Metabolic, drug-induced, and other noninflammatory myopathies
George Stojan, Lisa Christopher-Stine
▪ The differential diagnosis of polymyositis and other inflammatory myopathies includes metabolic disorders and drug- or toxin-induced myopathies in addition to endocrine disorders, muscular dystrophies, and infectious etiologies..
▪ The primary metabolic myopathies are inherited disorders of muscle glycogen, lipid, or mitochondrial metabolism; they may cause episodic exercise intolerance along with myoglobinuria or progressive proximal muscle weakness.
▪ Drugs and toxins can induce myopathies through direct toxic effects on muscle, induction of autoimmune-mediated inflammatory myopathy, or metabolic derangements.
Metabolic myopathies
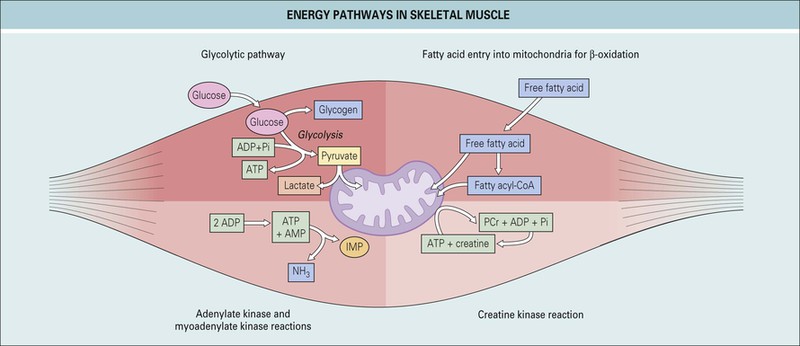
Muscle contraction and relaxation are energy-dependent processes that derive energy from the hydrolysis of adenosine triphosphate (ATP). The supply of ATP is crucial, and several pathways have evolved, with each supplying energy under specific conditions to ensure a continuous supply of energy under a variety of circumstances. The processes responsible for maintaining a continual supply of ATP include glycogen and glucose metabolism, oxidative phosphorylation, the creatine kinase (CK) reaction by which phosphocreatine is converted to ATP, the purine nucleotide cycle, and lipid metabolism.1
Glycogen/glucose metabolism (Fig. 151.1)
Anaerobic glycolysis is the main metabolic pathway used in the setting of limited oxygen supply during exercise. It is used during high-intensity, sustained, isometric muscle activity.1 It is inefficient from an energetic standpoint and produces only two ATP molecules per glucose molecule, which is 19 times less than the full energy potential of a glucose molecule. Despite its inefficiency, it is a rapid process, approximately 100 times faster than oxidative phosphorylation. The final step in the pathway is conversion of pyruvate to lactate, which leads to accumulation of lactic acid.
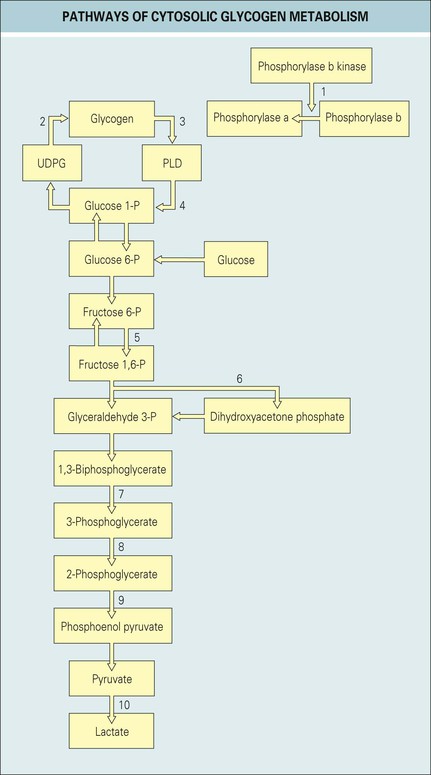
Aerobic glycolysis is more efficient; however, the price needed to maintain this system is high: it requires functional mitochondria, a functioning circulatory system with a constant oxygen supply, and the ability to eliminate carbon dioxide. It is used as the main supply of energy during sustained, dynamic forms of exercise such as walking, but if short bursts of energy are needed, the system is often overwhelmed and anaerobic glycolysis takes over.
The phosphocreatine pathway acts as a “buffer” of ATP stores by limiting changes in ATP and allowing rapid formation of ATP during high-intensity exercise. The amount of phosphocreatine in muscle is small, and it is not able to sustain activity independently.
ATP can also be produced by the adenylate kinase reaction, which catalyzes the conversion of two adenosine diphosphate (ADP) molecules into one ATP and one adenosine monophosphate (AMP); however its clinical significance is limited.
The oxidative phosphorylation system (Fig. 151.2), present in the inner mitochondrial membrane, is the principal source of energy in muscle and other tissues. The flow of electrons from the reduced form of nicotine adenine dinucleotide (NADH) to the last enzyme in the electron transport chain, cytochrome-c oxidase (complex IV), releases energy that is used in the synthesis of ATP.
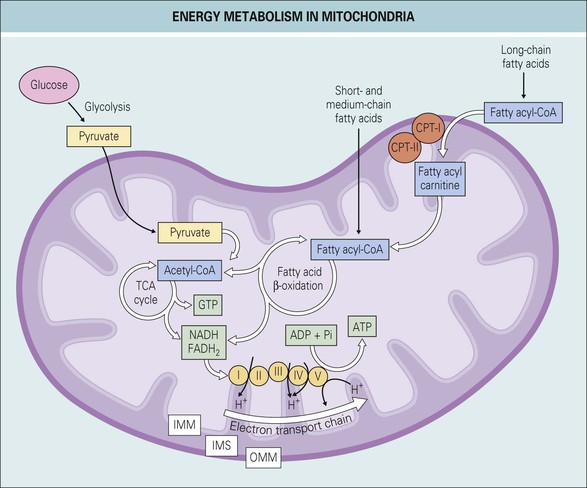
Lipid metabolism
The major energy substrates used by resting muscle are fatty acids derived from very-low-density lipoprotein (VLDL) or from stored triglycerides. The metabolism of short-, medium-, and long-chain fatty acids is slightly different, yet these biochemical details have great clinical relevance.
Long-chain fatty acids are the major source of energy for prolonged, low-intensity exercise lasting longer than 40 to 50 minutes.1 Unlike short- and medium-chain fatty acids, they are not able to freely cross the mitochondrial membrane and need to undergo a series of modifications. The first step is their conversion to coenzyme A (CoA) thioesters, which are then linked to carnitine by the enzyme carnitine palmitoyltransferase I (CPT-I), located on the inner side of the outer mitochondrial membrane. A translocase then transfers the acylcarnitine product across the inner mitochondrial membrane. Once in the mitochondrial matrix, CPT-II is required to convert the acylcarnitine molecule back into a CoA thioester and carnitine. Only then can the long-chain fatty acid derivative enter the β-oxidation pathway.
Short-chain and medium-chain fatty acids have a much less convoluted metabolic fate because they cross the mitochondrial membranes without difficulty and enter the β-oxidation pathway directly after activation to CoA thioesters. With each cycle of β-oxidation, a two-carbon fragment is cleaved and one acetyl-CoA molecule is generated. The acetyl-CoA then enters the citric cycle.
Part of the acetyl-CoA produced in the liver is used for ketone production at baseline. During periods of carbohydrate deprivation or fasting, ketone production is upregulated and it becomes an important source of energy for many tissues, including the brain.
General principles
The substrate used by muscle depends on the type of activity. Fatty acids are the predominant energy source at rest, whereas intense bursts of exercise will activate anaerobic glycolysis.2 Mild exercise will initially use aerobic glycolysis but, if sustained, will gradually switch to fatty acid oxidation as the main energy source after 4 hours. Submaximal exercise of low intensity will use both glucose and fatty acids, and high-intensity exercise will use mainly aerobic glycolysis.3,4
The metabolic myopathies are characterized, in general, by dynamic rather than static findings. This means that the symptoms and signs are usually triggered by activity. The most common phenotypes are exercise intolerance, exercise-induced weakness and muscle cramps, and only rarely, progressive, fixed weakness.1 Importantly, patients with exercise-induced symptoms may not have any evident signs of muscle disease, such as weakness or muscle atrophy, and may have normal findings on electromyography (EMG) and routine laboratory values.
The diagnostic approach to the metabolic myopathies includes blood tests (serum CK [Box 151.1], lactate, ammonia, acylcarnitine, and amino acid profile), urine tests (organic acids and myoglobin), muscle biopsy (light and electron microscopy and biochemical assays), EMG, exercise testing, genetic testing, and magnetic resonance imaging (MRI).5 Before proceeding with expensive or invasive diagnostic modalities such as genetic testing and muscle biopsy, simple exercise tests may be used for screening purposes.
The ischemic forearm exercise test is a highly sensitive and specific diagnostic screening test for muscle glycolytic disorders. The protocol involves inflating a blood pressure cuff on the upper part of the arm to 20 mm Hg higher than systolic blood pressure. The patient is given a handgrip dynamometer and told to squeeze it at maximum strength for 1 second followed by 1 second of rest. The cuff is released immediately after exercise. Blood samples from the tested arm are drawn at rest, during exercise, and during recovery, and blood levels of lactate and ammonia are measured.6 Normal subjects exhibit a threefold to fivefold rise in lactate and ammonia levels within 5 minutes after the end of the exercise, with a return to baseline about 10 to 15 minutes after cessation of the test.
If the lactate and ammonia responses are normal and suspicion for an underlying metabolic or mitochondrial myopathy persists, the next screening step is an aerobic exercise test. Patients with mitochondrial myopathies may have elevated resting lactate levels, but this finding is not universal. Cycle or treadmill ergometry is used to assess the cardiorespiratory response, arteriovenous oxygen difference, and lactate levels. Classically, patients with mitochondrial myopathies exhibit impaired oxygen extraction by the muscle, elevated lactate levels, and an intense cardiorespiratory response to exercise.7
The major categories of metabolic myopathies are defects in glucose/glycogen metabolism, disorders of fatty acid oxidation, mitochondrial diseases, and myoadenylate deaminase deficiency.5
Defects in glucose/glycogen metabolism
Depending on the specific enzyme deficiency, defects in glucose and glycogen metabolism can result in protean manifestations ranging from lethal, multiorgan involvement in infancy to isolated, dynamic or static muscle weakness in adulthood.
Patients with glycogen storage diseases affecting the muscles usually complain of random muscle aches and cramps at rest and easy fatigability with exertion and may occasionally have acute rhabdomyolysis induced either by brief, intense isometric exercise or by less intense but sustained dynamic exercise.8 Progressive muscle weakness mimicking polymyositis may occur. Serum CK levels may be elevated, EMG and histologic evaluation show myopathy with no inflammation, and even the Peter and Bohan criteria for polymyositis may be fulfilled.9 See Table 151.1 for an overview of glycogen storage diseases affecting skeletal muscle.
TABLE 151.1
Glycogen storage diseases affecting skeletal muscles
| Type | Defect | Clinical features | Laboratory findings | Treatment |
| Type II (Pompe disease) | Lysosomal acid maltase | Proximal muscle weakness with pelvic girdle, paraspinal, and diaphragmatic weakness | ↑ CK, ↑ AST Decreased or absent acid maltase activity in muscle or cultured skin fibroblasts | Enzyme replacement therapy with alglucosidase alfa, high-protein diet |
| Type V (McArdle disease) | Myophosphorylase deficiency | Exercise intolerance, muscle cramps, dark red urine after intense exercise Usually occurs in the 2nd or 3rd decade | Myoglobinuria, ↑ CK at rest and after exercise, ↑ NH3 and ↑ uric acid with exercise, abnormal enzyme assays in muscles | Ingestion of sucrose, glucose, or fructose immediately before exercise Low-dose creatine supplementation |
| Type VII (Tarui disease) | Phosphofructokinase | Same as McArdle disease plus severe exercise intolerance, compensated hemolytic anemia, acute exercise intolerance after a carbohydrate load | ↑ CK and ↑ bilirubin, reticulocytosis, hyperuricemia Abnormal enzyme assays in muscles | Avoid strenuous exercise for prevention of muscle cramps and myoglobinuria |
| Phosphoglycerate kinase deficiency | Phosphoglycerate kinase | Same as McArdle disease plus hemolytic anemia and CNS dysfunction | ↑ CK (not always), abnormal enzyme assays in muscles | Avoid strenuous exercise for prevention of muscle cramps and myoglobinuria |
| Lactate dehydrogenase deficiency | Lactate dehydrogenase, M subunit | Same as McArdle disease plus erythematous rash and obstetric difficulties because of uterine stiffness | ↑ CK (not always) | Avoid strenuous exercise for prevention of muscle cramps and myoglobinuria |
| Fructose-1,6-biphosphate aldolase A deficiency | Fructose-1,6-biphosphate aldolase A | Same as McArdle disease plus hemolytic anemia | ↑ CK (not always) | Avoid strenuous exercise for prevention of muscle cramps and myoglobinuria |
| Pyruvate kinase deficiency | Pyruvate kinase | Static symptoms, fixed muscle weakness, cramps | ↑ CK (not always) | Avoid strenuous exercise for prevention of muscle cramps and myoglobinuria |
| Muscle phosphorylase kinase deficiency | Muscle phosphorylase kinase | Same as McArdle disease with muscle atrophy | ↑ CK (not always) | Avoid strenuous exercise for prevention of muscle cramps and myoglobinuria |
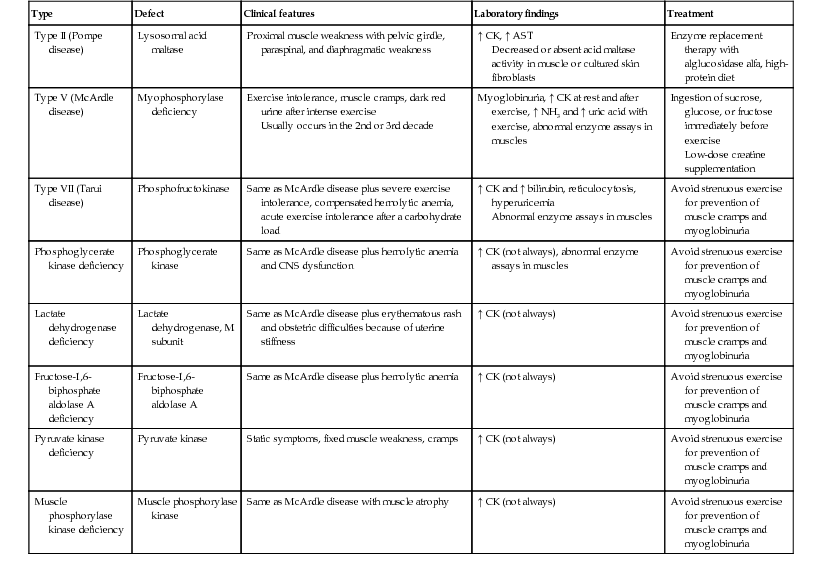
AST, aspartate aminotransferase; CK, creatine kinase; CNS, central nervous system; NH3, ammonia.
Adapted from Khan M. Carbohydrates. In: McPherson R, editor. Henry's clinical diagnosis and management by laboratory methods. 22nd ed. Philadelphia: Saunders; 2011, p. 1543.
Myophosphorylase deficiency (McArdle disease, glycogenosis type V)
McArdle disease is an autosomal recessive disease in which phosphorylase, the enzyme catalyzing the removal of 1,4-glucosyl residues from the outer branches of the glycogen molecule, is deficient. It is the most common disorder of skeletal muscle carbohydrate metabolism and one of the most frequent genetic myopathies.2
Onset occurs in the second or third decade with exercise intolerance and muscle cramps. Half of the patients describe episodes of burgundy-colored urine following intense exercise (as a result of rhabdomyolysis). Patients have no difficulty performing moderate exercise at their own pace even for long periods. Crises are induced by brief, intense exercise episodes such as sprinting, as well as by less intense activities such as stair climbing for longer periods. Acute crises are characterized by muscle cramps accompanied by a certain degree of rhabdomyolysis. The pathognomonic sign of McArdle disease is the “second wind” phenomenon, defined as marked improvement in exercise tolerance about 10 minutes into aerobic exercise involving large muscles.10 During the early phase of moderate exercise, glycogen and anaerobic glycolysis are the main source of energy. After 10 to 15 minutes of moderate to intense exercise, fatty acid metabolism becomes the main energy source, thus leading to a second-wind phenomenon.
Serum CK levels are usually high, even between acute crises. The forearm ischemic test is an excellent screening test and shows a flat venous lactate curve with exaggerated elevations in blood ammonia in McArdle disease (Table 151.2). There is a common misconception in the literature regarding the specificity of this test. McArdle disease is, in fact, indistinguishable from phosphofructokinase or debrancher enzyme deficiencies based on results of the forearm ischemic test, and hence diagnostic confirmation is always required. EMG shows myotonic discharges, fibrillations, and positive waves.11 Focal subsarcolemmal and intermyofibrillar accumulations of normally structured glycogen are seen in muscle biopsy specimens, but histochemical staining for phosphorylase reveals no activity.8
TABLE 151.2
The forearm ischemic test in metabolic myopathies
| Lactate | Ammonia | |
| Cori disease (GSD type III, liver and muscle involvement) | Impaired response | Normal or enhanced response |
| Cori disease (GSD type III, liver involvement only) | Normal | Normal |
| McArdle disease (GSD type V) | Severely impaired response | Normal or enhanced response |
| Tarui disease (GSD type VII) | Severely impaired response | Normal or enhanced response |
| Myoadenylate deaminase deficiency | Reduced response | Severely impaired response |
GSD, glycogen storage disease.
No specific treatment is available. Avoiding strenuous exercise is imperative. Glucose intake before exercise may diminish symptoms because the metabolic block is upstream of glucose metabolism.12 This intervention does not lead to consistent improvement and almost invariably results in weight gain. A high-protein diet and creatine supplementation may increase muscle endurance.13,14
Phosphofructokinase deficiency (glycogen storage disease type VII, Tarui disease)
Tarui disease is an autosomal recessive disease in which the key regulatory enzyme of the glycolytic pathway is deficient. Because of phosphofructokinase deficiency, the muscle is unable to use glucose as an energy source, so glucose intake in these patients leads to an “out-of-wind” phenomenon. The clinical features are similar to those of McArdle disease: muscle aches and cramps induced by exercise, as well as episodes of rhabdomyolysis. The characteristic features of Tarui disease include earlier onset, with the first symptoms occurring in childhood and more severe attacks frequently being associated with systemic symptoms such as nausea and vomiting. A compensated hemolytic anemia is characteristic and occurs secondary to decreased erythrocyte 2,3-biphosphoglycerate, which leads to a shortened erythrocyte life span.15 Serum CK is usually elevated between episodes. The forearm ischemic test shows no lactate elevation. Muscle biopsy demonstrates accumulation of normal glycogen. There is no effective therapy. A high-protein diet and aerobic conditioning have been used in case reports with variable success.16
Debrancher deficiency (Cori-Forbes disease, glycogenosis type III)
This is yet another autosomal recessive disease characterized by deficiency of the enzyme responsible for removal of the residual stubs of glycogen after digestion by phosphorylase. It is usually classified as a hepatic glycogenosis, but there are two subtypes: type IIIa, in which the enzyme deficiency affects both the liver and muscle, and type IIIb, in which only the liver is involved and debrancher activity in skeletal muscles is normal. The two subtypes can easily be distinguished with the forearm ischemic test, in which abnormal results are produced in type IIIa but not in type IIIb.
In contrast to McArdle and Tarui disease, muscle weakness in type IIIa is minimal in childhood and tends to become severe after the third or fourth decade. The weakness is static and additive. Rarely, exercise intolerance may be present.17 Patients generally have elevated transaminase levels at rest and are at high risk for liver failure, cirrhosis, and hepatocellular carcinoma.18 The serum CK level is usually elevated. EMG shows myopathy, whereas muscle biopsy specimens show free glycogen accumulation. No effective treatment is available. A high-protein diet is commonly recommended but has not been proved to be of consistent benefit.14
Acid maltase deficiency (glycogenosis type II, Pompe disease)
In contrast to the other glycogenosis just described, Pompe disease is an autosomal recessive deficiency of the lysosomal enzyme acid maltase that leads to intralysosomal accumulation of glycogen in various tissues.19 In its infantile form it is universally fatal. In the adult form it may have an onset between the second and sixth decade with progressive, proximal weakness that may mimic polymyositis.20 There is a wide range of phenotypic variability between these two forms. The serum CK level is elevated in all cases. There is no elevation in lactate levels with the forearm ischemic test, and EMG shows evidence of myopathic discharges with occasional myotonic and complex repetitive discharges.8 Muscle biopsy shows periodic acid–Schiff–positive vacuolar myopathy with glycogen storage within lysosomes. Muscle acid maltase activity is decreased to less than 10% of normal. Enzyme replacement with recombinant human α-glucosidase has had some success in treating the infantile form. In adults, benefits have been observed with a high-protein and low-carbohydrate diet.21
Fatty acid oxidation disorders
Although these disorders have many overlapping features, three predominant phenotypes can be recognized: hepatic, cardiac, and muscular. A detailed discussion of the first two is beyond the scope of this chapter. The muscle phenotype is characterized by hypotonia and recurrent episodes of rhabdomyolysis. Muscle pain and myoglobinuria are the usual initial signs and are characteristically induced by infection, general anesthesia, or a low-carbohydrate, high-fat diet.22 Patients have no muscle cramps or contractures, and there is no second-wind phenomenon.1
The major disorders of lipid metabolism that are manifested as isolated myopathy include (1) CPT-II deficiency, (2) very-long-chain acyl-CoA dehydrogenase (VLCAD) deficiency, and (3) long-chain acyl-CoA dehydrogenase (LCHAD) or trifunctional protein (TFP) deficiency.23
Carnitine palmitoyltransferase II deficiency
CPT-II deficiency is the most prevalent disorder of lipid metabolism and the most common overall cause of hereditary, recurrent myoglobinuria.24
CPT-II is located in the inner mitochondrial membrane and has the role of transporting long-chain fatty acids from the cytosolic compartment to the mitochondrial matrix so that they can undergo β-oxidation. A wide spectrum of phenotypes is noted in CPT-II deficiency, depending on the severity of the mutation.25 Patients are asymptomatic between episodes. Symptomatic episodes usually develop after prolonged exertion (typically longer than 30 minutes), and patients tolerate brief, intense isometric exercise without difficulty. The typical symptoms and signs of CPT-II deficiency consist of myalgia, cramps, muscle stiffness and tenderness, weakness, and occasionally, myoglobinuria.26 A second-wind or out-of-wind phenomenon is not seen. Similar crises may be induced by stress, fasting, viral infections, fever or exposure to cold, general anesthesia, use of nonsteroidal antiinflammatory drugs, and high doses of diazepam.8
A common laboratory finding that is used as a screening test in symptomatic patients is decreased total serum carnitine with an increased acylcarnitine fraction (elevated C16, C18:1, and C18:2). Findings on EMG and the ischemic forearm exercise test are normal. The diagnosis can be confirmed by whole blood DNA analysis, which detects known mutations in approximately 80% of patients.27
The mainstay of treatment is lifestyle changes, which include avoiding prolonged aerobic exercise (longer than 30 minutes), prolonged fasting, and cold exposure, as well as specific dietary requirements, including the use of a high-carbohydrate, low-fat, low-protein diet along with extra carbohydrate intake before sustained exercise.28
Very-long-chain acyl-CoA dehydrogenase deficiency
VLCAD deficiency involves the first step in the fatty acid oxidation of long-chain acyl-CoA intermediates of 20 to 12 carbons in length, thus affecting tissues dependent on fatty acid oxidation for cellular energy production, including the liver, heart, and skeletal muscle. The first symptoms usually occur in the neonatal period and consist of hypoglycemia and irritability. Affected patients have a high risk for recurrent rhabdomyolysis with exercise or other forms of stress, including illness. Dicarboxylic aciduria is universally present. The abnormal acylcarnitine profile is dominated by 5-cis-tetradecenoylcarnitine (C14:1).28 Treatment is based on restriction of long-chain fatty acids in the diet, accompanied by medium-chain triglyceride supplementation.
Long-chain acyl-CoA dehydrogenase deficiency and trifunctional protein deficiency
LCHAD resides in a TFP bound to the inner mitochondrial membrane, along with 2-enoyl-CoA hydratase and 3-ketoacyl-CoA thiolase. Isolated LCHAD deficiency is a common fatty acid oxidation disorder characterized by long-term complications caused by cardiomyopathy, neuropathy, and retinopathy, with a high risk for rhabdomyolysis associated with stress or exercise.29 The diagnosis is made by finding elevated hydroxyl long-chain acylcarnitines (OH-C16, OH-C18:1, and OH-C18:2) in the acylcarnitine profile.28 Enzyme and mutation testing will differentiate isolated LCHAD deficiency from TFP deficiency.
TFP deficiency is more severe, with a high risk for early mortality.29 It seems to be the only fatty acid oxidation disorder in which blood lactate levels are consistently elevated even when the patient is asymptomatic. Clinical features include hypoglycemia, hypotonia, dilated cardiomyopathy in the setting of hydroxydicarboxylic aciduria, and a normal blood acylcarnitine profile.28 Because of their systemic manifestations, LCHAD and TFP deficiencies are unlikely to be confused with the inflammatory myopathies.
Mitochondrial myopathies
This clinically heterogeneous group of disorders results from impairment of oxidative phosphorylation. It has a prevalence of 9.2 per 100,000, which makes it one of the most common inherited neuromuscular disorders.30 Primary mitochondrial myopathies may occur as a result of mitochondrial or nuclear DNA mutations. Those related to mitochondrial DNA mutations are inherited through maternal transmission and become apparent in late childhood or adult life. A smaller number are related to nuclear genes that encode proteins involved in the oxidative phosphorylation pathway, and they are manifested in early childhood. Secondary mitochondrial myopathies are caused by toxins, including zidovudine and clofibrate.
Clinically, they are well-defined syndromes associated with multisystem involvement and are not likely to be confused with the inflammatory myopathies.31 Mitochondrial myopathies include Kearns-Sayre syndrome; chronic progressive external ophthalmoplegia (CPEO); mitochondrial encephalomyopathy with lactic acidosis and strokelike episodes (MELAS); Leber hereditary optic neuropathy (LHON); neuropathy, ataxia, retinitis pigmentosa (NARP); myoclonic epilepsy with ragged red fibers (MERFF); mitochondrial neurogastrointestinal encephalomyopathy; and autosomal recessive cardiomyopathy-ophthalmoplegia.
An isolated mitochondrial myopathy may be accompanied by mild proximal limb weakness. Exercise intolerance marked by premature fatigue, headaches, nausea, and vomiting after submaximal exercise may be seen. Symptoms are worsened by fasting, minor illness, and stress.
The mitochondrial metabolic test battery includes CK, lactate and pyruvate, plasma carnitine, blood and urine amino acids, urine organic acids, and cerebrospinal fluid lactate and pyruvate.32 Fasting serum lactate levels are elevated in 70% of patients. The normal lactate-pyruvate ratio (<20 : 1) is increased. Serum CK is usually normal or mildly elevated. Carnitine levels are decreased with a relative increase in acylcarnitine levels. Muscle biopsy is often required for diagnosis. Histopathologic evaluation reveals characteristic ragged red fibers on Gomori trichrome stain (subsarcolemmal and intermyofibrillar accumulation of mitochondria that appear as bright red masses against the background of blue myofibrils).32 Confirmation of the diagnosis is achieved by molecular genetic testing for mutations most likely thought to be present based on the clinical and laboratory features. Treatment is mainly symptomatic, empirical, and palliative.33
Myoadenylate deaminase deficiency
AMP deaminase 1 catalyzes the conversion of AMP to inosine monophosphate in skeletal muscle and plays an important role in the purine nucleotide cycle. Deficiency of this enzyme is seen in 2% of muscle biopsy specimens regardless of the indication for biopsy, and it is also found in 2% of healthy, white individuals.34 The clinical manifestation of myoadenylate deaminase deficiency is thus heterogeneous, and it is most commonly asymptomatic. When symptomatic, exercise-induced fatigue, cramps, and myalgia are most common. Occasionally, myoglobinuria and rhabdomyolysis may occur after vigorous exercise. An increased serum creatine kinase level is seen in less than half of affected patients. The forearm ischemic test is highly sensitive and specific for the diagnosis of myoadenylate deficiency. AMP deaminase is the major ammonia-producing enzyme in muscle, so a flat ammonia response accompanied by a rise in plasma lactate is considered sufficient to ensure a reliable diagnosis.35 Findings on EMG are usually normal, with minor abnormalities reported in a small subset of patients. The diagnosis can be confirmed by histochemical stains or enzymatic assays. Myoadenylate deaminase deficiency can subsequently be confirmed in muscle biopsy specimens by histochemical stains or direct enzymatic assay.
Drug- and toxin-induced myopathies
A drug-induced myopathy is defined as an acute or subacute occurrence of myopathic symptoms, such as muscle weakness, myalgia, CK elevation, or myoglobinuria, in patients without muscle disease during or after exposure to therapeutic doses of certain drugs. Discontinuation of the offending agent leads to resolution of the symptoms in almost all cases.
The classification of drug- and toxin-induced myopathies according to the type of injury includes the following36:
1. Necrotizing myopathy—the prototype of drug-induced muscle injury. Muscle biopsy specimens are characterized by widespread necrosis with macrophage invasion. Inflammatory infiltrates are not seen. The typical example is statin-induced myopathy.
2. Inflammatory myopathy, which is indistinguishable histologically from polymyositis. It can be caused by statins, interferon alfa, or intramuscular injections of genes.
3. Thick filament loss myopathy. Critical care myopathy is the classic example.
4. Type II muscle fiber atrophy. This is the underlying abnormality in steroid-induced myopathies.
5. Mitochondrial myopathies. Ragged red, cyclooxygenase-negative fibers are seen on histologic examination, as well as increased lipid accumulation. Zidovudine is the typical representative.
6. Lysosomal storage myopathy, which is caused by the accumulation of amphiphilic drugs in lysosomes. Hydroxychloroquine is the classic representative.
7. Antimicrotubule myopathy, which involves disruption of the cytoskeleton. Microtubule inhibition is the main mechanism of action of colchicine.
8. Fasciitis, which is of historical interest because its main representatives include toxic oil syndrome and l-tryptophan. Clinically, these disorders mimic eosinophilic fasciitis.
Statins
Statins are a class of lipid-lowering agents that inhibit hydroxy-3-methylglutaryl–coenzyme A (HMG CoA) reductase (HMGCR). Statin myopathy is defined as muscle aches or muscle weakness in conjunction with increases in creatine phosphokinase (CPK) values to more than 10 times the upper limit of normal. According to the degree of muscle toxicity that they induce, the most myotoxic statin was cerivastatin, which was withdrawn from the market, followed by simvastatin, lovastatin, pravastatin, atorvastatin, and fluvastatin.37
Several clinically relevant scenarios may occur with statin therapy:
1. Asymptomatic elevation of CK up to 10 times the upper normal limit is the most common finding. Based on expert opinion, in this scenario no further changes in treatment are needed as long as the patient remains asymptomatic and the CK elevation remains stable.
2. Myalgia may occur in up to 25% of patients and usually resolves after switching to a different statin or discontinuing therapy.
3. Muscle weakness with CK elevation. Besides the well-described myopathies, an intriguing recent discovery of antibodies against HMGCR in a subset of patients with necrotizing myopathy38 highlighted the statins as the probable culprit of an immune-mediated necrotizing myopathy. If the myopathic symptoms and elevated CK persist with discontinuation of statin therapy, the possibility of an immune-mediated necrotizing myopathy must be considered. Nevertheless, this is a diagnosis of exclusion, and a meticulous workup for autoimmune, metabolic, paraneoplastic, or drug-induced myopathy should be pursued.
4. Rhabdomyolysis is a rare, but serious event characterized by highly elevated CK levels, myoglobinuria, and weakness. It may occur anytime during drug treatment, and the risk may be increased by a number of factors, including age, drug interactions, and renal dysfunction. The most important drug interactions that need to be avoided include the use of statins with amiodarone, cyclosporine, gemfibrozil, macrolides, and azoles.39 Pravastatin is metabolized by a different cytochrome P450 enzyme and seems to be the safest in this group, alongside atorvastatin.
Colchicine
Myopathy, peripheral neuropathy, and rhabdomyolysis have been reported in patients taking colchicine. The myotoxicity of colchicine is thought to be related to disruption of the microtubular cytoskeletal network, which results in intracellular accumulation of autophagic vacuoles. This myopathy may mimic polymyositis and classically occurs in patients taking colchicine for prolonged periods, with proximal muscle weakness and elevated CK developing insidiously. Muscle biopsy specimens are characterized by a vacuolar myopathy without prominent necrosis or inflammation. The myopathy resolves fully 3 to 4 weeks after discontinuing use of the drug. An acute myopathy has also been described in the literature but is very rare. The risk for colchicine myopathy is increased by age, duration of therapy, drug interactions (cyclosporine), and inappropriate dosing in patients with renal insufficiency.
Hydroxychloroquine
Prolonged use of high-dose chloroquine or hydroxychloroquine may lead to a reversible myopathy. Clinically and histologically, this myopathy resembles acid maltase deficiency.36 Progressive weakness and atrophy of proximal muscle groups may occur and be associated with mild sensory changes, tendon reflex depression, and abnormal nerve conduction. Importantly, serum CK levels are normal. Muscle biopsy specimens from patients with chloroquine- or hydroxychloroquine-induced myopathy demonstrate a vacuolar myopathy, often evident only on electron microscopy.40 Periodic examination of knee and ankle reflexes should be undertaken and any signs of muscle weakness investigated. Use of hydroxychloroquine should be discontinued if weakness occurs. The myopathy is reversible, but the symptoms may take months to resolve.
Corticosteroids
Exogenous or endogenous glucocorticoid excess leads to weakness secondary to type II fiber atrophy. The mechanism responsible for the steroid effects has not been completely elucidated. Some degree of muscle weakness may eventually develop in the majority of patients treated with corticosteroids, usually after a period of at least 4 weeks. The weakness is more severe and may develop more rapidly in those treated with fluorinated corticosteroids, such as dexamethasone, triamcinolone, and betamethasone, than in those treated with nonfluorinated corticosteroids, such as prednisone and methylprednisolone.41 The weakness is proximal and accompanied by atrophy of the affected muscles. The distal extremities and oculobulbar and facial muscles are spared. EMG shows low-amplitude motor unit potentials and the absence of any spontaneous electrical activity. Selective atrophy of type II muscle fibers and the absence of inflammation or muscle necrosis are characteristic histologic features.42 The recommended treatment of steroid myopathy is reducing the steroid dose or switching to a nonfluorinated steroid and encouraging exercise to prevent disuse atrophy.32
Toxicity from recreational drugs
Chronic alcoholism may lead to the development of both acute and chronic forms of myopathy. Chronic alcoholic myopathy is characterized by painless, progressive proximal muscle weakness and mild to moderate myopathic changes in muscle biopsy specimens (fiber necrosis, moth-eaten fibers, variability in fiber size, and type II fiber atrophy). Some long-term drinkers may experience an asymptomatic elevation in the serum CK level—as much as 20 times higher than normal levels—that is aggravated by physical activity.36 Acute ethanol myopathy is characterized by severe myalgia, highly elevated CK levels, proximal muscle weakness, and myoglobinuria. Usually, the illness is not severe and patients note resolution of the muscle swelling and pain over a period of 2 weeks. Patients who resume alcohol consumption are particularly prone to subsequent attacks of this form of myopathy.42
Endocrine myopathies
Thyroid disorders
More than 75% of hypothyroid patients and 67% of hyperthyroid patients have neuromuscular symptoms.43 Neuromuscular manifestations of hypothyroidism include proximal weakness, muscle stiffness and cramping, slow reflexes, and myoedema (local contracture after light percussion). Rarely, hypothyroidism may be accompanied by rhabdomyolysis. Another unusual finding that is sometimes seen in hypothyroid myopathy is muscular enlargement. Hoffman syndrome is defined as the combination of hypothyroidism with muscle stiffness, cramps, and muscular enlargement. Muscle enzymes are commonly high and rarely correlate with the weakness. EMG shows normal findings or mildly myopathic changes. The myopathy resolves with hormone replacement.
In hyperthyroidism, proximal muscular weakness with atrophy, brisk reflexes, and bulbar weakness may occur. Occasionally, distal weakness may be seen as well. Muscle enzymes are normal or low. EMG shows myopathic changes. The symptoms resolve a few months after normalization of thyroid hormone levels.44 A rare manifestation of muscular disease in hyperthyroidism is thyrotoxic periodic paralysis. Patients exhibit rapidly progressing paralysis, greater in the lower extremities and often worse in the proximal musculature. The signs and symptoms are clinically identical to those of familial periodic paralysis.
Adrenal disorders
Iatrogenic steroid myopathy is manifested as an insidious, proximal muscular atrophy with weakness. Myalgia may be present. Fluorinated steroid preparations, such as dexamethasone and betamethasone, are most often the culprits. CPK levels are normal and EMG shows no evidence of irritable myopathy. Muscle biopsy specimens typically show evidence of type II fiber atrophy. Cushing disease classically causes proximal weakness and may be severe. Clinically, the myopathy is indistinguishable from iatrogenic steroid myopathies.
Diabetes
Diabetic amyotrophy is a distinctive form of diabetic neuropathy typified by an abrupt onset of pain and asymmetric proximal weakness and wasting of the legs.45 It characteristically affects elderly patients with type 2 diabetes ranging from poor to good control and is often associated with weight loss. EMG shows multifocal denervation in the paraspinous and lower extremity muscles. Nerve conduction studies show an axonal neuropathy. Treatment includes optimizing diabetic control and the use of corticosteroids and intravenous immunoglobulins.
Skeletal muscle infarction is a very rare complication of poorly controlled diabetes mellitus. Patients have an acute onset of painful swelling of the affected muscle and, occasionally, a palpable mass. Because this entity is frequently misdiagnosed clinically, increased clinical awareness is important for early recognition, particularly in a diabetic patient with a painful thigh or leg swelling.46
Muscular dystrophies
Muscular dystrophies are a heterogeneous group of inherited muscular disorders that affect the stability of the sarcolemmal membrane.32
Duchenne muscular dystrophy
The prototypic example is Duchenne muscular dystrophy, an X-linked recessive disease characterized by the absence of dystrophin. Affected children are normal at birth, only to have a waddling gait, Gower sign (use of the arms to push oneself erect by moving the hands up the thighs), and calf pseudohypertrophy develop by 2 to 6 years of age. Because the stability of the sarcolemmal membrane is affected, there is ongoing degeneration and regeneration leading to elevated CK levels of up to 100 times normal. EMG shows myopathic changes. Muscle biopsy specimens with reduced or absent dystrophin staining are diagnostic. The diagnosis may be confirmed by demonstrating deletion of the dystrophin gene. No specific therapy is available. Death occurs at 14 to 20 years of age as a result of respiratory failure/cardiomyopathy.47
Becker muscular dystrophy
Becker muscular dystrophy is very similar to the Duchenne type but has a less severe phenotype because of a partial loss of dystrophin. Patients are typically ambulating until 15 years of age, but otherwise the clinical features are identical, with the exception of muscle biopsy specimens, which may reveal normal dystrophin staining. Dilated cardiomyopathy necessitating heart transplantation may occur.47
Emery-Dreifuss muscular dystrophy
Emery-Dreifuss muscular dystrophy is another X-linked recessive disorder that affects emerin. Emerin is an integral protein of the inner nuclear membrane in vertebrates and mediates anchorage of the membrane to the cytoskeleton. Clinically, patients have contractures of the elbows, neck, and spine, as well as scapulohumeroperoneal weakness. Cardiomyopathy and cardiac conduction defects are the most feared complications.47
Limb-girdle muscular dystrophies
Limb-girdle muscular dystrophies (LGMDs) are a heterogeneous group of diseases characterized by mutations affecting various structural proteins and enzymes. Their nomenclature is straightforward, with type 1 LGMD being autosomal dominant and type 2 being autosomal recessive. Subclassification with letters denotes specific genetic mutations. They include LGMD2A (calpainopathy; CAPN), LGMD2B (dysferlinopathy; DYSF), LGMD2C (γ-sarcoglycanopathy; SGCG), LGMD2D (α-sarcoglycanopathy; SGCA), LGMD2E (β-sarcoglycanopathy; SGCB), LGMD2F (δ-sarcoglycanopathy; SGCD), LGMD2G (telethoninopathy; TCAP), LGMD2H (TRIM32), LGMD2I (FKRP), LGMD2J (TTN), and LGMD2K (POMT1).47 Their prevalence approaches 1 in 100,000. The phenotypes cover the entire range between Duchenne and Becker muscular dystrophies. Patients generally have weakness and wasting restricted to the limb musculature, proximal greater than distal, with initial symptoms occurring in the second or third decade. Muscle biopsy specimens show muscle degeneration and regeneration. Muscle enzymes are typically elevated.42
LGMD2A (calpainopathy) and LGMD2B (dysferlinopathy) are the most common recessive LGMDs in North America. Calpainopathy is frequently associated with profound shoulder-girdle weakness and sometimes scapular winging. Dysferlin deficiency may be accompanied by distal weakness and, in a purely distal form, is associated with severe calf wasting and elevated serum CK levels.48 LGMD2B may be misdiagnosed as polymyositis because of its onset in adult life, marked elevation of serum CK levels, and the presence of inflammatory infiltrates in muscle biopsy specimens.49 Confirmation of the diagnosis begins with a muscle biopsy specimen, which demonstrates dystrophic changes, as well as reduced levels or absence of calpain or dysferlin on immunohistochemical staining. Molecular genetic studies are needed to confirm and define the character of the mutation.
The sarcoglycanopathies are forms of LGMD (types 2C, 2D, 2E, and 2F) caused respectively by mutations in the γ-, α-, β-, and δ-sarcoglycan genes, and they affect primarily white individuals. Onset is most often in early childhood, and loss of ambulation generally occurs before the age of 16 years. However, milder phenotypes do occur, with proximal muscle weakness first prompting medical attention in young adulthood. Muscle weakness is initially detectable in the pelvic girdle and later results in scapular winging in the shoulders. Respiratory muscle involvement occurs late in the disease and frequently leads to respiratory failure and death. Serum CK levels are elevated 10 to 100 times the upper limit of normal, typically higher than in the other forms of LGMD.
Myotonic dystrophy
Myotonic dystrophy (Steinert disease, myotonic dystrophy type I, dystrophia myotonica) is the most common adult dystrophy. It is an autosomal dominant disease caused by mutations in the gene that encodes for dystrophia myotonica protein kinase on chromosome 19 (DMPK). The mutation occurs in an untranslated region of the gene. Within the region are 5 to 30 repeating sequences of three nucleotides (CTG trinucleotide repeats). In myotonic dystrophy these CTG repeats expand into the hundreds or thousands. In general, the size of the expansion reflects the severity of the illness.
Clinically, slowly progressive limb weakness, distal more than proximal, is characteristic. The most commonly affected muscles are the facial muscles, distal muscles of the forearm, dorsiflexors of the foot, intrinsic muscles of the hand and feet, and the oropharyngeal and extraocular muscles.47 The pelvic girdle, hamstrings, soleus, and gastrocnemius are spared. Myotonia can be elicited by percussing the thenar eminence or by observing delayed muscular relaxation after forceful contractions. CK is normal to mildly elevated and EMG shows myotonic discharges. The extramuscular features have diagnostic and prognostic significance. Smooth and cardiac muscle is most commonly involved, but the central nervous system, bone, and endocrine system may also be affected. Cataracts are almost universal. Testicular atrophy, reduced intelligence, depression, personality disorders, male pattern baldness, diabetes, and sleep apnea are common manifestations.
Proximal myotonic myopathy
Proximal myotonic myopathy (myotonic dystrophy type 2) is milder than myotonic dystrophy. Patients have a slowly progressive proximal myopathy that spares the face. Muscle pain and stiffness may be prominent. Myotonia is not as prominent as myotonic dystrophy on examination. CK is normal to mildly increased, and EMG shows myotonic discharges. Common manifestations include cataracts, cardiac conduction defects, fatigue, and obstructive sleep apnea. Proximal myotonic myopathy may be a close clinical mimic of idiopathic inflammatory myopathies.
Infectious myopathies
The musculature is inherently resistant to infections. Infectious myositis is rare and tends to occur in the setting of muscular injury, ischemia, or trauma. Numerous infections can cause myopathy. It is helpful to categorize infectious myositis on the basis of clinical manifestations.50
Pyomyositis
Pyomyositis is a primary infection of skeletal muscle that is often associated with abscess formation. Intermuscular abscesses and abscesses extending into muscles by contiguous spread from nearby tissues are not classified as pyomyositis.
Pyomyositis is common in tropical areas and is responsible for up to 4% of hospital admissions in Africa. It is a rare diagnosis in the United States and most commonly described in human immunodeficiency virus (HIV)-infected and immunosuppressed patients, with approximately a third of cases occurring in previously healthy individuals.51
The most commonly identified etiologic agent is Staphylococcus aureus (75% in the United States), followed by group A streptococci, as well as rare cases of groups B, C, and G streptococci, Pneumococcus, Haemophilus, Aeromonas, Pseudomonas, Klebsiella, and Escherichia.
Most commonly, a single muscle is affected, but in up to 40% of cases, multiple muscles may be involved.52 The incidence of pyomyositis by body site may be reviewed in Figure 151.3.
In the initial, invasive stage, local, tender swelling develops with no fluctuation or erythema. Aspiration yields no pus, but 2 or 3 weeks later, the occurrence of high spiking fevers associated with extreme pain and swelling of the affected muscle is the harbinger of the suppurative period. Imaging studies demonstrate an abscess and aspiration yields pus.
Serum CK is usually normal and blood culture results are positive in less than 50% of cases.53,54 Besides nonspecific laboratory markers of infection (leukocytosis, C-reactive protein, erythrocyte sedimentation rate, etc.), which are invariably elevated, the most important tool in diagnosis is imaging. Ultrasound is used as the initial screening imaging modality but may show normal findings in the early invasive phase. It may reveal muscle enlargement, or hypoechoic areas consistent with abscesses. Computed tomography may show low-density areas with a surrounding rim of contrast enhancement characteristic of pyomyositis. MRI gives greater anatomic detail and is the de-facto imaging “gold standard” for the diagnosis of pyomyositis.
Treatment consists of urgent drainage of any identified abscess, as well as empirical antibiotic coverage with vancomycin in an immunocompetent host or broad-spectrum antibiotic coverage targeting both gram-positive and gram-negative bacteria in immunosuppressed or HIV-infected patients.
Gas gangrene
Gas gangrene is a rapidly progressive life-threatening infection of skeletal muscle caused by clostridia (principally Clostridium perfringens). It is due to wound contamination in the setting of severe tissue trauma, inadequate surgical débridement, immunosuppression, and impaired blood supply. Rarely, nontraumatic gas gangrene caused by Clostridium septicum may occur in patients with occult gastrointestinal malignancies and lead to transient bacteremia.
Symptoms and signs may evolve over a period of 2 to 3 days, but they may also be fulminant and achieve a peak within 6 hours. Pain out of proportion to the findings on physical examination is followed by the rapid development of septic shock. There is a foul-smelling, serosanguineous, dirty-appearing discharge with occasional gas bubbles. Crepitus on palpation is the classic hallmark described in the literature, but it may be masked by the massive surrounding tissue edema, and its absence does not rule out gas gangrene. Evidence of air in soft tissues on imaging or high clinical suspicion for gas gangrene is an absolute indication for urgent surgical intervention and débridement of nonviable tissues. Antibiotic therapy with penicillin G and clindamycin is an important adjunct to surgery.
Psoas abscess
A psoas abscess is defined as a purulent infectious collection within the psoas muscle. The psoas muscle is supplied by venous blood from the lumbar spine and has lymphatics from nearby intraabdominal organs overlying it. Secondary psoas abscesses develop as a result of spread of infection from contiguous structures, such as concurrent vertebral infections. Other routes may be from an intraabdominal source, most commonly gastrointestinal, including Crohn disease, cancer, appendicitis, or diverticulitis. Less commonly, psoas abscesses may develop in association with genitourinary infections, such as a perinephric abscess, vaginal delivery, cesarean surgery, abortion, or an infected retroperitoneal hematoma.
Myalgias without eosinophilia
Influenza
Influenza A and B viral myositis occurs most often in children. It is characterized by a sudden onset of calf pain and tenderness, which often results in difficulty walking. The myositis differs from the common initial complaint of myalgia by its later onset, more focal location, and more severe intensity.55 CK levels are elevated. If EMG is performed, myopathic changes can be noted. Histopathologic examination of muscle reveals degeneration and necrosis and overall little inflammatory infiltrates. The myositis resolves within a few days with symptomatic treatment.
Human immunodeficiency virus
A slowly progressive myopathy that resembles polymyositis may develop in patients infected with HIV. Patients have myalgia, proximal symmetric weakness, and elevation of serum CK levels. EMG may show an irritable myopathy indistinguishable from polymyositis.56 Muscle biopsy specimens show endomysial, perimysial, or perivascular mononuclear cell infiltrates with muscle fiber necrosis and phagocytosis. Differentiating HIV myopathy from nucleoside reverse transcriptase inhibitor (NRTI) myopathy is a difficult clinical challenge that often requires a trial without NRTIs.
Myalgias with eosinophilia
Among the parasites that cause myositis, Toxoplasma is the most common in the United States. Toxoplasmosis can cause an inflammatory myopathy in immunocompromised hosts, often accompanied by fever, encephalitis, and other organ involvement. If isolated, treatment is not generally warranted because the infection is usually self-limited. In severe cases, including progressive myositis, sulfadiazine and pyrimethamine are indicated.
Trichinosis occurs after the ingestion of undercooked pork or wild animal meat. The extraocular muscles are generally involved first, followed by the masseters and muscles of the diaphragm, neck, larynx, and limbs. Rarely, trichinosis can produce a clinical syndrome resembling polymyositis. Eosinophilia is an important diagnostic clue; serologic testing confirms the diagnosis.42
Acknowledgment
Box 151.1 and Figures 151.1 and 151.2 are from the previous edition of this chapter, authored by Alan N. Baer, MD.