Chapter 1
Properties and Overview of Immune Responses
The term immunity is derived from the Latin word immunitas, which referred to the protection from legal prosecution offered to Roman senators during their tenures in office. Historically, immunity meant protection from disease and, more specifically, infectious disease. The cells and molecules responsible for immunity constitute the immune system, and their collective and coordinated response to the introduction of foreign substances is called the immune response.
The physiologic function of the immune system is defense against infectious microbes. However, even noninfectious foreign substances can elicit immune responses. Furthermore, mechanisms that normally protect individuals from infection and eliminate foreign substances also are capable of causing tissue injury and disease in some situations. Therefore, a more inclusive definition of the immune response is a reaction to components of microbes as well as to macromolecules, such as proteins and polysaccharides, and small chemicals that are recognized as foreign, regardless of the physiologic or pathologic consequence of such a reaction. Under some situations, even self molecules can elicit immune responses (so-called autoimmune responses). Immunology is the study of immune responses in this broader sense and of the cellular and molecular events that occur after an organism encounters microbes and other foreign macromolecules.
Historians often credit Thucydides, in the fifth century BC in Athens, as having first mentioned immunity to an infection that he called plague (but that was probably not the bubonic plague we recognize today). The concept of protective immunity may have existed long before, as suggested by the ancient Chinese custom of making children resistant to smallpox by having them inhale powders made from the skin lesions of patients recovering from the disease. Immunology, in its modern form, is an experimental science in which explanations of immunologic phenomena are based on experimental observations and the conclusions drawn from them. The evolution of immunology as an experimental discipline has depended on our ability to manipulate the function of the immune system under controlled conditions. Historically, the first clear example of this manipulation, and one that remains among the most dramatic ever recorded, was Edward Jenner’s successful vaccination against smallpox. Jenner, an English physician, noticed that milkmaids who had recovered from cowpox never contracted the more serious smallpox. On the basis of this observation, he injected the material from a cowpox pustule into the arm of an 8-year-old boy. When this boy was later intentionally inoculated with smallpox, the disease did not develop. Jenner’s landmark treatise on vaccination (Latin vaccinus, of or from cows) was published in 1798. It led to the widespread acceptance of this method for inducing immunity to infectious diseases, and vaccination remains the most effective method for preventing infections (Table 1-1). An eloquent testament to the importance of immunology was the announcement by the World Health Organization in 1980 that smallpox was the first disease that had been eradicated worldwide by a program of vaccination.
Since the 1960s, there has been a remarkable transformation in our understanding of the immune system and its functions. Advances in cell culture techniques (including monoclonal antibody production), immunochemistry, recombinant DNA methodology, x-ray crystallography, and creation of genetically altered animals (especially transgenic and knockout mice) have changed immunology from a largely descriptive science into one in which diverse immune phenomena can be explained in structural and biochemical terms. In this chapter, we outline the general features of immune responses and introduce the concepts that form the cornerstones of modern immunology and that recur throughout this book.
TABLE 1-1
Effectiveness of Vaccines for Some Common Infectious Diseases
| Disease | Maximum Number of Cases (Year) | Number of Cases in 2009 | Percentage Change |
| Diphtheria | 206,939 (1921) | 0 | −99.99 |
| Measles | 894,134 (1941) | 61 | −99.99 |
| Mumps | 152,209 (1968) | 982 | −99.35 |
| Pertussis | 265,269 (1934) | 13,506 | −94.72 |
| Polio (paralytic) | 21,269 (1952) | 0 | −100.0 |
| Rubella | 57,686 (1969) | 4 | −99.99 |
| Tetanus | 1,560 (1923) | 14 | −99.10 |
| Haemophilus influenzae type B | ∼20,000 (1984) | 25 | −99.88 |
| Hepatitis B | 26,611 (1985) | 3,020 | −87.66 |

This table illustrates the striking decrease in the incidence of selected infectious diseases in the United States for which effective vaccines have been developed.
Data from Orenstein WA, Hinman AR, Bart KJ, Hadler SC: Immunization. In Mandell GL, Bennett JE, Dolin R (eds.): Principles and practices of infectious diseases, 4th ed. New York, 1995, Churchill Livingstone; and Morbidity and Mortality Weekly Report 58:1458–1469, 2010.
Innate and Adaptive Immunity
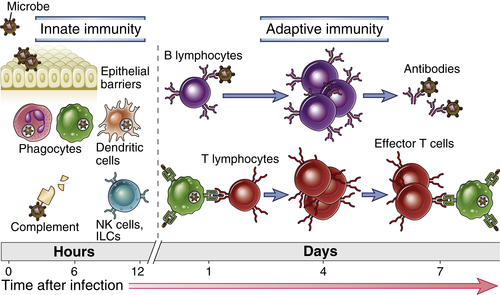
Defense against microbes is mediated by the early reactions of innate immunity and the later responses of adaptive immunity (Fig. 1-1 and Table 1-2). Innate immunity (also called natural or native immunity) provides the early line of defense against microbes. It consists of cellular and biochemical defense mechanisms that are in place even before infection and are poised to respond rapidly to infections. These mechanisms react to products of microbes and injured cells, and they respond in essentially the same way to repeated exposures. The mechanisms of innate immunity are specific for structures that are common to groups of related microbes and may not distinguish fine differences between microbes. The principal components of innate immunity are (1) physical and chemical barriers, such as epithelia and antimicrobial chemicals produced at epithelial surfaces; (2) phagocytic cells (neutrophils, macrophages), dendritic cells, and natural killer (NK) cells and other innate lymphoid cells; and (3) blood proteins, including members of the complement system and other mediators of inflammation.
In contrast to innate immunity, there are other immune responses that are stimulated by exposure to infectious agents and increase in magnitude and defensive capabilities with each successive exposure to a particular microbe. Because this form of immunity develops as a response to infection and adapts to the infection, it is called adaptive immunity (also called specific or acquired immunity). The adaptive immune system recognizes and reacts to a large number of microbial and nonmicrobial substances. The defining characteristics of adaptive immunity are the ability to distinguish different substances, called specificity, and the ability to respond more vigorously to repeated exposures to the same microbe, known as memory. The unique components of adaptive immunity are cells called lymphocytes and their secreted products, such as antibodies. Foreign substances that induce specific immune responses or are recognized by lymphocytes or antibodies are called antigens.
Cytokines are a large group of secreted proteins with diverse structures and functions, which regulate and coordinate many activities of the cells of innate and adaptive immunity. All cells of the immune system secrete at least some cytokines and express specific signaling receptors for several cytokines. The nomenclature for cytokines is inconsistent, with some named Interleukin followed by a number, and others named for a biological activity first attributed to them, such as tumor necrosis factor (TNF) or interferon. Among the many functions of cytokines we will discuss throughout this book are growth and differentiation of all immune cells, activation of effector functions of lymphocytes and phagocytes, and directed movement of immune cells from blood into tissues and within tissues. The large subset of structurally related cytokines that regulate cell migration and movement are called chemokines. Some of the most effective drugs developed recently to treat immunologic diseases target cytokines, which reflects the importance of these proteins in immune responses.
Mechanisms for defending the host against microbes are present in all multicellular organisms. The phylogenetically oldest mechanisms of host defense are those of innate immunity, which are present even in plants and insects. About 500 million years ago, jawless fish, such as lampreys and hagfish, developed an immune system containing lymphocyte-like cells that may function like lymphocytes in more advanced species and even respond to immunization. The antigen receptors on these cells are variable leucine-rich receptors that are capable of recognizing many antigens but are distinct from the antibodies and T cell receptors that appeared later in evolution. The more specialized defense mechanisms that constitute adaptive immunity are found in vertebrates only. Most of the components of the adaptive immune system, including lymphocytes with highly diverse antigen receptors, antibodies, and specialized lymphoid tissues, evolved coordinately within a short time in jawed vertebrates (e.g., sharks) about 360 million years ago.
Innate and adaptive immune responses are components of an integrated system of host defense in which numerous cells and molecules function cooperatively. The mechanisms of innate immunity provide effective initial defense against infections. However, many pathogenic microbes have evolved to resist innate immunity, and their elimination requires the more powerful mechanisms of adaptive immunity. There are numerous connections between the innate and adaptive immune systems. The innate immune response to microbes stimulates adaptive immune responses and influences the nature of the adaptive responses. Conversely, adaptive immune responses often work by enhancing the protective mechanisms of innate immunity, making them more capable of effectively combating pathogenic microbes.
Types of Adaptive Immune Responses
There are two types of adaptive immune responses, called humoral immunity and cell-mediated immunity, that are mediated by different components of the immune system and function to eliminate different types of microbes (Fig. 1-2). Humoral immunity is mediated by molecules in the blood and mucosal secretions, called antibodies, which are produced by cells called B lymphocytes (also called B cells). Antibodies recognize microbial antigens, neutralize the infectivity of the microbes, and target microbes for elimination by various effector mechanisms. Humoral immunity is the principal defense mechanism against extracellular microbes and their toxins because secreted antibodies can bind to these microbes and toxins and assist in their elimination. Antibodies themselves are specialized and may activate different mechanisms to combat microbes (effector mechanisms). For example, different types of antibodies promote the ingestion of microbes by host cells (phagocytosis), bind to and trigger the release of inflammatory mediators from cells, and are actively transported into the lumens of mucosal organs and through the placenta to provide defense against ingested and inhaled microbes and against infections of the newborn, respectively.
Cell-mediated immunity, also called cellular immunity, is mediated by T lymphocytes (also called T cells). Intracellular microbes, such as viruses and some bacteria, survive and proliferate inside phagocytes and other host cells, where they are inaccessible to circulating antibodies. Defense against such infections is a function of cell-mediated immunity, which promotes the destruction of microbes residing in phagocytes or the killing of infected cells to eliminate reservoirs of infection. Some T lymphocytes also contribute to eradication of extracellular microbes by recruiting leukocytes that destroy these pathogens and by helping B cells make effective antibodies.
TABLE 1-2
Features of Innate and Adaptive Immunity
| Innate | Adaptive | |
| Characteristics | ||
| Specificity | For molecules shared by groups of related microbes and molecules produced by damaged host cells | For microbial and nonmicrobial antigens |
| Diversity | Limited; germline encoded | Very large; receptors are produced by somatic recombination of gene segments |
| Memory | None | Yes |
| Nonreactivity to self | Yes | Yes |
| Components | ||
| Cellular and chemical barriers | Skin, mucosal epithelia; antimicrobial molecules | Lymphocytes in epithelia; antibodies secreted at epithelial surfaces |
| Blood proteins | Complement, others | Antibodies |
| Cells | Phagocytes (macrophages, neutrophils), natural killer cells, innate lymphoid cells | Lymphocytes |

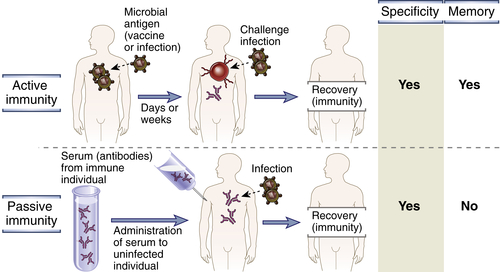
Protective immunity against a microbe is usually induced by the host’s response to the microbe (Fig. 1-3). The form of immunity that is induced by exposure to a foreign antigen is called active immunity because the immunized individual plays an active role in responding to the antigen. Individuals and lymphocytes that have not encountered a particular antigen are said to be naive, implying that they are immunologically inexperienced. Individuals who have responded to a microbial antigen and are protected from subsequent exposures to that microbe are said to be immune.

Immunity can also be conferred on an individual by transferring serum or lymphocytes from a specifically immunized individual in experimental situations, a process known as adoptive transfer (see Fig. 1-3). The recipient of such a transfer becomes immune to the particular antigen without ever having been exposed to or having responded to that antigen. Therefore, this form of immunity is called passive immunity. Passive immunization is a useful method for conferring resistance rapidly, without having to wait for an active immune response to develop. A physiologically important example of passive immunity is the transfer of maternal antibodies through the placenta to the fetus, which enables newborns to combat infections before they develop the ability to produce antibodies themselves. Passive immunization against toxins by the administration of antibodies from immunized animals is a lifesaving treatment for potentially lethal infections, such as rabies, and snake bites. The technique of adoptive transfer has also made it possible to define the various cells and molecules that are responsible for mediating specific immunity. In fact, humoral immunity was originally defined as the type of immunity that could be transferred to unimmunized, or naive, individuals with antibody-containing cell-free portions of the blood (i.e., plasma or serum) obtained from previously immunized individuals. Similarly, cell-mediated immunity was defined as the type of immunity that could be transferred to naive animals with cells (T lymphocytes) from immunized animals, but not with plasma or serum.
The first experimental demonstration of humoral immunity was provided by Emil von Behring and Shibasaburo Kitasato in 1890. They showed that if serum from animals that had been immunized with an attenuated form of diphtheria toxin was transferred to naive animals, the recipients became specifically resistant to diphtheria infection. The active components of the serum were called antitoxins because they neutralized the pathologic effects of the diphtheria toxin. This result led to the treatment of otherwise lethal diphtheria infection by the administration of antitoxin, an achievement that was recognized by the award of the first Nobel Prize in Physiology or Medicine to von Behring. In the 1890s, Paul Ehrlich postulated that immune cells use receptors, which he called side chains, to recognize microbial toxins and, subsequently, secrete these receptors to combat microbes. He also coined the term antibodies (antikörper in German) for the serum proteins that bound toxins, and substances that generated antibodies were called antigens. The modern definition of antigens includes substances that bind to specific lymphocyte receptors, whether or not they stimulate immune responses. According to strict definitions, substances that stimulate immune responses are called immunogens, but the term antigen is often used interchangeably with immunogen. The properties of antibodies and antigens are described in Chapter 5. Ehrlich’s concepts are a remarkably prescient model for the function of B cells in humoral immunity. This early emphasis on antibodies led to the general acceptance of the humoral theory of immunity, according to which host defense against infections is mediated by substances present in body fluids (once called humors).
Elie Metchnikoff initially championed the cellular theory of immunity, which stated that host cells are the principal mediators of immunity. His demonstration of phagocytes surrounding a thorn stuck into a translucent starfish larva, published in 1883, was perhaps the first experimental evidence that cells respond to foreign invaders. Ehrlich and Metchnikoff shared the Nobel Prize in 1908, in recognition of their contributions to establishing these fundamental principles of immunity. Sir Almroth Wright’s observation in the early 1900s that factors in immune serum enhanced the phagocytosis of bacteria by coating the bacteria, a process known as opsonization, lent support to the belief that antibodies prepare microbes for ingestion by phagocytes. These early cellularists were unable to prove that specific immunity to microbes could be mediated by cells. The cellular theory of immunity became firmly established in the 1950s, when it was shown that resistance to an intracellular bacterium, Listeria monocytogenes, could be transferred to animals with cells but not with serum. We now know that the specificity of cell-mediated immunity is due to lymphocytes, which often function in concert with other cells, such as phagocytes, to eliminate microbes.
In the clinical setting, immunity to a previously encountered microbe is measured indirectly, either by assaying for the presence of products of immune responses (such as serum antibodies specific for microbial antigens) or by administering substances purified from the microbe and measuring reactions to these substances. A reaction to an antigen is detectable only in individuals who have previously encountered the antigen; these individuals are said to be sensitized to the antigen, and the reaction is an indication of sensitivity. Such a reaction to a microbial antigen implies that the sensitized individual is capable of mounting a protective immune response to the microbe.
Cardinal Features of Adaptive Immune Responses
All humoral and cell-mediated immune responses to foreign antigens have a number of fundamental properties that reflect the properties of the lymphocytes that mediate these responses (Table 1-3).
• Specificity and diversity.Immune responses are specific for distinct antigens and, in fact, for different portions of a single complex protein, polysaccharide, or other macromolecule (Fig. 1-4). The parts of such antigens that are specifically recognized by individual lymphocytes are called determinants or epitopes. This fine specificity exists because individual lymphocytes express membrane receptors that can distinguish subtle differences in structure between distinct epitopes. Clones of lymphocytes with different specificities are present in unimmunized individuals and are able to recognize and respond to foreign antigens. This concept is the basic tenet of the clonal selection hypothesis, which is discussed in more detail later in this chapter.
TABLE 1-3
Cardinal Features of Adaptive Immune Responses
| Feature | Functional Significance |
| Specificity | Ensures that the immune response to a microbe (or nonmicrobial antigen) is targeted to that microbe (or antigen) |
| Diversity | Enables the immune system to respond to a large variety of antigens |
| Memory | Increases the ability to combat repeat infections by the same microbe |
| Clonal expansion | Increases the number of antigen-specific lymphocytes to keep pace with microbes |
| Specialization | Generates responses that are optimal for defense against different types of microbes |
| Contraction and homeostasis | Allows the immune system to recover from one response so that it can effectively respond to newly encountered antigens |
| Nonreactivity to self | Prevents injury to the host during responses to foreign antigens |
The total number of antigenic specificities of the lymphocytes in an individual, called the lymphocyte repertoire, is extremely large. It is estimated that the immune system of an individual can discriminate 107 to 109 distinct antigenic determinants. This ability of the lymphocyte repertoire to recognize a very large number of antigens is the result of variability in the structures of the antigen-binding sites of lymphocyte receptors for antigens, called diversity. In other words, there are many different clones of lymphocytes that differ in the structures of their antigen receptors and therefore in their specificity for antigens, contributing to a total repertoire that is extremely diverse. The expression of different antigen receptors in different clones of T and B cells is the reason that these receptors are said to be clonally distributed. The molecular mechanisms that generate such diverse antigen receptors are discussed in Chapter 8.
• Memory.Exposure of the immune system to a foreign antigen enhances its ability to respond again to that antigen. Responses to second and subsequent exposures to the same antigen, called secondary immune responses, are usually more rapid, larger, and often qualitatively different from the first, or primary, immune response to that antigen (see Fig. 1-4). Immunologic memory occurs because each exposure to an antigen generates long-lived memory cells specific for the antigen, which are more numerous than the naive lymphocytes specific for the antigen that exist before antigen exposure. In addition, memory cells have special characteristics that make them more efficient at responding to and eliminating the antigen than are naive lymphocytes that have not previously been exposed to the antigen. For instance, memory B lymphocytes produce antibodies that bind antigens with higher affinities than do antibodies produced in primary immune responses, and memory T cells react much more rapidly and vigorously to antigen challenge than do naive T cells.
• Clonal expansion.Lymphocytes specific for an antigen undergo considerable proliferation after exposure to that antigen. The term clonal expansion refers to an increase in the number of cells that express identical receptors for the antigen and thus belong to a clone. This increase in antigen-specific cells enables the adaptive immune response to keep pace with rapidly dividing infectious pathogens.

• Specialization.As we have already noted, the immune system responds in distinct and special ways to different microbes, maximizing the effectiveness of antimicrobial defense mechanisms. Thus, humoral immunity and cell-mediated immunity are elicited by different classes of microbes or by the same microbe at different stages of infection (extracellular and intracellular), and each type of immune response protects the host against that class of microbe. Even within humoral or cell-mediated immune responses, the nature of the antibodies or T lymphocytes that are generated may vary from one class of microbe to another. We will return to the mechanisms and functional significance of such specialization in later chapters.
• Contraction and homeostasis.All normal immune responses wane with time after antigen stimulation, thus returning the immune system to its resting basal state, a state called homeostasis (see Fig. 1-4). This contraction of immune responses occurs largely because responses that are triggered by antigens function to eliminate the antigens, thus eliminating an essential stimulus for lymphocyte survival and activation. Lymphocytes (other than memory cells) that are deprived of these stimuli die by apoptosis.
• Nonreactivity to self.One of the most remarkable properties of every normal individual’s immune system is its ability to recognize, respond to, and eliminate many foreign (non-self) antigens while not reacting harmfully to that individual’s own (self) antigenic substances. Immunologic unresponsiveness is also called tolerance. Tolerance to self antigens, or self-tolerance, is maintained by several mechanisms. These include eliminating lymphocytes that express receptors specific for some self antigens, inactivating self-reactive lymphocytes, or suppressing these cells by the actions of other (regulatory) cells. Abnormalities in the induction or maintenance of self-tolerance lead to immune responses against self (autologous) antigens, which may result in disorders called autoimmune diseases. The mechanisms of self-tolerance and its failure are discussed in Chapter 15.
These features of adaptive immunity are necessary if the immune system is to perform its normal function of host defense (see Table 1-3). Specificity and memory enable the immune system to mount heightened responses to persistent or recurring exposure to the same antigen and thus to combat infections that are prolonged or occur repeatedly. Diversity is essential if the immune system is to defend individuals against the many potential pathogens in the environment. Specialization enables the host to “custom design” responses to best combat different types of microbes. Contraction of the response allows the system to return to a state of rest after it eliminates each foreign antigen and to be prepared to respond to other antigens. Self-tolerance is vital for preventing harmful reactions against one’s own cells and tissues while maintaining a diverse repertoire of lymphocytes specific for foreign antigens.
Immune responses are regulated by a system of positive feedback loops that amplify the reaction and by control mechanisms that prevent inappropriate or pathologic reactions. When lymphocytes are activated, they trigger mechanisms that further increase the magnitude of the response. This positive feedback is important to enable the small number of lymphocytes that are specific for any microbe to generate the response needed to eradicate that infection. Many control mechanisms become active in immune responses to prevent excessive activation of lymphocytes, which may cause collateral damage to normal tissues, and to avoid responses against self antigens. In fact, a balance between activating and inhibitory signals is characteristic of all immune responses. We will mention specific examples of these fundamental features of the immune system throughout this book.
Cellular Components of the Adaptive Immune System
The principal cells of the adaptive immune system are lymphocytes, antigen-presenting cells, and effector cells. Lymphocytes are the cells that specifically recognize and respond to foreign antigens and are therefore the mediators of humoral and cellular immunity. There are distinct subpopulations of lymphocytes that differ in how they recognize antigens and in their functions (Fig. 1-5). B lymphocytes are the only cells capable of producing antibodies. They recognize extracellular soluble and cell surface antigens, and they differentiate into antibody-secreting plasma cells, thus functioning as the mediators of humoral immunity. T lymphocytes, the cells of cell-mediated immunity, recognize the antigens of intracellular microbes and the T cells either help phagocytes to destroy these microbes or they kill the infected cells. T cells do not produce antibody molecules. Their antigen receptors are membrane molecules distinct from but structurally related to antibodies (see Chapter 7). T lymphocytes have a restricted specificity for antigens; they recognize peptides derived from foreign proteins that are bound to host proteins called major histocompatibility complex (MHC) molecules, which are expressed on the surfaces of other cells. As a result, these T cells recognize and respond to cell surface–associated but not soluble antigens (see Chapter 6).
T lymphocytes consist of functionally distinct populations, the best defined of which are helper T cells and cytotoxic (or cytolytic) T lymphocytes (CTLs). In response to antigenic stimulation, helper T cells secrete cytokines, which are responsible for many of the cellular responses of innate and adaptive immunity and thus function as the “messenger molecules” of the immune system. The cytokines secreted by helper T lymphocytes stimulate the proliferation and differentiation of the T cells themselves and activate other cells, including B cells, macrophages, and other leukocytes. CTLs kill cells that produce foreign antigens, such as cells infected by viruses and other intracellular microbes. Some T lymphocytes, which are called regulatory T cells, function mainly to inhibit immune responses. A small population of T lymphocytes that express some cell surface proteins found on NK cells are called NKT cells; their specificities and role in host defense are not well understood. We will return to a more detailed discussion of the properties of lymphocytes in Chapter 2 and in later chapters. Different classes of lymphocytes can be distinguished by the expression of surface proteins that are named CD molecules and numbered (see Chapter 2).
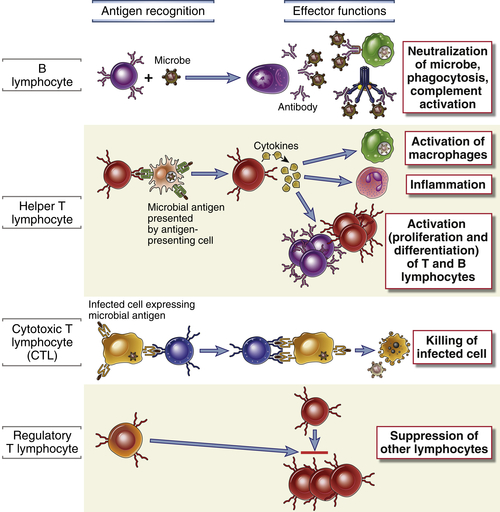
The initiation and development of adaptive immune responses require that antigens be captured and displayed to specific lymphocytes. The cells that serve this role are called antigen-presenting cells (APCs). The most specialized APCs are dendritic cells, which capture microbial antigens that enter from the external environment, transport these antigens to lymphoid organs, and present the antigens to naive T lymphocytes to initiate immune responses. Other cell types function as APCs at different stages of cell-mediated and humoral immune responses. We will describe the functions of APCs in Chapter 6.
The activation of lymphocytes by antigen leads to the generation of numerous mechanisms that function to eliminate the antigen. Antigen elimination often requires the participation of cells that are called effector cells because they mediate the final effect of the immune response, which is to get rid of the microbes. Activated T lymphocytes, mononuclear phagocytes, and other leukocytes function as effector cells in different immune responses.
Lymphocytes and APCs are concentrated in anatomically discrete lymphoid organs, where they interact with one another to initiate immune responses. Lymphocytes are also present in the blood; from the blood, they can recirculate through lymphoid tissues and home to peripheral tissue sites of antigen exposure to eliminate the antigen (see Chapter 3).
The cells of the immune system interact with one another and with other host cells during the initiation and effector stages of innate and adaptive immune responses. Many of these interactions are mediated by cytokines. We will describe the functions of individual cytokines when we discuss immune responses in which these proteins play important roles.
Overview of Immune Responses to Microbes
Now that we have introduced the major components of the immune system and their properties, it is useful to summarize the principles of immune responses to different types of microbes. Such a summary will be a foundation for the topics that are discussed throughout this book. The immune system has to combat many diverse microbes. As we will see shortly, immune responses to all infectious pathogens share some common features, and responses to different classes of these microbes may also have unique features. How these adaptive immune reactions are initiated, orchestrated, and controlled are the fundamental questions of immunology. We start with a discussion of the innate immune response.
The Early Innate Immune Response to Microbes
The innate immune system blocks the entry of microbes and eliminates or limits the growth of many microbes that are able to colonize tissues. The main sites of interaction between individuals and their environment—the skin, lungs, and gastrointestinal and respiratory tracts—are lined by continuous epithelia, which serve as barriers to prevent the entry of microbes from the external environment. If microbes successfully breach the epithelial barriers, they encounter other cells of innate immunity. The cellular innate immune response to microbes consists of two main types of reactions—inflammation and antiviral defense. Inflammation is the process of recruitment of leukocytes and plasma proteins from the blood, their accumulation in tissues, and their activation to destroy the microbes. Many of these reactions involve cytokines that are produced by dendritic cells, macrophages, and other types of cells during innate immune reactions. The major leukocytes that are recruited in inflammation are the phagocytes, neutrophils (which have short life spans in tissues) and monocytes (which develop into tissue macrophages). Phagocytes ingest microbes and dead cells and destroy these in intracellular vesicles. Antiviral defense consists of a cytokine-mediated reaction in which cells acquire resistance to viral infection and killing of virus-infected cells by specialized cells of the innate immune system, natural killer (NK) cells.
Microbes that are able to withstand these defense reactions in the tissues may enter the blood, where they are recognized by the circulating proteins of innate immunity. Among the most important plasma proteins of innate immunity are components of the complement system. When complement proteins are activated by microbial surfaces, proteolytic cleavage products are generated that mediate inflammatory responses, coat (opsonize) the microbes for enhanced phagocytosis, and directly lyse microbes. Other plasma proteins enter sites of infection during inflammatory reactions and help combat microbes in extravascular tissues.
The reactions of innate immunity are effective at controlling and even eradicating infections. However, as mentioned earlier, many pathogenic microbes have evolved to resist innate immunity. Defense against these pathogens requires the more powerful and specialized mechanisms of adaptive immunity.
The Adaptive Immune Response
The adaptive immune system uses three main strategies to combat most microbes.
• Antibodies.Secreted antibodies bind to extracellular microbes, block their ability to infect host cells, and promote their ingestion and subsequent destruction by phagocytes.
• Phagocytosis.Phagocytes ingest microbes and kill them, and antibodies and helper T cells enhance the microbicidal abilities of the phagocytes.
• Cell killing.Cytotoxic T lymphocytes (CTLs) destroy cells infected by microbes that are inaccessible to antibodies and phagocytic destruction.
The goal of the adaptive response is to activate one or more of these defense mechanisms against diverse microbes that may be in different anatomic locations, such as the intestines or airways, the circulation, or inside cells.
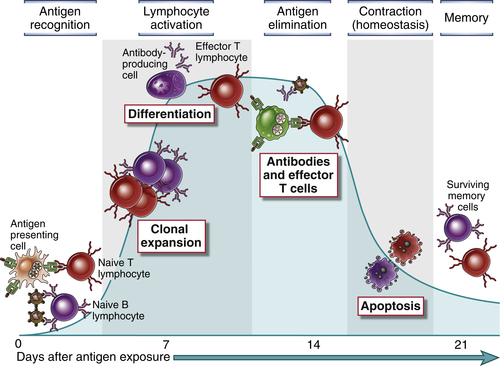
All adaptive immune responses develop in sequential steps, each of which corresponds to particular reactions of lymphocytes (Fig. 1-6). We start this overview of adaptive immunity with the first step, which is the recognition of antigens.
The Capture and Display of Microbial Antigens
Because the number of naive lymphocytes specific for any antigen is very small (on the order of 1 in 105 or 106 lymphocytes) and the quantity of the available antigen may also be small, special mechanisms are needed to capture microbes, to concentrate their antigens in the correct location, and to deliver the antigens to specific lymphocytes. Dendritic cells located in epithelia and connective tissues capture microbes, digest their proteins into fragments, and express on their surface microbial peptides bound to MHC molecules, which are the specialized peptide display molecules of the adaptive immune system. Dendritic cells carry their antigenic cargo to draining lymph nodes through which naive T lymphocytes continuously recirculate. Thus, the chance of a T cell with receptors for a particular antigen finding that antigen is greatly increased by concentrating many antigens and T cells in the same anatomic location. Dendritic cells also display microbial peptides in the spleen.
Intact microbes or microbial antigens that enter lymph nodes and spleen are recognized in unprocessed (native) form by specific B lymphocytes. Antigens may also be displayed to B lymphocytes by certain APCs in lymphoid organs.
Antigen Recognition by Lymphocytes
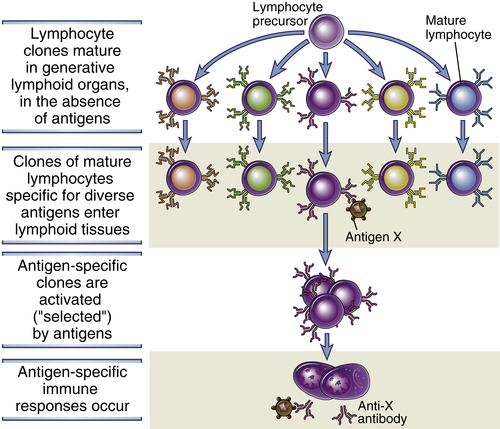
Lymphocytes specific for a large number of antigens exist before exposure to the antigen, and when an antigen enters a secondary lymphoid organ, it binds to (selects) the antigen-specific cells and activates them (Fig. 1-7). This fundamental concept is called the clonal selection hypothesis. It was suggested by Niels Jerne in 1955, and most clearly enunciated by Macfarlane Burnet in 1957, as a hypothesis to explain how the immune system could respond to a large number and variety of antigens. According to this hypothesis, antigen-specific clones of lymphocytes develop before and independent of exposure to antigen. A clone refers to a lymphocyte of one specificity and its progeny. A characteristic of the immune system is that a very large number of clones is generated during the maturation of lymphocytes, thus maximizing the potential for recognizing diverse microbes.
The activation of naive T lymphocytes requires recognition of peptide-MHC complexes presented on dendritic cells. Because T cell receptors are specific for MHC-associated peptides, these lymphocytes can interact only with cell-associated antigens (because MHC molecules are cell surface proteins) and not with free antigen. This feature is necessary because all the functions of T lymphocytes are dependent on their physical interactions with other cells. To respond, the T cells need to recognize not only antigens but also other molecules, called costimulators, which are induced on the APCs by microbes. Antigen recognition provides specificity to the immune response, and the need for costimulation ensures that T cells respond to microbes (the inducers of costimulatory molecules) and not to harmless substances.
B lymphocytes use their antigen receptors (membrane-bound antibody molecules) to recognize antigens of many different chemical types.
Engagement of antigen receptors and other signals trigger lymphocyte proliferation and differentiation. The reactions and functions of T and B lymphocytes differ in important ways and are best considered separately.
Cell-Mediated Immunity: Activation of T Lymphocytes and Elimination of Intracellular Microbes
Activated CD4+ helper T lymphocytes proliferate and differentiate into effector cells whose functions are mediated largely by secreted cytokines. When naive CD4+ T cells are activated by antigen, they secrete the cytokine interleukin-2 (IL-2), which is a growth factor that stimulates the proliferation (clonal expansion) of the antigen-specific T cells. Some of the progeny of these activated lymphocytes differentiate into effector cells that can secrete different sets of cytokines and thus perform different functions. Many of the effector cells leave the lymphoid organs where they were generated and migrate to sites of infection and accompanying inflammation. When these differentiated effector T cells again encounter cell-associated microbes, they are activated to perform the functions that are responsible for elimination of the microbes. Some CD4+ helper T cells secrete cytokines that recruit leukocytes and stimulate production of microbicidal substances in phagocytes. Thus, these T cells help phagocytes to kill the infectious pathogens. Other CD4+ helper T cells secrete cytokines that help B cells produce a type of antibody called immunoglobulin E (IgE) and activate leukocytes called eosinophils, which are able to kill parasites that may be too large to be phagocytosed. As we discuss later, some CD4+ helper T cells stay in the lymphoid organs and stimulate B cell responses.
Activated CD8+ lymphocytes proliferate and differentiate into CTLs that kill cells harboring microbes in the cytoplasm. These microbes may be viruses that infect many cell types or bacteria that are ingested by macrophages but escape from phagocytic vesicles into the cytoplasm (where they are inaccessible to the killing machinery of phagocytes, which is largely confined to vesicles). By destroying the infected cells, CTLs eliminate the reservoirs of infection.
Humoral Immunity: Activation of B Lymphocytes and Elimination of Extracellular Microbes
On activation by antigen, B lymphocytes proliferate and differentiate into cells that secrete different classes of antibodies with distinct functions. The response of B cells to protein antigens requires activating signals (help) from CD4+ T cells (which is the historical reason for calling these T cells helper cells). B cells can respond to many nonprotein antigens without the participation of helper T cells.
Some of the progeny of the expanded B cell clones differentiate into antibody-secreting plasma cells. Each plasma cell secretes antibodies that have the same antigen-binding site as the cell surface antibodies (B cell receptors) that first recognized the antigen. Polysaccharides and lipids stimulate secretion mainly of the antibody class called IgM. Protein antigens induce the production of antibodies of functionally different classes (IgG, IgA, IgE) from a single clone of B cells. The production of these classes of antibodies with different functions is called class switching. The process requires the action of helper T cells. It provides plasticity in the antibody response, enabling it to serve many functions. Helper T cells also stimulate the production of antibodies with increased affinity for the antigen. This process, called affinity maturation, improves the quality of the humoral immune response.
The humoral immune response combats microbes in many ways. Antibodies bind to microbes and prevent them from infecting cells, thus neutralizing the microbes. In fact, antibodies are the only mechanisms of adaptive immunity that prevent an infection from becoming established; this is why eliciting the production of potent antibodies is a key goal of vaccination. IgG antibodies coat microbes and target them for phagocytosis because phagocytes (neutrophils and macrophages) express receptors for parts of IgG molecules. IgG and IgM activate the complement system, and complement products promote phagocytosis and destruction of microbes. Some antibodies serve special roles at particular anatomic sites. IgA is secreted from mucosal epithelia and neutralizes microbes in the lumens of mucosal tissues, such as the respiratory and gastrointestinal tracts. Maternal IgG is actively transported across the placenta and protects the newborn until the baby’s immune system becomes mature. Most antibodies have half-lives of a few days, but most IgG antibodies have half-lives of about 3 weeks. Some antibody-secreting plasma cells migrate to the bone marrow and live for years, continuing to produce low levels of antibodies. The antibodies that are secreted by these long-lived plasma cells provide immediate protection if the microbe returns to infect the individual. More effective protection is provided by memory cells that are activated by the microbe and rapidly differentiate to generate large numbers of plasma cells.
Immunologic Memory
An effective immune response eliminates the microbes that initiated the response. This is followed by a contraction phase, in which the expanded lymphocyte clones die and homeostasis is restored.
The initial activation of lymphocytes generates long-lived memory cells, which may survive for years after the infection. Memory cells are more effective in combating microbes than are naive lymphocytes because, as mentioned earlier, memory cells represent an expanded pool of antigen-specific lymphocytes (more numerous than naive cells specific for the antigen) and respond faster and more effectively against the antigen than do naive cells. This is why generating memory responses is another important goal of vaccination. We will discuss the properties of memory lymphocytes in later chapters.
In the remainder of the book, we describe in detail the recognition, activation, regulation, and effector phases of innate and adaptive immune responses. The principles introduced in this chapter recur throughout this book.
