Imaging of The Peritoneum, Mesentery and Omentum
Nicholas Gourtsoyiannis, Panos Prassopoulos, Maria Daskalogiannaki
Anatomical Considerations
Introduction
The peritoneum is the largest serous membrane in the body. It consists of the parietal peritoneum, which lines the abdominal wall, and the visceral peritoneum, which envelops hollow and solid abdominal viscera. Between these two layers lies the peritoneal cavity. Peritoneal reflections interconnect the organs and viscera enclosed within the peritoneal cavity. The name of a particular ligament corresponds to the two major structures that it joins, e.g. hepatoduodenal, splenorenal ligament. Ligaments that attach the stomach to other structures are termed ‘omenta’. The mesenteries connect a portion of bowel with the posterior abdominal wall. The fatty tissue enclosed by peritoneal folds is in anatomic continuity with the retroperitoneal and properitoneal tissues.1 A potential space, termed the ‘subperitoneal space’ is enclosed within the peritoneal membrane of the mesenteries, bridges the peritoneal cavity with the retroperitoneum and represents a significant pathway for the spread of disease from the retroperitoneum to the peritoneal cavity and vice versa.2
The potential peritoneal spaces and the peritoneal reflections act as boundaries for pathological processes but may also become conduits for the spread of disease.1 Typically, the peritoneal folds are not directly visible on cross-sectional imaging, but they can be identified by either their typical position or organ relationships or by the anatomical landmarks provided by their major constituent vessels. When they become thickened by oedema, inflammation or neoplastic infiltration, they can be directly recognised on computed tomography (CT) or magnetic resonance imaging (MRI).
Peritoneal Spaces
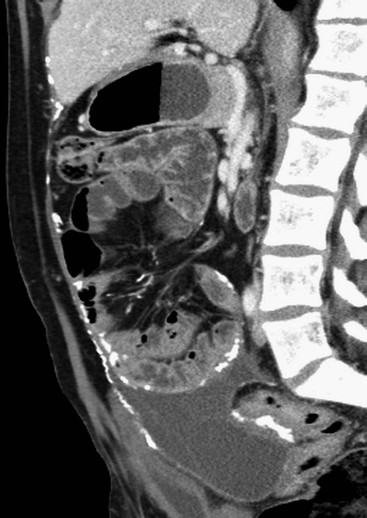
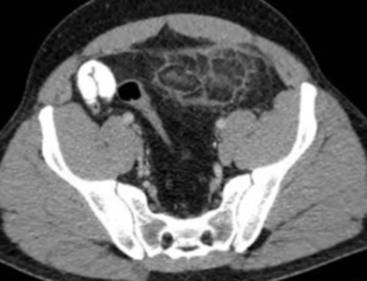
The peritoneal cavity is subdivided by peritoneal reflections into multiple compartments and recesses (Fig. 30-1). On cross-sectional imaging, the peritoneal spaces are not visualised unless they are distended by fluid (Fig. 30-2). The peritoneal cavity is divided into the supramesocolic and the inframesocolic compartments by the transverse colon and its mesentery.3
Supramesocolic Space
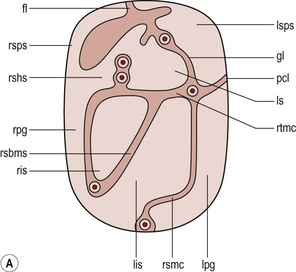
The supramesocolic space extends from the diaphragm to the transverse mesocolon. It is divided into right and left peritoneal compartments, which are arbitrarily subdivided into intercommunicating spaces.
The right supramesocolic space (Fig. 30-1A) includes the right perihepatic space and the lesser sac.1 The right perihepatic space includes the subphrenic and subhepatic spaces. The subphrenic space extends over the diaphragmatic surface of the right lobe of the liver and it is limited on the left by the falciform ligament and posteromedially by the right coronary ligament, which forms the right lateral margin of the bare area of the liver. The subhepatic space, also called the hepatorenal fossa or Morison's pouch, consists of the posteromedial continuation of the subphrenic space, extending between the liver and the right kidney. Gallbladder infections or collections after gallbladder surgery tend to accumulate in this space.2 The lesser sac is subdivided into a small superior recess and a larger inferior recess, by peritoneal reflection over the left gastric artery. The superior recess surrounds the caudate lobe of the liver and communicates with the right subhepatic space via the slit-like foramen of Winslow that is located between the inferior vena cava and portal vein. The larger inferior recess lies between the stomach, the visceral surface of the spleen and the pancreas. On the left it is bounded by the gastrosplenic ligament anteriorly and the splenorenal ligament posteriorly. Abnormalities of the transverse colon, pancreas, posterior wall of the stomach, duodenum and caudate lobe of the liver may extend into the lesser sac.
The left supramesocolic space is subdivided into four intercommunicating compartments.1 The left anterior perihepatic space is bounded on the right by the falciform ligament, posteriorly by the liver surface and anteriorly by the diaphragm (Fig. 30-2A). It is mainly affected by lesions arising from the left lobe of the liver and the stomach.2 The left posterior perihepatic space, also called the gastrohepatic recess, follows the posterior margin of the lateral segments of the left hepatic lobe. It is in close proximity to the lesser curve of the stomach, the anterior wall of the duodenal bulb and the anterior wall of the gallbladder. Abnormalities in any of these organs may extend into this space.3 The left anterior subphrenic space lies between the anterior wall of the stomach and the left hemidiaphragm, communicating inferiorly with the left anterior perihepatic space. Fluid collections in this space may result from perforation of the stomach or the splenic flexure of the colon.4 The left posterior subphrenic or perisplenic space is the posterior extension of the anterior subphrenic space (Fig. 30-2A).
Inframesocolic Space
The inframesocolic space is bordered superiorly by the transverse mesocolon and inferiorly by the pelvic rim. It contains the infracolic space and the paracolic gutters (Fig. 30-2B). The obliquely directed small-bowel mesentery, extending from the left upper midabdomen to the right iliac fossa, divides the infracolic space into a smaller right and a larger left space. The right infracolic space terminates at the ileocaecal junction. The left infracolic space is anatomically open to the pelvis except where it is restricted by the sigmoid mesocolon (Fig. 30-2C). The ascending and descending colon form the lateral borders of the right and left inframesocolic space, respectively.
The paracolic gutters are located alongside the lateral borders of the ascending and descending colon. The right paracolic gutter is continuous with the right perihepatic space and with the intraperitoneal pelvic space. Cephalad continuation of the left paracolic gutter is partially restricted by the phrenicocolic ligament. The pelvic peritoneal cavity consists of the lateral paravesical spaces and the pouch of Douglas—the rectovesical space in men, the rectouterine space in women.
Peritoneal Reflections
The Mesenteries
The mesenteries are double-layered peritoneal folds enclosing either the small bowel or portions of the colon and connecting them to the posterior abdominal wall. They contain a variable amount of adipose tissue, the superior or inferior mesenteric arteries and their branches, the associated veins, lymphatic vessels and nerves.
The small-bowel mesentery is a broad fan-shaped fold that suspends the jejunum and ileum from the posterior abdominal wall and contains the intestinal branches of the superior mesenteric vessels, lymph nodes, nerves and abundant fat.4 Its root originates at the duodenojejunal junction and extends downward in an oblique direction to the ileocaecal junction. The root is 15 cm long, while the intestinal border is 6–8 m in length. As a result, the mesentery has a pleated appearance along its intestinal border. The mesenteric folds are not discernible, unless they are separated by intervening fluid (Fig. 30-2B) or peritoneal thickening. Vasa recta can be identified within the fatty mesenteric tissue radiating in relationship to the mesenteric borders of small-bowel loops.3
The transverse mesocolon suspends the transverse colon from the posterior abdominal wall and provides an important route for the spread of disease across the mid-abdomen. On cross-sectional imaging, the transverse mesocolon can be identified as the fat-containing area extending from the uncinate process, the inferior border of the body and tail of the pancreas to the ventrally positioned transverse colon, containing the middle colic vessels.
The sigmoid mesocolon attaches the sigmoid colon to the posterior pelvic wall (Fig. 30-2C) and contains sigmoid and haemorrhoidal vessels. It has an inverted V-shape configuration with its apex lying anterior to the bifurcation of the left common iliac artery.
The Omentum
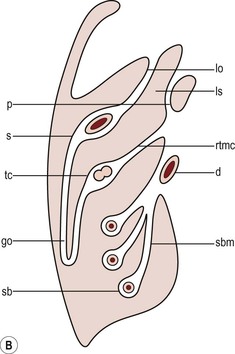
The greater omentum is a four-layered fold that descends from the greater curvature of the stomach, before turning superiorly again to insert into the anterosuperior aspect of the transverse colon. It provides an important pathway of disease spread from the greater curvature of the stomach to the transverse colon and vice versa. It has also an important role in limiting the spread of infectious diseases and confining bowel injuries, whereas it is a common site of involvement in metastatic peritoneal disease. On cross-sectional imaging, the greater omentum is identified as a fatty area extending behind the anterior abdominal wall, sometimes descending deep into the pelvis. There are portions of the greater omentum that are referred to with special names. The gastrocolic ligament is the segment of the greater omentum that links the stomach with the transverse colon. The duodenocolic ligament connects the first portion of the duodenum and the transverse colon. The gastrosplenic ligament extends from the stomach to the splenic hilum (Fig. 30-2A).
The lesser omentum or gastrohepatic ligament extends from the lesser curvature of the stomach deep to the fissure for the ligamentum venosum, between the caudate and left hepatic lobes (Fig. 30-1B). It contains the left gastric artery, the coronary vein and the left gastric nodal chain. Disease processes from the stomach extending along the gastrohepatic ligament may invade the liver, as the areolar tissue within the ligament is continuous with the hepatic capsule.1 The inferior edge of the gastrohepatic ligament, known as the hepatoduodenal ligament, bridges the upper duodenal flexure to the porta hepatis and contains the common hepatic duct, common bile duct, hepatic artery and portal vein.
Pathological Considerations
Ascites
The normal peritoneal cavity contains only a small amount of serous fluid—less than 100 mL. Free-fluid accumulation exceeding this amount is considered to be ascites. Accumulation of fluid in the peritoneal cavity is not a disease by itself, but it is the manifestation of a wide spectrum of processes that may involve intraperitoneal or extraperitoneal organs. Transudative collections may be associated with portal hypertension, cirrhosis, heart failure, nephrotic syndrome or obstruction of the inferior vena cava, hepatic vein or portal vein. Exudative fluid may be related to infection or peritoneal carcinomatosis. Blood collections may be the result of trauma, haemorrhagic diathesis or tumour rupture. Bile collections may follow rupture of the biliary tree; chylous collections may develop after lymphatic obstruction; pancreatic fluid collections may be the consequence of acute pancreatitis; urine collections may represent extension of a retroperitoneal urinoma; purulent collections may be the consequence of visceral inflammation, intestinal perforation or surgery. In general, haematoma, biliary, urinary or purulent collections may be limited by active peritoneal reaction; such reactions form adhesions which limit collections, isolate inflammatory processes and may plug perforations. Consequently, exudates may not move freely in the peritoneal cavity and are usually located or isolated at the area where they develop compared with transudates, which diffuse throughout the peritoneal cavity with no significant peritoneal reaction.
Peritoneal fluid moves along predictable pathways that are influenced by body habitus, gravity, intra-abdominal pressure gradients, adhesions and mesenteric reflections and attachments.3 At postmortem and at surgery ascites is mainly seen in the most dependent portion of the peritoneal cavity, namely the pouch of Douglas, which is the lowest and most posterior extension of the peritoneal reflections. In the intact abdomen, fluid migrates to the upper abdomen due to the lower hydrostatic pressure in the subdiaphragmatic related to respiratory movements. Fluid migration occurs along both the paracolic gutters and especially the right one, which is wider and deeper than the left, which in addition has an anatomical obstacle created by the phrenicocolic ligament at the level of the splenic flexure. Peritoneal fluid in the paracolic gutters is distinguished from retroperitoneal fluid by the preservation of the retroperitoneal fat posteriorly to the ascending or descending colon, provided there is not a complete ascending or descending mesocolon.
Ascites in the upper abdomen often accumulates in the pouch of Morison (or hepatorenal space, the most depended portion of the peritoneal cavity in the upper abdomen), the subdiaphragmatic spaces and the perihepatic and perisplenic spaces. Any amount of fluid that may be found in the inframesocolic compartment of the abdomen tends to move towards the lower pelvis; in the right inframesocolic compartment ascites flows along the surface of the small-bowel mesentery to a pouch at the ileocaecal conjunction and in continuation to the pelvis, while in the left inframesocolic compartment the fluid is directed to the surface of the sigmoid mesocolon and, thereafter, to the pelvis.
The CT attenuation values of ascites range from 0 to +30 HU. CT attenuation values are non-specific, although attenuation increases with increasing protein content as a general rule. Acute haemoperitoneum can be distinguished from other fluid collections by its high attenuation values (>30 HU), but lower values may be observed. In the presence of large ascites, the small-bowel loops are usually centrally positioned within the abdomen. However, in patients with very tense ascites, bowel loops can be displaced from the central position in the absence of intraperitoneal mass. Ascitic fluid under tension may result in extraperitoneal mass effect. Peritoneal fluid that becomes loculated due to benign or malignant adhesions may appear as a cystic lesion with mass effect.
Intraperitoneal Air
The most common location of free intraperitoneal air while the patient is in the supine position is anterior to the liver. CT is superior to plain radiographs in detecting minute quantities of pneumoperitoneum and negative window levels (lung settings) are very helpful in disclosing it. Free air demonstrated on CT can be distinguished from gas in the bowel because of its non-dependent location and lack of haustral or small-bowel folds. Perforation of the small bowel may be associated by inflammatory reaction in the mesentery in the form of streaky soft-tissue densities along with presence of extraluminal gas locally.
Developmental/Congenital Anomalies
Rotational Anomaly
Rotational anomalies of the small-bowel mesentery around the superior mesenteric artery occur when the normal process of fetal gut development is arrested. Intestinal malrotation in adults is generally asymptomatic. Reversal of the normal relationship between the superior mesenteric artery and vein, i.e. artery located to the right of vein, twisting of the mesentery around the artery and absence of the normal horizontal duodenum are characteristic findings.5
Developmental Defects
Internal hernias are formed by protrusion of a viscus through a peritoneal or mesenteric aperture. They include paraduodenal (53%), pericaecal (13%), foramen of Winslow (8%), transmesenteric and transmesocolic (8%), intersigmoid (6%) and retroanastomotic (5%) hernias. Internal hernias are often difficult to identify clinically. They are asymptomatic or cause symptoms ranging from intermittent and mild digestive complaints to acute intestinal obstruction. Imaging plays an important role in their diagnosis, with CT being the method of choice.
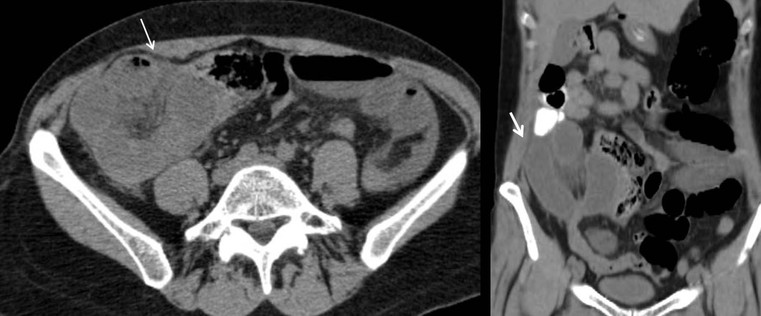
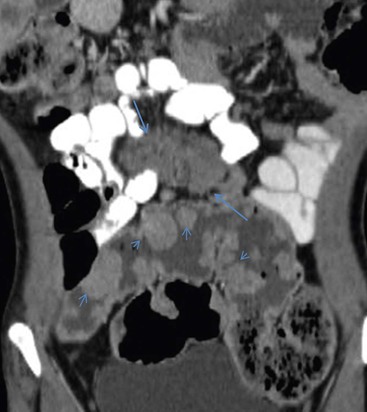
Paraduodenal, traditionally the most common type of internal hernias, result from congenital abnormalities in mesenteric peritoneal fixation. They are three times more frequent on the left side than on the right. Left paraduodenal hernias develop through the Landzert's fossa, which is present in approximately 2% of the population and is located at the duodenojejunal junction; the hernia sac lies posterior to the inferior mesenteric vessels. CT shows an abnormal cluster of dilated loops behind the stomach and pancreas, lateral to the duodenojejunal junction with anterior displacement of the stomach. Anterior displacement of the inferior mesenteric vein is a helpful sign.5,6
In right-sided paraduodenal hernias, bowel herniates through Waldeyer's fossa, behind the superior mesenteric artery and inferior to third portion of duodenum. It occurs most frequently in cases of a non-rotated small intestine. Imaging findings include encapsulated small-bowel loops in the right mid-abdomen with anterior displacement of the right colic vein, looping of the small intestine around the superior mesenteric vessels and abnormal position of the superior mesenteric vein relative to the artery.5
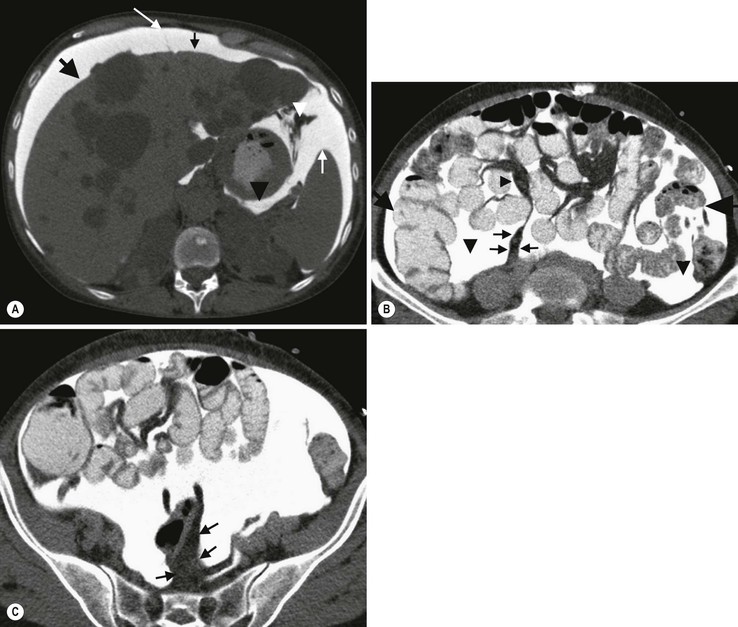
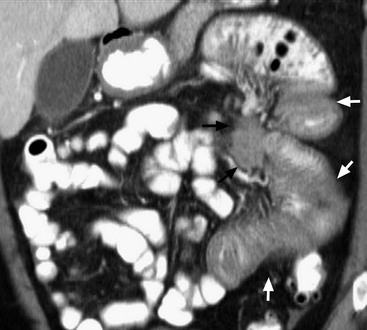
Transmesenteric hernias are increasing in incidence. They are more likely than other hernias to develop volvulus. In children, transmesenteric hernias are the most common type of internal hernia, related to congenital mesenteric defects. In adults, they are usually related to previous surgery, especially Roux-en-Y anastomoses. On CT, a cluster of dilated loops lying adjacent to the abdominal wall, without overlying omental fat lateral to the colon which is displaced centrally, provides an important clue (Fig. 30-3). The mesenteric vascular pedicle is characteristically engorged, stretched and crowded.7
Mesenteric Cysts
Lymphangioma is the most common mesenteric cystic lesion. Other mesenteric cysts like enteric duplication cyst, enteric cyst and mesothelial cyst are very uncommon.
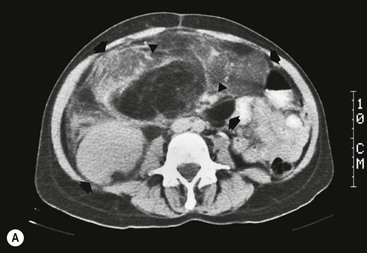
Lymphangioma represents a congenital malformation of the lymphatic vessels arising from the bowel. The typical imaging appearance is that of a large, thin-walled, single or multiloculated cystic mass, with contents of water-to-fat attenuation on CT (Fig. 30-4) and of high signal intensity on T2-weighted MR images. Enhancement of the cyst wall and septa is seen. It is frequently closely associated with the small bowel. US is helpful in demonstrating the internal septations of the cystic mass.8
Large mesenteric lymphangiomas can be differentiated from ascites by the presence of septa, compression on adjacent intestinal loops and lack of fluid in the dependent peritoneal recesses.
Infections–Inflammations
Infective/inflammatory infection of the peritoneal cavity may be localised (abscess) or generalised (peritonitis).
The CT appearance of a peritoneal abscess is variable, depending primarily on its age. In the earliest stages it may appear as a mass displaying attenuation values approximating those of soft tissue. As the process advances, the abscess undergoes liquefactive necrosis. A definable wall that may exhibit contrast enhancement and a nearly water attenuation centre are features of a mature abscess on CT. Accompanying findings include thickening or obliteration of adjacent fat planes and displacement of adjacent structures. Gas within a loculated fluid collection is highly suggestive of abscess, but is not pathognomonic, because a necrotic non-infected tumour or a mass that communicates with the bowel may also contain air.
The CT features of abscesses may overlap with other pathological processes such as haematomas, bilomas, urinomas, necrotic tumours or pseudocysts. MRI is superior to CT in differentiating a haematoma from an abscess, but may miss a small amount of gas within an abscess. A percutaneous fine needle aspiration may reveal the nature of a fluid collection and is important for abscess diagnosis by aspirating pus.
Peritonitis is characterised by a generalised collection of intraperitoneal fluid occurring secondary to bacterial, granulomatous or chemical causes. Although bacterial peritonitis may be primary, it usually results from an intraperitoneal abscess or rupture of a hollow viscus. CT features include ascites in association with peritoneal and mesenteric thickening. Gadolinium-enhanced MR images may show smooth peritoneal enhancement.
Tuberculosis
Tuberculosis of the peritoneum is an uncommon manifestation of tuberculosis that can occur after rupture of a caseous lymph node, from direct GI tract involvement by the disease or by lymphatic or haematogenous spread.
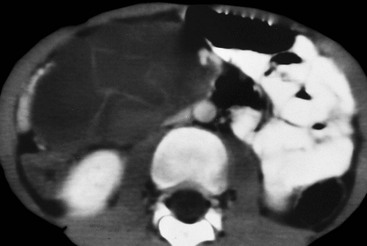
Common findings include the combination of free or loculated ascites, thickened strands with crowded vascular bundles within mesentery, smooth uniform thickening of the peritoneum and a smudged pattern of omental involvement infiltrated by small ill-defined soft tissue (Fig. 30-5).9 On CT, high-attenuation ascites (20–45 HU), reflecting its high protein content, may be seen. Lymphadenopathy is a common manifestation of abdominal tuberculosis and mesenteric nodes are frequently affected.
Peripheral enhancement with central low attenuation on CT may be seen and corresponds histologically to peripheral highly vascular inflammatory reaction around central liquefaction or caseous necrosis.10 This appearance is suggestive but not pathognomonic of tuberculosis as low-attenuation mesenteric lymph nodes may also be seen with Whipple's disease, necrotic metastases, infection with Mycobacterium avium-intracellulare, the cavitating mesenteric lymph node syndrome of coeliac disease and occasionally lymphoma.11
Hydatid Disease
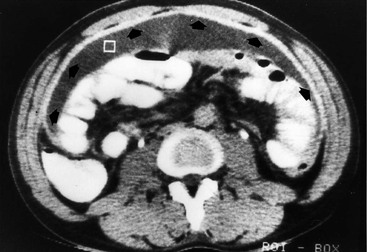
Hydatid disease is most commonly due to Echinococcus granulosus. Peritoneal hydatidosis is usually the result of traumatic or surgical rupture of hepatic hydatid disease and results in cystic, usually septated, thin-walled space-occupying lesions.12 CT is the method of choice in peritoneal seeding (Fig. 30-6). A calcifying rim is a suggestive feature. Ultrasound is useful for the detection of membranes, septa and hydatid sand within the cyst.
Crohn's Disease
Mesenteric abnormalities are commonly seen in Crohn's disease. Hypertrophied mesenteric adipose tissue, known as fibrofatty mesenteric proliferation, constitutes a characteristic feature of Crohn's disease. On cross-sectional imaging it appears as an increased quantity of mesenteric fat, producing a mass effect on adjacent loops, predominantly along the mesenteric side of the bowel (Fig. 30-7). Fibrofatty proliferation remains present in clinically quiescent disease, while engorged vasa recta (the ‘comb sign’) indicate active inflammation. MDCT/MRI can provide excellent information concerning mesenteric involvement and complications of Crohn's disease.13
Mesenteric Lymphadenitis
This is an appendicitis-mimicking clinical diagnosis, with self-limited symptoms that relate to benign inflammation of lymph nodes within the ileal mesentery. US and CT may demonstrate clustered, moderately enlarged mesenteric lymph nodes in the right lower quadrant without any identifiable inflammatory process. The diameter usually does not exceed 10 mm in short axis. Occasionally, an underlying terminal ileitis may be the cause of mesenteric adenitis.10
Acute Pancreatitis
In acute pancreatitis, extravasated pancreatic enzymes may dissect along the mesenteric pathways.1 An extrapancreatic acute fluid collection may spread along the root of transverse mesocolon and, at times, of small-bowel mesentery and extend along the vessels to the vasa recta of the transverse colon and small bowel (Fig. 30-8).5
Whipple's Disease
Whipple disease is a rare multisystemic infection caused by a Gram-positive bacillus. Low-attenuation mesenteric and retroperitoneal lymph nodes due to deposition of fat and fatty acids within the nodes are demonstrated on CT, in association with diffuse intestinal wall thickening (Fig. 30-9).10,14
Mesenteric Panniculitis–Sclerosing Mesenteritis
This complex disease entity is a rare, slowly progressive chronic inflammatory disorder of unknown origin involving the adipose tissue of the small-bowel mesentery and occasionally the mesocolon. It is now considered a single disease with two pathological subgroups. When inflammation and fatty necrosis are predominant components, it is known as mesenteric panniculitis. When fibrosis and retraction predominate, the disease is called sclerosing or retractile mesenteritis.
Various underlying causes, including prior abdominal surgery/trauma, autoimmune processes or malignancy, have been suggested. The disease is often asymptomatic. When present, symptoms are non-specific, including abdominal pain, diarrhoea, weight loss and fever of unknown origin.15 While a definite diagnosis requires surgical excision biopsy, in the majority of cases the disease is suggested on the basis of imaging features.
A well-delineated, heterogeneous fatty mass at the mesenteric root, envelopment of mesenteric vessels, absence of adjacent bowel loops involvement, which may or may not be displaced, and low-attenuation halo surrounding vessels (Fig. 30-10) are the characteristic CT features of mesenteric panniculitis.16 A hypointense capsule is demonstrated on T2-weighted MR images that shows contrast enhancement after intravenous gadolinium administration.
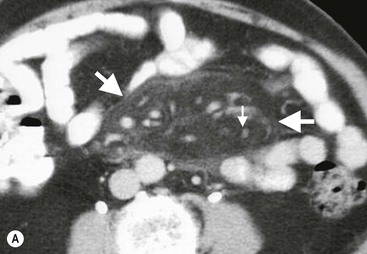
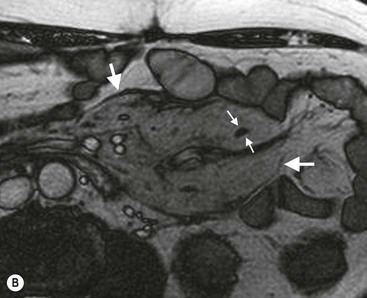
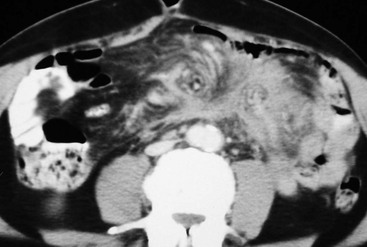
Retractile mesenteritis appears on CT as an infiltrative soft-tissue mass with associated radiating linear strands of soft-tissue attenuation (Fig. 30-11). These features may mimic those of fibromatosis or carcinoid tumours. MRI may help in the differentiation by showing low signal intensity on both T1- and T2-weighted images in sclerosing mesenteritis. Calcification may be present in the necrotic central portion of the mass (Fig. 30-12). The disease entity usually has a favourable prognosis and may be self-limiting.
Sclerosing Peritonitis
Sclerosing peritonitis, also known as encapsulating peritoneal sclerosis, is a rare chronic inflammatory disorder of the peritoneum associated with significant morbidity and mortality that occurs most commonly in patients who undergo continuous ambulatory peritoneal dialysis. Other rare causes include abdominal tuberculosis, recurrent peritonitis, long-term β-blocker treatment (practolol), sarcoidosis, etc. Patients may be asymptomatic or have nausea, anorexia, malnutrition, weight loss and recurrent episodes of small-bowel obstruction. CT is the optimal investigation for disclosing sclerosing peritonitis. Smooth or irregular and nodular peritoneal thickening and calcification, marked enhancement of the peritoneum, loculated fluid collections and tethered, thick-walled small-bowel loops are considered characteristic CT features (Fig. 30-13).17,18
Non-inflammatory Oedema
Mesenteric oedema is defined as a diffuse increase in attenuation throughout the mesentery that obscures visualisation of the normally well-defined mesenteric vessels as they course to the bowel walls. It may be secondary to various causes, including hypoalbuminaemia, cirrhosis, nephrotic syndrome, right-sided congestive heart failure, mesenteric ischaemia, vasculitis or trauma. Increased attenuation of the mesenteric fat and abnormal signal is seen on CT and MRI, respectively. The extent and site of involvement may contribute to the differential diagnosis.
In systemic diseases, such as cirrhosis or heart failure, mesenteric oedema is manifested as a diffuse haziness extending from the root of the mesentery to the bowel surface, associated with congestion and blurring of the mesenteric vascular markings. It is usually accompanied by ascites and generalised subcutaneous and retroperitoneal fat haziness secondary to generalised oedema.
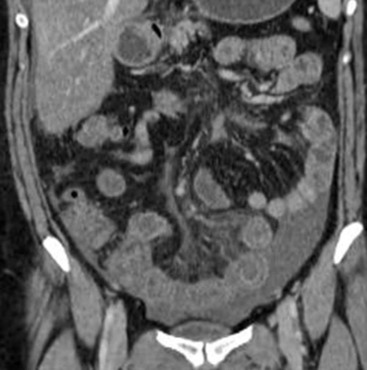
Hypoxia in acute mesenteric ischaemia causes a generalised increase in capillary permeability and consequently produces submucosal oedema. As ischaemia advances, venules in the mesentery become engorged with oedema and blood. If the ischaemic episode is not reversed, arterial flow to the involved segment becomes totally suspended. Mesenteric oedema secondary to mesenteric ischaemia may be focal or diffuse, depending on the site and extent of vascular compromise. As a result of oedema, increased attenuation of the mesenteric fat is seen on CT, together with poor definition of segmental mesenteric vessels (Fig. 30-14).
Epiploic Appendagitis
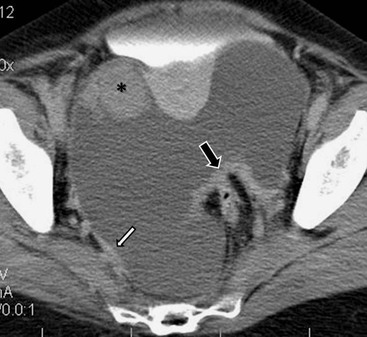
Epiploic appendagitis results from torsion or thrombosis of epiploic appendages. Clinical findings resemble acute diverticulitis or appendicitis. In the majority of cases, it is a self-limiting condition. CT findings of epiploic appendagitis are usually diagnostic, avoiding unnecessary surgery. The most common CT feature is an oval fatty lesion less than 5 cm in diameter surrounded by inflammatory changes and in contact with the serosal surface of the colon. A hyperattenuated rim that surrounds the mass and represents the inflamed visceral peritoneal lining may also be observed (Fig. 30-15). The lesion usually contains a central area of high attenuation corresponding to thrombosed vessels.19,20 Mild reactive thickening of the adjacent colonic wall is often found.
Omental Infarction
Segmental omental infarction is an uncommon cause of acute abdominal pain resulting from vascular compromise of the greater omentum. Primary omental torsion, attributed to congenital or vascular variations that predispose to venous thrombosis, is right-sided. Obesity has especially been postulated as an important risk factor. Secondary omental infarction is related to pre-existing abdominal pre-existing disease including surgery, abdominal inflammatory foci, tumours and hernial sacs and is located near the site of initial insult.
Typical CT findings include a large heterogeneous omental mass containing hyperattenuating streaks that is most often located in the right lower quadrant (Fig. 30-16). The mass is usually larger than 5 cm, differentiating it from epiploic appendagitis.10,19,20 The appropriate treatment is conservative.
Neoplastic Diseases
Neoplastic Diseases of Peritoneum
Nearly all peritoneal masses neoplasms are malignant. Secondary neoplasms are the most common malignancies involving the peritoneum. They can disseminate through the peritoneum by four pathways: direct invasion, intraperitoneal seeding, lymphatic permeation and embolic haematogenous spread.3 Although the imaging patterns of metastatic disease may coexist, many neoplasms metastasise predominantly by one particular route, indicating the primary site.
Direct Spread Along Mesenteric and Ligamentous Attachments
Malignant tumours of the stomach, colon, pancreas and ovary that have penetrated beyond the borders of these organs can spread directly along the adjacent visceral peritoneal surfaces to involve other structures. Early peritoneal invasion is manifested as linear strands in the fat adjacent to primary tumour. A mass contiguous with the primary neoplasm reflects more advanced dissemination. The mass may spread through the ligamentous attachments to involve other abdominal structures.1 In this way, gastric malignancy may extend to the spleen via the gastrosplenic ligament or invade the superior margin of the transverse colon along the gastrocolic ligament and vice versa (Fig. 30-17). Biliary tumours spread along the gastrohepatic and hepatoduodenal ligaments. Retroperitoneal tumours, such as pancreatic carcinoma, may directly invade the liver via the hepatoduodenal ligament or spread along the transverse mesocolon to the inferior border of the transverse colon. Pancreatic carcinoma may also directly invade the splenic hilum through the splenorenal ligament or the splenic flexure through the phrenicocolic ligament.21 Ovarian carcinoma may spread diffusely through all adjacent peritoneal surfaces.
Intraperitoneal Seeding and Peritoneal Carcinomatosis
Peritoneal seeding is a common mechanism of metastatic dissemination in advanced gastrointestinal and gynaecological malignancies. Peritoneal carcinomatosis (PC) is the term given to malignant tumour seeding of the peritoneum with most common primaries being the ovarian (71%), gastric (17%) and colorectal (10%) cancers. When cancer cells from a growing primary neoplasm reach the peritoneal surface, they are carried out by the peritoneal fluid and disseminated throughout the peritoneal cavity. Distribution of disease in PC is related to peritoneal fluid circulation along predetermined anatomical routes as previously described in the section ‘Ascites’. This peritoneal fluid circulation and the areas of temporal stasis of fluid explain the distribution of peritoneal seeding. Pooling of ascites favours the deposition and growth of seeded malignant cells. The most common seeding sites include the pouch of Douglas, the distal small-bowel mesentery near the ileocaecal junction, the sigmoid mesocolon, the right paracolic gutter, the pouch of Morison and the right subdiaphragmatic area.3 In general, more aggressive neoplasms exhibit malignant peritoneal deposits closer to the primary tumour, as opposed to less aggressive neoplasms that tend to manifest deposits in remote areas in the abdominal cavity. Variable amounts of ascites may accompany peritoneal seeding but ascites is not always present. Ascitic fluid is sometimes locculated and/or septated, and therefore it may be absent in dependent areas such as the pelvis. Peritoneal deposits (Fig. 30-18) may appear as thickening or enhancement of the peritoneum, nodules or plaques on the peritoneal surfaces, masses or merely stranding of the mesenteric fat.
MDCT with coronal and sagittal reformations (Fig. 30-19) is considered the optimal technique for detecting the presence, location and extent of peritoneal carcinomatosis; overall diagnostic accuracy of 94%, specificity of 92% and sensitivities between 75 and 81% have been reported;22 however, sensitivity for identifying small lesions by MDCT is significantly lower. Implantation of tumour deposits along the peritoneal surfaces of the diaphragm, liver and spleen results in smooth, nodular, or plaque-like thickening and contrast enhancement of the parietal peritoneal lining. However, these features are not specific for peritoneal carcinomatosis as they may be seen with other processes that seed the peritoneum, including tuberculosis, peritoneal mesothelioma and peritoneal lymphomatosis. Contrast-enhanced T1-weighted MR images with fat saturation can improve the detection of smaller or equivocal implants or of numerous very small implants that may be manifested as a contiguous ‘line-type’ enhancement along the peritoneal surfaces (Fig. 30-20). Peritoneal tumours often enhance slowly and are best seen on images obtained 5 min after injection of gadolinium. The combination of diffuse-weighted images (DWI) and conventional MRI improves the accuracy of MRI for depicting peritoneal implants;23 therefore, DWI is suggested as an indispensable part of any MRI evaluation in patients suspected for PC.23 DW-MRI and FDG-PET/CT are promising methods for the evaluation of peritoneal carcinomatosis,23,24 but still have a limited role, especially for the identification of disease relapse after treatment. Calcification within peritoneal implants before chemotherapy suggests that the primary site is either serous papillary cystadenocarcinoma of the ovary, or, rarely, gastric carcinoma (Fig. 30-21).
The evolution of cytoreductive surgery (CRS) has revolutionised the treatment of PC and is a challenge for imaging. Cytoreductive surgery aims at complete resection of tumour-bearing peritoneal surfaces and abdominal organs. Optimal CRS requires excision of any visible malignant peritoneal deposit. Complete CRS is often followed by hyperthermic intraperitoneal chemotherapy (HIPEC) to eliminate minimal residual PC. CRS with HIPEC is associated with significantly improved, progression-free and overall survival. Imaging techniques have an important role in the preoperative evaluation of PC and in the selection of candidates for CRS-HIPEC. Analysis of imaging findings should be performed on a site-by-site basis and the presence, type and size of peritoneal seeding should be reported in every region of the peritoneal cavity. Preoperative assessment of the overall disease burden and the extent of involvement in specific peritoneal areas are of decisive significance in the selection of patients that could benefit from CRS and in surgical planning.25 For example, extensive involvement of the hepatoduodenal ligament or of the small-bowel mesentery may preclude a complete CRS, while infiltration of the ligaments suspending the stomach by PC may indicate the need for gastrectomy (Fig. 30-22).
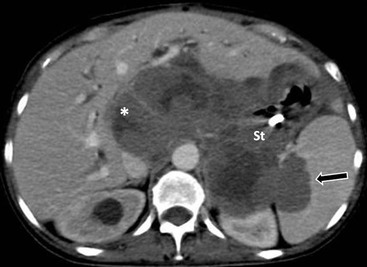
Pseudomyxoma peritonei is a specific type of peritoneal neoplasia due to ruptured benign or malignant mucin-producing tumour of the appendix, ovary and, occasionally, pancreas, stomach, colorectal or urachus. Suggestive imaging findings include masses of low attenuation on CT and moderately high signal intensity on MRI, accompanied by ascites with septations representing the margins of mucinous nodules, scalloping of visceral surfaces especially the liver by adjacent mucinous peritoneal implants, and soft-tissue thickening of the peritoneal surfaces, reflecting the more solid components of the tumour.26
Primary Neoplasms
Primary neoplasms of peritoneal origin are found less frequently than metastastic disease and include malignant mesothelioma, cystic mesothelioma, primary peritoneal serous carcinoma and desmoplastic small round cell tumour.
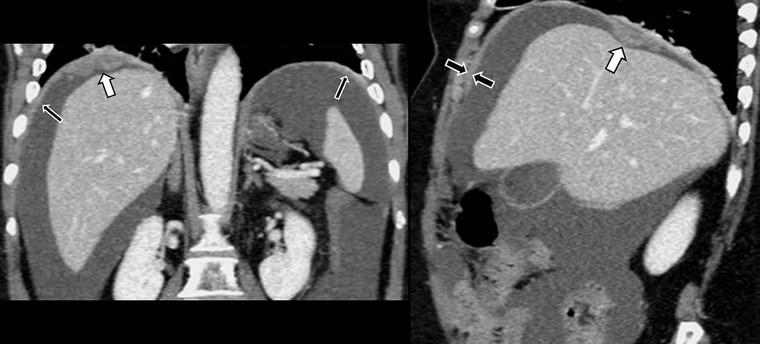
Malignant mesothelioma, most often seen in middle-aged men, is associated with asbestos exposure. Peritoneal involvement may occur, either alone or in association with pleural involvement. Imaging findings27,28 include diffuse or nodular thickening of the peritoneum, omental thickening, a stellate mesenteric appearance or peritoneal and omental masses, with variable ascites, usually mild to moderate (Fig. 30-23). Local invasion of adjacent abdominal organs may be seen.29 Enhancement of the peritoneal nodules or masses is typically seen on CT.27 Diffuse malignant mesotheliomas are highly aggressive in contrast to localised mesotheliomas that usually have a good prognosis.27 The differential diagnosis includes the more frequent peritoneal carcinomatosis and other abdominal processes such as tuberculous peritonitis, peritoneal lymphomatosis, peritoneal sarcomatosis, and pseudomyxoma peritonei. Malignant peritoneal mesothelioma appears similar to carcinomatosis but should be considered when sheet-like peritoneal thickening predominates. A smooth peritoneum with minimal thickening and marked enhancement suggests tuberculous peritonitis. Bulky mesenteric and retroperitoneal lymph node enlargement is the most prominent feature of lymphomatosis. Features suggesting sarcomatosis are heterogeneous bulky masses with hypervascularity, and variable ascites,30 while liver scalloping and septate ascites should raise the possibility of pseudomyxoma peritonei; leiomyomatosis peritonealis disseminata should be considered if uterine leiomyomas are present.27
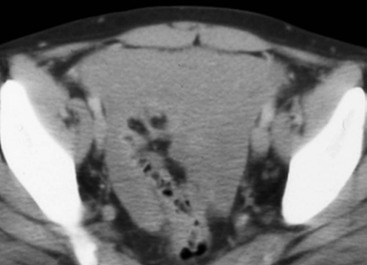
Cystic mesothelioma, also known as peritoneal inclusion cyst, is a rare non-malignant subtype of mesothelioma, most frequently found in the pelvis. It may be seen as a unilocular or complex cystic mass comprising cysts of 1 mm to 6 cm in size, simulating cystic lymphangioma or cystic epithelial neoplasms of the ovaries (Fig. 30-24).
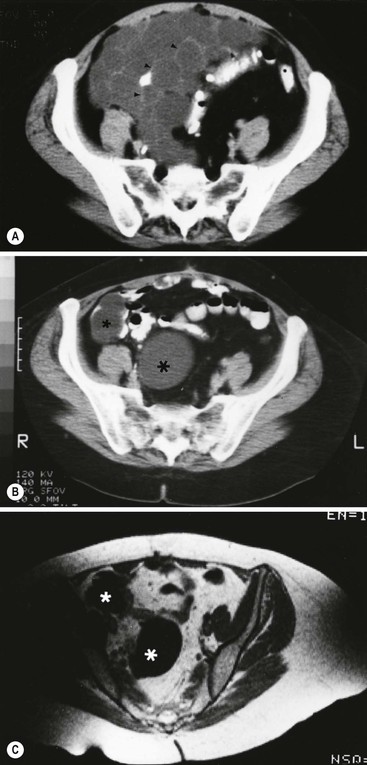
Primary peritoneal serous carcinoma is derived from extraovarian mesothelium and occurs in postmenopausal women. It is histologically identical to serous ovary adenocarcinoma. Imaging findings include ascites, peritoneal and omental thickening and peritoneal and omental enhancing nodules; psammomatous calcification may be seen.27
Desmoplastic small round cell tumour is a rare aggressive malignancy occurring in adolescents and young adults. Bulky heterogeneous peritoneal masses usually originating in the omentum or paravesical region are characteristic.31
Neoplastic Diseases of Mesentery
Primary neoplasms arising in the mesentery are rare and usually of mesenchymal origin.32 Benign primary mesenteric tumours such as desmoids, lipomas and neurofibromas are more common than malignant ones, such as fibrosarcoma (Fig. 30-25), liposarcoma or mesothelioma. Secondary neoplasms are more frequent than the primary malignancies and cystic tumours are more common than solid ones.
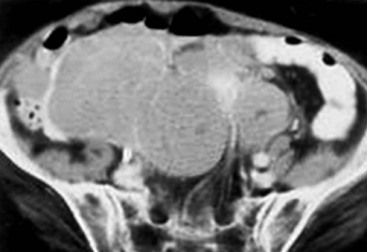
Primary Neoplasms
Primary neoplasms arising in the mesentery are rare and most are histologically benign, with fibromatosis being the commonest primary tumour.
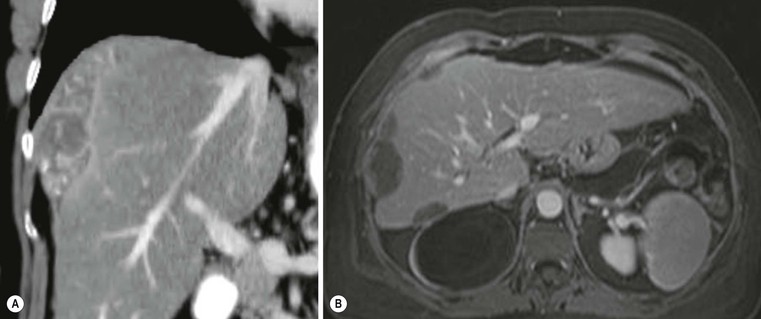
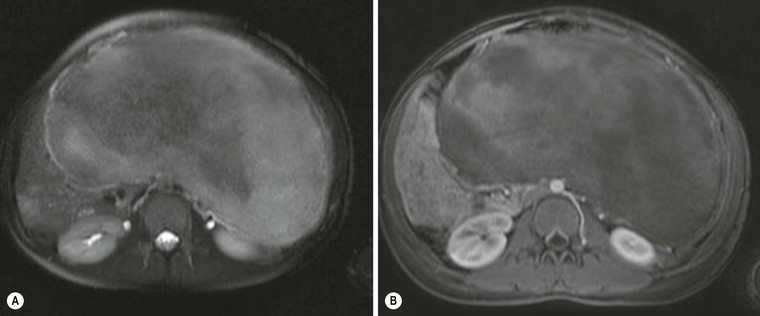
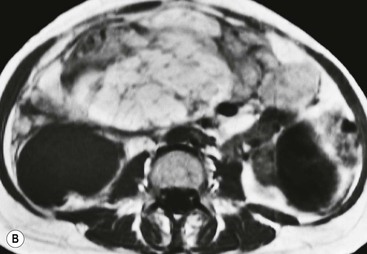
Fibromatosis, or desmoid tumour, results from a benign proliferation of fibrous tissue. Although it can occur sporadically, desmoid tumours are especially common in patients with familial adenomatous polyposis (FAP), particularly those with Gardner's syndrome. They are often locally aggressive but do not metastasise. The imaging features reflect the relative abundance of collagen or myxoid stroma within the lesion.33 On CT, lesions with highly collagenous stroma appear homogeneous, whereas those with myxoid stroma are hypoattenuated. A whorled appearance is described in lesions with both collagenous and myxoid areas. MRI shows low or intermediate signal intensity on T1-weighted images and variable signal intensity on T2 images (Fig. 30-26). The amount of hyperintensity on T2 images reflects the degree of cellularity and myxoid stroma.34 Contrast material enhancement is variable on both CT and MRI.33 It may be complicated by mesenteric ischaemia (Fig. 30-27) or intestinal obstruction. Fibromatosis accompanied by FAP (Fig. 30-28) is reported to be more invasive with higher local relapse rates.33
Lipomas are the second most common primary solid tumours of the mesentery, after fibromatosis. They appear as well-circumscribed homogeneous masses and are easy to diagnose on CT since they are composed entirely of low-attenuation fat. On MRI, they exhibit the characteristic high signal of fat on both T1- and T2-weighted images that turns to low signal when fat saturation techniques are applied. Internal septations are unusual.
Liposarcomas develop more frequently in the retroperitoneum than in the mesentery or peritoneum. Histologically, liposarcomas are classified, in increasing order of malignancy, as, myxoid, well-differentiated, pleomorphic, and round cell subtypes. They have variable CT and MR imaging appearances, reflecting their tissue composition, ranging from predominantly fat, fluid and soft-tissue elements to entirely soft-tissue density masses. Fat attenuation is less likely to be found in higher-grade liposarcomas such as the pleomorphic, and round cell subtypes (Fig. 30-29).35
Secondary Neoplasms
The most common malignancies spreading into the mesentery are secondary carcinoma, lymphoma and carcinoid tumour.
Secondary Carcinoma.
Primary neoplasms spread through mesentery by discrete pathways, producing characteristic imaging appearances.
Intraperitoneal Tumour Dissemination.
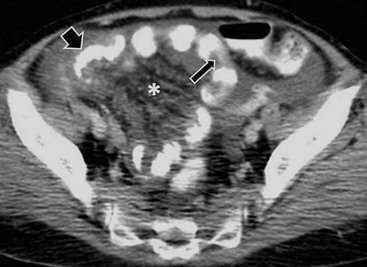
The small-bowel mesentery is frequently involved by intraperitoneally disseminated tumour. Common, although non-specific, imaging findings include scattered nodules, rounded, ill-defined soft-tissue or cystic masses and mesenteric fixation and thickening of the mesentery. The latter may demonstrate either a stellate or a pleated form.32 In the stellate appearance, a radiating configuration of the mesenteric folds with thickened rigid perivascular bundles and encased, straightened vascular structures is observed. A stellate pattern of infiltration has also been described in peritoneal lymphomatosis.32 In the pleated appearance, sheets of soft tissue produce thickening of the mesenteric folds. As metastatic tumour deposits involve the surface of the mesentery, the mesenteric fat is compressed rather than invaded. There is usually associated fluid, at times loculated. When ascites is extensive, it tends to surround the bowel loops, which are tethered centrally by the rigid mesentery. Diffuse infiltration of the mesentery by metastatic tumour may result in appearances resembling mesenteric oedema in some cases. Fixation of mesenteric folds, more apparent on repeated CT in the lateral or prone position, may aid the differential diagnosis. Peritoneal seeding may result in metastatic tumour nodules in the visceral peritoneal surfaces adherent to the serosa of small-bowel loops (Fig. 30-30). Larger metastatic mesenteric lesions maintain their relationship with the distal small-bowel mesentery and they may cause inferior and medial displacement of ileal loops. In cases of severe desmoplastic response to the seeded metastases, the marked fixation and angulation of ileal loops may lead to obstruction.
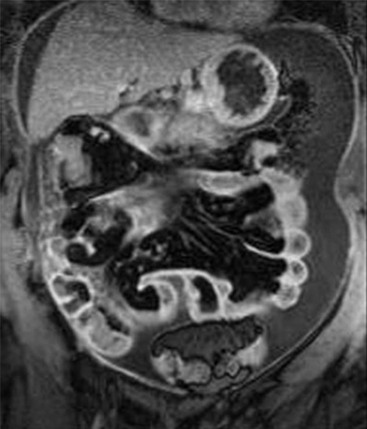
Involvement of the small bowel (SB) and SB mesentery is considered an independent prognostic factor of survival in patients with peritoneal carcinomatosis. The extent of SB/SB mesentery involvement by peritoneal seeding is crucial in the selection process of candidates for CRS and it is also important for surgical planning. Furthermore, SB mesentery is a common location of suboptimally debulked disease after CRS. As opposed to the clinical significance of an accurate preoperative evaluation of peritoneal seeding affecting the SB/SB mesentery, cross-sectional imaging presents the lowest sensitivity and diagnostic accuracy in this specific area. CT enteroclysis with SB distension by negative contrast medium results in increased sensitivity and specificity in the diagnosis of the extent of SB/SB mesentery involvement by peritoneal carcinomatosis36 and it has been proposed in the preoperative work-up in candidates for CRS (Fig. 30-31).
Embolic Metastases.
Embolic metastases from melanoma and lung or breast carcinoma can spread via mesenteric arteries to locate along the anti-mesenteric border of the small bowel. CT manifestations include focal bowel wall thickening and thickening of the mesenteric folds.29,32 Melanoma deposits may become large and ulcerated. Breast cancer deposits may cause multiple areas of small-bowel luminal narrowing with prestenotic dilatation.37
Lymphatic Dissemination.
Lymphatic permeation plays a minor role in the spread of metastatic carcinoma but it is the main pathway of dissemination of lymphoma to mesenteric lymph nodes. Enlarged mesenteric lymph nodes occur at presentation in approximately 50% of patients with non-Hodgkin's lymphoma. Confluent lymphomatous nodes may surround the superior mesenteric vessels, producing a sandwich-like appearance. Coexistent lymphomatous mural involvement of the small-bowel loops affects their mesenteric border.1
Carcinoid.
The small intestine is the commonest location for gastrointestinal carcinoid tumours. Approximately 40–80% of gastrointestinal carcinoids spread to the mesentery, either by direct extension or through the local lymphatics.32 Since the primary tumour in the small bowel can be quite small, and occasionally occult, metastasis to the small-bowel mesentery is usually discovered first. Secondary involvement of the mesentery usually leads to a desmoplastic reaction incited by local release of serotonin and other vasoactive substances. On barium studies, desmoplastic reaction appears as angulation, tethering and fixation of the involved small-bowel loops. CT/MR enterography is a valuable tool for evaluating patients suspected of having carcinoid tumours, since it may depict not only characteristic appearance of infiltrated mesentery but also the primary mural nodule. Carcinoid metastatic in the small-bowel mesentery manifests as hypervascular enhancing soft-tissue masses with well-defined or spiculated borders (Fig. 30-32). Radiating linear bands in the mesenteric fat result from the intense fibrotic proliferation and desmoplastic reaction. The degree of radiating strands tends to increase with the degree of fibrosis seen histopathologically.32,38 Segmental wall thickening of adjacent bowel loops resulting from chronic ischaemia or tumour infiltration as well as angulation can be seen. Dystrophic calcification is present in up to 40% of carcinoids on CT.38
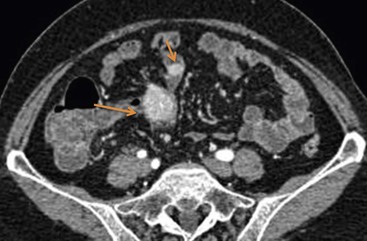
On MRI the lesions exhibit low T1 signal intensity, high T2 signal intensity and moderately intense gadolinium enhancement.39 Currently, somatostatin-receptor scintigraphy with 111In-octreotide, 111In-pentetreotide and 123I-metaiodobenzylguanidine is the study of choice for imaging carcinoid and its metastatic spread.40
Neoplastic Diseases of the Omentum
Primary neoplasms of the greater omentum, both benign and malignant, are similar to those that are also encountered in the mesentery. Benign neoplasms are usually well circumscribed and localised in the omentum. Malignant neoplasms frequently have indistinct margins and infiltrate surrounding structures.21
Secondary neoplasms of the omentum are more common than primary lesions. Secondary malignancies involving the omentum are similar to those responsible for peritoneal carcinomatosis, with the ovary being the most common source. The greater omentum is usually involved, since it is the site for re-absorption of the peritoneal fluid containing cancer cells. In addition, the greater omentum may be involved either by direct spread along the transverse mesocolon, the gastrosplenic, or gastrocolic ligaments or by peritoneal or haematogenous spread.
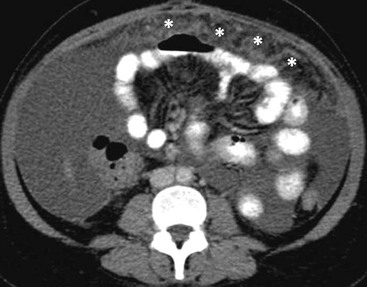
Early omental involvement is manifested on CT as omental thickening with increased attenuation values of the fat, fat stranding, nodules or irregular soft-tissue permeation of the omental fat (Fig. 30-33). In advanced seeding spread, the deposits range from discrete nodules to thick, confluent solid omental masses, the so-called omental ‘cake’. At CT, metastatic implants may enhance and be more readily seen within the low attenuation omentum. However, inflammatory thickening of the omentum may be indistinguishable from neoplastic infiltration. On MRI, implants present with low signal intensity within the high signal omental fat on T1-weighted images and exhibit enhancement. Extensive involvement of the omentum is manifested by a crescent-shaped mass, intermediate in signal intensity, exhibiting diffuse enhancement after administration of gadolinium. Fat-suppression post-gadolinium T1-weighted MR images facilitate the depiction of omental involvement.
For a full list of references, please see ExpertConsult.