Reproductive System
ANATOMY
Female Reproductive Anatomy
See Figure 16-1.
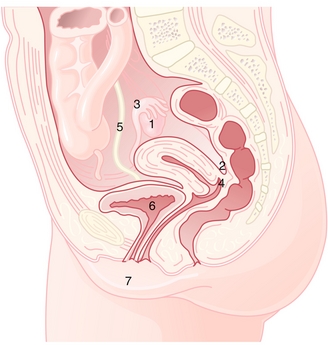
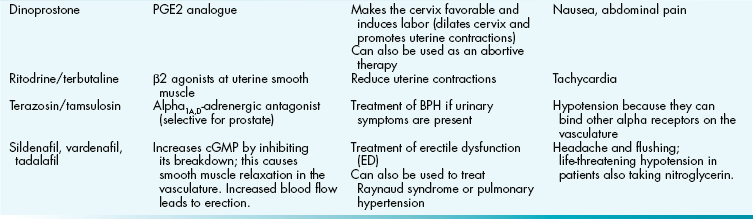
Figure 16-1 Sagittal view of female reproductive anatomy. 1, ovary; 2, pouch of Dougla; 3, fallopian tubes; 4, vagina; 5, ureter; 6, urinary bladder; 7, vulva. (From Hacker et al., Hacker & Moore's Essentials of Obstetrics and Gynecology, 5th ed. Philadelphia: Elsevier, 2009)
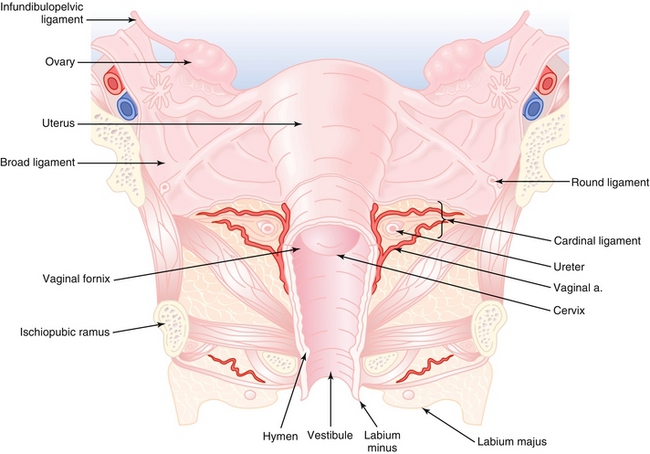
 Infundibulopelvic ligament: Also known as the suspensory ligament of ovaries. Connects the ovaries to the pelvic wall and contains the ovarian blood vessels.
Infundibulopelvic ligament: Also known as the suspensory ligament of ovaries. Connects the ovaries to the pelvic wall and contains the ovarian blood vessels.
 Ovarian ligament: Connects the ovaries to the lateral surface of the uterus. It does not contain any vessels.
Ovarian ligament: Connects the ovaries to the lateral surface of the uterus. It does not contain any vessels.
 Round ligament: Connects the uterine fundus to the labia majora by passing through the deep inguinal ring. The round ligament is a derivative of the embryologic gubernaculum.
Round ligament: Connects the uterine fundus to the labia majora by passing through the deep inguinal ring. The round ligament is a derivative of the embryologic gubernaculum.
 Cardinal ligament: Connects the cervix to the pelvic side wall. It contains the uterine blood vessels.
Cardinal ligament: Connects the cervix to the pelvic side wall. It contains the uterine blood vessels.
 Broad ligament: Connects the ovaries, fallopian tubes, and uterus to the pelvic floor and side wall (Fig. 16-2).
Broad ligament: Connects the ovaries, fallopian tubes, and uterus to the pelvic floor and side wall (Fig. 16-2).
 Pouch of Douglas: The anatomic space between the rectum and uterus. It can be palpated via a digital rectal exam. It is an important anatomic area because it can be a collecting site of blood (from ruptured ectopic pregnancy), pus (from pelvic inflammatory disease), malignant cells (from ovarian cancer), or endometrial implants (from endometriosis).
Pouch of Douglas: The anatomic space between the rectum and uterus. It can be palpated via a digital rectal exam. It is an important anatomic area because it can be a collecting site of blood (from ruptured ectopic pregnancy), pus (from pelvic inflammatory disease), malignant cells (from ovarian cancer), or endometrial implants (from endometriosis).
 Course of the female ureters: Cross over the bifurcation of the common iliac arteries, then lateral to the internal iliac arteries. They enter the retroperitoneal space in the uterosacral ligaments, then the cardinal ligaments, and dive under the uterine artery.
Course of the female ureters: Cross over the bifurcation of the common iliac arteries, then lateral to the internal iliac arteries. They enter the retroperitoneal space in the uterosacral ligaments, then the cardinal ligaments, and dive under the uterine artery.
Mnemonic: Water under the bridge.
The ureter is sometimes injured in surgeries involving ligation of the uterine artery because of their anatomic proximity. Surgeons must be wary!
Male Reproductive Anatomy
Pathway of sperm: Remembered by the mnemonic SEVEN UP—Seminiferous tubules (sperm created) → Epididymis (sperm stored) → Vas deferens → Ejaculatory ducts →“Nothing”→ Urethra → Penile urethra (Fig. 16-3).
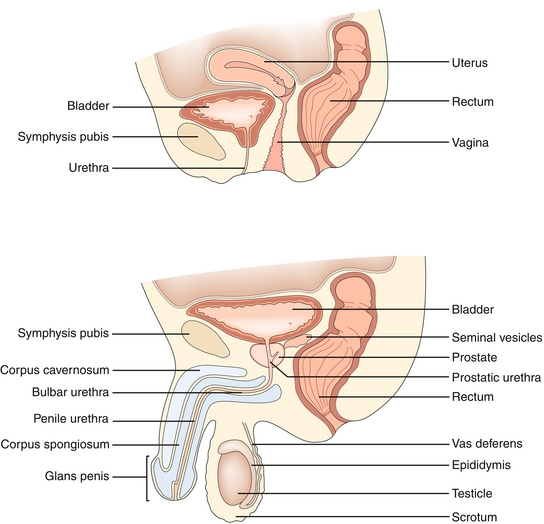
Figure 16-3 Pathway of sperm and anatomy of the male reproductive tract. (From Garden OJ, Bradbury AW, Forsythe JLR, Parks RW. Principles and Practice of Surgery. 5th ed. Philadelphia: Elsevier; 2007.)
Male Sexual Response
In order for an erection to occur, the parasympathetic fibers release nitric oxide (NO), which increases cyclic guanosine monophosphate, (cGMP), resulting in smooth muscle relaxation of the helicine artery, which results in blood flow into the paired corpus cavernosa and corpus spongiosum (Fig. 16-4).
Figure 16-4 Cross-sectional anatomy of the penis. (From Carroll RG. Elsevier’s Integrated Physiology. Philadelphia: Elsevier; 2006.)
During emission, smooth muscle contractions by the vas deferens and ejaculatory ducts push sperm into the prostatic urethra. This is a sympathetic response from the hypogastric nerveThis is a sympathetic response from the hypogastric nerve.
Remember the mnemonic: “Point and Shoot.” Erection (Point) is a Parasympathetic response while emission (Shoot) is a Sympathetic response.
During ejaculation, sperm is released through the urethra as a result of a somatic response from the pudendal nerve.
Male and Female Gonadal Drainage
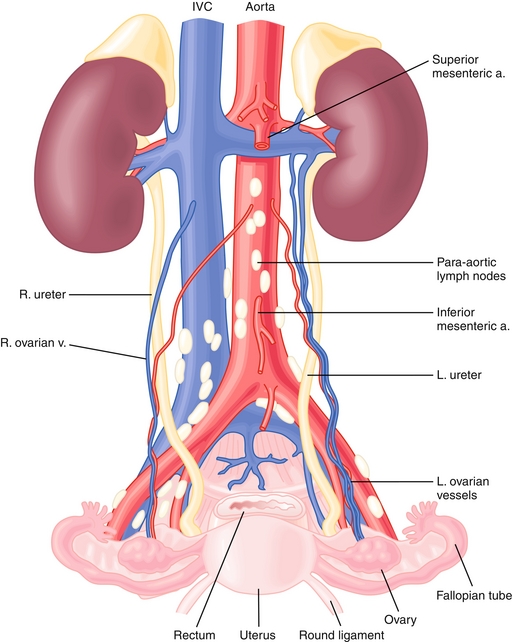
It is important to understand the venous and lymphatic drainage of the male and female gonads (Fig. 16-5). The venous drainage of the right ovary or testis is into the right ovarian or testicular vein and then into the inferior vena cava. The venous drainage of the left ovary or testis is into the left ovarian or testicular vein, then into the left renal vein, and then into the inferior vena cava. The extra step in left testicular venous drainage causes increased hydrostatic pressure. That is why varicoceles are more common on the left side.
The lymphatic drainage from the ovaries or testes is into the paraaortic nodes (this drainage pattern occurs because the ovaries and testes descended from the abdomen with their blood source from the aorta). The lymphatic drainage of the scrotum and distal third of the vagina is into the superficial inguinal nodes. The uterus and proximal two thirds of the vagina drain into the external iliac, obturator, and hypogastric nodes.
PHYSIOLOGY
Hypothalamic-Pituitary-Gonadal Axis
The hypothalamus secretes gonadotropin-releasing hormone (GnRH) in a pulsatile fashion to stimulate the anterior pituitary to produce luteinizing hormone (LH) and follicle-stimulating hormone (FSH). If GnRH were secreted continuously, it would paradoxically suppress the production of LH and FSH.
In males (Fig. 16-6A), LH stimulates Leydig cells to secrete testosterone. FSH stimulates Sertoli cells (Support cells) to maintain spermatogenesis. Some testosterone is further converted by 5-α-reductase into the more potent dihydrotestosterone. As in most of the endocrine system, a negative feedback loop maintains homeostasis. For example, administration of exogenous testosterone decreases LH, FSH, and testicular synthesis of testosterone. Interestingly, because spermatogenesis requires high local concentrations of testosterone in the seminiferous tubules, administering exogenous testosterone actually inhibits spermatogenesis because it will not be sufficiently concentrated in the testicles.
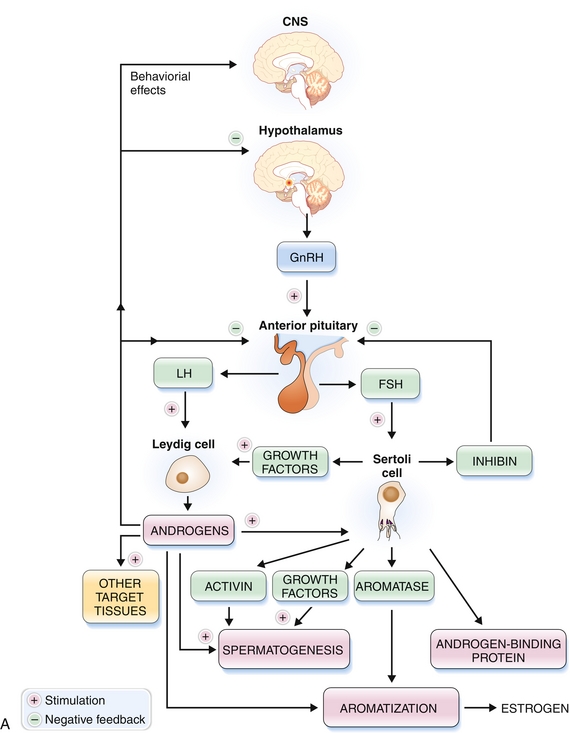
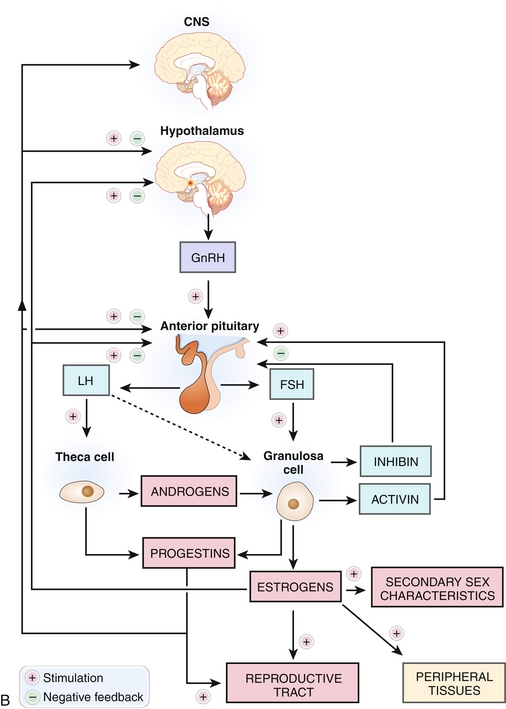
Figure 16-6 Hormonal control of the hypothalamic-pituitary-testicular axis (A) and hypothalamic-pituitary-ovarian axis (B). (From Damjanov I. Pathophysiology. Philadelphia: Elsevier; 2008.)
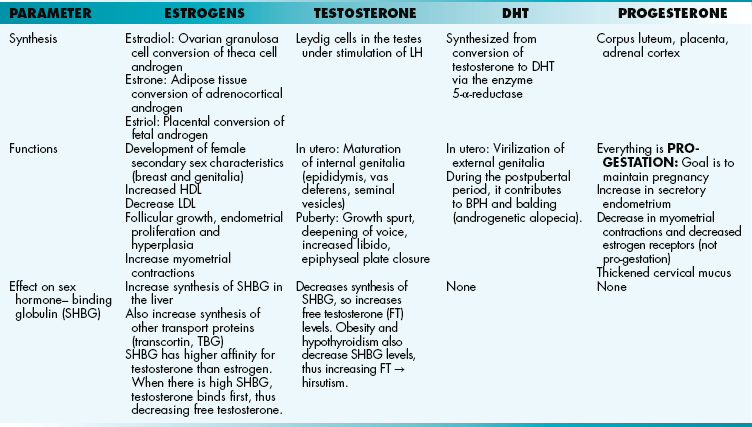
In females (Fig. 16-6B), LH stimulates theca cells to secrete androgens. FSH stimulates granulosa cells to convert those androgens into estradiol by aromatization. FSH, as the name implies, helps stimulate and mature the ovarian follicles.
Of note, estrogen refers to a group of hormones (unlike testosterone, which is a single molecule). In humans, estradiol is the most potent estrogen. It is produced by the ovary and is the most abundant estrogen in premenopausal women. Estrone is less potent. Adipose tissue produces estrone by aromatization of androstenedione (released from the adrenal cortex and gonads). Because estrone production does not require ovaries, it is the most abundant estrogen in postmenopausal females and males. Estriol is the least potent estrogen and is only present in significant levels during pregnancy. The placenta produces estriol by aromatization of fetal androgens. It can be used as a marker of fetal well-being.
Mnemonic: During reproductive years, there are two (husband and wife) in the house— estraDIol. During pregnancy there are three (husband, wife, and baby)—esTRIol. During menopause, sadly you might be a widow: estrONE.
Spermatogenesis
Sertoli cells are tall columnar Support cells that nurture spermatogenesis. They are located in the parenchyma of the seminiferous tubules and are stimulated by FSH. At puberty, Sertoli cells form tight junctions with each other, creating the basal and adluminal compartment and the blood-testis barrier. This barrier protects the maturing sperm in the adluminal compartment from autoimmune attack because they otherwise would be seen as foreign cells. Sertoli cells secrete three products: (1) inhibin, which provides negative feedback and inhibits FSH secretion that normally would stimulate the Sertoli cells; (2) androgen-binding protein (ABP), which maintains a high testosterone level in the seminiferous tubules; and (3) müllerian inhibiting hormone, which during fetal development inhibits the default female müllerian system from developing.
Leydig cells are endocrine cells in the interstitium or stroma that secrete testosterone. They are stimulated by LH. Testosterone helps increase sperm production and increases libido. (Remember, however, because spermatogenesis requires a high concentration of testosterone within the seminiferous tubules, exogenous administration of testosterone will decrease spermatogenesis.)
Spermatogonia are the most immature spermatogenic cells, located close to the basement membrane of the seminiferous tubules, and divide by mitosis to regenerate. Spermatogenesis begins at puberty and takes about 64 days for completion (see Fig. 16-6).
1. Spermatogonia (Fig. 16-7): Diploid, 2 N. The most immature spermatogenic cells, located close to the basement membrane of the seminiferous tubules. They divide by mitosis. One of the daughter cells serves as a stem cell and remains near the basement membrane. The other daughter cell starts the differentiation process and becomes a primary spermatocyte.
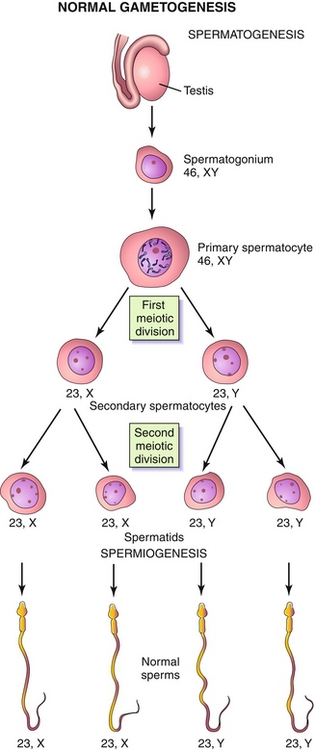
Figure 16-7 Spermatogenesis. Primordial germ cells mature into spermatogonia during puberty. Spermatogonia will undergo mitosis to become primary spermatocytes, which then undergo meiosis I to become secondary spermatocytes. Secondary spermatocytes undergo meiosis II to divide into spermatids. (From Moore KL, Persaud TVN, Torchia MG. Before We Are Born. 8th ed. Philadelphia: Elsevier; 2011.)
2. Primary spermatocyte: Diploid, 4 N. Migrates toward the adluminal compartment and undergo meiosis I to become a secondary spermatocyte. Failure of meiosis I results in primary spermatocyte accumulation.
3. Secondary spermatocyte: Haploid, 2 N. Undergoes meiosis II very quickly and becomes a spermatid. Failure of meiosis II results in accumulation of secondary spermatocytes.
4. Spermatids: Haploid, N. Undergo spermiogenesis, the process whereby spermatids transform into mature spermatozoa (haploid, N). Spermiogenesis results in a decreased amount of cytoplasm, formation of a flagellum (derived from a centriole), and an acrosome (derived from the Golgi apparatus). The head of a mature sperm contains chromatin and acrosomal enzymes needed for penetration of the ovum’s outer zona pellucida. The midpiece contains abundant mitochondria for adenosine triphosphate (ATP) production. The tail contains specialized cilia for efficient transport.
Female Physiology
Menstrual Cycle
The menstrual cycle (Fig. 16-8) normally lasts 28 days (± 7 days), with day 1 defined as the first day of menstruation. The cycle is divided into a follicular phase (before ovulation) and a luteal phase (after ovulation; Fig. 16-9).
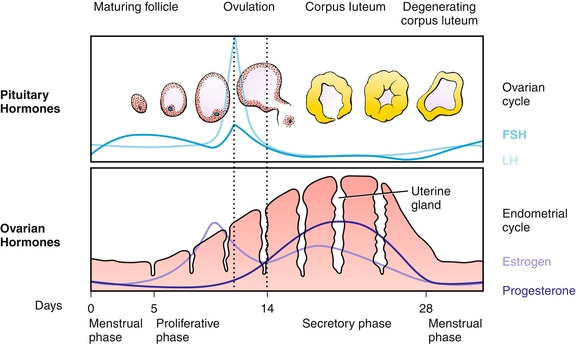
Figure 16-8 The menstrual cycle. (From Brauer PR, Francis-West PH, Schoenwolf GC, Bleyl SB. Larsen's Human Embryology. 4th ed. Philadelphia: Elsevier; 2008.)
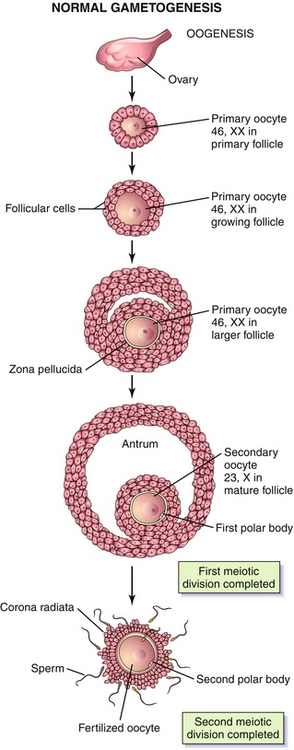
Figure 16-9 During the first month of fetal life, germ cells of a female embryo undergo extensive mitosis to become oogonia (diploid, 2 N). By the third month of gestation, these oogonia enter meiosis I and pass through the different phases of prophase I. They are now called primary oocytes (diploid, 4 N), and these primary oocytes are arrested in prophase I until puberty. Hours before ovulation, these primary oocytes complete meiosis I and become secondary oocytes (haploid, 2 N). Immediately, these secondary oocytes enter meiosis II and are arrested in metaphase II until fertilization. Once fertilization occurs, meiosis II is complete and becomes a mature ovum (haploid, N). (From Moore KL, Persaud TVN, Torchia MG. Before We Are Born. 8th ed. Philadelphia: Elsevier; 2011.)
Follicular (Proliferative) Phase
Ovarian follicles are spherical ovarian structures that contain oocytes, theca cells, and granulosa cells. They are the basic unit of female reproductive physiology. The follicular phase of the menstrual cycle refers to the growth of the follicles through various stages.
In the early follicular stage (during menstruation), estrogen levels are low, which means the anterior pituitary can produce abundant LH and FSH without much negative feedback. FSH stimulates several ovarian follicles to grow and will eventually create a dominant follicle. As the ovarian follicles grow, they will produce more and more estrogen. Although increasing estrogen will inhibit FSH production, the dominant follicle will upregulate FSH receptors, which will increase their sensitivity to FSH, so estrogen levels can continue to rise.
At the end of the follicular phase, estrogen switches from a negative feedback loop with the pituitary to a positive feedback loop. This creates a surge of LH, which will cause rupture of the dominant follicle and release of its oocyte into the fallopian tube.
The follicular phase is also known as the proliferative phase because, during this time, the endometrium (uterine lining) sees mostly estrogen, so the glands are “proliferating.” Histologically, the glands appear short, narrow and slightly wavy.
Of note, in the preovulatory phase, the LH surge causes the granulosa cells to make progesterone (released 24 hours prior to ovulation and causes the increase in basal body temperature) and prostaglandins.
Luteal (Secretory) Phase
The luteal phase refers to the corpus luteum, which forms from the follicle once the oocyte has been expelled. The corpus luteum (specifically the theca cells) produces estrogen and progesterone. This provides negative feedback on LH and FSH, resulting in their declining levels. The luteal phase is also known as the secretory phase because the elevated progesterone causes the endometrium to become secretory as it prepares for implantation.
The corpus luteum degenerates in a fixed 2 weeks if it is not rescued by human chorionic gonadotropin (hCG). The degenerated corpus luteum (yellow body) is now called the corpus albicans (white body). The loss of the corpus luteum causes a decline in estrogen and, more significantly, progesterone. Without progesterone to support it, the endometrial lining is sloughed off and menstruation occurs.
If the corpus luteum is rescued by pregnancy and hCG produced by the conceptus, it will linger for 8 to 10 weeks until the placenta takes over in the production of progesterone.
Of note, the luteal phase is fixed at 14 days long, whereas the follicular phase may differ. It is therefore the follicular phase that is responsible for the variability in the length of a woman’s menstrual cycle.
Pregnancy and hCG
Fertilization
Once ovulation has occurred, the secondary oocyte can typically survive for 24 hours before it degenerates. The ampullary portion of the fallopian tube is the most common location of fertilization. From there, the fertilized egg typically spends an average of 3 days in the fallopian tube and then 2 days in the uterine cavity, where it will implant on day 6.
Hormones of Pregnancy
Human growth hormone, human placental lactogen, prolactin, progesterone, and estriol are all increased during pregnancy.
 Human chorionic gonadotropin (hCG) is produced by the syncytiotrophoblast of the placenta, peaks at about 9 weeks gestation, and then falls. In the first trimester, hCG functions by binding to the LH receptor on the corpus luteum and stimulates it to produce progesterone. The beta subunit of hCG is used clinically as the first indicator of pregnancy. It is detectable in the blood by 1 week of gestation and in the urine by 2 weeks’ gestation. The peak level of β-hCG in a normal pregnancy is 90,000 to 100,000 mIU/mL. Anything significantly higher than its upper limit of normal usually signifies a pathologic state, such as a molar pregnancy or gestational trophoblastic disease (see later). The alpha subunit of hCG is identical to the alpha subunit of FH, LSH, and TSH.
Human chorionic gonadotropin (hCG) is produced by the syncytiotrophoblast of the placenta, peaks at about 9 weeks gestation, and then falls. In the first trimester, hCG functions by binding to the LH receptor on the corpus luteum and stimulates it to produce progesterone. The beta subunit of hCG is used clinically as the first indicator of pregnancy. It is detectable in the blood by 1 week of gestation and in the urine by 2 weeks’ gestation. The peak level of β-hCG in a normal pregnancy is 90,000 to 100,000 mIU/mL. Anything significantly higher than its upper limit of normal usually signifies a pathologic state, such as a molar pregnancy or gestational trophoblastic disease (see later). The alpha subunit of hCG is identical to the alpha subunit of FH, LSH, and TSH.
 Progesterone is initially produced by the corpus luteum, but by gestational week 12 the placenta produces enough progesterone to maintain pregnancy. Everything it does is pro-gestation. It increases secretory endometrium in preparation for implantation, it decreases myometrium contractility, and it thickens cervical mucus. It is a marker of fetal viability in early pregnancy.
Progesterone is initially produced by the corpus luteum, but by gestational week 12 the placenta produces enough progesterone to maintain pregnancy. Everything it does is pro-gestation. It increases secretory endometrium in preparation for implantation, it decreases myometrium contractility, and it thickens cervical mucus. It is a marker of fetal viability in early pregnancy.
 Estriol, as discussed earlier, is produced by the placenta by aromatization of fetal androgens. It can be used as a marker of fetal well-being later in pregnancy.
Estriol, as discussed earlier, is produced by the placenta by aromatization of fetal androgens. It can be used as a marker of fetal well-being later in pregnancy.
 Human placental lactogen is produced by the synctiotrophoblasts of the placenta. It decreases maternal insulin sensitivity and thus raises maternal serum glucose levels. Maintaining adequate glucose is crucial to fetal development.
Human placental lactogen is produced by the synctiotrophoblasts of the placenta. It decreases maternal insulin sensitivity and thus raises maternal serum glucose levels. Maintaining adequate glucose is crucial to fetal development.
 Prolactin is involved in milk production and simultaneously inhibits GnRH, serving as a natural form of contraception for the first 6 weeks postdelivery.
Prolactin is involved in milk production and simultaneously inhibits GnRH, serving as a natural form of contraception for the first 6 weeks postdelivery.
 Oxytocin is produced by the posterior pituitary. During pregnancy, the uterus upregulates oxytocin receptors so that an oxytocin surge at the onset of labor facilitates uterine contractions. It is also involved in the letdown of milk that was already produced through the actions of prolactin.
Oxytocin is produced by the posterior pituitary. During pregnancy, the uterus upregulates oxytocin receptors so that an oxytocin surge at the onset of labor facilitates uterine contractions. It is also involved in the letdown of milk that was already produced through the actions of prolactin.
 Corticotropin-releasing hormone is a hypothalamic hormone involved in the cortisol pathway thought to induce the beginning phases of labor.
Corticotropin-releasing hormone is a hypothalamic hormone involved in the cortisol pathway thought to induce the beginning phases of labor.
Menopause
Physiologic menopause occurs after 40 years of age, with the average age being 51 years of age. Most women experience perimenopausal symptoms for 3 to 5 years preceding menopause, after which time anovulation, amenorrhea, and menopausal symptoms occur (see later). The physiology behind menopause is the gradual loss of ovarian follicles, leading to decreased estradiol production, and thus increases in LH and FSH. The rise in LH and FSH can be used to diagnose menopause when the cause of amenorrhea is uncertain. Estrone becomes the principal estrogen that continues to circulate through peripheral conversion of androgens.
Symptoms of menopause include hot flashes, night sweats, amenorrhea, and mood swings. Conditions associated with menopause include atrophic vaginitis, urinary incontinence, cardiovascular disease (↓ high-density lipoprotein [HDL], ↑ low-density lipoprotein [LDL]), and osteoporosis. Risk factors for osteoporosis include fair skin, thin body habitus, smoking, alcohol and caffeine intake, and positive family history. The most common osteoporotic fracture site is the vertebral bodies.
Menopause before 40 years of age can indicate premature ovarian failure that can be caused by oophorectomy, radiation, chemotherapy, infection, autoimmune conditions, or mosaic karyotype.
FEMALE PATHOLOGY
Pathology in Pregnancy
Ectopic pregnancy is a condition in which the embryo implants outside the uterus. The most common site of ectopic pregnancy is the ampulla of the fallopian tube. Ectopic pregnancy often results in the triad of vaginal bleeding, pelvic pain, and adnexal mass. The patient with an ectopic pregnancy has elevated hCG levels; however, this level does not double in 48 hours as in most normal uterine pregnancies. Risk factors include pelvic inflammatory disease, prior ectopic pregnancy, tubal ligation, and adhesions from previous abdominal surgeries. Confirmation can be made with ultrasound, which may show an adnexal mass. Decidualized endometrium without chorionic villi may also be present because the pregnancy is not in the uterine cavity. Treatment is often necessary before ultrasonographic findings are present. Methotrexate is used to terminate the pregnancy or surgery can be used to remove the embryo.
Molar pregnancy is a benign proliferative abnormality of the placenta or chorionic epithelium that has malignant potential if it transforms to gestational trophoblastic tumor or choriocarcinoma. The most common form is complete mole, which is dispermic fertilization of an anuclear ovum, resulting in 46,XX or 46,XY. There are no fetal parts because it is a complete mole. There is a 20% chance of progressing to malignancy. On ultrasound, there are grapelike vesicles and a “snowstorm” appearance. Incomplete mole is dispermic fertilization of a normal ovum resulting in 69,XXY. An incomplete mole contains fetal parts and no vesicles. There is a 5% chance of progressing to malignancy. Presenting signs and symptoms include abnormal vaginal bleeding, nausea and vomiting (from extremely elevated hCG levels), theca lutein cysts, and hyperthyroidism (because hCG can mimic thyroid hormone and bind to receptor). Treatment is dilation and curettage and follow-up of hCG levels until they reach zero. This ensures that no tissue is retained that will progress to malignancy.
Miscarriages are extremely common, and one or two miscarriages do not imply maternal pathology. First-trimester losses are usually caused by aneuploidy, more specifically an autosomal trisomy. Second-trimester losses usually have maternal causes, such as uterine anomaly (bicornuate uterus) or cervical insufficiency.
Third-trimester bleeding is commonly secondary to placenta previa or placenta abruption.
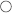 Placenta previa is when the placenta attaches to the lower uterine segment and partially covers the cervix. This commonly causes painless vaginal bleeding. Risk factors include prior cesarean section and multiparity. Patients with placenta previa are at higher risk for placenta accreta, in which the placenta attaches to the myometrium.
Placenta previa is when the placenta attaches to the lower uterine segment and partially covers the cervix. This commonly causes painless vaginal bleeding. Risk factors include prior cesarean section and multiparity. Patients with placenta previa are at higher risk for placenta accreta, in which the placenta attaches to the myometrium.
 Placental abruption is premature separation of the placenta, causing painful vaginal bleeding, fetal distress, and tetanic (constant maximal) contractions. Risk factors include cocaine use, smoking, hypertension, and trauma. The mother is at risk for disseminated intravascular coagulation (DIC) and shock.
Placental abruption is premature separation of the placenta, causing painful vaginal bleeding, fetal distress, and tetanic (constant maximal) contractions. Risk factors include cocaine use, smoking, hypertension, and trauma. The mother is at risk for disseminated intravascular coagulation (DIC) and shock.
Preeclampsia is a triad of pregnancy-induced hypertension, proteinuria, and edema that results from placental artery vasoconstriction. The hypertension is defined as more than 140/90 mm Hg and usually occurs after 20 weeks’ gestation and subsides 6 weeks’ postpartum. Other symptoms include headache, vision changes, and right upper quadrant (RUQ) pain. Risk factors include previous hypertension, diabetes, or kidney disease. Severe preeclampsia can be manifested as HELLP syndrome (Hemolysis, Elevated LFTs [liver function test], Low Platelets). Preeclampsia is treated with delivery whenever possible and magnesium sulfate is given to prevent progression to eclampsia. Eclampsia is preeclampsia with the addition of seizures. Magnesium levels must be monitored during treatment with magnesium to prevent toxicity. An early sign of magnesium toxicity is loss of deep tendon reflexes. Later signs are respiratory depression and cardiac arrest.
Amniotic Fluid Disorders
Polyhydramnios is an excess of amniotic fluid. Duodenal atresia and tracheoesophageal fistula lead to polyhydramnios because the fluid does not go through the fetal gastrointestinal tract. Anencephaly causes polyhydramnios as the central nervous system leaks excess fluid into the amnion. Maternal diabetes is also associated with polyhydramnios.
Duodenal atresia, TE fistula, anencephaly, and diabetes all can cause polyhydramnios.
Oligohydramnios is a deficit of amniotic fluid related to urogenital abnormalities (e.g., renal agenesis, posterior urethral valves), premature rupture of membranes, or placental insufficiency (preeclampsia or post-term pregnancy).
Benign Gynecologic Conditions
Mittelschmerz is abdominal pain secondary to ovulation. When ovulation occurs, blood from the ruptured follicle may cause peritoneal irritation.
Endometriosis is a condition in which endometrial glands and stroma are located outside the uterus, resulting in cyclic bleeding and dysmenorrhea. The most common sites are the ovaries, pouch of Douglas, fallopian tubes, and intestines. These growths can coalesce into small masses of ectopic endometrial tissue called endometriomas (or chocolate cysts because of their color and consistency). The pathogenesis is thought to be as follows: (1) retrograde menstrual flow through the fallopian tubes; (2) coelemic metaplasia into endometrial cells; or (3) vascular or lymphatic spread. Patients may experience dyspareunia (pain with intercourse) and are at increased risk for infertility. Definitive diagnosis is by laparoscopy, and treatment begins with oral contraceptive pills (OCPs; birth control pills). Adenomyosis is endometrial glands and stroma within the myometrium.
A leiomyoma (fibroid) is a common benign smooth muscle tumor usually found in African American women. These tumors are responsive to estrogen and commonly become larger during pregnancy and shrink in size after menopause. They are generally asymptomatic, but clinical findings include heavy menstrual bleeding, constipation, and urinary frequency or urgency. Complications include obstructing delivery or iron deficiency anemia from bleeding of submucosal fibroids. This tumor is commonly stained with smooth muscle actin. Leiomyosarcoma occurs de novo and not as malignant transformation of an existing leiomyoma. Leiomyosarcomas are aggressive, irregularly shaped tumors with atypical mitotic changes and necrotic areas. This tumor is commonly stained with desmin.
Anovulation occurs as a result of an increase in the estrogen-to-progesterone ratio, resulting in unopposed estrogen that puts the patient at increased risk of endometrial hyperplasia, and thus, cancer. Because there is not enough progesterone, ovulation does not occur. Regular menses also do not occur because of the absence of progesterone withdrawal. Therefore, anovulatory bleeding occurs when the proliferative endometrium outgrows its blood supply, resulting in irregular bleeding. Common causes of anovulation include obesity, hypothalamic-pituitary abnormalities, polycystic ovarian syndrome (PCOS), thyroid disorders, and Cushing’s syndrome.
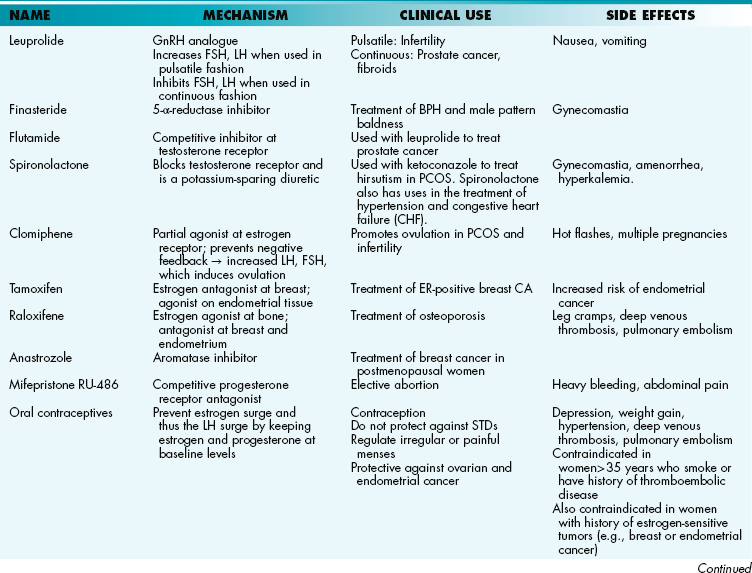
Polycystic ovarian syndrome is a disorder in which there is increased LH and decreased FSH released from the pituitary. Because of the increased LH, there is increased testosterone, which is converted into estrogen in adipose tissue (this is why patients with PCOS are at increased risk of endometrial cancer). This estrogen has negative feedback on the production of FSH, thus decreasing FSH levels, which results in follicular degeneration and bilateral cystic ovaries. Clinical findings associated with PCOS include obesity, insulin resistance, hirsutism, amenorrhea, and infertility. Treatment includes OCPs, leuprolide, and metformin.
Ovarian cysts are follicular cysts, the most common ovarian mass; they result from fluid buildup in a follicle, usually right before ovulation. They are unilateral and regress spontaneously. Corpus luteum cysts are similarly unilateral and regress spontaneously. Corpus luteum cysts are most common during pregnancy. Theca lutein cysts are usually bilateral and usually associated with molar pregnancies.
Gynecologic Malignancies
Vaginal cancer is the fourth most common gynecologic malignancy. Risk factors include human papillomavirus (HPV) 16 and 18 infection, obesity, and chronic hypertension. Squamous cell carcinoma is associated with HPV infection. Clear cell adenocarcinoma is associated with diethylstilbestrol exposure. Sarcoma botryoides is a rhabdomyosarcoma that typically occurs in very young females (< 8 years) and stains desmin-positive. The name botryoides comes from the Greek word for “grape bunches” because of the gross appearance of the tumor, resembling grape bunches.
Cervical dysplasia—cervical intraepithelial neoplasia (CIN) refers to the potentially premalignant transformation of the normal columnar epithelium to squamous epithelium at the transformation zone of the cervix. These dysplastic changes are mostly caused by HPV 16 and 18 (the most high risk of the HPV types), which inactivate p53 and Rb tumor suppressor genes. Risk factors include multiple sexual partners, early age of sexual intercourse, smoking, immunosuppression, and OCP use. Low-grade dysplasia normally regresses on its own. High-grade dysplasia has a greater chance of progressing to invasive cancer; however, this process usually takes about 10 years. Vaccines are now available that protect against HPV types 16 and 18.
Cervical cancer is the least common gynecologic malignancy. The most common tumor type is squamous cell carcinoma; less common is adenocarcinoma. Patients typically present with postcoital bleeding. The most common cause of death is renal failure from lateral invasion of the ureters.
Endometrial hyperplasia is abnormal proliferative endometrium related to prolonged estrogen exposure. Therefore, risk factors include nulliparity, late menopause, early menarche, anovulatory cycles, PCOS, and hormone replacement therapy, along with systemic diseases such as diabetes mellitus, chronic hypertension, and obesity. The most common presenting sign is postmenopausal bleeding. In fact, because endometrial hyperplasia progresses to endometrial carcinoma, any woman older than 35 years with abnormal vaginal bleeding requires an endometrial biopsy to rule out endometrial hyperplasia or cancer.
Endometrial carcinoma is the most common gynecologic malignancy, with adenocarcinoma being the most common tumor type. Risk factors are similar to endometrial hyperplasia, with the greatest risk factor being a history of endometrial hyperplasia. OCPs are protective.
Ovarian tumors (Table 16-3) are the second most common gynecologic malignancy. Risk factors include the BRCA1 gene mutation, positive family history, higher lifetime ovulation (early menarche, late menopause, nulliparity), and white ethnicity. OCPs are protective (less lifetime ovulation). Early disease is asymptomatic, which is why most patients present in later stages of the disease. Later disease has nonspecific symptoms and signs, such as bloating, constipation, nausea, and vomiting. The most common tumor type is epithelial (65% to 70%), followed by germ cell (15% to 20%), gonadal-stromal (3% to 5%), and metastatic (5%).
Breast Disorders and Malignancy
Common Breast Conditions
Acute mastitis is an infection around the areola that can lead to a breast abscess, usually from breastfeeding. Common organisms included Staphylococcus aureus and Streptococcus pyogenes. Purulent nipple discharge may be present. Fat necrosis is a painless lump secondary to breast trauma. Mammary duct ectasia occurs when the main ducts are filled with debris, causing inflammation and greenish nipple discharge. Gynecomastia is a glandular proliferation in the male breast caused by increased estrogen. Causes include puberty, old age, cirrhosis, Klinefelter syndrome, estrogen, marijuana, spironolactone, digitalis, cimetidine, and ketoconazole (displaces estrogen from sex hormone–binding globulin). The classic presentation of gynecomastia is a pubertal male presenting with a lump in his breast; the high levels of testosterone during puberty lead to high levels of estrogen when the aromatase enzyme converts the androgens into estrogens, causing the condition. This usually self-resolves and is of no consequence.
Benign Breast Disorders
Fibroadenoma is the most common benign tumor in young women. It is a small, well-circumscribed, mobile mass in the breast stroma. It is often tender during periods of increased estrogen, such as pregnancy and menstruation. Fibroadenoma does not commonly progress to breast cancer. Intraductal papilloma is a papillary growth within the lactiferous ducts of the breast, usually presenting with bloody discharge. It does not pose an increased risk for cancer. Phyllodes tumor is a large bulky tumor derived from the stroma that has malignant potential. Although a fibroadenoma is the most common benign tumor in young women, the most common cause of a breast lump in a young woman is fibrocystic change, which occurs in over half of women at some point during their reproductive years. This causes multiple lumps in the breast, which are usually painful, and vary in size and tenderness with the reproductive cycle. This is often concerning and bothersome for the patient, but is benign.
Breast Cancer
Breast cancer (Fig. 16-10) is the most common cancer in postmenopausal women and the second most common cancer resulting in death in women. The mean age at diagnosis is 64 years. Risk factors include increased estrogen exposure (nulliparity, early menarche, late menopause), BRCA1 and BRCA2 gene mutations, positive family history in first-generation relatives, obesity, and hormone replacement therapy. Malignancy is most commonly found in the upper outer quadrant, which drains into the axillary lymph nodes (important prognostic indicator). The inner quadrant drains to the internal mammillary lymph nodes and is a less common site for breast cancer. Breast cancer commonly metastasizes to lungs, bone, liver, brain, and ovaries. Breast cancers can also be classified by the presence or absence of hormone receptors—estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2/neu). ER-positive cancers carry a better prognosis and can be treated with estrogen blockers such as tamoxifen. HER2/neu-positive cancers carry a worse prognosis but can be treated with the monoclonal antibody trastuzumab, which binds the receptor.
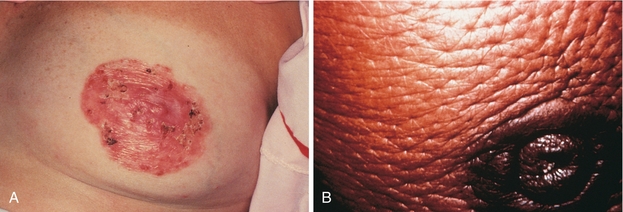
Figure 16-10 Breast cancer. A, Paget’s disease of nipple. B, Peau d’orange. French for “orange skin,” this finding is caused by lymphadema and dimpling secondary to lymphatic plugging. (From Swartz MH. Textbook of Physical Diagnosis. 6th ed. Philadelphia: Elsevier; 2009.)
Table 16-4
| Type | Description |
| Ductal carcinoma in situ (DCIS) | Ductal hyperplasia that can invade past basement membrane in one-third of cases Not palpable, detected via mammography because of microcalcifications |
| Paget disease of nipple | Dermal invasion of DCIS resulting in rash and possible nipple retraction (see Fig. 16-10A) Paget cells are large cells in the epidermis with halos. |
| Invasive ductal carcinoma | Most common type of breast cancer Firm, hard, sharply demarcated, invasive mass with stellate morphology One-third have HER2/neu amplification and can be treated with trastuzumab. |
| Lobular carcinoma in situ (LCIS) | Nonpalpable, often found incidentally on breast biopsy—not identified via mammography because there are no calcifications One-third invade. ER- and PR-positive. |
| Invasive lobular carcinoma | Row of cells or concentric circles that have a bull’s eye appearance on histology Often bilateral, although the other breast does not have to have lobular cancer |
| Medullary carcinoma | Related to BRCA1 mutation Typically HER2/neu–positive and ER-, PR-negative |
| Inflammatory | Poor prognosis Tumor plugs up dermal lymphatic lumens, resulting in lymphedema and dimpling (peau d’orange; see Fig. 16-10B) |
Sex Chromosome and Hormone Disorders
Turner syndrome (XO) is a disorder that results in follicle deterioration and ovarian dysgenesis (streak ovary) by 2 years of age. This puts those with this disorder at risk for dysgerminoma. Because of the nonfunctioning ovaries, there is decreased estrogen and increased FSH and LH. Clinical signs and symptoms include short stature, shield chest, preductal coarctation of aorta, bicuspid aortic valve, horseshoe kidney, cystic hygroma (from dilated lymphatics), amenorrhea, and a shortened fourth metacarpal.
Female pseudohermaphrodites (XX) have normal internal genitalia and virilized external genitalia from excessive exposure to androgens. Common causes include congenital adrenal hyperplasia and androgen exposure during pregnancy.
MALE PATHOLOGY
Summary of Breast Cancers
Benign Prostatic Hyperplasia (BPH)
Benign prostatic hyperplasia of the prostate gland is age-related. It is thought that in later adult male life, there is an increase in estrogen levels, along with an increased synthesis of androgen receptors that respond to dihydrotestosterone (DHT). Enlargement of the transitional and periurethral lobes (Fig. 16-11) results in increased urinary frequency, urgency, hesitancy, and nocturia from anatomic compression of the urethra. Complications include obstructive uropathy leading to bilateral hydronephrosis, bladder infections, and prostatic infarcts. PSA is normal (0 to 4 ng/mL) or mildly elevated (4 to 10 ng/mL). Medical treatment includes α-adrenergic antagonists (e.g., terazosin, tamsulosin), which relax the smooth muscle around the urethral sphincter or 5-α-reductase inhibitors (e.g., finasteride) that block the conversion of testosterone to DHT. Surgical treatment is most commonly transurethral resection of the prostate (TURP).
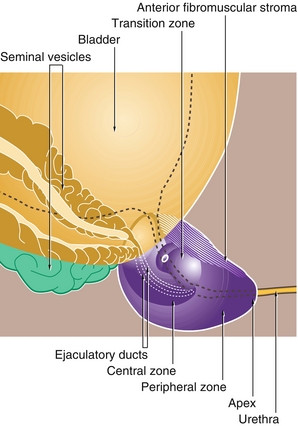
Figure 16-11 The prostate. Note the lobes of the prostate and their relation to pathology. Because prostate cancer affects the peripheral zone on the posterior of the prostate, it can be detected on digital rectal exam. Because BPH affects the transition zone, it is more likely to obstruct the urethra. (From Young B, Lowe JS, Stevens A, Heath JW. Wheater’s Functional Histology. 5th ed. Philadelphia: Elsevier; 2006.)
Prostatic Adenocarcinoma
Prostate cancer is the most common cancer in males, usually affecting the peripheral or posterior lobe of the prostate (see Fig. 16-11). For this reason, it is often diagnosed via digital rectal exam, in which a hard mass is palpated. Risk factors include increasing age, family history, and African American ethnicity. Clinically, prostate cancer is silent until advanced stages, at which time patients may report back pain from osteoblastic metastasis to the bone (spread via the Batson venous plexus), resulting in high alkaline phosphate levels. PSA levels are elevated (usually > 10 ng/mL). Diagnosis is made by needle biopsy. Radiation, chemotherapy, and surgery may all be used in treatment. Surgery has frequent side effects, including erectile dysfunction and urinary incontinence.
Testicular Tumors
Testicular cancer is the most common malignancy between ages 15 and 35 years in males (Table 16-5). The most common type of testicular tumors are germ cell in origin (95%), followed by gonadal-stromal (5%), which are mostly benign. Risk factors include cryptorchidism, Klinefelter syndrome, and inguinal hernias.
Table 16-5
| Name | Description | Tumor Marker |
| Seminoma | Most common germ cell tumor Malignant Affects men 30-35 years old Large cells with fried-egg appearance Good prognosis, radiosensitive |
Can have increase in hCG |
| Embryonal carcinoma | Big, painful tumor with bloody necrotic areas Mixed with other tumor types Worse prognosis |
AFP, hCG |
| Endodermal sinus tumor (yolk sac) | Most common testicular tumor in young children (< 5 years old) Schiller-Duvall bodies on histology |
AFP |
| Choriocarcinoma | Aggressive tumor of trophoblastic tissue that spreads hematogenously | hCG |
| Teratoma | Benign in children, malignant in adult. | AFP, hCG |
| Leydig cell | Androgen-producing tumor that causes precocious puberty in boys Reinke crystals on histology. |
Testosterone |
| Testicular lymphoma | Most common testicular tumor in men > 60 years old Bilateral testicle involvement Poor prognosis |
LDH (lymphoma marker) |
AFP, alpha-fetoprotein; hCG, human chorionic gonadotropin; LDH, lactate dehydrogenase.
Penile Disorders and Pathology
 Peyronie disease: Bending of the penis caused by fibrous tissue deposition secondary to previous trauma.
Peyronie disease: Bending of the penis caused by fibrous tissue deposition secondary to previous trauma.
 Priapism: Persistent penile erection secondary to sickle cell disease, spinal cord trauma, or medications (e.g., trazodone, sildenafil). Failure of detumescence leads to impaired venous drainage and causes compartment syndrome, which can eventually lead to ischemia and necrosis of penile tissue.
Priapism: Persistent penile erection secondary to sickle cell disease, spinal cord trauma, or medications (e.g., trazodone, sildenafil). Failure of detumescence leads to impaired venous drainage and causes compartment syndrome, which can eventually lead to ischemia and necrosis of penile tissue.
 Carcinoma in situ (CIS; Bowen disease): Leukoplakia (white plaques) of the penis and/or scrotum associated with HPV 16 that progresses to squamous cell carcinoma in 10% of cases.
Carcinoma in situ (CIS; Bowen disease): Leukoplakia (white plaques) of the penis and/or scrotum associated with HPV 16 that progresses to squamous cell carcinoma in 10% of cases.
 Erythroplasia of Queyrat: Erythroplakia (red plaques) on the glans penis, also associated with HPV 16.
Erythroplasia of Queyrat: Erythroplakia (red plaques) on the glans penis, also associated with HPV 16.
 Bowenoid papulosis: Red papular lesions on the external genitalia associated with HPV 16 that does not have the potential to become invasive squamous cell carcinoma (SCC).
Bowenoid papulosis: Red papular lesions on the external genitalia associated with HPV 16 that does not have the potential to become invasive squamous cell carcinoma (SCC).
 Squamous cell carcinoma: Associated with HPV 16 and 18. Other risk factors include uncircumcised status and smoking.
Squamous cell carcinoma: Associated with HPV 16 and 18. Other risk factors include uncircumcised status and smoking.
Scrotal Sac Disorders
A varicocele is dilation of the spermatic pampiniform venous plexus from increased pressure. It is most common on the left side because the left testicular vein drains into the left renal vein (not directly to the IVC), resulting in increased pressure. A varicocele feels like a “bag of worms.” Patients are asymptomatic or may complain of pain and a pulling sensation in the scrotum. If large, a varicocele can lead to infertility (theoretically because of elevated scrotal temperature from local pooling of blood that inhibits spermatogenesis).
Hydrocele is an incomplete fusion of the tunica vaginalis causing serous fluid accumulation around the testicle, resulting in scrotal swelling. Hydroceles are asymptomatic but are associated with inguinal hernias if there is total failure of fusion and therefore communication with the peritoneal cavity. Hydroceles will transilluminate, meaning that a bright light will pass through the fluid-filled sac (as opposed to testicular tumors, which will block light).
A spermatocele is a cystic mass derived from a tubule of the rete testis or the head of the epididymis. It can be palpated superior to the testicle. It contains spermatozoa and is generally asymptomatic.
Sex Chromosome and Hormone Disorders
Klinefelter syndrome (XXY) is a disorder in which hyalinization and dysgenesis of the seminiferous tubules leads to decreased inhibin. Because inhibin normally has a negative feedback on FSH, its absence causes an increase in FSH. There are also decreased testosterone levels from excess aromatization of testosterone to estrogen, leading to an increase in LH. Clinical symptoms include testicular atrophy, tall and long extremities (lack of testosterone to close epiphyseal plates), gynecomastia, female hair distribution, and infertility. Klinefelter’s syndrome is an example of primary hypogonadism (a defect of the gonad itself).
XYY syndrome occurs as a result of paternal nondisjunction. Patients are phenotypically tall and affected by acne. There are some reports of increased antisocial behavior. Fertility is unaffected.
Androgen insensitivity syndrome (46,XY) results in a nonfunctional androgen receptor, so there is no functional exposure to androgens. This allows for the development of a normal-appearing female with female external genitalia (because of peripheral conversion of testosterone to estrogen). The upper vagina, cervix, uterus, and fallopian tubes are absent because of müllerian-inhibiting hormone from the testes. Testicles are normally found in the labia majora and are removed surgically to prevent malignancy. There are high levels of LH and testosterone because testosterone cannot feed back negatively on the anterior pituitary to regulate LH levels because the testosterone receptors on the anterior pituitary are similarly defective. High LH levels thus stimulate even more testosterone. This is an example of male pseudohermaphroditism because the individual has testes (male) but female-appearing external genitalia.
In 5-α-reductase deficiency, there are normal male internal genitalia because of normal testosterone levels. However, they are phenotypically female-appearing males until puberty. This is because early in life, as a result of the defective enzyme, males cannot convert testosterone to DHT, which is important in developing male external genitalia and masculinization. However, when puberty occurs, there is so much testosterone that it stimulates these changes to occur, regardless of DHT levels.