Protozoa and Worms
Leishmaniasis
• Three major forms: (1) cutaneous (Fig. 70.1); (2) mucocutaneous (Fig. 70.2); and (3) visceral (e.g. liver, spleen).
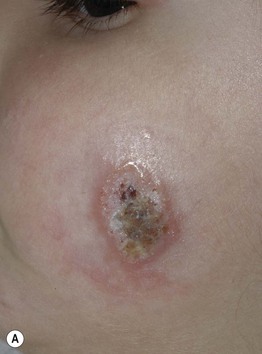
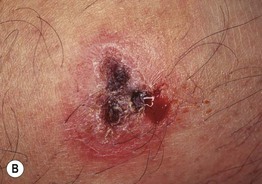
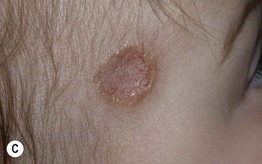
Fig. 70.1 Variable presentations of cutaneous leishmaniasis. Ulcerated plaques with rolled border (A) and central crusting (A, B). Plaque with translucent borders containing telangiectasias and central scarring (C). Cutaneous leishmaniasis is sometimes mistaken for a basal cell carcinoma in adults. A, C, Courtesy, Julie V. Schaffer, MD.
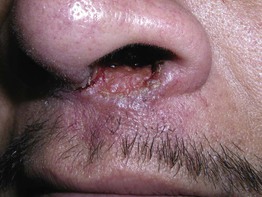
Fig. 70.2 Mucocutaneous leishmaniasis. Ulceration and induration of the nasal vestibule extending onto the cutaneous lip due to Leishmania braziliensis. Courtesy, Kalman Watsky, MD.
• Caused by more than 15 different species of Leishmania (Table 70.1).
Table 70.1
Four major species of Leishmania that cause cutaneous disease.
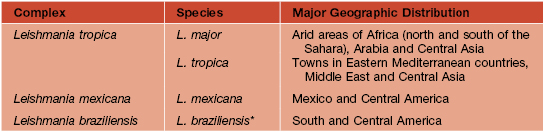
* Can also cause mucosal leishmaniasis or visceral leishmaniasis in immunocompromised individuals.
• Vector = sandfly (Phlebotomus and Lutzomyia spp.) (Fig. 70.3).
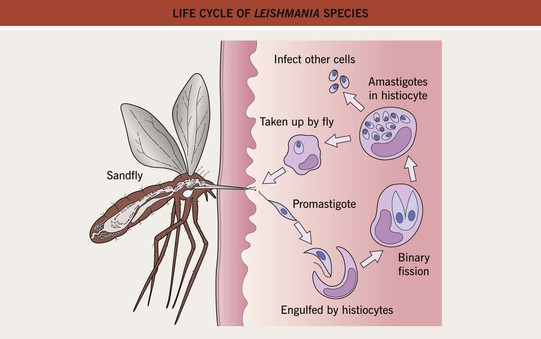
Fig. 70.3 Life cycle of Leishmania species. Promastigotes develop within the gut of the sandfly and then migrate to the proboscis.
• Disease seen worldwide but endemic in areas of Asia, Africa, Latin America, and the Mediterranean basin (Fig. 70.4).
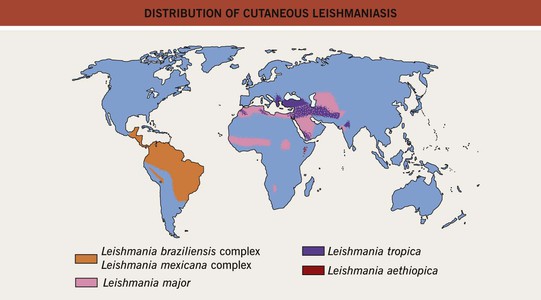
Fig. 70.4 Distribution of cutaneous leishmaniasis. Adapted with permission from Davidson RN, Leishmaniasis. In Cohen J, Powderly WG (Eds.), Infectious Diseases. Edinburgh, UK: Mosby, 2004.
• Cutaneous disease affects skin only and is commonly a papule that expands and ulcerates (Fig. 70.5); pattern may be sporotrichoid (Fig. 70.6); lesion(s) may heal spontaneously (Fig. 70.7).
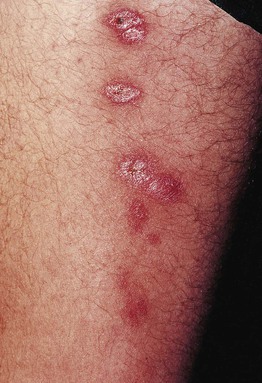
Fig. 70.6 Sporotrichoid form of cutaneous leishmaniasis. With permission from Tyring S, Lupi O, Hengge U (Eds.), Tropical Dermatology. Oxford: Churchill Livingstone, 2005.
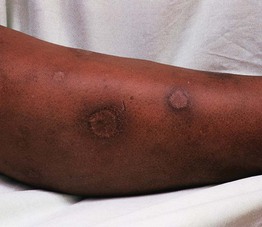
Fig. 70.7 Scars secondary to previous cutaneous leishmaniasis. Circular scars at previous sites of cutaneous leishmaniasis are often the only sign of a prior infection. Courtesy, Omar P. Sangüeza, MD.
• Mucocutaneous form, often due to Leishmania brasiliensis, involves mucosal (e.g. nose, lips, oropharynx) sites as well as the skin.
• Visceral leishmaniasis (kala-azar) affects the bone marrow, spleen, and liver and is commonly due to Leishmania donovani; symptoms include fever, cough, lymphadenopathy, and hepatosplenomegaly; post-kala-azar dermal leishmaniasis may follow treatment.
• Rx: for an isolated lesion, conservative therapy (e.g. observation, heat, cryotherapy) or topical paromycin can be used; for more extensive disease, IV or IM pentavalent antimony (sodium stibogluconate, meglumine antimonate), oral miltefosine.
• For assistance in diagnosis and treatment, helpful sources of information include the Centers for Disease Control and World Health Organization.
Amebiasis
• Protozoan infection (Entamoeba histolytica) that most commonly causes colitis; fecal–oral spread.
• Occasionally presents in the skin with necrotic ulcers that can resemble pyoderma gangrenosum or verrucous plaques that resemble squamous cell carcinoma or condyloma acuminatum.
• Skin involvement generally secondary to extension of rectal amebiasis to perianal or perigenital skin or extension of a liver abscess to skin of abdominal wall.
• Rx: for Entamoeba histolytica, metronidazole; other treatments include diloxanide, tinidazole.
Free-Living Ameba
• Balamuthia mandrillaris can infect immunocompetent (especially children) and immunocompromised patients; the typical cutaneous lesion is a slow-growing indurated plaque on the central face with eventual hematogenous spread to the central nervous system.
• Acanthamoeba spp. can cause cutaneous papulonodules and encephalitis in immunocompromised patients.
Trypanosomiasis – American
• Trypanosoma cruzi carried by reduviid (kissing) bugs.
• Endemic in areas of Central and South America.
• Systemic disease that can affect the autonomic nervous system, gastrointestinal tract, and heart.
• Primary acute phase: local erythema and edema at inoculation site ± regional lymphadenopathy; when periorbital, termed Romaña sign (Fig. 70.8).
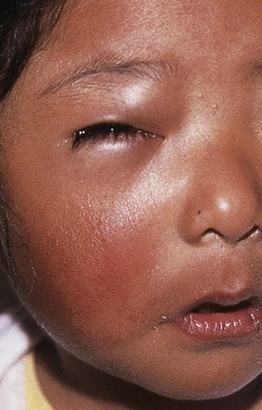
Fig. 70.8 Chagas disease. Young child with unilateral periorbital edema characteristic of this disease (Romaña sign) when the conjunctiva is the portal of entry. Courtesy, Omar P. Sangüeza, MD.
• Chronic phase seen after years to decades: congestive heart failure, arrhythmias, including heart block, megacolon, megaesophagus.
Trypanosomiasis – African
• Found in both West (Trypanosoma brucei gambiense) and East (Trypanosoma brucei rhodesiense) Africa.
• Skin findings: trypanosomal chancre (localized bite reaction; Fig. 70.9) and annular erythematous eruption with fever.
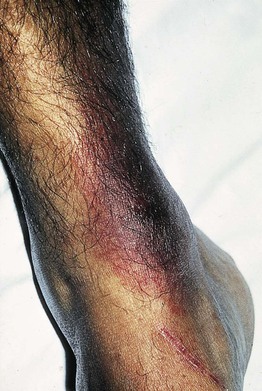
Fig. 70.9 Trypanosomal chancre. The bite reaction, the earliest clinical lesion, is known as a ‘trypanosomal chancre.’ It resembles a boil but is usually painless. Fluid aspirated from the nodule contains actively dividing trypanosomes. This reaction is seen more commonly in T. b. rhodesiense than in T. b. gambiense infection. With permission from Peters W, Pasvol G, Tropical Medicine and Parasitology, 6th ed. London: Mosby, 2007.
• Winterbottom's sign – enlargement of nodes of posterior cervical triangle – classic finding in West African form.
Toxoplasmosis
• Worldwide infection secondary to Toxoplasma gondii; oocytes present in cat feces or infected meat.
• Rare skin involvement; congenital infections present with necrotic or hemorrhagic papules on the trunk (‘T’ in TORCH complex).
• Common presentations include cervical lymphadenitis or chorioretinitis.
• Tissue cysts may lead to recrudescence in immunosuppressed individuals.
Cutaneous Larva Migrans
• Secondary to larvae of animal (e.g. usually wild/domestic dogs/cats) hookworms (intestinal nematodes), e.g. Ancylostoma braziliense.
• Worldwide, but especially common in tropical/subtropical areas and the southwestern United States.
• Larvae in infected soil, including sand, penetrate the skin.
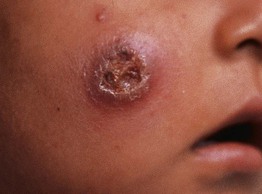
• Pruritic, inflamed, serpiginous tracks are produced by migrating organisms (Fig. 70.10); migration averages 1–2 cm/day.
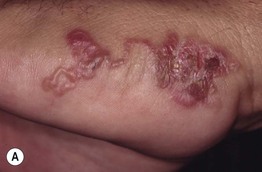
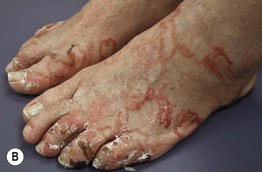
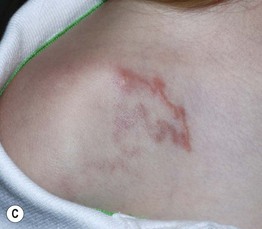
Fig. 70.10 Cutaneous larva migrans. Note the characteristic serpiginous erythematous tracks on the lateral foot (A), both feet (B), and the shoulder (C). Vesiculation and crusting (B) are sometimes seen. B, Courtesy, Peter Klein, MD; C, Courtesy, Julie V. Schaffer, MD.
• Most common locations are the lower extremities, especially the feet, and buttocks, due to walking and sitting at the beach.
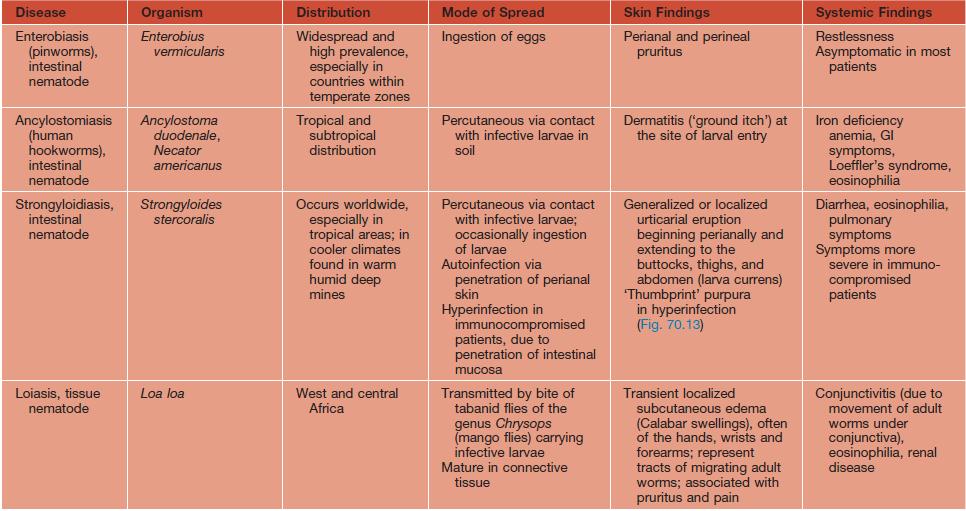
• DDx: not to be confused with larva currens (secondary to Strongyloides; see Table 70.2).
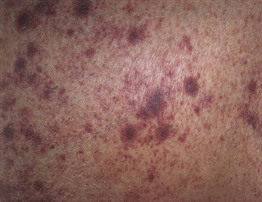
Fig. 70.13 Strongyloidiasis hyperinfection. Multiple purpuric lesions (sometimes referred to as ‘thumbprint’ purpura) on the abdomen of an immunocompromised patient. Courtesy, Jean L. Bolognia, MD.
• Disease is self-limited but treatment can include oral albendazole or ivermectin; topical thiabendazole for localized disease.
Onchocerciasis
• Secondary to Onchocerca volvulus, a tissue-dwelling nematode.
• Vector = black fly (Simulium).
• Endemic in Africa, Yemen, and some areas of Central and South America; near fast-flowing rivers.
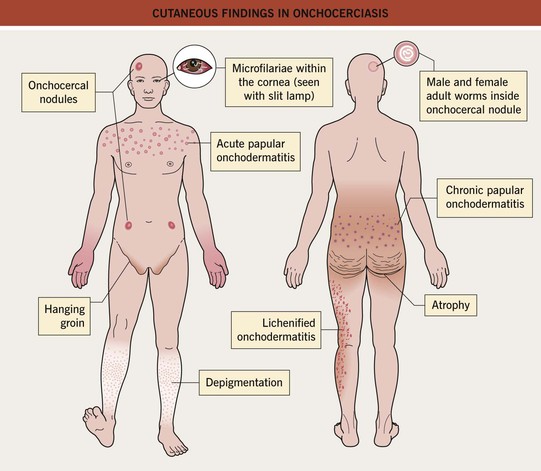
• Skin findings: subcutaneous nodules containing adult worms (onchocercomas), pruritic papular dermatitis, depigmented, lichenified skin, especially over the shins (sowda) (Fig. 70.11).
• Chronic ocular involvement leads to sclerosing keratitis, iridocyclitis, and ultimately blindness, hence the name river blindness.
Filariasis
• Tissue nematodes (Wuchereria bancrofti or Brugia malayi/B. timori) infect the lymphatic system.
• Vector = mosquito (Culex, Anopheles, Aedes spp.).
• Endemic in tropical and subtropical regions of India, the Americas, and Africa.
• Acute form: lymphangitis and orchitis.
• Chronic form: lymphedema, elephantiasis (the enlarged limb becomes indurated with skin folds and overlying verrucous changes), hydrocele, chyluria.
Schistosomiasis
• Trematode (fluke) infection secondary to three major species with specific geographic distributions (Africa – Schistosoma hematobium, Asia – S. japonicum, South America – S. mansoni); intermediate host is the freshwater snail; organisms penetrate skin.
• Variants of cutaneous disease – cercarial dermatitis (transient erythema, urticaria, or pruritic papules), Katayama fever (systemic allergic reaction with urticaria, fever, chills, sweats, headache, peripheral eosinophilia).
• Chronic fibro-occlusive disease in the liver (S. japonicum), intestine (S. mansoni), or urinary tract (S. hematobium).
Swimmer's Itch
• Cercariae of >20 species of animal schistosomes (e.g. Ornithobilharzia) can penetrate skin and cause swimmer's itch.
• Worldwide; endemic to the Great Lakes.
• Erythematous papules on exposed skin (Fig. 70.12).
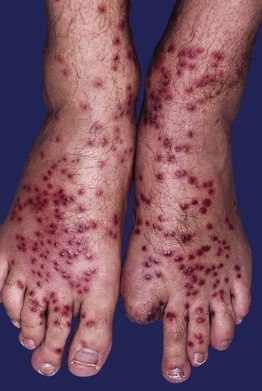
Fig. 70.12 Swimmer's itch. Numerous edematous dark red papules on the feet and ankles. Courtesy, Kalman Watsky, MD.
• Not to be confused with seabather's eruption (secondary to Linuche, Edwardsiella spp.), which affects areas under the swimsuit (see Chapter 72).
Cysticercosis
• Cestodes (tapeworms; e.g. Taenia solium) that more commonly infect animals may infect humans; spread from ingestion of contaminated meat or fecal–oral spread.
• Skin findings: small, asymptomatic papulonodules.
• May have systemic involvement of the brain, eye, heart, muscles, or peritoneal cavity.
Echinococcosis
• Cestode (tapeworm Echinococcus) that usually infects dogs; spread from ingestion of contaminated dog feces.
• Human may be an intermediate host and develop liver or lung hydatid cysts.
Gnathostomiasis
• Due to ingestion of raw or poorly cooked fresh water fish (e.g. ceviche) and eels or frogs that contain larvae of Gnathostoma spp. (nematodes).
• Transient subcutaneous swellings that may be pruritic are due to migration of the larvae.
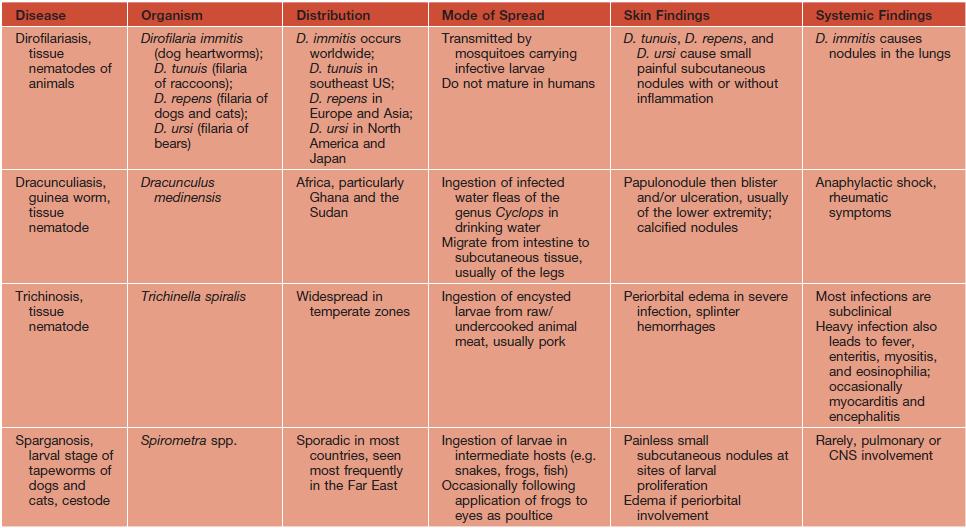
See Table 70.2 for a summary of other major parasitic worms that cause skin findings.
For further information see Ch. 83. From Dermatology, Third Edition.