Infection Prevention and Control
Cynthia Spry
Advancements in surgical interventions and the science of infection prevention and control continue to evolve and have become increasingly complex. The complexity is derived from the changes in the healthcare environment, the advancements in minimally invasive and advanced surgical procedures, and the development of new, highly technical instrumentation and equipment. This chapter provides an overview of the etiology of infection, including emerging drug-resistant bacteria, and identifies various methods to control infection in the perioperative environment. Use of Standard Precautions along with engineering and work-practice controls assists perioperative practitioners in reducing the transmission of pathogenic organisms. Perioperative patient care is based on surgical aseptic principles. Careful adherence to these principles supports infection prevention and control, ultimately improving surgical patient safety and outcomes. Each member of the surgical team must demonstrate the highest integrity in the application of this knowledge. Finally, the potential for bioterrorism has forced the United States along with the rest of the world to strengthen security and emergency readiness. If a bioterrorist attack resulted in massive exposure or illness, perioperative personnel could and would be mobilized to serve in a variety of locations. Perioperative personnel need to be familiar with the most likely biologic weapons and the levels of precautions required for patient and personnel safety.
Causes of Infection
The human body has three lines of defense to combat infection. The first line of defense consists of external barriers, such as the skin and mucous membranes, which are usually impervious to most pathogenic organisms. The second line of defense is the inflammatory response, which prevents an invading pathogen from reproducing and possibly involving other tissue. The third line of defense, the immune response, is triggered after the inflammatory response. When a break in this defense mechanism occurs, the possibility for infection increases (Box 4-1 lists term definitions).
Microorganisms That Cause Infection
Microorganisms are living organisms that are too small to be seen with the naked eye. These organisms include bacteria, fungi, protozoa, algae, and viruses. Microorganisms are classified to determine appropriate treatment for an infection. Each organism is assigned two names; the genus is the first name, and the specific epithet (species) is the second. Bacteria are further described by shape, motility, growth in aerobic or anaerobic environments, and reaction to staining processes (Figure 4-1). Scientific names can be assigned to organisms in various ways. Staphylococcus aureus is a microorganism commonly found on the skin. Staphylo describes the clustered arrangement of the cells; coccus (Greek kokkos, “berry”) indicates that they are shaped like spheres. The species aureus is Latin for “golden,” the color of the colonies of the bacterium. Bacteria cause most surgical site infections (SSIs). Gram-positive cocci, as a group, are the most common cause of SSIs. The organisms most commonly found in postoperative SSIs include staphylococcal, enterococcal, pseudomonal, and streptococcal species. S. aureus is the most frequently identified organism.
Staphylococci.
Staphylococci are gram-positive cocci. They are facultative anaerobes but grow best under aerobic conditions. Staphylococci can be found in the indigenous flora of the skin and mucous membranes of the nasopharynx, urethra, and vagina. They are resistant to drying, heat, and high salt concentrations.
The two recognized species of staphylococci are S. aureus and S. epidermidis. Staphylococci are called coagulase positive when they are capable of clotting plasma and coagulase negative when the plasma clumps them. Coagulase-positive staphylococci are more virulent or pathogenic than coagulase-negative staphylococci. S. aureus is hemolytic, parasitic, pathogenic, and coagulase positive. S. epidermidis is parasitic, less pathogenic, and coagulase negative. S. aureus is a long-recognized cause of surgical site infections. More recently, S. epidermidis has been implicated in infections of prosthetic devices and indwelling medical devices (CDC, 2008a; Geipl 2009; Widerstrom et al, 2012). The skin surface is the most common site of S. epidermidis. Approximately 30% to 70% of individuals carry staphylococci on their skin. This can lead to contamination of clothing and dispersal of the microorganisms. For no known reason individuals who are skin carriers of staphylococci differ in the rate at which they shed the microorganisms. There is no obvious difference in hygiene and skin condition between light and heavy shedders, and no other contributing factor is apparent. Heavy shedders seem to be in normal good health.
S. aureus infections in hospitals can lead to prolonged hospital stays and may result in death. S. aureus is a medically important human pathogen that is found in the nares of approximately one third of the population (Pynnonen et al, 2011). Human nasal and throat cavities are the most important reservoirs that continually replenish the external environment. Among perioperative personnel, S. aureus has been found most commonly in the respiratory passages. The potential for patient infection increases greatly as the personnel carrier rate increases. Nasal carriers also may be skin carriers. Carriers usually harbor either coagulase-positive (pathogenic) or coagulase-negative (nonpathogenic) staphylococci; seldom are there both types, and rarely more than one strain is identified. Because an individual may be a carrier of staphylococci one day and a noncarrier the next, frequent swab testing of the nose as an infection control measure is impractical. Staphylococci survive for long periods in the air, dust, debris, bedding, and clothing. Pathogenic staphylococci grow in the sweat, urine, and tissue and on the human skin. They are more difficult to destroy than many other non–spore-forming organisms. Cleanliness of the environment; proper handling, and, when appropriate, sterilization of linens and equipment; and adherence to adequate hand hygiene practices are important controls to prevent transmission of infection (AORN, 2013a).
Enterococci.
Enterococci are gram-positive organisms that are found in the normal flora of the gastrointestinal and female genital tracts. These organisms are responsible for many serious healthcare-associated infections, including surgical site infections, bacterial endocarditis, septicemia, and urinary tract infections (UTIs). Enterococci also have been implicated in polymicrobial wound infections (CDC, 2007a; Scheithauer et al, 2012). Enterococcal infections are most often healthcare associated and seen in patients with comorbid conditions.
Pseudomonads.
The most common aerobic species of pseudomonads pathogenic to humans is Pseudomonas aeruginosa. It thrives in moist environments and is found frequently in soil, water, sewage, debris, and air. It also can be found in the normal flora of the skin and intestines. P. aeruginosa was considered a microorganism of slight pathogenic power but is now known to be associated with many infections in humans, including surgical site infections. P. aeruginosa seems to be pathogenic only when it is introduced into areas where normal defenses are absent, when it is superimposed on staphylococcal infection, or when it is present in a mixed infection. It may attack a debilitated patient who has extensive burns or major trauma. The organism is often seen in critical care and burn units. P. aeruginosa is resistant to most antimicrobial agents. Other species of pseudomonads have been associated with infections caused by contaminated intravascular cannulae (as from the use of pressure transducers) or by the lumens of equipment (such as endoscopes used in gastrointestinal [GI] studies). An important preventive measure is proper environmental sanitation and strict adherence to aseptic technique.
Streptococci.
Most streptococci are gram-positive, non–spore-forming facultative microorganisms. They are normally found in the indigenous flora of the upper respiratory, genitourinary, and GI tracts. Streptococci are classified as alpha, beta, or gamma according to chemical factors, biochemical tests, and their action on red blood cells.
Group A streptococci account for most streptococcal infections in humans. Known as the flesh-eating bacteria because of their ability to cause necrotizing fasciitis, group A streptococci are of concern because of the sporadic and deadly outbreaks of community-acquired and healthcare-associated infection (CDC, 2006a; Ramirez et al, 2011). An example is Streptococcus pyogenes, which is responsible for most soft tissue infections, otitis media, pharyngitis, impetigo, septicemia, and surgical site infections. Virulent streptococci are more serious invaders than staphylococci. Streptococci tend to involve wide areas of tissue and cause necrosis without localization. Streptococci are usually sensitive to penicillin, whereas staphylococci may not be. Streptococci also occur in mixed infections with other pathogens.
In surgical wounds, streptococci can be introduced into the incision and spread via the lymph vessels and nodes. This distribution can result in inflammation and cellulitis. Streptococcal transmission occurs by droplet transmission and by contamination of the environment. Inhalation of infectious droplets expelled from the nose and mouth of an infected individual is considered direct contact. Indirect contact occurs when infected air and dust from the environment enter a susceptible host. Group A streptococcus can be carried in the nasal passages, anus, or vagina. The upper respiratory tract is not a significant reservoir for microorganisms that cause surgical site infections except in the presence of an acute upper respiratory tract infection. In several randomized controlled studies, it was found that surgical wound infection rates are unaffected by the use or nonuse of surgical masks by noninfected individuals during the operative procedure. It is not always possible to know who might be infected, and the use of masks during operative procedures continues to be strongly recommended in the prevention of SSIs. Most bacteria in the operating room (OR) environment are shed from the skin of perioperative personnel (AORN, 2013b).
Clostridia.
The clostridia bacteria are spore-forming, anaerobic, gram-positive bacilli that produce virulent toxins. Clostridium difficile is a bacterium in the Clostridium genus, which also includes C. perfringens (gas gangrene), C. tetani (tetanus), and C. botulinum (botulism). C. difficile is an anaerobic, gram-positive, spore-forming bacillus. C. difficile is an overgrowth in the colon that can manifest symptoms of diarrhea, colitis, toxic megacolon, dehydration, colonic perforation, and death. The overgrowth of C. difficile in the colon usually results from alterations in the normal flora of the colon that are associated with the use of antibiotics (CDC, 2006a; Gould et al, 2008). For this organism to cause disease, C. difficile must already be in the GI system. Subsequently, there must be a change in the normal flora of the colon to allow the organism to grow and produce toxins. The two toxins produced are toxin A, which is an enterotoxin causing excretion of large amounts of fluid from the bowel, and toxin B, which is cytotoxic. Toxin B attacks and disintegrates cells of the intestines. Additionally, C. difficile produces tissue-degrading enzymes, which produce an inflammatory response within the colon that can result in a spectrum of disease entities.
In its spore form, C. difficile can withstand drying and heat and may be resistant to many disinfectants. The spores can survive up to 5 months in the environment. C. difficile can be transmitted between individuals and by touching objects contaminated with the organism. In the healthcare environment, C. difficile has been cultured in rooms of infected individuals 40 days after discharge. C. difficile also has been cultured from healthcare workers' shoes, fingernails, fingertips, and the underside of rings (Mitchum, 2009).
Several interventions can assist in the prevention of C. difficile transmission in the healthcare environment, including following Contact Precautions, handwashing with antimicrobial soap and water, using personal protective equipment (PPE), cleaning and disinfecting all surfaces and equipment, and cleaning and disinfecting reusable devices in the perioperative suite. The use of disposable equipment only is recommended for infected patients (CDC, 2010a). Additionally, clinical practice guidelines from the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA) recommend that in a setting in which there is an outbreak or an increased rate of C. difficile infection to instruct visitors and healthcare workers caring for patients who are infected with C. difficile to wash their hands with soap, or antimicrobial soap and water. Further, they recommend that the frequency and duration of antimicrobial therapy be minimized and to implement an antimicrobial stewardship program (Cohen et al, 2010).
Mycobacterium Tuberculosis.
M. tuberculosis is a non–spore-forming, nonmotile, aerobic bacillus. These bacilli can infect almost any tissue—skin, bones, kidney, lymph nodes, intestinal tract, and fallopian tubes—and cause tuberculosis (TB). Tubercle bacilli spread in the host through the lymphatic channels and bloodstream and by way of the alveoli and GI tract. Tuberculosis is responsible for more than 3 million deaths a year and is the most opportunistic infection associated with human immunodeficiency virus (HIV) (Neil, 2008).
Tubercle bacilli are transmitted directly by means of discharge from the respiratory tract (e.g., by inhalation of droplets expelled during coughing or kissing) and, less frequently, through the digestive tract. M. tuberculosis is carried via airborne droplet nuclei when infected people sneeze, cough, or speak. These nuclei particles are less than 5 micrometers in size and contain up to three bacteria, allowing them to be kept airborne for a prolonged period (CDC, 2006a). Infection occurs when individuals inhale these infected droplet nuclei. The droplet nuclei travel through the nasal passages, upper respiratory tract, and bronchi to reach the lung alveoli, where they are engulfed by macrophages and spread throughout the body. Symptoms of TB are dependent on the part of the body infected, but may be vague at the onset and include fever, fatigue, loss of appetite, cough, fever, and night sweats.
Generally 2 to 10 weeks after initial infection, an immune response limits additional multiplication and spread of the bacilli. Some of the bacilli can remain dormant for many years, however, a condition referred to as latent tuberculosis infection. Individuals with latent TB usually have positive purified protein derivative (PPD) results, but they exhibit none of the symptoms of active TB and are considered noninfectious. The probability that a person who is exposed to M. tuberculosis will become infected depends on the concentration of infectious droplet nuclei and the duration of exposure. When infected, an individual has approximately a 10% risk of developing active TB in his or her lifetime. The risk is greatest during the first 2 years after initial exposure. In individuals with compromised immune systems, there is a higher risk that latent TB will progress into active TB (CDC, 2006a). Latent TB is treated with isoniazid or rifampin. Active TB is treated with a combination of medications (e.g., isoniazid, rifampin, ethambutol, pyrazinamide) (Neil, 2008).
Viruses.
Viruses are classified as small particles rather than living cells because viruses have no metabolic activity and must receive all sustenance for survival from a host cell. Viral pathogens are transmitted via the oral and respiratory tracts (e.g., poxvirus, rhinovirus), the intestinal and urinary tracts (e.g., poliovirus, hepatitis A virus, hepatitis E virus), and the genital tract (e.g., herpes simplex 2, HIV) and through blood and some blood products (e.g., HIV, hepatitis B virus, hepatitis C virus, hepatitis D virus, and others). Some viruses have multiple routes of transmission.
When a virus invades a host cell, it combines with the host cell's nucleic acid (deoxyribonucleic acid [DNA] or ribonucleic acid [RNA]) and reprograms the host cell metabolism to accommodate virus replication. Virus replication stimulates the antibody defense in the host. The presence of viruses may be detected by identifying the virus-specific antibodies that are produced by the infected individual's immune system, by detecting the antigens elaborated by the virus and present in the blood, or by growing a culture of the virus itself. Detection of virus-specific antibodies or antigens is termed seropositivity, or seroconversion. Viruses are susceptible to destruction by high-level disinfection—a process that destroys most disease-producing microorganisms.
Hepatitis.
Hepatitis is defined as a widespread inflammation of the liver, often caused by a virus. It can be acute or chronic. Symptoms include hepatomegaly, right upper quadrant abdominal pain, and jaundice. Hepatitis is a frequently reported viral disease in the United States, with six identified strains (A through E and G).
Hepatitis A virus (HAV) is the causative agent of what is referred to as infectious hepatitis. HAV is spread through the fecal-oral route and can be prevented by proper hand hygiene. It is very common in underdeveloped countries with poor sanitation practices. HAV constitutes slightly less than half of all hepatitis cases reported in the United States and usually has an uneventful course and recovery. Immunization against HAV is available and has helped decrease the incidence of this strain of hepatitis.
Hepatitis B virus (HBV) is the causative agent for what is sometimes referred to as serum hepatitis. This worldwide virus has reached near-epidemic levels but is preventable via vaccination and the strict use of Universal Precautions. HBV can be transmitted by blood or body fluids, such as serum, saliva, semen, and vaginal fluids. It has an incubation period of 6 weeks to 4 months, depending on the type and amount of exposure. Almost 30% of infected individuals are asymptomatic; individuals with symptoms experience fatigue, abdominal pain, nausea, vomiting, and jaundice. Chronic hepatitis infection leads to progressive liver disease and possible death and is a leading cause of liver transplants. Most adults with HBV recover without incidence and develop immunity, but a small percentage may not develop immunity and will become carriers of the virus. HBV vaccine is recommended, by government regulation, for healthcare workers who may be exposed to blood and body fluids. Postexposure prophylaxis for percutaneous or permucosal exposure to HBV depends on the vaccination status of the healthcare worker. The risk of a susceptible person developing clinical HBV ranges from 1% to 31%, depending on the source person's HBV status (Davenport and Myers, 2009). The Occupational Safety and Health Administration (OSHA) mandates that all healthcare workers report blood and body fluid exposures, and perioperative personnel should follow the institutional protocol for exposure reporting.
Hepatitis C virus (HCV) transmission is most often associated with direct exposure to blood (e.g., via intravenous or subcutaneous routes) or other infectious materials. The average incubation period is 7 weeks. Currently there is no vaccine for the prevention of HCV. Individuals who contract HCV infection are prone to develop chronic infection. A high proportion of this population is asymptomatic and develops chronic liver disease, such as cirrhosis, hepatocellular carcinoma, and portal hypertension. As with HBV, a small proportion of individuals infected with HCV die. Prevention of HCV requires strict use of Universal Precautions, implementation of and adherence to sharps safety programs, compliance with exposure reporting protocols, and completion of a postexposure treatment plan. It is crucial that all perioperative personnel be educated regarding the risk for and prevention of all bloodborne infections, including HCV. These education programs should be conducted routinely and updated for accurate information inclusive of the Centers for Disease Control and Prevention (CDC) guidelines. The risk of HCV transmission after percutaneous exposure is about 1.8% (Davenport and Myers, 2009).
Hepatitis D virus (HDV) requires the coinfection of HBV. HDV is transmitted by blood and body fluids. The incubation period is generally 14 to 56 days. Individuals immunized against HBV have protection against contracting HDV. HDV is suspected when individuals present with HBV superinfection. Treatment is supportive for acute infections. With chronic HDV infections, antiviral therapies and liver transplants may be required.
Hepatitis E virus (HEV) is spread by the fecal-oral route. HEV is similar to HAV in the transmission and disease process. Most outbreaks have occurred in developing countries, where fecally contaminated water is found to be the source. The incubation period is 15 to 64 days. No vaccine is available for the prevention of HEV. Prevention is similar to that for HAV, with the use of proper hand hygiene. Treatment for HEV is supportive and the infection generally resolves on its own. The prevalence of HEV in the United States is low.
Hepatitis G virus is a bloodborne pathogen, with a comparable rate of infection as that of HCV in units of banked blood. It is spread in a manner similar to HBV and HCV dissemination, by percutaneous or permucosal exposure. There is no vaccine to prevent hepatitis G virus infection. Similar to HBV and HCV, prudent and consistent use of barrier protection (e.g., Standard Precautions) is recommended. There are no concrete data at present on the chronic nature of this viral disease (CDC, 2006a).
Human Immunodeficiency Virus.
HIV is a retrovirus (e.g., a virus that carries genetic information in the RNA instead of the DNA) that attacks the immune system by destroying T-helper lymphocytes. Healthy individuals generally have 800 to 1000 T-helper cells/mm3 in their blood. Infection with HIV reduces these numbers, and when a person is HIV positive and his or her T-cell count falls below 200 cells/mm3 (or the person is HIV positive and is diagnosed with an atypical or opportunistic infection and/or cancer), a diagnosis of acquired immunodeficiency syndrome (AIDS) is made (Sampson and Workman, 2013). The period from HIV exposure to actual development of AIDS has been reported to be 12 years or longer. During this time the individual is a carrier of the virus. HIV has been isolated from all body fluids (e.g., blood, semen, vaginal secretions, saliva, tears, breast milk, cerebrospinal fluid, amniotic fluid, and urine) of infected individuals. Symptoms that result from HIV infection may be attributed to other causes and include fever, night sweats, chills, muscle aches, and headache.
Occupational exposure to HIV can occur by percutaneous injury (e.g., a needlestick or cut with a sharp instrument) or by mucous membrane or nonintact skin (e.g., exposed skin that is abraded) exposure to blood, tissue, or other body fluids that are potentially infectious. The CDC estimates that there is an average transmission rate of 0.3% per sharps injury (CDC, 2006b). Mucous membrane exposure is associated with a 0.09% transmission risk, and exposure through nonintact skin is probably lower (Davenport and Myers, 2009). If exposure occurs, appropriate postexposure management should take place as a part of the workplace safety program. The source patient should be informed of the incident. Serology testing should be done according to institutional policy and governmental requirements. Policies should be established for instances in which source patient consent cannot be obtained. The healthcare worker should be counseled about the risk of infection, and he or she should be evaluated clinically and serologically for evidence of HIV infection as soon as possible after exposure. Postexposure prophylaxis (PEP) regimens (Table 4-1) should be supervised by an expert in the management of exposures; follow-up should be provided for adherence to PEP; and adverse events, including seroconversion, should be monitored. Timely postexposure management and administration is important, and an exposure incident should be treated as an urgent medical condition.
TABLE 4-1
Postexposure Prophylaxis (PEP) for Occupational HIV Exposure*
| Basic PEP | Expanded PEP |
| Two drugs: zidovudine plus lamivudine or emtricitabine; or stavudine plus lamivudine or emtricitabine; or lopinavir/ritonavir | Three or more drugs: zidovudine plus lamivudine or emtricitabine plus lopinavir/ritonavir; or stavudine plus lamivudine or emtricitabine plus lopinavir/ritonavir; or tenofovir plus lamivudine or emtricitabine plus lopinavir/ritonavir |
| Small-volume (few drops) exposure to mucous membranes or nonintact skin Source patient is HIV positive, asymptomatic with known low viral load Source patient is HIV positive, symptomatic or AIDS with high viral load |
Large-volume (major splash) exposure to mucous membranes or nonintact skin Source patient is HIV positive, symptomatic or AIDS with high viral load |
| Large-volume (major splash) exposure to mucous membranes or nonintact skin Source patient is HIV positive, asymptomatic with known low viral load Optional when source patient is unknown or known with HIV factors |
Less severe percutaneous injury (solid needle or superficial injury) Source patient is HIV positive, symptomatic or AIDS with high viral load |
| Less severe percutaneous injury (solid needle or superficial injury) Source patient is HIV positive, asymptomatic with known low viral load Optional when source patient is unknown or known with HIV factors |
More severe percutaneous injury (large-bore hollow needle, deep punctures, visible blood on device, needle was in patient's artery or vein) Source patient is HIV positive, asymptomatic with known low viral load Source patient is HIV positive, symptomatic or AIDS with high viral load |
AIDS, Acquired immunodeficiency syndrome; HIV, human immunodeficiency virus.
*PEP should be supported by policies and procedures that include sharps management, exposure documentation, and a procedure for staff members to follow if a percutaneous needlestick occurs. The key to any PEP plan is for the staff member to be able to undergo assessment as soon as possible after exposure so that if the administration of antiretroviral medication is recommended that the staff member is able to begin taking them before 72 hours to prevent the virus from rapidly replicating within the body. Medications need to be taken for 28 days.
Modified from Sampson JG, Workman ML: Care of patients with HIV disease and other immune deficiencies. In Ignatavicius DD, Workman ML, editors: Medical-surgical nursing: patient-centered collaborative care, ed 7, St Louis, 2013, Saunders.
Multidrug-Resistant Organism Infections: Emerging and Resurging Organisms
Historical Perspective.
The first antibiotics, sulfonamides, were introduced in 1935 to treat staphylococcal and streptococcal infections. Penicillin was introduced in the 1940s during World War II, followed by the discovery of streptomycin in the mid- to late 1940s. Although the introduction of these medications was viewed as miraculous and did save many lives, in the mid-1940s strains of previously susceptible microorganisms began showing resistance to some of these medications. During the 1960s, cephalosporin and semisynthetic penicillins were developed, and it was believed that the problem of healthcare-associated infections was solved. Many gram-negative bacteria were found to be susceptible to these new drugs. By the late 1970s, however, strains of S. aureus resistant to penicillin, methicillin, cephalosporins, aminoglycosides, clindamycin, erythromycin, and other antibiotics were isolated in hospital outbreaks of infection. This situation led to the development of new antibiotics, including carbapenems, cephamycins, and fluoroquinolones. These broad-spectrum antimicrobials were bactericidal at low concentrations and led to a false sense of euphoria during the 1980s, when it was believed that, at last, resistance to these antimicrobials would be impossible (Sandlin, 2008).
In the 1990s it became evident that the potential for resistance to any and all antimicrobials existed as healthcare-associated outbreaks of multidrug-resistant tuberculosis (MDR-TB) occurred. Significant outbreaks of methicillin-resistant Staphylococcus aureus (MRSA) (Surgical Pharmacology) and vancomycin-resistant Enterococcus (VRE) also occurred in the 1990s. More recently Klebsiella, a gram-negative bacteria associated with surgical site infection, has shown resistance to the carbapenems (CDC, 2010b). In some cases, organisms seemed to acquire resistance almost immediately on exposure to the particular antibiotic. As fewer new antimicrobial drugs are being developed by the pharmaceutical industry, many consider the 1990s to be the beginning of the postantibiotic era (Sandlin, 2008) (see Surgical Pharmacology).
Surgical Pharmacology
Treating Methicillin-Resistant Staphylococcus aureus (MRSA)
| In the past, vancomycin was the only alternative for treating MRSA. With the increased use of vancomycin, however, strains of S. aureus with reduced susceptibility to vancomycin began to appear. Newer antibiotics include daptomycin, linezolid, teicoplanin, and quinupristin-dalfopristin. |
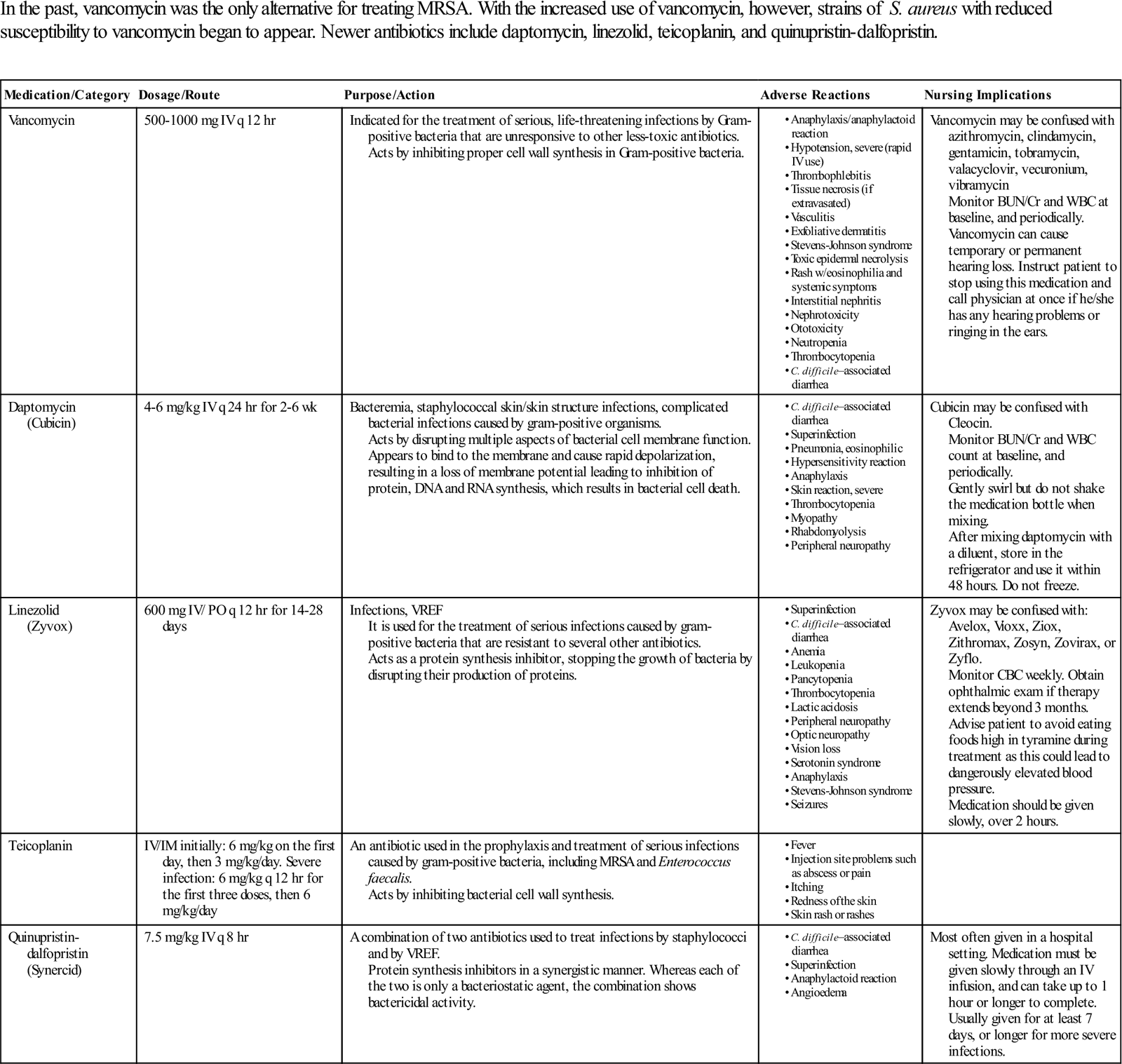
BUN, Blood urea nitrogen; CBC, complete blood count; Cr, creatinine; DNA, deoxyribonucleic acid; IV, intravenous; IM, intramuscular; PO, by mouth; RNA, ribonucleic acid; VREF, vancomycin-resistant Enterococcus faecium; WBC, white blood cell.
Modified from http://online.epocrates.com. Accessed December 13, 2012; http://www.nhs.uk/medicine-guides/pages/MedicineSideEffects.aspx?condition=Bacterialinfections&medicine=Teicoplanin&preparation=. Accessed December 13, 2012.
Mechanisms of Resistance.
Similar to their unwilling human hosts, pathogenic bacteria have an instinct to survive. When faced with an antimicrobial attack, they assemble their defensive resources. These microorganisms have the remarkable capability to incapacitate the threatening antibiotic and to mutate and outwit the most lethal clinical weapons. Although carrying only a single chromosome, bacteria by nature have extra minichromosomes called plasmids. These plasmids are hearty, and some may survive even the most aggressive antibiotic attack. Surviving plasmids are resistant and reproduce in kind, creating dominant organisms within the host. In addition, incompletely or ineffectively treated infections create the potential for reinfection with resistant organisms (CDC, 2006a).
Microbial resistance can be divided into three categories: (1) presence of a naturally resistant strain of an organism before any drugs are administered, (2) acquisition of a drug-resistant strain from an external source, and (3) drug resistance from treatment-related causes. Occurrence of naturally resistant strains of organisms without any exposure to drugs can occur by intrinsic resistance, genetic mutation, or transfer of genetic material. Some microorganisms possess genes that make them resistant to an antibiotic. These genes may always be present in the microorganism but remain in an inactive stage until challenged by an antibiotic. Genetic mutation can occur spontaneously in the course of rapid multiplication of the microbe. These antibiotic-resistant mutants reproduce within the host. New genetic material also can be transferred into bacteria by means of free DNA that contains resistant genes (CDC, 2006a).
Introduction of drug-resistant microorganisms can occur through a person or an inanimate object. Bacteria become mobile and accessible to humans on the hands or clothing of care providers, through instruments and procedures, or through food. Resistant microorganisms may travel from distant parts of the world by means of infected travelers. Surgical instruments that have been ineffectively cleaned and processed can contribute to the spread of resistant organisms. Use of antibiotics in agriculture and in animal feed products also has contributed to the development of resistant organisms. When the food is ingested, resistant genes may be spread to humans (CDC, 2006a).
Drug resistance from treatment-related causes is often the result of misuse (e.g., incorrect use, overuse, or underuse) of antibiotics. It is believed that 50% of all antibiotic use in the United States can be characterized by misuse in one form or another, and efforts to reduce surgical site infections include appropriate prophylactic antibiotic use in surgical patients (Evidence for Practice). It is estimated that half of all antibiotic prescriptions written are not warranted. During antibiotic therapy, the patient may have had a few resistant organisms. By natural selection, as the susceptible organisms are killed, the resistant organisms multiply and become predominant. Inadequate drug therapy may contribute to the phenomenon and may be the fault of the patient, the provider, or both. Failure to perform sensitivity testing along with inappropriate dosing can contribute to resistance. If the patient is noncompliant with the prescribed regimen or discontinues the drug prematurely, he or she may also be contributing to drug resistance (CDC, 2012a).
Perioperative Considerations
Methicillin-Resistant Staphylococcus aureus (MRSA).
MRSA infections, which occur in almost 130,000 cases in hospitals each year, lead to more than 5500 deaths annually (Ignatavicius, 2013). Common sites in which MRSA can be found include wounds (e.g., burns, surgical incisions, abrasions), chest tubes, intravenous catheter tips, intraabdominal abscesses, nasal passages, and the groin. S. aureus is one of the most frequently isolated organisms in postoperative surgical site infections. Surgical patients at risk for the development of MRSA infection include high-risk patients with underlying disease, patients who have a prolonged hospitalization, patients who have had previous antimicrobial therapy, patients who receive care in an intensive care unit, and patients who have been in proximity to another patient colonized with MRSA (Ignatavicius, 2013). Patients may present with unrecognized colonization or become infected by caregivers who carry MRSA in their nose or on their skin. The ambient environment is rarely a source of MRSA except for some reported cases in burn units.
The primary mode of transmission for MRSA is most likely direct contact transmission from the hands of healthcare personnel. The organism has been recovered from the hands of personnel after they touched contaminated material and before they washed their hands. It also has been shown that MRSA can be carried in the nares of personnel and transferred to patients by hand contact. The importance of hand hygiene cannot be overemphasized (AORN, 2013a; CDC, 2008a).
Because MRSA is transmitted by contact, perioperative protocols should be used when caring for these patients. They should include the following:
• Segregate the patient, using Contact Precaution guidelines.
• Wear a gown and gloves whenever there is potential for contact with contaminated fluids or materials.
• Implement strict hand hygiene practices.
• Limit patient transportation to essential movement only.
• Clean and disinfect patient care equipment as close as possible to the time of use (AORN, 2013c).
In healthcare facilities, MRSA causes serious and sometimes fatal infections, including necrotizing fasciitis. MRSA resists almost every antibiotic except intravenous vancomycin. One treatment for resistant MRSA is tigecycline (Tygacil). Tigecycline is an antibiotic related to the tetracycline family that was first used in 2005. This drug is formulated to retain activity against some bacteria that are resistant to tetracyclines and provides an expanded broad spectrum of activity for patients with complicated SSIs. Minocycline and doxycycline are also used in the treatment of MRSA.
Vancomycin-Resistant Enterococci.
Enterococci are intrinsically resistant to cephalosporins, semisynthetic penicillins, and clindamycin. Some strains also have acquired resistance to erythromycin, chloramphenicol, tetracycline, fluoroquinolones, and vancomycin (CDC, 2006c). The incidence of VRE infections in the United States has increased rapidly in recent years and now is difficult or impossible to treat. Patients at the greatest risk of VRE infection are those with a prolonged stay in the critical care unit, intraabdominal or cardiothoracic surgical procedures, presence of indwelling central venous catheters, presence of urinary catheters, extended hospital stays, multiple antimicrobial therapies, and proximity to infected individuals. Because the enterococcal plasmid carrying the resistant gene can be transferred to organisms such as S. aureus, VRE infections are of serious concern (CDC, 2006c; Droboni et al, 2009).
The CDC has issued recommendations regarding the use of vancomycin, including identifying situations in which vancomycin should and should not be used; establishing education programs for practitioners, susceptibility testing, isolation procedures, and dedication of equipment and devices; verifying procedures for cleaning and disinfecting the environment; and minimizing movement of personnel between patients with and without VRE (CDC, 2006c; Droboni et al, 2009).
VRE can be transmitted directly from patient to patient, through the hands of healthcare providers, or through contact with contaminated environmental surfaces and equipment used for patient care. As with MRSA, Contact Precautions should be followed when caring for patients infected with VRE. Perioperative protocols are similar to protocols for MRSA patients and should include the following:
• Segregate patients, using Contact Precaution guidelines.
• Wear a gown and gloves when in contact with contaminated materials.
• Implement strict hand hygiene practices.
• Limit patient transportation to essential movement only.
• Clean and disinfect patient care equipment as close as possible to the time of use (AORN, 2013c; CDC, 2007a).
Germicides commonly used for cleaning and disinfecting in hospitals are effective against VRE. Isopropyl alcohol and sodium hypochlorite (bleach) are highly effective. With a 10-minute exposure time, some phenolic and some quaternary ammonium compounds also are effective. These are less effective at shorter exposure times. Hydrogen peroxide has been found to be ineffective. No relationship seems to exist between microbial resistance to antibiotics and increased resistance to germicides (CDC, 2008a). Perioperative personnel should routinely consult the manufacturer's written instructions and follow Environmental Protection Agency (EPA) recommendations when selecting germicides for surface cleaning (AORN, 2013d).
Vancomycin Intermediate–Resistant Staphylococcus aureus.
In 1996 the first known incidence of vancomycin intermediate–resistant S. aureus (VISA) occurred in Japan when the organism was isolated from a surgical site infection and undrained abscess. The first occurrence of the organism in the United States was seen 1 year later. A patient that had been treated with vancomycin for multiple episodes of peritoneal MRSA developed bacterial strains moderately resistant to vancomycin, which was the treatment of choice for MRSA. Although the organism showed only intermediate levels of resistance, the CDC viewed this occurrence as an early warning that strains of S. aureus with full resistance to vancomycin may emerge (CDC, 2006c). Vancomycin is ineffective for the treatment of vancomycin-resistant S. aureus (VRSA) and VISA.
Multidrug-Resistant Mycobacterium tuberculosis (MDR-TB).
Outbreaks of TB have heightened concern about healthcare-associated transmission of this disease. Transmission is most likely to occur from patients with unrecognized pulmonary or laryngeal tuberculosis and those who do not take their TB medication regularly. Populations at greatest risk of developing TB or MDR-TB are the elderly, indigent, minorities, immigrants from countries where TB and MDR-TB are prevalent, and HIV-infected individuals (CDC, 2006b). Transmission also occurs as a result of procedures such as bronchoscopy, endotracheal intubation, endotracheal suctioning, and open abscess irrigation, inclusive of inadequate equipment disinfection. Extensively drug-resistant TB (XDR-TB) is now a threat in many Asian countries and the former Soviet Union. This strain of TB is very difficult to treat because of its resistance to the primary medications used to treat TB (e.g., isoniazid and rifampin) as well as many of the secondary medications used (e.g., fluoroquinolone, amikacin, kanamycin, and capreomycin) (Neil, 2008).
The CDC's Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Health-Care Settings emphasize the following:
• Importance of control measures, including engineering controls and personal respiratory protection, including fit-tested, personal respirators when indicated
• Use of risk assessment to develop a tuberculosis-control plan
• Early detection and treatment of patients with tuberculosis
• Screening programs for healthcare workers
• Training and education for healthcare workers
• Evaluation of the tuberculosis-control program (CDC, 2005)
Prions: Creutzfeldt-Jakob Disease.
Creutzfeldt-Jakob disease (CJD) is an infectious, human prion disease that is a fatal neurodegenerative disease of the central nervous system. CJD is one of a group of encephalopathies known as transmissible spongiform encephalopathies (TSEs). Other human forms of TSE are Gerstmann-Sträussler-Scheinker syndrome and new variant CJD (nvCJD) or variant CJD (vCJD) (CDC, 2008b).
CJD is caused by a self-replicating prion. Prions are a unique class of organisms that have no detectable DNA or RNA. These small proteinaceous agents are abnormal isoforms of normal cellular proteins. The incubation period for CJD varies from months to years to decades. Symptoms include rapidly progressing dementia, memory loss, rapid physical and mental deterioration, and a distinctive electroencephalogram reading. Positive diagnosis can be made only by direct examination of affected brain tissue. Most cases occur randomly and for unknown reasons when the patient is between 50 and 75 years old. Death typically occurs within 1 year of symptom onset (CDC, 2012b). In contrast, vCJD has an earlier onset (between 18 and 41 years of age). Patients exhibit initial psychiatric symptoms and then neurologic symptoms differing from those of CJD, and the course of illness averages 14 months. The disease is always fatal (CDC, 2008b). According to the CDC, there is strong epidemiologic and laboratory evidence to support a causal association between vCJD and bovine spongiform encephalopathy (also known as mad cow disease).
CJD can be familial (i.e., inherited in the form of a mutant gene) or sporadic (no family history and no known source of transmission). Approximately 90% of cases are sporadic. Only about 1% of cases result from person-to-person transmission, and those are primarily the result of iatrogenic (medically related) exposure. Exposures have occurred via transplantation of contaminated central nervous system tissue, such as dura mater or corneas, from injections of pituitary hormone extracts, and by use of contaminated surgical instruments or stereotactic depth electrodes (CDC, 2008b).
CJD and other TSEs are unusually resistant to conventional chemical and physical decontamination methods. The causative prions are resistant to steam autoclaving, dry heat, ethylene oxide gas, and chemical disinfection with formaldehyde or glutaraldehyde as normally used in the healthcare environment (AORN, 2013e). Glutaraldehyde and formaldehyde act as fixatives, causing the prions to become more stable and less susceptible to normal sterilization/disinfection protocols. Special protocols for instrument care after exposure to prions should be followed (AAMI, 2012). Some institutions use disposable instrument sets for diagnostic brain biopsies to rule out CJD or TSE. Processes being investigated for cleaning and sterilizing devices contaminated with prions include the use of an alkaline cleaning agent and vaporized hydrogen peroxide (VHP) sterilization. Protocols for handling CJD are evolving as researchers learn more about prions and their destruction. Table 4-2 lists options from which an acceptable protocol for care of instruments and equipment exposed to the CJD prion can be developed.
TABLE 4-2
Care of Items Exposed to the Creutzfeldt-Jakob Disease Prion
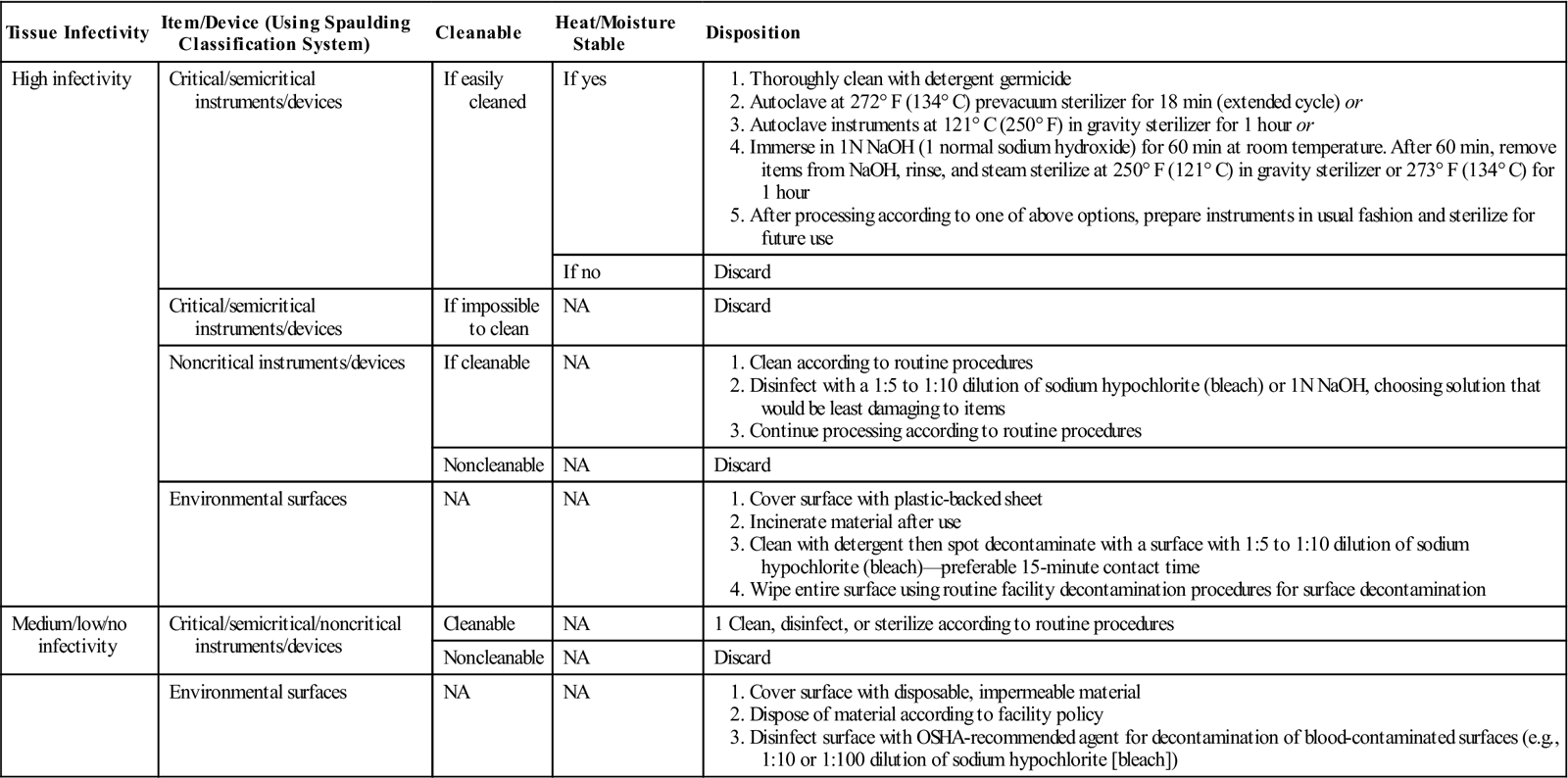
Research into processing of medical devices contaminated with prions is ongoing, and institutional policies relating to processing of prion contaminated devices should periodically consult the World Health Organization and the Centers for Disease Control to determine if recommendations have changed.
NA, Not applicable; OSHA, Occupational Safety and Health Administration.
Modified from Rutala W, Weber D: Guideline for disinfection and sterilization of prion-contaminated medical instruments, Infect Control Hosp Epidemiol 31(2), 2010.
Preventing Infection
Asepsis
The term asepsis means the absence of infectious organisms. Asepsis is directed at cleanliness and the elimination of all infectious agents. Surgical asepsis is designed to exclude all microbes, whereas medical asepsis is designed to exclude microbes associated with communicable diseases. Practices that restrict microorganisms in the environment and on equipment and supplies and that prevent normal body flora from contaminating the surgical wound are termed aseptic techniques. The goal of each aseptic practice is to optimize primary wound healing, prevent surgical infection, and minimize the length of recovery from surgery. For perioperative practitioners, surgical aseptic principles and practices are the foundation for infection control efforts in the perioperative arena.
Infection control practices should primarily focus on prevention. Transmission of infection involves a chain of events, including the presence of a pathogenic agent, reservoir, portal of exit, transmission, portal of entry, and host susceptibility. Prevention occurs when there is a break in the chain of transmission. Infection control practices involve personal and administrative measures. Personal measures should include fitness for work and application of aseptic principles. Administrative measures should include provision of adequate physical facilities, appropriate surgical supplies, and operational controls in the perioperative area. Standards for inpatient and outpatient settings should be established and followed (Ambulatory Surgery Considerations).
As new organisms emerge, and known organisms become more resistant, the importance of patient education as a risk reduction strategy cannot be minimized. In addition to practicing sound infection control practices, nurses partner with the patient through effective education about infection control measures to maximize the potential for perioperative safe care (Patient and Family Education).
Universal, Standard, and Transmission-Based Precautions
Universal Precautions.
In 1985, in response to the growing number of individuals testing positive for HIV, the CDC published recommendations for the use of Universal Precautions (CDC, 2006b). Reports of hospital personnel becoming infected with HIV after a needlestick or skin exposure to a patient's blood created an urgent need for new and better measures to protect personnel from patient transmission of infection. With knowledge that many patients with bloodborne infections are undiagnosed, Universal Precautions, for the first time, placed emphasis on applying Blood and Body Fluid Precautions (previously a category in the 1993 Guidelines for Isolation Precautions in Hospitals) universally to all individuals regardless of their presumed infection status. Universal Precautions expanded the Blood and Body Fluid Precautions by recommending masks and eye protection to prevent mucous membrane exposures in addition to the routine use of barrier protection, such as gowns and gloves. Universal Precautions also emphasized the prevention of needlestick injuries and the use of ventilation devices when resuscitation was performed. The CDC continued to recommend the use of Universal Precautions until 1987, when a new system of isolation called Body Substance Isolation (BSI) was proposed. BSI directed isolation of all moist and potentially infectious body substances (e.g., blood, feces, urine, sputum, saliva, wound drainage, other body fluids) for all individuals regardless of their infection status. This was accomplished primarily with the use of gowns and gloves. Because of the similarities yet differences between Universal Precautions and BSI, confusion reigned. In 2007 the CDC further expanded the Guidelines for Isolation Precautions.
OSHA issued a rule for Occupational Exposure to Bloodborne Pathogens to address workplace safety. This document was based on the concept of Universal Precautions. OSHA subsequently revised its final rule to include a special focus on needlestick and other percutaneous exposures to bloodborne pathogens (OSHA, 1991). The revised rule addressed “sharps with engineered sharps injury protections” (non-needle devices or needle devices with built-in safety features to reduce the potential for injury) and “needleless systems” (alternative delivery devices using blunt cannulae, catheter ports or connectors, or other non-needle devices). Other revisions focused on the review and updating of exposure control plans, solicitation of employee input, and improved record keeping. The following is a summary of the requirements of the Final Rule: Occupational Exposure to Bloodborne Pathogens (OSHA, 2001):
1. Each facility must develop and implement an exposure control plan that defines exposure and implements the requirements of the final rule. This plan is to be reviewed and revised annually with information provided to all employees. The plan must reflect changes in available technology to reduce exposure to bloodborne pathogens and implementation of appropriate technology to that end. Nonmanagerial employee input must be solicited in selecting technology to be implemented in the practice setting.
2. Engineering and work-practice controls must be used to eliminate or minimize employee exposure. Examples follow:
a. The employer must provide everything necessary for proper hand hygiene.
b. Contaminated needles must not be recapped or removed unless such action is required by a specific medical procedure. Such recapping or removal must be accomplished by the use of a mechanical device or one-handed technique.
c. A clamp or other mechanical device should be used to disassemble a knife blade and handle.
d. Sharps are to be placed in labeled or color-coded, puncture-resistant, leakproof containers for disposal.
e. Specimens of blood or body fluids must be placed in containers that prevent leakage and are labeled or color-coded. Warning labels must be affixed to containers of regulated waste, refrigerators and freezers containing blood or potentially infectious materials, and other containers used to transport blood or potentially infectious materials (Figure 4-2). The labels must be fluorescent orange or orange-red.
f. Food and drink are not to be kept in the same storage area where blood or other potentially infectious materials are present.
g. PPE must be provided by the employer at no cost to the employee. Appropriate PPE shall include but is not limited to gloves, gowns, face shields or masks, and eye protection. Protective eyewear must have solid side shields. Gloves are to be worn when contact with blood or body fluids is anticipated. Disposable gloves are to be replaced as soon as possible after contamination occurs. Disposable gloves are not to be washed or decontaminated for reuse. Some facilities may have educational signs posted to assist employees in recognition of appropriate PPE (Figure 4-3).
h. Signs must be posted at the entrance to work areas of potential contamination. These signs are to bear the biohazard legend with the following information: name of infectious agent, special requirements for entering the area, and name and telephone number of the responsible individual.
i. Housekeeping provisions are to ensure that the workplace is clean and sanitary. A written schedule for cleaning and a method of decontamination must be established. All equipment and working surfaces must be cleaned and decontaminated after contact with blood or other potentially infectious materials.
j. Contaminated laundry must be placed in a labeled or color-coded container that is recognized by all employees.
k. All employees are to receive education and training about safe handling of hazardous substances and materials. Information must be provided to all occupationally exposed employees at no cost to them. Individuals must receive training at the time of employment and annually thereafter. Individual employee training records are to be maintained by the employer for the duration of employment plus 30 years. The healthcare worker is highly encouraged to receive the HBV vaccine after obtaining the required information about the risk of exposure and about the vaccine. If the employee chooses not to accept the vaccination, the employer must have the employee sign a letter of declination.
l. Employees should report all exposures to blood and body fluids for postexposure evaluation.
m. Employers who are required to maintain a log of occupational injuries and illnesses must maintain a sharps injury log that acts as a tool for identifying high-risk practice areas and for evaluating various devices in use. This log must protect the confidentiality of the injured employee. Log information should include the type and brand of the device, the practice area in which the injury occurred, and an explanation of how the incident occurred.
The bloodborne pathogen regulation is enforceable by OSHA at the federal and state levels. This regulation is based on the concept of Universal Precautions to serve and protect healthcare providers and to minimize the transfer of pathogens from one patient to another. Surveyors for OSHA may engage in onsite visits to healthcare facilities. Unannounced visits may occur at any site where an employee exposure occurs. The visit may be a result of a verbal or written employee concern, referral from another regulatory agency, or random inspection.
Standard Precautions.
By the early 1990s the controversy regarding Universal Precautions and BSI had escalated. There was considerable confusion about which body fluids required special care under either Universal Precautions or BSI. There were also concerns about the need for additional precautions to prevent airborne, droplet, and contact transmission of other infectious agents. With this in mind, the CDC developed a single set of precautions incorporating the major features of Universal Precautions and BSI (CDC, 2007b). These precautions are called Standard Precautions, and they are designed to reduce the transmission risk of bloodborne and other pathogens. Additional precautions based on routes of transmission for patients known or suspected to be infected or colonized with highly transmissible or epidemiologically significant pathogens are included in the document.
Standard Precautions are intended to reduce the transmission of microorganisms from recognized and unrecognized sources of infection. Standard Precautions should be applied to all patients receiving care regardless of their diagnosis or presumed infection status. They are considered the first, and most important, tier of precautions and as such are a primary strategy for successful infection prevention and control. Standard Precautions apply to (1) blood, (2) all body fluids and secretions and excretions (except sweat) regardless of whether they contain visible blood, (3) mucous membranes, and (4) nonintact skin. Standard Precautions include the following:
1. Hand hygiene. Hand hygiene is the most important factor in preventing the spread of infection. Hands are to be washed whenever they are in contact with blood, body fluids, secretions, excretions, and contaminated items, whether or not gloves are worn. Hands are washed immediately after gloves are removed, between patient contacts, and when otherwise indicated to avoid transfer of microorganisms to other patients or environments. Sometimes it is necessary to wash hands between tasks and procedures on the same patient to prevent cross-contamination of different body sites. A plain (nonantimicrobial) soap should be used for routine handwashing. When special circumstances such as hyperendemic conditions occur, an antimicrobial soap or an antiseptic hand rub (waterless antiseptic agent) should be used. The hand rub antiseptic agent is most effective if the hands are clean before the antimicrobial agent is applied. For effectiveness, a sufficient amount of the agent must be used for the hand rub. Manufacturers' written instructions should be followed. An additional amount of hand rub agent may be necessary.
2. Gloves. Clean, nonsterile gloves should be worn when touching blood, body fluids, secretions, excretions, and contaminated items. Freshly donned gloves should be worn when touching mucous membranes and nonintact skin. Gloves should be changed between tasks and patient procedures and after contact with material that may contain high concentrations of organisms. Gloves should be removed immediately after use and hands washed before engaging in another task or giving care to another patient.
3. Masks, eye protection, face shields. A mask and eye protection or a face shield is to be worn at any time patient care activities are likely to generate sprays or splashes of blood or body fluids, secretions, and excretions. These protective devices help protect the mucous membranes of the nose, mouth, and eyes.
4. Gowns. Clean, nonsterile gowns are to be worn at any time patient care activities are likely to generate sprays or splashes of blood or body fluids, secretions, and excretions. Gowns help protect the skin and prevent soiling of clothing. The activity to be performed and the amount and type of fluid likely to be encountered dictate the degree of protective barrier necessary in the gown. Gowns should be removed immediately after use and hands washed before engaging in other activities or giving care to another patient.
5. Sharps. Needles, scalpels, and other sharps should be handled in a manner to avoid injury. Needles should never be recapped using any technique that directs the point of the needle toward any body part. If recapping is necessary, it should be done using a mechanical device or a one-handed scoop technique. Used needles should not be removed from disposable syringes, and they should not be bent, broken, or otherwise manipulated by hand. Used disposable sharps should be placed in puncture-resistant containers located as close as possible to the point-of-sharps use. Reusable sharps should be contained in a puncture-resistant container for transport to the point of decontamination.
6. Patient care equipment. Single-use items should be discarded after use. Reusable equipment must be cleaned and reprocessed to ensure safe use for another patient. Equipment soiled with blood, body fluids, secretions, and excretions should be handled carefully to prevent exposure of skin and mucous membranes, contamination of clothing, and transfer of organisms to patients, personnel, and the environment.
7. Linens. Linens soiled with blood, body fluids, secretions, or excretions should be handled in a manner to avoid skin and mucous membrane exposure, clothing contamination, and transfer of microorganisms to other patients, personnel, and the environment.
8. Environmental control. Adequate procedures for routine care and cleaning of environmental surfaces, beds, and associated equipment are to be developed, and the use of these procedures is monitored on a regular basis.
9. Patient placement. Patients who contaminate the environment or who are unable to maintain appropriate hygiene or environmental control are to be housed in a private room with appropriate air handling and ventilation. If a private room is unavailable, the infection control professional may determine a method for cohorting patients with similar infectious organisms (CDC, 2007b).
Transmission-Based Precautions.
Transmission-Based Precautions are the second tier of infection prevention, designed for patients known or suspected to be infected by epidemiologically important pathogens spread by airborne or droplet transmission or by contact with dry skin or contaminated surfaces. They may be used singly or in combination with one another if the patient has a disease that has multiple routes of transmission and are to be used in addition to Standard Precautions.
Airborne Precautions.
Airborne transmission occurs by dissemination of droplet nuclei from evaporated droplets that can remain suspended in the air for long periods or by dissemination of dust particles that contain the infectious agent. Droplet nuclei are particles 5 µm or smaller in size. Airborne microorganisms can be dispersed widely depending on air currents and can be inhaled by or deposited on a susceptible host. In addition to Standard Precautions, Airborne Precautions include the following:
1. Patients are to be placed in private, negative-pressure rooms. The air exchange should be at a rate of 6 to 12 exchanges per hour with air discharged to the outdoors or circulated through high-efficiency particulate-arresting (HEPA) filters before being circulated to other areas of the facility.
2. Caregivers must wear OSHA-specified respiratory protection when caring for patients with known or suspected tuberculosis. If susceptible personnel care for patients with rubeola (measles) or varicella (chickenpox), respiratory protection should be worn. If the caregiver is immune to rubeola and varicella, respiratory protection is unnecessary.
3. All precautions for preventing transmission of tuberculosis should be implemented if the patient is known or suspected to have tuberculosis.
4. A surgical mask should be placed over the patient's nose and mouth for Airborne Infection Isolation Precautions when the patient must be transported from one location to another. Patient transport should be limited to essential purposes only.
Droplet Precautions.
Droplet Precautions are used for patients known or suspected to be infected with microorganisms that are transmitted by large droplets (>5 µm). These droplets can be generated when the patient sneezes, coughs, or talks. Droplet Precautions are used in addition to Standard Precautions. Droplet Precautions include the following:
1. Patients are to be placed in private rooms when available. If this is not possible, the patient should be placed in a room with another patient who is infected with the same organism and with no other infection. If this is not possible, a 3-foot spatial separation should be maintained between the infected patient and other patients in the same room. For Droplet Precautions no special air handling is required.
2. Caregivers should wear a mask when working within 3 feet of the patient.
3. Patients should be transported only for essential purposes. When transport is necessary, a mask should be placed over the patient's nose and mouth to minimize dispersal of droplets (CDC, 2007b).
Contact Precautions.
In addition to Standard Precautions, Contact Precautions should be used for patients known or suspected to be infected or colonized with epidemiologically important organisms that can be transmitted by (1) direct contact, as occurs when the caregiver touches the patient's skin, or (2) indirect contact, as occurs when the caregiver touches patient care equipment or environmental surfaces in the patient's room. Contact Precautions include the following:
1. Patients should be placed in private rooms. If this is not possible, the patient should be placed in a room with another patient who is infected with the same organism and with no other infection. If this is not possible, patient placement must be determined on an individual basis, depending on the organism involved.
2. Gloves should be worn on entering the patient's room. Gloves should be changed after handling infective material that might contain a high concentration of microorganisms. When patient care activities have been completed, gloves should be removed before leaving the patient's room. Hands should be washed after glove removal. To avoid transferring microorganisms to others, no environmental surfaces in the patient's environment should be touched after the hands have been washed.
3. Gowns should be worn on entering the patient's room if there is a probability that the caregiver's clothing will be in contact with the patient or the environmental surfaces or if the patient is incontinent, has diarrhea, or has an ileostomy or colostomy. The gown should be removed before leaving the patient's room, and care should be exercised to avoid contact with environmental surfaces.
4. Patient transportation should be limited to essential transport only and Contact Precautions maintained to avoid contamination of personnel, visitors, or the environment.
5. Patient care equipment should be dedicated to a single patient and not be shared between patients. If this is impossible, equipment must be cleaned and disinfected thoroughly before being used for another patient (CDC, 2007b).
Perioperative staff members traditionally have relied on numerous types of precautions to protect themselves and others from bloodborne pathogens and other infectious diseases. Implementing these precautions within the surgical environment requires critical thinking skills and sound nursing judgment. Consistent application of these precautions by all members of the perioperative team serves to protect the healthcare provider and to minimize cross-infection of pathogens among patients (AORN, 2013f).
Infection Control and Bioterrorism.
The potential for bioterrorism is a reality. The CDC has identified agents that may pose a risk to the national security because of their (1) easy dissemination or transmission from person to person, (2) potential to cause high mortality and have a major public health impact, (3) potential to cause public panic and social disruption, and (4) necessity for special action for public health preparedness (CDC, 2007) (Table 4-3). Perioperative personnel should have a basic knowledge and understanding of the agents most likely to be encountered and the levels of precautions required for patient and personnel safety. Communication, collaboration, and coordination are required in the face of any bioterrorist event or natural disaster. Communities need emergency plans for such matters, such as guarding patient data and protecting information systems, linking quickly and easily with state and federal resources, ensuring culturally competent communication and care, accessing mental health resources, and identifying agencies that can partner with one another to provide services. Each institution must be compliant with The Joint Commission's (TJC) requirements for periodic testing of emergency management systems and their associated plans.
TABLE 4-3
Bioterrorism Agents*
| Agent | Transmission | Incubation | Symptoms | Management |
| Anthrax (Bacillus anthracis) | Cutaneous: Direct contact Inhalation: Droplet, aerosolization |
Cutaneous: 1-7 days Inhalation: 1-6 days on average, may be as late as 2 months |
Cutaneous: Itching, progressing to papular and vesicular lesions, eschar, edema, ulceration, and sloughing. If untreated may spread to lymph nodes and bloodstream. Fatality: 5%-20%. Inhalation: Influenza-like with progression to high fever dyspnea, stridor, cyanosis, and shock. Chest wall edema and hemorrhagic meningitis may be present. Death is universal in untreated cases and may occur in 95% of cases not treated within 24-36 hours. |
Standard Precautions. Special attention should be given to protection and containment of any draining wounds, inclusive of cutaneous lesions. |
| Smallpox (variola major/variola minor viruses) | Usually prolonged face-to-face contact is required. Also spread through direct contact with infected body fluids or contaminated objects. Rarely spread by virus carried in the air | Typically 12-14 days | High fever, malaise, head and body aches and sometimes vomiting. Rash develops over face and spreads to extremities. Rash soon becomes vesicular and then pustular. All lesions progress at same rate. Fever continues throughout course of disease, and growing and expanding pustules are very painful. Historical, variola major fatality rate is at 30%. | Standard, Droplet, Airborne, and Contact Precautions for patients with vesicular rash pending diagnosis. Avoid contact with organism while handling contaminated bedding. Wear protective attire to include gloves, gown, and N95 respirator. Vaccine does not give reliable lifelong immunity. Previously vaccinated persons are considered susceptible. After exposure, initiate Airborne Precautions and observe for unprotected contact (from days 10-17). Vaccinate within 2-3 days of exposure. One case is a public health emergency because of highly communicable nature of this disease. Consult CDC and local health agencies at earliest opportunity. |
| Plague (Yersinia pestis) | Pneumonic plague: Aerosolized bacteria carried on respiratory droplets from infected person Bubonic plague: Bites from fleas or infected rodent. Does not spread from person to person |
Pneumonic plague: 1-3 days Bubonic plague: 2-8 days |
Pneumonic: Fever, headache, and weakness, with rapid progression to pneumonia with dyspnea, chest pain, cough, and bloody or watery sputum. Respiratory failure and shock may follow within 2-4 days. Bubonic: Symptoms include swollen, tender lymph nodes (buboes), fever, headache, chills, and weakness. |
Standard and Droplet Precautions. Patients should be placed in private rooms or cohorted with “like” patients. Transport should be limited to essential movement only, and patient should wear mask during transport. Contact Precautions until decontamination is complete. |
| Tularemia (Francisella tularensis) | Bites of ticks, deerflies, and other arthropods that have eaten infected animal tissue; by handling infected animal carcasses; by eating or drinking contaminated food or water; or by inhaling infected aerosols | 1-14 days (average 3-5 days) | Depending on route of exposure, skin ulcers, inflamed eyes, sore throat, oral ulcers, swollen and painful lymph nodes, and pneumonia. When bacteria are inhaled, symptoms include sudden fever, chills, headache, muscle aches, joint pain, cough, and progressive weakness. Symptoms may progress to chest pain, dyspnea, bloody sputum, and respiratory failure. | Standard Precautions |
| Botulism (Clostridium botulinum) | Foodborne: Ingestion of preformed toxin Wound: When wounds are infected with C. botulinum that secretes toxin; injection drug users are at risk for wound botulism |
Foodborne: Within hours of ingesting food, but can occur up to 8 days later | Early symptoms of botulism include gastrointestinal distress, nausea, and vomiting. Symptoms progress to diplopia, blurred vision, drooping eyelids, slurred speech, difficulty swallowing, dry mouth, and muscle weakness. If not treated, the disease progresses to respiratory paralysis and paralysis of arms and legs. Botulinum toxin is most poisonous substance known. | Standard Precautions |
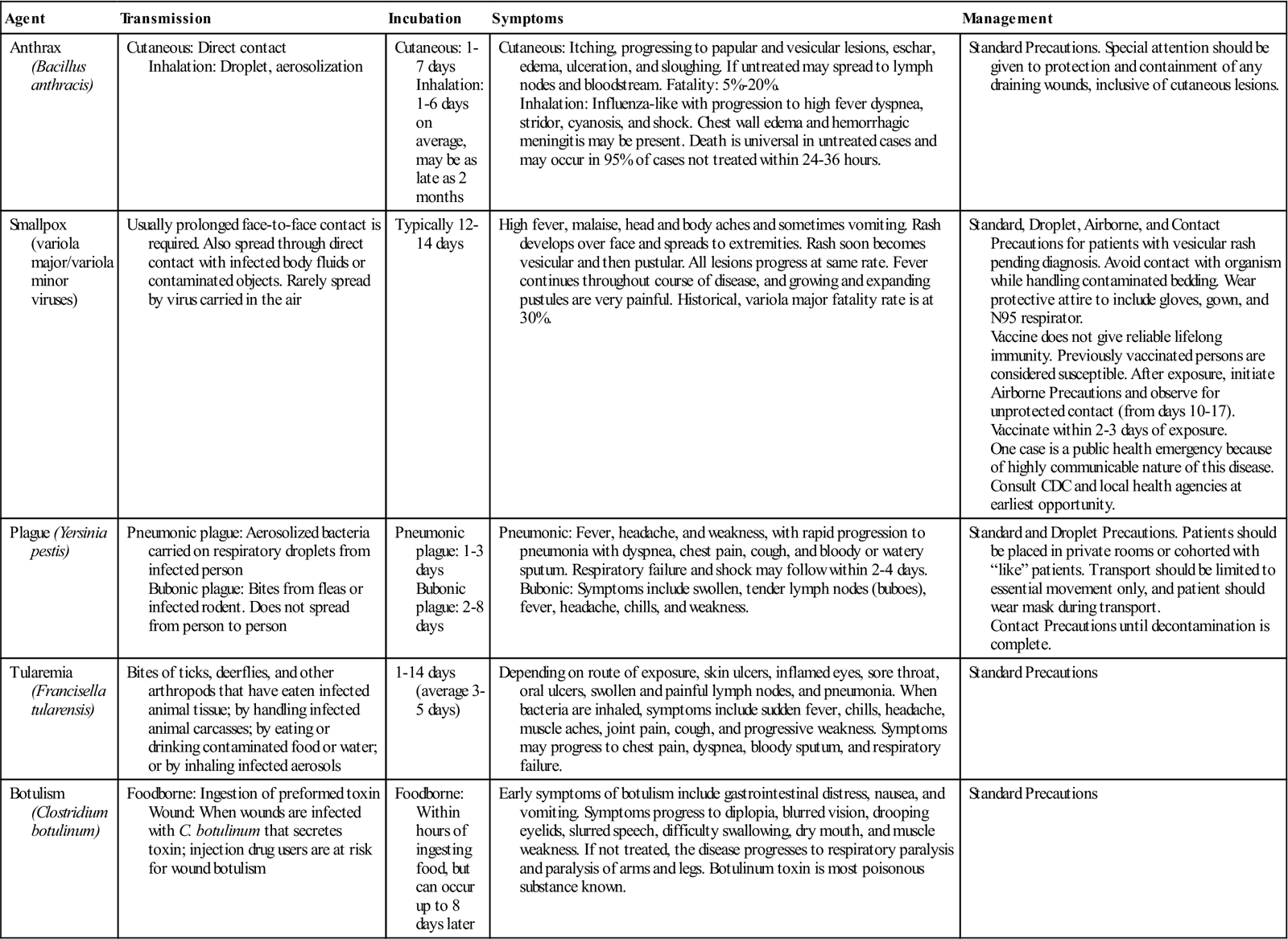
*The information about bioterrorism and the identified biologic weapons presented in this table represents current knowledge at the time of publication. The CDC updates its website with the most current information from throughout the world. Practitioners are encouraged to update their knowledge continually by consulting the CDC website (www.cdc.gov) and other experts in the field. The Health Alert Network (HAN) is the CDC's surveillance system. It connects local, state, and national public agencies with high-speed and satellite Internet access. The extent of precautions necessary can be quickly determined by using this system.
Modified from Centers for Disease Control and Prevention (CDC): Anthrax information for health care providers, available at www.bt.cdc.gov/agent/anthrax/anthrax-hcp-factsheet.asp. Accessed December 16, 2012; CDC: Smallpox overview, available at www.bt.cdc.gov/agent/smallpox. Accessed December 16, 2012; CDC: Plague overview, available at www.bt.cdc.gov/agent/plague. Accessed December 16, 2012; CDC: Key facts about tularemia, available at www.bt.cdc.gov/agent/tularemia. Accessed December 16, 2012; CDC: Facts about botulism, available at www.bt.cdc.gov/agent/botulism. Accessed December 16, 2012; Ignatavicius DD: Care of patients with infection. In Ignatavicius DD, Workman ML, editors: Medical-surgical nursing: patient-centered collaborative care, ed 7, St Louis, 2013, Saunders.
Engineering Practices to Prevent Infection
Environment of Care.
The surgical suite should be designed in such a way as to minimize and control the spread of infectious organisms. Either a central-core, race track, or a single-corridor design may be used. With the central-core design, sterile equipment and supplies should be contained within the central-core area, which is surrounded by ORs and a peripheral corridor. The single-corridor design places the ORs on either side of a single corridor, with separate storage rooms, usually along the corridor, to house sterile equipment and supplies. If a single-corridor design is used, sterile and contaminated items must be separated by either space or time. That is, sterile, wrapped, or containerized items can pass contaminated items in the corridor when the contaminated items are covered or otherwise contained.
Floors in the ORs should be hard, seamless, easily cleaned, and contiguous with the walls. This design eliminates the sharp angle where the floor and walls meet, where bacteria can become lodged and proliferate. Floors should be monolithic and joint free. Walls may be constructed of any hard surface that is easily cleaned and hard enough to withstand the impact of surgical equipment that may accidentally be pushed into the wall during transport. If ceramic tile is used, smooth-surface grouting mortar should be used. This grout provides a surface nearly as smooth as the tile itself; eliminating concerns that surface roughness may attract and retain bacteria. Painted walls are less desirable because the paint flakes and peels, particularly in areas of higher humidity. If a hard-finish epoxy paint is used, it is only as good as the surface beneath it. Equipment banged into a wall may cause damage and expose construction material to the environment. A soft-colored, matte-finished wall may be preferred to reduce reflectance and glare (AIA, 2010).
Doors in the ORs may swing or slide. If sliding doors are used, they should not recess into the wall but should slide over the adjoining wall to facilitate housekeeping. Cabinets should be recessed into the wall if possible. This configuration allows for maximum use of open floor space in ORs. Size and configuration of ORs are discussed in detail by the American Institute of Architects (AIA)/Academy of Architecture for Health. Stainless-steel cabinets are preferred because the surfaces remain smooth and are easily cleaned. Wooden cabinets quickly become damaged with cracks and crevices where bacteria can collect and proliferate. Wooden cabinets are difficult to clean and disinfect and should be avoided in ORs. Cabinet doors may be of either the swinging or the sliding type. A cleaning protocol should be established for the tracks if sliding doors are used. For noncabinet shelving, open wire shelves are preferred because dust and bacteria do not accumulate, and air can circulate freely around shelf contents (AIA, 2010).
Scrub sinks should be located adjacent to each OR, with a single area serving two ORs if possible. Ideally, scrub sinks are located in a room or alcove adjacent to the peripheral or single corridor of the OR. Scrub sinks should not be within the central-core area because aerosolization and splashing may occur where sterile items are stored, contaminating the environment.
Each surgical suite must contain an enclosed soiled workroom exclusive for its own use. The workroom should contain a flushing hopper, receptacles for waste and soiled linen, a handwashing sink, and a work counter. If the area is used as a holding area as part of a larger system for collection and disposal of soiled materials, the flushing hopper is not required (AIA, 2010).
Heating, Ventilation, Air Conditioning.
To control bioparticulate matter in the OR environment, ventilating air should be delivered to the room at the ceiling and exhausted near the floor and on walls opposite to those containing inlet vents. Airflow should be in a downward directional flow, moving down and through the location with a minimum of draft, to the floor and exhaust portals (AIA, 2010).
Air pressure in the OR should be greater than that in the surrounding corridor; this is called “positive pressure” in relation to corridors and adjacent areas. This positive pressure helps maintain the unidirectional airflow in the room and minimizes the amount of corridor air (less clean area) entering the OR (more clean area). Each OR should have a minimum of 15 total air exchanges per hour, with the equivalent of at least 3 replacements being of outside air to satisfy exhaust needs of the system. No recirculating devices, such as cooling fans or room humidifiers or dehumidifiers, are to be used. These units create a turbulent airflow and may recirculate settled bacteria. Doors to ORs should be kept closed to maintain correct ventilation, airflow, and air pressure. To minimize static electricity and to reduce the potential for bacterial growth, relative humidity in the OR should be maintained between 30% and 60%. A lower relative humidity may support accumulation of static electricity, whereas the presence of a higher humidity may cause condensation of ambient moisture, which may result in damp materials and supplies. This dampness supports bacterial growth. Temperatures in ORs should be maintained at 68° to 73° F (20° to 23° C) (AIA, 2010).
Practices to Prevent Infection
Preparing items for use in surgery and other invasive procedures requires thorough cleaning and drying of the items followed by either sterilization or high-level disinfection process. The efficacy of the sterilization or high-level disinfection process depends in part on cleaning to lower or limit the amount of bioburden present on the item to the lowest level possible.
Preparation of Items/Instruments for Sterilization and High-Level Disinfection
Cleaning.
To prevent infection, all items that come into contact with the patient or sterile field should be systematically decontaminated after a surgical procedure. Handling, transport, and cleaning methods must be selected to prevent cross-contamination to other patients, exposure of personnel to bloodborne pathogens, and damage to instruments. The cleaning and decontamination methods chosen should be economic and of demonstrated effectiveness (Research Highlight). Items may be cleaned manually, by mechanical means, or by a combination of the two (AAMI, 2012). Increased productivity, greater cleaning effectiveness, and increased employee safety may result from use of mechanical cleaning methods. Some mechanical cleaning equipment is designed to remove microorganisms through a cleaning and rinsing action, whereas others clean and destroy specific types of microorganisms by thermal or chemical means. The most commonly used mechanical cleaners are ultrasonic cleaners, washer disinfectors/decontaminators, and cart washers.
All workers handling soiled surgical instruments, whether in the OR, a substerile room, or a central decontamination area, must wear PPE sufficient to prevent contact with any blood or other body fluid. This generally means scrub attire covered with a liquid-resistant gown, coverall, or sleeved apron; hair covering; surgical face mask; eye protection; and gloves suitable to the task. In the event that fluids may pool on the floor, liquid-proof boots or shoe covers are recommended.
Instruments should be kept as free as possible from gross soil and other debris during the surgical procedure. Throughout the surgical procedure, the scrub person should wipe used instruments with sponges moistened with sterile water. When blood is allowed to dry on an instrument, it may cause pitting, rusting, or corrosion. Sterile water should be used rather than saline, which can cause pitting and damage to instrument surfaces. Initial decontamination should begin immediately on completion of the surgical procedure (AAMI, 2012; AORN, 2013e). All instruments that can be immersed are disassembled, and box locks are opened to allow solution to contact all soiled surfaces. These instruments should be placed in a basin, solid-bottom container system, or bin with a lid. Scissors and lightweight instruments should be placed on top. Heavy retractors should be placed in a separate tray.
Some instruments have sharp or pointed edges, such as scissors, forceps with teeth, perforating towel clamps, curettes, and rongeurs. These items can penetrate the gloves and skin, creating a portal of entry for infectious organisms. Sharp instruments must not be placed in a basin or tray in such a way that a worker would have to reach into the container to retrieve the instrument, risking injury. An enzyme solution, foam or spray, or a towel moistened with water can be added to the instruments to begin the process of breaking down any proteinaceous materials that may remain on the instruments and is useful in preventing debris from drying. Either should be used if there will be a delay in processing. Soiled instruments should be contained within leakproof containers, or trays inside plastic bags when they are transported from the OR for cleaning and decontamination. Soiled instruments should never be cleaned in a sink used for handwashing or a surgical scrub. Contaminated instruments should be transported to and cleaned only in a dedicated decontamination area. If sharps are being transported, the container should be puncture-resistant. Means of containing instruments include plastic, rubber, or metal bins with lids; solid-bottom sterilization container systems with the lids and filters in place; or simply placement of the instrument tray in a plastic bag. All soiled containment packages should be labeled with the biohazard symbol to warn handlers as to the nature of the contents. Transporting instruments while they are soaking is discouraged because of the possibility of a liquid spill and its associated clean-up problems as well as the difficulty of safely disposing the contaminated liquid (AAMI, 2012).
In the decontamination area, an initial cold water rinse with tap water or a soak in cool water with a protein-dissolving and blood-dissolving enzyme helps remove blood, tissue, and gross debris from device lumens, joints, and serrations. After completion of this pretreatment, the instruments should be processed in a mechanical washer or manually washed if a mechanical system is not available (AAMI, 2012) or if the device cannot tolerate mechanical washing.
Mechanical processing is usually accomplished with a washer disinfector/decontaminator and an ultrasonic cleaner. Washer disinfector/decontaminator cycles vary to some degree and may include an initial cool water rinse to remove protein debris, an enzymatic-solution soak, a detergent wash, ultrasonic cleaning, a hot water rinse, a liquid chemical germicide rinse, a lubrication cycle, and drying. Soiled utensils such as basins and trays should also pass through a mechanical washer. Some washers provide cycles specific to delicate or specialty instruments. When endoscopic devices or other items with a lumen are cleaned, a cleaning apparatus for these items can be attached to the many washer disinfectors/decontaminators (AAMI, 2012; CDC, 2008a).
The ultrasonic cleaning process is designed to remove fine soil from crevices and lumens of complex devices and from box-lock areas and serrations of instrumentation. It should be used only after gross debris has been removed. Ultrasonic energy occurs in waveforms and is generated by transducers on the sides or bottom of a specially constructed chamber that is filled with water or a water and detergent solution. The ultrasonic waves pass through the water, creating tiny bubbles that collapse or implode. This creates a negative pressure, which pulls debris away from surfaces. This process is known as cavitation. Ultrasonic cleaning is not biocidal, and after sonication the instruments should be rinsed to remove the loose debris.
Not all items tolerate the energy waves of the ultrasonic process. For example, lensed instruments should not be subject to ultrasonic cleaning because the energy can compromise the integrity of the lens seal. Dissimilar metals, such as stainless steel, titanium, copper, and lead, should not be ultrasonically processed at the same time. The energy waves, combined with the heat and detergent solution, can cause electrolysis to occur, plating one metal onto others, potentially ruining the instruments. Some manufacturers recommend that microsurgery instruments not be subjected to ultrasonic cleaning because of their delicate design and the fact that they may contain several types of metal. The detergent or enzyme cleaner used in the ultrasonic machine should be selected carefully and manufacturer's instructions should be consulted to determine compatibility with the ultrasonic cleaner machine and with the device. The corrosiveness and overall effectiveness of some solutions can be dramatically affected by the combination of heat and ultrasonic energy in such a machine. The water in the ultrasonic cleaner should be replaced when it is visibly soiled or at regularly scheduled intervals. Ultrasonic cleaners should be cleaned according to the manufacturer's written instructions for use.
Some instruments, such as air-powered and some microsurgical instruments, do not tolerate immersion in water or cannot tolerate the heat or pressures involved in mechanical processes. These items must be manually cleaned using an appropriate detergent for the type of material and the type of soil on the item. If protein or other organic soil is present, the detergent should have an alkaline pH (>7). If inorganic soil is present, the detergent should have an acid pH (<7). The degree of alkalinity or acidity should be selected so that the instrument or item itself is not damaged in the cleaning process. Using acidic or harshly alkaline solutions, however, can remove the protective passivation layer from the instrument, resulting in irreparable pitting and other corrosive activity. Although a neutral pH detergent is often selected for the foregoing reasons, written instructions from the instrument manufacturer should be consulted to determine appropriate cleaning products and procedures (AAMI, 2009b). If instruments are manually cleaned, they should be submerged in warm water with an appropriate detergent and then cleaned and rinsed while submerged. Cleaning in this fashion helps protect personnel from aerosolization or splashing of infectious material and from injury caused by sharp objects. Harsh abrasives should not be used for manual cleaning because they damage the protective surfaces of instruments, contribute to corrosion, and can impede sterilization (AORN, 2013e). The detergent manufacturer's instructions for concentration, water temperature, and contact time should be followed. Brushes used to clean lumens should be long enough to exit the distal end of the lumen. Brush diameter should be appropriate to lumen diameter. Brushing should be done under the surface of the water to prevent aerosolization and splashing of contaminants. Bristle diameter should be large enough to allow consistent contact with the inner walls of the lumen but small enough so as not to collapse when inserted. Brushes should be either single-use disposable or should be decontaminated at least daily.
Items that were soiled with blood or body fluids and that have been cleaned only may not have been sufficiently decontaminated to allow safe handling by workers not wearing protective attire and may require a microbicidal process, such as soaking in a liquid chemical germicide or liquid chemical sterilant. The device manufacturer's instructions should be consulted to determine if decontamination using a microbicidal process is required after cleaning and prior to packaging and sterilization. If there is no method suitable to further decontamination of the item, because of damage to the item, cost, or unavailability, workers in the preparation area should wear protective gloves when handling, inspecting, assembling, and packaging these items for sterilization.
The final step before sterilization for reuse includes instrument preparation and packaging. These activities should occur in a clean area, separate from the area where decontamination occurs. Instruments should be inspected for cleanliness and functionality. Soiled instruments should be returned to the decontamination area for further cleaning. Instruments with movable parts may be treated with a water-soluble lubricant solution that contains an antimicrobial agent to retard growth in the lubricant solution. Broken or worn instruments should be set aside for repair. Instruments are then assembled into sets according to set content lists prepared by perioperative nursing staff.
Packaging and Sterilizer Loading.
Packaging of surgical supplies and their arrangement in loads in the sterilizer are factors that govern the effectiveness of sterilization. The prime function of a package containing a surgical item is to permit sterilization of the contents and to ensure the sterility of the contents up to the time the package is opened. Provision must be made for the contents to be removed without contamination. To be effective, packaging material should have the following characteristics:
• Allows for adequate air removal and sterilant penetration
• Provides an adequate microbial barrier
• Resists tearing or punctures
• Has proven seal integrity (i.e., does not delaminate when opened and does not allow a reseal after opening)
• Allows for aseptic delivery of package contents
• Is free of toxic ingredients and nonfast dyes
• Is cost-effective by cost and value analysis (AORN, 2013h)
Rigid sterilization container systems are one method of packaging instrumentation. Rigid containers can be sterilized, stacked, and stored. Because of the rigid material of the container, they cannot be punctured, abraded, or easily contaminated by environmental microbes. Recommendations and written instructions for sterilizing should be obtained from the container manufacturer. Prior to purchase, performance testing should be carried out in the sterile processing department of the healthcare facility to ensure that all conditions essential for sterilization and drying can be achieved. The container manufacturer should be consulted for information regarding set preparation and the most challenging areas within the container for placement of chemical indicators. Chemical indicators serve to verify that the sterilant has reached the interior of the container and is evidence that one or more of the parameters of the sterilization process were sufficient to cause a color change (see Quality Monitoring Practices). Many in-hospital packaging materials—woven and nonwoven, reusable and disposable—are marketed and should be evaluated carefully before a product is chosen.
If textile wrappers are used, they must be laundered between sterilization exposures to ensure sufficient moisture content of the fibers. This prevents superheating, which can result in a process failure during steam sterilization. Rehydrated materials also deteriorate at a slower rate. All wrappers must be checked for holes or tears before use for packaging. Manufacturer's instructions for use should be consulted to determine acceptable size, weight and density of textile packs. Instrument sets, including the packaging should not exceed 25 pounds (AAMI 2010; 2012).
Wrappers should be held at room temperature (68° to 73° F [20° to 23° C]) at least 2 hours prior to sterilization. Temperature and humidity equilibration of packaging materials are needed to prevent superheating and possible sterilization failure. When textiles are used for wrapping, the items should be wrapped sequentially in two barrier-type wrappers, which may be disposable or reusable. A single-textile reusable wrapper is defined as one layer of 270- to 280-thread count woven fabric. Sequential double-wrapping creates a package within a package, providing for ease in presenting the wrapped item to the sterile field. A commonly used envelope wrap is made by placing the article diagonally in the center of the wrapper. The near corner, which should point toward the worker, is brought over the item, and the triangular tip is folded back to form a cuff. The two side flaps are folded to the center in like manner. The far corner of the wrapper is then folded on top of the other three. Careful wrapping to prevent tenting and gapping of the package is essential (Figure 4-4). The process is repeated with the second wrapper, and the package is secured with autoclave indicator tape.
When the pack is opened for use, the flaps at the corners are used to form a protective cuff over the person's hands during dispensing of the sterile contents.
Single-use disposable nonwoven wrappers made of synthetic materials have largely replaced textiles for wrap. These wrappers are available in a variety of sizes and may be supplied as a single sheet or a double wrap that is bonded together. Double-wrap sheets provide a bacterial barrier at least equivalent to the sequential double wrap and allow for safe and easy presentation of the package contents to the sterile field, providing an alternative to the sequential double-wrapping procedure. Wrapping technique is the same as when using a textile wrapper. Nonwoven, single-use wrappers should not be reused (AAMI, 2012).
Sterilization process (chemical) indicator tape should be used to hold wrappers in place on packages and to indicate that the packages have been exposed to the physical conditions of a sterilization cycle. When packages are opened, these tapes should be first torn so that the package cannot be retaped and then removed from reusable wrappers because they create laundry problems, such as occluding screens and filters. Tapes also may leave an adhesive residue that can interfere with future sterilization of the fabric.
Sterilization pouches, commonly manufactured from a combination of paper or Tyvek and plastic films, provide an option for the packaging of single (or small groups) or lightweight instruments or other objects (e.g., medicine cups). Pouches are selected based on material compatibility with the intended sterilization technology. Single or double pouching may be used; however, the pouch manufacturer's instructions for use should be followed to determine if double pouching is acceptable. A chemical indicator appropriate to the sterilization method is placed in the pouch, which is sealed either with heat lamination or with a self-adhesive strip (Figure 4-5). Unless validated by the pouch manufacturer, pouches should not be placed within sets because they cannot be positioned to ensure adequate air removal, steam penetration, or drying (AAMI, 2012). Any writing on the pouch should be done on the plastic side.
Every package intended for sterile use should be imprinted or labeled with a load-control number that identifies the date of sterilization, the sterilizer used, and the cycle or load number. Load-control numbers facilitate identification and retrieval of supplies, inventory control, and appropriate rotation to ensure that dated packages are used first (AAMI, 2012). Some instruments or packaging systems may present challenges to the sterilization process. Special preparation or loading procedures may be necessary to meet these challenges. Hinged instruments must be arranged so that the sterilant can contact all surfaces of the instrument, including the tips, hinged surfaces, and ratchets. To accomplish this, these instruments must be sterilized in the open position. If the instruments have ringed handles for the fingers, the instruments may be placed on a “stringer,” which is a U-shaped metal rod made especially for this purpose. When using container systems for sterilization, basket attachments may be used to immobilize instruments in the proper position for sterilization. Instruments with concave or other surfaces that can hold water must be carefully placed on edge to facilitate removal of air, which may become trapped in the concave surface. Placing the package on edge also facilitates drainage of condensate. Items should be positioned with sufficient space between all surfaces so that the sterilant can contact all surfaces (AAMI, 2012; CDC, 2008a).
When packaging is complete and the sterilizer chamber is loaded, the bundles and packages should be arranged to minimize resistance to sterilant contact or penetration and to enhance air removal. Items capable of holding water, such as basins or medicine cups, should be positioned, usually arranged on their sides, so that condensate can drain.
Rigid container systems should be placed flat on the sterilizer shelf. These containers should not be stacked during sterilization unless the container manufacturer specifically recommends this practice and then should only be stacked in the manner recommended by the manufacturer's written instructions. Stacking may interfere with air removal and sterilant penetration. In addition, during steam sterilization condensation from upper containers may drip down and contaminate lower containers when the sterilizer door is opened and the cooler room air contacts the containers (AAMI, 2012; CDC, 2008a).
When combining containers and pouches and or packs in a sterilizer load, hard goods should be placed on the bottom to prevent condensation from dripping onto lower packs.
Pouches should be positioned on edge in a rack. They should be placed paper to plastic, all facing in the same direction.
The Spaulding Classification System.
Items to be sterilized or high-level disinfected are classified as critical, semicritical, and noncritical, based on the risk of infection for the patient. This classification system, known as the Spaulding classification system and named for its developer, Earle Spaulding (Banerjee et al, 2008), has withstood the passage of time and continues to be used today to determine the correct processing method for preparing instruments and other items for patient use. According to the Spaulding system, the level of disinfection required is based on the nature of the item and the manner in which it is to be used.
Critical items are those that enter sterile tissue or the vascular system. These items should be subjected to a sterilization process and be sterile at the time of use. Examples of critical items include surgical instruments, needles, and implants (CDC, 2008a). Unsterile critical items may be sterilized using a variety of sterilization technologies. Examples of sterilization technology include steam, ethylene oxide, and hydrogen peroxide vapor. Many critical items are purchased from the manufacturer as sterile.
Semicritical items contact but do not penetrate mucous membranes. Examples include anesthesia breathing circuits, thermometers, GI endoscopes, and laryngoscopes. Semicritical instruments require high-level disinfection; that is, these items must be free of microorganisms other than bacterial spores. Examples of high-level disinfecting agents include glutaraldehyde, ortho-phthalaldehyde (OPA), stabilized hydrogen peroxide, peracetic acid, and chlorine or chlorine compounds (Banerjee et al, 2008).
Noncritical items are items that come into contact only with intact skin. Because skin is an effective barrier to most microorganisms, most noncritical, reusable items can be cleaned at the point of use. Intermediate-level or low-level disinfectants may be used to process noncritical items. Examples of noncritical items include blood pressure cuffs, bedpans, linens, utensils, furniture, and floors. Examples of low-level disinfectants include alcohols, sodium hypochlorite, phenolic solutions, iodophor solutions, and quaternary ammonium solutions (Banerjee et al, 2008).
Sterilization.
Sterilization is defined as the complete elimination or destruction of all forms of microbial life. The concept of what constitutes “sterile” is measured as the probability of sterility for each item to be sterilized. This probability is known as the sterility assurance level (SAL). For terminal steam sterilization processes, 10–6 is the recommended probability of survival for microorganisms on a sterilized device. A probability of microorganism survival of 10–6 means that there is less than or equal to a 1 in 1 million chance that any viable microorganisms remain after sterilization. The SAL of 10–6 is considered appropriate for items to be used on compromised body tissue. For items not intended to come into contact with compromised tissue, an SAL of 10–3 is sometimes accepted. An SAL of 10–3 represents a 1 in 1000 chance of a surviving microorganism (AAMI, 2012).
Steam Sterilization.
Steam sterilization is the oldest, safest, most economical, and most commonly used method of sterilization available in healthcare. It is the preferred method of sterilization for items that are heat and moisture tolerant. The efficacy of steam sterilization depends on lowering and limiting the bioburden on the item to be sterilized, using effective sterilization cycles, and preventing recontamination of sterile items before delivery to the point of use (AAMI, 2009a).
Theory of Microbial Destruction.
Microorganisms are believed to be destroyed by moist heat through a process of denaturation and coagulation of the enzyme-protein system within the bacterial cell. Microorganisms are killed at a lower temperature when moist heat is used as opposed to when dry heat is used. This fact is based on the theory that all chemical reactions, including coagulation of proteins, are catalyzed by the presence of water.
Principles and Mechanisms.
At standard atmospheric pressure (sea level) when water is heated, the water temperature increases as heat energy is added. After the boiling point of 212° F (100° C) is reached, additional heat energy evaporates the water to form steam. At this point, the steam and the water are at the same temperature, but the steam has more energy than the water. This difference in energy is known as the latent heat of vaporization. When a cold item is introduced into the steam, some of the steam releases its latent energy to the object and changes back to liquid water. This phenomenon allows items to be heated much more rapidly in steam than in dry heat. The phenomenon of steam changing to liquid water is called condensation, and the steam and the liquid water are at a temperature of 212° F (100° C) when this occurs. At this point, the steam is said to be saturated. This 212° F (100° C) temperature is insufficient to kill microorganisms, however. To achieve a saturation temperature sufficient to kill microorganisms (250° F [121° C]), it is necessary to have a sealed container. When water is boiled in a vessel from which the steam cannot escape, a higher temperature is reached. As more steam is generated with no escape route, pressure in the vessel increases. The higher the steam pressure, the higher the temperature. The steam is the sterilizing agent. Any compressed air remaining in the vessel mixes with the steam and lowers the steam temperature. This reduced-temperature steam is incapable of sterilization. Air acts as a barrier to steam sterilization (AAMI, 2009b).
Steam entering the sterilizer chamber should contain little or no trapped liquid water. The term steam quality describes the amount of steam vapor and liquid water in the mixture. A steam quality of 100% indicates that no liquid water is present in the steam. A steam quality of 97% or greater (i.e., <3% of the mixture is liquid water) is recommended to achieve an efficient sterilization process. Common causes of poor steam quality include improper boiler operation and inadequate or poorly maintained steam distribution lines to the sterilizer (AAMI, 2009b).
Sterilizing.
When steam enters the autoclave, it is at the same pressure as the atmosphere. As the valves and doors to the outside close, steam pressure rises inside the chamber, increasing the temperature of the steam. Evacuation of air from the sterilizer is necessary to permit proper permeation of steam. If a sterilizer is improperly loaded, mixing of air with steam acts as a barrier to steam penetration and prevents attainment of the sterilization temperature.
The three factors necessary to achieve steam sterilization are time, temperature, and saturated steam. The microbial destruction period is based on the known time and temperature necessary to accomplish sterilization in saturated steam. Authorities have shown that definite laws determine the order of death in a given bacterial population subjected to a sterilizing process. If the temperature is increased, the time may be decreased.
To provide a safety margin, the minimum estimated exposure is extended to cover the lag between the attainment of the selected temperature in the chamber and the temperature of the load. The length of exposure varies with the type of sterilizer; cycle design; altitude; bioburden; packaging; and size, design, and composition of items to be sterilized. Written instructions for sterilization parameters should be obtained from the device and the sterilizer manufacturer. If a closed container system is used as packaging for items to be sterilized, the container manufacturer's written instructions for exposure times should be consulted and reconciled with those of the sterilizer and the device manufacturer (AAMI, 2012).
Sterilizers vary in design and performance characteristics. Use of rigid container systems may alter the come-up and exposure times in steam sterilizers. The configuration of some instruments or medical devices may hinder air removal and steam penetration, making sterilization more difficult. In such circumstances the device manufacturer must be able to specify the necessary parameters to achieve sterility. Typical minimum cycle times for gravity-displacement steam sterilization and typical minimum cycle times for dynamic air-removal steam sterilization are provided in Table 4-4. Certain instruments, instrument sets, and implants require extended time cycles, however. The manufacturer's written instructions for extended cycle times must be followed.
TABLE 4-4
Common Time-Temperature Parameters for Steam Sterilization
| Type of Sterilization Cycle | Load Configuration | Temperature, in ° F (° C) | Time, in Minutes |
| Gravity displacement | Porous or nonporous | 250-254 (121-123) | 15-30 |
| 270-275 (132-135) | 10-25 | ||
| Prevacuum | Porous or nonporous | 270-275 (132-135) | 3-4 or manufacturer's instructions |
| Steam flush/pressure pulse | Porous or nonporous | 250-254 (121-123) | 20 |
| 270-275 (132-135) | 3-4 |
The recording mechanism on the sterilizer gives information about the run of the load and to what temperature the goods were exposed and the exposure time. To achieve sterilization the temperature inside the chamber must be maintained throughout the determined time of exposure. The sterilizer recording device (e.g., printout) documents the come-up time, exposure time, and exhaust time (AAMI, 2012).
Drying, Cooling, and Storing.
On completion of the sterilization exposure time, the steam inside the chamber is exhausted immediately so that it does not condense and produce wet packs. When the chamber has been exhausted and the pressure reduced, the door may be opened slightly to permit vapor to escape. Drying time can vary according to load contents. The minimum drying time is approximately 15 to 20 minutes.
After removal from the sterilizer, freshly sterilized packs should be left untouched on the loading carriage until they have cooled to room temperature. This is usually accomplished in 30 to 60 minutes but may take up to 2 hours, depending on the load contents (AAMI, 2012). If freshly sterilized packages are placed on cool surfaces such as metal tabletops, vapor still inside the essentially dry package may condense to water. This water may dampen the package from the inside to the outside. When the outside is wet, bacteria may follow the moist tract into the contents of the package. Because bacteria are capable of passing through layers of wet material, any packages that are wet must be considered unsterile.
A written record of existing conditions during each sterilization cycle should be maintained. It should include the sterilizer number, the cycle or load number, the time and temperature of the cycle, the date of sterilization, the contents of the load, and the initials of the operator. These records should be retained for the length of time designated by the statute of limitations in each state.
Sterile packages must be handled carefully and only as necessary. They should be stored in clean, dry, limited-access areas that are well ventilated and have controlled temperature and humidity. Closed cabinets are preferred to open shelves for sterile storage. If open shelves must be used, the lowest shelf should be 8 to 10 inches from the floor to avoid floor contamination. The highest shelf should be at least 18 inches from the ceiling or the sprinkler head to allow for circulation around the stored items. All shelves should be at least 2 inches from outside walls to facilitate air circulation and avoid any condensation that might accumulate on the walls during periods of severe temperature change (AAMI, 2012). Shelving should be smooth and well spaced, with no projections or sharp corners that might damage the wrappers. Sterilized packs should never be stacked in contact with each other and should only be stacked if the packaging instructions for use indicate this is acceptable. Their arrangement on the shelves should provide for air circulation on all sides of each package. Excessive handling, crowding, dropping, and stacking of sterile packs tend to force particles through the mesh or matrix of the wrapping material, which might contaminate the contents. Sterile items should not be stored in any area where they can become wet.
Shelf life refers to the length of time a pack may be considered sterile. Loss of package sterility is event related as opposed to time related; that is, what happens to the package after sterilization determines its continued sterility—not the length of time the package remains on the shelf ready for use. Variables that must be considered in determining shelf life are the type and number of layers of packaging material used, the presence or absence of impervious protective covers, the number of times a package is handled before use, and the conditions of storage. Impervious protective covers known as dust covers may extend shelf life by protecting the sterile package from a contaminating event. When used to protect sterilized items, impervious covers should be designated as such to prevent their being mistaken for a sterile wrap. They should be applied only to thoroughly cooled, dry packs at the time of removal from the sterilizer cart, after the required cooling period (AORN, 2013h).
A large number of commercially sterilized packages carry an expiration date after which the product should not be used. Expiration dating in these instances typically has to do with degradation of the product rather than sterility.
Supply standards should be planned to maintain adequate stock with prompt turnover. Appropriate volume and proper rotation of supplies reduce the need for concern about shelf life. The longer an item is stored, the greater the chances of contamination. For proper rotation, the most recently dated sterile packages should be placed behind those already on the shelves.
Quality Monitoring Practices.
Physical, chemical, and biologic monitors are used for quality monitoring of the sterilization process. These monitors assist in identifying process failures that may be a result of equipment malfunction or operator errors. Physical monitoring is accomplished with pressure recorders, digital printouts, and gauges. All mechanical parts of sterilizers, including gauges, steam lines, and drains, should be checked periodically by the engineering department within the facility. The individual responsible for the sterilizers should keep reports of these inspections. Temperature, humidity, and vacuum should be measured with control equipment, independently of the fixed gauges. The methods of keeping a constant check on the proper functioning of a sterilizer and ensuring the efficiency of the sterilizing process are described in Table 4-5.
TABLE 4-5
Physical, Chemical, and Biologic Sterilization Monitors




