Determination of the sun protection factor (SPF)

* Sunscreen product applied at 2 mg/cm2.
† Air drying in between immersions.
Relationship of the sun protection factor (SPF) to blockage of erythemal radiation
| SPF | Blockage of Erythemal Radiation (%) |
| 10 | 90 |
| 15 | 92.5 |
| 20 | 95 |
| 40 | 97.5 |
Labeling of sunscreens: ultraviolet B (UVB) and ultraviolet A (UVA) protection (2011).

* SPF is also referred to as sunburn protection factor.
Based on the 2011 FDA Final Rule for ‘Labeling and Effectiveness Testing; Sunscreen Drug Products for Over-the-Counter Human Use.’
Slicc* classification criteria for systemic lupus erythematosus (2012)
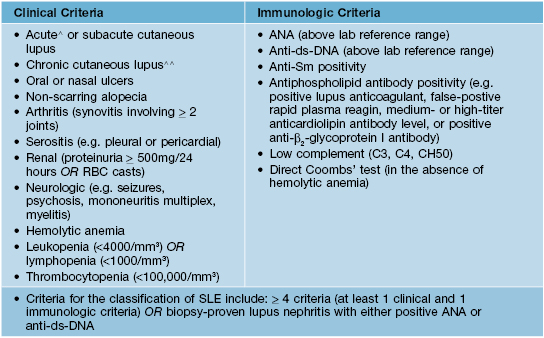
* SLICC, Systemic Lupus International Collaborating Clinics.
∧ Includes malar rash (but not discoid lesions), bullous lupus, toxic epidermal necrolysis variant of SLE, maculopapular lupus rash, photosensitive lupus rash (in the absence of dermatomyositis).
∧∧ Discoid lesions (localized or generalized), hypertrophic lupus, lupus panniculitis, mucosal lupus, LE tumidus, chilblains lupus, discoid lupus/lichen planus overlap.
These revised criteria are intended to be more clinically relevant and to incorporate new immunologic information. They have not been tested for the purposes of SLE diagnosis.
Adapted from Petri M et al. Arthritis Rheum 2012;64:2677.
Fitzpatrick scale of skin phototypes
| Skin Phototype | Skin Color | Response to UV Irradiation |
| I | White | Always burns, does not tan |
| II | White | Burns easily, tans with difficulty |
| III | Beige | Mild burns, tans gradually |
| IV | Brown | Rarely burns, tans easily |
| V | Dark brown | Very rarely burns, tans very easily |
| VI | Black | Never burns, tans very easily |
Special considerations for corticosteroid and antihistamine use during pregnancy

Courtesy, Christina M. Ambros-Rudolph, MD.
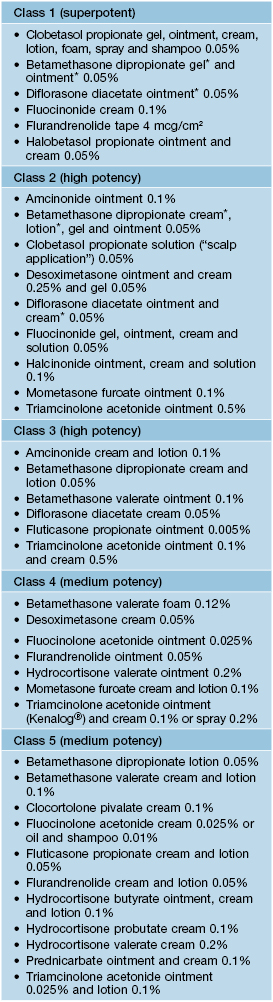
Corticosteroid classes
Suggested patch test screening agents are in italics for when allergic contact dermatitis to topical CS is suspected; all but hydrocortisone are in the NACDG screening series.

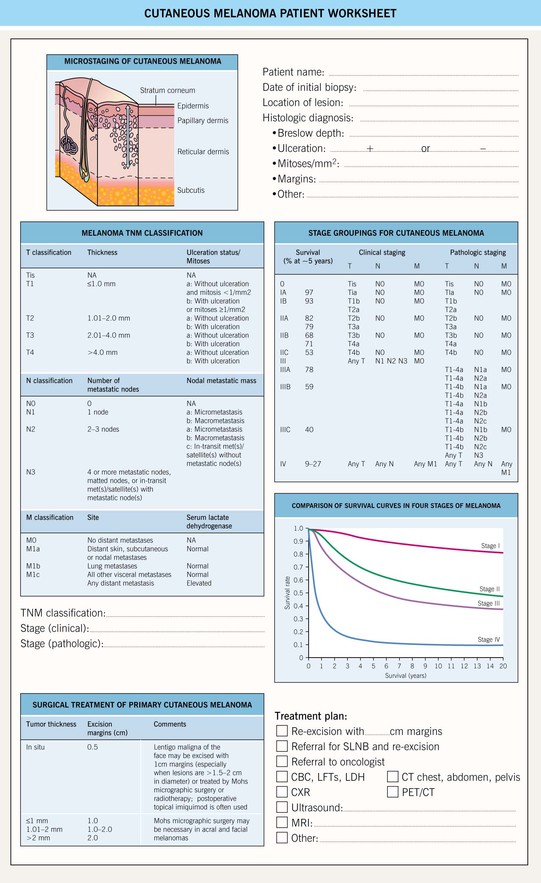
SLNB, sentinel lymph node biopsy; CBC, complete blood count; LFTs, liver function tests; LDH, lactate dehydrogenase; CXR, chest x-ray; CT, computed tomography; CT/PET, computed tomography/positron emission tomography; MRI, magnetic resonance imaging.
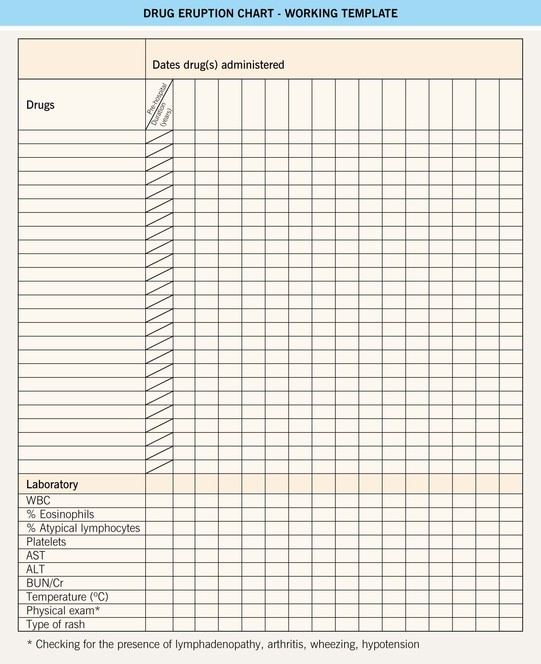
Drug Eruption Chart (blank template). This is a helpful working template for organizing all of the available patient information into one document for a patient with a suspected adverse cutaneous drug reaction (ACDR). Step 1: Compile all of the recently consumed or administered drugs (including prescription, over-the-counter, and supplements) into the chart. Step 2: Review and list the pertinent laboratory information and physical findings at the bottom of the chart. Step 3: Referring to Figure 17.1, exclude a SCAR and categorize the type of ACDR. Step 4: Based upon time intervals (see Table 17.3) and the most and least likely drugs to cause ACDR (see Table 17.2), begin to formulate the most likely culprit drugs and recommend their discontinuation. In addition, discontinue unnecessary drugs. Step 5: Longitudinal evaluation of the patient is necessary to: 1) exclude progression to a SCAR; 2) determine the response upon discontinuation of the culprit drug (noting that it might ‘get worse before it gets better’); and 3) to provide supportive care to the patient. SC, subcutaneous; WBC, white blood cell count; AST, aspartate aminotransferase; ALT, alanine aminotransferase.
Refer to www.expertconsult.com for a blank template of the drug eruption chart.