Rats
Chromodacryorrhea
Basic Information 
Definition
Porphyrin-pigmented tears secreted by the harderian glands of rats. Chromodacryorrhea literally means “excessive production of colored tears” (chromo Gk = color; dacryo Gk = gland; rhea = to pour out).
Epidemiology
Etiology and Pathophysiology
• Any disease or condition that results in chronic stress will result in chromodacryorrhea.
• Harderian glands of rodents with “red tears” exhibit a variety of histological autofluorescence patterns. In addition, their secretions are also affected by protoporphyrin binding to lipids, affecting fluorescence.
 Inflammation of the harderian gland (i.e., dacroadenitis) causes an increase in secretions. The most common cause is infection with SDAV, a coronavirus of rats.
Inflammation of the harderian gland (i.e., dacroadenitis) causes an increase in secretions. The most common cause is infection with SDAV, a coronavirus of rats.
 The tears are secreted via activation of the parasympathetic nervous system via muscarinic receptors. Anticholinergic drugs have been shown to block secretions.
The tears are secreted via activation of the parasympathetic nervous system via muscarinic receptors. Anticholinergic drugs have been shown to block secretions.
 Harderian glands’ secretions predominantly contain lipids that act as pheromones. The presence of porphyrins in Harderian gland secretions is more the exception than the rule when describing these secretions in rodents. Generally, porphyrins give color to secretions.
Harderian glands’ secretions predominantly contain lipids that act as pheromones. The presence of porphyrins in Harderian gland secretions is more the exception than the rule when describing these secretions in rodents. Generally, porphyrins give color to secretions.
Diagnosis 
Initial Database
• Wood’s lamp examination reveals bright orange-red fluorescence; allows differentiation from dried blood
• Further diagnostics will dependent on the clinical signs and suspected primary underlying disease, such as respiratory tract disease (see Respiratory Tract Disease, Acute, and Respiratory Tract Disease, Chronic).
Prognosis and Outcome 
Prognosis depends on underlying cause. If clinical signs are due to stress or husbandry, the prognosis is excellent if properly addressed. For other causes, the prognosis will vary from poor to good.
Pearls & Considerations 
Mammary and Pituitary Tumors
Client Education Sheet Available on Website ![]()
Basic Information 
Definition
Mammary gland tumors are the most frequently occurring tumors in female rats. Histologically, most are mammary fibroadenomas, although adenocarcinomas are also seen.
Special Species Consideration
Rats have 12 mammary glands along the mammary chain, which extends from the cervical region to the tail base. Mammary tumors can arise in any of these locations.
Epidemiology
Genetics and Breed Predisposition
In inbred rat strains susceptible to mammary tumors expression levels of several prolactin-regulated genes are significantly elevated (e.g., messenger RNA’s encoding prolactin and its cell surface receptor are amplified) indicating the presence of increased prolactin signaling in the mammary glands of mammary tumor susceptible rat strains.
Risk Factors
• Sex: females at higher risk than males
• Age: higher risk at greater than 2 years of age
• Nutrition: increased incidence with high-fat diets, reduced incidence with food restriction
• Prolactin-secreting pituitary tumors: increased risk of mammary tumors
• Neuter status: decreased risk if ovariectomized by 90 days of age; suspected to also have decreased risk even if ovariectomized after 90 days of age, but this has not been proven.
• The frequency of mammary tumors and pituitary tumors is significantly lower in 18- to 24-month-old ovariectomized (4%) versus sexually intact (mammary tumors, 49%; pituitary tumors, 59%) rats. Therefore, the decreased frequency of mammary tumor development could be related to the decreased frequency of prolactin-secreting pituitary tumors.
Etiology and Pathophysiology
• As with other species, most mammary gland development occurs during puberty primarily under influence of estrogen and pregnancy under the influence of progesterone and prolactin.
• Neutering sexually immature females removes estrogen influence during mammary growth and prevents mammary epithelial ductal elongation, bifurcation, and extension throughout the fat pad. Inhibition of ductal morphogenesis significantly reduces the risk of mammary tumors by limiting the amount of mammary tissue that develops.
• Estrogen is an important stimulator of prolactin secretion that acts directly on pituitary lactotrophs and via the hypothalamus. Experimentally prolactinomas can be induced in rats by chronic estrogen administration. In the mammary gland, prolactin stimulates alveolar epithelial proliferation with its fibrous connective tissue support structure.
• Aging female rats exhibit changes in estrous cycle and reproductive patterns. At 10-12 months of age, the once-regular ovulatory cycles gradually become lengthened and irregular and eventually develop into a prolonged period of constant estrus characterized by ovaries containing big follicles that secrete large quantities of estrogen. Neutering sexually mature females removes constant estrogen secretion and decreases prolactin secretion so benign mammary tumors either do not develop or do not increase in size.
• Female rats with benign mammary tumors have 27 times higher plasma levels of prolactin than 6-month-old virgin rats, and prolactin levels similar to that of rats on the seventh day postpartum. Because of this, rats with prolactin-secreting pituitary tumors are at increased risk of mammary tumor development.
• In aging rats, prolactin secretion is increased and is reflected in the high blood prolactin level in both sexes. This change is due to a reduction of hypothalamic dopamine activity. The escape from hypothalamic inhibitory control leads to lactotroph hyperplasia and a high incidence of prolactin cell adenomas in old rats.
• Gene expression of spontaneous fibroadenomas and adenocarcinomas compared to a normal rat mammary gland in the same developmental state has shown that fibroadenomas do not progress to adenocarcinoma.
• Adenocarcinomas arise de-novo (i.e., without prior adenoma stage) and represent fewer than 10% of mammary tumors in pet rats.
• Fibroadenomas can reach 8-10 cm in diameter and do metastasize.
Diagnosis 
Initial Database
• Fine-needle aspirate: caution during interpretation because large mammary tumors may be necrotic and can be difficult to differentiate from an abscess on cytology
• Blood work: complete blood count and serum biochemistry screening as a preoperative workup
• Thoracic radiographs/CT: preoperative workup if underlying respiratory disease is suspected
Treatment 
Acute General Treatment
• If necessary, stabilization of the patient if mammary tumor is infected/sepsis is present, or if blood loss has ensued from ulceration of the mass.
• Complete surgical removal of the tumor and any ulcerated/infected skin or tissue is the treatment of choice.
 Because tumors may be quite large, closure of dead space is important to prevent seroma formation.
Because tumors may be quite large, closure of dead space is important to prevent seroma formation.
 Some masses are difficult to remove if they are in close association with the vulva.
Some masses are difficult to remove if they are in close association with the vulva.
• Concurrent ovariectomy is recommended if patient is stable.
Chronic Treatment
Because recurrence of mammary tumors at different locations can frequently occur, repeated surgical removal might be necessary.
Prognosis and Outcome 
• In general, survival following mastectomy is good.
• Quality of life is improved post mastectomy; however, controversy continues over whether tumor removal actually prolongs survival time.
• Death can occur with sepsis or blood loss if the mammary mass is not surgically removed and becomes ulcerated or infected.
Controversy
• To date, the only proven treatment and prevention of mammary tumor development consists of surgical removal of the tumor and ovariectomy. Other treatments have been discussed but have not proven to be effective in preventing recurrence of spontaneous tumors or in decreasing their size once present.
• Cabergoline is a prolactin inhibitor that suppresses pituitary prolactin secretion and can be given orally. It has been successfully used in the palliative treatment of a pituitary adenoma in a rat at a dose of 0.6 mg/kg PO q 72 h, and thus may be helpful in rats that have mammary tumor development secondary to prolactin-secreting pituitary tumors and in those unable to undergo ovariectomy.
• Gonadotropin-releasing hormone (GnRH) agonists
 Deslorelin implants (4.7 mg) have been used experimentally to suppress estrus in rats for 1 year and may be useful in rats that cannot be ovariectomized.
Deslorelin implants (4.7 mg) have been used experimentally to suppress estrus in rats for 1 year and may be useful in rats that cannot be ovariectomized.
 Leuprolide acetate has been experimentally shown to suppress the ability of the pituitary-gonadal system to secrete gonadotropin and testosterone for over 5 weeks; similar to deslorelin because it may be useful in rats that cannot undergo ovariectomy
Leuprolide acetate has been experimentally shown to suppress the ability of the pituitary-gonadal system to secrete gonadotropin and testosterone for over 5 weeks; similar to deslorelin because it may be useful in rats that cannot undergo ovariectomy
• Melatonin induces apoptosis of rat prolactin-secreting tumors. Experimentally, SC melatonin administration in experimentally induced tumor-bearing rats significantly increased survival time and reduced prolactin levels but did not change the mammary tumor growth rate.
• Tamoxifen: antiestrogen used in the treatment of human breast cancer. This agent would be useful only in mammary adenocarcinomas that are estrogen receptor positive. This drug is NOT recommended, given the low incidence of adenocarcinoma in rats and the fact that it has been shown to induce hepatic cancer and proliferation of the rat uterus.
Pearls & Considerations 
Comments
Histopathologic examination of removed tumors should be performed because spontaneous mammary adenocarcinoma has a 5%-10% incidence. Individual genetic variability and environmental factors such as nutrition and maternal effects in utero and during lactation most likely affect quantitative trait loci (QTL) that control susceptibility to mammary adenocarcinoma. However, the individual genetic traits and QTL that influence gene function have not yet been elucidated.
Alkis, I, et al. Long term suppression of oestrus and prevention of pregnancy by deslorelin implant in rats. Bull Vet Inst Pulawy. 2011; 55:237–240.
Hotchkiss, C, et al. Effect of surgical removal of subcutaneous tumors on survival of rats. J Am Vet Med Assoc. 1995; 206:1575–1579.
Marxfeld, H, et al. Gene expression in fibroadenomas of the rat mammary gland in contrast to spontaneous adenocarcinomas and normal mammary gland. Exp Toxicol Pathol. 2006; 58:145–150.
Mayer, J, et al. Extralabel use of cabergoline in the treatment of a pituitary adenoma in a rat. J Am Vet Med Assoc. 2011; 239:656–660.
Saez, MC, et al. Melatonin increases the survival time of animals with untreated mammary tumours: neuroendocrine stabilization. Mol Cell Biochem. 2005; 278:15–20.

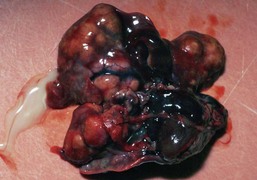
Mammary and Pituitary Tumors Large mammary fibroadenoma on a female rat. Note the close proximity to the left hind leg which impeded normal ambulation.
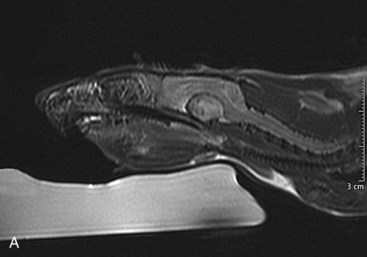
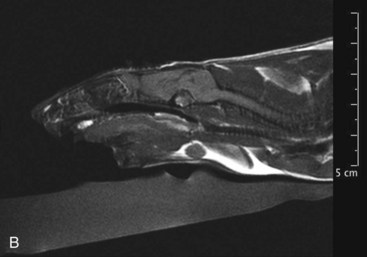
Mammary and Pituitary Tumors A, A pituitary tumor of a rat imaged with an MRI scan. B, The same tumor as in the previous image a few weeks after treatment with cabergoline. Note the significant shrinking of the tumor. (Photo courtesy Jörg Mayer, The University of Georgia, Athens.)
AUTHOR: NICOLE R. WYRE and THOMAS M. DONNELLY
Renal Disease
Basic Information 
Definition
Kidney or renal disease is a general term that describes any damage that reduces the functioning of the kidney.
Synonyms
Epidemiology
Species, Age, Sex
• Male rats develop a more severe form of chronic progressive nephrosis, usually earlier in life than females; lesions are more severe in rats over 12 months of age.
• Nephrocalcinosis is more common in females and can be found in animals as young as 7 weeks of age. Blood estrogen levels may play a role in that the disease can be prevented by ovariectomy and is induced in castrated male and female rats by estrogen administration.
Genetics and Breed Predisposition
A significantly higher prevalence of chronic progressive nephrosis is seen in the Sprague-Dawley strain of rat. Osborne-Mendel and Buffalo strains are relatively insusceptible. Elevated prolactin levels are suspected of contributing to more severe disease.
Risk Factors
• Chronic progressive nephrosis: high-protein diets designed for superior body growth result in earlier onset of more severe disease
• Nephrocalcinosis: may be the result of a number of dietary factors, including magnesium deficiency, elevated dietary phosphorus or calcium, and diet preparations with a low calcium-to-phosphorus ratio
• Suppurative pyelonephritis/nephritis: isosthenuria, urolithiasis, and lower urinary tract infections
Etiology and Pathophysiology
• Chronic progressive nephrosis
 A high protein diet may acutely increase the glomerular filtration rate, possibly causing intraglomerular hypertension, which would lead to progressive loss of renal function.
A high protein diet may acutely increase the glomerular filtration rate, possibly causing intraglomerular hypertension, which would lead to progressive loss of renal function.
 Albuminuria not only serves as a marker of glomerular injury but is also associated with tubulointerstitial injury.
Albuminuria not only serves as a marker of glomerular injury but is also associated with tubulointerstitial injury.
 In rats, no single mechanism has been identified that explains the association between all dietary factors that have been related to the prevalence of nephrocalcinosis.
In rats, no single mechanism has been identified that explains the association between all dietary factors that have been related to the prevalence of nephrocalcinosis.
 Nutritional studies have shown that diets high in phosphorus or low in calcium, with a net Ca : P molar ratio of less than 1.0, contribute to the development of nephrocalcinosis lesions. Increasing the calcium and phosphorus content and the Ca : P ratio to greater than 1.0 and closer to 1.3 markedly decreased the incidence and severity or prevented the occurrence of nephrocalcinosis lesions.
Nutritional studies have shown that diets high in phosphorus or low in calcium, with a net Ca : P molar ratio of less than 1.0, contribute to the development of nephrocalcinosis lesions. Increasing the calcium and phosphorus content and the Ca : P ratio to greater than 1.0 and closer to 1.3 markedly decreased the incidence and severity or prevented the occurrence of nephrocalcinosis lesions.
Diagnosis 
Initial Database
 Isosthenuria (normal specific gravity, 1.022-1.050)
Isosthenuria (normal specific gravity, 1.022-1.050)
 Proteinuria (mild proteinuria is normal in rats)
Proteinuria (mild proteinuria is normal in rats)
 Sediment analysis: casts, crystals, inflammatory or neoplastic cells, bacteria
Sediment analysis: casts, crystals, inflammatory or neoplastic cells, bacteria
• Complete blood count: may be normal
 Radiography: assess for increases or decreases in kidney size, radiopaque calculi within the urinary tract, abdominal masses associated with the urinary tract, and bladder distention.
Radiography: assess for increases or decreases in kidney size, radiopaque calculi within the urinary tract, abdominal masses associated with the urinary tract, and bladder distention.
 Ultrasonography: discern size, contour, and texture of the kidneys, allowing for differentiation of focal versus diffuse disease
Ultrasonography: discern size, contour, and texture of the kidneys, allowing for differentiation of focal versus diffuse disease
Treatment 
Acute General Treatment
• Discontinue any potentially nephrotoxic drugs.
• Identify and treat any prerenal or postrenal abnormalities.
• Identify any treatable conditions such as urolithiasis or pyelonephritis.
 To induce diuresis and correct azotemia, electrolyte, and acid-base imbalances
To induce diuresis and correct azotemia, electrolyte, and acid-base imbalances
 Subcutaneous administration: 60-100 mL/kg/d
Subcutaneous administration: 60-100 mL/kg/d
 Potassium supplementation of fluids based on blood potassium measurement
Potassium supplementation of fluids based on blood potassium measurement
 Indicated for cases of suppurative pyelonephritis/nephritis and cystitis
Indicated for cases of suppurative pyelonephritis/nephritis and cystitis
 Antibiotic selection should be based on culture and susceptibility whenever possible.
Antibiotic selection should be based on culture and susceptibility whenever possible.
 For empirical treatment, or for cases with negative urine culture, despite clinical suspicion, use antibiotics, which are effective against Gram-negative organisms and are renally excreted, to reach high tissue concentrations.
For empirical treatment, or for cases with negative urine culture, despite clinical suspicion, use antibiotics, which are effective against Gram-negative organisms and are renally excreted, to reach high tissue concentrations.
 Amoxicillin/clavulanic acid 15-20 mg/kg PO q 8-12 h
Amoxicillin/clavulanic acid 15-20 mg/kg PO q 8-12 h
• If hyperphosphatemic, alter diet and initiate enteric phosphate binders:
• Treat increased gastric acidity with H2 blockers:
• Multivitamin supplementation is recommended because the excessive amount of urine produced by failing kidneys commonly results in loss of water-soluble vitamins.
Chronic Treatment
• Maintain long-term dialysis with maintenance subcutaneous fluid therapy (owners can be taught to do this at home): 60-100 mL/kg/d SC.
• Antibiotic therapy for chronic pyelonephritis should be at least 4-6 weeks.
• Dietary management: high protein appears to be the major cause of severe nephropathy, and the term protein-overload nephropathy is often used. Changing the source of protein to one such as soy protein, restricting caloric intake, or modifying the diet to decrease protein consumption could decrease the severity of nephropathy. Changing the diet so that the Ca:P ratio is greater than 1.0 and is closer to 1.3 may decrease the incidence and severity of nephrocalcinosis in rats.
• The hyperphosphatemia that occurs in chronic renal failure is closely related to dietary protein intake because protein-rich diets are also high in phosphorus.
• Consider use of omega-3 fatty acid supplements based on studies showing their beneficial effects in other species.
Recommended Monitoring
• Overall condition and clinical response to therapy should be assessed in all patients with renal disease. Frequency of follow-up assessments varies with initial diagnosis and severity of disease. Periodic assessments for azotemia, anemia and phosphorus, and potassium and protein imbalances are recommended.
• Monitor body weight and condition, and adjust nutrition accordingly.
• Urinalysis and urine culture in patients being treated for pyelonephritis
Prognosis and Outcome 
• With any diagnosis of renal insufficiency or failure, prognosis varies with severity of clinical pathologic findings, duration of disease, and severity of primary renal failure. If secondary to infection or obstructive disease, prognosis is determined by duration of the disease process and success in treatment—medical or surgical—of the underlying condition of secondary renal insufficiency.
• Depending on initial diagnosis, disease severity, and response to therapy, quality of life issues and euthanasia should be discussed with the owner in terms of any patient with renal disease.
Controversy
Hematology, clinical chemistry, and urinalysis values may vary significantly with strain or breed of animal, nutritional status, sex, sampling site or frequency, time of day, stressors, age, health status, drug exposure, and environment. Therefore, normal values are broad; these variables should be kept in mind when interpreting individual animal values.
Pearls & Considerations 
• Many different terms are used to describe renal function and its deterioration.
 Azotemia refers to increased concentrations of urea nitrogen and creatinine and other nonproteinaceous nitrogenous waste products in the blood. Renal azotemia denotes azotemia caused by renal parenchymal changes.
Azotemia refers to increased concentrations of urea nitrogen and creatinine and other nonproteinaceous nitrogenous waste products in the blood. Renal azotemia denotes azotemia caused by renal parenchymal changes.
 Uremia is the presence of all urine constituents in the blood. Usually a toxic condition, it may occur secondary to renal failure or postrenal disorders, including urethral blockage.
Uremia is the presence of all urine constituents in the blood. Usually a toxic condition, it may occur secondary to renal failure or postrenal disorders, including urethral blockage.
 Renal reserve may be thought of as the percentage of “extra” nephrons—those not necessary to maintain normal renal function. Although it probably varies from animal to animal, this value is greater than 50% in most mammals.
Renal reserve may be thought of as the percentage of “extra” nephrons—those not necessary to maintain normal renal function. Although it probably varies from animal to animal, this value is greater than 50% in most mammals.
 Renal insufficiency begins when the renal reserve is lost. Animals with renal insufficiency outwardly appear normal, but have a reduced capacity to compensate for stresses such as infection or dehydration and have lost urine concentrating ability.
Renal insufficiency begins when the renal reserve is lost. Animals with renal insufficiency outwardly appear normal, but have a reduced capacity to compensate for stresses such as infection or dehydration and have lost urine concentrating ability.
 Renal failure is a state of decreased renal function that allows persistent abnormalities (azotemia and inability to concentrate urine) to exist; it refers to a level of organ function rather than a specific disease entity. Acute renal failure generally refers to cases of sudden decline of glomerular filtration rate resulting in an accumulation of nitrogenous waste products and inability to maintain normal fluid balance. Chronic renal failure generally refers to an insidious onset with slow progression (usually months to years) of azotemia and inadequately concentrated urine.
Renal failure is a state of decreased renal function that allows persistent abnormalities (azotemia and inability to concentrate urine) to exist; it refers to a level of organ function rather than a specific disease entity. Acute renal failure generally refers to cases of sudden decline of glomerular filtration rate resulting in an accumulation of nitrogenous waste products and inability to maintain normal fluid balance. Chronic renal failure generally refers to an insidious onset with slow progression (usually months to years) of azotemia and inadequately concentrated urine.
• It is important to realize that most of the renal diseases discussed can manifest as varying stages of compromise in renal reserve, renal insufficiency, or renal failure. If or when the disease process progresses depends on variables such as the specific disease in question, environmental factors, and the individual animal itself.
Comments
• NTP-2000 open formula is one diet available in laboratory medicine that is low in protein (14.0%) and has a Ca:P ratio approximating 1.3 : 1; it has been found to decrease the incidence of nephrocalcinosis in rats.
• Another laboratory rat diet, AIN-93G, has a lower phosphorus content (0.3%) and a higher Ca:P ratio and has been shown to lower the incidence of nephrocalcinosis.
• Dietary salt content has been found to have an effect on hypertension associated with hydronephrosis in rats. Hydronephrosis as a result of partial ureteral blockage led to increased blood pressure, which worsened significantly on a high-salt diet versus a low-salt diet.
• High levels of dietary soy isoflavones induced nephrocalcinosis formation, depending on the strain of laboratory rat.
Fisher, PG. Exotic mammal renal disease: causes and clinical presentation. Vet Clin North Am Exotic Anim Pract. 2006; 9:33–67.
Fisher, PG. Exotic mammal renal disease: diagnosis and treatment. Vet Clin North Am Exotic Anim Pract. 2006; 9:69–96.
Rao, GN. Diet and kidney diseases in rats. Toxicol Pathol. 2002; 30:651–656.
Respiratory Tract Disease, Acute
Basic Information 
Definition
Acute bacterial pneumonia in rats is caused by subclinical infection with Streptococcus pneumoniae and/or Corynebacterium kutscheri, which develops into clinical pneumonia and/or septicemia secondary concurrent infection or immunosuppression.
Epidemiology
Species, Age, Sex
Older animals are at increased risk for C. kutscheri. Younger animals are at increased risk for S. pneumoniae.
Clinical Presentation
Disease Forms/Subtypes
With both of these bacterial infections, animals can have no apparent clinical signs or can have severe respiratory disease and/or acute death.
Etiology and Pathophysiology
• Both S. pneumoniae and C. kutscheri colonize the upper respiratory tract (nasopharynx and tympanic bulla with S. pneumoniae and oropharynx, cervical, and submandibular lymph nodes with C. kutcheri) and can remain subclinical in the absence of concurrent disease.
• Concurrent infection with other pathogens (see Respiratory Tract Disease, Chronic) and confounding stressors lead to immune suppression, triggering a latent infection to become clinical.
• Suppurative inflammation of the upper respiratory tract is followed by infection of the lower respiratory tract, leading to bronchopneumonia and pleuritis.
• Bacteremia can lead to infection in other organs such as arthritis, meningitis, pericarditis, hepatitis, splenitis, and peritonitis or acute death.
Diagnosis 
Initial Database
• Thoracic radiographs/CT: findings consistent with pulmonary consolidation and/or pleural effusion
• Skull radiographs/CT/MRI: tympanic bullae sclerosis or effusion if otitis media is present
• Serologic testing: C. kutscheri (ELISA)
• Complete blood count: neutrophilia, neutropenia if septic
• Serum biochemistry: hypoglycemia if septic
• Submandibular lymph node aerobic culture for C. kutscheri: caution as nonclinical animals can harbor bacteria in these lymph nodes
Treatment 
Acute General Treatment
• Oxygen therapy if patient is dyspneic and/or cyanotic
• Fluid therapy: may require intraosseous administration if patient is severely compromised
• Antibiotic therapy should be based on aerobic culture and sensitivity results:
Prognosis and Outcome 
Little is known about the prognosis of pure acute bacterial pneumonia because co-infection with other respiratory pathogens is common, as are subclinical infections.
Pearls & Considerations 
Prevention
Because both of these bacteria can be present without causing clinical disease, preventive measures are focused on decreasing stress, avoiding immune suppressive drugs, and maintaining appropriate diet/husbandry to avoid conversion to clinical disease.
Client Education
All clients owning rats should understand the frequency of respiratory disease in rats and the importance of proper housing (good ventilation, avoidance of crowding, avoidance of dusty bedding such as wood shavings) and close observation for any signs of respiratory disease, so that treatment can be administered as soon as possible.
Amao, H, et al. Natural and subclinical Corynebacterium kutscheri infection in rats. Lab Anim Sci. 1995; 45:11–14.
Barthold, SW, et al. The effect of selected viruses on Corynebacterium kutscheri infection in rats. Lab Anim Sci. 1988; 38:580–583.
Borkowski, GL, et al. Diagnostic exercise: pneumonia and pleuritis in a rat [Streptococcus pneumoniae]. Lab Anim Sci. 1990; 40:323–325.
Corning, BF, et al. Group G streptococcal lymphadenitis in rats. J Clin Microbiol. 1991; 29:2720–2723.
Respiratory Tract Disease, Chronic
Client Education Sheet Available on Website ![]()
Basic Information 
Definition
Chronic respiratory disease (CRD) in rats is a multifactorial respiratory tract infection caused primarily by Mycoplasma pulmonis, commonly in association with other concurrent infections, resulting in chronic bronchitis and bronchiectasis.
Epidemiology
Etiology and Pathophysiology
• M. pulmonis colonizes the epithelial cells of the respiratory tract, middle ear, and epithelia of female genital tract.
• Although M. pulmonis causes upper and lower respiratory system lesions, the primary lesion is subacute chronic bronchitis that resembles chronic obstructive respiratory disease in humans.
• CRD in rats is a chronic inflammatory condition resulting in the hypersecretion and impaired clearance of mucus in which elevated levels and activation of macrophages and neutrophils play an important role.
• Once established in the lower respiratory tract, chronic bronchitis and bronchiolitis develop and progress to bronchiectasis and bronchiolectasis. Collections of mucus, leukocytes, and cellular debris accumulate in the lumen due to ciliostasis. There may be rupture of the bronchiolar walls, releasing inflammatory cells, mucus, and debris into the adjacent parenchyma, and developing pulmonary abscessation.
• As the airways become filled with mucus, bronchiolar lumen diameter decreases and a biofilm develops over bronchiolar epithelium, protecting secondary bacterial invaders from immune defenses and most antibiotics.
• M. pulmonis also causes an atrophic rhinitis in which the nasal turbinates become inflamed with a mixed pyogranulomatous infiltrate. The rhinitis accounts for the upper respiratory signs seen in CRD of rats. Because rats are obligate nose breathers, rhinitis results in open mouth breathing, hypoxia, and its associated metabolic disorders such as respiratory acidosis and myocyte irritability.
Diagnosis 
Initial Database
• Thoracic radiographs/CT: findings are consistent with bronchopneumonia, bronchitis, and/or atelectasis
• Skull radiographs/CT/MRI: tympanic bullae sclerosis or effusion if otitis media is present
• Complete blood count: may be normal or consistent with chronic inflammation (neutrophilia, monocytosis)
• Serologic testing: M. pulmonis, CAR bacillus, Sendai virus
• Bronchoalveolar lavage for PCR testing (M. pulmonis) and aerobic culture (for secondary bacterial pathogens). Culture for M. pulmonis requires special mycoplasma media.
Treatment 
Therapeutic Goal
Elimination of the disease is impossible. The goal of therapy is to improve the rat’s quality of life by controlling secondary bacterial infections and preventing acute dyspneic episodes.
Chronic Treatment
• Antibiotic therapy will not eliminate the pathogen. Antibiotic selection ideally is based on culture and sensitivity results.
• Doxycycline 5-10 mg/kg PO q 12 h: preferred antibiotic because it has additional antiinflammatory properties and is secreted by respiratory epithelial cells
• Enrofloxacin 10-20 mg/kg PO, IM, SC q 24 h: CAUTION with SC or IM injection as can cause severe pain and tissue necrosis. Dilute with sterile saline before injection.
• Tylosin 10 mg/kg PO, SC IM q 12-24 h: not recommended as use in drinking water because it may reduce water consumption
• Azithromycin 15-30 mg/kg PO q 24 h
• Nutritional support as animals may lose weight with chronic disease
Prognosis and Outcome 
Because many factors contribute to rat respiratory issues, the disease cannot be eliminated, but clinical signs may be ameliorated with antibiotics and supportive care.
Controversy
• Use of corticosteroids as antiinflammatory agents has been recommended to decrease the inflammation. Most (experimental) studies in rats have found steroids do not affect signs, function, and indices of inflammation. There is also significant concern that corticosteroid efficacy will be accompanied by consequential impairment of the rat’s immune defenses leading to fatal pulmonary abscessation and/or pneumonia.
• Use of bronchodilators (both oral and inhaled) has been recommended because these agents are helpful in humans with chronic bronchitis. Specific studies with bronchodilators have not been performed in rats with chronic respiratory disease, but they may be helpful.
• Nebulized hypertonic saline solution (7%) has been used successfully in humans with cystic fibrosis as a mucolytic agent. It breaks down the mucous biofilm and gives relief for ~8 hours.
• Concurrent nebulization with bronchodialators and/or antibiotics has been recommended to directly deliver medications. Specific studies using these nebulizations have not been performed in rats with chronic respiratory disease but may be helpful.
Pearls & Considerations 
Prevention
This disease complex (M. pulmonis) is thought to be ubiquitous in pet rats; thus prevention of infection is nearly impossible. Preventing contributing factors such as proper ventilation, bedding, and diet and decreasing stress can be helpful.
Client Education
All clients who own rats should be educated about the ubiquitous nature of M. pulmonis and the importance of ventilation, low cage ammonia levels, avoidance of dusty bedding such as wood shavings, and appropriate nutrition in decreasing the potential severity of chronic respiratory disease in rats.
Deeb, B. Respiratory disease in pet rats. Exotic DVM. 2005; 7:31–33.
Donnelly, TM. Application of laboratory animal immunoassays to exotic pet practice. Exotic DVM. 2006; 8:19–26.
Rempe, S, et al. Tetracyclines and pulmonary inflammation. Endocr Metab Immune Disord Drug Targets. 2007; 7:232–236.
Schoeb, TR, et al. Effects of viral and mycoplasmal infections, ammonia exposure, vitamin A deficiency, host age, and organism strain on adherence of Mycoplasma pulmonis in cultured rat tracheas. Lab Anim Sci. 1993; 43:417–424.
Wark, P, et al. Nebulised hypertonic saline for cystic fibrosis. Cochrane Database Syst Rev. 2009. [CD001506].
Skin Diseases
Basic Information 
Synonyms
Dermatitis, pyoderma, ulcerative dermatitis, ringworm, dermatophytosis, acariasis, ring tail, abscesses, bite wounds
Etiology and Pathophysiology
• Bacterial dermatitis/ulcerative dermatitis
 Usually secondary to self-trauma due to pruritus from mites or pruritus/pain over skin of salivary glands during sialodacryoadenitis (SDA) virus infection; dermatophytosis, fight wounds
Usually secondary to self-trauma due to pruritus from mites or pruritus/pain over skin of salivary glands during sialodacryoadenitis (SDA) virus infection; dermatophytosis, fight wounds
 All ectoparasitic infections can be complicated by secondary infections and self-mutilation. These secondary complications need to be identified and treated.
All ectoparasitic infections can be complicated by secondary infections and self-mutilation. These secondary complications need to be identified and treated.
 Rat fur mite (Radfordia ensifera): common; mild infestation produces few ill effects, but heavy infestation causes pruritus, leading to self-traumatization, and ulcerative dermatitis. Transmission is by direct contact.
Rat fur mite (Radfordia ensifera): common; mild infestation produces few ill effects, but heavy infestation causes pruritus, leading to self-traumatization, and ulcerative dermatitis. Transmission is by direct contact.
 Sarcoptic mites (e.g., Sarcoptes scabiei, Sarcoptes anacanthos, Trixacarus diversus): less common. Transmission is by direct contact. Leads to pruritus, crusting, and hyperkeratosis. Animals with clinical signs are often immune compromised.
Sarcoptic mites (e.g., Sarcoptes scabiei, Sarcoptes anacanthos, Trixacarus diversus): less common. Transmission is by direct contact. Leads to pruritus, crusting, and hyperkeratosis. Animals with clinical signs are often immune compromised.
 Notoedres muris: causes typical papulous lesions on ear pinnae
Notoedres muris: causes typical papulous lesions on ear pinnae
 Tropical rat mite (Ornithonyssus bacoti): Blood sucking mite; opportunistic ectoparasite. It spends a relatively short time on a host (usually at night) and penetrates the skin for feeding only. Cause severe pruritus. Animals appear nervous, particular in evening hours and at night. Severe infestations can cause anemia, debilitation, and death.
Tropical rat mite (Ornithonyssus bacoti): Blood sucking mite; opportunistic ectoparasite. It spends a relatively short time on a host (usually at night) and penetrates the skin for feeding only. Cause severe pruritus. Animals appear nervous, particular in evening hours and at night. Severe infestations can cause anemia, debilitation, and death.
 Demodectic mites (Demodex ratti, Demodex norvegicus, Demodex ratticola): rare
Demodectic mites (Demodex ratti, Demodex norvegicus, Demodex ratticola): rare
 Lice (Polyplax serrata, Polyplax spinulosa): Common; blood sucking lice. Located mainly at neck, at shoulders, and over back; poor fur condition and pruritus, which leads to self-mutilation
Lice (Polyplax serrata, Polyplax spinulosa): Common; blood sucking lice. Located mainly at neck, at shoulders, and over back; poor fur condition and pruritus, which leads to self-mutilation
 Pinworms (Syphacia obvelata): perianal pruritus and tail base mutilation
Pinworms (Syphacia obvelata): perianal pruritus and tail base mutilation
 Microsporum spp., Trichophyton mentagrophytes
Microsporum spp., Trichophyton mentagrophytes
 Clinical signs vary: alopecia, erythema, dandruff formation. Animals usually are not pruritic, unless secondary bacterial infection present.
Clinical signs vary: alopecia, erythema, dandruff formation. Animals usually are not pruritic, unless secondary bacterial infection present.
 Immune deficiency or stress may be underlying cause in chronic cases.
Immune deficiency or stress may be underlying cause in chronic cases.
• Neoplasia: fibroadenoma of the mammary glands (most common), mammary adenocarcinoma, lymphoma, etc. (see Mammary and Pituitary Tumors)
 Occurs in young rats (7-19 days) and is characterized by dry skin and formation of annular constrictions, which might progress to swelling, and tissue necrosis. Autoamputation might occur.
Occurs in young rats (7-19 days) and is characterized by dry skin and formation of annular constrictions, which might progress to swelling, and tissue necrosis. Autoamputation might occur.
 Low environmental relative humidity (less than 20%-40%) appears to be the cause; it is more often seen in rats housed in hanging cages and is rarely seen in pet rats.
Low environmental relative humidity (less than 20%-40%) appears to be the cause; it is more often seen in rats housed in hanging cages and is rarely seen in pet rats.
Diagnosis 
Differential Diagnosis
• Alopecia: trauma, dermatophytosis, chronic kidney disease, nutritional deficiency (low protein), neoplasia, barbering (behavorial)
• Ulcerative and crusting lesions: self-trauma, due to mites, secondary bacterial infections, fight wounds, neoplasia
• Pruritus: mites, secondary bacterial infections
• Crusting or flaking of skin: dermatophytosis, mites, nutritional deficiencies
• Cutaneous masses: neoplasia, inflammation, abscesses
• Localized erythema or pododermatitis: contact allergy, contact irritation (cleaners), trauma from bedding/cage material
Treatment 
Acute General Treatment
• If animal is self-mutilating: shorten and blunt nail tips. In severe case, temporarily apply bandages to hindfeet. Apply E-collar to prevent removal of bandages.
 Ivermectin 0.2-0.4 mg/kg SC, PO q 7-14 d
Ivermectin 0.2-0.4 mg/kg SC, PO q 7-14 d
 Selamectin 10-25 mg/kg topically q 21-28 d
Selamectin 10-25 mg/kg topically q 21-28 d
 Treat until clinical signs are resolved and no more parasites are found on the animals.
Treat until clinical signs are resolved and no more parasites are found on the animals.
 Treat the environment to prevent reinfection: regular bedding changes and cage cleaning. Discard cage furnishing that cannot be disinfected (e.g., wood-based furnishing).
Treat the environment to prevent reinfection: regular bedding changes and cage cleaning. Discard cage furnishing that cannot be disinfected (e.g., wood-based furnishing).
• Bacterial dermatitis/ulcerative dermatitis
 If indicated, provide systemic antibiotic therapy based on culture and sensitivity whenever possible
If indicated, provide systemic antibiotic therapy based on culture and sensitivity whenever possible
 Lance, débride, and flush or remove in toto if possible.
Lance, débride, and flush or remove in toto if possible.
 If indicated, provide systemic antibiotic therapy based on culture and sensitivity whenever possible.
If indicated, provide systemic antibiotic therapy based on culture and sensitivity whenever possible.
 Enilconazole (1 : 50, emulsion as spray or moist wipe)
Enilconazole (1 : 50, emulsion as spray or moist wipe)
 Miconazole/chlorhexidine shampoos
Miconazole/chlorhexidine shampoos
 Lime sulfur dips (1 : 40, q 7 d)
Lime sulfur dips (1 : 40, q 7 d)
 Used alone or in combination with systemic therapy
Used alone or in combination with systemic therapy
 Used preferably in cases of suspected dermatophytosis, while dermatophyte culture results are awaited
Used preferably in cases of suspected dermatophytosis, while dermatophyte culture results are awaited
 Environmental decontamination: frequent damp mopping of hard surfaces rather than sweeping can reduce environmental spread of spores; 1 : 10 bleach solution can be used to clean environment. Contact time: 10 minutes
Environmental decontamination: frequent damp mopping of hard surfaces rather than sweeping can reduce environmental spread of spores; 1 : 10 bleach solution can be used to clean environment. Contact time: 10 minutes
• Monitoring: once-weekly dermatophyte test medium (DTM) cultures. Discontinue treatment when two consecutive negative cultures are obtained.
• Antiinflammatory drugs: meloxicam 0.3-0.5 mg/kg PO, SC q 12-24 h
• Neoplasia: surgical mass removal (see Mammary and Pituitary Tumors)
• Nutritional deficiency: improve diet; provide access to commercial pelleted diet
Agren, MS, et al. Effect of topical zinc oxide on bacterial growth and inflammation in full-thickness skin wounds in normal and diabetic rats. Eur J Surg. 1991; 157:97–101.
Galler, JR, et al. Ulcerative dermatitis in rats with over fifteen generations of protein malnutrition. Br J Nutr. 1979; 41:611–618.
Honma, M, et al. Plantar decubitus ulcers in rats and rabbits. Jikken Dobutsu. 1989; 38:253–258.
Taylor, DK, et al. Lanolin as a treatment option for ringtail in transgenic rats. J Am Assoc Lab Anim Sci. 2006; 45:83–87.
Cross-References to Other Sections
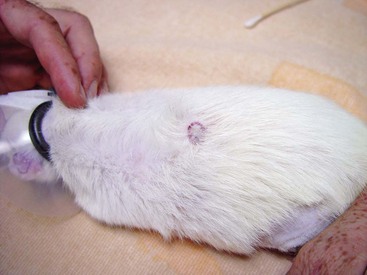
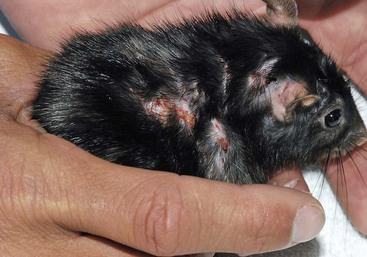
Skin Disease This skin lesion in a rat was caused by a subcutaneous injection of enrofloxacin; always dilute the drug if it needs to be injected SC or IM. (Photo courtesy Jörg Mayer, The University of Georgia, Athens.)