctor presents you with the treatment choices. Information about the risks and benefits of each choice is given and discussed with you. You ask questions and obtain all the information you want from the doctor. The doctor recommends a treatment that you accept.
Example 3
After looking at your medical records and examining you the doctor presents you with the treatment choices. Information about the risks and benefits of each choice is given and discussed with you. The doctor asks you to decide on a treatment and states that you are the best person to make the decision. You decide and inform the doctor of the treatment you prefer.
Example 4
After looking at your medical records and examining you the doctor presents you with treatment choices. Information about the risks and benefits of each choice is given and discussed with you. You ask questions and obtain all the information you want from the doctor. The doctor asks you about your preferences for treatment given your lifestyle and the issues that are important to you. Together you decide on the treatment that is best suited to you.
Example 1 was constructed as a paternalistic approach in which the physician dominated the process. In example 2, information was shared between patient and physician but the physician alone made the treatment decision (what we call some sharing). In example 3, the physician provided information to the patient but the latter was the sole decision maker (what we call an informed approach). In example 4, the patient and physician both participated in all phases of the decision-making process and together negotiated a treatment to implement (what we call a pure shared approach). None of the examples had any labels attached so physicians were unaware that we had deliberately constructed each scenario to represent a particular type of decision-making approach.
The study results indicated that over 95% of both oncologists and surgeons felt that a shared decision-making approach was illustrated in at least one of our clinical scenarios. Few physicians (less than 5%) described example 1, the paternalistic scenario, as shared. Example two, constructed to reflect a two way sharing of information but a single decision maker (physician) was identified as a shared approach by approximately 28% and 34% of surgeons and oncologists respectively. Example 3, constructed to reflect a two-way sharing of information but with a single patient decision maker, was considered shared by 27% of surgeons and 21% of oncologists. Example 4, illustrating a pure shared approach as defined in our model, was identified as shared by 94% of surgeons and 87% of oncologists.
From these results, we concluded that substantial congruence was found between the meaning of shared decision making as defined in our conceptual model and as perceived by study physicians. In recent years we have worked with physicians in other clinical areas such as general practice and diabetes to further refine our model and modify certain aspects of it to fit the respective decision-making contexts of different clinical areas (Montori et al 2006, Murray et al 2006).
The model can be used as a conceptual tool to guide research, compare different treatment decision-making approaches, clarify the meaning of shared decision making and enhance its translation into practice.
In the same study referred to above (Charles et al 2004), we also asked Ontario surgeons and oncologists the extent to which they practised shared decision making with their patients, their comfort level with this approach and perceived barriers and facilitators to implementation. More physicians from each specialty (89% of surgeons and 87% of oncologists) reported high comfort levels with example 4 (the pure shared approach) than with any other of the examples presented. Similarly, more surgeons and oncologists reported that their usual approach to treatment decision making was like example 4 than any other example presented (69% of surgeons and 56% of oncologists). Interestingly, reported comfort levels with example 4 were 20% higher for surgeons and 31% higher for oncologists than their reported use of this approach.
Physicians identified numerous barriers to implementing shared decision making, including lack of time, patient anxiety, patient lack of information or misinformation, and patient unwillingness or inability to participate. The latter barrier could be attributed to many factors, for example patient lack of interest and/or limitations in personal capacity, which in turn may be influenced by social circumstances, patient understanding, and the physician’s skill/ability to inform patients of relevant treatment options and to create an environment conducive to shared decision making. Many of the above factors and others (including physician factors) have also been identified in other studies as barriers to implementing a shared approach (Gwyn & Elwyn 1999, Stevenson et al 2000).
Based on the above results, it seems that there is still much to do to create clinical practice environments that are conducive to shared decision making between physicians and patients. Moreover, despite attempts like ours and those of others (Deber 1994, Edwards & Elwyn 2001) to clarify the meaning of shared decision making, there are still different perspectives on what it means and entails at the practice level.
THE DEVELOPMENT AND USE OF TREATMENT DECISION AIDS
There has been increasing interest and activity in the development, use and evaluation of decision aids as instruments or tools to assist patients to participate in the treatment decision-making process with their physicians (Whelan et al 2002). Whereas some studies (Whelan et al 2002, 2003, 2004) have shown that such tools improve patients’ knowledge and comfort with decision making, others have not (Goel et al 2001). The term decision aid is a general term applied to a broad array of different tools. Such tools are generally intended to help patients by providing them with evidence-based information about relevant treatment options and their risks and benefits, to structure the decision-making process in what the designers hope will be a useful and logical way, and to encourage patients to think about their treatment preferences and participate in the decision-making process. The number, types, formats, purposes and clinical contexts of their use have proliferated over the last 10 years (Charles et al 2005). Some of the more common formats include decision boards, interactive videos, pen and paper exercises, and coaching exercises to help patients interact with their physicians.
The development of decision aids, mostly by university academics or professional associations, has been so prolific that this growth has outpaced our ability to evaluate the rigor and success of such tools in achieving their stated goals. To help remedy this situation an international collaboration of scholars in different countries has been assembled to develop critical appraisal criteria (primarily methodological) for evaluating such aids, but this movement is still in its infancy (O’Connor et al 2005).
Methodological issues are not the only issues that need to be carefully thought through when developing and using various forms of decision aids. Other issues include, for example, the degree of fit of different aids with a variety of clinical and cultural contexts. Currently tool developers seem to commonly assume that a single type of aid will fit multiple contexts without the need for modification (a kind of ‘one size fits all’ mentality), but in reality this is unlikely to be the case. In addition, the theoretical underpinnings and assumptions underlying the development of such interventions often vary, may not be made explicit, or may be absent completely as a foundation on which applied decision-making tools are built. This is a little like putting the cart before the horse. No matter how rigorous the methodological steps are in developing a decision aid, the resultant tool will still be flawed if not guided initially by a clear statement about the goals of the instrument and a conceptual foundation of hypotheses and assumptions about the mechanisms to incorporate in the tool that are intended to produce the desired results. Unless these analytical processes are made clear, it is difficult for others to judge the thinking behind the development process or the extent to which such tools will resonate with physicians and patients. Finally, it is sometimes the case in this field of research that what we can measure drives what we should measure. However, more appropriately, desired goals of decision aids should drive measurement and not the other way round (Charles et al 2005).
One of the more recent developments in the design of decision aids is the attempt to structure into these tools exercises intended to help patients clarify their values (O’Connor et al 1999). Such exercises typically involve asking patients to evaluate the importance of various potential treatment risks and benefits and then to make, either implicitly or explicitly, trade-offs among them (for example, the importance of body image versus survival) to come up with a preferred decision. There are many variations on this exercise. The goal is to help patients assess whether the treatment decision they are leaning towards is consistent with the priorities they have identified in the exercise. This type of exercise assumes that undertaking a preference-based trade-off is the best method for determining individual treatment decisions.
There are several problems with such exercises that need to be addressed (Charles et al 2005). First, it is often not clear what the designers of such exercises mean by the concept of values and their various types and levels. Second, the alleged need for such exercises assumes that patients on their own do not know and cannot articulate their own values related to the desirability or undesirability of various treatments and need help to identify and weigh these. It is further assumed that all patients use a similar (universalistic) method to weigh the benefits and risks of various treatments, an assumption that may or may not be true. We wonder whether it is even possible to construct a valid test to assess the superiority of an explicit values clarification exercise over implicit methods. To do so would require that we first know what the patient’s true values are so that we could use them as a ‘gold standard’ by which to judge which approach resulted in a treatment decision most congruent with these values. But if we knew what the patient’s true values were in the first place, we would not need any explicit exercise to help the patient define them. Finally, the exercise of helping patients clarify their values may act inadvertently as an intervention, changing patients’ values through the exercise itself. Thus, while laudable in intent, many current values clarification exercises are fraught with difficulties and assumptions that require further evaluation.
CONCLUSION
There are a number of outstanding issues, both conceptual and empirical, that require further investigation in the field of shared decision making between physicians and patients. These include: the definition, types and levels of patient values to be considered when attempting to help patients clarify their preferences for different treatment decisions; the definition and influence of culture on patients’ preferences for decision-making processes and outcomes (Charles et al 2006); the fact that measurement activities often drive goal-setting activities for decision aids rather than the other way round; and the lack of precision in the stated rationale for and meaning of various goals suggested for decision aids, and for mechanisms through which they are intended to have an impact.
Further exploration of the above issues will enhance the development of practical tools to support shared decision making between clinicians and patients. We hope that this chapter has helped to inform readers of the many challenges facing researchers and clinicians working in this important field.
Charles CA, DeMaio S. Lay participation in health care decision-making: a conceptual framework. Journal of Health Policy and Law. 1993;18(4):881-904.
Charles C, Gafni A, Whelan T. Shared decision-making in the medical encounter: what does it mean? (or, it takes at least two to tango). Social Science and Medicine. 1997;44:681-692.
Charles C., Gafni A, Whelan T. Decision-making in the physician-patient encounter: revisiting the shared treatment decision-making model. Social Science and Medicine. 1999;49:651-661.
Charles C, Whelan T, Gafni A. What do we mean by partnership in making decisions about treatment? British Medical Journal. 1999;319:780-782.
Charles C, Gafni A, Whelan T. Self-reported use of shared decision-making among breast cancer specialists and perceived barriers and facilitators to implementing this approach. Health Expectations. 2004;7(4):338-348.
Charles C, Gafni A, Whelan T, et al. Treatment decision aids: conceptual issues and future directions. Health Expectations. 2005;8(2):114-125.
Charles C, Gafni A, Whelan T, et al. Cultural influences on the physician-patient encounter: the case of shared treatment decision-making. Patient Education and Counseling. 2006;63(3):262-267.
Chassin MR, Brook RH, Park RE. Variations in the use of medical and surgical services by the Medicare population. New England Journal of Medicine. 1986;314:285-290.
Chassin MR, Kosecoff J, Park RE, et al. Does inappropriate use explain geographic variations in the use of health services?: a study of three procedures. Journal of the American Medical Association. 1987;258:2533-2537.
Deber R. The patient–physician partnership: changing roles and the desire for information. Canadian Medical Association. 1994;151:171-176.
Eddy DM. Anatomy of a decision. Journal of the American Medical Association. 1990;263(3):441-443.
Edwards E, Elwyn G. Evidence-based patient choice. Oxford: Oxford University Press, 2001.
Gafni A, Charles CA, Whelan T. The physician–patient encounter: the physician as a perfect agent for the patient versus the informed treatment decision-making model. Social Science and Medicine. 1998;47:347-354.
Goel V, Sawka CA, Thiel EC, et al. Randomized trial of a patient decision aid for choice of surgical treatment for breast cancer. Medical Decision Making. 2001;21(1):1-6.
Gwyn R, Elwyn G. When is a shared decision not (quite) a shared decision? Negotiating preferences in a general practice encounter. Social Science and Medicine. 1999;49:437-447.
Haug MR, Lavin B. Practitioner or patient – who’s in charge? Journal of Health and Social Behaviour. 1981;22:212-229.
Haug M, Lavin B. Consumerism in medicine. Beverly Hills, CA: Sage, 1983.
Katz SJ, Lomas J, Charles C, et al. Physician relations in Canada: shooting inward as the circle closes. Journal of Health Policy and Law. 1997;22(6):1413-1431.
Levine MN, Gafni A, Markham B, et al. A bedside decision instrument to elicit a patient’s preference concerning adjuvant chemotherapy for breast cancer. Annals of Internal Medicine. 1992;117:53-58.
Lomas J, Lavis J. Guidelines in the midst. Hamilton, Ontario: McMaster University, 1996. CHEPA Working Paper #96-23, Centre for Health Economics and Policy Analysis
Makoul G, Clayman ML. An integrative model of shared decision making in medical encounters. Patient Education and Counseling. 2006;60(3):301-312.
Montori V, Gafni A, Charles C. A shared treatment decision-making approach between patients with chronic conditions and their clinicians: the case of diabetes. Health Expectations. 2006;9(1):25-36.
Murray E, Charles C, Gafni A. Clinical decision making in U.K. primary care. Patient Education and Counseling. 2006;62:205-211.
Nayfield SG, Bongiovanni GC, Alciati MH, et al. Statutory requirements for disclosure of breast cancer treatment alternatives. Journal of the National Cancer Institute. 1994;86:1202-1208.
O’Connor A, Wells G, Tugwell P, et al. The effects of an explicit values clarification exercise in a women’s decision aid regarding postmenopausal hormone therapy. Health Expectations. 1999;2:21-32.
O’Connor A, Llewellyn-Thomas H, Stacey D. International patient decision aid standards collaboration (IPDAS) collaboration background document. Online. Available: http://ipdas.ohri.ca/IPDAS_Background.pdf, 2005. 27 Feb 2005
Roos NP, Wennberg JE, McPherson K. Using diagnosis-related groups for studying variations in hospital admissions. Health Care Finance and Review. 1988;9:53-62.
Stevenson FA, Barry CA, Britten N, et al. Doctor–patient communication about drugs: the evidence for share decision-making. Social Science and Medicine. 2000;50:829-840.
Wennberg JE, Freeman JL, Culp WJ. Are hospital services rationed in New Haven or over-utilized in Boston? Lancet. 1987;329(8543):1185-1189.
Whelan T, O’Brien MA, Villasis-Keever M, et al. Impact of cancer-related decision aids. Evidence Report/Technology Assessment (Summary). 2002;46:1-4.
Whelan TJ, Sawka C, Levine M, et al. Helping patients make informed choices: a randomized trial of a decision aid for adjuvant chemotherapy in lymph node-negative breast cancer. Journal of the National Cancer Institute. 2003;95:581-587.
Whelan T, Levine M, Willan A, et al. Effect of a decision aid on knowledge and treatment decision-making for breast cancer surgery: a randomized trial. JAMA. 2004;292(4):435-441.