Anesthesia for Labor and Delivery
The goal of modern-day obstetric anesthesia practice is to provide the patient with analgesia as she requests it.67,62 That assumes that her choice is appropriate to the labor process, to the current conditions as evaluated by her obstetric provider, and has as little impact on the fetus as possible.59 Depending on the patient's predilections, labor might be approached with prepared responses to modify pain perception, a plan to use systemic medications or nerve blocks, or some combination of these. How and whether a mother's choice has an impact on fetal well-being and neonatal outcome is the focus of this chapter and includes a discussion of the newer anesthetic techniques because the neonatologist should understand them. It also reviews some of the more established pain relief approaches as well as the more popular alternatives currently in vogue.
Labor Pain Characteristics and Challenges
Research has shown that the experience of pain is a complex, subjective, multidimensional response to sensory stimuli.25 The perception of pain in pregnancy is influenced by numerous factors such as fear of pain, increased susceptibility to pain because of generalized anxiety, level of education, and age or parity.11 To appreciate the complexity of providing analgesia to the laboring patient and better understand successes and expectations when doing so, a considerably abbreviated and simplistic discussion of pain is in order. A clinically useful classification is to define it as being visceral or somatic. Stated very simply, visceral pain originates from the viscera and is often described as cramping, dull, and steady.25,11 Somatic pain is related to nonvisceral structures and is commonly described as sharp, intermittent, and well localized.25,11 During the dilation phase of labor, or first stage, visceral pain predominates.25,11 This pain stimulus arises from mechanical distention of the lower uterine segment and cervical dilation. Somatic pain prevails during the descent phase of labor, or second stage, and it is attributed to the distention and traction on pelvic structures surrounding the vagina and from distention of the pelvic floor and perineum.25,11
The challenge with labor pain is in understanding that the very dynamic nature of it requires adaptation of pain techniques to adequately manage it. Pain can evolve rapidly over a relatively short period of time, changing not only in intensity, but also from a visceral to a somatic source. To add to the challenge, not all medications are effective for all types of pain. Local anesthetics block nerve conduction and are effective where the nerves responsible for pain transmission are accessible. In labor, the nerves transmitting the pain are well described, and most of them can be reached with a needle, making some form of nerve block with a local anesthetic a common choice. On the other hand, opioids are very effective for visceral pain, but are of little value for somatic pain.58 Therefore, systemic opioids can aid a patient reasonably well during the first stage of labor, but have little effect if used during the second stage.
Neuraxial analgesia is widely accepted as the most effective and least depressant method of providing pain relief in labor.67 The term encompasses epidural, spinal, and combined spinal-epidural nerve blocks that can be administered only by those trained in anesthetic methods.41 With modern-day techniques, dosing can be reduced to such low levels that an analgesic state, which diminishes pain while minimizing the effect on other sensory pathways such as touch and proprioception, is possible and often the goal.6 The lower doses result in less absorption into the maternal bloodstream than occurs with heavier dosing regimens, and in some cases levels are nondetectable. These methods allow the parturient to experience pain-free labor with minimal side effects to both the mother and the fetus while permitting the patient to actively participate in the labor process.41 However, all procedures carry some risk, and dosing methods vary widely across the United States such that there can remain a fetal or neonatal effect from the medications used.
Despite the proven safety and efficacy of the lower-dose neuraxial techniques, not all obstetric patients wish to use them, nor are there always practitioners available to perform the procedures.59 Also, an obstetric patient may not be a reasonable candidate for a neuraxial block for a myriad of reasons. As a result, many of the tried-and-true approaches for managing labor pain are still commonplace in obstetric suites, as are some interesting alternative therapies.
Techniques That Modify Pain
Although nonpharmacologic techniques do not alter the actual transmission of pain sensation, they attempt to alter the person's perception and response to it, with some measured success. Many of these approaches require preparation beforehand, and their effectiveness is subject to several variables. As a result, studies comparing controlled breathing techniques, hypnosis, and other nonpharmacologic methods to nerve blocks and medications are hard to evaluate.
The assumption held by proponents of alternative pain management techniques and many expectant mothers is that nonpharmacologic techniques to control pain without the use of medications and nerve blocks will not have any adverse effect on the neonate and therefore are the safest and healthiest choices for themselves and their babies. Clinical studies have not proved this assumption to be true. Although most techniques are as benign as they appear, some can carry unintended consequences if not performed properly, making this pertinent knowledge for the well-informed neonatologist.
Nonpharmacologic Approaches
Natural Childbirth
Natural childbirth is a technique that was first defined by Ferdinand Lamaze in the 1950s and began its popular rise in the 1960s as an alternative to the heavy sedation that was often used at the time. The technique of controlled breathing and focal points to stay in a relaxed state during a contraction is based on the theory that one can modify the sensation of pain and one's response to it. Building upon the popularity of the Lamaze approach to labor pain, the Bradley technique was introduced in the 1970s as a variant and incorporated the woman's partner as her active coach. Today the term natural childbirth applies to any controlled breathing technique that the mother uses during labor to cope with pain and avoid pharmacologic methods.
Is natural childbirth an effective way to manage pain during labor? There is some objective evidence that Lamaze training as originally practiced is associated with β-endorphin elevations, which may help alter pain sensation.22 What is not always appreciated by those who teach and by couples who use this technique to prepare for labor is that incorrect performance can have negative consequences. Maternal and fetal Pco2 levels are positively correlated.53 If the mother deviates from the defined breathing technique and hyperventilates during her contractions, she will lower her Pco2 levels as a result.50 This situation has two effects. First, maternal hypocapnia causes uterine vasoconstriction, thereby reducing blood flow to the fetus; second, the fetus also develops hypocapnia in response to the maternal hypocapnia. As the fetal Pco2 falls, the fetal oxyhemoglobin dissociation curve shifts leftward, causing a reduction in fetal oxygenation and setting the stage for hypoxia and metabolic acidosis. If fetal acidosis develops during the first stage of labor, it will worsen during the second stage, especially if that stage exceeds 1 hour.71 Once hypoxia and a base deficit are present during the second stage of labor, it is difficult to reverse in utero.71 The result is a neonate who is in need of resuscitation. Obviously this series of events does not occur in every case, but the fetal tracing associated with this scenario can resemble that of cord compression and result in obstetric maneuvering.53 Effective analgesic techniques have been shown to improve these conditions if applied before the second stage.53
With the boom of the Internet, patients today are able to find a variety of information online about natural childbirth practices without any means to determine if the information presented is accurate. A common thread is to suggest that natural childbirth is empowering for women and has little potential for harm to the baby. Several factors have come to bear upon modern-day childbirth courses that teach the Lamaze or Bradley techniques and the information that is provided. Couples are no longer willing to take the full 6-week training course. Instead, weekend courses are offered that try to present all the information in 2 days. This alternative makes it nearly impossible to practice and learn the pain-controlling techniques that Lamaze and Bradley described. Additionally, information on all available forms of pain relief options is variable, because of not only some potential personal biases by the instructor, but also the instructor's knowledge of what is available at the maternity units in the area. As a result, couples may approach labor with a rigid plan that does not take into consideration informed alternatives for pain management should the fetus show signs of compromise. Unfortunately, a mother who envisions a natural childbirth for her labor often views resorting to any form of analgesia, especially an epidural, as a failure on her part. Studies show that natural childbirth preparation, including training in breathing and relaxation, did not decrease the use of epidural analgesia during labor.7 It has been shown that when mothers who planned for an unmedicated labor resort to a neuraxial block, it is as a result of labors that are frequently longer and associated with higher pain scores, which may catch them unprepared for the intensity of the process.37
Hypnosis
Hypnobirthing, or self-hypnosis for labor and delivery, enjoys a devoted following among those for whom the process has been successful, but certainly it is a technique that not many individuals can use. It requires time for training and practice, and is not a choice for those with only a weekend set aside for learning it. There are two main methods of providing hypnosis in the context of pain management for childbirth: hypnotherapy delivered in person by a known practitioner, and self-hypnosis, in which the mother is previously trained to self-induce a state of altered consciousness. Under such a state, there is less awareness of the environment and an increased susceptibility to suggestion. These may be verbal and nonverbal communications used to achieve specific therapeutic goals.15 In the context of childbirth, suggestions may focus on increasing feelings of safety, relaxation, and comfort, as well as potentially developing sensations of numbness.43
Further studies are warranted to clarify the impact of hypnosis on labor pain. The evidence available is difficult to evaluate.93 Studies lack randomization, hypnotic methods are not well described and may vary between studies, and the study populations are relatively small. A recent Cochrane Database review examined seven trials with a total of 1213 women comparing the use of hypnosis during labor with other existing therapies.86 Such therapies included placebo use, no treatment, or any other analgesic drug or technique.43 The review authors found no statistical differences between women in the hypnosis group and those in the control group with regard to several outcomes: use of pharmacologic pain relief, spontaneous vaginal birth, and satisfaction with pain relief. There was some evidence of benefits for women in the hypnosis group compared with the control group for pain intensity, length of labor, and maternal hospital stay, although these findings were based on studies with small numbers of women.
Water Birth
The water birth movement holds that submersion for labor, delivery, or both reduces the sensation of pain. It also holds that birthing from one water environment into another is less stressful for the infant. Unfortunately, there are no substantive studies supporting this claim. Like most alternative methods, almost everything reported in the literature is anecdotal. A Cochrane Database review article indicates that immersion in water during the first stage decreases maternal requests for neuraxial analgesia and can be supported for women at low risk of complications.13 However, immersion during the second stage of labor needs further investigation. At present, there is no clear evidence whether to support or reject a woman's decision to give birth in water.13 Questions regarding infection and neonatal outcomes are not addressed, and large collaborative trials are needed to answer these critical issues. A 2004 review article considered three clinical concerns: water aspiration, neonatal/maternal infection, and neonatal/maternal thermoregulation in the practice of water birthing. They also looked at two practical concerns: skills and education of midwives or health professionals assisting the water birth, and emergency procedures in case of maternal collapse. They did not find sufficient evidence to caution women away from this practice.99
Acupuncture, Saline Injections, and Transcutaneous Electrical Nerve Stimulation
This group of techniques has most often been used to address the issue of low back pain during labor and not that of labor pain management in general. In controlled trials, acupuncture has been demonstrated to be beneficial85 but time consuming and without consistent results.84 In addition, the study sizes have been small, indicating that more research is needed.85
Intracutaneous or subcutaneous injection of sterile water is rapidly gaining popularity as a method of pain relief in labor. The outcomes reported severely limit conclusions for clinical practice.19 There is little robust evidence that sterile water is effective for low back or any other labor pain. Present studies found no difference in delivery or other maternal or fetal outcomes. Further large, methodologically rigorous studies are required to determine the efficacy of sterile water to relieve pain in labor.19
Transcutaneous electrical nerve stimulation (TENS) for the management of back pain involves the placement of electrodes over the sacrum. A low electrical pulse is then transmitted that modifies the sensation of pain. Again, this method has been demonstrated to work well for low back pain in general but does not lessen the request for other analgesics in labor pain management. There is insufficient evidence to make judgments on whether TENS is more effective than placebo or other interventions for pain management in labor. None of these aforementioned techniques appears to have any negative effect on the neonate.
Systemic Medications
There are two classes of drugs commonly used by obstetric providers in modern practice for labor pain management that fall under the scope of this segment. They are opioids for pain relief and sedatives to relax and reduce anxiety. Under emergent surgical conditions, anesthesiologists use other classes of drugs, but these are not reviewed in any detail here. When considering the effect of any medication on the fetus, it is worth noting whether that medication has produced a cerebral effect on the mother. If so, that medication has the properties to cross the lipid blood-brain barrier and likely will cross the placental barrier to reach the fetus as the necessary transport mechanisms are the same.100 However, the extent to which a fetus is exposed depends on many factors, such as the drug's pharmacokinetic and pharmacodynamic properties, the dose, and the mode of delivery. The following segment reviews a few of the pharmacologic principles pertinent to medication choices and fetal exposure. A more complete discussion can be found elsewhere or in any major pharmacology text.
Pharmacokinetics, Pharmacodynamics, and the Fetus
Three pharmacologic characteristics are considered when weighing whether or not a medication has the properties to enter the maternal circulation from its primary site of administration and then leave that circulation to cross the placenta (see Chapter 51). These are how much of the drug is in the ionized versus the nonionized state as determined by its pH, whether it has lipophilic or hydrophilic tendencies, and whether it prefers to bind to protein. A medication that is ionized cannot cross a membrane barrier. If a drug's pH is physiologic (pKa = pH = 7.4), it means that 50% of the drug is ionized and 50% is nonionized. For the purposes of discussion, local anesthetics are a class of weak bases (pKa = 7.6-9.1), so they are more ionized when exposed to the physiologic pH. The ratio by which a local anesthetic is ionized as opposed to nonionized in the circulation depends on how great is the base of the drug. For instance, a local anesthetic with a pKa of 9.1 is mostly ionized, and more will stay in the maternal circulation than a local anesthetic with a pKa of 7.6. Because it is the nonionized portion of a drug that can cross a membrane barrier, the amount that crosses will reproportion itself into ionized and nonionized forms when in the fetal circulation. If the fetal pH is lower than the maternal pH because of acidosis, more of the drug converts to the ionized form while in the fetal circulation and cannot return to the maternal circulation. If maternal exposure to the medication persists, more of the drug passes from the mother to the fetus, and increasing amounts accumulate in the fetal circulation. This phenomenon is called fetal ion trapping and has been associated with some of the deleterious effects produced by medications in the compromised fetus.
Of the other pharmacodynamic properties mentioned, drugs that possess lipophilic properties as opposed to hydrophilic are more capable of crossing lipid-rich membranes. Most medications used in obstetrics are lipophilic, but to varying degrees. For example, fentanyl and sufentanil are lipophilic opioids, but sufentanil is much more so. As a result, epidural doses of sufentanil more readily leave the epidural space to enter the maternal circulation than fentanyl.
Protein binding is the third important pharmacologic principle. A drug that binds with protein molecules remains in the maternal circulation because when bound, it becomes a bulky molecule. Only the unbound drug is free to cross a membrane and produce an effect. So excluding active transport mechanisms, it is the unbound, nonionized portion of a lipophilic medication that can cross a membrane barrier to produce an effect.
The pharmacokinetic effects of metabolism and elimination are yet another regulator of how much or even whether a drug will exert an effect on the fetal brain. Most medications undergo extensive metabolism into inactive metabolites in the maternal circulation. As a result, very little if any effect may be found. One example of a rapidly metabolized medication is succinylcholine, a depolarizing muscle relaxant. This medication is metabolized to benign metabolites by pseudocholinesterase in the maternal plasma and has a half-life of about 90 seconds. As a result, nothing reaches the fetus. However, if a drug is metabolized into an active metabolite in the maternal system and then crosses into the fetal system, that metabolite can have fetal effects. Such is the case with normeperidine, the active metabolite of meperidine.
A useful measurement that helps estimate fetal medication exposure is the ratio of the umbilical vein drug concentration to the maternal vein (UV : MV). A ratio of 1 (UV : MV = 1) means that the amounts of medication in the umbilical vein equal those in the maternal vein. A low ratio means that a small amount has crossed the placenta to reach the umbilical vein. For example, the nondepolarizing muscle relaxants used in general anesthesia are highly ionized, hydrophilic compounds that do not cross the membrane barrier. Their UV : MV ratios tend to be on the order of 0.1. Table 28-1 lists the UV : MV ratios of some common anesthetics.
TABLE 28-1
Placental Passage of Commonly Used Anesthetic Medications
| Drug | Umbilical Vein to Maternal Vein Ratio (UV : MV) |
| Induction Agents | |
| Thiopental | 1.08 (range, 0.5-1.5) |
| Ketamine | 0.54 (range, 0.4-0.7) |
| Propofol | 0.7 |
| Etomidate | 0.5 |
| Nondepolarizing Neuromuscular Blocking Agents | |
| Pancuronium | 0.19 |
| Vecuronium | 0.11 |
| Opioids | |
| Morphine | 0.92 |
| Meperidine | 0.81 (may exceed 1.0 after 2-3 hours) |
| Fentanyl | 0.57 |
| Sufentanil | Levels too low to measure in humans |
| Butorphanol | 0.84 |
| Nalbuphine | 0.97 |
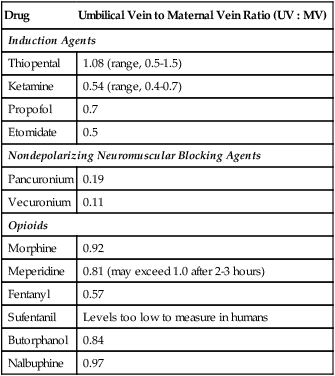
Adapted from data from Glosten B. Anesthesia for obstetrics. In: Longnecker DE, et al., eds. Principles and practice of anesthesiology, 2nd ed. St. Louis: Mosby; 1998.
Even if the umbilical vein levels are significant, they may not be great enough to produce any important neurobehavioral effect because two more protective barriers exist to shield the fetal brain from medication exposure. Approximately 40% to 60% of any medication entering the fetal circulation passes through the liver first and then travels through the inferior vena cava to the heart and into the circulation. The mature or nearly mature fetal liver can metabolize most drugs, and this first pass through the liver buffers the fetal brain from exposure.27 Additionally, the unique fetal circulation dilutes most of any drug as it travels into the general circulation, and this dilutional effect can be quite protective of the fetal brain.27
One final clinical note for the neonatologist: Not only does the dose have an impact on how much medication enters the maternal circulation for eventual fetal distribution, but so does the mode of administration. Although it is obvious that intravenous administration achieves the highest maternal blood levels, all blocks and injections distribute some level of the drug into the maternal circulation, and they differ markedly from one another. From the greatest to the least effect on maternal blood levels, drug administration modes have the following ranking27:

It should be noted that maternal local anesthetic levels after a spinal delivery are so low as to be clinically irrelevant.
Sedatives
The routine use of heavy sedatives in the active phase of labor has markedly diminished, making it unlikely that a neonatologist will be caring for a neonate with a recent exposure. In modern practice, the primary purpose of sedatives is to help the parturient rest during a prolonged latent phase of labor, as a sleep aid before starting an induction in a few hours, or as administered during a cesarean section either to an anxious patient or before the induction of a general anesthetic. Other than in the cesarean section scenario, enough time usually passes between administration and delivery so that any significant drug amounts clear the maternal and fetal circulations. The classes of medications most often encountered today are benzodiazepines.
Benzodiazepines are most likely encountered in an acute situation either because of maternal self-administration or from the anesthesia provider during a cesarean section. The two most commonly encountered benzodiazepines are diazepam and midazolam. Midazolam has a low UV : MV ratio, so it does not cross to the fetus well at all, making it essentially without clinical effect when given acutely. The most common use of midazolam is during a cesarean section that is unplanned. It is given by the anesthesia provider, and although a very good anxiolytic, it unfortunately causes maternal amnesia that will impair the mother's recall of the delivery if given beforehand. Therefore it is usually reserved for the highly anxious patient, and most providers attempt to delay administration until after delivery. Unfortunately diazepam has a high UV : MV ratio that reaches 1 within minutes and can increase to 2 within hours.100 Metabolites are active and can remain in the system for up to 8 days. When given in the intrapartum setting for eclamptic seizures, this medication can cause hypotonia, hypothermia, and respiratory depression in the neonate.21,46,94 Once thought to cause oral cleft malformations when used chronically during the first trimester, more recent prospective studies do not support this assertion.24,78 Regardless, diazepam is not as commonly prescribed by the obstetric provider as it once was, and its use is usually limited to the acute management of maternal seizures.
Barbiturates are used less commonly than before. Phenobarbital is rarely used, but remains an option for treatment of partial or generalized tonic-clonic seizures and status epilepticus.21 Thiopental is another barbiturate that used to be a common anesthetic induction agent; however, propofol is now more frequently used. The two have similar profiles as far as neonatal effects are concerned.
Some clinicians, even more in the past, felt that sedatives such as phenothiazines had value in early labor in reducing a woman's anxiety and promoting sleep, and thereby contributed to pain relief.60 Phenergan, also used for its antiemetic qualities, could be useful if there is a need to treat vomiting as well as provide pain relief.60
Opioids
The definition of an opioid is any natural, synthetic, or semisynthetic compound that acts on the same receptors as morphine and produces similar effects. Those agents that stimulate opioid receptors are known as pure agonists and include morphine, meperidine, and fentanyl. Those that act on only some of the receptors and may even block action on others are known as agonist-antagonists and include nalbuphine and butorphanol. Pure antagonists block action on all receptors and are not considered opioids but can reverse their actions. Naloxone defines this class.
Opioids do not block the transmission of pain, but rather stimulate receptors to alter the perception of pain and one's response to that perception. For that reason, opioids are considered incomplete analgesics. There are three known opioid receptor types and a possible fourth on which these drugs act.58,79 The µ-receptor provides the most complete analgesia, but also most of the known side effects of opioids such as pruritus, nausea and vomiting, euphoria, dysphoria, and respiratory depression to the point of apnea. The κ-receptor mediates less intense analgesia because of what is known as a “ceiling effect.” The dose-response studies performed early in the investigation of medications that stimulate this receptor determined that the curve stopped rising at a particular dose. This flattening of the dose-response curve means that more of the drug does not produce more of an expected effect. This “ceiling effect” holds not only for analgesia but also for respiratory depression. No other side effects appear to be associated with this receptor type. Both the µ- and κ-receptors have been divided into multiple subgroups. The δ-receptor mediates the effects of the endogenous endorphins, especially as they act on the spinal cord. The final receptor, named the σ-receptor, may be responsible for some of the side effects occasionally seen with some opioids. However, the existence of this receptor is dubious. Box 28-1 lists some of the opioids commonly used in labor and their classifications.
Opioids appear to be more effective on visceral pain than somatic pain.58 As a result, they are more useful during the first stage of labor, which tends to be visceral in nature, than the second stage, which is more somatic.41 Because all opioids can cross the placenta and produce an effect in the fetus, systemic opioid use has traditionally been limited to the first stage so that the opioid is metabolized from the fetus before delivery. However, not only has the selection of opioids expanded over the years, but also the thoughts on how they can be delivered. Intermittent intravenous administrations are still the mainstay on most units, but now intravenous patient-controlled analgesia (IV-PCA) is being offered on some obstetric units. Once thought to be taboo because of the depressive effects of opioids on the neonate, studies show that this administration technique can be provided reasonably well.63,83 But most agree that because of the neonatal effects, this method should be reserved for situations in which a regional technique is either not available or contraindicated.24
There are several reports in the literature describing the IV-PCA technique with meperidine, fentanyl, nalbuphine, alfentanil, and remifentanil.61,63,83,89 Those neonates whose mothers use this technique should be watched for signs of sedation, respiratory depression, and low oxygen saturation.
Opioid Agonists
Opioid agonists that may be used on the obstetric unit are morphine, meperidine, fentanyl, sufentanil, and remifentanil.88 Although formerly used, studies have shown that alfentanil is less effective than other opioids, such as fentanyl, for first-stage labor analgesia. In addition, when used in doses of 10 µg/kg in the mother, it can cause severe neonatal respiratory depression.45 Morphine is the only naturally occurring agonist and, if used at all during labor, is used early. The reason is that not only is it more depressive than any other opioid to the neonate, but it may also depress uterine contractions, making it a questionable choice for labor management.79
Meperidine (Demerol) is a synthetic opioid with some very interesting properties. This opioid is the one most commonly used systemically, and there is evidence that it enhances the effacement and dilation of the cervix.48 It has atropine-like properties that can produce tachycardia and myocardial depression in the mother. Also, when injected into the epidural or spinal space, it can produce a block similar to that seen with local anesthetics.12 For this reason, there are case reports that describe the use of meperidine in this manner in patients with a documented allergy to all forms of local anesthetics.
When used to provide systemic analgesia, meperidine is traditionally dosed in such a way as to limit fetal exposure before delivery. The drug's duration of action is 2 to 4 hours, with a half-life of 3 to 4.5 hours. Based on that and knowing that intramuscular administration requires about 45 minutes to an hour to absorb and have an effect, meperidine is not given unless delivery is expected within the hour or beyond 3 hours. Unfortunately, estimates of delivery time are an imprecise science, and sometimes the neonate is delivered during peak exposure. Even so, the need for naloxone to reverse the depressive effects is uncommon under this scenario. In any case, it would probably be best to closely observe neonates exposed to this form of analgesia. Meperidine is metabolized to an active metabolite called normeperidine. The fetal liver is capable of metabolizing meperidine into its active metabolite, or it can cross the placenta and accumulate. Its half-life is 15 to 40 hours, so its neurologic and depressive effects can be evident for some time after delivery.
Several commonly used opioids are analogues of meperidine. They are fentanyl, sufentanil, alfentanil, and remifentanil. Although only fentanyl and sufentanil are used for anesthetic nerve blocks, fentanyl, alfentanil, and remifentanil have been used systemically in the obstetric setting. All these meperidine analogues are lipid soluble and can cross the placenta. However, they are rapidly metabolized by the first pass through the liver to inactive metabolites, thereby making any depressive effects uncommon when these drugs are given as intermittent intravenous boluses.68 Because of its pharmacokinetic properties, remifentanil is so short-acting that it can be used only in a continuous infusion mode. In one study, fentanyl was injected as a maternal intravenous dose of 1 µg/kg just before the start of the cesarean section, and no neonatal depressive effects were detected.61
Fentanyl has been used in the IV-PCA format,49,76 as have alfentanil49 and remifentanil,24,36,61 with mild neonatal depression a common occurrence. This mode of administration is growing in use as more is reported in the literature and obstetric providers find it a more acceptable alternative to intermittent injections when anesthetic services are limited or neuraxial blocks are contraindicated. Remifentanil in particular may be well suited to the IV-PCA format because its half-life is only 4 minutes. Generally, oxygen supplementation and observation are all that is required for the neonate after such exposure.37,75
One final note about the opioid agonists: All of them are associated with the suppression of fetal beat-to-beat variability, which returns to normal as the drug is eliminated, suggesting a causal effect.
Opioid Agonist-Antagonists
All medications in this category stimulate the κ-receptor and thus have a ceiling effect on respiratory depression, as previously described. This information gives providers a sense of safety. However, the ceiling effect also applies to analgesic capabilities, making this class of drug limited in its effectiveness. The two most commonly used formulations on obstetric units in the United States are nalbuphine (Nubain) and butorphanol (Stadol). Both produce sedation in the mother and degrade to inactive metabolites, and although both cross the placenta, the fetal effects seem limited.27,58 However, nalbuphine reduces fetal beat-to-beat variability and can produce a sinusoidal pattern when used.20
It is important to note that these two medications are markedly different in how they act on the µ-receptor. Whereas butorphanol has essentially no effect on the µ-receptor, nalbuphine actively blocks it. As a result, it can be used in small doses in lieu of naloxone to counter µ-receptor side effects such as pruritus. The danger with nalbuphine is that it can place a narcotic-addicted patient into acute withdrawal at doses commonly used for labor analgesia. It can do the same to the addicted neonate.27,58 Therefore, caution should be exercised when using this medication if there is any indication of narcotic abuse in the mother.
Because of the sedative and respiratory depressive effects of these drugs as well as the inability to produce complete analgesia, heavy intrapartum use can lead to a situation in which maternal hyperventilation during a contraction is followed by hypoventilation as the contraction recedes. This condition will produce predictable results for maternal oxygen and carbon dioxide levels as well as those of the fetus. The end result can be significant fetal acidosis that begins during the first stage and worsens during the second.50,53,71
Opioid Antagonists
Although other antagonists exist, naloxone (Narcan) is used on obstetric units almost exclusively. This medication blocks the action of an opioid on all of the receptors and is commonly used to reverse the adverse effects of side effects and excessive amounts. It has a short half-life (30-45 minutes), which means that repeated boluses may be necessary in some instances. Naloxone is not a benign drug and has been known to cause pulmonary edema and cardiac failure in some situations, so caution should be exercised when using it.58 It crosses the placenta, so it will reverse any opioid effect in the fetus. As with nalbuphine, it is not a medication that should be given to a mother with a narcotic addiction. Not only will it produce withdrawal in the mother, but in the addicted fetus as well.50,58
Obstetric Nerve Blocks
Obstetric nerve blocks are invasive procedures to achieve analgesia that are performed by the obstetric provider managing the labor. There are only two nerve blocks used in obstetric practice, and these are used for different goals. The paracervical block provides relief for pain in the first stage of labor, whereas the pudendal block is used for analgesia during the delivery portion of the second stage.
Paracervical Blocks
The purpose of the paracervical technique is to block pain transmission via the visceral afferent fibers near the dilating cervix. This objective is reached by injecting local anesthetic lateral to the cervix at the vaginal fornices. Lidocaine without epinephrine is preferred in the United States because of its relative safety.77 In the early 1980s, US manufacturers of bupivacaine specifically contraindicated the use of the agent for paracervical blockade because of its risk for maternal cardiotoxicity.77
Paracervical block (PCB) was described by Europeans in the early part of the twentieth century and was first reported to be useful in providing labor analgesia in the United States in 1945. The technique became popular in the United States in the early 1960s.77 Numerous early case series, case reports, and clinical studies demonstrated good first-stage labor anesthesia in approximately 75% of women.77 However, careful reviews of the early reports also describe a post-blockade fetal bradycardia. Although the bradycardia has been described to be of a short duration and usually without adverse sequelae, fetal deaths have been reported.77 Some of these deaths resulted from direct injection of local anesthetic into the fetal head, resulting in profound fetal systemic toxicity. Other theories about the etiology of the fetal bradycardia included aortocaval compression caused by maternal positioning for the block, paracervical manipulation, increased uterine activity, and local anesthetic-induced constriction of uterine blood flow in the area adjacent to the nerves blocked.77
Paracervical blocks are less commonly performed for labor pain in the United States today than two decades ago. Studies show that parturients receiving paracervical block often continue to receive subsequent analgesia.34 Parturients who received paracervical blocks over neuraxial analgesia were less satisfied and less willing to have the same labor analgesia again.34 However, this technique might be useful when neuraxial blocks are not available or contraindicated.39 Further high-quality studies are needed to confirm the findings, assess other outcomes, and compare local anesthetic nerve blocks with various modalities for pain relief in labor.56
Pudendal Blocks
A pudendal block may be used for analgesia in the late stages of labor and is most typically used when an operative delivery with forceps is planned.80 The pudendal nerves are blocked using a transvaginal approach in which 5 to 10 mL of local anesthetic is injected at the site of the attachment of the sacrospinous ligament to the ischial spine. A significant potential complication of the procedure is maternal intravascular injection, so aspirations are frequently performed. Despite the avoidance of maternal injection and the typically short interval between injection and delivery, blood levels of the local anesthetic may be found in the neonate for up to 4 hours after delivery. However, studies included in a Cochrane analysis from 2012 found no significant adverse neonatal effects (based on an Apgar score of less than 7 at 5 minutes).56
Neuraxial Blocks
Any variety of epidural, spinal, or combination thereof falls under the general term neuraxial block. These nerve blocks are performed by an anesthesia provider and are the preferred techniques for cesarean deliveries because maternal airway management is avoided, sensorium is unimpaired, and little or no medication reaches the fetus. Any manner of neuraxial block is commonly offered to parturients when labor is established. The versatility of these blocks makes them the ideal choice, as how dense or sparing the block is determined by which medications are used and in what manner they are dosed. In most situations, only an analgesic state is desired; however, on occasion a denser block more closely resembling an anesthetic is preferred. This technical nuance defines two very different goals for the anesthesia provider. An anesthetic is that method designed to remove all sensation—pain, temperature, touch, vibratory sense, proprioception, and motor ability. The goal of an analgesic is to eliminate only the sensation of pain, leaving all other components intact. Logically, higher doses are more likely to produce the anesthetic state, whereas lower doses may produce only an analgesic state. This can be a difficult goal to achieve because individual patient needs vary greatly and small variations in dose can determine the difference between anesthesia and analgesia, as well as analgesia and no pain relief.
Techniques
How the most basic neuraxial blocks are administered has evolved over time to include several variations, of which the combined spinal-epidural and the patient-controlled epidural analgesic are just two. New on the horizon is the automated intermittent bolus approach that uses newer technology to administer boluses to the patient at preset intervals. It is not the purpose of this section to describe all aspects of these procedures in detail, but neonatologists should be aware of their most fundamental facts. A more in-depth description may be found in any anesthesia obstetric text.
Lumbar Epidurals
Lumbar epidural analgesia is usually initiated in either the sitting or lateral position.74 To block the necessary nerves that transmit labor pain in the first stage (T12-L2), the back is examined for the L4-5, L3-4, or L2-3 interspaces. A placement at any of these three levels will produce the necessary nerve block. The needle is removed over the catheter, which is tested with lidocaine or bupivacaine with or without epinephrine to rule out intrathecal or intravascular catheter placement. Once the catheter is tested, adequate labor analgesia is established by bolus injection of the selected anesthetic. Analgesia is maintained with any combination of intermittent bolus injections and/or a continuous infusion. There are numerous drug combinations, concentrations, and infusion rates cited in the literature, but the prevalent trend today is to use dilute local anesthetic concentrations most often in combination with a low dose of opioid in an attempt to maintain an analgesic state. This also improves the safety of the technique in that lower doses mean there is less of a chance of a deleterious effect on either the mother or fetus should the catheter migrate into either a vessel or the spinal space. The catheter is removed after delivery when there is no further need for analgesia or anesthesia.
Patient-Controlled Epidural Analgesia
First described for use in labor by Gambling et al. in 1988, patient-controlled epidural analgesia allows the parturient to self-administer intermittent boluses of epidural medication, thus providing flexibility to accommodate changing analgesic requirements as labor progresses.67 The anesthesia provider adjusts the patient-controlled epidural analgesic program settings such as demand bolus, lockout interval, and hourly maximum rate for individual patients.67 Practices vary on whether a background infusion is used, but when included, will often be set at a lower rate than would be set for a conventional infusion. Patient-controlled epidural analgesia allows better dose-demand matching as labor progresses, and has been shown to be associated with up to a 35% reduction in total volume of local anesthetic, especially in the first stage of labor.67
Intrathecal Injections (Spinals)
The word spinal is often replaced by the more accurate terms intrathecal and subarachnoid when anesthesia providers discuss the technique. Regardless of which term is used, intrathecal injections most often convey an image of complete paralysis and anesthesia from somewhere in the thoracoabdominal area on downward. However, with modern dosing techniques, a spinal as is performed for labor analgesia can preserve temperature, touch, proprioception, and even mobility while providing pain relief. The procedure is the same as that described for an epidural with the exception that a smaller, stiletted 25- to 27-gauge needle is used and a catheter is not left behind. The stylet is not removed until a “pop” is felt as the needle passes through the dura into the spinal space. Once the stylet is removed and the presence of cerebrospinal fluid (CSF) confirmed, the medication is injected and the needle removed.
Intrathecal injections are associated with the lowest maternal blood levels of all the analgesic methods used, resulting in clinically irrelevant fetal exposure. The most commonly used opioids for the induction of labor analgesia are highly lipophilic and travel to the µ-receptors within the central nervous system. As a result of this spread, maternal somnolence and even respiratory depression have been reported with small doses because the opioid acts on these receptors.30 Not appropriately managing this maternal complication will have an effect on the fetus, but there is no direct effect on the fetus from the opioid as a result of the intrathecal injection. Although intrathecal opioid injections have been tried as the sole labor analgesic, they are not very effective beyond the first stage, and they may not last long enough for the labor. Additionally, should there be a need for an operative delivery or cesarean section, another anesthetic procedure would be required. To resolve this issue, spinals are now often combined with epidurals.
Combined Spinal-Epidurals
With the combined spinal-epidural technique, both an epidural and spinal are performed at the same interspace so that only one local injection is required. The spinal needle may go through or under the epidural needle, or a special needle designed for this technique is now available. When the epidural tip is situated in the epidural space and before the catheter is threaded, the spinal portion is performed. After the intrathecal injection, the spinal needle is removed, the epidural catheter is threaded into the epidural space, and the epidural needle is removed. This procedure allows for a rapid analgesic response (the spinal), with the ability to bolus and infuse through the epidural catheter for longevity. An adaptation is to not administer a dose through the epidural catheter until the intrathecal effects begin to wear off.
Combined spinal-epidural analgesia has become increasingly popular over the past decade. Onset of analgesia is significantly faster with combined spinal-epidural compared with epidural analgesia by a factor of four (2-5 minutes vs. 15-20 minutes).97 Despite studies that have addressed many of the theoretical concerns with the technique, not all anesthesia providers embrace the technique. However, most agree that there are some parturients for whom it is better suited, such as the rapidly progressing multiparous patient who might not be offered any analgesic otherwise. Despite studies showing that epidural catheters placed during a combined spinal-epidural technique are more likely to function properly,67 the most consistently perceived detriment with the technique is that the epidural catheter may be untried. Should a problem arise, anesthesia providers may forgo using the untried catheter and subject the patient to a general anesthetic. Although the combined spinal-epidural and patient-controlled epidural analgesic have a slower onset of pain relief than a combined spinal-epidural, the epidural catheter is tried and functional should there be a sudden change in delivery plans.
Automated Intermittent Bolusing
The development of an automated intermittent bolusing (AIB) technique for maintenance of epidurals is based on studies that suggest that intermittent boluses, rather than continuous infusions, result in lower local anesthetic consumption, decrease the need for clinician-assisted epidural supplementation, and produce higher patient satisfaction.40,98 This may be because of the higher driving pressure generated by the bolus through the orifices of an epidural catheter at set times, which results in better dispersion of medication than can be achieved with a slow and steady continuous infusion.40 The use of intermittent boluses results in a more uniform epidural block compared with a similar hourly rate of continuous infusion.40 However, the optimal combination of bolus volume and dosing interval has yet to be determined as well as the marketable technology.40,98
Maternal Risks and Benefits
Benefits
Every study that compares the analgesic effect achieved with a neuraxial block against any other form of analgesic technique, whether it is pharmacologic or nonpharmacologic, documents a superior result.4 When done properly, these blocks allow the mother to relax and conserve her energy for the second stage of labor when she should have enough motor preservation to participate in that process. They also prevent the maternal hyperventilation often seen in response to pain and thus all the potential sequelae on the fetus associated with lower maternal Pco2 levels.
Although the medications used with an epidural can eventually be found in the maternal circulation, measured blood levels are low. The current trend is toward opioid and very dilute local anesthetic concentrations that further help to keep maternal blood levels to a minimum. As a result, mothers typically show very little if any systemic effect from the administered medications, and such low maternal levels usually indicate very limited exposure to the fetus.27
Current studies have shown either no change or an increase in uteroplacental blood flow after epidural analgesia, provided hypotension is avoided or promptly treated.23 Conversely, pain and the stress of labor may release excessive catecholamines into the circulation, which in turn decrease uteroplacental blood flow because of sympathetic stimulation.44 Adequate pain management with neuraxial analgesia decreases the level of circulating catecholamines. Moreover, it avoids maternal hyperventilation, which leads to maternal hypocapnia.4 Maternal hypocapnia causes uterine vasoconstriction, which reduces blood flow to the fetus if excessive.
Maternal Side Effects
Common side effects of neuraxial analgesia are hypotension, pruritus, and some degree of motor block. Of these, hypotension and pruritus are the most frequent.97 Hypotension is the result of local anesthetic block of the sympathetic nervous system, leading to vasodilation, increased venous capacitance, decreased preload, and decreased cardiac output. It is common after any neuraxial block but frequently more severe after a spinal. It is also more common after an anesthetic dose as opposed to an analgesic one. If the hypotension is allowed to persist untreated, there are significant consequences to both the mother and fetus. A decrease in maternal blood pressure would result in a decrease in uteroplacental perfusion. Thus, it would be below the standard of care not to treat hypotension when it occurs. To avoid hypotension, the mother is positioned laterally or in a semirecumbent position to prevent aortocaval compression, and decreases in blood pressure are treated with fluid boluses or intravenous vasopressors.
Pruritus is more common after spinal analgesia. The cause is unknown, but it is opioid-induced rather than histamine-related, and the incidence and severity are dose-dependent.97 It is usually self-limited, but if severe, it can be treated with an opioid antagonist such as naloxone or the agonist-antagonist nalbuphine.
Although the goal of an analgesic dose is to avoid a motor block, some level of weakness is likely if a local anesthetic is part of the dosing mixture. As a result, the policy at many institutions is to confine the parturient to bed after the neuraxial block is placed. Although the idea of a “walking epidural” was popular some time ago, studies show that ambulation per se does not alter the labor outcome96 and that most women did not care to get up once they were comfortable. As a result of this and the fears of potential liability should a fall occur, many providers and institutions have stopped offering this option.
Other irritating side effects occur primarily because of the opioid used. Besides the pruritus, they include nausea, vomiting, sedation, and urinary retention. All are caused by stimulation of the µ-receptor (opioid) and will dissipate with time. The only hazardous opioid-induced side effect is respiratory depression, and although it is rare, health care providers always should be alert for its presentation.
Maternal Risks
The anesthetic and nonanesthetic literature is full of discussions of documented as well as theoretical maternal risks resulting from neuraxial blocks. Although some are uncontested, such as epidural hematoma formation or nerve damage, others are quite fantastic, such as a reduction in the IQ of the neonate. The wide-ranging list of maternal risks includes unintentional intrathecal or intravascular injection, prolonged labor of either the first or second stage, backache, nerve damage, infection with possible abscess formation, epidural or subdural hematomas, arachnoiditis, an increased incidence of operative delivery, maternal temperature elevation, a cervical level that can affect respirations, and an impact on the cesarean section rate. Because the risks of backache, nerve damage, infection with possible abscess formation, epidural or subdural hematomas, and arachnoiditis are postdelivery problems, the reader is referred to any obstetric anesthesia text for a full discussion there. The following section considers only the maternal risks that are documented or have undergone scientific scrutiny, and pose a potential problem for the fetus or neonate. Of those left, some are significant, whereas others are of questionable clinical significance. Other mentioned “risks” are documented to no longer be associated with neuraxial blocks but are mentioned here because the reader may not be aware of the newer literature. Such is the situation with the rising cesarean section rate.
Cesarean Sections.
The effects of neuraxial blocks on the progress of labor, and particularly on the mode of delivery, have generated tremendous controversy in recent years. Selection bias confounded many studies, especially those that claimed epidurals increased the incidence of cesarean section. Now studies have determined that there is an association between epidurals and cesarean sections, but not causation.82 The association occurs because in situations of a prolonged or dysfunctional labor, women are more likely to request epidural analgesia. All agree that dysfunctional labors are prolonged and very painful. Because of that, women in dysfunctional labor patterns are more likely to request some form of neuraxial block. But dysfunctional labors are also associated with a greater cesarean delivery rate.92 Thus, current literature has shown that neuraxial blocks are more frequently requested once a dysfunctional labor pattern is in place. It is this labor pattern, not the epidural, that places the patient at risk for cesarean delivery.81
Labor Prolongation.
Prolongation of labor has long been blamed on neuraxial blocks. This claim is still undergoing some debate. Two meta-analyses and a Cochrane review reported no difference in the duration of the first stage of labor in women receiving epidural analgesia versus those receiving systemic opioid analgesia or no analgesia at all.9 Conversely, a 2004 meta-analysis claimed that epidural analgesia increased the length of first stage of labor by approximately a half-hour.84 One offered explanation of this result involved the frequency of cervical examination in these studies. Full cervical dilation is considered the end point of the first stage of labor. This would only be determined by cervical examination or when the parturient complains of rectal pressure, which is likely to be later in a woman with effective neuraxial analgesia.9 Thus, exams may be more frequently performed in the presence of an epidural to catch this end point. Clinical trials by Wong and Ohel found that the duration of the first stage of labor was significantly shorter when any form of neuraxial analgesia was administered early in labor.9 As for the second-stage prolongation, it is generally acknowledged that there is a statistically significant average increase of about 15 minutes in the presence of an epidural.9,81 Whether that amount of time is clinically relevant is debatable. Several studies suggest that a prolonged second stage of labor does not result in adverse maternal or fetal outcomes provided that the fetal status is reassuring, the mother is well hydrated and has adequate analgesia, and there is progress in descent of the fetal head.9 Also, whether pushing is initiated immediately upon recognition of full cervical dilation or delayed until descent of the head causes an urge to push can influence the duration of time in second stage. The American College of Obstetricians and Gynecologists (ACOG) recommends that if an epidural is in place and there are no signs of fetal compromise, the second stage should be allowed to continue until the fetus has descended to a lower fetal station to avoid maternal exhaustion.
Operative Vaginal Deliveries.
Neuraxial blocks have been associated with an increased need for operative vaginal deliveries. When studies report on the overall incidence of forceps deliveries and epidurals, there is no question that the incidence increases when an epidural is in place. However, multiple confounding factors contribute to these findings such as high doses of local anesthetic that will relax pelvic muscles, thereby preventing proper rotation of the fetus for delivery, the method of epidural analgesia maintenance, and obstetric factors.9 Why an obstetrician chooses a forceps delivery is not always well defined. For example, an obstetrician may request an epidural placement rather than perform a pudendal block if he or she feels that forceps are indicated for delivery. This may occur either because the obstetrician is uncomfortable performing a pudendal block or prefers the superior analgesia an epidural provides. As a result, the birth is recorded as a forceps delivery under an epidural. Impact studies have also been helpful in that they document that the introduction of a new epidural service does not increase the incidence of forceps deliveries for obstetric reasons. This strongly suggests that they have no effect.28 However, until more well-controlled studies are performed in which the indication for the forceps delivery is documented, it will be difficult to determine precisely what the risk is or even if one actually exists.
Maternal Temperature Elevation.
The gradual development of modest hyperthermia observed in laboring women with epidural analgesia is not seen in those electing other forms of analgesia or unmedicated labor.81 Selection bias confounds the association between epidural analgesia and fever because women at risk for fever, which may be caused by longer duration of ruptured membranes, longer labor, more frequent cervical examinations, and other interventions, are also more likely to select epidural analgesia.81 However, even randomized trials have confirmed a higher incidence of fever in epidural-exposed women, suggesting a causal relationship. The mechanisms of epidural-associated fever remain incompletely understood.81 Altered thermoregulation and an antipyretic effect of opioids given to women without epidural analgesia may explain part of the phenomenon, but the most likely etiology is inflammation, most commonly in the placenta and membranes (chorioamnionitis).81 The consequences of maternal fever are diverse. Obstetricians are more likely to intervene surgically in laboring women with fever, and neonatologists are more likely to evaluate neonates of febrile women for sepsis. Studies that looked at the neonatal results reported that there was no increased incidence of sepsis despite the maternal temperature elevation.28 As a result, many neonatal units have adjusted their policies to require other clinical indications besides maternal temperature before a neonatal sepsis workup is initiated.
Inadvertent Injections and Excessively High Levels.
Inadvertent intrathecal or intravascular injections when a neuraxial block is performed can have a serious impact on the fetus. The epidural space is a vascular as well as a potential space. Possible catheter or needle complications include placement into a vessel or through the dura into the intrathecal space. Unrecognized misplacement with subsequent injection of a large amount of local anesthetic can cause maternal hypotension, seizures, and cardiovascular collapse from an intravascular injection or respiratory compromise that may lead to apnea from an intrathecal injection.38 When an unrecognized intravascular injection occurs, the acute insult to the fetus appears to be caused by the maternal hypoxia97 that develops during the seizure or arrest and not the high levels of anesthetic in the maternal system, although some of it is sure to cross over. When a high block happens because of improper dosing or an unrecognized intrathecal injection, maternal respiratory muscles become paralyzed and inadequate respirations, including apnea, can occur. If this condition is not recognized and treated, the maternal arrest will have obvious consequences on the fetus. Fortunately, both of these complications are exceedingly rare and very preventable with attention to good technique.
Benefits and Potential Risks for the Fetus
When investigating different methods of maternal pain relief in labor, neonatal outcome has not always been at the forefront.72 Unfortunately, many laypersons and some health care providers assume that for every negative maternal effect, there must be an associated negative fetal or neonatal effect. However, the literature does not support such a presumption. Those studies that examine the fetal and neonatal effects typically report on the presence or absence of base excess to determine the recent intrauterine environment, the intrapartum fetal heart rate (FHR), the Apgar scores, and a host of neuroadaptive examinations. These neuroadaptive examinations often require operator training and have varying abilities to determine any prolonged effect on the neonate from an intrapartum event or medication. For example, the Neurologic and Adaptive Capacity Score (NACS) has poor sensitivity to distinguish between hypoxic and drug effects in the neonate.8,29 The following section discusses the effects of neuraxial block on the fetus and neonate as defined by the aforementioned indicators and concludes with a brief review on breastfeeding.
Acid-Base Balance.
One of the most notable benefits a fetus gains from a neuraxial block in the mother is that she no longer hyperventilates in response to painful contractions. Maternal hyperventilation in response to pain has long been known to have adverse fetal effects.72,73 Maternal and fetal CO2 levels are closely associated, and maternal hypocapnia as a result of hyperventilation leads to a maternal and fetal respiratory alkalosis resulting in a left shift in the oxygen dissociation curve. This leads to impaired placental transfer of oxygen. Furthermore, a compensatory metabolic acidosis develops in the maternal side and may be conveyed to the fetus, resulting in worsening acidosis and hypoxia as labor progresses. With a functioning neuraxial block in place, these events are less likely to occur, and studies have consistently shown an improved fetal acid-base balance when an epidural is used.72,73 Literature shows that fetal acid-base status is not only better with epidural than with systemic opioids, it is also better than with no analgesia.73 Neonatal base excess measurement at delivery is believed to best reflect the intrauterine environment just before delivery, and studies comparing epidurals against systemic medications or nonmedication labor management methods have documented that neonatal base excess levels stay within normal limits more consistently with epidural use than they do with other techniques.71 Other findings are that amounts of meconium are less or similar upon delivery, and Apgar scores are comparable or improved when a functioning epidural is in place. These findings further support the idea of a better intrauterine environment and less stress on the fetus with an effective neuraxial block present.
Fetal Heart Rate.
An alteration in fetal heart rate, most notably fetal bradycardia, can occur in approximately 10% to 12% of those parturients receiving an epidural, and has been a long-recognized phenomenon. It can occur within 15 to 45 minutes after initiation of any form of neuraxial analgesia. It has been hypothesized that as analgesia is established, there is an acute decrease in maternal plasma epinephrine levels. This acute decrease results in a temporary imbalance of uterine tocolytic and tocodynamic forces, resulting in uterine hypertonus, decreased uterine perfusion, and ultimately, fetal bradycardia.97 The bradycardia usually resolves with conservative therapy and is often significant enough to initiate intrauterine resuscitative measures, but rarely leads to an early delivery.10 Apgar scores are typically fine upon delivery.
The use of neuraxial opioids can also produce an effect on the FHR. Loss of beat-to-beat variability can be attributed to large doses of neuraxial opioids, but this side effect is also seen with systemic opioids and agonist-antagonist opioids. A comparison of analgesic techniques shows that FHR changes are more common with systemic medications.27 For instance, compared with epidural bupivacaine analgesia, use of systemic meperidine for analgesia is associated with a greater loss of FHR variability and fewer accelerations. More newborns of mothers receiving intravenous versus neuraxial opioids had lower 1-minute Apgar scores and needed naloxone and resuscitation.72,73 Although studies show that the neuraxial opioid effects on FHR are temporally limited, when very high doses are delivered repeatedly to the epidural space, enough can be absorbed systemically by the mother and cross the placenta. This can result in high levels in the neonate at birth and require the use of naloxone. As a result, anesthesia providers should be careful with the timing and use of neuraxial opioids during the second stage of labor.
Breastfeeding.
A frequent claim made by those opposed to the use of epidurals is that this mode of pain relief will temporarily or permanently damage the neonate's ability to breastfeed.90 Precisely how this occurs has never been elucidated, and those hypotheses that have been proposed and seemed plausible have not withstood scientific scrutiny. Studies reporting on the effect of neuraxial medications on breastfeeding have not been randomized, and those purporting to show adverse effects of epidural analgesia received disproportionate publicity.72,73 One of the most common theories is that medications used in the epidural space enter the maternal circulation, reaching such high levels that enough crosses to the fetus, where it remains in circulation after delivery and produces an adverse effect. The other common hypothesis is that high maternal systemic concentrations from epidural injections seep into the breast milk and affect the neonate. Reynolds reviewed 17 breastfeeding studies, from 1994 to 2010, looking at the effects of neuraxial blocks on breastfeeding versus other or no analgesia.72,73 Only two studies showed a dose-related detrimental effect of epidural fentanyl, but these findings were not confirmed by other retrospective studies.72,73 A Canadian study from 2010 showed that, with good support, a breastfeeding success rate of greater than 95% can be achieved after epidural bupivacaine plus fentanyl, with no discernible dose-related effect.72,73 Anecdotally, some of the patients who had undergone labor without an epidural were so exhausted after delivery that they were not able to breastfeed, whereas those who had an epidural were alert.
Other studies have not measured levels but examined whether nursing policies that require a different postpartum management of patients with epidurals might have a negative influence on breastfeeding behaviors. These policies were found to minimize early postpartum maternal-infant contact. Furthermore, where early maternal-infant separation is minimized, epidural analgesia was found to have no effects on lactation success.72,73 Although there are numerous reasons why mothers and neonates do not succeed at breastfeeding, the current peer-reviewed literature does not support the premise that neuraxial techniques have any effect on the abilities of the neonate to do so.
Delivery Modes and Anesthetic Techniques
When a mother presents for labor management, and delivery is not imminent, an important role for the obstetric team is to support whatever pain-relieving choices she makes as long as there is no perceived harm to her or her fetus. However, choices are not always well informed or circumstances dictate a change in labor management that conflicts with the mother's desires. For instance, the morbidly obese patient who has a difficult airway and a questionable fetal heart rate tracing should not pursue the water birth she planned but be counseled on oxygen supplementation and placement of a neuraxial block in anticipation of a possible emergency cesarean section. Labor is one of the most dynamic natural events in medicine, and what may begin as a routine process can change very rapidly. As a result analgesic techniques and management plans need to be adaptable. The following section outlines some of the anesthetic-based considerations the anesthesia provider faces when a spontaneous vaginal delivery is no longer a probability.
Operative Vaginal Delivery
Over the past decade, the rate of operative vaginal delivery has significantly decreased.14,57 There are two types: vacuum extraction and forceps delivery. The choice of instrument is often influenced by clinical circumstances, operator choice, and availability of specific instruments.56 In the case of a vacuum extraction, it cannot be performed until the head is very low in the pelvis.14 The extractor is generally a soft, pliable cup and the application is well tolerated by the mother so that additional analgesia is not necessary even if none was used for the labor. Some local anesthetic to the perineum in anticipation of an episiotomy may be all that is required.
Forceps application and delivery generally require some level of analgesia, and often the obstetric provider either requests a neuraxial block or performs a pudendal block. There are four levels of forceps delivery described, but currently, the two most commonly performed are low and outlet forceps deliveries.57,64 The placement of the forceps blades is very stimulating, and it is only in a dire emergency that an obstetric provider should perform such a delivery without some form of anesthesia in place. A pudendal block is a reasonable choice and can be performed in less time than a spinal or epidural can be administered. However, neuraxial blocks offer more complete pain relief and, in the case of an epidural or combined spinal-epidural, can be extended to a higher level should the need arise.
Cesarean Section
Cesarean sections can be performed electively, urgently, or emergently. The circumstances under which one occurs generally dictate the form of anesthetic used. Most anesthesia providers prefer not to use a general anesthetic because of concerns with the maternal airway and potential aspiration. The incidence of failed intubation among the pregnant population is estimated to be up to 8 to 10 times that of the nonpregnant population. Studies have reported between 1 per 238 and 1 per 750 general anesthetics, with 1 : 250 being the most widely quoted ratio.66 Several factors contribute to this number. The changes of pregnancy itself cause edematous airways resulting from fluid accumulation.47 Add to this the time spent in labor, especially after many Valsalva maneuvers in the second stage, and what was once a normal airway can become swollen with obscured intubation landmarks and easily obstructed with attempts at intubation. Other changes that contribute to maternal risk during induction include those of the pulmonary system, gastrointestinal system, and body habitus over the course of the pregnancy. These changes and the often emergent nature of an obstetric procedure may result in failed airway management, with or without aspiration. Airway fatalities have diminished secondary to the decreased use of general anesthesia for obstetric procedures and the use of rescue airway equipment such as the laryngeal mask airway (LMA) and video laryngoscopes.95 Because of these concerns, for most anesthesia providers, neuraxial anesthesia is the approach of choice whenever possible.
Neuraxial Anesthesia
In the elective or urgent situation, some form of neuraxial block is the anesthetic of choice, assuming that there are no existing contraindications (Box 28-2). If an epidural is already in place, it is an easy matter to change medications to a more concentrated, faster-onset local anesthetic. Opioids may or may not be added. Attention must still be paid to the dosing technique because moving the patient onto the operating bed may dislodge or shift the epidural catheter; therefore, test doses and aliquoted dosing amounts are still in order. If an epidural block is not in place and the need to proceed with the cesarean section is urgent or emergent, the preferred approach in most units is to perform an intrathecal injection or possibly even a combined spinal-epidural. These techniques give a faster onset and limit the medication exposure of the fetus. All neuraxial techniques have been shown to result in better neonatal parameters than when a general anesthetic is performed.1
General Anesthesia
At what level of surgical urgency a neuraxial block should not be attempted or should be abandoned is always a topic for discussion. That decision should never be made lightly because of issues with the maternal airway, effects of anesthetics on uterine tone, and the potential impact on the fetus.44 There are absolute contraindications to performing a neuraxial block as well (see Box 28-2), and when one is present, it directs the decision making. Neonatologists present at a nonelective cesarean section add the use of general anesthetics to their growing list of potential reasons for need to resuscitate the neonate. Fortunately, the literature supports the clinical observation that under the conditions of a short incision-to-delivery time frame, and with the current pharmacologic developments of short-acting anesthetics, the impact is small. Those anesthetic agents that are the most commonly used are discussed in the following.
Before the induction of a general anesthetic, the anesthesia provider usually gives the mother one or more agents to reduce the risk of aspiration if circumstances allow. The most commonly used agent is sodium citrate, which is an antacid taken orally just before induction. Other agents that may be given instead of or along with sodium citrate are some form of H2-blocker such as ranitidine and metoclopramide. None of these medications has been shown to have an impact on the fetus.26
Because it is only the induction phase and a short period of maintenance to which the fetus is exposed, the anesthetics of concern are induction agents, a rapid-onset neuromuscular blocker such as succinylcholine, nitrous oxide (N2O), a volatile agent, and occasionally opioids. The previous discussion on general pharmacologic properties and the UV : MV ratio should help gauge the potential impact on the fetus. Table 28-1 shows the UV : MV ratios of several of these drugs. Thiopental has become more difficult and expensive to obtain; hence, propofol is now the most commonly used induction agent.51 Ketamine and etomidate have been used as well. All cross the placenta, but there is rarely an effect seen in the neonate because of the first-pass effect through the fetal liver and the dilutional effects from the circulation. Some of propofol's benefits are its short half-life and minimal residual impact. Ketamine is generally reserved for the hemorrhaging or volume-depleted patient because it is less cardiodepressive and produces a sympathetic response that better maintains the maternal hemodynamic state. Etomidate is generally reserved for patients with cardiac disease. All of these induction agents depress the maternal myocardium, but to varying degrees. As a result, intervillous blood flow initially decreases, but returns to baseline as long as maternal blood pressures are monitored and any hypotension is treated.
To intubate and secure the airway in the most rapid manner possible, succinylcholine is the neuromuscular agent of choice because it has the fastest onset of action. It also has the shortest duration of action as it has a half-life of 90 seconds and is metabolized by pseudocholinesterase in the maternal plasma to benign metabolites. As a rule, no other neuromuscular blocking agent is needed during the short time from incision to delivery, so the use of succinylcholine alone will have no effect on the fetus. However, there are times when succinylcholine is contraindicated or delivery is delayed and a nondepolarizing neuromuscular blocking agent is required. This is a different class of neuromuscular blocker from succinylcholine, but this class of medications is hydrophilic and highly ionized. As a result, little if any crosses the placenta and again, the fetus is not affected (see Table 28-1).
N2O and a volatile agent such as isoflurane or sevoflurane are administered to maintain the anesthetic state. Although these agents pass to the fetus, they rarely have a direct effect unless the time from induction to delivery is prolonged. If more than 15 minutes pass since the induction, the concentrations of these agents in the fetus will equilibrate with the maternal levels and the neonate will be depressed at delivery. Ventilatory support for a few minutes is generally all that is required as these agents are expelled through the lungs. However, it is more important for the neonatologist to note that if the uterine incision to delivery time exceeds 180 seconds, the neonate will more likely be depressed from a compromised perfusion and less so from these maintenance agents.17
Ancillary Medications
Most commonly, intravenous opioids or sedatives are not administered to the mother before delivery, but on occasion it happens. Some patients demand some form of anxiolytic before the cesarean section, and in those cases a small dose of midazolam (Versed) is usual. A small dose (1-2 mg) will not have an impact on the neonate. At other times, the neuraxial block may not be adequate for the cesarean section because either the epidural catheter is poorly positioned or insufficient time has passed for the level to rise adequately, and the anesthesia provider may administer an opioid such as fentanyl or small doses of ketamine intravenously. These medications have already been discussed, and none has a significant effect on the neonate at the dosages commonly used.
Anesthetic Choices and Maternal Obstetric History
Preeclampsia
Preeclampsia is a disease unique to pregnancy.69 Although its etiology is still unclear, the placenta is the main pathogenic focus of the disease.18,65 Currently, the most widely accepted explanation is that the placenta fails to embed adequately and, in response to hypoxia caused by poor placental perfusion, releases factors into the circulation that damage the maternal endothelium and give rise to the multisystem manifestations of an accentuated inflammatory response.55,65,69 The classic presentation in the mother is proteinuria and hypertension.16,18 The severe systemic involvement of this disease can extend from mild to life-threateningly severe and involves multiple organ systems. One of the cardiovascular effects is an increase in the systemic vascular resistance (SVR).65 Depending on the severity of the disease, the increased SVR can reduce uteroplacental blood flow and compromise the fetus.65,70 As a result, it is not unusual to see a nonreassuring tracing during the evaluation or induction of a patient with severe preeclampsia.
Other clinical abnormalities associated with the disease that are particularly troublesome for the anesthesia provider are edema and thrombocytopenia.42 A severely preeclamptic patient can develop a generalized edema so severe that the tissues in the airway and around the vocal cords are involved.65 Clinically, the patient may notice hoarseness or other changes in her voice. To the attentive anesthesia provider, this manifestation signals a potential difficult airway in which attempts at intubation may fail. If a general anesthetic is the only option for a surgical delivery, and the airway evaluation suggests a difficult intubation, the anesthesia provider may try to secure the airway before induction using a fiberoptic laryngoscope in an awake patient.65 This gives the advantage of airway evaluation and appropriate management. However, the mother will be given opioids, sedatives, and anxiolytics such as midazolam during this process, all of which cross the placenta. Whether intubated awake or in the usual fashion, the anesthesia provider will likely attempt to block the sympathetic response to laryngoscopy by giving antihypertensives such as labetalol or a short-acting beta-blocker such as esmolol, which can cross to the fetus. Failing to manage the hypertensive response to laryngoscopy may produce such severe increases in maternal blood pressure that she experiences an intracranial bleed.
The thrombocytopenia that may occur with preeclampsia can be mild or very severe and significant enough that a regional anesthetic is contraindicated because of concerns for hematoma formation with subsequent nerve damage. The issue for the anesthesia provider has always been whether the platelets present are functional. Not all that long ago, many refused to place a neuraxial block if the platelet count was less than 100,000 because there were no accurate tests available to determine platelet function. Although there is still no standard test to evaluate platelet function at the bedside, current knowledge is that platelet function tends to be preserved, so the 100,000 platelet count limit is no longer valid. Most anesthesia providers place a neuraxial block at lower platelet counts, but at what lower limit a neuraxial block is contraindicated has not been determined.18
Another recent change in anesthesia management relates to the type of neuraxial block used. Older literature reported that spinals should be avoided, but it is now known from further studies and case reports that spinal-induced hypotension does not occur or is muted in preeclamptic patients. This is because the hypertension of preeclampsia is not caused by sympathetic tone but rather by the prostaglandin imbalance. Additionally, the favorable data on measured parameters seen in the neonates of non-preeclamptic mothers who receive a neuraxial block remains as favorable in the neonates of preeclamptic patients. Some studies have also shown that in the preeclamptic patient, intervillous blood flow improves with a regional block as long as maternal blood pressures are maintained.33,65 Thus, numerous options are acceptable for the preeclamptic patient as long as some aspect of the disease does not contraindicate the anesthetic choice.
Uterine Rupture
Between 1990 and 2011, the cesarean delivery rate in the United States increased nearly sevenfold.31 In response to this, obstetricians began to let go of the old adage “once a section, always a section.” In 2010, the American College of Obstetricians and Gynecologists (ACOG) published on the topic and updated their guidelines. A woman with one or two uncomplicated low transverse cesarean deliveries could attempt a trial of labor after cesarean delivery (TOLAC).31 If successful, the delivery was called a vaginal birth after cesarean (VBAC). It is recognized that the most serious complication of a TOLAC is the risk of uterine rupture.3,35 The risk is from 0.5% to 1% in women with one previous cesarean section.31,35 Even though in the first couple of years that VBACs were promoted, obstetric providers felt that women undergoing a trial of labor should not be allowed to receive an epidural, ACOG states that there is no high-quality evidence to suggest that epidural analgesia should be contraindicated for a TOLAC.31 The main concern was that epidural analgesia would mask the abdominal pain resulting from uterine rupture. However, two findings changed this mentality. First, the most common findings of either a rupture or scar dehiscence were rarely pain or even bleeding. They are a change in the fetal heart rate and contraction pattern. Second, with the advanced techniques in epidural analgesia, abnormal or pathologic pain during labor would “break through” the low analgesic doses of the epidural. As a result, current practice is not to deny women undergoing a trial of labor any form of epidural analgesia, but to aggressively examine them for other causes should they complain of unusual pain.
Once scar dehiscence or even a true uterine rupture is recognized, an epidural that is already in place may be safely extended for the cesarean section. However, it is common practice to proceed with a general anesthetic if nothing is in place as the fetus usually shows signs of severe distress, and the focus at this point is on delivery as fast as possible. When such an event occurs, it is more likely that the depressed neonate is suffering from the disruption in uteroplacental blood flow that occurs and less from adverse anesthetic effects because it would be unlikely that significant amounts of medication could cross.
Fetal Distress
Fetal distress is most often a result of some level of compromise in the intrauterine environment that may result from any of a large list of possibilities. It generally signals a progressive process that can lead to fetal acidosis or even asphyxia and result in severe neurologic damage or death. When signs of a stressed fetus are present, the obstetric provider evaluates the clinical situation and makes the determination of whether labor remains a viable option for delivery or a cesarean section should be undertaken, and how quickly. However, between the beginning of labor and that determination, a non-reassuring fetal tracing does not preclude a patient from receiving a neuraxial analgesic. As mentioned, the block can improve the intrauterine environment and keep the mother alert and active during her obstetric management, and the negative consequences of maternal hyperventilation, catecholamine elevations, and systemic medications are avoided.
Most anesthesia providers would prefer to meet and evaluate the mother before the fetus is in such severe distress that the call for a cesarean section is now an emergency, but that cannot always happen. General anesthesia can be thought to be the quickest anesthesia method in an emergency because it avoids the possibility of a failed regional block.2 Nonetheless, two arguments can be made against this. Literature shows that a spinal takes no longer to place than the induction of a general anesthetic provided it is successful on the first attempt.45 In addition, the infants most affected by general anesthesia are those who are already compromised in utero, as evidenced by fetal distress.2 General anesthesia not only increases the risk of neonatal intubation, but it also increases the chance that a neonate's Apgar score would be lower compared with a neonate intubated after a regional block.2 These findings support the concept that a neuraxial block should be pursued whenever possible in the absence of hemorrhage, and underscores the need for all members of the obstetric team to communicate. However, there will be times when the only option is to proceed with the general anesthetic.
Placenta Previa
Placenta previa is the covering of all or some portion of the internal cervical os by placental tissue that obstructs the fetal passage and places the mother at risk for hemorrhage. It has the potential to cause massive obstetric hemorrhage and maternal and fetal morbidity and mortality.32 Further classification can be made depending on the relationship of the cervical os and the placenta. The complete placenta previa covers the internal os completely and occurs 37% of the time. This form mandates an abdominal delivery. The partial placenta previa covers part, but not all, the cervical os, whereas the marginal placenta previa lies close, but does not cover the cervical os. Previously, these two types would have allowed for a vaginal delivery, but in today's obstetric environment, it is highly recommended that the obstetrician proceed with a cesarean delivery as well. Ultrasonography is the mainstay available to the obstetric provider to diagnose placenta previa.32 Because they are generally known in advance, an elective cesarean delivery under neuraxial anesthesia is the standard. Uncontrolled bleeding is rare, and there is no increased risk to the fetus. The dilemma arises if the patient starts to bleed. Because the bleeding is not necessarily brisk, yet the need to proceed to a cesarean section is obvious, there is no correct answer regarding whether to provide a neuraxial block or to perform a general anesthetic. The choice depends on the indication, urgency of the procedure, and degree of maternal bleeding and hypovolemia. The briskness of the bleeding will direct much of the decision making, as well as the ultimate insult to the neonate.
Placental Abruption
The most common cause of antepartum bleeding, placental abruption, is the premature separation of a normally sited placenta from its attachment to the uterus.52 It can be complete or partial and the bleeding might be obvious, but it can also present as a concealed hematoma.52 When the abruption is not complete and stable, the obstetric provider may prefer to observe the mother and watch for any signs of compromised blood flow to the fetus. If bleeding resumes, the separation extends, or there is fetal compromise, emergent delivery is required. Fetal hypoxia, premature labor, and sudden fetal death may occur.52 Under these conditions, most anesthesia providers will avoid a neuraxial block because it is difficult to determine the level of hemorrhage that is ongoing; therefore, a general anesthetic becomes the only choice. Because of the clinical situation, neonatal depression is caused by the severe compromise or loss of uteroplacental flow and not exposure to anesthetic medications.