Systemic glucocorticoids in rheumatology
Kenneth G. Saag, Frank Buttgereit
▪ Glucocorticoids act via a number of pathways including inhibition of transcription factors nuclear factor-κB and activator protein 1.
▪ Glucocorticoids decrease synthesis of proinflammatory cytokines and proinflammatory enzymes, decrease T-cell function, and reduce Fc receptor expression.
▪ Short-term use of low-dose glucocorticoids in rheumatoid arthritis decreases clinical disease activity. These agents also have a beneficial effect on radiographic activity.
▪ Adverse events are common with these drugs, and many of the side effects are dose dependent. Careful titration of dose and strategies to minimize toxicity are essential when glucocorticoids are used.
Introduction
Discovered over 50 years ago, synthetic cortisone was first shown to be remarkably effective in relieving the inflammation associated with rheumatoid arthritis (RA).1 This pioneering work by Hench, Kendall, and colleagues subsequently resulted in a Nobel Prize in Medicine. Despite the advent of many disease-specific therapies, the use of synthetic glucocorticoids in rheumatology remains a common, albeit controversial, therapy for many inflammatory disorders.
Nomenclature and pharmacology
Preparations and structure
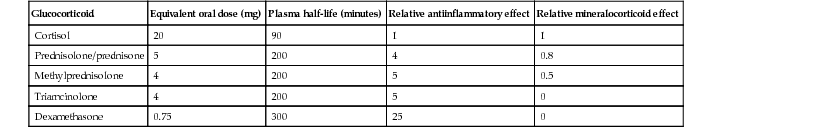
The term corticosteroid is often used synonymously with glucocorticoid, although glucocorticoid is the preferred designation when using exogenous agents therapeutically because corticosteroids encompass both glucocorticoid and mineralocorticoid hormones.2 A variety of synthetic glucocorticoid preparations are commercially available and have variable chemical structure (Fig. 54.1), potency, and half-life (Table 54.1). The 11β- and 17α-hydroxyl groups are important for glucocorticoid activity, and prednisone, a synthetic analogue of cortisone, is also hydroxylated before becoming biologically active. The additional double bond in ring A of the resultant prednisolone enhances glucocorticoid activity without increasing mineralocorticoid activity, an effect that is increased by 6α-methylation (to produce methylprednisolone) or 9α-fluorination (triamcinolone) or both (dexamethasone). In rheumatology, the most commonly used oral glucocorticoids are prednisone and prednisolone, whereas methylprednisolone is the predominant preparation used parenterally.
TABLE 54.1
Some commonly used glucocorticoids
| Glucocorticoid | Equivalent oral dose (mg) | Plasma half-life (minutes) | Relative antiinflammatory effect | Relative mineralocorticoid effect |
| Cortisol | 20 | 90 | 1 | 1 |
| Prednisolone/prednisone | 5 | 200 | 4 | 0.8 |
| Methylprednisolone | 4 | 200 | 5 | 0.5 |
| Triamcinolone | 4 | 200 | 5 | 0 |
| Dexamethasone | 0.75 | 300 | 25 | 0 |
Dosing
Thresholds for low, medium, and high dose are not well defined. Some rheumatologists consider 10 mg/day prednisone equivalent as the upper threshold of a low dose, whereas others use 7.5 mg/day3; this is actually somewhat above the daily endogenous production of cortisol (hydrocortisone) by the adrenal glands, which averages 5.7 mg/m2/day.4 A European League Against Rheumatism (EULAR) standing committee defined low dose as up to 7.5 mg/day, medium dose as more than 7.5 mg/day but less than or equal to 30 mg/day, high dose as more than 30 but mg/day less than or equal to 100 mg/day, and very high dose as more than 100 mg/day prednisolone equivalent.2 Dosing designations were considered based on the percent binding of the glucocorticoid receptors and the risk of perceived adverse effects. These dosing conventions are used throughout this chapter.
Bioequivalence and bioavailability
The different preparations of glucocorticoids have the same rate of gastrointestinal absorption. Most commercially available prednisone tablets are bioequivalent, independent of tablet strength,5 and the systemic bioavailability of prednisone and prednisolone are similar. Glucocorticoid bioavailability does not appear to be affected by pregnancy.
Metabolism and drug interactions
In contrast to endogenous cortisol, which is 80% bound, synthetic glucocorticoids bind poorly to cortisol-binding globulin (CBG, or transcortin). Prednisolone has about 60%, prednisone 5%, and methylprednisolone, dexamethasone, betamethasone, and triamcinolone less than 1% of the affinity of cortisol for CBG. Half-life ranges from about 1 hour for prednisone to over 4 hours for dexamethasone. Prednisone is rapidly metabolized in the liver by CYP3A4 (a cytochrome P450 isoform) to prednisolone; the elimination of prednisone is approximately 13 times faster than the elimination of prednisolone.6 Hyperthyroidism and nephrotic syndrome increase clearance, whereas aging and liver disease impair clearance. Prednisolone is preferred to prednisone in patients with significant hepatic dysfunction.
Enhancement of the rate of metabolism of some glucocorticoids has been noted with concurrent administration of phenobarbital, phenytoin, and rifampin (rifampicin). Concurrent use of aspirin and glucocorticoids decreases salicylate levels by causing an increased rate of salicylate metabolism.
Clearance and dose timing
The elimination half-life of prednisolone is approximately 3 hours.7 Total prednisolone clearance increases by 75% as the intravenous dose increased from 5 to 40 mg.8 Clearance also varies with the time of day. Both prednisolone and methylprednisolone clearance is up to 25% lower in the morning than in the evening.9 This property, in combination with the disruption of the usual cortisol diurnal rhythm by exogenous glucocorticoids, results in variations in efficacy when glucocorticoids are administered at different times during the day.10 Administration of short-acting glucocorticoids very early in the morning, in contrast to later in the day, better complements the normal diurnal variation in endogenous hormones and further blocks production of proinflammatory cytokines such as interleukin-6 (IL-6)11 and inflammatory symptoms.12 For example, a low-dose prednisone compound incorporating a modified-release mechanism (MR prednisone, i.e., drug is released 4 hours after ingestion) when taken in the evening reduced RA morning stiffness more than placebo at 12 weeks.13,14
MR prednisone plus disease-modifying antirheumatic drug (DMARD) therapy resulted in significantly higher ACR20 response rates (percentage of patients achieving a 20% improvement in signs and symptoms according to American College of Rheumatology criteria) and a greater relative reduction in morning stiffness that placebo plus DMARD.15 Significantly greater reductions in severity of RA and fatigue, and a greater improvement in physical function also were seen at week 12 with MR prednisone compared with placebo.
Timing of administration of the glucocorticoid dose also influences toxicity. For example, daily split-dose therapy is more immunosuppressive and should be used for a shorter duration due to its more profound disruption of the normal hypothalamic-pituitary-adrenal (HPA) axis function.
One study found no differences between MR prednisone and standard prednisone with regard to influence on the HPA axis as measured by corticotropin-releasing hormone tests. In both treatment groups, there was a normal response in 50% of the patients, the response was suppressed in about 37%, and there was no response in about 13%.16
Mechanisms of glucocorticoid action
Glucocorticoids act as antiinflammatory mediators via a number of pathways that continue to be further elucidated.17-22 Oral glucocorticoids at dosages of less than 30 mg/day prednisone equivalent circulate in the plasma and diffuse through the plasma membrane where they bind to the cytosolic glucocorticoid receptor, a 95-kDa phosphorylated protein. Diversity in the function of the glucocorticoid receptor is influenced by variations in genetic transcription mediated by alternative splice sites. The relative levels of the α and β receptors influence the sensitivity or resistance of cells to glucocorticoids. Glucocorticoid resistance is reported in patients with severe RA and has been associated with higher levels of glucocorticoid receptor β, activation of mitogen-activated protein kinases (MAPKs) that inhibit glucocorticoid signaling, or antibodies to lipocortin-1.23-26 Individuals with glucocorticoid receptor polymorphisms have higher sensitivity to exogenously administered glucocorticoids.27 Persons with relative glucocorticoid resistance may have diminished in-vitro response of their peripheral blood mononuclear cells to glucocorticoid stimulation.28 Imbalances in the two isoforms of 11β-hydroxysteroid dehydrogenase may play a central role in glucocorticoid resistance.29
Glucocorticoids act on the cell through genomic and nongenomic paths in at least three ways (Fig. 54.2).21
▪ Heat shock protein chaperons the glucocorticoid/glucocorticoid receptor complex to the nucleus, where it binds reversibly and exerts potent effects on transcription via binding to positive and negative glucocorticoid response elements in promoter or suppressor sites of target genes. The detailed mechanism of a direct transrepression via the binding of glucocorticoid receptors to simple DNA binding sequences has been elucidated.30
▪ The glucocorticoid/glucocorticoid receptor complex inhibits transcription factors such as nuclear factor-κB (NF-κB) and activator protein 1. Most antiinflammatory effects are thought to occur due to inhibited gene transcription via these effects on NF-κB (transrepression), whereas the adverse effects of glucocorticoids, such as metabolic side effects, are mediated in large part via activation of transcription of certain genes (transactivation) (Fig. 54.3). However, this concept has recently been challenged.30a
▪ Glucocorticoid signaling occurs through membrane-associated receptors and second messengers (nongenomic pathways). The glucocorticoid receptor gene encodes for both cytosolic and membrane-bound glucocorticoid receptor.31 At high glucocorticoid dosages (more than 100 mg/day prednisolone equivalent), glucocorticoid receptors become saturated and these nongenomic effects emerge. They are also mediated by incorporation of glucocorticoid molecules into cell membranes.19
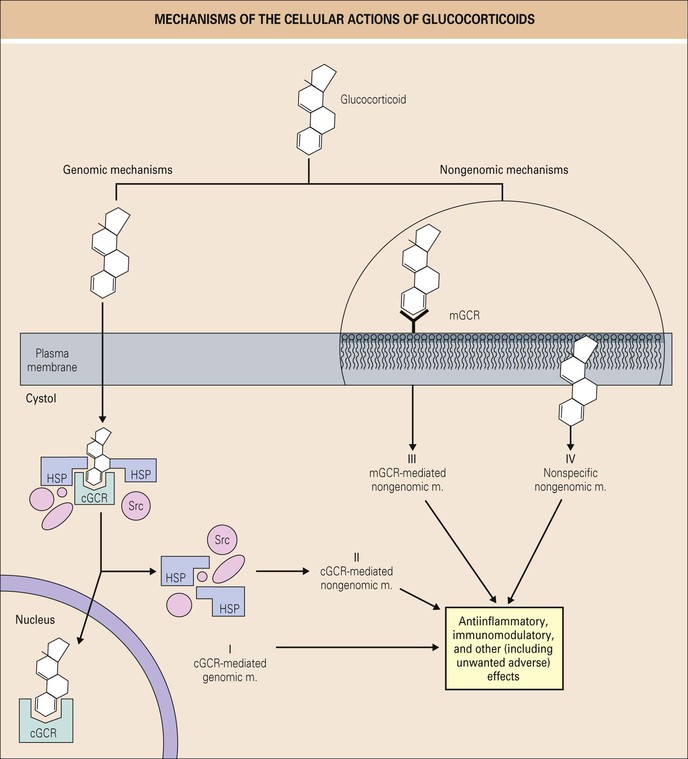
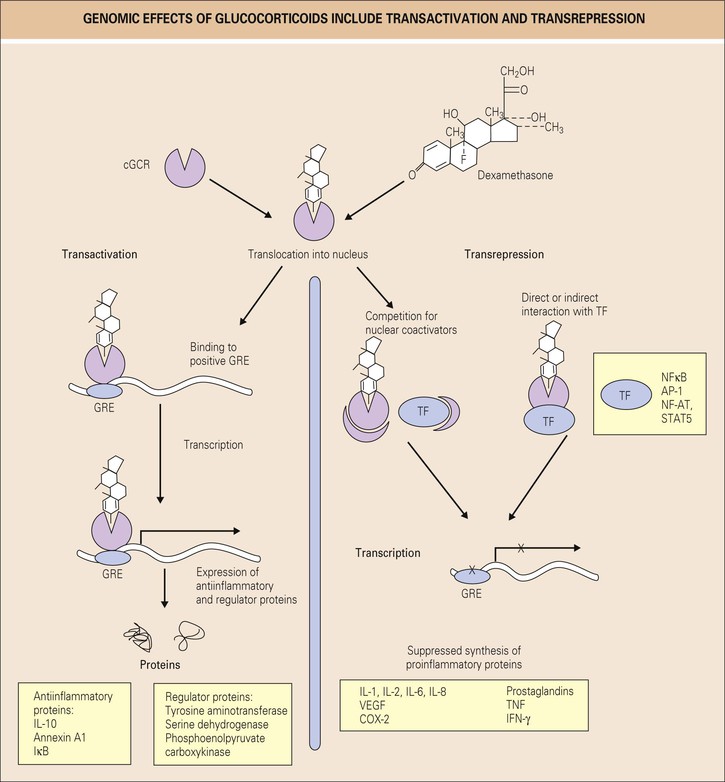
The relative contributions of these three mechanisms are not yet fully understood, but in combination they result in the decreased synthesis of proinflammatory cytokines such as IL-1, IL-2, IL-2 receptor, interferon-α, IL-6, and tumor necrosis factor-α (TNF-α).17,18,32 Glucocorticoids increase the rate of synthesis of lipocortin (annexin I), which inhibits phospholipase A2. Phospholipase A2 converts membrane-bound phospholipids into arachidonic acid with the subsequent intracellular production of prostaglandins, leukotrienes, and oxygen radicals.33 Glucocorticoids also induce MAPK phosphatase 1 (which inactivates all proteins of the MAPK family including Jun N-terminal kinase), and glucocorticoids repress transcription of cyclooxygenase 2.34 Independent of change in gene expression, glucocorticoids exert rapid effects via activation of endothelial nitric oxide as well as activation of β-adrenoreceptors, endonucleases, and neutral endopeptidases, which explains other observed effects.35 Glucocorticoids, even in very low concentrations, inhibit the synthesis of a variety of proinflammatory enzymes including the macrophage products collagenase, elastase, and plasminogen activator.36 Glucocorticoids have a direct effect on lymphocytes by decreasing T-cell function and circulating number. They further inhibit Fc receptor expression (reducing clearance of antibody-coated blood cells), increase the number of circulating neutrophils, and affect leukocyte adhesion to endothelial cells.37
Drug development efforts are attempting to exploit an improved knowledge of the molecular biology of glucocorticoids. Selective glucocorticoid receptor agonists that differentially affect levels of transactivation and transrepression have been under development.38,39 In preliminary experiments some compounds appeared to exhibit antiinflammatory effects similar to those of traditional glucocorticoids but may have fewer adverse effects such as blood glucose elevation or skin thinning.40,41 Liposomal glucocorticoids are being developed with the aim of providing more prolonged concentrations of glucocorticoids at the site of inflammation and lower plasma levels. Part of their mechanism of action may be via insertion of higher concentrations of glucocorticoids into cell membranes.42
Efficacy
Efficacy in rheumatoid arthritis
Short- to moderate-term glucocorticoid studies reveal improvement in disease activity and functional status compared with control therapy.43,44 In many past studies, the antiinflammatory benefits of glucocorticoids declined after the first year45-47 and often were not seen even beyond 5 months.48,49 In contrast to earlier reports, three more recent randomized controlled trials showed that at 2 years treatment with either 5 mg or 7.5 mg of prednisolone50,51 or 10 mg of prednisone52 resulted in variable levels of improvement in joint indices, functional status, and remission criteria compared with placebo.
In addition to disease activity, joint damage as assessed by radiographic criteria (bony erosions and joint space narrowing) is a critical efficacy outcome. Although it is known that methotrexate, the anti-TNF agents, and newer biologic response modifiers are DMARDs in RA, there is now consensus that glucocorticoids function similarly. Many of the RA studies evaluating the effects of glucocorticoids on radiographic progression are summarized in Table 54.2. Findings that DMARDs, biologic agents, and glucocorticoids had similar effects on radiographic progression in RA were confirmed in a meta-analysis.53 Many of the studies investigating the efficacy of glucocorticoids allowed concomitant use of DMARDs, which makes it difficult to discern the independent effects of glucocorticoids. Although many of the earlier investigations failed to demonstrate a benefit of low-dose glucocorticoids on radiographic progression, an increasing number of placebo-controlled randomized or open-label clinical trials have now demonstrated that low-dose glucocorticoids prevent radiographic joint destruction in RA. Although the overwhelming majority of glucocorticoid use internationally in RA is via lower-dose oral therapy,3,54 a few RA studies have examined other approaches such as pulse-dose and intramuscular depot administration.55,56 The addition of prednisone to a methotrexate-based therapy aimed at “tight control” resulted in a significantly greater reduction in radiographic damage.52
TABLE 54.2
Studies of oral glucocorticoid therapy and radiographic progression in rheumatoid arthritis (RA)
| Study | Experimental group | Control group | Study type (no. of subjects) | Effect on radiologic progression | Comments |
| MRC (1955)149 | Cortisone (69 mg/day) | ASA | CT (100) | No difference | Trend toward protective effect |
| MRC (1959)150 | Prednisolone (initially 20 mg, 12 mg/day by yr 1, 10 mg/day by yr 2) | ASA | CT (77) | Reduction after 2 yr, less after 3 yr | Control patients offered prednisolone in yr 3 |
| Berntsen & Freyberg (1961)151 | Various glucocorticoids, dose not reported | IM gold, analgesics | Retrospective (388) | Deterioration in all groups | Other treatments had already failed in many patients |
| Harris et al (1983)152 | Prednisone (5 mg/day) & DMARD | PBO & DMARD | DB RCT (34) | No significant difference | Trend toward reduction |
| Million et al (1984)153 | Prednisolone (10.3 mg/day) & DMARD | No prednisolone | RCT (103) | Significant reduction | 10-yr study |
| Kirwan (1995)45 | Prednisolone (7.5 mg/day) & DMARD | PBO & DMARD | DB RCT (106) | Significant reduction | |
| Boers et al (1997)48 | Prednisolone (60 mg/day with taper) & MTX, SSZ | PBO & SSZ | DB RCT (102) | Significant reduction | |
| Hansen et al (1999)49 | Prednisolone (6 mg/day) & DMARD | DMARD | RCT (102) | No significant difference | Trend toward reduction |
| van Everdingen et al (2002)96 | Prednisolone (10 mg/day) & SSZ as rescue | PBO & SSZ as rescue | DB RCT (81) | Significant reduction | Only study since the MRC studies not allow to background DMARDs; SSZ allowed after 6 mo |
| Cappell et al (2004)95 | Prednisolone (7 mg/day) & SSZ | PBO & SSZ | DB RCT (167) | No significant difference | Discordance between readers in radiographic interpretations |
| Wassenberg et al (2005)51 | Prednisolone (5 mg/day) & either AU or MTX | PBO & either AU or MTX | DB RCT (166) | Significant reduction | Relatively new-onset RA; one of lowest dosages to show radiographic protection |
| Svensson et al (2005)50 | Prednisolone (7.5 mg/day) & DMARD | No prednisone & DMARD | Open RT (225) | Significant reduction | Early RA |
| Bakker et al (2012)52 | Prednisone (10 mg/day) & MTX | PBO & MTX | DB RCT (236) | Significant reduction | Early RA; tight RA control sought with monthly methotrexate dose adjustments |
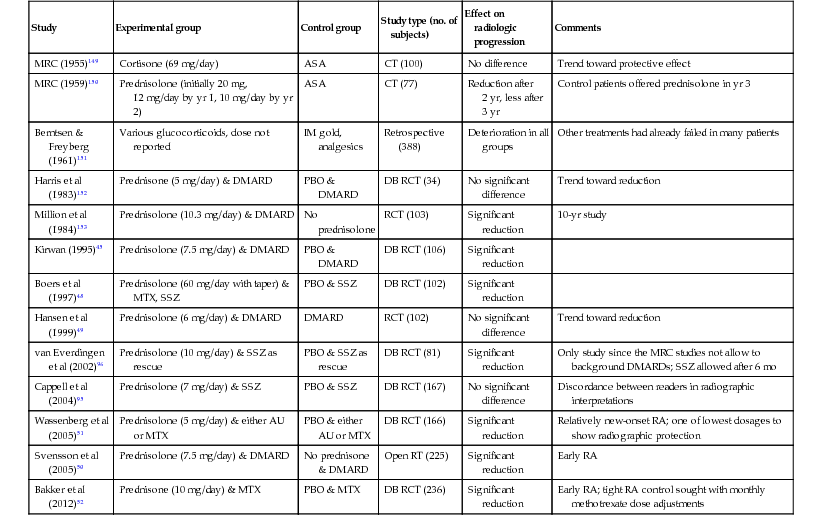
ASA, high-dose aspirin; AU, auranofin; CT, controlled trial; DB, double-blind; DMARD, disease-modifying antirheumatic drug; IM intramuscular; MRC, Medical Research Council; MTX, methotrexate; PBO, placebo; RCT, randomized controlled trial; RT, randomized trial; SSZ, sulfasalazine.
Efficacy in systemic connective tissue disorders and vasculitis
Medium- to high-dose glucocorticoids are the therapeutic mainstay in rheumatology for managing serious inflammatory disorders with life- or organ-threatening manifestations. Glucocorticoids have been used to treat serious extraarticular manifestations of RA including interstitial lung disease, bronchiolitis obliterans with organizing pneumonia, pericarditis, scleritis, and vasculitis.57
For these serious sequelae of RA, and in other major inflammatory disorders such as systemic lupus erythematosus (SLE), inflammatory myopathy, and systemic vasculitis, glucocorticoids are typically started at dosage in the range of 1 mg/kg/day of prednisone equivalent. Although highly effective in producing remission or improving control of these disorders, glucocorticoids do not inhibit the generation of platelet-derived thromboxane, and in certain forms of vasculitis patients can develop progressive ischemia despite glucocorticoid therapy.58
For life- or organ-threatening complications, intravenous pulse therapy is used at dosages of 250 to 1000 mg/day of methylprednisolone for a typical treatment period of 3 days. The intent of pulse therapy is to rapidly and aggressively suppress severe systemic inflammation while minimizing exposure to prolonged very-high-dose therapy. Rapidly progressive glomerulonephritis as well as pulmonary and central nervous system vasculitis are among the disorders for which very-high-dose pulse therapy has been advocated.59,60 Compared with prednisolone, methylprednisolone has more than threefold more nongenomic effects at equivalent dosages, which contributes to the preference for methylprednisolone over prednisone in this setting.61
Efficacy in polymyalgia rheumatica and large-vessel vasculitis
Glucocorticoids are the mainstay of therapy for polymyalgia rheumatica (PMR), giant cell (temporal) arteritis, and Takaysu arteritis.62,63 Due to the older age of most people with PMR and giant cell arteritis, glucocorticoids need to be used cautiously and monitored more closely in this population. Although a variety of other immunosuppressive agents have been tested as possible glucocorticoid-sparing agents, there are few compelling data that these agents are effective alternatives to glucocorticoids for treatment of these disorders. A typical prednisone starting dosage in giant cell arteritis is 60 mg/day, whereas 15 to 20 mg/day is usually adequate for PMR. Although efforts to taper glucocorticoids are initiated early in the disease course, over 6 months may be necessary to reach a low dose in persons with giant cell arteritis.64 It is not uncommon for patients with giant cell arteritis to require in excess of 2 years of glucocorticoid therapy. Glucocorticoid-tapering regimens for PMR are not well standardized, and symptoms often wax and wane over many years, remaining partially responsive to low-dose prednisone. Guidance from a meta-analysis suggests that once a prednisone dosage of 10 mg/day is reached, tapering by 1 mg/mo or less results in fewer relapses.65
Adverse effects
Despite the considerable benefits of glucocorticoids in controlling serious inflammation and improving functional status in a plethora of disorders, serious adverse effects dampen enthusiasm, particularly for its long-term use. Most studies of glucocorticoid toxicity tend to be retrospective and observational. The difficulty in differentiating adverse outcomes attributable to glucocorticoids from those occurring due to the underlying diseases or other comorbidities confounds potential associations. A strong physician selection bias for glucocorticoid use exists because physicians are inclined to treat patients with more severe disease with glucocorticoids (i.e., confounding by indication). The use of glucocorticoids at variable points in the disease course, limited data defining the threshold dose for particular adverse events,66 and toxicity reports for a heterogeneous group of glucocorticoid-treated diseases all further hinder interpretation of these data.
Several large retrospective reviews indicate that long-term, relatively low-dose glucocorticoid use is a significant independent predictor of numerous potentially serious adverse events.58,67-69 Lending further credibility to causality is the fact that the association between glucocorticoid use and adverse events was both dose and time dependent (Fig. 54.4). Both cumulative and mean glucocorticoid dose are independently associated with adverse events. There appear to be two distinct dose-related patterns of adverse events70: (1) a linear increase with increasing dose was found for a cushingoid phenotype, ecchymosis, leg edema, parchment-like skin, and sleep disturbance; (2) a threshold pattern showing an elevated frequency of events beyond a certain threshold value was observed for glaucoma, depression and listlessness, and increases in blood pressure. Dosages of 5 mg/day or more were associated with epistaxis and weight gain (Fig. 54.5).
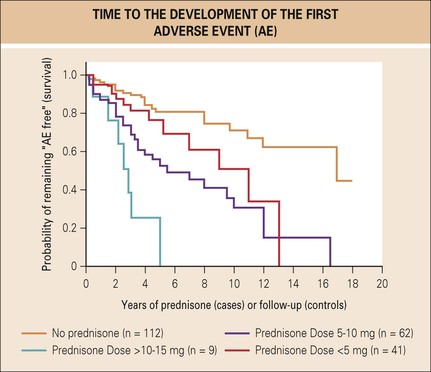
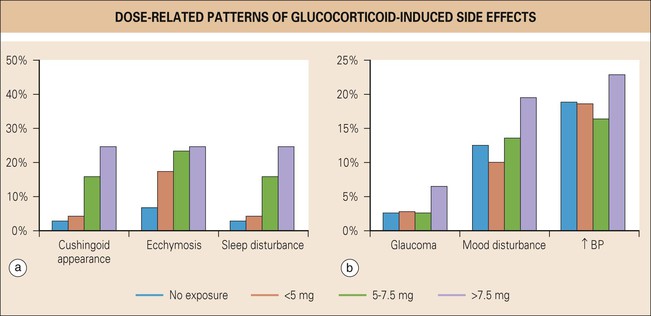
Less serious adverse effects (e.g., skin thinning, cushingoid appearance) are often of great concern to patients, whereas more debilitating toxicities, such as vertebral fractures and cataracts, initially may be unrecognized or asymptomatic.71 Compared with other antirheumatic agents, glucocorticoids have a low incidence of short-term symptomatic toxicity, and patients uncommonly discontinue therapy for this reason. What follows is an overview of the most common adverse effects that have been associated with glucocorticoid use. Recent international recommendations provide guidance on the safe use of glucocorticoids.72
Bone and muscle effects
Glucocorticoid-induced osteoporosis is the most potentially devastating complication of protracted glucocorticoid therapy. It is estimated from observational studies that as many as 40% of glucocorticoid users develop bone loss leading to fracture.73 Although glucocorticoids diminish sex hormone production and decrease circulating levels of receptor activator of NF-κB ligand (RANKL), both of which lead to enhanced osteoclast-mediated bone resorption, a direct glucocorticoid-induced defect in bone formation is thought to be the predominate pathway of importance.74,75 Glucocorticoid-induced osteoporosis initially affects trabecular bone. However, with long-term glucocorticoid use, cortical bone at sites such as the femoral neck is also affected.73 Comparison of the results of studies examining the bone effects of glucocorticoids is made difficult by the differences in timing of glucocorticoid initiation, variable dosing regimens, and use of different bone mass measurement techniques and disparities in the sites of measurement.
Observational studies of RA and other glucocorticoid-requiring diseases show a mean first-year loss of bone ranging from 1.5% to 20% at a dosage of 10 mg/day or less of prednisone.38
With continued use, bone loss remains greater than normal and is estimated to be approximately 1.5% to 3% per year, depending on dose, in subsequent years. Although a decline in bone mineral density (BMD) is strongly correlated with fracture risk, the rate of bone turnover, bone quality, and other factors also play important roles.76 Some data suggest that at very low doses, glucocorticoids prevent bone loss in RA because of their inhibitory effects on proinflammatory cytokines that modulate osteoclast activity as well as their beneficial effects on functional status, which promotes more weight-bearing activities.77,78 Several studies including a meta-analysis have failed to demonstrate an association of low-dose glucocorticoid use with low axial BMD, even in the setting of an increased fracture rate.78-81 However, the presence of biochemical changes in bone remodeling with very-low-dose oral82 or even intraarticular83 administration argues against a “safe” glucocorticoid dose from the standpoint of bone.84
The confounding effects in trying to identify an association between glucocorticoid treatment and osteoporosis in RA relate to two principal issues: the heightened risk of osteoporosis caused by the RA disease process itself85-87 and a higher rate of osteoporosis in postmenopausal women, the demographic group with the highest prevalence of RA.88
Given the inconsistent findings of BMD studies and the knowledge that fractures in glucocorticoid-treated patients occur at a higher BMD and depend on other factors,89,90 it is necessary to examine long-term studies that evaluate actual fracture incidence. In a large cohort of RA patients,91 34% of more than 300 women taking a mean dose of prednisone of 8.6 mg/day had a self-reported fracture within 5 years. Two case-control studies of hip fracture in patients both with and without RA showed a twofold increased risk even after adjustment for the presence of RA.85,92 Cohort studies identify fractures as one of the most commonly documented complications of supraphysiologic glucocorticoid use.67,68,93 Hip and other nonvertebral fractures occur with glucocorticoids in both a dose- and time-dependent fashion.84,94 Nevertheless, randomized controlled trials involving the most common glucocorticoid-requiring illness, RA, have not confirmed an increased rate of fracture.45,50,51,95,96 However, none of these trials has been large enough or long enough to fully clarify the magnitude of the fracture risk, some have not systematically assessed fracture outcomes in all patients, and in at least one,95 patients were allowed to use bone-protective medications.
Considerable advancement has occurred in the development of antiosteoporotic therapies, and numerous clinical trials have specifically tested such therapies in patients newly initiating glucocorticoid treatment and receiving long-term glucocorticoid therapy. All RA patients with or at risk of glucocorticoid-induced osteoporosis should receive initial therapy with adequate calcium and vitamin D,97,98 and strong evidence-based consideration should be given to the addition of a bisphosphonate99-103 or even potentially raloxifene as an alternative.104 Teriparatide, which may directly inhibit glucocorticoid-mediated deleterious effects on bone formation, is another treatment option for glucocorticoid users at high risk of fracture.105 Internationally, a variety of specialty societies have promulgated recommendations for appropriate testing and treatment to prevent glucocorticoid-induced osteoporosis.106-111
Osteonecrosis is a significant problem in patients receiving high-dose glucocorticoids, particularly for treatment of SLE. Osteonecrosis is more strongly associated with the peak dose of glucocorticoid than with the cumulative dose,112 perhaps owing to osteocyte and osteoblast apoptosis.75 The presence of antiphospholipid antibodies contributes to osteonecrosis independent of glucocorticoid use. Osteonecrosis is seldom seen when the prednisone dosage is maintained at less than 20 mg/day.113
Like osteonecrosis, myopathy occurs very rarely in patients receiving glucocorticoids at low doses. In patients with inflammatory myopathy it is often difficult to disentangle ongoing muscle inflammation from glucocorticoid myopathy. Gradual muscle improvement with dose reduction is often a diagnostic clue because electromyography and muscle biopsy findings are often insufficiently specific to differentiate these two entities. Based on small studies, fluorinated glucocorticoid preparations, such as triamcinolone, are more strongly associated with myopathy than prednisone.114 Myopathy has been reported to occur at a dosage as low as 8 mg/day of triamcinolone after only 3 months of treatment. In general, myopathy attributable to prednisone requires a higher dose and longer duration of treatment.
Cardiovascular effects
Glucocorticoids at medium to high dose promote fluid retention based in some115 but not all116,117 studies. This can be problematic for patients with underlying heart or kidney disease, although susceptibility is modified by both glucocorticoid dose and dietary factors. Patients with essential hypertension require closer surveillance of blood pressure and may need modification of their antihypertensive regimens while receiving glucocorticoid therapy. In some studies glucocorticoids, at even low or moderate doses, have been associated with hypertension, but this may be a reflection of channeling bias due to RA disease severity.118 Glucocorticoids can cause sodium retention and can potentiate vasopressive response to catecholamines and angiotensin II. In patients receiving less than 10 mg/day, age and elevated pretreatment blood pressure likely better explain significant hypertension than the use of glucocorticoids.119 There is inconsistent information on the effects of glucocorticoids on serum lipid profiles.120-123 To date, randomized controlled trials have not identified low-dose glucocorticoid therapy as an additional risk factor for dyslipidemia.
Another difficult-to-study potential toxicity of low-dose glucocorticoids is the development of premature atherosclerotic vascular disease. Increasing attention to the importance of accelerated atherosclerotic disease in RA and other inflammatory conditions has raised interesting questions about the role of chronic inflammation on the vascular endothelium.124 Although atherosclerotic vascular disease is known to be accelerated in patients with Cushing disease and at least one study showed increased carotid plaque and decreased arterial compressibility in RA patients receiving glucocorticoids,125 there are insufficient data to confirm an independent risk in patients taking low-dose glucocorticoids, in particular. A systematic review of low-dose glucocorticoid therapy,126 somewhat in contrast with the lipid results, reported that four out of six studies reviewed found low-dose glucocorticoid therapy to be associated with major cardiovascular events, including myocardial infarction, stroke, mortality and a composite index of cardiovascular events. Nonetheless, one of the largest and most carefully conducted cohort studies did not find an increased cardiovascular risk with dosages of less than 7.5 mg/day of prednisone.127 There is also evidence that glucocorticoids may tend to have a protective vascular effect in persons with polymyalgia rheumatica.128
Cardiac arrhythmias have been described during pulse-dose glucocorticoid therapy. This rare occurrence, reported mostly in patients with renal disease, calls for a preinfusion check of serum electrolyte levels and strong consideration of cardiac monitoring during infusion. The use of slower infusion rates (administration over 1 to 2 hours) may reduce this concern.
Dermatologic effects and appearance
Even at a low dose, skin thinning and ecchymoses are one of the most common glucocorticoid adverse events. A cushingoid appearance manifests as moon facies, truncal obesity, and buffalo hump and is very troubling to patients, but it is uncommon at doses below the physiologic glucocorticoid replacement range. Moon facies develops in slightly over 10% of patients receiving even short-term low-dose therapy. Use of an alternate-day schedule decreases the incidence of cushingoid appearance. Steroid acne and, to a lesser extent, hirsutism and stria are other undesirable dermatologic adverse effects that can occur even at lower doses. Glucocorticoids also impair wound healing by inhibiting the synthesis of matrix metalloproteinases and collagen.129 Weight gain and fat redistribution occur at moderate to high doses, in particular. The effects of hyperinsulinemia on leptin, increased adipocyte conversion of cortisone to cortisol, and increased appetite all contribute to weight gain and fat redistribution.
Gastrointestinal effects
Although glucocorticoids are considerably less toxic to the upper gastrointestinal (GI) tract than nonsteroidal antiinflammatory drugs (NSAIDs), glucocorticoids slightly increase the risk of adverse GI events such as gastritis, ulcers, and GI bleeding. The increased risk of GI events with glucocorticoid use is very small, with estimated relative risks varying from 1.1 (not significant) to 1.5 (marginally significant).130,131 In addition to reports of upper GI morbidity, there are case reports of intestinal rupture, diverticular perforation, and pancreatitis thought to be caused by glucocorticoids. Glucocorticoids frequently are used concurrently with NSAIDs in rheumatology, and the combination synergistically results in a higher risk of GI adverse events. Glucocorticoids cause a nearly twofold increased risk of GI adverse events among NSAID users,132 and the combined use of NSAIDs and glucocorticoids results in a more than fourfold increased risk of GI adverse events compared with nonusers.131
Infectious diseases
Moderate- to very-high-dose glucocorticoid therapy leads to an increased risk of serious infections. However, no studies have adequately explored the risk of infection in patients treated with low-dose glucocorticoids. The risk of infection appears to be lessened by initiating alternate-day therapy. The association of glucocorticoids with infectious adverse events becomes an even greater concern if glucocorticoids are used in combination with biologic antirheumatic agents.133 Among patients in the Consortium of Rheumatology Researchers of North America registry who were prescribed methotrexate, TNF antagonists, or other DMARDs, low-dose prednisone use overall was not associated with increased risk of infection.134 In a nested case-control study, the adjusted relative risk increased from 1.10 for dosages of less than 5 mg/day to 1.85 for dosages of more than 20 mg/day. Although the overall relative risk was low at 1.20, the absolute risk was high, with one additional infection seen for every 13 patients treated with glucocorticoids (at all dosage levels) for 1 year.135 In a study of RA patients comparing infection risk with biologic therapies to that with nonbiologic DMARDs, glucocorticoid use was the factor most associated with serious infection outcomes.136 A meta-analysis of 71 controlled clinical trials involving over 2000 patients randomly allocated to receive systemic glucocorticoids in the setting of different diseases, the relative risk for infectious complications overall was 1.6 (95% confidence interval, 1.3 to 1.9). The rate was not increased in patients given a daily dose of less than 10 mg or a cumulative dose of less than 700 mg of prednisone.137 Similarly, a different systematic review identified 15 studies assessing infection risk of low-dose glucocorticoid therapy in RA patients and did not find consistent associations between infection risk and low-dose glucocorticoid treatment.35,134-137 At higher doses, glucocorticoids appear to be a risk factor for tuberculosis.138
Metabolic and endocrine effects
Glucocorticoid users with diabetes mellitus commonly have higher blood glucose levels while taking glucocorticoids. Moreover, in patients with early diabetes or glucose intolerance new-onset hyperglycemia or, rarely, a nonketotic hyperosmolar state can develop de novo. Ketosis is very rare in glucocorticoid-associated diabetes, because the gluconeogenic and glycogenic effects of glucocorticoids offer protection against this complication. It is uncommon for frank diabetes to develop as a result of glucocorticoid therapy. A recent, small, randomized trial of short-term treatment with prednisolone 60 mg or 30 mg per day demonstrated no deterioration of glucose tolerance in patients with active RA.138a
A significant concern with long-term glucocorticoid use is HPA insufficiency. HPA insufficiency is both dose and duration specific. High-dose therapy results in protracted suppression of adrenocorticotropic hormone release and adrenal hyporesponsiveness in as little as 5 days. More typically, adrenal suppression can be detected in 6 weeks at 10 mg/day and in 4 weeks at 15 mg/day.66 Spontaneous recovery of the HPA axis is normal in patients taking 5 mg or less of prednisone; however, even subphysiologic dosages (less than 7.5 mg/day) given for long periods may lead to HPA blunting.139 HPA suppression is worsened if glucocorticoids are given twice daily.
Neuropsychiatric effects
Patients taking even low-dose glucocorticoid therapy report a slight increase in their overall sense of well-being, which appears to be independent of improvement in disease activity. Symptoms of akathisia (motor restlessness), insomnia, and depression are also occasionally observed in patients taking low-dose therapy. Memory impairment can occur even at low doses, particularly in older patients. Daily split-dose therapy, in particular, tends to be troublesome because the evening dose promotes sleep disturbances. True glucocorticoid psychosis is distinctly uncommon at dosages of less than 20 mg/day of prednisone.
Ophthalmologic effects
The development of posterior subcapsular cataracts is a well-described complication of prolonged glucocorticoid use. There is no minimal safe dose with respect to this complication, and reports exist of cataract formation even with the use of inhaled glucocorticoid preparations.140 Although a very-low-dose glucocorticoid threshold was seen for cataracts (less than 5 mg/day) in one study,54 cataracts rarely occur in patients taking less than 10 mg/day for less than 1 year.141,142 Nearly a third of RA patients taking a mean dosage of 8 mg/day for an average of 7 years developed cataracts.67 Cortical cataracts also have also been associated with glucocorticoid use.143
In addition to cataracts, glucocorticoid-treated patients can develop increased intraocular pressure, which can lead to visual disturbances. The development of frank glaucoma is rare, particularly with low-dose therapy, and it tends to appear in patients who are otherwise genetically predisposed.
Glucocorticoid use in pregnancy
Glucocorticoids are often used in pregnant patients to control peripartum inflammatory disease activity. Although glucocorticoids are safer than many other antirheumatic agents during pregnancy, their use can cause fetal growth retardation and low birth weight of offspring. It is difficult, however, to fully discern whether these adverse fetal outcomes are caused by the glucocorticoids or by the underlying chronic inflammatory disorder. Prednisone or prednisolone is preferred over other glucocorticoids if the aim is to treat a pregnant mother, because the placenta will convert active prednisolone back to prednisone and thereby limit fetal exposure. The American Academy of Pediatrics considers prednisone and its active metabolite prednisolone to be compatible with breastfeeding. Even at dosages above 1 mg/kg/day, the amount of glucocorticoid secreted into the breast milk is less than 10% of a nursing infant's endogenous cortisol production.
Practical recommendations
Evidence-based treatment guidelines
In an era of internationally rising health care costs and highly efficacious but very expensive biologic disease-modifying agents, glucocorticoids provide an apparently inexpensive approach to management of inflammatory and autoimmune disorders. Although the cost per dose of oral glucocorticoid is low, other costs such as those associated with adverse effects must also be considered.144 Monitoring for glucocorticoid adverse effects, as suggested by groups such as a EULAR task force,145 may mitigate some but not all toxicities.
Once the decision is made to initiate glucocorticoid treatment, every effort should be made to use these agents as safely and effectively as possible. Accumulating data argue that more aggressive use of glucocorticoids earlier rather than later in the course of RA might be best. However, practitioners skeptical of the disease-modifying benefits of glucocorticoid therapy or excessively concerned about glucocorticoid adverse effects should strive to use the lowest effective dose. Despite the evidence and an international call to action to prevent glucocorticoid-induced osteoporosis among even long-term glucocorticoids users, fewer than half of all long-term glucocorticoid users are receiving adequate bone prophylaxis or evaluation.146
Perioperative and stress-dose considerations
Long-term glucocorticoid users preparing for major surgery or undergoing considerable physiologic stress associated with severe illness should receive prophylactic stress-dose glucocorticoids, commonly in conjunction with a mineralocorticoid. Hydrocortisone at a dosage of 100 mg three times per day is empirically recommended, but for patients taking low-dose glucocorticoids or undergoing less significant physiologic insult, half this dose is often sufficient. Identifying which patients require stress-dose glucocorticoids requires consideration of both dose and duration of glucocorticoid use. Guidelines for low-dose use were discussed previously. Any person who has received more than 20 mg/day of prednisone equivalent for longer than 3 weeks or who has developed clinical Cushing syndrome should receive prophylaxis. If clinically feasible, the need for stress-dose glucocorticoids can be determined by testing for the responsiveness of the HPA axis using a low-dose synthetic adrenocorticotropic hormone (cosyntropin) stimulation test.
Glucocorticoid use in children
Growth retardation can occur in children receiving glucocorticoids because these agents inhibit linear bone growth and delay epiphyseal closure. Regular daily administration of more than 7.5 mg prednisolone is associated with inhibition of linear growth. The mechanism of action is unknown, although suppression of growth hormone secretion and other metabolic effects can contribute. Alternate-day administration of the same total dose reduces this effect147 and is preferred, if clinically practical.
Glucocorticoid withdrawal regimens and alternate-day therapy
There is little science to guide the necessary but challenging process of glucocorticoid dose reduction. In conditions such as RA, a very subtle dose reduction is most effective in promoting eventual glucocorticoid discontinuation. In SLE and other diseases in which immune complexes play a significant role in pathogenic consequences, alternate-day therapy is a consideration once disease control is achieved. Alternate-day therapy serves to minimize HPA axis suppression and, in turn, certain adverse effects, and it affords a reduction in cumulative glucocorticoid dose. In RA, however, alternate-day therapy often is not well tolerated due to an increase in joint symptoms on days off therapy.
Steroid withdrawal syndrome is not clearly associated with HPA insufficiency but presents as extreme weakness and arthralgias. Glucocorticoid tapering from 20 mg/day by increments of 2.5 mg/day every 2 weeks resulted in rebound deterioration in over half of RA patients.46 Although convenience and individual patient disease response should be accommodated, a relatively stable decrement of 10% to 20% of glucocorticoid dose every 1 to 2 weeks is recommended until the dose reaches 10 mg; then tapering should proceed more slowly for long-term users. Indeed, the difficulty in withdrawing patients from glucocorticoids is sometimes cited as a compelling reason for not initiating them.148 Many rheumatologists report significant difficulties in tapering glucocorticoids in most RA patients, and abrupt withdrawal can result in dramatic flares in disease activity. Thus it is recommended that in patients receiving long-term low-dose glucocorticoids for inflammatory arthritis or polymyalgia rheumatica the dose be lowered in increments as small as 1 mg/mo (sometimes facilitated by cutting the dose on alternate days) to maximize their prospects for successful withdrawal. However, further research on appropriate tapering regimens is greatly needed.
