Fasciae of the Thorax and Abdomen
Introduction
In the abdomen is the thickest superficial fascia of the body known as ‘Scarpa's fascia’. The superficial fascia is present throughout the trunk as a continuous fibroelastic layer. The deep fascia is arranged into three laminae: superficial, intermediate and deep. Considering that the deep fascia in the thorax is very thin and adherent to the muscles, whilst the superficial fascia is thick and strong, the superficial fascia is often confused with the deep fascia. In this chapter the following will be discussed: the classification and continuation of both the superficial and deep fascia from the thorax into the neck, abdomen and lumbar areas (Fig. 5.1).
Superficial Fascia of the Thorax and Abdomen
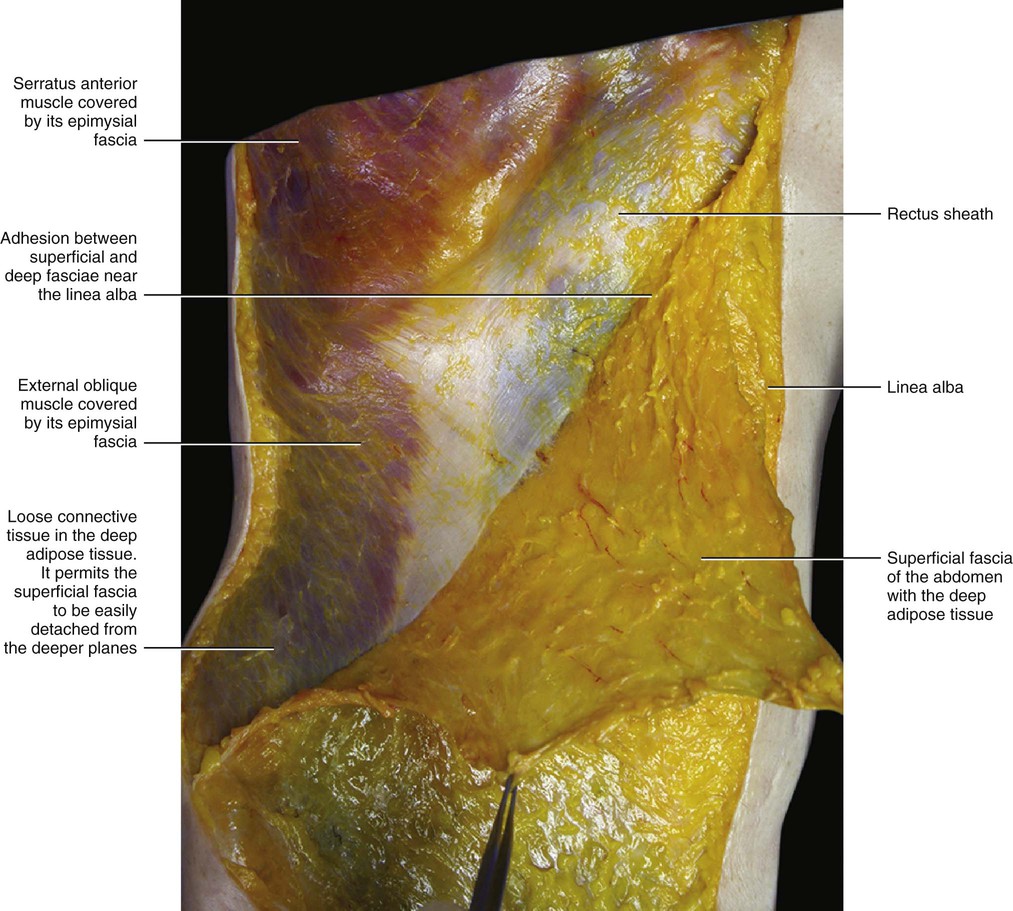
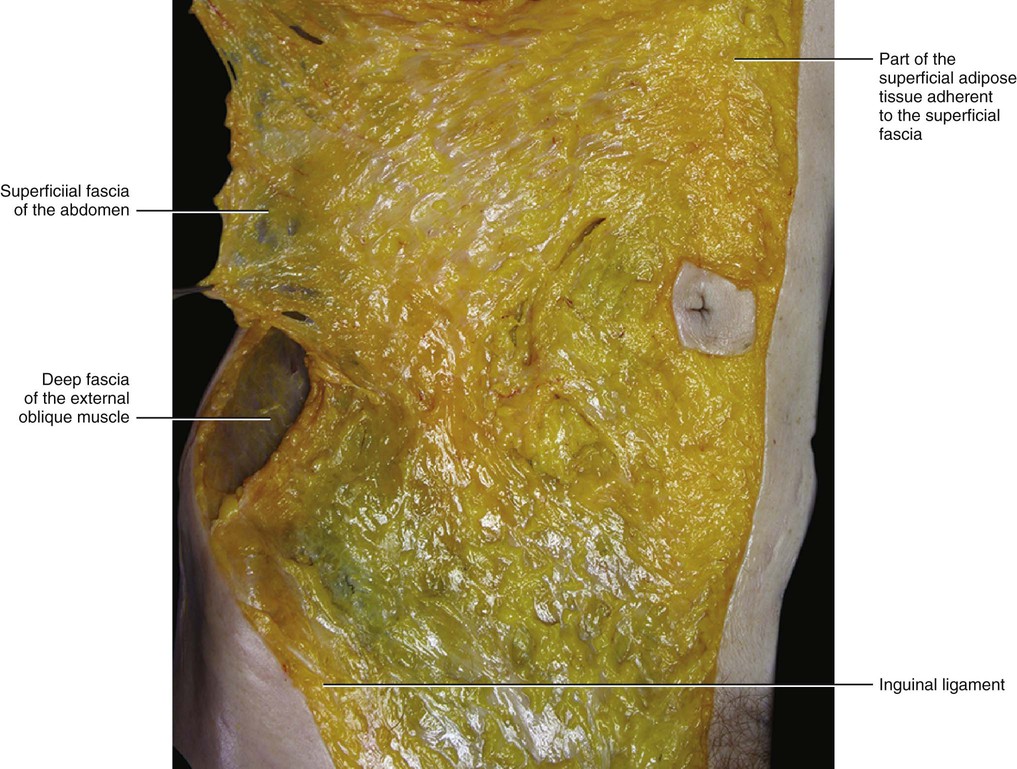
In the thorax and abdomen, the superficial fascia is easy to identify. It divides the subcutaneous tissue into two sublayers: the superficial adipose tissue (SAT) and the deep adipose tissue (DAT), and each has its characteristic features. The superficial fascia is a fibroelastic layer mainly composed of collagen and elastic fibres; however, small fat lobules are often observed between the fibres (Fig. 5.2). While macroscopically this fascia appears to be, and can be isolated as, a well-defined membrane (Fig. 5.3), microscopically its structure is better described as lamellar or highly packed and honeycomb like. The superficial fascia can be followed as a dissection plane from the thorax to the inguinal ligament, where it adheres to the deeper planes. It appears to have total continuity with the superficial fascia of the thigh.
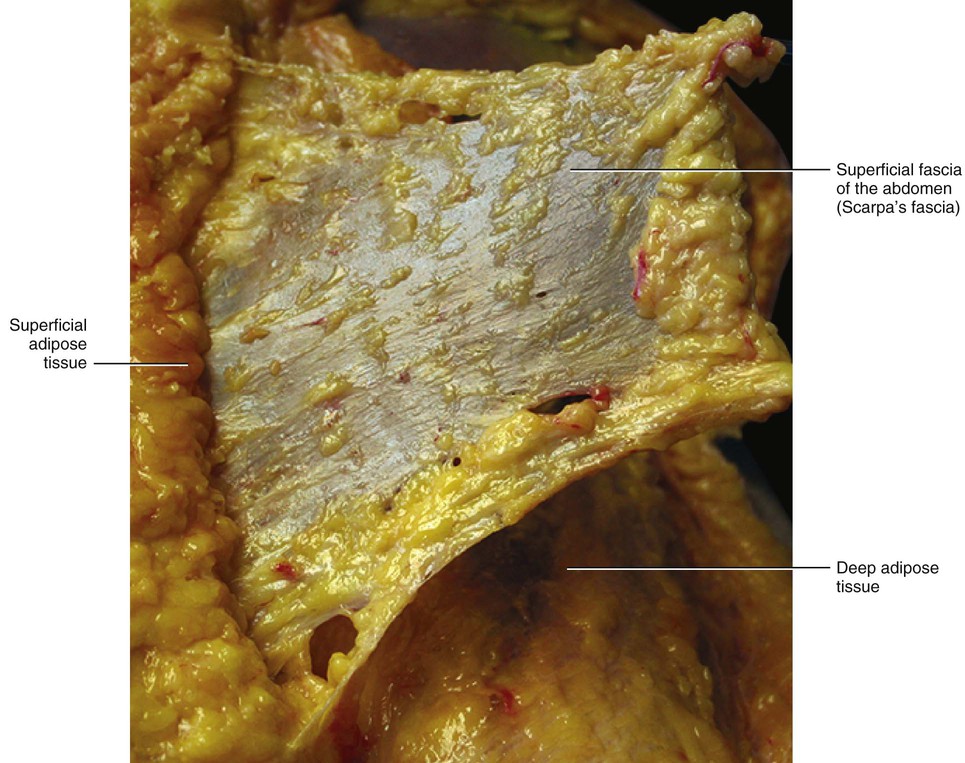
The superficial fascia does not have a uniform thickness. It is a well-defined, white layer in the lower abdomen where it is called Scarpa's fascia. It thickens towards the inguinal ligament where a multilayered structure of collagen bundles, extending in different directions, can be perceived both during dissection and in transparency (when a light goes through it). In the lower abdomen of cadavers, the mean resistance to traction of the isolated superficial fascia is 2.8 kg in a transverse direction and 5.5 kg in a craniocaudal direction. This difference demonstrates that the superficial fascia has specific spatial reinforcements rather than being considered a homogeneous tissue. In the upper abdomen, the superficial fascia is much thinner, appearing as a translucid collagen layer through which adipose tissue can be seen. Distally, in males it continues over the penis and spermatic cord to the scrotum, where it helps to form the dartos fascia. The dartos muscle is enveloped by the superficial fascia and in the same manner as the platysma muscle and the fundiform ligament. From the scrotum, it can be traced backwards and is continuous with the superficial fascia of the perineum (fascia of Colles).
According to Sterzi (1910), there are fibrous thickenings inside the superficial fascia of the trunk. These collagen fibrous bundles originate in the back and extend in an oblique direction to the anterior region of the trunk and in a craniocaudal direction. The distribution of these reinforcements of the superficial fascia corresponds to the lines in the dermis described by Langer in 1862.
Occasionally, inside the superficial fascia of the trunk some striated muscle fibres are found. According to Tobler (1902) and Ruge (1905), these muscular fibres inside the superficial fascia are the remains of the panniculus carnosus that is present in all superficial fasciae of mammals. To support this idea, they found well-represented axillary arches in gorillas and monkeys, which also have a large panniculus carnosus. The axillary arch can also be present in human axilla as an anatomical variation, where it can be considered as a possible cause of nerve or vascular compression.
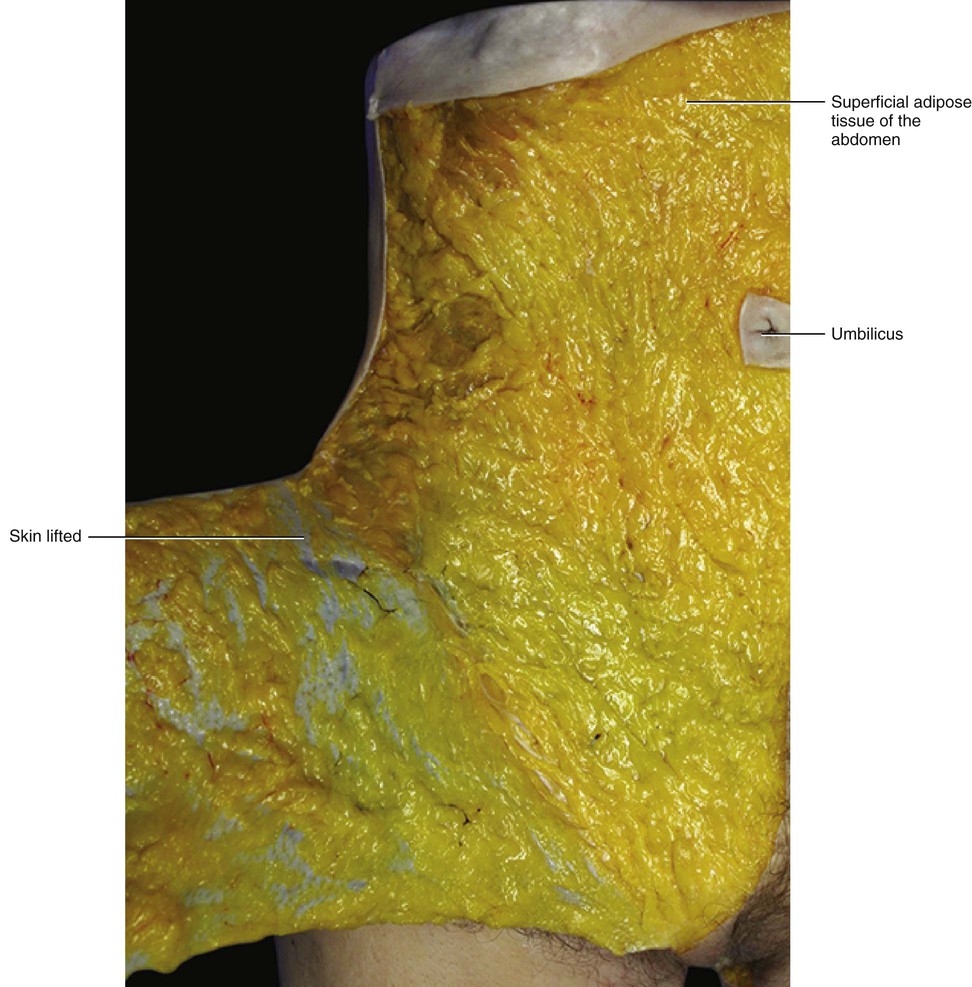
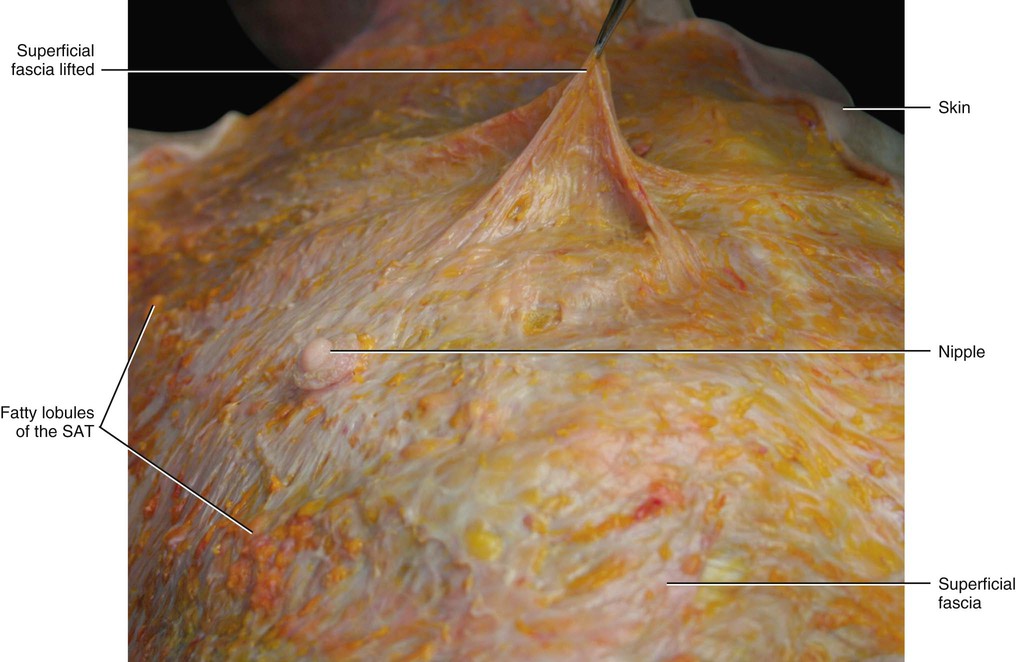
The SAT is typically well represented in both the thorax and the abdomen and shows nearly constant characteristics (Fig. 5.4). The fibrous septa, forming the retinaculum cutis superficialis, generally assume an arrangement perpendicular to the surface. The adipose lobules have an oval shape with their major axis perpendicular to the skin, and they are generally disposed in a single layer. This structure has high-structural stability, mechanical resilience and elastic properties. In particular, if a weight of 1 kg is placed over slices of the flap and then removed, the fat lobules return to their original position and shape. With the augmentation of fat deposition, the fat lobules increase in size and in the lower abdomen they assume a multilayered disposition. This causes a lengthening and thickening of the retinacula cutis, which assume a more oblique orientation forming a second fibrofatty layer parallel to the superficial fascia. This finding could explain why some authors describe an additional fibrofatty fascial layer in the subcutis of the abdomen (called ‘Camper’s fascia’). However, our dissections and imaging studies demonstrate that only one layer of superficial fascia is present; this was also recently confirmed by Chopra et al (2011) in their study of living subjects. These authors, using CT scans of the abdominal region, showed that a membranous layer (corresponding to the superficial fascia based on our findings) is present throughout the anterior abdominal wall. This membranous layer divides the subcutis into three layers: superficial fatty layer (the SAT, based on our findings), intermediate membranous layer (superficial fascia), and deep fatty layer (the DAT, based on our findings). If the membranous layer is not clear on a CT scan, this could be due to the absence of fat deposited in the deep compartment.
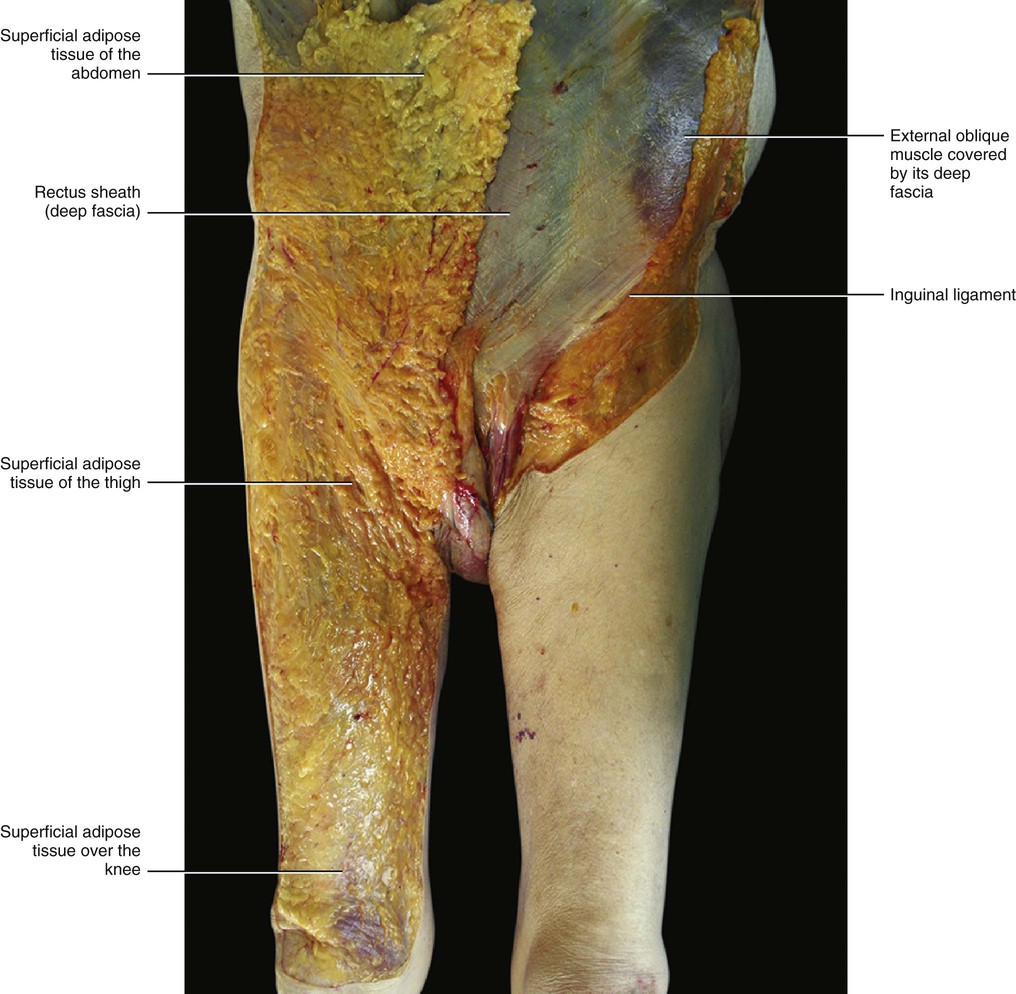
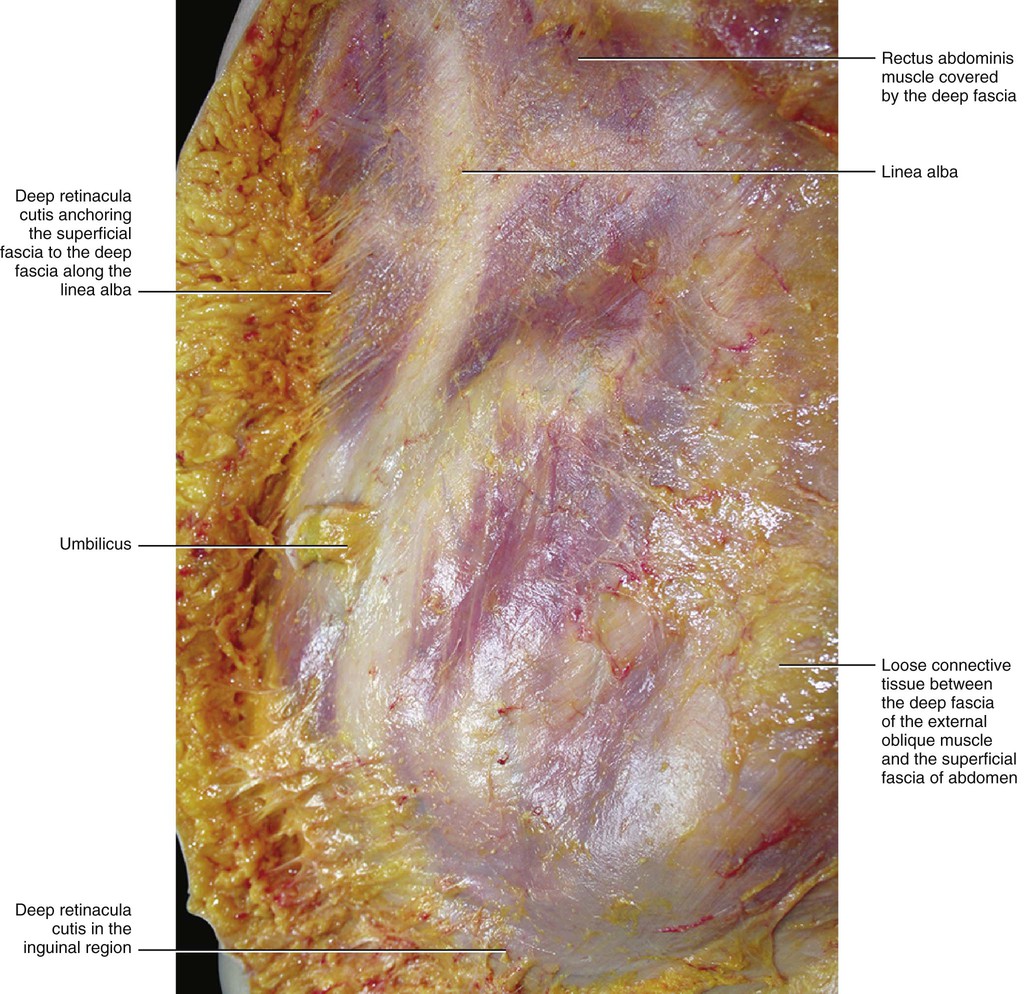
The SAT continues, with a similarly structured layer, over the inguinal ligament into the thigh (Fig. 5.5). No clear border to this layer can be identified either caudally or cranially. This layer is separated from the contralateral layer at the level of the linea alba, where the superficial fascia is adherent to the skin and to the deep fascia.
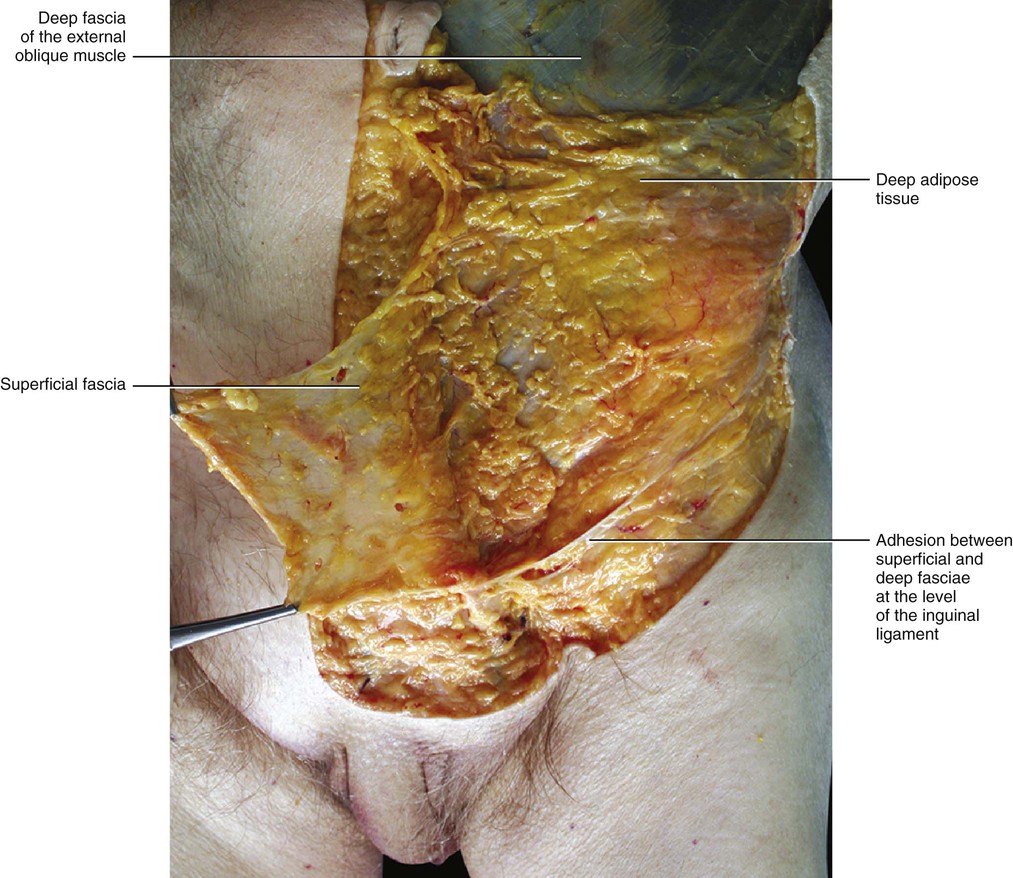
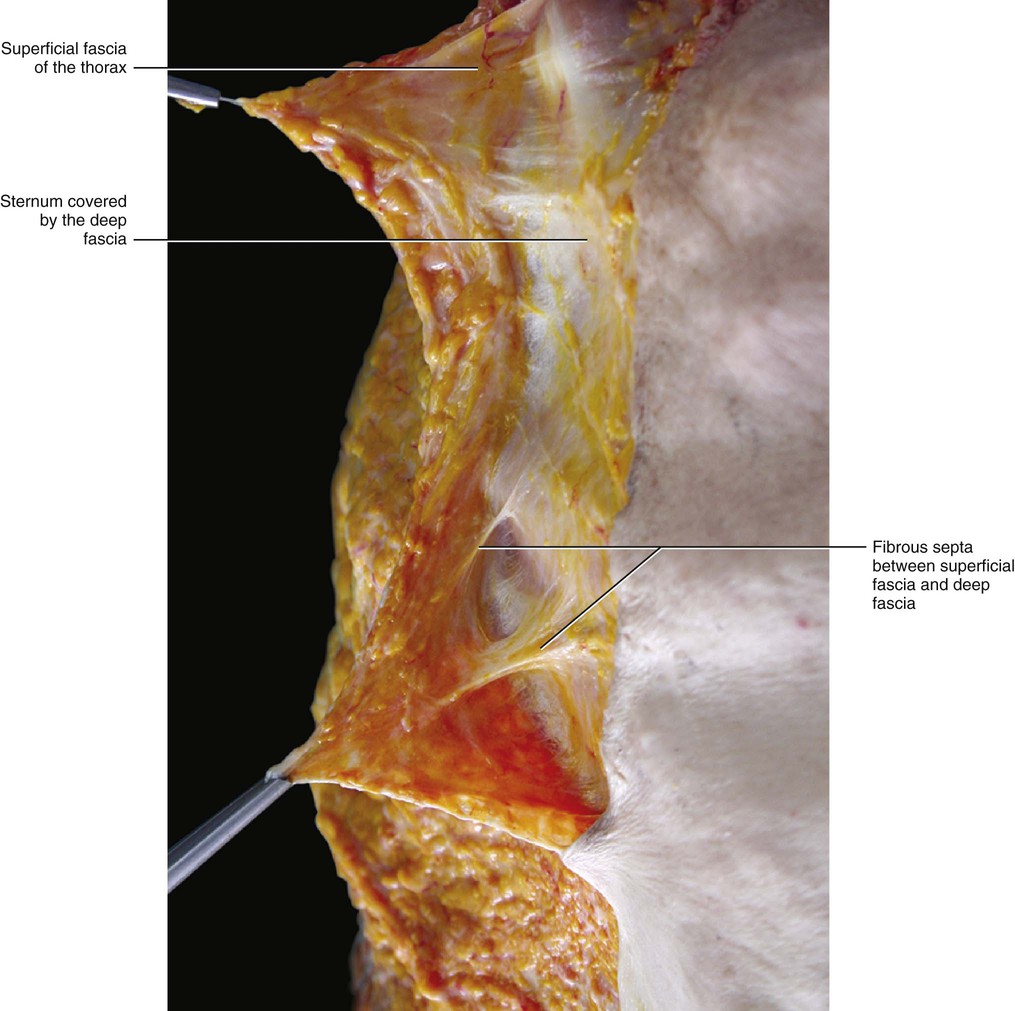
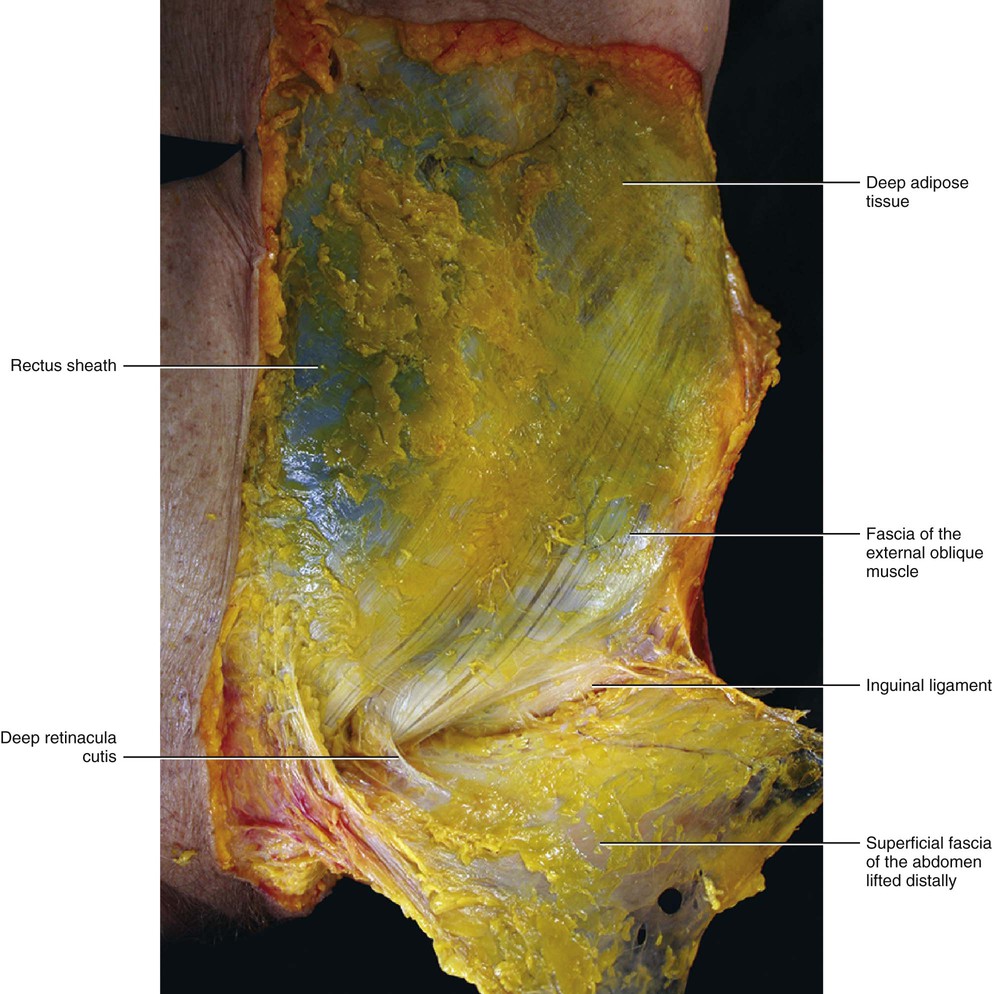
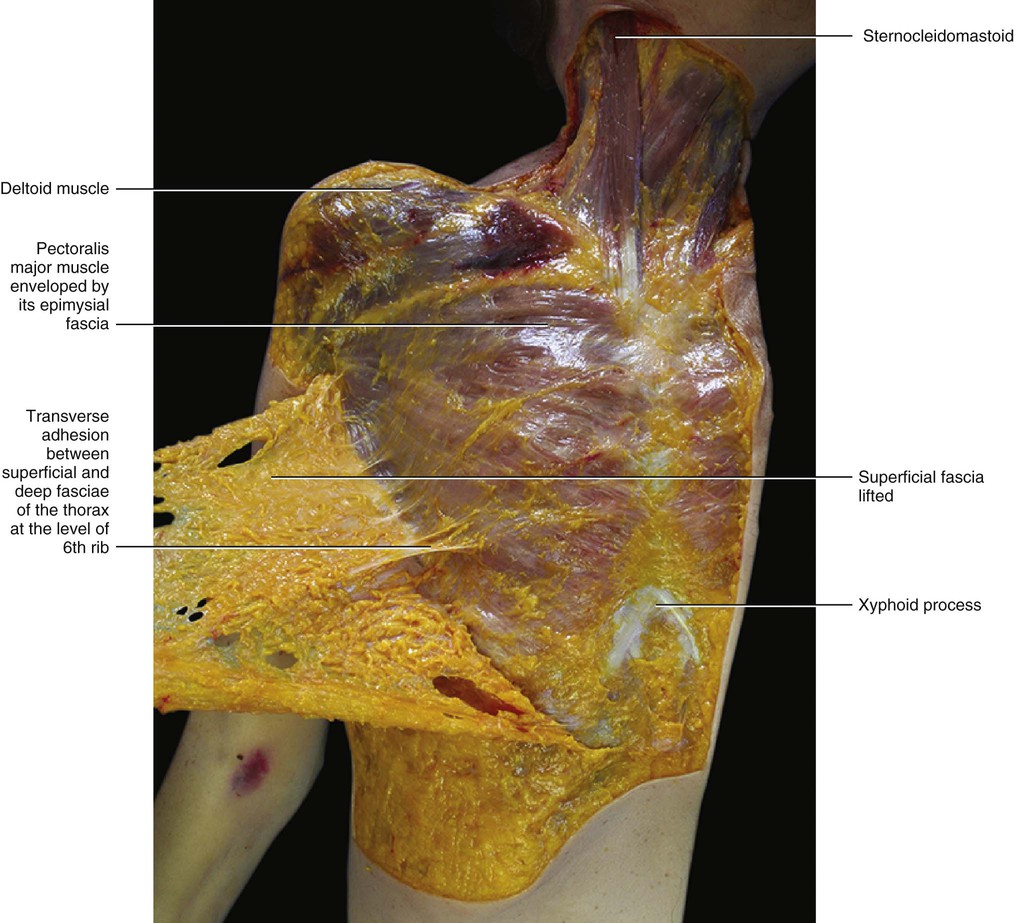
The deep compartment (DAT) appears to be very different from the SAT. It is usually thinner and formed mostly by loose connective tissue with a few fat cells (Fig. 5.6). The fat lobules are smaller, flatter and less defined, and the fibrous septa are less consistent and mostly orientated obliquely. The obliquity of the septa of the DAT, its adequate elastic properties and strength, and its lateral displacement of the fatty lobules creates a perfect plane for sliding of the subcutaneous tissue over the deep fascia. The DAT presents many variations in its structure and in the amount of fat depending on the particular area (Fig. 5.7). Along the sternum and the linea alba the DAT is absent and results in the superficial fascia adhering to the deep fascia (longitudinal adhesion, see Chapter 2). Transverse adhesions are also present between the two fasciae, in particular over the acromion and clavicle, at the level of the sixth rib and over the inguinal ligament (Figs 5.8–5.10). In this way, the DAT of the abdomen is completely divided from the DAT of the thigh, preventing infections or oedema passing from one region to the other. As no fatty tissue is embedded in the DAT of this region the inguinal fold is always easier to see, even in obese people. These adhesions also allow a division of the subcutis into cervical, thoracic and abdominal compartments. By understanding the various roles of the superficial fascia and subcutis, it may be hypothesized that the compartimentalization of the subcutis play an important role in lymphatic drainage and superficial venous return in this area.
Mammary Region
There is some confusion about the relationship between the superficial fascia and the mammary gland. For some authors the mammary gland is embedded within the superficial fascia, for others the superficial fascia is superficial to it, while others deny the existence of the superficial fascia (Beer et al 2002). Our dissections have revealed superficial fascia in the pectoral region of all subjects. This superficial fascia is a continuation of the superficial fascia of the neck, and the muscular fibres of the platysma muscle extend to the proximal third of the superficial fascia of the thoracic region (Fig. 5.11). The mammary gland at first appears to be enveloped by the superficial fascia. However, after careful dissection, it is evident that the fascia is always deeper respect to the mammary gland, and that the gland maintains its contact with the skin in the areola region (Figs 5.12 and 5.13). This organization is consistent with the embryologic development of the mammary gland, which is a cutaneous gland that has assumed a specific function. Our findings are confirmed by Sterzi (1910) who studied the superficial fascia of this region in cadavers of different ages. He found that in newborns the superficial fascia is deep with respect to the mammary gland, dividing the abundant SAT from a very thin DAT. In adult females, the superficial fascia adheres intimately to the inner side of the mammary gland. The retinacula cutis of the SAT divides the mammary gland into various lobules (Fig. 5.14). Inside the mammary gland, the fibrous septa of the superficial retinacula cutis are called Cooper's ligaments1 and they have the role of maintaining the structural integrity of the gland. Without the internal support of these septa, the breast tissue, which is heavier than the surrounding fat, sags under its own weight, and loses its normal shape and contours. Cooper’s ligaments play an important role in the changing appearance of the breast that often accompanies development of inflammatory carcinoma, in which blockage of the local lymphatic ducts causes swelling of the breast. As the skin remains tethered by these septa, it takes on a dimpled appearance reminiscent of an orange peel. Carcinomas can also decrease the length of Cooper's ligaments leading to a dimpling.
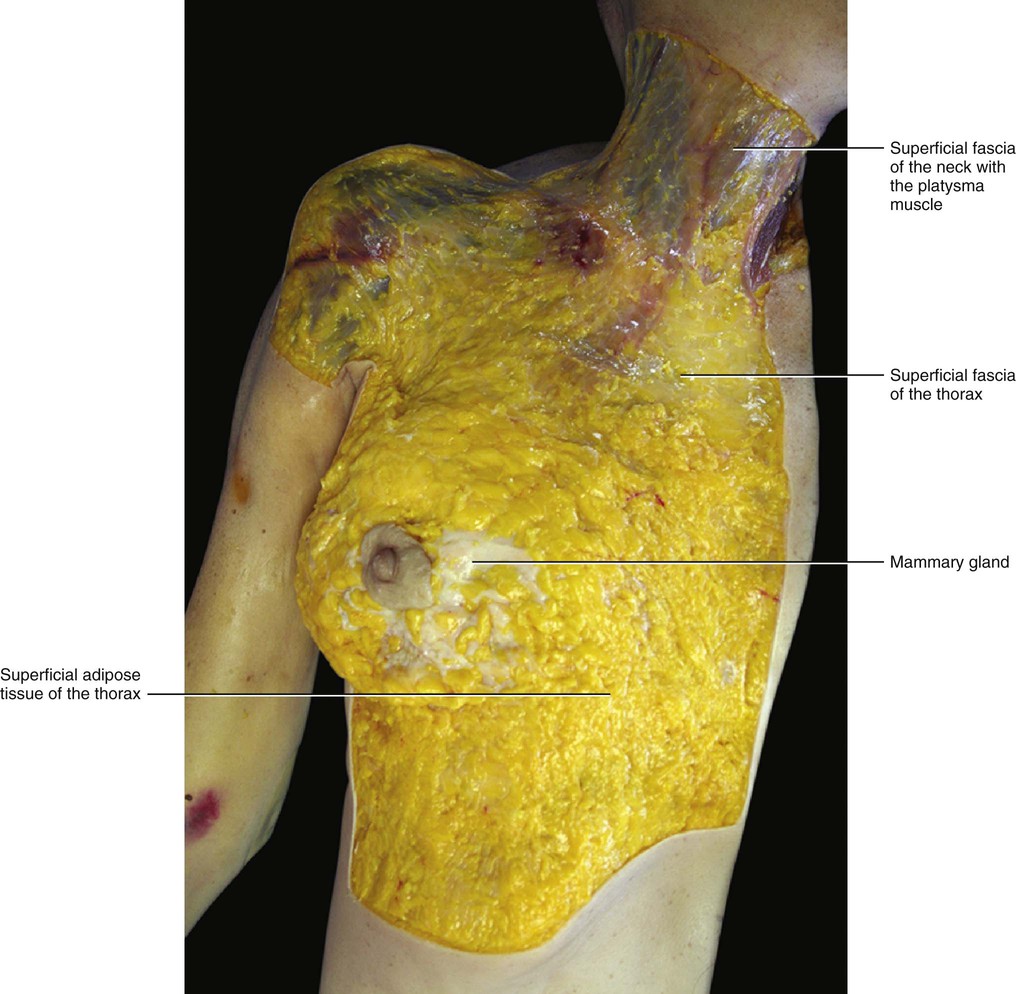
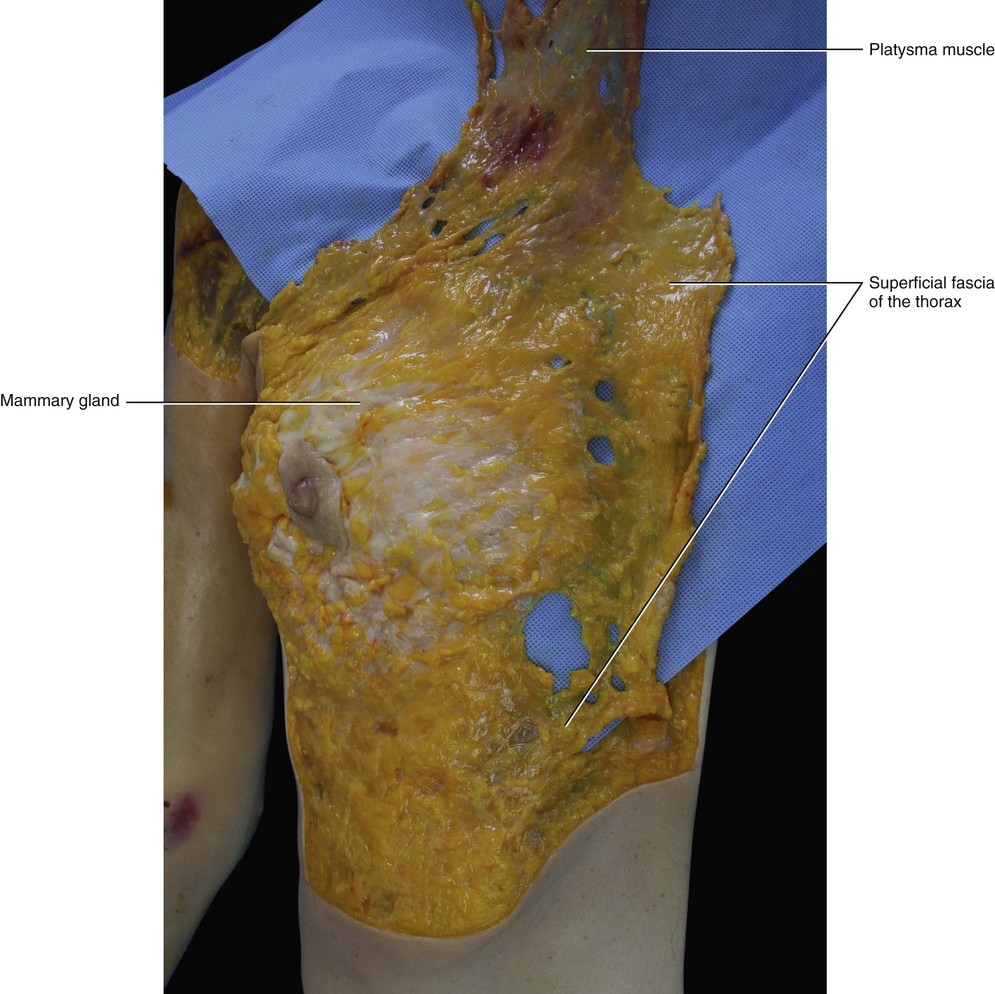
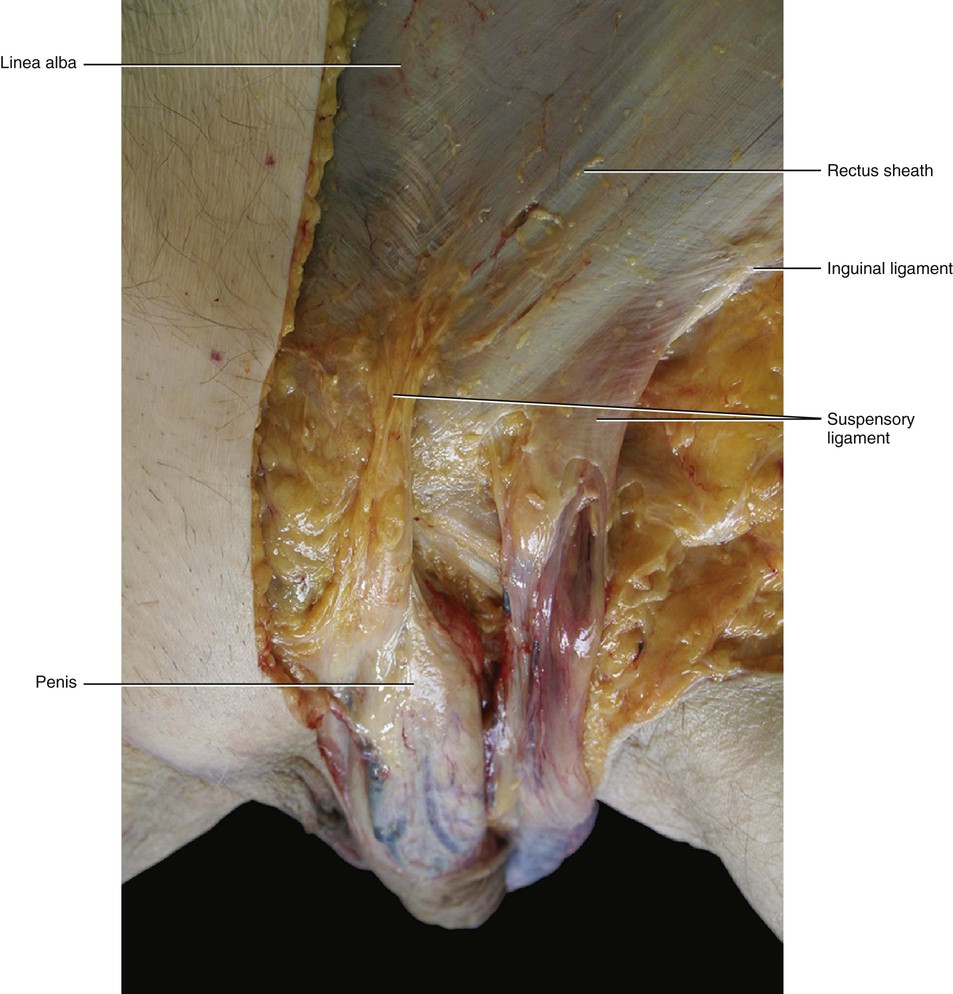
Between the clavicle and the mammary gland the superficial fascia is thicker, and this thickening forms the suspensory ligament of Giraldes (1851). This ligament is actually a reinforcement of the superficial fascia (rather than a true ligament), and its isolation is an artifact of the dissection. This ‘ligament’ has the effect of supporting the breast in its normal position and maintaining its normal shape, and it continues medially with the platysma muscle.
Between the superficial and deep fasciae of the thorax there is loose connective tissue that permits the movement of the mammary gland with respect to the underlying muscular plane. So, in the breast region the same organization of the subcutis can be identified as is present throughout the human body. These particular elements are: the presence of the mammary glands in the SAT and the major thickness of the superficial fascia with respect to the deep fascia.
Deep Fascia
The fascial anatomy of the trunk is more complex than that of the limbs. In the limbs, only one aponeurotic fascia connects the ipsodirectional motor units, while in the trunk there are three muscular planes with three fascial layers. Between these fasciae, loose connective tissue is present and permits the sliding between the various muscle bellies (Fig. 5.15). In some places these fasciae fuse, creating specific lines where all the muscular forces converge. The more important of these lines of fusion is the linea alba, but there are also fusions at the borders of the rectus sheath and over the sternum.
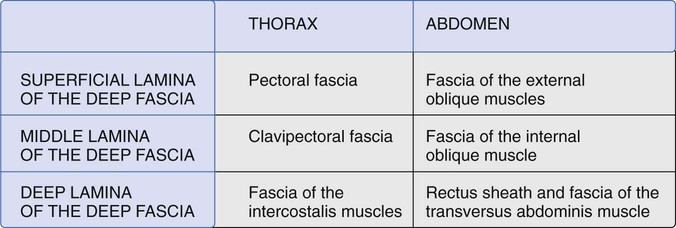
Each one of the three muscular layers (laminae) of the trunk have specific fascia with epimysial features. In particular, these fasciae are thin and adhere to the underlying muscles and to the flat tendons of these muscles. Thus, the anatomy and the function of these fascial layers cannot be separated from their respective muscles. Only two aponeurotic fasciae were identified in the trunk: the rectus sheath and the thoracolumbar fascia.
To understand the fascial anatomy of the trunk, it is useful to study their development. According to Skandalakis et al (2006), the deep, intermediate and superficial layers of deep fascia are produced in the embryo as muscular primordial, and originate from somites invading the somatopleura that penetrate the somatic wall connective tissue. This produces epimysial fascia on either side, from which arise the layers of investing fascia. Sato and Hashimoto (1984) report that the pectoralis major, latissimus dorsi and trapezius muscles form an additional myofascial layer with respect to the underlying muscular planes. Indeed, these muscles originate as muscles of the limbs, but then they grow inside the superficial lamina of the deep fasciae of the trunk just up to the connection with the midline (the interspinous processes of the spine in the back and with the sternum in the thorax). Thus an additional muscular plane in the trunk is formed. The development of these muscles in the trunk is probably determined by the necessity to connect firmly the upper and lower limbs to the trunk. The superficial lamina of the deep fascia, enveloping all these muscles, serves to reinforce these connections. The connections are spatially organized and very specific; for example, the expansions of the pectoralis major muscles into the rectus abdominis sheaths connect the muscles of the thorax and abdomen on both sides. The superficial layer of the pectoral fascia passes over the sternum to connect the two pectoralis major muscles.
We can identify three myofascial laminae in the trunk (superficial, middle and deep) (Fig. 5.16). The superficial lamina is formed by a fibrous layer, which is thicker at the level of the neck but becomes thinner as it descends into the abdomen. This fascia envelops all the large muscles of the trunk that work in spiral/rotational movements and connect the trunk with the limbs. At the cervical level it surrounds the sternocleidomastoid and trapezii muscles. At the level of the thorax it surrounds the pectoralis major and the latissimus dorsi muscles. At the level of the abdomen it surrounds the external oblique muscles and covers the rectus sheath on the anterior part. The middle lamina is formed by the infrahyoid, subclavius, pectoralis minor, serratus anterior and internal oblique muscles. These muscles primarily move in the frontal and horizontal planes. This middle layer of the fasciae of the trunk connects and coordinates all of them. The deep lamina is formed by the scalenes, intercostal, rectus abdominis, pyramidalis and transversus abdominis muscles. Most of the muscles in this plane have a longitudinal disposition of their muscular fibres and move primarily in the longitudinal plane. The transversus abdominis muscle and its fascia connect the rectus sheath with the iliopsoas, quadratus lumborum muscles and anterior layer of the thoracolumbar fascia. So, the transversus abdominis muscle becomes the joining element between the anterior and posterior muscular–fascial elements.
Pectoral Fascia
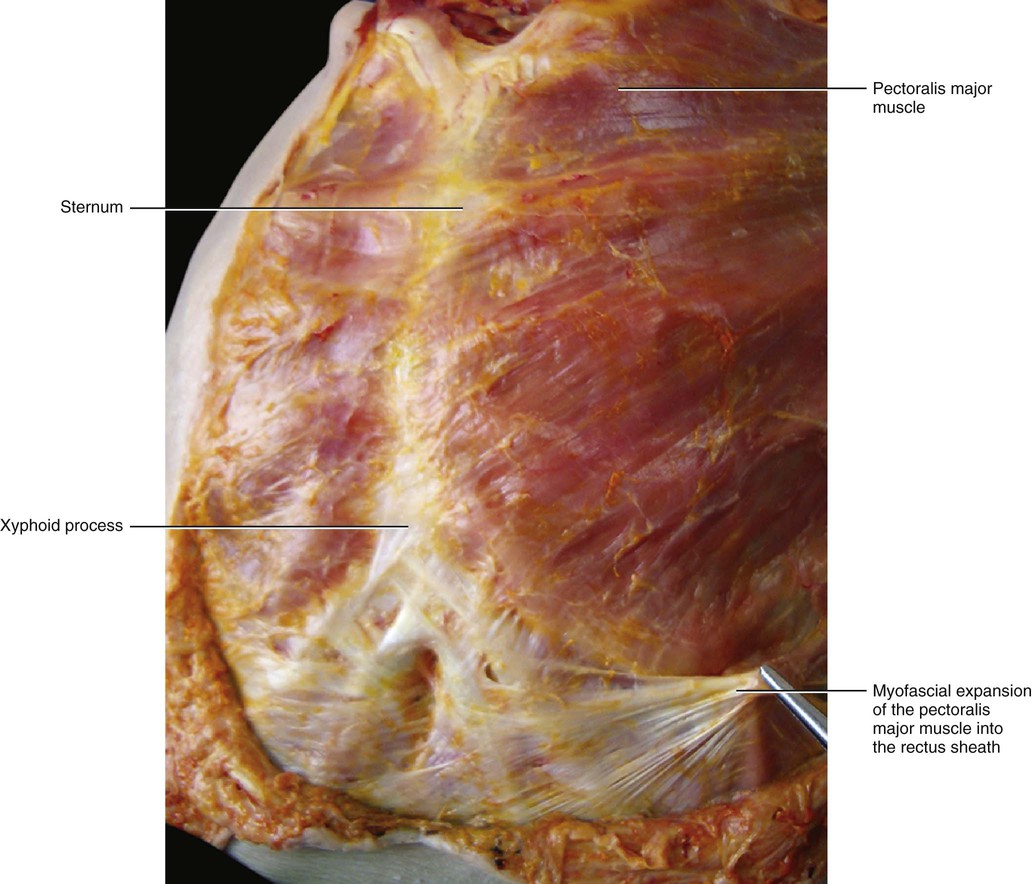
The fascia of the pectoralis major muscle is a thin fibro-elastic layer (mean thickness 151 μm ± 37) firmly connected to the underlying muscle by many intramuscular septa. They originate from its inner surface and penetrate between the muscular fibres, dividing the muscle itself into many bundles (Fig. 5.17). Many muscular fibres insert directly onto the pectoral fascia itself. The pectoral fascia originates from the clavicle and then divides into two layers to envelop the pectoralis major muscle. Proximally, only the deep layer of the pectoral fascia adheres to the clavicular periosteum. Its superficial layer continues with the superficial lamina of the deep cervical fascia, which surrounds the sternocleidomastoid muscle. Laterally, the pectoral fascia continues with the deltoid fascia and with the axillary fascia, extending as a fibrous expansion into the brachial fascia. The deltoid fascia envelops the deltoid muscle, just like the pectoral fascia does with the pectoralis major muscle. Over the serratus anterior, the two layers of the pectoral fascia adhere to form a single fascial lamina (Fig. 5.18) that some authors call the anterolateral thoracic fascia (Sebastien at al 1993). Posteriorly, this single layer divides itself again to enclose the latissimus dorsi muscle in the same manner as the pectoral fascia encloses the pectoralis major muscle. Medially, the deep layer of the pectoral fascia inserts into the periosteum of the sternum, while the superficial layer extends beyond the sternum to continue with the pectoral fascia on the other side (Figs 5.19 and 5.20). Distally, the pectoral fascia continues with some fibrous expansions into the rectus abdominis sheath and into the fascia of the controlateral external oblique muscle. Over the xyphoid process a criss-cross interweaving of fibres is clearly visible (Fig. 5.21).
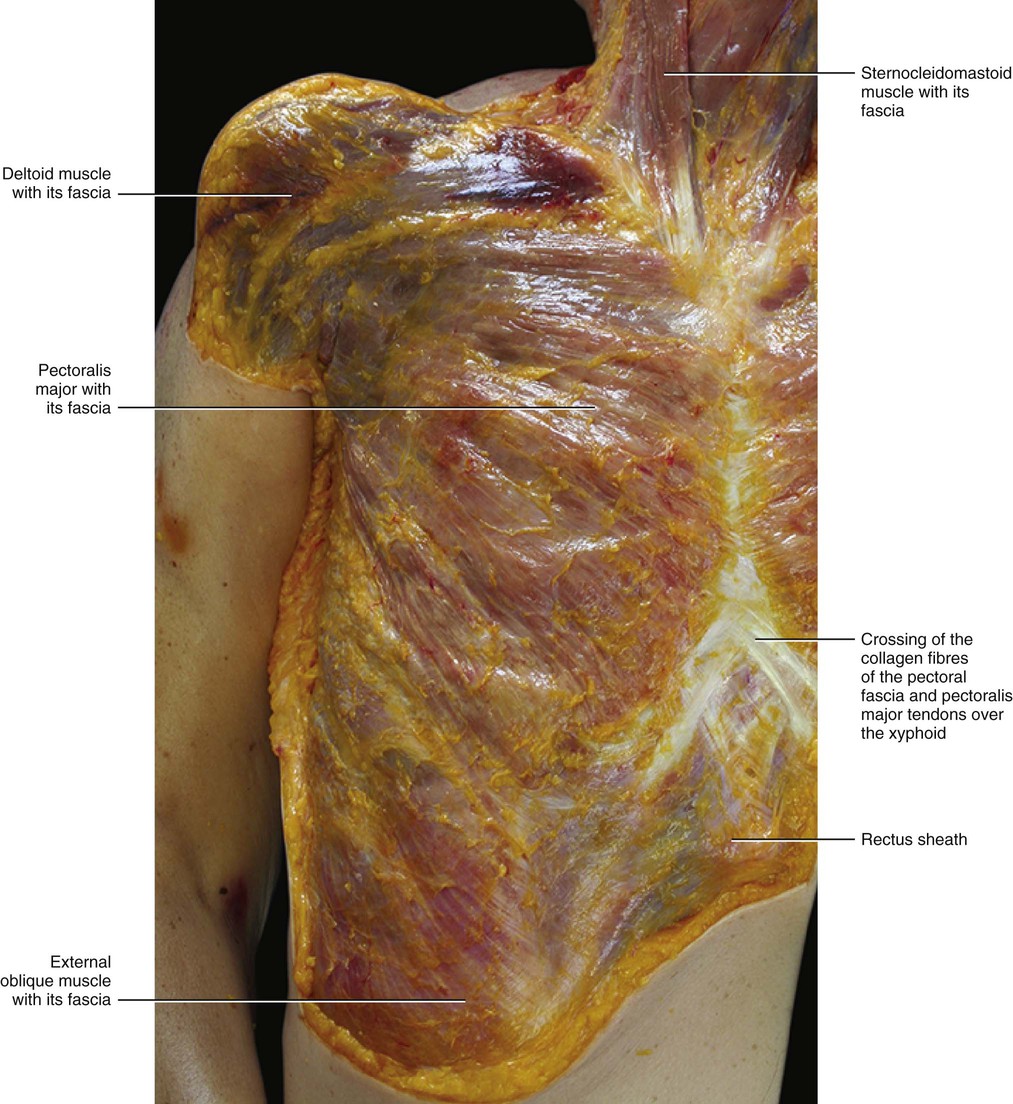
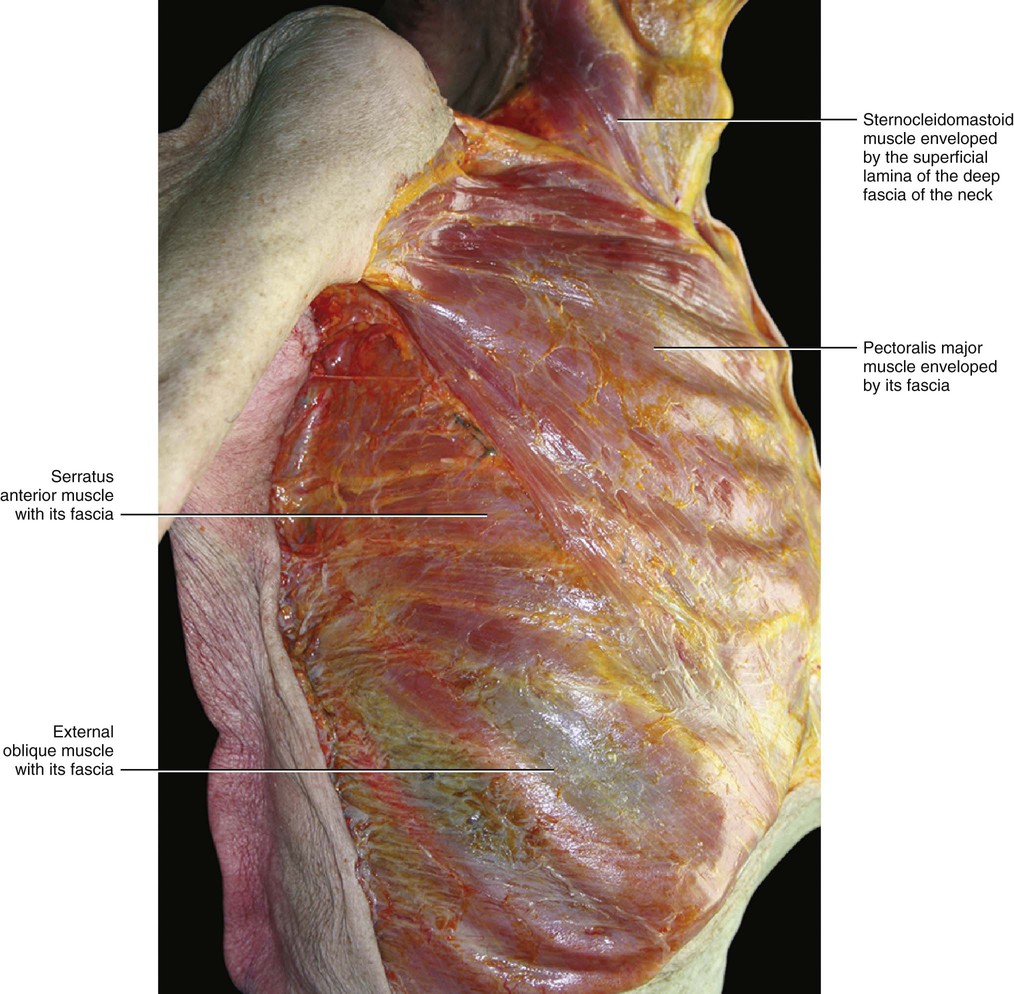
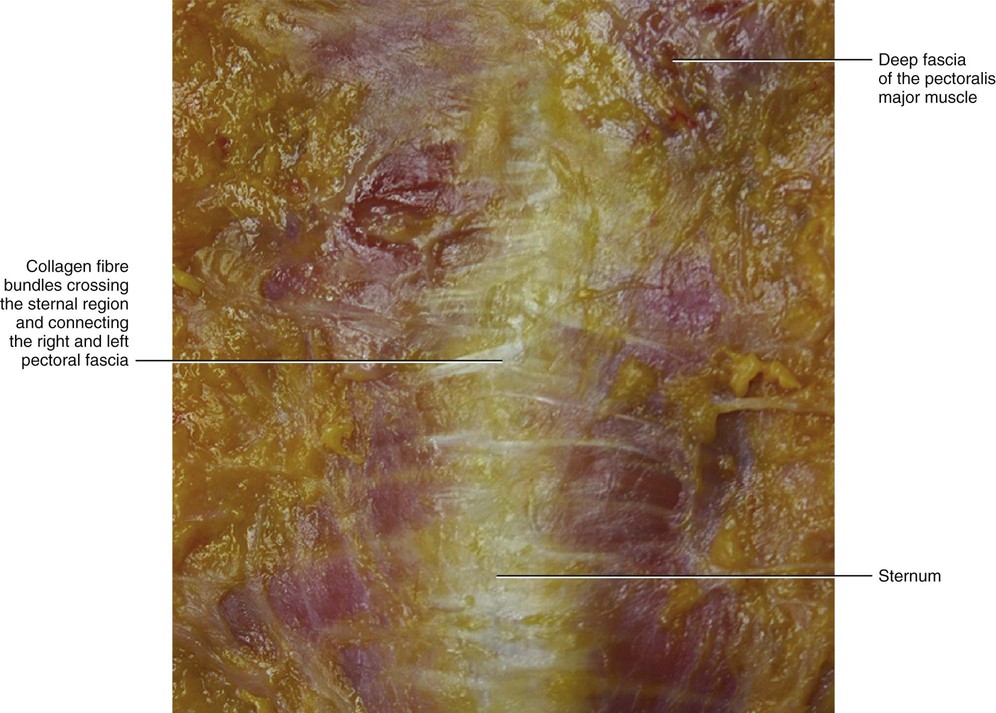
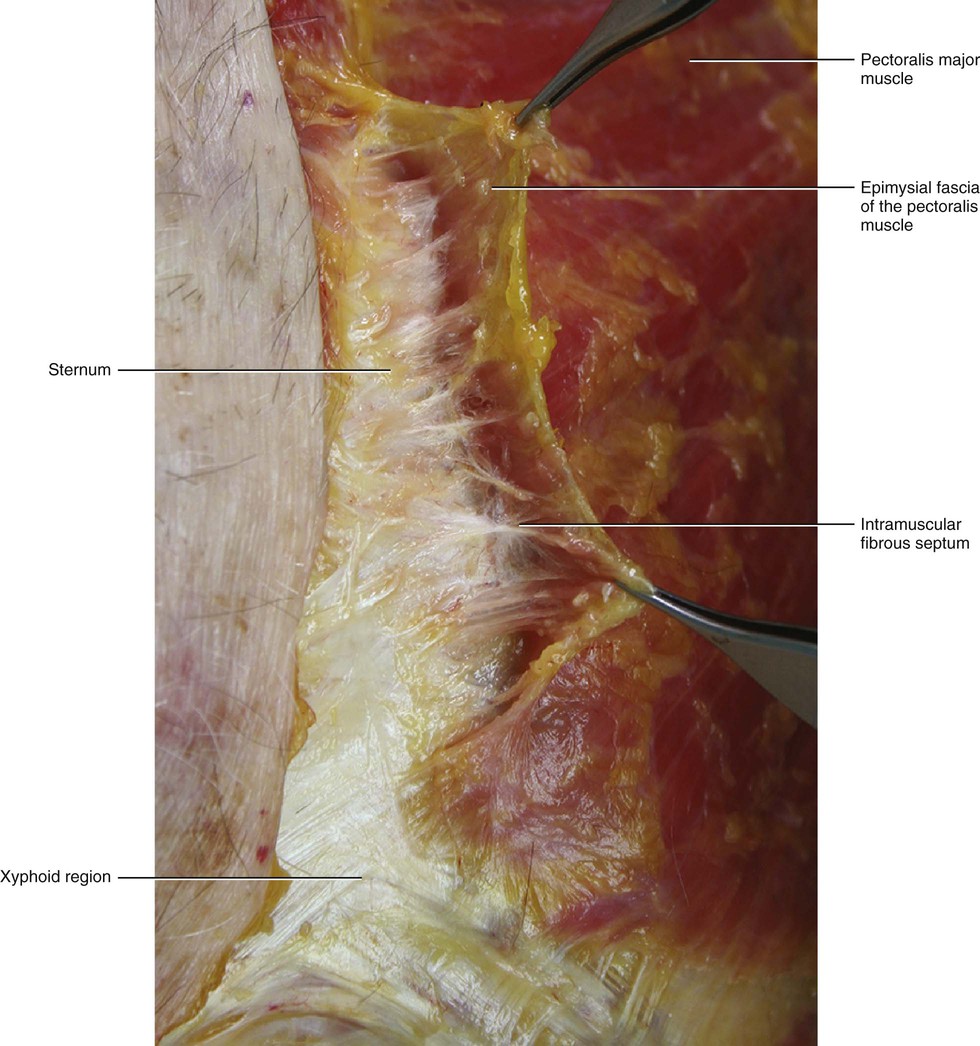
Histologically, the pectoral fascia appears to be formed by undulated collagen and elastic fibres in an irregular mesh. A true epimysium of the pectoralis major is not identifiable and the deep fascia itself acts as a surrogate.
Clavipectoral Fascia
Detaching the pectoralis major muscle exposes the clavipectoral fascia (Figs 5.22 and 5.23). There is an ample plane of cleavage between the pectoralis major muscle and this fascia due to the presence of loose connective tissue. This loose connective tissue allows the deep layer of the pectoral fascia to glide autonomously with respect to the clavipectoral fascia. The clavipectoral fascia is a strong connective tissue layer arising from the clavicle and extending distally to enclose the subclavius and pectoralis minor muscles. Under the subclavius muscle, the clavipectoral fascia continues with the middle lamina of the deep fascia of the neck. Laterally, the clavipectoral fascia forms the suspensory ligament of the axilla that drags the axillary fascia upwards when the arm is raised. This forms the ‘pit’ of the armpit. Singer (1935) divides the clavipectoral fascia into two parts: one covering the pectoralis minor muscle and one that forms a triangular-shaped layer between the upper border of this muscle and the clavicle, called the ‘coracoclavicular fascia’. The thicker lateral border of the coracoclavicular fascia extends from the coracoid process of the scapula to the cartilage of the first rib and is known as the costocoracoid ligament. It separates the cavity of the axilla from the anterior chest wall. The anterior thoracic artery and nerve and the cephalic vein pierce the coracoclavicular fascia. In the arm, the coracoclavicular fascia continues with the fascia of the coracobrachialis muscle.
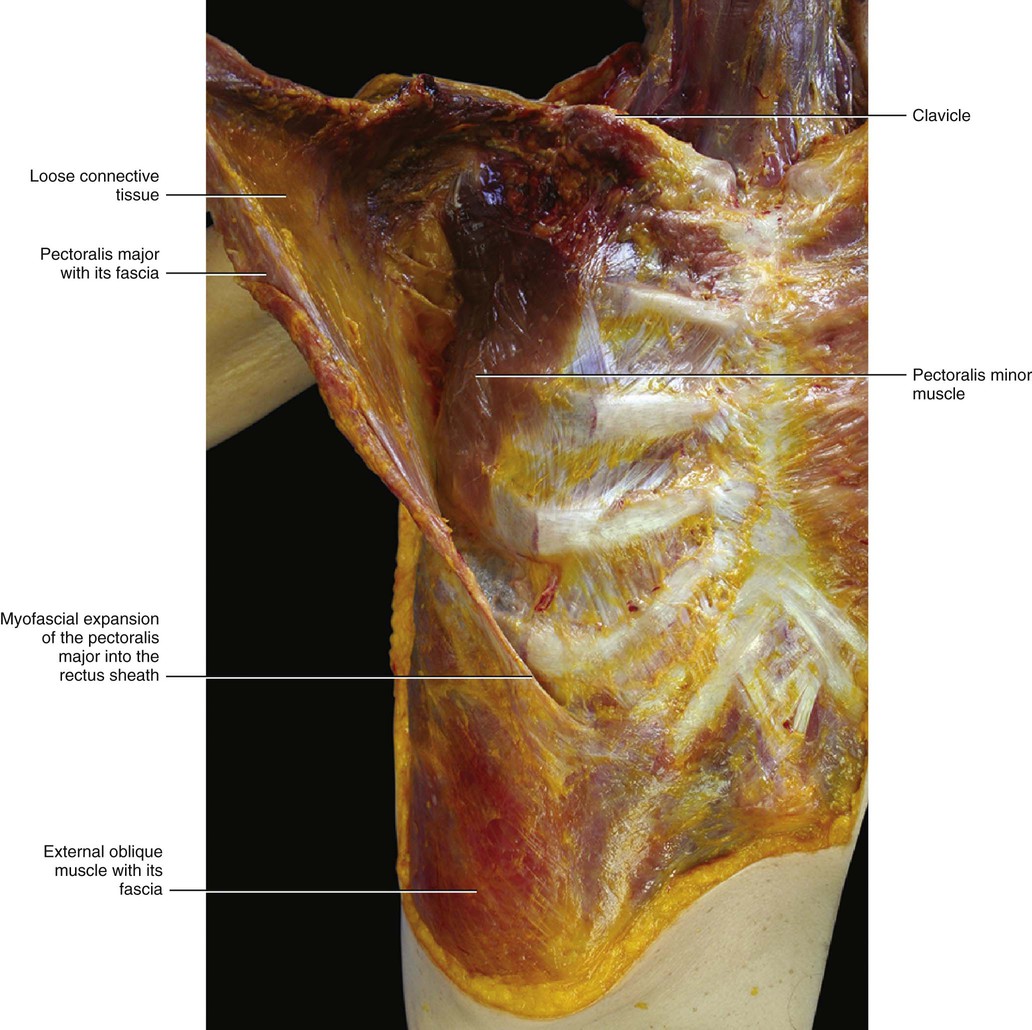
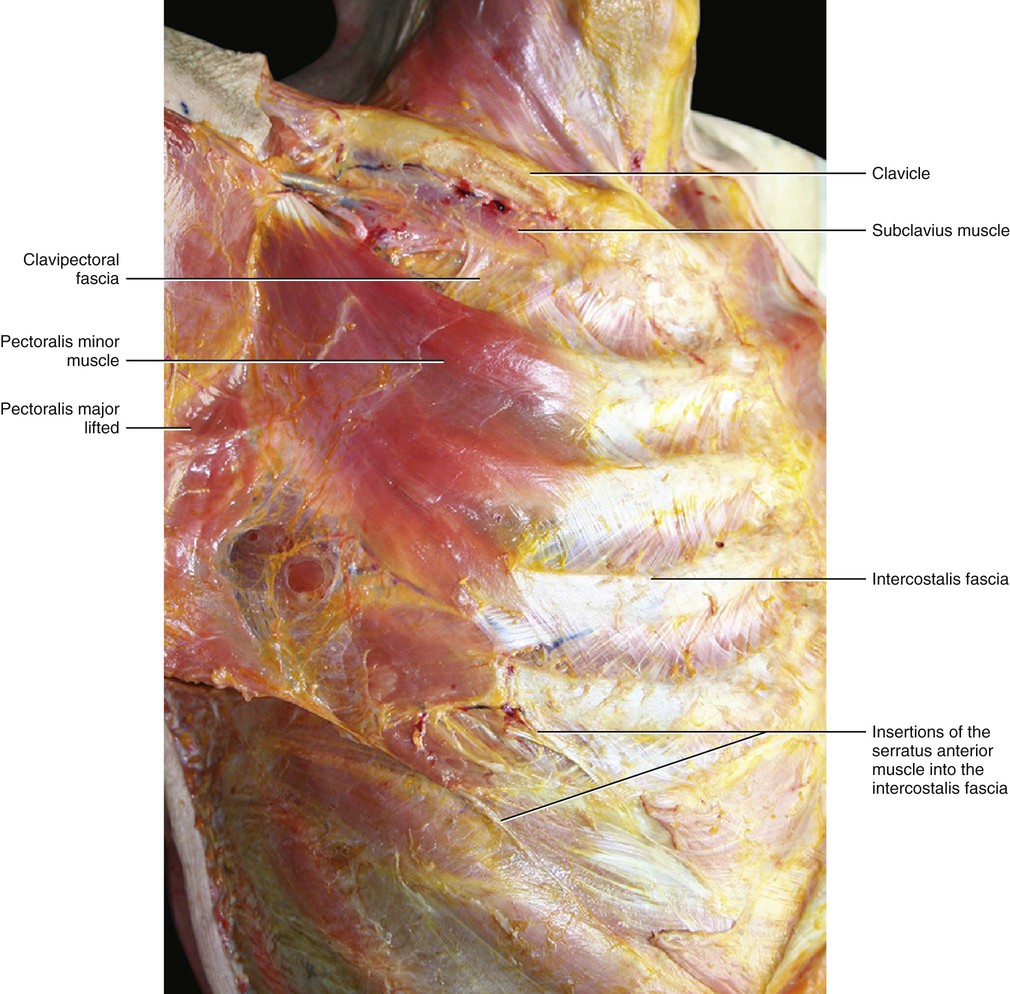
The continuation of the clavipectoral fascia envelops the serratus anterior muscle and then continues posteriorly with the fascia of the rhomboids (forming the ‘serratorhomboid complex’). This complex continues proximally with the intermediate layer of the deep fasciae of the neck. ‘Serratorhomboid complex’ was a term used by Nguyen in 1987 to emphasize the anatomical and functional continuity between the serratus anterior, rhomboid and levator scapulae muscles. Distally, the clavipectoral fascia continues with the fasciae of the internal oblique muscle.
Intercostal and Endothoracic Fasciae
The intercostal muscles are enveloped by a specific fascia that continues over the ribs as periosteum (Fig. 5.24). This fascia forms local fascial compartments between the various ribs, where the internal and external intercostal muscles are present. The intercostal nerves run along this fascia, as described by Kumaki et al (1979). The intercostal fascia continues with the fascia of the transversus abdominis muscle and with the rectus sheath. According to Chiarugi (1975), the lower internal intercostal muscles are in direct contact also with the internal abdominal oblique muscle. At the level of the sternum the intercostal fascia fuses with the periosteum. Internally, this fascia adheres to the parietal pleura to form the endothoracic fascia. Hence, an anatomical connection between visceral fasciae (parietal pleura) and muscular fasciae (intercostal fasciae) is created. This contrasts with the fascial anatomy of the abdomen, where there is a separation between the parietal peritoneum and the muscular fasciae. This difference is functionally significant as the lung needs the muscles to expand and in this way the parietal pleura follows all the movements of the thoracic wall.
Endothoracic fascia is a fascial layer deep to the intercostal spaces and ribs and represents the outermost membrane of the thoracic cavity. Testut (1905) described the endothoracic fascia as formed by three parts: a thin layer of loose connective tissue (probably corresponding to the epimysial fascia of the intercostal muscle), a fibroelastic layer (the true endothoracic fascia), and another layer of loose connective tissue (the parietal pleura). Stopar-Pintaric et al (2012) have studied the endothoracic fascia in rats using electronmicroscopy imaging. These authors located the endothoracic fascia between the parietal pleura and the innermost intercostal muscles or ribs. Its thickness ranges from 15 to 27 µm (mean, 20 ± 3 µm) and it appears as a condensed, fibroelastic lamina with its fibres primarily orientated transversely and obliquely.
The endothoracic fascia continues distally into the diaphragmatic fascia and, at the level of the oesophageal hiatus, it forms the phrenico-oesophageal ligament. The phrenico-oesophageal ligament is a strong structure firmly attached to the oesophageal wall. It surrounds the upper part of the distal oesophagus like a skirt. Thus, it is reasonable to assume that it plays an important role in the gastro-oesophageal sphincteric mechanism. Apaydin et al (2008) demonstrated that its collagen and elastic fibres decrease with age, which could mean a decrease in the resistance and the elasticity of the ligament. This situation may explain the predisposition towards developing hiatal hernias that is associated with increasing age.
Proximally, the endothoracic fascia forms the suprapleural membrane, which is attached in front to the internal border of the first rib and behind to the anterior border of the transverse process of the seventh cervical vertebra. It contains some muscular fibres which spread from the scalene muscles. Posteriorly, it is attached to the periosteum of the vertebral bodies, where it becomes continuous with the prevertebral fascia that covers the vertebrae and the intervertebral disks. Medially, it becomes part of the phrenopericardial membrane.
The endothoracic fascia has not been well defined as to its make-up, boundries or relationship to the spinal nerves. For example, Karmakar and Chung (2000) place the spinal nerves dorsal to the endothoracic fascia in the paravertebral space, while Naja et al (2004) claim that they are ventral.
Deep Fasciae of the Abdomen
The large muscles of the abdomen have a thin epimysial fascia with proprioceptive function and an aponeurotic fascia with a force transmission function formed by the fusion of their flat tendon and epimysial fascia. Loose connective tissue is present between the epymisial fasciae, permitting the gliding of the various muscular layers. The aponeurotic fasciae fuse with each other to form the rectus sheath. We can, therefore, identify a line of fusion between the three deep fasciae in the lateral border of the rectus muscle (lateral adhesion), and a larger line of fusion comprising the subcutis and skin, along the linea alba.
Fascia of the External Oblique Muscle
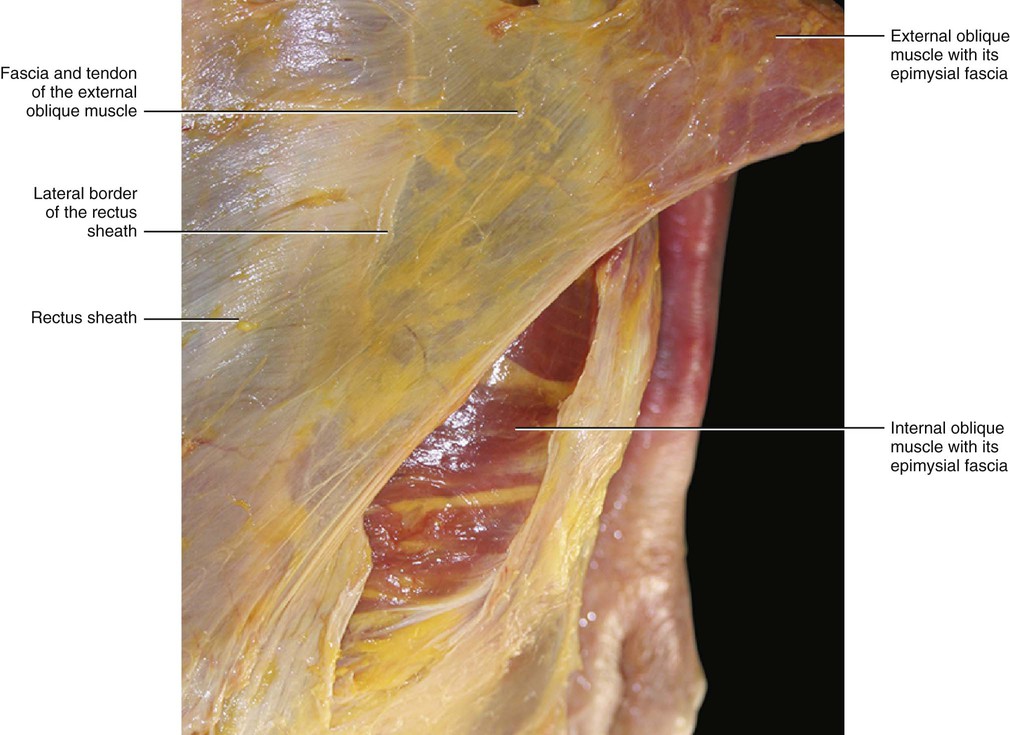
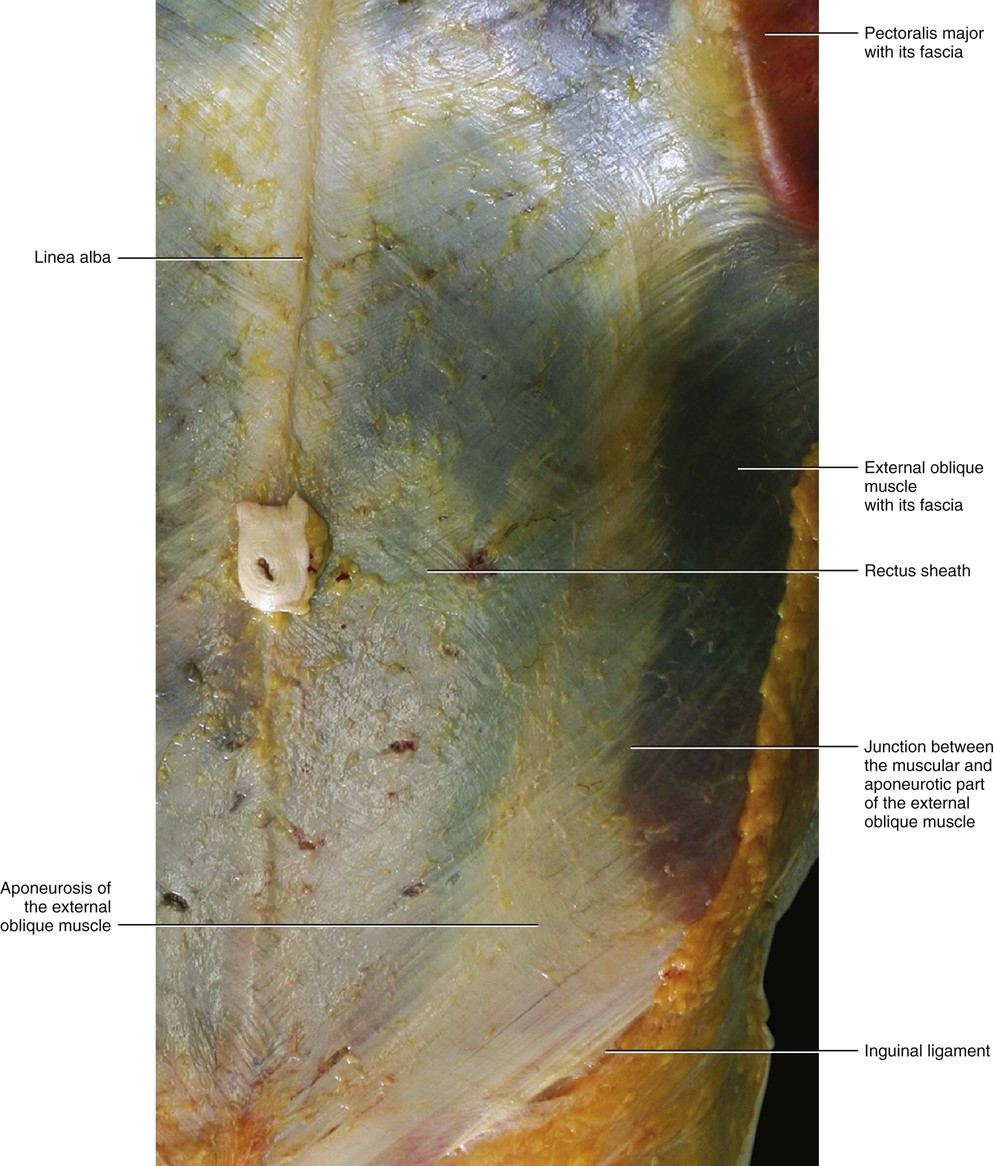
The fascia of the external oblique muscle is a thin but strong membranous structure, adherent to the underlying muscle and to the rectus sheath (Fig. 5.25). This fascia envelops the muscular belly and then continues to cover the tendinous insertions of the muscle. The oblique external component of the rectus sheath consists of two layers: a superficial layer formed by the true epimysial fascia of the muscle, and a deep layer, which is thicker and formed by the tendon of the oblique external muscle. This description agrees with Rizk (1980), who wrote:
The aponeurosis of the external oblique was formed of two layers: superficial and deep. The fibres of each layer were perpendicular to those of the other layer. The fibres of the deep layer were the direct continuation of the fleshy bundles of the external oblique muscle and extended downwards and medially. At the midline they crossed to the opposite side. Some fibres extended superficially to form the superficial layer of the opposite external oblique aponeurosis, while others passed deep to become directly continuous with the fibres of the anterior lamina of the internal oblique of the opposite side.
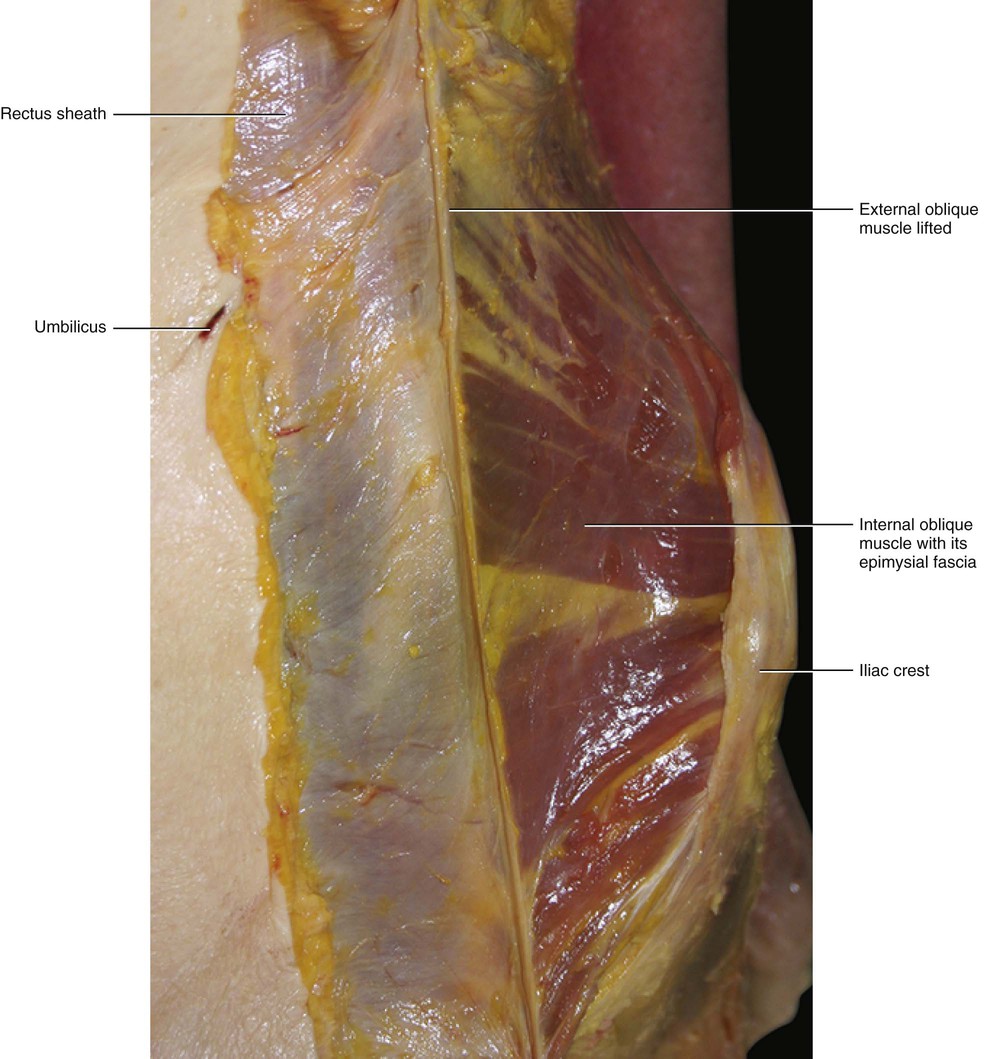
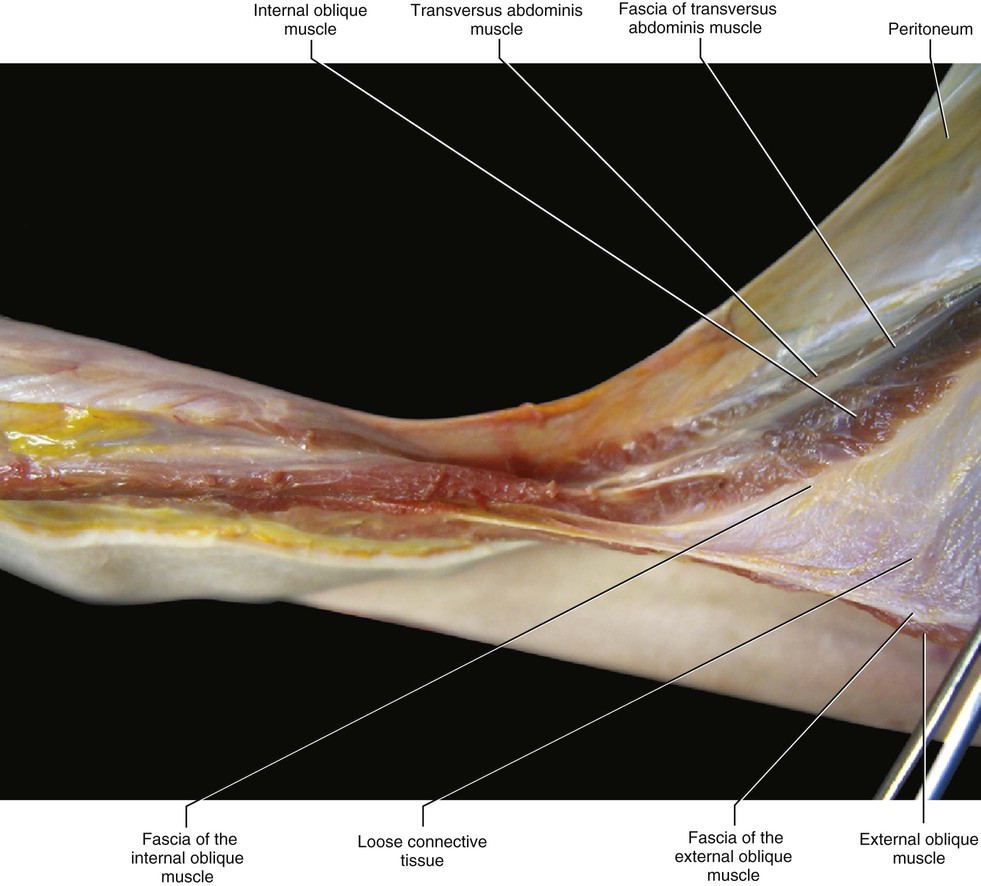
Rizk (1980) also described the myofascial expansion of the external oblique muscle into the fasciae of the controlateral external oblique and internal oblique muscles. This organization suggests that the muscles of the anterior abdominal wall are to be considered digastric-like muscles and the rectus sheath has the role of harmonizing the action of all these muscles. This prevents the action of an individual muscle or the muscles of one side from working independently. The external oblique muscle is free to glide with respect to the internal oblique muscle because of loose connective tissue between the two (Fig. 5.26). At the level of the rectus sheath, the various layers adhere and then completely merge at the linea alba.
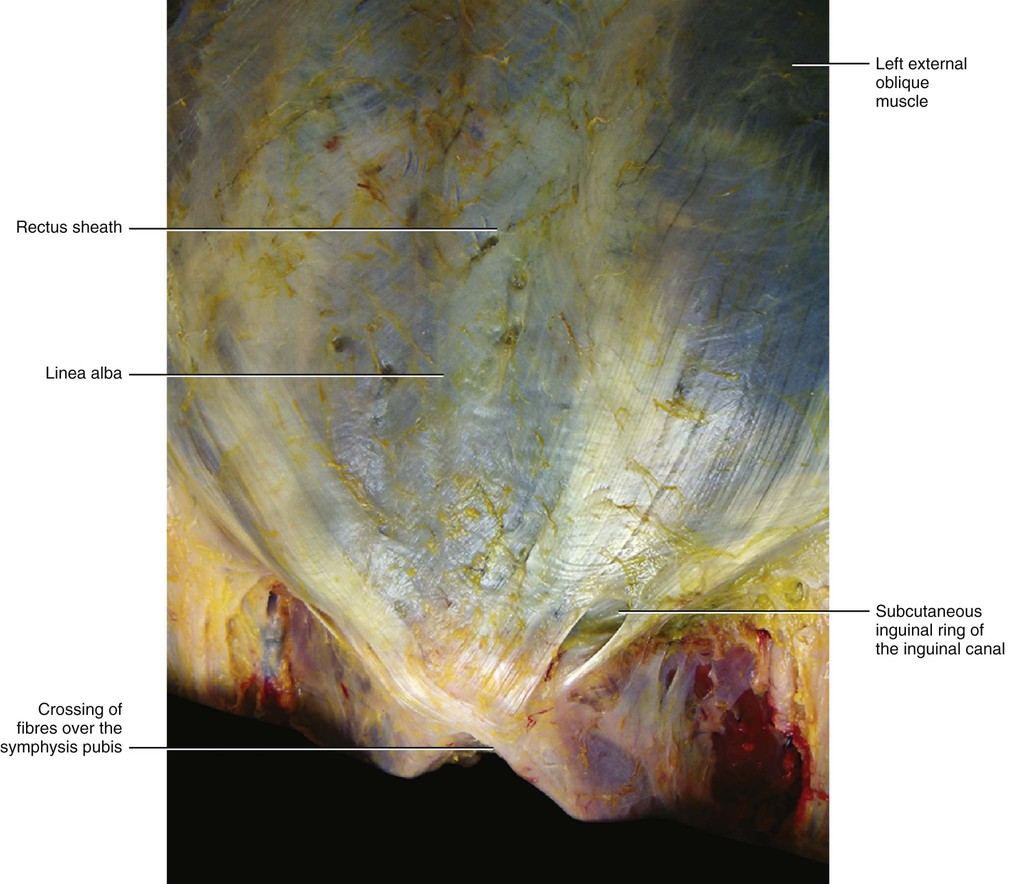
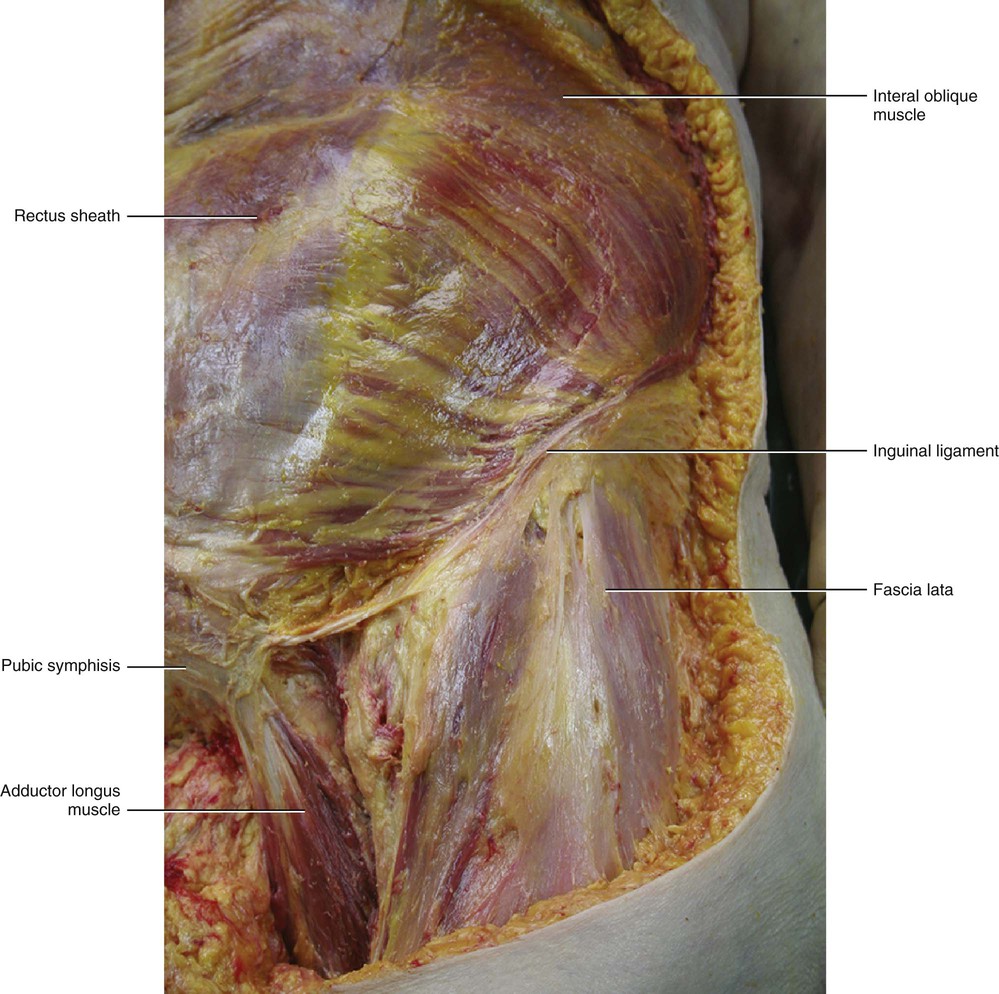
Distally, the fascia of the external oblique muscle is thicker and joins to form the inguinal ligament. The superficial fibres of this fascia continue distally to merge with the fascia lata, while the deeper fibres are attached to the anterior superior iliac spine and to the pubic tubercle. The inguinal ligament could thus be considered the lower border of the aponeurosis of the external oblique muscle, and it is also the point of conjunction between the fasciae of the abdomen and the fasciae of the thigh.
An expansion of the inguinal ligament connects the pubic tubercle with the pectinal line and forms the medial boundary of the femoral ring. Due to its expansion, the external oblique fascia is also continuous with the pectineal fascia, which originates from the pectineal line. The external oblique fascia, immediately above the crest of the pubis, has a triangular opening which forms the subcutaneous inguinal ring of the inguinal canal (Fig. 5.27).
Fascia of the Internal Oblique Muscle
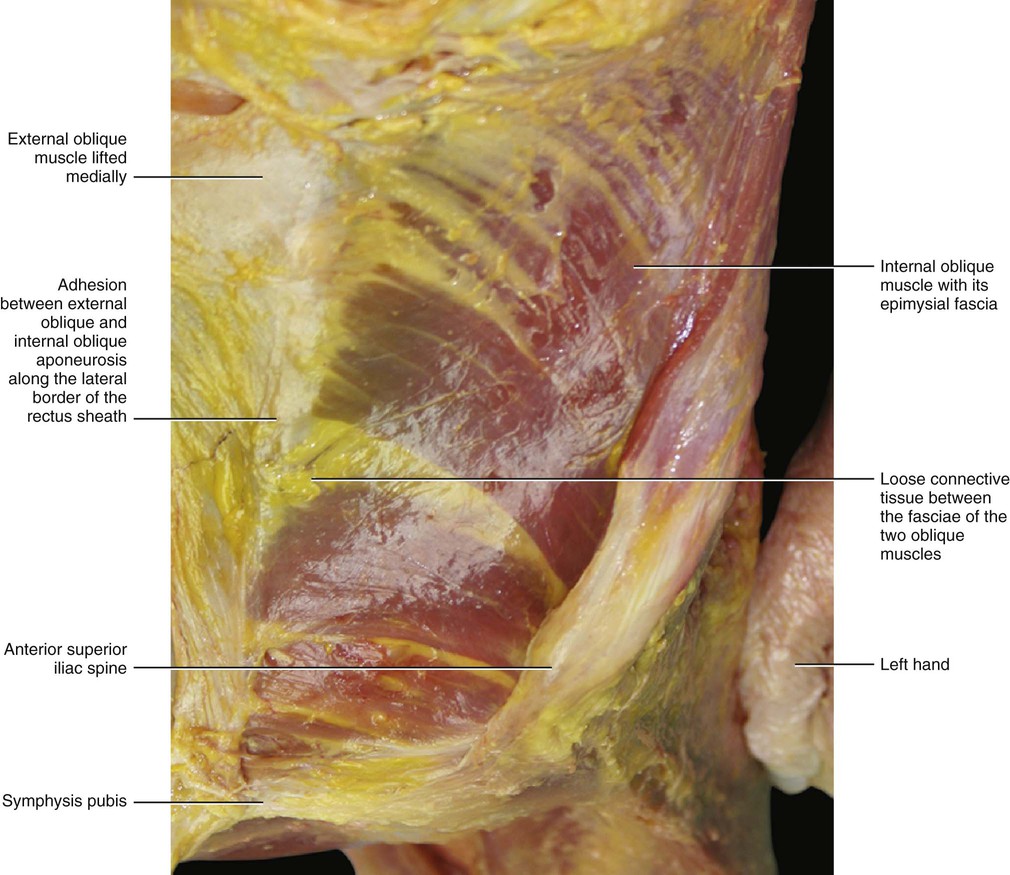
The fascia of the internal oblique muscle is a very thin fibrous layer that envelops the muscle from both sides. It is connected stongly to the muscle by many intermuscular septa and is separated from the fasciae of the external oblique and transversus abdominis muscles by a thin layer of loose connective tissue (Figs 5.28 and 5.29). This layer disappears over the rectus abdominis muscle where the various fasciae attach to one another to form the rectus sheath. The internal oblique fascia of one side joins with that of the opposite muscle along the midline, to form the rectus sheath and the linea alba. As for the external oblique, the fascia of this muscle, which is located over the rectus sheath, is also formed by two layers: the superficial (corresponding to the epimysial fascia), and the deep (which corresponds to the flat tendon of the muscle). The fascia of the internal oblique muscle arises together with the transversus abdominis muscle from the inguinal ligament, from the iliac crest and from the anterior layer of the thoracolumbar fascia. Some bundles of the internal oblique fascia arise from the inguinal ligament and arch downward and medially across the spermatic cord in the male and to the round ligament of the uterus in the female. They insert, joined together with those of the transversus abdominis fascia, into the crest of the pubis and the pectineal line. These form the inguinal aponeurotic falx.
Fascia of the Transversus Abdominis Muscle
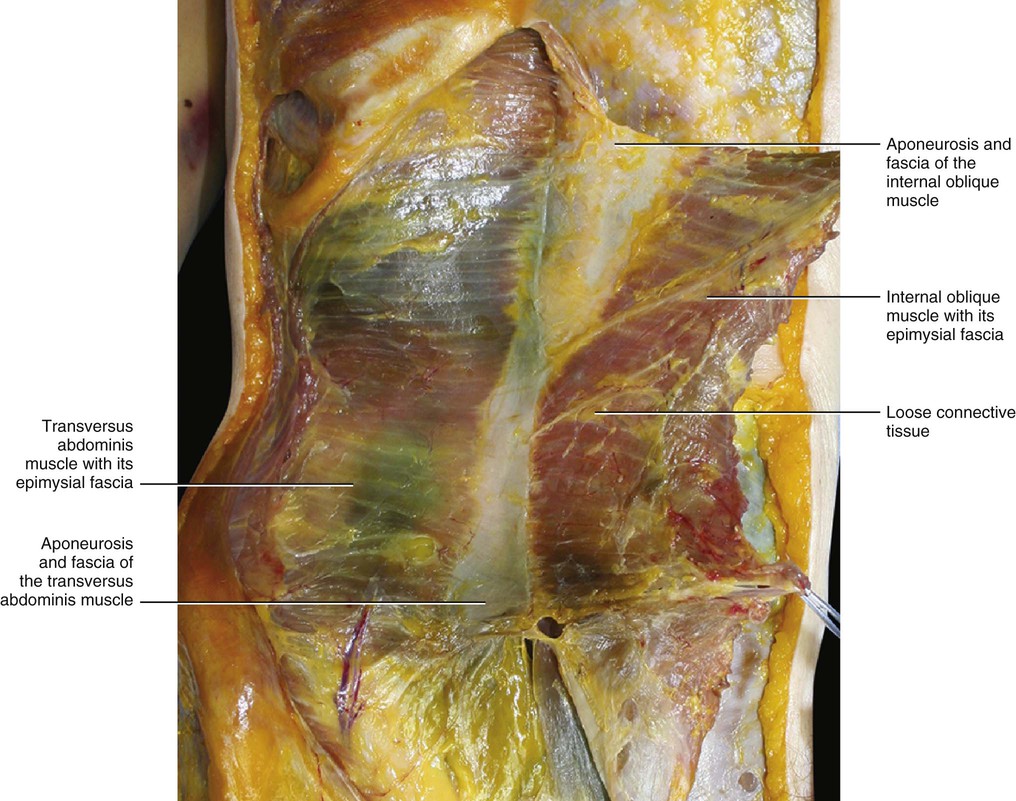
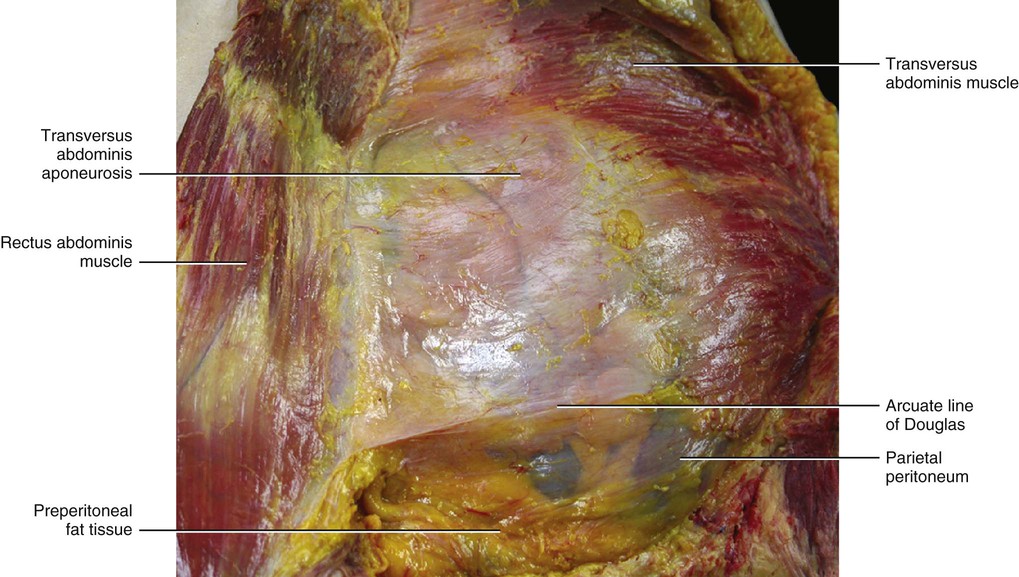
The transversus abdominis is the most internal of the flat muscles of the abdomen. It is enveloped by an epymisial fascia and usually separated from the internal oblique muscle by a layer of loose connective tissue (Figs 5.30 and 5.31). Distally, the two muscles sometime adhere. The transversus abdominis arises from the inguinal ligament, iliac crest, anterior layer of the thoracolumbar fascia, and inner surfaces of the cartilage of the lower six ribs to interdigrate with the diaphragm. This muscle ends in front in a broad aponeurosis, the lower fibres of which curve downwards and medially; they are inserted with those of the internal oblique into the crest of the pubis and pectineal line to form the inguinal aponeurotic falx. The rest of its aponeurosis passes horizontally to the midline crossing to the opposite side, and forming a digastric-like muscle linking the two sides together. This aponeurosis is commonly described as having its upper three-fourths lying behind the rectus abdominis and blending with the posterior layer of the aponeurosis of the internal oblique, and its lower fourth in front of the rectus. According to Askar (1977) and Rizk (1991), the rate of shifting of the fibres from the posterior to the anterior rectus sheath varies, and so the arcuate line (which demarcates the lower limit of the posterior layer of the rectus sheath) is often a dissector artifact.
Rectus Sheath
The rectus abdominis muscle is contained in a sheath made up of the aponeuroses of the three anterolateral abdominal muscles (external oblique, internal oblique and transversus abdominis). These aponeuroses fuse laterally to the rectus abdominis muscle and then pass, in part, over and under the rectus abdominis muscle to form its sheath (Fig. 5.32). Each large abdominal muscle contributes to the formation of the rectus sheath with two layers: the flat tendon and the epymisial fascia. This allows the rectus sheath to have both perceptive and force trasmission components. The layers of the external oblique muscle pass over the rectus abdominis muscle. The internal oblique muscle passes distally over the rectus abdominis, while proximally it divides into two laminae: a superficial layer that passes over the rectus abdominis muscle, and a deep layer that passes under the muscle. The transversus abdominis muscle passes proximally under the rectus abdominis muscle and distally over the rectus abdominis muscle. The lower border of the posterior layer is called the arcuate line or linea semicircularis of Douglas (Figs 5.33–5.35). It is a horizontal line that occurs about one-third of the distance from the umbilicus to the pubic crest, but this varies from person to person. Rizk (1991) affirms that the arcuate line is not an absolute point of termination of the posterior rectus sheath. It is also where the inferior epigastric vessels perforate the rectus abdominis.
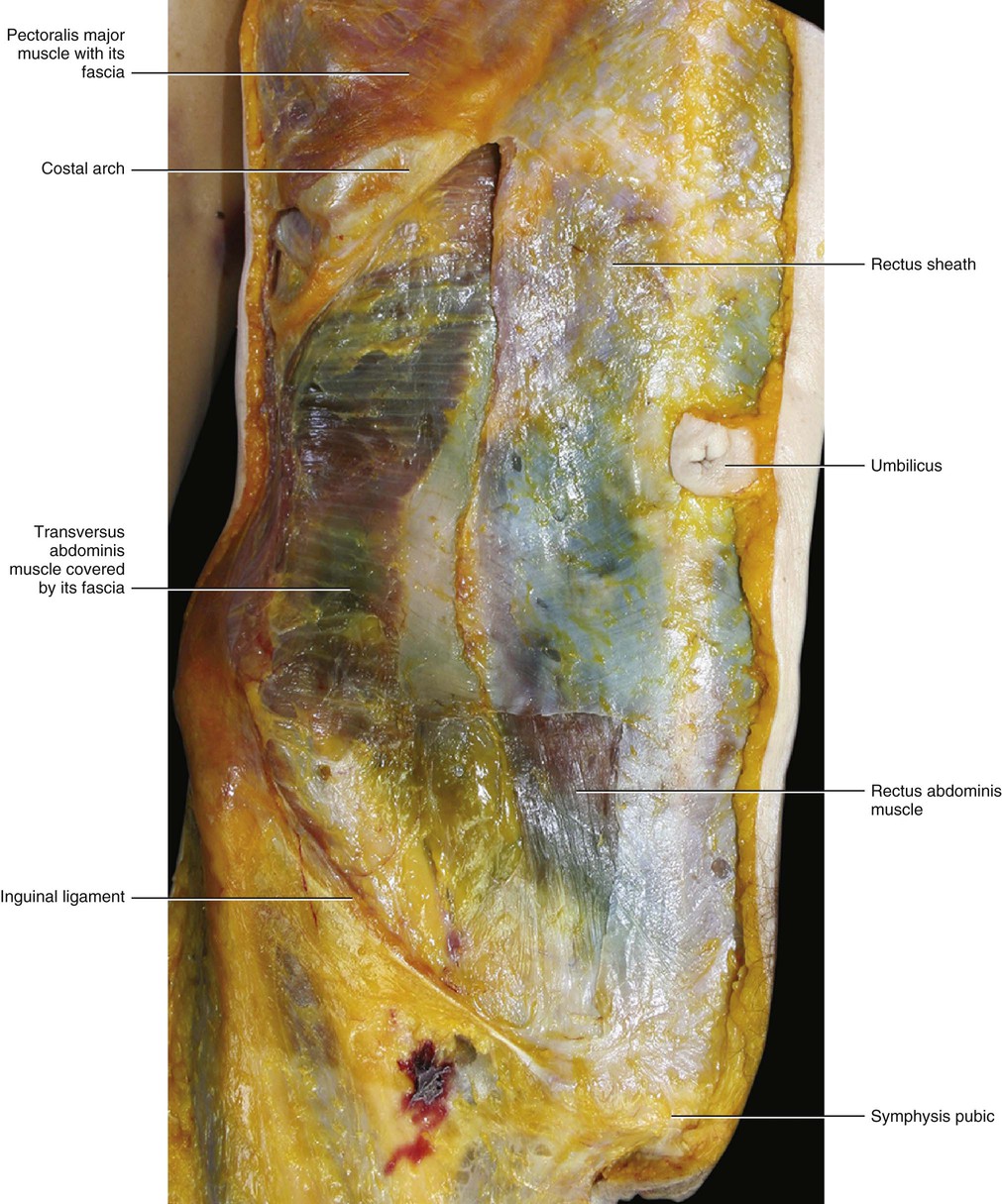
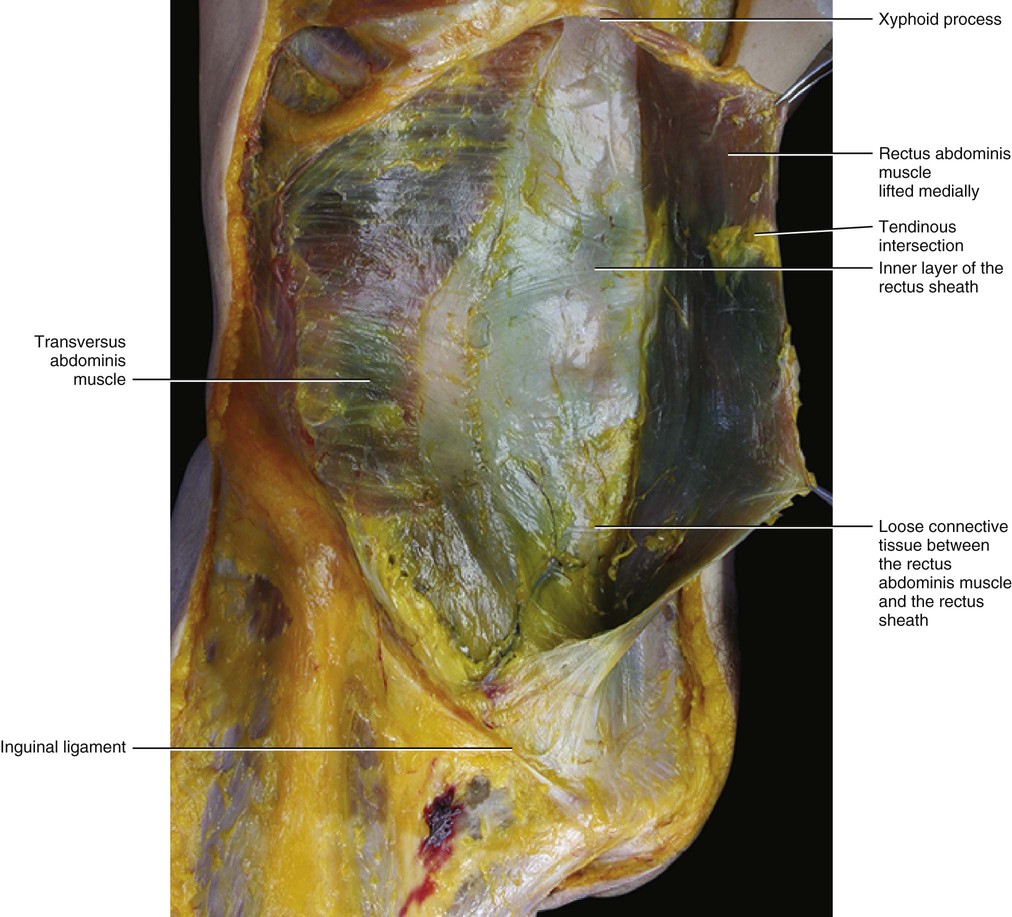

The current understanding of the composition of the rectus sheath is largely the result of the work by Askar (1977) and Rizk (1991), who independently reported their anatomical observations of the anterior abdominal wall and changed the traditional view of the rectus sheath. They described a bilaminar composition of the flat muscles of the abdominal wall, with each layer contributing fibres to the contralateral side. According to Askar (1977), the linea alba should be considered less as the insertion of the abdominal muscles and more as the common area of decussation of the intermediate aponeurosis. Consequently, the rectus sheath was discovered to be a trilaminar structure of variable thickness with decussating components from the external oblique, internal oblique and transversus abdominis muscles. They described a ‘plywood-like’ arrangement of the rectus sheath (similar to what we have described for the other aponeurotic fasciae), where there is a close collaboration of the layers without an actual fusion that would interfere with the free mobility of the abdominal wall (Fig. 5.36). In addition, histological analysis reveals a thin layer of loose connective tissue between the various sublayers of the rectus sheath, with complete adhesion only occurring at the level of the linea alba. Along the linea alba, also the superficial fascia connects with the deep fascia. This organization permits the free mobility of each layer, which allows an appreciable capacity for deformation in the resultant fabric of the rectus sheath. The oblique direction in which the collagen fibrous bundles are placed permits the adaptability of the rectus sheath to movements of the trunk. Due to this fascial organization, flexion of the trunk, for example, will produce folds in the skin and subcutis but not in the rectus sheath. This function may be lost as a result of rigid scarring following a midline incision.
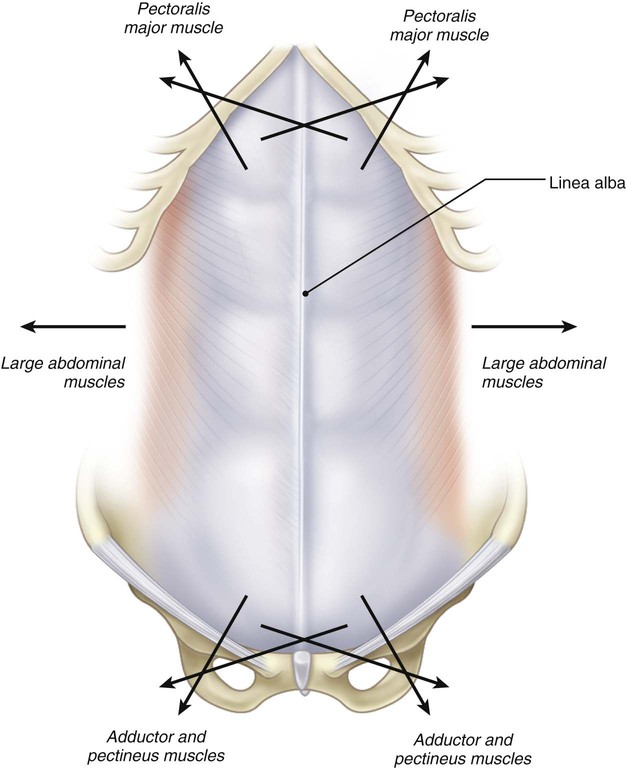
The rectus sheath is constantly stretched by the various muscular insertions. Obviously, the abdominal muscles stretch this sheath in a lateral direction, but it is also stretched in a longitudinal direction by the myofascial expansions of the pectoralis major and pyramidalis muscle. The anterior layer of the rectus sheath is reinforced proximally by the myofascial expansions of the pectoralis major muscles that form a cross in front of the xiphoid. Distally, the rectus sheath and particularly the linea alba are tensioned by the pyramidalis muscle, which may also be considered a fascial tensor (Fig. 5.37). This muscle is inside the rectus sheath (Rizk 1980) and is inserted into the pubis and anterior pubic ligament, and proximally it has just a fascial insertion into the linea alba.
There has been confusion over the position of the rectus abdominis with respect to the other muscular layers. Some authors hold that this is a superficial muscle. However, if we consider the embryological origin and the fascial anatomy, then the rectus abdominis is clearly a deep muscle corresponding to the erector spinae. The rectus abdominis originates from the hypaxial muscles and is the antagonist of the erector spinae that originates from the epaxial mass. The transversus abdominis muscle (a hypaxial muscle) with its fascia creates continuity between the epaxial muscles that moved around the visceral cavity during evolution. In addition, the fascia of the external oblique muscle is inserted posteriorly into the posterior (or ‘more superficial’) layer of the thoracolumbar fascia, and anteriorly forms the anterior (or ‘more superficial’) layer of the rectus sheath. The fasciae of the internal oblique and transversus abdominis muscles are inserted posteriorly to the anterior layer of the thoracolumbar fascia, and contribute anteriorly to form the rectus sheath. Thus, myofascial continuity between the thoracolumbar fascia and the rectus sheath is formed. This assures synchronization between the erector spinae and rectus abdominis muscles. Therefore, every time the oblique muscles contract, they stretch both the rectus sheath and the thoracolumbar fascia that envelops the erector spinae. This causes these fasciae to become more rigid, contributing to the increased force of the muscular contraction (see Clinical Pearl 6.4).
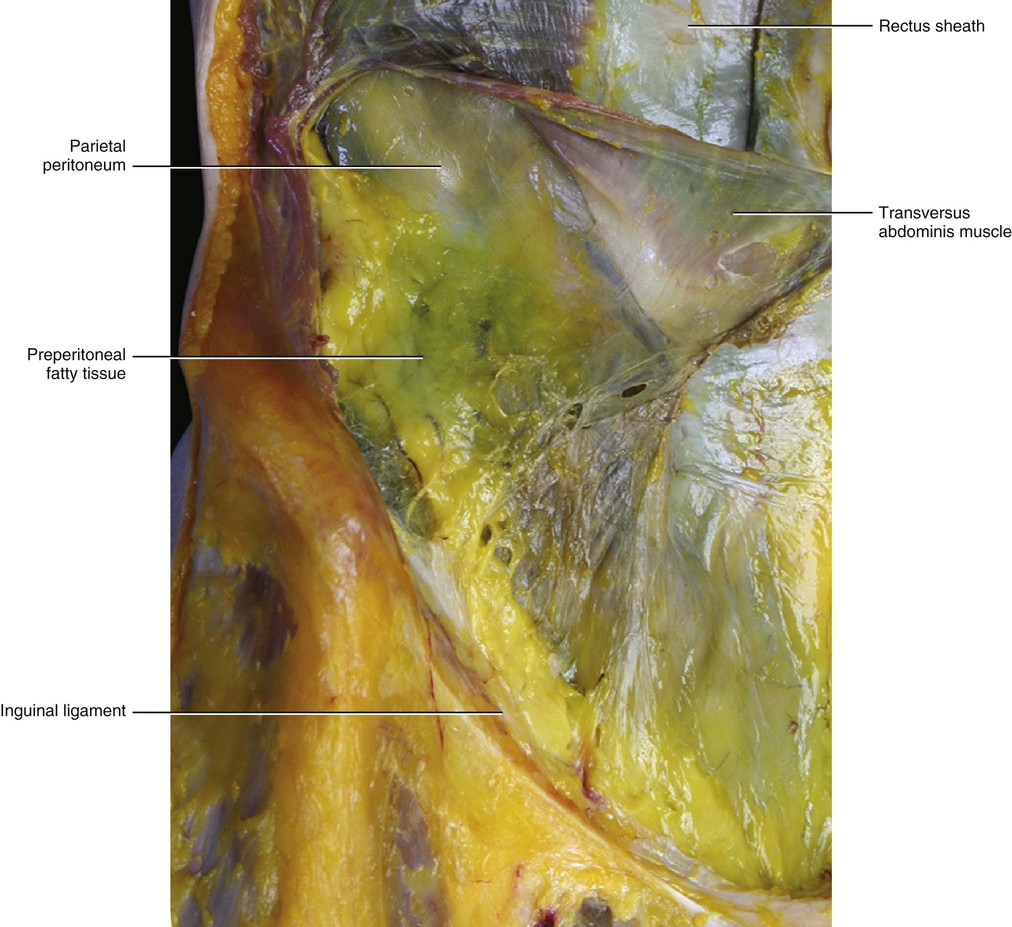
Transversalis (or Preperitoneal Fascia)
The transversalis fascia has been an object of discussion. According to Skandalakis et al (2006), the transversalis fascia is the epimysium of the transversus abdominis muscle. Tobin et al (1946) holds that the transversalis fascia is a fascial layer between the peritoneum and the abdominal wall. It forms a continuous lining for the abdomen, pelvis and spermatic cord (Fig. 5.39). He describes three strata: an inner stratum (peritoneum) associated with the digestive system; an intermediate stratum (transversalis fascia) embedding the adrenals, urogenital system, aorta and vena cava; and an outer stratum (muscular fascia) which is the intrinsic fascia of the transversus abdominis muscle. Our dissections reveal a distinct fascial layer between the internal surface of the transversus abdominis muscle and the peritoneum. This is especially evident in the distal part of the abdomen. This fascia is separated from the peritoneum by loose connective tissue that is typically scarce, but in some places may contain a great amount of fat, especially posteriorly and in the pelvis. Inferiorly, it is continuous with the pelvic andrenovesical fasciae. According to Bendavid (2001), the transversalis fascia corresponds to the investing layer of the bladder or the spermatic cord (Fig. 5.39). This fascia continues in the inguinal canal and in the scrotum. From an embryological point of view, we can affirm that the transversalis fascia comes from the septum transversum2, and is the element that guides the descent of the testis/ovaries into the pelvis.