Patient Safety and Risk Management
Ellen K. Murphy
Think about what happens in the perioperative setting. Patients' natural pain, communication, and reflex and infection defenses are purposefully diminished or obliterated. Their bodies are positioned on very firm flat surfaces and in unnatural positions. Then their bodies are further traumatized with instruments, fibers, drugs, and other foreign materials. It is no wonder that more than one quarter of the reported sentinel events from 2006 through 2012 occurred in the perioperative setting (Table 2-1).
TABLE 2-1
Summary Data of Perioperative Sentinel Events Reviewed by The Joint Commission
| Type of Sentinel Event | Number of Events from 2004 to 2012 |
| Anesthesia-related event | 94 |
| Fall | 538 |
| Fire | 98 |
| Infection-related event | 153 |
| Med equipment-related | 193 |
| Medication error | 378 |
| Op/postop complication | 719 |
| Transfusion error | 114 |
| Unintended retention of a foreign body* | 773 |
| Wrong-patient, wrong-site, wrong-procedure | 928 |
| All others† | 3006 |
| Total Incidents Reviewed | 6994 |
*Category added in 2005.
†Examples include criminal event, infant abduction, suicide, and perinatal death/injury.
Modified from www.jointcommission.org/assets/1/18/2004_4Q_2012_SE_Stats_Summary.pdf. Accessed February 11, 2013.
Although patient safety should be, and is, a paramount concern to nurses in all settings, nowhere is it more imperative than in the immediate perioperative period. Additionally, many elements of perioperative patient safety are coextensive with those of workplace safety (see Chapter 3).
This chapter describes the evolution of perioperative patient safety and the management of injury risks as a collective responsibility of the entire healthcare team.
History
Patient safety remains a primary concern for members of the surgical team. Primum non nocere or “First, do no harm,” is a long-standing imperative for physicians and nurses. Early operating room (OR) nursing textbooks written by OR nurses included counting sponges among nursing duties (e.g., Smith, 1924). Perioperative nursing textbooks and curricula continued to include substantial content on infection control, positioning, safe medication practices, and counts through World War II. The first edition of this very textbook by Edythe Alexander (1943) included content on asepsis, the importance of correct side surgery, proper blood handling, and proper tourniquet application. Alexander did not use the word “safety,” but she described the purpose of “perfecting every detail . . . to insure that patients . . . have every chance to overcome the disease or injury with which they are afflicted” (1943, p. 7).
After World War II, safety activities increased and became more formalized. The Joint Commission for the Accreditation of Hospitals (JCAH), now The Joint Commission, emerged. Nursing groups and publications increasingly emphasized their patient safety content. The American Nurses Association (ANA) published its Code for Nurses, which included provisions on patient safety and privacy, and the Association of Operating Room Nurses (now, the Association of periOperative Registered Nurses [AORN]) organized in the early 1950s. From the beginning, AORN's conferences and publications were replete with patient safety information. Its first conference in 1954 included programs on methods improvement, explosion prevention, bacteria destruction, the surgeon-nurse relationship, and positioning (Glass and Murphy, 2002). AORN published its first Standards of Operating Room Practice in 1974. Its Technical Standards soon followed, and by the 1980s it published and regularly updated its Recommended Practices (RPs). AORN soon became and remains the primary resource and a fundamental authority for evidence-based perioperative safety practices (e.g., the RP on sterile technique [AORN, 2013]).
Safety as an Individual Responsibility
Throughout the 1980s, healthcare authorities viewed professionals' errors and their effects on patient safety primarily as the individual practitioner's responsibility. The legal and professional licensure systems tended to reinforce this approach. Any finding of negligence required that at least one individual had failed to do what a similarly situated, reasonable, and prudent professional would have done under similar circumstances. Likewise, professional licensure related to the abilities and behavior of the individual licensee.
In 1991, Brennan, Leape, and others proposed that despite individual best efforts by professionals, mistakes continued and were, in fact, more common than previously thought (Brennan et al, 1991). Their findings, combined with James Reason's influential book Human Error (1990), spawned a plethora of new safety-related groups (Box 2-1), as well as fresh research and literature based on the role of systems, human factors, and their relationship to human error in healthcare. Researchers and authorities began to recognize human errors leading to patient injuries not so much as an individual's shortcoming, deserving of blame or punishment, but more as a result of system failures in patient care areas such as the perioperative setting. Similarly, Leape (1994), Cook and Woods (1994), and others urged that the legal and professional licensure systems' continued focus on individual error was misplaced if adverse patient events were to be effectively prevented. They urged an emphasis on transparent systems that required open reporting, investigation, innovation, and dissemination. The aviation and nuclear systems' parallel examination of human factors served as models for ideas that led to relative success in preventing injury attributable to human error. Communication and teamwork have been identified as key factors to promote patient safety.
In the first edition of this textbook, Alexander noted that “to get the best result from a surgical operation, the surgeon and his assistants, the operating room nurse and the entire staff must work as a team” (1943, p. 8). Despite AORN and its partner professional associations' emphasis on safety and teamwork, however, patient injuries caused by human error in perioperative settings continue (Pronovost and Colantuoni, 2009; TJC, 2012b).
Concentrated Emphasis on Systems
This earlier work took on urgency in 1999. The Institute of Medicine (IOM) landmark work, To Err Is Human, spurred major additional safety initiatives when it reported that at least 44,000, and perhaps as many as 98,000, patients died in U.S. hospitals every year as a result of preventable adverse events (IOM, 1999). Even the lower figure exceeded the number of deaths resulting from motor vehicle accidents, breast cancer, or acquired immunodeficiency syndrome (AIDS). The IOM report did not focus solely on perioperative errors. However, it did refer to Reason's (1990) theory that complex, tightly coupled systems are most prone to accidents, and specified the surgical suite, along with emergency departments and intensive care units, as examples of complex, tightly coupled systems. Moreover, when one considers the incredibly complex highly invasive surgical procedures and number of team members and disciplines involved, juxtaposed with anesthetized patients who are unable to detect pain or otherwise defend themselves, patient vulnerability and the consequent need for protection by all team members are strikingly apparent. More than 4000 surgical “never events” occurred each year between 1990 and 2010 in the United States, according to the findings of a retrospective study of national malpractice data (Johns Hopkins Malpractice Study, 2012).
Brennan and colleagues (1991) studied 30,121 randomly selected hospital records, found adverse events occurred in 3.7% of hospitalizations, and concluded that substandard care had caused substantial numbers of patient injuries. In part 2 of that study, Leape and colleagues (1991) found that nearly half (48%) of adverse events were associated with surgery. Their results were consistent with those of earlier investigators, who had found half of all hospital-based, potentially compensable events (i.e., injuries from substandard care) arose from treatment in the OR. The Joint Commission (TJC) findings from 2006 to 2012 (TJC, 2012b) are similarly consistent with those findings: Individual efforts of the best nurses, surgeons, and anesthesia providers, combined with a recognized need for teamwork, are not sufficient to prevent injury— and are especially insufficient in the perioperative setting.
This shift away from emphasis on individual responsibility to a broader systems approach that began in the 1990s has accelerated since 2000. Advances in surgical instrumentation and health information systems, as well as in social media and consumerism, have combined to alter the context of perioperative care dramatically. This change in context requires a new and more inclusive, global approach to safety.
New terms and phrases have joined the patient safety lexicon, such as “e-iatrogenesis,” “better systems for patient safety,” “shared decision making,” and C. diff. Checklists and tools for measuring safety practices have multiplied (e.g., the Institute for Healthcare Improvement (IHI) Global Trigger Tool for Measuring Adverse Events, Partnerships for Patients and IHI Improvement Blogs; the National Quality Forum's (NQF) Safe Practices Quality Positioning System; and National Priorities Partnerships to the Leapfrog Group's Health User Group Dashboards). Safety scholars have published influential treatises for the professions and the public that have raised everyone's awareness of checklist initiatives (e.g., Conley, 2011; Gawande, 2009).
Despite the impressive initial effectiveness of checklists (Haynes et al, 2009), barriers to their adoption (Fourcade et al, 2012) and a parallel need for flexibility, leadership, and teamwork different from current practice (Walker, 2012) have been identified. Various voluntary and governmental standards have so proliferated that matrices are published by still other groups to compare or provide crosswalks between them.
Major Association Safety Activities
TJC and TJC International, especially their National Patient Safety Goals (NPSGs), are excellent sources for safety information applicable to systems and facilities wherein invasive procedures take place. The AORN Perioperative Standards and Recommended Practices is the best source for information specific to perioperative nursing practices. Major government agencies that provide financial incentives and research resources for patient safety are also useful sources for safety information for facilities and individual professionals.
The Joint Commission
TJC has long been involved with quality and safety. It sharpened its systems-based safety focus in the mid-1990s when it established its Sentinel Event Policy. That policy first encouraged and then required self-reporting of medical errors and root cause analyses of them. Based on cumulative data, TJC published its first NPSGs in 2003. It then recognized the need for standardized methods of patient identification and established the Universal Protocol (discussed later in this chapter) in 2004. By 2005, the World Health Organization (WHO) formed the Collaborative Centre for Patient Safety Solutions, comprised of TJC and TJC International. TJC also is a founding member of the National Patient Safety Foundation (NPSF), collaborates with the National Quality Forum (NQF), and is an affiliate of Consumers Advancing Patient Safety (CAPS).
Sentinel Events.
TJC designated unexpected occurrences involving death or risk of serious physical or psychologic injuries as sentinel events. It chose the word “sentinel” to indicate that these events signal the need for immediate investigation and response through root cause analysis, a systematized process to identify variations in performance that cause or could cause a sentinel event. Suggested steps in such an analysis are briefly summarized in the Patient Safety box on page 19. Whereas TJC data collection focuses on sentinel events, it also recognizes the value of analyzing “close calls” to improve patient safety (Wu, 2011).
TJC categorizes errors reported to it and publishes their frequencies. Examples of perioperative care errors are those that are (1) related to anesthesia; (2) caused by medical equipment; (3) caused by medication error; (4) result in infection, fires, and transfusion reactions; (5) are operative or postoperative; or (6) give rise to unintended retained surgical items (RSIs) or wrong site/patient/procedure surgery. The summary of sentinel events published in 2013 reveals that of 6994 total incidents reviewed from 2004 through 2012, nearly 25% of all incidents reviewed most likely occurred in an operative setting (RSI, referred to by TJC as retained foreign object [RFO], and wrong site procedures). Add in the number of events that could have occurred in the perioperative setting (falls, transfusion and medication errors) and the percentage might reach closer to the 50% figure that Leape and Brennan found in the early 1990s.
National Patient Safety Goals.
Another TJC initiative relates to NPSGs for hospitals and office-based surgery, derived from reported sentinel events. NPSGs are reviewed, updated, or retired each year. See the Patient Safety box on page 19 for an overview of the 2013 NPSGs for hospitals and the Patient Safety box on page 20 for elements of performance for the NPSG related to labeling medications.
TJC also publishes NPSGs for office-based surgery. These are not excerpted here because the goals for patients in both settings are almost the same. Although initially surprising, this similarity is understandable. Even though patients in office-based surgery settings tend to be of healthier physical status classifications as set out by the American Society of Anesthesiologists (ASA) (see Chapter 5), one could argue that error prevention procedures in office-based surgery facilities nevertheless must be rigorous because these smaller facilities are less likely to have available the hospitals' wider array of emergency and corrective equipment and personnel. In addition, office-based and ambulatory perioperative staff provides most patient and family discharge education and preparation, unlike inpatient care settings (Ambulatory Surgery Considerations) (Patient and Family Education). Finally, causes of infection and patient defenses against infection do not differ based on the location of the surgical procedure.
Universal Protocol.
TJC introduced its required safety practice under the nomenclature, Universal Protocol, now incorporated into the NPSGs. Key features of the Universal Protocol, as codified in the NPSGs, are performing a preoperative verification process; marking the operative site; and conducting a time-out immediately before starting the procedure. A properly performed time-out includes information about the patient and procedure, as discussed in more detail later in this chapter.
U.S. Government Agencies
Centers for Medicare & Medicaid Services.
As part of the U.S. Department of Health and Human Services (DHHS), the Centers for Medicare & Medicaid Services (CMS) is the federal agency charged with administration (including regulations for payment) of the Medicare, Medicaid, and multiple state Children's Health Insurance Programs (CHIP), part of Medicaid. It also administers the Health Insurance Portability and Accountability Act (HIPAA) (discussed later) and several other health-related federal programs. Significant to patient safety is the decision by CMS to impose financial disincentives for selected unsafe patient care outcomes by refusing to pay for the extra cost of treatment to correct those outcomes. Conversely stated, the agency responsible for paying Medicare claims now provides a financial incentive for safe patient care.
Nonreimbursable claims most relevant to perioperative patient care include the following (CMS, 2012):
• Pressure ulcers, stages III and IV
• Surgical site infections (SSIs) after bariatric surgery, certain orthopedic procedures, and coronary artery bypass surgery (mediastinitis)
• Vascular catheter-associated infections
• Catheter-associated urinary tract infections (CAUTIs)
• Administration of incompatible blood
• Foreign objects unintentionally retained after surgery
• Wrong patient, wrong procedure, or wrong site surgeries
• Pulmonary emboli associated with knee and hip replacements
• High readmission rates to facility within 30 days of discharge
CMS regularly reviews and adds conditions pursuant to its federal rule-making process (e.g., it added specific types of readmissions within 30 days of discharge, effective 2013).
CMS notes that it does not consider these listed patient safety concerns more important than others. Rather, it has chosen the selected concerns to emphasize that the facilities deemed responsible now must bear directly otherwise avoidable financial costs of insufficient patient safety controls. Furthermore, CMS regulations prohibit passing these costs on to patients. Most private insurance companies have adopted similar provisions. Thus from a purely risk management standpoint, in addition to potential indirect costs arising from negligence awards and settlements (which can be insured against), facilities now bear the risk of direct, uninsurable, and potentially severe cost disincentives if they fail to avoid the listed conditions though the institution of safe practices.
Agency for Healthcare Research and Quality (AHRQ).
As one of the 12 agencies within DHHS, the AHRQ's mission is to improve the quality, safety, efficiency, and effectiveness of healthcare for all Americans. This agency is committed to improving care safety and quality by developing successful partnerships and generating the knowledge and tools required for long-term improvement. The central goal of its research is measurable improvements in healthcare in America. These measurable goals include improved quality of life and patient outcomes, lives saved, and value gained.
The overall focus of AHRQ activities is threefold:
• Safety and quality: reduce the risk of harm
• Effectiveness: improve healthcare outcomes by encouraging the use of evidence
This agency serves healthcare clinicians, facilities and systems, consumers, policymakers, purchasers and payers, and academic healthcare. AHRQ offers nurses and other providers extensive evidence-based resources related to patient safety on its website (ahrq.gov).
The World Health Organization
The United Nations (UN) created WHO to function as its health oversight and coordination authority for all UN member nations who in turn have joined WHO. In 2004, WHO launched the World Alliance on Patient Safety, by which it began to examine patient safety in acute as well as in primary care settings relevant to all WHO member nations. Its action initiatives include Clean Care is Safer Care and Safe Surgery Saves Lives. The focus of the Clean Care campaign is hand hygiene (also referenced in TJC's NPSGs); it resulted in release of a 2009 WHO guideline for surgical hand preparation (Evidence for Practice). Salient points in the guideline section titled Surgical Hand Preparation: State of the Art include discussion of the time required for preoperative hand antisepsis, encouragement of brushless hand scrubs, and review of antimicrobial hand scrub preparations.
WHO's Safe Surgery Saves Lives initiative led to publication of its Surgical Safety Checklist. Similar in content to TJC's Universal Protocol, the WHO checklist adds a third phase, the Sign Out that includes team reviews of outcomes and concerns to be included in the handover (the international term for “hand-off”) to postanesthesia recovery caregivers (Evidence for Practice). Haynes and colleagues (2009) hypothesized that implementation of the WHO Surgical Safety Checklist would reduce complications and deaths associated with surgery. They compared outcomes of surgical patients in eight countries representing widely diverse economic circumstances and patient populations, and found that a 1.5% prechecklist rate of death declined to 0.8% (p = 0.003) with use of the checklist. They also found inpatient complications occurred in 11% of patients before the checklist was implemented and in 7% after introduction of the checklist. They also confirmed tangible improvements in safety outcomes after implementation of the checklist (Haynes et al, 2009; 2011). Notable about WHO's entry into surgical patient safety is recognition that perioperative adverse events causing complications before, during, and after surgery is a public health problem worldwide.
The Association of periOperative Registered Nurses
For nearly 60 years AORN has addressed perioperative patient safety issues. It represents perioperative patients and perioperative nurses in multiple policy settings, collaborates with TJC, WHO, other nursing associations and surgical alliances, and many safety coalitions to formulate safety statements that directly affect perioperative patient care. AORN provides an array of standards, RPs, guidelines, publications, videos, and tool kits that specifically address patient safety from the perioperative team's point of view. Tool kits include subjects such as fire safety, correct site surgery, sharps safety, hand-off communications, safe patient handling, and cultural and human factors. RPs and tool kits are evidence based to the extent possible. These AORN undertakings aim to develop real-world strategies to implement perioperative patient care practices. Along with TJC and WHO recommendations, AORN recommendations should be reflected and adopted, to the extent possible, within institutional policies and procedures, and educational curricula.
Other Patient Safety Groups/Coalitions/Companies
Major professional associations representing other perioperative team members (e.g., American College of Surgeons [ACS], American Society of Anesthesiologists [ASA], American Association of Nurse Anesthetists [AANA], Association of Surgical Technologists [AST]) have also issued multiple position statements and clinical recommendations related to patient safety. Many of these are co-issued or mutually endorsed with AORN. This reflects the increasing systems approach to perioperative safety. Additionally, many coalitions with similar patient safety interests have emerged as not-for-profit entities to gather and disseminate data, guidelines, protocols, and goals. For-profit groups are also included in what now may fairly be called a “patient safety industry.” (A partial listing of additional patient safety groups is noted in Box 2-1.)
Perioperative Nursing Safety Issues
Communication and Teamwork
Communication underpins many patient safety issues. Implementation of both the Universal Protocol and the WHO checklist requires enhanced communication within a culture of teamwork (discussed later) (Research Highlight). Hand-off/handover protocols have joined traditional clinical written documentation records to improve communication further in perioperative settings. Research continues in the use of perioperative patient care checklists in a variety of settings and situations (Research Highlight).
Checklists and protocols alone, however, cannot enhance meaningful communication without an equal commitment to teamwork, trust, and respect. Ultimately, improved communication is imbedded in human factors, culture, and social systems, all of which are more complex than checklists, mnemonics, and acronyms (Bliss et al, 2012; de Vries et al, 2011).
Clinical Documentation
The written clinical record is the historical anchor of communicating perioperative patient information. Evidence suggests, however, that a written (or digital) record is inadequate as the sole perioperative communication tool. More enhanced communication initiatives such as safer surgery briefings and hand-offs now augment the written record. Nonetheless, whether documented on paper or digitally, the clinical record remains foundational in assuring a safer patient experience and provision of information to future care areas.
Facilities where operative and other invasive procedures occur maintain records of each operation that must comply with state and federal regulations as well as with accreditation requirements. Those operative records include preoperative diagnosis, surgery performed, a description of findings, specimens removed, postoperative diagnosis, and names of all individuals participating in intraoperative care. Additional key components include positioning and stabilizing devices, electrosurgical unit number and settings, medications, and evidence of ongoing assessment and additional actions taken (a sample intraoperative record is shown in Figure 2-1). The operative record is a permanent part of the patient's medical record. Note that nearly all components of perioperative clinical documentation relate directly or indirectly to patient safety and injury prevention.
Proper perioperative nursing documentation describes assessment, planning, and implementation of perioperative patient care reflecting individualization of care and evaluation of patient outcomes. Any and every unusual or significant incident pertinent to patient outcomes must be documented as well as all remediation efforts related to the patient's care. The facility's risk manager may require additional documentation. Only objective information directly related to the specific patient is included in the patient's record (e.g., it is inappropriate to record personal opinions or to describe circumstances surrounding an event except as they appear to affect the patient directly).
Thoughtfully designed perioperative nursing documentation tools include defined elements in a format to minimize time needed for the documentation process (e.g., checklists). Ideally, collaboration with preoperative, postanesthesia care unit (PACU), and postoperative nursing units will produce one documentation tool used across all areas, avoiding duplication of patient data by different nursing staff. Increasingly, settings where operative and other invasive procedures are performed use electronic records to enter and track patient care information. Coordination of the content included in the intraoperative record with that in the surgeon's and anesthesia provider's intraoperative content can reduce documentation time and provide a more integrated record that streamlines workflow and reduces documentation errors.
Documentation of perioperative patient care in the clinical record simultaneously serves risk management functions. Documentation requirements serve as reminders of actions needed to provide safe care, thus prompting risk reduction strategies and preventing injury. Information in the clinical record also enhances continuity of care, thus reducing future injury. If a patient injury does occur, documentation that preventive measures or other actions to mitigate risks were taken may lessen the likelihood of a successful lawsuit.
Hand-offs/Handovers
As noted, written documentation alone, however crucial, is insufficient to ensure patient safety as care responsibility passes from one team or individual caregiver to another. As they reviewed the evidence related to hand-offs, Friesen and colleagues (2008) defined the transfer of information and responsibility for care of the patient from one provider to another as a hand-off or (internationally) handover. Standardized approaches to hand-off communication further reduce risk for error.
TJC, AORN, and WHO uniformly recommend that time-outs (or “safer surgery briefings”), as well as pre- and postoperative hand-offs, be formalized. In healthcare settings, occasions for transfer-of-care processes, such as hand-offs, include nursing shift changes, temporary relief or coverage, nursing and physician hand-offs from one department to another, various other transfers of information in inpatient settings, and interhospital transfers. The purpose of hand-off communication and reports is to provide essential, up-to-date, and specific information about the patient. Standardized hand-off communication must include an opportunity to ask and respond to questions. For examples of strategies to assist in effective and efficient hand-off communication and reports, see the Patient Safety box on page 28.
Amato-Vealey and colleagues (2008) identified and further developed elements of effective hand-off communication for use at each perioperative hand-off stage, using the SBAR mnemonic (Situation, Background, Assessment, Recommendation). For identification of critical elements for hand-offs from preoperative to intraoperative, see Box 2-2; for hand-offs intraoperatively between scrub persons, see Box 2-3; and for hand-offs from intraoperative to PACU or another postanesthesia recovery area, see Box 2-4.
Like advancements in other segments of perioperative safety, communication, although improved, remains developmentally in process. Healy and associates (2012) argue that a new view of communication is needed to improve perioperative safety. They describe reengineering the system of communication for safety that includes not only immediate informational needs, but also an account of the broader ergonomic and human factors that shape improved communication (Shouhed et al, 2012).
Wrong Site, Person, or Procedure Surgery
Surgery that is the wrong procedure or surgery performed on the wrong site or person includes any operative or other invasive procedure performed on the wrong patient, wrong body part, wrong side of the body, or at the wrong level of the otherwise correctly identified anatomic structure, such as in spinal surgery. Evidence shows that wrong site surgery can not only devastate the patient and family but also negatively affect the entire perioperative team including the facility itself (Mulloy and Hughes, 2008). All institutions accredited by TJC must comply with the Universal Protocol found at TJC's NPSG at UP.01.01.01 Conduct a preprocedure verification process; UP.01.02.01 Mark the procedure site; and UP.01.03.01 A time-out is performed before the procedure begins.
• Preoperative verification processensures that all relevant documents (e.g., the history and physical examination, surgical consent, required laboratory studies) and imaging studies (properly labeled and displayed) are available before the start of the procedure. Preprocedure verification is best conducted when the patient can be involved and should be complete before the patient leaves the preprocedure area. The surgical team must agree that this is the correct patient and the planned procedure on the specified side and site. The preprocedure verification process also includes confirming availability of necessary equipment, which is reconfirmed during the time-out.
• Marking the surgical site must occur so that the intended site of incision or insertion is clear and unambiguous. Procedures that involve left/right distinction, multiple structures, or multiple levels (spinal surgery) require specific marking. The marking also must be unambiguous; initials, a “yes,” or a line at or near the incision site are all acceptable. The marking methodology should be consistently used throughout the facility. The person performing the procedure, who is accountable for it, and who will be present when the procedure is performed, must personally mark the site, and the patient should be involved in the marking if possible. The markings must remain visible after the patient has been prepped and draped. Facilities should have a provision in place for patients who refuse site marking.
• Taking the time-out. Facilities must have a procedure to implement the time-out before starting the procedure or making the surgical incision. The surgeon or other physician who is conducting the procedure must be present for the time-out. If separate teams are to perform distinct segments of a multiple procedure surgery, there must be a time-out before each procedure. This does not apply to patients in whom the same surgical team is performing multiple aspects of a single procedure. Specific situations requiring two time-outs are those in which hospital policy or law/regulation requires two separate consents (e.g., for a cesarean section followed by a tubal ligation). During the time-out, active involvement and communication among all surgical team members is expected while all participants confirm and agree that they have the correct patient, correct side and site, and the correct procedure to be performed. Many facility procedures include verification that the patient is in the correct position and that any needed equipment, supplies, or implants are correct and available, and sterile if indicated; the presence and review of relevant imaging studies; and that all prep solutions have dried to minimize surgical fire risks.
Since establishment of the Universal Protocol and WHO's Safe Surgery Saves Lives Checklist, the professional literature has asserted wide acceptance and general success in reducing errors (Haynes et al, 2011). Reducing errors does serve to reduce 30-day mortality (van Klei et al, 2012), but the authors caution that “the effect depended crucially on checklist compliance” (p. 49). Other articles have also identified barriers to implementation acceptance and a need for a broader systems approach if the checklist is to achieve its desired potential (e.g., Fourcade et al, 2012; McNamara, 2012; Walker et al, 2012). Increasingly comprehensive safety systems depend on compliance for effectiveness (Brooke et al, 2012). Unanticipated positive consequences have emerged from the implementation of checklists during surgery, such as reduction in conversion rates from laparoscopic cholecystectomy to open procedures (Robb et al, 2012) and the potential for reduced malpractice claims (de Vries et al, 2011) (Research Highlight).
Retained Surgical Items
RSIs are a concern of AORN, TJC, WHO, and all facilities where surgery is performed. Although clinically different, wrong-site surgery and RSIs are similar from safety and risk management perspectives. They both also share top positions in TJC's top 10 annual sentinel events. The physiologic, financial, and public relations consequences from both wrong-site surgery and RSIs are potentially severe. In ORs, conducting count processes according to evidence-based policies (audible, visual, and concurrent) are considered a key risk reduction and patient safety strategy (Grant-Orser, 2012).
Soft goods such as surgical sponges are one of the most frequent inadvertently retained items (Martindell, 2012). Moreover, in about 80% of retained sponge cases, counts were reported as “correct” at the conclusion of the procedure (Gibbs, 2011). Physiologic consequences of an RSI include possible infection, obstruction, fistula formation, perforation or consequent pain, suffering and possible death, and the likely need for additional surgery.
A voluntary national surgical patient safety initiative, NoThing Left Behind, investigated why RSIs continue and attempted to develop practices to reduce their persistence. Participating facilities aggregated data related to RSIs. Analysis revealed that the attribution of increased retention risk to patient or procedure characteristics such as those described by Rowlands (2012) (e.g., obesity, complexity of procedure, increased number of personnel emergencies) only partially explained incorrect surgical counts. Gibbs (2011) concludes that the OR culture and environment in which the patient has the procedure is also a major risk determinant. Thus prevention of RSIs, once thought of as a straightforward matter of counting to 10, has joined the human factors/systems analysis research cadre that focuses on perioperative patient safety issues. Gibbs also urges a greater effort at a common nomenclature that includes replacing the word “count” with “account” to more accurately reflect the complexity of the process.
As noted, CMS and many private insurers now refuse payment for remedial treatment such as otherwise unnecessary surgery to remove a sponge or to treat the physiologic consequences of its inadvertent retention. The healthcare facility must absorb the costs of extended hospitalization and required corrective treatment; it also may be deemed responsible for the entire cost of care for patients whose condition requires readmission within 30 days of discharge. Additional financial losses may arise from defending any resultant malpractice litigation, impaired public reputation, and the need to respond to licensing and regulatory body inquiries.
Prevention of RSIs.
Facilities should have an established system to prevent RSIs that reflects AORN's RP for Prevention of Retained Surgical Items (AORN, 2013). Key components of that RP appear in the Patient Safety box below, as does the sample count record in Figure 2-2. The policy should also incorporate human factors and keep interruptions, distractions, and repetition-induced attention deficits to their minimum (Coiera, 2012).
Two persons counting aloud, combined with the surgeon's manual exploration and the scrub person's mental tracking of sponges and other counted objects in the wound (situational awareness), have long been mainstay approaches to prevent RSIs. Situational awareness may be defined as the perioperative nurse's and scrub person's perception of relevant clinical cues in the count environment, their comprehension of the importance of those cues, and their identification of required interventions based on those cues (Sitterding et al, 2012). New sponge-tracking technologies have emerged to enhance risk reduction strategies to prevent inadvertent retention of RSIs. These include sequentially numbered sponges, bar coding, and radiofrequency identification (RFID) products (Goldberg and Feldman, 2012).
Typically, sequentially numbered sponge products are prepackaged, presterilized sponges in groups of 5 or 10 in sequentially numbered packages. When using such numbered systems, surgical teams are more likely to detect a missing/retained sponge because they must account for each numbered, individual sponge rather than account simply for an aggregate number. The usual tenets of counting remain: counts are performed audibly, concurrently, and visibly. Count policies and practices in the institution should be standardized with little variability among staff members (Edel, 2012). Distractions are minimized; some institutions take a “pause for the cause” while counts are in progress.
RFID and bar-code technologies are also incorporated into equipment to prevent RSIs. RFID systems tag instruments, soft goods such as sponges, and other items likely to enter the body during a procedure and use a scanning wand through tissues to ascertain the location of these items before the procedure ends. Bar-code scanning systems require the scanning of items as a procedure begins and/or as they are put into the body and again as they are removed from the body and/or as a procedure ends.
Other technologic approaches will continue to emerge. No matter which RSI prevention system a facility adopts, none is a cure-all or infallible. All technologies require precise use, as designed and in accordance with the manufacturer's instructions. As with any new technology or approach, all team members must be alert to possible unintended consequences of its use (e.g., electrical interference with other equipment or overreliance on the additional margin of safety provided by an approach, which may lead to less vigilance).
Incorrect Counts.
An “incorrect” or unreconciled count occurs when the number of items on the count record or worksheet fails to match the number of items recovered during a closing count. All incorrect closure counts must be reported immediately, and attempts made to resolve every discrepancy. If a count remains unresolved, the circulating nurse must again notify the surgeon of the unresolved count, and a search must be made for the missing item, including the surgical wound, field, floor, linen, and trash (this is the rationale that linen and trash must not leave the OR until the end of the procedure). All personnel must direct their immediate attention to locating the missing item. If it is not found, an x-ray film may be taken and read by the radiologist or surgeon as specified in institutional policy. If the x-ray is negative, the count is recorded as incorrect or unreconciled and the x-ray results noted on the patient's intraoperative record. An incident/occurrence/event report should be initiated according to institution policy. Accurate counting and recording of soft goods, sharps, and instruments are essential for the protection of the patient, personnel, and the institution, and are integral to effective risk management.
Care and Handling of Specimens
During the many years perioperative patient care has included safe handling of surgical specimens, laboratory medicine's safety practices focused on the analysis of the surgical specimen. The WHO World Alliance on Patient Safety now recognizes that to improve patient safety further in laboratory medicine, the pre- and postanalytic stages are equally important. As with checklists and communication, this WHO initiative urges adoption of requirements for multidisciplinary, multistage, and multisystem approaches (McCoy et al, 2009). Like other areas of patient safety, laboratory errors occur more frequently than commonly believed (Zarbo, 2009). Bixenstine and colleagues (2013) found specimen errors in 2.9% of cases they reviewed. The errors included problems with specimen containers (1.2% of containers with errors) and specimen requisitions (2.3% of requisitions with errors). Their analysis of errors only included specimens before they reached the pathology laboratory (from the OR to pathology). There is a wide potential for errors in surgical specimens, including unlabeled specimens, empty containers, incorrect side designation (called wrong “laterality”), incorrect or no identified tissue site, or incorrect or no patient names. As with preventing RSIs, technologies such as bar coding and RFID chips methods may reduce the frequency of specimen labeling errors (Layfield and Anderson, 2010).
TJC addresses specimen handling in the 2013 National Patient Safety Goals, requiring use of at least two patient identifiers when collecting specimens for clinical testing. It also requires action to improve timeliness of reporting critical laboratory results. Accurate and timely communication and delivery of specimens are presumptively necessary in timely reporting of results. The care and handling of specimens raise considerations unique to perioperative settings, and again AORN's RPs for Care and Handling of Specimens in the Perioperative Environment provides the best guidance. (The Patient Safety box on page 33 summarizes key points in safe specimen care and handling.)
Proper management of specimens is critical to the outcome of a patient's surgery. It is the responsibility of the surgical team to identify, document, and care properly for specimens. Common specimens include blood, soft tissue, bone, body fluids, and removed foreign bodies. Complete and accurate identification and labeling of specimens and timely delivery to the proper laboratory for analysis are imperative. A mislabeled specimen may result in misdiagnosis and consequently inappropriate treatment of the patient. At a minimum, each specimen must bear a label with the correct patient name and identification number, specific origin of the specimen, and laterality (e.g., Jane Doe, 100001, right breast biopsy). The surgeon should provide descriptive information about the specimen (e.g., “suture tag at 6 o'clock”). The nurse should “repeat back” to the surgeon information being sent to the laboratory (e.g., patient name, type of specimen, source/location, required tests, special handling needs). All specimens and their disposition must be documented in the patient's intraoperative record.
Handling of each specimen occurs according to specific protocols established by the receiving laboratory. Generally the surgical team handles all tissue in order to preserve its integrity, keeping specimens moist and transporting them to the laboratory as soon as possible. Standard transmission-based precautions govern specimen transport to protect individuals who must handle the specimen (see Chapter 4). Labels should identify the need for precautions and the presence of biohazardous material. Formalin is frequently used to preserve specimens if they are not taken to the laboratory immediately. It is a hazardous substance that can cause watery eyes and respiratory irritation. Direct contact will injure human tissue. Gloves must be worn and adequate ventilation provided in areas where formalin is handled. Institutional policy should describe procedures to follow in case of spills.
Specimens for Frozen Section.
When immediate tissue identification or identification of malignancy is needed, specimens are quick-frozen, sliced, stained, and examined in the laboratory under a microscope. This method of tissue examination is called frozen section. Specimens for frozen section usually are placed on moistened Telfa or into a dry covered specimen container to prevent drying. They are never placed in saline solution or formalin nor are they ever transported on a counted sponge. The results of frozen-section analysis are communicated to the surgeon intraoperatively. If the hospital has no system for direct pathologist-surgeon communication, the perioperative nurse receives the telephone report of a frozen section. Because this is considered an especially critical test result, the nurse “reads back” the test result to the pathologist and surgeon and receives a repeat-back from the pathologist and surgeon to verify accurate communication of the results.
Risks for Burns: Electrical and Light
Electrical hazards in the OR include electric shock, fire, burns, and explosions. Electrical burns result from current flowing through the body and emerging in a concentrated area to ground and can occur from touching an uninsulated wire, a damaged plug, or an ungrounded piece of metal equipment. Electrosurgical units (ESUs; sometimes called the “Bovie”) send a specified current from the ESU through the active electrode (often referred to as a “pencil”) or other ESU electrode to create sufficient heat to cut or coagulate the vessel touched by the pencil or other instrument. The current then exits the body through a dispersive electrode (sometimes called the “ground pad”) and returns to the ESU. Chapter 8 contains a thorough discussion of electrical safety.
Fires and Explosions
Surgical fires are one of the top 10 technology hazards identified by the ECRI Institute (ECRI, 2013). Fires and explosions in all settings require three components: (1) ignition source/heat, (2) fuel, and (3) oxygen (or another source of oxygenation [e.g., H2O2]). Nurses in perioperative settings can decrease risks of burns from fires by considering these “fire triangle” requirements during procedures and working with the perioperative team to reduce each to no more than necessary. Elimination of any single component of the triangle will prevent fire. If elimination of at least one component is not possible, the perioperative team's goal is to keep each component as distant from each other in either time and/or space as possible.
Ignition/heat sources serve to provide the energy to initiate the oxidation chemical reaction, that is, ignition. In the OR, concentrated sources of energy such as sparks from static electricity, ESUs, and lasers are key sources of heat and ignition. Oxygen or an oxygen-nitrogen composition such as nitrous oxide (N2O), administered via prong or mask, is the usual oxygenation source. Room air (21% O2) can serve as the oxygenation source but only if there is a sufficiently fine fuel (e.g., powders). A common fuel source is disposable drapes (which can also trap oxygenation sources in its folds). Fuels can literally be anything that burns, even mascara (Raqqad, 2010).
Fire and explosion prevention strategies involve separation of ignition/heat sources, fuel, and oxygen in time and/or space. Speed of the ignition reaction depends on temperature, particle size, and the concentration of the reactant fuel. Space separation strategies involve keeping (or eliminating) possible sources of igniting sparks (e.g., ESUs and lasers) as far from anesthesia and the patient's head and throat as possible and using room air rather than more concentrated oxygen or N2O as much as possible. Nitrous oxide, like free oxygen, is an oxidizing agent but only in the presence of a fire or explosion, which then breaks down its chemical bonds; an explosion, however, can be even more violent when N2O is present than with O2 alone because when a fuel burns in N2O, it produces more heat than burning in O2 alone.
Not surprisingly, procedures using ESUs and lasers around the face and neck (i.e., near O2 or O2/N2O) provide the greatest risk (Haith et al, 2012). Even so, as with all surgeries, ESU-active electrodes should be kept in holsters and not allowed to rest on drapes; lasers and ESU-active electrodes should not be allowed to touch fine gauze or sponges, especially alcohol-soaked sponges (Herman et al, 2009). See the Patient Safety box on page 34 for additional safe practice specifics.
Risk for Chemical Burns
Chemical burns most commonly occur during or after surgical site skin preparation (prep). Skin prep solutions should be applied with care to prevent pooling, which can lead to chemical burns to the skin. Towels should be tucked under the patient along the area to be prepped to catch any dripping solution and removed upon completion of skin prep (these towels are sometimes called “drip” towels). In addition to being a chemical irritant, prep solution can serve as the fuel component of the fire triangle that may be ignited by a spark from an active electrode of the ESU or even from a discharge of static electricity. Thus inappropriate use of prep solution can also cause thermal burns. Ignition of prep vapors can also occur as solution evaporates. Solutions used for skin prep must dry before application of surgical drapes.
Pressure Injuries
Pressure injuries include nerve compression (peroneal and ulnar nerves are particularly susceptible); nerve stretching (e.g., brachial plexus extension); and pressure ulcers (especially on the coccyx, heels, or back of head in supine position). Pressure or the lack thereof also may cause vessel stasis and DVT. Each surgical position has its own predictable pressure risks and methods of prevention. As a rule, however, the surgical team should remember that increasing the area of contact or reducing the force pushing on that area will decrease pressure. An easy to remember formula is pressure equals force divided by area (P = F/A). Thus both increasing area and decreasing force lessen pressure. This formula explains why both thin and obese patients are at risk for pressure injuries during surgery. Thin patients tend to have a smaller area (e.g., the coccyx) directly in contact with the OR bed or a positioning device. Obese patients, on the other hand, have more force from their increased weight and body mass. Padding typically reduces pressure by increasing the area over which a force is applied. Reminding scrubbed personnel not to lean on the patient reduces force applied to an area, thus also avoiding pressure increases.
Medication Administration
Like all perioperative safety concerns, medication administration safety must be examined within the context of its multidisciplinary system. That system includes medication prescribing, transcribing, procurement and dispensing, as well as administering (AORN, 2013). Medication administration in the OR/invasive procedure room is complicated, compared with that found in other nursing care areas, in that the OR often requires that the circulating nurse and scrub person jointly prepare a medication that will be administered by a third person, usually the surgeon. Moreover, two potentially lethal medications are frequently used on the field, for example epinephrine and heparin, and both are clear and come in widely disparate dosages. Additionally, aseptic technique requires the medication be administered by someone other than the person removing the medication from its original container. Checking for allergies, important in all areas, takes on new dimensions as well because anesthetics may disguise allergic reactions.
Vigilance in following established safe medication practices is crucial. Consider a “No interruption zone” or “Do not disturb” time during important activities in medication preparation (ISMP, 2012). Perioperative personnel must pay particular attention to acknowledging medication name, and concentrations, labeling solutions in syringes and containers on and off the sterile field, and performing read-backs/repeat-backs. Tedious as it may seem, the complete name and concentration of the drug should appear on the administration device; thus “lidocaine 2% with epinephrine 1 : 100,000” and not “epi.” The Patient Safety box above lists some potential risks of medication errors in the perioperative setting.
Team members also must follow policies and procedures strictly to double check the medication or solution before it is dispensed to the sterile field. Medications and medication containers (e.g., syringes, cups, basins), both on and off the sterile field, must be labeled fully and accurately with medication names and concentrations (see Patient Safety: Elements of Performance for Medication Safety, p. 20); furthermore, processes must be established and used to verify labels. Scrub persons should identify medications by announcing the name of the medication being passed to the surgeon, along with its concentration. A repeat-back confirmation from the surgeon ensures the correct dose of medication or solution. The institution's “Do not use” list of abbreviations must be observed and applied to all medication orders and medication-related documentation that are handwritten or on preprinted forms (Table 2-2).
TABLE 2-2
| Use | Do Not Use | Reason |
| units | U or u | Mistaken as “0”, “4,” or “cc” |
| 0.1 mg | .1 mg | No leading zero results in easily missed decimal point (i.e., 10 times the dose) |
| 0.1 mg or 1 mg | .10 mg or 1.0 mg | Trailing zero results in easily missed decimal point (i.e., 10-100 times the intended dose) |
| mcg or micrograms | µg | Mistaken for mg |
| morphine sulfate | MS | Mistaken for MSO4 and MgSO4 |
| morphine sulfate | MSO4 | Mistaken for MS and MgSO4 |
| magnesium sulfate | MgSO4 | Mistaken for MS and MSO4 |
| International unit | IU | Mistaken for IV and 10 |
| daily | Q.D. | Mistaken for Q.O.D.; the period after the Q can be mistaken for an “I” |
| every other day | Q.O.D. | Mistaken for Q.D.; the period after the Q can be mistaken for an “I” and the “O” can be mistaken for “I” |
| left ear | as | Confused with left eye (os) |
| right ear | ad | Confused with right eye (od) |
TJC's list of look-alike/sound-alike drugs, based on findings from the Institute for Safe Medication Practices (ISMP), should be readily available to all perioperative personnel (see Table 2-2 for potentially problematic drug names, potential errors and consequences, and specific safety strategies).
Federal agencies, TJC, and individual institutions increasingly look to bar-code medication administration (BCMA) systems to reduce medication errors. Adoption of BCMA systems is likely to accelerate because BCMA has become a criterion of the “meaningful use” of health information technology (CMS, 2012). Empiric studies have found a reduction in errors after BCMA implementation. Unfortunately, precursors to errors, such as workarounds, interruptions, and patient and technology characteristics, continue (IOM, 2011). As with adoption of any new system, perioperative nurses must recognize unique factors arising from the use of BCMA in perioperative settings and remain vigilant for possibly unintended consequences.
Risk for Fluid or Electrolyte Imbalance
Blood Loss.
Measuring blood loss is a vital procedure in the surgical management of critically ill or elderly patients, patients undergoing complex procedures, trauma and organ transplant patients, patients with abnormal bleeding or clotting time or with extensive renal/liver disease, and infants. Some anesthesia providers prefer to estimate blood loss via visual inspection of drapes, suction canisters, and used soft goods such as sponges. When blood loss estimates must be more accurate, weighing sponges provides a reliable means of judging the amount of blood lost and of gauging the need for transfusion. The weight of dry, unused sponges and the weight of the plastic bag for soiled sponges must first be determined and then excluded from the weight tally. Grams (g) measured are converted to milliliters (mL) on a 1 : 1 basis, and blood loss estimates are reported to the anesthesia provider. The setup to weigh sponges requires a gram scale and plastic bags to hold soiled sponges. (Box 2-5 details the procedure.) The estimated blood loss may be part of the hand-off report in ORs that use a surgical Apgar score.
Blood and Blood Products.
Maintenance of circulating blood volume is crucial during surgical procedures; this is accomplished with administration of whole blood or blood components. Blood products are literally life savers but can be fatal if improperly stored or administered. American Association of Blood Banks' (AABB, 2011) standards and technical manuals should be incorporated into facility policy and must be followed exactly. Whole blood is rarely administered unless the patient has an acute, massive loss (often empirically determined as a loss that exceeds one third of the circulating volume, or approximately 1500 mL for an adult). Instead, packed red blood cells (PRBCs) that improve oxygen-carrying capacity and oxygen transport to tissues, with or without crystalloid or colloid solutions, are administered to maintain intravascular blood volume. Crystalloid solutions include normal saline and lactated Ringer's solution; colloid solutions include albumin, purified protein factors, dextran, and hydroxyethyl starch (hetastarch). If coagulopathy is an issue, fresh frozen plasma (FFP) platelets and cryoprecipitate should be considered. When blood or any blood products must be given, appropriate precautions are necessary to reduce the hazards of its administration. Despite which product is administered, blood management is a patient-centered care measure provided by nurses (Tolich et al, 2013) (Patient-Centered Care).
Elective surgery patients who may need blood products during surgery have a pretransfusion sample taken 1 to 7 days (facility policy and patient history dependent) before surgery to ensure compatibility and to avoid antibodies that may emerge in response to exposure through blood transfusions, pregnancy, or environmental factors or as a consequence of the patient's disease process. Type refers to the test to determine the ABO and Rh blood type. Screen refers to the test for unexpected antibodies. Crossmatching refers to the test for the compatibility of the recipient's serum and the donor's red blood cells. It is crucial to identify the patient correctly before the pretransfusion blood sample is drawn and then to ensure that the sample is properly labeled. Improperly identified pretransfusion blood samples can result in acute hemolytic transfusion reactions at the time of transfusion, and death.
A patient having an elective surgical procedure for which blood has been requested should not be anesthetized without verification that the requested blood products are typed, crossmatched, and available, and that informed consent to receive blood products has been documented (Figure 2-3). This should be part of the preoperative verification process and then reverified during the time-out. An institutional blood requisition form, with complete and accurate patient identification information, should be sent to the blood bank when blood or blood products are being requested. Included on or with this requisition should be the number of units desired. Many institutions have computerized ordering and the same information is required.
If the patient is sent to the OR directly from the emergency department or trauma admitting area without a chart, all patient identification information must be printed plainly on a piece of paper. The perioperative nurse should contact the blood bank to explain the emergency situation and facilitate release of the needed blood products.
Precise and accurate blood product storage conditions are important to ensure patient safety and to avoid wasting improperly stored products. Whole blood and blood products must be stored under continuously monitored conditions in accordance with AABB rules and U.S. Food and Drug Administration (FDA) regulations. Storage temperatures must be documented and all storage equipment for blood products must be properly maintained and tested, including function checks on alarms. The return of unused blood products must comply with facility policy; further, each component presents specific requirements for proper maintenance and return (e.g., thawed cryoprecipitate and platelets must be maintained at room temperature [22° C]; and red cells and thawed plasma must be kept refrigerated at 4° C).
Before administration of any blood product, the circulating nurse and anesthesia provider (or a second licensed individual) must confirm that a signed consent is on the medical record, and then proceed with the following:
• The unit number on the blood product corresponds with the unit number on the blood requisition. Facilities using electronic records will return a “transfusion card” or “crossmatch card” as verification that this unit can be given to this patient in lieu of the requisition.
• The name, birth date, and number on the patient's identification band agree with the name, birth date, and number on the slip with the blood product.
• The patient's name on the slip with the blood product corresponds with the name on the requisition.
• The blood group indicated on the blood product corresponds with that of the patient.
• The date and time of expiration have not been reached.
• The blood product bag is free of leaks, damage, or signs of possible bacterial contamination (e.g., presence of fine gas bubbles, discoloration, clots, or excessive air in the bag).
Both individuals who verify this information must sign the slip that comes with the blood product. If a discrepancy arises with any of these checks, the blood product must not be infused until and unless resolution of the discrepancy occurs.
When it becomes apparent that more blood products will be needed than originally anticipated, the perioperative nurse should request the blood bank prepare a specified number of units in advance of the actual need to transfuse. This procedure allows the blood bank time to crossmatch the units carefully, without rushing and jeopardizing patient safety. Crossmatch requisitions should be sent for any additional units requested. If a significant amount of blood product has been required, a new, properly labeled sample with a blood grouping requisition may be needed to have an adequate sample for crossmatching although blood banks prefer using the original undiluted sample when possible.
Rapid blood transfusion requires warming of blood products to prevent hypothermia, which may induce cardiac arrest. Blood products should be warmed during passage through the transfusion set. The warming device must incorporate a temperature sensing device and a warning system to detect malfunction to prevent hemolysis or other damage to blood components (AABB, 2011). Blood must never be warmed in a microwave because its uneven, rapid heating can cause hemolysis. The probability of a transfusion reaction increases in direct proportion to the number of units transfused. The circulating nurse should remain alert for any signs of reaction, including the following:
If any such suspicious signs of reaction occur, the circulating nurse should assist the anesthesia provider to do the following:
1. Stop the transfusion. The tubing is disconnected and a new infusion of fluid such as 0.9% sodium chloride is begun to maintain venous access.
2. Report the reaction to the surgeon and blood bank immediately.
3. Anticipate possible order for stat antihistamines.
4. Return the unused portion of the blood product, the IV tubing used during the transfusion, and a properly labeled sample of the patient's blood to the blood bank.
5. Send a urine sample to the laboratory if requested.
6. Monitor the patient's reaction carefully.
7. Complete an incident/occurrence/event report covering the suspected reaction. The report might include time and date of reaction, type and amount of blood/blood product infused, and times transfusion started and stopped. Signs and symptoms, in the order of occurrence, along with the patient's vital signs, any urine or blood samples sent to the laboratory for analysis, any treatment given, and the patient's response should all be noted. A suspected transfusion reaction report must be sent to the blood bank.
Any unused blood product should be returned to the blood bank as soon as the patient leaves the OR. Returned blood can be reissued if it has not been allowed to warm to a temperature greater than 10° C. External blood bag thermometers (e.g., HemoTemp II), similar to a skin contact tape thermometer, are used on blood product bags by many blood banks to quickly identify blood that has exceeded safe storage temperatures.
Autotransfusion—the reinfusion of a patient's own blood intraoperatively—is used with increasing frequency in surgery. Predated autologous blood donation is in decreasing use. During intraoperative autotransfusion (cell salvage), blood is collected as it is lost during the surgical procedure and reinfused to the patient after it is filtered or washed. This technique can be lifesaving in emergency situations, as with major trauma, or in procedures with major blood loss, as in liver transplantation. It is also used for patients who refuse blood based on religious beliefs.
Maintaining Fluid and Electrolyte Balance
The body's fluids and electrolytes play a key role in maintaining homeostasis, transporting necessary oxygen and nourishment to the cells, removing waste products of cellular metabolism, and helping to maintain body temperature. Electrolytes are also essential to transmission of nerve impulses, regulation of water distribution, contraction of muscles, generation of adenosine triphosphate (ATP, needed for cellular energy), regulation of acid-base balance, and hemostasis (clotting blood). The intake, distribution, and output of water and electrolytes, regulated by the renal and pulmonary systems, normally maintain fluid and electrolyte balance.
Fluid and electrolyte imbalances may occur rapidly in the surgical patient, and can be caused by numerous factors, including preoperative fluid and food restrictions, intraoperative fluid loss, or the stress of surgery. The surgical patient is unable to regulate body fluid and electrolyte requirements by normal activities of drinking, eating, excreting, and breathing unaided. It is therefore imperative that the perioperative nurse collaborate in monitoring the fluid and electrolyte status of the patient intraoperatively.
Body Fluids.
The adult human body is approximately 60% water, although water content varies by age, gender, and body mass. In the elderly, body water content averages 45% to 55% of body weight, whereas it ranges from 70% to 80% in infants. Older adults are at higher risk for fluid imbalance because they have less fluid reserve, whereas the very young are at risk for fluid problems because a greater percentage of their body weight is water. Both age groups have a decreased ability to compensate for fluid loss. Muscular tissue contains more water than the same amount of adipose tissue; men generally have higher water content because they usually have more lean muscle mass than women.
Body fluids are distributed in two main functional compartments—intracellular and extracellular. Intracellular fluids (ICFs) are liquids within cell membranes that contain dissolved substances essential to fluid and electrolyte balance and metabolism. ICFs constitute approximately 70% of the body's fluid. Consequently, anything that affects water loss at the intracellular level has significant implications for the entire body. Extracellular fluids (ECFs) (30% of the body's fluid) are fluids in compartments outside the cells of the body, including plasma, intravascular fluids, fluids in the gastrointestinal (GI) tract, and cerebrospinal fluid (CSF).
Fluid spacing is a term used to classify distribution of body water. First spacing is the normal distribution of fluid in extracellular and intracellular compartments. Second spacing refers to excess accumulation of interstitial fluid (edema), and third spacing occurs when fluid accumulates in areas that normally have no fluid or only a minimal amount of fluid. This fluid accumulation occurs with burns, ascites, peritonitis, or small-bowel obstruction. Third spacing traps fluid away from normal fluid compartments and results in a deficit in useful fluid volume.
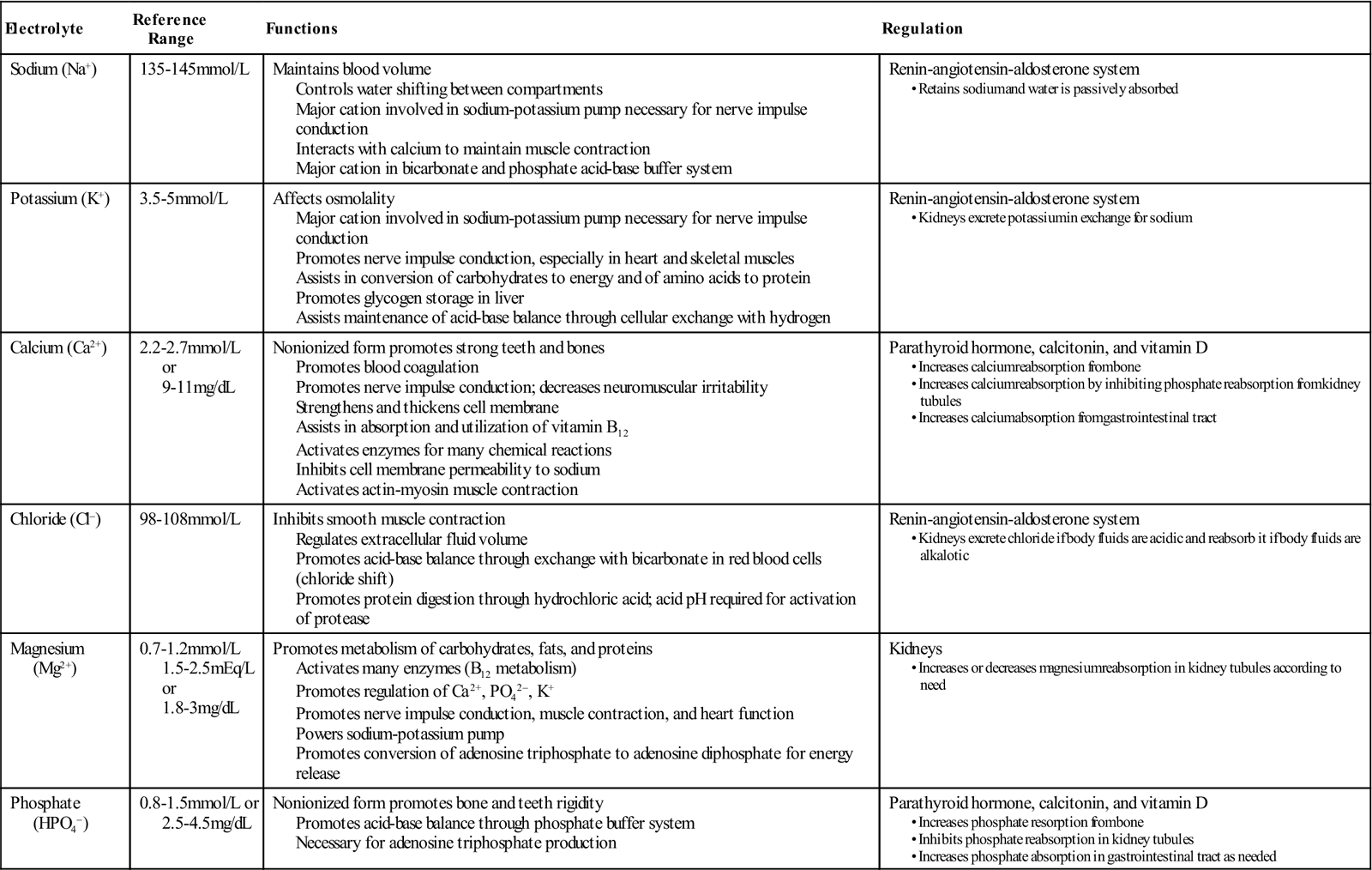
Electrolytes.
Electrolytes (Table 2-3) are found in both ICFs and ECFs. When dissolved in water, electrolytes dissociate into ions and are able to carry an electric charge. Positively charged ions are called cations, and negatively charged ions are anions.
TABLE 2-3
Electrolyte Functions and Regulators
Modified from Harkreader H et al: Fundamentals of nursing: caring and clinical judgment, ed 3, St Louis, 2007, Saunders.
The electrolytes found in ICFs and ECFs are essentially the same, but concentrations in each compartment differ. The primary ICF cation is potassium, and the primary ECF cation is sodium. The primary ICF anion is phosphate, and the primary ECF anion is chloride. Fluids and electrolytes move between the ICF and ECF spaces to facilitate body processes, such as acid-base balance, tissue oxygenation, response to drug therapies, and response to illness. Diffusion, active transport, and osmosis control these movements.
Diffusion, Active Transport, and Osmosis.
Diffusion is the movement of molecules from an area of high concentration to one of low concentration and can take place with or without a membrane separating the concentration gradient. When diffusion occurs, movement continues until there is an equal concentration of molecules. Diffusion across membranes is particularly important in regulating fluid transport and balance in the body.
Active transport is a process by which molecules move across a cell membrane against a concentration gradient (i.e., from an area of low concentration to an area of high concentration) with the impetus of external energy. A so-called “sodium-potassium pump” moves sodium out of the cell and potassium into the cell to maintain the intracellular and extracellular concentration differences of sodium and potassium. ATP is the energy source for this sodium-potassium pump.
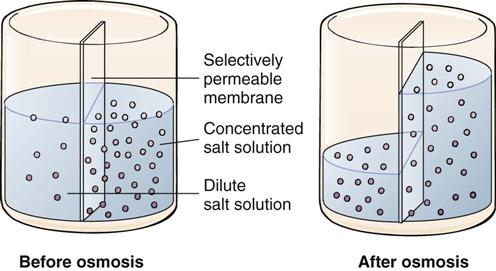
Osmosis is the movement of a fluid through a semipermeable membrane from a solution that has a lower solute concentration to one that has a higher solute concentration (Figure 2-4). The semipermeable membrane prevents movement of solute particles. The concentration of these particles is measured in a unit called the osmole (osm). Osmolality is the term used to express the concentration of a solution in milliosmoles per kilogram (mOsm/kg) of water. A solution with the same osmolality as blood plasma is called isotonic. Isotonic solutions, such as 0.9% normal saline or lactated Ringer's solution administered intravenously, prevent the shift of fluid and electrolytes from intracellular compartments. A hypotonic IV solution (0.45% saline or 2.5% dextrose) has a lower concentration of solutes than that found in plasma and moves water into the cells. Administration of a hypertonic IV solution (5% dextrose in normal saline or 5% dextrose in lactated Ringer's solution) with a greater concentration of solutes than that of plasma moves water out of the cells.
Preoperative Considerations.
Preoperative laboratory analysis of electrolyte levels should be checked and abnormalities corrected to within normal limits before any surgical procedure, unless the surgery is needed to correct a life-threatening problem. Preexisting conditions, such as diabetes mellitus, liver disease, or renal insufficiency, may worsen with surgical stress, increasing a patient's risk of fluid and electrolyte imbalances. Diagnostic procedures that require the administration of IV dyes may produce osmotic diuresis, with resulting urinary excretion of water and electrolytes. Preoperative steroids or diuretics affect the excretion of water and electrolytes; diuretics, used in the management of hypertension, cause the loss of potassium. Preoperative surgical regimens, such as bowel-cleansing routines, may increase fluid loss from the GI tract. Medical management, such as gastric suction, can also affect fluid and electrolyte balance in the surgical patient, as can preoperative fluid restriction.
Preoperative fluid restrictions are used to reduce nausea, vomiting, and aspiration risks in the surgical patient. Many healthcare facilities and providers are changing this practice because newer anesthetic agents tend to cause less nausea and vomiting than in the past. Prolonged fluid restrictions may not be necessary in healthy patients before surgery; thus black coffee or pulp-free juice may be ingested safely 2 to 3 hours before surgery, without an increase in gastric volume (see Chapter 5).
Deficient Fluid Volume.
The most common patient problems associated with fluid and electrolyte imbalances during surgery include deficient fluid volume (DFV), water imbalance, and potassium imbalance. DFV is an imbalance in isotonic body fluids related to decreased intravascular, interstitial, or intracellular fluid. Very young and very old surgical patients are affected most rapidly by fluid losses from bleeding; inadequate intake because of previous nothing per os (mouth) (NPO) status; inadequate IV fluid replacement; excessive cutaneous losses from fever and sweating; third-space fluid losses attributable to bowel obstructions, ascites, or peritonitis; excessive GI losses resulting from diarrhea, vomiting, GI suctioning, or fistulas; evaporation of fluid from the exposed peritoneum during open abdominal surgery; shifting of intravascular fluid into the surgical site (third-space edema); and inhalation of dry gases. Third-space fluid losses cannot be measured directly, but can be considerable after extensive dissection of tissue. Intraoperative use of an electrolyte solution, such as lactated Ringer's solution, for fluid replacement can help to correct intraoperative third-space fluid losses.
The effect of fluid loss on the surgical patient depends on the amount of fluid lost and the speed at which the fluid is lost. A patient who loses a large amount of fluid (>500 mL) or loses fluid rapidly exhibits symptoms of shock; immediate fluid replacement therapy is required. A slow loss of fluid may be compensated through albumin synthesis and erythropoiesis.
Sodium and Water Imbalances.
Sodium is a cation in ECF; sodium plays a major role in maintaining the osmolality and water balance of ECF. Because cell membranes are permeable to water, sodium also affects ICF volume, as well as helps to maintain acid-base balance in the body. The sodium-potassium pump plays a vital role in neuromuscular activity.
Hyponatremia (serum sodium level <135 mEq/L) can be caused by increased excretion of sodium with diuretic therapy and the abnormal loss of sodium through nasogastric (NG) suctioning and third spacing. Patients undergoing procedures such as transurethral resection of the prostate (TURP) or similar procedures are at risk for dilutional hyponatremia and volume overload, caused by absorption of the irrigation solution used. Given that saline is a good conductor of electrical current, fluids that contain no electrolytes (e.g., glycine, sorbitol, and mannitol) must be used to irrigate when using electrical current in tissue dissection during these procedures.
Potassium Imbalances.
Potassium is the major ICF cation and is necessary for contraction of skeletal, smooth, and cardiac muscle. It is also necessary for cardiac contractions and for peristaltic movements of the GI tract. Potassium plays a major role as well in the transmission of nerve impulses by regulating neuromuscular excitability and in the formation of muscle protein by transporting glucose into the cells with insulin. It is also elemental in maintaining acid-base balance and ICF osmotic pressures.
Hypokalemia (serum potassium concentration <3.5 mEq/L) can occur intraoperatively as a result of the suctioning of large amounts of body fluids or using diuretic therapy and other drugs (e.g., mannitol) that increase renal flow. Signs and symptoms of hypokalemia include cardiac effects, such as ectopy, dysrhythmias, conduction abnormalities, and altered sensitivity to digitalis. The neuromuscular effects of hypokalemia include muscle weakness; its smooth muscle effects include gastric distention, paralytic ileus, and urinary retention.
Treatment of hypokalemia includes IV replacement when the deficit is severe, as in the development of cardiovascular or other serious symptoms. Potassium irritates veins on infusion; the infusion site therefore requires monitoring for redness, heat, swelling, and site pain—all signs of chemical phlebitis. Overcorrection and subsequent hyperkalemia must be avoided by monitoring serum potassium levels at frequent intervals (often every 2 to 4 hours).
Hyperkalemia (serum potassium level of more than 5 mEq/L) can arise during surgery via massive transfusions of stored blood; decreased excretion of potassium caused by hypovolemia or renal failure; and shifting of potassium from the cells into the ECF caused by acidosis, tissue breakdown from surgery, crush injuries, or burns. Drugs infused during surgery can also induce hyperkalemia; such drugs include antiinflammatory agents, β-adrenergic receptor blockers, digitalis, succinylcholine, heparin, and the penicillins.
Signs and symptoms of hyperkalemia include neuromuscular symptoms such as weakness and paresthesias in the arms and legs. Smooth muscle symptoms include diarrhea and abdominal distention. Excess potassium can cause serious cardiac symptoms such as ventricular dysrhythmias, heart block, and asystole. Cardiac side effects of hyperkalemia are treated by calcium gluconate infusion. Sodium bicarbonate or insulin-glucose infusions can shift ECF potassium into the cells, reducing the plasma's potassium level.
Patient Rights
Protection of patients' personal, ethical, and legal rights underlies patient safety and risk management. Many nurses equate patients' legal rights with negligence, malpractice, and informed consent. As described in the following text, these are only some of the many legal and ethical concerns in myriad perioperative settings, although legal and ethical issues interrelate as do all components in healthcare.
Freedom from Negligent Treatment
Malpractice is negligence by a professional in the performance of a professional act. Provision of safe care to the patient is the best protection for the nurse and facility from malpractice claims. This is because it is impossible to sue healthcare providers for malpractice without an injury; almost always this must be a physical injury. To be successful in a malpractice case, the injured patient must prove that the member(s) of the surgical team departed from the standard(s) of care applicable to their profession and that this breach of applicable standard(s) caused the injury.
If an injury occurs, both the professional licensure and civil legal systems require the team to act to prevent it from worsening. Risk management should be notified and become involved immediately after injury occurs in order to minimize the likelihood of the patient or his or her representative bringing suit and if that fails, to prepare a defense.
Many injuries that do occur in the immediate perioperative period are extremely difficult to defend successfully. Injuries resulting from RSIs, wrong site surgery, wrong patient surgery, wrong side surgery, fires and other burns, and intraoperative pressure ulcers are all subject to a legal doctrine, in Latin called res ipsa loquitur (“the thing speaks for itself”), because these injuries ordinarily do not occur in the absence of some form of negligence. This means that unless the defense can prove there was no negligence (proving a negative is always difficult, if not impossible), the injured party will prevail. Even in potential res ipsa loquitur cases, patients who feel they have been treated with respect and dignity may be likely to accept sometimes less than perfect results, may be less inclined to litigate, and may be more likely to cooperate with postinjury treatment, regardless of fault.
Privacy
A patchwork of state statutes attempts to protect confidentiality, to ensure patients' access to their healthcare records, and to preserve healthcare data privacy. Simultaneously, federal law provides that patients must consent for their health information to be used for treatment, payment, or business purposes. The federal Health Insurance Portability and Accountability Act (HIPAA) imposes privacy and security rules that limit use or disclosure of protected health information in order to ensure patient privacy rights with respect to this information. Healthcare facilities accordingly must have administrative procedures to protect the privacy and confidentiality of all patient information. Facility policies should include protocols specific to perioperative settings, such as those governing or limiting the presence of healthcare industry representatives or other visitors in the OR.
Another federal statute, the Patient Safety and Quality Improvement Act (PSQIA) established a voluntary medical error reporting system to enhance the data available to assess and resolve patient safety and quality issues. To encourage reporting and analysis of medical errors, PSQIA created federal confidentiality protections for patient safety information. That law also sets forth an exception to HIPAA rules requiring patients' consent to disclose information about an error that occurred in the course of their treatment. Many states also have mandatory medical error reporting systems that likewise make exceptions to their confidentiality rules. Additional information on PSQIA is available at www.hhs.gov/ocr/privacy/psa/understanding.
Perioperative nurses ethically must protect patient privacy even if HIPAA, state laws, or facility protocols do not specifically address a privacy issue. Team members exercise care to protect patients' bodies from any more exposure than is required by a procedure. Likewise, comments made in or out of patient care areas about patient appearance, lifestyle, or social status, for example, are never appropriate unless required for safe care of that patient.
Privacy and Social Media
Widespread use of social media has raised a new challenge for patient privacy protection. Adoption and even the definition of social media is rapidly evolving (von Muhlen and Ohno-Machado, 2012). Although social media provide many benefits, including patient access to their records and facilitating patient partnership in their care, confidentiality pitfalls are rife. Inappropriate discussions of patient information in elevators, in cafeterias, or in social settings after work can now wind up being “overheard” worldwide. In that light, all healthcare professionals, employees, and facilities themselves must recognize that they have a heightened obligation to protect patient privacy by avoiding such inappropriate discussions entirely and absolutely. A refinement of HIPAA was passed in 2009 called the HITECH Act. Students, unlicensed and licensed healthcare workers, and retired healthcare workers uniformly have a continuing duty to observe and protect patient privacy rights under HIPAA and HITECH (Terry, 2011-2012).
The consequences of violating patient privacy rights via inappropriate use of social media may result in both employer and licensure disciplinary actions, possibly including termination of employment, suspension and revocation of license, fines, and other serious penalties. A 2010 survey of state boards of nursing by the National Council of State Boards of Nursing (NCSBN) revealed that 33 of 46 responding state boards had received complaints of nurses violating patient privacy by posting patient information and/or photographs on social networking sites. Twenty six of those 33 state boards reported taking disciplinary action based on these complaints, ranging from reprimand to license suspension (NCSBN, 2011) (Box 2-6).
In addition to violating federal and state patient privacy laws, posting anonymous patient photographs on social media can also violate another federal statute, the Video Voyeurism Prevention Act of 2004, which provides that whoever photographs, films, or records (by any means), and disseminates an image of a private area of an individual who has a reasonable expectation of privacy without the individual's consent shall be fined or imprisoned up to 1 year or both. Every healthcare facility should have a social media use policy. Several professional associations have developed or are developing such a policy. In addition to legal and ethical considerations, inappropriate postings can harm the reputation of the student/professional posting them and jeopardize future graduate program admission and employment prospects.
Patient Autonomy
Courts in the United States have long recognized that every adult has the right to determine what happens to his or her body (e.g., Schloendorff v. Society of New York Hospitals, 105 N.E.92 [N.Y., 1914]). This legal right is also an ethical right recognized under the ethical principle of respect for persons as autonomous beings (Beauchamp and Childress, 2008). In perioperative settings, these rights enjoy protections via the informed consent process for the procedure itself and/or for any research interventions; and via patient wishes expressed in advance directives for healthcare.
Informed Consent
The current professional literature may seem to place less emphasis on informed consent as it focuses more on other aspects of patient safety. However, courts continue to affirm the patient's right to informed consent (e.g., Singer v Rodriguez et al, 2012). Usually, the informed consent case also involves a physical injury due to other lapses in the safety system, but both aspects of such cases are equally important in the respectful treatment of patients.
Except in emergencies, surgical procedures should not be performed without documentation of the patient's consent in the chart (Figure 2-5). How to document this “informed consent” should be described in facility policy, but documentation is typically accomplished by means of a signed consent form and/or the primary surgeon's entry into the progress notes, which may or may not also be signed by the patient. The surgeon or practitioner performing the procedure is legally responsible to inform the patient about the proposed operation or other invasive procedure and its inherent risks, benefits, alternatives, and complications before obtaining the patient's oral and documented consent.
The patient must receive this information in terms that he or she can understand. TJC (2012a) also suggests, that to meet health literacy needs, consent materials be written at a fifth grade or lower reading level, subjected to readability tests, designed with bullet points to divide complex material, and translated into the patient's language whenever possible.
The patient also must be informed who will perform the procedure and when practitioners other than the primary surgeon will perform important parts of the procedure, even when under the primary surgeon's supervision. Nurses with concerns about the adequacy of the patient's understanding should report their concerns to the operating surgeon, or anesthesia provider if the concerns involve the anesthesia consent.
Consent documentation must be complete before administration of preoperative medications. On the patient's arrival in the OR, the circulating nurse and anesthesia provider are responsible to verify that consent documentation is in the chart and is correct, properly signed, and witnessed before administration of anesthesia. Typically, consent documentation encompasses the following:
• The patient's name (and legal guardian, if applicable)
• Name of facility where the procedure is being performed
• Specific name of the surgical procedure (or when multiple procedures are being done, the names of those procedures) in terms the patient understands
• Site/side of the planned procedure
• Name of the practitioner(s) performing the procedure or important aspects of it
• Alternative procedures, treatments, or therapies
• Signature of the patient (or legal guardian, if applicable)
• Date and time consent is obtained
• Statement that the procedure was explained to the patient (or legal guardian or both if applicable)
• Name and signature of the person who explained the procedure, usually the primary surgeon or physician performing the procedure
• Signature of person witnessing the consent, often a perioperative team member
Nurses who are involved as witnesses to the signing of the consent form attest only to the validity of the patient signature, time, and date, and not to the adequacy of the patient's understanding because that assurance remains the duty of the surgeon or physician performing the procedure.
Advance Directives and DNR Orders
Many of the 50 states had statutes to allow patients to make their wishes regarding their future known in a legally recognized fashion if they were unable to do so when a life-threatening situation arose. Then, in the wake of the first U.S. Supreme Court case to deal with the issue, Cruzan v. Director, Missouri Department of Health, 497 U.S. 261 (1990), Congress in 1991 passed the Patient Self-Determination Act (PSDA) to extend legal protection to all U.S. citizens and residents. Under PSDA, patients have the legal right to accept or refuse medical treatment, including resuscitation, even if refusal will likely result in death. That law also provides that all patients admitted to Medicare and Medicaid recipient providers must be asked whether they have executed, or wish to be given information about, such an advance healthcare directive, and that information must be included in the patient's healthcare record. If a patient arrives in a preoperative area with such a directive, the perioperative nurse should immediately clarify with the surgeon and/or anesthesia provider if there has been a presedation discussion with the patient as to the effect the directive should have in the immediate perioperative period. The law does not address this issue, but an automatic suspension or continuation of advance directives or DNR orders is not justified under either legal or ethical analysis. Again, facility policy should describe a perioperative procedure that preserves the patient's right to legal and ethical self-determination.
Veracity and Fidelity
Veracity includes providing accurate information. Fidelity (or loyalty) carries with it an implicit promise of truth telling and promise keeping. Both principles relate to respect for patients and their autonomy—that patients are entitled to full and accurate information and disclosure; and both principles are likewise inherent in trust, which also is foundational for patient safety. These principles relate most directly to the patient's right to informed consent. Within the patient safety and risk management context, however, veracity and fidelity also demand transparency in disclosure of adverse events to the patient/family. Some states (e.g., Pennsylvania, Florida, and Utah) legally require patient notification when an error occurs (Griffin, 2008).
Griffin reviewed data that demonstrated transparency, and disclosure after an error has occurred can minimize the consequences of litigation. In fact, articles encouraging full disclosure often do so more as a risk management issue than as an issue concerning patient rights. In any event, one consequence of the renewed emphasis on patient safety and patient rights has been to merge the heretofore separate systems of perioperative care provision, risk management, and quality improvement under a patient safety rubric that has enhanced the effectiveness of each.
Key Points
• Management of patient safety and prevention of injury risks is a collective responsibility of the entire perioperative care team.
• Major government, regulatory and accrediting agencies, and associations address key elements of patient safety.
• The AORN Perioperative Standards and Recommended Practices is the best source for information specific to perioperative nursing practices.
• Meaningful communication and a commitment to teamwork, trust, and respect are characteristic in workplace cultures that emphasize patient safety.
• Enhanced communication initiatives such as safer surgery briefings, clinical perioperative records (written or digital) and handoffs are foundational in assuring a safer patient experience and care transitions between providers and units responsible for perioperative patient care.
• Communication for safety in perioperative settings includes a wide array of protocols and topics (such as the time-out, counts, surgical specimens, electrical and chemical safety, and proper positioning) to prevent patient harm.
• Maintaining fluid and electrolyte balance is a collaborative effort that prevents deficient volume in the surgical patient.
• Protection of a patient's personal, ethical, and legal rights underlies patient safety and risk management in perioperative care settings.
Critical Thinking Question
At 1600, Nurse Steve tweeted: awful day. fat guy fell off table. busted shoulder. MD blames me! Strap on!!!! pooled prep. 2d degree burns! allergic to I !!!!!! Camouflaged d/t drape fire.
Earlier that day:
Mike Jones, a 56-year-old construction worker enters the OR for removal of a suspicious mass left upper scapular areas under MAC. History and physical review reveals 40-year history for smoking and has COPD and hypertension. Ht = 70 inches and wt = 120 kg.
Mr. Jones was not yet sedated and able to move himself onto the table and lay supine attended by Carole, the anesthesia provider. When surgeon came in, Carole says, “I note you suggested prone; I'd rather do this right lateral—OK with you?” The surgeon responded, “Sure, whatever,” and left to scrub.
Steve, the circulating nurse, gathers the pillows and axillary roll, then stands by Jones's side and loosens the safety strap. The anesthesia provider tells Mr. Jones to lie on his right side and bend his knees up until comfortable. Anesthesia positioned his right arm extended on an armboard. Steve places the axillary roll, a pillow between Jones's legs and under left elbow, and reattaches safety strap across Jones's upper thighs. He looks at Carole to say, “OK?” “Yup, thanks” was the response. Steve then proceeded with the prep; there was a little pooling of the iodine-based solution but since the case was so short and the surgeon was already gowning and gloving and said ready to drape, Steve just left it. After draping finished, Steve helped the scrub move the Mayo and back table to the left of the surgeon and the incision site as Carole adjusted the oxygen mask on the patient's face and announced she was now injecting the fentanyl and midazolam (Versed). Steve rolled the ESU unit next to the back table and reminded everyone to avoid ESU use when the patient was receiving enriched oxygen. He then called the time-out and read the checklist. All checked out, except his heart sank when Carole answered “Iodine” to allergies question. But everything was ready for the local injection and it was a short case.
Steve returned to his computer, facing the wall, to document. He glanced over to check for incision time and as he did so he heard a thump. He immediately checked the field and saw Mr. Jones's legs sliding down the other side of the table. He said, “What?” and ran to the patient's side. Carole had been turned from the patient documenting her sedatives and drawing up a sustaining dose. The surgeon and scrub tech were looking at each other at the Mayo stand discussing whether the deeper incision required a 10 blade or 15. Everyone's attention immediately turned to Jones. First they assessed breathing, then alignment, and an orthopedic surgeon came from the next room to assess Jones's shoulder, then the incision site and a second-degree skin burn was noted. A team was called to safely get the patient back on the table. His shoulder was x-rayed as positioned by the orthopedic surgeon; he was repositioned, reprepped, and the surgery proceeded.
Mr. Jones required extra sedative but was still hyperventilating. Carole began bagging with enriched O2 under the drape so as not to interfere with the sterile field. The mass involved more bleeding than anticipated. The surgeon requested the ESU. In the confusion of the fall everyone forgot the time-out warning to avoid ESU use when the patient was receiving enriched oxygen. The surgeon activated the ESU tip and ignited a flame flashback from under the drape.
1. Could this fall have been prevented?
2. Could these burns have been prevented?
3. What did the team do correctly immediately after the fall?
4. Was lateral really the indicated position for this patient?
5. What should Steve have done differently throughout Mr. Jones's perioperative care and why? Are there ethical and privacy issues that should be addressed with Steve?
![]() The answers to the Critical Thinking Question can be found at http://evolve.elsevier.com/Rothrock/Alexander.
The answers to the Critical Thinking Question can be found at http://evolve.elsevier.com/Rothrock/Alexander.