Special collections and procedures
Objectives
After completing this chapter, you should be able to:
2. Define and explain the uses of:
a. Fasting specimens
b. Timed specimens
c. 2-hour postprandial specimens
3. Describe the procedure for performing the various tolerance tests.
4. Define diurnal variation, and list the blood constituents that may be affected by it.
5. Define therapeutic drug monitoring (TDM), describe the differences among a random level and peak and trough levels, and explain how TDM samples are collected.
6. Describe the reasons and procedures for collecting blood for culture.
7. Explain the steps in collecting blood from donors for transfusion.
8. Define and explain the uses of autologous donation and therapeutic phlebotomy.
9. Explain how samples to be tested for or suspected of containing cold agglutinins, cryofibrinogen, or cryoglobulin should be handled.
10. List samples that should be chilled until tested.
11. List samples that are light sensitive, and explain how they should be handled.
12. Describe the precautions to be taken when collecting legal or forensic specimens.
13. List samples that are time sensitive, and explain how they should be handled.
14. Explain how to prepare blood smears, describe features of unacceptable smears, and list the possible causes.
15. Explain how to prepare smears to be examined for malaria.
Key terms
aerobic bacteria
agglutination
anaerobic bacteria
bacteremia
basal state
blood culture (BC)
chain of custody (COC)
cold agglutinins
cryofibrinogen
cryoglobulin
differential count (diff)
diurnal variation
epinephrine tolerance test
fasting specimen
feathered edge
fever of unknown origin (FUO)
gestational diabetes
glucagon tolerance test
glucose tolerance test (GTT)
half-life
hemochromatosis
hyperglycemia
hypoglycemia
lactose tolerance test (LTT)
oral glucose tolerance test (OGTT)
peak level
polycythemia
sepsis
sodium polyanethole sulfonate (SPS)
therapeutic drug monitoring (TDM)
therapeutic phlebotomy
trough level
2-hour postprandial test
Although routine venipuncture is the most common procedure you will perform as a phlebotomist, special collecting or handling procedures are needed in many situations for samples that involve one or more special circumstances. Fasting specimens, timed specimens, blood cultures, and blood donor specimens all require collection procedures specific to the sample being collected. A variety of samples require special handling, which may involve keeping the sample warm, cool, or away from light, or providing immediate delivery or legal documentation. In this chapter, you will learn when and why these special procedures are needed and the details of how to perform them.
Abbreviations
Fasting specimens and the basal state
As detailed in Box 14-1, many factors influence the composition of blood. Diurnal variation refers to the normal daily fluctuations in body chemistry related to hormonal cycles, sleep–wake cycles, and other regular patterns of change.
To minimize the variations introduced by normal fluctuations in blood composition, reference ranges for blood tests are based on healthy patients in what is known as the basal state. The basal state is defined as the body’s state after 8 to 12 hours of fasting and abstention from strenuous exercise. Routine phlebotomy rounds are scheduled for the early morning, because most patients are in the basal state at that time.
Some test results are more affected than others when a patient has not been scrupulously fasting for 12 hours. Glucose and triglycerides are especially affected. For this reason, a fasting specimen may be requested that is drawn after a 12-hour complete fast. Caffeine and nicotine are also prohibited during the fasting period, as these are metabolic stimulants. If a fasting specimen is requested, the phlebotomist must ask the patient if he or she has had anything to eat or drink other than water, or has had any caffeine or nicotine, within the past 12 hours. It is better to ask the question in this form than to ask, “Have you been fasting for 12 hours?” because some patients may not consider an evening snack or morning juice to be a violation of their fast. If the patient has violated the fast, you can still draw the sample, but make a note on the requisition. You may also contact your supervisor to see whether the physician should be notified before the draw. Often, the physician will reschedule the lab work, particularly if a lipid profile is ordered.
Timed specimens
Timed specimens are taken to determine changes in the level of some substance of interest over time. Timed specimens are most often used to monitor:
• Medication levels (e.g., digoxin for heart disease or levodopa for Parkinson disease).
• Changes in the patient’s condition (e.g., a decrease in hemoglobin level).
• Normal diurnal variation in blood levels at different times of the day (e.g., cortisol or other hormones).
• Cardiac enzymes, used to diagnose or rule out myocardial infarction (heart attack); these are tested at admission and then twice more at 8-hour intervals.
2-hour postprandial test
The 2-hour postprandial test is used to test for diabetes mellitus. It compares the fasting glucose level with the level 2 hours after consuming glucose, either by eating a meal or by ingesting a measured amount of glucose (postprandial means “after a meal”). In patients with diabetes mellitus, the glucose level will be higher than normal, whereas the level in normal patients will have returned to the fasting level. After the phlebotomist has obtained a fasting specimen, the patient is instructed to eat a full meal and then return to the lab 2 hours after eating for collection of the second specimen.
Glucose tolerance test
The glucose tolerance test (GTT), also called the oral glucose tolerance test (OGTT), tests for both diabetes mellitus and other disorders of carbohydrate metabolism. Hyperglycemia, or abnormally elevated blood sugar, is most commonly caused by diabetes; hypoglycemia, or abnormally lowered blood sugar, may be due to one of several endocrine disorders or other metabolic disruptions. Hyperglycemia is detected with a 3-hour GTT, and hypoglycemia is detected with a 5-hour GTT. Longer testing periods are sometimes used as well to identify a variety of metabolic disorders. The GTT has fallen out of general use for the diagnosis of diabetes mellitus, and has been replaced by either a fasting glucose sample (at least 8 hours without caloric intake) or a random glucose sample.
Glucose tolerance testing is still widely used in pregnant women, to diagnose gestational diabetes. Gestational diabetes is insulin-resistant diabetes that develops in almost 20% of women late in pregnancy. The initial screen is a 1-hour GTT, performed without fasting. The patient drinks an intensely sweetened liquid, and a sample is taken 1 hour later. Elevated glucose in the sample indicates potential gestational diabetes. That finding usually prompts the physician to order a fasting GTT.
Patients will be instructed by their physicians regarding pretest preparation, which includes eating high-carbohydrate meals for several days and then fasting for 12 hours immediately before the test.
Testing begins between 0700 (pronounced “oh-seven-hundred”) and 0900 (7 am to 9 am), with the collection of a fasting blood specimen and sometimes a urine specimen. These specimens should be tested before the GTT proceeds. In the event that the glucose level is severely elevated, the physician may decide not to proceed with the test.
The patient then drinks a standardized amount of glucose solution within 5 minutes. Timing for the rest of the procedure begins after the drink is finished. The phlebotomist makes a collection schedule. The first collection is 1 hour after glucose ingestion. The second collection is 2 hours after glucose ingestion, and so on, for the number of hours the physician has indicated. Note that some procedures may call for collection of the first sample at 30 minutes after ingestion of the glucose solution, followed by another collection 30 minutes later at the 1 hour mark, and then hourly samples after that. Follow the protocol of your facility. The phlebotomist gives the patient the collection schedule and instructs the patient to return to the collection station at the appropriate times (Table 14-1). Patients also should be instructed to continue to fast and drink plenty of water so that they remain adequately hydrated throughout the test.
TABLE 14-1
Collection Schedule for Oral Glucose Tolerance Tests
| Test | Schedule |
| 2-Hr glucose tolerance test (GTT) | Fasting, 1 hr, and 2 hr |
| 3-Hr GTT | Fasting, 1 hr, 2 hr, and 3 hr |
| 5-Hr GTT | Fasting, 1 hr, 2 hr, 3 hr, 4 hr, and 5 hr |
Some patients do not tolerate the test well. Any vomiting should be reported to the physician ordering the test. If the patient vomits shortly after the test begins, the procedure will have to be started again.
All collections should be made on time and using the same collection method (i.e., venipuncture or dermal puncture) and anticoagulant for each sample. A urine specimen may be collected at the same time. Samples should be labeled with the time from test commencement (1 hour, 2 hours, and so forth).
Other tolerance tests
Similar procedures are used for other tolerance tests. The epinephrine tolerance test determines the patient’s ability to mobilize glycogen from the liver. In response to a dose of the hormone epinephrine, glycogen is converted to glucose and released into the bloodstream. The test begins with a fasting specimen, followed by the epinephrine injection administered by the physician. Specimen collection begins 30 minutes later. The glucagon tolerance test is identical in purpose and procedure, except that the hormone glucagon is injected instead of epinephrine.
The lactose tolerance test (LTT) determines whether the lactose-digesting enzyme lactase is present in the gut. A 3-hour GTT is performed first to produce a baseline glucose uptake graph. The following day a lactose tolerance test is performed. The procedure is identical to the GTT except that lactose is consumed and a tourniquet may not be used. Be sure to follow the protocol for your facility. Because lactose is broken down into glucose and galactose, the timed samples should produce an identical glucose uptake graph. Lower glucose levels indicate a problem with lactose metabolism.
Lactose intolerance can also be determined using the hydrogen breath test, which does not require a blood sample. In this test, the patient drinks a lactose solution and then exhales into a collection bag. Hydrogen in the breath is a sign of undigested lactose in the gut.
A stool acidity test may be ordered for patients and children who are unable to undergo the other tests. Lactic acid and other acids from undigested lactose can be detected in a stool, or fecal, sample.
Diurnal variation
Many substances in the blood (especially hormones) show diurnal variation, or regular changes throughout the day (Box 14-2). Cortisol, for instance, is usually twice as high in the morning as in the late afternoon. The time for the draw is usually scheduled for the diurnal peak or trough. Cortisol is usually drawn at 1000 or 1600, for instance.
Therapeutic drug monitoring
Patients differ greatly in the rate at which they metabolize or excrete medications. In addition, the “margin of safety,” or the difference between the level at which a drug is therapeutic and the level at which it becomes toxic, may be narrow. To maintain constant therapeutic plasma drug levels and ensure that the drug does not reach toxic levels, a patient may require timed specimens to measure the levels of the medication. This is known as therapeutic drug monitoring (TDM). Results of TDM are used by the pharmacy to adjust drug dosing. Table 14-2 lists some commonly monitored drugs. The rate of metabolism is often given in terms of the drug’s half-life, the time for half of the drug to be metabolized. Drugs with long half-lives, including digoxin, often require only one timed specimen. Drugs with short half-lives, such as the aminoglycoside antibiotics (including gentamicin, tobramycin, and vancomycin) require the most careful monitoring. Monitoring for these rapidly metabolized antibiotics is done with a pair of specimens, known as a peak and a trough.
TABLE 14-2
Commonly Monitored Therapeutic Drugs
| Drug Name | Therapeutic Purpose |
| Dilantin and valproic acid | Anticonvulsant |
| Gentamicin | Antibiotic |
| Procainamide and digoxin | Heart medication |
| Theophylline | Antiasthmatic |
| Tobramycin | Antibiotic |
| Vancomycin | Antibiotic |
Collection is usually timed to coincide with either the trough or the peak serum level. The trough level is the lowest serum level and occurs immediately before the next dose of medicine is given. The requisition will specify the actual collection time, which is usually 30 minutes before the dose. The peak level, or highest serum level, occurs sometime after the dose is given; exactly when depends on the characteristics of the drug, the patient’s own metabolism, and the method of administration. The tube should be labeled with the draw time in all cases. Careful attention to the correct timing of draw, and accurate labeling, is vital to the patient’s health. TDM results are needed promptly, because the pharmacy is usually waiting for the results to determine both the timing and the concentration of the next dose of drug. By properly timing the administration of medications, the doctor can have the maximum beneficial effect with the fewest side effects. Most drugs need to be collected in a red top tube because the gel in a serum separator tube interferes with the analysis of the drugs.
Blood cultures
A blood culture (BC) is ordered to test for the presence of microorganisms in the blood, a potentially life-threatening situation. Such microorganisms include bacteria, fungi, and protozoa. Bacteremia refers specifically to the presence of bacteria in the blood. Bacteria occasionally enter the bloodstream, for instance through the gut or from excessively vigorous toothbrushing, which breaks capillaries. These rarely cause illness. Septicemia is a life-threatening infection caused by rapid multiplication of pathogens in the bloodstream. When microorganisms in the blood trigger a systemic inflammatory response, it is called sepsis. Patients with symptoms of chills and fever, or fever of unknown origin (FUO), may require a BC. BCs are ordered as short turnaround time (stat) or timed specimens.
Isolating pathogenic organisms from blood is difficult because the number of organisms may be low (leading to false-negative results) and the potential for sample contamination is high (leading to false-positive results). To increase the likelihood of finding pathogens and decrease the number of false positives, collection is performed at timed intervals and from multiple sites. Aseptic collection technique is critical for meaningful results. Drawing the correct volume is also critical, because the ratio of blood to culture media depends on the system used. Always check your institution’s guidelines. Volumes for pediatric patients differ from those for adults.
Types of collection containers
There are three basic types of containers for collecting BCs:
1. A long-necked bottle, which accepts a BD Vacutainer needle and tube holder.
2. A shorter bottle, which accepts a winged infusion device, such as a BD Bactec, using a special adapter.
3. A standard evacuated tube with sodium polyanethole sulfonate (SPS) anticoagulant.
Figure 14-1 shows these three types of collection containers.
Timing
The number of organisms in the bloodstream is often highest just before a spike in the patient’s temperature. By frequently recording the temperature, these spikes can often be predicted and collection scheduled accordingly. In other situations, collection may be timed at regular intervals, often hourly or just before antibiotic administration.
Multiple sites
Contamination of the sample by skin bacteria is a frequent complication of BC collection. However, distinguishing contaminants from true pathogens can be difficult because some contaminants can grow on indwelling devices, causing infection in the patient. To reduce errors caused by this contamination, a known skin contaminant must be cultured from at least two different sites to be considered a blood pathogen. It is even better to collect two pairs of samples from two different sites. This helps detect contamination of samples by skin bacteria. The physician should be consulted about the exact sites and timing of the two sets.
Sample collection
Procedure 14-1 outlines the steps in BC collection. As noted earlier, samples are collected either directly into a bottle containing culture media or indirectly into a sterile anticoagulated tube for later transfer to culture media in the laboratory. The ratio of blood to culture media is crucial to the culture, so be sure to collect the sample size indicated on the bottle. In addition to culture media, some tubes contain activated charcoal, which absorbs antibiotics from the patient’s blood so that they do not inhibit growth of the bacteria in the culture tube. Also, some bacteria, called anaerobic bacteria, cannot tolerate oxygen, while others, called aerobic bacteria, use it. Some samples you collect will exclude oxygen and will be used to grow anaerobic bacteria. Other samples you collect will include oxygen and will be used to grow aerobic bacteria.
PROCEDURE 14-1
![]()
![]()
![]()
![]()
![]()
For direct collection into culture media, two samples, one aerobic and one anaerobic, are collected from each site. When using a syringe, collect the anaerobic sample first and the aerobic sample second, since the second sample will be more likely to have been exposed to air. The transfer must be performed with the appropriate transfer safety device. When using a butterfly, the opposite order is used: Collect the aerobic sample first, since there is air inside the butterfly tube, and then collect the anaerobic sample. Be sure to label the samples to reflect their order of collection.
For anticoagulated tube collections, only one tube is collected per site. At the laboratory, the specimen is cultured onto the appropriate media.
Blood donor collection
Blood donation is a vital link in the health care system, and the phlebotomist plays a central role in the collection of donated blood. Blood banks collect and store donated blood for use in both emergency and scheduled transfusions. Guidelines for uniform collection procedures and safeguards have been established by the American Association of Blood Banks (AABB) and the U.S. Food and Drug Administration (FDA).
Potential donors must be screened to ensure that the donation is not harmful to the donor or the recipient. Donors must be at least 17 years old (16 years in some states), weigh a minimum of 110 pounds, and not have donated blood in the past 8 weeks. Donor screening is usually done by the phlebotomist. All information provided during the screening is confidential. The screening process is performed every time a person donates blood. Screening involves the following:
• Registration. The donor must provide his or her name, date of birth, address, other identifying information, and written consent. All information must be kept on file for at least 10 years.
• Interview and medical history. This is done in private by a trained interviewer. All responses are kept confidential. Potential donors may be rejected for a variety of reasons, including exposure to human immunodeficiency virus (HIV) or viral hepatitis, current drug use, or cardiovascular conditions.
• Physical examination. The donor’s weight, temperature, blood pressure, pulse, and hemoglobin level are determined. Hemoglobin is usually measured with a drop of whole blood from a dermal puncture. A hematocrit may be substituted for the hemoglobin determination.
Collection procedure
Blood is collected by the unit, whose volume is 400 to 500 mL, or approximately 0.5 liter (Figure 14-2). It is collected directly into a sterile plastic bag, which hangs below the collection site and fills by gravity. The weight of the filled bag triggers a clamp that stops the collection. A 16- to 18-gauge needle is used for collection. This large needle speeds the collection and prevents hemolysis.
A large vein in the antecubital area is used for donor collection. The site is cleaned first with soap and water and then with iodine. After the puncture, the needle is secured to the arm with tape to prevent motion during the collection. The donor is instructed to pump his or her fist to increase flow. (Hemoconcentration may result from this action, but this is not a concern with blood collected for transfusion.) The phlebotomist stays with the donor during the collection and observes for any signs of distress, anxiety, or pale skin. After needle removal, the phlebotomist instructs the patient to apply firm pressure to the site and bandages the site when the bleeding has stopped.
Autologous donation
An autologous donation is blood donated by a patient for his or her own use later. Patients planning surgery often make autologous donations before the procedure. This may reduce the likelihood of complications and may be especially useful for patients with rare blood types. Because multiple units might be needed during surgery, patients may need to donate several times. A patient can donate as often as every 72 hours, assuming that his or her health is good. Hemoglobin is checked during the donation series and should not fall below 11 g/dL.
Therapeutic phlebotomy
Therapeutic phlebotomy is the removal of blood from a patient’s system as part of the treatment for a disorder. The principal disorders treated by therapeutic phlebotomy are polycythemia, a disease characterized by excessive production of red blood cells (RBCs), and hemochromatosis, an excess of iron in the blood. In both cases, periodic removal of a unit of blood may be part of the treatment program. Because such a large volume must be removed, therapeutic phlebotomy is performed in the donor center, although a special area may be set aside for this purpose. This blood cannot be used for transfusion in most instances.
Special specimen handling
Cold agglutinins
Cold agglutinins are antibodies often formed in response to infection with Mycoplasma pneumoniae, a cause of atypical pneumonia. The antibodies created by the immune system during the infection may also react with RBCs at temperatures below body temperature, causing them to stick together, hence cold agglutinins (agglutination is the process of sticking together). Because the agglutinins attach to RBCs at cold temperatures, the specimen must be kept warm until the serum is separated from the cells to avoid falsely lowering the agglutinin levels. A patient with cold agglutinins is sometimes asymptomatic, but the presence of the antibodies can cause problems if the patient’s body temperature falls. This may occur when a patient undergoes cardiopulmonary bypass, for example, which requires cooling the patient to slow the metabolic activity of the heart. If the patient has undetected cold agglutinins, this hypothermia will cause dangerous agglutination of red blood cells. A presurgical screening is therefore sometimes required to detect these cold agglutinins.
To collect a cold agglutinin sample, prewarm a plain red-topped tube (containing no gel) in a 37° C incubator for 30 minutes. To keep the specimen warm, it can be wrapped in an activated heel-warmer pack or placed in the incubator. Deliver the specimen as quickly as possible to the laboratory.
Cryofibrinogen and cryoglobulin
Warm collection and storage are required for two other types of samples: cryofibrinogen (an abnormal type of fibrinogen) and cryoglobulin (an abnormal serum protein). Both precipitate when cold and redissolve when warmed. These samples should be collected and handled in the same manner as a cold agglutinin sample.
Chilled specimens
A number of tests require that the specimen be chilled immediately after collection (Box 14-3). Chilling is used to prevent chemical changes that would alter test results. The sample should be placed in crushed ice or in an ice and water mixture and immediately delivered to the laboratory. The temperature should be 1° C to 5° C, and the sample should be transported to the laboratory for testing within 5 minutes of collection (Figure 14-3). A lactic acid sample should also be collected without a tourniquet, and without the patient making a fist, since both of these can raise lactic acid.
Light-sensitive specimens
Exposure to light can break down or alter certain blood constituents (Box 14-4). Specimens to be tested for these constituents must be protected from light after collection. This is done by wrapping the tube in aluminum foil immediately after collection (Figure 14-4). An amber-colored microtube can be used for dermal collection (e.g., of bilirubin samples).
Time-sensitive specimens
Some analytes are unstable or volatile. Because of this, the tests must be performed rapidly after the sample is taken. Box 14-5 lists analytes that are time sensitive.
Legal and forensic specimens
Blood specimens may be collected for use as evidence in legal proceedings, including alcohol and drug testing, deoxyribonucleic acid (DNA) analysis, or paternity or parentage testing. Such samples must be handled with special procedures designed to prevent tampering, misidentification, or interference with the test results.
The most important concept in handling forensic specimens is the chain of custody (COC), a protocol that ensures that the sample is always in the custody of a person legally entrusted to be in control of it. The chain begins with patient identification and continues through every step of the collection and testing process. COC documentation includes special containers, seals, and forms, as well as the date, time, and identification of the handler (Figure 14-5).

The National Institute on Drug Abuse (NIDA) has established requirements for patient preparation and specimen handling in COC samples. These requirements include the following:
• The purpose and procedure of the test must be explained to the patient.
• The patient must sign a consent form.
• The patient must present picture identification.
• The specimen must be labeled appropriately to establish a COC.
• The specimen must be sealed in such a way that any tampering can be identified.
• The specimen must be placed in a locked container before transport to the testing site.
Legal alcohol collection
Collection for alcohol testing is most common in emergency departments. This collection requires special handling to prevent alteration of the test results. Important features of alcohol testing include the following:
• The site must not be cleaned with alcohol, as this would falsely elevate the result. Instead, use soap, water, and sterile gauze or another nonalcoholic antiseptic solution.
• Tubes must be filled as full as the vacuum allows to minimize the escape of alcohol from the specimen into the space above.
• Note on the requisition form that the site was cleansed with soap and water or another a nonalcoholic solution.
Blood smears
Blood smears are made to allow microscopic examination of the blood cells. The blood smear is used for determining the proportion of the various blood cell types, called a differential count; counting reticulocytes; and performing special staining procedures.
Blood smear preparation
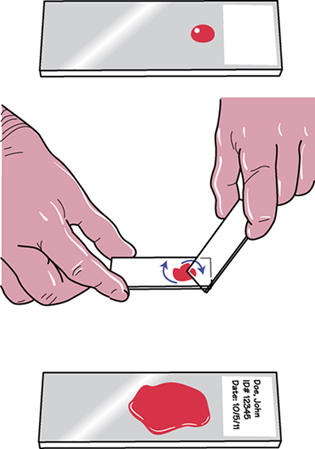
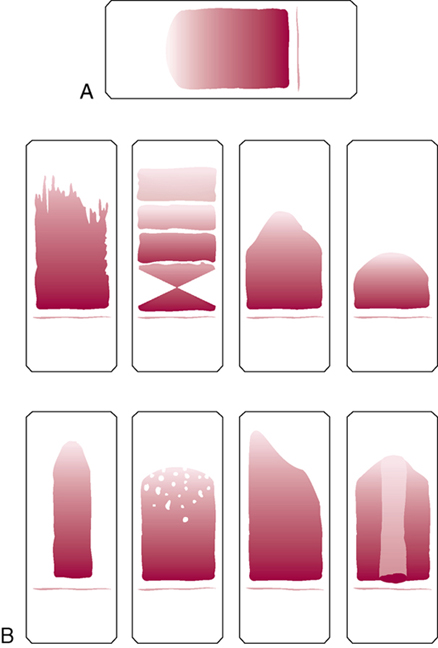
Blood smears are usually prepared following dermal puncture. Slides are made in pairs. Figure 14-6 illustrates a good smear, as well as several examples of unacceptable smears. Procedure 14-2 outlines how to prepare blood smears.
PROCEDURE 14-2
![]()
![]()
![]()
![]()
![]()
 | |
|
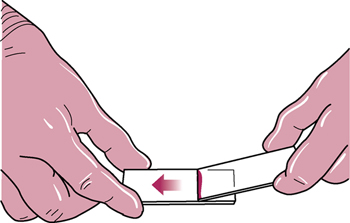
Place the edge of a second slide, the “spreader,” onto the first slide in front of the blood at a 25- to 30-degree angle, and draw it back to just contact the drop. Move the spreader slide forward, away from the drop, in one continuous movement to the end of the slide. The blood will be drawn along over the slide. To make the second slide, place a drop of blood onto the spreader slide. Using the first slide as the spreader, repeat the procedure. |
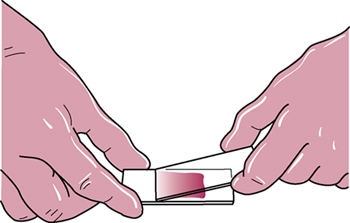 |
 | |
|
An acceptable smear must have a feathered edge, meaning that the cells appear to thin out farther from the original drop. At the far end, you should see a very thin transparent layer. This is the area from which the differential count is made. See Figure 14-6 for examples of blood smears. Table 14-3 describes common problems with smears and their likely causes. |
 |
TABLE 14-3
Unacceptable Smears and Their Causes
| Result | Cause |
| Holes in the smear | Dirty slide |
| No feathered edge | Drop that is too large Drop that is not placed close enough to the far edge of the slide
Spreader slide that is lifted before it reaches the end of the sample slide |
| Smear that is too thick and long | Drop of blood that is too big Angle of the spreader slide >30 degrees |
| Smear that is too thin and short | Drop of blood that is too small Angle of the spreader slide <25 degrees |
| Streaks in the feathered edge | Chipped or dirty spreader slide Spreader slide that is not placed flush against smear slide
Drop of blood that is in front of the spreader slide |
| Uneven distribution of blood | Uneven pressure on the spreader slide Uneven movement of the spreader slide |
Malaria smears
Malaria is caused by blood-borne protozoa of the genus Plasmodium. A patient with malaria has cycles of fever and chills that coincide with the life cycle of the parasite in the bloodstream. Malaria is diagnosed with a blood smear, drawn as a stat or timed collection just before the onset of fever or chills. The test requires two to three regular smears, plus a thick smear. To make the thick smear, use a larger drop of blood, and spread it out to only about the size of a dime (Figure 14-7). The sample must be allowed to dry for at least 2 hours; it is then stained to reveal the parasites.
Review for certification
Routine phlebotomy specimens are often collected in the early morning for inpatients and nursing home patients because most patients are in the basal state at that time. A 12-hour complete fast may be required for some tests, especially glucose and triglycerides. The phlebotomist must ensure that the patient has complied with the fast and note any irregularities on the requisition. Timed specimens include the 2-hour postprandial test for diabetes mellitus and the GTT for diabetes mellitus, gestational diabetes, hyperglycemia, and hypoglycemia. TDM tests are timed to coincide with the peak or trough of serum drug levels. Testing for the presence of microorganisms in the blood requires a BC, with collection at timed intervals from multiple sites, using aseptic collection of the proper volume. Units of blood may be collected for donation to the blood bank or for use by the patient during later surgery. Therapeutic phlebotomy removes blood from the patient, most often to treat polycythemia or hemochromatosis. Samples to test for cold agglutinins, antibodies formed against Mycoplasma pneumoniae, must be kept warm after collection, as must those for cryofibrinogen and cryoglobulin. Other specimens may require chilling or protection from light.
Legal or forensic samples require documentation of the COC, a protocol that ensures that the sample is always in the custody of a person legally entrusted to be in control of it. Alcohol specimens must be handled carefully to avoid contamination of the sample with alcohol and prevent escape of alcohol from the blood.
Blood smears are used for differential counts, counting reticulocytes, and special staining procedures. An acceptable smear must have a feathered edge, prepared by careful drawing of the blood drop across the slide using another slide.
Study questions
See answers in Appendix F.
2. List 10 factors that influence blood composition.
3. What three things are timed specimens most often used to monitor?
5. What is TDM, and why would this be ordered?
7. What collection technique is critical for meaningful BC results?
8. Describe the screening process a potential blood donor goes through.
9. What special handling procedure is necessary for cold agglutinin samples?
10. List five tests that require transport on ice.
12. Describe the important features of blood alcohol testing.
13. Explain the procedure for preparing a blood smear.
14. Describe the procedure for preparing a thick smear to test for malaria.
15. List the disorders for which therapeutic phlebotomy is used. Define the diseases based on the word roots, prefixes, suffixes, or a combination of these.
Certification examination preparation
See answers in Appendix F.
1. Timed specimens are most frequently collected to monitor
2. A 2-hour postprandial test is used to test for
4. For a 3-hour GTT, how many blood samples will be collected?
5. Within what time period should a patient drink the glucose solution required for a GTT?
6. The epinephrine tolerance test determines the patient’s ability to
8. Therapeutic phlebotomies are commonly performed on patients with
9. To collect a cold agglutinin sample,
a. warm the sample tube for 30 minutes before collection.
b. pack the sample tube in ice before collection.
10. Which specimen requires chilling during transfer to the laboratory?
11. When making smears for malaria,
a. prepare two regular smears only.
b. prepare two to three regular smears and one thick smear.
12. It is necessary for a patient to fast for 12 hours before
13. Factors influencing blood composition include all of the following except:
14. Which of the following specimens is not sensitive to light?